The novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) pandemic has spread rapidly across the world (Zhu et al., 2020). The clinical manifestations of Covid-19 appear to be extensive, encompassing asymptomatic infection, circulatory and renal injury, mild upper respiratory tract illness, and severe viral pneumonia with respiratory failure or death and poses a particular risk to people living with preexisting conditions (Zhu et al., 2020). The severity of Covid-19 results from a profound lymphopenia, the downregulation of type I IFN-mediated antiviral defense, as well as an excessive inflammatory response orchestrated by monocytes and macrophages (Merad and Martin, 2020, Vabret et al., 2020). The potent activation of inflammatory monocytes/macrophages leads to a cytokine storm of IL-6, MCP-1, TNFα (Merad and Martin, 2020, Vabret et al., 2020). This uncontrolled release of cytokines was previously reported during severe infections mediated by other pathogenic human coronavirus as SARS-CoV and MERS-CoV, which, contrary to SARS-CoV2, also triggered high levels of IL-1β (Merad and Martin, 2020, Perlman and Dandekar, 2005).
The early innate immune evasion strategies used by SARS-CoV2 to circumvent type I signaling in order to gain a window of opportunity for virus propagation, remains to be elucidated. SARS-CoV2 could express some immune evasion proteins capable of reducing the interferon regulatory factor 3 (IRF3) and IRF7 activation to limit the type I IFN stimulation (Figure 1), as previously observed for SARS-CoV, and MERS-CoV. (Merad and Martin, 2020, Perlman and Dandekar, 2005, Schäfer and Baric, 2017, Schulz and Mossman, 2016, Vabret et al., 2020). In addition, the epigenetic control of type I IFN innate immune response by SARS-CoV2 could be hypothesized, as MERS-CoV infection mediates repressive histone modification in promoter regions of interferon stimulating genes (ISG) for the shutdown of innate immune signaling (Figure 1) (Merad and Martin, 2020, Perlman and Dandekar, 2005, Schäfer and Baric, 2017, Vabret et al., 2020). Moreover, the kinetics of the systemic and the local IFN responses, an important parameter that determines SARS-CoV2 infection outcomes, needs clinical and experimental clarification. Variations in severity of infection and mortality by SARS-CoV2 are broadly reported worldwide but remain unexplained up to now. One hypothesis associated Covid-19 incidence and mortality with Bacille Calmette-Guérin (BCG) vaccinations (Miller et al., 2020, O’Neill and Netea, 2020). Countries maintaining long-standing universal BCG vaccination policies, such as South Korea and Japan, showed lower SARS-CoV2-associated fatalities than nations such as Italy or USA, which interrupted global BCG vaccination campaigns (Miller et al., 2020). BCG, a live attenuated vaccine against tuberculosis (TB), is nowadays one of the most widely used vaccines globally, covering more than 90% of newborns and infants. Recent epidemiological and clinical findings suggested that, in addition to TB-specific protection, BCG vaccinations trigger as well non-specific effects through the ability to enhance immune protection against other pathogens (Aaby and Benn, 2012, Aaby et al., 2011, Anderson et al., 2012, Colditz et al., 1995, Stensballe et al., 2005). Notably, randomized trials revealed that BCG vaccinations of infants could reduce the risk of lethal respiratory infections by 50% and these protective effects were clearly not related only to tuberculosis prevention (Aaby and Benn, 2012, Aaby et al., 2011). Protective effects of BCG- was evidenced for a wide range of pathogens, including Staphylococcus aureus, Candida albicans, and Yellow fever virus (Covián et al., 2019, Moorlag et al., 2019). Also, mice vaccinated with BCG reduced influenza A virus viral titers and generated protective immunity against herpes simplex virus type 2 (HSV-2) (Covián et al., 2019, Moorlag et al., 2019).
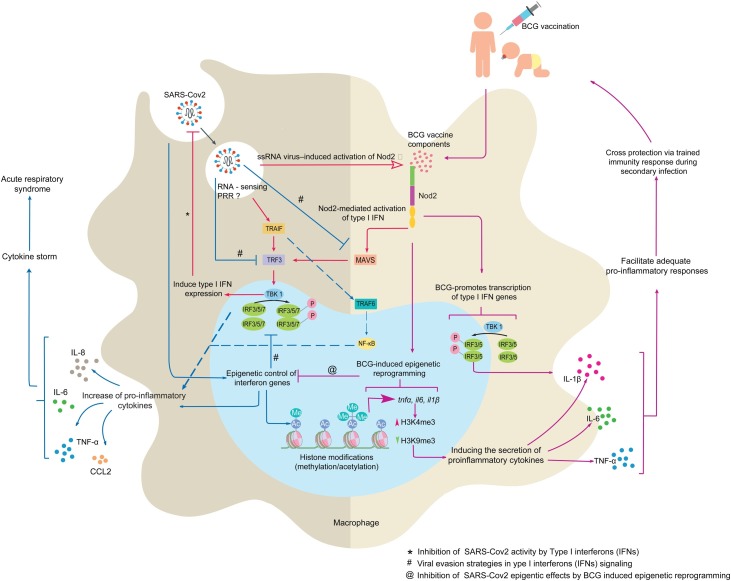
Figure 1.
NOD2-dependent BCG-induced trained immunity may be determinant for the control of SARS-CoV2 infection. SARS-CoV2 could adopt different strategies (immune evasion protein expression and epigenetic changes) to delay or fully inhibit host type I interferon (IFN) antiviral defenses, resulting in the development of pulmonary hyper-inflammation in severe Covid-19. We suggest that NOD2 signaling following BCG vaccinations could trigger epigenetic reprogramming of innate immune responses in macrophages/monocytes to prevent the cytokine storm. Abbreviation; CCL2, CC chemokine ligand 2, IRF, interferon regulatory factors, NOD2, nucleotide-binding oligomerization domain-containing protein 2, NF-kB, nuclear factor kappa-light-chain-enhancer of activated B cells, MAVS, mitochondrial antiviral-signaling protein, PRR, pattern recognition receptor, TRAF, tumor necrosis factor receptor (TNFR)-associated factor, TBK 1, serine/threonine-protein kinase 1, TRF3, telomere binding protein 3.
Although the mechanisms of these BCG-induced immunomodulatory effects remain to be fully elucidated, induction of innate immune memory appeared to be the most plausible explanation (Aaby and Benn, 2012). This T- and B-cell independent trained immunity results from epigenetic reprogramming of innate immune cells, specifically through histone modifications and leading to upregulation of proinflammatory cytokine production (IL-1, IL-6, TNFα) (Figure 1) (Covián et al., 2019, Kleinnijenhuis et al., 2014, Kleinnijenhuis et al., 2012, Moorlag et al., 2019). Notably, BCG vaccinations were shown to trigger H3K4me3 variation in human peripheral blood monocytes (Covián et al., 2019, Kleinnijenhuis et al., 2014, Kleinnijenhuis et al., 2012, Moorlag et al., 2019). The epigenetic control of proinflammatory cytokine gene expression by BCG is mainly dependent on the nucleotide-binding oligomerization domain 2 receptor (NOD2) signaling in monocytes (Covián et al., 2019, Kleinnijenhuis et al., 2012) as evidenced by the following data: (1) ablation of TLR2, TLR4 or Dectin-1 signaling did not abolish cross-protection induced by BCG (Kleinnijenhuis et al., 2012); (2) homozygous 1022insC frameshift mutation leading to complete NOD2 deficiency, abolished BCG-induced cytokine upregulation in human monocytes (Kleinnijenhuis et al., 2012); (3). The training effects elicited by BCG could be reproduced with NOD2 agonist muramyl dipeptide (MDP) from the mycobacterial cell wall (Covián et al., 2019, Kleinnijenhuis et al., 2012). Of note, these epigenetic modifications were shown to increase NOD2 expression (Figure 1). Virulent Mycobacterium tuberculosis strains induce expression of type I IFN mediated by NOD2 together with cyclic GMP-AMP synthase (cGAS) signaling (Moreira-Teixeira et al., 2018). Such type I IFN responses were shown to downregulate proinflammatory cytokines and impaired antibacterial Th1 responses (Moreira-Teixeira et al., 2018). Interestingly, BCG is capable of eliciting type I IFN expression in both human and mouse primary macrophages, albeit to a significantly lesser degree than pathogenic M. tuberculosis (Moreira-Teixeira et al., 2018).
Based on these data, we speculate that BCG-trained innate immunity could similarly control SARS-CoV2 infection by appropriate control of type I IFN and proinflammatory cytokine responses (Figure 2). According to the critical role of NOD2 in BCG-induced innate memory, we hypothesize that NOD2 signaling in macrophages/monocytes could be determinant to tune innate immune responses against SARS-CoV2 (Figure 1).
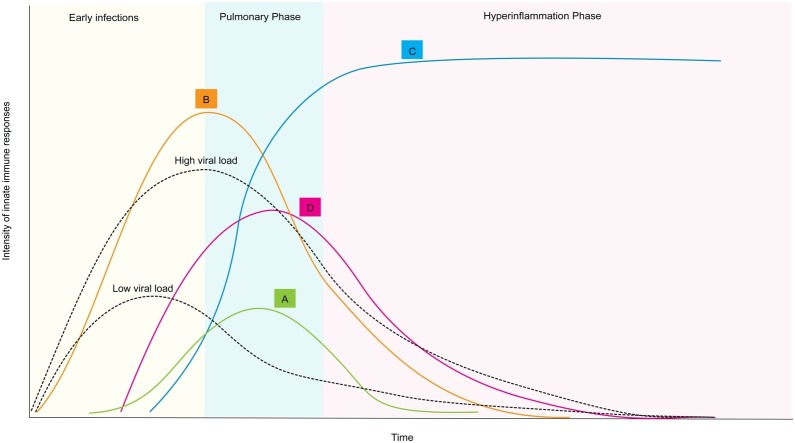
Figure 2.
Time course of innate immune responses to primary SARS-CoV2 infection may be modulated by BCG-induced trained immunity. Delayed or suboptimal antiviral defenses (A) cannot regulate the progression of SARS-CoV2 infection (high viral load). This may result in the development of cytokine storm (overproduction of proinflammatory cytokines as IL-6, IL-8, TNFα), resulting in the influx of various immune cells such as macrophages, neutrophils, and T cells to trigger to acute respiratory distress syndrome (ARDS) and widespread tissue damage (C). Alternatively, in a BCG-vaccinated individual, trained immunity finely tunes type I IFNs/proinflammatory cytokine response (B) to control the SARS-CoV2 infection (low viral load) and, consequently, prevent ARDS (D).
The role of NOD2 signaling in SARS-CoV2 infection remains altogether unknown up to now. However, NOD2 was reported to sense ssRNA from viruses as Respiratory Syncytial Virus (RSV), Vesicular Stomatitis Virus (VSV), Parainfluenza, Influenza A, or Foot-and-mouth disease virus (FMDV) to stimulate type I IFNs responses through the mitochondrial membrane-anchored CARD protein (MAVS)/IRF3/IRF7 signaling (Keestra-Gounder and Tsolis, 2017, Moreira and Zamboni, 2012, Sabbah et al., 2009). Interestingly, MERS-CoV is capable of decreasing NOD2 expression in macrophages (Kim et al., 2019).
We thus propose that BCG vaccinations, through the induction of NOD2-dependent epigenetic reprogramming, inhibit SARS-CoV2 evasion strategies and prevent excessive accumulation of monocytes, macrophages, and neutrophils in the lungs and consequently the cytokine storm induction (Figure 1). Particularly, BCG could early elicit type I IFN expression by NOD2 signaling (Figure 2). This early antiviral response would be critical for the control of SARS-CoV2 propagation as delayed IFN-I production is associated with the pathology of Covid-19 (Acharya et al., 2020, Schulz and Mossman, 2016) (Figure 2). The in-depth characterization of the molecular crosstalk between type-I IFN and proinflammatory cytokine signaling together with epigenetic changes associated with SARS-CoV2 infection will be decisive to clearly measure the potential of BCG-trained innate memory for the control of Covid-19. Results from large clinical trials (ClincialTrials.gov identifiers NCT04328441 and NCT04327206) currently underway will be influential in recommending BCG vaccinations to prevent Covid-19. Together with the extensive characterization of innate immune activation by SARS-CoV2, several key unanswered research questions need to be addressed: What is the role of NOD2 in SARS-CoV2 infection? Could BCG-induced trained immunity positively influence NOD2 signaling for the early induction of IFN-I inflammatory responses? What are the epigenetic changes triggered by SARS-CoV2 in innate immune cells? Could the epigenetic reprogramming by BCG vaccinations interfere with epigenetic changes modulated by SARS-CoV2? Is BCG-trained immunity in the context of Covid-19 dependent on the type of BCG vaccine strains? As Covid-19 progresses to severe disease, particularly in elderly patients, is the strength of BCG-induced memory age-dependent?
BCG represents an elegant platform as well for the development of recombinant vaccines, including auto-adjuvant capacities (Covián et al., 2019). Consequently, vaccine candidates based on recombinant BCG expressing SARS-CoV2 antigens could simultaneously drive potent innate immune memory and adaptive responses. Finally, NOD2 synthetic ligands could be selected for the control of the innate immune response during SARS-CoV2 infection and could also be used as vaccine adjuvants. Together with the development of vaccine(s) against Covid-19, we believe that NOD2-dependent BCG-induced trained immunity will strengthen antiviral defenses to prevent any future episode of coronavirus pandemics.
Innate immunity, acting as the first element of defense, can represent, together with other mechanisms, a promising target for the treatment of patients or at least to better understand the pathogenesis of the disease.
Authors’ contributions
The authors contributed equally to all aspects of the article.
Competing interest
The authors declare no competing interests.
Acknowledgments
Dhammika Leshan Wannigama was supported by Chulalongkorn University (Second Century Fund- C2F Fellowship), and the University of Western Australia (Overseas Research Experience Fellowship). Alain Jacquet was supported by the Ratchadapisek Sompotch Fund, Faculty of Medicine, Chulalongkorn University (RA61/123), and Chulalongkorn Academic Advancement (2nd Century Project-CUAASC).
References
- Aaby P., Benn C.S. Saving lives by training innate immunity with Bacille Calmette-Guerin vaccine. Proc Natl Acad Sci U S A. 2012;109(43):17317–17318. doi: 10.1073/pnas.1215761109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaby P., Roth A., Ravn H., Napirna B.M., Rodrigues A., Lisse I.M. Randomized trial of BCG vaccination at birth to low-birth-weight children: beneficial non-specific effects in the neonatal period? J Infect Dis. 2011;204(2):245–252. doi: 10.1093/infdis/jir240. [DOI] [PubMed] [Google Scholar]
- Acharya D., Liu G., Gack M.U. Dysregulation of type I interferon responses in COVID-19. Nat Rev Immunol. 2020;20(7):397–398. doi: 10.1038/s41577-020-0346-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson E.J., Webb E.L., Mawa P.A., Kizza M., Lyadda N., Nampijja M. The influence of BCG vaccine strain on mycobacteria-specific and non-specific immune responses in a prospective cohort of infants in Uganda. Vaccine. 2012;30(12):2083–2089. doi: 10.1016/j.vaccine.2012.01.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colditz G.A., Berkey C.S., Mosteller F., Brewer T.F., Wilson M.E., Burdick E. The efficacy of Bacillus Calmette-Guérin vaccination of newborns and infants in the prevention of tuberculosis: meta-analyses of the published literature. Pediatrics. 1995;96(1 Pt 1):29–35. [PubMed] [Google Scholar]
- Covián C., Fernández-Fierro A., Retamal-Díaz A., Díaz F.E., Vasquez A.E., Lay M.K. BCG-induced cross-protection and development of trained immunity: implication for vaccine design. Front Immunol. 2019;10:2806. doi: 10.3389/fimmu.2019.02806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keestra-Gounder A.M., Tsolis R.M. NOD1 and NOD2: beyond peptidoglycan sensing. Trends Immunol. 2017;38(10):758–767. doi: 10.1016/j.it.2017.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Yang Y.L., Jang Y.-S. Human β-defensin 2 is involved in CCR2-mediated Nod2 signal transduction, leading to activation of the innate immune response in macrophages. Immunobiology. 2019;224(4):502–510. doi: 10.1016/j.imbio.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinnijenhuis J., Quintin J., Preijers F., Benn C.S., Joosten L.A., Jacobs C. Long-lasting effects of BCG vaccination on both heterologous Th1/Th17 responses and innate trained immunity. J Innate Immun. 2014;6(2):152–158. doi: 10.1159/000355628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinnijenhuis J., Quintin J., Preijers F., Joosten L.A., Ifrim D.C., Saeed S. Bacille Calmette-Guerin induces NOD2-dependent non-specific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci U S A. 2012;109(43):17537–17542. doi: 10.1073/pnas.1202870109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20(6):355–362. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller A., Reandelar M.J., Fasciglione K., Roumenova V., Li Y., Otazu G.H. Correlation between universal BCG vaccination policy and reduced morbidity and mortality for COVID-19: an epidemiological study. medRxiv. 2020 2020.03.24.20042937. [Google Scholar]
- Moorlag S., Arts R.J.W., van Crevel R., Netea M.G. Non-specific effects of BCG vaccine on viral infections. Clin Microbiol Infect. 2019;25(12):1473–1478. doi: 10.1016/j.cmi.2019.04.020. [DOI] [PubMed] [Google Scholar]
- Moreira L.O., Zamboni D.S. NOD1 and NOD2 signaling in infection and inflammation. Front Immunol. 2012;3:328. doi: 10.3389/fimmu.2012.00328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreira-Teixeira L., Mayer-Barber K., Sher A., O’Garra A. Type I interferons in tuberculosis: foe and occasionally friend. J Exp Med. 2018;215(5):1273–1285. doi: 10.1084/jem.20180325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Neill L.A.J., Netea M.G. BCG-induced trained immunity: can it offer protection against COVID-19? Nat Rev Immunol. 2020;20(6):335–337. doi: 10.1038/s41577-020-0337-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlman S., Dandekar A.A. Immunopathogenesis of coronavirus infections: implications for SARS. Nat Rev Immunol. 2005;5(12):917–927. doi: 10.1038/nri1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabbah A., Chang T.H., Harnack R., Frohlich V., Tominaga K., Dube P.H. Activation of innate immune antiviral responses by Nod2. Nat Immunol. 2009;10(10):1073–1080. doi: 10.1038/ni.1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schäfer A., Baric R.S. Epigenetic landscape during coronavirus infection. Pathogens. 2017;6(1) doi: 10.3390/pathogens6010008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulz K.S., Mossman K.L. Viral evasion strategies in type I IFN signaling — a summary of recent developments. Front Immunol. 2016;7:498. doi: 10.3389/fimmu.2016.00498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stensballe L.G., Nante E., Jensen I.P., Kofoed P.E., Poulsen A., Jensen H. Acute lower respiratory tract infections and respiratory syncytial virus in infants in Guinea-Bissau: a beneficial effect of BCG vaccination for girls community based case-control study. Vaccine. 2005;23(10):1251–1257. doi: 10.1016/j.vaccine.2004.09.006. [DOI] [PubMed] [Google Scholar]
- Vabret N., Britton G.J., Gruber C., Hegde S., Kim J., Kuksin M. Immunology of COVID-19: current state of the science. Immunity. 2020;52(6):910–941. doi: 10.1016/j.immuni.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu N., Zhang D., Wang W., Li X., Yang B., Song J. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]