Abstract
Objective
We sought to review the literature on cerebrospinal fluid (CSF) testing in patients with COVID-19 for evidence of viral neuroinvasion by SARS-CoV-2.
Methods
We performed a systematic review of Medline and Embase between December 1, 2019 and November 18, 2020 to identify case reports or series of patients who had COVID-19 diagnosed based on positive SARS-CoV-2 polymerase chain reaction (PCR) or serologic testing and had CSF testing due to a neurologic symptom.
Results
We identified 242 relevant documents which included 430 patients with COVID-19 who had acute neurological symptoms prompting CSF testing. Of those, 321 (75%) patients had symptoms that localized to the central nervous system (CNS). Of 304 patients whose CSF was tested for SARS-CoV-2 PCR, there were 17 (6%) whose test was positive, all of whom had symptoms that localized to the central nervous system (CNS). The majority (13/17, 76%) of these patients were admitted to the hospital because of neurological symptoms. Of 58 patients whose CSF was tested for SARS-CoV-2 antibody, 7 (12%) had positive antibodies with evidence of intrathecal synthesis, all of whom had symptoms that localized to the CNS. Of 132 patients who had oligoclonal bands evaluated, 3 (2%) had evidence of intrathecal antibody synthesis. Of 77 patients tested for autoimmune antibodies in the CSF, 4 (5%) had positive findings.
Conclusion
Detection of SARS-CoV-2 in CSF via PCR or evaluation for intrathecal antibody synthesis appears to be rare. Most neurological complications associated with SARS- CoV-2 are unlikely to be related to direct viral neuroinvasion.
Keywords: COVID-19, SARS-CoV-2, Cerebrospinal fluid, Neuroinvasion
1. Introduction
Although viruses commonly infect the respiratory tract, zoonoses, which present in animals and then cross the species barrier into humans, have the ability to adapt to new environments, including the central nervous system (CNS) [1]. The most prevalent viruses that infect the CNS are herpesviruses, arboviruses and enteroviruses. The potential for viral neuroinvasion has also been documented in human, mouse and porcine coronaviruses (SARS-CoV, MERS-CoV, HcoV-229E, HcoV-OC43, MHV and porcine HEV) [[1], [2], [3]]. The mechanism for viral entry into the CNS has been postulated to be through the olfactory nerve, retrograde transmission via other cranial or peripheral nerves (such as the trigeminal nerve, which has nociceptive cells in the nasal cavity, or the vagus nerve, which innervates the respiratory and the gastrointestinal tracts), hematogenous spread, lymphatic spread or entry via the choroid plexus [[1], [2], [3], [4]]. Viral neuroinvasion can have acute consequences (such as viral encephalitis), subacute consequences (such as post-infectious acute disseminated encephalomyelitis) or delayed consequences (such as subacute sclerosing panencephalitis, which can present 6–10 years after initial viral infection), and while no specific virus is considered the causal agent for neurodegenerative diseases, herpesviruses have been associated with Alzheimer’s disease and multiple sclerosis [1,3]. Viruses can also induce neurological sequelae indirectly due to hypoxic-ischemic injury, stroke, toxic-metabolic changes, cytokine storming or through molecular mimicry against neuronal, glial or peripheral nerve cells [[4], [5], [6], [7], [8], [9], [10]].
Although there have been multiple publications about neurological symptoms associated with SARS-CoV-2, the novel coronavirus responsible for COVID-19, there is a need for a summary of the literature on cerebrospinal fluid (CSF) testing for SARS-CoV-2 detection via polymerase chain reaction (PCR) assay, evaluation for intrathecal SARS-CoV-2 antibodies and measurement of CSF biomarkers of inflammation and neuronal injury. We sought to review CSF results in patients with COVID-19 who had acute neurological symptoms to evaluate for evidence of neuroinvasion.
2. Methods
To identify documents that included CSF results from patients with COVID-19, we searched Medline and Embase using the population search terms “COVID-19” or “SARS-CoV-2” and the intervention search terms “cerebrospinal fluid” or “csf” or “spinal puncture” or “spinal tap” or “lumbar puncture” or “meningitis” or “encephalitis” or “encephalomyelitis” or “seizure” or “encephalopathy” or “myelitis” or “Guillain Barre” or “polyradiculitis” or “Miller Fisher.” The search period included documents published between December 1, 2019 and November 18, 2020. After removal of duplicates, two board-certified neurointensivists (AL and KM) independently screened references using Covidence Systematic Review Software and performed full-text review to identify documents that provided details on at least one unique patient with COVID-19 diagnosed based on positive SARS-CoV-2 PCR or serologic testing who had a neurological symptom and CSF testing [11]. Documents were excluded if they did not present clinical data on an original patient with COVID-19 diagnosed via positive SARS-CoV-2 PCR or serologic testing who had a neurological symptom, did not include any CSF results, described a patient with another acute neurological diagnosis that could potentially impact CSF results (such as subarachnoid hemorrhage or another intracranial infection) or were not in English. Additional documents were identified based on review of the reference lists in screened documents, ultimately resulting in inclusion of 242 documents. This search was performed in accordance with PRISMA guidelines (Fig. 1 ) [12]. Cases were reviewed and organized based on localization of neurological symptoms and CSF findings. All laboratory test results were converted to a common unit to facilitate comparison.
Fig. 1.
Document selection.
3. Results
After review of the 242 documents, we identified 430 patients with COVID-19 diagnosed based on PCR or serologic testing who had acute neurological symptoms prompting CSF testing [[5], [6], [7], [8], [9], [10],[13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92], [93], [94], [95], [96], [97], [98], [99], [100], [101], [102], [103], [104], [105], [106], [107], [108], [109], [110], [111], [112], [113], [114], [115], [116], [117], [118], [119], [120], [121], [122], [123], [124], [125], [126], [127], [128], [129], [130], [131], [132], [133], [134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144], [145], [146], [147], [148], [149], [150], [151], [152], [153], [154], [155], [156], [157], [158], [159], [160], [161], [162], [163], [164], [165], [166], [167], [168], [169], [170], [171], [172], [173], [174], [175], [176], [177], [178], [179], [180], [181], [182], [183], [184], [185], [186], [187], [188], [189], [190], [191], [192], [193], [194], [195], [196], [197], [198], [199], [200], [201], [202], [203], [204], [205], [206], [207], [208], [209], [210], [211], [212], [213], [214], [215], [216], [217], [218], [219], [220], [221], [222], [223], [224], [225], [226], [227], [228], [229], [230], [231], [232], [233], [234], [235], [236], [237], [238], [239], [240], [241], [242], [243], [244], [245], [246]]. The patients ranged in age from 40-weeks+3-days gestational age to 96-years-old and lived on all six continents. There were 321 (75%) patients with symptoms that localized to the CNS and 109 (25%) patients with symptoms that localized to the peripheral nervous system (PNS; Supplemental Tables 1 and 2). The most common primary neurological symptoms that precipitated CSF testing was encephalopathy/coma (242, 56%; Fig. 2 ).
Fig. 2.
Primary neurological symptom prompting evaluation of cerebrospinal fluid.
3.1. CSF pleocytosis
Although the CSF RBC count was only reported for 61/430 (14%) patients [22,28,29,31,35,39,40,44,73,74,77,78,81,89,92,102,110,118,[120], [121], [122], [123],128,133,134,[138], [139], [140],156,165,166,[172], [173], [174], [175], [176],180,184,188,191,192,198,202,204,208,215,220,225,227,233,241,242,246], the CSF white blood cell (WBC) count was provided for 409/430 (95%) patients, 270 (66%) of whom had >0 cells/μL, >1 WBC:1,000 RBC or were noted to have “pleocytosis” (Fig. 3 ) [[5], [6], [7], [8],10,13,[15], [16], [17], [18], [19], [20],[22], [23], [24], [25], [26], [27], [28], [29],[31], [32], [33], [34], [35], [36],[38], [39], [40],[42], [43], [44], [45], [46], [47], [48],[50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92], [93], [94], [95],[98], [99], [100], [101], [102], [103], [104], [105], [106], [107], [108], [109], [110], [111], [112], [113], [114], [115], [116], [117], [118], [119], [120], [121], [122], [123], [124], [125], [126], [127], [128], [129], [130], [131], [132], [133], [134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144], [145], [146], [147], [148], [149], [150]] [[151], [152], [153], [154], [155], [156], [157], [158], [159], [160], [161], [162], [163], [164], [165], [166], [167], [168],[170], [171], [172], [173], [174], [175], [176], [177], [178], [179], [180], [181], [182], [183], [184], [185], [186], [187],[190], [191], [192], [193], [194], [195],[197], [198], [199], [200], [201], [202], [203], [204], [205], [206], [207], [208], [209], [210], [211],[213], [214], [215], [216],[218], [219], [220], [221], [222], [223], [224], [225],227,[229], [230], [231], [232], [233], [234], [235], [236],[238], [239], [240], [241], [242], [243], [244], [245], [246]]. CSF pleocytosis may be indicative of infection, inflammation or other CNS injury [247]. There were 29/409 patients (7%) who had a CSF WBC count of 21–100 cells/μL, all of whom had symptoms that localized to the CNS; 10 (34%) of these patients had CSF RBC counts provided, all of whom had a CSF RBC count of <300 cells/μL; Naz et al. described a patient with headache and indicated that CSF was consistent with viral meningitis, but the CSF WBC count was unclear, as it was noted that he had 90 lymphocytes and 10 neutrophils (no units provided so we were unsure if these were percentages or numbers of cells), so we classified these findings as “increased WBC.” [10,[27], [28], [29],44,75,79,[127], [128], [129],140,145,154,160,161,170,173,183,184,187,198,205,208,216]
Fig. 3.
Cerebrospinal fluid white blood cell count results.
The CSF WBC count was >100 cells/μL for 8/409 patients (2%). Although Abdel-Mannan et al. noted that two patients with encephalopathy had a CSF WBC count of 8,000 cells/μL and 2,000 cells/μL in a table, they indicated in the text that CSF was acellular; we attempted to reach the authors for clarification, but were unsuccessful, so we classified these two patients as having “normal WBC”) [31,75,127,130,171,199,222,241,245]. Mardani et al. reported a 64-year-old woman with encephalopathy who had a CSF WBC count of 1,920 cells/μL with neutrophilic predominance and was found to have a positive CSF SARS-CoV-2 PCR [199]. Similarly, Yousefi et al. described a 9-year-old girl who presented with headache and had a CSF WBC count of 1,870 cells/μL with neutrophilic predominance and a CSF RBC count of 350 cells/μL who was found to have a positive CSF SARS-CoV-2 PCR [241]. These patients are discussed further below. Rebeiz et al. presented a patient with encephalopathy and fever with no respiratory symptoms who had a CSF WBC count of 350 cells/μL (98% mononuclear cells; CSF RBC count not provided) [222]. CSF SARS-CoV-2 PCR was not performed, but he had a progressively worsening leukoencephalopathy over 49 days, then was declared brain dead. Benameur et al. described a 31-year-old woman who had a lumbar puncture due to persistent coma after discontinuation of sedation and was found to have a CSF WBC count of 115 cells/μL with a CSF RBC count of 7,374 cells/μL [31]. She had a negative CSF SARS-CoV-2 PCR, but a positive CSF SARS-CoV-2 antibody, though no testing was done to distinguish intrathecal antibody synthesis from antibody transmission to the CSF due to breakdown of the blood-brain barrier. Imaging showed diffusion changes and edema in the right hemisphere and edema in the cervical cord. Sarma et al. reported a 28-year-old woman with decreased sensation below the fifth thoracic vertebral level, but preserved strength, whose spine imaging showed “widespread elongated signal changes throughout the spinal cord to the conus medullaris and involving the medulla,” who had a CSF WBC count of 125 cells/μL (CSF RBC count was not reported) and “negative antibodies.” [75] Bellon et al. presented a 60-year-old man with a “meningeal syndrome” who had a CSF WBC count of 2,840 cells/μL; CSF SARS-CoV-2 PCR was negative and CSF protein was 460 mg/dL, but no additional data was provided [127]. Zoghi et al. reported a 21-year-old man who presented with quadriparesis and encephalopathy 2 weeks after onset of cough and sore throat and was found to have a CSF WBC count of 150 cells/μL (60% lymphocytes) which increased 3 days later to 250 cells/μL (60% lymphocytes; no CSF RBC count reported) [245]. Imaging revealed a hyperintense lesion in the spinal cord extending >3 segments and bilateral corticospinal tract hyperintensities in the pons and corpus callosum. CSF SARS-CoV-2 PCR, oligoclonal bands, aquaporin-4 antibodies and myelin oligodendrocyte glycoprotein were all negative. Lastly, Guilmot et al. reported a patient who had polyradiculitis involving the cranial nerves and cauda equina 10 days after onset of cough and fever and had a CSF WBC count of 101 cells/μL (CSF RBC count was not reported) [171]. CSF SARS-CoV-2 PCR was negative, CSF protein was normal and oligoclonal bands were matched. Repeat lumbar puncture 12 days later showed improved pleocytosis (WBC count 28 cells/μL).
Of 14 other patients who had quantitative results for CSF WBC count reported on more than one lumbar puncture, it was unchanged for 2/14 (14%) patients (both ≤6 cells/μL), decreased for 6/14 (43%) patients and increased for 6/14 (43%) patients [10,45,51,74,77,78,170,180,184,188,205,218,220,233,246]. The patients whose CSF WBC count increased included 1) a patient with quadriplegia due to Guillain-Barré Syndrome reported by Toscano et al. (0 cells/μL➔4 cells/μL 10 days later), 2) a patient with transverse myelitis reported by Munz et al. (16 cells/μL➔27 cells/μL after an unspecified timeframe), 3) a patient with encephalopathy for 11 days who had normal intracranial imaging reported by Pilotto et al. (18 cells/μL➔38 cells/μL 10 days later), 4) a patient with transverse myelitis reported by Zachariadis et al. (16 cells/μL (92% lymphocytes)➔36 cells/μL (94% lymphocytes) and 5) a patient with a leukoencephalopathy 45 days after diagnosis of COVID-19 by nasopharyngeal SARS-CoV-2 PCR (WBC count 1 cell/μL with RBC count 0 cells/μL➔WBC count 2 cells/μL with RBC count 51 cells/μL 1 week later) reported by Carroll et al. [10,45,51,78,220]
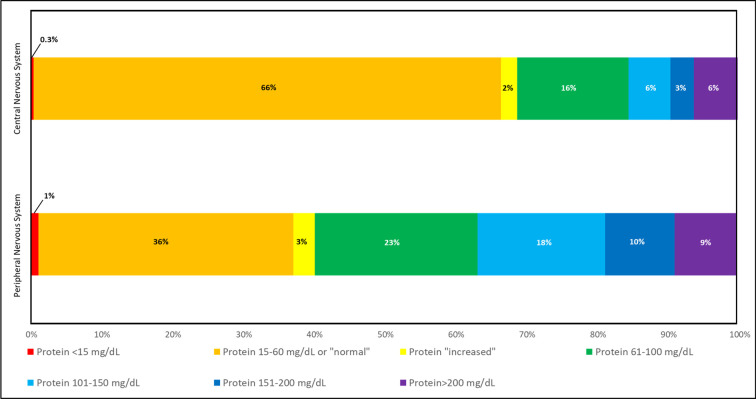
3.2. CSF protein
The protein was included in the CSF results for 397/430 (92%) patients, 160/397 (40%) of whom were noted to have “increased protein” or protein >60 mg/dL (Fig. 4 ) [[5], [6], [7], [8], [9], [10],[13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25],[27], [28], [29], [30], [31], [32], [33], [34], [35], [36],[38], [39], [40],[42], [43], [44], [45], [46], [47], [48],[50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92],[94], [95], [96], [97], [98], [99], [100], [101], [102], [103], [104], [105], [106], [107], [108], [109], [110], [111], [112], [113], [114], [115], [116], [117], [118], [119], [120], [121], [122], [123], [124], [125], [126], [127], [128], [129], [130], [131],[133], [134], [135], [136],[138], [139], [140], [141], [142], [143], [144], [145], [146], [147], [148], [149], [150], [151], [152], [153], [154], [155], [156],158,[160], [161], [162], [163], [164], [165], [166], [167], [168], [169], [170], [171], [172], [173], [174],[176], [177], [178], [179], [180], [181], [182], [183], [184], [185], [186], [187], [188], [189], [190], [191], [192], [193], [194], [195],[197], [198], [199], [200], [201], [202], [203], [204], [205], [206], [207], [208], [209], [210], [211],[213], [214], [215], [216],[218], [219], [220], [221], [222],224,225,[227], [228], [229], [230], [231], [232], [233], [234], [235], [236], [237], [238], [239], [240], [241], [242], [243], [244], [245], [246]]. Hyperproteinorrachia can be indicative of the presence of inflammation or axonal injury and may reflect the existence of intrathecal antibodies [248]. Although Abdel-Mannan et al. indicated in the text of their report that CSF for two patients with encephalopathy had normal protein, their table indicated that the CSF protein was 2.0 g/L (200 mg/dL) and 1.9 g/L (190 mg/dL); we attempted to reach the authors for clarification, but were unsuccessful, so we classified these two patients as having CSF protein 151–200 mg/dL [130]. Similarly, Demirci Otluoglu et al. noted CSF protein was 0.4 mg/L (0.04 mg/dL) and Dharsandiya et al. indicated the CSF protein was 39 g/dL (3,900 mg/dL), but described the protein as normal, and Matos et al. reported a CSF protein of 0.78 mg/dL, but noted it was mildly elevated; we presumed these were meant to be 0.4g/L (40 mg/dL), 39 mg/dL, and 78 mg/dL, respectively [150,151,200]. There were 28/397 (7%) patients who had CSF protein >200 mg/dL (20/28 (71%) with symptoms that localized to the CNS and 8/28 (29%) with symptoms that localized to the PNS) [16,21,31,32,34,44,53,58,78,81,89,104,113,127,128,160,170,173,176,184,199,[220], [221], [222],245].
Fig. 4.
Cerebrospinal fluid protein.
There were 5 patients with CSF protein > 1,000 mg/dL [53,128,176,199]. The aforementioned patient with encephalopathy, a CSF WBC count of 1,920 cells/μL and a positive SARS-CoV-2 PCR reported by Mardani et al., who is discussed further below, had a CSF protein of 9,480 mg/dL [199]. Bodro et al. reported two patients who had fever and a few days of encephalopathy that self-resolved and normal neuroimaging whose lumbar punctures revealed CSF protein of 1,055 mg/dL and 1,155 mg/dL [128]. Both patients had a lymphocytic pleocytosis in the CSF (CSF WBC count of 95 cells/μL with CSF RBC count of 0 cells/μL and CSF WBC count of 90 cells/μL with RBC count of 260 cells/μL, respectively) and a negative CSF SARS-CoV-2 PCR. Hoelscher et al. described a comatose patient who developed bithalamic edema in the setting of thrombosis of the vein of Galen and the bilateral internal cerebral veins who had a CSF protein of 1,104 mg/dL [176]. The CSF WBC count was 10 cells/μL and the CSF RBC count was 401 cells/μL. CSF SARS-CoV-2 PCR was not tested.
Lastly, Pfefferkorn et al. presented a patient who remained quadriplegic and ventilator dependent 1-month after presentation with Guillain-Barré Syndrome who had a normal CSF protein on admission, but it increased to 1,023.1 mg/dL 13 days later [53]. Khodamoradi et al. also documented a marked increase in CSF protein on serial lumbar punctures (the text notes the initial CSF protein was 0.2 mg/dL, but we assume this was meant to be 20 mg/dL, then the subsequent CSF protein was 685 mg/dL (confirmed via personal communication with the authors) and a third CSF protein was normal) in a patient who presented with headache and was found to have a positive CSF SARS-CoV-2 PCR (discussed further below).” [184,249] CSF protein also increased between taps, but not as dramatically, for 11 other patients who had serial CSF protein values reported [10,14,45,51,74,78,135,180,205,220,233]. However, it decreased between taps for a patient with multifocal strokes (314 mg/dL➔normal), a patient with multifocal intracranial and spinal cord hyperintense lesions (281 mg/dL➔111 mg/dL) and much less notably so for a patient with leukoencephalopathy (22 mg/dL➔19 mg/dL) [170,188,245].
3.3. CSF SARS-CoV-2 PCR testing
SARS-CoV-2 PCR testing was performed in the CSF on 303/430 (70%) patients (238/321 (74%) patients with symptoms that localized to the CNS and 65/109 (60%) patients with symptoms that localized to the PNS (Table 1 )) [[5], [6], [7], [8], [9], [10],15,17,22,24,[26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38],[43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59],[68], [69], [70], [71],73,74,[76], [77], [78],81,84,88,89,95,97,98,101,103,105,108,109,111,113,116,119,122,123,125,[127], [128], [129],131,[134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144],[147], [148], [149], [150],152,154,156,157,161,164,165,169,171,177,178,[180], [181], [182],[184], [185], [186], [187],190,191,193,[195], [196], [197], [198], [199], [200], [201],203,204,[205], [206], [207],209,[212], [213], [214], [215], [216],[218], [219], [220], [221],[224], [225], [226], [227],230,[233], [234], [235], [236], [237], [238],[240], [241], [242], [243], [244], [245], [246]]. The CSF SARS-CoV-2 PCR resulted positive for 17/303 (6%) patients, all of whom had symptoms that localized to the CNS (17/238 (7%) patients with symptoms that localized to the CNS): 8/17 (47%) who had encephalopathy/coma +/- seizures, 4/17 (24%) who had headache +/- encephalopathy, 2/17 (12%) who had seizures without encephalopathy, 1/17 (6%) who had cerebellar symptoms, 1/17 (6%) who had paresthesias and 1/17 (6%) who had vision loss (Table 2 ) [[26], [27], [28], [29],150,164,180,195,199,204,224,225,227,236,238,241]. The cycle threshold (Ct; the number of amplification cycles required for the target gene to exceed the threshold, which is inversely related to viral load) was only provided for 2/17 (12%) patients with a positive CSF SARS-CoV-2 PCR (37.12, 37.52 and 36.44 for the N1 gene, but negative for N2 for the patient reported by Moriguchi et al. and 34.29 for the patient reported by Virhammar et al.), though Fadakar et al. noted that a Ct of 35 was the cutoff for a positive SARS-CoV-2 PCR [[26], [27], [28], [29],150,164,180,184,195,199,204,224,225,227,236,238,241]. There were 6/17 (35%) patients who did not have a positive nasopharyngeal/oropharyngeal SARS-CoV-2 PCR, 1 of whom had positive SARS-CoV-2 serum antibodies [26,27,153,184,241,250]. The majority (13/17, 76%) of patients with a positive CSF SARS-CoV-2 PCR were admitted to the hospital because of neurological symptoms [[26], [27], [28], [29],150,164,180,195,204,224,236,241]. There were 11/17 (65%) patients who had acute findings on neuroimaging and 5/17 (29%) patients who had acute findings on chest imaging [26,27,150,153,164,180,199,204,225,227,236,238]. CSF WBC count, when provided, ranged from 0 to 90 cells/μL for every patient except the patients reported by Mardani et al. and Yousefi et al., who had a CSF WBC count of 1,920 cells/μL (90% neutrophils) and 1,870 cells/μL (90% neutrophils, CSF RBC count of 350 cells/μL), respectively [[26], [27], [28], [29],150,164,180,195,199,204,224,225,227,238,241]. The CSF RBC count was provided for 8/17 (47%) patients, 1 of whom had 0 cells/μL; the remainder had a CSF RBC count of 50–1,685 cells/μL [28,29,180,184,204,227,241]. The CSF protein was normal or mildly increased (maximally 115 mg/dL) for 15/17 (88%) patients; the other 2 patients had a CSF protein of 685 mg/dL and 9,480 mg/dL, the latter of which was the same patient who had a CSF WBC count of 1,920 cells/μL [[26], [27], [28], [29],150,164,180,195,199,204,224,225,227,236,238,241]. That patient also had hypoglycorrhachia (10 mg/dL), but the other patients who had a CSF glucose reported had normal glycorrhachia. Notably, the patient who had marked neutrophilic pleocytosis, hyperproteinorrachia and hypoglycorrhachia had a tracheal aspiration culture that grew multidrug resistant Klebsiella pneumoniae [199]. There were 6/17 (35%) patients who had testing to evaluate for intrathecal antibody synthesis: 1 patient had a “high intrathecal oligoclonal IgG synthesis (13.2 mg/dL, reference <3.4 mg/dL);” 1 patient had an elevated CSF:serum IgG ratio (4.48; reported normal 0.82–3.26) and a slightly elevated CSF:serum albumin ratio (7.74, reported normal 1.92–7.3) demonstrating evidence of both possible intrathecal antibody synthesis and breakdown of the blood-brain barrier; 1 patient initially had elevated CSF:serum albumin demonstrating breakdown of the blood-brain barrier, though this subsequently normalized on another lumbar puncture, at which time CSF IgG was noted to be increased and oligoclonal bands were noted to be positive (though no information was provided about whether they were matched); the other 3 patients had oligoclonal bands which were not consistent with intrathecal antibody synthesis (2 had no bands and the third had bands matched to serum) [27,153,225,227,236,238]. CSF biomarkers were measured in 1 patient; he had elevated glial fibrillary acidic protein (GFAP), interleukin-6 (IL-6), neurofilament light chain and tau [236]. The majority of patients with a positive CSF SARS-CoV-2 PCR (14/17 (82%)) recovered, but 1 patient died due to multiorgan failure and 1 remained unresponsive after 15 days (no follow up data was provided for the last patient) [[26], [27], [28], [29],150,164,180,195,199,204,224,225,227,236,238,241].
Table 1.
Cerebrospinal fluid testing for evidence of SARS-CoV-2.
| Test | Central nervous system (n=321) |
Peripheral nervous system (n=109) |
||||
|---|---|---|---|---|---|---|
| Number of patients tested | Number of patients with evidence of intrathecal virus or intrathecal antibody production n (%) | Notes | Number of patients tested | Number of patients with evidence of intrathecal virus or intrathecal antibody production n (%) | Notes | |
| SARS-CoV-2 PCR |
238 |
17 (7%) Positive |
|
65 |
0 Positive/Indeterminate |
|
| 3 (1%) Indeterminate |
|
|||||
| SARS-CoV-2 Antibodies |
49 |
7 (14%) Positive SARS-CoV-2 Antibodies with Evidence of Intrathecal Antibody Synthesis |
|
9 |
0 Positive SARS-CoV-2 Antibodies with Evidence of Intrathecal Antibody Synthesis |
|
| 7 (14%) Positive SARS-CoV-2 Antibodies but Indeterminate if there was Intrathecal Antibody Synthesis |
|
3 (33%) Positive SARS-CoV-2 Antibodies but Indeterminate if there was Intrathecal Antibody Synthesis |
|
|||
| 23 (47%) Positive SARS-CoV-2 Antibodies with no Evidence of Intrathecal Antibody Synthesis |
|
2 (22%) Positive SARS-CoV-2 Antibodies with no Evidence of Intrathecal Antibody Synthesis |
|
|||
| Oligoclonal Bands |
119 |
2 (2%) Positive Oligoclonal Bands with Evidence of Intrathecal Antibody Synthesis |
|
13 |
1 (8%) Positive Oligoclonal Bands with Evidence of Intrathecal Antibody Synthesis |
|
| 3 (3%) Positive Oligoclonal Bands but Indeterminate if there was Intrathecal Antibody Synthesis |
|
0 Positive Oligoclonal Bands but Indeterminate if there was Intrathecal Antibody Synthesis |
||||
| 42 (35%) Positive Oligoclonal Bands with no Evidence of Intrathecal Antibody Synthesis |
|
4 (31%) Positive Oligoclonal Bands with no Evidence of Intrathecal Antibody Synthesis |
|
|||
| Immunoglobulins | 53 | 3 (6%) Immunoglobulin Results Consistent with Intrathecal Antibody Synthesis |
|
9 | 1 (11%) Immunoglobulin Results Consistent with Intrathecal Antibody Synthesis |
|
| 7 (13%) Immunoglobulin Results Consistent with Possible Intrathecal Antibody Synthesis |
|
0 Immunoglobulin Results Consistent with Possible Intrathecal Antibody Synthesis | ||||
| 5 (9%) Immunoglobulin Results Not Suggestive of Intrathecal Antibody Synthesis |
|
2 (22%) Immunoglobulin Results Not Suggestive of Intrathecal Antibody Synthesis |
|
|||
Table 2.
Patients with a positive CSF SARS-CoV-2 PCR.
| Author | Age/Sex | SARS-CoV-2 testing | WBC | Other CSF/neuroimaging | Bloodwork/Chest imaging | Reason for admission | Systemic Symptoms | Neurological symptoms | Time between systemic symptoms and neurological symptoms (Days) | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Demirci Otluoglu et al. [150] | 48M |
|
0 cells/μL |
|
|
Neuro |
|
|
0 | Treated with steroids, hydroxychloroquine, favipiravir, acyclovir, antibiotics and levetiracetam and remained neurologically normal |
| Domingues et al. [153] | 42F |
|
1 cell/μL |
|
|
Neuro |
|
|
N/A | Recovery after 3 weeks without treatment |
| Duong et al. [28] and Huang et al. [29] | 41F |
|
70 cells/μL (RBC 65 cells/μL) |
|
|
Neuro |
|
|
0 | Treated with hydroxychloroquine and levetiracetam and returned to normal after 12 days |
| Fadakar et al. [164] | 47M |
|
10 cells/μL |
|
|
Neuro |
|
|
3 | Treated with lopinavir/ritonavir and symptoms improved markedly after 1 month |
| Kamal et al. [180] | 31M |
|
<5 cells/μL (RBC 150 cells/μL)➔ <5 cells/μL (RBC 50 cells/μL 2 weeks later |
|
|
Neuro |
|
|
2 | Treated with chloroquine, lopinavir/ritonavir, acyclovir and levetiracetam and returned to normal after 2 weeks |
| Khodamoradi et al. [184,249] | 49F |
|
90 cells/μL (RBC 0 cells/μL)➔ 23 cells/μL (RBC 57 cells/μL) 1 week later➔ Normal prior to discharge |
|
|
Neuro |
|
|
N/A | Treated with hydroxychloroquine, azithromycin, Lopinavir/ritonavir, acyclovir and antibiotic and was discharged after 15 days |
| Lo Monaco et al. [195] | 56M |
|
4 cells/μL |
|
|
Neuro |
|
|
7 | Treated with tocilizumab, antibiotics and antivirals and improved rapidly |
| Mardani et al. [199] | 64F |
|
1,920 cells/μL (90% neutrophils) |
|
|
Respiratory |
|
|
21 | Treated with hydroxychloroquine, lopinavir/ritonavir and antibiotics but no follow up data available |
| Mirzaee et al. [204] | 12M |
|
0 cells/uL (RBC 100 cells/uL) |
|
|
Neuro | N/A |
|
N/A | Treated conservatively then discharged home but remained hemiparetic |
| Moriguchi et al. [26] | 24M |
|
12 cells/μL |
|
|
Neuro |
|
|
9 | No improvement after 15 days despite administration of steroids, acyclovir, favipiravir, ceftriaxone, vancomycin and levetiracetam |
| Novi et al. [27] | 64F |
|
22 cells/μL |
|
|
Neuro |
|
|
21 | Vision improved after 2 weeks following administration of steroids and IVIG |
| Rifino et al. [224] | 60M |
|
Normal |
|
|
Neuro | N/A |
|
N/A | Fully recovered after 6 days following treatment with hydroxychloroquine and ariprazole |
| Saitta et al. [225] | 68M |
|
0 cells/μL (RBC 1 cell/μL) |
|
|
Respiratory |
|
|
9 (after sedation discontinued) | Death 21 days after admission |
| Sattar et al. [227] | 44M |
|
11 cells/μL (75% neutrophils; RBC 1,685 cells/μL) |
|
N/A | Respiratory |
|
|
20 | Improved neurologically after initiation of antiepileptic drugs (previously treated with hydroxychloroquine, azithromycin, vitamin C, zinc) and was discharged after 34 days |
| Virhammar et al. [236] | 55F |
|
No pleocytosis noted on 4 lumbar punctures |
|
|
Neuro |
|
|
7 | Gradually improved following treatment with IVIG, acyclovir, plasmapheresis and convalescent plasma and discharged to rehab after 35 days |
| Westhoff et al. [238] | 69M |
|
1 cell/μL |
|
|
Respiratory |
|
|
10 | Improved after treatment with hydroxychloroquine and antiepileptic drugs and was discharged after 38 days |
| Yousefi et al. [241] | 9F |
|
1,870 cells/μL (90% neutrophils; RBC 350 cells/μL) |
|
|
Neuro |
|
|
0 | Improved after treatment with antibiotics and hydroxychloroquine and was discharged after 10 days |
CSF: cerebrospinal fluid; CT: computed tomography; IL: interleukin; IVIG: intravenous immunoglobulin; MRI: magnetic resonance imaging; N/A: not available; PCR: polymerase chain reaction; RBC: red blood cells; WBC: white blood cells
Of the 17 patients with a positive CSF SARS-CoV-2 PCR, 3 (18%) had more than one lumbar puncture [180,184,236]. The patients reported by Kamal et al. and Khodamoradi et al. had a positive CSF SARS-CoV-2 PCR on the first lumbar puncture, but the one reported by Virhammar et al. had two negative CSF SARS-CoV-2 PCRs then a positive CSF SARS-CoV-2 PCR with Ct 34.29 for the N1 gene (although this finding was not reproducible using a commercial PCR assay), and a fourth CSF SARS-CoV-2 PCR was subsequently negative. For all three patients, the CSF WBC count decreased or remained stable and the CSF protein increased over time, though after the patient reported by Khodamoradi et al. had a substantial increase in the CSF protein between the first and second lumbar puncture (20 mg/dL to 685 mg/dL), it normalized on a third lumbar puncture.
In addition to these 17 patients who had a positive CSF SARS-CoV-2 PCR, Eden et al. described 3 patients with encephalopathy whose initial real-time CSF SARS-CoV-2 PCR resulted as indeterminate (Ct 37–40) with a QS6 instrument (Applied Biosystems, Foster City, CA, USA) prompting retesting of stored samples using the Xpert®-Xpress SARS-CoV-2 test (Cepheid, Sunnyvale, USA) which did not detect SARS-CoV-2 RNA [157]. None of these patients had CSF pleocytosis and they all had a normal albumin ratio, reflecting an intact blood-brain barrier, and IgG index, indicative of absence of intrathecal IgG synthesis. The authors noted that these findings are provocative, but that failure to detect viral RNA when the CSF was retested calls the initial findings into question, though retesting of low viral load samples after freezing and thawing can yield inaccurate results. All 3 patients had elevated CSF β2 microglobulin and neopterin.
Lastly, McAbee et al. reported a case of “encephalitis associated with COVID-19” and noted that “nasopharyngeal swab was positive for COVID-19 as well as rhinovirus/enterovirus, but the latter was absent in the CSF PCR.” [202] We attempted to clarify with the authors whether this patient had a CSF SARS-CoV-2 PCR performed and if it was positive, but were unsuccessful, so we did not include him amongst the patients who had a CSF SARS-CoV-2 PCR test performed.
3.4. CSF SARS-CoV-2 antibody testing
CSF antibodies to SARS-CoV-2 were tested in 58/414 (14%) patients who did not have a positive CSF SARS-CoV-2 PCR (49/58 (84%) of whom had symptoms that localized to the CNS and 9/58 (16%) of whom had symptoms that localized to the PNS) [6,13,25,31,43,46,98,101,113,119,127,141,182,193,197,209,220,224,231,233,237,246]. Of these, 42/58 (72%) tested positive for SARS-CoV-2 antibodies in the CSF [6,25,31,43,46,98,119,127,193,197,209,224,231,237]. Keller et al. noted that the 5 patients they reported did not have intrathecal synthesis of SARS-CoV-2 IgG, but did not comment on whether any SARS-CoV-2 antibodies were present in the CSF [182]. Additional testing was performed on the CSF from 32/42 (76%) patients with positive CSF SARS-CoV-2 antibodies in an attempt to distinguish intrathecal antibody synthesis from antibody transmission to the CSF due to breakdown of the blood-brain barrier [25,43,119,127,193,231].
For 7/32 (22%) of these patients, all of whom had symptoms that localized to the CNS, the additional testing was consistent with intrathecal antibody synthesis [25,231]. Using a SARS-CoV-2 epitope Luminex panel, Song et al. evaluated CSF and serum SARS-CoV-2 antibodies in 6 patients and found there were antibodies specific to different regions of the spike protein in both compartments [231]. The 552–589 region of the spike protein was an elevated target specific to the CSF, while the 818–855 region was an elevated target in the serum. Alexopoulos et al. identified 1 patient with encephalopathy who had SARS-CoV-2 antibodies in the CSF and an IgG index of 1.85, indicative of some intrathecal protein synthesis (the authors noted the threshold to identify intrathecal synthesis was an IgG index>1) [25]. Song et al. did not provide outcome data, but the patient described by Alexopoulos et al. patient died [25,231].
The remaining 25/32 (78%) patients with positive SARS-CoV-2 antibodies in the CSF who had additional testing to evaluate if the antibodies were synthesized intrathecally did not have evidence of intrathecal antibody synthesis. Bellon et al. identified 14 patients with SARS-CoV-2 antibodies in the CSF (12 with encephalopathy, 1 with a “meningeal syndrome” and 1 with a neuromuscular disorder) [127]. They evaluated the CSF:serum SARS-CoV-2 IgG index and found that every patient had a ratio <2 (they considered a ratio >2 to be consistent with intrathecal production); 3 patients had a ratio between 1 and 1.22, but none had a ratio >1.22. Oligoclonal bands were tested in 13/14 patients and they all had absent or matched oligoclonal bands. The albumin quotient was tested in 13/14 patients; it was elevated for 8 (62%) patients, indicating breakdown of the blood-brain barrier. Thus, they concluded none of these patients had intrathecal SARS-CoV-2 antibody synthesis. Alexopoulos et al. identified 7 patients with encephalopathy/coma who had SARS-CoV-2 antibodies in the CSF, but a normal IgG index (reported normal <0.77). Of these 7 patients, 2 did not have SARS-CoV-2 antibodies in the CSF initially but had them on a second lumbar puncture 1 week later when the CSF was diluted 1:10, but not when it was diluted 1:100 [25]. To determine if the SARS-CoV-2 CSF antibodies for 2 patients originated due to intrathecal synthesis or transudation, Andriuta et al. calculated the Tibbling-Link, Delpech and transudation indices. For both patients, the indices reflected antibody entry to the CSF via transudation [43]. Senel et al. concluded that though their patient, who had Miller-Fisher Syndrome, had positive SARS-CoV-2 antibodies, these did not develop in the CSF because 1) the SARS-CoV-2 IgG and IgA CSF to blood antibody index was <1; 2) evaluation of all CSF immunoglobulins with the Reiber diagram showed no evidence of intrathecal synthesis; and 3) oligoclonal bands were negative [119]. Lastly, although Lin et al. detected increased levels of IgM (1:64) for SARS-CoV-2 S1 and E proteins in the CSF, the IgG index was normal and oligoclonal bands were negative, suggesting there was no intrathecal synthesis [193].
3.5. CSF oligoclonal bands
CSF oligoclonal bands were tested in 114 patients who did not have a positive CSF SARS-CoV-2 PCR or positive CSF antibodies to SARS-CoV-2 (103 (90%) who had symptoms that localized to the CNS and 11 (10%) who had symptoms that localized to the PNS) [9,10,19,20,35,36,42,44,45,58,69,74,79,81,101,112,113,116,117,126,127,130,135,136,147,149,152,154,160,[168], [169], [170], [171],177,181,182,[185], [186], [187], [188],191,194,203,205,207,209,212,213,215,216,218,233,242,245]. Of these, 35 (31%) had matched CSF and serum bands [9,35,79,112,113,127,147,170,171,177,182,185,187,203,205,207,213,215].
There were 2/114 (2%) patients who did not have a positive CSF SARS-CoV-2 PCR or positive CSF antibodies to SARS-CoV-2 who had oligoclonal bands that were unique to the CSF, reflecting intrathecal antibody synthesis [101,171]. First, a 21-year-old woman who developed ptosis and diplopia a week after she had mild respiratory symptoms was found to have SARS-CoV-2 IgG and IgA antibodies in her serum (but not in her CSF) and positive oligoclonal bands type 2 (not matched in serum) in her CSF [101]. Second, an 80-year-old patient who presented with hallucinations, memory problems and a seizure was found to have a positive nasopharyngeal SARS-CoV-2 PCR, CSF-specific oligoclonal bands and anti-contactin-associated protein 2 (Caspr2) IgG antibodies in serum and CSF [171]. Both patients recovered [101,171].
There were two additional patients who did not have a positive CSF SARS-CoV-2 PCR or positive CSF antibodies to SARS-CoV-2 and may have had oligoclonal bands that were unique to the CSF [154,212]. Dono et al. described an 81-year-old man who developed new-onset refractory status epilepticus while hospitalized with COVID-19 who was noted to have positive oligoclonal bands in his CSF [154]. No further information was provided to clarify if the bands were matched in serum. Lastly, Palao et al. reported a 29-year-old woman with no prior neurologic symptoms who presented with optic neuritis 2 weeks after acute onset of anosmia and myalgia and was found to have positive SARS-CoV-2 serum antibodies and oligoclonal bands in the CSF [212]. Imaging revealed enhancement of the optic nerve and periventricular demyelinating lesions, only one of which was enhancing. This prompted diagnosis of multiple sclerosis. Though the authors assumed her pathogenic process began prior to infection with COVID-19, we cannot conclusively explain the correlation between her imaging findings and clinical presentation. As serum oligoclonal bands were not reported, it is unclear if her bands were matched, but if they were not, it is possible that this reflects intrathecal synthesis of antibodies to SARS-CoV-2.
3.6. CSF immunoglobulins
Of the patients who did not have a positive CSF SARS-CoV-2 PCR, CSF SARS-CoV-2 antibodies or CSF-specific oligoclonal bands, 45 had CSF immunoglobulins measured [9,10,42,45,49,57,58,62,69,74,81,113,117,148,181,182,187,194,205,215,220,242,246]. Of these, 29 (64%) patients had normal immunoassays [42,45,49,57,58,62,69,74,81,113,181,182,187,194,215,242,246].
There were 2/45 (4%) patients who did not have a positive CSF SARS-CoV-2 PCR, CSF SARS-CoV-2 antibodies or CSF-specific oligoclonal bands who had immunoglobulin results consistent with intrathecal antibody synthesis [117,205]. Rajdev et al. presented a patient with Guillain-Barré Syndrome who had an elevated IgG index (14.67; reported normal <0.86) and markedly elevated IgG synthesis (293.5 mg/day; reported normal 9.2 mg/day), with normal albumin levels and absence of oligoclonal bands [117]. The authors commented that these findings suggest intrathecal IgG synthesis, as there was no clear evidence of blood-brain barrier breakdown, but concluded that his polyneuropathy was postinfectious. Monti et al. reported a patient with encephalopathy who had positive CSF N-Methyl-D-aspartate (NMDA) antibodies and an IgG index of 1.45 (reported normal <0.7) 5 weeks after presentation [205]. It was 0.67 1 month earlier. Oligoclonal bands were initially matched, then were absent. Both patients improved and were discharged from the hospital [117,205].
Another 6/45 (13%) patients who did not have a positive CSF SARS-CoV-2 PCR, CSF SARS-CoV-2 antibodies or CSF-specific oligoclonal bands had CSF immunoglobulins consistent with possible intrathecal antibody synthesis [42,117,168,187]. Kremer et al. identified four patients with encephalopathy who had elevated CSF IgG levels from 3.8 to 16.9 mg/dL [187]. They did not stipulate the normal value or provide serum IgG or CSF albumin, so it is not feasible to interpret whether these findings reflect intrathecal IgG synthesis. However, two of these patients had matched oligoclonal bands in CSF and serum, suggesting these patients did not have unique intrathecal antibodies. The authors felt their findings were likely immune-mediated. Dogan et al. described a patient with encephalopathy who had an IgG index of 0.78 (upper limit of normal reported by the authors was 0.6), but the patient also had a CSF IgG of 3.23 mg/dL (upper limit of normal reported by the authors of 3.4 mg/dL) and negative oligoclonal bands [42,117]. The authors concluded that these findings were likely the result of autoimmune/antibody-mediated inflammation. Similarly, Ghosh et al. described a patient who presented with coma and seizures and was found to have a frontoparietal hyperintense lesion with surrounding edema and foci of hemorrhage who had an elevated IgG index [168]. The value of the index was not provided. Additionally, oligoclonal bands were absent in the CSF. The authors did not conclude these findings were consistent with intrathecal antibody synthesis.
3.7. CSF autoimmune antibodies
Autoimmune antibodies were tested in the CSF of 77/430 (18%) patients [10,25,30,34,44,45,74,122,135,136,141,148,154,164,169,171,177,182,185,186,191,193,194,[205], [206], [207],209,213,215,216,220,233,234,242,245,246]. Of the 77 patients who had autoimmune antibodies tested in the CSF, 4 (5%) had positive autoimmune antibodies [30,169,171,205].
There were two patients with NMDA receptor antibodies in the CSF [30,205]. Both patients presented with fever and psychosis and were found to have COVID-19 then developed dyskinesias. Imaging was normal. They improved after treatment with intravenous immunoglobulins (IVIG) and steroids.
Guilmot et al. reported the aforementioned patient who presented with hallucinations, memory problems and a seizure and had anti-contactin-associated protein 2 (Caspr2) IgG antibodies in serum and CSF [171]. Neuroimaging was normal and an electroencephalogram showed slowing. Steroids were administered and plasmapheresis was performed after which there were no seizures.
Lastly, a 72-year-old man who presented with cerebellar symptoms 17 days after fever and odynophagia and, in addition to having a positive nasopharyngeal SARS-CoV-2 PCR and serum IgG and IgM to SARS-CoV-2, he had autoantibodies directed against the nuclei of Purkinje cells, striatal and hippocampal neurons in the CSF (1:96) and serum (1:25,000) [169]. The intensity and reactivity in the serum and CSF observed at the same IgG concentration ruled out intrathecal autoantibody synthesis, as did the absence of CSF specific oligoclonal bands. MRI was normal. He was treated with IVIG and steroids and his symptoms improved.
3.8. Other CSF biomarkers
Other CSF biomarkers were tested in the CSF of 57/430 (13%) patients (Table 3 ), 45 (79%) of whom were reported to have elevation of at least one CSF biomarker [10,24,25,30,31,98,111,119,128,135,136,149,157,165,182,191,205,215,217,227,231,236]. Notably, there is not consensus on the reference values for biomarkers and reported norms vary between labs.
Table 3.
CSF biomarkers in patients with COVID-19 based on PCR or serologic testinga.
ACE: angiotensin converting enzyme; AMS: altered mental status; CSF: cerebrospinal fluid; CXCL: chemokine ligand; GFAP: Glial fibrillary acidic protein; HEM: hemiparesis; I: indeterminate (virus present but with Ct above cutoff of 37); IL: interleukin; INF: interferon; IP: interferon gamma-induced protein; M: matched in serum and CSF; N: noted to be normal with no specific value/range provided by authors; NfL: neurofilament light chain; OPH: ophthalmoparesis; PAR: paraparesis; PCR: polymerase chain reaction; pNfH: phosphorylated neurofilament heavy chain; QUAD: quadriparesis; RBC: red blood cells; SZ: seizures; TNF: tumor necrosis factor; WBC: white blood cells; ~: value interpreted based on review of graph; K: thousand;
Values that are elevated are bolded (normal ranges specified by authors are noted here with alternate ranges included when there was discrepancy in normal limits between authors): [10,25,30,98,111,128,135,149,157,165,182,191,205,215] IgG Index 0.1–0.7 or <0.77 or <0.63; ACE CSF 0-2.5 U/L; Amyloid-β-42 CSF 650–2000 pg/mL; β2 microglobulin serum <2.1 mg/L and CSF <1.8 mg/L; IL-1β serum <0.21 pg/mL (or <0.001 pg/mL) and CSF 0.1–0.5 pg/mL (or <2.56 pg/mL); IL-2R serum 440–1435 pg/mL and CSF range assumed equal to serum due to absence of standardized values; IL-6 serum 0.76–6.38 pg/mL (or <4 pg/mL or <5.9 pg/mL or 0–6.5 pg/mL or <10 pg/mL) and CSF 2.1–9.6 pg/mL (or <1.3 pg/mL or <2.5 pg/mL or <4 pg/mL or <5.9 pg/mL or <6.5 pg/mL or <7 pg/mL); IL-8 serum 6.7–16.2 pg/mL (or <70 pg/mL) and CSF 32.6–88 pg/mL (or <72 pg/mL or <70 pg/mL); IL-10 serum 1.8–3.8 pg/mL (or <9 pg/mL) and CSF range assumed equal to serum due to absence of standardized values (or <2.5 pg/mL or <9 pg/mL); IL-17A control data serum ~0 pg/mL and CSF ~10 pg/mL: INF-α serum <0.99 pg/mL and CSF range assumed equal to serum due to absence of standardized values; IP-10 serum 37.2–222 pg/mL and CSF range assumed equal to serum due to absence of standardized values; MCP-1 control data serum ~0 pg/mL and CSF ~140 pg/mL; Neopterin serum <8.8 nmol/L and CSF <5.8 nmol/L; NfL CSF <1850 pg/mL (or <1,577 pg/mL); pNfH CSF <560 pg/mL; Tau CSF <479 pg/mL (or 150–450 pg/mL); TNF-α serum 7.78–12.2 pg/mL (or <8.1 pg/mL) and CSF 0.2–3.7 pg/mL (or <0.2 pg/mL or <8.1 pg/mL).
There were 27 patients who had CSF IL-6 measured, 20 (74%) of whom had an increased concentration based on the author’s reported normal; 11/20 patients with increased CSF IL-6 concentration had serum IL-6 concentration measured, and this was elevated for all 11 patients [10,30,31,98,111,128,135,149,165,182,191,205,215,217,236]. Keller et al. measured IL-6 in the CSF and serum of 5 patients (2 with hemiparesis, 1 who was comatose, 1 who was encephalopathic and 1 who had seizures) and found it was elevated in 4/5 patients, but was lower than it was in serum [182]. CSF IL-6 was mildly elevated in two patients with encephalopathy reported by Delorme et al. (13 pg/mL and 16 pg/mL; reported normal <6.5 pg/mL; serum levels not reported) [149]. However, Le Guennec et al. reported that the second patient reported by Delorme et al. also had an elevated serum IL-6 (28.8 pg/mL with reported normal <6.5 pg/mL). The two patients presented by Bodro et al. who had CSF protein >1,000 mg/dL in the setting of transient encephalopathy had increased CSF IL-6 (190 pg/mL and 25 pg/mL; reported normal <7 pg/mL); serum values were not reported [128]. Perrin et al. found that 3/5 patients with encephalopathy had an elevated CSF IL-6 [215]. Only 2 of these patients had IL-6 measured in the serum, but both had an elevated concentration. Cani et al. measured IL-6 in the CSF and serum of a patient who initially had akinetic mutism after being weaned off sedation for acute respiratory failure, then improved and had only mild dysexecutive syndrome and finally recovered after 43 days [135]. CSF WBC count was normal, CSF SARS-CoV-2 PCR and oligoclonal bands were negative and CSF protein was mildly increased. CSF IL-6 was elevated on initial lumbar puncture 21 days after symptoms onset (55.1 pg/mL; reported normal <5.9 pg/mL) at which time serum IL-6 was slightly above normal (9.1 pg/mL; same limits as CSF), but repeat lumbar puncture 14 days later revealed normal IL-6 in both CSF and serum. Farhadian et al. compared biomarker concentration in the CSF and serum of a patient who had encephalopathy and seizures to the CSF and serum from two healthy controls and found that IL-6 was elevated in both CSF and serum in the patient with COVID-19 as compared to the control patients [165]. Lastly, both of the patients with NMDA receptor antibodies had elevated CSF IL-6: one had a concentration of 39 pg/mL (normal reported <7 pg/mL) and did not have serum levels reported [30]; the second had an elevated CSF IL-6 compared to 12 subjects with functional neurologic disorders (4.58 pg/mL 1 week after presentation and 5.75 pg/mL 1 month later) and elevated serum IL-6 compared to the established norm of <10 pg/mL (52 pg/mL and 206 pg/mL, respectively) [205]. He also had elevated CSF interleukin-8 (IL-8) compared to the control patients.
In addition to that patient, there were 16 other patients who had CSF IL-8 measured, 16/17 (94%) of whom had elevated CSF IL-8; 6 patients with elevated CSF IL-8 had serum IL-8 measured, all of whom had elevated serum concentrations too [10,31,98,111,135,149,165,205,217]. In comparison to historical controls with normal cognition and no viral illness, Benameur et al. found that 3 patients with CSF SARS-CoV-2 antibodies who were comatose or profoundly encephalopathic had increased levels of CSF IL-6, IL-8, interleukin-10 (IL-10), interferon gamma-induced protein-10 (IP-10) and TNF-α [31]. CSF cytokine levels for these patients were compared to those from patients with HIV-associated neurocognitive disorders and it was noted that elevated CSF IL-8 and IL-10 appeared to be unique to the patients with neurological complications of COVID-19. Inflammatory makers were not evaluated in blood. Gigli et al. performed cytokine testing in the CSF and serum of a 53-year-old man who presented to a hospital with paraparesis, paresthesias and ataxic gait 55 days after he had fever and diarrhea and was found to have a positive CSF SARS-CoV-2 IgG and IgM with specific reactivity against nucleocapsid and spike 2 glycoprotein [98]. IL-8 was markedly elevated (121 pg/mL; normal reported 32.6–88 pg/mL) in CSF and mildly elevated in serum (26 pg/mL; normal reported 6.7–16.2 pg/mL). Manganotti et al. measured IL-8 in the CSF and serum of 3 patients who had Guillain-Barré Syndrome [111]. IL-8 was elevated in CSF for all three patients, and was higher than it was in serum (42.6 pg/mL in CSF and 17.8 pg/mL in serum; 96 pg/mL in CSF and 55 pg/mL in serum; and 22.7 pg/mL in CSF and 20 pg/mL in serum). Pilotto et al. evaluated CSF cytokines in a 60-year-old man who presented with five days of encephalopathy, fever and cough, who was found to have lymphocytic pleocytosis (WBC count of 18 cells/μL) and hyperproteinorrachia (127.2 mg/dL) [10,217]. He had a negative CSF SARS-CoV-2 PCR and no evidence of intrathecal antibody synthesis, oligoclonal bands or autoimmune antibodies. MRI of the brain was normal. CSF IL-8 was markedly increased (>1,100 pg/mL; normal reported elsewhere <72 pg/mL) and was over ten times higher than the serum concentration (98.3 pg/mL) prompting the authors to equate their findings to immune effector cell-associated neurotoxicity syndrome (ICANS, also known as CAR T-cell neurotoxicity) [251]. After treatment with five days of steroids, lopinavir/ritonavir, hydroxychloroquine, acyclovir and ampicillin, he returned to normal, and repeat CSF testing showed IL-8 decreased substantially to 97 pg/mL. Lastly, Song et al. compared CSF and serum concentration of IL-8, IL-1β, interleukin-12 (IL-12) and fibroblast growth factor-2 (FGF-2) for six patients with positive SARS-CoV-2 antibodies (3 with encephalopathy, 2 with headache and 1 with seizure) to CSF from controls [231]. Data was presented cumulatively, but they noted that all of these cytokines were elevated in the CSF of patients with COVID-19 compared with controls, but were not elevated in the plasma.
β2 microglobulin, a biomarker for monocytic activation in the CNS, was increased in the CSF of 6/7 patients [10,119,157,217]. Pilotto et al. found that CSF β2 microglobulin was increased in a patient with encephalopathy (3.06 mg/L; no reference range provided) [10,217]. Eden et al. found elevated CSF and serum β2 microglobulin in 5/5 patients with encephalopathy (3 of whom had an initial CSF SARS-CoV-2 PCR result as indeterminate (Ct 37-40), prompting retesting which did not detect SARS-CoV-2 RNA) [157]. Because these findings were not associated with viral detection, pleocytosis or disruption of the blood-brain barrier (based on the albumin index), the authors concluded they were reflective of an indirect effect of systemic infection.
To evaluate for axonal injury, the CSF was tested for 1) 14-3-3 in 9 patients with encephalopathy, 2) neurofilament light chain in 8 patients with encephalopathy and 3) phosphorylated neurofilament heavy chain in 1 patient with ophthalmoplegia [10,24,25,119,157,217]. There were 4/9 (44%) patients who were positive for 14-3-3 in the CSF, 1 of whom was negative on an initial lumbar puncture then positive when a second CSF sample was obtained [24,25]. All of the patients with 14-3-3 detected in the CSF also had SARS-CoV-2 antibodies in the CSF at a 1:10 dilution and 3/4 had SARS-CoV-2 antibodies detected in the CSF at 1:100 dilution, though they all had a CSF:serum SARS-CoV-2 IgG index <1, and 2/4 had an elevated albumin index [25]. Eden et al. found that neurofilament light chain was elevated in the CSF of 2/6 patients with encephalopathy (including one whose initial CSF SARS-CoV-2 PCR resulted as indeterminate), which the authors felt may have been attributed to hypoxia [157]. Virhammar et al. found that CSF neurofilament light chain was markedly increased in a patient who was comatose on admission and was found to have a positive CSF SARS-CoV-2 PCR on the third lumbar puncture performed; serum concentration was not reported [236]. Senel et al. found elevated phosphorylated neurofilament heavy chain in the CSF (2,131 pg/mL; reported normal <30 pg/mL) of a patient with Miller-Fisher Syndrome who had positive CSF SARS-CoV-2 IgG and IgA, but no evidence of intrathecal antibody production [119]. After treatment with IVIG for five days, his symptoms resolved. The authors checked neurofilament light chain in blood 2 days later, and 16 days after that, and found that it was elevated (61 and 58 pg/mL, respectively; no reference range provided).
There were two patients who had the concentration of GFAP, a biomarker for gliosis, measured in the CSF [10,217,236]. The patient presented by Virhammar et al. who had a positive CSF SARS-CoV-2 PCR on the third lumbar puncture had markedly elevated GFAP on day 12 (~4,000 pg/mL) which then decreased to normal by day 30 (~500 pg/mL) [236]. Serum levels were not reported.
Amyloid-β-42 and tau, biomarkers for Alzheimer-type pathologic changes in the CNS, were both tested in the CSF of 4 patients [10,119,149,217,227,236]. Delorme et al. described a patient who had markedly elevated CSF tau (2,000 pg/mL; reported normal 150–450 pg/mL) [149]. Virhammar et al. presented a patient who was comatose on admission and was found to have a positive CSF SARS-CoV-2 PCR on the third lumbar puncture performed [236]. The patient had elevated CSF tau which continued to increase over the course of 30 days from ~2,000 pg/mL to ~7,000 pg/mL. The patient gradually improved and was discharged to rehab after 35 days.
Because it is postulated that SARS-CoV-2 enters cells via the angiotensin-converting enzyme-2 (ACE2) receptor, Bodro et al. measured ACE levels in the CSF of the two aforementioned patients with transient encephalopathy who had CSF protein >1,000 mg/dL [128]. Both patients had elevated CSF ACE (15.5 U/L and 10.9 U/L; reported normal 0–2.5 U/L). The authors acknowledged that it is unclear how to interpret this abnormality, particularly given that these patients had negative CSF SARS-CoV-2 PCRs and recovered fully after a few days, making infection of the CNS unlikely, but suggested it could be relevant.
4. Discussion
In this systematic review, we identified 430 patients with COVID-19 diagnosed based on PCR or serology who had CSF obtained due to a neurological symptom [[5], [6], [7], [8], [9], [10],[13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92], [93], [94], [95], [96], [97], [98], [99], [100], [101], [102], [103], [104], [105], [106], [107], [108], [109], [110], [111], [112], [113], [114], [115], [116], [117], [118], [119], [120], [121], [122], [123], [124], [125], [126], [127], [128], [129], [130], [131], [132], [133], [134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144], [145], [146], [147], [148], [149], [150], [151], [152], [153], [154], [155], [156], [157], [158], [159], [160], [161], [162], [163], [164], [165], [166], [167], [168], [169], [170], [171], [172], [173], [174], [175], [176], [177], [178], [179], [180], [181], [182], [183], [184], [185], [186], [187], [188], [189], [190], [191], [192], [193], [194], [195], [196], [197], [198], [199], [200], [201], [202], [203], [204], [205], [206], [207], [208], [209], [210], [211], [212], [213], [214], [215], [216], [217], [218], [219], [220], [221], [222], [223], [224], [225], [226], [227], [228], [229], [230], [231], [232], [233], [234], [235], [236], [237], [238], [239], [240], [241], [242], [243], [244], [245], [246]]. A paucity of patients in this cohort had a positive CSF SARS-CoV-2 PCR; of 303 patients who had SARS-CoV-2 PCR tested in the CSF, 17 (6%) patients, all of whom had symptoms that localized to the CNS, had a positive result and an additional 3 (1%) had an indeterminate result [[5], [6], [7], [8], [9], [10],15,17,22,24,[26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38],[43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59],[68], [69], [70], [71],73,74,[76], [77], [78],81,84,88,89,95,97,98,101,103,105,108,109,111,113,116,119,122,123,125,[127], [128], [129],131,[134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144],[147], [148], [149], [150],152,154,156,157,161,164,165,169,171,177,178,[180], [181], [182],[184], [185], [186], [187],190,191,193,[195], [196], [197], [198], [199], [200], [201],203,204,[205], [206], [207],209,[212], [213], [214], [215], [216],[218], [219], [220], [221],[224], [225], [226], [227],230,[233], [234], [235], [236], [237], [238],[240], [241], [242], [243], [244], [245], [246]]. Furthermore, only 14/252 (6%) patients who had direct or indirect evaluation for CSF SARS-CoV-2 antibodies (oligoclonal bands or immunoglobulins), had evidence of intrathecal antibody synthesis [6,9,10,13,19,20,25,31,35,36,[42], [43], [44], [45], [46],49,57,58,62,69,74,79,81,98,101,112,113,116,117,119,126,127,130,135,136,141,[147], [148], [149],152,154,160,[168], [169], [170], [171],177,181,182,[185], [186], [187], [188],191,193,194,197,203,205,207,209,212,213,215,216,218,220,224,231,233,237,242,245,246]. Inconsistencies in the evaluation of CSF and serum and challenges associated with interpretation of laboratory results preclude determination of a finite rate of viral neuroinvasion in this cohort. However, neuroinvasion by SARS-CoV-2 appears to be rare, and it is likely that neurological symptoms in patients with COVID-19 are usually the result of hypoxic-ischemic injury, toxic-metabolic changes, stroke, or a parainfectious or post-infectious inflammatory response due to cytokine release syndrome or molecular mimicry between COVID-19 antibodies and peripheral nerve glycolipids, neuronal or glial cells [[5], [6], [7], [8], [9], [10],[13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75], [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92], [93], [94], [95], [96], [97], [98], [99], [100], [101], [102], [103], [104], [105], [106], [107], [108], [109], [110], [111], [112], [113], [114], [115], [116], [117], [118], [119], [120], [121], [122], [123], [124], [125], [126], [127], [128], [129], [130], [131], [132], [133], [134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144], [145], [146], [147], [148], [149], [150], [151], [152], [153], [154], [155], [156], [157], [158], [159], [160], [161], [162], [163], [164], [165], [166], [167], [168], [169], [170], [171], [172], [173], [174], [175], [176], [177], [178], [179], [180], [181], [182], [183], [184], [185], [186], [187], [188], [189], [190], [191], [192], [193], [194], [195], [196], [197], [198], [199], [200], [201], [202], [203], [204], [205], [206], [207], [208], [209], [210], [211], [212], [213], [214], [215], [216], [217], [218], [219], [220], [221], [222], [223], [224], [225], [226], [227], [228], [229], [230], [231], [232], [233], [234], [235], [236], [237], [238], [239], [240], [241], [242], [243], [244], [245], [246]].
As is evident by the fact that 303/430 (70%) patients we identified had CSF SARS-CoV-2 PCR testing performed, it is feasible to perform PCR testing for SARS-CoV-2 in CSF [[5], [6], [7], [8], [9], [10],15,17,22,24,[26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38],[43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59],[68], [69], [70], [71],73,74,[76], [77], [78],81,84,88,89,95,97,98,101,103,105,108,109,111,113,116,119,122,123,125,[127], [128], [129],131,[134], [135], [136], [137], [138], [139], [140], [141], [142], [143], [144],[147], [148], [149], [150],152,154,156,157,161,164,165,169,171,177,178,[180], [181], [182],[184], [185], [186], [187],190,191,193,[195], [196], [197], [198], [199], [200], [201],[203], [204], [205], [206], [207],209,[212], [213], [214], [215], [216],[218], [219], [220], [221],[224], [225], [226], [227],230,[233], [234], [235], [236], [237], [238],[240], [241], [242], [243], [244], [245], [246]]. In fact, PCR testing for the N2 gene target of SARS-CoV-2 was noted to have the highest sensitivity in CSF when compared with a nasopharyngeal swab, bronchoalveolar lavage, sputum, plasma or stool [252]. Despite this, many hospitals lacked the ability to perform SARS-CoV-2 PCR testing in CSF early in the pandemic [63]. Additionally, viral detection via PCR testing is not 100% sensitive due to genetic variability in the virus itself and technical factors [253]. It has also been suggested that isolation of SARS-CoV-2 in CSF may be challenging because of rapid CSF clearance, low titers or delayed sampling [33,37,254]. Further, CSF SARS-CoV-2 PCR testing is not 100% specific for intrathecal virus, in part because a sample can be contaminated from shed airborne virus or blood contamination [255]. The CSF RBC count was only available for 8/17 (47%) patients we identified who had a positive SARS-CoV-2 CSF PCR, one of whom had a CSF RBC count of 1,685 with a CSF WBC count of 11 cells/μL, which may indicate the CSF SARS-CoV-2 PCR was falsely positive due to a traumatic lumbar puncture [[26], [27], [28], [29],153,164,180,184,195,199,204,224,225,227,236,238,241]. The SARS-CoV-2 PCR can also be falsely positive if there is a pre-analytical error [256]. The process by which CSF SARS-CoV-2 PCR was evaluated varied and this information was not always provided, so we did not delineate the testing technique for each patient who had CSF SARS-CoV-2 PCR testing performed in this review; however, it is important to note that the sensitivity and specificity varies between tests.
Data on the Ct was only available for 2/17 (12%) patients with a positive CSF SARS-CoV-2 PCR, and they both had a high Ct (37.12, 37.52 and 36.44 for one patient and 34.29 for the second (whose positive result was not reproducible)), which is indicative of a low viral load, though the author of another report on a patient with a positive CSF SARS-CoV-2 PCR noted that a cycle threshold of 35 was the cutoff for a positive SARS-CoV-2 PCR [[26], [27], [28], [29],150,164,180,184,195,199,204,224,225,227,236,238,241]. While many labs use a cutoff of 40 to consider a test positive, some classify a Ct of 37-40 as indeterminate [157,257]. Accordingly, when Eden et al. repeated CSF SARS-CoV-2 PCR testing for three patients who had a Ct of 37-40, they found no evidence of SARS-CoV-2 RNA in the CSF [157].
There was considerable variability in the CSF WBC count of patients we identified who had a positive CSF SARS-CoV-2 PCR [[26], [27], [28], [29],150,164,180,184,195,199,204,224,225,227,238,241]. There were 2 patients who had a CSF WBC count >1,500 cells/μL, 90% of which were neutrophils [199,241]. Viral encephalitis is typically characterized by a CSF lymphocytic pleocytosis, while a neutrophilic pleocytosis is generally associated with a bacterial infection, but a retrospective review of 182 patients with confirmed viral infections in the CNS found that 25% had CSF neutrophilic pleocytosis [258]. However, it is worth noting that the patient reported by Mardani et al. had hypoglycorrhachia (CSF glucose of 10 mg/dL when blood glucose was 162 mg/dL) and a tracheal aspiration culture that grew Klebsiella pneumoniae in conjunction with her neutrophilic pleocytosis which raises the questions of whether 1) she, in fact, had culture negative meningitis and 2) her CSF SARS-CoV-2 PCR was a true positive [199]. On the other end of the spectrum, there were 6 patients with a positive CSF SARS-CoV-2 PCR who had a CSF WBC count of only 1-5 cells/μL, 2 with “no pleocytosis” and 2 with a CSF WBC count of 0 cells/μL [150,153,180,195,204,224,225,236,238]. Absence of pleocytosis in general, and acellular CSF in particular, is atypical in the setting of viral encephalitis [259,260]. However, there have been reports of patients without pleocytosis who had a positive CSF PCR for Enterovirus, Echovirus, Adenovirus and Herpes and CSF can be acellular in the early stages of herpes encephalitis [259,261]. Nonetheless, it is also feasible that the patients with acellular CSF had a false-positive CSF SARS-CoV-2 PCR, particularly given that for one patient, this was the only means employed to diagnose COVID-19 since a rapid COVID-19 test was negative and no other PCR or serologic testing was performed [262]. However, 5 of the other 16 (31%) patients with a positive CSF SARS-CoV-2 PCR also had negative nasopharyngeal swabs, though one had positive serum SARS-CoV-2 antibodies [[26], [27], [28], [29],153,164,180,184,195,199,204,225,227,236,241]. Chest imaging was consistent with viral pneumonia for 2/5 (40%) patients who were only diagnosed with COVID-19 based on a positive SARS-CoV-2 PCR in the CSF [26,150,153,184,241]. Domingues et al. postulated that SARS-CoV-2 may be more persistent in the CNS because it is an immunoprivileged site [153].
Serological testing for viruses may have a higher sensitivity and specificity than PCR testing, but like PCR testing, the sensitivity and specificity among serological tests vary [263,264]. The mere presence of antibodies in CSF is not indicative of antibody synthesis in the CSF, as antibodies, or the cells that secrete them, can be transmitted to the CSF via a damaged blood-brain barrier or a traumatic tap [31,265,266]. The presence of CSF SARS-CoV-2 antibodies, oligoclonal bands or elevated CSF IgG is notable, but measuring IgG synthesis or comparing CSF and serum IgG or oligoclonal bands can help facilitate a distinction between intrathecal synthesis of antibodies and transudation of antibodies through a damaged blood-brain barrier [25,43,119,127,193,231]. However, it is important to note that evidence of intrathecal antibody synthesis based on oligoclonal band or immunoglobulin analysis in this patient population does not elucidate the type of antibodies being synthesized, so it is feasible some patients had intrathecal synthesis of antibodies other than SARS-CoV-2 antibodies, such as antibodies to myelin or autoimmune antibodies [169,171,205]. Song et al.’s finding that anti-SARS-CoV-2 antibodies in the CSF of six patients had different epitope specificity compared with antibodies in the serum, suggesting that there are compartment-specific immune responses, is provocative and warrants further investigation in a larger cohort and consideration about how or if this finding can be used translationally [231].
Although CSF testing of biomarkers is not universally available, we identified 57 patients who had CSF biomarkers measured, 45 (79%) of whom had elevation of at least one CSF biomarker suggesting the presence of inflammation, axonal injury, gliosis and Alzheimer-type pathologic changes [10,24,25,30,31,98,111,119,128,135,136,149,157,165,182,191,205,215,217,227,231,236]. Interestingly, some of these patients had elevation of biomarkers in the CSF, but not the serum, which is similar to ICANS, the neurotoxicity that occurs after treatment with chimeric antigen receptor T-cell therapy [10,217,231]. While further data is needed, these findings suggest that there are compartment-specific immune responses to SARS-CoV-2 [231]. Further, Gigli et al. suggested that some patients may have a specific genetic predisposition to postinfectious inflammatory syndromes and associated intrathecal cytokine responses after COVID-19 [98]. Of course, it is imperative to recognize that elevated CSF and serum biomarkers is not clearly indicative of neuroinvasion as this can result from other mechanisms such as hypoxic injury [267,268]. Additional research into treatment implications and the diagnostic and prognostic value of CSF biomarkers in patients with COVID-19 who have neurological symptoms is needed.
Of course, this review of the neuroinvasiveness of COVID-19 and associated CSF changes is limited based on publication bias, in general, and our search methodology, in particular. There were two pre-print case reports of patients who had CSF testing by Xiang et al. and Cai et al. cited in reviews that we did not include in our discussion, as one was not accessible and one was not in English. However, both of these patients reportedly had a positive SARS-CoV-2 PCR in CSF [269,270]. Additionally, we noted three letters-to-the-editor that mentioned patients in China who had positive CSF SARS-CoV-2 PCRs, but we did not include these cases as no details were provided; similarly Finsterer et al. mentioned an unpublished report of a patient in India with a positive SARS-CoV-2 PCR in CSF [[271], [272], [273], [274]].
On the other hand, we excluded 1) documents that described cases in which CSF was obtained from a patient with COVID-19 who did not have neurological symptoms, such as a report by Bajwa et al. who described two patients with COVID-19 who had CSF collected during administration of spinal anesthesia [275]; and 2) those that provided cumulative CSF data for a series of patients, but did not include clinical details for individual patients.
A number of cumulative analyses were published by authors in France [[276], [277], [278], [279], [280]]. We report these results here, but it is important to acknowledge that we believe it is very likely some of these patients were described in the case reports/case series included in our review. Further, as there are multiple cumulative analyses using data from one country in a brief time period, some of which have overlapping authors, we are unsure if any patients were included in more than one cumulative analysis. For example, our review includes 1) 15 patients that were described by Kremer et al. in a case series on patients with neurologic manifestations of COVID-19 who were admitted to 11 French hospitals from March 16 to April 9, 2020 and 2) 5 patients with COVID-19 and neurologic symptoms admitted to one French hospital between March 9 and April 9, 2020 reported by Perrin et al. (on which Kremer was a co-author), but Kremer was also an author on two cumulative analyses [187,215,278,279]. In the first, Kremer et al. studied CSF results from 37 patients with COVID-19 and neurological symptoms admitted to 16 hospitals between March 23 and April 27, 2020 and found that 1/28 (4%) had a positive CSF SARS-CoV-2 PCR, 14/31 (45%) had a high CSF WBC count, 11/31 (36%) had elevated CSF protein, 7/16 (44%) had elevated CSF IgG, 6/13 (46%) had oligoclonal bands with a mirror pattern, 2/6 (33%) had elevated CSF IL-6 and 1/6 (17%) had elevated CSF IL-10 [278]. In the second, Helms et al. evaluated CSF in 25 patients with encephalopathy admitted to 2 hospitals between March 3 and May 5, 2020 and found that 1 (4%) had a positive SARS-CoV-2 PCR in CSF, 3 (12%) had elevated CSF WBC, 8 (32%) had elevated CSF protein, 9 (36%) had elevated CSF IgG, 15 (52%) had oligoclonal bands with a mirror pattern, 7 (28%) had elevated CSF IL-6 and 2 (8%) had elevated CSF IL-10 [279,280]. In another French series which includes overlapping patients from the aforementioned reports, Meppiel et al. retrospectively reviewed data from adult patients with COVID-19 who had neurological symptoms and were admitted to 46 hospitals in France from March 16 to April 27, 2020 and identified: 21 patients with encephalitis (defined as altered mental status lasting ≥24 h and either CSF WBC >5 cells/μL or compatible acute lesion on brain MRI) and found that 2 (10%) had a positive CSF SARS-CoV-2 PCR, 14 (67%) had a CSF WBC count >5 cells/μL and 12 (57%) had CSF protein>45 mg/dL; 36 patients with COVID-encephalopathy (defined as altered mental status lasting ≥24 h that does not meet criteria for encephalitis and was not attributed to another toxic or metabolic etiology) who had CSF testing, none of whom had a positive SARS-CoV-2 PCR in CSF or a CSF WBC count >5 cells/μL, but 8 (22%) who had CSF protein>45 mg/dL; and 14 patients with Guillain-Barré Syndrome who had CSF testing, none of whom had a positive CSF SARS-CoV-2 PCR, 1 (7%) who had a CSF WBC count >5 cells/μL and 8 (57%) who had CSF protein>45 mg/dL [277]. Lastly, Destras et al. described the results of a retrospective screen of CSF samples obtained between February and May from 555 patients (23 of whom had COVID-19 diagnosed by respiratory PCR) at one hospital in France that demonstrated only 2 patients had a slightly positive CSF SARS-CoV-2 PCR (with Ct of 32 and 35); both of these patients died and their brain biopsies showed no evidence of viral invasion, prompting the conclusion that the CSF was contaminated by blood [276].
In addition to these reports from France, cumulative CSF results from patients with COVID-19 have been described by other authors, a few examples of which are noted here [224,[281], [282], [283]]. Espindola et al. analyzed the CSF of 58 patients in Brazil with COVID-19 diagnosed via nasopharygneal SARS-CoV-2 PCR who had a variety of neurological complaints and found: 10 (17%) patients had a CSF WBC count >5 cells/μL (median 2 cells/μL (interquartile range (IQR) 1–4 cells/μL); 16 (28%) patients had CSF protein >45 mg/dL (median 35 mg/dL (IQR 25.8-48 mg/dL); 2 (3%) patients had detectable SARS-CoV-2 RNA in the CSF (one with persistent headache and one with acute disseminated encephalomyelitis); 3/38 (8%) patients had oligoclonal bands restricted to the CSF consistent with intrathecal IgG synthesis, 4/38 (11%) patients had matched CSF and serum oligoclonal bands while 31/38 (82%) patients had no oligoclonal bands; and median CSF neurofilament light chain was 1,694 pg/mL (IQR 1,091–3,358 pg/mL) and median CSF tau was 318.3 pg/mL (IQR 173-457.4 pg/mL) [281].
Neumann et al. also published a cumulative analysis of CSF data from 30 patients with COVID-19 admitted to six hospitals in Germany from March to June 2020 [282]. There were two patients with a CSF WBC count >8 cells/μL, one who had herpes encephalitis and one who had an intracranial hemorrhage. CSF protein was <200 mg/dL for all patients. CSF SARS-CoV-2 PCR was negative for all patients. Although the patient with herpes encephalitis had unique oligoclonal bands in the CSF, the remaining 24 patients who had testing for oligoclonal bands had matched or absent oligoclonal bands.
In a review of the neurological manifestations of COVID-19 observed in a hospital in Italy from February 23 to April 30, 2020, Rifino et al. noted that 2/21 (10%) patients with encephalopathy had a positive CSF SARS-CoV-2 PCR (details on one patient were provided so he is included in our review) and 0/8 patients with Guillain-Barré Syndrome had a positive CSF SARS-CoV-2 PCR [224].
Remsik et al. analyzed the CSF of 13 patients with cancer, COVID-19 and neurological symptoms [283]. None of the patients had a positive CSF SARS-CoV-2 PCR. There was one patient who had antibodies to SARS-CoV-2, but the authors attributed this to breakdown of the blood-brain barrier. CSF biomarkers were obtained and compared to CSF biomarkers from 1) patients matched by age, cancer type and presence of brain metastases, 2) patients with ICANS and 3) patients with autoimmune encephalitis. The patients with COVID-19 had a significant accumulation of 12 inflammatory mediators in the CSF (IL-6, IL-8, IFN-γ, CXCL-1, CXCL-6, CXCL-9, CXL-10, CXCL-11, CCL-8, CCL-20, MMP-10 and 4E-BP1), similar to the changes seen in patients with ICANS.
The most definitive means to assess for viral neuroinvasion in patients with COVID-19 is via neuropathology studies. In a review of 81 postmortem neuropathological examinations, Al-Sarraj et al. concluded that it was difficult to parse out whether the findings were the result of viral neuroinvasion, hypoxia, multiorgan failure, cytokine storming, reduced immune response or strokes; they recommended the need for large-scale molecular and cellular investigations of brain tissue and CSF in conjunction with data on the neurological evaluation and neuroimaging for each patient [284]. Notably, Paniz-Mondolfi reported the presence of SARS-CoV-2 identified in brain tissue via ultrastructural analysis and molecular testing despite a negative SARS-CoV-2 PCR in CSF [285].
It is important to note that CSF results can change over the course of a patient’s illness. This is evident in the results from some of the patients included in this review who had more than one lumbar puncture performed [10,14,25,45,51,53,74,135]. Although we documented the number of days between onset of symptoms of COVID-19 and development of neurological symptoms in Supplementary Tables 1 and 2, we did not indicate the number of days after onset of neurological symptoms that CSF was obtained. Further, the fact that CSF results were not obtained at a specific time point relative to development of neurological symptoms certainly impacts our results. It has previously been suggested that detection of viral neuroinvasion via a positive CSF PCR is highest when CSF is obtained 5 days after onset of neurological symptoms [286].
Additionally, CSF (particularly CSF protein) can be abnormal due to a number of etiologies other than COVID-19 which were not addressed herein. These include, but are not limited to, male sex, body mass index, diabetes mellitus, hypothyroidism, hypoparathyroidism, Cushing’s disease, uremia, medications (such as phenytoin and phenothiazines) [287,288]. Elevated CSF protein has been observed in a wide range of neuropsychiatric disorders and we did not document past medical/neuropsychiatric history for patients included in this review [288,289]. Further, though perspectives on the relationship between CSF protein and age vary, it has been suggested that CSF protein may increase with age throughout adulthood and we did not take age into consideration when we reviewed CSF protein [[287], [288], [289], [290]].
Finally, it has been postulated that viruses in the CSF of patients with neurological disorders can sometimes be bystanders, rather than causative agents of disease, so a positive SARS-CoV-2 PCR or evidence of intrathecal antibody production does not definitively indicate neuroinvasion is responsible for a given constellation of symptoms [291]. In a review of 43 brains postmortem, Marschke et al. found that the presence of SARS-CoV-2 did not seem to be associated with the severity of neuroimmune activation [292].
5. Conclusion
Neurological symptoms in patients with COVID-19 can be attributed to hypoxic-ischemic injury, stroke, toxic-metabolic changes, a parainfectious or post-infectious inflammatory response or direct neuroinvasion. Based on this review of 430 patients with COVID-19 who had neurological symptoms and CSF studies performed, detection of viral neuroinvasion via CSF SARS-CoV-2 PCR or evaluation for intrathecal antibody synthesis is rare. Though data is limited, CSF biomarkers may have diagnostic, therapeutic and prognostic benefit in patients with COVID-19 who have neurological symptoms. Additional research and postmortem studies are needed.
Author contributions
Ariane Lewis was responsible for conception and design, data collection, analysis and interpretation of data, drafting the manuscript and final approval of the manuscript. Kara Melmed was responsible for data collection, critical revision of the manuscript and final approval of the manuscript. Jennifer Frontera, Dimitris Placantonakis, Jennifer Lighter, Steven Galetta and Laura Balcer were responsible for critical revision of the manuscript and final approval of the manuscript.
Declaration of competing interest
None.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jns.2021.117316.
Appendix A. Supplementary data
Supplemental Table 1: CSF Results for Patients with COVID-19 and Disorders of the Central Nervous System and Supplemental Table 2: CSF Results for Patients with COVID-19 and Disorders of the Peripheral Nervous System
References
- 1.Desforges M., Le Coupanec A., Dubeau P., et al. Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses. 2019;12(1):14. doi: 10.3390/v12010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;92(6):552–555. doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Desforges M., Le Coupanec A., Brison E., Meessen-Pinard M., Talbot P.J. Neuroinvasive and neurotropic human respiratory coronaviruses: potential neurovirulent agents in humans. Adv. Exp. Med. Biol. 2014;807:75–96. doi: 10.1007/978-81-322-1777-0_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dalakas M.C. Guillain-Barré syndrome: the first documented COVID-19-triggered autoimmune neurologic disease: more to come with myositis in the offing. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) doi: 10.1212/NXI.0000000000000781. e781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oguz-Akarsu E., Ozpar R., Mirzayev H., et al. Guillain-Barré syndrome in a patient with minimal symptoms of COVID-19 infection. Muscle Nerve. 2020;62(3):e54–e57. doi: 10.1002/mus.26992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Helbok R., Beer R., Löscher W., et al. Guillain-Barré syndrome in a patient with antibodies against SARS-COV-2. Eur. J. Neurol. 2020;27(9):1754–1756. doi: 10.1111/ene.14388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chan J.L., Ebadi H., Sarna J.R. Guillain-Barré syndrome with facial diplegia related to SARS-CoV-2 infection. Can. J. Neurol. Sci. 2020;47(6):852–854. doi: 10.1017/cjn.2020.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gutiérrez-Ortiz C., Méndez A., Rodrigo-Rey S., et al. Miller Fisher Syndrome and polyneuritis cranialis in COVID-19. Neurology. 2020;95(5):e601–e605. doi: 10.1212/WNL.0000000000009619. [DOI] [PubMed] [Google Scholar]
- 9.Assini A., Benedetti L., Di Maio S., Schirinzi E., Del Sette M. New clinical manifestation of COVID-19 related Guillain-Barrè syndrome highly responsive to intravenous immunoglobulins: two Italian cases. Neurol. Sci. 2020;41(7):1657–1658. doi: 10.1007/s10072-020-04484-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pilotto A., Odolini S., Masciocchi S., et al. Steroid-responsive encephalitis in coronavirus disease 2019. Ann. Neurol. 2020;88(2):423–427. doi: 10.1002/ana.25783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Covidence Systematic Review Software. Veritas Health Innovation; Melbourne, Australia: 2020. Available at www.covidence.org. [Google Scholar]
- 12.Moher D., Liberati A., Tetzlaff J., Altman D.G., Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J. Clin. Epidemiol. 2009;62(10):1006–1012. doi: 10.1016/j.jclinepi.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 13.Siepmann T., Kitzler H.H., Lueck C., Platzek I., Reichmann H., Barlinn K. Neuralgic amyotrophy following infection with SARS-CoV-2. Muscle Nerve. 2020;62(4):E68–E70. doi: 10.1002/mus.27035. [DOI] [PubMed] [Google Scholar]
- 14.Tard C., Maurage C.A., de Paula A.M., et al. Anti-pan-neurofascin IgM in COVID-19-related Guillain-Barré syndrome: evidence for a nodo-paranodopathy. Neurophysiol. Clin. 2020;50(5):397–399. doi: 10.1016/j.neucli.2020.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tiet M.Y., Alshaikh N. Guillain-Barre syndrome associated with COVID-19 infection: a case from the UK. BMJ Case Rep. 2020;13(7) doi: 10.1136/bcr-2020-236536. e236536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wada S., Nagasaki Y., Arimizu Y., et al. Neurological disorders identified during treatment of a SARS-CoV-2 infection. Intern. Med. 2020;59(17):2187–2189. doi: 10.2169/internalmedicine.5447-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Webb S., Wallace V.C., Martin-Lopez D., Yogarajah M. Guillain-Barré syndrome following COVID-19: a newly emerging post-infectious complication. BMJ Case Rep. 2020;13(6) doi: 10.1136/bcr-2020-236182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yaqoob H., Jain A., Epelbaum O. A unique case of Guillain-Barre syndrome related to Covid-19 infection. Chest. 2020;158(4 Supplement):A771. [Google Scholar]
- 19.Zito A., Alfonsi E., Franciotta D., et al. COVID-19 and Guillain-Barré Syndrome: a case report and review of literature. Front. Neurol. 2020;11:909. doi: 10.3389/fneur.2020.00909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abdi S., Ghorbani A., Fatehi F. The association of SARS-CoV-2 infection and acute disseminated encephalomyelitis without prominent clinical pulmonary symptoms. J. Neurol. Sci. 2020;416:117001. doi: 10.1016/j.jns.2020.117001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Abdulsalam M.A., Abdulsalam A.J., Shehab D. Generalized status epilepticus as a possible manifestation of COVID-19. Acta Neurol. Scand. 2020;142(4):297–298. doi: 10.1111/ane.13321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abel D., Shen M.Y., Abid Z., et al. Encephalopathy and bilateral thalamic lesions in a child with MIS-C associated with COVID-19. Neurology. 2020;95(16):745–748. doi: 10.1212/WNL.0000000000010652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abenza-Abildúa M.J., Novo-Aparicio S., Moreno-Zabaleta R., et al. Encephalopathy in severe SARS-CoV2 infection: inflammatory or infectious? Int. J. Infect. Dis. 2020;98:398–400. doi: 10.1016/j.ijid.2020.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Affes Z., Bouvard E.J., Levy P., Dussaule C., Grateau G., Haymann J.P. COVID-19 presenting with confusion: an unusual but suggestive electroencephalography pattern of encephalitis. J. Clin. Neurophysiol. 2020 doi: 10.1097/WNP.0000000000000795. [DOI] [PubMed] [Google Scholar]
- 25.Alexopoulos H., Magira E., Bitzogli K., et al. Anti-SARS-CoV-2 antibodies in the CSF, blood-brain barrier dysfunction, and neurological outcome: studies in 8 stuporous and comatose patients. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(6) doi: 10.1212/NXI.0000000000000893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moriguchi T., Harii N., Goto J., et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Novi G., Rossi T., Pedemonte E., et al. Acute disseminated encephalomyelitis after SARS-CoV-2 infection. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) doi: 10.1212/NXI.0000000000000797. e797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duong L., Xu P., Liu A. Meningoencephalitis without respiratory failure in a young female patient with COVID-19 infection in Downtown Los Angeles, early April 2020. Brain Behav. Immun. 2020;87 doi: 10.1016/j.bbi.2020.04.024. 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huang Y.H., Jiang D., Huang J.T. SARS-CoV-2 detected in cerebrospinal fluid by PCR in a case of COVID-19 encephalitis. Brain Behav. Immun. 2020;87 doi: 10.1016/j.bbi.2020.05.012. 149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Panariello A., Bassetti R., Radice A., et al. Anti-NMDA receptor encephalitis in a psychiatric Covid-19 patient: a case report. Brain Behav. Immun. 2020;87:179–181. doi: 10.1016/j.bbi.2020.05.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Benameur K., Agarwal A., Auld S.C., et al. Encephalopathy and encephalitis associated with cerebrospinal fluid cytokine alterations and coronavirus disease, Atlanta, Georgia, USA, 2020. Emerg. Infect. Dis. 2020;26(9):2016–2021. doi: 10.3201/eid2609.202122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dixon L., Varley J., Gontsarova A., et al. COVID-19-related acute necrotizing encephalopathy with brain stem involvement in a patient with aplastic anemia. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) doi: 10.1212/NXI.0000000000000789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ye M., Ren Y., Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav. Immun. 2020;88:945–946. doi: 10.1016/j.bbi.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bernard-Valnet R., Pizzarotti B., Anichini A., et al. Two patients with acute meningoencephalitis concomitant with SARS-CoV-2 infection. Eur. J. Neurol. 2020;27(9):e43–e44. doi: 10.1111/ene.14298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Parsons T., Banks S., Bae C., Gelber J., Alahmadi H., Tichauer M. COVID-19-associated acute disseminated encephalomyelitis (ADEM) J. Neurol. 2020;267(10):2799–2802. doi: 10.1007/s00415-020-09951-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Afshar H., Yassin Z., Kalantari S., et al. Evolution and resolution of brain involvement associated with SARS- CoV2 infection: a close clinical – paraclinical follow up study of a case. Mult. Scler. Relat. Disord. 2020;43:102216. doi: 10.1016/j.msard.2020.102216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zanin L., Saraceno G., Panciani P.P., et al. SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir. 2020;162(7):1491–1494. doi: 10.1007/s00701-020-04374-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Karimi N.S.R.A., Rouhani N. Frequent convulsive seizures in an adult patient with COVID-19: a case report. Iran. Red. Crescent Med. J. 2020;22(3) [Google Scholar]
- 39.Filatov A., Sharma P., Hindi F., Espinosa P.S. Neurological complications of coronavirus disease (COVID-19): encephalopathy. Cureus. 2020;12(3) doi: 10.7759/cureus.7352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Espinosa P.S., Rizvi Z., Sharma P., Hindi F., Filatov A. Neurological complications of coronavirus disease (COVID-19): encephalopathy, MRI brain and cerebrospinal fluid findings: case 2. Cureus. 2020;12(5) doi: 10.7759/cureus.7930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020:e119–e120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dogan L., Kaya D., Sarikaya T., et al. Plasmapheresis treatment in COVID-19-related autoimmune meningoencephalitis: case series. Brain Behav. Immun. 2020;87:155–158. doi: 10.1016/j.bbi.2020.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Andriuta D., Roger P.A., Thibault W., et al. COVID-19 encephalopathy: detection of antibodies against SARS-CoV-2 in CSF. J. Neurol. 2020;267(10):2810–2811. doi: 10.1007/s00415-020-09975-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sotoca J., Rodríguez-Álvarez Y. COVID-19-associated acute necrotizing myelitis. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) doi: 10.1212/NXI.0000000000000803. e803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Munz M., Wessendorf S., Koretsis G., et al. Acute transverse myelitis after COVID-19 pneumonia. J. Neurol. 2020;267(8):2196–2197. doi: 10.1007/s00415-020-09934-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gigli G.L., Bax F., Marini A., et al. Guillain-Barré syndrome in the COVID-19 era: just an occasional cluster? J. Neurol. 2020:1–3. doi: 10.1007/s00415-020-09911-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.El Otmani H., El Moutawakil B., Rafai M.A., et al. Covid-19 and Guillain-Barré syndrome: more than a coincidence! Rev. Neurol. 2020;176(6):518–519. doi: 10.1016/j.neurol.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ottaviani D., Boso F., Tranquillini E., et al. Early Guillain-Barré syndrome in coronavirus disease 2019 (COVID-19): a case report from an Italian COVID-hospital. Neurol. Sci. 2020;41(6):1351–1354. doi: 10.1007/s10072-020-04449-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Coen M., Jeanson G., Culebras Almeida L.A., et al. Guillain-Barré syndrome as a complication of SARS-CoV-2 infection. Brain Behav. Immun. 2020;87:111–112. doi: 10.1016/j.bbi.2020.04.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sancho-Saldaña A., Lambea-Gil Á., Liesa J.L.C., et al. Guillain-Barré syndrome associated with leptomeningeal enhancement following SARS-CoV-2 infection. Clin. Med. 2020;20(4):e93–e94. doi: 10.7861/clinmed.2020-0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Toscano G., Palmerini F., Ravaglia S., et al. Guillain-Barré syndrome associated with SARS-CoV-2. N. Engl. J. Med. 2020;382(26):2574–2576. doi: 10.1056/NEJMc2009191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Alberti P., Beretta S., Piatti M., et al. Guillain-Barré syndrome related to COVID-19 infection. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(4) doi: 10.1212/NXI.0000000000000741. e741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pfefferkorn T., Dabitz R., von Wernitz-Keibel T., Aufenanger J., Nowak-Machen M., Janssen H. Acute polyradiculoneuritis with locked-in syndrome in a patient with Covid-19. J. Neurol. 2020;267(7):1883–1884. doi: 10.1007/s00415-020-09897-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Juliao Caamaño D.S., Alonso Beato R. Facial diplegia, a possible atypical variant of Guillain-Barré Syndrome as a rare neurological complication of SARS-CoV-2. J. Clin. Neurosci. 2020;77:230–232. doi: 10.1016/j.jocn.2020.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bigaut K., Mallaret M., Baloglu S., et al. Guillain-Barre syndrome related to SARS-CoV-2 infection. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) doi: 10.1212/NXI.0000000000000785. e785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fernández-Domínguez J., Ameijide-Sanluis E., García-Cabo C., García-Rodríguez R., Mateos V. Miller-Fisher-like syndrome related to SARS-CoV-2 infection (COVID 19) J. Neurol. 2020;267(9):2495–2496. doi: 10.1007/s00415-020-09912-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Arnaud S., Budowski C., Ng Wing Tin S., Degos B. Post SARS-CoV-2 Guillain-Barré syndrome. Clin. Neurophysiol. 2020;131(7):1652–1654. doi: 10.1016/j.clinph.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Su X.W., Palka S.V., Rao R.R., Chen F.S., Brackney C.R., Cambi F. SARS-CoV-2-associated Guillain-Barré syndrome with dysautonomia. Muscle Nerve. 2020;62(2):e48–e49. doi: 10.1002/mus.26988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Riva N., Russo T., Falzone Y.M., et al. Post-infectious Guillain-Barré syndrome related to SARS-CoV-2 infection: a case report. J. Neurol. 2020;267(9):2492–2494. doi: 10.1007/s00415-020-09907-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lascano A.M., Epiney J.B., Coen M., et al. SARS-CoV-2 and Guillain-Barré syndrome: AIDP variant with favorable outcome. Eur. J. Neurol. 2020;27(9):1751–1753. doi: 10.1111/ene.14368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhao H., Shen D., Zhou H., Liu J., Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 2020;19(5):383–384. doi: 10.1016/S1474-4422(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Scheidl E., Canseco D.D., Hadji-Naumov A., Bereznai B. Guillain-Barré syndrome during SARS-CoV-2 pandemic: a case report and review of recent literature. J. Peripher. Nerv. Syst. 2020;25(2):204–207. doi: 10.1111/jns.12382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Padroni M., Mastrangelo V., Asioli G.M., et al. Guillain-Barré syndrome following COVID-19: new infection, old complication? J. Neurol. 2020;267(7):1877–1879. doi: 10.1007/s00415-020-09849-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Camdessanche J.P., Morel J., Pozzetto B., Paul S., Tholance Y., Botelho-Nevers E. COVID-19 may induce Guillain-Barré syndrome. Rev. Neurol. 2020;176(6):516–518. doi: 10.1016/j.neurol.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Manganotti P., Pesavento V., Buoite Stella A., et al. Miller Fisher syndrome diagnosis and treatment in a patient with SARS-CoV-2. J. Neuro-Oncol. 2020;26(4):605–606. doi: 10.1007/s13365-020-00858-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Reyes-Bueno J.A., García-Trujillo L., Urbaneja P., et al. Miller-Fisher syndrome after SARS-CoV-2 infection. Eur. J. Neurol. 2020;27(9):1759–1761. doi: 10.1111/ene.14383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Abdelhady M., Elsotouhy A., Vattoth S. Acute flaccid myelitis in COVID-19. BJR Case Rep. 2020;6(3):20200098. doi: 10.1259/bjrcr.20200098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Águila-Gordo D., Manuel Flores-Barragán J., Ferragut-Lloret F., et al. Acute myelitis and SARS-CoV-2 infection. A new etiology of myelitis? J. Clin. Neurosci. 2020;80:280–281. doi: 10.1016/j.jocn.2020.07.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Baghbanian S.M., Namazi F. Post COVID-19 longitudinally extensive transverse myelitis (LETM)-a case report. Acta Neurol. Belg. 2020:1–2. doi: 10.1007/s13760-020-01497-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chakraborty U., Chandra A., Ray A.K., Biswas P. COVID-19-associated acute transverse myelitis: a rare entity. BMJ Case Rep. 2020;13(8):e238668. doi: 10.1136/bcr-2020-238668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chow C.C.N., Magnussen J., Ip J., Su Y. Acute transverse myelitis in COVID-19 infection. BMJ Case Rep. 2020;13(8):e236720. doi: 10.1136/bcr-2020-236720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Durrani M., Kucharski K., Smith Z., Fien S. Acute transverse myelitis secondary to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): a case report. Clin. Pract. Cases Emerg. Med. 2020;4(3):344–348. doi: 10.5811/cpcem.2020.6.48462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kaur H., Mason J.A., Bajracharya M., et al. Transverse myelitis in a child with COVID-19. Pediatr. Neurol. 2020;112:5–6. doi: 10.1016/j.pediatrneurol.2020.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Maideniuc C., Memon A.B. Acute necrotizing myelitis and acute motor axonal neuropathy in a COVID-19 patient. J. Neurol. 2020:1–3. doi: 10.1007/s00415-020-10145-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sarma D., Bilello L.A. A CASE report of acute transverse myelitis following novel coronavirus infection. Clin. Pract. Cases Emerg. Med. 2020;4(3):321–323. doi: 10.5811/cpcem.2020.5.47937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Shaw V.C., Chander G., Puttanna A. Neuromyelitis optica spectrum disorder secondary to COVID-19. Br. J. Hosp. Med. 2020;81(9):1–3. doi: 10.12968/hmed.2020.0401. [DOI] [PubMed] [Google Scholar]
- 77.Valiuddin H., Skwirsk B., Paz-Arabo P. Acute transverse myelitis associated with SARS-CoV-2: a case-report. Brain Behav. Immun. Health. 2020;5 doi: 10.1016/j.bbih.2020.100091. 100091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zachariadis A., Tulbu A., Strambo D., Dumoulin A., Di Virgilio G. Transverse myelitis related to COVID-19 infection. J. Neurol. 2020:1–3. doi: 10.1007/s00415-020-09997-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhou S., Jones-Lopez E.C., Soneji D.J., Azevedo C.J., Patel V.R. Myelin oligodendrocyte glycoprotein antibody-associated optic neuritis and myelitis in COVID-19. J. Neuroophthalmol. 2020;40(3):398–402. doi: 10.1097/WNO.0000000000001049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Abbaslou M.A., Karbasi M., Mozhdehipanah H. A rare axonal variant of guillain-barre syndrome as a neurological complication of covid-19 infection. Archiv. Iran. Med. 2020;23(10):718–721. doi: 10.34172/aim.2020.93. [DOI] [PubMed] [Google Scholar]
- 81.Abrams R.M.C., Kim B.D., Markantone D.M., et al. Severe rapidly progressive Guillain-Barré syndrome in the setting of acute COVID-19 disease. J. Neuro-Oncol. 2020;26(5):797–799. doi: 10.1007/s13365-020-00884-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Agosti E., Giorgianni A., D’Amore F., Vinacci G., Balbi S., Locatelli D. Is Guillain-Barrè syndrome triggered by SARS-CoV-2? Case report and literature review. Neurol. Sci. 2020:1–6. doi: 10.1007/s10072-020-04553-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ameer N., Shekhda K.M., Cheesman A. Guillain-Barré syndrome presenting with COVID-19 infection. BMJ Case Rep. 2020;13(9):236978. doi: 10.1136/bcr-2020-236978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Atakla H.G., Noudohounsi M., Sacca H., Tassiou N.R.A., Noudohounsi W.C., Houinato D.S. Acute Guillain-Barré polyradiculoneuritis indicative of COVID-19 infection: a case report. Pan Afr. Med. J. 2020;35(Suppl. 2):150. doi: 10.11604/pamj.supp.2020.35.150.25745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Boostani R., Talab F.R., Meibodi N.T., Zemorshidi F. COVID-19 associated with sensorimotor polyradiculoneuropathy and skin lesions: a case report. J. Neuroimmunol. 2020;350:577434. doi: 10.1016/j.jneuroim.2020.577434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bueso T., Elmassry M., El Nawaa S.E.N., Ball S., Solis X., Test V. Covid-19 infection complicated by guillain-barre syndrome: a case report. Chest. 2020;158(4 Supplement):A522–A523. [Google Scholar]
- 87.Busillo V., Lerza M.C., Gargiulo M.G., et al. A case of Guillain-Barre syndrome associated with SARS-CoV-2 infection. Pharmacologyonline. 2020;2:321–324. [Google Scholar]
- 88.Civardi C., Collini A., Geda D.J., Geda C. Antiganglioside antibodies in Guillain-Barré syndrome associated with SARS-CoV-2 infection. J. Neurol. Neurosurg. Psychiatry. 2020;91(12):1361–1362. doi: 10.1136/jnnp-2020-324279. [DOI] [PubMed] [Google Scholar]
- 89.Curtis M., Bhumbra S., Felker M.V., et al. Guillain-Barré syndrome in a child with COVID-19 infection. Pediatrics. 2020 doi: 10.1542/peds.2020-015115. e2020015115. [DOI] [PubMed] [Google Scholar]
- 90.Defabio A.C., Scott T.R., Stenberg R.T., Simon E.L. Guillain-Barré syndrome in a patient previously diagnosed with COVID-19. Am. J. Emerg. Med. 2020:S0735–6751-0. doi: 10.1016/j.ajem.2020.07.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Diez-Porras L., Vergés E., Gil F., Vidal M.J., Massons J., Arboix A. Guillain-Barré-Strohl syndrome and COVID-19: case report and literature review. Neuromusc. Disord. 2020;30(10):859–861. doi: 10.1016/j.nmd.2020.08.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Ebrahimzadeh S.A., Ghoreishi A., Rahimian N. Guillain-Barré Syndrome associated with the coronavirus disease 2019 (COVID-19) Neurol. Clin. Pract. 2020;209 doi: 10.1212/CPJ.0000000000000879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Elkhouly A., Kaplan A.C. Noteworthy neurological manifestations associated with COVID-19 infection. Cureus. 2020;12(7) doi: 10.7759/cureus.8992. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 94.Faqihi F., Alharthy A., Memish Z.A., Kutsogiannis D.J., Brindley P.G., Karakitsos D. Peripheral neuropathy in severe COVID-19 resolved with therapeutic plasma exchange. Clin. Case Rep. 2020;8(12):3234–3239. doi: 10.1002/ccr3.3397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Frank C.H.M., Almeida T.V.R., Marques E.A., et al. Guillain-Barré syndrome associated with SARS-CoV-2 infection in a pediatric patient. J. Trop. Pediatr. 2020 doi: 10.1093/tropej/fmaa044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Garcia-Manzanedo S., Lopez De La Oliva Calvo L., Ruiz Alvarez L. Guillain-Barre syndrome after covid-19 infection. Med. Clin. 2020;155(8) doi: 10.1016/j.medcle.2020.06.019. 366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Garnero M., Del Sette M., Assini A., et al. COVID-19-related and not related Guillain-Barré syndromes share the same management pitfalls during lock down: the experience of Liguria region in Italy. J. Neurol. Sci. 2020;418:117114. doi: 10.1016/j.jns.2020.117114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Gigli G.L., Vogrig A., Nilo A., et al. HLA and immunological features of SARS-CoV-2-induced Guillain-Barre syndrome. Neurol. Sci. 2020;41(12):3391–3394. doi: 10.1007/s10072-020-04787-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Granger A., Omari M., Jakubowska-Sadowska K., Boffa M., Zakin E. SARS-CoV-2-associated guillain-barre syndrome with good response to plasmapheresis. J. Clin. Neuromuscul. Dis. 2020;22(1):58–59. doi: 10.1097/CND.0000000000000310. [DOI] [PubMed] [Google Scholar]
- 100.Hirayama T., Hongo Y., Kaida K., Kano O. Guillain-Barré syndrome after COVID-19 in Japan. BMJ Case Rep. 2020;13(10):e239218. doi: 10.1136/bcr-2020-239218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Huber M., Rogozinski S., Puppe W., et al. Postinfectious onset of myasthenia gravis in a COVID-19 patient. Front. Neurol. 2020;11 doi: 10.3389/fneur.2020.576153. 576153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hutchins K.L., Jansen J.H., Comer A.D., et al. COVID-19-associated bifacial weakness with paresthesia subtype of Guillain-Barré syndrome. AJNR Am. J. Neuroradiol. 2020;41(9):1707–1711. doi: 10.3174/ajnr.A6654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Khaja M., Gomez G.P.R., Santana Y., et al. A 44-year-old hispanic man with loss of taste and bilateral facial weakness diagnosed with Guillain-Barré Syndrome and Bell’s Palsy associated with SARS-CoV-2 infection treated with intravenous immunoglobulin. Am. J. Case Rep. 2020;21 doi: 10.12659/AJCR.927956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Khalifa M., Zakaria F., Ragab Y., et al. Guillain-Barré Syndrome associated with severe acute respiratory syndrome coronavirus 2 detection and coronavirus disease 2019 in a child. J. Pediat. Infect. Dis. Soc. 2020;9(4):510–513. doi: 10.1093/jpids/piaa086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Kilinc D., van de Pasch S., Doets A.Y., Jacobs B.C., van Vliet J., Garssen M.P.J. Guillain-Barré syndrome after SARS-CoV-2 infection. Eur. J. Neurol. 2020;27(9):1757–1758. doi: 10.1111/ene.14398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Korem S., Gandhi H., Dayag D.B. Guillain-Barré syndrome associated with COVID-19 disease. BMJ Case Rep. 2020;13(9):e237215. doi: 10.1136/bcr-2020-237215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lampe A., Winschel A., Lang C., Steiner T. Guillain-Barré syndrome and SARS-CoV-2. Neurol. Res. Pract. 2020;2(1):19. doi: 10.1186/s42466-020-00066-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Liberatore G., De Santis T., Doneddu P.E., Gentile F., Albanese A., Nobile-Orazio E. Clinical reasoning: a case of COVID-19 associated pharyngeal-cervical-brachial variant of Guillain-Barre syndrome. Neurology. 2020;95(21):978–983. doi: 10.1212/WNL.0000000000010817. [DOI] [PubMed] [Google Scholar]
- 109.Lima M.A., Silva M.T.T., Soares C.N., et al. Peripheral facial nerve palsy associated with COVID-19. J. Neuro-Oncol. 2020:1–4. doi: 10.1007/s13365-020-00912-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lowery M.M., Taimur Malik M., Seemiller J., Tsai C.S. Atypical variant of Guillain Barre syndrome in a patient with COVID-19. J. Crit. Care Med. 2020;6(4):231–236. doi: 10.2478/jccm-2020-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Manganotti P., Bellavita G., D’Acunto L., et al. Clinical neurophysiology and cerebrospinal liquor analysis to detect Guillain-Barré syndrome and polyneuritis cranialis in COVID-19 patients: a case series. J. Med. Virol. 2021;93(2):766–774. doi: 10.1002/jmv.26289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Masuccio F.G., Barra M., Claudio G., Claudio S. A rare case of acute motor axonal neuropathy and myelitis related to SARS-CoV-2 infection. J. Neurol. 2020:1–4. doi: 10.1007/s00415-020-10219-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Naddaf E., Laughlin R.S., Klein C.J., et al. Guillain-Barre syndrome in a patient with evidence of recent SARS-CoV-2 infection. Mayo Clin. Proc. 2020;95(8):1799–1801. doi: 10.1016/j.mayocp.2020.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Nanda S., Handa R., Prasad A., et al. Covid-19 associated Guillain-Barre Syndrome: contrasting tale of four patients from a tertiary care centre in India. Am. J. Emerg. Med. 2021;39:125–128. doi: 10.1016/j.ajem.2020.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Paybast S., Gorji R., Mavandadi S. Guillain-Barré syndrome as a neurological complication of novel COVID-19 Infection: a case report and review of the literature. Neurologist. 2020;25(4):101–103. doi: 10.1097/NRL.0000000000000291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Pelea T., Reuter U., Schmidt C., Laubinger R., Siegmund R., Walther B.W. SARS-CoV-2 associated Guillain-Barré syndrome. J. Neurol. 2020:1–4. doi: 10.1007/s00415-020-10133-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Rajdev K., Victor N., Buckholtz E.S., et al. A case of Guillain-Barré syndrome associated with COVID-19. J. Investig. Med. High Impact Case Rep. 2020;8 doi: 10.1177/2324709620961198. 2324709620961198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ray A. Miller Fisher syndrome and COVID-19: is there a link? BMJ Case Rep. 2020;13(8):e236429. doi: 10.1136/bcr-2020-236419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Senel M., Abu-Rumeileh S., Michel D., et al. Miller-Fisher syndrome after COVID-19: neurochemical markers as an early sign of nervous system involvement. Eur. J. Neurol. 2020;27(11):2378–2380. doi: 10.1111/ene.14473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Anand P., Al-Faraj A., Sader E., et al. Seizure as the presenting symptom of COVID-19: a retrospective case series. Epilepsy Behav. 2020;112 doi: 10.1016/j.yebeh.2020.107335. 107335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Anand P., Lau K.H.V., Chung D.Y., et al. Posterior reversible encephalopathy syndrome in patients with coronavirus disease 2019: two cases and a review of the literature. J. Stroke Cerebrovasc. Dis. 2020;29(11):105212. doi: 10.1016/j.jstrokecerebrovasdis.2020.105212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Avula A., Gill A., Nassar R., Nalleballe K., Siddamreddy S., Chalhoub M. Locked-in with COVID-19. J. Clin. Neurosci. 2020;79:80–83. doi: 10.1016/j.jocn.2020.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ayatollahi P., Tarazi A., Wennberg R. Possible autoimmune encephalitis with claustrum sign in case of acute SARS-CoV-2 infection. Can. J. Neurol. Sci. 2020:1–3. doi: 10.1017/cjn.2020.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Babar A., Lewandowski U., Capin I., et al. SARS-CoV-2 encephalitis in a 20-year old healthy female. Pediatr. Infect. Dis. J. 2020;39(10):e320–e321. doi: 10.1097/INF.0000000000002855. [DOI] [PubMed] [Google Scholar]
- 125.Balloy G., Leclair-Visonneau L., Péréon Y., et al. Non-lesional status epilepticus in a patient with coronavirus disease 2019. Clin. Neurophysiol. 2020;131(8):2059–2061. doi: 10.1016/j.clinph.2020.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bektaş G., Akçay N., Boydağ K., Şevketoğlu E. Reversible splenial lesion syndrome associated with SARS-CoV-2 infection in two children. Brain and Development. 2021;43(2):230–233. doi: 10.1016/j.braindev.2020.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Bellon M., Schweblin C., Lambeng N., et al. Cerebrospinal fluid features in SARS-CoV-2 RT-PCR positive patients. Clin. Infect. Dis. 2020:ciaa1165. doi: 10.1093/cid/ciaa1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Bodro M., Compta Y., Llansó L., et al. Increased CSF levels of IL-1β, IL-6, and ACE in SARS-CoV-2-associated encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5):e821. doi: 10.1212/NXI.0000000000000821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Borroni B., Gazzina S., Dono F., et al. Diaphragmatic myoclonus due to SARS-CoV-2 infection. Neurol. Sci. 2020;41(12):3471–3474. doi: 10.1007/s10072-020-04766-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Abdel-Mannan O., Eyre M., Löbel U., et al. Neurologic and radiographic findings associated with COVID-19 infection in children. JAMA Neurol. 2020;77(11):1–6. doi: 10.1001/jamaneurol.2020.2687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Brun G., Hak J.-F., Coze S., et al. COVID-19—White matter and globus pallidum lesions. Demyelination or small-vessel vasculitis? Neurol. Neuroimmunol. Neuroinflamm. 2020;7(4) doi: 10.1212/NXI.0000000000000777. e777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Byrnes S., Bisen M., Syed B., et al. COVID-19 encephalopathy masquerading as substance withdrawal. J. Med. Virol. 2020;92(11):2376–2378. doi: 10.1002/jmv.26065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Caan M.P., Lim C.T., Howard M. A case of catatonia in a man with COVID-19. Psychosomatics. 2020;61(5):556–560. doi: 10.1016/j.psym.2020.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Canham L.J.W., Staniaszek L.E., Mortimer A.M., Nouri L.F., Kane N.M. Electroencephalographic (EEG) features of encephalopathy in the setting of Covid-19: a case series. Clin. Neurophysiol. Pract. 2020;5:199–205. doi: 10.1016/j.cnp.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Cani I., Barone V., D’Angelo R., et al. Frontal encephalopathy related to hyperinflammation in COVID-19. J. Neurol. 2020:1–4. doi: 10.1007/s00415-020-10057-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Cao A., Rohaut B., Le Guennec L., et al. Severe COVID-19-related encephalitis can respond to immunotherapy. Brain J. Neurol. 2020:awaa337. doi: 10.1093/brain/awaa337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Casez O., Willaume G., Grand S., et al. SARS-CoV-2 related encephalitis: MRI pattern of the olfactory tract involvement. Neurology. 2020 doi: 10.1212/WNL.0000000000011150. [DOI] [PubMed] [Google Scholar]
- 138.Cebrián J., Gonzalez-Martinez A., García-Blanco M.J., et al. Headache and impaired consciousness level associated with SARS-CoV-2 in CSF: A case report. Neurology. 2020;95(6):266–268. doi: 10.1212/WNL.0000000000010213. [DOI] [PubMed] [Google Scholar]
- 139.Chauffier J., Poey N., Husain M., et al. First case of mild encephalopathy with reversible splenial lesion in SARS-CoV-2 infection. Med. Mal. Infect. 2020:S0399–77-2. doi: 10.1016/j.medmal.2020.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Chaumont H., Etienne P., Roze E., Couratier C., Roger P.M., Lannuzel A. Acute meningoencephalitis in a patient with COVID-19. Rev. Neurol. 2020;176(6):519–521. doi: 10.1016/j.neurol.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Cohen M.E., Eichel R., Steiner-Birmanns B., et al. A case of probable Parkinson’s disease after SARS-CoV-2 infection. Lancet Neurol. 2020;19(10):804–805. doi: 10.1016/S1474-4422(20)30305-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Conklin J., Frosch M.P., Mukerji S., et al. Cerebral microvascular injury in severe COVID-19. medRxiv. 2020 doi: 10.1101/2020.07.21.20159376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Conte G., Avignone S., Carbonara M., et al. COVID-19-associated PRES-like encephalopathy with perivascular gadolinium enhancement. Am. J. Neuroradiol. 2020;41(12):2206–2208. doi: 10.3174/ajnr.A6762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.de Miranda Henriques-Souza A.M., de Melo A.C.M.G., de Aguiar Coelho Silva Madeiro B., Freitas L.F., Sampaio Rocha-Filho P.A., Goncalves F.G. Acute disseminated encephalomyelitis in a COVID-19 pediatric patient. Neuroradiology. 2020:1–5. doi: 10.1007/s00234-020-02571-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.de Oliveira F.A.A., Palmeira D.C.C., Rocha-Filho P.A.S. Headache and pleocytosis in CSF associated with COVID-19: case report. Neurol. Sci. 2020;41(11):3021–3022. doi: 10.1007/s10072-020-04694-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.de Oliveira R.D.M.C., Santos D.H., Olivetti B.C., Takahashi J.T. Bilateral trochlear nerve palsy due to cerebral vasculitis related to covid-19 infection. Arq. Neuropsiquiatr. 2020;78(7):385–386. doi: 10.1590/0004-282X20200052. [DOI] [PubMed] [Google Scholar]
- 147.De Stefano P., Nencha U., De Stefano L., Mégevand P., Seeck M. Focal EEG changes indicating critical illness associated cerebral microbleeds in a Covid-19 patient. Clin. Neurophysiol. Pract. 2020;5:125–129. doi: 10.1016/j.cnp.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Delamarre L., Gollion C., Grouteau G., et al. COVID-19-associated acute necrotising encephalopathy successfully treated with steroids and polyvalent immunoglobulin with unusual IgG targeting the cerebral fibre network. J. Neurol. Neurosurg. Psychiatry. 2020;91(9):1004–1006. doi: 10.1136/jnnp-2020-323678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Delorme C., Paccoud O., Kas A., et al. Covid-19-related encephalopathy: a case series with brain FDG-PET/CT findings. Eur. J. Neurol. 2020;27(12):2651–2657. doi: 10.1111/ene.14478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Demirci Otluoglu G., Yener U., Demir M.K., Yilmaz B. Encephalomyelitis associated with Covid-19 infection: case report. Br. J. Neurosurg. 2020:1–3. doi: 10.1080/02688697.2020.1787342. [DOI] [PubMed] [Google Scholar]
- 151.Dharsandiya M., Shah K., Patel K., Patel T., Patel A., Patel A. SARS-CoV-2 viral sepsis with meningoencephalitis. Indian J. Med. Microbiol. 2020;38(2):219–221. doi: 10.4103/ijmm.IJMM_20_291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Djellaoui A., Seddik L., Cleret De Langavant L., Cattan S., Bachoud-Lévi A.C., Hosseini H. Posterior reversible encephalopathy syndrome associated with SARS-CoV-2 infection. J. Neurol. Neurosurg. Psychiatry. 2020 doi: 10.1136/jnnp-2020-323923. jnnp-2020-323923. [DOI] [PubMed] [Google Scholar]
- 153.Domingues R.B., Mendes-Correa M.C., de Moura Leite F.B.V., et al. First case of SARS-COV-2 sequencing in cerebrospinal fluid of a patient with suspected demyelinating disease. J. Neurol. 2020;267(11):3154–3156. doi: 10.1007/s00415-020-09996-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Dono F., Carrarini C., Russo M., et al. New-onset refractory status epilepticus (NORSE) in post SARS-CoV-2 autoimmune encephalitis: a case report. Neurol. Sci. 2020:1–4. doi: 10.1007/s10072-020-04846-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Doo F.X., Kassim G., Lefton D.R., Patterson S., Pham H., Belani P. Rare presentations of COVID-19: PRES-like leukoencephalopathy and carotid thrombosis. Clin. Imaging. 2021;69:94–101. doi: 10.1016/j.clinimag.2020.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Dugue R., Cay-Martínez K.C., Thakur K.T., et al. Neurologic manifestations in an infant with COVID-19. Neurology. 2020;94(24):1100–1102. doi: 10.1212/WNL.0000000000009653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Edén A., Kanberg N., Gostner J., et al. CSF biomarkers in patients with COVID-19 and neurological symptoms: a case series. Neurology. 2020 doi: 10.1212/WNL.0000000000010977. [DOI] [PubMed] [Google Scholar]
- 158.El Aoud S., Sorial D., Selmaoui A., et al. A first case of Mild Encephalitis with Reversible Splenial Lesion(MERS) as a presenting feature of SARS-CoV-2. Rev. Neurol. 2020 doi: 10.1016/j.neurol.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Elgamasy S., Kamel M.G., Ghozy S., Khalil A., Morra M.E., Islam S.M.S. First case of focal epilepsy associated with SARS-coronavirus-2. J. Med. Virol. 2020;92(10):2238–2242. doi: 10.1002/jmv.26113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Elkady A., Rabinstein A.A. Acute necrotizing encephalopathy and myocarditis in a young patient with COVID-19. Neurol. Neuroimmunol. Neuroinflamm. 2020;7(5) e801. [Google Scholar]
- 161.El-Zein R.S., Cardinali S., Murphy C., Keeling T. COVID-19-associated meningoencephalitis treated with intravenous immunoglobulin. BMJ Case Rep. 2020;13(9):e237364. doi: 10.1136/bcr-2020-237364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Emami A., Fadakar N., Akbari A., et al. Seizure in patients with COVID-19. Neurol. Sci. 2020;41(11):3057–3061. doi: 10.1007/s10072-020-04731-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Faber I., Brandão P.R.P., Menegatti F., de Carvalho Bispo D.D., Maluf F.B., Cardoso F. Coronavirus disease 2019 and Parkinsonism: a non-post-encephalitic case. Mov. Disord. 2020;35(10):1721–1722. doi: 10.1002/mds.28277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Fadakar N., Ghaemmaghami S., Masoompour S.M., et al. A first case of acute cerebellitis associated with coronavirus disease (COVID-19): a case report and literature review. Cerebellum. 2020;19(6):911–914. doi: 10.1007/s12311-020-01177-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Farhadian S., Glick L.R., Vogels C.B.F., et al. Acute encephalopathy with elevated CSF inflammatory markers as the initial presentation of COVID-19. BMC Neurol. 2020;20(1):248. doi: 10.1186/s12883-020-01812-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Freire-Álvarez E., Guillén L., Lambert K., et al. COVID-19-associated encephalitis successfully treated with combination therapy. Clin. Infect. Pract. 2020:100053. doi: 10.1016/j.clinpr.2020.100053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.García-Howard M., Herranz-Aguirre M., Moreno-Galarraga L., et al. Case report: benign infantile seizures temporally associated with COVID-19. Front. Pediatr. 2020;8:507. doi: 10.3389/fped.2020.00507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Ghosh R., Dubey S., Finsterer J., Chatterjee S., Ray B.K. SARS-CoV-2-associated acute hemorrhagic, Necrotizing Encephalitis (AHNE) presenting with cognitive impairment in a 44-year-old woman without comorbidities: a case report. Am. J. Case Rep. 2020;21 doi: 10.12659/AJCR.925641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Grimaldi S., Lagarde S., Harle J.R., Boucraut J., Guedj E. Autoimmune encephalitis concomitant with SARS-CoV-2 infection: insight from (18)F-FDG PET imaging and neuronal autoantibodies. J. Nucl. Med. 2020;61(12):1726–1729. doi: 10.2967/jnumed.120.249292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Guillan M., Villacieros-Alvarez J., Bellido S., et al. Unusual simultaneous cerebral infarcts in multiple arterial territories in a COVID-19 patient. Thromb. Res. 2020;193:107–109. doi: 10.1016/j.thromres.2020.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Guilmot A., Maldonado Slootjes S., Sellimi A., et al. Immune-mediated neurological syndromes in SARS-CoV-2-infected patients. J. Neurol. 2020:1–7. doi: 10.1007/s00415-020-10108-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Haddad S., Tayyar R., Risch L., et al. Encephalopathy and seizure activity in a COVID-19 well controlled HIV patient. IDCases. 2020;21 doi: 10.1016/j.idcr.2020.e00814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Hafizi F., Kherani S., Shams M. Meningoencephalitis from SARS-CoV-2 infection. IDCases. 2020;21 doi: 10.1016/j.idcr.2020.e00919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Haider A., Siddiqa A., Ali N., Dhallu M. COVID-19 and the brain: acute encephalitis as a clinical manifestation. Cureus. 2020;12(10) doi: 10.7759/cureus.10784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Hayat U., Ahmed S., Hussain M.A., Hameed N. Encephalopathy as the presenting symptom of COVID-19. Kansas J. Med. 2020;13:272–274. doi: 10.17161/kjm.vol13.14766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Hoelscher C., Sweid A., Ghosh R., et al. Cerebral deep venous thrombosis and COVID-19: case report. J. Neurosurg. 2020:1–4. doi: 10.3171/2020.5.JNS201542. [DOI] [PubMed] [Google Scholar]
- 177.Hosseini A.A., Shetty A.K., Sprigg N., Auer D.P., Constantinescu C.S. Delirium as a presenting feature in COVID-19: neuroinvasive infection or autoimmune encephalopathy? Brain Behav. Immun. 2020;88:68–70. doi: 10.1016/j.bbi.2020.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Jang K., Khatri A., Majure D.T. COVID-19 leading to acute encephalopathy in a patient with heart transplant. J. Heart Lung Transplant. 2020;39(8):853–855. doi: 10.1016/j.healun.2020.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Kakadia B., Ahmed J., Siegal T., Jovin T.G., Thon J.M. Mild encephalopathy with reversible splenium lesion (MERS) in a patient with COVID-19. J. Clin. Neurosci. 2020;79:272–274. doi: 10.1016/j.jocn.2020.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Kamal Y.M., Abdelmajid Y., Al Madani A.A.R. Cerebrospinal fluid confirmed COVID-19-associated encephalitis treated successfully. BMJ Case Rep. 2020;13(9):e237378. doi: 10.1136/bcr-2020-237378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Karapanayiotides T., Geka E., Prassopoulos P., et al. Concentric demyelination pattern in COVID-19-associated acute haemorrhagic leukoencephalitis: a lurking catastrophe? Brain. 2020:awaa375. doi: 10.1093/brain/awaa375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Keller E., Brandi G., Winklhofer S., et al. Large and small cerebral vessel involvement in severe COVID-19: detailed clinical workup of a case series. Stroke. 2020 doi: 10.1161/STROKEAHA.120.031224. Strokeaha120031224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kest H., Kaushik A., DeBruin W., Colletti M., Goldberg D. Multisystem inflammatory syndrome in children (MIS-C) associated with 2019 novel coronavirus (SARS-CoV-2) infection. Case Rep.Pediatr. 2020;2020 doi: 10.1155/2020/8875987. 8875987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Khodamoradi Z., Hosseini S.A., Gholampoor Saadi M.H., Mehrabi Z., Sasani M.R., Yaghoubi S. COVID-19 meningitis without pulmonary involvement with positive cerebrospinal fluid PCR. Eur. J. Neurol. 2020;27(12):2668–2669. doi: 10.1111/ene.14536. [DOI] [PubMed] [Google Scholar]
- 185.Khoo A., McLoughlin B., Cheema S., et al. Postinfectious brainstem encephalitis associated with SARS-CoV-2. J. Neurol. Neurosurg. Psychiatry. 2020;91(9):1013–1014. doi: 10.1136/jnnp-2020-323816. [DOI] [PubMed] [Google Scholar]
- 186.Kihira S., Delman B.N., Belani P., et al. Imaging features of acute encephalopathy in patients with COVID-19: a case series. Am. J. Neuroradiol. 2020;41(10):1804–1808. doi: 10.3174/ajnr.A6715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Kremer S., Lersy F., Anheim M., et al. Neurologic and neuroimaging findings in patients with COVID-19: a retrospective multicenter study. Neurology. 2020;95(13):e1868–e1882. doi: 10.1212/WNL.0000000000010112. [DOI] [PubMed] [Google Scholar]
- 188.Kumar A., Olivera A., Mueller N., Howard J., Lewis A. Delayed SARS-COV-2 leukoencephalopathy without severe Hypoxia. J. Neurol. Sci. 2020;418:117146. doi: 10.1016/j.jns.2020.117146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Kumar N., Kumar S., Pati B.K., Kumar A., Singh C., Sarfraz A. Acute necrotizing encephalitis as a probable association of covid-19. Indian J. Crit. Care Med. 2020;24(10):991–994. doi: 10.5005/jp-journals-10071-23636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Lang M., Buch K., Li M.D., et al. Leukoencephalopathy associated with severe COVID-19 infection: sequela of hypoxemia? Am. J. Neuroradiol. 2020;41(9):1641–1645. doi: 10.3174/ajnr.A6671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Le Guennec L., Devianne J., Jalin L., et al. Orbitofrontal involvement in a neuroCOVID-19 patient. Epilepsia. 2020;61(8):e90–e94. doi: 10.1111/epi.16612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Lim S.T., Janaway B., Costello H., Trip A., Price G. Persistent psychotic symptoms following COVID-19 infection. BJPsych Open. 2020;6(5) doi: 10.1192/bjo.2020.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Lin J., Lawson E.C., Verma S., Peterson R.B., Sidhu R. Cytotoxic lesion of the corpus callosum in an adolescent with multisystem inflammatory syndrome and SARS-CoV-2 infection. Am. J. Neuroradiol. 2020;41(11):2017–2019. doi: 10.3174/ajnr.A6755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Llorente Ayuso L., Torres Rubio P., Beijinho do Rosário R.F., Giganto Arroyo M.L., Sierra-Hidalgo F. Bickerstaff encephalitis after COVID-19. J. Neurol. 2020:1–3. doi: 10.1007/s00415-020-10201-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Lo Monaco M.R., Di Giambenedetto S., Martone A.M., De Gaetano Donati K., Landi F. Encephalopathy as neurological involvement of SARS-COV-2 infection. Clin. Infect. Pract. 2020;7 doi: 10.1016/j.clinpr.2020.100034. 100034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Lorenz N., Treptow A., Schmidt S., et al. Neonatal early-onset infection with SARS-CoV-2 in a newborn presenting with encephalitic symptoms. Pediatr. Infect. Dis. J. 2020;39(8) doi: 10.1097/INF.0000000000002735. [DOI] [PubMed] [Google Scholar]
- 197.Lu S., Wei N., Jiang J., et al. First report of manic-like symptoms in a COVID-19 patient with no previous history of a psychiatric disorder. J. Affect. Disord. 2020;277:337–340. doi: 10.1016/j.jad.2020.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Lyons S., O’Kelly B., Woods S., et al. Seizure with CSF lymphocytosis as a presenting feature of COVID-19 in an otherwise healthy young man. Seizure. 2020;80:113–114. doi: 10.1016/j.seizure.2020.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Mardani M., Nadji S.A., Sarhangipor K.A., Sharifi-Razavi A., Baziboroun M. COVID-19 infection recurrence presenting with meningoencephalitis. New Microbes New Infect. 2020;37:100732. doi: 10.1016/j.nmni.2020.100732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Matos A.R., Quintas-Neves M., Oliveira A.I., et al. COVID-19 associated central nervous system vasculopathy. Can. J. Neurol. Sci. 2020:1–2. doi: 10.1017/cjn.2020.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Mawhinney J.A., Wilcock C., Haboubi H., Roshanzamir S. Neurotropism of SARS-CoV-2: COVID-19 presenting with an acute manic episode. BMJ Case Rep. 2020;13(6):e236123. doi: 10.1136/bcr-2020-236123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.McAbee G.N., Brosgol Y., Pavlakis S., Agha R., Gaffoor M. Encephalitis associated with COVID-19 infection in an 11-year-old child. Pediatr. Neurol. 2020;109:94. doi: 10.1016/j.pediatrneurol.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.McCuddy M., Kelkar P., Zhao Y., Wicklund D. Acute demyelinating encephalomyelitis (ADEM) in COVID-19 infection: a case series. Neurol. India. 2020;68(5):1192–1195. doi: 10.4103/0028-3886.299174. [DOI] [PubMed] [Google Scholar]
- 204.Mirzaee S.M.M., Gonçalves F.G., Mohammadifard M., Tavakoli S.M., Vossough A. Focal cerebral arteriopathy in a pediatric patient with COVID-19. Radiolog. 2020;297(2):e274–e275. doi: 10.1148/radiol.2020202197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Monti G., Giovannini G., Marudi A., et al. Anti-NMDA receptor encephalitis presenting as new onset refractory status epilepticus in COVID-19. Seizure. 2020;81:18–20. doi: 10.1016/j.seizure.2020.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Muccioli L., Pensato U., Bernabè G., et al. Intravenous immunoglobulin therapy in COVID-19-related encephalopathy. J. Neurol. 2020:1–5. doi: 10.1007/s00415-020-10248-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Muccioli L., Pensato U., Cani I., et al. COVID-19-related encephalopathy presenting with aphasia resolving following tocilizumab treatment. J. Neuroimmunol. 2020;349 doi: 10.1016/j.jneuroim.2020.577400. 577400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Naz S., Hanif M., Haider M.A., Ali M.J., Ahmed M.U., Saleem S. Meningitis as an Initial presentation of COVID-19: a case report. Front. Public Health. 2020;8:474. doi: 10.3389/fpubh.2020.00474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Noone R., Cabassa J.A., Gardner L., Schwartz B., Alpert J.E., Gabbay V. Letter to the Editor: New onset psychosis and mania following COVID-19 infection. J. Psychiatr. Res. 2020;130:177–179. doi: 10.1016/j.jpsychires.2020.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Sripadma P., Rai A., Wadhwa C. Postpartum atypical posterior reversible encephalopathy syndrome in a COVID-19 patient – an obstetric emergency. J. Stroke Cerebrovasc. Dis. 2020;29(12):105357. doi: 10.1016/j.jstrokecerebrovasdis.2020.105357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Packwood R., Galletta G., Tennyson J. An Unusual case report of COVID-19 presenting with meningitis symptoms and shingles. Clin. Pract. Cases Emerg. Med. 2020;4(3):316–320. doi: 10.5811/cpcem.2020.4.47557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Palao M., Fernandez-Diaz E., Gracia-Gil J., Romero-Sanchez C.M., Diaz-Maroto I., Segura T. Multiple sclerosis following SARS-CoV-2 infection. Mult. Scler. Relat. Disord. 2020;45 doi: 10.1016/j.msard.2020.102377. 102377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Paterson R.W., Brown R.L., Benjamin L., et al. The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain J. Neurol. 2020;143(10):3104–3120. doi: 10.1093/brain/awaa240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Pensato U., Muccioli L., Pasini E., et al. Encephalopathy in COVID-19 presenting with acute aphasia mimicking stroke. Front. Neurol. 2020;11:587226. doi: 10.3389/fneur.2020.587226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Perrin P., Collongues N., Baloglu S., et al. Cytokine release syndrome-associated encephalopathy in patients with COVID-19. Eur. J. Neurol. 2021;28(1):248–258. doi: 10.1111/ene.14491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Pilotto A., Masciocchi S., Volonghi I., et al. Clinical presentation and outcomes of SARS-CoV-2 related encephalitis: the ENCOVID multicentre study. J. Infect. Dis. 2021;223(1):28–37. doi: 10.1093/infdis/jiaa609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Pilotto A., Padovani A. Reply to the Letter “COVID-19-associated encephalopathy and cytokine-mediated neuroinflammation”. Ann. Neurol. 2020;88(4):861–862. doi: 10.1002/ana.25856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Pinto A.A., Carroll L.S., Nar V., Varatharaj A., Galea I. CNS inflammatory vasculopathy with antimyelin oligodendrocyte glycoprotein antibodies in COVID-19. Neurology. 2020;7(5):e813. doi: 10.1212/NXI.0000000000000813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Ramakrishna J.M., Libertin C.R., Siegel J., et al. Three-tier stratification for CNS COVID-19 to help decide which patients should undergo lumbar puncture with CSF analysis: a case report and literature review. Rom. J. Intern. Med. 2020 doi: 10.2478/rjim-2020-0031. [DOI] [PubMed] [Google Scholar]
- 220.Carroll E., Neumann H., Aguero-Rosenfeld M.E., et al. Post-COVID-19 inflammatory syndrome manifesting as refractory status epilepticus. Epilepsia. 2020;61(10):e135–e139. doi: 10.1111/epi.16683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Chan M., Han S.C., Kelly S., Tamimi M., Giglio B., Lewis A. A case series of Guillain-Barré Syndrome following Covid-19 infection in New York. Neurol. Clin. Pract. 2020 doi: 10.1212/CPJ.0000000000000880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Rebeiz T., Lim-Hing K., Khazanehdari S., Rebeiz K. Behavioral changes without respiratory symptoms as a presenting sign of COVID-19 encephalitis. Cureus. 2020;12(9) doi: 10.7759/cureus.10469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Regev T., Antebi M., Eytan D., et al. Pediatric inflammatory multisystem syndrome with central nervous system involvement and hypocomplementemia following SARS-COV-2 infection. Pediatr. Infect. Dis. J. 2020;39(8):e206–e207. doi: 10.1097/INF.0000000000002804. [DOI] [PubMed] [Google Scholar]
- 224.Rifino N., Censori B., Agazzi E., et al. Neurologic manifestations in 1760 COVID-19 patients admitted to Papa Giovanni XXIII Hospital, Bergamo, Italy. J. Neurol. 2020:1–8. doi: 10.1007/s00415-020-10251-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Saitta L., Molin A., Villani F., et al. Brain microvascular occlusive disorder in COVID-19: a case report. Neurol. Sci. 2020;41(12):3401–3404. doi: 10.1007/s10072-020-04795-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Santos De Lima F., Issa N., Seibert K., et al. Epileptiform activity and seizures in patients with COVID-19. J. Neurol. Neurosurg. Psychiatry. 2020 doi: 10.1136/jnnp-2020-324337. [DOI] [PubMed] [Google Scholar]
- 227.Sattar S.B.A., Haider M.A., Zia Z., Niazi M., Iqbal Q.Z. Clinical, radiological, and molecular findings of acute encephalitis in a COVID-19 patient: a rare case report. Cureus. 2020;12(9) doi: 10.7759/cureus.10650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Scullen T., Keen J., Mathkour M., Dumont A.S., Kahn L. Coronavirus 2019 (COVID-19)-associated encephalopathies and cerebrovascular disease: the new orleans experience. World Neurosurg. 2020;141:e437–e446. doi: 10.1016/j.wneu.2020.05.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Shahbaznejad L., Navaeifar M.R., Abbaskhanian A., Hosseinzadeh F., Rahimzadeh G., Rezai M.S. Clinical characteristics of 10 children with a pediatric inflammatory multisystem syndrome associated with COVID-19 in Iran. BMC Pediatr. 2020;20(1):513. doi: 10.1186/s12887-020-02415-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Silva M.T.T., Lima M.A., Torezani G., et al. Isolated intracranial hypertension associated with COVID-19. Cephalalgia. 2020;40(13):1452–1458. doi: 10.1177/0333102420965963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Song E., Chow R.D., Jiang R., et al. Immunologically distinct responses occur in the CNS of COVID-19 patients. bioRxiv. 2020 doi: 10.1101/2020.09.11.293464. [DOI] [Google Scholar]
- 232.Soto Insuga V., Cantarin-Extremera V., Solis-Munoz I., et al. Pseudotumor Cerebri Caused by SARS-CoV-2 Infection in a Boy. J. Pediatr. Neurol. 2020 doi: 10.1055/s-0040-1715857. [DOI] [Google Scholar]
- 233.Umapathi T., Quek W.M.J., Yen J.M., et al. Encephalopathy in COVID-19 patients; viral, parainfectious, or both? eNeurologicalSci. 2020;21:100275. doi: 10.1016/j.ensci.2020.100275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Vandervorst F., Guldolf K., Peeters I., et al. Encephalitis associated with the SARS-CoV-2 virus: A case report. Interdisciplinary Neurosurgery: Advanced techniques and case management. 2020;22:100821. doi: 10.1016/j.inat.2020.100821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Vaschetto R., Cena T., Sainaghi P.P., et al. Cerebral nervous system vasculitis in a Covid-19 patient with pneumonia. J. Clin. Neurosci. 2020;79:71–73. doi: 10.1016/j.jocn.2020.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Virhammar J., Kumlien E., Fällmar D., et al. Acute necrotizing encephalopathy with SARS-CoV-2 RNA confirmed in cerebrospinal fluid. Neurology. 2020;95(10):445–449. doi: 10.1212/WNL.0000000000010250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Wang M., Li T., Qiao F., Wang L., Li C., Gong Y. Coronavirus disease 2019 associated with aggressive neurological and mental abnormalities confirmed based on cerebrospinal fluid antibodies: A case report. Medicine. 2020;99(36) doi: 10.1097/MD.0000000000021428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Westhoff T.H., Seibert F.S., Bauer F., et al. Allograft infiltration and meningoencephalitis by SARS-CoV-2 in a pancreas-kidney transplant recipient. Am. J. Transplant. 2020;20(11):3216–3220. doi: 10.1111/ajt.16223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Wong P.F., Craik S., Newman P., et al. Lessons of the month 1: A case of rhombencephalitis as a rare complication of acute COVID-19 infection. Clin. Med. 2020;20(3):293–294. doi: 10.7861/clinmed.2020-0182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Yin R., Feng W., Wang T., et al. Concomitant neurological symptoms observed in a patient diagnosed with coronavirus disease 2019. J. Med. Virol. 2020;92(10):1782–1784. doi: 10.1002/jmv.25888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Yousefi K., Poorbarat S., Abasi Z., Rahimi S., Khakshour A. Viral meningitis associated with COVID-19 in a 9-year-old child: a case report. Pediatr. Infect. Dis. J. 2020 doi: 10.1097/INF.0000000000002979. [DOI] [PubMed] [Google Scholar]
- 242.Zambreanu L., Lightbody S., Bhandari M., et al. A case of limbic encephalitis associated with asymptomatic COVID-19 infection. J. Neurol. Neurosurg. Psychiatry. 2020;91(11):1229–1230. doi: 10.1136/jnnp-2020-323839. [DOI] [PubMed] [Google Scholar]
- 243.Zayet S., Ben Abdallah Y., Royer P.Y., Toko-Tchiundzie L., Gendrin V., Klopfenstein T. Encephalopathy in patients with COVID-19: ‘Causality or coincidence?’. J. Med. Virol. 2021;93(2):1193. doi: 10.1002/jmv.26027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Zhang T., Hirsh E., Zandieh S., Rodricks M.B. COVID-19-associated acute multi-infarct encephalopathy in an asymptomatic CADASIL patient. Neurocrit. Care. 2020:1–4. doi: 10.1007/s12028-020-01119-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Zoghi A., Ramezani M., Roozbeh M., Darazam I.A., Sahraian M.A. A case of possible atypical demyelinating event of the central nervous system following COVID-19. Mult. Scler. Relat. Disord. 2020;44:102324. doi: 10.1016/j.msard.2020.102324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Zuhorn F., Omaimen H., Ruprecht B., et al. Parainfectious encephalitis in COVID-19: “The Claustrum Sign”. J. Neurol. 2020:1–4. doi: 10.1007/s00415-020-10185-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Brown B., Fidell A., Ingolia G., Murad E., Beckham J.D. Defining diagnostic approaches and outcomes in patients with inflammatory CSF: a retrospective cohort study. Clin. Neurol. Neurosurg. 2018;172:105–111. doi: 10.1016/j.clineuro.2018.06.039. [DOI] [PubMed] [Google Scholar]
- 248.Blakeley J., Irani D.N. In: Cerebrospinal Fluid in Clinical Practice. Irani D.N., editor. W.B. Saunders; Philadelphia: 2009. CHAPTER 32 – Approach to the patient with abnormal cerebrospinal fluid protein content; pp. 287–294. [Google Scholar]
- 249.Personal Communication with Dr. Shole Yaqubi. 12/9/2020. [Google Scholar]
- 250.Demir G., Balaban O., Tekeci M.H., Issı Z., Erdem A.F. Fahr’s syndrome presenting with seizures in SARS-CoV-2 (COVID-19) pneumonia-a case report. Neurol. Sci. 2020;41(11):3063–3065. doi: 10.1007/s10072-020-04733-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Sajjad M.U., Blennow K., Knapskog A.B., et al. Cerebrospinal fluid levels of interleukin-8 in delirium, dementia, and cognitively healthy patients. J. Alzheimers Dis. 2020;73(4):1363–1372. doi: 10.3233/JAD-190941. [DOI] [PubMed] [Google Scholar]
- 252.Perchetti G.A., Nalla A.K., Huang M.L., et al. Validation of SARS-CoV-2 detection across multiple specimen types. J. Clin. Virol. 2020;128:104438. doi: 10.1016/j.jcv.2020.104438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Penarrubia A.L., Ruiz M., Porco R., et al. Multiple assays in a real-time RT-PCR SARS-CoV-2 panel can mitigate the risk of loss of sensitivity by new genomic variants during the COVID-19 outbreak. Int. J. Infect. Dis. 2020;97:225–229. doi: 10.1016/j.ijid.2020.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Panciani P.P., Saraceno G., Zanin L., et al. SARS-CoV-2: “Three-steps” infection model and CSF diagnostic implication. Brain Behav. Immun. 2020;87:128–129. doi: 10.1016/j.bbi.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Needham E.J., Chou S.H., Coles A.J., Menon D.K. Neurological implications of COVID-19 infections. Neurocrit. Care. 2020;32(3):667–671. doi: 10.1007/s12028-020-00978-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Joob B., Wiwantikit V. COVID-19 PCR test, cluster of false positive and importance of quality control. Clin. Lab. 2020;66(10) doi: 10.7754/Clin.Lab.2020.200629. [DOI] [PubMed] [Google Scholar]
- 257.Tom M.R., Mina M.J. To interpret the SARS-CoV-2 test, consider the cycle threshold value. Clin. Infect. Dis. 2020;71(16):2252–2254. doi: 10.1093/cid/ciaa619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Jaijakul S., Salazar L., Wootton S.H., Aguilera E., Hasbun R. The clinical significance of neutrophilic pleocytosis in cerebrospinal fluid in patients with viral central nervous system infections. Int. J. Infect. Dis. 2017;59:77–81. doi: 10.1016/j.ijid.2017.04.010. [DOI] [PubMed] [Google Scholar]
- 259.Troendle M., Pettigrew A. A systematic review of cases of meningitis in the absence of cerebrospinal fluid pleocytosis on lumbar puncture. BMC Infect. Dis. 2019;19(1) doi: 10.1186/s12879-019-4204-z. 692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Tyler K.L., Pape J., Goody R.J., Corkill M., Kleinschmidt-DeMasters B.K. CSF findings in 250 patients with serologically confirmed West Nile virus meningitis and encephalitis. Neurology. 2006;66(3):361–365. doi: 10.1212/01.wnl.0000195890.70898.1f. [DOI] [PubMed] [Google Scholar]
- 261.Barnes S., Senanayake S., Budge M. Acellular cerebral spinal fluid in the early stages of herpes simplex encephalitis. Intern. Med. J. 2009;39(11):783–784. doi: 10.1111/j.1445-5994.2009.02049.x. [DOI] [PubMed] [Google Scholar]
- 262.Tahamtan A., Ardebili A. Real-time RT-PCR in COVID-19 detection: issues affecting the results. Expert. Rev. Mol. Diagn. 2020;20(5):453–454. doi: 10.1080/14737159.2020.1757437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Plebani M., Padoan A., Negrini D., Carpinteri B., Sciacovelli L. Diagnostic performances and thresholds: The key to harmonization in serological SARS-CoV-2 assays? Clin. Chim. Acta. 2020;509:1–7. doi: 10.1016/j.cca.2020.05.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Das S.J., Cabral-Castro M.J., Guimarães M.A., Peralta J.M., Puccioni-Sohler M. Cerebrospinal fluid challenges for the diagnosis of herpes simplex infection in the central nervous system. Arq. Neuropsiquiatr. 2020;78(3):163–168. doi: 10.1590/0004-282X20190179. [DOI] [PubMed] [Google Scholar]
- 265.Obermeier B., Daneman R., Ransohoff R.M. Development, maintenance and disruption of the blood-brain barrier. Nat. Med. 2013;19(12):1584–1596. doi: 10.1038/nm.3407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Eeg-Olofsson O., Wigertz A., Link H. Immunoglobulin abnormalities in cerebrospinal fluid and blood in children with febrile seizures. Neuropediatrics. 1982;13(1):39–41. doi: 10.1055/s-2008-1059593. [DOI] [PubMed] [Google Scholar]
- 267.Kanberg N., Ashton N.J., Andersson L.M., et al. Neurochemical evidence of astrocytic and neuronal injury commonly found in COVID-19. Neurology. 2020;95(12):e1754–e1759. doi: 10.1212/WNL.0000000000010111. [DOI] [PubMed] [Google Scholar]
- 268.DeKosky S.T., Kochanek P.M., Valadka A.B., et al. Blood biomarkers for detection of brain injury in COVID-19 patients. J. Neurotrauma. 2021;38(1):1–43. doi: 10.1089/neu.2020.7332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Lahiri D., Ardila A. COVID-19 pandemic: a neurological perspective. Cureus. 2020;12(4) doi: 10.7759/cureus.7889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Romoli M., Jelcic I., Bernard-Valnet R., et al. A systematic review of neurological manifestations of SARS-CoV-2 infection: the devil is hidden in the details. Eur. J. Neurol. 2020;27(9):1712–1726. doi: 10.1111/ene.14382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Sun T., Guan J. Novel coronavirus and the central nervous system. Eur. J. Neurol. 2020:e52. doi: 10.1111/ene.14227. [DOI] [PubMed] [Google Scholar]
- 272.Zhou L., Zhang M., Wang J., Gao J. Sars-Cov-2: underestimated damage to nervous system. Travel Med. Infect. Dis. 2020:101642. doi: 10.1016/j.tmaid.2020.101642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Finsterer J., Stollberger C. Update on the neurology of COVID-19. J. Med. Virol. 2020;92(11):2316–2318. doi: 10.1002/jmv.26000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Liu K., Pan M., Xiao Z., Xu X. Neurological manifestations of the coronavirus (SARS-CoV-2) pandemic 2019-2020. J. Neurol. Neurosurg. Psychiatry. 2020;91(6):669–670. doi: 10.1136/jnnp-2020-323177. [DOI] [PubMed] [Google Scholar]
- 275.Bajwa S.J.S., Gupta R., Wahi A., Goraya S.P.S. Exploring the unknown territories in the new normal world of COVID. J. Anaesthesiol. Clin. Pharmacol. 2020;36(Suppl. 1):S77–S80. doi: 10.4103/joacp.JOACP_350_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Destras G., Bal A., Escuret V., Morfin F., Lina B., Josset L. Systematic SARS-CoV-2 screening in cerebrospinal fluid during the COVID-19 pandemic. The Lancet Microbe. 2020;1(4) doi: 10.1016/S2666-5247(20)30066-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Meppiel E., Peiffer-Smadja N., Maury A., et al. Neurological manifestations associated with COVID-19: a multicentric registry. Clin. Microbiol. Infect. 2020 doi: 10.1016/j.cmi.2020.11.005. S1198-743X(20)30698-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Kremer S., Lersy F., de Sèze J., et al. Brain MRI findings in severe COVID-19: a retrospective observational study. Radiology. 2020;297(2):e242–e251. doi: 10.1148/radiol.2020202222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Helms J., Kremer S., Merdji H., et al. Delirium and encephalopathy in severe COVID-19: a cohort analysis of ICU patients. Crit. Care. 2020;24(1) doi: 10.1186/s13054-020-03200-1. 491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Brouwer M.C., Ascione T., Pagliano P. Neurologic aspects of covid-19: a concise review. Infez. Med. 2020;28(Suppl. 1):42–45. [PubMed] [Google Scholar]
- 281.Espíndola O.M., Brandão C.O., Gomes Y.C.P., et al. Cerebrospinal fluid findings in neurological diseases associated with COVID-19 and insights into mechanisms of disease development. Int. J. Infect. Dis. 2021;102:155–162. doi: 10.1016/j.ijid.2020.10.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Neumann B., Schmidbauer M.L., Dimitriadis K., et al. Cerebrospinal fluid findings in COVID-19 patients with neurological symptoms. J. Neurol. Sci. 2020;418:117090. doi: 10.1016/j.jns.2020.117090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Remsik J., Wilcox J.A., Babady N.E., et al. Inflammatory leptomeningeal cytokines mediate delayed COVID-19 encephalopathy. medRxiv. 2020 doi: 10.1101/2020.09.15.20195511. [DOI] [Google Scholar]
- 284.Al-Sarraj S., Troakes C., Hanley B., et al. Invited review: the spectrum of neuropathology in COVID-19. Neuropathol. Appl. Neurobiol. 2020 doi: 10.1111/nan.12667. [DOI] [PubMed] [Google Scholar]
- 285.Paniz-Mondolfi A., Bryce C., Grimes Z., et al. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) J. Med. Virol. 2020;92(7):699–702. doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Davies N.W., Brown L.J., Gonde J., et al. Factors influencing PCR detection of viruses in cerebrospinal fluid of patients with suspected CNS infections. J. Neurol. Neurosurg. Psychiatry. 2005;76(1):82–87. doi: 10.1136/jnnp.2004.045336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Seyfert S., Kunzmann V., Schwertfeger N., Koch H.C., Faulstich A. Determinants of lumbar CSF protein concentration. J. Neurol. 2002;249(8):1021–1026. doi: 10.1007/s00415-002-0777-2. [DOI] [PubMed] [Google Scholar]
- 288.Pitts A.F., Carroll B.T., Gehris T.L., Kathol R.G., Samuelson S.D. Elevated CSF protein in male patients with depression. Biol. Psychiatry. 1990;28(7):629–637. doi: 10.1016/0006-3223(90)90401-m. [DOI] [PubMed] [Google Scholar]
- 289.Brooks J.A., McCudden C., Breiner A., Bourque P.R. Causes of albuminocytological dissociation and the impact of age-adjusted cerebrospinal fluid protein reference intervals: a retrospective chart review of 2627 samples collected at tertiary care centre. BMJ Open. 2019;9(2) doi: 10.1136/bmjopen-2018-025348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Giovannoni G. Cerebrospinal fluid analysis. Handb. Clin. Neurol. 2014;122:681–702. doi: 10.1016/B978-0-444-52001-2.00029-7. [DOI] [PubMed] [Google Scholar]
- 291.Delbue S., Franciotta D., Giannella S., et al. Human polyomaviruses in the cerebrospinal fluid of neurological patients. Microorganisms. 2019;8(1):16. doi: 10.3390/microorganisms8010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Matschke J., Lütgehetmann M., Hagel C., et al. Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. The Lancet Neurology. 2020;19(11):919–929. doi: 10.1016/S1474-4422(20)30308-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Table 1: CSF Results for Patients with COVID-19 and Disorders of the Central Nervous System and Supplemental Table 2: CSF Results for Patients with COVID-19 and Disorders of the Peripheral Nervous System