Abstract
Background
Because of their anti-pyretic effects, some individuals prophylactically use non-steroidal anti-inflammatory drugs (NSAIDs) to blunt core temperature (Tc) increases during exercise, thus, potentially improving performance by preventing hyperthermia and/or exertional heat illness. However, NSAIDs induce gastrointestinal damage, alter renal function, and decrease cardiovascular function, which could compromise thermoregulation and increase Tc. The aim of this systematic review was to evaluate the effects of NSAIDs on Tc in exercising, adult humans.
Methods
We conducted searches in MEDLINE, PubMed, Cochrane Reviews, and Google Scholar for literature published up to November 2020. We conducted a quality assessment review using the Physiotherapy Evidence Database scale. Nine articles achieved a score ≥ seven to be included in the review.
Results
Seven studies found aspirin, ibuprofen, and naproxen had no effect (p > .05) on Tc during walking, running, or cycling for ≤ 90 min in moderate to hot environments. Two studies found significant Tc changes. In one investigation, 81 mg of aspirin for 7–10 days prior to exercise significantly increased Tc during cycling (p < .001); final Tc at the end of exercise = 38.3 ± 0.1 °C vs. control = 38.1 ± 0.1 °C. In contrast, participants administered 50 mg rofecoxib for 6 days experienced significantly lower Tc during 45 min of cycling compared to placebo (NSAID Tc range ≈ 36.7–37.2 °C vs control ≈ 37.3–37.8 °C, p < 0.05).
Conclusions
There are limited quality studies examining NSAID effects on Tc during exercise in humans. The majority suggest taking non-selective NSAIDs (e.g., aspirin) 1–14 days before exercise does not significantly affect Tc during exercise. However, it remains unclear whether Tc increases, decreases, or does not change during exercise with other NSAID drug types (e.g., naproxen), higher dosages, chronic use, greater exercise intensity, and/or greater environmental temperatures.
Keywords: Thermoregulation, Anti-pyretic, Heat illness, Physical activity, Cyclooxygenase inhibitor
Introduction
Non-steroidal anti-inflammatory drugs (NSAIDs) reduce pain and inflammation by inhibiting prostaglandins integral in signaling vasodilation, increased vascular permeability, and fever.1, 2, 3 Specifically, NSAIDs target two cyclooxygenase isoforms (COX-1 and COX-2) that catalyze prostaglandin production from arachidonic acid. COX-1, inherently present in tissues, yields prostaglandins that regulate gastric mucosa, platelet aggregation, and renal blood flow. COX-2, present in small amounts and upregulated by inflammatory mediators, produces prostaglandins that promote pain, inflammation, and fever during an injury/illness.1,4 Non-selective NSAIDs (e.g., aspirin, naproxen, ibuprofen) indiscriminately inhibit both COX isoforms.3 However, due to varying chemical make-ups, non-selective NSAIDs have a greater affinity toward one COX isoform over the other. For instance, aspirin has a greater affinity for COX-1 whereas naproxen has greater affinity for COX-2.3 A selective NSAID (e.g., celecoxib) targets COX-2, inhibiting the inflammatory responses while maintaining the important regulatory and protective COX-1 functions.5
Physically active individuals are known to prophylactically take NSAIDs in an attempt to blunt pain and inflammation during exercise, and controversy exists as to whether individuals may also use NSAIDs to try to mitigate core temperature (Tc) increases during exercise.6 Theoretically, NSAIDs’ may prevent exertional hyperthermia, including exertional heat stroke (EHS), via their anti-pyretic effect.6 It is important to briefly mention EHS pathophysiology to understand how NSAIDs may positively or negatively alter Tc and exertional heat illness risk. The gastrointestinal (GI) and immune systems largely influence EHS development.7, 8, 9, 10 During exercise, increased metabolic heat production, hypoxia, and mechanical pressures damage the GI epithelial barrier.11, 12, 13, 14 As GI permeability increases, endotoxin leaks from the GI tract into the blood stream.10 The immune system and liver remove endotoxin from the blood, allowing the person to continue activity. When the body becomes overloaded, pro-inflammatory mediators (e.g., cytokines) are released to counteract endotoxin. Unfortunately, cytokines also promote necrosis, vasodilation, further induce GI damage, and promote fever.7,8,15 If unmitigated, the end result is a systemic inflammatory response that propagates EHS and, in severe cases, will lead to multi-system organ failure.10 Theoretically, NSAIDs could improve a person’s heat tolerance during exercise by mitigating fever and the overall inflammatory response. On the other hand, NSAIDs induce GI damage and decrease cardiovascular function,16, 17, 18, 19, 20 which would compromise thermoregulation and increase Tc.10,21, 22, 23
Animal studies evaluating NSAIDs’ effect on Tc while under heat and/or exercise stress are conflicting. In monkeys exposed to passive heat, controls experienced a 1.6 ± 0.14 °C increase in Tc whereas acetylsalicylate resulted in as little as a 0.2 ± 0.05 °C increase.24 Rats running in a temperate environment administered indomethacin experienced a 0.4 °C Tc increase compared to controls who experienced a 0.8–1.0 °C increase.25 In contrast, high oral indomethacin doses decreased mice survival rate by 45% during passive heat stress26 and resulted in a 67% mortality rate when mice exercised for 90 min.27 The latter investigation did not examine Tc or GI damage markers that may explain why the combination of strenuous exercise and indomethacin decreased survival; however, the former investigation attributed decreased survival to hemorrhaging in the GI tract caused by combining indomethacin with heat stress.26
NSAID use is prevalent among physically active adolescents and adults and from recreational to elite athletes.28, 29, 30, 31, 32 NSAIDs are perceived as performance enhancing, but using NSAIDs to prevent hyperthermia or EHS remains controversial and the risks (i.e., GI, increased cardiovascular strain) may outweigh any potential benefits towards improved heat-tolerance.6 The purpose of this systematic review was to examine existing literature on the effects that selective and non-selective NSAIDs have on Tc in exercising adult humans.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and checklist were used to guide this review.33
Inclusion and exclusion criteria
We utilized the participants, interventions, comparisons, and study design (PICOS) framework. The population of interest was exercising humans ≥18 years old. The intervention was any selective or non-selective NSAID compared to a placebo or control group. The outcome was Tc measured through a valid assessment (i.e., rectal thermistor, ingestible thermistor, or esophageal thermistor).34 All studies had to be in the English language, original, and utilize a randomized control trial design. Excluded studies consisted of case report studies and review articles; studies using passive hyperthermia only, animals, steroidal anti-inflammatories, and/or acetaminophen; and studies using other tools to measure body temperature (e.g., oral, aural, temporal) were not included, because they can produce invalid results in exercising individuals.34
Search strategy and study selection
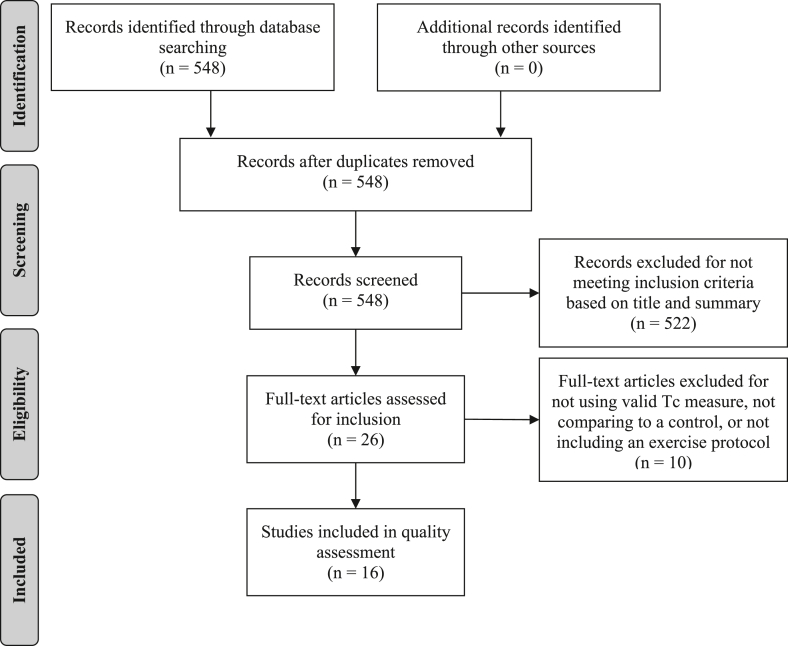
In November 2020, the following databases were searched: MEDLINE, PubMed, Google Scholar, and the Cochrane Reviews. Key words for searches included: NSAIDs, non-steroidal anti-inflammatory drugs, ibuprofen, naproxen, salicylate, aspirin, acetylsalicylic acid, indomethacin, core temperature, temperature, heat, heat illness, hyperthermia, cold, thermoregulation, exercise, and physical activity. The flow-chart for inclusion of relevant studies is provided in Fig. 1. A total of 16 articles were included for a quality assessment review.
Fig. 1.
PRISMA flow chart illustrating the different phases of the literature search and study selection.
Quality assessment
Two researchers independently assessed the methodologic quality of studies using the Physiotherapy Evidence Database (PEDro) scale.35 An inclusion score of 7/10 was selected for an article to be included in data synthesis. If discrepancy existed between reviewers, a discussion occurred to determine whether a reviewer missed or misunderstood a study aspect. A third independent review was conducted if an agreement was not reached; this reviewer was blinded to the previous reviews. Cohen’s κ determined agreement between the two reviewers and revealed very good agreement (κ = 0.886, p < 0.001). After a consensus meeting, agreement improved (κ = 0.952, p < 0.001). The third independent reviewer was required to review 1 article to resolve disagreement. Although some studies included other outcome variables, the focus of this review is on Tc; therefore, only Tc data is discussed.
Results
Table 1 contains final PEDro scores for the 16 articles. Mean PEDro score for the 16 articles = 7.3 ± 2.5, indicating moderate quality research in this area. Specific results are presented in Table 2 for the nine studies meeting inclusion criteria and achieving a PEDro score of 7/10. Seven studies showed no significant Tc difference between non-selective NSAIDs and controls.36, 37, 38, 39, 40, 41, 42 Across these seven studies, drug dosage varied from low to high doses administered between 1 and 14 days; exercise mode included running, walking, or cycling and ranged from 45 to 90 min; and environment ranged from thermoneutral (18 °C) to hot (38.9 °C).
Table 1.
Physiotherapy evidence database (PEDro) scale scores of critically reviewed articles.
| Bass & Jacobson66 | Bradford et al.43 | Bruning et al.18 | de Meersman40 | Downey & Darling67 | Emerson et al.42 | Farquhar & Kenney38 | Farquhar et al.39 | |
|---|---|---|---|---|---|---|---|---|
| Eligibility criteria specified (no points) | ||||||||
|
X |  |
 |
 |
X |  |
 |
 |
|
X |  |
 |
X |  |
 |
X | |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
X |  |
 |
 |
X |  |
 |
 |
|
X |  |
 |
 |
X |  |
 |
X |
|
X |  |
 |
 |
X |  |
 |
X |
|
 |
 |
 |
 |
 |
X |  |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
X |  |
 |
 |
 |
 |
 |
 |
| Total PEDro scale score |
4 |
10 |
10 |
10 |
5 |
9 |
10 |
7 |
| Fujii et al.68 |
Fujii et al.69 |
Hostler et al.37 |
Jacobson & Bass70 |
Lambert et al.41 |
McEntire et al.36 |
Ryan et al.71 |
Veltmeijer et al.72 |
|
| Eligibility criteria specified (no points) | ||||||||
|
X | X |  |
X |  |
 |
X |  |
|
X | X |  |
X | X |  |
X | X |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
X | X |  |
X |  |
 |
 |
X |
|
X | X |  |
X |  |
 |
X | X |
|
X | X |  |
X |  |
 |
X | X |
|
X |  |
X | X |  |
X |  |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
 |
 |
 |
 |
 |
 |
 |
 |
|
 |
 |
 |
X |  |
 |
 |
 |
| Total PEDro scale score | 4 | 5 | 9 | 3 | 9 | 9 | 6 | 6 |
X = article did not meet criteria,  = article met criteria.
= article met criteria.
Table 2.
Results from included articles on NSAIDs and Tc during exercise.
| Bruning et al.18 | de Meersman40 | Hostler et al.37 | Lambert et al.41 | McEntire et al.40 | |
|---|---|---|---|---|---|
| Participants | N = 14 Males and females Age = 55 ± 1 years V̇O2peak = 29.5 ± 1.7 mL/kg/min | N = 8 Males Age 21–34 years Baseline V̇O2max not reported | N = 102 Male and female firefighters Age = 31.9 ± 9.1 years V̇O2max = 44.6 ± 7.2 mL/kg/min | N = 17 Males and females Age = 27 ± 1 years V̇O2max = 62.2 ± 2.1 mL/kg/min | N = 15 Male firefighters Age = 30.5 ± 9.3 years V̇O2max = 46.6 ± 7.0 mL/kg/min |
| NSAID | Aspirin | Aspirin | Aspirin | Aspirin | Aspirin |
| Dose | 81 mg 1 x day for 7–10 days | 500 mg 6 x day for 1 day | 81 mg/day for 14 days | 325 mg 4 x day for 1 day | 81 mg/day for 14 days |
| Timinga | Morning of trial | Last dose not reported; mean drug concentration = 17 ± 8 mg/100 mL | Morning of trial | Morning of trial | Last dose not reported |
| Exercise | 120 min recumbent cycle at 60% V̇O2peak | 60 min run at 50% V̇O2max | 50 min intermittent walking at 4.5 km/h with 2.5% incline and 2.5 km/h with 0% incline | 60 min run at 70% V̇O2max | 45 min walk at 6.4 km/h |
| Environment | 30 °C dry/22 °C wet bulb, 40% RH | 18 ± 0.5 °C, 43 ± 2% RH | 38.9 ± 1.1 °C, 19.8 ± 3.8% RH | 22.4 °C, 48% RH | 38.8 ± 2.1 °C, 24.9 ± 9.1% RH |
| Tc Measure | Esophageal | Rectal | Ingestible thermistor | Rectal | Ingestible thermistor |
| Tc Results | NSAID range ≈37.1–38.2 °C, significantly higher than control range ≈36.6–37.9 °C, p < .001 | At 60 min NSAID = 38.2 ± 0.52 °C and control = 37.9 ± 0.56 °C No significant difference, p > .05 |
Range ≈36.7–39.4 °C No significant difference, p > .05 |
Mean = 38.4 ± 0.1 °C No significant difference, p > .05 |
At 45 min NSAID = 39.0 ± 0.7 °C and control = 39.1 ± 0.6 °C No significant difference, p = .69 |
| Emerson et al.42 | Farquhar & Kenney38 | Farquhar et al.39 | Bradford et al.43 | |
|---|---|---|---|---|
| Participants | N = 11 Males and females Age = 27.8 ± 6.5 years V̇O2max = 41.4 ± 5.7 mL/kg/min |
N = 16 Females Younger age = 24 ± 2 years Older age = 64 ± 2 years Younger V̇O2max = 41.2 ± 2.2 mL/kg/min Older V̇O2max = 29.1 ± 1.7 mL/kg/min |
N = 12 Males and females Age = 25 ± 1 years V̇O2max = 54 ± 1 mL/kg/min |
N = 10 Males Age = 23 ± 5 years V̇O2max = 53.1 ± 5.3 mL/kg/min |
| NSAID | Naproxen | Ibuprofen | Ibuprofen | Rofecoxib |
| Dose | 220 mg 3 x day for 1 day | 400 mg 3 x day for 3 days | 400 mg 3 x day for 4 days | 25 mg 2 x day for 6 days |
| Timinga | ∼1 h before | ∼30 min before | Morning of trial | ∼1½ hours before |
| Exercise | 80 min cycling at 70% V̇O2max then 10 min at max effort | 60 min walk at 60% V̇O2max | 45 min run at 65% V̇O2max | 45 min run at ∼75% V̇O2max then 45 min cycle at matched perceived intensity |
| Environment | 35.7 ± 1.3 °C, 53.2 ± 3.2% RH 22.7 ± 1.8 °C, 52.4 ± 5.5% RH |
36 °C dry/24 °C wet bulb | 36 °C dry/24 °C wet bulb | 28 °C, 50% RH |
| Tc Measure | Rectal | Rectal | Rectal | Rectal |
| Tc Results | At 90 min mean = 38.2 ± 0.3 °C No significant difference, p > .05 |
At 60 min, younger NSAID = 38.74 ± 0.08 °C and control = 38.81 ± 0.10 °C; older NSAID = 38.50 ± 0.16 °C and control = 38.45 ± 0.13 °C No significant difference, p > .05 |
At 45 min NSAID = 39.2 ± 0.1 °C and control = 38.9 ± 0.1 °C No significant difference, p > .05 |
Run range ≈37.2–38.0 °C No significant difference, p > .05 Cycling NSAID range ≈36.7–37.2 °C significantly lower than control range ≈37.3–37.8 °C, p < .05 |
Abbreviations: RH = relative humidity; Tc = core temperature; V̇O2max = maximal oxygen consumption; V̇O2peak = peak oxygen consumption.
Timing refers to when the last NSAID dose was taken prior to exercise.
Two of the nine studies identified significant differences between NSAID and control groups. One study found significantly greater Tc with aspirin use during passive heating (p < .005) that remained higher during exercise (p < .001).18 In the one study that used a selective COX-2 inhibitor,43 no Tc difference occurred when participants ran in a warm environment. However, Tc was 0.33 °C lower during the subsequent 45 min cycling bout in the NSAID group versus control (p = .03), and Tc remained significantly lower throughout the 1-h recovery (p < .001).43
Discussion
It is difficult to make a definitive statement on whether NSAIDs significantly affect Tc during exercise. In general, prophylactic non-selective NSAID use for 14 days or less does not significantly increase or decrease Tc during exercise compared to controls. However, it is important to consider the context of this supposition, because there are limited high quality studies examining this topic and the methodology varies considerably among the existing investigations.
As some of the oldest, most inexpensive, and widely used NSAIDs, aspirin and ibuprofen are often researched. Aspirin has a greater affinity for COX-1.44,45 Daily low-doses (∼81 mg/day) will completely inhibit all platelets in ∼10 days.44 As a result, daily low-dose aspirin is recommended for individuals with cardiovascular disease risk to prevent strokes and myocardial infarctions.45 Using a methodological approach where participants take aspirin daily for 7–14 days and complete some type of physical activity, whether planned exercise or as part of their job (e.g., firefighters),36,37 is applicable to a large population. Interestingly, the three studies with similar aspirin doses over 7–14 days yielded different Tc results.18,36,37 Participant characteristics and exercise may explain the discrepancy. Bruning et al.18 used passive heat exposure then long duration, continuous, moderate-intense cycling in a warm environment. Participants were older and euhydrated with fair to average V̇O2peak. This was the only study to show Tc was higher during exercise. NSAIDs may increase Tc by impairing sweat responses.46,47 However, based on a lack of significant sweat rate or skin temperature responses during exercise between control and NSAIDs,18,37,43 Tc differences likely occurred through other mechanisms. Bruning et al.18 attributed higher Tc to decreased skin blood flow, which prevented the body from dissipating heat as it would if it had a normal vascular response. Appropriate cardiovascular function is essential to thermoregulation, as the body redistributes blood flow to dissipate heat while also supplying working muscles and maintaining cardiac output.48 Poor cardiovascular fitness is highly associated with increased thermoregulatory strain.49,50 Therefore, it is possible the Tc results in Bruning et al.18 were confounded by the participants’ being older and exercising for longer with poor cardiovascular conditioning compared to the younger, more aerobically fit participants in Hostler et al.37 and McEntire et al.36
Before we discuss potential reasons why many of the reviewed studies did not find NSAIDs altered Tc during exercise, it is important to understand that the fever mechanism is different than the mechanism for exertional hyperthermia. A fever occurs when the body initiates a cascade of events that promote heat gain, and this is predominately mediated by prostaglandins.51 These events trigger the set-point temperature in the hypothalamus to increase from ∼37 °C to, for example, 40 °C in order to fight off an infection. In contrast, during exercise, the hypothalamic set-point temperature is not changed. Instead, the body is producing metabolic heat, and when temperatures exceed ∼37 °C, physiological responses are initiated to promote heat loss. The most important heat loss mechanism is sweat evaporation.48 When evaporation is sufficient to maintain Tc (heat loss ≈ heat gain) this is referred to as compensable heat strain.48,52 When evaporative heat loss is impaired, for instance due to high relative humidity or wearing protective clothing, an uncompensable heat situation occurs (heat gain > heat loss).48,53,54 Beyond simply being in a hot, humid environment, many factors, including the person’s heat acclimatization status, hydration status, aerobic fitness, pre-existing medical conditions, or, as it relates to this review, medication use can impair thermoregulation and increase hyperthermia risk.52,53,55
Our first consideration for the lack of Tc affect among studies is related to environmental temperatures and humidity. Wet-bulb globe temperature (WBGT) provides the most accurate indication of the environmental heat strain, accounting for dry bulb, wet bulb, and black bulb temperatures.56 Moderate risk for heat illness begins around 29 °C WBGT with extreme risk above 32 °C WBGT.56 Impaired sweat evaporation begins around 50% relative humidity,57 and, as we mentioned, the ability to effectively evaporate sweat (lose heat) is what makes a condition either compensable or uncompensable. The majority of investigations used hot to extremely hot environments (>30 °C dry, wet, or WBGT).18,36, 37, 38, 39,42 However, only two studies reported >50% humidity.42,43 The environmental strain among many of the investigations was likely not enough to lead to an uncompensable condition.
Even in the absence of a febrile state, NSAIDs lower Tc by inhibiting prostaglandin production.58,59 All non-selective NSAID doses in the reviewed studies should have been enough to reduce fever and theoretically alter Tc. No studies reported differences in baseline or pre-exercise temperature between NSAIDs and control, suggesting NSAIDs did not elicit an anti-pyretic effect or mask a response by “cooling” individuals prior to exercise. During exercise, it is possible inflammation is driving Tc responses. Therefore, NSAID doses would need to be sufficient enough to elicit an anti-inflammatory affect. Ibuprofen study dosages (1.3 and 3.0 g/day)38,39 were less than recommended (3.6 g/day)60 to inhibit inflammation. Similarly, only one study40 administered an aspirin dose within the recommended 2.4–5.4 g/day range for anti-inflammatory affects.60 While the dosage administered by Emerson et al. (220 mg)42 should have provided an anti-inflammatory effect, higher doses (550 mg) may be necessary.60,61
Our next consideration is regarding drug plasma concentrations. Drug effects on Tc would most likely come at peak plasma concentrations, which is influenced by the drug formulation (e.g., tablet, enteric coated, rapid-release) and when the last dose was administered prior to exercise. Ibuprofen peak plasma concentrations occur about 2 h after administration.62 In Farquhar & Kenney,38 the peak plasma concentration likely occurred after exercise ended. This may have occurred in Farquhar et al.39 as well, but we are unable to determine exactly when the last dose was administered. Peak naproxen plasma concentrations occur 1–3 h after administration.61 A lack of Tc difference in Emerson et al.42 may be because peak concentration occurred at the end of exercise. Oral, uncoated aspirin’s affects are rapid, with peak plasma concentration occurring ∼30 min after administration.63 Discrepancy between investigations that administered aspirin for 7–10 days18,36,37 could be related to peak concentration, where aspirin taken the morning of a trial would peak before exercise began. When further considering dose and timing, if aspirin’s effect on Tc during exercise is attributed to inhibited platelet function and skin blood flow,18 a one-day, acute aspirin dose may not alter platelet aggregation and skin blood flow to the extent that Tc would be negatively affected.
Rofecoxib was the only COX-2 selective NSAID among the articles reviewed.43 COX-2 selective NSAIDs are extremely effective at reducing pain and inflammation.3 The anti-inflammatory affect could explain the decreased Tc during cycling and 1-h recovery after exercise.43 COX-2 inhibitors cause less GI damage because they do not inhibit the GI protective prostaglandins produced by COX-1.3 Less GI damage could also explain the lower Tc among those administered rofecoxib; however, GI damage and plasma endotoxin were not measured in the study. The investigators attributed the Tc difference to lower cardiovascular strain,43 but this was not seen when participants ran. We suggest another explanation for the Tc results could be the timing of the last dose. Rofecoxib plasma concentration peaks within 2–3 h.64 Participants in Bradford et al.43 would experience peak plasma concentrations toward the end of the running/beginning of the cycling and throughout recovery, which aligns with when Tc was significantly lower. It should be noted rofecoxib was withdrawn from the market in 2004 due to an increased risk of cardiovascular events.65
Limitations and future research
Several limitations exist in the available research on NSAIDs and Tc during exercise. No studies were included in the critical appraisal that used indomethacin or other commonly used NSAIDs or prescription strength dosages. Indomethacin is extremely potent66 and generally more effective at reducing pain and inflammation than aspirin or ibuprofen.3 To date, celecoxib remains one of the only available selective COX-2 inhibitors on the market and is only available through prescription. Therefore, celecoxib use is not as predominant in physically active populations. In general, the amount of drug available to elicit effects at a given time (e.g., during exercise) is highly dependent on timing, dosage, administration route, formulation, and individual factors that affect how the person will respond to the drug (e.g., body composition, nutritional status, history of medication use). For research to determine to what extent NSAIDs affect Tc, investigators need to administer drug doses based on body weight and/or measure drug plasma concentrations.
Although hyperthermia or EHS can occur in cooler environments, few of the reviewed studies used environments that were hot with high humidity to investigate the compounding effects of exercise and uncompensable heat stress with NSAID use. Due to restrictions in human subjects’ research, participants are rarely allowed to exceed Tc > 39.5 °C and EHS cannot be induced. Five studies averaged a Tc < 39 °C18,38,40,42,67 and only one had participants who exceeded 39.5 °C.36 By limiting the Tc, participants in controlled laboratory studies may inherently have less thermoregulatory strain than participants in field or case studies. Responses to exercise and NSAIDs will differ between individuals who are well-conditioned vs recreationally active, heat acclimatized or non-acclimatized, or have previously used NSAIDs or have never used NSAIDs. Hydration status, nutrition, pre-existing or current illness, sleep, and numerous other factors can also influence thermoregulation by compromising the cardiovascular, GI, and/or immune systems. Not only do these factors need to be controlled for, more research needs to examine the physiological mechanisms responsible for potential interactions between NSAIDs, heat stress, and exercise.
Conclusion
The overall results of this review do not support the belief that NSAIDs decrease Tc and therefore provide some performance benefit to exercising individuals by mitigating hyperthermia. It is possible non-selective NSAIDs may increase Tc by altering thermoregulatory function. However, more research is needed, and results may vary between different populations. We recommend individuals participating in intense exercise in a thermal environment taking NSAIDs use caution. Additionally, sports medicine professionals working with individuals known to be using NSAIDs, who may also have other intrinsic or extrinsic exertional heat illness risks, should be aware of the potential consequences associated with combining NSAIDs, exercise, and heat.
Declaration of competing interest
The authors have no other conflict of interests to disclose.
Contributor Information
Dawn M. Emerson, Email: demerson@kumc.edu.
Stephen CL. Chen, Email: Schen@bju.edu.
Melani R. Kelly, Email: melani.kelly@ku.edu.
Bryant Parnell, Email: parnellb311@gmail.com.
Toni M. Torres-McGehee, Email: torresmc@mailbox.sc.edu.
References
- 1.Brock T.G., McNish R.W., Peters-Golden M. Arachidonic acid is preferentially metabolized by cyclooxygenase-2 to prostacyclin and prostaglandin E2. J Biol Chem. 1999;274(17):11660–11666. doi: 10.1074/jbc.274.17.11660. [DOI] [PubMed] [Google Scholar]
- 2.Ricciotti E., FitzGerald G.A. Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol. 2011;31(5):986–1000. doi: 10.1161/ATVBAHA.110.207449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Botting R.M. Inhibitors of cyclooxygenases: mechanisms, selectivity and uses. J Physiol Pharmacol. 2006;57:113–124. [PubMed] [Google Scholar]
- 4.Constanti A., Bartke A., Khardori R. Harwood Academic Publishers; Amsterdam, Netherlands: 1998. Basic Endocrinology. [Google Scholar]
- 5.Shah A.A., Thjodleifsson B., Murray F.E. Selective inhibition of COX-2 in humans is associated with less gastrointestinal injury: a comparison of nimesulide and naproxen. Gut. 2001;48:339–346. doi: 10.1136/gut.48.3.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Laitano O., Leon L.R., Roberts W.O., Sawka M.N. Controversies in exertional heat stroke diagnosis, prevention, and treatment. J Appl Phys. 2019;127(5):1338–1348. doi: 10.1152/japplphysiol.00452.2019. [DOI] [PubMed] [Google Scholar]
- 7.Bouchama A., Al-Sedairy S., Siddiqui S., Shail E., Rezeig M. Elevated pyrogenic cytokines in heatstroke. Chest. 1993;104:1498–1502. doi: 10.1378/chest.104.5.1498. [DOI] [PubMed] [Google Scholar]
- 8.Bouchama A., Knochel J.P. Heat stroke. N Engl J Med. 2002;346(25):1978–1988. doi: 10.1056/NEJMra011089. [DOI] [PubMed] [Google Scholar]
- 9.Bouchama A., Parhar R.S., El-Yazigi A., Sheth K., Al-Sedairy S. Endotoxemia and release of tumor necrosis factor and interleukin Ia in acute heatstroke. J Appl Physiol. 1991;70(6):2640–2644. doi: 10.1152/jappl.1991.70.6.2640. [DOI] [PubMed] [Google Scholar]
- 10.Lim C.L., Mackinnon L.T. The roles of exercise-induced immune system disturbances in the pathology of heat stroke : the dual pathway model of heat stroke. Sports Med. 2006;36(1):39–64. doi: 10.2165/00007256-200636010-00004. [DOI] [PubMed] [Google Scholar]
- 11.de Oliveira E.P., Burini R.C. The impact of physical exercise on the gastrointestinal tract. Curr Opin Clin Nutr Metab Care. 2009;12(5):533–538. doi: 10.1097/MCO.0b013e32832e6776. [DOI] [PubMed] [Google Scholar]
- 12.Baumgart D.C., Dignass A.U. Intestinal barrier function. Curr Opin Clin Nutr Metab Care. 2002;5(6):685–694. doi: 10.1097/00075197-200211000-00012. [DOI] [PubMed] [Google Scholar]
- 13.ter Steege R.W., Kolkman J.J. Review article: the pathophysiology and management of gastrointestinal symptoms during physical exercise, and the role of splanchnic blood flow. Aliment Pharmacol Ther. 2012;35(5):516–528. doi: 10.1111/j.1365-2036.2011.04980.x. [DOI] [PubMed] [Google Scholar]
- 14.Dokladny K., Moseley P.L., Ma T.Y. Physiologically relevant increase in temperature causes an increase in intestinal epithelial tight junction permeability. Am J Physiol Gastrointest Liver Physiol. 2006;290(2):G204–212. doi: 10.1152/ajpgi.00401.2005. [DOI] [PubMed] [Google Scholar]
- 15.Marshall J.C. The gut as a potential trigger of exercise-induced inflammatory responses. Can J Physiol Pharmacol. 1998;76:479–484. doi: 10.1139/cjpp-76-5-479. [DOI] [PubMed] [Google Scholar]
- 16.Bjarnason I., Williams P., Smethurst P., Peters T.J., Levi A.J. Effect of non-steroidal anti-inflammatory drugs and prostaglandins on the permeability of the human small intestine. Gut. 1986;27:6. doi: 10.1136/gut.27.11.1292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Smecuol E., Bai J.C., Sugai E. Acute gastrointestinal permeability responses to different non-steroidal anti-inflammatory drugs. Gut. 2001;49:6. doi: 10.1136/gut.49.5.650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bruning R.S., Dahmus J.D., Kenney W.L., Alexander L.M. Aspirin and clopidogrel alter core temperature and skin blood flow during heat stress. Med Sci Sports Exerc. 2013;45(4):674–682. doi: 10.1249/MSS.0b013e31827981dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim S., Wook Joo K. vol. 5. Electrolyte Blood Press; 2007. pp. 116–125. (Electrolyte and Acid-Base Disturbances Associated with Non-steroidal Anti-inflammatory Drugs). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cheng H.F., Harris R.C. Renal effects of non-steroidal anti-inflammatory drugs and selective cyclooxygenase-2 inhibitors. Curr Pharmaceut Des. 2005;11(14):1795–1804. doi: 10.2174/1381612053764922. [DOI] [PubMed] [Google Scholar]
- 21.Armstrong L.E., Maresh C.M., Gabaree C.V. Thermal and circulatory responses during exercise: effects of hypohydration, dehydration, and water intake. J Appl Phys. 1997;82(6):2028–2035. doi: 10.1152/jappl.1997.82.6.2028. [DOI] [PubMed] [Google Scholar]
- 22.Casa D.J., Stearns R.L., Lopez R.M. Influence of hydration on physiological function and performance during trail running in the heat. J Athl Train. 2010;45(2):147–156. doi: 10.4085/1062-6050-45.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lambert G.P. Role of gastrointestinal permeability in exertional heatstroke. Exerc Sport Sci Rev. 2004;32(4):185–190. doi: 10.1097/00003677-200410000-00011. [DOI] [PubMed] [Google Scholar]
- 24.Lin M.T., Chai C.Y. Effects of sodium acetylsalicylate on body temperature of monkeys under heat exposure. J Pharmacol Exp Therapeut. 1975;194(1):165–170. [PubMed] [Google Scholar]
- 25.Tanaka Y., Yanase-Fujiwara O., Kanosue K. Effects of centrally and systemically administered indomethacin on body temperature in exercising rats. Am J Physiol. 1993;265:R230–R234. doi: 10.1152/ajpregu.1993.265.1.R230. [DOI] [PubMed] [Google Scholar]
- 26.Audet G.N., Dineen S.M., Stewart D.A. Pretreatment with indomethacin results in increased heat stroke severity during recovery in a rodent model of heat stroke. J Appl Phys. 2017;123(3):544–557. doi: 10.1152/japplphysiol.00242.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Enos R.T., Davis J.M., McClellan J.L., Lake J.L., Carmichael M.D., Murphy E.A. Negative interaction between indomethacin and exercise in mice. Int J Sports Med. 2013;34(3):191–195. doi: 10.1055/s-0032-1323718. [DOI] [PubMed] [Google Scholar]
- 28.Warden S.J. Prophylactic use of NSAIDs by athletes: a risk/benefit assessment. Physician Sportsmed. 2010;1(38):1–7. doi: 10.3810/psm.2010.04.1770. [DOI] [PubMed] [Google Scholar]
- 29.Feucht C.L., Patel D.R. Analgesics and anti-inflammatory medications in sports - use and abuse. Pediatr Clin. 2010;57:751–774. doi: 10.1016/j.pcl.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 30.Warner D.C., Schnepf G., Barrett M.S., Dian D., Swigonski N.L. Prevalence, attitudes, and behaviors related to the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in student athletes. J Adolesc Health. 2002;30:150–153. doi: 10.1016/s1054-139x(01)00325-1. [DOI] [PubMed] [Google Scholar]
- 31.Nakahura T., Griswold W., Lemire J., Mendoza S., Rezni V. Nonsteroidal anti-inflammatory drug use in adolescence. J Adolesc Health. 1998;23:307–310. doi: 10.1016/s1054-139x(98)00036-6. [DOI] [PubMed] [Google Scholar]
- 32.Chambers C.T., Reid G.J., McGrath P.J., Finley G.A. Self-administration of over-the-counter medication for pain among adolescents. Arch Pediatr Adolesc Med. 1997;151:449–455. doi: 10.1001/archpedi.1997.02170420019003. [DOI] [PubMed] [Google Scholar]
- 33.Moher D., Liberati A., Tetzlaff J., Altman D.G. Preferred reporting Items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):1–6. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Casa D.J., Becker S.M., Ganio M.S. Validity of devices that assess body temperature during outdoor exercise in the heat. J Athl Train. 2007;42(3):333–342. [PMC free article] [PubMed] [Google Scholar]
- 35.PEDro scale. https://www.pedro.org.au/english/downloads/pedro-scale/
- 36.McEntire S.J., Reis S.E., Suman O.E., Hostler D. Effects of low-dose aspirin therapy on thermoregulation in firefighters. Saf Health Work. 2015;6(3):256–262. doi: 10.1016/j.shaw.2015.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hostler D., Gallagher M., Jr., Goss F.L. The effect of hyperhydration on physiological and perceived strain during treadmill exercise in personal protective equipment. Eur J Appl Physiol. 2009;105(4):607–613. doi: 10.1007/s00421-008-0940-2. [DOI] [PubMed] [Google Scholar]
- 38.Farquhar W.B., Kenney W.L. Age and renal prostaglandin inhibition during exercise and heat stress. J Appl Phys. 1999;86(6):1936–1943. doi: 10.1152/jappl.1999.86.6.1936. [DOI] [PubMed] [Google Scholar]
- 39.Farquhar W.B., Morgan A.L., Zambraski E.J., Kenney W.L. Effects of acetaminophen and ibuprofen on renal function in the stressed kidney. J Appl Phys. 1999;86(2):598–604. doi: 10.1152/jappl.1999.86.2.598. [DOI] [PubMed] [Google Scholar]
- 40.de Meersman R.E. Thermal, ventilatory, gluco-regulatory responses during exercise following short-term acetylsalicylic acid ingestion. Int J Clin Pharm. 1988;8(6):477–483. [PubMed] [Google Scholar]
- 41.Lambert G.P., Broussard L.J., Mason B.L., Mauermann W.J., Gisolfi C.V. Gastrointestinal permeability during exercise: effects of aspirin and energy-containing beverages. J Appl Phys. 2001;90(6):2075–2080. doi: 10.1152/jappl.2001.90.6.2075. [DOI] [PubMed] [Google Scholar]
- 42.Emerson D.M., Davis J.M., Chen S.C.L. A 24 hour naproxen dose on gastrointestinal distress and performance during cycling in the heat. Sports Med Health Sci. 2020;2(1):19–24. doi: 10.1016/j.smhs.2020.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bradford C.D., Cotter J.D., Thorburn M.S., Walker R.J., Gerrard D.F. Exercise can be pyrogenic in humans. Am J Physiol Regul Integr Comp Physiol. 2007;292(1):R143–R149. doi: 10.1152/ajpregu.00926.2005. [DOI] [PubMed] [Google Scholar]
- 44.Patrono C., Coller B., Dalen J.E. Platelet-active drugs: the relationship among dose, effectiveness, and side effects. Chest. 2001;119(1 Suppl):39S–63S. doi: 10.1378/chest.119.1_suppl.39s. [DOI] [PubMed] [Google Scholar]
- 45.Patrono C., Garcia Rodriguez L.A., Landolfi R., Baigent C. Low-dose aspirin for the prevention of atherothrombosis. N Engl J Med. 2005;353:2373–2383. doi: 10.1056/NEJMra052717. [DOI] [PubMed] [Google Scholar]
- 46.Fujii N., McGinn R., Stapleton J.M., Paull G., Meade R.D., Kenny G.P. Evidence for cyclooxygenase-dependent sweating in young males during intermittent exercise in the heat. J Physiol. 2014;592(23):5327–5339. doi: 10.1113/jphysiol.2014.280651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fujii N., Pastore O.L., McGarr G.W. Cyclooxygenase-1 and -2 modulate sweating but not cutaneous vasodilation during exercise in the heat in young men. Phys Rep. 2018;6(17) doi: 10.14814/phy2.13844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.McArdle W.D., Katch F.I., Katch V.L. fifth ed. Lippincott Williams & Wilkins; Baltimore, MD: 2001. Exercise Physiology: Energy, Nutrition, and Human Performance. [Google Scholar]
- 49.Kenney W.L. Thermoregulation at rest and during exercise in healthy older adults. Exerc Sport Sci Rev. 1997;25(1):41–76. [PubMed] [Google Scholar]
- 50.Greenhaff P.L. Cardiovascular fitness and thermoregulation during prolonged exercise in man. Br J Sports Med. 1989;23(2):109–114. doi: 10.1136/bjsm.23.2.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Engblom D., Saha S., Engstrom L. Microsomal prostaglandin E synthase-1 is the central switch during immune-induced pyresis. Nat Neurosci. 2003;6(11):1137–1138. doi: 10.1038/nn1137. [DOI] [PubMed] [Google Scholar]
- 52.Cheung S.S., McLellan T.M. Heat acclimation, aerobic fitness, and hydration effects on tolerance during uncompensable heat stress. J Appl Phys. 1998;84(5):1731–1739. doi: 10.1152/jappl.1998.84.5.1731. [DOI] [PubMed] [Google Scholar]
- 53.Cheung S.S., McLellan T.M., Tenaglia S. The thermophysiology of uncompensable heat stress. Physiological manipulations and individual characteristics. Sports Med. 2000;29(5):329–359. doi: 10.2165/00007256-200029050-00004. [DOI] [PubMed] [Google Scholar]
- 54.Robinson S., Turrell E.S., Gerking S.D. Phyiological equivalent conditions of air temperature and humidity. Am J Physiol. 1944;143(1):21–32. [Google Scholar]
- 55.Kraning K.K., Gonzalez R.R. Physiological consequences of intermittent exercise during compensable and uncompensable heat stress. J Appl Phys. 1991;71(6):2138–2145. doi: 10.1152/jappl.1991.71.6.2138. [DOI] [PubMed] [Google Scholar]
- 56.Sawka M.N., Wenger C.B., Montain S.J. Department of the Army; 2003. Heat Stress Control and Heat Caualty Management. [Google Scholar]
- 57.Armstrong L.E. Heat and humidity. In: Bahrke M.S., editor. Performance in Extreme Environments. Human Kinetics; Champaign, IL: 2000. pp. 15–70. [Google Scholar]
- 58.Satinoff E. Salicylate: action on normal body temperature in rats. Science. 1972;176:532–533. doi: 10.1126/science.176.4034.532. [DOI] [PubMed] [Google Scholar]
- 59.Bizzi A., Garattini S., Veneroni E. The action of salicylate in reducing plasma free fatty acids and its pharmacological consequences. Br J Pharmacol Chemother. 1965;25:187–196. doi: 10.1111/j.1476-5381.1965.tb01771.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ciccone C.D. F. A. Davis; Philadelphia, PA: 2013. Davis’s Drug Guide for Rehabilitation Specialists. [Google Scholar]
- 61.Davies M.N., Anderson K.E. Clinical pharmacokinetics of naproxen. Clin Pharmacokinet. 1997;32(4):268–293. doi: 10.2165/00003088-199732040-00002. [DOI] [PubMed] [Google Scholar]
- 62.Davies M.N. Clinical pharmacokinetics of ibuprofen: the first 30 years. Clin Pharmacokinet. 1998;34(2):101–154. doi: 10.2165/00003088-199834020-00002. [DOI] [PubMed] [Google Scholar]
- 63.Sagar K.A., Smyth M.R. A comparative bioavailability study of different aspirin formulations using on-line multidimensional chromatography. J Pharm Biomed. 1999;21:383–392. doi: 10.1016/s0731-7085(99)00177-6. [DOI] [PubMed] [Google Scholar]
- 64.Davies M.N., Teng X.W., Skjodt N.M. Pharmacokinetics of rofecoxib: a specific cyclo-oxygenase-2 inhibitor. Clin Pharmacokinet. 2003;42(6):545–556. doi: 10.2165/00003088-200342060-00004. [DOI] [PubMed] [Google Scholar]
- 65.Kaufman M. Merck withdraws arthritis medication. Wash Post. 2004 [Google Scholar]
- 66.Cryer B., Feldman M. Cyclooxygenase-1 and cyclooxygenase-2 selectivity of widely used nonsteroidal anti-inflammatory drugs. Am J Med. 1998;104:413–421. doi: 10.1016/s0002-9343(98)00091-6. [DOI] [PubMed] [Google Scholar]
- 67.Lambert G.P., Schmidt A., Schwarzkopf K., Lanspa S. Effect of aspirin dose on gastrointestinal permeability. Int J Sports Med. 2012;33(6):421–425. doi: 10.1055/s-0032-1301892. [DOI] [PubMed] [Google Scholar]
- 68.Fujii N., McGinn R., Stapleton J.M., Paull G., Meade R.D., Kenny G.P. Evidence for cyclooxyegenase-dependent sweating in young males during intermittenet exercise in the heat. J Physiol. 2014;592(23):5327–5339. doi: 10.1113/jphysiol.2014.280651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fujii N., Paull G., Meade R.D. Do nitric oxide synthase and cyclooxygenase contribute to the heat loss response in older males exercising in the heat? J Physiol. 2015;593(14):3169–3180. doi: 10.1113/JP270330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jacobson E.D., Bass D.E. Effects of sodium salicyalte on physiological resposnes to work in heat. J Appl Physiol. 1964;19(1):33–36. doi: 10.1152/jappl.1964.19.1.33. [DOI] [PubMed] [Google Scholar]
- 71.Ryan A.J., Chang R.-T., Gisolfi C.V. Gastrointestinal permeability following aspirin intake and prolonged running. Med Sci Sports Exerc. 1995;28(6):698–705. doi: 10.1097/00005768-199606000-00009. [DOI] [PubMed] [Google Scholar]
- 72.Veltmeijer M.T.W., Veeneman D., Bongers C.C.C.W. Int J Sports Physiol Perform. 2017;12(5):662–667. doi: 10.1123/ijspp.2016-0382. [DOI] [PubMed] [Google Scholar]