Abstract
Macrophages play major roles to produce several pro-inflammatory and inflammatory mediators in chronic inflammatory diseases. All current anti-inflammatory drugs target these mediators to alleviate inflammation. Searching for new anti-inflammatory agents is always needed due to problems from the clinical use of current anti-inflammatory drugs. We intended to evaluate the anti-inflammatory potential of three main compounds, arborinine, methylatalaphylline, and S-deoxydihydroglyparvin (DDGP), from Glycosmis parva leaves and branches on macrophage stimulated by lipopolysaccharide (LPS). Only DDGP demonstrated a potent inhibitor of LPS-activated macrophages. Results indicated that the mRNA level of inducible nitric oxide synthase (iNOS) was inhibited by the treatment in accompany with the decreased nitric oxide (IC50 at 3.47 ± 0.1 μM). DDGP was shown to suppress tumor necrosis factor-α, interleukin (IL)-1, and IL-6 at the mRNA expression and at the released protein levels. In addition, DDGP inhibited the several chemokines, monocyte chemoattractant protein-1 and macrophage inflammatory proteins-1α, and enzymes for prostaglandin (PG) synthesis. It also inhibited PGE2 production. On LPS signaling pathways, DDGP profoundly decreased phosphorylation of p38 mitogen-activated protein kinase (MAPK) in the LPS-treated cells. It had little or no effect on the activation of JNK, ERK and nuclear factor kappa B. In conclusion, results suggested that DDGP from G. parva inhibited expression and production of inflammatory molecules in LPS-activated macrophages through suppressing p38 MAPK activation. DDGP should be a good candidate anti-inflammatory agent in the future.
Key words: Anti-inflammation, Glycosmis parva, lipopolysaccharide, macrophage, S-deoxydihydroglyparvin
INTRODUCTION
Inflammation is a defense mechanism of the body against harmful stimuli. Dysregulated inflammation is involved in various chronic inflammatory diseases.[1] Activated macrophages are crucial cells for the pathogenesis of chronic inflammation by generating several inflammatory molecules.[2] The mitogen-activated protein kinase (MAPK) signaling pathways and activation of nuclear factor kappa B (NF-κB) are the main intracellular signaling cascades involve in the production of these inflammatory mediators.[3] Syntheses and activities of these mediators become the targets of current anti-inflammatory drugs. Side effects and high cost of these drugs present a burden for patients. Therefore, the search for novel anti-inflammatory agents is still needed.[4] Medicinal plants are rich sources for searching new anti-inflammatory agents.[5] Glycosmis parva Craib is a plant in the Rutaceae distributed mainly in Thailand. The ethyl acetate extracts of G. parva leaves and branches previously demonstrated antiviral activities against herpes simplex viruses.[6] The extract of G. parva leaves arrested the cell cycle and induced apoptosis of colorectal cancer HT-29 cells in part by suppressing cyclooxygenase-2 (COX-2) expression.[7] We found that the ethyl acetate extracts of G. parva leaves and branches potently suppressed lipopolysaccharide (LPS)-induced macrophage J774A.1 cell activation by decreasing pro-inflammatory cytokines, COX-2, and inducible nitric oxide synthase (iNOS) expressions (unpublished data). Six acridone alkaloids and four sulfur-containing propanamide derivatives were isolated from the extracts, and their structures were well elucidated.[6] A new sulfur-containing propanamide S-deoxydihydroglyparvin (DDGP) from the leaves, and two acridone alkaloids, arborinine (ABN) from the leaves and N-methylatalaphylline (MPL) from the branches, were main compounds chosen for this study. None of them have been reported for anti-inflammatory activities. We predicted those some of these compounds may have anti-inflammatory activities similar to that of the ethyl acetate extracts. Therefore, we intended to study the anti-inflammatory potential and the mechanisms of actions of the compounds using LPS-induced macrophage model.
MATERIALS AND METHODS
Dulbecco's modified eagle medium (DMEM) and medium reagents were from Gibco (USA). Dexamethasone (DEX), dimethyl sulfoxide (DMSO), LPS (Escherichia coli O26:B6), and resazurin were from Sigma-Aldrich (USA). Griess reagent system and ImProm-II™ reverse transcription system were from Promega Corporation (USA). Primers for real-time polymerase chain reaction (PCR) were from Bio Basic Inc. (Canada). Tumor necrosis factor-α (TNF-α), interleukin (IL)-1, IL-6, and PGE2 ELISA kits were from ImmunoTools (Germany), Thermo Fisher Scientific (USA), and R&D Systems (USA). RIPA lysis buffer was from Abcam (UK). Cell fractionation kit and all antibodies (against ERK1/2, phospho-ERK1/2, p38, phospho-p38, JNK, phospho-JNK, NF-κB p65, IκB-α, proliferating cell nuclear antigen [PCNA], and GAPDH) were purchased from Cell Signaling Technology (USA). HRP substrate was from Merck Millipore (USA).
Cell culture
Mouse macrophage RAW 264.7 cells (ATCC, USA) were used in the present study. The cells were cultured in DMEM supplemented with 100 U/ml penicillin and 100 μg/mL streptomycin and 10% fetal bovine serum, in appropriate cell culture condition. The cells were prepared at 4 × 105 cells/ml in suitable well plates for 24 h and treated with 100 ng/ml of LPS.
Test compounds
DDGP, ABN, and MPL were isolated from ethyl acetate extracts of G. parva and well characterized Figure 1.[6] These compounds were stocked in DMSO and made to the final concentrations containing 0.2% DMSO. The positive control was 10 μM DEX.
Figure 1.
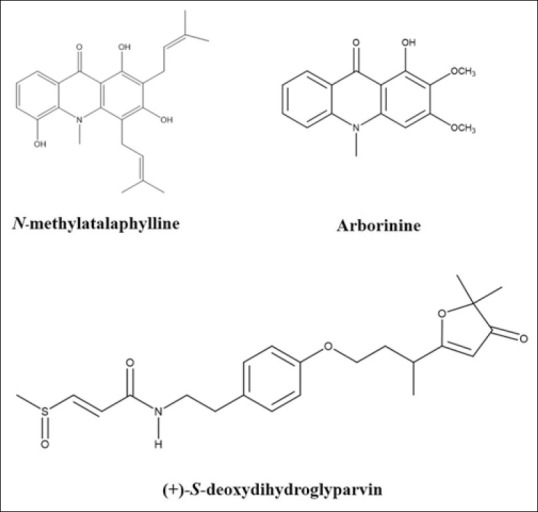
Structures of compounds isolated from ethyl acetate extracts of Glycosmis parva leaves and branches
Nitric oxide determination
LPS-activated cells were incubated with 3 and 10 μM of DDGP, ABN, and MPL for 24 h. Nitric oxide (NO) level was assessed by the Griess reaction. Concentrations of NO were derived from the standard curve of standard nitrite solutions.
For determining the IC50 value of DDGP, LPS-activated cells were treated with DDGP at 0.3125–10 μM for determining NO production as mentioned above. After assessing the IC50, DDGP at 1.25, 2.5, 5, and 10 μM was used in all experiments.
Cell viability assay
The cytotoxic effect of the test compounds was evaluated by resazurin assay.[8] The remaining treated cells from NO determination were incubated with 50 μg/ml resazurin for 4 h at 37°C and analyzed the results of measurement at 570 nm and 600 nm.
Quantitative polymerase chain reaction
LPS-activated cells were treated with 1.25–10 μM DDGP for 4 h and/or 24 h at 37°C. Total RNA of the cells was collected and extracted using RNA isolation solution and converted to cDNA using Improm-II Reverse Transcription system. The cDNA samples were used to amplify genes of interest by real-time PCR in StepOnePlus real-time-PCR System (Thermo, USA) using qPCR green master mix and the primers of the investigated genes showed in Table 1. A loading control was β-actin. Levels of gene expression were quantified using 2-ΔΔ CT method and indicated as the percentage changes from LPS-activated control.
Table 1.
Primer sequences of the investigated genes for real-time polymerase chain reaction
| Gene | Forward primer 5’-3’ | Reverse primer 5’-3’ |
|---|---|---|
| TNF-α | TTGACCTCAGCGCTGAGTTG | CCTGTAGCCCACGTCGTAGC |
| IL-1β | CAGGATGAGGACATGAGCACC | CTCTGCAGACTCAAACTCCAC |
| IL-6 | AGGACCAAGACCATCCAATTCA | GCTTAGGCATAACGCACTAGG |
| MCP-1 | ACTGAAGCCAGCTCTCTCTTCCTC | TTCCTTCTTGGGGTCAGCACAGAC |
| MIP-1α | GCCCTTGCTGTTCTTCTCTGT | GCCCTTGCTGTTCTTCTCTGT |
| iNOS | CCCTTCCGAAGTTTCTGGCAG | GGTGTCAGAGCCTCGTGGCT |
| COX-2 | CACTACATCCTGACCCACTT | ATGCTCCTGCTTGAGTATGT |
| mPGES-1 | CAGCCTAATGTTCAGCGACA | CCTTGAGCTGACAGCCTACC |
| Β-actin | GTGGGCCGCCCTAGGCACCAG | GGAGGAAGAGGATGCGGCAGT |
TNF-α: Tumor necrosis factor-α, IL: Interleukin, MCP-1: Monocyte chemoattractant protein-1, MIP-1α: Macrophage inflammatory proteins-1α, iNOS: Inducible nitric oxide synthase, COX-2: Cyclooxygenase-2
Enzyme-linked immunosorbent assay
LPS-activated cells were incubated with 1.25–10 μM DDGP for 24 h at 37°C. The concentrations of IL-1β, IL-6, TNF-α, and PGE2 were assessed with ELISA kits followed the manufacturer manuals.
Western blot analysis
LPS-activated cells were treated with 1.25–10 μM DDGP for 30 min. Whole cell proteins were isolated using RIPA lysis buffer. Cytosolic and nuclear proteins were fractionated using cell fractionation kit. Ten micrograms protein per sample was run in 7.5% sodium dodecyl sulfate-polyacrylamide gel and transferred to a nitrocellulose membrane. The membrane was blocked and blotted with appropriated specific primary antibodies against ERK, JNK, p38, IKB, p65 NF-κB, GAPDH, PCNA, phosphorylated ERK, phosphorylated JNK, and phosphorylated p38 (1:1000 dilution) at 4°C overnight, washed, blotted with HRP secondary antibody (1:2000 dilution) for 1 h, washed, and added HRP substrate. The density of each protein was determined using a chemiluminescence detector (C-DiGit® Blot Scanner, USA).
Statistical analysis
The data are shown as mean with the standard error of the mean of three independent experiments. Results of tested compounds were compared to the suitable control using one-way analysis of variance followed by post hoc test (Turkey's). IBM SPSS software version 22 (IBM Corp., USA) was utilized.
RESULTS
Effects of the compounds on nitric oxide production
We first evaluated the inhibitory potential of MPL, ABN, and DDGP on LPS-induced macrophage activation. DDGP had the most inhibitory effect on NO production [Figure 2a] without any cytotoxicity Figure 2b. At 10 μM, MPL, ABN, and DDGP inhibited NO production to 52%, 78%, and 19%, respectively, compared to the LPS-activated control. DDGP demonstrated a good candidate to investigate its anti-inflammatory activity.
Figure 2.

Effects of N-methylatalaphylline, arborinine, and S-deoxydihydroglyparvin on nitric oxide production (a) and cell viability (b) in lipopolysaccharide-activated RAW 264.7 macrophages
Effect of DDGP on nitric oxide production
We identified the optimal concentrations of DDGP for evaluating its anti-inflammatory activity in detail by determining IC50 value of NO inhibition. DDGP suppressed NO production with IC50 3.47 ± 0.1 μM [Figure 3a]. DDGP at 1.25–10 μM was chosen for further evaluation. The suppressive effect of DDGP on NO generation was confirmed by determining iNOS expression, which is an inducible enzyme for NO generation. iNOS did not express in resting macrophages [Figure 3b]. DDGP decreased iNOS mRNA level in the LPS-activated cells in a similar pattern to its effect on NO production [Figure 3b].
Figure 3.

Effects of S-deoxydihydroglyparvin on nitric oxide production (a) and inducible nitric oxide synthase expression (b) in lipopolysaccharide-activated RAW 264.7 macrophages
Effect of S-deoxydihydroglyparvin on the pro-inflammatory cytokines
We elucidated the possible target site of DDGP beyond iNOS/NO production by investigating its effect on pro-inflammatory cytokines. The mRNA and protein levels of key cytokines including IL-1, IL-6, and TNF-α were evaluated by real-time PCR and ELISA. DDGP downregulated the expression of these cytokines in LPS-activated cells after 4 [Figure 4.1a’c] and 24 h [Figure 4.2a’c]. It also decreased protein levels of these cytokines in the supernatant [Figure 4.3a’c]. These results imply that DDGP may act at the early step of LPS-induced macrophage activation by suppressing the protein levels of pro-inflammatory cytokines, which can induce the generation of chemokines, enzymes, and molecules involved in the inflammatory process.
Figure 4.
Effects of S-deoxydihydroglyparvin on pro-inflammatory cytokine expression and production. The expression of tumor necrosis factor-α (A), interleukin-1β (B), and interleukin-6 (C) was determined after 4 h (4.1) and 24 h (4.2) of treatment. The cytokine levels were determined after 24 h exposure (4.3)
Effect of S-deoxydihydroglyparvin on chemokine expression
DDGP at 5 and 10 μM downregulated the mRNA expression of both monocyte chemoattractant protein-1 (MCP-1) shown in Figure 5a and macrophage inflammatory proteins-1α (MIP-1α) in Figure 5b in LPS-activated macrophages.
Figure 5.

Effects of S-deoxydihydroglyparvin on the expression of chemokines, monocyte chemoattractant protein-1 (a), and macrophage inflammatory proteins-1α. (b) In lipopolysaccharide-activated RAW 264.7 macrophages
Effect of S-deoxydihydroglyparvin on cyclooxygenase-2 and mPEGS-1 expression and on PGE2 production
DDGP significantly decreased mRNA levels of inducible enzymes, COX-2 [Figure 6a], and mPGES-1 [Figure 6b]. It also decreased the production of PGE2 [Figure 6c].
Figure 6.
Effects of S-deoxydihydroglyparvin on the expression of cyclooxygenase-2 (a), mPGES-1 (b), and the production of PGE2 (c) in lipopolysaccharide-activated RAW 264.7 macrophages
Effect of S-deoxydihydroglyparvin on mitogen-activated protein kinase signaling molecules and nuclear factor kappa B activation
To verify the mechanism of action, the proteins in regulation of macrophage activation were further verified by western blot analysis after treating the cells with DDGP for 30 min. DDGP profoundly suppressed the phosphorylation of p38 MAPK [Figure 7c]. At 10 μM, it significantly decreased ERK phosphorylation >50% compared to the LPS control [Figure 7a]. DDGP did not have an effect on JNK phosphorylation [Figure 7b] and NF-κB activation [Figure 8]. These results propose that DDGP suppresses LPS-induced macrophage activation mainly by blocking p38 signaling pathway.
Figure 7.
Effects of S-deoxydihydroglyparvin on lipopolysaccharide-induced phosphorylation of ERK (a), JNK (b), and p38 MAPK in RAW264.7 cells. GAPDH was used as a loading control
Figure 8.
Effects of S-deoxydihydroglyparvin on lipopolysaccharide-induced the degradation of cytosolic iκB (a) and the activation of nuclear factor kappa B p65 in the nucleus (b). GAPDH and PCNA were used as loading controls of cytosolic and nuclear proteins, respectively
DISCUSSION
Activated macrophages and their products are key targets for developing novel anti-inflammatory agents. LPS-induced macrophage activation is a classical model used for evaluating candidate anti-inflammatory compounds. LPS activates macrophages to generate pro-inflammatory cytokines which induce the production of chemokines, adhesion molecules, inducible enzymes iNOS, and COX-2 for NO and PGE2 production.[3,9] RAW264.7 macrophages were used in the present study for evaluating anti-inflammatory effects of ABN, MPL, and DDGP.
Only DDGP demonstrated a potent inhibitor on NO production in LPS-activated macrophages. In inflammatory process, NO is mainly generated by iNOS, which expresses in macrophages at activated stage.[10] DDGP suppressed NO production through downregulating iNOS expression. It was shown that LPS and pro-inflammatory cytokines induced iNOS expression through MAPK and NF-κB activation.[9] Excessive pro-inflammatory cytokine generation was shown to involve in pathogenesis and progression of many inflammatory diseases.[11,12] Therefore, inhibiting the induction as well as effects of pro-inflammatory cytokine should be a potential therapeutic strategy. We found that DDGP decreased the induction of TNFα, IL-6, and IL-1β by downregulating their gene expression. Production of these cytokines is the early step of LPS-induced macrophage activation. These cytokines amplify LPS activity by upregulating the production of chemokines, iNOS/NO, and COX-2/PGE2.[2,10,13] These results suggest that DDGP inhibits in the early step of LPS-induced macrophage activation.
DDGP downregulated the expression of MCP-1 and MIP-1α, which are potent chemokines for recruiting monocytes and macrophages to inflammation areas, leading to the continuation of the inflammatory process.[2,14] DDGP also downregulated mPGES-1 and COX-2 expression leading to the decrease of PGE2 production. In activated macrophage, COX-2 catalyzes arachidonic acid to PGH2 and mPGES-1 catalyzes PGH2 to PGE2.[13] At the inflamed site, activated macrophages produce high levels of PGE2, which is a potent inflammatory mediator.[15]
LPS acts through toll-like receptor-4 to activate two main signaling pathways, MAPK signaling pathways and NF-κB activation, to induce inflammation.[3,16,17] TNF-α and IL-1 also act through these two pathways to amplify the inflammatory process.[18,19] MAPKs are protein serine/threonine kinases with three subtypes: JNK, ERK, and p38 MAPK.[16] Many evidences revealed that activation of MAPKs and NF-κB upregulates pro-inflammatory cytokines and inflammatory mediators.[16,17] Several compounds demonstrated their anti-inflammatory effects through inactivating these pathways.[15,20] DDGP profoundly suppressed phosphorylated p38. It had little or no effect on ERK and JNK phosphorylation and on NF-κB activation.
Several reports suggested that p38 activation involved in inflammation by activating pro-inflammatory cytokines and COX-2 expression.[16,21] A strong link between the p38 activation and many inflammatory diseases was also reported.[22,23] Several natural compounds exhibited anti-inflammatory activities in part by inactivating p38.[15,20] Our results suggest that DDGP may inhibit the early step of LPS-induced macrophage activation through inactivating p38 MAPK, leading to the suppression of the production of inflammation-related cytokines and inflammatory mediators. [Figure 9].
Figure 9.
Proposed mechanisms of anti-inflammatory action of S-deoxydihydroglyparvin
CONCLUSION
We revealed for the first time that DDGP from G. parva inhibited LPS-induced macrophage activation by suppressing p38 MAPK activation. DDGP could be considered a potential anti-inflammatory agent.
Financial support and sponsorship
This study was supported by Ratchadapiseksompotch Fund, Faculty of Medicine, Chulalongkorn University, Grant number RA62/119 and the 90th Anniversary Fund of Chulalongkorn University.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Freire MO, Van Dyke TE. Natural resolution of inflammation. Periodontol 2000. 2013;63:149–64. doi: 10.1111/prd.12034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arango Duque G, Descoteaux A. Macrophage cytokines: Involvement in immunity and infectious diseases. Front Immunol. 2014;5:491. doi: 10.3389/fimmu.2014.00491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9:7204–18. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dinarello CA. Anti-inflammatory Agents: Present and Future. Cell. 2010;140:935–50. doi: 10.1016/j.cell.2010.02.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tasneem S, Liu B, Li B, Choudhary MI, Wang W. Molecular pharmacology of inflammation: Medicinal plants as anti-inflammatory agents. Pharmacol Res. 2019;139:126–40. doi: 10.1016/j.phrs.2018.11.001. [DOI] [PubMed] [Google Scholar]
- 6.Chansriniyom C, Ruangrungsi N, Lipipun V, Kumamoto T, Ishikawa T. Isolation of acridone alkaloids and N-[(4-monoterpenyloxy) phenylethyl]-substituted sulfur-containing propanamide derivatives from Glycosmis parva and their anti-herpes simplex virus activity. Chem Pharm Bull (Tokyo) 2009;57:1246–50. doi: 10.1248/cpb.57.1246. [DOI] [PubMed] [Google Scholar]
- 7.Buranabunwong N, Ruangrungsi N, Chansriniyom C, Limpanasithikul W. Ethyl acetate extract from Glycosmis parva leaf induces apoptosis and cell-cycle arrest by decreasing expression of COX-2 and altering BCL-2 family gene expression in human colorectal cancer HT-29 cells. Pharm Biol. 2015;53:540–7. doi: 10.3109/13880209.2014.931442. [DOI] [PubMed] [Google Scholar]
- 8.Anoopkumar-Dukie S, Carey JB, Conere T, O'sullivan E, van Pelt FN, Allshire A. Resazurin assay of radiation response in cultured cells. Br J Radiol. 2005;78:945–7. doi: 10.1259/bjr/54004230. [DOI] [PubMed] [Google Scholar]
- 9.Li X, Jiang S, Tapping RI. Toll-like receptor signaling in cell proliferation and survival. Cytokine. 2010;49:1–9. doi: 10.1016/j.cyto.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coleman JW. Nitric oxide in immunity and inflammation. Int Immunopharmacol. 2001;1:1397–406. doi: 10.1016/s1567-5769(01)00086-8. [DOI] [PubMed] [Google Scholar]
- 11.Smolen JS, Redlich K, Zwerina J, Aletaha D, Steiner G, Schett G. Pro-inflammatory cytokines in rheumatoid arthritis: Pathogenetic and therapeutic aspects. Clin Rev Allergy Immunol. 2005;28:239–48. doi: 10.1385/CRIAI:28:3:239. [DOI] [PubMed] [Google Scholar]
- 12.Ramji DP, Davies TS. Cytokines in atherosclerosis: Key players in all stages of disease and promising therapeutic targets. Cytokine Growth Factor Rev. 2015;26:673–85. doi: 10.1016/j.cytogfr.2015.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Park JY, Pillinger MH, Abramson SB. Prostaglandin E2 synthesis and secretion: The role of PGE2 synthases. Clin Immunol. 2006;119:229–40. doi: 10.1016/j.clim.2006.01.016. [DOI] [PubMed] [Google Scholar]
- 14.White GE, Iqbal AJ, Greaves DR. CC chemokine receptors and chronic inflammation-therapeutic opportunities and pharmacological challenges. Pharmacol Rev. 2013;65:47–89. doi: 10.1124/pr.111.005074. [DOI] [PubMed] [Google Scholar]
- 15.Choi YH, Kim GY, Lee HH. Anti-inflammatory effects of cordycepin in lipopolysaccharide-stimulated RAW 264.7 macrophages through Toll-like receptor 4-mediated suppression of mitogen-activated protein kinases and NF-κB signaling pathways. Drug Des Devel Ther. 2014;8:1941–53. doi: 10.2147/DDDT.S71957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hommes DW, Peppelenbosch MP, van Deventer SJ. Mitogen activated protein (MAP) kinase signal transduction pathways and novel anti-inflammatory targets. Gut. 2003;52:144–51. doi: 10.1136/gut.52.1.144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu T, Zhang L, Joo D, Sun SC. NF-κB signaling in inflammation. Signal Transduct Target Ther. 2017;2:17023. doi: 10.1038/sigtrans.2017.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kalliolias GD, Ivashkiv LB. TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nat Rev Rheumatol. 2016;12:49–62. doi: 10.1038/nrrheum.2015.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Acuner Ozbabacan SE, Gursoy A, Nussinov R, Keskin O. The structural pathway of interleukin 1 (IL-1) initiated signaling reveals mechanisms of oncogenic mutations and SNPs in inflammation and cancer. PLoS Comput Biol. 2014;10:e1003470. doi: 10.1371/journal.pcbi.1003470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bajpai VK, Alam MB, Quan KT, Ju MK, Majumder R, Shukla S, et al. Attenuation of inflammatory responses by (+)-syringaresinol via MAP-Kinase-mediated suppression of NF-κB signaling in vitro and in vivo. Sci Rep. 2018;8:9216. doi: 10.1038/s41598-018-27585-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang Y, Kim SC, Yu T, Yi YS, Rhee MH, Sung GH, et al. Functional roles of p38 mitogen-activated protein kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014;2014:352371. doi: 10.1155/2014/352371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hollenbach E, Neumann M, Vieth M, Roessner A, Malfertheiner P, Naumann M. Inhibition of p38 MAP kinase- and RICK/NF-kappaB-signaling suppresses inflammatory bowel disease. FASEB J. 2004;18:1550–2. doi: 10.1096/fj.04-1642fje. [DOI] [PubMed] [Google Scholar]
- 23.Johnson GV, Bailey CD. The p38 MAP kinase signaling pathway in Alzheimer's disease. Exp Neurol. 2003;183:263–8. doi: 10.1016/s0014-4886(03)00268-1. [DOI] [PubMed] [Google Scholar]







