Abstract
The ongoing global pandemic of COVID-19 disease, which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), mainly infect lung epithelial cells, and spread mainly through respiratory droplets. However, recent studies showed potential intestinal infection of SARS-CoV-2, implicated the possibility that the intestinal infection of SARS-CoV-2 may correlate with the dysbiosis of gut microbiota, as well as the severity of COVID-19 symptoms. Here, we investigated the alteration of the gut microbiota in COVID-19 patients, as well as analyzed the correlation between the altered microbes and the levels of intestinal inflammatory cytokine IL-18, which was reported to be elevated in the serum of in COVID-19 patients. Comparing with healthy controls or seasonal flu patients, the gut microbiota showed significantly reduced diversity, with increased opportunistic pathogens in COVID-19 patients. Also, IL-18 level was higher in the fecal samples of COVID-19 patients than in those of either healthy controls or seasonal flu patients. Moreover, the IL-18 levels were even higher in the fecal supernatants obtained from COVID-19 patients that tested positive for SARS-CoV-2 RNA than those that tested negative in fecal samples. These results indicate that changes in gut microbiota composition might contribute to SARS-CoV-2-induced production of inflammatory cytokines in the intestine and potentially also to the onset of a cytokine storm.
Keywords: COVID19, SARS2, Gut microbiota, IL18
1. Introduction
The ongoing global pandemic of COVID-19 disease is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which has already infected over 20 million people and caused 700 000 deaths worldwide as of August 2020 [1]. Cytokine overproduction triggered by SARS-CoV-2 infection, known as a “cytokine storm”, is highly associated with disease severity. However, how SARS-CoV-2 infection induces a cytokine storm remains unclear. Accumulating in vitro and in vivo evidence suggests that the gut is also susceptible to SARS-CoV-2 infection. For example, human intestinal organoids, an in vitro model that mimics intestinal cellular and spatial structures, are susceptible to SARS-CoV-2 infection [2], while a significant fraction of COVID-19 patients reported experiencing gut symptoms. Additionally, viral RNA may persist for more than 30 days and infectious virus can be isolated from fecal samples [3,4]. The gastrointestinal tract is the primary site of interaction between the host immune system and symbiotic and pathogenic microorganisms. The bacteria resident in the gastrointestinal tract, known as the gut microbiota, are important for the maintenance of immune homeostasis. A gut microbiota imbalance, or dysbiosis, is associated with multiple inflammatory diseases [5]. It is feasible that SARS-CoV-2 infection may alter the composition of the gut microbiota, thereby worsening COVID-19 symptoms. Interleukin (IL)-18 is a proinflammatory cytokine produced by different types of enteric cells, including intestinal epithelial cells, immune cells, and cells of the enteric nervous system [6,7], and IL-18 levels are upregulated in the serum of COVID-19 patients [8]. Here, we investigated the changes occurring in the gut microbiota of COVID-19 patients, and the correlation between these alterations and the levels of IL-18.
2. Results
2.1. Alternation of the intestinal microbiota in COVID-19 patients
We enrolled 62 COVID-19 patients from the First Affiliated Hospital of the University of Science and Technology of China in Hefei, as well as 33 seasonal flu patients and 40 healthy controls from Hefei. Fresh fecal and serum samples from the three groups were immediately stored at −80 °C for further analysis. We used next-generation sequencing of the V4 region of the 16S ribosomal RNA gene to investigate the potential effect of intestinal SARS-CoV-2 infection on the composition of the gut microbiota. The raw sequencing data were first trimmed and quality-filtered to remove adaptor or low-quality sequences, resulting in an average of 28 054 sequences per sample. The sequences were then further processed using a customized pipeline that combined usearch (v8.1) [7], vsearch (v2.13.0)[8], and QIIME (v1.9.1)[9]. Alpha-diversity analysis showed that the mean species diversity (Chao1 index) was significantly decreased in COVID-19 patients compared with both flu patients and healthy controls (Fig. 1 A). Beta diversity values, as measured by weighted UniFrac, and a histogram depicting the relative taxonomic fraction at the genus level in each group, showed that the abundance and composition of fecal bacteria in COVID-19 patients differed from those of both healthy controls and seasonal flu patients (Fig. 1B and C). To investigate which gut bacteria were associated with COVID-19 patients, linear discriminant analysis effect size (LEfSe) was performed in galaxy (http://huttenhower.sph.harvard.edu/galaxy/) to identify biomarkers for each group. The threshold was set at LDA >2 to filter the features that differed significantly between COVID-19 patients and seasonal flu patients or healthy controls. Compared with healthy controls, the abundance of members of the genera Streptococcus, Clostridium, Lactobacillus, and Bifidobacterium was increased, whereas that of members of the genera Bacteroidetes, Roseburia, Faecalibacterium, Coprococcus, and Parabacteroides was decreased, in SARS-CoV-2-infected patients (Fig. 1D and E). Compared with seasonal flu patients, the abundance of members of the genera Streptococcus, Veillonella, Fusobacterium, Clostridium, Bifidobacterium, and Escherichia was increased, whereas that of members of the genera Parabacteroides and Sutterella was decreased, in SARS-CoV-2 patients (Fig. 1F and G). The increased abundance of Streptococcus in COVID-19 patients was indicative of the risk of infection by opportunistic pathogenic bacteria in this group [10].
Fig. 1.
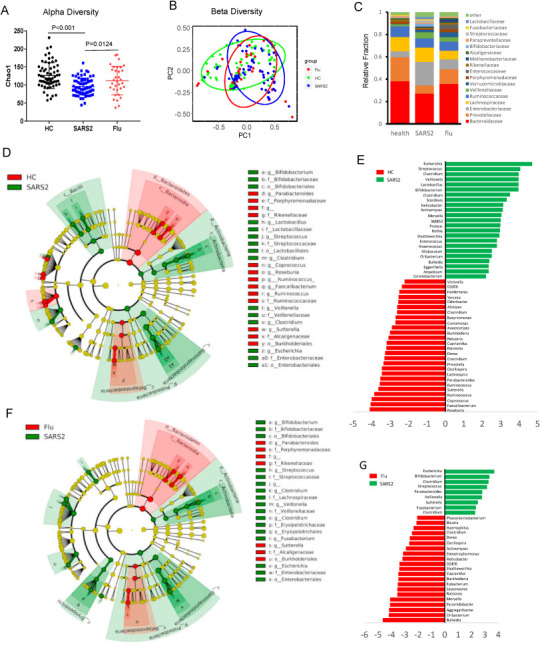
Changes in the gut microbiota of COVID-19 patients. Fecal samples from healthy controls (HC), severe acute respiratory syndrome coronavirus 2 patients (SARS2), and seasonal flu patients (Flu) were collected for analyzed of gut microbiota. Alpha diversity (Chao1) (A) and weighted Unifrac beta diversity (B) of fecal bacteria, as well the taxonomic fraction in each group (C). Linear discriminant analysis (LDA) effect size (LEfSe) was performed in galaxy to identify biomarkers for each group. (D, E) Features differing significantly (LDA >3.5 for Cladogram plot) between COVID-19 patients and HC. (F, G) Features differing significantly (LDA >3.5 for Cladogram plot) between COVID-19 patients and seasonal flu patients. (H) The fecal concentrations of interleukin (IL)-18 from each group, or (I) from fecal viral RNA positive or negative COVID-19 patients. (J)The correlation among microbiota composition, gut IL18 concentration and disease severity were analyzed using Spearman's correlation coefficient.
2.2. Correlation of gut microbiota with the inflammatory factor IL-18
Intestinal infection by viruses can also induce the production of the proinflammatory factor IL-18 [11]. Here, we used ELISA to measure the concentrations of IL-18 in the fecal supernatants of the three groups. Notably, IL-18 production was higher in fecal supernatants of COVID-19 patients than in those of either healthy controls or seasonal flu patients (Fig. 2 A). Moreover, the IL-18 levels were higher in the fecal supernatants obtained from COVID-19 patients that tested positive for SARS-CoV-2 RNA than those that tested negative in fecal samples (Fig. 2B), indicating that SARS-CoV-2 induced strong intestinal inflammation. We next analyzed the correlation among fecal microbiota composition, Fecal IL-18 levels, and disease severity in COVID-19 patients. Bilophila and Citrobacter was negatively correlate with disease severity. Intriguingly, fecal IL-18 levels positively correlated with the relative abundance of Peptostreptococcus, Fusobacterium and Citrobacter at the genus level (Fig. 2C), indicating that changes in gut microbiota composition might contribute to SARS-CoV-2-induced production of inflammatory cytokines in the intestine and potentially also to the onset of a cytokine storm.
Fig. 2.
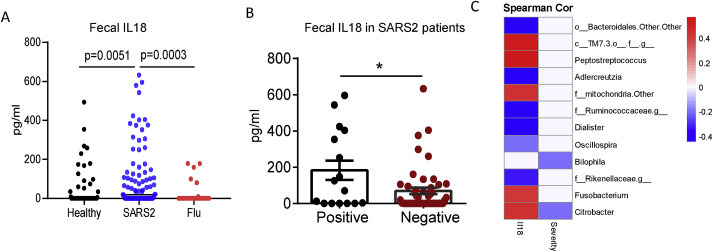
Correlation of gut microbiota with fecal IL-18 in COVID19 patients. (A) The fecal concentrations of interleukin (IL)-18 from each group, or (B) from fecal viral RNA positive or negative COVID-19 patients. (C)The correlation among microbiota composition, gut IL18 concentration and disease severity were analyzed using Spearman's correlation coefficient.
3. Discussion
In conclusion, we found SARS-CoV-2-dependent changes in the composition of the gut microbiota of COVID-19 patients. Moreover, higher relative levels of Streptococcus, Clostridium, Lactobacillus, and Bifidobacterium, and lower relative levels of Bacteroidetes, Roseburia, Faecalibacterium, Coprococcus, and Parabacteroides, were found in COVID-19 patients when compared with both seasonal flu patients and healthy controls. While this manuscript was under preparation, other groups also reported alternation of gut microbiota in COVID-19 patients [12,13]. Possibly due to observation from different geographic locations, our studies showed unique microbiota pattern comparing with others, such as higher Helicobacter in COVID-19 patients than Healthy controls. Notably, there are also some consistent microbiota change across different studies. For example, higher level of Streptococcus, Clostridium and Bifidobacterium was also observed in COVID-19 patients from other site [13]. The observation of enriched opportunity pathogenic bacteria from different hospitals indicate potential contribution of gut microbiota to disease progress. Interestingly, the concentration of IL-18, a key proinflammatory factor produced by gut cells, was increased in COVID-19 patients, but not in seasonal flu patients. Additionally, fecal supernatant IL-18 levels were higher in COVID-19 patients that tested positive for SARS-CoV-2 RNA than in those that tested negative. Our results suggested that gut microbiota dysbiosis due to SARS-CoV-2 infection may contribute to disease severity, and that IL-18 might serve as an indicator of intestinal infection in COVID-19 patients. Notably, SARS-CoV-2-infected patients were under medical care when their fecal samples were collected, and further studies with treatment-naïve patients are needed to exclude the possibility that changes in the gut microbiota were caused by the administered drugs.
4. Methods
4.1. Cases
This is an observational cohort study. This study is part of the project of “Construction of a bio-information platform for novel coronavirus pneumonia (COVID-19) patients follow-up in Anhui” (ChiCTR2000030331). This study was approved by the institutional board of the First Affiliated Hospital of University of Science and Technology of China (2020-XG(H)-009). Patient history, physical findings, and hematological, biochemical, and microbiological investigation results were recorded and analyzed. All laboratory procedures for clinical samples have been previously reported [12,14,15]. Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
4.2. 16S rRNA gene sequencing of fecal microbiota
Fecal samples were stored at −80 °C until 16S rRNA gene analysis. Fecal samples from healthy controls (HC), severe acute respiratory syndrome coronavirus 2 patients (SARS2), and seasonal flu patients (Flu) were first heated at 65 °C to inactivate potential live viruses. DNA was extracted from fecal pellets with a QIAamp stool DNA Mini kit (QIAGEN) according to the manufacturer's instructions. 16S rRNA gene amplicons were generated using the primer pair 515 F/806 R as recommended by Earth Microbiome Project [16]. PCR products were quantified, pooled, cleaned using the PCR Cleanup kit (QIAGEN), and subsequently sequenced on Illumina MiSeq (2 × 250bp). Custom primers were added to the Illumina MiSeq kit resulting in a depth of 28 054 ± 15 086 reads (mean ± SD). The raw sequencing data were trimmed and processed using a customized pipeline that combined vsearch (v2.13.3), usearch (v8.1) [7], and QIIME (v1.9.1)[9] for analysis of alpha diversity (Chao1) and weighted Unifrac beta diversity of fecal bacteria, as well as to determine the taxonomic fraction in each group. The linear discriminant analysis effect size Galaxy module (http://huttenhower.sph.harvard.edu/galaxy/) was used for additional statistical analyses [17].
4.3. Enzyme-linked immunosorbent assay (ELISA) analysis of IL-18
Fecal samples (0.5 g) were then suspended in 3 ml of sterile PBS buffer and the supernatants used for further analysis. Fecal extracts were prepared as described above and IL-18 levels were measured using an IL-18 ELISA Kit (Sino Biological) according to the instructions of the vendor. The correlation among microbiota composition, gut IL18 concentration and disease severity were analyzed using Spearman's correlation coefficient.
Credit author statement
WT and SZ designed the experiments. WT, XW and GZ performed the experiments and interpreted the data. LL, JW, AP, YW, XM and JW collected all the clinical samples; JW, XM, DC, KZ provided critical comments and suggestions; TJ, ZX, WZ and DZ performed the serological analyses; SZ, WT, and MG wrote the manuscript; SZ, LL and JW supervised the project.
Declaration of competing interest
We declare no competing interests.
Acknowledgements
This work was supported by a grant from the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB29030101) (SZ), the National Key R&D Program of China (2018YFA0508000) (SZ), the National Natural Science Foundation of China (81822021, 91842105, 31770990, 81821001) (SZ), and the Fundamental Research Funds for the Central Universities (WK2070000159) (SZ).
References
- 1.Dodd D. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature. 2017;551:648–652. doi: 10.1038/nature24661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lamers M.M. Science; New York, N.Y.: 2020. SARS-CoV-2 productively infects human gut enterocytes; p. abc1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu Y. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. The lancet. Gastroenterol Hepatol. 2020 doi: 10.1016/s2468-1253(20)30083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Xiao F. Infectious SARS-CoV-2 in feces of patient with severe COVID-19. Emerg Infect Dis. 2020;26 doi: 10.3201/eid2608.200681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu H.J., Wu E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microb. 2012;3:4–14. doi: 10.4161/gmic.19320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stadnyk A.W. Intestinal epithelial cells as a source of inflammatory cytokines and chemokines. Canadian journal of gastroenterology = Journal canadien de gastroenterologie. 2002;16:241–246. doi: 10.1155/2002/941087. [DOI] [PubMed] [Google Scholar]
- 7.Edgar R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics. 2010;26:2460–2461. doi: 10.1093/bioinformatics/btq461. [DOI] [PubMed] [Google Scholar]
- 8.Rognes T., Flouri T., Nichols B., Quince C., Mahe F. VSEARCH: a versatile open source tool for metagenomics. PeerJ. 2016;4 doi: 10.7717/peerj.2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caporaso J.G. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weiser J.N., Ferreira D.M., Paton J.C. Streptococcus pneumoniae: transmission, colonization and invasion. Nat Rev Microbiol. 2018;16:355–367. doi: 10.1038/s41579-018-0001-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhu S. Nlrp9b inflammasome restricts rotavirus infection in intestinal epithelial cells. Nature. 2017;546:667–670. doi: 10.1038/nature22967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zuo T. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gu S. Alterations of the gut microbiota in patients with COVID-19 or H1N1 influenza. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu X. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. U.S.A. 2020;117:10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tao W. Re-detectable positive SARS-CoV-2 RNA tests in patients who recovered from COVID-19 with intestinal infection. Protein & Cell. 2020 doi: 10.1007/s13238-020-00778-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thompson L.R. A communal catalogue reveals Earth's multiscale microbial diversity. Nature. 2017;551:457–463. doi: 10.1038/nature24621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Segata N. Metagenomic biomarker discovery and explanation. Genome Biol. 2011;12:R60. doi: 10.1186/gb-2011-12-6-r60. [DOI] [PMC free article] [PubMed] [Google Scholar]


