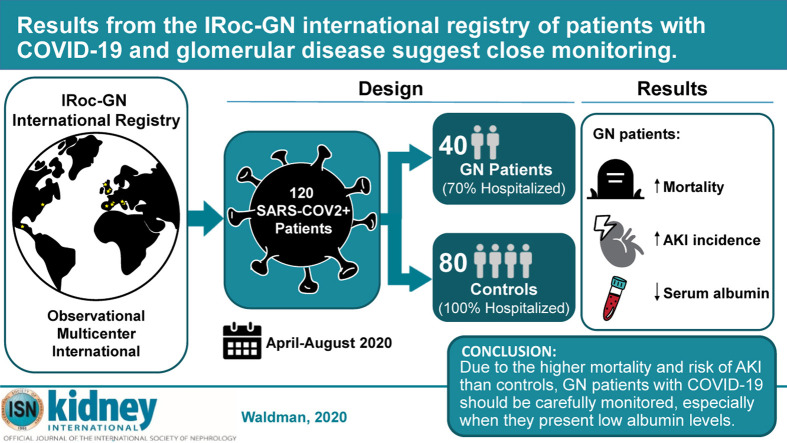
Abstract
The effects of SARS-CoV-2 infection on individuals with immune-mediated glomerulonephritis, who are often undergoing immunosuppressive treatments, are unknown. Therefore, we created the International Registry of COVID infection in glomerulonephritis (IRoc-GN) and identified 40 patients with glomerulonephritis and COVID-19 followed in centers in North America and Europe. Detailed information on glomerulonephritis diagnosis, kidney parameters, and baseline immunosuppression prior to infection were recorded, as well as clinical presentation, laboratory values, treatment, complications, and outcomes of COVID-19. This cohort was compared to 80 COVID-positive control cases from the general population without glomerulonephritis matched for the time of infection. The majority (70%) of the patients with glomerulonephritis and all the controls were hospitalized. Patients with glomerulonephritis had significantly higher mortality (15% vs. 5%, respectively) and acute kidney injury (39% vs. 14%) than controls, while the need for kidney replacement therapy was not statistically different between the two groups. Receiving immunosuppression or renin-angiotensin-aldosterone system inhibitors at presentation did not increase the risk of death or acute kidney injury in the glomerulonephritis cohort. In the cohort with glomerulonephritis, lower serum albumin at presentation and shorter duration of glomerular disease were associated with greater risk of acute kidney injury and need for kidney replacement therapy. No differences in outcomes occurred between patients with primary glomerulonephritis versus glomerulonephritis associated with a systemic autoimmune disease (lupus or vasculitis). Thus, due to the higher mortality and risk of acute kidney injury than in the general population without glomerulonephritis, patients with glomerulonephritis and COVID-19 should be carefully monitored, especially when they present with low serum albumin levels.
Keywords: AKI, COVID-19, glomerulonephritis, IRoc-GN, proteinuria, SARS-CoV-2
Graphical abstract
Since the emergence of the novel coronavirus disease 2019 (COVID-19) due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in December 2019, our understanding of the clinical consequences of COVID-19 on the kidney and the implications of infection in patients with kidney disease have grown. Urinary abnormalities and acute kidney injury are common in patients with COVID-19 from the general population.1, 2, 3 The susceptibility of individuals with kidney disease to SARS-CoV-2 is unclear, but available evidence suggests that patients with chronic kidney disease,4 renal transplantation,5, 6, 7, 8 or on dialysis9 have higher mortality from COVID-19 than the general population.
Nonetheless, there are no published data regarding the outcomes of COVID-19 in adult patients with underlying glomerular diseases.10 , 11 Although this is a heterogeneous group of patients spanning a wide age range and encompassing diverse disease mechanisms, it is reasonable to believe that these patients may represent a particularly vulnerable group with a heightened risk of severe disease manifestations due to an interplay of factors related to the immunosuppressive medications, underlying immune dysfunction, and comorbidities.
Given the relative rarity of glomerular diseases, we created an International Registry of COVID infection in glomerulonephritis (IRoc-GN) to characterize the clinical course of COVID-19, identify risk factors associated with severe complications, and evaluate nonrenal and renal outcomes in glomerulonephritis (GN) patients. We compared outcomes of GN patients with those of a control cohort of COVID-19–positive individuals from the general population without GN.
Results
Patients
Among more than 5000 patients with GN actively followed at the participating centers in 5 countries (Supplementary Table S1), 48 cases with biopsy-proven GN and COVID-19 and 80 hospitalized controls without GN were entered into the registry from 20 April 2020 to 20 August 2020. Eight GN cases (2 on hemodialysis before COVID-19, 3 renal transplant recipients, 1 with thrombotic microangiopathy, 2 duplicates) were excluded from the analysis. Therefore, this study included 120 patients: 40 patients with GN and 80 controls.
Age and sex were similar between GN patients and controls (Table 1 ). GN patients had lower Hispanic/Latino representation than controls (48% vs. 79%, P < 0.001). Hypertension and diabetes were the most common comorbidities and were present with similar frequency in both groups. There was a higher prevalence of obesity in the GN group, but BMI was not statistically different from controls.
Table 1.
Baseline characteristics of GN patients and controls with COVID-19
| Characteristics | GN (n = 40) | Death |
Controls (n = 80) | Death |
P value | ||
|---|---|---|---|---|---|---|---|
| Yes (n = 6) | No (n = 34) | Yes (n = 4) | No (n = 76) | ||||
| Age (yr) | 60.3 ± 17.7 | 70.2 ± 10.5 | 58.5 ± 18.2 | 62.5 ± 15.6 | 72.0 ± 13.6 | 62.0 ± 15.6 | 0.21 |
| Sex, n (%) | |||||||
| Female | 23 (57.5) | 3 (50.0) | 20 (58.8) | 30 (37.5) | 1 (25.0) | 29 (38.2) | 0.051 |
| Male | 17 (42.5) | 3 (50.0) | 14 (41.2) | 50 (62.5) | 3 (75.0) | 47 (61.8) | |
| Race | 33 (82.5) | 6 (100.0) | 27 (79.4) | 72 (90.0) | 4 (100.0) | 68 (89.5) | 0.38 |
| White | 3 (7.5) | 0 | 3 (8.8) | 5 (6.3) | 0 | 5 (6.6) | |
| Black/AAm | 3 (7.5) | 0 | 3 (8.8) | 3 (3.8) | 0 | 3 (4.0) | |
| Other | 1 (2.5) | 0 | 1 (2.9) | 0 | 0 | 0 | |
| Ethnicity | <0.001 | ||||||
| Hispanic/Latino | 19 (47.5) | 4 (66.7) | 15 (44.1) | 63 (78.8) | 4 (100.0) | 56 (73.7) | |
| BMI | 27.7 ± 6.3 (n = 26) | 32.0 ± 7.2 (n = 5) | 26.7 ± 5.8 (n = 21) | 32.0 ± 16.9 (n = 67) | 42.1 ± 6.3b | 29.4 ± 6.3 (n = 63) | 0.16 |
| Glomerular disease | |||||||
| Lupus nephritis | 10 (25.0) | 2 (33.3) | 8 (23.5) | ||||
| Vasculitis | 6 (15.0) | 2 (33.3) | 4 (11.8) | ||||
| FSGS | 5 (12.5) | 0 | 5 (14.7) | ||||
| IgA nephropathy | 5 (12.5) | 0 | 5 (14.7) | ||||
| Membranous nephropathy | 4 (10.0) | 0 | 4 (11.8) | NA | NA | NA | NA |
| Minimal change | 3 (7.5) | 0 | 3 (8.8) | ||||
| Postinfectious | 3 (7.5) | 1 (16.7) | 2 (5.9) | ||||
| AA amyloidosis | 2 (5.0) | 1(16.7) | 1 (2.9) | ||||
| Membranoproliferative | 2 (5.0) | 0 | 2 (5.9) | ||||
| Kidney parameters (pre-COVID-19) | |||||||
| sCr (mg/dl) | 1.41 (0.89–1.91) | 1.72 (1.40–1.87) | 1.29 (0.87–1.93) | 0.90 (0.79–1.01) (n = 70) |
0.90 (0.76–1.14) | 0.90 (0.79–1.01) (n = 66) |
0.006 |
| eGFR (ml/min/1.73 m2) | 47.9 (30.6–80.2) | 32.6 (29.3–39.1) | 49.1 (34.5–87.8) | 83.4 (72.1–92.3) (n = 70) |
79.2 (62.7–86.8) | 83.4 (72.1–97.0) (n = 66) |
<0.001 |
| Serum albumin (g/dl) | 3.54 ± 0.62 (n = 38) | 3.47 ± 0.43 | 3.56 ± 0.66 (n = 32) | 3.92 ± 0.55 (n = 35) | 3.23a (n = 1) | 3.94 ± 0.54 (n = 34) | 0.018 |
| Hematuria | |||||||
| No/negative | 18 (47.4) (n = 38) | 2 (33.3) | 16 (50.0) (n = 32) | ||||
| 1+ | 5 (13.2) | 0 | 5 (15.6) | ||||
| 2+ | 4 (10.5) | 2 (33.3) | 2 (6.3) | ||||
| 3+ | 3 (7.9) | 1 (16.7) | 2 (6.3) | ||||
| Comorbidities | |||||||
| Hypertension | 22 (55.0) | 4 (66.7) | 18 (52.9) | 42 (52.5) | 4 (100.0) | 38 (50.0) | 0.78 |
| Diabetes | 8 (20.0) | 2 (33.3) | 6 (17.7) | 21 (26.3) | 1 (25.0) | 20 (26.3) | 0.51 |
| Obesity | 7 (17.5) | 2 (33.3) | 5 (14.7) | 4 (5.0) | 1 (25.0) | 3 (4.0) | 0.015 |
| Cardiovascular disease | 6 (15.0) | 2 (33.3) | 4 (11.8) | 17 (21.3) | 2 (50.0) | 15 (19.7) | 0.56 |
| COPD | 6 (15.0) | 4 (66.7)c | 2 (5.9) | 3 (3.8) | 0 | 3 (4.0) | 0.081 |
| Asthma | 3 (7.5) | 1 (16.7) | 2 (5.9) | 5 (6.3) | 0 | 5 (6.6) | 0.87 |
| Other chronic lung disease | 2 (5.0) | 1 (16.7) | 1 (2.9) | 2 (2.5) | 0 | 2 (2.6) | 0.41 |
| Liver disease | 3 (7.5) | 2 (33.3) | 1 (2.9) | 3 (3.8) | 0 | 3 (4.0) | 0.38 |
| Stroke | 3 (7.5) | 0 | 3 (8.8) | 0 | 0 | 0 | 0.032 |
| Cancer | 2 (5.0) | 1 (16.7) | 1 (2.9) | 1 (1.3) | 0 | 1 (1.3) | 0.41 |
| HIV/AIDS | 1 (2.5) | 0 | 1 (2.9) | 0 | 0 | 0 | 0.086 |
| Current smoker | 1 (2.5) | 1 (16.7) | 0 | 4 (5.0) | 0 | 4 (5.3) | 0.12 |
| Ex-smoker | 7 (17.5) | 1 (16.7) | 6 (17.7) | 0 | 0 | 0 | <0.001 |
| Clinical presentation | |||||||
| Fever | 31 (77.5) | 6 (100.0) | 25 (73.5) | 61 (76.3) | 3 (75.0) | 58 (76.3) | 0.46 |
| Cough | 25 (62.5) | 4 (66.7) | 21 (61.8) | 21 (26.3) | 1 (25.0) | 20 (26.3) | <0.001 |
| Dyspnea | 16 (40.0) | 5 (83.3)c | 11 (32.4) | 53 (66.3) | 3 (75.0) | 50 (65.8) | 0.016 |
| Fatigue | 14 (35.0) | 3 (50.0) | 11 (32.4) | 4 (5.0) | 0 | 4 (5.3) | <0.001 |
| Myalgia | 13 (32.5) | 3 (50.0) | 10 (29.4) | 6 (7.5) | 0 | 6 (7.9) | <0.001 |
| GI symptoms | 11 (27.5) | 1 (16.7) | 10 (29.4) | 24 (30.0) | 0 | 24 (31.6) | 0.92 |
| Anorexia | 8 (20.0) | 1 (16.7) | 7 (20.6) | 2 (2.5) | 0 0 |
2 (2.6) | 0.006 |
| Chills | 4 (10.0) | 1 (16.7) | 3 (8.8) | 4 (5.0) | 0 | 4 (5.3) | 0.14 |
| Nasal congestion | 3 (7.5) | 1 (16.7) | 2 (5.9) | 0 | 1 (25.0) | 0 | 0.010 |
| Sore throat | 3 (7.5) | 0 | 3 (8.8) | 3 (3.8) | 0 | 2 (2.6) | 0.38 |
| Anosmia | 2 (5.0) | 0 | 1 (2.9) | 13 (16.3) | 0 | 13 (17.1) | 0.18 |
| Neurologic symptoms | 1 (2.5) | 0 | 2 (5.9) | 0 | 0 | 0.086 | |
| Use of RAASi at diagnosis | 18 (45.0) | 1 (16.7) | 17 (50.0) | 4 (5.0) | 1 (25.0) | 3 (4.0) | <0.001 |
| Laboratory values at presentation | |||||||
| sCR, mg/dl | 1.93 (1.09–4.36) (n = 27) |
3.67 (2.30–5.04) (n = 2) |
1.93 (1.09–3.81) (n = 25) |
0.83 (0.75–1.06) (n = 77) |
1.03 (0.89–1.73) | 0.83 (0.75–1.05) (n = 73) |
0.022 |
| Serum albumin, g/dl | 2.83 ± 0.88 (n = 22) | 2.75 ± 0.78 (n = 2) | 2.83 ± 0.90 (n = 20) | 3.60 ± 0.86 (n = 62) | 2.67 (n = 1) | 3.61 ± 0.86 (n = 61) | 0.019 |
| Lymphocytes (×1000/ul) | 0.9 ± 0.6 (n = 30) | 0.8 ± 0.4 (n = 5) | 0.9 ± 0.6 (n = 25) | 1.2 ± 0.8 (n = 78) | 0.7 ± 0.2c | 1.3 ± 0.8 (n = 74) | 0.041 |
| Neutrophils (×1000/ul) | 6.4 ± 3.6 (n = 30) | 7.4 ± 3.1 (n = 5) | 6.2 ± 379 (n = 25) | 6.2 ± 2.5 (n = 28) | 4.44 (n = 1) | 6.3 ± 2.5 (n = 27) | 0.86 |
| Hemoglobin (g/dl) | 11.1 ± 2.0 (n = 30) | 11.0 ± 1.7 (n = 5) | 11.2 ± 2.1 (n = 25) | 12.9 ± 2.7 (n = 30) | 14.6 (n = 1) | 12.9 ± 2.8 (n = 29) | 0.009 |
| Platelets (×109/l) | 242.6 ± 108.1 (n = 30) | 230.2 ± 107.5 (n = 5) | 245.1 ± 110.3 (n = 25) | 246.2 ± 130.5 (n = 31) | 140.0 (n = 1) | 249.7 ± 131.2 (n = 30) | 0.96 |
| Ferritin (ng/ml) | 694 (455–1637) (n = 19) |
694 (506–1708) (n = 3) |
720 (338–1564) (n = 16) |
544.5 (220.5–1015.5) (n = 60) |
364.7 (n = 1) | 555.0 (201.0–1042.0) (n = 59) |
0.88 |
| CRP (mg/L) | 23.1 (6.1–72.6) (n = 27) |
19.8 (2.7–40.5) (n = 4) |
23.1 (6.3–94.0) (n = 23) |
82.5 (24.9–141.1) (n = 76) |
320.0 (170.7–396.0) | 77.5 (24.9–136.5) (n = 72) |
0.014 |
| D-dimer (ng/ml) | 1170.0 (440.0–2916.5) (n = 20) |
2200.0 (1460.0–2733.0) (n = 3) |
649.0 (409.0–3080.0) (n = 17) |
751.0 (377.0–1207.0) (n = 70) | 1294.0 (592.0–1445.0) (n = 3) |
739.0 (375.0–1109.0) (n = 67) |
0.91 |
AA, amyloid A protein; AAm, African American; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; COVID-19, coronavirus disease 2019; CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; FSGS, focal segmental glomerulosclerosis; GI, gastrointestinal; GN, glomerulonephritis; NA, not applicable; RA, rheumatoid arthritis; RAASi: renin–angiotensin–aldosterone system inhibitors; sCR, serum creatinine; SLE, systemic lupus erythematosus.
Data are mean ± SD, or median (interquartile range); frequencies are n (%). Full age range is 18–90 vs. 32–90 for GN patients versus controls, respectively. Sample sizes are listed if they are fewer than the respective denominators shown in the header row. Percentages may not add to 100 due to rounding or missing or not applicable responses. Pre-COVID refers to the latest values before infection. P values are for GN versus control subjects; comparisons between GN and controls were adjusted for ethnicity. We did not report variables with <40% values available.
Cannot be computed due to n = 1 in 1 of the comparison groups (died n = 1).
P < 0.01 versus number of deaths in the same cohort.
P < 0.05 versus number of deaths in the same cohort.
The most common GN diagnoses were lupus nephritis (25%) and vasculitis (15%), followed by focal segmental glomerulosclerosis, IgA nephropathy, and membranous nephropathy (Table 1). More than one-third of patients suffered from GN for more than 5 years. Before COVID-19, median proteinuria was in the subnephrotic range (1.0 [0.33–3.20] g/d), and hematuria was present in 43% of patients. Half of GN patients were taking immunosuppressants at the time of COVID-19 diagnosis (Table 2 ); the majority (65%) of patients on immunosuppression had a diagnosis of lupus nephritis or vasculitis (Supplementary Table S2). One-third of GN patients were on oral corticosteroids (85% of whom were on 5–10 mg/d), 18% were on mycophenolate mofetil, 5% on calcineurin inhibitor, 5% on azathioprine, and 18% were on hydroxychloroquine. Four (10%) patients had received rituximab before COVID-19 diagnosis, and 3 of them received it within 1 month of diagnosis.
Table 2.
Immunosuppression and use of RAAS inhibitors at time of COVID-19 diagnosis in all GN patients versus hospitalized GN patients
| All GN (n = 40) | Death |
P value | Hospitalized (n = 28) | Death |
P value | |||
|---|---|---|---|---|---|---|---|---|
| Yes (n = 6) | No (n = 34) | Yes (n = 5) | No (n = 23) | |||||
| Immunosuppressiona | 20 (50.0) | 5 (83.3) | 15 (44.1) | 0.18 | 15 (53.6) | 5 (100.0) | 10 (43.5) | 0.044 |
| Steroids | 13 (32.5) | 4 (66.7) | 9 (26.5) | 0.075 | 11 (39.3) | 4 (80.0) | 7 (30.4) | 0.062 |
| Mycophenolate mofetil/mycophenolic acid | 7 (17.5) | 1 (16.7) | 6 (17.7) | 1.0 | 7 (25.0) | 1 (20.0) | 6 (26.1) | 1.0 |
| Hydroxychloroquine | 7 (17.5) | 1 (16.7) | 6 (17.7) | 1.0 | 5 (17.9) | 1 (20.0) | 4 (17.4) | 1.0 |
| Rituximab | 4 (10.0) | 2 (33.3) | 2 (5.9) | 0.10 | 4 (14.3) | 2 (40.0) | 2 (8.7) | 0.14 |
| Calcineurin inhibitor | 2 (5.0) | 0 | 2 (5.9) | 1.0 | 1 (3.6) | 0 | 1 (4.4) | 1.0 |
| Azathioprine | 2 (5.0) | 1 (16.7) | 1 (2.9) | 0.28 | 1 (3.6) | 1 (20.0) | 0 | 0.18 |
| Methotrexate | 2 (5.0) | 0 | 2 (5.9) | 1.0 | 1 (3.6) | 0 | 1 (4.4) | 1.0 |
| Sulfasalazine | 1 (2.5) | 0 | 1 (2.9) | 1.0 | 1 (3.6) | 0 | 1 (4.4) | 1.0 |
| Use of RAASi at diagnosis | ||||||||
| ARB | 11 (27.5) | 1 (16.7) | 10 (29.4) | 1.0 | 8 (28.6) | 1 (20.0) | 7 (30.4) | 1.0 |
| ACE inhibitors (n = 39) | 7 (18.0) | 0 | 7 (21.2) | 0.57 | 4 (14.8) (n = 27) |
0 | 4 (18.2) (n = 22) |
0.56 |
ACEI, angiotensin converting enzyme inhibitors; ARB, Angiotensin receptor blockers; COVID-19, coronavirus disease 2019; GN, glomerulonephritis; RAAS, renin–angiotensin–aldosterone system; RAASi, renin–angiotensin–aldosterone system inhibitors.
Data are n (%).
At time of infection.
Before COVID-19, GN patients had higher serum creatinine (1.41 [0.89–1.91] vs. 0.90 [0.79–1.01] mg/dl, respectively, P = 0.006), lower glomerular filtration rate (47.9 [30.6–80.2] vs. 83.4 [72.1–92.3] ml/min/1.73 m2, P < 0.001), and lower serum albumin (3.54 ± 0.62 vs. 3.92 ± 0.55 g/dl, P = 0.018) than controls (Table 1). At the time of COVID-19 diagnosis, almost half of the GN patients compared with only 5% of controls were on renin–angiotensin–aldosterone inhibitors (RAASi) (P < 0.001).
COVID-19 clinical presentation and disease course
The most common presenting symptom of COVID-19 in both cohorts was fever (Table 1). Fatigue, cough, myalgia, anorexia, and nasal congestion were more common in GN patients, whereas dyspnea was more common in controls.
The majority of GN patients (70%) and all controls were hospitalized. At COVID-19 presentation, serum creatinine in GN patients was higher [1.93 (1.09–4.36) vs. 0.83 (0.75–1.05) mg/dl, P = 0.022], whereas serum albumin (2.83 ± 0.88 vs. 3.60 ± 0.86 g/dl, P = 0.019), hemoglobin (11.1 ± 2.0 vs. 12.9 ± 2.7 g/dl, respectively, P = 0.009), and lymphocyte counts (0.9 ± 0.6 vs. 1.2 ± 0.8 × 1000/ul, P = 0.041) were significantly lower than in controls (Table 1). C-reactive protein values were higher in the control group. There were no differences in other inflammatory markers, but the interpretation is limited by missing values.
Management
COVID-19–targeted therapies
The most frequently used therapies for COVID-19 were hydroxychloroquine, antivirals, and azithromycin (Table 3 ). There was more frequent use of hydroxychloroquine and azithromycin in controls but similar use of corticosteroids, interleukin-6 receptor antagonist (tocilizumab), and antibiotics in both groups. Almost half of GN and control patients received prophylactic anticoagulation.
Table 3.
COVID-19–related therapies among GN and control patients
| GN patients (n = 40) | Controls (n = 80) | P value | |
|---|---|---|---|
| Antivirals | |||
| Hydroxychloroquine | 15 (37.5) | 60 (75.0) | <0.001 |
| Azithromycin | 10 (25.0) | 37 (46.3) | 0.008 |
| Lopinavir | 5 (12.5) | 12 (15.0) | 0.72 |
| Ritonavir | 3 (7.5) | 10 (12.5) | 0.27 |
| Other antivirals | 2 (5.0) | 1 (1.3) | 0.21 |
| Darunavir | 1 (2.5) | 2 (2.5) | 0.35 |
| Antiinflammatory | |||
| Oral steroids | 7 (17.5) | 9 (11.3) | 0.47 |
| IV methylprednisolonea | 5 (12.5) | 25 (31.3) | 0.052 |
| Tocilizumab | 2 (5.0) | 11 (13.8) | 0.095 |
| Antibiotics | 2 (5.0) | 10 (12.5) | 0.23 |
| Anticoagulants | 19 (48.7) (n = 39) |
39 (48.8) | 0.58 |
COVID-19, coronavirus disease 2019; GN, glomerulonephritis.
Data are n (%) and may not add up to 100 due to rounding or missing/not available responses. Sample sizes are listed if they are fewer than the respective denominators shown in the header row. Comparisons between GN and controls were adjusted for ethnicity.
Range for total IV methylprednisolone dosage was 200 to 750 mg for GN patients and 300 to 1000 mg for controls.
Management of immunosuppression in GN patients during infection
Among 20 GN patients on immunosuppression at the time of COVID-19 presentation, antimetabolites (azathioprine or mycophenolate) were discontinued in most (89%), and calcineurin inhibitors were discontinued in 1 patient. Steroids were maintained with the same dosage in 61%, increased in 31% (predominantly due to concerns of adrenal insufficiency or flare of underlying GN), and discontinued in 8%. Patients on hydroxychloroquine before infection were maintained on this treatment. RAASi were discontinued in 7 GN patients (39%) due to the presence of acute kidney injury (57%), hypotension (29%), hyperkalemia (14%), and theoretical concerns about enhancing SARS-CoV-2 viral entry (14%).
Death and nonrenal complications
More deaths occurred in GN patients compared with controls (15 % vs. 5%, P = 0.026) (Table 4 ). On the basis of multivariable analyses, the odds ratio (OR) of death in GN patients was 6.33 times higher than in controls (95% confidence interval [CI]: 1.38–29.04, P = 0.018) and older age associated with death in the study cohort overall (OR = 1.05. 95% CI: 1.002–1.102 P = 0.042). Among GN patients, nonsurvivors were more likely to have chronic obstructive pulmonary disease (67% vs. 6%, P = 0.002) and dyspnea at COVID-19 presentation (83% vs. 32%, P = 0.029) (Table 1). Among controls, nonsurvivors had higher BMI (42.1 ± 6.3 vs. 29.4 ± 6.3, P = 0.004) and lower lymphocytes (0.7 ± 0.2 vs. 1.3 ± 0.8 × 1000/ul, P = 0.023) than survivors. The use of RAASi was not associated with a significant difference in mortality in GN cases (P = 0.20) or in controls (P = 0.19).
Table 4.
Nonrenal and renal complications and outcomes of GN and control patients during COVID-19 infection
| GN patients (n = 40) | Controls (n = 80) | P value | |
|---|---|---|---|
| Hospitalized | 28 (70.0) | 80 (100.0) | <0.001 |
| Description of illness | |||
| Pneumonia | 21 (52.5) | 41 (51.3) | 0.66 |
| ARDS, noninvasive ventilation | 3 (7.5) | 22 (27.5) | 0.12 |
| ARDS, intubated | 3 (7.5) | 8 (10.0) | 0.27 |
| Days of intubation | 9.0 (4.0–14.0) (n = 2) | 16.0 (10.0–37.0) | 0.99 |
| Septic shock | 2 (5.0) | 3 (3.8) | 0.86 |
| Inotropes or other vasopressors | 2 (7.1) (n = 28) | 6 (8.1) | 0.55 |
| Developed AKI | 15 (38.5) (n = 39) | 11 (13.9) | 0.047 |
| Stage 1 | 3 (25.0) | 2 (18.2) | 0.82 |
| Stage 2 | 3 (25.0) | 2 (18.2) | |
| Stage 3 | 6 (50.0) (n = 12) | 7 (63.6) (n = 11) |
|
| RRT new onset | 7 (17.5) | 5 (6.3) | 0.40 |
| Days of RRT | 25.0 (15.0–33.0) (n = 6)a | 30.0 (26.0–30.0) | 0.40 |
| Discharged on RRT | 4 (80.0) (n = 5) | 1 (20.0) | 0.13 |
| Nonrenal complications | |||
| Cardiac | |||
| MI | 2 (5.6) | 0 | 0.12 |
| Arrhythmia | 1 (2.8) | 6 (7.9) | 0.034 |
| GI | 3 (8.1) (n = 36) | 5 (6.6) (n = 76) | 0.76 |
| CNS | 1 (2.7) (n = 36) | 2 (2.6) (n = 76) | 0.44 |
| Hematologic | |||
| PE | 2 (5.0) | 0 | 0.16 |
| DVT | 2 (5.0) | 3 (5.0) | 0.60 |
| Superimposed bacterial infection | 6 (15.8) (n = 38) | 8 (10.7) | 0.91 |
| Length of hospital stay | 15.0 (8.0–21.0) (n = 27) | 7.0 (5.0–12.0) (n = 77) | 0.29 |
| Death | 6 (15.0) | 4 (5.0) | 0.026 |
AKI, acute kidney injury; ARDS, acute respiratory distress syndrome; CNS, central nervous system; COVID-19, coronavirus disease 2019; DVT, deep vein thrombosis; GI, gastrointestinal; GN, glomerulonephritis; MI, myocardial infarction; PE, pulmonary embolism; RRT, renal replacement therapy.
Data are n (%), or median (interquartile range). Sample sizes are listed if they are fewer than the respective denominators shown in the header row. Percentages may not add to 100 due to rounding or missing/not available responses. Comparisons of GN versus controls adjust for ethnicity. All comparisons are adjusted for ethnicity.
One patient died while on RRT.
In GN patients, GN diagnosis, estimated glomerular filtration rate, proteinuria, or serum albumin before COVID-19 or at presentation did not significantly differ between survivors and nonsurvivors (Table 1). There was no difference in death between those on immunosuppression (including use of steroids) at the time of COVID-19 versus those not on immunosuppression (P = 0.18) (Tables 2 and 5 ); however, among 15 hospitalized GN patients, all 5 who died were on immunosuppression (100% of 5 vs. 44% of 10, P = 0.044) (Table 2).
Table 5.
Duration of disease and immunosuppression in GN patients stratified by outcome
| GN (n = 40) | AKI any stage (n = 39)a |
RRT requirement |
Death |
||||
|---|---|---|---|---|---|---|---|
| Yes (n = 15) | No (n = 24) | Yes (n = 7) | No (n = 33) | Yes (n = 6) | No (n = 34) | ||
| Duration of GN disease | |||||||
| <6 mo | 11 (27.5) | 7 (46.7)b | 4 (16.7) | 5 (71.4)b | 6 (18.2) | 3 (50.0) | 8 (23.5) |
| 6–12 mo | 2 (5.0) | 1 (6.7) | 1 (4.2) | 0 | 2 (6.1) | 0 | 2 (5.9) |
| 12–24 mo | 6 (15.0) | 2 (13.3) | 4 (16.7) | 0 | 6 (18.2) | 2 (33.3) | 4 (11.8) |
| 2–5 yr | 6 (15.0) | 1 (6.7) | 4 (16.7) | 1 (14.3) | 5 (15.2) | 0 | 6 (17.7) |
| >5 yr | 15 (37.5) | 4 (26.7) | 11 (45.8) | 1 (14.3) | 14 (42.4) | 1 (16.7) | 14 (41.2) |
| Immunosuppression at presentation | 20 (50.0) | 6 (40.0) | 13 (54.2) | 4 (57.1) | 16 (48.5) | 5 (83.3) | 15 (44.1) |
| Steroids | 13 (32.5) | 5 (33.3) | 8 (33.3) | 2 (28.6) | 11 (33.3) | 4 (66.7) | 9 (26.5) |
| Mycophenolate mofetil | 7 (17.5) | 4 (26.7) | 3 (12.5) | 1 (14.3) | 6 (18.2) | 1 (16.7) | 6 (17.7) |
| Hydroxychloroquine | 7 (17.5) | 2 (13.3) | 5 (20.8) | 2 (28.6) | 5 (15.2) | 1 (16.7) | 6 (17.7) |
| Rituximab | 4 (10.0) | 2 (13.3) | 2 (8.3) | 1 (14.3) | 3 (9.1) | 2 (33.3) | 2 (5.9) |
| Calcineurin inhibitor | 2 (5.0) | 0 | 1 (4.2) | 1 (14.3) | 1 (3.0) | 0 | 2 (5.9) |
| Azathioprine | 2 (5.0) | 0 | 2 (8.3) | 0 | 2 (6.1) | 1 (16.7) | 1 (2.9) |
| Methotrexate | 2 (5.0) | 0 | 2 (8.3) | 0 | 2 (6.1) | 0 | 2 (5.9) |
| Sulfasalazine | 1 (2.5) | 0 | 1 (4.2) | 0 | 1 (3.1) | 0 | 1 (2.9) |
| On immunosuppression | |||||||
| Yes | 20 (50.0) | 6 (40.0) | 13 (54.2) | 4 (57.1) | 16 (48.5) | 5 (83.3) | 15 (44.1) |
| No | 20 (50.0) | 9 (60.0) | 11 (45.8) | 3 (42.8) | 17 (51.) | 1 (16.7) | 19 (55.9) |
AKI, acute kidney injury; GN, glomerulonephritis; RRT, renal replacement therapy.
Data are n (%). Sample sizes are listed if they are fewer than the respective denominators shown in the header row. Percentages may not add to 100 due to rounding or missing/not available responses.
One patient died at home; therefore, no information for development of AKI was available.
Observed differences in subsets: duration of GN disease was associated with AKI yes versus no (P = 0.043) and RRT yes versus no (P = 0.023).
Thirteen percent of GN patients and 10% of controls required admission to the intensive care unit. Acute respiratory distress syndrome developed in 15% of GN patients and in 38% of controls (Table 4). There were no differences in percentages of patients intubated, requirement for vasopressors, length of hospital stay, and nonrenal complications in GN versus controls, except for arrhythmias, which were more frequent in controls (3% vs. 8%, P = 0.034). Five of six (83%) controls who developed arrhythmias were receiving treatment with hydroxychloroquine; arrhythmia in the GN patient was not associated with antimalarial treatment. Four (10%) GN patients had venous thromboembolic events, despite prophylactic anticoagulation in 3. Serum albumin levels during infection were lower in GN patients who experienced venous thromboembolic events (1.67 ± 0.38 vs. 2.94 ± 0.83 g/dl, P = 0.048).
There were no significant difference in death between patients with kidney-limited GN versus GN associated with systemic diseases (lupus or vasculitis) (8% vs. 25%, respectively, P = 0.20), or in other non-renal complications (Supplementary Table S3) despite the greater overall density of immunosuppressive agents (81% vs. 29%, P = 0.003), in particular, steroids (63% vs. 13%, P = 0.002) and mycophenolate mofetil (38% vs. 4%, P = 0.011), in those with systemic disease (Supplementary Table S2).
Acute kidney injury and renal replacement therapy
Acute kidney injury occurred more frequently in GN patients than in controls (38% vs. 14%, P = 0.047). The peak stages of acute kidney injury (AKI) are shown in Table 4. There were no significant differences in the need for renal replacement therapy (RRT) between GN patients and controls (18% vs. 6%, P = 0.40). Incidence of AKI was similar between those treated versus not treated with RAASi among GN patients (33% vs. 43%, respectively; P = 0.74) or controls (25% vs. 13%, P = 0.46).
Among GN patients, shorter duration of GN (<6 months) was associated with a higher frequency of AKI (P = 0.043) as well as RRT (P = 0.023). The presence of diabetes was also associated with a higher frequency of RRT in GN patients (57% vs. 12%, P = 0.020). All 5 GN patients treated with i.v. steroids for acute COVID management developed AKI. However, the use of steroids for baseline immunosuppression was not associated with a significant difference in the occurrence of AKI (33% vs. 33%, P = 1.0). Immunosuppression at presentation was similar between those who developed AKI and those who did not (32% vs. 45%, P = 0.51) (Table 5). There was also no difference in the incidence of AKI (38% vs. 40%, P = 1.0) and need for RRT (13% vs. 25%, P = 0.41) between patients with renal limited GN versus GN associated with systemic diseases (lupus or vasculitis) (Supplementary Table S3).
Among controls, AKI occurred more frequently in those with chronic obstructive pulmonary disease (P = 0.049), liver disease (P = 0.049), lower lymphocytes (P = 0.035), and higher D-dimer (P = 0.006); those with higher neutrophil counts were more likely to require RRT (P = 0.012). The use of i.v. steroids for COVID management was not associated with increased risk of AKI in controls (16% vs. 84% for patients who did vs. did not use i.v. steroids, respectively; P = 0.74).
Among GN patients, those with AKI had higher serum creatinine at presentation that those without AKI (2.57 [1.93–5.04] vs. 1.23 [0.76–1.81] mg/dl, P = 0.003) (Supplementary Table S4). They also had significantly lower serum albumin at presentation (2.29 ± 0.86 vs. 3.44 ± 0.44 g/dl, P = 0.001) and after COVID-19 (1.96 ± 0.40 vs. 3.37 ±0.57 g/dl, P < 0.001) compared to those without AKI. In contrast, GN patients who did not develop AKI had serum albumin levels consistently >3 g/dl. Among controls, those with AKI had higher serum creatinine pre-COVID (1.20 [0.80–1.30] vs. 0.89 [0.79–1.00] mg/dl, P = 0.024), and at presentation (2.00 [0.94–6.20] vs. 0.83 [0.74–0.98] mg/dl, P < 0.001) than patients without AKI, but there were no differences in serum albumin.
Changes in renal parameters over time
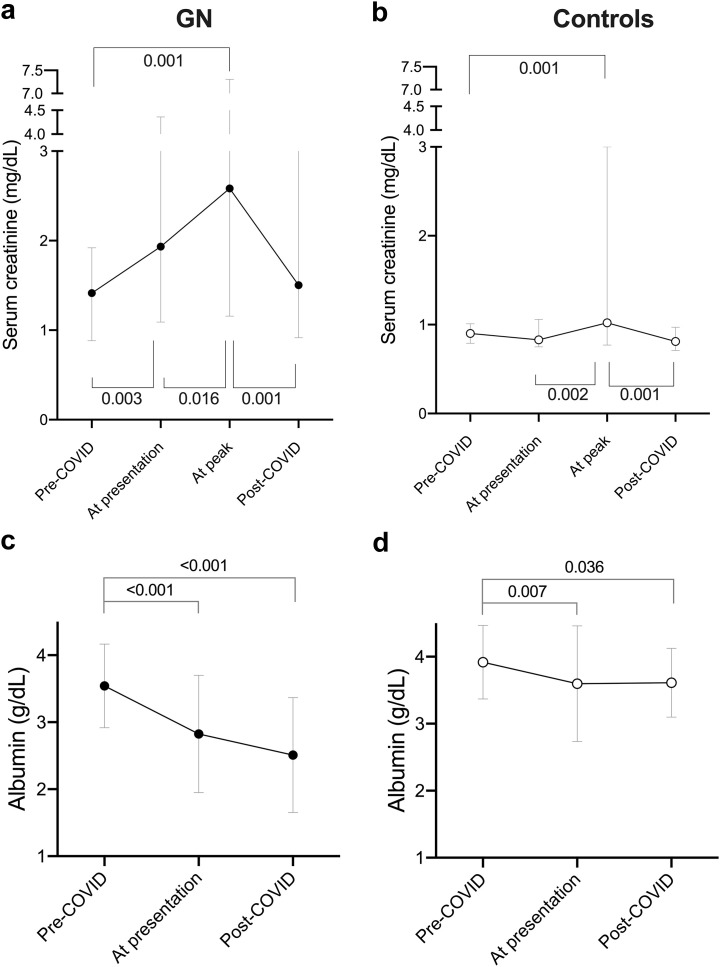
In GN patients, serum creatinine significantly increased from pre-COVID-19 levels to presentation (1.41 [0.89–1.91] vs. 1.93 [1.09–4.36] mg/dl, P = 0.003), peaked at 2.58 [1.23–7.03] mg/dl (P = 0.001 vs. pre-COVID) after a median of 1.0 (0–6.0) days from diagnosis, and returned to pre-COVID levels (1.50 [0.94–3.51] mg/dl, P = 0.17) after median follow-up of 17.0 (9.0–22.0) days (Figure 1 a; Supplementary Table S4). In contrast, in controls, there was no significant increase in serum creatinine at presentation compared with pre-COVID (P = 0.54); serum creatinine peaked at 1.02 (0.77–3.00) mg/dl (P = 0.001 vs. pre-COVID) and, at last follow-up, was similar to pre-COVID levels (Figure 1b; Supplementary Table S4). At all time points, serum creatinine was higher in GN patients than in controls (pre-COVID: P = 0.006, at presentation: P = 0.022 and post-COVID: P = 0.014).
Figure 1.
Changes in (a,b) serum creatinine and (c,d) serum albumin levels pre–coronavirus disease 2019 (COVID-19) infection, at presentation, at peak during COVID-19 infection, and post-COVID-19 in glomerulonephritis patients and controls. Post-COVID is defined by absence of, or marked improvement in, original COVID-19-related symptoms or signs. Data are median mean ± 95% confidence interval.
In GN patients, serum albumin significantly declined from pre-COVID levels to presentation (3.54 ± 0.62 vs. 2.83 ± 0.88 g/dl, P < 0.001) and was significantly lower at the last follow-up (2.51 ± 0.86 g/dl, P < 0.001 vs. pre-COVID) (Figure 1C; Supplementary Table S4). Similarly, in control patients, serum albumin decreased from pre-COVID to presentation (3.92 ±0.55 vs. 3.60 ± 0.86 g/dl, P = 0.007) and persisted at follow-up (3.61 ± 0.51 g/dl, P = 0.036 vs. pre-COVID) (Figure 1D; Supplementary Table S4). However, at all time points, serum albumin was lower in GN patients than in controls (pre-COVID: P = 0.018, at presentation: P = 0.019, post-COVID: P < 0.001)
We had limited repeated measurements of proteinuria in GN patients during COVID-19 (n = 6) and after recovery (n = 4). On the basis of available measurements, median protein excretion appeared to increase from subnephrotic levels (pre-COVID) to nephrotic range at presentation (3.82 [1.48–9.91] g/d, P = 0.064) and appeared to improve after the acute infection (2.08 [0.58–6.12] g/d, P = 0.32) (Supplementary Table S4). However, there was no statistical evidence for these changes due to missing values in the majority. Proteinuria was not assessed in most control patients.
Discussion
To the best of our knowledge, this is the first report of the impact of COVID-19 in patients with underlying GN. We found that the overall mortality rate in the GN cohort was significantly higher compared with controls, despite the fact that all controls were hospitalized. GN and control patients had similar major risk factors for COVID-19 mortality, including older age, male sex, obesity, hypertension, diabetes, cardiovascular disease, and chronic lung disease.12 , 13 Importantly, GN patients and controls were matched for the time of infection, to account for the potentially improved patient management over time in each center. Therefore, our data support the mounting evidence that, in addition to the aforementioned risk factors, preexisting kidney disease is independently associated with severe COVID-19 and mortality.2 , 13 , 14 Consistently, GN patients had lower hemoglobin and serum albumin at presentation compared with controls, findings partially attributable to the degree of chronic kidney disease and proteinuria in GN patients, respectively. Both anemia and hypoalbuminemia have been associated with increased in-hospital mortality in other populations.15, 16, 17, 18
The higher rate of AKI in GN patients than in controls may have contributed to the increased mortality. On the basis of a recent meta-analysis of more than 13,000 COVID-19 patients, the prevalence of AKI is 17%,19 although there is considerable heterogeneity across populations. Nevertheless, the burden of AKI in our GN patients is in the higher range of estimates. COVID-associated AKI portends a worse prognosis20 , 21 and is associated with increased odds of death.20
Among GN patients, those who developed AKI had higher serum creatinine at presentation compared with those without AKI and were more likely to have received IV steroids as part of the management of COVID-19. This latter finding, also reported in other populations,2 may reflect the severity of illness rather than a causal association. However, this trend was not seen in controls in our study, and accruing data suggest reduced mortality associated with corticosteroid treatment for severe COVID-19.22 We also found that the duration of GN diagnosis was shorter in those GN patients that developed AKI. This provocative observation, with many potential explanations, needs confirmation in larger studies.
The etiology of AKI in COVID-19 has not been fully elucidated.23 It is unclear whether unique factors in GN patients (or subgroups), beyond conventional risk factors, increase susceptibility to AKI, particularly in light of the association with shorter disease duration. Notably, our data do not indicate a difference in outcomes between those with systemic versus kidney-limited GN, and the levels of proteinuria and hypoalbuminemia pre-COVID-19 were not associated with increased risk of AKI. However, serum albumin at presentation was significantly lower in GN patients than controls. Moreover, among GN patients, serum albumin was lower in those who developed AKI, those who required RRT, and those with venous thromboembolic events. The extent to which the observed differences in hypoalbuminemia in GN patients is driven by urinary protein excretion, which increased during COVID-19, versus other nonrenal factors (e.g., critical illness, liver dysfunction) is difficult to determine given the limited dataset and lack of characterization of proteinuria (tubular vs. albuminuria). Nevertheless, the presence of hypoalbuminemia may further complicate fluid management in these tenuous patients and may potentiate COVID-19 associated hypercoagulability, both of which could conceivably increase the risk of AKI in GN patients.
Immunosuppressive treatment before COVID-19 was not associated with greater mortality or AKI in GN patients. Although the published data regarding risks of COVID-19 and immunosuppression are still limited,10 our finding is in accordance with other reports of infected patients on immunosuppressive medications for other autoimmune diseases24, 25, 26, 27, 28 or underlying cancer.29 However, prednisone dose ≥10 mg/day was associated with higher odds of hospitalization in the COVID-19 Global Rheumatology Alliance.26 More data are needed to confirm our findings and to determine interactions between different immunosuppressive regimens and COVID-19 severity. Of particular interest in this regard is the effect of B-cell-depleting therapies in COVID-19, given its wide application in GN patients.30 We also found no relationship between the use of RAASi at disease presentation and death or AKI risk, a finding that has been substantiated also in other populations.2 , 31, 32, 33 Therefore, our data do not support the discontinuation of these medications in GN patients with COVID-19.
Perhaps the most unexpected finding was the small number of cases of COVID-19 in GN patients, considering that contributing centers were located in areas with high prevalence of COVID-19 and treat large numbers of GN patients. Given the numbers of GN patients treated at the various contributing centers, the incidence of infection among GN patients during the study period varied widely from 0.3% to 4.1%. However, the true rate of COVID-19 in GN patients is difficult to ascertain for several reasons. Most important, GN patients were not systematically tested for COVID-19. The registry has been active for a short period, dissemination of the surveys has not reached widespread scale, and underreporting may occur as the health care systems are strained. Moreover, during this early period of the pandemic, testing in the ambulatory setting was limited in most countries, likely leading to underestimation of true rates of infection in this population and by extension, the rates of complications. Expanded, widespread SARS-CoV-2 RNA testing of all GN patients and systematic antibody testing for SARS-CoV-2 antibodies will help provide a better estimate of infection rates and characterize the spectrum of the clinical phenotype of infection. We can only speculate whether patients with GN practice better social distancing or if there is a potential “protective” role of certain immunosuppressive medications that prevents or limits the excessive immune activation responsible for severe clinical manifestations.26 , 28 , 30 , 34 Thus far, there is no convincing evidence that any agents protect against severe COVID-19 infection, but these issues warrant further investigation.
To our knowledge, this is the first analysis of COVID-19 in patients with GN with global representation and a diverse population. Inclusion of a control group increased the validity of our findings, and the definitions used for AKI are validated, allowing for meaningful comparison across cohorts.
The study has limitations. The sample size is relatively small, although our network included centers following high volumes of patients with GN. Most participating centers are from Europe with a greater percentage of Spanish patients. Therefore, the generalizability of our findings to other populations may not apply, particularly in light of emerging data regarding racial/ethnic disparities in COVID-19 incidence and mortality.35 The timing of our study cohort deserves consideration. The registry was launched early in the pandemic, leading to selection bias toward more severe hospitalized cases rather than the full spectrum of the disease phenotype. Moreover, there was limited understanding of the disease complications and few proven effective therapies, which influenced the outcomes under investigation. Inflammatory markers were unavailable for many patients, and there was an inconsistent collection of proteinuria and hematuria during infection. Finally, small numbers on immunosuppression preclude meaningful association of each of the individual medications and outcomes.
In conclusion, our data indicate that GN patients have a significant risk of mortality and renal impairment associated with SARS-CoV2 infection. Further studies are required to confirm our findings; improve our understanding of the impact of COVID-19 on this unique population; and determine interactions among COVID-19, underlying GN, and different immunosuppressive regimens, as well as the long-term consequences of COVID-19 on glomerular disease activity and serologic activity.
Methods
Study design and participants
This retrospective cohort study included patients with biopsy-proven GN diagnosed with COVID-19 from centers participating in the IRoc-GN registry. We included cases managed as inpatients or outpatients. For each GN patient entered in the registry, reporters were asked to enter at least 1 age- and sex-matched control patient who was positive for COVID-19 but without GN and with estimated glomerular filtration rate >60 ml/min/1.73 m2. Controls were hospitalized within ± 2 weeks of the case to account for changes in treatment strategies over time that may influence outcomes. Patients on maintenance hemodialysis before infection and kidney transplant recipients were excluded.
Data were deidentified, and a computer-generated code was assigned for each patient entry, allowing reporters to edit or modify entries in the event of a change in patient status. For quality control, erroneous or duplicate reports were identified based on matching age, sex, and glomerular disease diagnosis; reviewed manually; and removed after verification with a reporter.
Data collection
The registry was launched on 20 April 2020. Providers practicing in centers with large numbers of GN patients in North America, Europe, and Australia were sent a link to the secure public survey (https://redcapsurvey.niddk.nih.gov/surveys/?s = FPM87NK7T4) and encouraged to voluntarily report cases of COVID-19 (confirmed by polymerase chain reaction or highly suspected based on symptoms and lung imaging) occurring in adult patients with biopsy-proven GN. We used REDCap (Research Electronic Data Capture),36 , 37 a secure web-based electronic data capture tool hosted by the National Institute of Diabetes and Digestive Kidney Disease, National Institutes of Health, to collect and manage study data. Registry data elements include sociodemographic information, comorbidities, GN diagnosis and duration, and use of RAASi and immunosuppressive medications at the time of infection. We collected data on COVID-19–related symptoms, therapies, management of immunosuppression during infection, outcomes, the maximum level of care, nonrenal complications, AKI, need for RRT, disposition at the time of report (i.e., recovery, death). Laboratory parameters collected were serum creatinine, serum albumin, proteinuria, hematuria, ferritin, complete blood count, interleukin-6 concentrations, D-dimer, lactate dehydrogenase, C-reactive protein, erythrocyte sedimentation rate, and serologies if relevant. Renal parameters were collected at various time points: (i) pre-COVID (latest available before infection onset), (ii) at presentation, (iii) peak, and (iv) post-COVID as defined by the absence of, or marked improvement in, original COVID-related symptoms or signs. All data were checked for quality by 2 physicians (PC and MW) and a third researcher (NS).
Definitions
AKI is defined by Kidney Disease Improving Global Outcomes (KDIGO) criteria as follows: stage 1, increase in baseline serum creatinine by ≥0.3 mg/dl within 48 h or a 1.5 to 1.9 times increase in serum creatinine from baseline within 7 days; stage 2, 2 to 2.9 times increase in baseline serum creatinine within 7 days; stage 3, ≥3 times increase in serum creatinine within 7 days or increase to ≥4 mg/dl or the initiation of RRT. Patients were stratified according to the highest AKI stage reached during their course. The estimated glomerular filtration rate was calculated using the Chronic Kidney Disease Epidemiology Collaboration creatinine equation.38
Ethical considerations
The project was reviewed by the National Institutes of Health Office of Institutional Review Board Operations (IRBO) and determined that the storage and analysis of anonymous or deidentified data collected by the registry did not qualify as human subjects research defined under federal regulations (45 CFR 46.102) and did not require institutional review board approval.
Statistical Analysis
Descriptive statistics summarized demographic and clinical characteristics of the study population and are described as frequency (percentage), mean (±SD) or median (interquartile range: 25th–75th percentiles). The primary outcome was death and secondary outcomes were AKI and/or need for RRT. To identify risk factors associated with the development of death or AKI/RRT, a series of statistical tests were performed in an exploratory manner. We analyzed a variety of COVID-19 outcomes including hospitalization, intensive care unit admission, ventilator requirement, and COVID-19–related complications including cardiovascular and thromboembolic events. Categorical data were compared between groups within the GN and control subsets (e.g., patients with and without the outcomes; GN subgroups) using Fisher’s exact test or, if ordinal, Kruskal-Wallis tests. Continuous data were compared using t tests or Wilcoxon rank-sum tests, as appropriate. Changes in renal parameters before COVID-19 infection, at the time of COVID-19 diagnosis, at peak values achieved during infection (if applicable), and after recovery were assessed using repeated measures mixed models. Mixed models also controlled for potential confounders (i.e., ethnicity) when comparing GN patients and controls. Post hoc pairwise comparisons corrected for multiplicity using the Stepdown Bonferroni method; otherwise, due to the exploratory aspect of the risk factor identifications, P values were uncorrected. Multivariable logistic regression models tested predictive relations of pertinent risk factors (age, sex, pre-COVID serum creatinine and serum albumin, hemoglobin, and ethnicity) when comparing the outcome of death between GN patients and controls. Results from the regression analyses are reported as OR and 95% CI. Data were analyzed using SAS v9.4 (SAS Institute, Inc, Cary, NC).
Disclosure
All the authors declared no competing interests.
Acknowledgments
The authors thank Frank Velez (National Institutes of Health [NIH]/National Institute of Diabetes and Digestive Kidney Disease [NIDDK]) and Matthew Breymaier (NIH/ Eunice Kennedy Shriver National Institute of Child Health and Human Development) for their assistance with creation of the registry using REDCap. Thank you to Dr. Damien Ashby, and all in the ICHNT Renal COVID Group, for use of the COVID database and care of the patients.
This research was supported in part by the Intramural Research Program of the NIH, NIDDK, and by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London.
Footnotes
Table S1. Numbers of patients with glomerular diseases followed at each center.
Table S2. Density of immunosuppression based on glomerular disease diagnosis and duration of disease.
Table S3. Complications in “kidney limited” (local) versus systemic GN patients with COVID-19.
Table S4. Changes in kidney parameters in GN patients and controls stratified according to main outcomes.
Supplementary Material
References
- 1.Chen T., Wu D., Chen H., et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ. 2020;368:m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cheng Y., Luo R., Wang K., et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97:829–838. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Benedetti C., Waldman M., Zaza G., et al. COVID-19 and the kidneys: an update. Front Med (Lausanne) 2020;7:423. doi: 10.3389/fmed.2020.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yamada T., Mikami T., Chopra N., et al. Patients with chronic kidney disease have a poorer prognosis of coronavirus disease 2019 (COVID-19): an experience in New York City. Int Urol Nephrol. 2020;52:1405–1406. doi: 10.1007/s11255-020-02494-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Banerjee D., Popoola J., Shah S., et al. COVID-19 infection in kidney transplant recipients. Kidney Int. 2020;97:1076–1082. doi: 10.1016/j.kint.2020.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alberici F., Delbarba E., Manenti C., et al. A single center observational study of the clinical characteristics and short-term outcome of 20 kidney transplant patients admitted for SARS-CoV2 pneumonia. Kidney Int. 2020;97:1083–1088. doi: 10.1016/j.kint.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hartzell S., Bin S., Benedetti C., et al. Evidence of potent humoral immune activity in COVID-19-infected kidney transplant recipients. Am J Transplant. 2020;20:3149–3161. doi: 10.1111/ajt.16261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cravedi P., Mothi S.S., Azzi Y., et al. COVID-19 and kidney transplantation: results from the TANGO International Transplant Consortium. Am J Transplant. 2020;20:3140–3148. doi: 10.1111/ajt.16185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Valeri A.M., Robbins-Juarez S.Y., Stevens J.S., et al. Presentation and outcomes of patients with ESKD and COVID-19. J Am Soc Nephrol. 2020;31:1409–1415. doi: 10.1681/ASN.2020040470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kronbichler A., Gauckler P., Windpessl M., et al. COVID-19: implications for immunosuppression in kidney disease and transplantation. Nat Rev Nephrol. 2020;16:365–367. doi: 10.1038/s41581-020-0305-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Angeletti A., Drovandi S., Sanguineri F., et al. COVID-19 in children with nephrotic syndrome on anti-CD20 chronic immunosuppression. Clin J Am Soc Nephrol. 2020;15:1494–1495. doi: 10.2215/CJN.06400420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chow N., Fleming-Dutra K., Gierke R., et al. Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12–March 28, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:382–386. doi: 10.15585/mmwr.mm6913e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Williamson E.J., Walker A.J., Bhaskaran K., et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Flythe JE, Assimon MM, Tugman MJ, et al. Characteristics and outcomes of individuals with pre-existing kidney disease and COVID-19 admitted to intensive care units in the United States [e-pub ahead of print]. Am J Kidney Dis. https://doi.org/10.1053/j.ajkd.2020.09.003. Accessed October 1, 2020. [DOI] [PMC free article] [PubMed]
- 15.Hariyanto T.I., Kurniawan A. Anemia is associated with severe coronavirus disease 2019 (COVID-19) infection. Transf Apher Sci. 2020;59:102926. doi: 10.1016/j.transci.2020.102926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lippi G., Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hematol Transf Cell Ther. 2020;42:116–117. doi: 10.1016/j.htct.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Aziz M., Fatima R., Lee-Smith W., Assaly R. The association of low serum albumin level with severe COVID-19: a systematic review and meta-analysis. Crit Care. 2020;24:255. doi: 10.1186/s13054-020-02995-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Violi F, Cangemi R, Romiti GF, et al. Is albumin predictor of mortality in COVID-19? [e-pub ahead of print]. Antioxid Redox Signal. https://doi.org/10.1089/ars.2020.8142. Accessed June 30, 2020. [DOI] [PubMed]
- 19.Robbins-Juarez S.Y., Qian L., King K.L., et al. Outcomes for patients with COVID-19 and acute kidney injury: a systematic review and meta-analysis. Kidney Int Rep. 2020;5:1149–1160. doi: 10.1016/j.ekir.2020.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cheng Y., Luo R., Wang X., et al. The incidence, risk factors, and prognosis of acute kidney injury in adult patients with coronavirus disease 2019. Clin J Am Soc Nephrol. 2020;15:1394–1402. doi: 10.2215/CJN.04650420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chan L, Chaudhary K, Saha A, et al. AKI in Hospitalized Patients with COVID-19 [e-pub ahead of print]. J Am Soc Nephrol. https://doi.org/10.1681/asn.2020050615. Accessed September 15, 2020.
- 22.Sterne J.A.C., Murthy S., Diaz J.V., et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA. 2020;324:1330–1341. doi: 10.1001/jama.2020.17023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Batlle D., Soler M.J., Sparks M.A., et al. Acute kidney injury in COVID-19: emerging evidence of a distinct pathophysiology. J Am Soc Nephrol. 2020;31:1380–1383. doi: 10.1681/ASN.2020040419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Norsa L., Indriolo A., Sansotta N., et al. Uneventful course in IBD patients during SARS-CoV-2 outbreak in northern Italy. Gastroenterology. 2020;159:371–372. doi: 10.1053/j.gastro.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.D’Silva K.M., Serling-Boyd N., Wallwork R., et al. Clinical characteristics and outcomes of patients with coronavirus disease 2019 (COVID-19) and rheumatic disease: a comparative cohort study from a US ‘hot spot’. Ann Rheum Dis. 2020;79:1156–1162. doi: 10.1136/annrheumdis-2020-217888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gianfrancesco M., Hyrich K., Al-Adely S., et al. Characteristics associated with hospitalisation for COVID-19 in people with rheumatic disease: data from the COVID-19 Global Rheumatology Alliance physician-reported registry. Ann Rheum Dis. 2020;79:859–866. doi: 10.1136/annrheumdis-2020-217871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kastritis E., Kitas G.D., Vassilopoulos D., et al. Systemic autoimmune diseases, anti-rheumatic therapies, COVID-19 infection risk and patient outcomes. Rheumatol Int. 2020;40:1353–1360. doi: 10.1007/s00296-020-04629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Simon D., Tascilar K., Krönke G., et al. Patients with immune-mediated inflammatory diseases receiving cytokine inhibitors have low prevalence of SARS-CoV-2 seroconversion. Nat Commun. 2020;11(1):3774. doi: 10.1038/s41467-020-17703-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee L.Y.W., Baptiste Cazier J., et al. COVID-19 mortality in patients with cancer on chemotherapy or other anticancer treatments: a prospective cohort study. Lancet. 2020;395:P1919–P1926. doi: 10.1016/S0140-6736(20)31173-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mehta P., Porter J.C., Chambers R.C., et al. B-cell depletion with rituximab in the COVID-19 pandemic: where do we stand? Lancet Rheumatol. 2020;2:e589–e590. doi: 10.1016/S2665-9913(20)30270-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hirsch J.S., Ng J.H., Ross D.W., et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020;98:209–218. doi: 10.1016/j.kint.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hippisley-Cox J., Young D., Coupland C., et al. Risk of severe COVID-19 disease with ACE inhibitors and angiotensin receptor blockers: cohort study including 8.3 million people. Heart. 2020;106:1503–1511. doi: 10.1136/heartjnl-2020-317393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lam K.W., Chow K.W., Vo J., et al. Continued in-hospital angiotensin-converting enzyme inhibitor and angiotensin II receptor blocker use in hypertensive COVID-19 patients is associated with positive clinical outcome. J Infect Dis. 2020;222:1256–1264. doi: 10.1093/infdis/jiaa447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Willicombe M., Thomas D., McAdoo S. COVID-19 and calcineurin inhibitors: should they get left out in the storm? J Am Soc Nephrol. 2020;31:1145–1146. doi: 10.1681/ASN.2020030348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moore J.T., Ricaldi J.N., Rose C.E., et al. Disparities in incidence of COVID-19 among underrepresented racial/ethnic groups in counties identified as hotspots during June 5–18, 2020—22 states, February–June 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1122–1126. doi: 10.15585/mmwr.mm6933e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Harris P.A., Taylor R., Thielke R., et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Harris P.A., Taylor R., Minor B.L., et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi: 10.1016/j.jbi.2019.103208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Levey A.S., Deo A., Jaber B.L. Filtration markers in acute kidney injury. Am J Kidney Dis. 2010;56(4):619–622. doi: 10.1053/j.ajkd.2010.08.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.