Abstract
Common manifestations of COVID-19 are respiratory and can extend from mild symptoms to severe acute respiratory distress. The severity of the illness can also extend from mild disease to life-threatening acute respiratory distress syndrome (ARDS). SARS-CoV-2 infection can also affect the gastrointestinal tract, liver and pancreatic functions, leading to gastrointestinal symptoms. Moreover, SARS-CoV-2 can cause central and peripheral neurological manifestations, affect the cardiovascular system and promote renal dysfunction. Epidemiological data have indicated that cancer patients are at a higher risk of contracting the SARS-CoV-2 virus. Considering the multitude of clinical symptoms of COVID-19, the objective of the present review was to summarize their pathophysiology in previously healthy patients, as well as in those with comorbidities. The present review summarizes the current, though admittedly fluid knowledge on the pathophysiology and symptoms of COVID-19 infection. Although unclear issues still remain, the present study contributes to a more complete understanding of the disease, and may drive the direction of new research. The recognition of the severity of the clinical symptoms of COVID-19 is crucial for the specific therapeutic management of affected patients.
Keywords: SARS-C0V-2, Clinical signs, COVID-19 pandemic, Cancer, Cardiovascular system, Pulmonary system, Gastrointestinal system, Neurological system
Graphical abstract
1. Introduction
Coronaviruses form a large family of viruses that can infect both humans and animals. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) belongs to the family of Coronaviridae and causes COVID-19 disease, which is a respiratory illness (Wang et al., 2020a; Wu and McGoogan, 2020).
SARS-CoV-2 is mainly transmitted from person to person. Respiratory droplets are commonly spread by coughing and sneezing; however, there is some evidence to indicate that transmission via aerosols is also possible, since infectious concentrates of viral particles can be detected in aerosols for up to 3 h or even longer (van Doremalen et al., 2020). Direct contact with infected patients or to a certain extent with surfaces, may also lead to transmission (Ong et al., 2020). Specifically, fomite transmission is possible due to the ability of viral particles to remain on surfaces for a period of time according to the material of the surface (Ong et al., 2020). For example, latex, aluminum, or copper viral particles can remain infectious for approximately 8 h, and plastic or stainless steel surfaces for 1–3 days (Ong et al., 2020; Goumenou et al., 2020; Tsatsakis et al., 2020; Kostoff et al., 2020a). Recent data have also demonstrated that fecal-oral transmission is possible (Yeo et al., 2020). The incubation period for COVID-19 has been estimated at approximately 2–14 days, and its transmission is possible even when patients are asymptomatic or exhibit only minimal symptoms (Bai et al., 2020; Lauer et al., 2020). A previous study demonstrated that the estimated mean incubation time for COVID-19 is 5.1 days (95% CI: 4.5–5.8 days), with almost all the infected patients developing symptoms within 11.5 days (95% CI: 8.2–15.6 days) (Lauer et al., 2020).
SARS-CoV-2, similar to SARS-CoV, has characteristic transmembrane trimeric proteins that facilitate the binding and fusion of the viral particle to the surface of the cell membrane of the host-cell. SARS-CoV-2 attaches to the angiotensin-converting enzyme 2 (ACE2) receptor that is highly expressed in lung epithelial cells, as well as in other tissues (Zou et al., 2020; Stancioiu et al., 2020). Specifically, spike protein S is responsible for the attachment to the ACE2 receptor (Hoffmann et al., 2020). Similar to SARS-CoV, protein S of the novel coronavirus employs host cell transmembrane protease serine 2 (TMPRSS2) that cleavage ACE2, promoting SARS-CoV-2 fusion with the epithelial cell (Hoffmann et al., 2020).
Common manifestations of COVID-19 are respiratory and can extend from mild symptoms to severe acute respiratory distress (Wang et al., 2020a; Wu and McGoogan, 2020; Bai et al., 2020; Farsalinos et al., 2020; Abedi et al., 2020). COVID-19 has a wide spectrum of symptoms. The majority of patients infected with COVID-19 are asymptomatic or present with mild symptoms. Anosmia, hyposmia and dysgeusia can be part of the initial presentation (Lechien et al., 2020; Vaira et al., 2020). Shortness of breath and non-productive cough accompanied by constitutional symptoms, such as fever and fatigue were initially suggested as the classic symptomatology of COVID-19 (Huang et al., 2020a). However, it has recently been demonstrated that this symptomatology is usually found in only 15% of cases (Wang et al., 2020a). Although fever has been proposed to be one of the most common symptoms present in 98% of symptomatic cases, recent data suggest that actually only 30% of COVID-19 patients present with fever at triage (Richardson et al., 2020). Other constitutional symptoms include generalized fatigue and myalgia (Wang et al., 2020a).
Zhou et al. performed a retrospective cohort study on 191 inpatients describing the stages of the disease and the associated symptoms (Zhou et al., 2020a). Mild upper respiratory symptoms, such as cough and fever are commonly present during the viral stage of the disease as the virus infects the epithelial cells of the respiratory tract (Zhou et al., 2020a). However, as the patient transitions to the later phase, a progressive worsening of shortness of breath manifests with a mean duration of 12 days (Zhou et al., 2020a). In the same study, it was noted that the average time from the onset of illness to discharge was 22 days (IQR 18·–25·) (Zhou et al., 2020a). Although specific symptoms may be closely associated with the stage of the disease, the duration of symptoms cannot be used to predict the course of the disease (Zhou et al., 2020a).
A growing body of evidence also suggests that the COVID-19 may not be limited to the respiratory tract, since the SARS-CoV-2 can invade the central nervous system (CNS), thus causing a multitude of diverse neurological manifestations as acute cerebrovascular diseases, encephalopathy, acute polyneuropathy, hypogeusia, encephalitis and hyposmia, as well as certain non-specific symptoms (such as headaches, myalgia, fatigue and unsteadiness) (Carod-Artal, 2020). Whether these features are an indirect and unspecific consequence of the pulmonary disease or of a generalized inflammatory state on the CNS, or whether they rather reflect a direct SARS-CoV-2-related neuronal damage, remains to be determined. Overall, the different level and severity of human neurotropism and neurovirulence in these patients may be explained by a combination of viral and host factors and their interaction (Wu et al., 2020a).
A number of COVID-19 associated gastrointestinal (GI) manifestations have been reported (Azwar et al., 2020; Lin et al., 2020a). ACE-2 (Li et al., 2003) receptors act as the viral entry sites for SARS-CoV-2, which are abundantly expressed in epithelial cells of the stomach, small and large intestines. SARS-CoV-2 has been shown to cause a plethora of GI symptoms, such as ageusia, nausea, vomiting, abdominal pain and diarrhea. A number of studies have attempted to estimate the prevalence of GI symptoms in patients with COVID-19; however, their estimates are widely variable (Gu et al., 2020; Lin et al., 2020b; Ng and Tilg, 2020). Jin et al., in a study on 651 patients infected with SARS-CoV-2, 11.4% described at list one GI symptoms, while from those GI symptoms as diarrhea, vomiting and nausea, without respiratory symptoms were observed in 28.38% of these patients (Jin et al., 2020). Therefore, during the current pandemic, a high index of clinical suspicion is warranted for clinicians evaluating patients presenting with GI symptoms, as these could be the sole manifestation of COVID-19.
The COVID-19 pandemic also has a strong impact on cancer patients. Indeed, it has been shown that cancer patients have a greater risk of contracting the COVID-19 infection and of developing severe respiratory syndromes due to the immunosuppression related to anticancer treatments or to the tumor itself (Liang et al., 2020; Xia et al., 2020a; Yeoh et al., 2020). Numerous studies continue to establish the safety of chemotherapy administration during the pandemic. In addition, leading medical oncology societies have issued guidelines to follow during the pandemic in order to reduce the risk of infection in a category of fragile patients as that of cancer patients (Hanna et al., 2020).
Some patients with COVID-19 have also experienced ophthalmic damage. Several reports published in scientific journals and recent news have suggested that SARS-CoV-2 may cause conjunctivitis, that can appear during hospitalization or as an early sign of infection. Thus, SARS-CoV-2 may be transmitted to the conjunctiva by aerosols or hand-eye contact. There is also evidence of the presence of SARS-CoV-2 RNA in the tears of patients with COVID-19 who have conjunctivitis. However, to date, the infectious virus has not yet been cultured in the conjunctiva of any patient with COVID-19 (Dockery et al., 2020).
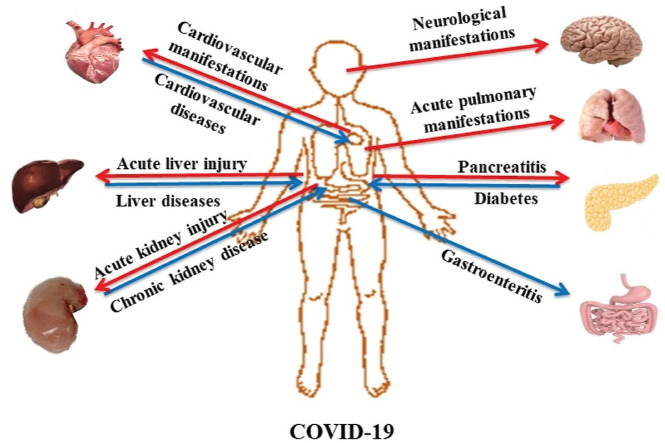
Given the multitude of clinical signs of COVID-19 disease, the present review aimed to summarize the pathophysiology and clinical signs of the disease in both previously healthy subjects and those with comorbidities, such as kidneys or neoplastic diseases. The recognition of the severity of the clinical symptoms of COVID-19 is crucial for the specific therapeutical management of affected patients.
2. COVID-19 and pulmonary manifestations
The lungs are in direct contact with the external environment and thus function as the primary host site for a number of airborne pathogens, including viruses such as coronaviruses.
The innate defenses of the lungs include physical and mechanical barriers. The cellular elements of the innate defenses, include the lung epithelial lining, pulmonary macrophages and dendritic cells (DCs) (Channappanavar et al., 2014). The first line of the cellular immune response to viral respiratory infections, such as SARS-CoV-2 is facilitated by DCs and macrophages (Channappanavar et al., 2014). The second line or adaptive immune response to coronavirus infection involves the T cell response and activation, and was previously investigated by Channappanavar R. et al., in 2014 (Channappanavar et al., 2014). Reactive oxygen species can be one of the trigger factor for these toxic responses to the virus (Nasi et al., 2020).
Recent immunological studies on adult patients with severe COVID-19 disease have provided further evidence of the immune response against SARS-CoV-2. High levels of proinflammatory cytokines, such as interleukin (IL)-6, IL-10, tumor necrosis factor-α (TNF- α), monocyte chemoattractant protein 1 (MCP1), granulocyte-colony stimulating factor (G-CSF) and macrophage inflammatory protein (MIP)1α have also been detected in the plasma of adult patients (Qin et al., 2020; Zhou et al., 2020b; Gong et al., 2020). Ulhaq et al. also described an association between IL-6 plasma levels in COVID-19 patients and their prognosis (Ulhaq and Soraya, 2020). The higher the IL-6 levels, the worse the prognosis and the severity of their clinical condition (Ulhaq and Soraya, 2020). The chemotaxis and infiltration of neutrophils into infected epithelial cells induce lung injury. Additionally, the attracted cytotoxic T cells, as a result of the activation of the adaptive immunity, can cause lung injury to the infected lung (Fang et al., 2012; Small et al., 2001). Patients with COVID-19 have been found to have a high percentage of specific inflammatory CD14+ and CD16+ monocytes, which also produce IL-6, and therefore induce the systemic inflammatory response (Nile et al., 2020). Therefore, it is clear that in severe COVID-19 disease, a massive production and release of pro-inflammatory cytokines is the crux of the “cytokine storm”. This can result in a destructive immune response and can eventually lead to acute respiratory distress syndrome (ARDS) and multi-organ dysfunction syndrome (MODS) (Nile et al., 2020).
2.1. Pulmonary symptomatology
COVID-19 pneumonia and ARDS: It has been suggested that approximately 80% of cases will present with mild or no symptoms, while 20% will have more severe courses, requiring hospitalization and approximately 5% of these requiring ICU admission (Richardson et al., 2020).
The severe course of COVID-19 is associated with pneumonia with signs of dyspnea and hypoxia developing 5–7 days after the first appearance of symptoms (Marini and Gattinoni, 2020). Gattinoni et al. described it as a viral pneumonia with near-normal pulmonary compliance, which indicates hypoxia with a compliance >50 ml/cmH2O, as a result of V/Q mismatch (Marini and Gattinoni, 2020). A lung CT scan of patients with the severe course of the disease previously revealed no areas to recruit and venous admixture levels were approximately 50%. The average duration of pneumonia associated with severe COVID-19 is 3–6 weeks (Marini and Gattinoni, 2020).
COVID-19 may progress to a more critical course, commonly due to various comorbidities, including an older age of the infected patients. If the course becomes critical, the signs of severe pneumonia and therefore, respiratory failure can lead to shock and possibly, MODS. Approximately 20–30% of patients in critical care will progress to severe hypoxemia with a <40 ml/cmH2O lung compliance consistent with ARDS (Maiolo et al., 2018).
A Chest X-ray (CXR) is routinely used as part of the standard and initial clinical assessment of lung infections. According to Wong et al., the sensitivity of the chest x-ray is estimated to be 69% (Wong et al., 2019). Vancheri et al. analyzed 240 CXRs from patients who tested positive with symptoms and found that ground-glass opacities were the most common lesion in patients with COVID-19. However, the CXR can be negative in 25% of cases (Jacobi et al., 2020).
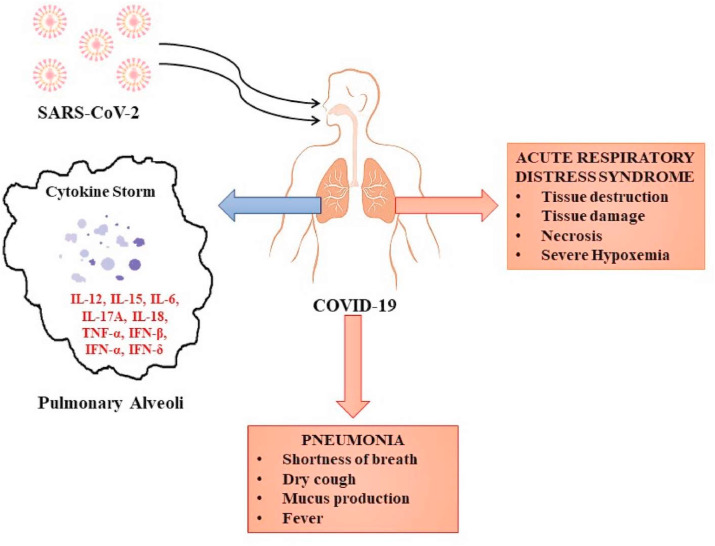
Computer tomography (CT) is another imaging method used in the diagnosis and clinical assessment of patients COVID-19. In COVID-19-related pneumonia, one can clearly identify certain typical radiographic features in the majority of patients with COVID-19, including ground-glass opacities, interstitial changes with a peripheral distribution and multifocal patchy consolidation (Chung et al., 2020; Ai et al., 2020). These typical CT findings may also be observed in negative RT-PCR-tested patients that present with clinical symptoms. A chest CT scan has been recommended in cases of initial negative RT-PCR results when the clinical picture highly suggests a COVID-19 diagnosis, since it has a high sensitivity (Xie et al., 2020; Huang et al., 2020b). Indeed, several studies have demonstrated that RT-PCR often fails in detecting SARS-CoV-2 infection due to some technical limits (Tahamtan and Ardebili, 2020); therefore, other high-sensitive molecular methods already used for the detection of small amount of mutated DNA, small RNA or viral RNA (Salemi et al., 2018; Filetti et al., 2020), like droplet digital PCR, and clinical investigations are needed in all the negative patients with COVID-19-like symptomatology (Falzone et al., 2020; Wang et al., 2020b). Other, less commonly observed CT findings, include bronchiectasis, pleural thickening and septal thinning, to name a few. Gattinoni et al. recently demonstrated that in the case of CT scan unavailability, lung compliance and the response to positive end-expiratory pressure (PEEP) are two highly recommended surrogates that may assist in the assessment of the severity of COVID-19 infection (Gattinoni et al., 2020). Fig. 1 presents the main pulmonary clinical manifestations of COVID-19.
Fig. 1.
The main pulmonary clinical manifestations of COVID-19. Note: IL-12 – interleukin-12; IL-15 - interleukin-15; IL-6- interleukin-6; IL-17A - interleukin-17A; IL-18 - interleukin-18; TNF-α – tumor necrosis factor alpha; IFN-β – interferon beta; IFN-α – interferon alpha; IFN-γ – interferon gamma.
There are multiple risk factors for poor clinical outcomes, including advanced age (OR 1.14 [1.09–1.18]), hypercoagulability (prothrombin time ≥16 OR 4.62 [1.29–16.50]), organ dysfunction (chronic obstructive lung disease OR 5.4 [0.96–20.40]; coronary heart disease OR 21.4 [4.64–98.76]), elevated D-dimer (values > 0.5 μg/mL OR 1.96 [0.52–7.43] and values > 1 μg/mL OR 20.04 [6.52–61.56]) and lactate dehydrogenase (LDH) levels (values > 245 U/L OR 45.43 [6.1–338.44]), and lymphopenia (OR 3.7 [2.44–5.63]) that are associated with patients developing ARDS and eventually succumbing to the disease (Zhou et al., 2020a; Maiolo et al., 2018; Huang and Pranata, 2020). The poor clinical outcome was defined in these studies as death. Several comorbidities, such as hypertension, obesity and diabetes, as well as laboratory parameters, such as elevated AST, ALT and serum ferritin levels, prothrombin time and other laboratory values have also been associated with ARDS and a worse prognosis of patients with COVID-19. However, the clear prognostic value of all these factors remains unclear (Zheng et al., 2020a).
3. COVID-19 and cardiovascular clinical manifestations
SARS-CoV-2 infection not only causes respiratory manifestations, but also commonly affects the cardiovascular system. Patients with cardiovascular risk factors, such as male sex, advanced age, hypertension, diabetes, obesity, and those with pre-existing cardiovascular or cerebrovascular disease are prone to developing COVID-19-associated complications, and these factors have been shown to be associated with an increased morbidity and mortality. Inpatient cardiac complications have been noted with the increased in-hospital mortality (Torequl Islam et al., 2020a).
SARS-CoV-2 mainly affects the respiratory system and specifically, the alveoli, either by directly destructing the infected epithelial cells or most importantly due to the extreme inflammatory response imitated by the host, leading to respiratory distress (Calina et al., 2020). In severe cases, the patients will eventually suffer from hypoxic respiratory failure (Docea et al., 2020). Hypoxia can directly affect the tissue oxygen demand, increasing the risk for myocardial infarction and stroke (Salehi et al., 2020a).
There are data to support the notion that the immune system of individuals with pre-existing cardiovascular conditions, such as heart failure, may function in a less effective manner in combatting COVID-19 compared to the immune system of healthy individuals, thus making them more prone to viral and bacterial infection (Sharifi-Rad et al., 2020a). In addition, infectious diseases are closely related to hemodynamic changes that can lead to the decompensation of pre-existing cardiovascular conditions, such as heart failure (Zlatian et al., 2018).
There is evidence to indicate that pre-existing cardiovascular conditions, such as heart failure, coronary artery disease and hypertension increase the risk of cardiac complications in patients suffering from COVID-19 (Bansal, 2020; Sharifi-Rad et al., 2020b). However, the association between associated heart damage, COVID-19 and mortality remains unclear.
Cardiovascular adverse events, common in COVID-19 infection, play a role in prognosis, similar to other viral infections, such as influenza (Kwong et al., 2018). The exact mechanisms of action are complex, multi-factorial and bidirectional. In addition, COVID-19 infection can trigger unique pathways for this pathogen, which contributes to the evolution of patients with cardiovascular disease. As previously discussed, SARS-CoV-2 facilitates its entry into cells by attaching to the ACE 2 receptor. ACE2 is a target for the anti-hypertensive class of drugs, known as angiotensin-converting enzyme inhibitors (ACE inhibitors). Given the high prevalence of hypertension among severe cases of COVID-19, it has been hypothesized that hypertensive patients receiving these treatments may have an increased risk of acquiring SARS-CoV-2 infection and of developing more severe manifestations of COVID 19, given the possible upregulation of ACE2.
ACE inhibitors are commonly administered to patients with diabetes, given their known micro- and macrovascular protective effects on the heart and kidneys (Jarred and Kennedy, 2010). Although initially a concern was raised regarding the use of ACE inhibitors in patients with COVID 19, given that SARS-CoV-2 attaches to the ACE2 receptor, there are no data available to date to suggest the discontinuation of the regiment or the adjustment of doses (Cordonnier et al., 2001).
The progression of atherosclerotic disease is highly related to chronic inflammation and oxidative damage (Sharifi-Rad et al., 2020c; Padureanu et al., 2019). COVID-19 is associated with the hyperactivation of the immune system and the acceleration of inflammatory cascades, promoting an optimal environment for acute atherosclerotic complications and thromboembolic phenomena (Wojciechowski et al., 2017). In addition, common cardiovascular risk factors, such as aging, diabetes, obesity and hyperlipidemia, may potentially affect immune function and in turn, may affect the susceptibility to the virus and potentially, the course of the disease (Saltiel and Olefsky, 2017).
3.1. Pathophysiology of cardiac heart damage in patients with COVID-19
Although the morphopathology and pathophysiology of cardiovascular damage induced by SARS-CoV-2 are only partially known, researchers have described 3 possible mechanisms linking cardiovascular pathology and COVID-19. The possible mechanisms include the following:
-
i)
Severe infection is associated with a severe inflammatory response that triggers a massive discharge of cytokines. As with other diseases encompassing a systemic inflammatory response, such as sepsis, these cytokines can lead to cardiomyocyte dysfunction (Fried et al., 2020).
-
ii)
SARS-CoV-2 can cause inflammation of the heart tissue, i.e., myocarditis by recruiting cells of the immune system to cardiomyocytes (Inciardi et al., 2020). Data from a recently published study suggest that the development of myocarditis is possible in the setting of COVID-19. The main cellular receptor for SARS-CoV-2 virus is ACE2, highly present in the lungs and heart tissue. It is known that the coronavirus can potentially infect the myocardium. Previous studies have demonstrated that the RNA of a SARS-CoV a virus with similar behavior to the novel coronavirus, was found in the myocardium of 35% of patients who died due to infection from the same virus (Nicin et al., 2020; Oudit et al., 2009).
-
iii)
SARS-CoV-2 infection can also affect small blood vessels through ACE2, leading to ischemic phenomena and injury to the myocardial tissue, and potential ventricular dysfunction, as well as arrhythmias (Haga et al., 2008).
Approximately 10% of hospitalized patients with COVID-19 have cardiovascular disease at the time of diagnosis of the disease; however, the number of patients who develop cardiovascular sequelae, such as acute myocardial injury, cardiomyopathy, myocarditis, congestive heart failure acute coronary syndrome, cardiogenic shock and arrhythmias varies (Driggin et al., 2020).
A recently published study demonstrated the association between heart damage and mortality in patients with COVID-19. It was demonstrated that 19.7% of hospitalized patients with COVID 19 suffered from cardiac injury. In that same study, cardiac injury was also associated with an increased risk of mortality during hospitalization (Shi et al., 2020).
A recently published study with 138 patients critically ill with COVID-19, revealed that 25% of these patients had a history of coronary heart disease and 58.3% of them had hypertension (Wang et al., 2020a).
Another study reported that 12% of patients with COVID-19 had acute heart disease associated with the viral infection, manifested by a decreased ejection fraction and increased troponin I levels (Huang et al., 2020a).
Another study reported that 7.2% of the 138 patients hospitalized with COVID-19 had acute heart damage. Patients admitted to the intensive care unit more often exhibited heart damage (22.2%) compared to the other patients. That study retrospectively analyzed data collected at a single center in Wuhan, China, to examine the possible association between heart injury and mortality in patients with COVID-19. The study included a cohort of 416 patients confirmed with COVID-19, of whom 82 patients (19.7%) had heart damage. Compared to the patients without heart damage, these patients were older (74 vs. 60 years; the average age of all the patients was 64 years) and with greater frequency of comorbidities (Paramasivam et al., 2020).
Several smaller cohort studies have revealed similar results, suggesting a higher risk of adverse events in these patients, although there were differences in testing and data standardization. Although reports outside China are limited, data from Italy suggest similar mortality rates and an increased risk of death in patients with comorbidities (Porcheddu et al., 2020). These data suggest that cardiac impairment may be associated with the clinical course of COVID-19 (Wang et al., 2020a).
Although there is limited evidence available for the establishment of a direct link between acute heart damage associated with COVID-19 and cardiovascular comorbidities, patients with coronary heart disease and heart failure are likely to be more susceptible to acute heart damage (Guzik et al., 2020). Once these patients are infected and develop severe pneumonia, myocardial ischemia or heart failure is more likely to occur, eventually leading to devastating complications. Concomitantly, an intense inflammatory response overlapping with pre-existing cardiovascular disease may precipitate heart damage in patients with SARS-CoV-2 infections (Gupta et al., 2020).
3.2. Cardiovascular manifestations in COVID-19 disease
Arrhythmias are a risk factor in COVID-19 infection, with implications for survival. Palpitations are one of the most common symptoms of onset. Atrial fibrillation does not increase the risk of SARS-CoV-2 infection. However, a large number of patients with atrial fibrillation are elderly and this is associated with other pathologies, which can lead to a more severe evolution of the infection. Atrial fibrillation may be triggered by COVID-19 infection through associated myocardial injury. Certain antiviral drugs can also cause arrhythmias (Guzik et al., 2020).
Even patients without cardiovascular disease may develop heart dysfunction during SARS-CoV-2 infection. In a number of infected patients, the levels of biomarkers of myocardial injury may increase. These levels can increase due to non-ischemic causes, such as myocarditis or ischemic causes, such as myocardial infarction caused by the rupture of atheroma plaque (Guzik et al., 2020).
COVID-19 disease is strongly associated with thrombosis: Thrombosis in large vessels, deep vein thrombosis, pulmonary embolism, arterial events and possible microvascular thrombosis may occur (Klok et al., 2020a). It has also recently been suggested that the use of systemic anticoagulation is associated with the survival of hospitalized patients with COVID- 19 (Paranjpe et al., 2020).
There are currently limited data available on the occurrence of arrhythmias in SARS-CoV-2 infection. Severe forms of COVID-19 at the setting of myocarditis can be complicated by cardiac arrhythmias. During infection, elevated levels of biomarkers of myocardial injury can be observed, and are probably associated with myocarditis and myocardial ischemia.
In a study based on a group of 137 patients in Hubei Province, 7.3% of patients had palpitations as their first symptom (Liu et al., 2020a). Another study, performed on 138 patients with COVID-19 in Wuhan, demonstrated that arrhythmias were present in 16.7% of patients, and in 44% of those admitted to intensive care units (Wang et al., 2020a). In both studies, however, the specific causes of palpitations and the type of arrhythmia were not identified. In another study from the same institution, ventricular tachycardia and ventricular fibrillation were reported as complications of COVID-19 in 11 of 187 patients (5.9%), with a higher incidence in patients with elevated troponin T levels (Guo et al., 2020a).
However, one of the largest studies in China, based on 1099 patients in 552 hospitals, did not report arrhythmias in patients with COVID-19 (Guan et al., 2020a). By contrast, among 393 hospitalized patients in New York, USA, atrial arrhythmias were more common in patients who required mechanical ventilation (17.6 vs. 1.8%) (Goyal et al., 2020). The possible mechanisms of cardiac arrhythmia in COVID-19 include acute myocardial injury, and currently used therapies are considered the main mechanisms in increasing the risk of arrhythmias in patients with COVID-19 (Babapoor-Farrokhran et al., 2020).
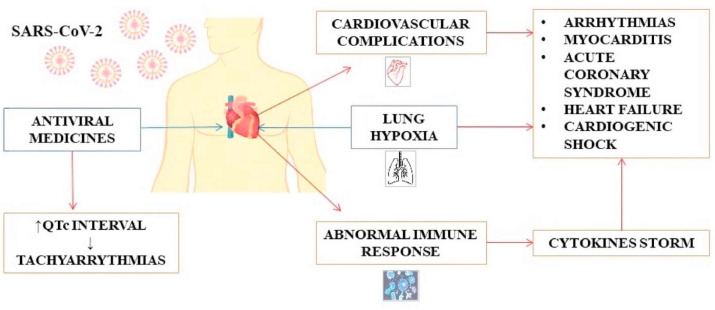
Myocardial injury can be the direct result of the cardiac invasion by the virus, but also an indirect consequence of major lung damage (damage to heart cells caused by hypoxia). In addition, certain medications that were initially recommended for SARS-CoV-2 infection (until therapy is approved off-label), such as antimalarials (hydroxychloroquine), protease inhibitors (lopinavir/ritonavir) and macrolides (azithromycin), may prolong the QT interval and increase the risk of a particularly dangerous ventricular arrhythmia, known as torsade de points (Cunningham et al., 2020) (Fig. 2 ).
Fig. 2.
SARS-CoV-2 infection and its association with the cardiovascular system.
In this context, the immune response, the abnormal systemic inflammatory response of the body, with the role of counteracting infection [cytokine storm (CS)], can further amplify the risk of arrhythmic phenomena, including malignant ventricular arrhythmias. In addition to their role in myocardial injury, certain inflammatory cytokines (particularly IL-6) may promote QT prolongation, both directly by modulating myocyte ion channels and indirectly by increasing the availability of molecules that increase the range of QT (by inhibiting CYP450-3A4). In addition, as a result of the direct stimulation of the vegetative nervous system, pro-inflammatory cytokines can induce the hyperactivation of the sympathetic nervous system, with an increase in cardiac electrical instability (Babapoor-Farrokhran et al., 2020).
Elderly patients hospitalized due to COVID-19 infection, who present with a combination of pneumonia, SDR and sepsis, frequently develop de novo or recurrent fibroadenoma (FiA), which may further complicate the course of the disease. Precipitating factors, in this case, are hypokalemia and hypomagnesemia (induced nausea, anorexia, diarrhea and diuretic medication), metabolic acidosis, use of inotropic agents, ventilation-induced cardiac asynchrony, volume overload, increased sympathetic tone, inflammation, hypoxia, ischemia and myocardial injury (Kwenandar et al., 2020).
In addition, a number of patients with FiA have associated cardiovascular damage, increasing the risk of more severe infection from the start. In addition, all patients with associated heart disease appear to have a weaker immune system, thus compromising their bodies’ efforts to fight the virus (Boriani et al., 2018).
4. COVID 19 and gastrointestinal and liver manifestations
4.1. Pathophysiology of gastrointestinal disorders in patients with COVID-19
The reason why only a certain proportion of patients with COVID-19 develop GI symptoms, while others do not, remains largely unknown, and whether there are underlying pathophysiological mechanisms contributing to the development of gastroenteritis remains to be determined. A possible explanation may be related to the high expression of the ACE2 receptor in GI epithelial cells (Hamming et al., 2004), and the extensive infection of enterocytes, leading to malabsorption and diarrhea.
The underlying mechanisms of acute hepatic injury associated with COVID-19 have not yet been determined. The involvement of the abundant expression of ACE-2 receptors in biliary system cells (cholangiocytes) has been suggested (Hamming et al., 2004). However, it is noteworthy that elevated levels of alkaline phosphatase and gamma-glutamyl transferase (GGT) are found in a small number of patients compared to elevated levels of aminotransferases (Zhang et al., 2020a).
One of the most significant complications associated with COVID-19 is the cytokine storm, namely, the hyperactivation of the immune system, leading to a profound inflammatory response. This leads to a significant increase in the levels of IL-6, IL -10, IL-2 and IFN-γ that can potentially induce tissue injury (Zhang et al., 2020a). Recent data suggest that higher levels of ILs are associated with more severe disease and liver failure, suggesting the underlying mechanism of the cytokine storm in these patients l (Liu et al., 2020b). Furthermore, lymphopenia and elevated C-reactive protein (CRP) levels in patients with COVID-19 have also been associated with the cytokine storm and liver injury, leading to a generally poorer prognosis (Li et al., 2020).
It also remains unclear whether pre-existing liver diseases, such as viral hepatitis and non-alcoholic fatty liver disease are associated with a prolonged disease course or more severe COVID-19 infection (Boettler et al., 2020). There are data to suggest that patients with pre-existing viral hepatitis are more prone to severe liver injury, possibly due to the SARS-CoV-2-related enhancement of viral replication (Chau et al., 2004); however, extensive studies are required to clarify this phenomenon.
The role of NF-κB inhibition in SARS-CoV-2 has also been demonstrated. It has been shown that COVID-19 activates the NF-κB pathway (Guo et al., 2020b). Previous studies have also supported the use of NF-κB inhibitors as promising antivirals in infection caused by SARS-CoV and potentially other pathogenic human coronaviruses (DeDiego et al., 2014). On the other hand, in COVID-19 pandemic, the blockade of IL-6 has been proposed to manage the COVID-19 cytokine storm (Liu et al., 2020c). The use of corticosteroids has also been suggested as a possible therapeutic agent for COVID 19-related lung and acute liver injury (Russell et al., 2020).
Therefore, targeting the NF-κB signaling pathway represents an attractive approach for anti-inflammatory and cancer therapies (Xia et al., 2014; Mititelu et al., 2020). However, due to its central role in maintaining normal cellular functions, the components of the NF-κB pathway need to be carefully selected and evaluated for the design of optimally targeted therapies (Padureanu et al., 2019; Rogoveanu et al., 2018). Therefore, the exploration of NF-κB and IL-6 inhibitors in the treatment of patients with COVID-19, synergistically with antiviral therapy may be feasible options (Stancioiu et al., 2020; Nitulescu et al., 2020; Dehelean et al., 2020; Torequl Islam et al., 2020b; Sarkar et al., 2020). The role of NF-κB in inflammation-associated GI malignancies has been previously documented (Doukas et al., 2018; Gambhir et al., 2015; Vageli et al., 2018; Zhang and Rigas, 2006). In particular, the NF-κB/IL6 pathway links inflammatory disease and GI cancer (De Simone et al., 2015; Yang et al., 2014; Zlatian et al., 2015). Nevertheless, further data are necessary to clarify the significance of NF-κB inhibition in the management of patients with COVID 19, and further studies are required to evaluate the effectiveness of this approach.
4.2. Gastrointestinal manifestations in patients with COVID-19
Gastroenteritis: RNA of SARS-CoV- 2 has been detected in the stool samples of the majority of patients with COVID-19 infection several days after the respiratory tests had yielded negative results. The fecal-oral transmission of SARS-CoV-2 has been reported; therefore, consistent handwashing and the use of separate bathrooms, if possible, for infected patients has been suggested to limit the spread (Gu et al., 2020; Hindson, 2020).
Stool samples isolated from patients with COIVD 19, with or without GI symptoms, have often been found to be positive for SARS-CoV-2 (Leung et al., 2003). Although studies have indicated that viral RNA is detected in stool samples days after the resolution of symptoms (Leung et al., 2003; Wu et al., 2020b; Wei et al., 2020), it is not yet clear whether these patients remain infective. It has been demonstrated that patients with GI symptoms have a prolonged COVID-19-related illness compared to those without GI symptoms (Wei et al., 2020).
However, it remains unknown whether the presence of GI symptoms is associated with a worse prognosis and/or inflammatory storm. A previous epidemiological study demonstrated that patients with diarrhea typically had higher levels of C-reactive protein (CRP) and lactate dehydrogenase (LDH) (Wei et al., 2020). However, a number of aspects of COVID-19 particularly in relation to GI symptoms are currently being investigated, and future studies may provide further insight into the risk factors, disease severity and asymptomatic spread of SARS-CoV-2 in patients with concomitant GI manifestations.
As regards the liver manifestation of COVID-19, elevations of liver enzyme levels have been reported to vary degrees in patients with COVID-19. Studies have demonstrated that the elevation of liver enzyme levels can be observed in up to 78% of cases (Zhang et al., 2020a; Musa, 2020; Xu et al., 2020a). In the majority of cases, the liver involvement of COVID-19 is generally asymptomatic with the modest elevation of aminotransferase levels. Elevated levels of bilirubin have also been reported in some cases (Musa, 2020; Xu et al., 2020a). The elevation of aminotransferase levels has been shown to be associated with the increased severity of COVID-19 and a potentially poorer prognosis (Fan et al., 2020; Cai et al., 2020).
Several studies have demonstrated the findings of microvesicular steatosis, and mild portal and lobular activity that may be attributed either to viral injury or drug-induced injury on pathological specimens (Tian et al., 2020; Lagana et al., 2020; Li and Xiao, 2020). Additionally, molecular analysis detected SARS-CoV-2 in the hepatocytes of infected patients.
As regards COVID-19 and pancreatitis, a recent single center study in China demonstrated that 17% of patients with COVID-19-associated pneumonia had evidence of pancreatic injury with elevated amylase and lipase levels. However, it is unclear whether any of those patients had acute pancreatitis, as no imaging studies were performed in that patient cohort. While the true prevalence of acute pancreatitis remains unknown in patients with COVID-19, ACE-2 receptors were found in pancreatic islet cells, suggesting that pancreatic infection is possible (Wang et al., 2020c).
5. COVID-19 and neurological clinical symptoms
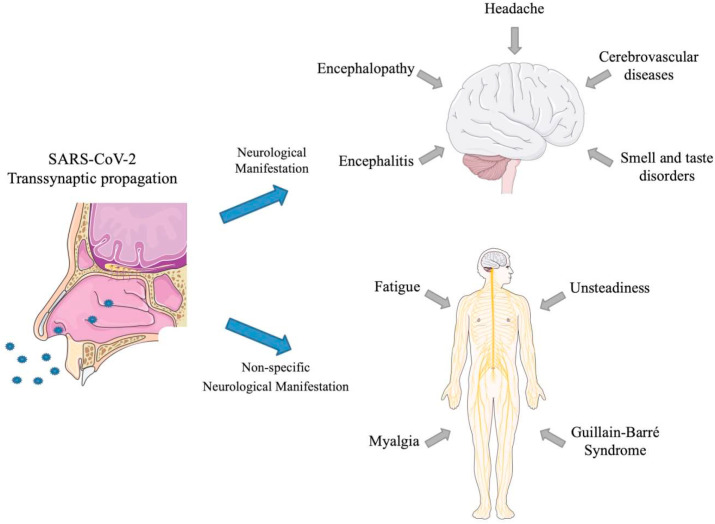
Various experimental and clinical studies have suggested that seven types of coronavirus can affect brain and other central nerve structure (CNS) (Carod-Artal, 2020). In the majority of animal and human coronaviruses, a great documentation of their neuroinvasion potential exists (Tsai et al., 2005). Recent epidemiological data has demonstrated that some COVID-19 patients developed neurological symptoms including psychosis, sometimes difficult to differentiate from other type of psychosis (Nussbaum et al., 2017; Brown et al., 2020). However, clinical features, pathogenic mechanisms and therapeutic options have yet to be fully characterized. Preliminary studies have indicated that COVID-19 is associated with a plethora of neurological symptoms (hypogeusia, hyposmia acute polyneuropathy, headaches, etc.) and CNS-related disorders, including encephalopathy, encephalitis, acute cerebrovascular diseases, etc. (Pennisi et al., 2020) (Fig. 3 ).
Fig. 3.
COVID-19 neurological manifestations.
5.1. SARS-CoV-2 neurological routes of propagation and neurological pathogenesis
5.1.1. Hematogenous vs. transsynaptic propagation
SARS-CoV-2 is able to directly invade the CNS at the beginning of viral infection or in the late phases (Baig et al., 2020), although the mechanisms responsible for this development are not yet well understood. There are evidence that some coronaviruses are cappable to invade the CNS by penetrating the anatomical structures of ethmoid affecting the olfactory bulb ability to control viral invasion and supporting a retrograde transsynaptic propagation (Li et al., 2020b). Other retrograde route of propagation may be mediated by SARS-CoV-2 invasion of sensorial receptors located in lung and airways (Li et al., 2020b).
In contrast, other researchers doen't support the neurogenic hypothesis of respiratory failure (Turtle, 2020) because the observed increased levels of CO2 and the consequent hypoxia are observed also in patients with COVID-19 pneumonia without neurological symptoms. Compared with patients suffering from respiratory failure of neurological origin that presents reduced respiratory rate, high CO2 levels and low oxygen levels, these patients do not need artificial ventilation although they manifest weak respiratory difficulties (Turtle, 2020). Further studies are warranted to demonstrate a neurotropism to the respiratory control centers after COVID-19 infection.
5.1.2. ACE2-mediated neuroinvasion
It has been widely demonstrated that SARS-CoV-2 invasion and replication are facilitated by the presence of ACE2 cell surface receptor (Yan et al., 2020). The depletion of ACE2 receptor mediated by SARS-CoV-2 increase the levels of angiotensin II thus increasing the worsening of respiratory functions. Accordingly, the ACE2 receptor downregulation and the consequent angiotensin II increasing, worsening also the health status of COVID-19 patients with hypertension and diabetes (Zhou et al., 2020a). Yan et al. hypothese that the administration of ACE inhibitors in COVID-19 patients can up-regulate ACE2 expression increasing the vulnerability of the cells to virus penetration (Yan et al., 2020).
It is speculated that the expression levels of the ACE2 receptor in the nervous system is associatd with SARS-CoV2 neurovirulence, although the ACE2 receptor is expressed in endothelial cells. Further studies aiming to explore the molecular basis of neurological complications after COVID-19 infection, such as stroke, are needed (Carod-Artal, 2020). It has been shown that the interactions existing between ACE2 receptors located in brain endotial cells with viral particle are mediated by S spike protein that permit the infection of the endothelial cells and disemination in the neurons after endothelial damage (Baig et al., 2020).
5.1.3. Other mechanisms
Indirect neuronal damage is produced mainly by COVID-19-induced hypoxia. Hypoxia is responsible for the accumulation of toxic metabolites due to anaerobic metabolic processes occurring in neuronal cells leading to vasodilation, ischemia, cerebral edema, etc., with severe consequence for the patients (Wu et al., 2020a; Tsatsakis et al., 2019; Trejo-Gabriel-Galán, 2020).
Due to the capability of SARS-CoV-2 to invade immune cells, the immune-mediated response could play a fundamental role in COVID-19 neurological manifestations. Supporting this notion, patients who died because of COVID-19 infection manifested cytokine storm syndrome and multiple organ failure (Mehta et al., 2020). In addition, in vitro experiments have indicated that glial cells can secrete inflammatory factors, including IL-6, 12, 15 and TNF-α following infection with coronavirus (Bohmwald et al., 2018).
Finally, it was also speculated that the persistence of COVID-19 infection could be due to the permanence of SARS-CoV-2 withi neuronal cells where the virus aggravates the neurological symptoms. Supporting this notion, in multiple sclerosis, Parkinson's disease and optic neuritis patients several coronaviruses have been identified (Dessau et al., 1999; Salmi et al., 1982; Fazzini et al., 1992; Fleming et al., 1982). Therefore, a persistent coronavirus infection may lead to the onset of neurological diseases, as in some susceptible patients, the immune system may exacerbate the disease (Desforges et al., 2019).
5.2. Main neurological manifestations of COVID-19
5.2.1. Encephalopathy
It was demonstrated that COVID-19 infection has severe health consequence for elderly patients with previous neurological disorders, co-morbidities, or cardiovascular diseases (Mao et al., 2020a; Filatov et al., 2020). In addition, one of the first COVID-19 sympthoms may be represented by encephalopathy resulting from the SARS-CoV-2 infection itself or as a consequence of antiviral treatments, hypoxia and virus-related symptoms (Mao et al., 2020a).
Toxic and metabolic causes, as well as the effects of drugs, hypoxia, or subclinical crises, may be the result of COVID-19-associated encephalopathy (Guan et al., 2020a). In a previous study, an electroencephalogram (EEG) revealed a diffuse slow wave in the bilateral temporal region (Filatov et al., 2020), whereas signs of edema without inflammatory and neuronal degeneration are the pathological findings in autopsies of deceased patients with COVID-19-related encephalopathy (Xu et al., 2020b).
Poyiadji N and colleagues described a patient with acute hemorrhagic necrotizing encephalopathy diagnosed by detecting SARS-CoV-2 by RT-PCR in a nasopharyngeal sample (Poyiadji et al., 2020a). A brain CT scan showed thalamic bilateral hypodense area, while the clinical features after MRI included multifocal and symmetrical disposition in different brain areas (thalami, insula and temporal lobes) (Poyiadji et al., 2020a). The cytokine storm has been postulated to play a relevant role in the pathogenesis and clinical manifestations of the disease (Mehta et al., 2020; Buga et al., 2019).
5.2.2. Encephalitis
Encephalitis has been associated with some viral infections. On this bases some researchers have proposed the evaluation of SARS-COV-2 in patients with encephalitis in order to early diagnose this neurological complication and increase the survival rate of patients (Carod-Artal, 2020). Xiang and colleagues have diagnosed SARS-CoV-2 encephalitis in a 56-year-old woman from Wuhan without any clinical evidence of encephalitis observed with CT scan. Indeed, the diagnosis was formulated after the detection of SARS-CoV-2 on cerebrospinal fluid sample by NGS (Xiang et al., 2020).
Another case report of Moriguci showed that in a young male the detection of SARS-CoV-2 failed in the nasopharyngeal swab while positive signals were obtained from CSF (Moriguchi et al., 2020). This patient presented also lower consciousness suggesting a possible meningoencephalitis, however, in this case, MRI revealed abnormalities in the hippocampus and in the medial region of the right temporal lobe (Moriguchi et al., 2020).
5.2.3. Cerebrovascular diseases
Cerebrovascular diseases can be present either during the early or late stages in the course of COVID-19. Moreover, cerebrovascular diseases can be present in either patient with known risk factors for cerebrovascular diseases or in patients with COVID-19 with a negative medical history. It has been demonstrated that when compared to younger subjects without co-morbidities and with a high mortality rate, elderly COVID-19 patients with cardiovascular disease seem to have an increased risk of neurological symptoms (Carod-Artal, 2020). In a study of more tha 200 COVID-19 patients (Li et al., 2020c), the ischemic stroke was reposted in 11 patients, cerebral venous thrombosis in one patients and cerebral hemorrhage in 1 patient. Ealderly patients (mean age,71.6 years), pulmonary impairments, diabetes, cardiovascular disease, pro-inflammatory status, alteration of the coagulant cascade and previous cerebrovascular events were the identified risk factors for stroke (Li et al., 2020c). Another cohort of patients showed that in four patients with ischemic stroke and in one patient with hemorragic, associated with acute COVID-19 infection, multiple organ failure, high D-dimer levels and thrombocytopenia (Mao et al., 2020b). The incidence of ischemic stroke has been determined to be lower in other studies (1.6 and 2.5% in the study by Klok et al. and Lodigiani et al. respectively) (Klok et al., 2020b; Lodigiani et al., 2020). Notably, large-vessel stroke has also been reported to be present in younger-aged patients with Covid-19 (<50 years) (Oxley et al., 2020). Oxley et al. described 5 patients with stroke with Covid-19 (aged 33–49 years), with 2 of them (aged 33 and 37 years) without a medical history and risk factors for stroke (Oxley et al., 2020). Acute necrotizing encephalopathy (ANE), has been reported in patients with severe COVID-19 infection, possibly as a result of the cytokine storm syndrome (Poyiadji et al., 2020b).
Pathomechanisms have indicated that SARS-CoV-2 is able to recognize ACE2 receptors on vascular endothelial cells leading to high blood pressure that in turn predispose to an increased risk of ischemic and hemorrhagic stroke (Carod-Artal, 2020).
5.2.4. Guillain-Barre syndrome (GBS)
The neurological complications of COVID-19 infection comprise also GBS diagnosed in a COVID-19 patients of 62-year-old patient manifesting muscular weakness in the lower limbs (Zhao et al., 2020a). An increase of protein levels and the lack of cells (124 mg/dl) was evidenced in the CSF together with the absecence of F-waves and distal latency. Despite these observations, the onset of the GBS symptoms could be not related to SARS-CoV-2 infection but probably occurring concomitantly, thus, further studies are warranted on this matter (Zhao et al., 2020a).
A few cases GBS related to COVID 19 infection have been reported to date (Alberti et al., 2020; Toscano et al., 2020; Camdessanche et al., 2020; Virani et al., 2020; Zhao et al., 2020b; Sedaghat and Karimi, 2020; Juliao Caamaño and Alonso Beato, 2020). Taking into consideration that only a few GBS cases related to other coronaviruses have been reported, it can be hypothesized that GBS associated with SARS-COV-2 is not an uncommon manifestation, and physicians may perform a test for SARS-COV-2 in patients with GBS (Zhao et al., 2020b). Therefore, larger epidemiological studies are required to establish a clear causal evidence of the association between SARS-COV-2 and GBS (Zhao et al., 2020b).
Recently, two cases (a 36-year-old male and 71-year-old female) with SARS-CoV-2 infection developed cranial neuropathies a few days following the onset of respiratory symptoms. The symptoms in the above-mentioned patients may have been unrelated to SARS-CoV-2 infection; however, it should be noted that cranial neuropathies may be a rare clinical presentation of SARS-CoV-2 (Dinkin et al., 2020).
5.2.5. Epilepsy
Potential pathophysiological processes through which COVID-19 could lead to seizures are hypoxia, brain damage, polyorganic failure and organ metabolic derangements. While the reported cases of seizures are limited (Mao et al., 2020b), this number may increase, following the paradigm of epilepsy-related to other coronaviruses (Hung et al., 2003; Lau et al., 2004).
5.2.6. Smell and taste disorders
Even with the absence of nasal symptoms, research indicates that olfactory and taste disorders are quite prevalent in COVID-19 patients (Giacomelli et al., 2020; Skalny et al., 2020). In a case registry of 12 European hospitals on 417 patients with mild-to-moderate COVID-19, 85.6 and 88% reported disorders of smell and taste, respectively (Lechien et al., 2020b). Notably, 12% complained of smell dysfunction also in absence of nasal obstruction or other nasal symptoms (Lechien et al., 2020b).
5.2.7. Nonspecific neurological symptoms
In a Wuhan hospital study, 36.4% of 214 patients with COVID-19 exhibited some form of neurological manifestation (Mao et al., 2020b), which was categorized as CNS involvement (24.8%), peripheral (10.7%) and musculoskeletal (10.7%). The most common symptoms were dizziness (36 patients), headaches (28), hypogeusia (12) and hyposmia (5), more frequently observed in severe than in mild patients (45.5% vs. 30%).
In a previous study, the majority of Chinese patients with COVID-19 reported having headaches (13.6%), whereby 15% of these were severe (Guan et al., 2020b), although that study did not encompass a previous history of headaches or other brain signs. Among other symptoms, myalgia was observed in 15% of patients without severe. Creatine kinase levels, were high in 13.7% of pateints (Benny and Khadilkar, 2020). Finally, two cases of rhabdomyolysis (0.2%) were also reported (Jin and Tong, 2020).
6. COVID-19 and diabetes
6.1. Possible mechanisms through which diabetes complicates the evolution of COVID-19
Although the mechanisms responsible for this increase in susceptibility are not yet known, studies have suggested that elevated blood glucose levels lead to immunosuppression (Salehi et al., 2020b). In general, diabetes is associated with other chronic diseases, such as cardiovascular diseases and/or obesity, which further increases the risk of infection. Elevated blood glucose levels are potentially associated with more severe manifestations of infectious diseases.
Research on infections associated with diabetes describes hyperglycemia as an initiator of immunosuppression (Cristelo et al., 2020).
Most often, diabetes is associated with severe cardiovascular disease, kidney disease, aging and these factors contribute to the unfavorable course of infections. The association of diabetes with the novel coronavirus is complex and it is not known exactly whether hyperglycemia contributes to the virulence of the new coronavirus or whether it alters carbohydrate metabolism. Often, the metabolic imbalances produced by this disease are multiple and lead to a proinflammatory and prooxidative state in the body, which can decrease the resistance to infection with the novel coronavirus. (Cristelo et al., 2020).
6.2. The impact of diabetes on the evolution of SARS-CoV-2 infection
The risk of infection in diabetic patients depends on a number of factors: glycemic control, the duration of the disease, associated comorbidities (heart disease, kidney damage, cerebrovascular disease), as well as age, body weight and tobacco use (Sharifi-Rad et al., 2020a, 2020c; Petrakis et al., 2017).
An analysis of 72,314 cases of COVID-19 in Wuhan, the epicenter of the epidemic, led to the observation that the disease was associated with a mortality rate of 7.3% (Wu and McGoogan 2020) among diabetic patients. Studies using smaller cohorts have led to similar conclusions: The incidence of diabetes in a group of 26 patients that succumbed to COVID 19 infection was 42.3% (Deng and Peng, 2020). In a follow-up study on 191 patients, diabetes was confirmed to increase the risk of lethal evolution of COVID-19 by 2.85-fold (Singh et al., 2020).
7. COVID-19 and kidney diseases
The kidneys are frequently affected in symptomatic patients suffering from COVID 19. Proteinuria is a common finding in these patients and acute renal failure (ARF) often occurs at the onset or later during the course of the disease.
7.1. Main mechanisms of renal impairment in COVID-19
-
i)
The novel coronavirus can directly affect the kidneys. This is supported by the detection of PCR fragments of coronavirus in the blood and urine of patients with COVID-19. SARS-CoV-2 uses ACE2 as a cellular penetration receptor (Zhang et al., 2020b). It is known that ACE2 expression is almost 100-fold higher in the kidneys than in the lungs (Jia et al., 2005). Thus, it is possible that SARS-CoV-2, apart from infecting lung epithelial cells, can also potentially infect renal tubular epithelial cells. A previous histological analysis of renal tissues from autopsies of patients with COIVD-19 revealed acute renal tubular lesions in 6 cases of COVID-19 (Jia et al., 2005). Therefore, renal failure can be caused due to the direct infection and destruction of ACE2-presenting kidney cells.
-
ii)
Renal dysfunction can accelerate the progression of inflammation commencing in the lungs, not only as a side-effect of lung inflammation. On the one hand, the inflammatory reaction following lung damage can damage the kidneys; the damage to and death of renal tubular epithelial cells can also cause severe damage to the lungs and other organs through a large amount of inflammatory substances. Above a certain critical level, the kidney-lung association can lead to a storm of cytokines, irreversible and self-amplifying, which rapidly leads to multiple organ failure and death.
Renal dysfunction may be the result of an extensive immune reaction, also known as the “cytokine storm”. This often occurs at the late stages of inflammation and presents with multi-organ injury, including injury to the lungs, liver and kidneys (Jia et al., 2005). Although the exact mechanisms responsible for this are not yet well understood, possibly the enormous release of cytokines induces inflammatory changes in multiple tissues, including the kidneys, leading to acute kidney injury (Jia et al., 2005).
-
iii)
The kidneys can be affected by the deposition of viral antigen immune complexes or by specific virus-induced immune mechanisms (specific T lymphocytes or antibodies). However, in patients with SARS, a normal glomerular histology without electrodense deposits was observed (Chu et al., 2005). In theory, considering that SARS-CoV-2 behaves similarly to the previous SARS coronavirus (SARS-CoV), the possibility of immune-mediated glomerulonephritis is less likely. Further renal histologic evaluation is required to clarify this point of view.
-
iv)
Severe sepsis due to COVID-19 can lead to hypoxia, shock and rhabdomyolysis, leading to acute kidney failure. It has been demonstrated that the elevation of creatinine levels is a common finding in patients with COVID-19 (Dallan et al., 2020).
A recent study found that kidney dysfunction in patients with COVID-19 was generally mild, but more severe compared to other pneumonias. Patients with a severe presentation of COVID-19 infection can rapidly develop kidney dysfunction, which then progresses to acute renal failure, which has been associated with a worse prognosis, and high morbidity and mortality (Guan et al., 2020c).
In a previous study on 193 patients with COVID-19, with a mean age of 57 years, 54% had chronic kidney disease. Patients with severe COVID-19 (34%) were significantly older (median age, 66 vs. 55 years) and were more likely to have other comorbidities, such as respiratory disease (26 vs. 8%). A number of patients had complications, including ARDS (28%), IRA (28%), septic shock (18%) and acute heart damage (12%), particularly in patients with severe COVID-19 infection (Guan et al., 2020c).
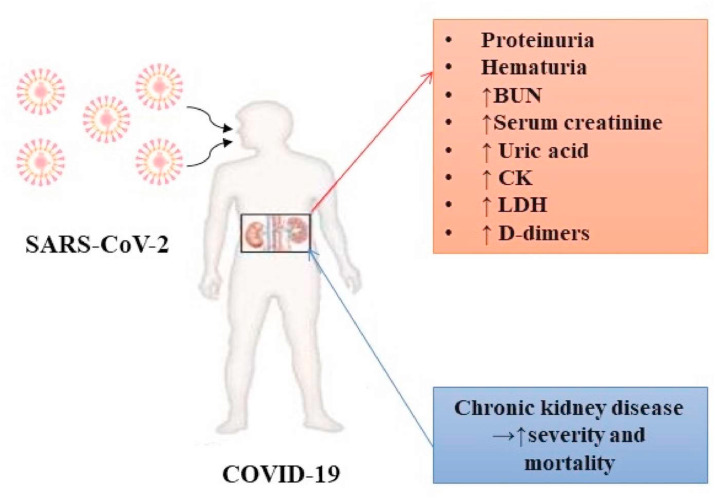
As regards renal impairment since admission, a previous study revealed elevated levels of blood urea nitrogen (BUN; urea nitrogen in the blood) and serum creatinine levels in 14 and 10% of infected patients, respectively, suggesting the presence of renal dysfunction before or during hospitalization. In addition, elevated levels of uric acid, creatinine kinase (CK) and LDH in some patients with COVID-19 infection were observed. Proteinuria was also detected in 59% of patients and hematuria in 44% (Guan et al., 2020c).
7.2. Clinical signs of renal disorders in patients with COVID-19
The high prevalence of renal failure during hospitalization can be explained by the fact that a number of patients with COVID-19 could not be hospitalized in the early stages of the disease due to a large number of patients and the limited number of beds available at the Wuhan hospital where the study was conducted.
In addition, some patients with COVID-19 may have a history of chronic kidney disease, with a pro-inflammatory status, and innate and adaptive immune defects. Moreover, these patients have a higher risk of upper respiratory tract infections and pneumonia. Proteinuria was detected in 60% of patients during hospitalization. Data analysis revealed no association between the presence of proteinuria and the severity of COVID-19. Hematuria was present in 48% of patients and its severity was associated with the severity of the disease. Proteinuria and hematuria were detected from the early days of hospitalization, suggesting that there was a potential time window to intervene and protect renal function. In total, 31% of patients with COVID-19 had a high level of BUN. By contrast, only one patient (4%) with other causes of pneumonia had elevated BUN levels and 22% of patients with COVID-19 had elevated serum creatinine (SCr) levels (Guan et al., 2020c). The duration from admission to the increase in SCr levels was between 0 and 20 days (average of 5 days). Severe cases (including deaths) had significantly higher levels of SCr during the course of the disease, suggesting that the increase in SCr levels was a potential marker of a poor prognosis of patients infected with COVID-19. By contrast, in other cases of pneumonia, there were no patients with high SCr levels (Guan et al., 2020c).
A total of 20% of patients with COVID-19 had elevated levels of uric acid, and 70% had elevated levels of D-dimers (Guan et al., 2020c). By contrast, only 11% of the other patients with other causes of pneumonia had elevated levels of uric acid, and 63% had elevated levels of D-dimers. The duration from the admission to the increase in the levels of uric acid and D-dimers was between 0 and 20 days (average of 7 days), and between 0 and 22 days (0 days on average), respectively. Severe cases (including deaths) had significantly higher levels of uric acid and D-dimers (Guan et al., 2020c). This was consistent with the finding of recent reports suggesting that high levels of D-dimers may indicate a poor prognosis of patients with COVID-19 (Guan et al., 2020c).
In another study, approximately 96% of patients had radiographic abnormalities of the kidneys. Inflammation and edema of the renal parenchyma are frequently observed in patients with COVID-19. The analysis of kidney CT images revealed that the group of patients with COVID-19 exhibited significant differences compared to the group of patients with other pneumonias, and to healthy patients (Chen et al., 2020a). The main renal clinical manifestations of COVID-19 are summarized in Fig. 4 .
Fig. 4.
The most prominent renal clinical manifestations in COVID-19. Note:BUN – blood urea nitrogen; CK- creatine kinase; LDH – lactate dehydrogenase.
8. New manifestations on the official list of clinical symptoms suggestive of COVID-19
Initial symptoms on the CDC list of characteristic manifestations of COVID-19 include fever, cough and difficulty breathing. Recently, additional symptoms have been added to the clinical presentation of the disease, such as a sore throat, headaches, muscle aches, chills, tremors and loss of taste or smell (C, 2020).
8.1. Ophthalmic damage
Recent studies have suggested that SARS-CoV-2 may cause conjunctivitis, either as an early sign of infection or during hospitalization. SARS-CoV-2 may be transmitted to the conjunctiva by aerosols or direct hand-eye contact (Xia et al., 2020b). There is also evidence of the presence of SARS-CoV-2 RNA in the tears of patients with COVID-19 associated with conjunctivitis. However, to date, the virus has not yet been cultured form the conjunctiva of any patient with COVID-19. Recent data also suggest SARS-CoV-2 conjunctivitis is often associated with the severity of the disease, and highlight the need for eye protection for all individuals potentially exposed to the virus (Aiello et al., 2020).
8.2. Dermatological lesions
Some health experts have reported a dermatological sign known as “COVID toes”, a blue or reddish discoloration of the toes, and at times the hands. When present, the condition is associated with itching and pain, while other times absent of any other symptoms. Studies thus far have indicated that it predominantly affects young individuals. The American Academy of Dermatology has requested that medical staff document cases in which patients “develop dermatological manifestations” of the infection. It is assumed that there is a link between dermatological manifestations and COVID-19. Galvan Casas et al. (2020) recently published a collection of images and a description of the dermatological manifestations associated with COVID-19 infection, and also linked specific dermatological manifestations with the disease stages. Specifically, the authors described that vesicular eruptions usually appeared early in the course of the disease, and even prior to the presentation of other typical symptomatology, and pseudo-chilblain patterns commonly present later in the course of the disease (Galvan Casas et al., 2020).
The referred association is not yet understood; however, two possible explanations could be that the lesions are an inflammatory response to the virus or may related t thromboembolic phenomena, given the known coagulopathies associated with COVID-19 infection (Seirafianpour et al., 2020). The lesions are localized to small blood vessels in the hands and feet, and are likely related to an inflammatory reaction, leading to consequent vasoconstriction. The vasoconstriction of these small blood vessels may occur in response to inflammation in the body's reaction to the virus that causes COVID-19 (Seirafianpour et al., 2020).
9. Clinical implications of COVID-19 infection in cancer patients
The previous paragraphs on the pathogenetic mechanisms of SARS-CoV-2 virus clearly highlight that the immune system plays a crucial role against this infection. Starting from this fundamental notion, several studies worldwide have demonstrated that subjects who present with immunodeficiencies or severe comorbidities are more susceptible to SARS-CoV-2 infection (Guan et al., 2020d; Carnero Contentti and Correa, 2020).
One of the categories of patients most at risk of developing a severe COVID-19 infection is that of cancer patients, where the disease state and the anticancer pharmacological and surgical treatments can induce long-term immunodeficiency which exposes cancer patients to various microbial and severe fungal infections (Zlatian et al., 2018; Maschmeyer and Haas, 2008; Tanase et al., 2016). In particular, chemotherapeutic and radiotherapeutic regimens have a direct antiproliferative action towards cancer cells (Sani et al., 2017); however, these drugs, including cytotoxic agents, selective inhibitors and the new immune checkpoint inhibitors, have high toxicity also for normal cells in constant replication such as all progenitor cells and white blood cells in the bone marrow, and the epithelial cells of the digestive tract (Gao et al., 2019; Falzone et al., 2018). Thus, anticancer treatments are able to directly suppress the immune system by killing white blood cells and indirectly, secondary to cachexia, nutritional deficiencies and malabsorption syndromes, including vitamin B12 deficiency and a folate low intake, due to damage to the digestive tract (Ibrahim et al., 2016; Rasmussen and Arvin, 1982).
In order to clearly establish the risk of acquiring SARS-CoV-2 and to predict the clinical evolution of COVID-19 infection in cancer patients, an increasing number of epidemiological and clinical studies have been performed worldwide, beginning in Wuhan in China, the first outbreak of COVID-19 infection, followed by European countries and the USA (Moujaess et al., 2020).
Compared to the general population, these studies suggest that the immunosuppression associated with the type of tumor and anticancer treatments significantly increases the risk of acquiring the SARS-CoV-2 virus in patients with the disease (Liang et al., 2020; Xia et al., 2020a; Yeoh et al., 2020). However, conflicting evidence exists on this topic. Indeed, Liang et al. report that cancer patients had an odds ratio (OR) of 5.34 of developing COVID-19 infection, adjusting the data for age, smoking history and other comorbidities. Notably, the authors demonstrated that among the cancer patients analyzed, those subjected to surgical resection or chemotherapy over the past month had a 75% probability of developing severe COVID-19 manifestations if infected (Liang et al., 2020). On the other hand, Xia et al. critically analyzed the data obtained by Liang et al. (2020) In particular, Xia et al., 2020 suggested that the high number of cancer patients found in the COVID-19 cohort compared to the normal population group did not correspond to a real increased risk of COVID-19 infection in cancer patients. Other concerns were related to the small number of cancer patients analyzed and to the heterogeneity of the clinicopathological features of these patients. Indeed, variable tumors, treatments and cancer pathology histories were taken into account (Xia et al., 2020a). Finally, Xia et al., 2020 concluded that the higher incidence of severe COVID-19 clinical manifestations observed in cancer patients may be due to the high percentage of cancer patients with smoking habits examined in the study conducted by Liang et al., as it is known that the chronic inflammation caused by smoking induces severe lung and respiratory damages, such as chronic obstructive pulmonary disease, associated with a worse prognosis of patients with COVID-19 (Xia et al., 2020a).
On these bases, in order to establish the real risk of infection in cancer patients, different factors should be considered. In particular, variability in the rate of COVID-19-positive cancer patients in different geographical regions may be related to the different policies and health protocols adopted by countries worldwide (Peng et al., 2020). These different preventive strategies may be responsible for the discrepancies observed in the COVID-19 epidemiological data in cancer patient cohorts, introducing significant bias in the interpretation of the results. In particular, the case fatality rate (CFR) for cancer patients with COVID-19 infection (all types of cancer) observed in China is of 5.6% compared to the 0.9% of CFR in COVID-19 patients without comorbidities (The Novel Coronavirus Pneumonia Emergency Response Epidemiology T, 2019). Contrasting data derived from the analysis of 3200 COVID-19 deaths performed by the Italian Istituto Superiore della Sanità (data updated until March 20, 2020). The members of the Italian COVID-19 Surveillance Group demonstrated that in Italy, the case fatality rates (CFRs) of all patients with COVID-19 were higher compared to those of other countries, reaching a percentage equal to 7.2%. In agreement with these data, the authors demonstrated that in a subsample of 355 patients with COVID-19 who died in Italy, 87 of them had active cancer (24.5%), thus suggesting that the CFRs in cancer patients with COVID-19 infection in Italy may be reasonably higher than those observed in China (Onder et al., 2020). In addition, other studies have demonstrated that the CFRs in cancer patients are dependent on the type of tumor taken into account. He et al. (2020) have demonstrated that in a cohort of 128 subjects with hematological cancers, only 10% (13 patients) acquired SARS-CoV-2 virus; however, in these onco-hematological COVID-19-positive patients, the CFR was 62% (He et al., 2020). Another study by Yu et al. (2020) demonstrated that in solid tumors (non-small cell lung cancer and renal cancer), the CFR of cancer COVID-19-positive patients was 25%; however, only a limited number of samples was analyzed (Yu et al., 2020).
As already mentioned, the reasons for these high CFR values in cancer patients are certainly related to the myelosuppression and immune deficiencies often observed in cancer patients (Wargo et al., 2015; Wang et al., 2006). In addition, it has been observed that a significant fraction of COVID-19-positive cancer patients experiences severe respiratory manifestations with the adoption of invasive respiratory support, including invasive ventilation and admission to the intensive care unit (Sidaway, 2020). These severe respiratory manifestations are prompted both by the direct action of the SARS-CoV-2 virus within the pulmonary epithelium and interstitial cavities, causing chronic inflammation which results in bilateral interstitial pneumonia (Lomoro et al., 2020), a well as by indirect cancer-related mechanisms, where tumor cells and the tumor microenvironment produce immunosuppressive factors and pro-inflammatory cytokines that impair the immune surveillance, worsening the COVID-19 infection (Xia et al., 2020a; Liu et al., 2019; Schreiber et al., 2011).
Finally, COVID-19 interstitial pneumonia in cancer patients may be further exacerbated by other pulmonary cellular and molecular alterations, including the overexpression of intracellular and membrane receptors in neoplastic cells, particularly in lung cancer patients (Ibrahim et al., 2020; Rizzo et al., 2020; Ulrich and Pillat, 2020). The overexpression of transmembrane proteins recognized by SARS-CoV-2 envelope proteins may facilitate the penetration of virus; of these, ACE2 receptors seem to be the most prominent proteins in SARS-CoV-2 pulmonary cells invasion, particularly in cancer patients with a long history of smoking (Zheng et al., 2020b; Cai, 2020).
Other important clinical implications of COVID-19 infection for cancer patients are related to anticancer surgical, pharmacological and radiation therapies. The global “lockdown” following the worldwide spread of COVID-19 infections resulted in a substantial change to hospital access for cancer patients (Kostoff et al., 2020b). Scheduled cancer screening visits, surgical interventions, some pharmacological treatments and appointments are being postponed or canceled in order to make available as many hospital beds as possible for COVID-19 patients with respiratory symptoms. In addition, the shortage of oncological health care workers due to the spread of COVID-19 within hospitals or due to the enforced quarantine measures for suspicious cases contributed to the delay of therapies and follow-up visits (Cannizzaro and Puglisi, 2020). Therefore, a case-by-case decision was often made to try to lighten as much as possible the workload of the health systems of most of the affected countries (The Lancet, 2020).
All the above-mentioned limitations represent a severe risk for the health status of cancer patients, who in some cases, experienced delays in the access to pharmacological treatments or, in more severe patients, were even prohibited to access to intensive care units (where the risk of COVID-19 infections is significantly high) with harmful consequences. This emergency led the main cancer scientific organizations, including the Italian Association of Medical Oncology (AIOM), the American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO), among others, to define specific guidelines in order to guarantee the necessary treatments for cancer patients by ensuring pharmacological treatments when strictly necessary, and rescheduled or shifted to telemedicine other evaluation or follow-up visits (Moujaess et al., 2020; Cannizzaro and Puglisi, 2020).
The consequence of COVID-19 infection in cancer patients is also dependent on the type of treatment to which they are subjected. The studies cited above refer only to a worse prognosis for COVID-19-positive cancer patients treated surgically or with chemotherapy [Lian W et al., 2020]; however little is known regarding the dual interaction existing between COVID-19 infection and the new immune checkpoint inhibitors (ICIs) in cancer patients (Vivarelli et al., 2020). These ICIs, and in particular anti-PD-1/PD-L1 and anti-CTLA-4 antibodies, are currently used in a wide cohort of patients for the treatment of different advanced and metastatic tumors (Christofi et al., 2019). In contrast to chemotherapeutic regimens, ICIs do not negatively affect the immune system, as such treatments re-establish immune cell competence, counteracting potential infectious diseases, as demonstrated with other viral infections (Bersanelli et al., 2020; Bersanelli, 2020; Wykes and Lewin, 2018). Preliminary evaluations on COVID-19 and ICIs have suggested that treatments with anti-PD-1/PD-L1 and anti-CTLA-4 antibodies, alone or in combination in cancer patients may also be administered during COVID-19 infections if no severe symptoms are present. However, the administration of these drugs should be evaluated in a case-by-case scenario, taking into account the risk/benefit ratio for the patient (Bersanelli, 2020; Kattan et al., 2020). If the pulmonary side-effects of ICIs or cytokine-release syndrome are detected, it is advisable to terminate the anticancer immunotherapy (Chen et al., 2020b). In this context, a growing body of evidence has elucidated some molecular mechanisms behing the interaction of ICIs, and in particular anti-PD-1/PD-L1, with SARS-CoV-2 and the immune cells responsible for the onset of cytokine storm in COVID-19 patients. Through the analysis of previous studies performed on mice and humans, a recent review of the literature proposed a possibile mechanism of action of anti-PD-1 treatement in contrasting viral infections (Vivarelli et al., 2020). Indeed, the administration of anti-PD-1/PD-L1 agents (e.g. ipilimumab, nivolumab, pembrolizumab, etc.) in animal models of lymphocytic choriomeningitis virus or in HIV patients has demonstrated beneficial effects thanks to the indirect regulatory actions on CD4+ and CD8+ T-cell responses that counteract virus propagation. of ICIs against virus antibodies administered to subjects chronically infected with viruses (i.e., lymphocytic choriomeningitis virus in mice or HIV in humans), enhance the viral control and virus-specific CD8+ T-cell responses, suggesting that a similar molecular action could be could also happen in case of COVID-19 infection. Other studies have demonstrated that different viral infection, including that of SARS-CoV-2, stimulate the expression of PD1 on infected cell surface thus inhibiting the immune action of T-cells (Schönrich and Raftery, 2019a; Allegra et al., 2020). Therefore, the administration of anti-PD-1/PD-L1 could reactivate CD4+ and CD8+ T-cell improving the host immune response towards SARS-Cov-2 infected cells with the activation of an immune cascade and the stimulation of other immune cells and receptors like tool like receptors (Schönrich and Raftery, 2019b; Onofrio et al., 2020). As already mentioned, although not properly defined as immunotherapeutic drugs, other monoclonal antibodies have proved efficacy in counteracting SARS-CoV-2 infection reducing the correlated symtomps, including Tocilizumab, Eculizumab, etc. (Di Giambenedetto et al., 2020). Through the stimulation of the immune system or the reduction of inflammatory cytokines, these monoclonal represent an effective therapeutic option in patients with severe respiratory symtomps.
By contrast, for patients treated with standard chemotherapy, dose reduction or discontinuation of treatment during the pandemic has been recommended as these drugs, in particular alkylating agents, taxol derivates, antimetabolites, etc., are capable of inducing strong neutropenia and myelosuppression, which can aggravate the health of patients in the case of COVID-19 infection (Yeoh et al., 2020; Falzone et al., 2018). For some tumors, specific recommendations were provided. For example, Curigliano et al., 2020 suggested avoiding or reducing the use of anthracyclines, docetaxel or platinum due to the risk of immunosuppression and, in ER+/HER2-negative patients, refraining from administering adjuvant chemotherapy (Curigliano et al., 2020). These authors further suggested the use of chemotherapy only in advanced breast cancer, preferring the administration of oral agents or capecitabine with a lower risk of immunosuppression, coupled with the administration of hematopoietic growth factors to reduce the risk of febrile neutropenia (Curigliano et al., 2020).
Finally, for radiation therapy, no clear recommendations on postponing or avoiding radiation therapy during the COVID-19 pandemic have been proposed. However, it is known that high-dose irradiation is associated with hematopoietic toxicity (Costa and Reagan, 2019); therefore, although proton beam therapy or stereotactic radiation are less toxic, anticancer radiation protocols were also generally delayed in order to avoid the risk of COVID-19 infection during centering visits or radiotherapy sessions (Rivera et al., 2020). On these bases, treatments should be planned by first assessing the disease status of each patient, thus reducing the number of radiotherapy sessions to the strict minimum (Rivera et al., 2020).
On the whole, the above corroborate that the abundance of data has been collected on the clinical implication of COVID-19 infection for cancer patients. However, the different epidemiological spread of the infection in various countries and the lack of standardized protocols for the management of cancer patients during the COVID-19 pandemic has generated conflicting data that should be carefully analyzed and normalized.
Overall, there is still no robust evidence available to define a specific association between cancer patients and COVID-19 infection. However, cancer patients have always been considered a subclass of high-risk subjects for COVID-19 infection and severe respiratory syndrome. Therefore, it is necessary to implement preventive strategies in order to better manage cancer patients, optimizing the administration of anticancer treatments and thus reducing the risk of immunosuppression in such patients.
10. Overall conclusions and future perspectives
At the beginning of the COVID-19 pandemic, the virus appeared to be a respiratory disease; however, SARS-CoV-2 infection can have a number of clinical effects, from heart damage to GI-related issues and neurological disorders, and the list of non-respiratory symptoms has grown considerably. For this reason, the present review presents an update of the most relevant information on the most important pathophysiology and symptoms of COVID-19 infection. Even so, although a more complete picture of the disease exists nowadays, unclear issues remain and it is expected that at least some of the information presented in this comprehensive review will change with the results of new research.
The respiratory manifestations of COVID-19 are directly associated with the severity of the disease. The further understanding of the pathophysiology of the disease and determination of the prognostic utility of clinical and laboratory parameters associated with COVID-19-related ARDS will possibly lead to the development of more precise and effective management approaches for the disease.
Studies have demonstrated that a small percentage of patients hospitalized with COVID-19 have cardiovascular disease at the time of the diagnosis of COVID-19 infection; however, the number of patients who develop cardiovascular sequelae varies, consisting of acute myocardial injury, myocarditis, acute coronary syndrome, cardiomyopathy, congestive heart failure, cardiogenic shock and arrhythmias.
Overall, although COVID-19-related GI manifestations, such as gastroenteritis and liver injury are known, further evidence is required to assess the effects of COVID-19 under existing chronic GI and liver diseases, as well as the determination of the utility of liver enzymes in the management of patients with COVID-19.
In addition, the kidneys are frequently affected in subjects who test positive and are symptomatic. Proteinuria often occurs at the beginning or during the infection and some patients develop acute renal failure, which may be a predictor of the mortality of infected patients.
The available studies on COVID-19 neurological manifestations are limited. However, a growing number of evidence indicates that patients with COVID-19 may exhibit a wide range of neurological complications affecting both the CNS and peripheral nervous system. The most accredited hypothesis behind the development of neurological symptoms is related to the nasal transsynaptic propagation of SARS-CoV-2, together with the binding of the ACE2 receptor and viral persistency within the neuronal cells. The moderate incidence of COVID-19 neurological manifestations has prompted researchers involved in the field of neurosciences to shed the light onto the molecular and pathological mechanisms responsible for these common and eventually severe neurological manifestations. In addition, specific clinical examinations (lumbar puncture, EEG, MRI, etc.) are currently undergoing in patients with encephalopathy, encephalitis, acute cerebrovascular diseases and other neurological disorders.
The epidemiological data obtained from cancer patients have revealed that both incidence and CFR are higher in cancer patients compared to the general population. In these patients, numerous efforts have been made to limit the number of infections, including changing treatment plans, carrying out visits with the aid of telemedicine and isolating cancer patients from hospital wards considered at risk. Furthermore, studies are trying to characterize the immunological profile of patients undergoing immunotherapy, as in these patients the treatment does not seem to determine an increase in risk and therefore, suspension or reduction of the dose may not be necessary.
However, the management of cancer patients and the administration of anticancer drugs should be evaluated, taking into account a patient-by-patient scenario and considering the risk/benefit ratio for the patient.
As further information is learned about the virus and the pathophysiology associated with COVID-19, the further understanding of the processes associated with its organ-specific damage may soon be likely, and the exisiting treatment modalities may have to be re-evaluated.
Funding
The present study was supported in part by grants from the national Institute of Environmental Health Sciences (NIEHS) R01ES10563 and R01ES07331 (to MA).
CRediT authorship contribution statement
Aristides Tsatsakis: Conceptualization, Methodology, Investigation, Writing - review & editing, Supervision. Daniela Calina: Conceptualization, Methodology, Investigation, Writing - review & editing, Supervision. Luca Falzone: Methodology, Investigation, Data curation, Writing - original draft. Dimitrios Petrakis: Methodology, Investigation, Data curation, Writing - original draft. Radu Mitrut: Methodology, Investigation, Data curation, Writing - original draft. Vasileios Siokas: Investigation, Data curation, Writing - original draft. Manuela Pennisi: Investigation, Data curation, Writing - original draft. Giuseppe Lanza: Investigation, Writing - review & editing, Supervision. Massimo Libra: Investigation, Writing - review & editing, Supervision. Sotirios G. Doukas: Methodology, Investigation, Data curation, Writing - original draft. Panagiotis G. Doukas: Methodology, Investigation, Data curation, Writing - original draft. Leena Kavali: Conceptualization, Methodology, Writing - review & editing, Supervision. Amar Bukhari: Conceptualization, Methodology, Writing - review & editing, Supervision. Chiranjeevi Gadiparthi: Conceptualization, Methodology, Investigation, Writing - review & editing, Supervision. Dimitra P. Vageli: Conceptualization, Methodology, Investigation, Writing - review & editing, Supervision. Diamantis P. Kofteridis: Methodology, Investigation, Data curation, Writing - review & editing, Supervision. Demetrios A. Spandidos: Methodology, Investigation, Data curation, Writing - review & editing, Supervision. Monica M.B. Paoliello: Methodology, Investigation, Data curation, Writing - original draft, Supervision. Michael Aschner: Methodology, Investigation, Data curation, Writing - review & editing, Supervision. Anca Oana Docea: Methodology, Investigation, Data curation, Writing - review & editing, Supervision.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors would like to thank all healthcare providers worldwide, who, with self-sacrifice and responsibility, have provided care to patients with COVID 19.
References
- Abedi F., Rezaee R., Karimi G. Plausibility of therapeutic effects of rho kinase inhibitors against severe acute respiratory syndrome coronavirus 2 (COVID-19) Pharmacol. Res. 2020;156:104808. doi: 10.1016/j.phrs.2020.104808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ai T., Yang Z., Hou H., Zhan C., Chen C., Lv W., et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: a report of 1014 cases. Radiology. 2020:200642. doi: 10.1148/radiol.2020200642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aiello F., Gallo Afflitto G., Mancino R., Li J.-P.O., Cesareo M., Giannini C., et al. Coronavirus disease 2019 (SARS-CoV-2) and colonization of ocular tissues and secretions: a systematic review. Eye. 2020;34(7):1206–1211. doi: 10.1038/s41433-020-0926-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alberti P., Beretta S., Piatti M., Karantzoulis A., Piatti M.L., Santoro P., et al. Guillain-Barré syndrome related to COVID-19 infection. Neurology - Neuroimmunology Neuroinflammation. 2020;7(4):e741. doi: 10.1212/NXI.0000000000000741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allegra A., Di Gioacchino M., Tonacci A., Musolino C., Gangemi S. Immunopathology of SARS-CoV-2 infection: immune cells and mediators, prognostic factors, and immune-therapeutic implications. Int. J. Mol. Sci. 2020;21(13):4782. doi: 10.3390/ijms21134782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azwar M.K., Kirana F., Kurniawan A., Handayani S., Setiati S. Gastrointestinal presentation in COVID-19 in Indonesia: a case report. Acta Med. Indones. 2020;52(1):63–67. [PubMed] [Google Scholar]
- Babapoor-Farrokhran S., Gill D., Walker J., Rasekhi R.T., Bozorgnia B., Amanullah A. Myocardial injury and COVID-19: possible mechanisms. Life Sci. 2020;253:117723. doi: 10.1016/j.lfs.2020.117723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai Y., Yao L., Wei T., Tian F., Jin D.Y., Chen L., et al. Presumed asymptomatic carrier transmission of COVID-19. Jama. 2020;323(14):1406–1407. doi: 10.1001/jama.2020.2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baig A.M., Khaleeq A., Ali U., Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11(7):995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- Bansal M. Cardiovascular disease and COVID-19. Diabetes Metab Syndr. 2020;14(3):247–250. doi: 10.1016/j.dsx.2020.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benny R., Khadilkar S.V. COVID 19: neuromuscular manifestations. Ann. Indian Acad. Neurol. 2020;23(Suppl. 1):S40–S42. doi: 10.4103/aian.AIAN_309_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bersanelli M. Controversies about COVID-19 and anticancer treatment with immune checkpoint inhibitors. Immunotherapy. 2020;12(5):269–273. doi: 10.2217/imt-2020-0067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bersanelli M., Scala S., Affanni P., Veronesi L., Colucci M.E., Banna G.L., et al. Immunological insights on influenza infection and vaccination during immune checkpoint blockade in cancer patients. Immunotherapy. 2020;12(2):105–110. doi: 10.2217/imt-2019-0200. [DOI] [PubMed] [Google Scholar]
- Boettler T., Newsome P.N., Mondelli M.U., Maticic M., Cordero E., Cornberg M., et al. Care of patients with liver disease during the COVID-19 pandemic: EASL-ESCMID position paper. JHEP reports : innovation in hepatology. 2020;2(3):100113. doi: 10.1016/j.jhepr.2020.100113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohmwald K., Gálvez N.M.S., Ríos M., Kalergis A.M. Neurologic alterations due to respiratory virus infections. Front. Cell. Neurosci. 2018;12:386. doi: 10.3389/fncel.2018.00386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boriani G., Fauchier L., Aguinaga L., Beattie J.M., Blomstrom Lundqvist C., Cohen A., et al. European heart rhythm association (EHRA) consensus document on management of arrhythmias and cardiac electronic devices in the critically ill and post-surgery patient, endorsed by heart rhythm society (HRS), asia pacific heart rhythm society (APHRS), cardiac arrhythmia society of southern africa (CASSA), and Latin American heart rhythm society (LAHRS) EP Europace. 2018;21(1):7–8. doi: 10.1093/europace/euy110. [DOI] [PubMed] [Google Scholar]
- Brown E., Gray R., Lo Monaco S., O'Donoghue B., Nelson B., Thompson A., et al. The potential impact of COVID-19 on psychosis: a rapid review of contemporary epidemic and pandemic research. Schizophr. Res. 2020;S0920–9964(20) doi: 10.1016/j.schres.2020.05.005. 30257-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buga A.-M., Docea A.O., Albu C., Malin R.D., Branisteanu D.E., Ianosi G., et al. Molecular and cellular stratagem of brain metastases associated with melanoma (Review) Oncol Lett. 2019;17(5):4170–4175. doi: 10.3892/ol.2019.9933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- C C.D. 2020. Symptoms of Coronavirus.https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html Available from: [Google Scholar]
- Cai G. Bulk and single-cell transcriptomics identify tobacco-use disparity in lung gene expression of ACE2, the receptor of 2019-nCov. medRxiv. 2020 2020.02.05.20020107, PPR: PPR112394. [Google Scholar]
- Cai Q., Huang D., Ou P., Yu H., Zhu Z., Xia Z., et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province. China. 2020;75(7):1742–1752. doi: 10.1111/all.14309. [DOI] [PubMed] [Google Scholar]
- Calina D., Docea A.O., Petrakis D., Egorov A.M., Ishmukhametov A.A., Gabibov A.G., et al. Towards effective COVID-19 vaccines: updates, perspectives and challenges (Review) Int. J. Mol. Med. 2020;46(1):3–16. doi: 10.3892/ijmm.2020.4596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camdessanche J.P., Morel J., Pozzetto B., Paul S., Tholance Y., Botelho-Nevers E. COVID-19 may induce Guillain-Barré syndrome. Rev. Neurol. (Paris) 2020;176(6):516–518. doi: 10.1016/j.neurol.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cannizzaro R., Puglisi F. Covid-19 and cancer patients: choosing wisely is the key. Digestive and liver disease. official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2020;52(6):595–596. doi: 10.1016/j.dld.2020.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carnero Contentti E., Correa J. Immunosuppression during the COVID-19 pandemic in neuromyelitis optica spectrum disorders patients: a new challenge. Multiple sclerosis and related disorders. 2020;41:102097. doi: 10.1016/j.msard.2020.102097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carod-Artal F.J. Neurological complications of coronavirus and COVID-19. Rev. Neurol. 2020;70(9):311–322. doi: 10.33588/rn.7009.2020179. [DOI] [PubMed] [Google Scholar]
- Channappanavar R., Zhao J., Perlman S. T cell-mediated immune response to respiratory coronaviruses. Immunol. Res. 2014;59(1–3):118–128. doi: 10.1007/s12026-014-8534-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chau T.-N., Lee K.-C., Yao H., Tsang T.-Y., Chow T.-C., Yeung Y.-C., et al. SARS-associated viral hepatitis caused by a novel coronavirus: report of three cases. Hepatology. 2004;39:302–310. doi: 10.1002/hep.20111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X., Tang Y., Mo Y., Li S., Lin D., Yang Z., et al. A diagnostic model for coronavirus disease 2019 (COVID-19) based on radiological semantic and clinical features: a multi-center study. Eur. Radiol. 2020;30(9):4893–4902. doi: 10.1007/s00330-020-06829-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C., Zhang X.R., Ju Z.Y., He W.F. [Advances in the research of mechanism and related immunotherapy on the cytokine storm induced by coronavirus disease 2019] Zhonghua Shaoshang Zazhi. 2020;36(6):471–475. doi: 10.3760/cma.j.cn501120-20200224-00088. [DOI] [PubMed] [Google Scholar]
- Christofi T., Baritaki S., Falzone L., Libra M., Zaravinos A. Current perspectives in cancer immunotherapy. Cancers. 2019;11(10):1472. doi: 10.3390/cancers11101472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu K.H., Tsang W.K., Tang C.S., Lam M.F., Lai F.M., To K.F., et al. Acute renal impairment in coronavirus-associated severe acute respiratory syndrome. Kidney Int. 2005;67(2):698–705. doi: 10.1111/j.1523-1755.2005.67130.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung M., Bernheim A., Mei X., Zhang N., Huang M., Zeng X., et al. CT imaging features of 2019 novel coronavirus (2019-nCoV) Radiology. 2020;295(1):202–207. doi: 10.1148/radiol.2020200230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cordonnier D.J., Zaoui P., Halimi S. Role of ACE inhibitors in patients with diabetes mellitus. Drugs. 2001;61(13):1883–1892. doi: 10.2165/00003495-200161130-00001. [DOI] [PubMed] [Google Scholar]
- Costa S., Reagan M.R. Therapeutic irradiation: consequences for bone and bone marrow adipose tissue. Front. Endocrinol. 2019;10:587. doi: 10.3389/fendo.2019.00587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cristelo C., Azevedo C., Marques J.M., Nunes R., Sarmento B. SARS-CoV-2 and diabetes: new challenges for the disease. Diabetes Res. Clin. Pract. 2020;164:108228. doi: 10.1016/j.diabres.2020.108228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham A.C., Goh H.P., Koh D. Treatment of COVID-19: old tricks for new challenges. Crit. Care. 2020;24(1):91. doi: 10.1186/s13054-020-2818-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curigliano G., Cardoso M.J., Poortmans P., Gentilini O., Pravettoni G., Mazzocco K., et al. Recommendations for triage, prioritization and treatment of breast cancer patients during the COVID-19 pandemic. Breast. 2020;52:8–16. doi: 10.1016/j.breast.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallan C., Romano F., Siebert J., Politi S., Lacroix L., Sahyoun C. Septic shock presentation in adolescents with COVID-19. The Lancet Child & Adolescent Health. 2020;4(7):e21–e23. doi: 10.1016/S2352-4642(20)30164-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeDiego M.L., Nieto-Torres J.L., Regla-Nava J.A., Jimenez-Guardeño J.M., Fernandez-Delgado R., Fett C., et al. Inhibition of NF-κB-mediated inflammation in severe acute respiratory syndrome coronavirus-infected mice increases survival. J. Virol. 2014;88(2):913–924. doi: 10.1128/JVI.02576-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dehelean C.A., Lazureanu V., Coricovac D., Mioc M., Oancea R., Marcovici I., et al. SARS-CoV-2: repurposed drugs and novel therapeutic approaches—insights into chemical structure—biological activity and toxicological screening. J. Clin. Med. 2020;9(7):2084. doi: 10.3390/jcm9072084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng S.-Q., Peng H.-J. Characteristics of and public health responses to the coronavirus disease 2019 outbreak in China. J. Clin. Med. 2020;9(2):575. doi: 10.3390/jcm9020575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desforges M., Le Coupanec A., Dubeau P., Bourgouin A., Lajoie L., Dubé M., et al. Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses. 2019;12(1):14. doi: 10.3390/v12010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dessau R.B., Lisby G., Frederiksen J.L. Coronaviruses in spinal fluid of patients with acute monosymptomatic optic neuritis. Acta Neurol. Scand. 1999;100(2):88–91. doi: 10.1111/j.1600-0404.1999.tb01043.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinkin M., Gao V., Kahan J., Bobker S., Simonetto M., Wechsler P., et al. COVID-19 presenting with ophthalmoparesis from cranial nerve palsy. Neurology. 2020;95(5):221–223. doi: 10.1212/WNL.0000000000009700. [DOI] [PubMed] [Google Scholar]
- Docea A.O., Tsatsakis A., Albulescu D., Cristea O., Zlatian O., Vinceti M., et al. A new threat from an old enemy: Re-emergence of coronavirus (Review) Int. J. Mol. Med. 2020;45(6):1631–1643. doi: 10.3892/ijmm.2020.4555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dockery D.M., Rowe S.G., Murphy M.A., Krzystolik M.G. The ocular manifestations and transmission of COVID-19: recommendations for prevention. J. Emerg. Med. 2020;59(1):137–140. doi: 10.1016/j.jemermed.2020.04.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Doremalen N., Bushmaker T., Morris D.H., Holbrook M.G., Gamble A., Williamson B.N., et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020;382(16):1564–1567. doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doukas S.G., Vageli D.P., Sasaki C.T. NF-κB inhibition reverses acidic bile-induced miR-21, miR-155, miR-192, miR-34a, miR-375 and miR-451a deregulations in human hypopharyngeal cells. J. Cell Mol. Med. 2018;22(5):2922–2934. doi: 10.1111/jcmm.13591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driggin E., Madhavan M.V., Bikdeli B., Chuich T., Laracy J., Biondi-Zoccai G., et al. Cardiovascular considerations for patients, health care workers, and health systems during the COVID-19 pandemic. J. Am. Coll. Cardiol. 2020;75(18):2352–2371. doi: 10.1016/j.jacc.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falzone L., Salomone S., Libra M. Evolution of cancer pharmacological treatments at the turn of the third millennium. Front. Pharmacol. 2018;9:1300. doi: 10.3389/fphar.2018.01300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falzone L., Musso N., Gattuso G., Bongiorno D., Palermo C.I., Scalia G., et al. Sensitivity assessment of droplet digital PCR for SARS-CoV-2 detection. Int. J. Mol. Med. 2020;46(3):957–964. doi: 10.3892/ijmm.2020.4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan Z., Chen L., Li J., Cheng X., Yang J., Tian C., et al. Clinical features of COVID-19-related liver functional abnormality. Clin. Gastroenterol. Hepatol. : the official clinical practice journal of the American Gastroenterological Association. 2020;18(7):1561–1566. doi: 10.1016/j.cgh.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang M., Siciliano N.A., Hersperger A.R., Roscoe F., Hu A., Ma X., et al. Perforin-dependent CD4+ T-cell cytotoxicity contributes to control a murine poxvirus infection. Proc. Natl. Acad. Sci. U.S.A. 2012;109(25):9983–9988. doi: 10.1073/pnas.1202143109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farsalinos K., Niaura R., Le Houezec J., Barbouni A., Tsatsakis A., Kouretas D., et al. Editorial: nicotine and SARS-CoV-2: COVID-19 may be a disease of the nicotinic cholinergic system. Toxicology reports. 2020;7:658–663. doi: 10.1016/j.toxrep.2020.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fazzini E., Fleming J., Fahn S. Cerebrospinal fluid antibodies to coronavirus in patients with Parkinson's disease. Mov. Disord. 1992;7(2):153–158. doi: 10.1002/mds.870070210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filatov A., Sharma P., Hindi F., Espinosa P.S. Neurological complications of coronavirus disease (COVID-19): encephalopathy. Cureus. 2020;12(3) doi: 10.7759/cureus.7352. e7352-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filetti V., Falzone L., Rapisarda V., Caltabiano R., Eleonora Graziano A.C., Ledda C., et al. Modulation of microRNA expression levels after naturally occurring asbestiform fibers exposure as a diagnostic biomarker of mesothelial neoplastic transformation. Ecotoxicol. Environ. Saf. 2020;198:110640. doi: 10.1016/j.ecoenv.2020.110640. [DOI] [PubMed] [Google Scholar]
- Fleming J.O., Stohlman S.A., Weiner L.P. Coronaviruses and multiple sclerosis. Arch. Neurol. 1982;39(7):455. doi: 10.1001/archneur.1982.00510190073030. [DOI] [PubMed] [Google Scholar]
- Fried J.A., Ramasubbu K., Bhatt R., Topkara V.K., Clerkin K.J., Horn E., et al. The variety of cardiovascular presentations of COVID-19. Circulation. 2020;141(23):1930–1936. doi: 10.1161/CIRCULATIONAHA.120.047164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gambhir S., Vyas D., Hollis M., Aekka A., Vyas A. Nuclear factor kappa B role in inflammation associated gastrointestinal malignancies. World J. Gastroenterol. 2015;21(11):3174–3183. doi: 10.3748/wjg.v21.i11.3174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Q., Zhou G., Lin S.J. How chemotherapy and radiotherapy damage the tissue: comparative biology lessons from feather and hair models. 2019;28(4):413–418. doi: 10.1111/exd.13846. [DOI] [PubMed] [Google Scholar]
- Gattinoni L., Chiumello D., Rossi S. COVID-19 pneumonia: ARDS or not? Crit. Care. 2020;24(1):154. doi: 10.1186/s13054-020-02880-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giacomelli A., Pezzati L., Conti F., Bernacchia D., Siano M., Oreni L., et al. Self-reported olfactory and taste disorders in patients with severe acute respiratory coronavirus 2 infection: a cross-sectional study. Clin. Infect. Dis. 2020;71(15):889–890. doi: 10.1093/cid/ciaa330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Giambenedetto S., Ciccullo A., Borghetti A., Gambassi G., Landi F., Visconti E., et al. Off-label use of tocilizumab in patients with SARS-CoV-2 infection. J. Med. Virol. 2020 doi: 10.1002/jmv.25897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong J., Dong H., Xia S.Q., Huang Y.Z., Wang D., Zhao Y., et al. Correlation analysis between disease severity and inflammation-related parameters in patients with COVID-19 pneumonia. medRxiv. 2020 doi: 10.1186/s12879-020-05681-5. 2020.02.25.20025643, PPR: PPR114815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goumenou M., Spandidos D.A., Tsatsakis A. [Editorial] Possibility of transmission through dogs being a contributing factor to the extreme Covid-19 outbreak in North Italy. Mol. Med. Rep. 2020;21(6):2293–2295. doi: 10.3892/mmr.2020.11037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goyal P., Choi J.J., Pinheiro L.C., Schenck E.J., Chen R., Jabri A., et al. Clinical characteristics of covid-19 in New York city. N. Engl. J. Med. 2020;382(24):2372–2374. doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu J., Han B., Wang J. COVID-19: gastrointestinal manifestations and potential fecal-oral transmission. Gastroenterology. 2020;158(6):1518–1519. doi: 10.1053/j.gastro.2020.02.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan W-j, Ni Z-y, Hu Y., Liang W-h, Ou C-q, He J-x, et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan W.-J., Ni Z.-Y., Hu Y., Liang W.-H., Ou C.-Q., He J.-X., et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan W-j, Ni Z-y, Hu Y., Liang W-h, Ou C-q, He J-x, et al. medRxiv; 2020. Clinical Characteristics of 2019 Novel Coronavirus Infection in China. 2020.02.06.20020974. [Google Scholar]
- Guan W.J., Liang W.H., Zhao Y., Liang H.R., Chen Z.S., Li Y.M., et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. Eur. Respir. J. 2020;55(5) doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo T., Fan Y., Chen M., Wu X., Zhang L., He T., et al. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):1–8. doi: 10.1001/jamacardio.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y.-R., Cao Q.-D., Hong Z.-S., Tan Y.-Y., Chen S.-D., Jin H.-J., et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak – an update on the status. Military Medical Research. 2020;7(1):11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta M.D., Girish M.P., Yadav G., Shankar A., Yadav R. Coronavirus disease 2019 and the cardiovascular system: impacts and implications. Indian Heart J. 2020;72(1):1–6. doi: 10.1016/j.ihj.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzik T.J., Mohiddin S.A., Dimarco A., Patel V., Savvatis K., Marelli-Berg F.M., et al. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020;116(10):1666–1687. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haga S., Yamamoto N., Nakai-Murakami C., Osawa Y., Tokunaga K., Sata T., et al. Modulation of TNF-alpha-converting enzyme by the spike protein of SARS-CoV and ACE2 induces TNF-alpha production and facilitates viral entry. Proc. Natl. Acad. Sci. U.S.A. 2008;105(22):7809–7814. doi: 10.1073/pnas.0711241105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamming I., Timens W., Bulthuis M.L.C., Lely A.T., Navis G.J., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203(2):631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanna T.P., Evans G.A., Booth C.M. Cancer, COVID-19 and the precautionary principle: prioritizing treatment during a global pandemic. Nat. Rev. Clin. Oncol. 2020;17(5):268–270. doi: 10.1038/s41571-020-0362-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He W., Chen L., Chen L., Yuan G., Fang Y., Chen W., et al. COVID-19 in persons with haematological cancers. Leukemia. 2020;34(6):1637–1645. doi: 10.1038/s41375-020-0836-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hindson J. COVID-19: faecal-oral transmission? Nat. Rev. Gastroenterol. Hepatol. 2020;17(5):259. doi: 10.1038/s41575-020-0295-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280. doi: 10.1016/j.cell.2020.02.052. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang I., Pranata R. Lymphopenia in severe coronavirus disease-2019 (COVID-19): systematic review and meta-analysis. Journal of Intensive Care. 2020;8(1):36. doi: 10.1186/s40560-020-00453-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang P., Liu T., Huang L., Liu H., Lei M., Xu W., et al. Use of chest CT in combination with negative RT-PCR assay for the 2019 novel coronavirus but high clinical suspicion. Radiology. 2020;295(1):22–23. doi: 10.1148/radiol.2020200330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hung E.C.W., Chim S.S.C., Chan P.K.S., Tong Y.K., Ng E.K.O., Chiu R.W.K., et al. Detection of SARS coronavirus RNA in the cerebrospinal fluid of a patient with severe acute respiratory syndrome. Clin. Chem. 2003;49(12):2108–2109. doi: 10.1373/clinchem.2003.025437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibrahim U.A., Yusuf A.A., Ahmed S.G. The pathophysiologic basis of anaemia in patients with malignant diseases. The Gulf journal of oncology. 2016;1(22):80–89. [PubMed] [Google Scholar]
- Ibrahim I.M., Abdelmalek D.H., Elshahat M.E., Elfiky A.A. COVID-19 spike-host cell receptor GRP78 binding site prediction. J. Infect. 2020;80(5):554–562. doi: 10.1016/j.jinf.2020.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inciardi R.M., Lupi L., Zaccone G., Italia L., Raffo M., Tomasoni D., et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):819–824. doi: 10.1001/jamacardio.2020.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobi A., Chung M., Bernheim A., Eber C. Portable chest X-ray in coronavirus disease-19 (COVID-19): a pictorial review. Clin. Imag. 2020;64:35–42. doi: 10.1016/j.clinimag.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarred G., Kennedy R.L. Therapeutic perspective: starting an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker in a diabetic patient. Therapeutic advances in endocrinology and metabolism. 2010;1(1):23–28. doi: 10.1177/2042018810369437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia H.P., Look D.C., Shi L., Hickey M., Pewe L., Netland J., et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J. Virol. 2005;79(23):14614–14621. doi: 10.1128/JVI.79.23.14614-14621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin M., Tong Q. Rhabdomyolysis as potential late complication associated with COVID-19. Emerg. Infect. Dis. 2020;26(7):1618–1620. doi: 10.3201/eid2607.200445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin X., Lian J.-S., Hu J.-H., Gao J., Zheng L., Zhang Y.-M., et al. Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut. 2020;69(6):1002–1009. doi: 10.1136/gutjnl-2020-320926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juliao Caamaño D.S., Alonso Beato R. Facial diplegia, a possible atypical variant of Guillain-Barré Syndrome as a rare neurological complication of SARS-CoV-2. J. Clin. Neurosci. 2020;77:230–232. doi: 10.1016/j.jocn.2020.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kattan J., Kattan C., Assi T. Do checkpoint inhibitors compromise the cancer patients' immunity and increase the vulnerability to COVID-19 infection? Immunotherapy. 2020;12(6):351–354. doi: 10.2217/imt-2020-0077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klok F.A., Kruip M., van der Meer N.J.M., Arbous M.S., Gommers D., Kant K.M., et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020;191:145–147. doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klok F.A., Kruip M.J.H.A., van der Meer N.J.M., Arbous M.S., Gommers D.A.M.P.J., Kant K.M., et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020;191:145–147. doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostoff R.N., Briggs M.B., Porter A.L., Hernández A.F., Abdollahi M., Aschner M., et al. The under-reported role of toxic substance exposures in the COVID-19 pandemic. Food Chem. Toxicol. 2020:111687. doi: 10.1016/j.fct.2020.111687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kostoff R.N., Briggs M.B., Porter A.L., Aschner M., Spandidos D.A., Tsatsakis A. COVID-19: post-lockdown guidelines. Int. J. Mol. Med. 2020;46(2):463–466. doi: 10.3892/ijmm.2020.4640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwenandar F., Japar K.V., Damay V., Hariyanto T.I., Tanaka M., Lugito N.P.H., et al. Coronavirus disease 2019 and cardiovascular system: a narrative review. Int J Cardiol Heart Vasc. 2020;29:100557. doi: 10.1016/j.ijcha.2020.100557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwong J.C., Schwartz K.L., Campitelli M.A., Chung H., Crowcroft N.S., Karnauchow T., et al. Acute myocardial infarction after laboratory-confirmed influenza infection. N. Engl. J. Med. 2018;378(4):345–353. doi: 10.1056/NEJMoa1702090. [DOI] [PubMed] [Google Scholar]
- Lagana S.M., Kudose S., Iuga A.C., Lee M.J., Fazlollahi L., Remotti H.E., et al. Hepatic pathology in patients dying of COVID-19: a series of 40 cases including clinical, histologic, and virologic data. Modern Pathology. 2020:1–9. doi: 10.1038/s41379-020-00649-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau K.-K., Yu W.-C., Chu C.-M., Lau S.-T., Sheng B., Yuen K.-Y. Possible central nervous system infection by SARS coronavirus. Emerg. Infect. Dis. 2004;10(2):342–344. doi: 10.3201/eid1002.030638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lauer S.A., Grantz K.H., Bi Q., Jones F.K., Zheng Q., Meredith H.R., et al. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann. Intern. Med. 2020;172(9):577–582. doi: 10.7326/M20-0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lechien J.R., Chiesa-Estomba C.M., De Siati D.R., Horoi M., Le Bon S.D., Rodriguez A., et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur. Arch. Oto-Rhino-Laryngol. 2020;277(8):2251–2261. doi: 10.1007/s00405-020-05965-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lechien J.R., Chiesa-Estomba C.M., Hans S., Barillari M.R., Jouffe L., Saussez S. Loss of smell and taste in 2013 European patients with mild to moderate COVID-19. Ann. Intern. Med. 2020 doi: 10.7326/M20-2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung W.K., To K-f, Chan P.K.S., Chan H.L.Y., Wu A.K.L., Lee N., et al. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection1. Gastroenterology. 2003;125(4):1011–1017. doi: 10.1016/j.gastro.2003.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Xiao S.-Y. Hepatic involvement in COVID-19 patients: pathology, pathogenesis, and clinical implications. J. Med. Virol. 2020;92(9):1491–1494. doi: 10.1002/jmv.25973. [DOI] [PubMed] [Google Scholar]
- Li L., Li S., Xu M., Yu P., Zheng S., Duan Z., et al. medRxiv; 2020. Risk Factors Related to Hepatic Injury in Patients with Corona Virus Disease 2019. PPR: PPR115458. [Google Scholar]
- Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;92(6):552–555. doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Li M., Wang M., Zhou Y., Chang J., Xian Y., et al. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke and vascular neurology. 2020 doi: 10.1136/svn-2020-000431. svn-2020-000431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang W., Guan W., Chen R., Wang W., Li J., Xu K., et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21(3):335–337. doi: 10.1016/S1470-2045(20)30096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin L., Jiang X., Zhang Z., Huang S., Zhang Z., Fang Z., et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut. 2020;69(6):997–1001. doi: 10.1136/gutjnl-2020-321013. [DOI] [PubMed] [Google Scholar]
- Lin L., Jiang X., Zhang Z., Huang S., Zhang Z., Fang Z., et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. 2020;69(6):997–1001. doi: 10.1136/gutjnl-2020-321013. [DOI] [PubMed] [Google Scholar]
- Liu C.C., Steen C.B., Newman A.M. Computational approaches for characterizing the tumor immune microenvironment. Immunology. 2019;158(2):70–84. doi: 10.1111/imm.13101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu K., Fang Y.-Y., Deng Y., Liu W., Wang M.-F., Ma J.-P., et al. Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin. Med. J. 2020;133(9):1025–1031. doi: 10.1097/CM9.0000000000000744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Li S., Liu J., Liang B., Wang X., Wang H., et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B., Li M., Zhou Z., Guan X., Xiang Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J. Autoimmun. 2020;111:102452. doi: 10.1016/j.jaut.2020.102452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodigiani C., Iapichino G., Carenzo L., Cecconi M., Ferrazzi P., Sebastian T., et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lomoro P., Verde F., Zerboni F., Simonetti I., Borghi C., Fachinetti C., et al. COVID-19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: single-center study and comprehensive radiologic literature review. European journal of radiology open. 2020;7:100231. doi: 10.1016/j.ejro.2020.100231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maiolo G., Collino F., Vasques F., Rapetti F., Tonetti T., Romitti F., et al. Reclassifying acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2018;197(12):1586–1595. doi: 10.1164/rccm.201709-1804OC. [DOI] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, China. JAMA Neurol. 2020;77(6):1–9. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, China. JAMA Neurology. 2020;77(6):683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marini J.J., Gattinoni L. Management of COVID-19 respiratory distress. Jama. 2020;323(22):2329–2330. doi: 10.1001/jama.2020.6825. [DOI] [PubMed] [Google Scholar]
- Maschmeyer G., Haas A. The epidemiology and treatment of infections in cancer patients. Int. J. Antimicrob. Agents. 2008;31(3):193–197. doi: 10.1016/j.ijantimicag.2007.06.014. [DOI] [PubMed] [Google Scholar]
- Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mititelu R.R., Pădureanu R., Băcănoiu M., Pădureanu V., Docea A.O., Calina D., et al. Inflammatory and oxidative stress markers-mirror tools in rheumatoid arthritis. Biomedicines. 2020;8(5) doi: 10.3390/biomedicines8050125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriguchi T., Harii N., Goto J., Harada D., Sugawara H., Takamino J., et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. : IJID : official publication of the International Society for Infectious Diseases. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moujaess E., Kourie H.R., Ghosn M. Cancer patients and research during COVID-19 pandemic: a systematic review of current evidence. Crit. Rev. Oncol. Hematol. 2020;150:102972. doi: 10.1016/j.critrevonc.2020.102972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Musa S. Hepatic and gastrointestinal involvement in coronavirus disease 2019 (COVID-19): what do we know till now? Arab journal of gastroenterology : the official publication of the Pan-Arab Association of Gastroenterology. 2020;21(1):3–8. doi: 10.1016/j.ajg.2020.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasi A., McArdle S., Gaudernack G., Westman G., Melief C., Rockberg J., et al. Reactive oxygen species as an initiator of toxic innate immune responses in retort to SARS-CoV-2 in an ageing population, consider N-acetylcysteine as early therapeutic intervention. Toxicology reports. 2020;7:768–771. doi: 10.1016/j.toxrep.2020.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S.C., Tilg H. COVID-19 and the gastrointestinal tract: more than meets the eye. 2020;69(6):973–974. doi: 10.1136/gutjnl-2020-321195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicin L., Abplanalp W.T., Mellentin H., Kattih B., Tombor L., John D., et al. Cell type-specific expression of the putative SARS-CoV-2 receptor ACE2 in human hearts. Eur. Heart J. 2020;41(19):1804–1806. doi: 10.1093/eurheartj/ehaa311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nile S.H., Nile A., Qiu J., Li L., Jia X., Kai G. COVID-19: pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020;53:66–70. doi: 10.1016/j.cytogfr.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitulescu G.M., Paunescu H., Moschos S.A., Petrakis D., Nitulescu G., Ion G.N.D., et al. Comprehensive analysis of drugs to treat SARS-CoV-2 infection: mechanistic insights into current COVID-19 therapies (Review) Int. J. Mol. Med. 2020;46(2):467–488. doi: 10.3892/ijmm.2020.4608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nussbaum L., Hogea L., Calina D., Andreescu N., Gradinaru R., Ștefănescu R., et al. Modern treatment approaches in psychoses. Pharmacogenetic, neuroimagistic and clinical implications. FARMACIA. 2017;65:75–81. [Google Scholar]
- Onder G., Rezza G., Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. J. Am. Med. Assoc. 2020;323(18):1775–1776. doi: 10.1001/jama.2020.4683. [DOI] [PubMed] [Google Scholar]
- Ong S.W.X., Tan Y.K., Chia P.Y., Lee T.H., Ng O.T., Wong M.S.Y., et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. Jama. 2020;323(16):1610–1612. doi: 10.1001/jama.2020.3227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onofrio L., Caraglia M., Facchini G., Margherita V., Placido S.D., Buonerba C. Toll-like receptors and COVID-19: a two-faced story with an exciting ending. Future Science OA. 2020;6(8):FSO605. doi: 10.2144/fsoa-2020-0091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oudit G.Y., Kassiri Z., Jiang C., Liu P.P., Poutanen S.M., Penninger J.M., et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur. J. Clin. Invest. 2009;39(7):618–625. doi: 10.1111/j.1365-2362.2009.02153.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oxley T.J., Mocco J., Majidi S., Kellner C.P., Shoirah H., Singh I.P., et al. Large-vessel stroke as a presenting feature of covid-19 in the young. N. Engl. J. Med. 2020;382(20) doi: 10.1056/NEJMc2009787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padureanu R., Albu C.V., Mititelu R.R., Bacanoiu M.V., Docea A.O., Calina D., et al. Oxidative stress and inflammation interdependence in multiple sclerosis. J. Clin. Med. 2019;8(11):1815. doi: 10.3390/jcm8111815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paramasivam A., Priyadharsini J.V., Raghunandhakumar S., Elumalai P. A novel COVID-19 and its effects on cardiovascular disease. Hypertens. Res. 2020;43(7):729–730. doi: 10.1038/s41440-020-0461-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paranjpe I., Fuster V., Lala A., Russak A.J., Glicksberg B.S., Levin M.A., et al. Association of treatment dose anticoagulation with in-hospital survival among hospitalized patients with COVID-19. J. Am. Coll. Cardiol. 2020;76(1):122–124. doi: 10.1016/j.jacc.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng F., Tu L., Yang Y., Hu P., Wang R., Hu Q., et al. Management and treatment of COVID-19: the Chinese experience. Can. J. Cardiol. 2020;36(6):915–930. doi: 10.1016/j.cjca.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pennisi M., Lanza G., Falzone L., Fisicaro F., Ferri R., Bella R. SARS-CoV-2 and the nervous system: from clinical features to molecular mechanisms. Int. J. Mol. Sci. 2020;21(15):5475. doi: 10.3390/ijms21155475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrakis D., Vassilopoulou L., Mamoulakis C., Psycharakis C., Anifantaki A., Sifakis S., et al. Endocrine disruptors leading to obesity and related diseases. Int. J. Environ. Res. Publ. Health. 2017;14(10):1282. doi: 10.3390/ijerph14101282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porcheddu R., Serra C., Kelvin D., Kelvin N., Rubino S. Similarity in case fatality rates (CFR) of COVID-19/SARS-COV-2 in Italy and China. J Infect Dev Ctries. 2020;14(2):125–128. doi: 10.3855/jidc.12600. [DOI] [PubMed] [Google Scholar]
- Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology. 2020;296(2):E119–E120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19–associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology. 2020;296(2):E119–E120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020;71(15):762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmussen L., Arvin A. Chemotherapy-induced immunosuppression. Environ. Health Perspect. 1982;43:21–25. doi: 10.1289/ehp.824321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W., et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. J. Am. Med. Assoc. 2020;323(20):2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera A., Ohri N., Thomas E., Miller R., Knoll M.A. The impact of COVID-19 on radiation oncology clinics and cancer patients in the U.S. Advances in radiation oncology. 2020;5(4):538–543. doi: 10.1016/j.adro.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rizzo P., Vieceli Dalla Sega F., Fortini F., Marracino L., Rapezzi C., Ferrari R. COVID-19 in the heart and the lungs: could we "Notch" the inflammatory storm? Basic Res. Cardiol. 2020;115(3):31. doi: 10.1007/s00395-020-0791-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogoveanu O.C., Calina D., Cucu M.G., Burada F., Docea A.O., Sosoi S., et al. Association of cytokine gene polymorphisms with osteoarthritis susceptibility. Exp Ther Med. 2018;16(3):2659–2664. doi: 10.3892/etm.2018.6477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell C.D., Millar J.E., Baillie J.K. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395(10223):473–475. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salehi B., Calina D., Docea A.O., Koirala N., Aryal S., Lombardo D., et al. Curcumin's nanomedicine formulations for therapeutic application in neurological diseases. J. Clin. Med. 2020;9(2) doi: 10.3390/jcm9020430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salehi B., Rescigno A., Dettori T., Calina D., Docea A.O., Singh L., et al. Avocado–soybean unsaponifiables: a panoply of potentialities to Be exploited. Biomolecules. 2020;10(1):130. doi: 10.3390/biom10010130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salemi R., Falzone L., Madonna G., Polesel J., Cinà D., Mallardo D., et al. MMP-9 as a candidate marker of response to BRAF inhibitors in melanoma patients with BRAFV600E mutation detected in circulating-free DNA. Front. Pharmacol. 2018;9(856) doi: 10.3389/fphar.2018.00856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmi A., Ziola B., Hovi T., Reunanen M. Antibodies to coronaviruses OC43 and 229E in multiple sclerosis patients. Neurology. 1982;32(3):292–295. doi: 10.1212/wnl.32.3.292. [DOI] [PubMed] [Google Scholar]
- Saltiel A.R., Olefsky J.M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Invest. 2017;127(1):1–4. doi: 10.1172/JCI92035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sani T.A., Mohammadpour E., Mohammadi A., Memariani T., Yazdi M.V., Rezaee R., et al. Cytotoxic and apoptogenic properties OF dracocephalum kotschyi aerial part different fractions ON CALU-6 and MEHR-80 lung cancer cell lines. FARMACIA. 2017;65(2):189–199. [Google Scholar]
- Sarkar C., Mondal M., Torequl Islam M., Martorell M., Docea A.O., Maroyi A., et al. Potential therapeutic options for COVID-19: current status, challenges, and future perspectives. Front. Pharmacol. 2020;11(1428) doi: 10.3389/fphar.2020.572870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönrich G., Raftery M.J. The PD-1/PD-L1 Axis and virus infections: a delicate balance. Front Cell Infect Microbiol. 2019;9:207. doi: 10.3389/fcimb.2019.00207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schönrich G., Raftery M.J. The PD-1/PD-L1 Axis and virus infections: a delicate balance. Frontiers in Cellular and Infection Microbiology. 2019;9(207) doi: 10.3389/fcimb.2019.00207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber R.D., Old L.J., Smyth M.J. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science. 2011;331(6024):1565–1570. doi: 10.1126/science.1203486. [DOI] [PubMed] [Google Scholar]
- Sedaghat Z., Karimi N. Guillain Barre syndrome associated with COVID-19 infection: a case report. J. Clin. Neurosci. 2020;76:233–235. doi: 10.1016/j.jocn.2020.04.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seirafianpour F., Sodagar S., Pour Mohammad A., Panahi P., Mozafarpoor S., Almasi S., et al. Cutaneous manifestations and considerations in COVID-19 pandemic: a systematic review. Dermatol. Ther. 2020 doi: 10.1111/dth.13986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharifi-Rad M., Anil Kumar N.V., Zucca P., Varoni E.M., Dini L., Panzarini E., et al. Lifestyle, oxidative stress, and antioxidants: back and forth in the pathophysiology of chronic diseases. Front. Physiol. 2020;11(694) doi: 10.3389/fphys.2020.00694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharifi-Rad M., Lankatillake C., Dias D.A., Docea A.O., Mahomoodally M.F., Lobine D., et al. Impact of natural compounds on neurodegenerative disorders: from preclinical to pharmacotherapeutics. J. Clin. Med. 2020;9(4) doi: 10.3390/jcm9041061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharifi-Rad J., Rodrigues C.F., Sharopov F., Docea A.O., Can Karaca A., Sharifi-Rad M., et al. Diet, lifestyle and cardiovascular diseases: linking pathophysiology to cardioprotective effects of natural bioactive compounds. Int. J. Environ. Res. Publ. Health. 2020;17(7):2326. doi: 10.3390/ijerph17072326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi S., Qin M., Shen B., Cai Y., Liu T., Yang F., et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in wuhan, China. JAMA Cardiol. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sidaway P. COVID-19 and cancer: what we know so far. Nat. Rev. Clin. Oncol. 2020;17(6):336. doi: 10.1038/s41571-020-0366-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Simone V., Franzè E., Ronchetti G., Colantoni A., Fantini M.C., Di Fusco D., et al. Th17-type cytokines, IL-6 and TNF-α synergistically activate STAT3 and NF-kB to promote colorectal cancer cell growth. Oncogene. 2015;34(27):3493–3503. doi: 10.1038/onc.2014.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh A.K., Gupta R., Ghosh A., Misra A. Diabetes in COVID-19: prevalence, pathophysiology, prognosis and practical considerations. Diabetes & metabolic syndrome. 2020;14(4):303–310. doi: 10.1016/j.dsx.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skalny A.V., Rink L., Ajsuvakova O.P., Aschner M., Gritsenko V.A., Alekseenko S.I., et al. Zinc and respiratory tract infections: perspectives for COVID-19 (Review) Int. J. Mol. Med. 2020;46(1):17–26. doi: 10.3892/ijmm.2020.4575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small B.A., Dressel S.A., Lawrence C.W., Drake D.R., 3rd, Stoler M.H., Enelow R.I., et al. CD8(+) T cell-mediated injury in vivo progresses in the absence of effector T cells. J. Exp. Med. 2001;194(12):1835–1846. doi: 10.1084/jem.194.12.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stancioiu F., Papadakis G.Z., Kteniadakis S., Izotov B.N., Coleman M.D., Spandidos D.A., et al. A dissection of SARS-CoV2 with clinical implications (Review) Int. J. Mol. Med. 2020;46(2):489–508. doi: 10.3892/ijmm.2020.4636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tahamtan A., Ardebili A. Real-time RT-PCR in COVID-19 detection: issues affecting the results. Expert Rev. Mol. Diagn. 2020;20(5):453–454. doi: 10.1080/14737159.2020.1757437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanase A., Colita A., Ianosi G., Neagoe D., Branisteanu D.E., Calina D., et al. Rare case of disseminated fusariosis in a young patient with graft vs. host disease following an allogeneic transplant. Exp Ther Med. 2016;12(4):2078–2082. doi: 10.3892/etm.2016.3562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Lancet O. COVID-19: global consequences for oncology. Lancet Oncol. 2020;21(4):467. doi: 10.1016/S1470-2045(20)30175-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Novel Coronavirus Pneumonia Emergency Response Epidemiology T. Vital Surveillances: the Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19) — China, 20202020; 2(8):[113-122 pp.]. [PMC free article] [PubMed]
- Tian S., Xiong Y., Liu H., Niu L., Guo J., Liao M., et al. Pathological study of the 2019 novel coronavirus disease (COVID-19) through postmortem core biopsies. Mod. Pathol. 2020;33(6):1007–1014. doi: 10.1038/s41379-020-0536-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torequl Islam M., Nasiruddin M., Khan I.N., Mishra S.K., Kudrat-E-Zahan M., Alam Riaz T., et al. A perspective on emerging therapeutic interventions for COVID-19. Frontiers in Public Health. 2020;8(281) doi: 10.3389/fpubh.2020.00281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torequl Islam M., Nasiruddin M., Khan I.N., Mishra S.K., Kudrat-E-Zahan M., Alam Riaz T., et al. A perspective on emerging therapeutic interventions for COVID-19. Frontiers in public health. 2020;8:281. doi: 10.3389/fpubh.2020.00281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toscano G., Palmerini F., Ravaglia S., Ruiz L., Invernizzi P., Cuzzoni M.G., et al. Guillain–barré syndrome associated with SARS-CoV-2. N. Engl. J. Med. 2020;382(26):2574–2576. doi: 10.1056/NEJMc2009191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trejo-Gabriel-Galán J.M. Stroke as a complication and prognostic factor of COVID-19. Neurologia. 2020;35(5):318–322. doi: 10.1016/j.nrl.2020.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai L.K., Hsieh S.T., Chang Y.C. Neurological manifestations in severe acute respiratory syndrome. Acta Neurol. Taiwan. 2005;14(3):113–119. [PubMed] [Google Scholar]
- Tsatsakis A., Docea A.O., Calina D., Tsarouhas K., Zamfira L.-M., Mitrut R., et al. A mechanistic and pathophysiological approach for stroke associated with drugs of abuse. J. Clin. Med. 2019;8(9):1295. doi: 10.3390/jcm8091295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsatsakis A., Petrakis D., Nikolouzakis T.K., Docea A.O., Calina D., Vinceti M., et al. COVID-19, an opportunity to reevaluate the correlation between long-term effects of anthropogenic pollutants on viral epidemic/pandemic events and prevalence. Food Chem. Toxicol. 2020;141:111418. doi: 10.1016/j.fct.2020.111418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turtle L. Respiratory failure alone does not suggest central nervous system invasion by SARS-CoV-2. J. Med. Virol. 2020;92(7):705–706. doi: 10.1002/jmv.25828. [DOI] [PubMed] [Google Scholar]
- Ulhaq Z.S., Soraya G.V. Interleukin-6 as a potential biomarker of COVID-19 progression. Med. Maladies Infect. 2020;50(4):382–383. doi: 10.1016/j.medmal.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulrich H., Pillat M.M. CD147 as a target for COVID-19 treatment: suggested effects of azithromycin and stem cell engagement. 2020;16(3):434–440. doi: 10.1007/s12015-020-09976-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vageli D.P., Doukas S.G., Spock T., Sasaki C.T. Curcumin prevents the bile reflux-induced NF-κB-related mRNA oncogenic phenotype, in human hypopharyngeal cells. J. Cell Mol. Med. 2018;22(9):4209–4220. doi: 10.1111/jcmm.13701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaira L.A., Salzano G., Deiana G., De Riu G. Anosmia and ageusia: common findings in COVID-19 patients. Laryngoscope. 2020;130(7):1787. doi: 10.1002/lary.28692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virani A., Rabold E., Hanson T., Haag A., Elrufay R., Cheema T., et al. Guillain-Barré Syndrome associated with SARS-CoV-2 infection. IDCases. 2020;20 doi: 10.1016/j.idcr.2020.e00771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vivarelli S., Falzone L., Grillo C.M., Scandurra G., Torino F., Libra M. Cancer management during COVID-19 pandemic: is immune checkpoint inhibitors-based immunotherapy harmful or beneficial? Cancers. 2020;12(8):2237. doi: 10.3390/cancers12082237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Probin V., Zhou D. Cancer therapy-induced residual bone marrow injury-Mechanisms of induction and implication for therapy. Curr. Canc. Ther. Rev. 2006;2(3):271–279. doi: 10.2174/157339406777934717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in wuhan, China. J. Am. Med. Assoc. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y.Y., Jin Y.H., Ren X.Q., Li Y.R., Zhang X.C., Zeng X.T., et al. Updating the diagnostic criteria of COVID-19 "suspected case" and "confirmed case" is necessary. Mil Med Res. 2020;7(1):17. doi: 10.1186/s40779-020-00245-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F., Wang H., Fan J., Zhang Y., Wang H., Zhao Q. Pancreatic injury patterns in patients with coronavirus disease 19 pneumonia. Gastroenterology. 2020;159(1):367–370. doi: 10.1053/j.gastro.2020.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wargo J.A., Reuben A., Cooper Z.A., Oh K.S., Sullivan R.J. Immune effects of chemotherapy, radiation, and targeted therapy and opportunities for combination with immunotherapy. Semin. Oncol. 2015;42(4):601–616. doi: 10.1053/j.seminoncol.2015.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei X.-S., Wang X., Niu Y.-R., Ye L.-L., Peng W.-B., Wang Z.-H., et al. Diarrhea is associated with prolonged symptoms and viral carriage in corona virus disease 2019. Clin. Gastroenterol. Hepatol. : the official clinical practice journal of the American Gastroenterological Association. 2020;18(8) doi: 10.1016/j.cgh.2020.04.030. 1753-9.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wojciechowski V.V., Calina D., Tsarouhas K., Pivnik A.V., Sergievich A.A., Kodintsev V.V., et al. A guide to acquired vitamin K coagulophathy diagnosis and treatment: the Russian perspective. Daru. 2017;25(1):10. doi: 10.1186/s40199-017-0175-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H.Y.F., Lam H.Y.S., Fong A.H.-T., Leung S.T., Chin T.W.-Y., Lo C.S.Y., et al. Frequency and distribution of chest radiographic findings in COVID-19 positive patients. Radiology. 2019:201160. doi: 10.1148/radiol.2020201160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese center for disease control and prevention. J. Am. Med. Assoc. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- Wu Y., Xu X., Chen Z., Duan J., Hashimoto K., Yang L., et al. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020;87:18–22. doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Guo C., Tang L., Hong Z., Zhou J., Dong X., et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;5(5):434–435. doi: 10.1016/S2468-1253(20)30083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wykes M.N., Lewin S.R. Immune checkpoint blockade in infectious diseases. Nat. Rev. Immunol. 2018;18(2):91–104. doi: 10.1038/nri.2017.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia Y., Shen S., Verma I.M. NF-κB, an active player in human cancers. Cancer Immunol Res. 2014;2(9):823–830. doi: 10.1158/2326-6066.CIR-14-0112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia Y., Jin R., Zhao J., Li W., Shen H. Risk of COVID-19 for patients with cancer. Lancet Oncol. 2020;21(4) doi: 10.1016/S1470-2045(20)30150-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia J., Tong J., Liu M., Shen Y., Guo D. Evaluation of coronavirus in tears and conjunctival secretions of patients with SARS-CoV-2 infection. J. Med. Virol. 2020;92(6):589–594. doi: 10.1002/jmv.25725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiang P., Xu X., Gao L., Wang H., Xiong H., Li R. First case of 2019 novel coronavirus disease with encephalitis. China. 2020;202003 [Google Scholar]
- Xie X., Zhong Z., Zhao W., Zheng C., Wang F., Liu J. Chest CT for typical 2019-nCoV pneumonia: relationship to negative RT-PCR testing. Radiology. 2020:200343. doi: 10.1148/radiol.2020200343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu L., Liu J., Lu M., Yang D., Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. : official journal of the International Association for the Study of the Liver. 2020;40(5):998–1004. doi: 10.1111/liv.14435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan R., Zhang Y., Li Y., Xia L., Guo Y., Zhou Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science (New York, NY) 2020;367(6485):1444–1448. doi: 10.1126/science.abb2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H., Qi H., Ren J., Cui J., Li Z., Waldum H.L., et al. Involvement of NF-κB/IL-6 pathway in the processing of colorectal carcinogenesis in colitis mice. Int. J. Inflamm. 2014;2014:130981. doi: 10.1155/2014/130981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeo C., Kaushal S., Yeo D. Enteric involvement of coronaviruses: is faecal-oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol Hepatol. 2020;5(4):335–337. doi: 10.1016/S2468-1253(20)30048-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeoh C.B., Lee K.J., Rieth E.F., Mapes R., Tchoudovskaia A.V., Fischer G.W., et al. COVID-19 in the cancer patient. Anesth. Analg. 2020;131(1):16–23. doi: 10.1213/ANE.0000000000004884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J., Ouyang W., Chua M.L.K., Xie C. SARS-CoV-2 transmission in patients with cancer at a tertiary care hospital in wuhan, China. JAMA Oncology. 2020;6(7):1108–1110. doi: 10.1001/jamaoncol.2020.0980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z., Rigas B. NF-κB, inflammation and pancreatic carcinogenesis: NF-κB as a chemoprevention target (Review) Int. J. Oncol. 2006;29(1):185–192. [PubMed] [Google Scholar]
- Zhang C., Shi L., Wang F.S. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5(5):428–430. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46(4):586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H., Shen D., Zhou H., Liu J., Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 2020;19(5):383–384. doi: 10.1016/S1474-4422(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H., Shen D., Zhou H., Liu J., Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 2020;19(5):383–384. doi: 10.1016/S1474-4422(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Z., Peng F., Xu B., Zhao J., Liu H., Peng J., et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J. Infect. 2020;S0163–4453(20):30234–30236. doi: 10.1016/j.jinf.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y.-Y., Ma Y.-T., Zhang J.-Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020;17(5):259–260. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Fu B., Zheng X., Wang D., Zhao C., qi Y., et al. Pathogenic T cells and inflammatory monocytes incite inflammatory storm in severe COVID-19 patients. National Science Review. 2020:nwaa041. doi: 10.1093/nsr/nwaa041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zlatian O.M., Comănescu M.V., Roşu A.F., Roşu L., Cruce M., Găman A.E., et al. Histochemical and immunohistochemical evidence of tumor heterogeneity in colorectal cancer. Rom. J. Morphol. Embryol. 2015;56(1):175–181. [PubMed] [Google Scholar]
- Zlatian O., Balasoiu A.T., Balasoiu M., Cristea O., Docea A.O., Mitrut R., et al. Antimicrobial resistance in bacterial pathogens among hospitalised patients with severe invasive infections. Exp Ther Med. 2018;16(6):4499–4510. doi: 10.3892/etm.2018.6737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou X., Chen K., Zou J., Han P., Hao J., Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front. Med. 2020;14(2):185–192. doi: 10.1007/s11684-020-0754-0. [DOI] [PMC free article] [PubMed] [Google Scholar]