Abstract
The COVID-19 is a novel infectious disease caused by SARS-CoV-2 and is known as a pandemic emergency that has led to a high rate of mortality throughout the world. Evidence has indicated that hyperinflammatory responses triggered by SARS-CoV-2 are the main cause of pathogenicity in the severe cases of patients who have died during the current viral disease. Monocytes and macrophages as the most important cells of the innate arm of the immune system play a substantial part in the body's defense against viral infections. They mainly respond to the microbial antigens by producing inflammatory mediators to remove pathogens and repair tissue injury. Nevertheless, aberrant alterations in their function such as cytokine storm can be so harmful to the host in the acute respiratory distress syndrome cases caused by SARS-CoV-2. Moreover, inflammatory responses stimulated by SARS-CoV-2 have affected the other vital organs of the body including the heart. As cardiovascular complications in COVID-19 patients have been reported in several studies. During the infection, monocytes and macrophages may be involved in the hypersensitive and exacerbated reactions that contribute to the tissue damage, especially lung injury resulted in its dysfunction and respiratory disorder. In this review, we discuss both advantageous and disadvantageous about the pathological potential of monocytes and macrophages during the infection of SARS-CoV-2 to clarify their mutual effects on immune processing as a fist line defender in the current disease.
Keywords: COVID-19, SARS-CoV-2, Monocyte, Macrophage, Inflammation
Graphical abstract
1. Introduction
COVID-19 is a disease caused by a new coronavirus (SARS-CoV-2), which according to the declaration of the World Health Organization (WHO) on March 11, 2020, has known as a pandemic threat and global emergency [1]. As yet, SARS-CoV-2 infection has transmitted throughout the world and endangered the life of humans. Its clinical manifestations and severity are varied from person to person including fever, cough, dyspnea, and fatigue as common symptoms. Moreover, the following symptoms are also characterized in COVID-19: Sputum production, headache, diarrhea, hemoptysis, and the loss of smell or taste [2].
The main route of novel coronavirus transmission is via inhalation of the infectious respiratory droplets from infected patients with symptomatic and asymptotic conditions. However, it can indirectly spread through other ways such as post-natal and fecal-oral routes by contaminated things or surfaces [3]. Nevertheless, the findings have demonstrated that pulmonary inflammation and consequently acute lung injury are the important causes of mortality resulted from this disease. Furthermore, the clinical manifestation of cardiovascular disorder such as myocardial injury has been observed in COVID-19 cases [4]. Although the pathogenicity pattern of coronavirus is so intricate and variable, noteworthy data are available in this regard [2]. According to the studies, the spike (S) protein of this virus is the most potent immunogenic proteins during current infection. A human host cell receptor described for SARS-CoV-2 entry is angiotensin-converting enzyme 2 (ACE2) that probably leads to infect airway epithelial cells as the most commonly injured tissue due to their abundant ACE2 receptors via attaching to the S protein receptor-binding domain (RBD) of the virus [5,6]. Monocytes and macrophages as two subpopulations of immune cells that have pivotal functions in the immunity and host defense against various microbes in the lung. The Lung resident macrophages are accompanying by different subgroups of monocyte that provide effectual lung immune responses to eliminate alien materials from the airways and blood. Indeed, they can represent a suitable reaction (activator or modulator) through diverse mechanisms depending on the microenvironment and type of pathogen [7]. However, it has been shown that in addition to the beneficial roles of monocytes and macrophages to defend from the host as soldiers serving the immune system during the severe respiratory infection caused by SARS-CoV-2, they represent pathological activity, potentially [8]. It is possible that SARS-CoV-2 can infect macrophages and monocytes through ACE2-independent and ACE2-dependent pathways and thus they will lack their function to combat against viruses and also these infected cells won't have the ability to evoke adaptive immune responses. On the other hand, their dysregulated functions can lead to patients' organ damages, mainly through flaming the acute inflammation, promoting cytokine storm and extending fibrotic complications. Therefore, they worsen the disease to a high extent and cause acute respiratory distress syndrome (ARDS), which is often resulted in death in patients with COVID-19, consequently [9,10].
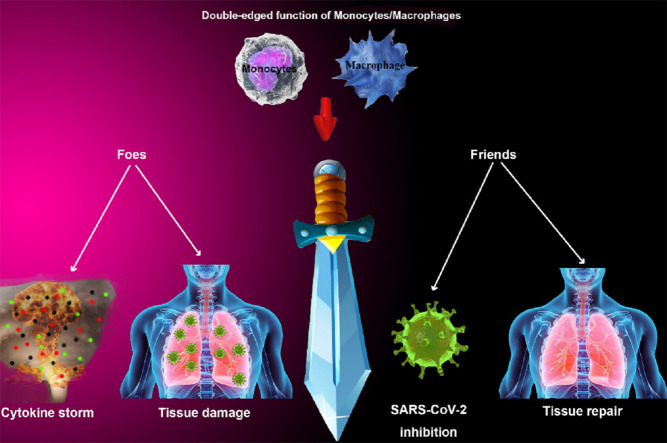
In this review, we are going to explain the double-edged role of macrophages and monocytes during the infection of SARS-CoV-2 in COVID-19 cases to gather studies with the aim of clarifying their functions as friends and foes of the current infection.
1.1. Monocytes and macrophages in viral immune response
Monocytes are a subset of leukocytes mostly originated from myeloid progenitors in the bone marrow and exist in the bloodstream. They are able to differentiate into macrophages in tissue and dendritic cells (DCs) [11]. Based on the amount of expression of distinct surface markers, monocytes in humans are divided into the two main subgroups with different characterization. Classical or CD14++CD16− monocytes are known as the most conventional monocytes that are similar to LY6Chi monocytes in mice. It has found in animal studies that LY6Chi has good potential for developing CD103+ DCs in the lung, and specifically expresses C-C chemokine receptor type 2 (CCR2). The second subtype of human monocytes is the CD16+ monocyte population that itself consists of intermediate (CD14++CD16+) and non-classical (CD14+CD16++) monocytes which have been defined to resemble mouse LY6C low monocyte regarding in vivo patrolling and can give rise to CD11b+ DCs [12,13]. Also, they have differentiation capability in the lung to turn into parenchymal macrophages that in the following stage cause the formation of alveolar macrophages [14]. Therefore, with considering available knowledge, under the steady condition, monocytes are renovating DC and macrophage sources of the body. The results of numerous investigations have revealed that monocytes are momentous players in the strategies of immune responses to infections such as protection against viral pathogens. They traffic and recruit regularly to the position of infection for accomplishing their functions and exert notable antimicrobial actions [15].
Macrophages are other cells of the innate immune system that are derived from monocytes and specialize in phagocytosis. Moreover, these cells have the ability to exert an extensive range of immune functions such as antigen presentation, the production of chemokine and cytokine, and activation of the other immune cells including T and B cells [16,17]. Macrophages can be found in different tissues throughout the body such as the respiratory system. Under steady-state, at least two main types including interstitial macrophages (IM) and alveolar macrophages (AM) form the macrophages population in the healthy lung that their phenotypes, localization, and function are distinguishable [18,19]. Although in presence of the pathogen, alveolar macrophages are polarized toward the M1 or M2 subgroup according to the microenvironment existence and required immunological responses. It is established that M2 cells belong to producing allergic cytokines which are related to tissue remodeling and pathology [19,20]. Generally, alveolar macrophages are residents in pulmonary alveoli and exhibit a pivotal effect in the defending process against respiratory infections through scavenging and phagocytosis of different kinds of pathogens particles such as viruses in the lung. They secrete a broad range of cytokines to induce inflammatory reactions and tissue repairing. Furthermore, macrophages are a powerful activator for triggering adaptive immune responses and killing microbes [20,21]. According to the points mentioned above, both monocytes and macrophages have a fundamental role in the first line of immunological responses toward viral infections [17]. During the viral infections, those subpopulations of DCs derived from nonresident monocytes aggregate as inflammatory monocytes in the lungs. Dunbar et al. illustrated in a study that classical monocytes are necessary to the lung resident memory CD8 T cells development (TRM) as essential defensive agents in response to respiratory viruses after being infected with influenza A and they have indicated a significant effect of pulmonary monocytes in TRM establishment via the lung T cells direct interaction [22]. A study conducted in 2009 showed that Ly6Chi inflammatory monocytes can mediate an exclusive response to the viral pathogens by driving Toll like receptor (TLR2)-dependent type I interferon production which is led to distinguishing between pathogen types and stimulation of antiviral specific mechanism [23]. Masson F et al. in 2008 stated monocytes as the main origins of DCs that have a great impact on the immune defensive process against viral infection. They referred to the substantial role of DCS in cross-presentation that viral antigens present to the CD8+ T cells via the major histocompatibility complex (MHC) class I pathway and powerful specific responses induce against them, finally [24]. Nonetheless, numerous researches have been demonstrated the unfavorable effects of monocytes during the responses to viral infections. Therefore, both profitable and detrimental activity are defined for these innate immune cells. In this regard, it was shown that the morbidity of CCR2 deficient mice infected with influenza virus was decreased significantly in the animal study and it was related to disturbed monocytes recruitment to the lung. Therefore, results have illustrated that diminishing monocytes recruitment to the virus contaminated tissue led to decrease the immunopathology complications of the disease. Generally, CCR2 and its ligand monocyte chemoattractant protein 1 (MCP-1) are recognized as inducer factors to the employment of inflammatory monocytes from the blood circulation to the infectious lungs or lymph nodes during the inflammation and infection [25]. One of the most threatening human health during the last decades is human immunodeficiency virus (HIV) which causes acquired immunodeficiency syndrome (AIDS) and there is no certain therapeutic approach for it [26]. Scientifics have found that this retrovirus can infect those cells of the immune system which express CD4 marker on their surface. It is defined that tissue-resident macrophages and MDM (monocytes derived macrophages) are extremely susceptible to infection with HIV, while the blood monocytes represent less vulnerability [27]. The results obtained from several studies have shown that HIV entry is associated with the amount of CCR5 or expression of CD4, and the very low surface expression of them can limit the virus-cell entry and diminish the severity of the infection [[28], [29], [30]]. The study by Fear et al. has demonstrated that HIV-1 infection in fresh monocytes is blocked in the beginning steps, before performing integration and reverse transcription [31]. These data also have been confirmed by another study by Sonza S et al. that was carried out on one-day-old monocytes and investigated their susceptibility to HIV infection compared with mature MDM [32].
Unfortunately, infected tissue macrophages are defined as permanent reservoirs of HIV. Due to their long half-life, they are able to spread the virus throughout the body and lead to the progression of AIDS [25,33]. This duration is estimated for about two months for alveolar macrophages [34]. In line with this argument, the results of some experiments were shown that the detected viral loads in the cerebrospinal fluid due to the presence of tissue macrophages in the relevant site was dramatically higher than viral loads in the other biological fluid such as plasma and semen [35,36]. Also, studies have reported that macrophages and monocytes have a critical duty in the HIV-associated neuro disorders such as dementia as a target for this virus via facilitating neuroinflammatory mechanisms. Overall, these immune cells represent a central pathogenesis action during the HIV infection [37]. Respiratory syncytial virus (RSV) is a part of the Pneumoviridae family that can cause pneumonia in elderly people, children, and persons with immunodeficiency disease which are classified into high-risk groups. This unusual virus leads to repeated infections in patients even for years after the first affliction. Joleen M et al. have suggested in their study that producing some chemokines such as macrophage inflammatory protein (MIP1α) and chemokines RANTES by airway epithelial cells during RSV infection causes the monocytes recruitment to the infection sites to expose suitable immune responses against the current pathogen in order to restrict the progression of the infection [38]. It is demonstrated that the secretion of interleukin 16 (IL-16) by monocytes helps the attraction of CD4 positive cells [39]. Moreover, increasing some receptors such as CD40, CD80 and CD 86 on the surface of monocytes resulted in enhancement of migration of these cells to the infection area and differentiation into the DCs to promote immunological reactions [40]. One of the other viruses considered as a human pathogen is the dengue virus which is responsible for dengue fever [41]. The results of Mohit Singla et al. research in 2016 have illustrated that monocyte which could be infected with dengue virus had a significant role in the pathogenesis of it and these findings were useful for the exploration of novel therapeutic approaches through the prediction of marker to attenuate severe infection [42].
Zaire Ebolavirus (ZEBOV) is responsible for EVD (Ebola virus disease) which often causes a severe hemorrhagic fever in humans and other mammals and can lead to death [43]. It has been indicated in the Menicucci et al. study that monocytes could be a target for this virus and play a central role in the pathogenesis and replication of ZEBOV [44]. Furthermore, they intensified clinical manifestation of EVD via enhancement of inflammation and coagulation related genes. The exacerbating effect of monocytes on the filoviral hemorrhagic disease was also revealed in the other investigations [45].
In summary, monocytes, and macrophages are known as crucial immune cells with a wide range of immunological function that can be in favor of extending viral infection within the body or limiting pathogen activity through exhibiting beneficial effects [8].
1.2. Alteration in blood monocytes in COVID-19
Peripheral blood monocytes are fundamental mononuclear cells to provide appropriate control of pathogens such as viruses [46]. Nonetheless, it has been discovered that following the SARS-CoV-2 infection, some of their functions are disrupted including cytokine production and chemotaxis. In other words, a pattern of altered chemokine and cytokine profiles have been observed in the blood monocytes of COVID-19 patients. This contributes to a series of inefficient response which subsequently enhances the pathogenesis of COVID-19 and leads to an increase in mortality rates [9,47].
The reports of alteration in the number of blood monocytes during the coronavirus infection depend on the stage of the disease and are variable in different manuscripts. Flow cytometry analyses of blood samples of COVID-19 were demonstrated that monocytes count didn't change, while the size of monocytes was larger than normal monocytes which were related to an inflammatory phenotype [48]. However, the results of some study such as the research that has been accomplished by Paliogiannis et al. indicated that the blood monocytes count in COVID-19 patients significantly decreased [49]. Moreover, diminished monocyte count has been reported in COVID-19 patients by the other studies and they revealed that the phenotype of circulating monocytes in severe-stage cases mostly consists of CD14+, and CD16+ inflammatory monocytes subpopulation which exerts inflammatory activity especially through producing IL-6 in intensive care unit (ICU) admitted COVID-19 patients [50]. Henry and colleagues have revealed in their meta-analysis study that circulating monocytes number in the COVID-19 patient decreased, while it has defined that MDW (monocyte distribution width) remarkably elevated through current disease [51]. Whereas, the number of blood mononuclear cells exhibited enhancement in the investigation performed by Andonegui-Elguera et al. They showed that the other lymphocytes including neutrophils, natural killer (NK), and T cell count have diminished, dramatically [52].
Data from the study which has done by Wen et al. showed that cytokine activation has occurred regarding the expansion of the inflammatory monocytes' population (especially CD14 phenotypes) which is considered as a clinical manifestation of blood monocytes dysregulation and led to increase inflammation in the SARS-COV-2 infection. It also revealed that inflammatory monocytes can induce inflammatory responses through interaction with adaptive immune cells consisting of B and T cells. Furthermore, T cells are capable to activate monocytes via expressing colony stimulating factor 2 and 1 (CSF2 and CSF1) as ligands to their receptors which exist on the monocytes and thus cause the inflammatory storm. Another exclusive interaction between CD14 inflammatory monocytes and T cells detected in the erythrocyte sedimentation rate (ESR) of patients with COVID-19 is through binding IL1RAP expressed by T cells to its ligand on the surface of monocytes that induce immune responses. In addition, results have illustrated a large amount of IL-6, lymphotoxin beta (LTB), and LTA secretion from B cells which bind to their receptor in monocytes. High doses of IL-6 can stimulate the production of inflammatory chemokines and cytokines such as interferon gamma (IFN-γ), IL-1β from T cells. These activated immune cells were able to move via the bloodstream to the various organs especially the lungs and led to tissue damage that has been manifested in the patients infected with SARS-CoV-2 [53]. These results have been proven by the other study that was carried out by Chuang Guo et al. They identified specific monocytes subset and related cytokine or chemokine which may in accompany with plasma B cells and T cells cause inflammatory storms in COVID-19 severe cases. It also suggested that these cytokines and chemokines and their receptors could be suitable candidates for designing a drug for the treatment of COVID-19. Chemokines that were reported in this regard including CCL4, CCL3, and CCL4L2, and their respective receptors and cytokines such as tumor necrosis factor (TNF), IL10, and IL6 were also identified in association with severe stage-specific monocytes [54].
Zhou et al. investigated the pathological role of inflammatory CD14 ++CD16 ++ monocyte and granulocyte-macrophage colony-stimulating factor (GM-CSF) T cells that secret a great amount of IL-6 to induce inflammation in patients with COVID-19 that had the severe pulmonary syndrome. They demonstrated that during the coronavirus infection CD4 T cells shift to the pathogenic T helper1 which can produce GM-CSF. It was recently revealed that GM-CSF has an effective role in the inflammatory diseases pathogenesis via adjusting the various pathological potential of inflammatory myeloid cells such as monocytes. They are capable to commence the detrimental mechanisms which led to tissue damage in the mouse and human. Therefore, the entrance of pathogenic T cells and monocytes in the lung circulation has been considered as a pulmonary disorder, dysfunction, and morbidity in 2019-nCoV patients [50]. According to this, a monoclonal antibody against GM-CSF or IL-6 could be a good therapeutic approach to attenuate the immunopathogenesis of SARS-CoV-2 and help virus clearance by the host immune system [55]. Zhang et al. identified the CD14+CD16+ monocytes in blood samples of patients with COVID-19 which expressed some of the macrophage markers represented differentiation into the macrophages in the circulation of SARS-CoV-2 infected individuals. This population also exhibited notable potency in contributing cytokine storms through producing a high level of TNF-α, IL-10, and IL-6 that are related to the deterioration of patients and increasing their admission to the ICU [48].
Zhang and colleagues also suggested in their study that ACE2 expression may be downregulated by SARS-CoV-2 which is associated with the disease severity. ACE2 is a receptor for SARS-CoV-2 and the above-mentioned study has been identified that the monocytes isolated from the blood of COVID-19 patients expressed the lower level of ACE2 compared with the healthy control group [6,56]. Moreover, data from the comprehensive review which has been done recently by Verdecchia et al. have supported this argument that diminishing in ACE2 level can result in the infiltration of lung inflammatory cells and subsequently lead to ARDS [57].
Giamarellos-Bourboulis et al. showed that in all patients who were under investigation have significant immune dysregulation that the altered monocytes' function has a principal effect in these aberrant responses. The study by Sanchez-Cerrillo et al. suggested that inflammatory monocytes that were trafficked to the lung have participated in the pathogenesis of novel coronavirus [58].
Overall, alteration in monocyte count, phenotype, and function plays a significant role in the SARS-CoV pathogenesis. Indeed, infected circulating monocytes can induce acute inflammatory responses through exerting inappropriate activity and cause cytokine storm which enhances the pathogenicity of the virus and disease worsening in patients via severe tissue damage and consequently lead to the increased mortality rate of SARS-CoV-2 infection [59].
1.3. Macrophages in respiratory system
Lungs immunity is considered the most sensitive part of protection by the immune system and requires tightening control due to continuous exposure with the air-foreign antigens that could be a pathogen. Large populations of immune cells are placed within the lung tissue, in its blood circulation, and related lymph nodes [60]. As above-mentioned in the current manuscript, the innate immune system substantial cells are macrophages found in all mammals that their phenotypes are regulated by tissue-specific manner [61]. Based on the previous description, there are two main subpopulations of macrophages in the respiratory system, including alveolar macrophages that reside in the airspace lumen, near the epithelial cells and are derived from fetal liver monocytes during embryogenesis, and interstitial macrophages which are more localized in the lung tissue, the space between alveolar epithelium and microvascular endothelium [62,63]. They are divided into the three subgroups according to their surface markers, IM1 (CD206+MHCII−), IM2 (CD206+MHCII+), and IM3 (CD206−CD11c+) [64]. Both IM and AM significantly have self-renewal ability and are essential to maintain lung homeostasis and immunity, although despite numerous functional researches which have been done in this regard, clear in vivo activity of macrophages is still a challenge [64,65].
Alveolar macrophages are defined as the first immune defenders against pathogens and foreign particles that enter into the respiratory system and cause the following immune responses priming in the lung [66]. Alveolar macrophages adapt themselves to the physiological manner via the high potency of phagocytosis and diminishing cytokine production to prevent inflammation [62], whereas the number of alveolar macrophages increase during the inflammatory condition through monocyte recruitment to the lung. Moreover, their functional pattern is changed depending on the dominant microenvironment of the pulmonary organ [[67], [68], [69]]. Indeed, several factors modulate the alveolar macrophage function such as close contact with epithelial cells, hyperoxia, and exposure to surfactant-containing fluids [70]. Alveolar macrophages represent fundamental multiple activities that are necessary to provide suitable immunity in the respiratory system such as antigen recognition and presentation, helping to initiation of inflammation, activation of adaptive immune cells, and contributing to tissue repairing and homeostasis [70]. It has been established that alveolar macrophages are classified into the two main phenotypes: M1 macrophage and M2 macrophage activate in classical and alternative pathways, respectively. M1 macrophages are specialized in cooperation with T helper1 to produce pro-inflammatory cytokines, intensify bacterial removing and also enhancement of the other immune cell migration to the lung parenchyma and alveolus. M2 macrophages are activated via antigen-presenting to the T helper 2 and induced by its cytokines leading to secret anti-inflammatory cytokines, clearance of apoptotic cells, eliminating inflammation, and tissue remodeling. Interestingly, reports have been characterized that alveolar macrophages could represent the signs of both their typical phenotypes (M1 and M2 cells), synchronously depending on the nature of received environmental signals [66,71,72].
Considerably, the lung microenvironment plays a pivotal role in determining the phenotype and functional properties of alveolar macrophages [73]. They exert different responses through recognizing pathogen/damage-associated molecular patterns and to the varied mediators or growth factors that exist in their milieu. Therefore, they can balance immune reaction as pro/anti-inflammatory inducers and pro/anti-fibrotic agents, besides exhibit tissue repairing effects [74,75]. Nonetheless, under the pathogen-associated lung inflammatory condition, alveolar macrophages try to adjust tissue homeostasis and pose an effectual immunity via communicating with the other involved cells such as lymphocytes, neutrophils, fibroblasts, epithelial and microvascular endothelial cells [14,[76], [77], [78], [79]]. It has been suggested that AM is a pivotal cell that contributes to antigen transportation to the lung-associated lymph nodes [80]. Recent studies have reported that lung resident macrophages as the main modifier of pulmonary inflammatory responses that can induce the termination of inflammation. They also play an important role to initiate parenchymal repair mechanisms that are substantial for returning back from inflammatory condition to homeostasis after lung injury (Fig. 1 ).
Fig. 1.
The activity of macrophages to attenuate alveolar inflammation after lung injury and regulation of repair process which is necessary for hemostasis. For attenuating alveolar inflammation and repairing tissue damages, macrophages are involved directly and indirectly. Blockage of monocyte and granulocyte (PMN) recruitment, apoptotic parenchymal and neutrophils phagocytosis, alveolar edema fluid clearance, clearance of fibrin are macrophages activities in this process. In addition, the repairing process of the epithelial and endothelial barrier is induced by junctional sealing, ACE2 proliferation or differentiation, and angiogenesis [81].
AMs own exclusive attributes that we have referred to them. Moreover, their anatomical localization at the air-tissue interface render some momentous duties to mediate pulmonary immune responses [20,82]. Defensive role of macrophages against respiratory viral infection has attracted a wide range of attention during recent years [83]. Alveolar macrophages have been reported to be important effectors of cell-mediating immune responses by capturing, processing, and presenting the virus-derived antigens to the lymphocytes and DCs to trigger adaptive immune responses [84]. Furthermore, they are experts in producing IFN (type II) as an effective cytokine to protect body against viral pathogens [85], probably through helping to modulate immunological processing and inhibition of viral replication, however, the precise nature of macrophage capability to limit the virus replication is yet unclear. To answer this question, better insight into researches will be acquired for the treatment of diverse virus infections [86].
Despite all the antimicrobial benefits of macrophages, the detrimental role of them that result in lung lesions has been established in many types of research through various diagnostic approaches, including assessment of bronchoalveolar lavage (BAL), transbronchial and surgical lung biopsy [87]. For example, it has revealed that pulmonary macrophages are capable of mediating lung inflammatory lesions during mechanical ventilation via activating Nod-like receptor protein 3 (NLRP3) inflammasome and increasing pro-inflammatory responses [88]. Furthermore, few studies have illustrated the pathological role of alveolar macrophages exerted through their macrophage metalloproteinase, MMP-12, and they are also expressed in smooth muscle cells of the airway and bronchial epithelial cells. It has defined that MMP-12 was contributed to tissue remodeling or pulmonary fibrosis in chronic asthma and allergen-induced airway inflammation in mice model, thereby it causes tissue-damages during some respiratory disease [89].
1.4. Macrophages in COVID-19
According to the most important function of lung resident macrophages, we are going to discuss the role of them in the infection of SARS-CoV-2 in the following three main subtitles.
1.4.1. Antibody-dependent cellular cytotoxicity (ADCC)
ADCC occurs when the Fc domain of an immunoglobulin, which is linked to the membrane-viral antigen on the infected cell, attaches to the Fc gamma receptors (FcγRs) on the surface of activated innate immune cells comprising monocytes, neutrophils, natural killer (NK) cells, and macrophages. This attachment causes the secretion of the cytolysis granules containing perforins and granzymes to destruct infected cells. Although the most effector cells to ADCC performing in vivo are NK cells, macrophages are also important cells involved in this interaction [90,91]. It has been considered that ADCC has an effectual role that contributes to the immune responses toward the various type of viruses and it has been investigated in some studies on RSV infection such as the designing of the anti-RSV vaccine [90]. In regards to the SARS coronavirus, antibodies that are produced against this type of virus, exhibit a double-edged effect on the process of infection and represent both of favorable and undesirable activities through cooperation with the immune system components including NK cells mediated by antibody-dependent cell cytotoxicity pathway and macrophages especially alveolar macrophages by Fc gamma receptor (FcγRs)-bearing cells, macrophages, monocytes-derived infiltrating, monocytes, and complement–antibody complex pathways, respectively [92]. Results from Yasui et al. study have shown that phagocytic cells included alveolar macrophages and monocyte-derived infiltrating macrophages through SARS-CoV specific antibodies play a principal part in the clearance of pulmonary cells that were infected with the current virus. Generally, in addition to the elimination of infected cells from the body, excessive Fc-mediated effector response may induce immunopathological complications and tissue-damaging [92].
1.4.2. Antibody-dependent enhancement
It has been established that neutralizing antibodies against the virus have a beneficial effect on supporting the body's immunity but if the antiviral antibodies cannot neutralize the virus, the antibody-dependent enhancement (ADE) phenomenon happens [93,94]. ADE is able to boost the infection of cells that have FCR through the attachment between this type of receptor and the immune complex of virus-antibody [93]. The interesting point is that ADE may be induced by both non-neutralizing and neutralizing antibodies [95]. It has been indicated that ADE could definitely occur during some of the viral infections with the Ebola virus, Zika virus, dengue viruses, HIV, and influenza virus [93]. Also, researchers have recently found that some disorders are resulted from novel coronavirus infection. They showed that produced antibodies due to the prior immunity or transferred antibodies by passive approaches cause to trigger the severity of the disease [96]. It has been defined in several studies that macrophages have a considerable role in the intensifying immunopathogenesis of SARS-CoV-2 through increasing ADE owing to their FCR. It has been proposed that alveolar macrophages might contribute to exacerbating COVID-19 through mediating ADE by their high level of FcγRIIA(CD32A). IgG binding to the FcγRIIA leads to induce the main inflammatory cytokines such as IL-6, TNF-α, IL-1, and IFN-γ through the immunoreceptor tyrosine-based activation motif (ITAM)-related signaling pathway [95,97]. Surprisingly, antibodies against SARS-CoV-2 represent a notable effect on the enhancement of replication in macrophages under in vitro conditions [98]. The anti-spike protein of SARS-CoV-2 ability to infect monocytic cell lines and human macrophages are also defined in the study designed by Jaume et al. [99]. According to the extracted data from several investigations, it seems that inflammatory responses throughout the infection of SARS-CoV-2 are segregated into the two certain phases; primary inflammatory responses that resulted from virus replication, cellular death mediated by apoptosis, or host defensive responses that cause inflammation and it happens when the antiviral antibodies have not still appeared [100]. The secondary phase is initiated from the time point that anti-coronavirus antibodies are produced and detectable. These immunoglobulins (Ig) are capable to restrict virus replication, but they unfortunately make an immune complex with the virus antigens and bind to the surface FCR on cells expressing these receptors. Finally, it leads to uptake virus into the cells especially macrophages, and increases the pro-inflammatory phenotype of M1 macrophages in the lung which could be major secretors of inflammatory mediators including IL-8 and MCP-1 and contribute to the lung damage [100]. The significant correlation between SARS-CoV-2 immunopathogenesis and antibodies against it was discovered in the vaccinated rhesus macaques with the SARS-CoV-2 vaccine manufactured based on anti-S IgG. It has been defined that vaccination before the viral clearance was led to the development of acute lung injury through the recruitment of pro-inflammatory alveolar macrophages which produced a high amount of MCP-1 and IL-8 as inflammation inducer chemokines, while such complications had not been reported in the unvaccinated group [100,101]. Furthermore, data gathered from couple of studies have been demonstrated similar results in which lung damage was escalated in mice and African green monkeys after SARS coronavirus vaccination [102,103]. The same pattern of inflammatory responses has been almost indicated in the COVID-19 sever-stage cases. In addition, dead patients from current infection exhibited profound elevation of MCP-1and IL-8 in their serum samples that were produced by human macrophages which are responsible for the wound healing process [101]. Indeed, in 80% of patients who were afflicted with ARDS, increasing anti-SARS coronavirus IgG was detected in their sera as an accompanying risk factor. Consequently, the mortality rate of patients with SARS-CoV-2 has a direct relationship with the acceleration of antiviral antibody production [104,105].
1.4.3. Cytokine storm
Valuable data obtained from a large number of studies indicated various degrees of cytokine storm occurrence during the COVID-19 [106]. Cytokine storm also called by other names including cytokine release syndrome (CRS) and hyper-cytokinemia, is known as a systematic inflammatory response mediated via immune cells, chiefly monocytes, and macrophages which are excessively activated due to infectious or non-infectious agents and release a great deal of pro-inflammatory cytokines that lead to hyperinflammation through triggering recruitment of leucocytes and production of inflammatory mediators in a positive stimulating cycle [[107], [108], [109], [110], [111], [112]]. It has been reported that the cytokine storm has a principal role in the severity of SARS-CoV-2 infection and lung injury resulted from ARDS, especially in ICU patients [113,114].
It has been discovered that there is a correlation between the sudden worsening of clinical complications in the late stage of disease and cytokine storm [115]. During this phenomenon, a series of cytokines including IL-17, IL-7, IL-1β, IL-9, IL-2, IL-10, TNF-α, GM-CSF, G-CSF, IFN-γ, MCP1, MIP1A, MIP1B, CXCL10 and CXCL8 are distinguished in the novel corona virus-infected patients. Almost all of these chemical mediators generated by monocytes and macrophages can lead to the immunopathological enhancement of COVID-19 [48]. Among this, IL-6 has been introduced as the main driver of CRS via escalating the secretion of many acute-phase proteins and fever-related responses in the severe cases of COVID-19 [2,116]. Moreover, CRS is closely associated with macrophage activation syndrome (MAS). It is defined by several clinical symptoms such as liver failure, pancytopenia, hyperferritinemia, coagulopathy, and neurological disorder. MAS is resulted from the excessive proliferation of differentiated macrophages which cause hypercytokinemia and hemophagocytosis [117,118]. This could induce the pathogenesis of SARS-CoV-2 through ARDS in the patients with COVID-19 that has been reported as a severity manifestation of current infection [119,120]. Another cytokine that has a critical function in mediating cytokine storm is GM-CSF, which is an important activity in the adjustment of monocyte and macrophage differentiation. Data have indicated the increase in the GM-CSF level in the COVID-19 patients who have required ICU admission [119].
According to the investigation of severe cases of COVID-19, it is debatable that inappropriate responses of the patient's immune system, which lead to severe inflammation in the form of cytokine storm, can involve the other vital organs of the body including the heart in addition to the lungs and respiratory system damages. Therefore, it enhances the mortality rate. For example, the study which has been done by Duerra et al. reported that aberrant antiviral immunity can be a cause of heart injury through myocardial inflammation in COVID-19 patients. Fluorescence-activated cell sorting (FACS) evaluation in plasma exchange (PE) cases indicated the enhancement of CD3+CD8+ T cells frequencies among all CD3+ T-cells, while frequencies of regulatory T cells (Tregs) and CD14+HLA−DR+-monocytes were decreased. Also, elevated levels of antiviral cytokines were notably related to high CD8/Treg/monocyte ratio and severity course of disease in PE patients which is considered as a prediction sign of imminent cardiac lesion in COVID-19 cases. In conclusion, the valuable findings of this study revealed the correlation between hyperinflammatory associated heart damage and dysfunctional immune responses such as monocyte alteration during the SARS-CoV-2 infection [4].
In addition, data from the study by Luetkens et al. showed the possibility of a causal relationship between myocardial inflammation and myocardial injury with fatal outcomes in COVID-19 patients. They monitored symptoms of a 79 years old COVID-19 patient with diffuse myocardial inflammation which could result from the direct involvement of cardiomyocyte by virus or myocardial damage due to cytokine storm and imbalanced activity of T helper cells (TH1 and TH2) following the SARS-CoV-2 infection. Furthermore, it was diagnosed by mapping with CMR (Cardiac Magnetic Resonance Imaging) [121].
Sorokin et al. have assessed the other aspect of dysregulated immune responses role in cardiovascular damages. They mentioned in their study that aberrant adaptive immune reactions can increase the individual susceptibility to viral infections. On the other hand, virus triggered acute inflammation contributes to the enhancement of metabolic demands which lead to ischemic injury, and vascular disorder due to myocardial oxygenation. The immune-mediated inflammatory dyslipoproteinemia which is resulted from SAS-CoV-2 stimulated cytokine storm causes the set of risk factors for cardiovascular disease including high serum level of triglycerides, elevated lipoprotein oxidation, low apolipoprotein E (ApoE), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C) levels, and disrupted inflammation resolution due to diminished specialized pro-resolving lipid mediators (SPMs) biosynthesis. They together increase the mortality ratio of dead patients from COVID-19 [122].
Lindner et al. in their cohort study investigated the correlation between cardiac infection and SARS-CoV-2 in 39 autopsy cases of COVID-19 to discover whether SARS-CoV-2 present in the myocardial tissue or not. They also evaluated the cardiac related immune responses against current virus through assessment of TNF-α, IFN-γ, chemokine ligand 5 (CCL-5), IL-6, IL-8, and IL-18 gene expression. The results have shown that SAS-CoV-2 was found in heart tissue and immune reaction was observed in cases with higher virus load [123].
The study that investigated the pathological features of COVID-19 in the body of two dead cases have suggested that major tissue manifestations are bleeding and exudation infiltration of macrophages, epithelium injuries, and lung fibrosis. Also, the activation of alveolar macrophages was obvious [124].
Collectively, based on existing data, macrophages are considered the chief promoters of the cytokine storm during the infection of SARS-CoV-2 that potentially cause the enhancement of its pathogenesis and increase the number of COVID-19 sever cases which most probably lead to death [48]. Therefore, the blockage of macrophage activation pathways could be an effective therapeutic procedure, and finding about the role of macrophages in coronavirus pathogenesis can help to have a better perspective for the treatment of this complicated viral disease [8,125].
After SARS-CoV-2 infection, the cytokines are released and lymphocytosis (lymphopenia occurs especially in severe patients) happens as immune system responses that are triggered by dendritic cells, and macrophages. Cytokine storm damages lung cells and also other organs such as the kidney, heart, blood vessels and brain [126]. Cytokine storm pathological mechanisms are illustrated in Fig. 2 .
Fig. 2.
Cytokine storm damage mechanisms and new pharmaceutical interventions. Due to the attachment of SARS-CoV-2 to ACE2 in epithelial cells, the hyperinflammatory response is stimulated and epithelial cell-mediated reactive oxygen species (ROS) are produced that lead to cell death. Moreover, ROS can induce NF-Kβ and NLRP3 production which are important to trigger cytokine storm. Cytokine storm leads to some clinical condition such as sepsis, ARDS, MOF, and death. Some new clinical interventions like Emodin and Camostat mesilate have attachment prevention activity of SARS-CoV-2 to ACE2 and TMPRSS2. Flavonoid compounds modulate NLRP3 activity. Tocilizumab and GM-CSF blockers can block IL-6 and GM-CSF, respectively. NRF-2 activator drugs downregulate IL-6 and IL-1β in LPS stimulated macrophages [126]. SARS-CoV-2, severe acute respiratory syndrome 2, ROS, reactive oxygen species, MOF, multiple organ failure, PAMP, pathogen associated molecular pattern, DAMP, damaged associated molecular pattern, TNF, tumor necrosis factor, PRR, pattern recognition receptor, IL, interleukin, NLRP3, (NOD)like receptor protein 3 inflammasome, NFkβ, nuclear factor kappa B-light chain enhancer of activated B cells, NRF2, nuclear factor erythroid 2 p45-related factors 2 (NRF2), GM-CSF, granulocyte-macrophage colony-stimulating factor.
1.5. Pharmacological interventions of macrophages in COVID-19
Because of the described features of macrophages to trigger the SARS-CoV-2 infection, scientists are exploring the efficient procedures for regulating macrophage activity in order to suppress their impressive effects on the pathogenesis of COVID-19 and the efforts in this regard have been somewhat successful [59].
Some studies were reported that during the SARS-CoV infection, dysregulated type I interferon response occurred and the most notable agents of the immunopathology of the virus were the inflammatory phenotypes of macrophages and monocytes accumulation. By this argument, macrophages are known as an effective weapon to use for target therapy procedures in severe cases of COVID-19 [127,128]. Analyzing CRS carried out after antigen receptor-modified T (CAR T) therapy was also helpful to reveal the substantial role of monocytes and macrophages in CRS. Related studies have been identified that monocytes could drive the CRS process mainly through IL-1and IL-6 production, hence depletion of monocytes, IL-1, L-6, and macrophages led to limit CRS and cytokine storm in the different studies [[129], [130], [131]]. Understanding multiple pathophysiological pathways associated with cytokine storm could be a helpful way to design targeted therapeutical approaches and novel pharmacological interventions for severe COVID-19 cases to decline complications such as MOF and ARDS resulted from cytokine storm and led to serious tissue damage in patients (Fig. 2).
The important cytokines involved in the activation of monocytes and inducing their differentiation into the macrophages are IL-6 and GM-CSF. These cytokines are detected as increased factors in the severe patients of COVID-19 that have a fundamental role in the contribution of an inflammatory condition included cytokine storm [59,119]. So, they could be a suitable therapeutic target for blocking cytokine storm-related pathways [132]. Evidence indicated the successful usage of Tocilizumab as an IL-6 blocking monoclonal antibody to diminish immunopathogenesis of SARS-CoV-2 infection via suppressing cytokine storm [2,133]. Also, it has been shown that blocking GM-CSF is a beneficial method for restraining the CRS and their fallowing neuroinflammatory disorder [134]. Several pre-clinical types of research and clinical trial studies have suggested the inhibiting action of GM-CSF as a promising therapeutic approach to attenuate inflammatory-resulted complications [135,136]. Therefore, it has a good potential to be a candidate drug for ARDS treatment of patients with SARS-CoV-2 [59,137]. Furthermore, based on the different reports, IL-6 and TNF-α blockers have been suggested as possible drugs to attenuate the severity of outcome in COVID-19 patients [138,139]. Another pharmaceutical intervention that can be utilized to the improvement of SARS-CoV-2 complications is the therapy based on nuclear factor erythroid 2 p45-related factors 2 (NRF2) activity [140]. NRF2 is known as a pivotal transcription factor that plays a part in the regulation of antioxidant proteins expression to decline oxidative damages resulted from inflammation [141]. Besides, it is capable of downregulating proinflammatory genes such as IL1B and IL6. This is considered in lipopolysaccharide (LPS)-stimulated macrophage cells, which their accumulated anti-inflammatory immunometabolite itaconate can activate NRF2. Indeed, the remedial effect of activated NRF2 has been demonstrated in several animal studies on respiratory infection and ARDS. Also, the good potential of some NRF2 activator drugs including sulforaphane and bardoxolone methyl has been revealed to fight toward SARS-CoV-2 infection in several pre-clinical models [140,142,143].
Camostat mesilate is one of the transmembrane protease serine 2 (TMPRSS2) inhibitors, known as a therapeutic tool that has been introduced as a candidate to protect body against COVID-19 by preventing attachment of S protein to the ACE2 receptor on the surface of cells such as macrophages. Therefore, it may be helpful to the immunopathogenesis of coronavirus. It is noteworthy that TMPRSS2 is the host serine protease that facilitate the S proteins binding to the ACE2 receptors [6,144,145].
Emodin (6-methyl-1,3,8-trihydroxyanthraquinone) (CAS number: 518−82-1) has been reported as another possible inhibitor of SARS-CoV-2 S protein and ACE2 receptor interaction. It is found in Chinese traditional medicine and is an anthraquinone natural product isolated from many genera of fungi such as Aspergillus, Pyrenochaeta, and Pestalotiopsis. Emodin could exhibit its effects in a dose-dependent manner [146]. Also, Chinese scientists have introduced a pharmaceutical capsule called Lianhua Qingwen with the capability of reducing macrophage and DC related inflammation through inhibition of HTR7, CDK6, and ALOX15 in 7%, 14%, and 32% patients, respectively. Therefore, it may be a potential drug to diminish SARS-CoV-2 infection by regulating macrophage activity [147].
It has been established that one of the causes of enhanced inflammatory cytokines during the ARDS in COVID-19 patients is the NLRP3 inflammasome activation in induced T helper-1 lymphocytes and macrophages. Based on this, several studies have been focused on modulating NLRP3 inflammasome activity by using flavonoid compounds [148,149] including luteolin, myricetin, apigenin, quercetin, kaempferol, baicalin, and wogonoside. These drugs have been shown to be effective in the regulation of macrophage associated inflammatory response in the infection of SARS-CoV-2 [145].
In regards to pharmacological intervention for the treatment of COVID-19, novel therapeutical methods by mesenchymal stem cells (MSCs) derived exosomes have attracted the attention of many researchers. MSCs are known as multipotent stem cells with a wide range of properties such as tissue-regenerative ability, anti-inflammatory, and anti-microbial attributions. MSCs are able to produce IL-10, vascular endothelial growth factor (VEGF), hepatocyte growth factor, and keratinocytes growth factors that are helpful to ameliorate ARDS and lung injuries improvement [54]. During the last decades, exosomes are recognized as beneficial therapeutic tools for drug delivery. The MSCs derived exosomes can replicate beneficial effects of them after MSC phagocytosis by tissue-resident macrophages. Due to the immunomodulatory activity of MSCs, they are able to inhibit the activation of macrophages and derive their differentiation into the anti-inflammatory phenotypes in favor of suppressing inflammation. Hence, inducing upregulation of cytokines such as the transforming growth factor beta (TGF-β) and IL-10 can evacuate the lungs from detrimental macrophages and prevent DC-associated systemic immune responses through regulating both phenotype and function of lung infiltrating DCs. Therefore, all of the above-mentioned properties of MSCs derived exosomes can make them popular candidates with unique therapeutic aspects for the treatment of SARS-CoV-2 related pathogenicity [54,150].
2. Conclusion
In summary, monocytes and macrophages are pivotal innate immune cells with a wide range of defensive activities against various antimicrobial agents such as viruses. Besides their beneficial activities, it has been established that they exert unfavorable effects on the host in some of the viral infections such as COVID-19 caused by SARS-CoV-2, which has been considered nowadays as a life-threatening emergency to human health. Investigation of many studies has revealed that monocytes and macrophages can induce pathogenesis during the COVID-19 through dysregulated function including enhancement of inflammatory cytokines and chemokines, excessive interaction with adaptive immune cells, and their products that ultimately lead to serious tissue damage and enhancement of clinical manifestation of the disease. Therefore, discovering involved mechanisms and mediators can be significantly helpful to develop novel therapeutic methods and cell-based drugs for alleviating SARS-CoV-2 infection, but its hazardous complications require further researches in the future.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data of material
All data generated or analyzed during this study are included in this published article.
Funding
Not applicable.
Declaration of competing interest
None.
Acknowledgments
Acknowledgement
None.
References
- 1.Saxena S.K. Springer Nature; 2020. Coronavirus Disease 2019 (COVID-19): Epidemiology, Pathogenesis, Diagnosis, and Therapeutics. [Google Scholar]
- 2.Jin Y., et al. Virology, epidemiology, pathogenesis, and control of COVID-19. Viruses. 2020;12(4):372. doi: 10.3390/v12040372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McIntosh K., Hirsch M.S., Bloom A. Coronavirus disease 2019 (COVID-19) UpToDate Hirsch MS Bloom. 2020:5. [Google Scholar]
- 4.Duerr G., et al. Parameters predicting COVID-19-induced myocardial injury and mortality. Life Sci. 2020;260:118400. doi: 10.1016/j.lfs.2020.118400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li W., et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoffmann M., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Suzuki T., Chow C.-W., Downey G.P. Role of innate immune cells and their products in lung immunopathology. Int. J. Biochem. Cell Biol. 2008;40(6–7):1348–1361. doi: 10.1016/j.biocel.2008.01.003. [DOI] [PubMed] [Google Scholar]
- 8.Merad M., Martin J.C. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat. Rev. Immunol. 2020:1–8. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schulte-Schrepping J., et al. Severe COVID-19 is marked by a dysregulated myeloid cell compartment. Cell. 2020;182(6):1419–1440. doi: 10.1016/j.cell.2020.08.001. (e23) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McKechnie J.L., Blish C.A. The innate immune system: fighting on the front lines or fanning the flames of COVID-19? Cell Host Microbe. 2020;27:863–869. doi: 10.1016/j.chom.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Auffray C., Sieweke M.H., Geissmann F. Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Annu. Rev. Immunol. 2009;27 doi: 10.1146/annurev.immunol.021908.132557. [DOI] [PubMed] [Google Scholar]
- 12.Boyette L.B., et al. Phenotype, function, and differentiation potential of human monocyte subsets. PLoS One. 2017;12(4) doi: 10.1371/journal.pone.0176460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ingersoll M.A., et al. Comparison of gene expression profiles between human and mouse monocyte subsets. Blood, The Journal of the American Society of Hematology. 2010;115(3):e10–e19. doi: 10.1182/blood-2009-07-235028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Misharin A.V., et al. Monocyte-derived alveolar macrophages drive lung fibrosis and persist in the lung over the life span. J. Exp. Med. 2017;214(8):2387–2404. doi: 10.1084/jem.20162152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shi C., Pamer E.G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011;11(11):762–774. doi: 10.1038/nri3070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arango Duque G., Descoteaux A. Macrophage cytokines: involvement in immunity and infectious diseases. Front. Immunol. 2014;5:491. doi: 10.3389/fimmu.2014.00491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tosi M.F. Innate immune responses to infection. J. Allergy Clin. Immunol. 2005;116(2):241–249. doi: 10.1016/j.jaci.2005.05.036. [DOI] [PubMed] [Google Scholar]
- 18.Crowell R., et al. Alveolar nd interstitial macrophage populations in the murine lung. Exp. Lung Res. 1992;18(4):435–446. doi: 10.3109/01902149209064338. [DOI] [PubMed] [Google Scholar]
- 19.McCubbrey A.L., et al. Promoter specificity and efficacy in conditional and inducible transgenic targeting of lung macrophages. Front. Immunol. 2017;8:1618. doi: 10.3389/fimmu.2017.01618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fels A., Cohn Z. The alveolar macrophage. J. Appl. Physiol. 1986;60(2):353–369. doi: 10.1152/jappl.1986.60.2.353. [DOI] [PubMed] [Google Scholar]
- 21.Brieland J.K., Kunkel R.G., Fantone J.C. Pulmonary alveolar macrophage function during acute inflammatory lung injury. Am. Rev. Respir. Dis. 1987;135(6):1300–1306. doi: 10.1164/arrd.1987.135.6.1300. [DOI] [PubMed] [Google Scholar]
- 22.Dunbar P.R., et al. Pulmonary monocytes interact with effector T cells in the lung tissue to drive T RM differentiation following viral infection. Mucosal Immunol. 2020;13(1):161–171. doi: 10.1038/s41385-019-0224-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Barbalat R., et al. Toll-like receptor 2 on inflammatory monocytes induces type I interferon in response to viral but not bacterial ligands. Nat. Immunol. 2009;10(11):1200–1207. doi: 10.1038/ni.1792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Masson F., et al. Dendritic cells: driving the differentiation programme of T cells in viral infections. Immunol. Cell Biol. 2008;86(4):333–342. doi: 10.1038/icb.2008.15. [DOI] [PubMed] [Google Scholar]
- 25.Dawson T.C., et al. Contrasting effects of CCR5 and CCR2 deficiency in the pulmonary inflammatory response to influenza A virus. Am. J. Pathol. 2000;156(6):1951–1959. doi: 10.1016/S0002-9440(10)65068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Deeks S.G., Lewin S.R., Havlir D.V. The end of AIDS: HIV infection as a chronic disease. Lancet. 2013;382(9903):1525–1533. doi: 10.1016/S0140-6736(13)61809-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kedzierska K., Crowe S.M. The role of monocytes and macrophages in the pathogenesis of HIV-1 infection. Curr. Med. Chem. 2002;9(21):1893–1903. doi: 10.2174/0929867023368935. [DOI] [PubMed] [Google Scholar]
- 28.Kozak S.L., et al. CD4, CXCR-4, and CCR-5 dependencies for infections by primary patient and laboratory-adapted isolates of human immunodeficiency virus type 1. J. Virol. 1997;71(2):873–882. doi: 10.1128/jvi.71.2.873-882.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Platt E.J., et al. Effects of CCR5 and CD4 cell surface concentrations on infections by macrophagetropic isolates of human immunodeficiency virus type 1. J. Virol. 1998;72(4):2855–2864. doi: 10.1128/jvi.72.4.2855-2864.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Naif H.M., et al. Definition of the stage of host cell genetic restriction of replication of human immunodeficiency virus type 1 in monocytes and monocyte-derived macrophages by using twins. J. Virol. 1999;73(6):4866–4881. doi: 10.1128/jvi.73.6.4866-4881.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fear W.R., et al. Differential tropism and chemokine receptor expression of human immunodeficiency virus type 1 in neonatal monocytes, monocyte-derived macrophages, and placental macrophages. J. Virol. 1998;72(2):1334–1344. doi: 10.1128/jvi.72.2.1334-1344.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sonza S., et al. Human immunodeficiency virus type 1 replication is blocked prior to reverse transcription and integration in freshly isolated peripheral blood monocytes. J. Virol. 1996;70(6):3863–3869. doi: 10.1128/jvi.70.6.3863-3869.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hufert F.T., et al. Human Kupffer cells infected with HIV-1 in vivo. J. Acquir. Immune Defic. Syndr. 1993;6(7):772–777. [PubMed] [Google Scholar]
- 34.Lassmann H., et al. Bone marrow derived elements and resident microglia in brain inflammation. Glia. 1993;7(1):19–24. doi: 10.1002/glia.440070106. [DOI] [PubMed] [Google Scholar]
- 35.García F., et al. Cerebrospinal fluid HIV-1 RNA levels in asymptomatic patients with early stage chronic HIV-1 infection: support for the hypothesis of local virus replication. Aids. 1999;13(12):1491–1496. doi: 10.1097/00002030-199908200-00008. [DOI] [PubMed] [Google Scholar]
- 36.Liuzzi G., et al. Analysis of HIV-1 load in blood, semen and saliva: evidence for different viral compartments in a cross-sectional and longitudinal study. Aids. 1996;10(14):F51–F56. doi: 10.1097/00002030-199612000-00001. [DOI] [PubMed] [Google Scholar]
- 37.Glass J.D., et al. Clinical-neuropathologic correlation in HIV-associated dementia. Neurology. 1993;43(11):2230. doi: 10.1212/WNL.43.11.2230. [DOI] [PubMed] [Google Scholar]
- 38.Soukup J.M., Becker S. Role of monocytes and eosinophils in human respiratory syncytial virus infection in vitro. Clin. Immunol. 2003;107(3):178–185. doi: 10.1016/s1521-6616(03)00038-x. [DOI] [PubMed] [Google Scholar]
- 39.Bandeira-Melo C., et al. IL-16 promotes leukotriene C4 and IL-4 release from human eosinophils via CD4-and autocrine CCR3-chemokine-mediated signaling. J. Immunol. 2002;168(9):4756–4763. doi: 10.4049/jimmunol.168.9.4756. [DOI] [PubMed] [Google Scholar]
- 40.Holt P., Stumbles P. Characterization of dendritic cell populations in the respiratory tract. Journal of Aerosol Medicine. 2000;13(4):361–367. doi: 10.1089/jam.2000.13.361. [DOI] [PubMed] [Google Scholar]
- 41.Normile D. American Association for the Advancement of Science; 2013. Surprising New Dengue Virus Throws a Spanner in Disease Control Efforts. [DOI] [PubMed] [Google Scholar]
- 42.Singla M., et al. Immune response to dengue virus infection in pediatric patients in New Delhi, India—association of viremia, inflammatory mediators and monocytes with disease severity. PLoS Negl. Trop. Dis. 2016;10(3) doi: 10.1371/journal.pntd.0004497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Na W., et al. Ebola outbreak in Western Africa 2014: what is going on with Ebola virus? Clinical and experimental vaccine research. 2015;4(1):17–22. doi: 10.7774/cevr.2015.4.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Menicucci A.R., et al. Transcriptome analysis of circulating immune cell subsets highlight the role of monocytes in Zaire Ebola virus Makona pathogenesis. Front. Immunol. 2017;8:1372. doi: 10.3389/fimmu.2017.01372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ströher U., et al. Infection and activation of monocytes by Marburg and Ebola viruses. J. Virol. 2001;75(22):11025–11033. doi: 10.1128/JVI.75.22.11025-11033.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Parihar A., Eubank T.D., Doseff A.I. Monocytes and macrophages regulate immunity through dynamic networks of survival and cell death. Journal of innate immunity. 2010;2(3):204–215. doi: 10.1159/000296507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pence B.D. Severe COVID-19 and aging: are monocytes the key? GeroScience. 2020;42(4):1051–1061. doi: 10.1007/s11357-020-00213-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wu A., et al. Genome composition and divergence of the novel coronavirus (2019-nCoV) originating in China. Cell Host Microbe. 2020;27:325–328. doi: 10.1016/j.chom.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paliogiannis P., et al. Laboratory test alterations in patients with COVID-19 and non COVID-19 interstitial pneumonia: a preliminary report. The Journal of Infection in Developing Countries. 2020;14(07):685–690. doi: 10.3855/jidc.12879. [DOI] [PubMed] [Google Scholar]
- 50.Zhou Y., et al. BioRxiv; 2020. Aberrant Pathogenic GM-CSF+ T Cells and Inflammatory CD14+ CD16+ Monocytes in Severe Pulmonary Syndrome Patients of a New Coronavirus. [Google Scholar]
- 51.Henry B.M., et al. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clinical Chemistry and Laboratory Medicine (CCLM) 2020;58(7):1021–1028. doi: 10.1515/cclm-2020-0369. [DOI] [PubMed] [Google Scholar]
- 52.Andonegui-Elguera S., et al. Molecular alterations prompted by SARS-CoV-2 infection: induction of hyaluronan, glycosaminoglycan and mucopolysaccharide metabolism. Arch. Med. Res. 2020;51:645–653. doi: 10.1016/j.arcmed.2020.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wen W., et al. Immune cell profiling of COVID-19 patients in the recovery stage by single-cell sequencing. Cell discovery. 2020;6(1):1–18. doi: 10.1038/s41421-020-0168-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Guo C., et al. Single-cell analysis of two severe COVID-19 patients reveals a monocyte-associated and tocilizumab-responding cytokine storm. Nat. Commun. 2020;11(1):1–11. doi: 10.1038/s41467-020-17834-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jafarzadeh A., et al. Contribution of monocytes and macrophages to the local tissue inflammation and cytokine storm in COVID-19: lessons from SARS and MERS, and potential therapeutic interventions. Life Sci. 2020;257:118102. doi: 10.1016/j.lfs.2020.118102. 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhang J.-j., et al. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. 2020;75:1730–1741. doi: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- 57.Verdecchia P., et al. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. European Journal of Internal Medicine. 2020;76:14–20. doi: 10.1016/j.ejim.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Giamarellos-Bourboulis E.J., et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27:992–1000.e3. doi: 10.1016/j.chom.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gómez-Rial J., et al. Role of monocytes/macrophages in covid-19 pathogenesis: implications for therapy. Infection and Drug Resistance. 2020;13:2485. doi: 10.2147/IDR.S258639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thepen T., Van Rooijen N., Kraal G. Alveolar macrophage elimination in vivo is associated with an increase in pulmonary immune response in mice. J. Exp. Med. 1989;170(2):499–509. doi: 10.1084/jem.170.2.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hu G., Christman J.W. Alveolar macrophages in lung inflammation and resolution. Front. Immunol. 2019;10:2275. doi: 10.3389/fimmu.2019.02275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hussell T., Bell T.J. Alveolar macrophages: plasticity in a tissue-specific context. Nat. Rev. Immunol. 2014;14(2):81–93. doi: 10.1038/nri3600. [DOI] [PubMed] [Google Scholar]
- 63.Misharin A.V., Scott Budinger G., Perlman H. American Thoracic Society; 2011. The Lung Macrophage: A Jack of All Trades. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gibbings S.L., et al. Three unique interstitial macrophages in the murine lung at steady state. Am. J. Respir. Cell Mol. Biol. 2017;57(1):66–76. doi: 10.1165/rcmb.2016-0361OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Abt M.C., et al. Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity. 2012;37(1):158–170. doi: 10.1016/j.immuni.2012.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Viola A., et al. The metabolic signature of macrophage responses. Front. Immunol. 2019:10. doi: 10.3389/fimmu.2019.01462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bain C.C., et al. Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat. Immunol. 2014;15(10):929–937. doi: 10.1038/ni.2967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hashimoto D., et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity. 2013;38(4):792–804. doi: 10.1016/j.immuni.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hong M., et al. Protective role of γδ T cells in cigarette smoke and influenza infection. Mucosal Immunol. 2018;11(3):894–908. doi: 10.1038/mi.2017.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Joshi N., Walter J.M., Misharin A.V. Alveolar macrophages. Cell. Immunol. 2018;330:86–90. doi: 10.1016/j.cellimm.2018.01.005. [DOI] [PubMed] [Google Scholar]
- 71.Xue J., et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity. 2014;40(2):274–288. doi: 10.1016/j.immuni.2014.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wynn T.A., Vannella K.M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity. 2016;44(3):450–462. doi: 10.1016/j.immuni.2016.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lavin Y., et al. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell. 2014;159(6):1312–1326. doi: 10.1016/j.cell.2014.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mosser D.M., Edwards J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008;8(12):958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Duffield J.S., et al. Host responses in tissue repair and fibrosis. Annual Review of Pathology: Mechanisms of Disease. 2013;8:241–276. doi: 10.1146/annurev-pathol-020712-163930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Eguíluz-Gracia I., et al. Long-term persistence of human donor alveolar macrophages in lung transplant recipients. Thorax. 2016;71(11):1006–1011. doi: 10.1136/thoraxjnl-2016-208292. [DOI] [PubMed] [Google Scholar]
- 77.Gordon S., Taylor P.R. Monocyte and macrophage heterogeneity. Nat. Rev. Immunol. 2005;5(12):953–964. doi: 10.1038/nri1733. [DOI] [PubMed] [Google Scholar]
- 78.Divangahi M., King I.L., Pernet E. Alveolar macrophages and type I IFN in airway homeostasis and immunity. Trends Immunol. 2015;36(5):307–314. doi: 10.1016/j.it.2015.03.005. [DOI] [PubMed] [Google Scholar]
- 79.Davies L.C., et al. Tissue-resident macrophages. Nat. Immunol. 2013;14(10):986. doi: 10.1038/ni.2705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Marsland B.J., et al. Nippostrongylus brasiliensis infection leads to the development of emphysema associated with the induction of alternatively activated macrophages. Eur. J. Immunol. 2008;38(2):479–488. doi: 10.1002/eji.200737827. [DOI] [PubMed] [Google Scholar]
- 81.Herold S., Mayer K., Lohmeyer J. Acute lung injury: how macrophages orchestrate resolution of inflammation and tissue repair. Front. Immunol. 2011;2:65. doi: 10.3389/fimmu.2011.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hocking W.G., Golde D.W. The pulmonary-alveolar macrophage. N. Engl. J. Med. 1979;301(12):639–645. doi: 10.1056/NEJM197909203011205. [DOI] [PubMed] [Google Scholar]
- 83.Byrne A.J., et al. Pulmonary macrophages: key players in the innate defence of the airways. Thorax. 2015;70(12):1189–1196. doi: 10.1136/thoraxjnl-2015-207020. [DOI] [PubMed] [Google Scholar]
- 84.Mogensen S.C. Role of macrophages in natural resistance to virus infections. Microbiol. Rev. 1979;43(1):1. doi: 10.1128/mr.43.1.1-26.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Epstein L.B., Stevens D.A., Merigan T.C. Selective increase in lymphocyte interferon response to vaccinia antigen after revaccination. Proc. Natl. Acad. Sci. 1972;69(9):2632–2636. doi: 10.1073/pnas.69.9.2632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Unanue E. Advances in Immunology. Elsevier; 1972. The regulatory role of macrophages in antigenic stimulation; pp. 95–165. [DOI] [PubMed] [Google Scholar]
- 87.Rossi G., et al. The role of macrophages in interstitial lung diseases: number 3 in the series “pathology for the clinician” edited by Peter Dorfmüller and Alberto Cavazza. Eur. Respir. Rev. 2017;26(145) doi: 10.1183/16000617.0009-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Bouchery T., et al. ILC2s and T cells cooperate to ensure maintenance of M2 macrophages for lung immunity against hookworms. Nat. Commun. 2015;6(1):1–13. doi: 10.1038/ncomms7970. [DOI] [PubMed] [Google Scholar]
- 89.Pouniotis D., et al. Alveolar macrophage function is altered in patients with lung cancer. Clinical & Experimental Immunology. 2006;143(2):363–372. doi: 10.1111/j.1365-2249.2006.02998.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Van Erp E.A., et al. Fc-mediated antibody effector functions during respiratory syncytial virus infection and disease. Front. Immunol. 2019;10:548. doi: 10.3389/fimmu.2019.00548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Smyth M.J., et al. Activation of NK cell cytotoxicity. Mol. Immunol. 2005;42(4):501–510. doi: 10.1016/j.molimm.2004.07.034. [DOI] [PubMed] [Google Scholar]
- 92.Yasui F., et al. Phagocytic cells contribute to the antibody-mediated elimination of pulmonary-infected SARS coronavirus. Virology. 2014;454:157–168. doi: 10.1016/j.virol.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mehta P., et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet (London, England) 2020;395(10229):1033. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jafarzadeh A., et al. 2015. Human Leukocyte Antigens Influence the Antibody Response to Hepatitis B Vaccine. [PubMed] [Google Scholar]
- 95.Kadkhoda K. COVID-19: an Immunopathological view. Msphere. 2020;5(2) doi: 10.1128/mSphere.00344-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Weiss R.C., Scott F.W. Antibody-mediated enhancement of disease in feline infectious peritonitis: comparisons with dengue hemorrhagic fever. Comp. Immunol. Microbiol. Infect. Dis. 1981;4(2):175–189. doi: 10.1016/0147-9571(81)90003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yip M.S., et al. Antibody-dependent infection of human macrophages by severe acute respiratory syndrome coronavirus. Virol. J. 2014;11(1):1–11. doi: 10.1186/1743-422X-11-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Hohdatsu T., et al. Antibody-dependent enhancement of feline infectious peritonitis virus infection in feline alveolar macrophages and human monocyte cell line U937 by serum of cats experimentally or naturally infected with feline coronavirus. J. Vet. Med. Sci. 1998;60(1):49–55. doi: 10.1292/jvms.60.49. [DOI] [PubMed] [Google Scholar]
- 99.Jaume M., et al. Anti-severe acute respiratory syndrome coronavirus spike antibodies trigger infection of human immune cells via a pH-and cysteine protease-independent FcγR pathway. J. Virol. 2011;85(20):10582–10597. doi: 10.1128/JVI.00671-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fu Y., Cheng Y., Wu Y. Understanding SARS-CoV-2-mediated inflammatory responses: from mechanisms to potential therapeutic tools. Virol. Sin. 2020:1–6. doi: 10.1007/s12250-020-00207-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu L., et al. Anti–spike IgG causes severe acute lung injury by skewing macrophage responses during acute SARS-CoV infection. JCI insight. 2019;4(4) doi: 10.1172/jci.insight.123158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tseng C.-T., et al. Immunization with SARS coronavirus vaccines leads to pulmonary immunopathology on challenge with the SARS virus. PLoS One. 2012;7(4):e35421. doi: 10.1371/journal.pone.0035421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bolles M., et al. A double-inactivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge. J. Virol. 2011;85(23):12201–12215. doi: 10.1128/JVI.06048-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Peiris J.S.M., et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361(9371):1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zhang L., et al. Antibody responses against SARS coronavirus are correlated with disease outcome of infected individuals. J. Med. Virol. 2006;78(1):1–8. doi: 10.1002/jmv.20499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ye Q., Wang B., Mao J. Cytokine storm in COVID-19 and treatment. J. Infect. 2020;80:607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Farsalinos K., Barbouni A., Niaura R. Systematic review of the prevalence of current smoking among hospitalized COVID-19 patients in China: could nicotine be a therapeutic option? Intern. Emerg. Med. 2020:1–8. doi: 10.1007/s11739-020-02355-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wong J.P., et al. Current and future developments in the treatment of virus-induced hypercytokinemia. Future Med. Chem. 2017;9(2):169–178. doi: 10.4155/fmc-2016-0181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu Q., Zhou Y.-h., Yang Z.-q. The cytokine storm of severe influenza and development of immunomodulatory therapy. Cellular & molecular immunology. 2016;13(1):3–10. doi: 10.1038/cmi.2015.74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Tisoncik J.R., et al. Into the eye of the cytokine storm. Microbiol. Mol. Biol. Rev. 2012;76(1):16–32. doi: 10.1128/MMBR.05015-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Shimabukuro-Vornhagen A., et al. Cytokine release syndrome. Journal for immunotherapy of cancer. 2018;6(1):56. doi: 10.1186/s40425-018-0343-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lee D.W., et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124(2):188–195. doi: 10.1182/blood-2014-05-552729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wiersinga W.J., et al. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. Jama. 2020;324(8):782–793. doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- 114.Inandiklioglu N., Akkoc T. Immune responses to SARS-CoV, MERS-CoV and SARS-CoV-2. Adv. Exp. Med. Biol. 2020;9:5–12. doi: 10.1007/5584_2020_549. [DOI] [PubMed] [Google Scholar]
- 115.Jamilloux Y., et al. Should we stimulate or suppress immune responses in COVID-19? Cytokine and anti-cytokine interventions. Autoimmun. Rev. 2020;19:102567. doi: 10.1016/j.autrev.2020.102567. 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Eskilsson A., et al. Immune-induced fever is mediated by IL-6 receptors on brain endothelial cells coupled to STAT3-dependent induction of brain endothelial prostaglandin synthesis. J. Neurosci. 2014;34(48):15957–15961. doi: 10.1523/JNEUROSCI.3520-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ravelli A. Macrophage activation syndrome. Curr. Opin. Rheumatol. 2002;14(5):548–552. doi: 10.1097/00002281-200209000-00012. [DOI] [PubMed] [Google Scholar]
- 118.Lerkvaleekul B., Vilaiyuk S. Macrophage activation syndrome: early diagnosis is key. Open access rheumatology: research and reviews. 2018;10:117. doi: 10.2147/OARRR.S151013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Yang X., et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir. Med. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.McGonagle D., et al. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. The Lancet Rheumatology. 2020;2:e437–e445. doi: 10.1016/S2665-9913(20)30121-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Alexander, J., et al., Diffuse Myocardial Inflammation in COVID-19 Associated Myocarditis Detected by Multiparametric Cardiac Magnetic Resonance Imaging. [DOI] [PubMed]
- 122.Sorokin A.V., et al. COVID-19—associated dyslipidemia: implications for mechanism of impaired resolution and novel therapeutic approaches. FASEB J. 2020;34(8):9843–9853. doi: 10.1096/fj.202001451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lindner D., et al. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020;5(11):1281–1285. doi: 10.1001/jamacardio.2020.3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wang C., et al. Alveolar macrophage dysfunction and cytokine storm in the pathogenesis of two severe COVID-19 patients. EBioMedicine. 2020;57:102833. doi: 10.1016/j.ebiom.2020.102833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.McGonagle D., et al. Interleukin-6 use in COVID-19 pneumonia related macrophage activation syndrome. Autoimmun. Rev. 2020:102537. doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bhaskar S., et al. Cytokine storm in COVID-19—immunopathological mechanisms, clinical considerations, and therapeutic approaches: the REPROGRAM Consortium position paper. Front. Immunol. 2020;11:1648. doi: 10.3389/fimmu.2020.01648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Channappanavar R., et al. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice. Cell Host Microbe. 2016;19(2):181–193. doi: 10.1016/j.chom.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Cheung C.Y., et al. Cytokine responses in severe acute respiratory syndrome coronavirus-infected macrophages in vitro: possible relevance to pathogenesis. J. Virol. 2005;79(12):7819–7826. doi: 10.1128/JVI.79.12.7819-7826.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Norelli M., et al. Monocyte-derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nat. Med. 2018;24(6):739–748. doi: 10.1038/s41591-018-0036-4. [DOI] [PubMed] [Google Scholar]
- 130.Giavridis T., et al. CAR T cell–induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade. Nat. Med. 2018;24(6):731–738. doi: 10.1038/s41591-018-0041-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sumaili H., et al. Outcome of pediatric patients with lymphoma following stem cell transplant: a single institution report. Leukemia & Lymphoma. 2015;56(5):1327–1334. doi: 10.3109/10428194.2014.951846. [DOI] [PubMed] [Google Scholar]
- 132.J Gómez-Rial M.-T. A strategy targeting monocyte-macrophage differentiation to avoid pulmonary complications in SARS-Cov2 infection. Clinical Immunology (Orlando, Fla.) 2020;216:108442. doi: 10.1016/j.clim.2020.108442. 1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Crisafulli S., et al. Potential role of anti-interleukin (IL)-6 drugs in the treatment of COVID-19: rationale, clinical evidence and risks. BioDrugs. 2020;34(4):415–422. doi: 10.1007/s40259-020-00430-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Sterner R.M., et al. GM-CSF inhibition reduces cytokine release syndrome and neuroinflammation but enhances CAR-T cell function in xenografts. Blood, The Journal of the American Society of Hematology. 2019;133(7):697–709. doi: 10.1182/blood-2018-10-881722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lacey D.C., et al. Defining GM-CSF–and macrophage-CSF–dependent macrophage responses by in vitro models. J. Immunol. 2012;188(11):5752–5765. doi: 10.4049/jimmunol.1103426. [DOI] [PubMed] [Google Scholar]
- 136.Hamilton J.A. GM-CSF-dependent inflammatory pathways. Front. Immunol. 2019;10:2055. doi: 10.3389/fimmu.2019.02055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Toniati P., et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia, Italy. Autoimmun. Rev. 2020:102568. doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Messina G., et al. Functional role of dietary intervention to improve the outcome of COVID-19: a hypothesis of work. Int. J. Mol. Sci. 2020;21(9):3104. doi: 10.3390/ijms21093104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Russell B., et al. Associations between immune-suppressive and stimulating drugs and novel COVID-19—a systematic review of current evidence. ecancermedicalscience. 2020;14 doi: 10.3332/ecancer.2020.1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Cuadrado A., et al. Can activation of NRF2 be a strategy against COVID-19? Trends Pharmacol. Sci. 2020;41:598–610. doi: 10.1016/j.tips.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Otsuki A., Yamamoto M. Cis-element architecture of Nrf2–sMaf heterodimer binding sites and its relation to diseases. Arch. Pharm. Res. 2020:1–11. doi: 10.1007/s12272-019-01193-2. [DOI] [PubMed] [Google Scholar]
- 142.Cuadrado A., et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019;18(4):295–317. doi: 10.1038/s41573-018-0008-x. [DOI] [PubMed] [Google Scholar]
- 143.Rabbani P.S., et al. Dysregulation of Nrf2/Keap1 redox pathway in diabetes affects multipotency of stromal cells. Diabetes. 2019;68(1):141–155. doi: 10.2337/db18-0232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Báez-Santos Y.M., John S.E.S., Mesecar A.D. The SARS-coronavirus papain-like protease: structure, function and inhibition by designed antiviral compounds. Antivir. Res. 2015;115:21–38. doi: 10.1016/j.antiviral.2014.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.McKee D.L., et al. Candidate drugs against SARS-CoV-2 and COVID-19. Pharmacol. Res. 2020:104859. doi: 10.1016/j.phrs.2020.104859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ho T.-Y., et al. Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2007;74(2):92–101. doi: 10.1016/j.antiviral.2006.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Wang Y., et al. The pros and cons of traditional Chinese medicines in the treatment of COVID-19. Pharmacol. Res. 2020;157:104873. doi: 10.1016/j.phrs.2020.104873. 1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Lim H., et al. Flavonoids interfere with NLRP3 inflammasome activation. Toxicol. Appl. Pharmacol. 2018;355:93–102. doi: 10.1016/j.taap.2018.06.022. [DOI] [PubMed] [Google Scholar]
- 149.Chen I.-Y., et al. Severe acute respiratory syndrome coronavirus viroporin 3a activates the NLRP3 inflammasome. Front. Microbiol. 2019;10:50. doi: 10.3389/fmicb.2019.00050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Gupta S., et al. Mesenchymal stem cell derived exosomes: a nano platform for therapeutics and drug delivery in combating COVID-19. Stem Cell Rev. Rep. 2020:1–11. doi: 10.1007/s12015-020-10002-z. [DOI] [PMC free article] [PubMed] [Google Scholar]