Abstract
Background
Patients with COVID-19 caused by SARS-CoV-2 exhibit diverse clinical manifestations and severity including enteric involvement. Commensal gut bacteria can contribute to defense against potential pathogens by promoting beneficial immune interactions. Interventions targeting the gut microbiome may have systemic anti-viral effects in SARS-CoV-2 infection.
Scope and approach
To summarise alterations of gut microbiota in patients with COVID-19 including impact of specific bacteria on disease severity, discuss current knowledge on the role of probiotics, prebiotics and dietary approaches including vitamin D in preventing and reducing disease susceptibility and review clinical studies using probiotics to target coronavirus. A literature review on SARS-CoV-2, COVID-19, gut microbiome and immunity was undertaken and relevant literature was summarised and critically examined.
Key findings and conclusions
Integrity of gut microbiome was perturbed in SARS-CoV-2 infections and associated with disease severity. Poor prognosis in SARS-CoV-2 infection was observed in subjects with underlying co-morbidities who had increased gut permeability and reduced gut microbiome diversity. Dietary microbes, including probiotics or selected prebiotics of Chinese origin, had anti-viral effects against other forms of coronavirus, and could positively impact host immune functions during SARS-CoV-2 infection. Numerous studies are investigating the role of probiotics in preventing and reducing susceptibility to SARS-CoV-2 infection in healthcare workers, household contacts and affected patients. An approach to strengthen intestinal barrier and lower pro-inflammatory states by adopting a more diversified diet during COVID-19 pandemic.
SARS-CoV-2 infection is associated with immune dysfunction and gut microbiota alterations. Delineating mechanisms of probiotics, prebiotics and diet with anti-SARS-CoV-2 immunity present opportunities for discovery of microbial therapeutics to prevent and treat COVID-19.
Keywords: SARS-CoV-2, Probiotics, Prebiotics, Diet, Nutrition
1. Introduction
In December 2019, the Coronavirus disease 2019 (COVID-19), caused by a novel coronavirus SARS-CoV-2, emerged in Wuhan, Hubei province, China (Li, Fan, et al., 2020) and soon spread rapidly across the world (Li, Guan, et al., 2020). To date, more than 9.2 million people have contracted COVID-19 infection with over 470,000 deaths worldwide. Therapeutic options are limited and clinically proven vaccine are lacking. In particular, there is a need for safe and effective interventions to prevent, reduce susceptibility and lessen the severity of COVID-19 (Amanat & Krammer, 2020). A striking observation during the pandemic is the heterogenous presentation and clinical outcomes of infected subjects across different geographic locations. Elderly individuals, along with those with pre-existing conditions, such as diabetes mellitus, hypertension, cardiovascular diseases and cancer have a demonstrated higher risk for developing more severe disease as well as suffering a higher risk of mortality, suggesting that the immune system may play a crucial role (Wang, Hu, et al., 2020). Patients with severe disease commonly presented with lymphopenia and a cytokine storm leading to acute respiratory distress syndrome and multi-organ failure. Lymphopenia can result in defects in anti-viral immunity and compromise innate and adaptive immune response contributing to poor prognosis (Azkur et al., 2020).
The gastrointestinal (GI) tract is the largest immunological organ in the body and home to trillions of microbes; the link between the GI tract and COVID-19 has been supported by reports of GI symptoms such as diarrhea in COVID-19 patients (Ng & Tilg, 2020). Microbes that reside in the human gut are key contributors to host metabolism and immunity and represent potential targets for novel therapeutics (Grifoni et al., 2020). In January 2020, the China National Health Commission and National Administration of Traditional Chinese Medicine Guidelines recommended the use of probiotics along with conventional treatment in patients with COVID-19 infection to improve the balance of intestinal flora and prevent secondary bacterial infections. The use of probiotics has also been suggested to flatten the curve of COVID-19 but currently evidence remain scarce (Baud et al., 2020). Since the outbreak of COVID-19, over 21,000 Google searches on “probiotics and immunity” have been performed in March 2020 by several channels and vehicles on probiotic products that can boost immunity. This simple finding suggests that the field of microbiome research is not only blossoming but also strongly highlights the need for advancement. One of the major caveats remains the lack of perspective from the society, scientists and healthcare professionals, who may misinterpret data or are anticipating a direct translation of such complex research from the bench to the bedside. To date, the rationale for using probiotics in COVID-19 has been mostly derived from indirect evidence (Mak et al., 2020). The current understanding is that the gut microbiome (referred to as the collective genomes of the diverse microbiota that reside in the gastrointestinal tracts of humans) can both regulate and be regulated by invading viruses, facilitating either stimulatory or suppressive effects. Some commensal gut bacteria can contribute to defense against potential pathogens by communicating with human cells and promoting beneficial immune interactions (Cabinian et al., 2018). This depicts a mechanism whereby exposure to dietary microbes, including probiotics or prebiotics, could positively impact immune function during infections (Jespersen et al., 2015).
In this perspective review, we described recent evidence showing altered gut microbiota in patients with COVID-19 including impact of specific bacteria and their involvement in immunity and disease severity. Secondly, we debated current knowledge on the role of probiotics, prebiotics, nutritional modulation in preventing and reducing susceptibility to COVID-19. Finally, we reviewed clinical studies using probiotics to target coronavirus and implications for future study design and introduction of precision nutrition in COVID-19.
2. Gut microbiome responses to SARS-CoV-2 infection
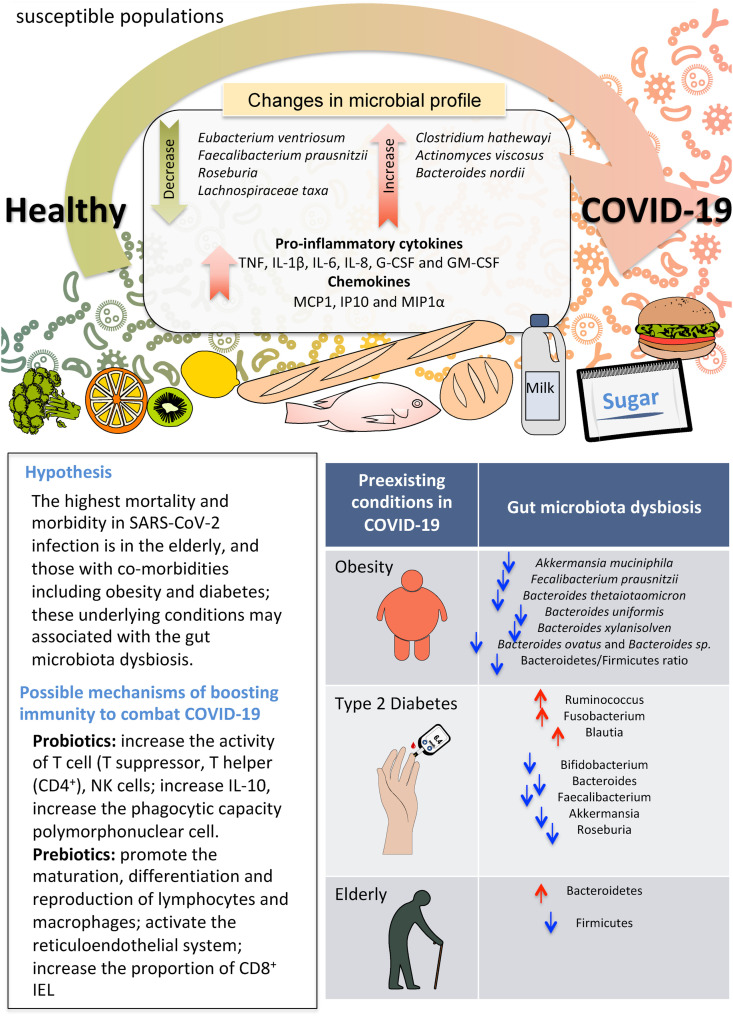
As with other infectious diseases, it is conceivable that the integrity of the gut microbiome could be perturbed by SARS-CoV-2 leading to gut dysbiosis in the host (Fig. 1 ). Fig. 1 shows the possible mechanisms of dysbiosis by SAR-CoV-2 virus infection and susceptible populations. Emerging signs show a link between gut functionality and microbiome responses to SARS-CoV-2. For instance, some patients with SARS-CoV-2 present with gastrointestinal (GI) symptoms including diarrhea and vomiting. The viral entry receptor, angiotensin converting enzyme 2 (ACE2) and virus nucleocapsid protein are present in gastrointestinal epithelial cells, and SARS-CoV-2 virus are repeatedly detected in fecal samples across different cohorts (Ng & Tilg, 2020). The highest mortality and morbidity in SARS-CoV-2 infection is in the elderly, and those with co-morbidities including obesity and diabetes; these underlying conditions have been associated with increased gut permeability, intestinal inflammation and reduced gut microbiome diversity (Liu et al., 2017). The gut-lung crosstalk has also been proposed to contribute to the pathogenesis of SARS-CoV-2 infection. Considering that pulmonary inflammation is common in patients in SARS-CoV-2 infection, direct suppression or promotion of viral infection by the gut microbiome may play a role (Dang & Marsland, 2019). Theoretically, these data point to an opportunity for interventions targeting the gut microbiome that may have systemic anti-viral effects in SARS-CoV-2 infection.
Fig. 1.
Possible mechanisms of dysbiosis by SAR-CoV-2 virus infection and susceptible populations.
A recent study from Hong Kong showed that patients with SARS-CoV-2 infection had significant alterations in fecal microbiomes compared with controls, characterized by enrichment of opportunistic pathogens (Clostridium hathewayi, Actinomyces viscous, and Bacteroides nordii) and depletion of beneficial commensals (Faecalibacterium prausnitzii, Lachnospiraceae bacterium, Eubacterium rectale, Ruminococcus obeum, and Dorea formicigenerans) (Zuo, Zhan, et al., 2020). Depleted symbionts and gut dysbiosis persisted even after clearance of SARS-CoV-2 in respiratory samples and resolution of respiratory symptoms. Gut bacteria that correlated with efficient viral clearance such as Bacteroidetes species may have a protective role in combating SARS-CoV-2 infection by hampering host entry through ACE2 (Zuo, Zhang, et al., 2020). Of interest was the observation that three bacterial members of the phylum Firmicutes (specifically, the genus Coprobacillus, and the two species Clostridium ramosum and Clostridium hathewayi) were associated with increased clinical severity of SARS-CoV-2 infection. This association is of particular interest, since Coprobacillus has been shown to upregulate colonic ACE-2 in mice (Naama et al., 2017). Faecalibacterium prausnitzii, which was found to have a strong negative correlation with the severity of COVID-19, had anti-inflammatory activity in the gut (Sokol et al., 2008). Another finding of interest was a negative correlation between fecal SARS-CoV-2 load and the abundance of specific gut bacteria such as certain Bacteroides species (Bacteroides dorei, Bacteroides thetaiotaomicron, Bacteroides massiliensis and Bacteroides ovatus). These species were associated with a reduction in ACE2 expression in the mouse gut (Naama et al., 2017), suggesting that they may limit the ability of SARS-CoV-2 to enter into enterocytes. These findings corroborated with that of a separate study showing lowered gut microbiota diversity and levels of butyrate-producers, and higher levels of opportunistic pathogens in patients with SARS-CoV-2 infection compared with H1N1 patients and healthy controls. In addition, seven bacterial strains (Streptococcus, Fusicatenibacter, Collinsella, Dorea, Agathobacter, Eubacterium hallii group, Ruminococcus torques group) could be regarded as a COVID-19 diagnostic biomarker (Gu et al., 2020). To date, it remains unclear whether the dysbiotic microbial communities are a cause or a consequence of COVID-19. However, metabolic alterations associated with inflammation might act as microbial stressors and promote the outgrowth of dysbiotic species. These data indicate that strategies to alter the intestinal microbiota might reduce the disease severity of COVID-19 (Zuo, Zhang, et al., 2020).
3. Interactions of gut microbiome and lung immunity in SARS-CoV-2 infection
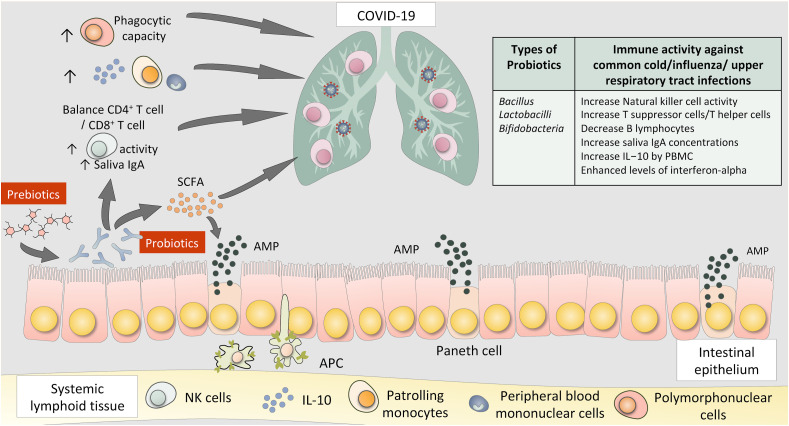
Manipulation of gut microbiota by antibiotics, probiotics, prebiotics, and natural products or diets represents a promising therapeutic strategy for lung diseases in clinical and experimental studies (Zhang, Li, Wang, Tan, Zhang and Feng, 2020). The pathogenesis of SARS-CoV-2 infection relates to dysfunctional immune response including excessive infiltration of monocytes, macrophages and T cells, systemic cytokine storm, pneumonia and multi-organ damage (Zhang, Tan, et al., 2020). Defining these key immune cell subsets and their states in SARS-CoV-2 infection is a crucial step in obtaining insights into the immune clearance mechanism and developing new therapeutic strategies for patients with COVID-19. Xu et al. described pathological findings in severe SARS-CoV-2 and demonstrated aberrant immune cell infiltrates in the lung (Xu et al., 2020). In this regard, reduction and functional exhaustion of CD8+ T cells was shown in parallel with a rise in inflammatory cytokine levels in patients with severe COVID-19 (Diao et al., 2020). Importantly, gut microbiota influences the balance between pro-inflammatory and regulatory responses and shape host immune system (Zheng et al., 2020). Fig. 2 shows interactions between the human gut and lung and potential positive immune responses triggered by probiotics or prebiotics. Prebiotics provide the energy for probiotics growth. Several probiotics including Bacillus, Lactobacilli, Bifidobacteria have been reported to have positive immune activity against common upper respiratory tract infections, via increasing Natural killer cell activity, increasing T suppressor or T helper cells, decreasing B lymphocytes, increasing saliva IgA concentrations, increasing IL-10 production and enhancing levels of interferon-alpha.
Fig. 2.
Interactions between the human gut and lung and potential positive immune response triggered by probiotics and prebiotics.
4. Role of probiotics and SARS-CoV-2 infection
While the beneficial effect of probiotics (defined as live microorganisms that when administered in adequate amounts confer a health benefit on the host) was originally thought to stem from improvement in the intestinal microbial balance, there is now substantial evidence that probiotics can also provide benefits by modulating host immune functions (Klaenhammer et al., 2012). Probiotics has been shown to shape T cell subsets (Lee & Kim, 2017), stimulates antimicrobial peptide production by Paneth cells (Bevins & Salzman, 2011) and direct Th17 cells differentiation in the small intestinal tract (Ivanov et al., 2008). Understanding the fundamental immunological process underlying clinical manifestations of SARS-CoV-2 infection is vital for the identification and rational design of effective therapies. Laboratory studies have reported that certain probiotics have anti-viral effects including against other forms of coronavirus (Chai et al., 2013). Others have reported the potential of probiotics to interact with ACE2, the host entry receptor of SARS-CoV-2 virus; for instance, some lactobacilli have been reported to release peptides with high affinity for ACE during milk fermentation (Li et al., 2019). Recently, Paenibacillus bacteria were shown to naturally produce carboxypeptidases homologous to ACE2 in structure and function (Minato et al., 2020). A recent study showed that Toll-like Receptor (TLR4) signaling was essential for the effects of preventive intranasal treatment with probiotic Lacticaseibacillus rhamnosus GG in a neonatal mouse model of influenza infection (Kumova et al., 2019). Interestingly, human cathelicidin, LL-37, is one of the best-studied human antimicrobial peptides that have a wide-spectrum activity against bacteria and viruses (Mookherjee et al., 2020) and could be effectively delivered by food-grade probiotics including Lactococcus lactis expression system (Mierau & Kleerebezem, 2005). The protective immunomodulatory effect of cathelicidin-expressing Lactococcus lactis (Wong et al., 2012), may help open new avenues of combating SARS-CoV-2 infection using antimicrobial peptides-expressing probiotics delivery system. Whether these or other immunomodulatory effects, following local or oral administration, could be effective in SARS-CoV-2 infected individuals remains unknown. Some clinical trials have demonstrated that specific probiotics can reduce the incidence and duration of common upper respiratory tract infections, especially in children but also with some data in adults (King et al., 2014) and long-term nursing home residents (Van Puyenbroeck et al., 2012).
Recently, new guidelines from the WHO recommends against antibiotic therapy or prophylaxis for patients with mild and moderate COVID-19 unless there is concurrent suspected bacterial infection (https://www.who.int/publications/i/item/clinical-management-of-covid-19). Another setting whereby probiotics may play a role relates to perturbed microbiota caused by antibiotics (Palleja et al., 2018). As empirical antibiotics are widely used in the early phase of COVID-19 outbreak and may lead to more severe and unfavorable dysbiosis, reinforcement of colonic microbiota using probiotics has been proposed to reduce susceptibility to subsequent secondary co-infections. More clinical trials are necessary to confirm the role of probiotics and to determine the optimal strain(s), dosing regimens, time and duration of intervention for SARS-CoV-2 infection.
5. Role of prebiotics and SARS-CoV-2 infection
Prebiotics play a widely recognized role in the regulation of intestinal microbiota. Prebiotics are special fibers with selective fermenting ingredients that are needed by gut microbes. It selectively stimulates favourable growth and/or enhance activities of indigenous probiotic bacteria. Prebiotic therapies have been shown to be associated with improvement in gut-related diseases such as constipation, diarrhea, as well as non-gut disorders including reduction of the risks of osteoporosis, atherosclerotic cardiovascular disease associated with dyslipidemia, insulin resistance, obesity, and possibly type 2 diabetes (Wang, Xiao, Tian, Zhao and Chen, 2020). Large amounts of prebiotic fiber provide an abundance of food for a range of gut bacteria and when utilized this can result in an excess of gas in the intestine. The most common side effects of prebiotics are abdominal bloating and discomfort, occurring when large doses are consumed (Marteau & Seksik, 2004). Compared to probiotics, there has been less information on the use of prebiotics for addressing respiratory infections. Some studies support the use of galactans and fructans in infant formulae in reducing upper respiratory infections (Shahramian et al., 2018) and a meta-analysis of synbiotics demonstrated efficacy in lowering respiratory tract infections (Chan et al., 2020). Research in mice showed that the dietary fiber, inulin, conferred protection against influenza A by shaping Ly6c (−) patrolling monocyte hematopoiesis and CD8+ T Cell (Trompette et al., 2018). Traditional Chinese medicine has been playing an important role in the prevention, treatment and rehabilitation of COVID-19 (Ren et al., 2020). Recent data from the National Administration of Traditional Chinese Medicine showed that a Traditional Chinese Medicine (TCM) named “Lung Cleansing and Detoxifying Decoction (Qing Fei Pai Du Tang)”, which consists of carbohydrate polymers may have therapeutic effects in SARS-CoV-2 infection. The “Lung Cleansing and Detoxifying Decoction” was given to 214 COVID-19 patients, and the symptoms of 90 percent of the patients were notably relieved after 3-day-treatment of this Traditional Chinese Medicine (Zhao et al., 2020). These carbohydrate polymers are polysaccharides within the family of prebiotics comprising of oligosaccharides and polysaccharides. Considering that cellular immunity plays a critical role in eliminating viral infections, the most significant properties of polysaccharides rest in their immunomodulatory activities (Li, Fan, et al., 2020). For example, Chinese yam polysaccharides carry immunomodulatory and immune enhancement functions and have served as adjuvants in developing vaccines (Wu et al., 2020). Glycyrrhiza polysaccharide could activate the immune system by promoting the maturation, differentiation and reproduction of immune cells such as lymphocytes and macrophages, as well as activating the reticuloendothelial system (Luo et al., 2017). Guan et al. analyzed 1099 cases of COVID-19 in China and found lymphocytopenia to be one of the most common features and subjects with lymphopenia were less likely to survive (Li, Peri, et al., 2020). IL-6 and IL-8 negatively correlated with lymphocyte count and IL-6 kinetics was associated with disease severity (Zhang, Tan, et al., 2020). For example, dietary sugar beet fiber has been shown to increase the proportion of CD8+ IEL, especially in cecal mucosa (Ishizuka & Tanaka, 2002).
Importantly, prebiotics is closely related to the growth and function of probiotics. Gut microbes, especially probiotics, degrade various prebiotics such as fructan, glucan, arabinoxylan. The end products, short chain fatty acids (SCFAs) have been demonstrated to have regulatory effects on host immunity. SCFAs are involved in host immune response by different pathways through specific receptors, and vary in intensity of G protein-coupled receptors (GPCRS) receptor activation (Trompette et al., 2014). In addition, butyric acid and propionic acid from prebiotics fermentation by gut microbiota can affect differentiation or functions of T cells, macrophages and dendritic cells. These SCFAs directly impact B cell intrinsic functions to moderately enhance class-switch DNA recombination (CSR). In human and mouse B cells, butyrate and propionate decrease B cell Aicda and Prdm1 by upregulating select miRNAs that target Aicda and Prdm1 mRNA-3′ UTRs through inhibition of histone deacetylation (HDAC) of miRNA host genes. By acting as HDAC inhibitors, SCFAs can impair intestinal and systemic T-dependent and T-independent antibody responses (Sanchez et al., 2020).
Glycans, or polysaccharides, are included in prebiotics. They are carbohydrate-based polymers that regulate a variety of processes including immunity (Lauc et al., 2016). Glycan diversity represents one of the main defenses of all higher organisms against infection, and the repertoire of glycans changes with age, especially in the age ranges that are most susceptible to SARS-CoV2 infection. Furthermore, both SARS-Cov-2 virus and its principal cellular target ACE2 are known to be highly glycosylated (Walls et al., 2020). A recent study analyzed site-specific N-linked glycosylation of MERS and SARS S glycoproteins, indicating that each of these glycosylation sites can be occupied by up to ten different glycans (called glycoforms), which greatly extends epitope diversity (Watanabe et al., 2020). Furthermore, glycans are one of the principal regulators of antibody effector functions and many other aspects of the immune system (Lauc & Sinclair, 2020).
6. Nutritional and dietary approaches to restore a healthy microbiota
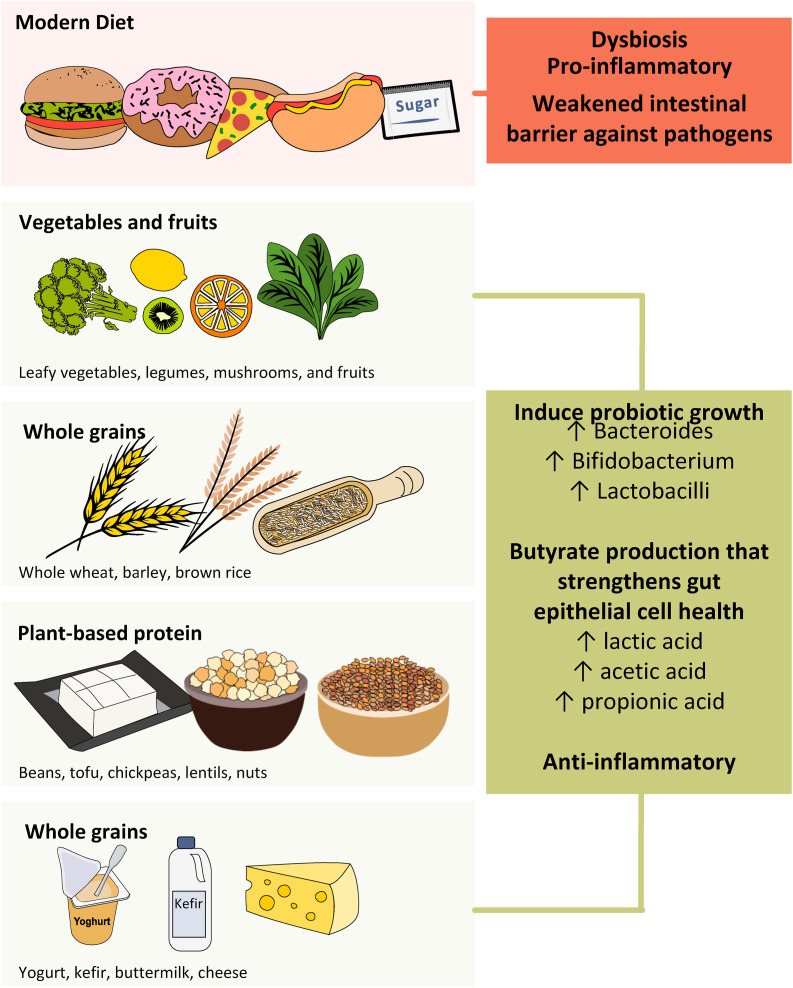
Diet has a key impact on the gut microbiota and can influence gut microbial richness and diversity. High intake of animal proteins, saturated fat, sugar, and salt has been reported to stimulate the growth of pathogenic bacteria leading to alterations of the intestinal barrier, whereas consumption of complex polysaccharides and plant protein was associated with an increase of beneficial bacteria which stimulate SCFA production. Omega-3, polyphenols, and micronutrients can confer health benefits via modulation of the gut microbiota (Rinninella et al., 2019). In addition, branched-chain amino acids can increase intestinal immunoglobulin levels hence improving gut barrier (Elmadfa & Meyer, 2019). High-quality proteins are a crucial component of an anti-inflammatory diet. Nutritional dietary components known to exert anti-inflammatory and antioxidant properties include omega-3 fatty acids, vitamin C, vitamin E, and phytochemicals such as carotenoids and polyphenols that are commonly found in plant-based foods. Plant-based fiber also has prebiotic effects such as promoting the growth of bacteria that are associated with health benefits, such as Bifidobacterium and Lactobacillus spp. Moreover, reduce potential pathogens such as Clostridium spp (Iddir et al., 2020). Most microbial species in the GI tract belong to Firmicutes, Bacteroidetes, Proteobacteria and Actinobacteria and these microbial populations aim to provide nutrients and maintain homeostasis in the host (Gombart et al., 2020). Modern diets in developed nations have shifted away from diets high in phytochemical rich fruits, vegetables, polysaccharides and fibres, lean meats to one that is low in fibres and high in processed foods with exposure to food additives, refined sugar and hydrogenated fats (Jew et al., 2009). One approach key during COVID-19 pandemic is to strengthen intestinal barrier against pathogens, increase intestinal motility and lower a pro-inflammatory state (Trottein & Sokol, 2020) by adopting a more diversified diet with moderate increase in high-fiber and plant-based foods.
Food diversity is the corner stone for every being against COVID-19 pandemic. In light of the complexity of the gut microbiota ecosystem, a single food item may not dramatically shift its overall landscape (McCann, 2000). A diversified diet such like the Mediterranean diet has positive effect on immunity with abundant supply of dietary fibers. It was found in vitro that galactooligosaccharides (GOS) and arabinoxylan increased the production of T helper cell 1 IFN-γ, whereas the T helper cell 1 cytokine TNF-α was decreased (Bermudez-Brito, 2015). Following the Mediterranean dietary pattern, characterized by high consumption of whole grains, vegetables, legume, nuts, extra-virgin olive oil (rich in polyphenols), consumption of fats where unsaturated fatty acids dominate, and low consumption of processed meats and refined sugars, can have favourable modulation of relative gut microbial abundance and diversity to maintain its homeostasis (De Filippis, 2016). Moreover, micronutrients obtained from such diet, namely vitamins A, B6, B12, C, D, E, folate, iron, selenium, magnesium, zinc and copper optimize the innate and adaptive immunity since many micronutrients cast functional roles in our immune system (Maggini et al., 2018). Lastly, a diversified diet includes fermented dairy products, fermented soybeans and vegetables that contain naturally occurred probiotics, Lactobacillus bulgaricus and Streptococcus thermophiles (Ghosh et al., 2019). Considering the synergetic effect of microbial-diet-host interaction, food diversity is important to maintain immunity by creating a nurturing environment for microbiota cultivation (De Filippo et al., 2010).
Of particular relevance is the enhancement of intestinal butyrate production through promotion of microbial interactions using dietary approaches, which can strengthen gut epithelial cell health. Universally, Bifidobacteria and Lactobacilli have been considered beneficial species with butyrate production. Faecalibacterium (F) prausnitzii has been shown to be reduced in patients with COVID -19 (Zuo, Zhang, et al., 2020). The role of F. prausnitzii in stimulating butyrate (Lopez-Siles et al., 2017) and their anti-inflammatory properties (Sokol et al., 2008) in the intestine have been consistently reported and attracted increasing interest in restoring this beneficial gut bacterium to facilitate treatment for inflammatory bowel disease (Lopez-Siles et al., 2017). F. prausnitzii could be stimulated by different substrates derived from diet or prebiotics, including inulin (Ramirez-Farias et al., 2009), dietary fibre (Benus et al., 2010), polydextrose and soluble corn fiber (Hooda et al., 2012). Table 1 shows natural foods containing high content of prebiotics (such as fibers, oligosaccharides) or probiotics (such as bifidobacteria and lactobacillus) beneficial to build a resilient and diversified gut microbiota.
Table 1.
Natural foods containing high content of prebiotics (such as fibers, oligosaccharides) or probiotics (such as bifidobacterial and lactobacillus).
| Food Group (and their associated nutrients) | Specific Food to Consume |
|---|---|
| Whole grains (fibers, oligosaccharides, minerals, and vitamins) | whole wheat, barley, brown rice |
| Vegetables and fruits (fibers, oligosaccharides, and vitamins A, B, C, D, E, K) | most leafy vegetables, legumes, mushrooms, and fruits |
| Plant-based protein (soluble fibers and amino acids) | beans, tofu, chickpeas, lentils, nuts |
| Fiber-fortified food (oligosaccharides) | cereals, breads, drinks, supplements |
| Fermented dairy (bifidobacterial and lactobacillus) | yogurt, kefir, sweet acidophilus milk, buttermilk; cheese: cheddar, Gouda, mozzarella, and cottage |
| Fermented beans or vegetables (lactobacillus) | miso, sauerkraut, sour pickles soybeans (tempeh) |
7. Dietary approaches during COVID-19 pandemic
The WHO published a set of recommendations to promote a shift from a Westernized modern diet to a more diversified diet to protect against malnutrition and non-communicable diseases (Organization WHO). Dietitians in Canada recommend a healthy diet rich in fruit and vegetables, protein foods and whole grains to support a strong immune system. Academy of Dietetics and Nutrition suggests a variety of foods to obtain key nutrients in supporting the immune system. Due to the limited data on the beneficial or harmful strains of bacteria in SARS-CoV-2 infection, researches in this area and diets adhering to modest qualities of naturally fermented food needs to be urgently investigated. A diversified diet for a healthy individual daily includes five servings or more of fruits, vegetables (excluding starchy vegetables) including legumes, nuts, and whole grains; minimize free sugars (all sugar added to foods in the manufacturing or cooking processes); up to 30% of total energy intake comes from fats while unsaturated fats are preferred over saturated fats; avoid industrially produced trans-fat; and less than 5 g of salt (Mozaffarian et al., 2014). As subjects with type 2 diabetes mellitus, obesity, cardiovascular disease, and other chronic diseases had a worst prognosis and higher mortality when infected with SARS-CoV-2, hydration and adequate nutrients are key components to prevent malnutrition and maintain homeostasis of the metabolism and physiology in these individuals (Volkert et al., 2019). In a recent study among 7300 individuals with COVID-19 (among which nearly 1000 had type 2 diabetes mellitus) in Hubei Province, China, subjects with well-controlled blood glucose (upper limit ≤ 10 mmol/L) had a reduced risk for complications and mortality (Zhu et al., 2020). The overall nutrition management for suspected or infected cases is to control blood sugar in those with diabetes or metabolic diseases and to maintain nutrition and hydration. In subjects with gastrointestinal symptoms with sensory loss – anosmia, hyposmia, and dysgeusia, food high in calories and proteins in small bouts every two to 3 h and fluid intake of 60–120 mL every 15 min to compensate water loss through fever are recommended (Reithner, 1981). In infected patients who require intensive care, a timely manner to initiate enteral nutrition (EN) within 24–36 h of admission to ICU or within 12 h of intubation/placement of mechanical ventilator is recommended (Barazzoni et al., 2020). A fiber free formula is more tolerable to patients with significant gastrointestinal dysfunction. In addition, evidence linking vitamin D deficiency with COVID-19 severity is growing with data suggesting a correlation between insufficient vitamin D level and COVID-19 mortality (Rhodes et al., 2020). Therefore, vitamin D supplementation at moderate dose is recommended for individuals at risk of deficiency, including those living in institutions or individuals with darker skin. Fig. 3 illustrates dietary and nutritional recommendations during COVID-19.
Fig. 3.
Dietary and nutritional recommendations during COVID-19.
8. Clinical trials of probiotics for prevention or treatment of SARS-CoV-2 infection
At a time when drugs are being tested with no clear success as yet, probiotics have been considered in clinical trials to reduce the burden and severity of COVID-19. It is highly likely that inter-individual variation in gut microbiome will impact on the efficacy of proposed microbiota interventions for SARS-COV-2 infection. No published studies so far have reported the use of probiotics as add-on therapy for the management of COVID-19. However, the International Scientific Association of Probiotics and Prebiotics (IASPP) has highlighted that scientists and clinicians globally are investigating the relationship between the gut microbiome and susceptibility to COVID-19 and assessing the role of various probiotics strains to lower viral load via different mechanisms. Scientists in University of Antwerp, Belgium are exploring whether specific strains of lactobacilli in the nasopharynx and oropharynx could potentially reduce viral activity via a multifactorial mode of action, including barrier-enhancing and anti-inflammatory effects, and reduce the risk of secondary bacterial infections in COVID-19. They are also investigating the potential of specific strains of lactobacilli with immunostimulatory effects as adjuvants for intranasal SARS-CoV-2 vaccination, or the potential of a genetically engineered antigen-producing organism for vaccine delivery. Other researchers at the North Carolina State University, USA, are actively engineering Lactobacillus acidophilus probiotics expressing COVID-19 proteins to be tested as potential vaccines. In the United Kingdom, a Phase II randomized, double-blind, placebo-controlled trial is being conducted to determine the efficacy and safety of oral Live Biotherapeutic MRx-4DP0004 in addition to standard supportive care for hospitalized patients with COVID-19. Researchers from Duke University are developing protocols to study prevention and treatment of COVID-19 in a range of at-risk populations including healthcare providers, hospitalized patients, and nursing home and nursing home workers. An initial multicenter, randomized, double blind, placebo-controlled phase 2 trial will assess if the probiotic Lactobacillus rhamnosus GG has a role in prevention and treatment of COVID-19 infection. Researchers from the Chinese University of Hong Kong has identified a probiotic formula derived from analysis of depleted bacteria in fecal samples of COVID-19 patients that persisted even after recovery and discharge form hospitals, and this formula will be investigated in a randomized controlled trial for patients who have recovered from COVID-19 to rebalance their gut microbiota. Table 2 summarises ongoing clinical studies of probiotic intervention in COVID-19.
Table 2.
Ongoing clinical studies of probiotic intervention in COVID-19.
| Country | ClinicalTrials.gov ID | Study type | Study subjects | Age group | Sample size | Intervention | Dose | Duration | Primary outcome |
|---|---|---|---|---|---|---|---|---|---|
| Spain | NCT04390477 | Open label RCT | COVID-19 patients requiring hospitalization | ≥18 years | 40 | Dietary Supplement: Probiotic vs No intervention | 1 × 10∧9 CFU/day | 30 days | Cases with discharge to ICU |
| Spain | NCT04366180 | Double-blind RCT | Healthcare workers without COVID-19 | ≥20 years | 314 | Probiotic (Lactobacillus) vs Control (Maltodextrin) | 3 × 10∧9 CFU/day | 8 weeks | Incidence of SARS-CoV-2 infection in healthcare workers |
| Italy | NCT04366089 | Single-blind RCT | COVID-19 patients requiring hospitalization | ≥18 years | 152 | Standard of care (Azithromycin and hydroxychloroquine) vs Oxygen-ozone therapy and probiotic (SivoMixx 200 billion: Streptococcus thermophiles, 2 Bifidobacterium strains, 5 Lactobacillus strains) |
1.2 × 10∧12 CFU/day | 21 days | Delta in the number of patients requiring orotracheal intubation despite treatment |
| Austria | NCT04420676 | Double-blind RCT | COVID-19 patients with diarrhea | ≥18 years | 108 | Synbiotic (Omnibiotic AAD: 2 Bifidobacterium strains, Enterococcus, 7 Lactobacillus strains) vs Placebo | Not mentioned | 30 days | Duration of diarrhea |
| United States | NCT04399252 | Double-blind RCT | People with household contact of COVID-19 patient | ≥1 year | 1000 | Probiotic (Lactobaciltus rhamnosus GG) vs Placebo | 2 capsules per day | 28 days | Changes in Shannon bacteria diversity |
| Canada | NCT04334980 | Double-blind RCT | Healthy subjects without COVID-19 | 19–45 years old | 84 | bacTRL-Spike (Bifidobacterium engineered to deliver plasmids encoding spike protein from SARS-CoV-2) vs Placebo | 1 × 10∧9, 3 × 10∧9 or 1 × 10∧10 | 12 months | Frequency of adverse events (specifically including incidence of gastrointestinal-associated events) |
| Italy | NCT04368351 | Retrospective Case-Control | COVID-19 patients requiring hospitalization | ≥18 years | 70 | Standard of care (Azithromycin and hydroxychloroquine) vs Bacteriotherapy (SivoMixx: Streptococcus thermophiles, 2 Bifidobacterium strains, 5 Lactobacillus strains; Azithromycin and Hydroxychloroquine) |
Not mentioned | 21 days | Delta of time of disappearance of acute diarrhea |
| Mexico | NCT04517422 | RCT | COVID-19 patients requiring hospitalization | 18–60 year old | 300 | Combination of Lactobacillus plantarum CECT7481, Lactobacillus plantarum CECT 7484, Lactobacillus plantarum CECT 7485, and Pediococcus acidilactici CECT 7483 vs Placebo | Not mentioned | 30 days | Severity progression of COVID-19, Stay at ICU, Mortality ratio. |
| Hong Kong | aApproved by CUHK-NTEC CREC: CRE-2020.407 | Pilot study | COVID-19 Patients requiring hospitalization | ≥18 years | 50 | Synbiotic (CUHK-Synbiotic vs Standard care) | 2 × 10∧11 CFU/day | 28 days | Composite outcome of symptom score improvement within 4 weeks. |
| Hong Kong | aApproved by CUHK-NTEC CREC: CRE-2020.400 | A single-arm, open-labelled interventional study | Recovered/Discharged COVID-19 patients | ≥18 years | 20 | Synbiotic (CUHK-Synbiotic) | 2 × 10∧11 CFU/day | 28 days | Changes in gut microbiome (bacteria, virome and fungome) at week 5 compared to baseline. |
The Joint Chinese University of Hong Kong – New Territories East Cluster Clinical Research Ethics Committee: CUHK-NTEC CREC.
9. Conclusion and future directions
SARS-CoV-2 associated-immune dysfunction and alterations in the gut microbiota present an exciting opportunity for studies and discovery of microbial therapeutics to prevent and treat SARS-CoV-2 infection. There is a pressing need to characterize specific microbial species, prebiotics, or a combination that can boost immunity, and better understand mechanisms of precision symbiotic with anti-viral immunity for SARS-CoV-2 infection. In addition to bacteria, other key microorganisms, such as viruses, phages, yeast and fungi, are also present in the gut. These microorganisms, which likely regulate the activity of the host and of the other gut microbes may be as important as bacteria. It was recently shown that patients with COVID-19 had significantly altered gut mycobiome with a 3-fold increase in fungal diversity and pathogens from the genus Aspergillus and patients who had more than two Aspergillus pathogens in high abundance had more severe disease. These fungi were also present in the feces despite nasopharyngeal clearance of SARS-CoV-2, hinting to persistent colonization of fungal pathogen and a potential threat to the host in the longer term (Zuo, Zhan, et al., 2020). The gut mycobiome offer an additional dimension to the investigation of host–microorganism interactions in SARS-CoV-2 infection.
Importantly, different methodologies should be adopted to study associations between dietary and microbiome effects and susceptibility to SARS-CoV-2 infection and disease severity. An international approach will allow inclusion of populations from different locations and various background, dietary patterns, and environmental exposures. Adequately powered studies that recruit COVID-19 patients and healthy individuals to collect clinical data, host genetic parameters, dietary datasets, immune phenotyping, and multi-omics microbiome markers are needed. A collaborative international approach is essential for unravelling the determinants of clinical outcomes of SARS-CoV-2 infection and for designing targeted therapeutic and preventative microbial measures. The moderating effects of high fiber especially the choice of the high-fiber food type, freshly fermented, and diverse foods should also be investigated as preventative and mitigating measures. Novel technologies and delivery systems such as nanotechnology can efficiently help in designing intelligent functional foods with the possibility of localized delivery in the gut (Kalantar-Zadeh et al., 2020).
Another consideration is whether the composition of one's microbial profile can influence the immunomodulatory treatment and response to drugs or vaccine. Several anti-viral drugs have been investigated to combat SARS-COV2 virus and on May 1, 2020, the anti-viral Remdesivir became the first drug to receive emergency FDA approval for the treatment of COVID-19 (Grein et al., 2020). Although there is no direct evidence of gut microbiome interaction with Remdesivir, there is precedent for gut microbiome modulation of other anti-viral drugs (Dominguez-Diaz et al., 2019). It has been reported that enzymes belonging to the common gut microbiota constituents Bacteroides thetaiotaomicron and Bacteroides ovatus functionally impact activity and toxicity of the oral antiviral Brivudine. The ability of 76 human gut bacteria to metabolise 176 drugs including several with antiviral activity has also recently been characterized (Zimmermann et al., 2019). In addition, a clinical phase I study demonstrated that the gut microbiota plays a major role in generating an optimal antibody response against influenza shot, which indicated vaccines response could be affected by gut microbiota (Hagan et al., 2019). For instance, probiotics and/or prebiotics could positively boost the immune system to increase influenza vaccine efficacy in humans (Van Puyenbroeck et al., 2012). These questions that remain are challenging and will require innovative approaches. Ultimately, these efforts should lead to deeper insight into host-microbial relationships and provide exciting new opportunities to improve host immunity to conquer COVID-19, either through microbiota modulation, for prevention, or enhancing vaccine response. Nutritional and dietary strategies directed at restoring established beneficial microbiota, which can possibly suppress viral infection in the elderly and those with underlying health problems, may be an effective strategy to mitigate the unwanted effects of this virus. Ultimately, a comprehensive mechanistic-based approach, coupled with carefully conducted multicenter randomized, controlled trials will advance our knowledge to address the potential of microbiota and dietary manipulation as an alternative management option for SARS-CoV-2 infection.
Authors’ contributions
Jielun Hu, Lin Zhang, Winnie Lin were responsible for conception, literature review, writing and revising the manuscript. Whitney assisted in the graphical figures’ preparation. Siew C Ng and Francis KL Chan provided critical revisions of the final manuscript. All authors agreed to the final version of manuscript.
Declaration of competing interest
The authors declare that they have no conflict of interest.
References
- Amanat F., Krammer F. SARS-CoV-2 vaccines: Status report. Immunity. 2020;52(4):583–589. doi: 10.1016/j.immuni.2020.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azkur A.K., Akdis M., Azkur D., Sokolowska M., Van D.V.W., Brüggen M., et al. 2020. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy, 2020, publish online May 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barazzoni R., Bischoff S.C., Breda J., Wickramasinghe K., Krznaric Z., Nitzan D., et al. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clinical Nutrition. 2020;39:1631–1638. doi: 10.1016/j.clnu.2020.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baud D., Dimopoulou A.V., Gibson G.R., Reid G., Giannoni E. Using probiotics to flatten the curve of coronavirus disease COVID-2019 pandemic. Frontiers in Public Health. 2020;8:186. doi: 10.3389/fpubh.2020.00186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benus R.F.J., van der Werf T.S., Welling G.W., Judd P.A., Taylor M.A., Harmsen H.J.M., et al. Association between Faecalibacterium prausnitzii and dietary fibre in colonic fermentation in healthy human subjects. British Journal of Nutrition. 2010;104:693–700. doi: 10.1017/S0007114510001030. [DOI] [PubMed] [Google Scholar]
- Bermudez-Brito M., Sahasrabudhe N.M., Rosch C., Schols H.A., Faas M.M., de Vos P. The impact of dietary fibers on dendritic cell responses in vitro is dependent on the differential effects of the fibers on intestinal epithelial cells. Molecular Nutrition & Food Research. 2015;59(4):698–710. doi: 10.1002/mnfr.201400811. [DOI] [PubMed] [Google Scholar]
- Bevins C.L., Salzman N.H. Paneth cells, Antimicrobial peptides and maintenance of intestinal homeostasis. Nature Reviews Microbiology. 2011;9(5):356–368. doi: 10.1038/nrmicro2546. [DOI] [PubMed] [Google Scholar]
- Cabinian A., Sinsimer D., Tang M., Jang Y., Choi B., Laouar Y., et al. Gut symbiotic microbes imprint intestinal immune cells with the innate receptor SLAMF4 which contributes to gut immune protection against enteric pathogens. Gut. 2018;67(5):847–859. doi: 10.1136/gutjnl-2016-313214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai W.-D., Burwinkel M., Wang Z.-Y., Palissa C., Esch B., Twardziok S., et al. Antiviral effects of a probiotic Enterococcus faecium strain against transmissible gastroenteritis coronavirus. Archives of Virology. 2013;158(4):799–807. doi: 10.1007/s00705-012-1543-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan C.K.Y., Tao J., Chan O.S., Li H.-B., Pang H. Preventing respiratory tract infections by synbiotic interventions: A systematic review and meta-analysis of randomized controlled trials. Advances in Nutrition. 2020;2020 doi: 10.1093/advances/nmaa003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang A.T., Marsland B.J. Microbes, metabolites, and the gut-lung axis. Mucosal Immunology. 2019;12(4):843–850. doi: 10.1038/s41385-019-0160-6. [DOI] [PubMed] [Google Scholar]
- De Filippis F., Pellegrini N., Vannini L., Jeffery I.B., La Storia A., Laghi L., et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. 2016;65(11):1812–1821. doi: 10.1136/gutjnl-2015-309957. [DOI] [PubMed] [Google Scholar]
- De Filippo C., Cavalieri D., Di Paola M., Ramazzotti M., Poullet J.B., Massart S., et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proceedings of the National Academy of Sciences. 2010;107(33):14691–14696. doi: 10.1073/pnas.1005963107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diao B., Wang C.-H., Tan Y.-J., Chen X.-W., Liu Y., Ning L.-F., et al. Reduction and functional exhaustion of T Cells in patients with coronavirus disease 2019 (COVID-19) Frontiers in Immunology. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominguez-Diaz C., Garcia-Orozco A., Riera-Leal A., Padilla-Arellano J.R., Fafutis-Morris M. Microbiota and its role on viral evasion: Is it with us or against us? Frontiers in Cellular and Infection Microbiology. 2019;9:256. doi: 10.3389/fcimb.2019.00256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmadfa I., Meyer A.L. The role of the status of selected micronutrients in shaping the immune function. Endocrine, Metabolic & Immune Disorders - Drug Targets. 2019;19(8):1100–1115. doi: 10.2174/1871530319666190529101816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh T., Beniwal A., Semwal A., Navani N.K. Mechanistic insights into probiotic properties of lactic acid bacteria associated with ethnic fermented dairy products. Frontiers in Microbiology. 2019;10:502. doi: 10.3389/fmicb.2019.00502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gombart A.F., Pierre A., Maggini S. A review of micronutrients and the immune system–working in harmony to reduce the risk of infection. Nutrients. 2020;12(1):236. doi: 10.3390/nu12010236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grein J., Ohmagari N., Shin D., Diaz G., Asperges E., Castagna A., et al. Compassionate Use of remdesivir for patients with severe covid-19. New England Journal of Medicine. 2020;382:2327–2336. doi: 10.1056/NEJMoa2007016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grifoni A., Weiskopf D., Ramirez S.I., Mateus J., Dan J.M., Moderbacher C.R., et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-disease and unexposed individuals. Cell. 2020 doi: 10.1016/j.cell.2020.05.015. 2020, publish online May 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu S., Chen Y., Wu Z., Chen Y.-B., Gao H.-N., Lv L.-X., et al. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America; 2020. Alterations of the gut microbiota in patients with COVID-19 or H1N1 influenza. 2020, publish online June 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagan T., Cortese M., Rouphael N., Boudreau C., Linde C., Maddur M.S., et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178:1313–1328. doi: 10.1016/j.cell.2019.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hooda S., Boler B.M.V., Serao M.C.R., Brulc J.M., Brulc J.M., Staeger M.A., et al. 454 pyrosequencing reveals a shift in fecal microbiota of healthy adult men consuming polydextrose or soluble corn fiber. Journal of Nutrition. 2012;142:1259–1265. doi: 10.3945/jn.112.158766. [DOI] [PubMed] [Google Scholar]
- Iddir M., Brito A., Dingeo G., Campo S.S.F.D., Samouda H., Frano M.R.L., et al. Strengthening the immune system and reducing inflammation and oxidative stress through diet and nutrition: Considerations during the COVID-19 crisis. Nutrients. 2020;12(6):1562. doi: 10.3390/nu12061562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuka S., Tanaka S. Modulation of CD8(+) intraepithelial lymphocyte distribution by dietary fiber in the rat large intestine. Experimental Biology and Medicine. 2002;227(11):1017–1021. doi: 10.1177/153537020222701110. [DOI] [PubMed] [Google Scholar]
- Ivanov I.I., Frutos R.de L., Manel N., Yoshinaga K., Rifkin D.B., Sartor R.B., et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host & Microbe. 2008;4(4):337–349. doi: 10.1016/j.chom.2008.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jespersen L., Tarnow I., Eskesen D., Morberg C.M., Michelsen B., Bügel S., et al. Effect of lactobacillus paracasei subsp. paracasei, L. casei 431 on immune response to influenza vaccination and upper respiratory tract infections in healthy adult volunteers: A randomized, double-blind, placebo-controlled, parallel-group study. American Journal of Clinical Nutrition. 2015;101(6):1188–1196. doi: 10.3945/ajcn.114.103531. [DOI] [PubMed] [Google Scholar]
- Jew S., AbuMweis S.S., Jones P.J. Evolution of the human diet: Linking our ancestral diet to modern functional foods as a means of chronic disease prevention. Journal of Medicinal Food. 2009;12(5):925–934. doi: 10.1089/jmf.2008.0268. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K., Ward S.A., Kalantar-Zadeh K., El-Omar E.M. Considering the effects of microbiome and diet on SARS-CoV-2 infection: Nanotechnology roles. ACS Nano. 2020;14:5179–5182. doi: 10.1021/acsnano.0c03402. [DOI] [PubMed] [Google Scholar]
- King S., Glanville J., Sanders M.E., Fitzgerald A., Varley D. Effectiveness of probiotics on the duration of illness in healthy children and adults who develop common acute respiratory infectious conditions: A systematic review and meta-analysis. British Journal of Nutrition. 2014;112(1):41–54. doi: 10.1017/S0007114514000075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaenhammer T.R., Kleerebezem M., Kopp M.V., Rescigno M. The impact of probiotics and prebiotics on the immune system. Nature Reviews Immunology. 2012;12(10):728–734. doi: 10.1038/nri3312. [DOI] [PubMed] [Google Scholar]
- Kumova O.K., Fike A.J., Thayer J.L., Nguyen L.T., Mell J.C., Pascasio J., et al. Lung transcriptional unresponsiveness and loss of early influenza virus control in infected neonates is prevented by intranasal Lactobacillus rhamnosus GG. PLoS Pathogens. 2019;15(10) doi: 10.1371/journal.ppat.1008072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lauc G., Pezer M., Rudan I., Campbell H. Mechanisms of disease: The human N-glycome. Biochimica et Biophysica Acta (BBA) - General Subjects. 2016;1860(8):1574–1582. doi: 10.1016/j.bbagen.2015.10.016. [DOI] [PubMed] [Google Scholar]
- Lauc G., Sinclair D. Biomarkers of biological age as predictors of COVID-19 disease severity. Aging (U.S.A.) 2020;12(8):6490–6491. doi: 10.18632/aging.103052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee N., Kim W.U. Microbiota in T-cell homeostasis and inflammatory diseases. Experimental & Molecular Medicine. 2017;49(5):e340. doi: 10.1038/emm.2017.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li G., Fan Y.-H., Lai Y.-N., Han T.-T., Li Z.-H., Zhou P.-W., et al. Coronavirus infections and immune responses. Journal of Medical Virology. 2020;92(4):424–432. doi: 10.1002/jmv.25685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q., Guan X.-H., Wu P., Wang X.-Y., Zhou L., Tong Y.-Q., et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. New England Journal of Medicine. 2020;382(13):1199–1207. doi: 10.1056/NEJMoa2001316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li R.-Y., Pei S., Chen B., Song Y.-M., Zhang T., Yang W., et al. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2) Science. 2020;368(6490):489–493. doi: 10.1126/science.abb3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu R., Hong J., Xu X., Feng Q., Zhang D.-Y., Gu Y.-Y., et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nature Medicine. 2017;23(7):859–868. doi: 10.1038/nm.4358. [DOI] [PubMed] [Google Scholar]
- Li J.-Q., Zhao J.-J., Wang X.-D., Qayum A., Hussain M.A., Liang G.-Z., et al. Novel angiotensin-converting enzyme-inhibitory peptides from fermented bovine milk started by lactobacillus helveticus KLDS.31 and lactobacillus casei KLDS.105: Purification, identification, and interaction mechanisms. Frontiers in Microbiology. 2019;10:2643. doi: 10.3389/fmicb.2019.02643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez-Siles M., Duncan S.H., Garcia-Gil L.J., Martinez-Medina M. Faecalibacterium prausnitzii: From microbiology to diagnostics and prognostics. The ISME Journal. 2017;11(4):841–852. doi: 10.1038/ismej.2016.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo L., Qin T., Huang Y.-F., Zheng S.-S., Bo R.-N., Liu Z.-G., et al. Exploring the immunopotentiation of Chinese yam polysaccharide poly (lactic-co-glycolic acid) nanoparticles in an ovalbumin vaccine formulation in vivo. Drug Delivery. 2017;24(1):1099–1111. doi: 10.1080/10717544.2017.1359861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maggini S., Pierre A., Calder P. Immune function and micronutrient requirements change over the life course. Nutrients. 2018;10:1531. doi: 10.3390/nu10101531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mak J.W.Y., Chan F.K.L., Ng S.C. Probiotics and COVID-19: One size does not fit all. The Lancet Gastroenterology & Hepatology. 2020;5(7):644–645. doi: 10.1016/S2468-1253(20)30122-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marteau P., Seksik P. Tolerance of probiotics and prebiotics. Journal of Clinical Gastroenterology. 2004;38(6 Suppl):S67–S69. doi: 10.1097/01.mcg.0000128929.37156.a7. [DOI] [PubMed] [Google Scholar]
- McCann K.S. The diversity-stability debate. Nature. 2000;405(6783):228–233. doi: 10.1038/35012234. [DOI] [PubMed] [Google Scholar]
- Mierau I., Kleerebezem M. 10 years of the nisin-controlled gene expression system (NICE) in Lactococcus lactis. Applied Microbiology and Biotechnology. 2005;68(6):705–717. doi: 10.1007/s00253-005-0107-6. [DOI] [PubMed] [Google Scholar]
- Minato T., Nirasawa S., Sato T., Yamaguchi T., Hoshizaki M., Inagaki T., et al. B38-CAP is a bacteria-derived ACE2-like enzyme that suppresses hypertension and cardiac dysfunction. Nature Communications. 2020;11(1):1058. doi: 10.1038/s41467-020-14867-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mookherjee N., Anderson M.A., Haagsman H.P., Davidson D.J. Antimicrobial host defence peptides: Functions and clinical potential. Nature Reviews Drug Discovery. 2020;19(5):311–332. doi: 10.1038/s41573-019-0058-8. [DOI] [PubMed] [Google Scholar]
- Mozaffarian D., Fahimi S., Singh G.M., Micha R., Khatibzadeh S., Engell R.E., et al. Global sodium consumption and death from cardiovascular causes. New England Journal of Medicine. 2014;371:624–634. doi: 10.1056/NEJMoa1304127. [DOI] [PubMed] [Google Scholar]
- Naama G., Esen S., Lindsay K., Lesley P., Tze G.T., Adriana O., et al. Mining the human gut microbiota for immunomodulatory organisms. Cell. 2017;168(5):928–943. doi: 10.1016/j.cell.2017.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S.C., Tilg H. COVID-19 and the gastrointestinal tract: More than meets the eye. Gut. 2020;69(6):973–974. doi: 10.1136/gutjnl-2020-321195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palleja A., Mikkelsen K.H., Forslund S.K., Kashani A., Allin K.H., Nielsen T., et al. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nature Microbiology. 2018;3(11):1255–1265. doi: 10.1038/s41564-018-0257-9. [DOI] [PubMed] [Google Scholar]
- Ramirez-Farias C., Slezak K., Fuller Z., Duncan A., Holtrop G., Louis P. Effect of inulin on the human gut microbiota: Stimulation of bifidobacterium adolescentis and faecalibacterium prausnitzii. British Journal of Nutrition. 2009;101(4):541–550. doi: 10.1017/S0007114508019880. [DOI] [PubMed] [Google Scholar]
- Reithner L. Insensible water loss from the respiratory tract in patients with fever. Acta Chirurgica Scandinavica. 1981;147 163-116. [PubMed] [Google Scholar]
- Ren J.-L., Zhang A.-H., Wang X.-J. Traditional Chinese medicine for COVID-19 treatment. Pharmacological Research. 2020;155:104743. doi: 10.1016/j.phrs.2020.104743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhodes J., Dunstan F., Laird E., Kenny R.A. Prevention & Health; 2020. COVID-19 mortality increases with northerly latitude after adjustment for age suggesting a link with ultraviolet and vitamin D. BMJ Nutrition. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rinninella E., Cintoni M., Raoul P., Lopetuso L.R., Scaldaferri F., Pulcini G., et al. Food components and dietary habits: Keys for a healthy gut microbiota composition. Nutrients. 2019;11(10):2393. doi: 10.3390/nu11102393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez H.N., Moroney J.B., Gan H.-Q., Shen T., Im J.L., Li T.-B., et al. B cell-intrinsic epigenetic modulation of antibody responses by dietary fiber-derived short-chain fatty acids. Nature Communications. 2020;11(1):60. doi: 10.1038/s41467-019-13603-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahramian I., Kalvandi G., Javaherizadeh H., Khalili M., Noori N.M., Delaramnasab M., et al. The effects of prebiotic supplementation on weight gain, diarrhoea, constipation, fever and respiratory tract infections in the first year of life. Journal of Paediatrics and Child Health. 2018;54(8):875–880. doi: 10.1111/jpc.13906. [DOI] [PubMed] [Google Scholar]
- Sokol H., Pigneur B., Watterlot L., Lakhdari O., Bermúdez-Humarán L.G., Gratadoux J., et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(43):16731–16736. doi: 10.1073/pnas.0804812105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trompette A., Gollwitzer E.S., Pattaroni C., Lopez-Mejia I.C., Riva E., Pernot J., et al. Dietary fiber confers protection against flu by shaping Ly6c (-) patrolling monocyte hematopoiesis and CD8(+) T cell metabolism. Immunity. 2018;48(5):992–1005. doi: 10.1016/j.immuni.2018.04.022. e8. [DOI] [PubMed] [Google Scholar]
- Trompette A., Gollwitzer E.S., Yadava K., Sichelstiel A.K., Sprenger N., Ngom-Bru C., et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nature Medicine. 2014;20(2):159–166. doi: 10.1038/nm.3444. [DOI] [PubMed] [Google Scholar]
- Trottein F., Sokol H. Potential causes and consequences of gastrointestinal disorders during a sars-cov-2 infection. Cell Reports. 2020;32(3):107915. doi: 10.1016/j.celrep.2020.107915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Puyenbroeck K., Hens N., Coenen S., Michiels B., Beunckens C., Molenberghs G., et al. Efficacy of daily intake of lactobacillus casei shirota on respiratory symptoms and influenza vaccination immune response: A randomized, double-blind, placebo-controlled trial in healthy elderly nursing home residents. American Journal of Clinical Nutrition. 2012;95(5):1165–1171. doi: 10.3945/ajcn.111.026831. [DOI] [PubMed] [Google Scholar]
- Volkert D., Beck A.M., Cederholm T., Cruz-Jentoft A., Goisser S., Hooper L., et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clinical Nutrition. 2019;38:10–47. doi: 10.1016/j.clnu.2018.05.024. [DOI] [PubMed] [Google Scholar]
- Walls A.C., Park Y.J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181(2):281–292. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D., Hu B., Hu C., Zhu F.-F., Liu X., Zhang J., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. Jama. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S., Xiao Y., Tian F., Zhao J., Chen W. Rational use of prebiotics for gut microbiota alterations: Specific bacterial phylotypes and related mechanisms. Journal of Functional Foods. 2020;66:103838. [Google Scholar]
- Watanabe Y., Berndsen Z.T., Raghwani J., Seabright G.E., Allen J.D., Pybus O.G., et al. Vulnerabilities in coronavirus glycan shields despite extensive glycosylation. Nature Communications. 2020;11(1):2688. doi: 10.1038/s41467-020-16567-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong C.C.M., Zhang L., Li Z.-J., Wu W.K.K., Ren S.-X., Chen Y.-C., et al. Protective effects of cathelicidin-encoding Lactococcus lactis in murine ulcerative colitis. Journal of Gastroenterology and Hepatology. 2012;27(7):1205–1212. doi: 10.1111/j.1440-1746.2012.07158.x. [DOI] [PubMed] [Google Scholar]
- Wu M.-Q., Feng H.-F., Song J.-X., Chen L.-X., Xu Z.-Z., Xia W., et al. Structural elucidation and immunomodulatory activity of a neutral polysaccharide from the Kushui Rose (Rosa setate x Rosa rugosa) waste. Carbohydrate Polymers. 2020;232:115804. doi: 10.1016/j.carbpol.2019.115804. [DOI] [PubMed] [Google Scholar]
- Xu Z., Shi L., Wang Y., Zhang J.-Y., Huang L., Zhang C., et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. The Lancet Respiratory Medicine. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D., Li S., Wang N., Tan H.Y., Zhang Z., Feng Y. The cross-talk between gut microbiota and lungs in common lung diseases. Frontiers in Microbiology. 2020;11:301. doi: 10.3389/fmicb.2020.00301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X.-N., Tan Y., Ling Y., Lu G., Liu F., Yi G.-Z., et al. Viral and host factors related to the clinical outcome of COVID-19. Nature. 2020;2020 doi: 10.1038/s41586-020-2355-0. publish online May 20. [DOI] [PubMed] [Google Scholar]
- Zhao J., Tian S.-S., Yang J., Liu J.-F., Zhang W.-D. Investigating mechanism of Qing-Fei-Pai-Du-Tang for treatment of COVID-19 by network pharmacology. Chinese Traditional and Herbal Drugs. 2020;51(4):829–835. [Google Scholar]
- Zheng D.-P., Liwinski T., Elinav E. Interaction between microbiota and immunity in health and disease. Cell Research. 2020;30(6):492–506. doi: 10.1038/s41422-020-0332-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu L.-H., She Z.-G., Cheng X., Qin J.-J., Zhang X.-J., Cai J.-J., et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metabolism. 2020;31:1068–1077. doi: 10.1016/j.cmet.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmermann M., Zimmermann-Kogadeeva M., Wegmann R., Goodman A. Mapping human microbiome drug metabolism by gut bacteria and their genes. Nature. 2019;570:462–467. doi: 10.1038/s41586-019-1291-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo T., Zhang F., Grace C.Y.L., Yun K.Y., Amy Y.L.L., Zhan H., et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159(3):944–955.e8. doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo T., Zhan H., Zhang F., Liu Q., Tso E.Y.K., Liu G.C.Y., et al. Alterations in fecal fungal microbiome of patients with COVID-19 during time of hospitalization until discharge. Gastroenterology. 2020;159(4):1302–1310.E5. doi: 10.1053/j.gastro.2020.06.048. [DOI] [PMC free article] [PubMed] [Google Scholar]