Abstract
Background
Patients receiving biologic therapies are at risk for viral infections. This study investigated the impact of the SARS-CoV-2 infection and the serum prevalence of SARS-CoV-2 antibodies in patients with inflammatory bowel disease (IBD) treated with biologic drugs.
Methods
Information on demography, co-morbidities, clinical data regarding IBD, symptoms suggestive of the SARS-CoV-2 infection, close contacts with SARS-CoV-2 positive patients, hospitalization, and therapies administered for COVID-19 was collected for all patients who were being treated with biologic drugs. All patients underwent SARS-CoV-2 antibody testing.
Results
Two hundred and fifty-nine patients (27 children) with a mean age of 42.2 ± 16.7 years (range 9 - 88) and a mean duration of disease of 13.4 ± 10 years (range 0.2 – 49) were enrolled. One hundred four patients (40.2%) had ulcerative colitis, and 155 (59.8%) had Crohn's disease. About the therapy: 62 patients were receiving infliximab, 89 adalimumab, 20 golimumab, 57 vedolizumab, 27 ustekinumab, 1 thalidomide, and 3 an experimental compound. The mean Charlson Comorbidity Index was 2. Thirty-two patients (12.3%) reported respiratory symptoms, and 2 of them were hospitalized (0.77%). Two patients resulted positive for IgG against SARS-CoV-2 (0.77%).
Conclusions
In patients with IBD, treatment with biologic drug does not represent a risk factor for the SARS-CoV-2 infection.
Keywords: Biologic therapy, IBD, SARS-CoV-2
1. Introduction
The 2019–2020 Coronavirus disease (COVID-19) outbreak is an ongoing pandemic caused by a novel Coronavirus named Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2), initially identified in Wuhan, China, where the first 5 patients were hospitalized in December 2019 [1]. At the end of January 2020, 7734 cases were confirmed in China, and 90 other cases were reported from several European countries, such as Germany, France, and Finland [2]. The first 16 Italian patients infected with SARS-CoV-2 were registered on February 21, 2020, in Codogno (Northern Italy). Since then, the virus has spread throughout Italy. By July 19, 2020, over 244.000 individuals had been infected, of whom 35.000 died [3]. The median age of infected patients was 64 years, and about one third of them presented with a severe disease which required admission to an intensive care unit in 5% of cases [3]. Factors associated with an aggressive course of the infection were: older age, male sex, concomitant co-morbidities (cardiomyopathy, hypertension, kidney failure, and chronic obstructive pulmonary disease), obesity, and active smoking [4], [5], [6], [7]. The role of air pollution is still under debate [8].
Patients with inflammatory bowel disease (IBD) treated with biologics and/or immunosuppressant drugs are at higher risk for opportunistic infections [9]. A single-center study, conducted on 522 IBD patients (both adult and pediatric subjects) living in an urban area with a high prevalence of COVID-19 infection, found no infected subjects either among those receiving immunosuppressant drugs (no.=22%) or biologics (no.=16%), or among those not treated with this class of compounds [10]. A multicenter study carried out by the Italian Group for Inflammatory Bowel Disease (IG-IBD) collected 79 cases of IBD patients with the SARS-CoV-2 infection, ensuing in death in 6 patients [11]. No IBD-specific features resulted associated with a poor outcome (pneumonia, need for respiratory therapies, hospitalization, and death), whereas older age, male sex, and presence of co-morbidities were all significant predictors of a worse outcome [11].
Despite the current pandemic, scientific societies recommend maintaining IBD patients on their ongoing therapies, be these based on immunosuppressant or biologic drugs, as no evidence has yet incriminated these drugs as a potential factor favoring and/or worsening the Coronavirus disease [12,13]. Nevertheless, this indication needs to be backed by real-world data exploring the safety of these therapies during the current pandemic [14,15]. Two studies investigated the serum prevalence of SARS-CoV-2 infection in IBD patients [16,17]. In the first one, 90 out of 103 patients under current biologics therapy were investigated for the presence of IgG and or IgM against SARS-CoV-2 in the circulation: 19 of them resulted positive for IgG, IgM, or both (21%), suggesting that the majority of patients had gone through an asymptomatic course of infection [16]. Of note, this seroprevalence data was similar to that encountered in a healthy control population. At multivariate analysis, male sex was confirmed as protective for the COVID-19 infection, while older age as more likely associated with a positive serological result [16]. Bertè et al. tested, with a homemade ELISA assay for the detection of anti-SARS-CoV-2 specific IgG and IgA, 354 patients with IBD from 3 different center treated with biologics: no significant differences were found in the IBD patients when compared with a control population of healthy subjects [17]. Only the presence of anosmia/ageusia was an independent predictor of IgG seropositivity at multivariate analysis (RR54.5, 95%CI 2.1–1434.9, p = 0.016) [17].
The aim of our study was to explore the risk of acquiring the SARS-CoV-2 infection and to evaluate the severity of the disease in patients with IBD treated with biologics.
2. Materials and methods
All patients followed up at the IBD center at the Casa Sollievo della Sofferenza Research Hospital (San Giovanni Rotondo, Italy) who received at least one injection of a biologic drug for IBD from February 1st, 2020 on, were enrolled. Data on age, sex, IBD (type, duration of disease, smoking habit, and ongoing therapies), and comorbidities according to the Charlson Comorbidity Index (CCI) [17] were collected. All patients were accurately interviewed (by phone for patients treated with subcutaneous biologics) to investigate the possible onset of at least one of the following symptoms during the study period: cough, sneeze, dysosmia, dysgeusia, sore throat, fever, diarrhea, chest pain, dyspnea, musculoskeletal pain, and asthenia. In addition, all information regarding close contacts with individuals who tested positive for SARS-CoV-2, needed quarantine, had a diagnosis of COVID-19 and were consequently hospitalized and administered therapies, were carefully investigated. The diagnosis of SARS-CoV-2 infection was confirmed if the genome of this novel Coronavirus was found in a nasopharyngeal swab. If the test was negative, the diagnosis was considered probable if patients had close contacts with an infected person and/or a person having 3 or more of the following signs and symptoms: cough, dyspnea, fever, dysosmia, and dysgeusia, or if the computerized tomography (CT) chest scan revealed the signs commonly described for COVID-19 pneumonia.
From May 2020 on, patients have also been asked to donate a blood sample for SARS-CoV-2 IgM/IgG antibody testing. In addition, human sera of healthy controls matched for age and sex, collected during the pre-COVID era (2018), were also analysed. Each sample (a 5 ml standard citrated plasma collection tube) was immediately centrifuged for 10 min at 3000 g and stored at −20 Celsius degrees. Furthermore, serum samples were collected from 9 patients with a confirmed diagnosis of SARS-CoV-2 infection, as they all tested positive on the nasopharyngeal swab.
2.1. Reorganization of the infusion administration
From mid-March 2020 on, all patients receiving an infusion of a biologic compound had to follow dedicated paths and use masks (surgical or FFP2) and gloves. Before entering the hospital, all patients and healthcare personnel had their body temperature checked; subjects with a temperature above 37.5 °C were not allowed to access. Social distance between patients (> 2 m) was maintained before and during the infusion. The health personnel who dealt with infusions was equipped with personal protective equipment (gloves, FFP2 masks, and facial shields).
2.2. SARS-CoV-2 rapid test
The rapid serological test POCT Diagnostic Kit for SARS-CoV-2 IgM/IgG antibody (Colloidal Gold) (KHB, Shanghai Kehua Bio-engineering Co., China, Cod. REF-423-25-C-CE) was used. The kit performance declared a sensitivity of about 90% and a specificity of 96.3%. For all the samples, the recommended serum volume was added to the specimen well on the individual test cassettes followed by the addition of the supplied buffer, in accordance with the manufacturer's instructions.
2.3. SARS-CoV-2 ELISA
To verify the reliability of positive results obtained by the rapid serological tests and to avoid the risk of false positive data, we tested all positive samples by means of the Enzyme-Linked Immunosorbent Assay (ELISA) technique, using commercially available kits (SARS-CoV-2 ELISA IgM and IgG: Cat. No. 30177448, and Cat. No. 30177447, respectively; Tecan, IBL) according to the manufacturer's recommendations. The results were interpreted as positive and negative; borderline results were interpreted as positive.
2.4. Urea dissociation test of qualitative and semi-quantitative assays
The urea dissociation test, recently proposed by Wang et al., is based on the principle that urea dissociates the antigen-antibody reactions [18,19]. The purpose of this test is to identify false positive results. The urea treatment was performed in sera from positive samples for IgM and/or IgG. The concentration (6 M) and the dissociated time of urea (25 min) were used in the same way as they were used in previous studies [18,19]; whereas, for ELISA, each sample/control was tested for two concentrations of urea in different wells: 4 M (reported as being the best one by Wang et al.) [18], and 6 M. Results were expressed as Affinity Index (AI) and interpreted according to the threshold values calculation method described by Wang et al. [18]. Finally, we also checked rheumatoid factor (RF) plasma levels of all IBD tested patients [18].
2.5. Study end points
The primary endpoint of our investigation was to highlight the prevalence of the COVID-19 infection in IBD patients treated with advanced targeted therapies. The secondary endpoint was the evaluation of the clinical course of the SARS-CoV-2 infection in IBD patients treated with biologic compounds.
2.6. Ethical considerations
The local Ethics Committee discussed and approved the study protocol in May 2020 (Prot n73/CE). The written informed consent was obtained from all patients.
3. Results
3.1. Patients population
Two hundred and fifty-nine patients were enrolled, of whom 232 (89.6%) were adults and 27 (10.4%) were pediatric subjects. The mean age of the overall population was of 42.2 ± 16.7 years (range 9 - 88): in particular, 18 patients (6.9%) were older than 65 years, and 165 subjects (63.7%) were males. Forty-seven subjects were current smokers (18.1%). The mean duration of the disease was 13.4 ± 10 years (range 0.2 – 49). One hundred six patients had significant comorbidities (40.9%) with a mean CCI score of 2. One hundred four patients (40.2%) had ulcerative colitis, and the remaining 155 (59.8%) had Crohn's disease. Regarding the type of biologic therapy administered, 62 (24.0%) patients were on infliximab; 89 (34.4%) on adalimumab; 20 (7.7%) on golimumab; 57 (22.0%) on vedolizumab; and 27 (10.4%) on ustekinumab; 3 adult patients (1.1%) were participating in clinical trials and were receiving a new experimental biologic drug; and 1 pediatric patient (0.4%) was treated with thalidomide. Thirty-two patients (12.3%) were under current treatment with a combination therapy (biologic plus immunosuppressant drug). Three hundred forty-three infusions of biologics were performed at our hospital between February and June 2020. Baseline information is shown in Table 1 .
Table 1.
Demographic and clinical characteristics of the enrolled patients.
| IBD patients | Patients with symptoms | Healthy controls | |
|---|---|---|---|
| Number of patients, n (%) | 259 | 32 (12.3) | 214 |
| Adult subjects (%) | 232 (89.6%) | 32 (100) | |
| Pediatric subjects (%) | 27 (10.4%) | 0 (0.0) | |
| Male (%) | 165 (63.7%) | 20 (62.5) | 117 (54.7) |
| Mean age, years ± SD, (range) | 42.2 ± 16.7 (9–88) | 44.2 ± 12.8 (20–67) | 58.1 ± 16.3 (20–90) |
| Diagnosis: | |||
| CD, n (%) | 155 (59.8%) | 21 (65.6) | |
| UC, n (%) | 104 (40.2%) | 11 (34.4) | |
| Mean disease duration, years ± SD (range) | 13.4 ± 10.0 (0.2–49) | 16.2 ± 11.1 (2–36) | |
| Smoking habit: | |||
| non smokers, n (%) | 151 (58.3) | 14 (43.7) | |
| current smokers, n (%) | 47 (18.1) | 13 (40.6) | |
| ex smokers, n (%) | 61 (23.5) | 5 (15.6%) | |
| Co-morbidities | 106 (40.9) | 15 (46.9%) | |
| cardiovascular disease, n (%) | 32 (12.3) | 2 (6.2%) | |
| Diabetes, n (%) | 12 (4.6) | 2 (6.2%) | |
| respiratory disease, n (%) | 5 (1.9) | 0 (0.0) | |
| renal disease, n (%) | 6 (2.3) | 1 (3.1%) | |
| Others, n (%) | 87 (33.6) | 12 (37.5%) | |
| Biologic therapy: | |||
| Infliximab, n (%) | 62 (23.9) | 8 (25) | |
| Adalimumab, n (%) | 89 (34.4) | 11 (34.4) | |
| Golimumab, n (%) | 20 (7.7) | 3 (9.4) | |
| Vedolizumab, n (%) | 57 (22.0) | 7 (21.9) | |
| Ustekinumab, n (%) | 27 (10.4) | 3 (9.4) | |
| Others, n (%) | 4 (1.5) | 0 (0.0) | |
| Combination therapy with IS, n (%) | 32 (12.3) | 5 (15.6) | |
| Concomitant therapy for co-morbidities, n (%) | 54 (20.8) | 5 (15.6) | |
| Symptoms: | |||
| Dysosmia and/or dysgeusia, n (%) | – | 2 (6.3) | |
| Cough, n (%) | – | 12 (37.5) | |
| Fever, n (%) | – | 10 (31.1) | |
| Dyspnea, n (%) | – | 2 (6.2) | |
| Diarrhea, n (%) | – | 2 (6.2) | |
| Others, n (%) | – | 21 (65.6) |
The control group consisted of 214 subjects with a mean age of 58.1 ± 16.3 years (range 20–90). Of these 214 subjects,117 (54.7%) were male.
3.2. COVID-19 infection and suspected symptoms of infection
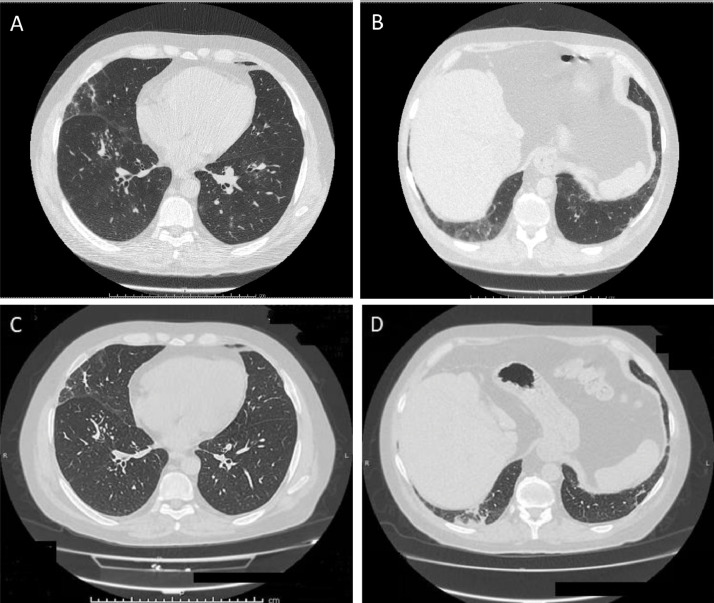
Thirty-two patients (12.3%) reported one or more symptoms suggestive for COVID-19, as shown in Table 1. One of them was on infliximab when he presented fever, cough, dyspnea, and dysosmia. He was hospitalized and underwent a nasopharyngeal swab (positive for SARS-CoV-2 infection) and the chest CT scan (negative for interstitial pneumonitis). The treatment included oral steroids and non-invasive ventilation for 3 days. A patient being treated with ustekinumab presented cough and underwent a nasopharyngeal swab, which tested positive. The patient was in good clinical conditions and did not require hospitalization. Another patient (treated with vedolizumab) was admitted for an interstitial pneumonia, though negative for the SARS-CoV-2 infection by real-time polymerase chain reaction (RT-PCR) on nasopharyngeal swab. Due to the rapid development of severe respiratory depression, he was urgently admitted to the intensive care unit, where he received a non-invasive ventilation for 3 days. The medical treatment included, also, hydroxychloroquine 200 mg twice a day for 10 days, low molecular weight heparin, and methylprednisolone 40 mg/day per 5 days with subsequent tapering. He was discharged at home after 3 weeks, where he continued systemic steroids (prednisone 25 mg/day), low molecular weight heparin and oxygen supplementation of 3 L/h for 16 h/day. CT scan images obtained at admission and before discharge are shown in Fig. 1 . The serology test for COVID-19 antibodies resulted negative.
Fig. 1.
Chest CT scan images at the time of hospital admission (A and B) and before discharge (C and D). Baseline CT images (A-B) show bilateral ground glass opacities with a peripheral and posterior distribution, mainly in the lower lobes, in association with thickened interlobular lines. Small pulmonary nodules are also present in the right lower lobe, many of them distributed in the secondary lobules. Ground glass opacifications localized in lower lobes evolve into parenchymal bands and perilobular consolidations at follow-up CT images (C-D), suggesting the possible presence of organizing pneumonia.
Another patient underwent a nasopharyngeal swab for SARS-CoV-2 and resulted negative. One more patient recalled having had a close contact with a confirmed case of COVID-19 infection and to have been consequently quarantined for 14 days. During this period, he remained symptoms-free. One additional patient, a healthcare professional exposed to numerous close contacts with individuals positive for COVID-19 disease, started complaining of fever, cough and asthenia despite wearing full protective disposable equipment, in compliance with by the internal guidelines and local protocols, but he was not tested with the nasopharyngeal swab.
3.3. The COVID-19 serological test
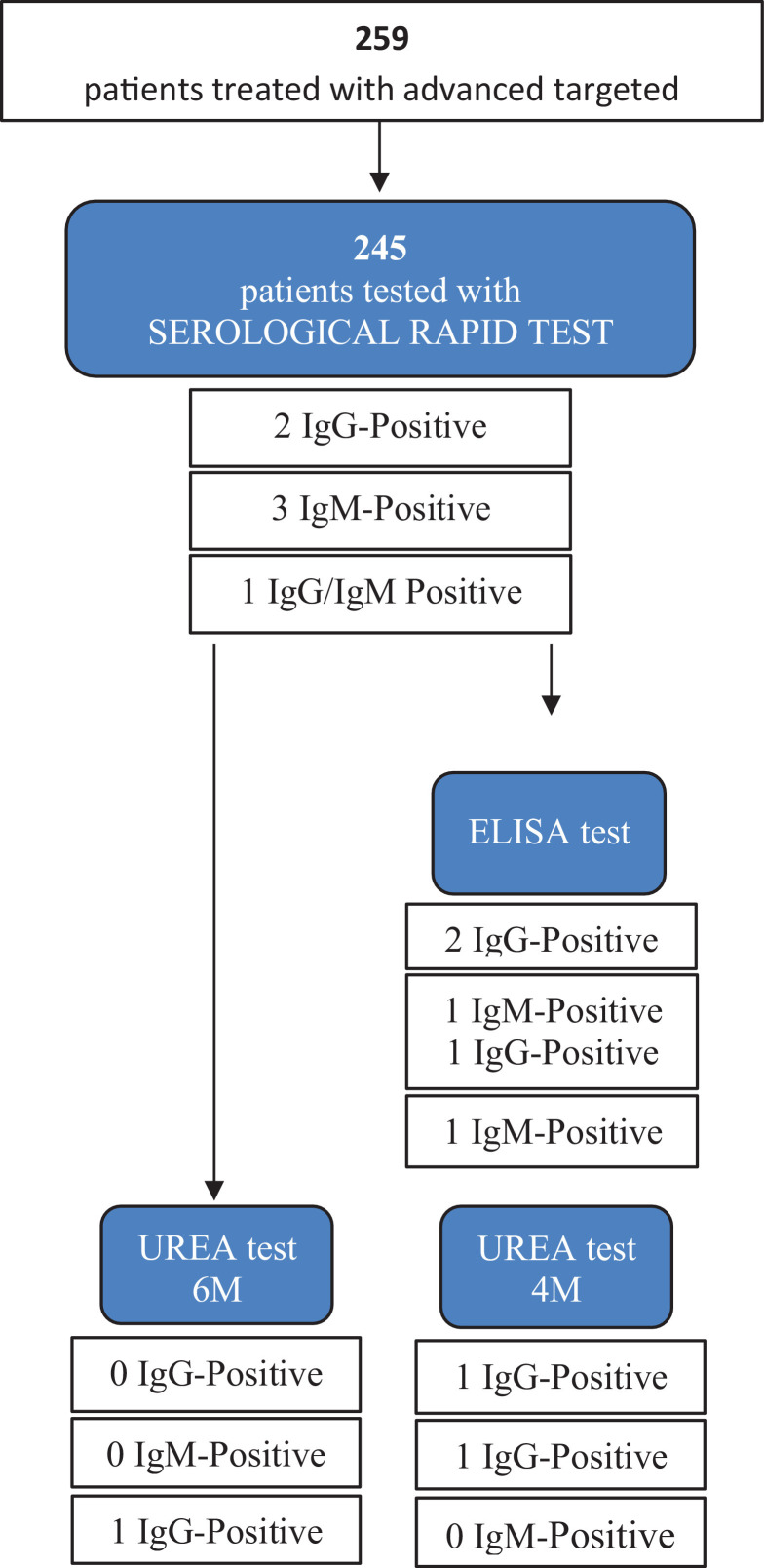
Two hundred and forty-five patients (96.1%) underwent the SARS-CoV-2 antibody test; twenty-six tested patients had previously reported one or more symptoms reminiscent of COVID-19 disease. Nine hospitalized patients with a confirmed diagnosis of COVID-19 (positive RT-PCR on nasopharyngeal swab) underwent the same serological rapid test and were used as controls. Six patients resulted positive at the SARS-CoV-2 IgM/IgG antibody rapid test (2.9% of the tested population). Of these, only 1 reported cough, sneeze and asthenia during the study period. All positive patients were subsequently re-tested with the ELISA kit for IgM and IgG, and 2 of them (0.88%) were positive for IgG (this result was confirmed also by the urea dissociation test). In all ymptomatic patients, the positivity at the rapid test was not confirmed by the ELISA assay. All the results of serological positive tests are shown in the Fig. 2 .
Fig. 2.
SARS-CoV-2 IgM/IgG antibodies serum results detected using the rapid serological test and the ELISA test after urea dissociation in sera positive on both methods. At the final evaluation, only 2 patients were considered definitely positive, as confirmed by the ELISA test after urea dissociation. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
All the healthy subjects underwent the serological rapid test and tested negative for both IgG and IgM.
4. Discussion
During the COVID-19 pandemic, patients on immunosuppressant therapy were considered to be at increased risk. The British Society of Gastroenterology (BSG) and the American Gastroenterological Association (AGA) issued guidelines for the appropriate management of IBD patients during the COVID-19 outbreak [12,13]. Both position papers were in favor of continuing administering biologic therapies to IBD patients as there is no evidence of increased risk associated with these drugs. Danese et al. suggested the IBD Units may be reorganized in a way that the safety of IBD patients is guaranteed during the COVID-19 pandemic while they continue receiving all the necessary therapies for IBD [20]. In particular, the authors recommend establishing checkpoints at the hospital entrances to screen people with suspected upper respiratory symptoms, to allow entry only to patients without accompanying people, to investigate close contacts with confirmed or suspected COVID-19 infected patients, to respect social distancing during visits and therapy administration, and to use protective personal equipment (surgical masks and gloves) [20].
The Surveillance Epidemiology of Coronavirus under Research Exclusion (SECURE)-IBD registry has reported 1760 cases of COVID-19 in IBD patients (85 registered in Italy), of whom 497 were hospitalized (28%) and 63 died (4%) [21]. Recently, a preliminary report of the SECURE-IBD registry on the initial 525 patients from 33 countries has been published. In this study, the multivariate analysis identified advanced age, the presence of at least 2 comorbidities, and the use of systemic steroids or sulfasalazine/5-aminosalicylate as risk factors associated with a worst course of the SARS-Cov-2 [22].
Regarding the spread of the COVID-19 infection in the Foggia province - the area in which our hospital is located-, 1171 cases of SARS-CoV-2 infection were reported by July 19th, 2020, with a mortality rate of 11.4% [23]. Comparing the death rate of the first 3 months of 2020 with the mean death rate registered between 2015 and 2019 during the same range of time, an increase of 20.6% of deaths can be observed [24]. On a total number of 622.000 inhabitants in Foggia [25], one case of SARS-Cov-2 (0.19%) every 537 residents was documented. However, it is possible that the total number of infected patients is much higher, since several studies showed that a portion of the population had an asymptomatic course of the COVID-19 infection [26,27], and a recent study has reported a nasopharyngeal swab sensitivity of 56–83% [28]. The rate of the COVID-19 infection in our cohort was comparable to that reported in the general population of the town of Foggia (0.77 vs 0.19; p = 0.5). Only 2 patients were admitted to hospital (one for a severe pneumonia, and the other one for fever and cough). None of the other 30 patients who reported signs or symptoms suggestive of COVID-19 was hospitalized, and in all cases the symptoms recovered spontaneously without treatment. Only 2 of these patients underwent nasopharyngeal swab, which resulted positive in 1 case. Only 2 patients were positive for IgG antibodies, but none of them experienced symptoms. Due to the lack of IgM antibodies, it is not possible to make any assumption about the time of infection. The serum prevalence of anti SARS-CoV-2 antibody in our cohort was similar to that reported in the Apulia Region -where the town of Foggia is located- using the CLIA or the ELISA assay (0.77 vs 0.9) [29]. We studied the largest cohort of IBD patients treated with biologic therapy in a single IBD center; also, we included both adults and children. The study by Berté et al. included a higher number of patients but from different 3 centres (2 in Italy and 1 in Germany) and with different epidemiological data. The study by Norsa et al. reported data on serum prevalence from a single center and included both adult and children, but only 90 patients were finally analysed. A major shortcoming of our study is the limited availability of nasopharyngeal swabs (only 4 patients of our cohort were tested).
Finally, the commercial immunoassay kits usually used the full-length protein (N or S, or even combined) as the coating antigen, which substantially increases the possibility of catching nonspecific antibodies. In addition, many β-Coronaviruses (more than 170 recorded in Pubmed) share similarities at sequencing. For these reasons, we cannot rule out a misdiagnosis due to interference factors and cross-reactivity with different Coronaviruses in our patients.
Conclusion
The frequency of the SARS-CoV-2 infection in patients with IBD on biologic therapy seems to be the same as that of the general population. No additional risks for COVID-19 emerged in our cohort of IBD patients treated with advanced targeted therapies.
Conflict of interest
None declared.
Acknowledgements
We thank Cristina Borrelli and Annalisa Simeone for their support in the interpretation of the chest CT scan images. We thank Chiara Di Giorgio for the expert revision of the English language.
References
- 1.Du Toit A. Outbreak of a novel coronavirus. Nat Rev Microbiol. 2020;18:123. doi: 10.1038/s41579-020-0332-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bassetti M., Vena A., Giacobbe D.R. The novel Chinese coronavirus (2019-nCoV) infections: challenges for fighting the storm. Eur J Clin Invest. 2020;50:e13209. doi: 10.1111/eci.13209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.2020. https://www.iss.it/infografiche.
- 4.Zhang J., Wang X., Jia X. Risk factors for disease severity, unimprovement, and mortality of COVID-19 patients in Wuhan, China. Clin Microbiol Infect. 2020 doi: 10.1016/j.cmi.2020.04.012. Apr 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Simonnet A., Chetboun M., Poissy J. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity. 2020 doi: 10.1002/oby.22831. Apr 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zheng Y., Xu H., Yang M. Epidemiological characteristics and clinical features of 32 critical and 67 noncritical cases of COVID-19 in Chengdu. J Clin Virol. 2020 doi: 10.1016/j.jcv.2020.104366. Apr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao Q., Meng M., Kumar R. The impact of COPD and smoking history on the severity of Covid-19: a systemic review and meta-analysis. J Med Virol. 2020 doi: 10.1002/jmv.25889. Apr 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ogen Y. Assessing nitrogen dioxide (NO2) levels as a contributing factor to coronavirus (COVID-19) fatality. Sci Total Environ. 2020;726 doi: 10.1016/j.scitotenv.2020.138605. Apr 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rahier J.F., Magro F., Abreu C. Second European evidence-based consensus on the prevention, diagnosis and management of opportunistic infections in inflammatory bowel disease. J. Crohn's Colitis. 2014;8:443–468. doi: 10.1016/j.crohns.2013.12.013. [DOI] [PubMed] [Google Scholar]
- 10.Noarsa L., Indriolo A., Sansotta N. Uneventful course in IBD patients during SARS-CoV-2 outbreak in northern Italy. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.03.062. Apr 2. pii: S0016-5085(20)30445-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bezzio C., Saibeni S., Variola A. Outcomes of Covid-19 in 71 patients with inflammatory bowel disease in Italy. Gut. 2020 [Google Scholar]
- 12.Kennedy N.A., Jones G., Lamb C.A. British society of gastroenterology guidance for management oif inflammatory bowel disease during the Covid-19 pandemic. Gut. 2020;0:1–7. doi: 10.1136/gutjnl-2020-321244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rubin D.T., Feuerstein J.D., Wang A.Y. AGA clinical practice update on management of inflammatory bowel disease during the COVID-19 pandemic: expert commentary. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.04.012. Apr 10;S0016-5085(20)30482-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Winthrop K.L., Melmed G.Y., Vermeire S. Herpes zoster infection in patients with ulcerative colitis receiving tofacitinib. Inflamm Bowel Dis. 2018;24(10):2258–2265. doi: 10.1093/ibd/izy131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pauly M.P., Tucker L.-.Y, Szpakowski J.-.L. Incidence of hepatitis b virus reactivation and hepatotoxicity in patients receiving long-term treatment with tumor necrosis factor antagonists. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc. 2018;16(12):1964–1973.e1. doi: 10.1016/j.cgh.2018.04.033. [DOI] [PubMed] [Google Scholar]
- 16.Norsa L., Cosimo P., Indriolo A. Asymptomatic severe acute respiratory syndrome Coronavirus 2 in patients with inflammatory bowel disease under biologic treatment. Gastroenterology. 2020;159:2229–2231. doi: 10.1053/j.gastro.2020.08.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Berté R., Mazza S., Stefanucci M.R. Seroprevalence of SARS-CoV2 in IBD patients treated with biological therapy. J Crohns Colitis. 2020 doi: 10.1093/ecco-jcc/jjaa237. Nov 19;jjaa237Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Charlson M.E., Pompei P., Ales K.L. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 19.Wang Q., Du Q., Guo B. A method to prevent SARS-CoV-2 IgM false positives in gold immunochromatography and enzyme-linked immunosorbent assays. J Clin Microbiol. 2020;58(6) doi: 10.1128/JCM.00375-20. e00375-20. Published 2020 May 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang Q., Lei Y., Lu X., Wang G., Du Q., Guo X., Xing Y., Zhang G., Wang D. Urea-mediated dissociation alleviate the false-positive Treponema pallidum-specific antibodies detected by ELISA. PLoS ONE. 2019;14 doi: 10.1371/journal.pone.0212893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Danese S., Cecconi M., Spinelli A. Management of IBD during the Covid-19 outbreak: resetting clinical priorities. Nat Rev Gastroenterol Hepatol. 2020;17:253–255. doi: 10.1038/s41575-020-0294-8. May. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.SECURE-IBD Registry. SECURE-IBD registry: surveillance epidemiology of coronavirus (COVID-19) under research exclusion, 2020. Available: https:// covidibd. org/ currentdata/ [Accessed 05th May 2020].
- 23.Brenner E.J., Ungaro R.C., Gearry R.B. Corticosteroids, but not TNF antagonists, are associated with adverse COVID-19 outcomes in patients with inflammatory bowel diseases: results from an international registry. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.05.032. May 18;S0016-5085(20)30655-7Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.2020. https://www.covid19puglia.it/.
- 25.2020. https://www.epicentro.iss.it/coronavirus/pdf/Rapp_Istat_Iss_9luglio.pdf.
- 26.Dobbiamo cambiare rotta, tampone a chi non ha sintomi. Corriere della Sera, Mar 2020. Https://corrierefiorentino.corriere.it/firenze/notizie/cronaca/20_marzo15/dobbiamo-cambiare-rotta-ef23a500-669a-11ea-a40-86d505f82a96.shtml.
- 27.Wong J., Abdul Aziz A.B.Z., Chaw L. High proportion of asymptomatic and presymptomatic Covid-19 infections in travelers and returning resident to Brunei. J Travel Med. 2020:066. doi: 10.1093/jtm/taaa066. May 5Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kokkinakis I., Selby K., Favrat B. Covid-19 diagnosis: clinical recommendations and performance of nasopharyngeal swab-PCR. Rev Med Suisse. 2020;16(689):699–701. Apr 8. [PubMed] [Google Scholar]
- 29.LL S. 2020. Primi risultati dell'indagine di sieroprevalenza SARS-CoV-2. [Google Scholar]