Abstract
The absence of a specific treatment for SARS-CoV-2 infection led to an intense global effort in order to find new therapeutic interventions and improve patient outcomes. One important feature of COVID-19 pathophysiology is the activation of immune cells, with consequent massive production and release of inflammatory mediators that may cause impairment of several organ functions, including the brain. In addition to its classical role as a neurotransmitter, serotonin (5-hydroxytryptamine, 5-HT) has immunomodulatory properties, downregulating the inflammatory response by central and peripheral mechanisms. In this review, we describe the roles of 5-HT in the regulation of systemic inflammation and the potential benefits of the use of specific serotonin reuptake inhibitors as a coadjutant therapy to attenuate neurological complications of COVID-19.
Keywords: Serotonin, Cytokines, SARS-CoV-2, Inflammation
1. Introduction
Serotonin (5-hydroxytryptamine, 5-HT) has been associated with a plethora of neurological and physiological responses (Berger et al., 2009) and it also has immunomodulatory properties (Baganz and Blakely, 2013; Wu et al., 2019). We now know that different stages of SARS-CoV-2 infection take place, namely infection phase, asymptomatic or low-symptomatic phase, followed by a mildly symptomatic phase and eventually a severe phase (when an exacerbated inflammation is a major concern), which can be lethal (Ayres, 2020). One important aspect observed in severe patients is the positive association with cytokine release syndrome,i.e., an exacerbated systemic inflammatory response that may be triggered by infections, for instance (Chatenoud et al., 1989), in particular the increase of peripheral interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α levels. Interestingly, TNF-α levels highly correlate with SARS-CoV-2 load (Lucas et al., 2020) and IL-6 has been suggested as an early inflammatory marker to predict severe illness in patients with SARS-CoV-2 infection (Aziz et al., 2020; McElvaney et al., 2020; Tay et al., 2020). This hyper-inflammatory state that is also observed in the severe phase of SARS-CoV-2 infection can damage tissues beyond the lungs, including the brain, and has deleterious neurological and neuropsychiatric consequences (Wu et al., 2020; Yuan et al., 2020). The psychosocial effects of the striking economic and health system crises caused by the pandemic along with the pre-existing worldwide psychosocial factors must also be remembered as potential threats to psychiatric disorders (Ajilore and Thames, 2020; Batty et al., 2020; Lassale et al., 2020).
Given the broad spectrum of central and peripheral 5-HT actions and the urgency to find new perspectives for the management and treatment of COVID-19 patients, here we discuss the potential use of selective 5-HT reuptake inhibitors not only for treating anxiety/depressive disorders but also as an adjuvant therapy to decrease SARS-CoV-2-induced exacerbated inflammatory response.
2. Central 5-HT and its role in systemic inflammation
Serotonin arises from tryptophan metabolism and plays different roles in the brain. Sleep, body temperature, motor control and several behavioral effects (mood, memory, fear, perception, stress) are some of the central functions regulated by 5-HT (Berger et al., 2009). This neurotransmitter is mainly known due to the intense research over the last 40 years investigating the modulatory effects of serotonergic system by exploiting the effects of certain drugs aimed at the treatment of depression, a key area of research in psychiatry (Mann, 1999). Moreover, it is well-known that 5-HT is effective not only to treat depression but also to decrease the susceptibility to depression and suicide (Li and He, 2007). Interestingly, stimulation of the serotonergic pathway is thought to be so beneficial that the use of non-pharmacological methods to increase 5-HT levels have been suggested as a potential strategies to prevent depression and/or improve mood and social functioning, by means of exercise (Sbolli et al., 2020) or exposure to bright light (Harrison et al., 2015), for instance.
2.1. Central 5-HT in severe systemic inflammation (as observed in cytokine release syndrome)
Adding to this scenario, we have recently reported the potential effects of central 5-HT in lipopolysaccharide (LPS)-induced severe septic like systemic inflammation (Mota et al., 2019). We have found reduced 5-HT levels in the hypothalamus that coincided with hypotension, increased heart rate, hypothermia followed by fever, increased plasma prostaglandin E2, nitric oxide, and cytokine levels after LPS intravenous administration. Interestingly, exogenous 5-HT given intracerebroventricularly prevented hypotension, reduced plasma nitric oxide (NO), blunted hypothermia, and potentiated fever, which were associated with reduced LPS-induced TNF-α, IL-6, and IL-1β surges.
2.2. Central 5-HT in mild systemic inflammation
These aforementioned data add support to previous studies about the role of brain 5-HT during systemic inflammation (Mota et al., 2017; Voronova et al., 2016). The role of central 5-HT in inflammation was first described by evaluating serotonin 5-HT2A receptor activation reversing LPS-induced neuroinflammation by attenuating NO levels (Voronova et al., 2016). However, the first anti-inflammatory effect in the periphery in a model of LPS-induced mild systemic inflammation was described by Mota et al. (2017), by observing that intracerebroventricular injection of 5-HT effectively reduces pro-inflammatory cytokines surges.
2.3. Central 5-HT in low-grade systemic inflammation
Low-grade systemic inflammation is characterized by sustained low-levels of some peripheral cytokines. This phenomenon is observed in a myriad of diseases such as neuropsychiatric and metabolic disorders (Minihane et al., 2015; Osimo et al., 2019; Rorato et al., 2017). There is an understanding that this inflammatory response has a significant impact in neuropsychiatric alterations (Heneka et al., 2015; Miller and Raison, 2016; Perry, 2010; Shastri et al., 2013) and metabolic disorders (Hotamisligil, 2006) in some cases. Importantly, adipose macrophage abundance is correlated with adiposity in high-fat diet (HFD) model (Weisberg et al., 2003). HFD also increases hypothalamic inflammation (De Souza et al., 2005; Milanski et al., 2009). Interestingly, obesity is associated with a range of neurodegenerative and psychiatric disorders, such as anxiety and depression (Gariepy et al., 2010; Hryhorczuk et al., 2013). In a systematic review about obesity as a factor that raises the risk of developing anxiety disorders, a mild association was observed between these two disorders (Gariepy et al., 2010). More recently, it was speculated that one of the connections between obesity and anxiety is inflammation (Capuron and Miller, 2011; Lasselin and Capuron, 2014), and metabolic disorders have been suggested as a link between obesity and depression (Hryhorczuk et al., 2013).
This is relevant information since severe SARS-CoV-2 infection has also been correlated with increased body mass index (Lucas et al., 2020) which can also be epidemiologically translated by the high death prevalence between patients with metabolic disorders (KHAN et al., 2020 preprint). It is suggested that the pre-existing underlying inflammation in obese patients may result in a dysfunctional immune response during COVID-19, predisposing to a more severe condition (Seidu et al., 2020). Obesity is often associated with type 2 diabetes and it has been shown that uncontrolled glucose levels promote SARS-CoV-2 replication, increase cytokine release and induce T cell dysfunction (Codo et al., 2020; Seidu et al., 2020). Hypercoagulopathy, an important component of COVID-19 pathophysiology, is also exacerbated in people with obesity (Pasquarelli-do-Nascimento et al., 2020). Metabolic disorders have increased in low and middle-income countries populations (Miranda et al., 2019), which are also greatly impacted by the economic crises initiated by the pandemic outbreak.
There are plenty of articles using HFD as a pre-clinical model to evaluate depressive and anxiogenic-like behavior in obesity (Haleem and Mahmood, 2019; Kim et al., 2013; Zemdegs et al., 2016). Most of them have demonstrated that HFD decreases plasma and brain 5-HT levels (Haleem and Mahmood, 2019; Kesic et al., 2020) followed by an increase in anxiogenic and depressive-like behaviors (de Noronha et al., 2017; Gomes et al., 2020; Zemdegs et al., 2016). Interestingly, peripheral and central 5-HT have opposite roles on metabolic homeostasis: while circulating 5-HT favors the onset of obesity (Crane et al., 2015), brain serotonin diminishes weight gain by regulating food intake and brown adipose tissue activity (Kesic et al., 2020). Pharmacological interventions that restore central serotoninergic levels in obese people could be helpful not only to regulate immune response during an infection, like COVID-19, but also to treat and prevent consequent neuropsychiatric alterations.
Altogether, taking the effect of central 5-HT in attenuating severe septic like inflammation and mild systemic inflammation, we believe that this decrease in central 5-HT levels might have a role in low-grade inflammation observed in these disorders.
Reconciling available data in the literature, a potential central effect of 5-HT on systemic inflammation is suggested and the mechanisms would involve inhibition of pro-inflammatory cytokines production that would compensate a reduced endogenous central serotonin production that take place in systemic inflammation.
2.4. Central 5-HT and its anti-inflammatory mechanism
One may wonder how does brain 5-HT exerts its anti-inflammatory effects in the periphery in such an effective way that LPS-induced hypotension and hypothermia are abolished. The answer may reside in the finding that pro-inflammatory responses seem to actually be modulated by neural circuits that link the areas of physiology, immunology, and neuroscience (Martelli et al., 2014; Okusa et al., 2017; Tracey, 2002). The “inflammatory reflex” has been documented as an effective anti-inflammatory mechanism in systemic inflammation (Tracey, 2002). The afferent pathway of the “inflammatory reflex” relies on the vagal nerve that is sensitized by circulating cytokines (Komegae et al., 2018), leading to reduced production of pro-inflammatory cytokines in the spleen (Okusa et al., 2017). Mota et al. (2019) showed that 5-HT may activate the splenic inflammatory reflex, since i.c.v. 5-HT administration reduced the production of spleen TNF-α, which is in agreement with the notion that specific signaling pathways inhibit spleen TNF-α production (de Jonge et al., 2005; Lu et al., 2014). The effect of hypothalamic 5-HT has been elegantly reviewed by Rummel (2019).
3. Peripheral 5-HT and systemic inflammation
Besides the central effects of 5-HT, it is important to mention that most of the body's 5-HT is actually found in the circulating platelets of the bloodstream (Berger et al., 2009) and this peripheral 5-HT plays several roles. Systemic 5-HT arises from the enterochromaffin cells of the gastrointestinal tract. Under resting conditions, plasma 5-HT concentration is about 10 nmol, but it increases to micromolar levels when platelets become activated by inflammation, for instance (Mossner and Lesch, 1998). Once released, 5-HT functions are mediated by at least 7 (so far documented in mammals) serotonin 5-HT receptors classes (5-HT1 through 5-HT7), as well as by the 5-HT transporter (SERT) located presynaptically on serotonergic axon terminals.
Circulating serotonin modulates numerous functions not only in the cardiovascular system, respiration and gastrointestinal functions but also in nociception, coagulation and reproduction (Berger et al., 2009). In addition to regulating hemostasis, several immunoregulatory functions of peripheral 5-HT have been reported to be mediated through a direct effect of 5-HT on circulating immune cells (Roszman et al., 1985).
These immune cells express different serotonin receptors subtypes, SERT, the enzymes for serotonin synthesis, i.e., tryptophan hydroxylase, and for serotonin degradation, i.e., monoamine oxidase (Herr et al., 2017). In the innate immune system, the first line of defense against pathogens, it has been shown that 5-HT inhibits LPS-induced proinflammatory cytokines and induces M2 (anti-inflammatory) polarization of human macrophages through serotonin 5-HT2B and 5-HT7 receptors (Quintero-Villegas and Valdes-Ferrer, 2019). The use of a serotonin 5-HT7 receptor agonist in experimental sepsis reduces plasma IL-1β and IL-6 and also lung NFκB, increasing survival (Cadirci et al., 2013). Dendritic cells express several receptor subtypes and are able to use serotonin to promote T-cell proliferation and naive T-cell differentiation, showing a serotoninergic action also in adaptative immunity (O'Connell et al., 2006). Interestingly, some studies report that serotonin can have inflammatory effects in the gut through serotonin 5-HT3 and 5-HT4 receptors (Salaga et al., 2019), showing that serotoninergic action is tissue- and receptor-specific.
4. 5-HT and viral infections
The bidirectional communication between the serotoninergic system and viral infections has also been investigated, although to a lesser extent. Polyinosinic: polycytidylic acid (poly I:C) is a potent immunostimulant and has been used as a methodological approach to mimic viral infections, given its interaction with toll-like receptor 3 (TLR3), which is involved in the activation of the immune response following virus recognition (Lever et al., 2015). Systemic administration of poly I:C increases the expression of the serotonin transporter (SERT) and reduces extracellular 5-HT in the prefrontal cortex (PFC) of rats, an area associated with essential cognitive and limbic functions (Katafuchi et al., 2006). The increased SERT expression in astrocytes of this brain region is at least partially mediated by TLR3 pathway signaling, microglial activation, and IL-1β production (Ifuku et al., 2014). Furthermore, poly I:C increases tryptophan in the hippocampus and PFC, but this amino acid is directed to the kynurenine pathway which produces several metabolites that may disrupt normal brain function (Gibney et al., 2013).
Peripheral 5-HT is also able to modulate several mechanisms of different viral infections. Subpopulations of human dendritic cells express serotonin 5-HT2B receptor that when activated suppresses the production of inflammatory cytokines and chemokines during a viral stimulus (Szabo et al., 2018). Serotonin also increases the cytotoxicity of natural killers (NK) cells, probably through serotonin 5-HT1A receptor (Hellstrand and Hermodsson, 1990).
The role of 5-HT in the immune response in specific viral infections has also been addressed. For example, 5-HT decreases HIV infection in human macrophages by down-regulating the expression of CCR5, an essential receptor for virus entry in the cell, probably via serotonin 5-HT1A receptor (Maneglier et al., 2008). Additionally, buspirone, a serotonin-receptor agonist, was able to decrease T-CD8 cell count and increase CD4/CD8 ratios in HIV-infected patients (Eugen-Olsen et al., 2000). Inhibition of chikungunya (Bouma et al., 2020), reovirus (Mainou et al., 2015), and parainfluenza (Rabbani and Barik, 2017) infection has also been shown to be mediated by this monoamine. However, 5-HT immunomodulatory actions may be virus and/or receptor-dependent, as exemplified by JC virus infection, where virus internalization is stimulated by serotonin 5-HT2A receptors (Assetta et al., 2013; O'Hara and Atwood, 2008).
5SARS-CoV-2 and neuropsychiatry disorders
The outbreak of the COVID-19 pandemic raised global attention after the rapid spread of this disease, reaching more than 15 million cases and 600,000 deaths less than eight months after the first documented cases in December 2019 in Wuhan, China. The infection by this new virus, named SARS-CoV-2 led to worldwide efforts to rapidly understand the pathophysiology of this disease and the search for efficient treatments.
Primary targets of SARS-CoV-2 are airway epithelial cells and pneumocytes, which are invaded after the virus binding to the angiotensin-converting enzyme 2 (ACE2) receptor and TMPRSS2 activation (Hoffmann et al., 2020). Following replication in the infected cells, new virions are released and this acute and severe infection promotes initially a local inflammatory response, with the release of proinflammatory cytokines (Wiersinga et al., 2020). Macrophages and mononuclear cells infiltrate alveolar tissue and induce edema formation, which may explain the respiratory symptoms of this disease, like cough, difficulty breathing, and even pneumonia (Wiersinga et al., 2020; Xu et al., 2020).
This progressive and dysregulated host response to infection, with a sustained innate inflammatory response, extrapolate to a systemic level, with progressive immune effects and excessive circulating inflammatory cytokines. Plasma concentrations of IL-1β, IL-1 receptor antagonist, IL-6, IL-8, IL-10, IL-17, IFN-γ, and other mediators are increased in severe COVID-19 patients (Huang et al., 2020). At the cellular level, it is reported that SARS-CoV-2 infection decreases TCD4+ and TCD8+ lymphocytes, monocytes and eosinophils (Chen et al., 2020; Qin et al., 2020; Zhao et al., 2020). Patients with metabolic disorders (e.g. obesity and type 2 diabetes) are at a higher risk of developing a more severe form of the disease, since they are predisposed to an even more uncontrolled inflammatory response, with additional production of cytokines and deficient cell immunity in COVID-19 and other infections (Andersen et al., 2016; Codo et al., 2020).
This abnormal immune state and the cytokine release syndrome play an important role in the clinical manifestations, including neurological alterations. One of the early symptoms in patients with COVID-19 is the loss or reduction of smell and taste (Lechien et al., 2020; Spinato et al., 2020). Although not yet proved to occur in humans, SARS-CoV-2 is able to invade the olfactory bulb of transgenic mice expressing ACE2 receptor and spread to other brain regions (Netland et al., 2008).
Some of the most common complications of SARS-CoV-2 infection are the cerebrovascular events, mainly ischemic stroke (Beyrouti et al., 2020; Bridwell et al., 2020). These events could be associated with coagulation alterations, given that COVID-19 infection is characterized by high fibrinogen and D-dimer (a fibrin degradation product) concentrations that lead to a prothrombotic state and disseminated intravascular coagulation (Goshua et al., 2020). Cytokine release syndrome is a major component of coagulopathy since it activates the coagulation cascade and promotes endothelial dysfunction (Colantuoni et al., 2020).
The inadequate blood supply and concomitant impaired pulmonary function may critically decrease cerebral oxygenation and have deleterious consequences in brain function. Low oxygen levels may result in tissue hypoxia, which further causes cell death, activation of brain immune cells, oxidative stress and the consequent production of inflammatory mediators, like cytokines and chemokines (Liu and McCullough, 2013). Post-mortem analysis of COVID-19 patients revealed astrocytosis and loss of neurons in the hippocampus, cerebral cortex, and cerebellum (Solomon et al., 2020). Increased cytokine release during COVID-19 could induce the onset of cerebrovascular and neurological alterations or worsen pre-existing conditions, since these disorders are associated with the production of inflammatory mediators (Deleidi and Isacson, 2012; Ellul et al., 2020).
In addition to neurological disorders, neuropsychiatric complications are also a concern in SARS-CoV-2 infection. Individual reports and case series have described alterations including delirium, mild cognitive impairment, psychosis, and mood swings (Dinakaran et al., 2020). A nationwide surveillance study identified altered mental status in 31% of COVID-19 patients, including syndromic diagnosis like encephalitis but also primary psychiatric disorders like psychosis, dementia, and mania (Varatharaj et al., 2020).
6. Therapeutic perspectives
The rapid spread of the disease and the absence of immediate therapeutic interventions to effectively treat SARS-CoV-2 infection led the scientific and medical community to rethink the use of already available drugs in order to improve clinical outcomes. In this scenario, the use of selective 5-HT reuptake inhibitors (SSRI) could be considered an adjuvant in COVID-19 pharmacological therapy.
This class of drugs was launched in the market more than three decades ago and has well described pharmacodynamic and pharmacokinetic properties, making it a safer option as a possible treatment. Clinical and experimental studies support the hypothesis that 5-HT could help to dampen the excessive production of cytokines during the systemic inflammatory condition caused by COVID-19 and diminish its deleterious consequences. Serotonin could not only act directly in circulating peripheral immune cells by binding to specific serotonin 5-HT receptors (Herr et al., 2017) but also through central neural mechanisms like the anti-inflammatory vagal reflex (Mota et al., 2019).
Selective 5-HT reuptake inhibitors increase brain 5-HT availability by crossing the blood-brain barrier and inhibiting central SERT (Hervas and Artigas, 1998), but it has been shown that vagus nerve stimulation can augment central production of 5-HT in some brain areas, indicating an alternative neural mechanism of monoaminergic system control (Manta et al., 2013). It must be highlighted that the decrease of anxiety and depressive-like symptoms during fluoxetine and sertraline treatment is partially dependent on indirect CNS activity by vagus nerve signaling (McVey Neufeld et al., 2019) and that vagal stimulation has been recently described as a therapeutic approach to treat depression (Aaronson et al., 2017; Krahl et al., 2004). Interestingly, one main feature of vagal stimulation is systemic inflammation attenuation (Pavlov and Tracey, 2012). However, more studies must be conducted to evaluate if SSRI/vagus association might also have a role increasing central 5-HT levels and thus, attenuating systemic inflammation.
In agreement with this perspective, fluoxetine (the first and one of the most prescribed 5-HT reuptake inhibitors) inhibits viral replication (Bauer et al., 2019; Zuo et al., 2012) and increases NK cells activity in HIV patients (Evans et al., 2008; Frank et al., 1999). Centrally, this drug inhibits microglial activation and decreases cytokine production by these cells (Liu et al., 2011). Interestingly, an in vitro study showed that fluoxetine has a specific action inhibiting SARS-CoV-2 infection (Zimniak et al., 2020 preprint). A clinical trial (NCT04377308) is currently investigating the effect of this drug on clinical outcomes and cytokine storm in COVID-19 patients. Obviously, other selective 5-HT reuptake inhibitors may also regulate immune function during viral infections (Greeson et al., 2016; Jiang et al., 2014; Sharma et al., 2013).
Furthermore, selective 5-HT reuptake inhibitors may modulate the inflammatory response not only by direct serotonergic mechanisms. Fluvoxamine, for example, has significant immunomodulatory properties by downregulating inflammatory pathways and cytokine production in monocytes through its high affinity to sigma-1 receptor (S1R) (Rosen et al., 2019). Also, interestingly, an ongoing clinical trial (NCT04342663) is currently exploring the potential benefits of this drug in COVID-19. Several other selective 5-HT reuptake inhibitors interact with S1R and could also have anti-inflammatory properties (Ishima et al., 2014).
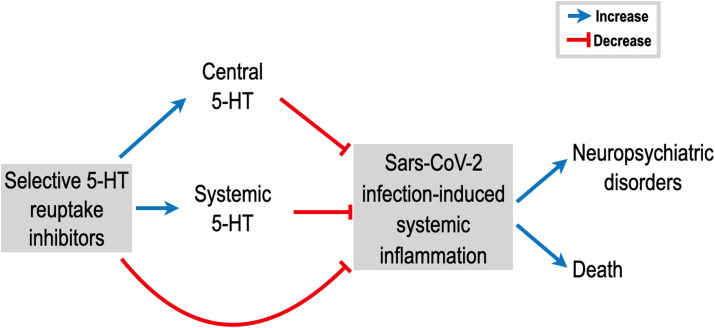
To summarize, there is significant evidence in the scientific literature to encourage the use of 5-HT reuptake inhibitors as a complementary intervention during SARS-CoV-2 infection. The direct immunomodulatory action of central and peripheral 5-HT, associated with other indirect mechanisms, could effectively dampen the exacerbated immune response and prevent neurological complications (Fig. 1 ). Additionally, it would be relevant to consider that patients under long-term SSRI therapy should continue the use of their medication when hospitalized due to COVID-19.
Fig. 1.
Potential role of selective serotonin (5-HT) reuptake inhibitors decreasing systemic inflammation induced by SARS-CoV-2 infection and ameliorating patient prognosis. The excessive production of inflammatory mediators may be attenuated by augmenting central and systemic 5-HT levels and also by the activation of non-serotoninergic pathways in immune cells. We believe that 5-HT reuptake inhibitors can blunt the exacerbated immune response in COVID-19 and ameliorate clinical consequences, such as the development of neuropsychiatric disorders and death.
As always, for any treatment, the adverse effects must be kept in mind, especially when patients are being treated with several combined drugs that may increase the risk to develop serotonin syndrome, for example (Mas Serrano et al., 2020). Serotonin syndrome (SS) is a potentially lethal drug-induced disorder caused by serotoninergic over-activity at synapses of both central and peripheral nervous systems (Scotton et al., 2019). In SS an altered mental state, tachycardia, hyperthermia, hyperreflexia with clonus, ocular clonus, myoclonus, tremors, or rigidity may be observed (Buckley et al., 2014). This serious condition is usually caused by the combination of selective 5-HT reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors, monoamine oxidase inhibitors, opiates, or tricyclic antidepressants among others drugs (Scotton et al., 2019).
Funding
This work was financially supported by Grants #2019/27231-9 and #2016/17681-9, São Paulo Research Foundation (FAPESP) and by Grants # 301040/2018-4 and # 404927/2018-1 National Council for Scientific and Technological Development (CNPq), Brazil.
Declaration of competing interest
None.
References
- Aaronson S.T., Sears P., Ruvuna F., Bunker M., Conway C.R., Dougherty D.D., Reimherr F.W., Schwartz T.L., Zajecka J.M. A 5-year observational study of patients with treatment-resistant depression treated with vagus nerve stimulation or treatment as usual: comparison of response, remission, and suicidality. Am. J. Psychiatr. 2017;174:640–648. doi: 10.1176/appi.ajp.2017.16010034. [DOI] [PubMed] [Google Scholar]
- Ajilore O., Thames A.D. The fire this time: the stress of racism, inflammation and COVID-19. Brain Behav. Immun. 2020 doi: 10.1016/j.bbi.2020.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen C.J., Murphy K.E., Fernandez M.L. Impact of obesity and metabolic syndrome on immunity. Adv Nutr. 2016;7:66–75. doi: 10.3945/an.115.010207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assetta B., Maginnis M.S., Gracia Ahufinger I., Haley S.A., Gee G.V., Nelson C.D., O'Hara B.A., Allen Ramdial S.A., Atwood W.J. 5-HT2 receptors facilitate JC polyomavirus entry. J. Virol. 2013;87:13490–13498. doi: 10.1128/JVI.02252-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ayres J.S. A metabolic handbook for the COVID-19 pandemic. Nat Metab. 2020;2:572–585. doi: 10.1038/s42255-020-0237-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aziz M., Fatima R., Assaly R. Elevated interleukin-6 and severe COVID-19: a meta-analysis. J. Med. Virol. 2020 doi: 10.1002/jmv.25948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baganz N.L., Blakely R.D. A dialogue between the immune system and brain, spoken in the language of serotonin. ACS Chem. Neurosci. 2013;4:48–63. doi: 10.1021/cn300186b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batty G.D., Deary I.J., Luciano M., Altschul D.M., Kivimaki M., Gale C.R. Psychosocial factors and hospitalisations for COVID-19: prospective cohort study based on a community sample. Brain Behav. Immun. 2020 doi: 10.1016/j.bbi.2020.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer L., Manganaro R., Zonsics B., Strating J., El Kazzi P., Lorenzo Lopez M., Ulferts R., van Hoey C., Mate M.J., Langer T., Coutard B., Brancale A., van Kuppeveld F.J.M. Fluoxetine inhibits enterovirus replication by targeting the viral 2C protein in a stereospecific manner. ACS Infect. Dis. 2019;5:1609–1623. doi: 10.1021/acsinfecdis.9b00179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger M., Gray J.A., Roth B.L. The expanded biology of serotonin. Annu. Rev. Med. 2009;60:355–366. doi: 10.1146/annurev.med.60.042307.110802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beyrouti R., Adams M.E., Benjamin L., Cohen H., Farmer S.F., Goh Y.Y., Humphries F., Jager H.R., Losseff N.A., Perry R.J., Shah S., Simister R.J., Turner D., Chandratheva A., Werring D.J. Characteristics of ischaemic stroke associated with COVID-19. J. Neurol. Neurosurg. Psychiatry. 2020;91:889–891. doi: 10.1136/jnnp-2020-323586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouma E.M., van de Pol D.P.I., Sanders I.D., Rodenhuis-Zybert I.A., Smit J.M. Serotonergic drugs inhibit chikungunya virus infection at different stages of the cell entry pathway. J. Virol. 2020;94 doi: 10.1128/JVI.00274-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bridwell R., Long B., Gottlieb M. Neurologic complications of COVID-19. Am. J. Emerg. Med. 2020;38:1549 e1543–1549 e1547. doi: 10.1016/j.ajem.2020.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckley N.A., Dawson A.H., Isbister G.K. Serotonin syndrome. BMJ. 2014;348:g1626. doi: 10.1136/bmj.g1626. [DOI] [PubMed] [Google Scholar]
- Cadirci E., Halici Z., Bayir Y., Albayrak A., Karakus E., Polat B., Unal D., Atamanalp S.S., Aksak S., Gundogdu C. Peripheral 5-HT7 receptors as a new target for prevention of lung injury and mortality in septic rats. Immunobiology. 2013;218:1271–1283. doi: 10.1016/j.imbio.2013.04.012. [DOI] [PubMed] [Google Scholar]
- Capuron L., Miller A.H. Immune system to brain signaling: neuropsychopharmacological implications. Pharmacol. Ther. 2011;130:226–238. doi: 10.1016/j.pharmthera.2011.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatenoud L., Ferran C., Reuter A., Legendre C., Gevaert Y., Kreis H., Franchimont P., Bach J.F. Systemic reaction to the anti-T-cell monoclonal antibody OKT3 in relation to serum levels of tumor necrosis factor and interferon-gamma [corrected] N. Engl. J. Med. 1989;320:1420–1421. doi: 10.1056/NEJM198905253202117. [DOI] [PubMed] [Google Scholar]
- Chen G., Wu D., Guo W., Cao Y., Huang D., Wang H., Wang T., Zhang X., Chen H., Yu H., Zhang X., Zhang M., Wu S., Song J., Chen T., Han M., Li S., Luo X., Zhao J., Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Codo A.C., Davanzo G.G., Monteiro L.B., de Souza G.F., Muraro S.P., Virgilio-da-Silva J.V., Prodonoff J.S., Carregari V.C., de Biagi Junior C.A.O., Crunfli F., Jimenez Restrepo J.L., Vendramini P.H., Reis-de-Oliveira G., Bispo Dos Santos K., Toledo-Teixeira D.A., Parise P.L., Martini M.C., Marques R.E., Carmo H.R., Borin A., Coimbra L.D., Boldrini V.O., Brunetti N.S., Vieira A.S., Mansour E., Ulaf R.G., Bernardes A.F., Nunes T.A., Ribeiro L.C., Palma A.C., Agrela M.V., Moretti M.L., Sposito A.C., Pereira F.B., Velloso L.A., Vinolo M.A.R., Damasio A., Proenca-Modena J.L., Carvalho R.F., Mori M.A., Martins-de-Souza D., Nakaya H.I., Farias A.S., Moraes-Vieira P.M. Elevated glucose levels favor SARS-CoV-2 infection and monocyte response through a HIF-1 alpha/Glycolysis-Dependent Axis. Cell Metabol. 2020;32:498–499. doi: 10.1016/j.cmet.2020.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colantuoni A., Martini R., Caprari P., Ballestri M., Capecchi P.L., Gnasso A., Lo Presti R., Marcoccia A., Rossi M., Caimi G. COVID-19 sepsis and microcirculation dysfunction. Front. Physiol. 2020;11:747. doi: 10.3389/fphys.2020.00747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crane J.D., Palanivel R., Mottillo E.P., Bujak A.L., Wang H., Ford R.J., Collins A., Blumer R.M., Fullerton M.D., Yabut J.M., Kim J.J., Ghia J.E., Hamza S.M., Morrison K.M., Schertzer J.D., Dyck J.R., Khan W.I., Steinberg G.R. Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat. Med. 2015;21:166–172. doi: 10.1038/nm.3766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Jonge W.J., van der Zanden E.P., The F.O., Bijlsma M.F., van Westerloo D.J., Bennink R.J., Berthoud H.R., Uematsu S., Akira S., van den Wijngaard R.M., Boeckxstaens G.E. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat. Immunol. 2005;6:844–851. doi: 10.1038/ni1229. [DOI] [PubMed] [Google Scholar]
- de Noronha S.R., Campos G.V., Abreu A.R., de Souza A.A., Chianca D.A., Jr., de Menezes R.C. High fat diet induced-obesity facilitates anxiety-like behaviors due to GABAergic impairment within the dorsomedial hypothalamus in rats. Behav. Brain Res. 2017;316:38–46. doi: 10.1016/j.bbr.2016.08.042. [DOI] [PubMed] [Google Scholar]
- De Souza C.T., Araujo E.P., Bordin S., Ashimine R., Zollner R.L., Boschero A.C., Saad M.J., Velloso L.A. Consumption of a fat-rich diet activates a proinflammatory response and induces insulin resistance in the hypothalamus. Endocrinology. 2005;146:4192–4199. doi: 10.1210/en.2004-1520. [DOI] [PubMed] [Google Scholar]
- Deleidi M., Isacson O. Viral and inflammatory triggers of neurodegenerative diseases. Sci. Transl. Med. 2012;4:121ps123. doi: 10.1126/scitranslmed.3003492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinakaran D., Manjunatha N., Naveen Kumar C., Suresh B.M. Neuropsychiatric aspects of COVID-19 pandemic: a selective review. Asian J Psychiatr. 2020;53:102188. doi: 10.1016/j.ajp.2020.102188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellul M., Varatharaj A., Nicholson T.R., Pollak T.A., Thomas N., Easton A., Zandi M.S., Manji H., Solomon T., Carson A., Turner M.R., Kneen R., Galea I., Pett S., Thomas R.H., Michael B.D., CoroNerve Steering C. Defining causality in COVID-19 and neurological disorders. J. Neurol. Neurosurg. Psychiatry. 2020;91:811–812. doi: 10.1136/jnnp-2020-323667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eugen-Olsen J., Benfield T., Axen T.E., Parner J., Iversen J., Pedersen C., Nielsen J.O. Effect of the serotonin receptor agonist, buspirone, on immune function in HIV-infected individuals: a six-month randomized, double-blind, placebo-controlled trial. HIV Clin. Trials. 2000;1:20–26. doi: 10.1310/UFDA-PU6H-2B0U-K9E6. [DOI] [PubMed] [Google Scholar]
- Evans D.L., Lynch K.G., Benton T., Dube B., Gettes D.R., Tustin N.B., Lai J.P., Metzger D., Douglas S.D. Selective serotonin reuptake inhibitor and substance P antagonist enhancement of natural killer cell innate immunity in human immunodeficiency virus/acquired immunodeficiency syndrome. Biol. Psychiatr. 2008;63:899–905. doi: 10.1016/j.biopsych.2007.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank M.G., Hendricks S.E., Johnson D.R., Wieseler J.L., Burke W.J. Antidepressants augment natural killer cell activity: in vivo and in vitro. Neuropsychobiology. 1999;39:18–24. doi: 10.1159/000026555. [DOI] [PubMed] [Google Scholar]
- Gariepy G., Nitka D., Schmitz N. The association between obesity and anxiety disorders in the population: a systematic review and meta-analysis. Int. J. Obes. 2010;34:407–419. doi: 10.1038/ijo.2009.252. [DOI] [PubMed] [Google Scholar]
- Gibney S.M., McGuinness B., Prendergast C., Harkin A., Connor T.J. Poly I:C-induced activation of the immune response is accompanied by depression and anxiety-like behaviours, kynurenine pathway activation and reduced BDNF expression. Brain Behav. Immun. 2013;28:170–181. doi: 10.1016/j.bbi.2012.11.010. [DOI] [PubMed] [Google Scholar]
- Gomes J.A.S., Silva J.F., Marcal A.P., Silva G.C., Gomes G.F., de Oliveira A.C.P., Soares V.L., Oliveira M.C., Ferreira A.V.M., Aguiar D.C. High-refined carbohydrate diet consumption induces neuroinflammation and anxiety-like behavior in mice. J. Nutr. Biochem. 2020;77:108317. doi: 10.1016/j.jnutbio.2019.108317. [DOI] [PubMed] [Google Scholar]
- Goshua G., Pine A.B., Meizlish M.L., Chang C.H., Zhang H., Bahel P., Baluha A., Bar N., Bona R.D., Burns A.J., Dela Cruz C.S., Dumont A., Halene S., Hwa J., Koff J., Menninger H., Neparidze N., Price C., Siner J.M., Tormey C., Rinder H.M., Chun H.J., Lee A.I. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020 doi: 10.1016/S2352-3026(20)30216-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greeson J.M., Gettes D.R., Spitsin S., Dube B., Benton T.D., Lynch K.G., Douglas S.D., Evans D.L. The selective serotonin reuptake inhibitor citalopram decreases human immunodeficiency virus receptor and coreceptor expression in immune cells. Biol. Psychiatr. 2016;80:33–39. doi: 10.1016/j.biopsych.2015.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haleem D.J., Mahmood K. Brain serotonin in high-fat diet-induced weight gain, anxiety and spatial memory in rats. Nutr. Neurosci. 2019:1–10. doi: 10.1080/1028415X.2019.1619983. [DOI] [PubMed] [Google Scholar]
- Harrison S.J., Tyrer A.E., Levitan R.D., Xu X., Houle S., Wilson A.A., Nobrega J.N., Rusjan P.M., Meyer J.H. Light therapy and serotonin transporter binding in the anterior cingulate and prefrontal cortex. Acta Psychiatr. Scand. 2015;132:379–388. doi: 10.1111/acps.12424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellstrand K., Hermodsson S. Enhancement of human natural killer cell cytotoxicity by serotonin: role of non-T/CD16+ NK cells, accessory monocytes, and 5-HT1A receptors. Cell. Immunol. 1990;127:199–214. doi: 10.1016/0008-8749(90)90125-b. [DOI] [PubMed] [Google Scholar]
- Heneka M.T., Golenbock D.T., Latz E. Innate immunity in Alzheimer's disease. Nat. Immunol. 2015;16:229–236. doi: 10.1038/ni.3102. [DOI] [PubMed] [Google Scholar]
- Herr N., Bode C., Duerschmied D. The effects of serotonin in immune cells. Front Cardiovasc Med. 2017;4:48. doi: 10.3389/fcvm.2017.00048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hervas I., Artigas F. Effect of fluoxetine on extracellular 5-hydroxytryptamine in rat brain. Role of 5-HT autoreceptors. Eur. J. Pharmacol. 1998;358:9–18. doi: 10.1016/s0014-2999(98)00579-2. [DOI] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotamisligil G.S. Inflammation and metabolic disorders. Nature. 2006;444:860–867. doi: 10.1038/nature05485. [DOI] [PubMed] [Google Scholar]
- Hryhorczuk C., Sharma S., Fulton S.E. Metabolic disturbances connecting obesity and depression. Front. Neurosci. 2013;7:177. doi: 10.3389/fnins.2013.00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang D., Lian X., Song F., Ma H., Lian Z., Liang Y., Qin T., Chen W., Wang S. Clinical features of severe patients infected with 2019 novel coronavirus: a systematic review and meta-analysis. Ann. Transl. Med. 2020;8:576. doi: 10.21037/atm-20-2124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ifuku M., Hossain S.M., Noda M., Katafuchi T. Induction of interleukin-1 beta by activated microglia is a prerequisite for immunologically induced fatigue. Eur. J. Neurosci. 2014;40:3253–3263. doi: 10.1111/ejn.12668. [DOI] [PubMed] [Google Scholar]
- Ishima T., Fujita Y., Hashimoto K. Interaction of new antidepressants with sigma-1 receptor chaperones and their potentiation of neurite outgrowth in PC12 cells. Eur. J. Pharmacol. 2014;727:167–173. doi: 10.1016/j.ejphar.2014.01.064. [DOI] [PubMed] [Google Scholar]
- Jiang H.Y., Deng M., Zhang Y.H., Chen H.Z., Chen Q., Ruan B. Specific serotonin reuptake inhibitors prevent interferon-alpha-induced depression in patients with hepatitis C: a meta-analysis. Clin. Gastroenterol. Hepatol. 2014;12:1452–1460. doi: 10.1016/j.cgh.2013.04.035. e1453. [DOI] [PubMed] [Google Scholar]
- Katafuchi T., Kondo T., Take S., Yoshimura M. Brain cytokines and the 5-HT system during poly I:C-induced fatigue. Ann. N. Y. Acad. Sci. 2006;1088:230–237. doi: 10.1196/annals.1366.020. [DOI] [PubMed] [Google Scholar]
- Kesic M., Bakovic P., Horvaticek M., Proust B.L.J., Stefulj J., Cicin-Sain L. Constitutionally high serotonin tone favors obesity: study on rat sublines with altered serotonin homeostasis. Front. Neurosci. 2020;14:219. doi: 10.3389/fnins.2020.00219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan M., Khan N., Mustagir G., Rana J., Islam S., Kabir I. medRxiv. 2020;520 doi: 10.1101/2020.05.08.20095968. 05.08.20095968. [Preprint] [DOI] [Google Scholar]
- Kim M., Bae S., Lim K.M. Impact of high fat diet-induced obesity on the plasma levels of monoamine neurotransmitters in C57BL/6 mice. Biomol Ther (Seoul) 2013;21:476–480. doi: 10.4062/biomolther.2013.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komegae E.N., Farmer D.G.S., Brooks V.L., McKinley M.J., McAllen R.M., Martelli D. Vagal afferent activation suppresses systemic inflammation via the splanchnic anti-inflammatory pathway. Brain Behav. Immun. 2018;73:441–449. doi: 10.1016/j.bbi.2018.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krahl S.E., Senanayake S.S., Pekary A.E., Sattin A. Vagus nerve stimulation (VNS) is effective in a rat model of antidepressant action. J. Psychiatr. Res. 2004;38:237–240. doi: 10.1016/j.jpsychires.2003.11.005. [DOI] [PubMed] [Google Scholar]
- Lassale C., Gaye B., Hamer M., Gale C.R., Batty G.D. Ethnic disparities in hospitalisation for COVID-19 in England: the role of socioeconomic factors, mental health, and inflammatory and pro-inflammatory factors in a community-based cohort study. Brain Behav. Immun. 2020 doi: 10.1016/j.bbi.2020.05.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasselin J., Capuron L. Chronic low-grade inflammation in metabolic disorders: relevance for behavioral symptoms. Neuroimmunomodulation. 2014;21:95–101. doi: 10.1159/000356535. [DOI] [PubMed] [Google Scholar]
- Lechien J.R., Chiesa-Estomba C.M., Hans S., Barillari M.R., Jouffe L., Saussez S. Loss of smell and taste in 2013 European patients with mild to moderate COVID-19. Ann. Intern. Med. 2020 doi: 10.7326/M20-2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lever A.R., Park H., Mulhern T.J., Jackson G.R., Comolli J.C., Borenstein J.T., Hayden P.J., Prantil-Baun R. Comprehensive evaluation of poly(I:C) induced inflammatory response in an airway epithelial model. Phys. Rep. 2015;3 doi: 10.14814/phy2.12334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li D., He L. Meta-analysis supports association between serotonin transporter (5-HTT) and suicidal behavior. Mol. Psychiatr. 2007;12:47–54. doi: 10.1038/sj.mp.4001890. [DOI] [PubMed] [Google Scholar]
- Liu D., Wang Z., Liu S., Wang F., Zhao S., Hao A. Anti-inflammatory effects of fluoxetine in lipopolysaccharide(LPS)-stimulated microglial cells. Neuropharmacology. 2011;61:592–599. doi: 10.1016/j.neuropharm.2011.04.033. [DOI] [PubMed] [Google Scholar]
- Liu F., McCullough L.D. Inflammatory responses in hypoxic ischemic encephalopathy. Acta Pharmacol. Sin. 2013;34:1121–1130. doi: 10.1038/aps.2013.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu B., Kwan K., Levine Y.A., Olofsson P.S., Yang H., Li J., Joshi S., Wang H., Andersson U., Chavan S.S., Tracey K.J. alpha 7 nicotinic acetylcholine receptor signaling inhibits inflammasome activation by preventing mitochondrial DNA release. Mol. Med. 2014;20:350–358. doi: 10.2119/molmed.2013.00117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucas C., Wong P., Klein J., Castro T.B.R., Silva J., Sundaram M., Ellingson M.K., Mao T., Oh J.E., Israelow B., Takahashi T., Tokuyama M., Lu P., Venkataraman A., Park A., Mohanty S., Wang H., Wyllie A.L., Vogels C.B.F., Earnest R., Lapidus S., Ott I.M., Moore A.J., Muenker M.C., Fournier J.B., Campbell M., Odio C.D., Casanovas-Massana A., Yale I.T., Herbst R., Shaw A.C., Medzhitov R., Schulz W.L., Grubaugh N.D., Dela Cruz C., Farhadian S., Ko A.I., Omer S.B., Iwasaki A. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020 doi: 10.1038/s41586-020-2588-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mainou B.A., Ashbrook A.W., Smith E.C., Dorset D.C., Denison M.R., Dermody T.S. Serotonin receptor agonist 5-nonyloxytryptamine alters the kinetics of reovirus cell entry. J. Virol. 2015;89:8701–8712. doi: 10.1128/JVI.00739-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maneglier B., Guillemin G.J., Clayette P., Rogez-Kreuz C., Brew B.J., Dormont D., Advenier C., Therond P., Spreux-Varoquaux O. Serotonin decreases HIV-1 replication in primary cultures of human macrophages through 5-HT(1A) receptors. Br. J. Pharmacol. 2008;154:174–182. doi: 10.1038/bjp.2008.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann J.J. Role of the serotonergic system in the pathogenesis of major depression and suicidal behavior. Neuropsychopharmacology. 1999;21:99S–105S. doi: 10.1016/S0893-133X(99)00040-8. [DOI] [PubMed] [Google Scholar]
- Manta S., El Mansari M., Debonnel G., Blier P. Electrophysiological and neurochemical effects of long-term vagus nerve stimulation on the rat monoaminergic systems. Int. J. Neuropsychopharmacol. 2013;16:459–470. doi: 10.1017/S1461145712000387. [DOI] [PubMed] [Google Scholar]
- Martelli D., Yao S.T., McKinley M.J., McAllen R.M. Reflex control of inflammation by sympathetic nerves, not the vagus. J. Physiol. 2014;592:1677–1686. doi: 10.1113/jphysiol.2013.268573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mas Serrano M., Perez-Sanchez J.R., Portela Sanchez S., De La Casa-Fages B., Mato Jimeno V., Perez Tamayo I., Grandas F. Serotonin syndrome in two COVID-19 patients treated with lopinavir/ritonavir. J. Neurol. Sci. 2020;415:116944. doi: 10.1016/j.jns.2020.116944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McElvaney O.J., McEvoy N., McElvaney O.F., Carroll T.P., Murphy M.P., Dunlea D.M., Ni Choileain O., Clarke J., O'Connor E., Hogan G., Ryan D., Sulaiman I., Gunaratnam C., Branagan P., O'Brien M.E., Morgan R.K., Costello R.W., Hurley K., Walsh S., de Barra E., McNally C., McConkey S., Boland F., Galvin S., Kiernan F., O'Rourke J., Dwyer R., Power M., Geoghegan P., Larkin C., O'Leary R.A., Freeman J., Gaffney A., Marsh B., Curley G.F., McElvaney N.G. Characterization of the inflammatory response to severe COVID-19 illness. Am. J. Respir. Crit. Care Med. 2020 doi: 10.1164/rccm.202005-1583OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McVey Neufeld K.A., Bienenstock J., Bharwani A., Champagne-Jorgensen K., Mao Y., West C., Liu Y., Surette M.G., Kunze W., Forsythe P. Oral selective serotonin reuptake inhibitors activate vagus nerve dependent gut-brain signalling. Sci. Rep. 2019;9:14290. doi: 10.1038/s41598-019-50807-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milanski M., Degasperi G., Coope A., Morari J., Denis R., Cintra D.E., Tsukumo D.M., Anhe G., Amaral M.E., Takahashi H.K., Curi R., Oliveira H.C., Carvalheira J.B., Bordin S., Saad M.J., Velloso L.A. Saturated fatty acids produce an inflammatory response predominantly through the activation of TLR4 signaling in hypothalamus: implications for the pathogenesis of obesity. J. Neurosci. 2009;29:359–370. doi: 10.1523/JNEUROSCI.2760-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller A.H., Raison C.L. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2016;16:22–34. doi: 10.1038/nri.2015.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minihane A.M., Vinoy S., Russell W.R., Baka A., Roche H.M., Tuohy K.M., Teeling J.L., Blaak E.E., Fenech M., Vauzour D., McArdle H.J., Kremer B.H., Sterkman L., Vafeiadou K., Benedetti M.M., Williams C.M., Calder P.C. Low-grade inflammation, diet composition and health: current research evidence and its translation. Br. J. Nutr. 2015;114:999–1012. doi: 10.1017/S0007114515002093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miranda J.J., Barrientos-Gutierrez T., Corvalan C., Hyder A.A., Lazo-Porras M., Oni T., Wells J.C.K. Understanding the rise of cardiometabolic diseases in low- and middle-income countries. Nat. Med. 2019;25:1667–1679. doi: 10.1038/s41591-019-0644-7. [DOI] [PubMed] [Google Scholar]
- Mossner R., Lesch K.P. Role of serotonin in the immune system and in neuroimmune interactions. Brain Behav. Immun. 1998;12:249–271. doi: 10.1006/brbi.1998.0532. [DOI] [PubMed] [Google Scholar]
- Mota C.M.D., Borges G.S., Amorim M.R., Carolino R.O.G., Batalhao M.E., Anselmo-Franci J.A., Carnio E.C., Branco L.G.S. Central serotonin prevents hypotension and hypothermia and reduces plasma and spleen cytokine levels during systemic inflammation. Brain Behav. Immun. 2019;80:255–265. doi: 10.1016/j.bbi.2019.03.017. [DOI] [PubMed] [Google Scholar]
- Mota C.M.D., Rodrigues-Santos C., Fernandez R.A.R., Carolino R.O.G., Antunes-Rodrigues J., Anselmo-Franci J.A., Branco L.G.S. Central serotonin attenuates LPS-induced systemic inflammation. Brain Behav. Immun. 2017;66:372–381. doi: 10.1016/j.bbi.2017.07.010. [DOI] [PubMed] [Google Scholar]
- Netland J., Meyerholz D.K., Moore S., Cassell M., Perlman S. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J. Virol. 2008;82:7264–7275. doi: 10.1128/JVI.00737-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connell P.J., Wang X., Leon-Ponte M., Griffiths C., Pingle S.C., Ahern G.P. A novel form of immune signaling revealed by transmission of the inflammatory mediator serotonin between dendritic cells and T cells. Blood. 2006;107:1010–1017. doi: 10.1182/blood-2005-07-2903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Hara B.A., Atwood W.J. Interferon beta 1-a and selective anti-5HT(2a) receptor antagonists inhibit infection of human glial cells by JC virus. Virus Res. 2008;132:97–103. doi: 10.1016/j.virusres.2007.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okusa M.D., Rosin D.L., Tracey K.J. Targeting neural reflex circuits in immunity to treat kidney disease. Nat. Rev. Nephrol. 2017;13:669–680. doi: 10.1038/nrneph.2017.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osimo E.F., Baxter L.J., Lewis G., Jones P.B., Khandaker G.M. Prevalence of low-grade inflammation in depression: a systematic review and meta-analysis of CRP levels. Psychol. Med. 2019;49:1958–1970. doi: 10.1017/S0033291719001454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquarelli-do-Nascimento G., Braz-de-Melo H.A., Faria S.S., Santos I.O., Kobinger G.P., Magalhaes K.G. Hypercoagulopathy and adipose tissue exacerbated inflammation may explain higher mortality in COVID-19 patients with obesity. Front. Endocrinol. 2020;11:530. doi: 10.3389/fendo.2020.00530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavlov V.A., Tracey K.J. The vagus nerve and the inflammatory reflex--linking immunity and metabolism. Nat. Rev. Endocrinol. 2012;8:743–754. doi: 10.1038/nrendo.2012.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry V.H. Contribution of systemic inflammation to chronic neurodegeneration. Acta Neuropathol. 2010;120:277–286. doi: 10.1007/s00401-010-0722-x. [DOI] [PubMed] [Google Scholar]
- Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.S. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in wuhan, China. Clin. Infect. Dis. 2020;71:762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quintero-Villegas A., Valdes-Ferrer S.I. Role of 5-HT7 receptors in the immune system in health and disease. Mol. Med. 2019;26:2. doi: 10.1186/s10020-019-0126-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabbani M.A.G., Barik S. 5-Hydroxytryptophan, a major product of tryptophan degradation, is essential for optimal replication of human parainfluenza virus. Virology. 2017;503:46–51. doi: 10.1016/j.virol.2017.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rorato R., Borges B.C., Uchoa E.T., Antunes-Rodrigues J., Elias C.F., Elias L.L.K. LPS-induced low-grade inflammation increases hypothalamic JNK expression and causes central insulin resistance irrespective of body weight changes. Int. J. Mol. Sci. 2017;18 doi: 10.3390/ijms18071431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen D.A., Seki S.M., Fernandez-Castaneda A., Beiter R.M., Eccles J.D., Woodfolk J.A., Gaultier A. Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis. Sci. Transl. Med. 2019;11 doi: 10.1126/scitranslmed.aau5266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roszman T.L., Jackson J.C., Cross R.J., Titus M.J., Markesbery W.R., Brooks W.H. Neuroanatomic and neurotransmitter influences on immune function. J. Immunol. 1985;135:769s–772s. [PubMed] [Google Scholar]
- Rummel C. Does hypothalamic serotonin govern septic inflammation via the splanchnic anti-inflammatory reflex? Brain Behav. Immun. 2019;81:10–11. doi: 10.1016/j.bbi.2019.06.023. [DOI] [PubMed] [Google Scholar]
- Salaga M., Binienda A., Piscitelli F., Mokrowiecka A., Cygankiewicz A.I., Verde R., Malecka-Panas E., Kordek R., Krajewska W.M., Di Marzo V., Fichna J. Systemic administration of serotonin exacerbates abdominal pain and colitis via interaction with the endocannabinoid system. Biochem. Pharmacol. 2019;161:37–51. doi: 10.1016/j.bcp.2019.01.001. [DOI] [PubMed] [Google Scholar]
- Sbolli M., Fiuzat M., Cani D., O'Connor C.M. Depression and heart failure: the lonely comorbidity. Eur. J. Heart Fail. 2020 doi: 10.1002/ejhf.1865. [DOI] [PubMed] [Google Scholar]
- Scotton W.J., Hill L.J., Williams A.C., Barnes N.M. Serotonin syndrome: pathophysiology, clinical features, management, and potential future directions. Int. J. Tryptophan Res. 2019;12 doi: 10.1177/1178646919873925. 1178646919873925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidu S., Gillies C., Zaccardi F., Kunutsor S.K., Hartmann-Boyce J., Yates T., Singh A.K., Davies M.J., Khunti K. The impact of obesity on severe disease and mortality in people with SARS-CoV-2: a systematic review and meta-analysis. Endocrinol Diabetes Metab. 2020 doi: 10.1002/edm2.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma G., Champalal Sharma D., Hwei Fen L., Pathak M., Bethur N., Pendharkar V., Peiris M., Altmeyer R. Reduction of influenza virus-induced lung inflammation and mortality in animals treated with a phosophodisestrase-4 inhibitor and a selective serotonin reuptake inhibitor. Emerg. Microb. Infect. 2013;2:e54. doi: 10.1038/emi.2013.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shastri A., Bonifati D.M., Kishore U. Innate immunity and neuroinflammation. Mediat. Inflamm. 2013;2013:342931. doi: 10.1155/2013/342931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon I.H., Normandin E., Bhattacharyya S., Mukerji S.S., Keller K., Ali A.S., Adams G., Hornick J.L., Padera R.F., Jr., Sabeti P. Neuropathological features of covid-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMc2019373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spinato G., Fabbris C., Polesel J., Cazzador D., Borsetto D., Hopkins C., Boscolo-Rizzo P. Alterations in smell or taste in mildly symptomatic outpatients with SARS-CoV-2 infection. J. Am. Med. Assoc. 2020 doi: 10.1001/jama.2020.6771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabo A., Gogolak P., Koncz G., Foldvari Z., Pazmandi K., Miltner N., Poliska S., Bacsi A., Djurovic S., Rajnavolgyi E. Immunomodulatory capacity of the serotonin receptor 5-HT2B in a subset of human dendritic cells. Sci. Rep. 2018;8:1765. doi: 10.1038/s41598-018-20173-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tay M.Z., Poh C.M., Renia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tracey K.J. The inflammatory reflex. Nature. 2002;420:853–859. doi: 10.1038/nature01321. [DOI] [PubMed] [Google Scholar]
- Varatharaj A., Thomas N., Ellul M.A., Davies N.W.S., Pollak T.A., Tenorio E.L., Sultan M., Easton A., Breen G., Zandi M., Coles J.P., Manji H., Al-Shahi Salman R., Menon D.K., Nicholson T.R., Benjamin L.A., Carson A., Smith C., Turner M.R., Solomon T., Kneen R., Pett S.L., Galea I., Thomas R.H., Michael B.D., CoroNerve Study G. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry. 2020 doi: 10.1016/S2215-0366(20)30287-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voronova I.P., Khramova G.M., Kulikova E.A., Petrovskii D.V., Bazovkina D.V., Kulikov A.V. 5-HT2A receptors control body temperature in mice during LPS-induced inflammation via regulation of NO production. Pharmacol. Res. 2016;103:123–131. doi: 10.1016/j.phrs.2015.11.018. [DOI] [PubMed] [Google Scholar]
- Weisberg S.P., McCann D., Desai M., Rosenbaum M., Leibel R.L., Ferrante A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 2003;112:1796–1808. doi: 10.1172/JCI19246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiersinga W.J., Rhodes A., Cheng A.C., Peacock S.J., Prescott H.C. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. J. Am. Med. Assoc. 2020 doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- Wu H., Denna T.H., Storkersen J.N., Gerriets V.A. Beyond a neurotransmitter: the role of serotonin in inflammation and immunity. Pharmacol. Res. 2019;140:100–114. doi: 10.1016/j.phrs.2018.06.015. [DOI] [PubMed] [Google Scholar]
- Wu Y., Xu X., Chen Z., Duan J., Hashimoto K., Yang L., Liu C., Yang C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020;87:18–22. doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., Tai Y., Bai C., Gao T., Song J., Xia P., Dong J., Zhao J., Wang F.S. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan B., Li W., Liu H., Cai X., Song S., Zhao J., Hu X., Li Z., Chen Y., Zhang K., Liu Z., Peng J., Wang C., Wang J., An Y. Correlation between immune response and self-reported depression during convalescence from COVID-19. Brain Behav. Immun. 2020 doi: 10.1016/j.bbi.2020.05.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zemdegs J., Quesseveur G., Jarriault D., Penicaud L., Fioramonti X., Guiard B.P. High-fat diet-induced metabolic disorders impairs 5-HT function and anxiety-like behavior in mice. Br. J. Pharmacol. 2016;173:2095–2110. doi: 10.1111/bph.13343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Q., Meng M., Kumar R., Wu Y., Huang J., Deng Y., Weng Z., Yang L. Lymphopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: a systemic review and meta-analysis. Int. J. Infect. Dis. 2020;96:131–135. doi: 10.1016/j.ijid.2020.04.086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimniak M., Kirschner L., Hilpert H., Seibel J., Bodem J. bioRxiv. 2020;742 doi: 10.1101/2020.06.14.150490. 06.14.150490. [Preprint] [DOI] [Google Scholar]
- Zuo J., Quinn K.K., Kye S., Cooper P., Damoiseaux R., Krogstad P. Fluoxetine is a potent inhibitor of coxsackievirus replication. Antimicrob. Agents Chemother. 2012;56:4838–4844. doi: 10.1128/AAC.00983-12. [DOI] [PMC free article] [PubMed] [Google Scholar]