Coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has taken an immeasurable toll on humanity, with the number of deaths recently surpassing 1,588,000 globally and 293,000 in the United States, and many survivors suffering lingering symptoms and side effects (e.g., respiratory issues, post-infection fatigue syndrome, post-intensive care syndrome, blood clots, muscle aches, and cardiac damage). Even with this high incidence of morbidity and mortality, clinicians and scientists are still struggling to understand why some COVID-19 patients are asymptomatic and others require intensive care.
There has become an increasing appreciation of the vasculature in COVID-19 pathogenesis, and particularly the endothelium, in many of the complications that arise [1]. Specifically, endothelial dysfunction after SARS-CoV-2 infection includes barrier breakdown, leading to tissue edema and infiltration inflammatory cells, activation of intravascular coagulation pathways [2], and endotheliitis [3].
Recently, it has been revealed that the endothelium-dependent vasodilator, bradykinin, may also be involved [4,5]. Bradykinin, first discovered by three Brazilian scientists from Bothrops jararaca snake venom [6], is a downstream product of the kallikrein-kinin system, generated in the plasma by the proteolytic cleavage of the liver-derived kininogen by kallikrein serine proteases. Kallikrein exists in two distinct forms, plasma kallikrein and tissue kallikrein, and are separate gene products [7]. In fact, these two proteases have little amino acid sequence homology, even though they have related functions (i.e., cleavage of kininogens) [7]. Tissue kallikrein is secreted by many cells throughout the body and initiates the cleavage of predominantly low-molecular weight kininogen into Lys-bradykinin and then aminopeptidase generates bradykinin. On the other hand, plasma kallikrein is generated via intersection with the intrinsic coagulation pathway and cleaves high-molecular weight kininogen directly into bradykinin. Bradykinin is subsequently broken down by kininase I and kininase II, the latter also being known as angiotensin converting enzyme (ACE). Interestingly, ACE has a higher affinity for bradykinin than it does for angiotensin I [8] and therefore under conditions where ACE is low, bradykinin is preserved and the vasopressor effects of the renin-angiotensin-aldosterone system (RAAS) are minimized.
Bradykinin and its metabolites act on G protein-coupled receptors, B1 and B2 to exert vasoactive effects such as vasodilation and increased vascular permeability. Specifically, bradykinin and Lys-bradykinin are preferred B2 receptor agonists, where as their degradation metabolites, des-Arg9-BK and Lys-des-Arg9-BK, respectively, have greater high affinity and selectivity for B1 receptor [9] and the B1 receptor shows a greater preference for des-Arg9-BK than for bradykinin itself [10].
In addition to its role in pressure and fluid homeostasis via its intersection with the RAAS, bradykinin is part of the inflammatory response after injury and acts to induce pain [10], neutrophil recruitment, and increases in vascular permeability [11,12]. Specifically, the B2 receptor has been shown to play a role in the early stages of inflammatory pain, while in sustained inflammation the des-Arg9-BK and B1 receptor activity becomes more important for the hyperalgesia. Therefore, both the B1 and B2 receptors participate in inflammatory and pain response depend on stimuli or phase [13].
Recently, an important paper was published by Garvin and colleagues in eLife [4] reporting that bronchoalveolar lavage fluid (BALF) from COVID-19 patients have decreased gene expression of ACE in combination with increases in ACE2, renin, angiotensin, angiotensin receptors I and II, kininogen and many kallikrein enzymes that activate it, and both bradykinin receptors. As a result, the authors surmised that this milieu would likely induce a bradykinin storm, subsequently promoting vasodilation, vascular permeability, and hypotension. A possible source of this bradykinin in BALF from COVID-19 patients could be bronchiole and alveoli-resident mast cells. It is well known that as tissue resident granulocytes, mast cells can synthesize bradykinin via the secretion of heparin, activation of coagulation factor XII, and formation of plasma kallikrein [14,15]. Therefore, the increase in bradykinin may be due to the increased mast cells density in the lungs of COVID-19 patients [16].
The work by Garvin et al. expanded on an earlier publication by van de Veerdonk and colleagues, also published in eLife [5], that hypothesized that the bradykinin metabolite, des-Arg9-BK, could contribute to the inflammation, vasodilation, vascular permeability via activation of bradykinin receptors. The hypothesis proposed by van de Veerdonk et al. is significant because des-Arg9-BK is inactivated by ACE2 [17] and, in conflict with what was observed by Garvin [4], it has been well established that membrane bound ACE2 decreases after coronavirus infection [18]. One possible reason why Garvin measured increased ACE2 in BALF from COVID-19 patients could be because it is either the membrane shed form (via ADAM17-mediated proteolytic cleavage) or the soluble form. Nonetheless, the exact form of ACE2 was not delineated [4].
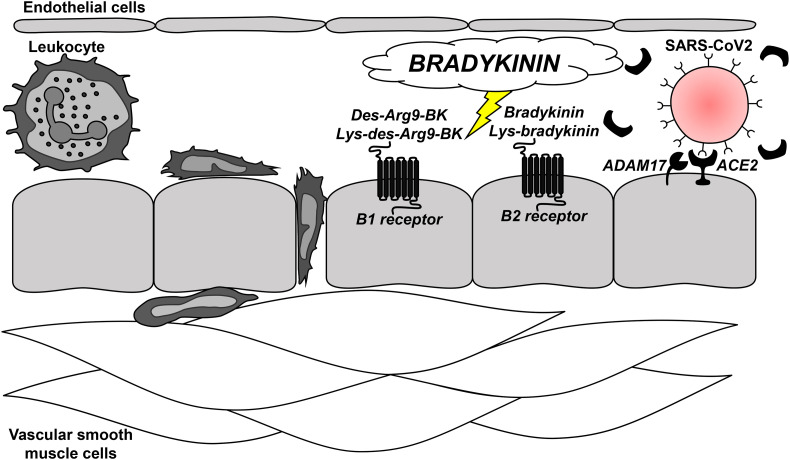
While the most publicized contribution of the RAAS to SARS-CoV-2 infection has been ACE2 due to its “Trojan Horse” like properties that allow it and other coronaviruses to enter into host cells [19], the work by Garvin [4], van de Veerdonk [5], and their colleagues reveal that bradykinin may be an important extension of this system to consider (Fig. 1). These papers are supported by a more recent single cell transcriptome analysis that observed that the kallikrein-kinin system, the RAAS, and the coagulation system are all co-expressed with ACE2 in alveolar cells [20]. These associations could help to explain how changes in ACE2 promoted by SARS-CoV-2 infection could manifest some of COVID-19's most severe clinical symptoms such as, lung edema, cardiovascular dysfunction, and thromboembolism.
Fig. 1.
Bradykinin signaling after SARS-CoV2 infection in pulmonary arterioles. Recent, high profile publications in eLife [4,5] have revealed that changes in the renin-angiotensin-aldosterone system after SARS-CoV2 infection could predispose bradykinin storms. Given that bradykinin and its metabolites are well-known inducers of endothelium-dependent vasodilation, vascular permeability, and pain via the activation of the G protein-coupled receptors B1 and B2, this signaling could be a novel therapeutic target in the treatment of COVID-19, including lung edema, cardiovascular dysfunction, and thromboembolism.
Acknowledgments
Acknowledgements
We would like to thank the reviewers for their helpful comments and expertise.
Disclosures
None.
Sources of funding
This work was supported by National Institutes of Health (K99HL151889, R00GM118885, and P01HL134604).
Conflict of interest
None.
References
- 1.Libby P, Luscher T. COVID-19 is, in the end, an endothelial disease. Eur.Heart J. 2020;41:3038–3044. doi: 10.1093/eurheartj/ehaa623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Teuwen LA, Geldhof V, Pasut A, Carmeliet P, et al. COVID-19: the vasculature unleashed. Nat.Rev.Immunol. 2020;20:389–391. doi: 10.1038/s41577-020-0343-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garvin MR, Alvarez C, Miller JI, Prates ET, Walker AM, et al. A mechanistic model and therapeutic interventions for COVID-19 involving a RAS-mediated bradykinin storm. Elife. 2020;9 doi: 10.7554/eLife.59177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van de Veerdonk FL, Netea MG, van Deuren M, van der Meer JW, de Mast Q, Bruggemann RJ, et al. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife. 2020;9 doi: 10.7554/eLife.57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rocha e Silva M, Beraldo WT, Rosenfeld G. Bradykinin, a hypotensive and smooth muscle stimulating factor released from plasma globulin by snake venoms and by trypsin. Am.J.Physiol. 1949;156:261–273. doi: 10.1152/ajplegacy.1949.156.2.261. [DOI] [PubMed] [Google Scholar]
- 7.Kaplan AP, Joseph K, Silverberg M. Pathways for bradykinin formation and inflammatory disease. J.Allergy Clin.Immunol. 2002;109:195–209. doi: 10.1067/mai.2002.121316. [DOI] [PubMed] [Google Scholar]
- 8.Jaspard E, Wei L, Alhenc-Gelas F. Differences in the properties and enzymatic specificities of the two active sites of angiotensin I-converting enzyme (kininase II). Studies with bradykinin and other natural peptides. J.Biol.Chem. 1993;268:9496–9503. [PubMed] [Google Scholar]
- 9.Dutra RC. Kinin receptors: key regulators of autoimmunity. Autoimmun.Rev. 2017;16:192–207. doi: 10.1016/j.autrev.2016.12.011. [DOI] [PubMed] [Google Scholar]
- 10.Dray A, Perkins M. Bradykinin and inflammatory pain. Trends Neurosci. 1993;16:99–104. doi: 10.1016/0166-2236(93)90133-7. [DOI] [PubMed] [Google Scholar]
- 11.Araujo RC, Kettritz R, Fichtner I, Paiva AC, Pesquero JB, Bader M. Altered neutrophil homeostasis in kinin B1 receptor-deficient mice. Biol.Chem. 2001;382:91–95. doi: 10.1515/BC.2001.014. [DOI] [PubMed] [Google Scholar]
- 12.Stuardo M, Gonzalez CB, Nualart F, Boric M, Corthorn J, Bhoola KD, et al. Stimulated human neutrophils form biologically active kinin peptides from high and low molecular weight kininogens, J. Leukoc.Biol. 2004;75:631–640. doi: 10.1189/jlb.1103546. [DOI] [PubMed] [Google Scholar]
- 13.Goncalves ECD, Vieira G, Goncalves TR, Simoes RR, Brusco I, Oliveira SM, et al. Bradykinin Receptors Play a Critical Role in the Chronic Post-ischaemia Pain Model. Cell.Mol.Neurobiol. 2020 doi: 10.1007/s10571-020-00832-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Brunnee T, Reddigari SR, Shibayama Y, Kaplan AP, Silverberg M. Mast cell derived heparin activates the contact system: a link to kinin generation in allergic reactions. Clin.Exp.Allergy. 1997;27:653–663. [PubMed] [Google Scholar]
- 15.Oschatz C, Maas C, Lecher B, Jansen T, Bjorkqvist J, Tradler T, et al. Mast cells increase vascular permeability by heparin-initiated bradykinin formation in vivo. Immunity. 2011;34:258–268. doi: 10.1016/j.immuni.2011.02.008. [DOI] [PubMed] [Google Scholar]
- 16.Motta Junior JDS, Miggiolaro AFRDS, Nagashima S, CBV de Paula, Baena CP, Scharfstein J, et al. Mast Cells in Alveolar Septa of COVID-19 Patients: A Pathogenic Pathway That May Link Interstitial Edema to Immunothrombosis. Front.Immunol. 2020;11:574862. doi: 10.3389/fimmu.2020.574862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sodhi CP, Wohlford-Lenane C, Yamaguchi Y, Prindle T, Fulton WB, Wang S, et al. Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg(9) bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration. Am.J.Physiol.Lung Cell.Mol.Physiol. 2018;314:L17–L31. doi: 10.1152/ajplung.00498.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat.Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.D Sidarta-Oliveira, CP Jara, AJ Ferruzzi, MS Skaf, WH Velander, EP Araujo, et al. SARS-CoV-2 receptor is co-expressed with elements of the kinin-kallikrein, renin-angiotensin and coagulation systems in alveolar cells, Sci.Rep. 10 (2020) 19522–020–76488-2. [DOI] [PMC free article] [PubMed]