Abstract
Aim
To describe the clinical characteristics and risk factors associated with the progression of COVID-19 in elderly diabetes patients.
Methods
This was a retrospective cohort study, including elderly COVID-19 patients admitted to Wuhan Huoshenshan Hospital between February 10 and 13, 2020. Demographic data, medical history, signs and symptoms, and laboratory parameters were collected and analysed.
Results
We included 131 elderly COVID-19 patients (50 patients with diabetes). COVID-19 diabetes patients experienced more severe pneumonia and abnormal organ functions than non-diabetes patients (P < 0.05 or P < 0.01). Most function indicators were significantly different between the mild to moderate and severely ill groups in diabetes patients (P < 0.05 or P < 0.01). Python analysis confirmed diabetes was the independent risk factor of COVID-19 progression in elderly patients. All blood glucose (BG) indices went into the risk factor equation. The cut-off values of COVID-19 progression were BG value on admission > 8.0 mmol/L or maximum BG value > 12.0 mmol/L in all elderly patients, and BG value on admission > 5.1 mmol/L or maximum BG value > 5.4 mmol/L in non-diabetes patients.
Conclusions
Diabetes is an independent important risk factor, and glucose levels associate closely with COVID-19 progression in elderly patients.
Keywords: Diabetes, COVID-19, Progression, Glucose levels, Risk factors
Abbreviations: COVID-19, coronavirus disease 2019; MERS, Middle East respiratory syndrome; CHD, coronary atherosclerotic heart disease; COPD, chronic obstructive pulmonary disease; CT, computed tomography; RNA, ribonucleic acid; ICU, intensive care unit; ARDS, acute respiratory distress syndrome; MODS, multiple-organ dysfunction syndrome; BG, blood glucose; T2DM, type 2 diabetes mellitus; cTNI, cardiac troponin I; cTNT, cardiac troponin T; ULN, upper limit of normal; MRI, magnetic resonance imaging; IQR, interquartile range; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; LDH, lactate dehydrogenase; CRP, C-reactive protein; BNP, brain natriuretic peptide; PT, prothrombin time; APPT, activated partial thromboplastin time; OR, odds ratio
1. Introduction
In December 2019, a cluster of cases with pneumonia caused by a novel RNA beta-coronavirus (SARS-CoV-2) emerged. The World Health Organization (WHO) named it coronavirus disease 2019 (COVID-19) and declared it a pandemic recently. Severe cases of COVID-19 can rapidly progress to acute respiratory distress syndrome (ARDS), septic shock, and multiple organ dysfunction syndrome (MODS) [1].
It has previously been observed that patients with older age and chronic underlying conditions might have a considerable risk of experiencing severe cases and higher mortality [1], [2], [3], [4]. Moreover, recent evidence suggested that diabetes might be a risk factor for the progression and prognosis of COVID-19 [5], [6]. Patients with diabetes are more susceptible to infections and exhibit worse prognosis than patients without diabetes [7], which is believed to be due to the detrimental effects of hyperglycaemia to control viremia and inflammation [8]. Data from several studies showed that diabetes has become one of the most common comorbidities in infected individuals [2], [9]. As the COVID-19 pandemic progresses, the high incidence of diabetes worldwide makes this problem particularly worrying. Previous clinical trials examining the effects of glucose control on mortality have controversial results [8]. Despite the multiple epidemiological studies regarding COVID-19 in patients with diabetes, the risk factors for COVID-19 in elderly patients with diabetes have not been well described. In elderly individuals with COVID-19 and pre-existing type 2 diabetes mellitus (T2DM), a key challenge for clinicians is to improve outcomes in the face of uncertainty regarding the degree of glycaemic management that should be maintained and its benefits and risks.
To our knowledge, no previous studies have been performed to estimate the risk factors for severe disease in elderly COVID-19 diabetes patients. Therefore, in this study, we aim to report the clinical characteristics and risk factors associated with the progression of COVID-19 in elderly diabetes patients.
2. Materials and methods
2.1. Study design and participants
This was a single-centred, retrospective cohort study, and the study protocol was approved by the Institutional Ethics Committee of Huoshenshan Hospital (HSSLL030). Given the urgency of the COVID-19 pandemic, the need for informed consent forms were waived by the ethics board of the hospitals.
The study screened all patients with COVID-19 from two regular wards and one intensive care unit (ICU) hospitalised in Wuhan Huoshenshan Hospital between February 10 and 13, 2020. COVID-19 was diagnosed based on chest computed tomography (CT) manifestations and/or reverse transcription-polymerase chain reaction (RT-PCR) following the criteria of the New Coronavirus Pneumonia Prevention and Control Program (fifth and sixth edition at that time, National Health Commission of China, 2020; World Health Organization, 2020) [10], [11].
2.2. Data collection
Collected data including demographic data, medical history, signs and symptoms, laboratory parameters, CT scans, and outcomes were extracted from electronic medical records. The outcomes were followed up until March 04, 2020. The patients’ medical records were analysed by an integrated research team, including physicians, data scientists, and statisticians. After the deidentification process of removing the participants’ personal information (e.g. name and ID) and using a coding system, the data were analysed.
2.3. Definitions
The COVID-19 diagnostic criteria of the New Coronavirus Pneumonia Prevention and Control Program were as follows: (1) fever and/or other typical respiratory symptoms, (2) typical CT image manifestations of viral pneumonia, and (3) a positive RT-PCR result for COVID-19 RNA and/or gene diagnosis. Criteria for severe COVID-19 patients were as follows: (1) respiratory distress, with respiratory rate ≥ 30 breaths/min, (2) finger oxygen saturation ≤ 93% in the resting state, and (3) arterial blood oxygen partial pressure (PaO2/oxygen concentration (FiO2) ≤ 300 mmHg.
The T2DM status was designated based on the patient’s medical history and guideline for the prevention and control of T2DM in China (2017) (Chinese Diabetes Society, 2018). The timing and frequency of obtaining the fasting and 2-h postprandial blood glucose (BG) (2-hPG) varied between individuals, depending on the necessity related to the severity of the comorbid T2DM.
Hypertension, cardiac injury, acute liver injury, and acute kidney injury status were designated based on the patient’s medical history and guideline for diagnosis [12]. Cardiac injury was defined when the serum levels of the cardiac injury biomarkers cardiac troponin I (cTNI) and cardiac troponin T (cTNT) were higher than the upper limit of normal (ULN). Acute liver injury was defined when an acutely increased level of serum alanine aminotransferase (ALT) and serum alkaline phosphatase of ULN was observed [13]. Acute kidney injury was indicated by the value of serum creatinine level when it exceeded the ULN (μmmol/L).
2.4. Statistical analysis
All statistical analyses were performed using SPSS Statistics (version 23.0, IBM, Armonk, NY, USA) and Python 3.6 (Python Software Foundation, Delaware, USA). Data with continuous variables were presented as mean ± SD or median (interquartile range [IQR]), and data with categorical variables were presented as frequency rates and percentage (%). Comparison between two groups was analysed using Student’s t-tests (normally distributed) or Mann-Whitney U test (non-normally distributed) for continuous variables. Comparison of categorical variables was analysed by Fisher’s exact test or χ2 test. The risk and corresponding hazard ratio (HR) for severe condition were analysed using Python by fourfold cross-validation. Cut-off values of BG to predict COVID-19 severity were analysed using Python by the SHAP (SHapley Additive exPlanations) method [14]. A difference with a two-tailed P value < 0.05 was considered statistically significant.
3. Results
3.1. Population
A total of 143 patients were initially screened for the study, and 12 patients younger than 60 years old were excluded. The 131 patients were divided into two groups: diabetes patients (n = 50) and non-diabetes patients (n = 81) according to the clinical diagnosis and/or medical history on admission (Fig. 1 ). Differences were compared between two groups, and risk factors were analysed.
Fig. 1.
Diagram of study inclusion criteria.
3.2. General conditions of elderly participants and difference between diabetes and non-diabetes patients
Of the 131 inpatients with COVID-19, the median age was 67 (IQR, 62–72). There were 60 (45.8%) female patients; 89 were mild to moderate patients (67.9%), and 42 were severely ill patients (32.1%). All patients were scanned using chest CT. The three most common symptoms were cough (110 [84.0%]), fever (106 [80.9%]), and shortness of breath (86 [65.6%]); the less common symptoms were haemoptysis (1 [0.8%] of 131 patients), rhinorrhoea (2 [1.5%]), and headache (4 [3.1%]). The most common comorbidities were hypertension, diabetes, and coronary atherosclerotic heart disease (38.9%, 38.2%, and 11.5%) (Table 1 ).
Table 1.
Demographics and symptomatic characteristics of patients with COVID-19.
| Total (%) | Diabetes (%) | Non-diabetes (%) | P value | |
|---|---|---|---|---|
| (n = 131) | (n = 50) | (n = 81) | ||
| Age, median (IQR) | 67 (62–72) | 68.5 (62–75) | 67 (62–71) | 0.137 |
| Gender | 0.745 | |||
| Male | 71 (54.2) | 28 (56.0) | 43 (53.1) | |
| Female | 60 (45.8) | 22 (44.0) | 38 (46.9) | |
| Diagnosis method | 0.087 | |||
| RT-PCR | 118 (90.1) | 42 (82.0) | 76 (93.8) | |
| Gene | 1 (0.8) | 1 (2.0) | 0 (0.0) | |
| Clinical diagnosis | 12 (9.2) | 7 (14.0) | 5 (6.2) | |
| CT side | 0.348 | |||
| Unilateral | 6 (4.6) | 1 (2.0) | 5 (6.2) | |
| Bilateral | 122 (93.1) | 47 (94.0) | 75 (92.6) | |
| Unclear | 3 (2.3) | 2 (4.0) | 1 (1.2) | |
| Comorbidities | ||||
| Diabetes | 50 (38.2) | 50 (100) | 0 (0.0) | / |
| Hypertension | 51 (38.9) | 24 (48.0) | 27 (33.3) | 0.094 |
| CHD | 15 (11.5) | 6 (12.0) | 9 (11.1) | 0.877 |
| Cerebrovascular disease | 6 (4.6) | 3 (6.0) | 3 (3.7) | 0.417 |
| Liver disease | 3 (2.3) | 1 (2.0) | 2 (2.5) | 0.675 |
| Kidney disease | 5 (3.8) | 3 (6.0) | 2 (2.5) | 0.283 |
| COPD | 3 (2.3) | 1 (2.0) | 2 (2.5) | 0.675 |
| Malignancy | 7 (5.3) | 1 (2.0) | 6 (7.4) | 0.177 |
| Signs and symptoms | ||||
| Fever | 106 (80.9) | 38 (76.0) | 68 (84.0) | 0.261 |
| Cough | 110 (84.0) | 41 (82.0) | 69 (85.2) | 0.629 |
| Expectoration | 23 (17.6) | 12 (24.0) | 11 (13.6) | 0.128 |
| Rhinorrhoea | 2 (1.5) | 1 (2.0) | 1 (1.5) | 0.619 |
| Haemoptysis | 1 (0.8) | 1 (2.0) | 0 (0.0) | 0.382 |
| Shortness of breath | 86 (65.6) | 40 (80.0) | 46 (56.8) | 0.007 |
| Fatigue | 83 (63.4) | 34 (68.0) | 49(60.5) | 0.386 |
| Myalgia | 52 (39.7) | 20 (40.0) | 32 (39.5) | 0.955 |
| Diarrhoea | 13 (9.9) | 4 (8.0) | 9 (11.1) | 0.781 |
| Nausea and vomiting | 12 (9.2) | 7 (14.0) | 5 (6.2) | 0.231 |
| Arrhythmias | 9 (6.9) | 8 (16.0) | 1 (1.2) | 0.004 |
| Palpitation | 6 (4.6) | 2 (4.0) | 4 (4.9) | 0.583 |
| Dizziness | 5 (3.8) | 2 (4.0) | 3 (3.7) | 0.634 |
| Headache | 4 (3.1) | 2 (4.0) | 2 (2.5) | 0.494 |
| Outcome | <0.001 | |||
| Still in hospital | 22 (16.8) | 13 (26.0) | 9 (11.1) | |
| Discharged | 93 (71.0) | 25 (50.0) | 68 (84.0) | |
| Death | 16 (12.2) | 12 (24.0) | 4 (4.9) |
Abbreviation: IQR, interquartile range; CHD, coronary atherosclerotic heart disease; COPD, chronic obstructive pulmonary diseases.
There was no difference in age, gender, and diagnostic methods between diabetes and non-diabetes patients (P > 0.05). Among all influenza-like symptoms, respiratory symptoms, neuromuscular symptoms, and circulatory symptoms, only shortness of breath and arrhythmia had statistically significant differences between diabetes and non-diabetes patients (P < 0.01) (Table 1). There were 46% diabetes patients with mild to moderate symptoms and 54% with severe symptoms, compared to the 81.5% non-diabetes patients with mild to moderate symptoms and 18.5% with severe symptoms (Table 3).
Table 3.
Difference of mild to moderate and severely ill groups in diabetes and non-diabetes elderly patients.
| Reference values | Diabetes (n = 50) |
Non-diabetes (n = 81) |
||||||
|---|---|---|---|---|---|---|---|---|
| Mild to moderate (n = 23, 46%) | Severe (n = 27, 54%) | P value | Mild to moderate (n = 66, 81.5%) | Severe (n = 15, 18.5%) | P value | |||
| Blood glucose(mmol/L) | ||||||||
| Maximum BG | 3.9–6.11 | 16.7 (13.21–19.3) | 17.37 (14.83–24.06) | 0.169 | 5.03 (4.67–5.38) | 7.42 (5.44–8.75) | <0.001 | |
| BG at the time of admission | 3.9–6.11 | 7.3 (5.95–9) | 9.6 (6.02–13.7) | 0.088 | 4.86 (4.48–5.13) | 5.71 (4.91–7.78) | 0.018 | |
| Average BG | 3.9–6.11 | 9 (7.42–11.99) | 11.4 (7.75–14.61) | 0.191 | 4.86 (4.48–5.13) | 5.71 (4.91–7.7) | 0.018 | |
| Biochemical detection | ||||||||
| Total protein (g/L) | 65–85 | 61.2 (58.5–64.55) | 58.4 (54–62.4) | 0.025 | 61.85 (58.6–67) | 58.6 (55.7–61.6) | 0.02 | |
| ALT (IU/L) | 7–40 | 19.9 (14.45–31.46) | 36.2 (20.1–47.7) | 0.067 | 22.45 (16.4–37.4) | 34.3 (24.95–88.6) | 0.02 | |
| AST (IU/L) | 7–45 | 17.8 (15.55–28.65) | 30.2 (24.8–38.6) | 0.002 | 21.45 (15.4–27.4) | 30.8 (28.92–51) | 0.002 | |
| Albumin (g/L) | 40–55 | 34.88 ± 3.8 | 30.68 ± 2.82 | <0.001 | 34.7 (32.7–37.4) | 30.7 (27.55–33.05) | 0.001 | |
| BUN (mmol/L) | 2.6–7.5 | 5.56 (4.17–6) | 6.9 (5.5–9.64) | 0.031 | 4.55 (3.75–5.42) | 5.82 (4.87–8.39) | 0.007 | |
| Creatine kinase (IU/L) | 24–170 | 34.8 (26.65–44) | 69.3 (36.1–99.8) | 0.004 | 42.05 (31.3–67.06) | 59 (26.7–118.9) | 0.293 | |
| Creatine kinase-MB (IU/L) | 0–24 | 8.8 (6.65–11.25) | 11.6 (10.1–15.15) | 0.003 | 8.9 (6.7–11.2) | 13.6 (10.53–18.25) | <0.001 | |
| LDH (IU/L) | 120–250 | 207.09 ± 58.01 | 461.01 ± 146.89 | <0.001 | 190.8 (165.6–227.6) | 360 (264.05–434.75) | <0.001 | |
| Inflammatory indices | ||||||||
| CRP (mg/L) | 0–4 | 6.02 (3.82–34.52) | 75.68 (34.42–127.67) | <0.001 | 4.32 (1.26–10.64) | 61.71 (21.34–148.34) | <0.001 | |
| Blood routine examination | ||||||||
| White blood cell count (×109/L) | 3.5–9.5 | 6.26 ± 1.82 | 8.38 ± 3.08 | 0.006 | 5.5 (4.1–6.8) | 9.7 (7.15–13.3) | <0.001 | |
| Neutrophil percentage (%) | 40–75 | 67.2 (59.05–72.85) | 86.2 (79.5–92.6) | <0.001 | 62.5 (57.4–71.1) | 88.6 (82.95–92.05) | <0.001 | |
| Lymphocyte percentage (%) | 20–50 | 21 (16.7–31.95) | 8.1 (4.2–13.45) | <0.001 | 27.8 (19–31.8) | 6.6 (4.75–11.7) | <0.001 | |
| Neutrophil count (×109/L) | 1.8–6.3 | 3.96 (2.88–5.55) | 7.13 (5.55–8.4) | <0.001 | 3.55 (2.33–4.81) | 8.83 (5.97–11.77) | <0.001 | |
| Lymphocyte count (×109/L) | 1.1–3.2 | 1.41 ± 0.52 | 0.73 ± 0.4 | <0.001 | 1.37 ± 0.48 | 0.76 ± 0.32 | <0.001 | |
| Eosinophil count (×109/L) | 0.02–0.52 | 0.09 (0.06–0.15) | 0.02 (0–0.04) | <0.001 | 0.07 (0.04–0.11) | 0.01 (0–0.05) | <0.001 | |
| Red blood cell count (×109/L) | 4.3–5.8 | 3.93 (3.72–4.13) | 4.22 (3.94–4.64) | 0.029 | 3.94 (3.63–4.17) | 4.12 (3.85–4.49) | 0.173 | |
| Platelet count (×109/L) | 125–350 | 265.59 ± 100.73 | 182.44 ± 84.51 | 0.003 | 269.5 ± 76.21 | 263.15 ± 97.19 | 0.403 | |
| Coagulation indices | ||||||||
| PT (seconds) | 9.2–15 | 12.59 (12.2–13.95) | 14.26 (13.9–16.73) | <0.001 | 13.25 (12.46–13.95) | 13.95 (12.7–14.21) | 0.151 | |
| D-dimer (mg/L) | 0–0.55 | 1.17 (0.41–2.57) | 2.57 (2.3–6.43) | 0.002 | 0.69 (0.36–2.57) | 2.57 (0.71–4.09) | 0.004 | |
Abbreviation: IQR, interquartile range; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, urea nitrogen; LDH, lactate dehydrogenase; CRP, C-reactive protein; PT, prothrombin time.
3.3. Clinical laboratory differences between diabetes and non-diabetes elderly patients
Laboratory tests after admission was shown in Table 2 . Of the 131 cases, a medium level of maximum BG and inflammatory indices (procalcitonin, c-reactive protein [CRP]) were higher than the normal range (Table 2).
Table 2.
Laboratory parameters of patients with COVID-19.
| Reference values | Total (n = 131) | Diabetes (n = 50) | Non-diabetes (n = 81) | P value | |
|---|---|---|---|---|---|
| Blood oxygen desaturation, n (%) | 52 (39.7) | 28 (56.0) | 24 (29.6) | 0.003 | |
| Blood glucose | |||||
| Maximum BG, median (IQR), mmol/L | 3.9–6.11 | 6.34 (5.02–15.2) | 17.15 (14.2–21.3) | 5.11 (4.72–5.76) | <0.001 |
| BG at the time of admission, median (IQR), mmol/L | 3.9–6.11 | 5.27 (4.81–7.8) | 8.1 (5.93–12.8) | 4.92 (4.5–5.32) | <0.001 |
| Average BG at the day of admission, median (IQR), mmol/L | 3.9–6.11 | 5.37 (4.81–8.83) | 9.91 (7.41–14.21) | 4.92 (4.5–5.32) | <0.001 |
| Biochemical detection | |||||
| Total protein, median (IQR), g/L | 65–85 | 60.6 (57.2–65.2) | 59.85 (57.1–63.6) | 61.15 (57.7–66.15) | 0.225 |
| ALT, median (IQR), IU/L | 7–40 | 25.1 (16.6–42) | 24.1 (16.2–44.4) | 25.3 (16.95–38.7) | 0.789 |
| AST, median (IQR), IU/L | 7–45 | 23.4 (16.8–33.9) | 26 (18.4–38) | 22.3 (16.7–31.5) | 0.102 |
| Albumin, median (IQR), g/L | 40–55 | 33.5 (30.3–36.4) | 31.9 (30.1–35.5) | 34.2 (30.9–36.95) | 0.046 |
| BUN, median (IQR), mmol/L | 2.6–7.5 | 5.01 (4.05–6.9) | 5.93 (4.61–8.43) | 4.73 (3.83–5.89) | 0.001 |
| Creatinine, median (IQR), μmol/L | 41–73 | 67.9 (57.6–82.6) | 71.55 (61.3–82.85) | 67.6 (55.6–81.75) | 0.356 |
| Uric acid, median (IQR), μmol/L | 142–340 | 267 (192–336) | 247 (182–351) | 270 (205–332.5) | 0.404 |
| Total bile acid, median (IQR), μmol/L | 0–10 | 3.7 (2.5–5.6) | 4 (2.5–6.5) | 3.65 (2.5–5.45) | 0.445 |
| Creatine kinase, median (IQR), IU/L | 24–170 | 42.8 (29.3–71.3) | 42.35 (27.7–75.9) | 42.45 (31.05–68.73) | 0.861 |
| Creatine kinase-MB, median (IQR), IU/L | 0–24 | 10.1 (7.4–12.7) | 10.88 (8.1–12.7) | 9.7 (7.3–12.8) | 0.142 |
| LDH, median (IQR), IU/L | 120–250 | 227.6 (174.1–325) | 284.1 (192.4–461.7) | 201.35 (172.35–272.85) | <0.001 |
| Troponin I, median (IQR), ng/mL | 0–0.04 | 0.01 (0.01–0.34) | 0.025 (0.01–0.046) | 0.01 (0.01–0.022) | 0.557 |
| Myohaemoglobin, median (IQR), ng/mL | 0–65 | 10.53 (5.11–33.30) | 21.89 (8.23–47.67) | 9.35 (3.47–14.7) | 0.175 |
| BNP, median (IQR), pg/mL | 0–100 | 54.21 (0.28–123.08) | 68.77 (13.29–123.08) | 48.65 (0.01–123.08) | 0.336 |
| Inflammatory indices | |||||
| Procalcitonin, median (IQR), ng/mL | 0–0.05 | 0.06 (0.04–0.18) | 0.95 (0.05–0.24) | 0.04 (0.03–0.14) | 0.703 |
| CRP, median (IQR), mg/L | 0–4 | 9.99 (2.72–54) | 36.48 (5.77–87.72) | 7.12 (1.86–36.03) | 0.002 |
| Blood routine examination | |||||
| White blood cell count, median (IQR), ×109/L | 3.5–9.5 | 6.5 (4.9–8.1) | 6.97 (5.2–9) | 6 (4.2–7.4) | 0.023 |
| Neutrophil percentage, median (IQR), % | 40–75 | 69.8 (60.1–82.2) | 75.95 (65.4–87.1) | 65.35 (58.15–79.05) | 0.004 |
| Lymphocyte percentage, median (IQR), % | 20–50 | 20.2 (10.3–30.6) | 14.8 (7–23.8) | 24.85 (12.6–31.4) | 0.006 |
| Neutrophil count, median (IQR), ×109/L | 1.8–6.3 | 4.65 (2.83–6.33) | 5.62 (3.53–7.72) | 3.96 (2.38–5.62) | 0.005 |
| Lymphocyte count, mean ± SD, ×109/L | 1.1–3.2 | 1.18 (0.73–1.59) | 1.04 ± 0.57 | 1.26 ± 0.51 | 0.025 |
| Monocyte count, median (IQR), ×109/L | 0.1–0.6 | 0.43 (0.31–0.54) | 0.42 (0.3–0.54) | 0.44 (0.31–0.57) | 0.509 |
| Eosinophil count, median (IQR), ×109/L | 0.02–0.52 | 0.06 (0.02–0.11) | 0.05 (0.01–0.12) | 0.06 (0.03–0.1) | 0.662 |
| Red blood cell count, median (IQR), ×109/L | 4.3–5.8 | 3.99 (3.7–4.33) | 4.06 (3.74–4.34) | 3.97 (3.64–4.28) | 0.172 |
| Haemoglobin, median (IQR), g/L | 130–175 | 122.023 (111–132) | 124 (114–136) | 120.5(109.5–131) | 0.111 |
| Platelet count, mean ± SD, ×109/L | 125–350 | 259 (187–319) | 220.69 ± 100.49 | 269.3 ± 79.41 | 0.002 |
| Coagulation indices | |||||
| PT, median (IQR), seconds | 9.2–15 | 13.39 (12.44–14.16) | 13.95 (12.42–14.73) | 13.29 (12.46–13.95) | 0.136 |
| APPT, median (IQR), seconds | 21–37 | 28.44 (25.81–30.07) | 27.93 (25.23–29.17) | 28.44 (26.37–30.23) | 0.198 |
| D-dimer, median (IQR), mg/L | 0–0.55 | 1.36 (0.52–2.57) | 2.57 (0.83–3.88) | 0.85 (0.43–2.57) | 0.001 |
Abbreviation: IQR, interquartile range; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, urea nitrogen; LDH, lactate dehydrogenase; BNP, brain natriuretic peptide; CRP, C-reactive protein; PT, prothrombin time; APPT, activated partial thromboplastin time.
All BG indices showed a significant difference between diabetes and non-diabetes patients (P < 0.01). Blood oxygen saturation was significantly lower in diabetes patients (P < 0.01). Albumin level, kidney function (blood urea nitrogen [BUN]), indicators of myocardial injury (lactate dehydrogenase [LDH]), inflammatory factors (CRP), and coagulation indicator (D-dimer) for biochemical detection in the diabetes groups were all inferior to the non-diabetes group (P < 0.05 or P < 0.01). All indices for blood routine examination, except monocyte count, eosinophil count, and red blood cell count, showed a significant difference between the diabetes and non-diabetes groups (P < 0.05 or P < 0.01) (Table 2).
3.4. Differences of mild to moderate and severely ill groups between diabetes and non-diabetes elderly patients
Among all the indices, nutritional indices (total protein, albumin levels), liver and kidney function tests (AST, BUN, and LDH), myocardial injury indices (creatine kinase, creatine kinase-MB, LDH), inflammatory marker (CRP), most routine blood test indicators (white blood cell count, neutrophil count and percentage, lymphocyte count and percentage, eosinophil count, red blood cell count, and platelet count), and coagulation indicators (prothrombin time [PT], D-dimer) were expressed significantly differently between the mild to moderate and severely ill groups in diabetes patients (P < 0.05 or P < 0.01) (Table 3).
Compared with diabetes patients, all BG indices (BG value at the time of admission, maximum BG value, average BG value on the day of admission) and ALT level were significantly different between the mild to moderate and severely ill groups in non-diabetes patients (P < 0.05 or P < 0.01) (Table 3), but not in diabetes patients. Conversely, a difference in creatine kinase level, red blood cell count, and PT, as seen in diabetes patients, could not be found in the non-diabetes subgroups between the mild to moderate and severely ill groups (P > 0.05).
3.5. Risk factors and blood glucose cut-off values associated with COVID-19 progression
All difference indicators in univariate comparison were included in the Python analysis to obtain the risk and corresponding HR. Among all indicators that showed a difference, decreased oxygen saturation, shortness of breath, and respiratory rate were excluded because they were the diagnostic clinical presentation of severely ill patients. Among the neutrophil ratio, lymphatic ratio, neutrophil value, and lymphatic value, two values were selected for presentation (Table 4 ).
Table 4.
Python analysis of risk factors associated with severe COVID-19 progression in elderly patients.
| Factors | Total |
Diabetes |
Non-diabetes |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Rank | ß-value | HR value | Rank | ß-value | HR value | Rank | ß-value | HR value | |
| Diabetes | 7 | 0.58 | 1.79 | ||||||
| Maximum BG | 9 | 0.55 | 1.73 | 2 | 0.57 | 1.77 | |||
| BG at the time of admission | 8 | 0.56 | 1.75 | 8 | 0.32 | 1.38 | |||
| Average BG | 11 | 0.52 | 1.68 | 9 | 0.32 | 1.38 | |||
| Systolic pressure | 6 | 0.59 | 1.8 | ||||||
| Hypertension | 10 | 0.53 | 1.7 | 1 | 0.65 | 1.92 | |||
| CRP | 1 | 1.14 | 3.12 | 3 | 0.85 | 2.34 | 3 | 0.56 | 1.76 |
| LDH | 2 | 1.08 | 2.94 | 1 | 1.08 | 2.95 | 6 | 0.42 | 1.52 |
| D-dimer | 5 | 0.73 | 2.07 | 5 | 0.6 | 1.81 | 11 | 0.30 | 1.35 |
| Neutrophil count | 3 | 0.89 | 2.42 | 2 | 0.95 | 2.57 | 5 | 0.52 | 1.68 |
| ALT | 4 | 0.84 | 2.32 | 9 | 0.2 | 1.22 | 4 | 0.54 | 1.71 |
| AST | 13 | 0.51 | 1.66 | 10 | 0.08 | 1.08 | 7 | 0.36 | 1.43 |
| Body temperature | 4 | 0.68 | 1.97 | ||||||
| Heart rate | 6 | 0.46 | 1.59 | ||||||
| PT | 7 | 0.37 | 1.45 | ||||||
| Creatine kinase | 15 | 0.46 | 1.58 | 8 | 0.35 | 1.41 | |||
| CT description | 14 | 0.50 | 1.64 | 10 | 0.3 | 1.35 | |||
Abbreviation: BG, blood glucose; CRP, C-reactive protein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; LDH, lactate dehydrogenase; PT, prothrombin time.
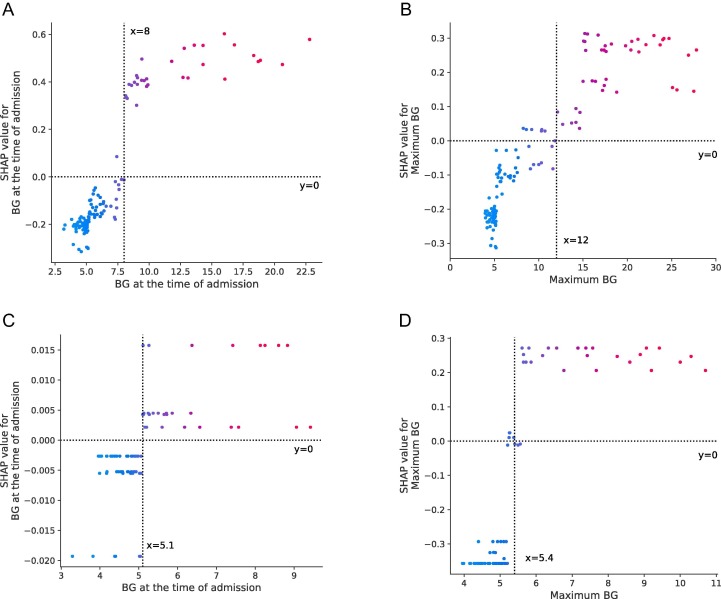
In the total population, diabetes was an independent risk factor among the top 10 risk factors associated with COVID-19 progression (hazard ratio, HR 1.79). All glucose indices went into the risk factor equation among all the elderly patients (HR 1.38–1.77, rank 8, 9, 11). The area under the curve (AUC) of the test set reached 92.24%. Cut-off results of the SHAP analysis showed that elderly patients with BG value on admission > 8.0 mmol/L or maximum BG value > 12.0 mmol/L experienced dramatically higher trends of severity (Fig. 2 A–B).
Fig. 2.
Cut-off values of blood glucose analysed by SHAP. (A–B) SHAP values for BG at the time of admission and maximum BG in all elderly COVID-19 patients; (C–D) SHAP values for BG at the time of admission and maximum BG in non-diabetes elderly COVID-19 patients. SHAP value represents the contribution of each element to the model.
In patients with diabetes, neutrophil count and LDH, CRP, D-dimer, ALT, and AST levels became one of the top 10 risk factors (HR 1.81–2.95), which also became the top factors in the overall and/or non-diabetes population. Body temperature, heart rate, PT, and creatine kinase level solely became the top influential factors of progression in diabetes patients (HR 1.08–1.59). Hypertension was not included in the top factors affecting diabetes patients.
Furthermore, in non-diabetes elderly patients, all glucose indices went into the risk factor equation (HR 1.38–1.77, rank 2, 8, 9). BG value on admission > 5.1 mmol/L or maximum BG value > 5.4 mmol/L was the cut-off value for COVID-19 progression (Fig. 2 C–D).
4. Discussion
In this study, we reported the clinical characteristics of elderly COVID-19 patients. The most frequent comorbidities were hypertension, diabetes, and cardiovascular diseases, similar to the previously reported results [2], [15], [16]. However, in the present cohort, patients with diabetes reached 38.2% (50 of 131), and 32.1% patients were severe cases, which were higher than other studies [5], [15], [17]. Furthermore, 75% of deceased cases had diabetes, which was higher than that in survivors (26.9%). The reasons might be that Huoshenshan Hospital was responsible for receiving diagnosed patients and prefers to treat severely ill patients, and all enrolled individuals were aged over 60 years. There was no obvious demographic and symptomatic difference between diabetes and non-diabetes patients.
Univariable and regression analyses were performed to compare laboratory parameters in the diabetes and non-diabetes groups and mild to moderate and severely ill subgroups. There were three collections of results: (1) Factors that showed a difference in all three comparisons might be the influential factors in the progression of COVID-19 in all patients. Some factors were different between diabetes and non-diabetes patients, such as blood cell counts and albumin, BUN, LDH, CRP, and D-dimer levels, while others are not, like ALT, AST, and CK-Mb (Creatine kinase – MB) levels. (2) Factors that influenced non-diabetes patients, with difference between the diabetes and non-diabetes groups, were the influential factors that diabetes patients have specifically or at a significantly higher level, like all BG indices. (3) Factors that showed a difference only in the diabetes subgroup obviously changed in diabetes patients and led to a severe condition (PT and CK; PLT also displayed the same result but was not included in the influential factors).
From the univariable and regression analyses, it could be confirmed notably that diabetes diagnosis is an independent risk factor for COVID-19 progression in elderly patients. Diabetes patients had 79% higher risk than non-diabetes patients to develop into severe cases. This finding is in accordance with other researches with its highlights [5], [24]. It is known that diabetes patients are more susceptible to infectious diseases and tend to experience faster progression and worse prognosis [18], [19], [20]. Concurrently, a dysregulated immune response caused by diabetes is likely also responsible for the increased disease severity of COVID-19 in patients with diabetes [21]. The SHAP analysis revealed that elevated BG value on admission (>8.0 mmol/L) or maximum BG value (>12.0 mmol/L) was associated with poor progression in elderly COVID-19 patients. These values were consistent with the diagnostic criteria of diabetes. The SHAP analysis result is a brand-new support evidence for diabetes as an independent risk factor in COVID-19 patients. At the same time, these results also suggest that in COVID-19 patients with a history of diabetes, the glycaemic control targets might be slightly extended compared with the conventional control target [22].
Our research reveals a new interesting effect of glucose levels in elderly COVID-19 patients: BG levels are also an important risk factor for progression in non-diabetes patients. BG levels at the time of admission are independent risk factors, which means the former pancreatic status or glucose metabolic abilities relate closely with the progression of COVID-19, even if they do not have diabetes (BG value on admission > 5.1 mmol/L is associated with poor progression). Maximum BG value is an independent risk factor in non-diabetes patients with the highest HR. The population with a maximum BG value > 5.4 mmol/L showed a higher risk of worse progression. This may be because the maximum BG value presents the worst pancreatic conditions. It is partly consistent with a former study reporting that a well-controlled BG level could reduce adverse outcomes and death in COVID-19 patients [23]. Yang et al. studied COVID-19 patients without a history of diabetes and reported that BG level is representative of the clustered indicators of multi-organ injury and an earlier predictor of poor outcomes and death in COVID-19 patients [24], [25], [26], [27]. As reported, elevation of glucose level may be caused by “stress hyperglycaemia” during acute illness in patients with COVID-19 and relates to both insulin secretion dysfunction and insulin resistance [22]. The concentration of BG in airway epithelial secretion would increase as BG level increased, which might break the defensiveness of the epithelia [28].
There were several other prognostic factors in elderly COVID-19 patients with diabetes that is worthy of emphasis. (1) LDH is an enzyme, existing in the myocardia, which is released into the blood when the heart is injured. It became the most effective factor in the progression of COVID-19 diabetes patients in this cohort out of our expectation. This result might be due to the high prevalence of heart injury in severe COVID-19 patients. (2) D-dimer level was also included in the top rank due to its association with COVID-19 progression in overall and diabetes patients, but not in non-diabetes patients. D-dimer level is an accepted risk factor [23], while PT is not mentioned in former studies. Further attention should be paid to disordered coagulation in diabetes patients to prevent COVID-19 progression [29]. (3) Inflammatory factors, CRP level, and neutrophil count were associated with cytokine storms caused by virus invasion, which may be related to the death of COVID-19 patients [15], [21], [23]. (4) Hypertension also showed a significant effect on the overall population and non-diabetes patients (the first ranking risk factor), but not in diabetes patients (there was no difference between mild to moderate and severely ill diabetes patients). The effect of hypertension might be an interesting research point in elderly COVID-19 patients. (5) Age was not included in the top 10 risk factors in neither group analysis, which might be because patients were all elderly, and the age span was only 12 years.
Our study has some limitations. First, owing to the small sample size, more clinical trials are needed to validate the findings. Second, due to the retrospective study design, not all laboratory tests were done in all patients, including those for insulin, C-peptide, HbA1c, and IL-6 levels. Therefore, their roles in predicting disease progression might be underestimated. Third, some patients were transferred from other hospitals at a later period, so the results might be different with researches where patients were included at their first visit. Fourth, we were not able to retrieve the pre-hospital status of diabetes patients from the current cohort due to the urgent circumstance of the COVID-19 pandemic.
5. Conclusions
In conclusion, diabetes is an important independent risk factor for COVID-19 progression in elderly individuals. BG indices are important predictive factors for disease severity.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgements
Not applicable.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Contributors
WM responded to the care of the patient. WM, LW, and YG were involved in the diagnosis, management, and treatment. WM and LW collected the data. PZ, YW, and TL explained the data. YG, JZ and CL designed the study. JZ and YW conducted the analysis. JZ and TL reviewed the relevant papers. YG, PZ, and CL wrote the first draft. All authors contributed equally to writing the manuscript.
References
- 1.Guan W., Liang W., Zhao Y., Liang H., Chen Z., Li Y. Comorbidity and its impact on 1,590 patients with COVID-19 in China: a nationwide analysis. medRxiv. 2020 doi: 10.1101/2020.02.25.20027664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y.i., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S., Huang H., Zhang L.i., Zhou X., Du C., Zhang Y., Song J., Wang S., Chao Y., Yang Z., Xu J., Zhou X., Chen D., Xiong W., Xu L., Zhou F., Jiang J., Bai C., Zheng J., Song Y. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Du Y., Tu L., Zhu P., Mu M.i., Wang R., Yang P., Wang X.i., Hu C., Ping R., Hu P., Li T., Cao F., Chang C., Hu Q., Jin Y., Xu G. Clinical features of 85 fatal cases of COVID-19 from Wuhan. A retrospective observational study. Am J Respir Crit Care Med. 2020;201(11):1372–1379. doi: 10.1164/rccm.202003-0543OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guo W., Li M., Dong Y., Zhou H., Zhang Z., Tian C. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020 doi: 10.1002/dmrr.3319. e3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muniyappa R., Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol-Endocrinol Metab. 2020;318(5):E736–E741. doi: 10.1152/ajpendo.00124.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kumar Nathella P., Babu S. Influence of diabetes mellitus on immunity to human tuberculosis. Immunology. 2017;152(1):13–24. doi: 10.1111/imm.12762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Forbes A., Murrells T., Mulnier H., Sinclair A.J. Mean HbA1c, HbA1c variability, and mortality in people with diabetes aged 70 years and older: a retrospective cohort study. The Lancet Diabetes & Endocrinology. 2018;6(6):476–486. doi: 10.1016/S2213-8587(18)30048-2. [DOI] [PubMed] [Google Scholar]
- 9.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y.i., Zhang L.i., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L.i., Xie J., Wang G., Jiang R., Gao Z., Jin Q.i., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.China NHC. New Coronavirus Pneumonia Prevention and Control Program (5th edition), http://www.nhc.gov.cn/yzygj/s7653p/202002/d4b895337e19445f8d728fcaf1e%0A3e13a/files/ab6bec7f93e64e7f998d802991203cd6.pdf; 2020.
- 11.World Health Organization (WHO). Coronavirus disease (COVID-2019) situation reports. n.d., https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports [accessed April 5, 2020].
- 12.Unger T., Borghi C., Charchar F., Khan N.A., Poulter N.R., Prabhakaran D., Ramirez A., Schlaich M., Stergiou G.S., Tomaszewski M., Wainford R.D., Williams B., Schutte A.E. 2020 International Society of Hypertension global hypertension practice guidelines. J Hypertens. 2020;38(6):982–1004. doi: 10.1097/HJH.0000000000002453. [DOI] [PubMed] [Google Scholar]
- 13.Marrone G, Vaccaro FG, Biolato M, Miele L, Liguori A, Araneo C, et al. Drug-induced liver injury 2017: the diagnosis is not easy but always to keep in mind. Eur Rev Med Pharmacol Sci 2017;21:122–34 [PubMed]
- 14.Li R., Shinde A., Liu A.n., Glaser S., Lyou Y., Yuh B., Wong J., Amini A. Machine learning–based interpretation and visualization of nonlinear interactions in prostate cancer survival. JCO Clinical Cancer Informatics. 2020;(4):637–646. doi: 10.1200/CCI.20.00002. [DOI] [PubMed] [Google Scholar]
- 15.Wang L., He W., Yu X., Hu D., Bao M., Liu H., Zhou J., Jiang H. Coronavirus disease 2019 in elderly patients: Characteristics and prognostic factors based on 4-week follow-up. J Infect. 2020;80(6):639–645. doi: 10.1016/j.jinf.2020.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang J.-J., Dong X., Cao Y.-Y., Yuan Y.-D., Yang Y.-B., Yan Y.-Q., Akdis C.A., Gao Y.-D. Clinical characteristics of 140 patients infected with SARS‐CoV‐2 in Wuhan, China. Allergy. 2020;75(7):1730–1741. doi: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- 17.Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? The Lancet Respir Med. 2020;8(4):e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xu M., Liu P.P., Li H. Innate immune signaling and its role in metabolic and cardiovascular diseases. Physiol Rev. 2019;99(1):893–948. doi: 10.1152/physrev.00065.2017. [DOI] [PubMed] [Google Scholar]
- 19.Knapp S. Diabetes and infection: is there a link? - A mini-review. Gerontology. 2013;59(2):99–104. doi: 10.1159/000345107. [DOI] [PubMed] [Google Scholar]
- 20.Fadini G.P., Morieri M.L., Longato E., Avogaro A. Prevalence and impact of diabetes among people infected with SARS-CoV-2. J Endocrinol Invest. 2020;43(6):867–869. doi: 10.1007/s40618-020-01236-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kulcsar K.A., Coleman C.M., Beck S.E., Frieman M.B. Comorbid diabetes results in immune dysregulation and enhanced disease severity following MERS-CoV infection. JCI Insight. 2019;4:1–18. doi: 10.1172/jci.insight.131774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bornstein S.R., Rubino F., Khunti K., Mingrone G., Hopkins D., Birkenfeld A.L., Boehm B., Amiel S., Holt R.IG., Skyler J.S., DeVries J.H., Renard E., Eckel R.H., Zimmet P., Alberti K.G., Vidal J., Geloneze B., Chan J.C., Ji L., Ludwig B. Practical recommendations for the management of diabetes in patients with COVID-19. The Lancet Diabetes & Endocrinology. 2020;8(6):546–550. doi: 10.1016/S2213-8587(20)30152-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhu L., She Z.-G., Cheng X.u., Qin J.-J., Zhang X.-J., Cai J., Lei F., Wang H., Xie J., Wang W., Li H., Zhang P., Song X., Chen X.i., Xiang M., Zhang C., Bai L., Xiang D.a., Chen M.-M., Liu Y., Yan Y., Liu M., Mao W., Zou J., Liu L., Chen G., Luo P., Xiao B., Zhang C., Zhang Z., Lu Z., Wang J., Lu H., Xia X., Wang D., Liao X., Peng G., Ye P., Yang J., Yuan Y., Huang X., Guo J., Zhang B.-H., Li H. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31(6):1068–1077.e3. doi: 10.1016/j.cmet.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang J-K, Jin J-M, Liu S, Bai P, He W, Wu F, et al. Blood glucose is a representative of the clustered indicators of multi-organ injury for predicting mortality of COVID-19 in Wuhan, China. SSRN Electron J 2020. https://doi.org/10.2139/ssrn.3569852
- 25.Bindom S.M., Lazartigues E. The sweeter side of ACE2: Physiological evidence for a role in diabetes. Mol Cell Endocrinol. 2009;302(2):193–202. doi: 10.1016/j.mce.2008.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roca-Ho H, Riera M, Palau V, Pascual J, Soler MJ. Characterization of ACE and ACE2 expression within different organs of the NOD mouse. Int J Mol Sci 2017;18. https://doi.org/10.3390/ijms18030563 [DOI] [PMC free article] [PubMed]
- 27.Yang J.-K., Lin S.-S., Ji X.-J., Guo L.-M. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010;47(3):193–199. doi: 10.1007/s00592-009-0109-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Philips B.J., Meguer J.-X., Redman J., Baker E.H. Factors determining the appearance of glucose in upper and lower respiratory tract secretions. Intensive Care Med. 2003;29(12):2204–2210. doi: 10.1007/s00134-003-1961-2. [DOI] [PubMed] [Google Scholar]
- 29.Wang T., Chen R., Liu C., Liang W., Guan W., Tang R., Tang C., Zhang N., Zhong N., Li S. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID-19. The Lancet Haematology. 2020;7(5):e362–e363. doi: 10.1016/S2352-3026(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]