Abstract
Angiotensin-converting enzyme 2 (ACE2) is an important player of the renin-angiotensin-aldosterone system (RAAS) in regulating the conversion of angiotensin II into angiotensin (1-7). While expressed on the surface of human cells, such as lung, heart, kidney, neurons, and endothelial cells (EC), ACE2 is the entry receptor for SARS-CoV-2. Here, we would like to highlight that ACE2 is predominant on the EC membrane. Many of coronavirus disease 2019 (COVID-19) symptoms have been associated with the large recruitment of immune cells, directly affecting EC. Additionally, cytokines, hypoxia, and complement activation can trigger the activation of EC leading to the coagulation cascade. The EC dysfunction plus the inflammation due to SARS-CoV-2 infection may lead to abnormal coagulation, actively participating in thrombo-inflammatory processes resulting in vasculopathy and indicating poor prognosis in patients with COVID-19. Considering the intrinsic relationship between EC and the pathophysiology of SARS-CoV-2, EC-associated therapies such as anticoagulants, fibrinolytic drugs, immunomodulators, and molecular therapies have been proposed. In this review, we will discuss the role of EC in the lung inflammation and edema, in the disseminate coagulation process, ACE2 positive cancer patients, and current and future EC-associated therapies to treat COVID-19.
Keywords: Endothelial cells, SARS-CoV-2, ACE2, Inflammation, Coagulation
Graphical abstract
1. Introduction
In December 2019, in the city of Wuhan, China, the first cases of a new respiratory infection were reported [1]. Experts determined that the disease, later termed COVID-19, was caused by a novel coronavirus. Since COVID-19 can cause severe acute respiratory distress syndrome (SARS), the new coronavirus was named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. COVID-19 has since led to the deaths of thousands of people and had caused a serious global impact on both health and economies. Further investigation into the evolution of symptoms in patients with COVID-19 is necessary, since patients may be asymptomatic or have mild, moderate or severe symptoms. Patients with mild and moderate symptoms have fever, fatigue, and cough. Patients with viral pneumonia or systemic inflammation are considered to be severe symptoms [2]. Patients with dysfunctional coagulation are considered to be in the critical phase [3]. The SARS-CoV-2 virus can penetrate the host cell by binding to cell surface receptors [4]. As described by Yuki et al (2020) [3], the virus enters the host cell through endocytosis or membrane fusion (penetration), and the viral RNA is then released into the cells and initiates synthesis of viral proteins.
Proteins, glycoproteins and proteoglycans on the surface of host cells, such as the serine transmembrane protein 2 (TMPRSS2) and Heparan Sulfate Proteoglycans (HSPG), are important for the initial interaction between viruses and cells [[5], [6], [7], [8], [9], [10], [11]]. Other proteins that can act as viral receptors, such as sialic acid receptors [12,13], the matrix metalloproteinase inducer CD147 [14] and ACE2, then mediate viral entry into the host cell [15]. ACE2, which is part of the RAAS [16,17] is currently the most studied receptor in the context of SARS-CoV-2 [17] and is considered to be important for viral infection [18]. There is evidence that the virus interacts with ACE2 through its spike protein, a trimeric transmembrane glycoprotein that is important for determining viral diversity and host tropism [3,15,16,19]. Experimentally, the spike protein of SARS-CoV-2 has been shown to have high affinity for human ACE2 [7,20]. Thus, it is possible to use this affinity to estimate the density of ACE2 in different types of tissue [15] and it has been suggested that the density of ACE2 in each tissue may correlate with the severity of disease in that tissue [[21], [22], [23], [24], [25]]. Additionally, it has been demonstrated that binding of heparin or heparan sulfate can induce significant conformational changes in the spike protein structure and that the receptor binding domain of the spike subunit contains a heparan sulfate binding site [26,27]. Recently, evidence has demonstrated that HSPG is a cell surface proteoglycan co-receptor with the ACE2 protein for recognition of SARS-CoV-2 spike protein [[28], [29], [30]].
ACE2 is expressed in several organs, including the lung, heart, kidney and intestine, and is also expressed in EC [31]. Although ACE2 is highly expressed in the lung [3,22,32], levels of ACE2 expression in enterocytes are higher than in the lung [[33], [34], [35], [36]]. Based on brain transcriptome data, Chen et al. (2020) [37] reported that, in rats, ACE2 is expressed in the substantia nigra, choroid plexus, non-neuronal cells and many neurons, both excitatory and inhibitory. Expression of ACE2 in the brain has been suggested to contribute to the neurological symptoms associated with COVID-19 [33], as well as the development of neurogenic hypertension [38]. Other studies have indicated the presence of SARS-CoV-2 particles in brain neurons of infected patients [33,[39], [40], [41]]. It has also been suggested that interaction of SARS-CoV-2 with ACE2 in the epithelium of the oral and nasal mucosa leads to the damage reported in these regions [21,33].
Here, we would like to highlight that ACE2 is predominantly expressed in EC [37], which make up the lining of blood and lymph vessels. Endothelial cells are of fundamental importance in the functioning of the vascular endothelium, which regulates vasodilation, fibrinolysis, aggregation and thrombosis [3,15,42]. ACE2 is present in EC in lung tissue, which represent one third of cells present in the lung [3]. COVID-19 leads to systemic changes in the organization and basic functions of EC, suggesting that these cells are important for the pathophysiology of this disease [2,31,43].
COVID-19 has been associated with extensive recruitment of immune cells, which directly affect EC or act in an immune-mediated manner [31,44]. Inflammatory processes play an important role in the development of COVID-19 and EC are key players in these processes. In this article, we will discuss the role of EC in lung inflammation and edema, disseminated coagulation, and in patients with ACE2-positive cancer. We will also discuss current EC-related therapies to treat COVID-19 and possibilities for the future.
2. Endothelial cells in SARS-CoV-2 infection: Inflammation and pulmonary edema
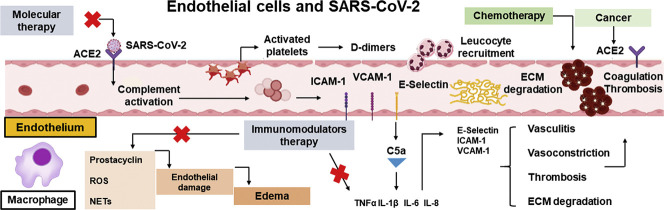
When the immune system generates an effective adaptive response against SARS-CoV-2, the infection can be eliminated and clinical manifestations will become absent or may completely disappear [45]. A successful antiviral response is dependent on the expression of type I interferons (IFNs) [[46], [47], [48]] and on the activation of T lymphocytes (TCD4+ helper cells (Th) and TCD8+ cytotoxic cells), as well as specific antibody-producing B lymphocytes. If the host organism does not develop an adequate adaptive immune response, the prolongation and amplification of innate mechanisms, combined with dysfunctional adaptive responses, lead to a hyperinflammatory state [45]. The resulting intense and continuous release of pro-inflammatory cytokines underlies the cytokine storm [45] that is characteristic of acute respiratory distress syndrome (ARDS) [[45], [46], [47], [48]], and is responsible for lung damage [45]. Patients with severe COVID-19 have decreased IFN production, as well as aberrant polarization of Th cells (predominantly Th17), increased expression of exhaustion-related surface markers, such as TIM3 and PD-1, and changes in the pattern of cytokine secretion [45,48,49]. Histochemical analysis of lung tissues from patients who died from COVID-19 showed deposition of mannose-binding lectin (MBL), C3, C4 and C5b-C9, complement system proteins, in alveolar epithelial cells, as well as in pneumocytes, inflammatory cells and exudates of alveolar spaces, which indicates activation of the complement system in response to SARS-CoV-2 infection [50]. (Fig. 1 ).
Fig. 1.
Endothelial cells and pulmonary epithelium in SARS-CoV-2 infection. (A) The binding of SARS-CoV-2 to the ACE2 receptor on EC leads to the activation of the complement system, a set of plasma proteins with opsonization functions in the immune system. The activation of the complement system induces pro-inflammatory cytokines release (e.g.: TNFα, IL-1β, IL-6, IL-8) and the recruitment of phagocytic cells. The release of these cytokines promotes the activation of the endothelium leading to the expression of selectins (E-selectin and P-selectin) and integrins (ICAM and VCAM) and promote the recruitment of neutrophils and monocytes, causing damage to the endothelium through the release of ROS, production of NETs, degradation of the extracellular matrix (ECM) and more pro-inflammatory cytokines release. The lesion in the endothelium induces the activation of the coagulation cascade and platelet activation leading to coagulation and thrombosis and increasing D-dimers formation. (B) The SARS-CoV-2 infection activates NLRP3, a cytosolic receptor in phagocyte cells, leading to the inflammasome, activating caspase 1 and exacerbating the production of IL-1β and TNFα, which in turn, lead to the recruitment of neutrophils, increased production of ROS and NETs, causing endothelial damage, pulmonary edema, and cell death.
Epithelial and endothelial tissues are important airway barriers, regulating tissue homeostasis and acting to protect against pathogens and aggressive agents. In response to a viral infection, epithelial cells signal to the endothelium, which starts the recruitment of cells from the immune system, in a highly temporally regulated process [51]. Endothelium and epithelium, form a cell monolayer connected by tight junctions (TJ), which regulate the permeability of the monolayer and adherent junctions (AJ) that mediate intercellular adhesion interactions [52,53]. The pulmonary endothelium consists of macro and microvascular EC [54]. These pulmonary microvascular cells are part of the alveolar-capillary/epithelial-endothelial barrier [55].
Although SARS-CoV-2 primarily infects cells of the pulmonary epithelium, these cells act as a gateway, and alveolar damage is mediated mainly by endothelial damage, which results in activation of cytokines and chemokines and recruitment of cells from the immune system [49] SARS-CoV-2 infection is not the primary cause of tissue damage associated with COVID-19 [46,47]. Instead, the damage results from acute hyperinflammation caused by the host immune response, which is mediated by massive release of cytokines, such as interleukin (IL)-1β, IL-6 and tumor necrosis factor-α (TNF-α) [46]. These cytokines affect cells of the lung parenchyma, oxygen uptake and EC, and lead to endotheliitis, thrombotic events and intravascular coagulation [56]. Epithelial/endothelial lesions mediated by cytokines/chemokines can impair the integrity of the blood/air barrier, promoting vascular permeability, alveolar edema, leukocyte infiltration (macrophages and neutrophils) and hypoxia [45]. The injury caused by SARS-CoV-2 to epithelial cells expressing ACE2 leads to activation of the NLRP3 inflammasome, an acute immune response to the infection, which is directed by alveolar monocytes/macrophages [49,57]. These cells secrete TNF-α and IL-1β, leading to disseminated activation of NLRP3 and positive feedback that causes cell damage and death, extending to the vasculature and causing leakage and pulmonary edema [57]. TNF-α, generated mainly by activated macrophages, is present at high concentrations in the bronchoalveolar lavage fluid of patients with ARDS and can cause apoptotic death of both epithelial and EC in the lung [57,58]. IL-1β, generated by resident macrophages in response to activation of the NLRP3 pattern recognition receptor, plays a role in the recruitment and activation of neutrophils and is a key inducer of neutrophil extracellular traps (NETs) [57]. NETs are extracellular structures produced by extrusion of DNA, histones, cytotoxic proteins, myeloperoxidase and cathepsin by neutrophils [59,60]. Uncontrolled production of NETs causes damage to the endothelium, coagulation and thrombosis, and contributes to ARDS [60].
Large numbers of neutrophils in patients with severe COVID-19 are also associated with high levels of reactive oxygen species (ROS), which activate a cascade of reactions, leading to tissue damage, thrombosis and blood dysfunction [61]. The healthy endothelium produces antithrombotic molecules such as nitric oxide (NO) and prostacyclin (PGI2) [62]. The endothelial dysfunction observed in COVID-19 patients is related to oxidation and is associated with a decrease in NO levels and an increase in prothrombotic adhesion molecules [63]. Endothelial dysfunction, inflammation, ROS production and increased risk of thromboembolism are intricately linked. ROS stimulate the expression of tissue factor and inhibit protein C, a major anticoagulant [62]. During SARS-CoV-2 infection, activation of the angiotensin 1 receptor also causes increased ROS release, as well as excessive activation of the NLRP3 inflammasome and death of lung EC by pyroptosis [64], as demonstrated experimentally by Ratajczak et al. (2020) [65].
Acute inflammation in response to SARS-CoV-2 infection also damages EC through overactivation of the complement system, which is involved in intravascular coagulation and thrombosis. The complement system, which consists of a collection of plasma proteins, eliminates microorganisms through opsonization, recruitment of neutrophils and macrophages, generation of proinflammatory mediators, activation of the membrane attack complex and increased humoral immunity. This system is activated by three distinct pathways: the classical, alternative and lectin pathways, which lead to production of C3 and C5 [66]. These proteins are subsequently cleaved by their respective convertases, generating the anaphylatoxins, C3a and C5a [56,[66], [67], [68]]. Systemic activation of the complement system via the lectin pathway involves a pattern recognition protein, MBL which, together with the MBL-associated serine protease 2 (MASP-2), recognizes mannose residues expressed on various pathogens [67,69], possibly including the SARS-CoV-2 spike protein [50,67]. SARS-CoV-2 binds to its receptor ACE2 on EC and activates first the lectin pathway and then the classical pathway, leading to deposition of C3b [50]. The formation of C3b supports the alternative pathway and participates in formation of the convertases that cleave C5 in the terminal products of the complement system, C5a and C5b-C9 [56]. C5a activates proinflammatory cells, such as neutrophils, and stimulates release of cytokines, such as TNF-α, IL-1β, IL-6 and IL-8, thereby promoting microvascular thrombosis and fibrinolysis.
In cells such as neutrophils, EC and platelets, C5a stimulates the expression of adhesion molecules, such as ICAM-1, VCAM-1 and E-selectin [50]. These adhesion molecules favor the recruitment, adhesion and transendothelial migration of neutrophils and macrophages, which contributes to destruction of the subendothelial matrix and vasculitis through production of ROS [56,67]. ICAM-1 promotes fibrinogen and fibrin adhesion to EC, which contributes to leukocyte transmigration, endothelial dysfunction and increased risk of thrombus formation and vasoconstriction, when linked to fibrinogen and D-dimer [70]. C5a and the membrane attack complex lead to release of P-selectin and von Willebrand Factor (VWF) by EC, which promotes platelet aggregation and release of thrombomodulin, triggering the coagulation cascade [66]. Finally, C5b-C9 directly promotes platelet activation, leading to platelet aggregation, vascular injury and dysfunction, and coagulation [56].
Therefore, SARS-CoV-2 can induce injury to the epithelium- endothelium barrier and a rupture of this system. Studies comparing lung tissues in patients who died from COVID-19, demonstrated significant changes in endothelial morphology with loss of contact with the basement membrane and rupture of intercellular junctions [43]. This finding indicates that SARS-CoV-2 can penetrate further into the pulmonary microvasculature and infect EC, as they also express high ACE2 levels at the main entry-receptor. Consequently, the inflammation reaches endothelial levels and then intensifies. However, it is proposed that endothelial injury is due much more to inflammatory responses from the host due to epithelium infection rather than viral replication or increased viral load in EC [71]. In summary, direct or indirect activation of EC by SARS-CoV-2 infection culminates in pulmonary edema and triggers a coagulation cascade that is implicated in severe cases of COVID-19.
3. SARS-CoV-2 infection: Development of thromboembolic, cardiovascular and cerebrovascular complications
Endothelial cells play a crucial role in several physiologic processes, including maintaining homeostasis, blood rheology and vascular barrier function [72]. Physiological and pathological stimulation of ACE2 receptors, which are expressed by ECs in several organs, including the lungs, heart and brain [17,[73], [74], [75]], leads to activation of ECs and subsequent activation of the coagulation cascade [17,76].
Pro-inflammatory cytokines, hypoxia and complement activation can trigger activation of ECs, leading to activation of the coagulation cascade, which depends on interactions between ECs, platelets and coagulation factors [77,78]. Briefly, following stimulation, enhanced fibrin or fibrinogen synthesis activates plasminogen, and plasmin then cleaves the network into soluble fragments, including D-dimer [72]. Binding of D-dimer to platelet receptors then leads to the secretion of molecules stored in platelet granules, resulting in platelet activation (Fig. 1). Stored VWF and P-selectin are secreted by exocytosis of Weibel–Palade bodies from EC, and VWF tethers platelets and leucocytes to the vessel wall [76,78], leading to platelet aggregation and thrombus formation [56,70,78].
Dysfunction of ECs, together with inflammation caused by SARS-CoV-2 infection, may lead to abnormal coagulation and actively participate in thrombo-inflammatory processes that result in vasculopathy and sepsis, indicating a poor prognosis in patients with COVID-19 [31,72]. D-dimer, a fibrin degradation product that is normally not present in blood unless coagulation has occurred, serves as a biomarker for thrombosis [70] and the presence of D-dimer is associated with poor prognosis and mortality. Laboratory examinations have shown that patients with newly diagnosed SARS-CoV-2 infection have increased levels of D-dimer and that levels of D-dimer increase rapidly as the disease progresse [70,74,79]. Pathological analyses showed that coagulopathy was characterized by high levels of D-dimer, P-selectin and fibrinogen [2,70].
Patients with the severe form of COVID-19 present with a hyperinflammatory state and blood hyperviscosity [79]. Myocardial infarction (MI) [31,78] and stroke have been associated with patients who developed severe COVID-19 and presented with elevated levels of IL-6, D-dimers, troponin and N-terminal pro-brain natriuretic peptide, and erythrocytes [74,79,80], since erythrocyte aggregation represents an important cardiovascular risk factor [81].
Analyzing 68 patients with COVID-19, Goshua et al. (2020) [2] found COVID-19-associated coagulopathy and arterial thrombosis in up to 69% of critically ill patients. In their cohort study, concentrations of D-dimer and thrombin-antithrombin complex were elevated in all patients and were significantly higher in intensive care unit (ICU) patients than in non-ICU patients. VWF parameters were also increased in critically ill patients. It is noteworthy to mention that patients undergoing bed rest for long periods are at a higher risk of developing deep-vein thrombosis (DVT) even without the added pressure from COVID-19 [82,83], and DVT can also be caused by other types of viruses [84].
Varga et al. (2020) [31] demonstrated EC involvement across vascular beds of different organs in a post-mortem analysis of patients with COVID-19. Histological analysis showed an accumulation of inflammatory cells associated with the endothelium, as well as apoptotic bodies in the heart, among other organs, revealing lymphocytic endotheliitis and evidence of MI. They found viral elements within EC and an accumulation of inflammatory cells, with evidence of endothelial and inflammatory cell death. From these data, they suggest that SARS-CoV-2 infection directly facilitates the induction of endotheliitis in several organs. Klok and colleagues (2020) [85] observed 184 ICU patients over approximately seven days and found that 31% of these patients demonstrated acute pulmonary embolism, DVT, ischemic stroke, MI or systemic arterial embolism.
A case-report describes a 82-year-old patient with no previous disease reported who presented with hypercoagulable status, endothelial dysfunction, and MI within one week of COVID-19 diagnosis [86]. Stefanini et al. (2020) [81] analyzed 28 cases of COVID-19 patients who suffered MIs in Lombardy, Italy in which where 24 patients presented MI before confirmation of COVID-19 infection and four presented MI during COVID-19 infection. They concluded that MI may represent the first clinical manifestation of COVID-19. During hospitalization, 17 of these patients presented with thrombotic stenosis, being 11 of the 20 patients who had previously presented with arterial hypertension, two of the three patients who had reported previous MI, three of the four patients with percutaneous coronary intervention history and one patient who had previously undergone coronary artery bypass grafting. These authors did not report medicines administered to the patients in this study.
In turn, a retrospective study that included 78 patients with COVID-19 and MI from four hospitals in Spain, Italy, Lithuania and Iraq was performed. All patients had advanced age and high levels of comorbidities, such as stroke or previous coronary artery bypass grafting (15%), diabetes mellitus (21%), hypertension (57%), and/or coronary artery disease (61%). Upon admission, these patients received medications, such as novel oral anticoagulant (1%), warfarin (4%), clopidogrel (10%), aspirin (22%), calcium channel blocker (17%), angiotensin receptor blocker (21%), beta blocker and ACE2 inhibitor (39%) and statin (42%). Six percent of these patients died during hospitalization and 85% of the 59 patients initially treated with fibrinolytic therapy had successful fibrinolysis. It is believed that COVID-19 could trigger MI in these patients [81,86,87].
In addition, a 58 years-old patient who had previous diabetes, arterial hypertension, and obesity developed progressive respiratory failure due to COVID-19 and followed by circulatory failure and MI after 16 days of infection, leading to the patient’s death [31]. COVID-19 patients with previous cardiovascular disease are more likely to have worse prognosis, since they have observed that heart failure patients are more susceptible to heart infection by SARS-CoV-2 compared to normal heart [37], and that MI can manifest in both the early and late stages of COVID-19 [31].
Autopsies of patients who died from COVID-19 have shown brain hyperemic and edematous tissue and degenerated neurons [74]. In the first detailed report of neurological manifestations of COVID-19 in Wuhan, China, Mao et al. (2020) [74] found neurologic manifestations in 78/218 patients (36%) with SARS-CoV-2 infection treated in their hospital. These manifestations were more common in patients with severe infection than in patients with milder infection. Four of the patients had a stroke and one had a cerebral hemorrhage. These cerebrovascular manifestations may be due to impairment of the blood-brain-barrier (BBB) and brain vasculature [88]. The BBB, which protects the central nervous system (CNS) from pathogens, is composed of specialized brain microvascular endothelial cells (BMECs) joined by TJ and surrounded by astrocyte and pericytes. The stability of the BBB structure and function is of great importance for the brain homeostasis [[89], [90], [91]]. TJ prevent both free paracellular passage through the vessel wall and expression of transporters that regulate the entry and exit of substrates. A TJ also limit transcellular transport through the vessel wall and assists maintain low levels of leukocyte adhesion molecules expression, with a focus on limiting the entry of immune cells into the brain [89,[92], [93], [94]].
Hematogenous and neuronal routes have been proposed as the two main pathways by which neurotropic respiratory viruses enter the CNS [95]. To date, the specific neuroinvasive mechanism of SARS-CoV-2 is still unclear. However, it is postulated that this mechanism occurs through trans-synaptic transfer, in the olfactory nerve, with infection of the vascular endothelium or migration of immune cells through the BBB [96,97]. Recently, the expression of ACE2 was detected in the capillaries of brain tissue and primary human BMECs in in vitro cultures [98], since virus-like particles were detected in the frontal lobe endothelium [99]. Interestingly, the SARS-CoV-2 entry-associated protease cathepsin B (CTSB) but not TMPRSS2 is highly expressed in brain vascular EC [100]. When the virus invades the CNS, this invasive process can lead to the release of viral proteins that affect structure and function of EC, degrade TJ components, and lead to an increase in BBB permeability [101,102]. All of these observations suggest a potential mechanism of SARS-CoV-2 entry into EC and the brain that might underlie the neurovascular and vascular symptoms observed in some COVID-19 patients [88]. However, further studies are still needed regarding changes in the BBB resulting from SARS-CoV-2 entry and subsequent interactions with CNS.
Despite reports showing that older patients and/or patients with comorbidities are at higher risk of complications from COVID-19, it is important to highlight that patients without previous diseases may also present with the most aggressive form of the disease [103]. Deliwala et al. (2020) [25]reported a case of a 31-year-old patient with no risk factors who developed several disease and stroke. Additionally, in two weeks of this pandemic, five patients under the age of 50, diagnosed with SARS-CoV-2 infection in New York City, showed symptoms indicative of onset of ischemic stroke of large vessels. Over the last 12 months, the New York City health service received an average of 0.73 patients under the age of 50 every two weeks with stroke of the large vessels [79]. These authors showed that a diagnosis of cryptogenic stroke was twice as prevalent in COVID-19 positive patients (65.6%), as in COVID-19 negative (30.4%) patients hospitalized in the same hospital system between March 15, 2019 and April 15, 2019. Impressively, these COVID-19 positive patients were younger and had more severe conditions, attributed to the higher prevalence of occlusion of large vessels.
Based on these data, it has been suggested that patients with severe SARS-CoV-2 infection may develop a prothrombotic state, leading to thromboembolic complications, and have a greater propensity to develop cardiovascular and cerebrovascular disease [31,74,104].
4. Tumors and COVID-19 risk: Do ACE2 levels on endothelial cells matter?
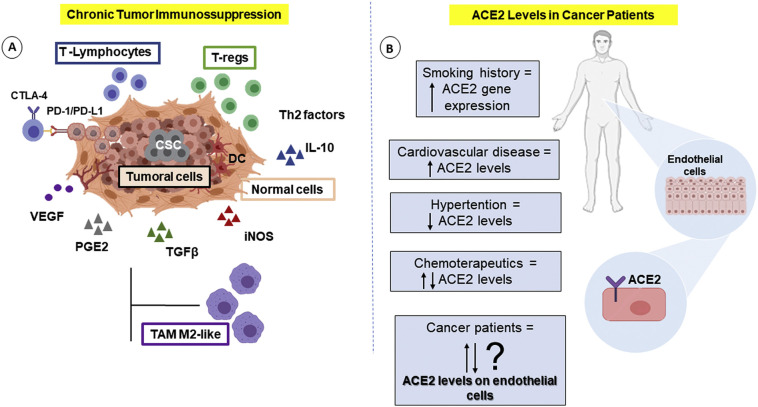
Several human disorders can be influenced by the levels of ACE2 [105]. For instance, it has been described that patients who suffer with major advanced cardiovascular events and cardiovascular disease have increased ACE2 expression. Thus, comorbid conditions that influence the levels of this protein might affect COVID-19 severity [106]. ACE2 is considered to be a regulator of tumor angiogenesis [4] and this receptor is abnormally high in lung tumors [107]. Additionally, higher levels of vascular endothelial growth factor (VEGF) have been found in COVID-19 patients compared with healthy controls [108] and EC can influence the state of inflammation and immunocompetence in cancer individuals [109,110]. Therefore, here, we hypothesize that the level of ACE2 in the vascular endothelium of cancer patients may influence the risk associated with COVID-19 (Fig. 2 ).
Fig. 2.
Cancer patient risk to SARS-CoV-2 infection could be influenced by ACE2 levels on endothelial cells? (A) Cancer therapeutics and tumor microenvironment can cause immunosuppression, vascular complications and may modulate ACE2 levels in cancer patients. Cancer cells with modified immunogenicity selected immune cells to release immunosuppressive molecules, such as TGFβ, VEGF, PGE2, IL-10, and iNOS, suppressing the proliferation and the cytotoxic response from T-lymphocytes and leading to an anti-inflammatory phenotype (Regulatory T-cells, T-regs). This immunosuppression environment can also induce the recruitment of tumor-associated macrophages with an anti-inflammatory phenotype (TAM-M2) and immature dendritic cells [159]. The chronic immunosuppression in tumor patients can facilitate the infection by SARS-CoV-2 and the COVID-19 severity. (B) Several human disorders can be influenced by levels of ACE2. However, very few are known about ACE2 levels on EC in cancer patients, and this possible increased risk of severity of COVID-19
Cancer patients infected with SARS-CoV-2 have a high chance of developing serious symptoms, a high risk of mortality and a worse prognosis [1,[111], [112], [113], [114], [115]]. Liang et al. (2020) [35] reported that cancer patients in China had a higher risk of serious events of COVID-19 than patients without cancer (39% vs. 8%, P = 0.0003). Lung tumors were the most common form (28%). In the patients with tumors and COVID-19, 25% had been subjected to surgery or chemotherapy within the past month. Compared to subjects without tumors, patients with tumors were older (63 vs. 49 years in average) and were more likely to have a history of smoking (22% vs. 7% of patients). Smoking, in addition to being an independent risk factor, increases the gene expression of ACE2 [114], suggesting greater susceptibility to infection by SARS-CoV-2.
In turn, Meng et al. (2020) [112] also demonstrated that cancer patients with COVID-19 had a higher risk of mortality (29.4% vs. 10.2%, P < 0.0001). These authors used a binary logistic regression analysis to show that cancer history is an independent risk factor for mortality in hospitalized COVID-19 patients. Furthermore, the clinical outcomes of patients with hematological malignancies were worse, with 2-fold increased mortality rate compared with patients with solid tumors (50% vs. 26.1%) [112]. According to Mehta et al. (2020) [116], the mortality rate associated with COVID-19 is higher in patients with lung, colorectal, or breast cancer. Notably, overexpression of ACE2 has been observed in lung and colon adenocarcinomas [1] and estrogen receptor-positive breast cancer [117]. Also, subjects with metastatic cancer had the greatest incidence of critical events [118,119]. However, to date, a consensus has not been found in the literature. Miyashita et al. (2020) found that patients with tumors were intubated considerably more frequently, but the death rate was not significantly different than in non-cancer patients. The authors stratified patients by age and demonstrated a significantly augmented risk of intubation in patients with tumors if they were between 66 and 80 years. Unexpectedly, patients who were younger than 50 years and had tumors had a higher mortality percentage than older ones without tumors [120]. In addition, non-metastatic cancer patients presented similar rates of critical events as patients without tumors [118,119]. Therefore, some authors state that it is not clear whether cancer is an independent risk factor, or it would mainly be driven by gender, age, obesity, uncontrolled diabetes, cardiovascular disease, and/or previous/current therapy [121,122].
Cancer patients who are receiving treatment or who have undergone surgery are more vulnerable to infection because of their immunosuppressed status and coexisting medical conditions. The immune status of cancer patients is characterized by overexpression of pro-inflammatory cytokines and a decrease in functional immunosuppressive leukocytes [114]. These characteristics lead to more complications when associated with COVID-19, since infection with this virus is considered to lead to a hyperinflammatory process. Laboratory results were significantly different in cancer and non-cancer patients indicating immune and inflammatory reactions in cancer patients, with high levels of IL-2 and IL-6 receptors, together with possible changes in the prothrombotic state, such as elevation of prothrombin time [35,112,114,123]. As mentioned earlier, all these inflammatory events can be orchestrated by EC [77,78].
It is known that several drugs, including chemotherapeutics such as cisplatin, modulate ACE2 levels in cells [124]. In addition, trials are currently evaluating the action of the chemotherapeutic anti-VEGF bevacizumab on SARS-CoV-2 patients (NCT04275414) [108]. However, many of the chemotherapeutics, such as the antiangiogenic drugs that inhibit VEGF signaling are also associated with cardiovascular toxicities, including hypertension, thrombosis, heart failure, cardiomyopathy, and arrhythmias, that increase cardiovascular risk in cancer patients [125,126]. Therefore, the application of such medications should be evaluated and considered with caution in cancer patients compromised with COVID-19.
Although it is known that several human disorders can be influenced by levels of ACE2 on EC, very few reports of ACE levels in cancer patients can be found in the literature, especially those that are related to COVID-19. It would be interesting, therefore, to further investigate the correlation between EC ACE2 levels in patients with tumors and the possible increased risk of severity of COVID-19 in this patient cohort.
5. COVID-19 treatment: EC-associated therapies
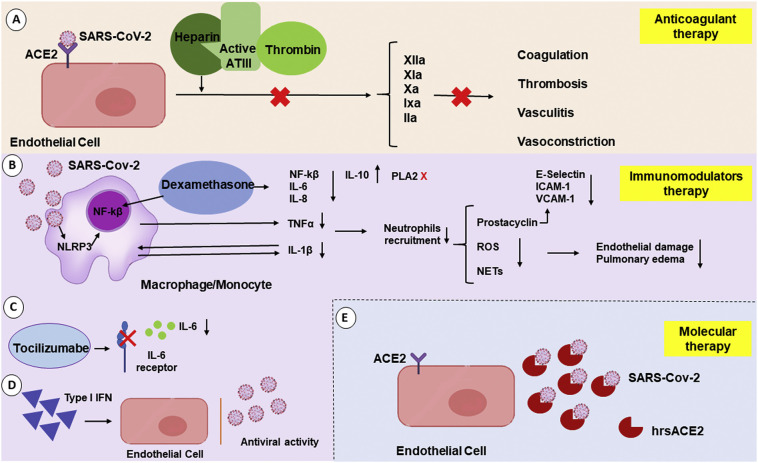
The unexpected pandemic generated by SARS-CoV-2 has created a global sense of urgency. Current efforts have focused on control and prevention of viral infection, with scientists and physicians responsible for developing protocols, treatments and vaccines. There has been a large global demand for therapies and this demand has been continually increasing in line with the growing number of cases worldwide. Since there has been insufficient time to develop new drugs, health professionals have been challenged to repurpose drugs used to treat other diseases to treat patients with COVID-19 [36]. Because of the intrinsic relationship between EC and the pathophysiology of SARS-CoV-2 and, consequently, the development of pathologies such as perivascular inflammation and disseminated intravascular coagulation, treatments with drugs targeting EC have been tested in COVID-19 patients. As examples, anticoagulants have been administered to patients infected with SARS-CoV-2 to decrease coagulopathy, and fibrinolytic drugs, immunomodulators and molecular therapies have also been investigated (Fig. 3 ) [[127], [128], [129], [130]].
Fig. 3.
Inflammatory environment triggered by SARS-CoV-2 infection and EC-associated therapeutic models. (A) The binding of heparin to antithrombin 3 (ATIII) increases ATIII activity. This leads to exposure of the ATIII catalytic site, which will interact with thrombin and inhibit its activity. This process leads to the inhibition of important factors for the coagulation process, such as IIa, IXa, Xa, XIa, XIIa. (B) Glucocorticoids such as dexamethasone have an important anti-inflammatory action by modulating inflammatory cytokines and COX-2 and PLA2 activity. Cells from the immune system increase the expression of anti-inflammatory molecules and decrease pro-inflammatory expression. Activation of the glucocorticoid receptor blocks the NFκB gene expression and impairs the expression of proinflammatory cytokines such as IL-1, IL-6, IL-8, and TNF-a. Dexamethasone can bind to Annexin I, which inhibits PLA2 activity impairing the conversion of phospholipids in arachidonic acid, which decreases the production of prostaglandins. (C) Tocilizumab is a monoclonal anti-IL-6 (IL-6) antibody which reduces the proinflammatory activity of this cytokine. (D) Type I IFNs are a large family of cytokines that mediate innate antiviral immune responses. Type I IFNs activate the transcription of several genes giving cells resistance to viral infection (antiviral status). Besides, they promote an increase in the lymphocyte population and its toxicity. (E) Soluble recombinant human ACE2 (hrsACE2) may impair the entry of SARS CoV-2 to the host cell.
5.1. Anticoagulant therapy
Heparin has been of great pharmacological importance for decades because of its role as an anticoagulant and antithrombotic [131,132]. Heparin is a member of the sulfated linear glycosaminoglycan polysaccharide family and is composed of repeating disaccharide units of uronic acid and D-glucosamine [133]. The anticoagulant action of heparin is due to its ability to bind and enhance the activity of coagulation inhibitors, such as the plasma protein antithrombin [132], and this characteristic has shown promise for the treatment of patients hospitalized with COVID-19 once disseminated intravascular coagulation is frequent in these patients [134]. Added to that, as already mentioned, long-term bed rest may increase the risk of venous thromboembolism.
In a study carried out in the city of Wuhan, administration of low molecular weight heparin for at least seven days resulted in a lower mortality rate in patients with severe SARS-CoV-2 infection type, i.e., those with elevated D-dimer resulting from fibrin degradation [135]. In addition to its anticoagulant activity, the use of heparin as a treatment has other important benefits. The polyanionic character of heparin allows it to bind to various proteins [132] and enables it to act as an effective inhibitor of viral infection by competing with the virus for binding sites on the cell surface [136]. A recent study confirmed that the S1 subunit of the SARS-CoV-2 spike protein, which contains a receptor-binding domain, interacts with heparin [137]. Heparin can also antagonize histones released by cells damaged by pathogenic infection. There is thus a reduced chance of endothelial injury when generating a “protection” to the endothelium [138,139].
However, studies are needed to determine whether this heparin-mediated endothelial protection would also be beneficial in SARS-Cov-2 infection. There are indications that anticoagulant therapy with heparin may be important to prevent the formation of microthrombin and to treat prothrombotic complications but it is unable to remove advanced clusters of fibrin deposited in the alveolar space in patients with severe infection [140]. In this case, therapies that increase plasminogen activation or downregulate fibrinolytic inhibitors can be used [141].
5.2. Immunomodulators
As discussed above, inflammation is a prominent aspect of SARS-CoV-2 infection, and EC have a unique role in modulating the inflammatory response. As well as harboring established receptors for pro-inflammatory cytokines [44] and playing a role in increased vascular permeability, EC have an intrinsic role in the inflammatory process and in activation of the coagulation cascade in COVID-19 patients. Treatments with immunomodulators, such as dexamethasone, tocilizumab and IFN, have been tested in COVID-19 patients [142,143].
Dexamethasone is an inexpensive and commonly used synthetic adrenal corticosteroid with anti-inflammatory or immunosuppressive properties, depending on whether a low or high dose is administered [144]. Dexamethasone acts as a glucocorticoid agonist and, on binding to the glucocorticoid receptor, inhibits leukocyte infiltration at the site of inflammation and suppresses humoral immune responses [145]. According to the latest results from the RECOVERY trial, in hospitalized patients with COVID-19 receiving invasive mechanical ventilation at randomization and those who were receiving oxygen without invasive mechanical ventilation, the use of dexamethasone for up to 10 days resulted in lower 28-day mortality [144]. Severely ill COVID-19 patients usually have an aberrant inflammatory response and overactivation of the immune system [44,67]. The correct dose of dexamethasone can, therefore, benefit these patients because of the anti-inflammatory and immunosuppressive effects mentioned earlier. Administration of dexamethasone to patients who were not receiving respiratory support at randomization provided no benefit, and the drug should not be used in this situation. Because of the widespread availability of dexamethasone and the positive clinical evidence, the use of this medication can be indicated in severe clinical cases that require supplemental oxygen or mechanical ventilation [144].
Tocilizumab is a monoclonal anti-IL-6 receptor antibody, which exerts an immunosuppressive effect by reducing the proinflammatory activity of IL-6 [146]. Tocilizumab is used in the treatment of rheumatoid arthritis, systemic juvenile idiopathic arthritis and polyarticular juvenile idiopathic arthritis [147]. Tocilizumab was considered as a possibility for the treatment of SARS-CoV-2 infection because it may soothe the so-called cytokine storm, which, for example, increases endothelial permeability. Scientific studies on the use of tocilizumab are, however, controversial.
In a study carried out in China, a single dose of tocilizumab, administered together with the corticosteroid methylprednisolone, was found to reduce serum levels of IL-6 and C-reactive protein [148]. Although the group claimed that the treatment provided therapeutic benefit, it was suggested that repeated doses of tocilizumab may be more effective in improving the condition of critically ill patients or patients with high levels of IL-6. In another study, administration of tocilizumab provided an improvement in patients' recovery times and a decrease in levels of C-reactive protein. In this study, however, the patients had previously also received hydroxychloroquine and azithromycin, so that the concomitant use of these drugs may have influenced the results [149].
Type I IFN is a protein produced and secreted by the immune system after a viral infection, and an impaired type I IFN response has been identified in patients with severe SARS-CoV-2 infection [150]. An excessive inflammatory response and high viral load in the blood have been also observed and may be associated with increased production of cytokines [150,151]. Drugs that increase the amount of IFN have, therefore, been tested as a treatment for COVID-19. It is, however, unclear whether the virus blocks the action of IFNs, or whether people with a deficiency of this protein develop the disease more severely. In a study of 446 patients in China, early administration of IFN reduced mortality, whereas late administration of IFN was associated with increased mortality and delayed recovery [152]. Another group investigated the consequences of treatment with nebulized IFN, alone and in combination with the antiviral agent arbidol, in 77 patients. A decrease in the inflammatory biomarker IL-6 and greater viral clearance from the respiratory tract were observed in both cases [153]. There is, therefore, some evidence that the use of IFN as a therapy produces favorable outcomes, but there is still insufficient knowledge to prove its efficacy. It also appears to be important to confirm the ideal stage of the disease at which to administer type I IFN.
5.3. Molecular therapy
As discussed above, the SARS-CoV-2 spike protein binds to ACE2, thereby increasing cell membrane fusion and endocytosis. After activation of the spike protein by a cellular protease (TMPRSS2), ACE2 promotes entry of the virus into the cell [153,154]. Treatments targeting ACE2 have been tested, mainly because this receptor is present in epithelial cells of the respiratory tract, such as type 2 pneumocytes, that are susceptible to SARS-CoV-2 infection [155,156]. One option is the use of recombinant soluble human ACE2 (hrsACE2), which, if present in excess, would intercept the virus-receptor binding and decrease the rate of viral infection [130]. Administration of hrsACE2 has been observed to reduce the infection of capillary and renal organoids by SARS-CoV-2 in vitro. As well as acting on the alveolar tract, hrsACE2 may also act on EC and thus prevent viral spread through the bloodstream [157,158]. This molecular therapy thus has potential in the treatment of SARS-CoV-2 infection.
6. Conclusions
In summary, ACE2 mediates SARS-CoV-2 entry into the host cell and it is a predominant receptor on the EC membrane. In turn, EC has a critical role in the inflammatory and disseminate coagulation process reported in COVID-19 patients, which culminate in a higher risk of thromboembolic, cardiovascular, and cerebrovascular complications in these patients. Several human disorders can be influenced by levels of ACE2. However, ACE2 levels on EC in cancer patients, who have a high chance of developing serious symptoms have not been well explored. Finally, EC-related therapies such as anticoagulants and corticosteroids have been used to treat severe COVID-19 patients and other immunomodulators and molecular therapies have also been studied to this purpose.
Funding
This study was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Associação Mahatma Gandhi.
Author contributions
Designed the manuscript: VF. Wrote the manuscript: LB, TL, LP, LR and VF. Designed the figures: TL and LB. Supervised the involved coworkers and edited the manuscript: VF.
Declaration of Competing Interest
None.
References
- 1.Chai P., Yu J., Ge S., Jia R., Fan X. Genetic alteration, RNA expression, and DNA methylation profiling of coronavirus disease 2019 (COVID-19) receptor ACE2 in malignancies: a pan-cancer analysis. J. Hematol. Oncol.J Hematol Oncol. 2020;13:43. doi: 10.1186/s13045-020-00883-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goshua G., Pine A.B., Meizlish M.L., Chang C.-H., Zhang H., Bahel P., Baluha A., Bar N., Bona R.D., Burns A.J., Dela Cruz C.S., Dumont A., Halene S., Hwa J., Koff J., Menninger H., Neparidze N., Price C., Siner J.M., Tormey C., Rinder H.M., Chun H.J., Lee A.I. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020;7:e575–e582. doi: 10.1016/S2352-3026(20)30216-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yuki K., Fujiogi M., Koutsogiannaki S. COVID-19 pathophysiology: A review. Clin. Immunol. 2020;215:108427. doi: 10.1016/j.clim.2020.108427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang Q., Lu S., Li T., Yu L., Zhang Y., Zeng H., Qian X., Bi J., Lin Y. ACE2 inhibits breast cancer angiogenesis via suppressing the VEGFa/VEGFR2/ERK pathway. J. Exp. Clin. Cancer Res. 2019;38:173. doi: 10.1186/s13046-019-1156-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matsuyama S., Nagata N., Shirato K., Kawase M., Takeda M., Taguchi F. Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2. J. Virol. 2010;84:12658–12664. doi: 10.1128/JVI.01542-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Glowacka I., Bertram S., Muller M.A., Allen P., Soilleux E., Pfefferle S., Steffen I., Tsegaye T.S., He Y., Gnirss K., Niemeyer D., Schneider H., Drosten C., Pohlmann S. Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response. J. Virol. 2011;85:4122–4134. doi: 10.1128/JVI.02232-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Iwata-Yoshikawa N., Okamura T., Shimizu Y., Hasegawa H., Takeda M., Nagata N. TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection. J. Virol. 2019;93 doi: 10.1128/JVI.01815-18. e01815-18, /jvi/93/6/JVI.01815-18.atom. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Matsuyama S., Nao N., Shirato K., Kawase M., Saito S., Takayama I., Nagata N., Sekizuka T., Katoh H., Kato F., Sakata M., Tahara M., Kutsuna S., Ohmagari N., Kuroda M., Suzuki T., Kageyama T., Takeda M. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. 2020;117:7001–7003. doi: 10.1073/pnas.2002589117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Walls A.C., Park Y.-J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281–292. doi: 10.1016/j.cell.2020.02.058. e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Milewska A., Zarebski M., Nowak P., Stozek K., Potempa J., Pyrc K. Human coronavirus NL63 utilizes heparan sulfate proteoglycans for attachment to target cells. J. Virol. 2014;88:13221–13230. doi: 10.1128/JVI.02078-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tortorici M.A., Walls A.C., Lang Y., Wang C., Li Z., Koerhuis D., Boons G.-J., Bosch B.-J., Rey F.A., de Groot R.J., Veesler D. Structural basis for human coronavirus attachment to sialic acid receptors. Nat. Struct. Mol. Biol. 2019;26:481–489. doi: 10.1038/s41594-019-0233-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hulswit R.J.G., Lang Y., Bakkers M.J.G., Li W., Li Z., Schouten A., Ophorst B., van Kuppeveld F.J.M., Boons G.-J., Bosch B.-J., Huizinga E.G., de Groot R.J. Human coronaviruses OC43 and HKU1 bind to 9- O -acetylated sialic acids via a conserved receptor-binding site in spike protein domain A. Proc. Natl. Acad. Sci. 2019;116:2681–2690. doi: 10.1073/pnas.1809667116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen Z., Mi L., Xu J., Yu J., Wang X., Jiang J., Xing J., Shang P., Qian A., Li Y., Shaw P.X., Wang J., Duan S., Ding J., Fan C., Zhang Y., Yang Y., Yu X., Feng Q., Li B., Yao X., Zhang Z., Li L., Xue X., Zhu P. Function of HAb18G/CD147 in invasion of host cells by severe acute respiratory syndrome coronavirus. J. Infect. Dis. 2005;191:755–760. doi: 10.1086/427811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sardu C., Gambardella J., Morelli M.B., Wang X., Marfella R., Santulli G. Is COVID-19 an endothelial disease? Clinical and basic evidence. Med. Pharmacol. 2020 doi: 10.20944/preprints202004.0204.v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huertas A., Montani D., Savale L., Pichon J., Tu L., Parent F., Guignabert C., Humbert M. Endothelial cell dysfunction: a major player in SARS-CoV-2 infection (COVID-19)? Eur. Respir. J. 2020;56:2001634. doi: 10.1183/13993003.01634-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gosain R., Abdou Y., Singh A., Rana N., Puzanov I., Ernstoff M.S. COVID-19 and cancer: a comprehensive review. Curr. Oncol. Rep. 2020;22:53. doi: 10.1007/s11912-020-00934-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ziegler C.G.K., Allon S.J., Nyquist S.K., Mbano I.M., Miao V.N., Tzouanas C.N., Cao Y., Yousif A.S., Bals J., Hauser B.M., Feldman J., Muus C., Wadsworth M.H., Kazer S.W., Hughes T.K., Doran B., Gatter G.J., Vukovic M., Taliaferro F., Mead B.E., Guo Z., Wang J.P., Gras D., Plaisant M., Ansari M., Angelidis I., Adler H., Sucre J.M.S., Taylor C.J., Lin B., Waghray A., Mitsialis V., Dwyer D.F., Buchheit K.M., Boyce J.A., Barrett N.A., Laidlaw T.M., Carroll S.L., Colonna L., Tkachev V., Peterson C.W., Yu A., Zheng H.B., Gideon H.P., Winchell C.G., Lin P.L., Bingle C.D., Snapper S.B., Kropski J.A., Theis F.J., Schiller H.B., Zaragosi L.-E., Barbry P., Leslie A., Kiem H.-P., Flynn J.L., Fortune S.M., Berger B., Finberg R.W., Kean L.S., Garber M., Schmidt A.G., Lingwood D., Shalek A.K., Ordovas-Montanes J., Banovich N., Barbry P., Brazma A., Desai T., Duong T.E., Eickelberg O., Falk C., Farzan M., Glass I., Haniffa M., Horvath P., Hung D., Kaminski N., Krasnow M., Kropski J.A., Kuhnemund M., Lafyatis R., Lee H., Leroy S., Linnarson S., Lundeberg J., Meyer K., Misharin A., Nawijn M., Nikolic M.Z., Ordovas-Montanes J., Pe’er D., Powell J., Quake S., Rajagopal J., Tata P.R., Rawlins E.L., Regev A., Reyfman P.A., Rojas M., Rosen O., Saeb-Parsy K., Samakovlis C., Schiller H., Schultze J.L., Seibold M.A., Shalek A.K., Shepherd D., Spence J., Spira A., Sun X., Teichmann S., Theis F., Tsankov A., van den Berge M., von Papen M., Whitsett J., Xavier R., Xu Y., Zaragosi L.-E., Zhang K. SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell. 2020;181:1016–1035. doi: 10.1016/j.cell.2020.04.035. e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.-L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu H., Zhong L., Deng J., Peng J., Dan H., Zeng X., Li T., Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020;12:8. doi: 10.1038/s41368-020-0074-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jia H.P., Look D.C., Shi L., Hickey M., Pewe L., Netland J., Farzan M., Wohlford-Lenane C., Perlman S., McCray P.B. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J. Virol. 2005;79:14614–14621. doi: 10.1128/JVI.79.23.14614-14621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Perico L., Benigni A., Remuzzi G. Should COVID-19 concern nephrologists? Why and to what extent? The emerging impasse of angiotensin blockade. Nephron. 2020;144:213–221. doi: 10.1159/000507305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gheblawi M., Wang K., Viveiros A., Nguyen Q., Zhong J.-C., Turner A.J., Raizada M.K., Grant M.B., Oudit G.Y. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020;126:1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Deliwala S., Abdulhamid S., Abusalih M.F., Al-Qasmi M.M., Bachuwa G. 2020. Encephalopathy as the Sentinel Sign of a Cortical Stroke in a Patient Infected With Coronavirus Disease-19 (COVID-19), Cureus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mycroft-West C., Su D., Elli S., Li Y., Guimond S., Miller G., Turnbull J., Yates E., Guerrini M., Fernig D., Lima M., Skidmore M. 2020. The 2019 coronavirus (SARS-CoV-2) surface protein (Spike) S1 Receptor Binding Domain undergoes conformational change upon heparin binding; p. 9. [Google Scholar]
- 27.Mycroft-West C.J., Su D., Li Y., Guimond S.E., Rudd T.R., Miller G., Nunes Q.M., Procter P., Bisio A., Forsyth N.R., Turnbull J.E., Guerrini M., Fernig D.G., Yates E.A., Lima M.A., Skidmore M.A. 2020. SARS-CoV-2 spike S1 receptor binding domain undergoes conformational change upon interaction with low molecular weight heparins; p. 15. [Google Scholar]
- 28.Guimond S.E., Mycroft-West C.J., Gandhi N.S., Tree J.A., Buttigieg K.R., Coombes N., Elmore M.J., Nyström K., Said J., Setoh Y.X., Amarilla A.A., Modhiran N., Sng J.D.J., Chhabra M., Watterson D., Young P.R., Khromykh A.A., Lima M.A., Yates E.A., Karlsson R., Chen Y.-H., Zhang Y., Hammond E., Dredge K., Carroll M.W., Trybala E., Bergström T., Ferro V., Skidmore M.A., Turnbull J.E. Synthetic heparan sulfate mimetic pixatimod (PG545) potently inhibits SARS-CoV-2 by disrupting the spike-ACE2 interaction. Microbiology. 2020 doi: 10.1101/2020.06.24.169334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tavassoly O., Safavi F., Tavassoly I. Heparin-binding peptides as novel therapies to stop SARS-CoV-2 cellular entry and infection. Mol. Pharmacol. 2020;98:612–619. doi: 10.1124/molpharm.120.000098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tiwari V., Tandon R., Sankaranarayanan N.V., Beer J.C., Kohlmeir E.K., Swanson-Mungerson M., Desai U.R. Preferential recognition and antagonism of SARS-CoV-2 spike glycoprotein binding to 3- O -sulfated heparan sulfate. Biochemistry. 2020 doi: 10.1101/2020.10.08.331751. [DOI] [Google Scholar]
- 31.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., Mehra M.R., Schuepbach R.A., Ruschitzka F., Moch H. Endothelial cell infection and endotheliitis in COVID-19. The Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hamming I., Timens W., Bulthuis M., Lely A., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.DosSantos M.F., Devalle S., Aran V., Capra D., Roque N.R., Coelho-Aguiar J. de M., e Spohr T.C.L. de S., Subilhaga J.G., Pereira C.M., D’Andrea Meira I., Filho P. Niemeyer Soares, Moura-Neto V. Neuromechanisms of SARS-CoV-2: A review. Front. Neuroanat. 2020;14:37. doi: 10.3389/fnana.2020.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lamers M.M., Beumer J., van der Vaart J., Knoops K., Puschhof J., Breugem T.I., Ravelli R.B.G., Paul van Schayck J., Mykytyn A.Z., Duimel H.Q., van Donselaar E., Riesebosch S., Kuijpers H.J.H., Schipper D., van de Wetering W.J., de Graaf M., Koopmans M., Cuppen E., Peters P.J., Haagmans B.L., Clevers H. SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369:50–54. doi: 10.1126/science.abc1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liang W., Guan W., Chen R., Wang W., Li J., Xu K., Li C., Ai Q., Lu W., Liang H., Li S., He J. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335–337. doi: 10.1016/S1470-2045(20)30096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou P., Yang X.-L., Wang X.-G., Hu B., Zhang L., Zhang W., Si H.-R., Zhu Y., Li B., Huang C.-L., Chen H.-D., Chen J., Luo Y., Guo H., Jiang R.-D., Liu M.-Q., Chen Y., Shen X.-R., Wang X., Zheng X.-S., Zhao K., Chen Q.-J., Deng F., Liu L.-L., Yan B., Zhan F.-X., Wang Y.-Y., Xiao G.-F., Shi Z.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen R., Wang K., Yu J., Howard D., French L., Chen Z., Wen C., Xu Z. The spatial and cell-type distribution of SARS-CoV-2 receptor ACE2 in human and mouse brain. Neuroscience. 2020 doi: 10.1101/2020.04.07.030650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xia H., Sriramula S., Chhabra K.H., Lazartigues E. Brain angiotensin-converting enzyme type 2 shedding contributes to the development of neurogenic hypertension. Circ. Res. 2013;113:1087–1096. doi: 10.1161/CIRCRESAHA.113.301811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ding Y., He L., Zhang Q., Huang Z., Che X., Hou J., Wang H., Shen H., Qiu L., Li Z., Geng J., Cai J., Han H., Li X., Kang W., Weng D., Liang P., Jiang S. Organ distribution of severe acute respiratory syndrome(SARS) associated coronavirus(SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J. Pathol. 2004;203:622–630. doi: 10.1002/path.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z., Zhuang H., Wu B., Zhong H., Shao H., Fang W., Gao D., Pei F., Li X., He Z., Xu D., Shi X., Anderson V.M., Leong A.S.-Y. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li K., Wohlford-Lenane C., Perlman S., Zhao J., Jewell A.K., Reznikov L.R., Gibson-Corley K.N., Meyerholz D.K., McCray P.B. Middle east respiratory syndrome coronavirus causes multiple organ damage and lethal disease in mice transgenic for human dipeptidyl peptidase 4. J. Infect. Dis. 2016;213:712–722. doi: 10.1093/infdis/jiv499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang M., Hao H., Leeper N.J., Zhu L. Thrombotic regulation from the endothelial cell perspectives. Arterioscler. Thromb. Vasc. Biol. 2018;38 doi: 10.1161/ATVBAHA.118.310367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F., Vanstapel A., Werlein C., Stark H., Tzankov A., Li W.W., Li V.W., Mentzer S.J., Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N. Engl. J. Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pober J.S., Sessa W.C. Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol. 2007;7:803–815. doi: 10.1038/nri2171. [DOI] [PubMed] [Google Scholar]
- 45.Pelaia C., Tinello C., Vatrella A., De Sarro G., Pelaia G. Lung under attack by COVID-19-induced cytokine storm: pathogenic mechanisms and therapeutic implications. Ther. Adv. Respir. Dis. 2020;14 doi: 10.1177/1753466620933508. 175346662093350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Felsenstein S., Herbert J.A., McNamara P.S., Hedrich C.M. COVID-19: immunology and treatment options. Clin. Immunol. 2020;215:108448. doi: 10.1016/j.clim.2020.108448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Florindo H.F., Kleiner R., Vaskovich-Koubi D., Acúrcio R.C., Carreira B., Yeini E., Tiram G., Liubomirski Y., Satchi-Fainaro R. Immune-mediated approaches against COVID-19. Nat. Nanotechnol. 2020;15:630–645. doi: 10.1038/s41565-020-0732-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.N. Vabret, G.J. Britton, C. Gruber, S. Hegde, J. Kim, M. Kuksin, R. Levantovsky, L. Malle, A. Moreira, M.D. Park, L. Pia, E. Risson, M. Saffern, B. Salomé, M. Esai Selvan, M.P. Spindler, J. Tan, V. van der Heide, J.K. Gregory, K. Alexandropoulos, N. Bhardwaj, B.D. Brown, B. Greenbaum, Z.H. Gümüş, D. Homann, A. Horowitz, A.O. Kamphorst, M.A. Curotto de Lafaille, S. Mehandru, M. Merad, R.M. Samstein, M. Agrawal, M. Aleynick, M. Belabed, M. Brown, M. Casanova-Acebes, J. Catalan, M. Centa, A. Charap, A. Chan, S.T. Chen, J. Chung, C.C. Bozkus, E. Cody, F. Cossarini, E. Dalla, N. Fernandez, J. Grout, D.F. Ruan, P. Hamon, E. Humblin, D. Jha, J. Kodysh, A. Leader, M. Lin, K. Lindblad, D. Lozano-Ojalvo, G. Lubitz, A. Magen, Z. Mahmood, G. Martinez-Delgado, J. Mateus-Tique, E. Meritt, C. Moon, J. Noel, T. O’Donnell, M. Ota, T. Plitt, V. Pothula, J. Redes, I. Reyes Torres, M. Roberto, A.R. Sanchez-Paulete, J. Shang, A.S. Schanoski, M. Suprun, M. Tran, N. Vaninov, C.M. Wilk, J. Aguirre-Ghiso, D. Bogunovic, J. Cho, J. Faith, E. Grasset, P. Heeger, E. Kenigsberg, F. Krammer, U. Laserson, Immunology of COVID-19: current state of the science, Immunity. 52 (2020) 910–941. 10.1016/j.immuni.2020.05.002. [DOI] [PMC free article] [PubMed]
- 49.De Biasi S., Meschiari M., Gibellini L., Bellinazzi C., Borella R., Fidanza L., Gozzi L., Iannone A., Tartaro D. Lo, Mattioli M., Paolini A., Menozzi M., Milić J., Franceschi G., Fantini R., Tonelli R., Sita M., Sarti M., Trenti T., Brugioni L., Cicchetti L., Facchinetti F., Pietrangelo A., Clini E., Girardis M., Guaraldi G., Mussini C., Cossarizza A. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat. Commun. 2020;11:3434. doi: 10.1038/s41467-020-17292-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gao T., Hu M., Zhang X., Li H., Zhu L., Liu H., Dong Q., Zhang Z., Wang Z., Hu Y., Fu Y., Jin Y., Li K., Zhao S., Xiao Y., Luo S., Li L., Zhao L., Liu J., Zhao H., Liu Y., Yang W., Peng J., Chen X., Li P., Liu Y., Xie Y., Song J., Zhang L., Ma Q., Bian X., Chen W., Liu X., Mao Q., Cao C. Highly pathogenic coronavirus N protein aggravates lung injury by MASP-2-mediated complement over-activation. Infect. Dis. (except HIV/AIDS) 2020 doi: 10.1101/2020.03.29.20041962. [DOI] [Google Scholar]
- 51.Blume C., Reale R., Held M., Loxham M., Millar T.M., Collins J.E., Swindle E.J., Morgan H., Davies D.E. 2017. Cellular crosstalk between airway epithelial and endothelial cells regulates barrier functions during exposure to double-stranded RNA; p. 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chowdhury F., Howat W.J., Phillips G.J., Lackie P.M. Interactions between endothelial cells and epithelial cells in a combined cell model of airway mucosa: effects on tight junction permeability. Exp. Lung Res. 2010;36:1–11. doi: 10.3109/01902140903026582. [DOI] [PubMed] [Google Scholar]
- 53.García-Ponce A., Chánez Paredes S., Castro Ochoa K.F., Schnoor M. Regulation of endothelial and epithelial barrier functions by peptide hormones of the adrenomedullin family. Tissue Barriers. 2016;4 doi: 10.1080/21688370.2016.1228439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Aird W.C. Phenotypic heterogeneity of the endothelium: II. Representative vascular beds. Circ. Res. 2007;100:174–190. doi: 10.1161/01.RES.0000255690.03436.ae. [DOI] [PubMed] [Google Scholar]
- 55.Millar F.R., Summers C., Grif M.J., Proudfoot A.G. The pulmonary endothelium in acute respiratory distress syndrome: insights and therapeutic opportunities. Thorax. 2016;71:462–473. doi: 10.1136/thoraxjnl-2015-207461. [DOI] [PubMed] [Google Scholar]
- 56.Noris M., Benigni A., Remuzzi G. The case of complement activation in COVID-19 multiorgan impact. Kidney Int. 2020;98:314–322. doi: 10.1016/j.kint.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Freeman T.L., Swartz T.H. Targeting the NLRP3 inflammasome in severe COVID-19. Front. Immunol. 2020;11:1518. doi: 10.3389/fimmu.2020.01518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Meduri G.U., Headley S., Kohler G., Stentz F., Tolley E., Umberger R., Leeper K. Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS. Chest. 1995;107:1062–1073. doi: 10.1378/chest.107.4.1062. [DOI] [PubMed] [Google Scholar]
- 59.Yaqinuddin A., Kashir J. Novel therapeutic targets for SARS-CoV-2-induced acute lung injury: targeting a potential IL-1β/neutrophil extracellular traps feedback loop. Med. Hypotheses. 2020;143:109906. doi: 10.1016/j.mehy.2020.109906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Middleton E.A., He X.-Y., Denorme F., Campbell R.A., Ng D., Salvatore S.P., Mostyka M., Baxter-Stoltzfus A., Borczuk A.C., Loda M., Cody M.J., Manne B.K., Portier I., Harris E.S., Petrey A.C., Beswick E.J., Caulin A.F., Iovino A., Abegglen L.M., Weyrich A.S., Rondina M.T., Egeblad M., Schiffman J.D., Yost C.C. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–1179. doi: 10.1182/blood.2020007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Laforge M., Elbim C., Frère C., Hémadi M., Massaad C., Nuss P., Benoliel J.-J., Becker C. Tissue damage from neutrophil-induced oxidative stress in COVID-19. Nat. Rev. Immunol. 2020;20:515–516. doi: 10.1038/s41577-020-0407-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Panfoli I. Potential role of endothelial cell surface ectopic redox complexes in COVID-19 disease pathogenesis. Clin. Med. 2020;20:e146–e147. doi: 10.7861/clinmed.2020-0252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Xavier A.R., Silva J.S., Almeida J.P.C.L., Conceição J.F.F., Lacerda G.S., Kanaan S. COVID-19: clinical and laboratory manifestations in novel coronavirus infection. J. Bras. Patol. E Med. Lab. 2020 doi: 10.5935/1676-2444.20200049. [DOI] [Google Scholar]
- 64.Ratajczak M.Z., Kucia M. SARS-CoV-2 infection and overactivation of Nlrp3 inflammasome as a trigger of cytokine “storm” and risk factor for damage of hematopoietic stem cells. Leukemia. 2020;34:1726–1729. doi: 10.1038/s41375-020-0887-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ratajczak M.Z., Bujko K., Ciechanowicz A., Sielatycka K., Cymer M., Marlicz W., Kucia M. SARS-CoV-2 entry receptor ACE2 is expressed on very small CD45− precursors of hematopoietic and endothelial cells and in response to virus spike protein activates the Nlrp3 inflammasome. Stem Cell Rev. Rep. 2020 doi: 10.1007/s12015-020-10010-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Fletcher-Sandersjöö A., Bellander B.-M. Is COVID-19 associated thrombosis caused by overactivation of the complement cascade? A literature review. Thromb. Res. 2020;194:36–41. doi: 10.1016/j.thromres.2020.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chauhan A.J., Wiffen L.J., Brown T.P. COVID-19: a collision of complement, coagulation and inflammatory pathways. J. Thromb. Haemost. 2020;18:2110–2117. doi: 10.1111/jth.14981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Marchetti M. COVID-19-driven endothelial damage: complement, HIF-1, and ABL2 are potential pathways of damage and targets for cure. Ann. Hematol. 2020;99:1701–1707. doi: 10.1007/s00277-020-04138-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.McKechnie J.L., Blish C.A. The innate immune system: fighting on the front lines or fanning the flames of COVID-19? Cell Host Microbe. 2020;27:863–869. doi: 10.1016/j.chom.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Grobler C., Maphumulo S.C., Grobbelaar L.M., Bredenkamp J.C., Laubscher G.J., Lourens P.J., Steenkamp J., Kell D.B., Pretorius E. Covid-19: The rollercoaster of fibrin(ogen), D-dimer, Von Willebrand factor, P-selectin and their interactions with endothelial cells, platelets and erythrocytes. Int. J. Mol. Sci. 2020;21:5168. doi: 10.3390/ijms21145168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kaur S., Tripathi D.M., Yadav A. The enigma of endothelium in COVID-19. Front. Physiol. 2020;11:989. doi: 10.3389/fphys.2020.00989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pons S., Fodil S., Azoulay E., Zafrani L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit. Care. 2020;24:353. doi: 10.1186/s13054-020-03062-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kai H., Kai M. Interactions of coronaviruses with ACE2, angiotensin II, and RAS inhibitors—lessons from available evidence and insights into COVID-19. Hypertens. Res. 2020;43:648–654. doi: 10.1038/s41440-020-0455-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with Coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wang B., Li R., Lu Z., Huang Y. Does comorbidity increase the risk of patients with COVID-19: evidence from meta-analysis. Aging. 2020;12:6049–6057. doi: 10.18632/aging.103000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Teuwen L.-A., Geldhof V., Pasut A., Carmeliet P. COVID-19: the vasculature unleashed. Nat. Rev. Immunol. 2020;20:389–391. doi: 10.1038/s41577-020-0343-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang J., Saguner A.M., An J., Ning Y., Yan Y., Li G. Dysfunctional coagulation in COVID-19: from cell to bedside. Adv. Ther. 2020;37:3033–3039. doi: 10.1007/s12325-020-01399-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.O’Sullivan J.M., Gonagle D.M., Ward S.E., Preston R.J.S., O’Donnell J.S. Endothelial cells orchestrate COVID-19 coagulopathy. Lancet Haematol. 2020;7:e553–e555. doi: 10.1016/S2352-3026(20)30215-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yaghi S., Ishida K., Torres J., Mac Grory B., Raz E., Humbert K., Henninger N., Trivedi T., Lillemoe K., Alam S., Sanger M., Kim S., Scher E., Dehkharghani S., Wachs M., Tanweer O., Volpicelli F., Bosworth B., Lord A., Frontera J. SARS-CoV-2 and stroke in a New York healthcare system. Stroke. 2020;51:2002–2011. doi: 10.1161/STROKEAHA.120.030335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Qin C., Zhou L., Hu Z., Yang S., Zhang S., Chen M., Yu H., Tian D.-S., Wang W. Clinical characteristics and outcomes of COVID-19 patients with a history of stroke in Wuhan, China. Stroke. 2020;51:2219–2223. doi: 10.1161/STROKEAHA.120.030365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Stefanini G.G., Montorfano M., Trabattoni D., Andreini D., Ferrante G., Ancona M., Metra M., Curello S., Maffeo D., Pero G., Cacucci M., Assanelli E., Bellini B., Russo F., Ielasi A., Tespili M., Danzi G.B., Vandoni P., Bollati M., Barbieri L., Oreglia J., Lettieri C., Cremonesi A., Carugo S., Reimers B., Condorelli G., Chieffo A. ST-elevation myocardial infarction in patients with COVID-19: clinical and angiographic outcomes. Circulation. 2020;141:2113–2116. doi: 10.1161/CIRCULATIONAHA.120.047525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gong J.-M., Du J.-S., Han D.-M. Implications of bed rest for patients with acute deep vein thrombosis: a qualitative study. Patient Prefer. Adherence. 2020;14:1659–1667. doi: 10.2147/PPA.S271481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Partsch H. Bed rest versus ambulation in the initial treatment of patients with proximal deep vein thrombosis. Curr. Opin. Pulm. Med. 2002;8:389–393. doi: 10.1097/00063198-200209000-00008. [DOI] [PubMed] [Google Scholar]
- 84.Marques M.A., Adami de Sá F.P., Lupi O., Brasil P., von Ristow A. Trombose venosa profunda e vírus chicungunha. J. Vasc. Bras. 2017;16:60–62. doi: 10.1590/1677-5449.009616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Klok F.A., Kruip M.J.H.A., van der Meer N.J.M., Arbous M.S., Gommers D.A.M.P.J., Kant K.M., Kaptein F.H.J., van Paassen J., Stals M.A.M., Huisman M.V., Endeman H. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020;191:145–147. doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ueki Y., Otsuka T., Windecker S., Räber L. ST-elevation myocardial infarction and pulmonary embolism in a patient with COVID-19 acute respiratory distress syndrome. Eur. Heart J. 2020;41:2134. doi: 10.1093/eurheartj/ehaa399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hamadeh A., Aldujeli A., Briedis K., Tecson K.M., Sanz-Sánchez J., Dujeili M. al, Al-Obeidi A., Diez J.L., Žaliūnas R., Stoler R.C., McCullough P.A. Characteristics and outcomes in patients presenting with COVID-19 and ST-segment elevation myocardial infarction. Am. J. Cardiol. 2020;131:1–6. doi: 10.1016/j.amjcard.2020.06.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ghobrial M., Charish J., Takada S., Valiante T., Radovanovic I., Bader G.D., Wälchli T. The human brain vasculature shows a distinct expression pattern of SARS- CoV-2 entry factors. BioRxiv. 2020 doi: 10.1101/2020.10.10.334664. 2020.10.10.334664. [DOI] [Google Scholar]
- 89.Chen Z., Li G. Immune response and blood–brain barrier dysfunction during viral neuroinvasion. Innate Immun. 2020 doi: 10.1177/1753425920954281. 175342592095428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.L.V. De Oliveira Rosario, B.G. Da Rosa, T.L. Goncalves, D.I.L. Matias, C. Freitas, V.P. Ferrer, Glioblastoma factors increase the migration of human brain endothelial cells in vitro by increasing MMP-9/CXCR4 levels, Anticancer Res. 40 (2020) 2725–2737. 10.21873/anticanres.14244. [DOI] [PubMed]
- 91.L. Pellegrini, A. Albecka, D.L. Mallery, M.J. Kellner, D. Paul, A.P. Carter, L.C. James, M.A. Lancaster, SARS-CoV-2 infects brain choroid plexus and disrupts the blood-CSF-barrier, 2020.08.20.259937. 10.1101/2020.08.20.259937. [DOI] [PMC free article] [PubMed]
- 92.Alquisiras-Burgos I., Peralta-Arrieta I., Alonso-Palomares L.A., Zacapala-Gómez A.E., Salmerón-Bárcenas E.G., Aguilera P. Neurological complications associated with the blood-brain barrier damage induced by the inflammatory response during SARS-CoV-2 infection. Mol. Neurobiol. 2020 doi: 10.1007/s12035-020-02134-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Siegenthaler J.A., Sohet F., Daneman R. ‘Sealing off the CNS’: cellular and molecular regulation of blood–brain barriergenesis. Curr. Opin. Neurobiol. 2013;23:1057–1064. doi: 10.1016/j.conb.2013.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Langen U.H., Ayloo S., Gu C. Development and cell biology of the blood-brain barrier. Annu. Rev. Cell Dev. Biol. 2019;35:591–613. doi: 10.1146/annurev-cellbio-100617-062608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Desforges M., Le Coupanec A., Dubeau P., Bourgouin A., Lajoie L., Dubé M., Talbot P.J. Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses. 2019;12:14. doi: 10.3390/v12010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Alquisiras-Burgos I., Peralta-Arrieta I., Alonso-Palomares L.A., Zacapala-Gómez A.E., Salmerón-Bárcenas E.G., Aguilera P. Neurological complications associated with the blood-brain barrier damage induced by the inflammatory response during SARS-CoV-2 infection. Mol. Neurobiol. 2020 doi: 10.1007/s12035-020-02134-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Zubair A.S., McAlpine L.S., Gardin T., Farhadian S., Kuruvilla D.E., Spudich S. Neuropathogenesis and neurologic manifestations of the coronaviruses in the age of coronavirus disease 2019: a review. JAMA Neurol. 2020;77:1018. doi: 10.1001/jamaneurol.2020.2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Buzhdygan T.P., DeOre B.J., Baldwin-Leclair A., McGary H., Razmpour R., Galie P.A., Potula R., Andrews A.M., Ramirez S.H. The SARS-CoV-2 spike protein alters barrier function in 2D static and 3D microfluidic in vitro models of the human blood–brain barrier. BioRxiv. 2020 doi: 10.1101/2020.06.15.150912. 2020.06.15.150912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Paniz-Mondolfi A., Bryce C., Grimes Z., Gordon R.E., Reidy J., Lednicky J., Sordillo E.M., Fowkes M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) J. Med. Virol. 2020;92:699–702. doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ghobrial M., Charish J., Takada S., Valiante T., Radovanovic I., Bader G.D., Wälchli T. 2020. The human brain vasculature shows a distinct expression pattern of SARS-CoV-2 entry factors. 10.10.334664. [DOI] [Google Scholar]
- 101.Chen Z., Li G. Immune response and blood–brain barrier dysfunction during viral neuroinvasion. Innate Immun. 2020 doi: 10.1177/1753425920954281. 175342592095428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Puerta-Guardo H., Glasner D.R., Espinosa D.A., Biering S.B., Patana M., Ratnasiri K., Wang C., Beatty P.R., Harris E. Flavivirus NS1 triggers tissue-specific vascular endothelial dysfunction reflecting disease tropism. Cell Rep. 2019;26:1598–1613. doi: 10.1016/j.celrep.2019.01.036. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Froldi G., Dorigo P. Endothelial dysfunction in Coronavirus disease 2019 (COVID-19): gender and age influences. Med. Hypotheses. 2020;144:110015. doi: 10.1016/j.mehy.2020.110015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tsivgoulis G., Katsanos A.H., Ornello R., Sacco S. Ischemic stroke epidemiology during the COVID-19 pandemic: navigating uncharted waters with changing tides. Stroke. 2020;51:1924–1926. doi: 10.1161/STROKEAHA.120.030791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Al-Zaidan L., Mestiri S., Raza A., Merhi M., Philipose V., Fernandez Q., Taib N., Uddin S., Dermime S. 2020. SARS-CoV-2 S protein binding hACE2: viral entry, pathogenesis, prognosis, and potential therapeutic targets; p. 43. [DOI] [PubMed] [Google Scholar]
- 106.Ghafouri-Fard S., Noroozi R., Omrani M.D., Branicki W., Pośpiech E., Sayad A., Pyrc K., Łabaj P.P., Vafaee R., Taheri M., Sanak M. Angiotensin converting enzyme: a review on expression profile and its association with human disorders with special focus on SARS-CoV-2 infection. Vascul Pharmacol. 2020 Jul;130:106680. doi: 10.1016/j.vph.2020.106680. Epub 2020 May 11. PMID: 32423553; PMCID: PMC7211701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zhang H., Quek K., Chen R., Chen J., Chen B. Expression of the SAR2-Cov-2 receptor ACE2 reveals the susceptibility of COVID-19 in non-small cell lung cancer. J. Cancer. 2020;11:4. doi: 10.7150/jca.49462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cancer Labs Pivot to battle COVID-19. Cancer Discov. 2020;10:634. doi: 10.1158/2159-8290.CD-ND2020-006. [DOI] [PubMed] [Google Scholar]
- 109.Hua Y., Bergers G. Tumors vs. chronic wounds: an immune cell’s perspective. Front. Immunol. 2019;10:2178. doi: 10.3389/fimmu.2019.02178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wilkinson A.L., Qurashi M., Shetty S. The role of sinusoidal endothelial cells in the axis of inflammation and cancer within the liver. Front. Physiol. 2020;11:990. doi: 10.3389/fphys.2020.00990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Chakravarty D., Nair S.S., Hammouda N., Ratnani P., Gharib Y., Wagaskar V., Mohamed N., Lundon D., Dovey Z., Kyprianou N., Tewari A.K. Sex differences in SARS-CoV-2 infection rates and the potential link to prostate cancer. Commun. Biol. 2020;3:374. doi: 10.1038/s42003-020-1088-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Meng Y., Lu W., Guo E., Liu J., Yang B., Wu P., Lin S., Peng T., Fu Y., Li F., Wang Z., Li Y., Xiao R., Liu C., Huang Y., Lu F., Wu X., You L., Ma D., Sun C., Wu P., Chen G. Cancer history is an independent risk factor for mortality in hospitalized COVID-19 patients: a propensity score-matched analysis. J. Hematol. Oncol. 2020;13:75. doi: 10.1186/s13045-020-00907-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Desai A., Sachdeva S., Parekh T., Desai R. COVID-19 and cancer: lessons from a pooled meta-analysis. JCO Glob. Oncol. 2020:557–559. doi: 10.1200/GO.20.00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Wang H., Zhang L. Risk of COVID-19 for patients with cancer. Lancet Oncol. 2020;21 doi: 10.1016/S1470-2045(20)30149-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ged Y., Markowski M.C., Pierorazio P.M. Advanced renal cell carcinoma and COVID-19 — a personal perspective. Nat. Rev. Urol. 2020;17:425–427. doi: 10.1038/s41585-020-0345-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Mehta V., Goel S., Kabarriti R., Cole D., Goldfinger M., Acuna-Villaorduna A., Pradhan K., Thota R., Reissman S., Sparano J.A., Gartrell B.A., Smith R.V., Ohri N., Garg M., Racine A.D., Kalnicki S., Perez-Soler R., Halmos B., Verma A. Case fatality rate of cancer patients with COVID-19 in a New York Hospital System. Cancer Discov. 2020;10:935–941. doi: 10.1158/2159-8290.CD-20-0516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Rao S., Lau A., So H.-C. Exploring diseases/traits and blood proteins causally related to expression of ACE2, the putative receptor of SARS-CoV-2: a Mendelian randomization analysis highlights tentative relevance of diabetes-related traits. Diabetes Care. 2020;43:1416–1426. doi: 10.2337/dc20-0643. [DOI] [PubMed] [Google Scholar]
- 118.Allegra A., Pioggia G., Tonacci A., Musolino C., Gangemi S. Cancer and SARS-CoV-2 infection: diagnostic and therapeutic challenges. Cancers. 2020;12:1581. doi: 10.3390/cancers12061581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Dai M., Liu D., Liu M., Zhou F., Li G., Chen Z., Zhang Z., You H., Wu M., Zheng Q., Xiong Y., Xiong H., Wang C., Chen C., Xiong F., Zhang Y., Peng Y., Ge S., Zhen B., Yu T., Wang L., Wang H., Liu Y., Chen Y., Mei J., Gao X., Li Z., Gan L., He C., Li Z., Shi Y., Qi Y., Yang J., Tenen D.G., Chai L., Mucci L.A., Santillana M., Cai H. Patients with cancer appear more vulnerable to SARS-COV-2: a multi-center study during the COVID-19 outbreak. Cancer Discov. 2020 doi: 10.1158/2159-8290.CD-20-0422. CD-20-0422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Miyashita H., Mikami T., Chopra N., Yamada T., Chernyavsky S., Rizk D., Cruz C. Do patients with cancer have a poorer prognosis of COVID-19? An experience in New York City. Ann. Oncol. 2020;31:1088–1089. doi: 10.1016/j.annonc.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kuderer N.M., Choueiri T.K., Shah D.P., Shyr Y., Rubinstein S.M., Rivera D.R., Shete S., Hsu C.-Y., Desai A., de Lima Lopes G., Grivas P., Painter C.A., Peters S., Thompson M.A., Bakouny Z., Batist G., Bekaii-Saab T., Bilen M.A., Bouganim N., Larroya M.B., Castellano D., Del Prete S.A., Doroshow D.B., Egan P.C., Elkrief A., Farmakiotis D., Flora D., Galsky M.D., Glover M.J., Griffiths E.A., Gulati A.P., Gupta S., Hafez N., Halfdanarson T.R., Hawley J.E., Hsu E., Kasi A., Khaki A.R., Lemmon C.A., Lewis C., Logan B., Masters T., McKay R.R., Mesa R.A., Morgans A.K., Mulcahy M.F., Panagiotou O.A., Peddi P., Pennell N.A., Reynolds K., Rosen L.R., Rosovsky R., Salazar M., Schmidt A., Shah S.A., Shaya J.A., Steinharter J., Stockerl-Goldstein K.E., Subbiah S., Vinh D.C., Wehbe F.H., Weissmann L.B., Wu J.T.-Y., Wulff-Burchfield E., Xie Z., Yeh A., Yu P.P., Zhou A.Y., Zubiri L., Mishra S., Lyman G.H., Rini B.I., Warner J.L., Abidi M., Acoba J.D., Agarwal N., Ahmad S., Ajmera A., Altman J., Angevine A.H., Azad N., Bar M.H., Bardia A., Barnholtz-Sloan J., Barrow B., Bashir B., Belenkaya R., Berg S., Bernicker E.H., Bestvina C., Bishnoi R., Boland G., Bonnen M., Bouchard G., Bowles D.W., Busser F., Cabal A., Caimi P., Carducci T., Casulo C., Chen J.L., Clement J.M., Chism D., Cook E., Curran C., Daher A., Dailey M., Dahiya S., Deeken J., Demetri G.D., DiLullo S., Duma N., Elias R., Faller B., Fecher L.A., Feldman L.E., Friese C.R., Fu P., Fu J., Futreal A., Gainor J., Garcia J., Gill D.M., Gillaspie E.A., Giordano A., Glace (Mary) Grace, Grothey A., Gulati S., Gurley M., Halmos B., Herbst R., Hershman D., Hoskins K., Jain R.K., Jabbour S., Jha A., Johnson D.B., Joshi M., Kelleher K., Kharofa J., Khan H., Knoble J., Koshkin V.S., Kulkarni A.A., Lammers P.E., Leighton J.C., Lewis M.A., Li X., Li A., Lo K.M.S., Loaiza-Bonilla A., LoRusso P., Low C.A., Lustberg M.B., Mahadevan D., Mansoor A.-H., Marcum M., Markham M.J., Marshall C. Handy, Mashru S.H., Matar S., McNair C., McWeeney S., Mehnert J.M., Menendez A., Menon H., Messmer M., Monahan R., Mushtaq S., Nagaraj G., Nagle S., Naidoo J., Nakayama J.M., Narayan V., Nelson H.H., Nemecek E.R., Nguyen R., Nuzzo P.V., Oberstein P.E., Olszewski A.J., Owenby S., Pasquinelli M.M., Philip J., Prabhakaran S., Puc M., Ramirez A., Rathmann J., Revankar S.G., Rho Y.S., Rhodes T.D., Rice R.L., Riely G.J., Riess J., Rink C., Robilotti E.V., Rosenstein L., Routy B., Rovito M.A., Saif M.W., Sanyal A., Schapira L., Schwartz C., Serrano O., Shah M., Shah C., Shaw G., Shergill A., Shouse G., Soares H.P., Solorzano C.C., Srivastava P.K., Stauffer K., Stover D.G., Stratton J., Stratton C., Subbiah V., Tamimi R., Tannir N.M., Topaloglu U., Van Allen E., Van Loon S., Vega-Luna K., Venepalli N., Verma A.K., Vikas P., Wall S., Weinstein P.L., Weiss M., Wise-Draper T., Wood W.A., Xu W. (Vincent), Yackzan S., Zacks R., Zhang T., Zimmer A.J., West J. Clinical impact of COVID-19 on patients with cancer (CCC19): a cohort study. The Lancet. 2020;395:1907–1918. doi: 10.1016/S0140-6736(20)31187-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.van Dam P.A., Huizing M., Mestach G., Dierckxsens S., Tjalma W., Trinh X.B., Papadimitriou K., Altintas S., Vermorken J., Vulsteke C., Janssens A., Berneman Z., Prenen H., Meuris L., Vanden Berghe W., Smits E., Peeters M. SARS-CoV-2 and cancer: are they really partners in crime? Cancer Treat. Rev. 2020;89:102068. doi: 10.1016/j.ctrv.2020.102068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Yu J., Ouyang W., Chua M.L.K., Xie C. SARS-CoV-2 transmission in patients with cancer at a tertiary care hospital in Wuhan, China. JAMA Oncol. 2020;6:1108. doi: 10.1001/jamaoncol.2020.0980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Saheb Sharif-Askari N., Saheb Sharif-Askari F., Alabed M., Tayoun A.A., Loney T., Uddin M., Senok A., Al Heialy S., Hamoudi R., Kashour T., Alsheikh-Ali A., Hamid Q., Halwani R. Effect of common medications on the expression of SARS-CoV-2 entry receptors in kidney tissue. Clin. Transl. Sci. 2020 doi: 10.1111/cts.12862. cts.12862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Cameron A.C., Touyz R.M., Lang N.N. Vascular complications of cancer chemotherapy. Can. J. Cardiol. 2016;32:852–862. doi: 10.1016/j.cjca.2015.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Herrmann J., Yang E.H., Iliescu C.A., Cilingiroglu M., Charitakis K., Hakeem A., Toutouzas K., Leesar M.A., Grines C.L., Marmagkiolis K. Vascular toxicities of cancer therapies. Circulation. 2016;133:1272–1289. doi: 10.1161/CIRCULATIONAHA.115.018347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kollias A., Kyriakoulis K.G., Dimakakos E., Poulakou G., Stergiou G.S., Syrigos K. Thromboembolic risk and anticoagulant therapy in COVID-19 patients: emerging evidence and call for action. Br. J. Haematol. 2020;189:846–847. doi: 10.1111/bjh.16727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Barrett C.D., Moore H.B., Moore E.E., McIntyre R.C., Moore P.K., Burke J., Hua F., Apgar J., Talmor D.S., Sauaia A., Liptzin D.R., Veress L.A., Yaffe M.B. Fibrinolytic therapy for refractory COVID-19 acute respiratory distress syndrome: scientific rationale and review. Res. Pract. Thromb. Haemost. 2020;4:524–531. doi: 10.1002/rth2.12357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Rizk J.G., Kalantar-Zadeh K., Mehra M.R., Lavie C.J., Rizk Y., Forthal D.N. Pharmaco-immunomodulatory therapy in COVID-19. Drugs. 2020;80:1267–1292. doi: 10.1007/s40265-020-01367-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Batlle D., Wysocki J., Satchell K. Soluble angiotensin-converting enzyme 2: a potential approach for coronavirus infection therapy? Clin. Sci. 2020;134:543–545. doi: 10.1042/CS20200163. [DOI] [PubMed] [Google Scholar]
- 131.Gray E., Hogwood J., Mulloy B. In: Heparin - Century Prog. Lever R., Mulloy B., Page C.P., editors. Springer Berlin Heidelberg; Berlin, Heidelberg: 2012. The anticoagulant and antithrombotic mechanisms of heparin; pp. 43–61. [DOI] [Google Scholar]
- 132.Mulloy B., Hogwood J., Gray E., Lever R., Page C.P. Pharmacology of heparin and related drugs. Pharmacol. Rev. 2016;68:76–141. doi: 10.1124/pr.115.011247. [DOI] [PubMed] [Google Scholar]
- 133.Meneghetti M.C.Z., Hughes A.J., Rudd T.R., Nader H.B., Powell A.K., Yates E.A., Lima M.A. Heparan sulfate and heparin interactions with proteins. J. R. Soc. Interface. 2015;12:20150589. doi: 10.1098/rsif.2015.0589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Perico L., Benigni A., Casiraghi F., Ng L.F.P., Renia L., Remuzzi G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat. Rev. Nephrol. 2020 doi: 10.1038/s41581-020-00357-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Tang N., Bai H., Chen X., Gong J., Li D., Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020;18:1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Shukla D., Spear P.G. Herpesviruses and heparan sulfate: an intimate relationship in aid of viral entry. J. Clin. Invest. 2001;108:503–510. doi: 10.1172/JCI200113799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Mycroft-West C.J., Su D., Pagani I., Rudd T.R., Elli S., Guimond S.E., Miller G., Meneghetti M.C.Z., Nader H.B., Li Y., Nunes Q.M., Procter P., Mancini N., Clementi M., Bisio A., Forsyth N.R., Turnbull J.E., Guerrini M., Fernig D.G., Vicenzi E., Yates E.A., Lima M.A., Skidmore M.A. Heparin inhibits cellular invasion by SARS-CoV-2: structural dependence of the interaction of the surface protein (spike) S1 receptor binding domain with heparin. Biochemistry. 2020 doi: 10.1101/2020.04.28.066761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Iba T., Hashiguchi N., Nagaoka I., Tabe Y., Kadota K., Sato K. Heparins attenuated histone-mediated cytotoxicity in vitro and improved the survival in a rat model of histone-induced organ dysfunction. Intensive Care Med. Exp. 2015;3:36. doi: 10.1186/s40635-015-0072-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zhu C., Liang Y., Li X., Chen N., Ma X. Unfractionated heparin attenuates histone-mediated cytotoxicity in vitro and prevents intestinal microcirculatory dysfunction in histone-infused rats. J. Trauma Acute Care Surg. 2019;87:614–622. doi: 10.1097/TA.0000000000002387. [DOI] [PubMed] [Google Scholar]
- 140.Whyte C.S., Morrow G.B., Mitchell J.L., Chowdary P., Mutch N.J. Fibrinolytic abnormalities in acute respiratory distress syndrome (ARDS) and versatility of thrombolytic drugs to treat COVID-19. J. Thromb. Haemost. 2020;18:1548–1555. doi: 10.1111/jth.14872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wang J., Hajizadeh N., Moore E.E., McIntyre R.C., Moore P.K., Veress L.A., Yaffe M.B., Moore H.B., Barrett C.D. Tissue plasminogen activator (tPA) treatment for COVID-19 associated acute respiratory distress syndrome (ARDS): a case series. J. Thromb. Haemost. 2020;18:1752–1755. doi: 10.1111/jth.14828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Toniati P., Piva S., Cattalini M., Garrafa E., Regola F., Castelli F., Franceschini F., Airò P., Bazzani C., Beindorf E.-A., Berlendis M., Bezzi M., Bossini N., Castellano M., Cattaneo S., Cavazzana I., Contessi G.-B., Crippa M., Delbarba A., De Peri E., Faletti A., Filippini M., Filippini M., Frassi M., Gaggiotti M., Gorla R., Lanspa M., Lorenzotti S., Marino R., Maroldi R., Metra M., Matteelli A., Modina D., Moioli G., Montani G., Muiesan M.-L., Odolini S., Peli E., Pesenti S., Pezzoli M.-C., Pirola I., Pozzi A., Proto A., Rasulo F.-A., Renisi G., Ricci C., Rizzoni D., Romanelli G., Rossi M., Salvetti M., Scolari F., Signorini L., Taglietti M., Tomasoni G., Tomasoni L.-R., Turla F., Valsecchi A., Zani D., Zuccalà F., Zunica F., Focà E., Andreoli L., Latronico N. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia, Italy. Autoimmun. Rev. 2020;19:102568. doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Trouillet-Assant S., Viel S., Gaymard A., Pons S., Richard J.-C., Perret M., Villard M., Brengel-Pesce K., Lina B., Mezidi M., Bitker L., Belot A., Mouton W., Oriol G., Compagnon C., Generenaz L., Cheynet V., Ader F., Becker A., Benech N., Chauvelot P., Chidiac C., Conrad A., Ferry T., Miailhes P., Perpoint T., Perry M., Pouderoux C., Roux S., Triffault-Fillit C., Valour F., Hodane Y., Chauvelot L., Chabert P., Provoost J., David G., Folliet L., Lecam P., Billaud G., Bouscambert M., Escuret V., Frobert E., Bal A., Destras G., Josset L., Morfin F., Munier C., Valette M., Venet F., Garnier L., Pescarmona R., Lombard C., Walzer T. Type I IFN immunoprofiling in COVID-19 patients. J. Allergy Clin. Immunol. 2020;146:206–208. doi: 10.1016/j.jaci.2020.04.029. e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.The RECOVERY Collaborative Group Dexamethasone in hospitalized patients with Covid-19 — preliminary report. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2021436. NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.National Library of Medicine (US) National Center for Biotechnology Information, PubChem Compound Summary for CID 5743, Dexamethasone. 2004. https://pubchem.ncbi.nlm.nih.gov/compound/Dexamethasone (accessed September 24, 2020)
- 146.Sebba A. Tocilizumab: the first interleukin-6-receptor inhibitor. Am. J. Health. Syst. Pharm. 2008;65:1413–1418. doi: 10.2146/ajhp070449. [DOI] [PubMed] [Google Scholar]
- 147.Sheppard M., Laskou F., Stapleton P.P., Hadavi S., Dasgupta B. Tocilizumab (Actemra) Hum. Vaccines Immunother. 2017;13:1972–1988. doi: 10.1080/21645515.2017.1316909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Luo P., Liu Y., Qiu L., Liu X., Liu D., Li J. Tocilizumab treatment in COVID-19: a single center experience. J. Med. Virol. 2020;92:814–818. doi: 10.1002/jmv.25801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Kewan T., Covut F., Al-Jaghbeer M.J., Rose L., Gopalakrishna K.V., Akbik B. Tocilizumab for treatment of patients with severe COVID–19: a retrospective cohort study. EClinicalMedicine. 2020;24:100418. doi: 10.1016/j.eclinm.2020.100418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hadjadj J., Yatim N., Barnabei L., Corneau A., Boussier J., Smith N., Péré H., Charbit B., Bondet V., Chenevier-Gobeaux C., Breillat P., Carlier N., Gauzit R., Morbieu C., Pène F., Marin N., Roche N., Szwebel T.-A., Merkling S.H., Treluyer J.-M., Veyer D., Mouthon L., Blanc C., Tharaux P.-L., Rozenberg F., Fischer A., Duffy D., Rieux-Laucat F., Kernéis S., Terrier B. Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients. Science. 2020;369:718–724. doi: 10.1126/science.abc6027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Blanco-Melo D., Nilsson-Payant B.E., Liu W.-C., Uhl S., Hoagland D., Møller R., Jordan T.X., Oishi K., Panis M., Sachs D., Wang T.T., Schwartz R.E., Lim J.K., Albrecht R.A., ten Oever B.R. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020;181:1036–1045. doi: 10.1016/j.cell.2020.04.026. e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wang N., Zhan Y., Zhu L., Hou Z., Liu F., Song P., Qiu F., Wang X., Zou X., Wan D., Qian X., Wang S., Guo Y., Yu H., Cui M., Tong G., Xu Y., Zheng Z., Lu Y., Hong P. Retrospective multicenter cohort study shows early interferon therapy is associated with favorable clinical responses in COVID-19 patients. Cell Host Microbe. 2020;28:455–464. doi: 10.1016/j.chom.2020.07.005. e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Zhou P., Yang X.-L., Wang X.-G., Hu B., Zhang L., Zhang W., Si H.-R., Zhu Y., Li B., Huang C.-L., Chen H.-D., Chen J., Luo Y., Guo H., Jiang R.-D., Liu M.-Q., Chen Y., Shen X.-R., Wang X., Zheng X.-S., Zhao K., Chen Q.-J., Deng F., Liu L.-L., Yan B., Zhan F.-X., Wang Y.-Y., Xiao G.-F., Shi Z.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Hoffmann M., Kleine-Weber H., Krüger N., Müller M., Drosten C., Pöhlmann S. The novel coronavirus 2019 (2019-nCoV) uses the SARS-coronavirus receptor ACE2 and the cellular protease TMPRSS2 for entry into target cells. Mol. Biol. 2020 doi: 10.1101/2020.01.31.929042. [DOI] [Google Scholar]
- 155.Rodríguez-Puertas R. ACE2 activators for the treatment of COVID 19 patients. J. Med. Virol. 2020;92:1701–1702. doi: 10.1002/jmv.25992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Zhao Y., Zhao Z., Wang Y., Zhou Y., Ma Y., Zuo W. Single-cell RNA expression profiling of ACE2, the receptor of SARS-CoV-2. Bioinformatics. 2020 doi: 10.1101/2020.01.26.919985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Monteil V., Kwon H., Prado P., Hagelkrüys A., Wimmer R.A., Stahl M., Leopoldi A., Garreta E., Hurtado del Pozo C., Prosper F., Romero J.P., Wirnsberger G., Zhang H., Slutsky A.S., Conder R., Montserrat N., Mirazimi A., Penninger J.M. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181:905–913. doi: 10.1016/j.cell.2020.04.004. e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., Huan Y., Yang P., Zhang Y., Deng W., Bao L., Zhang B., Liu G., Wang Z., Chappell M., Liu Y., Zheng D., Leibbrandt A., Wada T., Slutsky A.S., Liu D., Qin C., Jiang C., Penninger J.M. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus–induced lung injury. Nat. Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Gras Navarro A., Björklund A.T., Chekenya M. Therapeutic potential and challenges of natural killer cells in treatment of solid tumors. Front. Immunol. 2015;6 doi: 10.3389/fimmu.2015.00202. [DOI] [PMC free article] [PubMed] [Google Scholar]