Dear Editor,
In a recent review, Ye and colleagues discussed in this journal the inflammatory response mechanisms underlying the coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and its implications for therapy1, consistently with the current worldwide necessity of understanding and finding clinical therapies for the disease as it has been declared by the World Health Organization (WHO) a Public Health Emergency of International Concern, that to date has triggered the infection of more than 5 million people and over 300.000 deaths around the world.
Impaired and exacerbated immune responses have been associated with the clinical features and severity of the disease, as the SARS-CoV-2 can lead to innate and adaptative immune system activation, resulting in an excessive release of proinflammatory cytokines, known as cytokine storm (CS)2. The exaggerated cytokine response plays a major role in the immunopathology of the lung injury and the acute respiratory distress syndrome (ARDS) developed in critically ill patients, where the cytokine profile involves a variety of proinflammatory and anti-inflammatory mediators, including IL-1β, IL-2, IL-4, IL-6, IL-8, IL-12, IL-13, IL-17, G-CSF, GM-CSF, IFN-γ, IFN-α, IP-10, MCP-1 and TNF-α2 , 3.
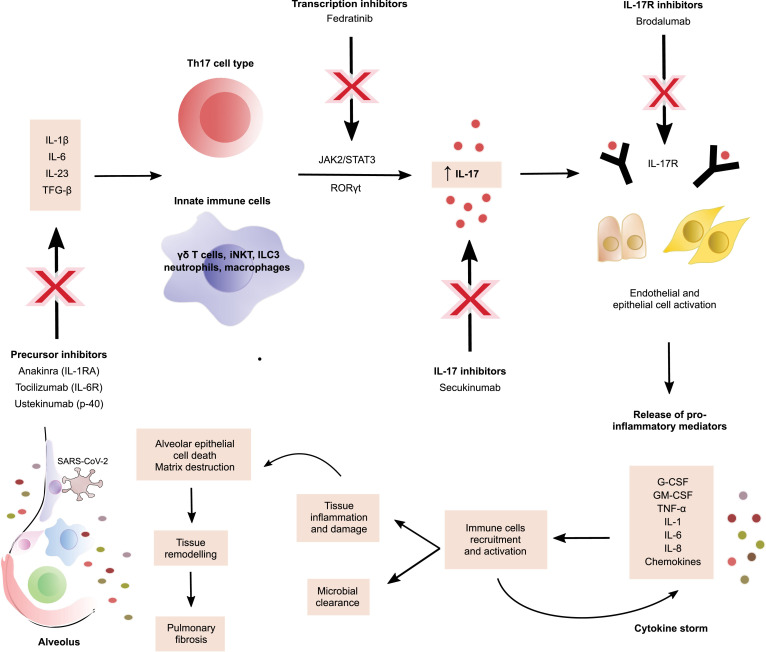
Acute lung injury observed in patients with severe COVID-19 is characterized by inflammation and tissue damage in the respiratory tract that has is highly correlated to T-helper type 17 (Th17) cell responses, as IL-17 can prompt the apoptosis of alveolar epithelial cells and the progression to pulmonary fibrosis, affecting the normal alveolar architecture, the alveolar-capillary gas exchange and consequently the normal oxygenation process, contributing to the respiratory symptoms of the disease4. IL-17A is a proinflammatory cytokine belonging to the IL-17 family of cytokines, which is mainly secreted by Th17 lymphocytes under the stimulus of TGF-β, IL-23 and IL-6 derived from phagocytes, although several innate immune cells such as γδ T cells, invariant natural killer T cells (iNKT), type 3 innate lymphoid cells (ILC3), neutrophils and macrophages can produce it too when stimulated by IL-1β and IL-235. Th17 subtype differentiation in early stages of inflammation is mostly mediated by IL-6-induced activation via JAK2/STAT3 pathway, which increases the production of IL-17A, IL-17F and IL-22 through the nuclear receptor and transcriptional regulator RORγt in a STAT3-dependent way6. Consequently, IL-17A downstream signaling stimulates the production of many proinflammatory agents and antimicrobial peptides from many cell types that participate in the recruitment of immune cells to the inflammation site, granulopoiesis, microbial clearance, mucosal defense and tissue inflammation such as CXCL1, CXCL5, CXCL12, MIP3A, G-CSF, GM-CSF, IL-1, IL-6, IL-8, TNF-α and IP-106 , 7, which may also have a paradoxical role as they participate in the hyperinflammation state of COVID-19 by promoting constant immune infiltrations and tissue destruction that exacerbates the manifestations of SARS-CoV-2 infection8. Moreover, a disbalance in IL-17 functions has been associated with a variety of illnesses, including cancer and immune-mediated inflammatory diseases (IMIDs) such as psoriasis, asthma, inflammatory bowel disease and rheumatoid arthritis (Fig. 1 ).
Fig. 1.
IL-17 mechanism in COVID-19 and potential therapeutic approaches.
Thereby, the detrimental immunological implications have led to the use of immunomodulatory therapy, also used in IMIDs, as management of severe COVID-19 considering there is no international established treatment guideline and that most of the specific strategies are under investigation. Suppression of proinflammatory mediators involved in the CS has shown effectiveness in the clinical improvement of severely ill patients with COVID-19, and its blockade may be the key to treat complications and reduce mortality associated to the hypercytokinemia, as revealed by an observation study in Anhui, China, where they evaluated the clinical outcomes of severe and critical COVID-19 patients after using tocilizumab, a recombinant humanized monoclonal antibody against IL-6 receptor employed in the treatment of cytokine release syndrome produced with the chimeric antigen receptor T-cell (CART) immunotherapy and other immunopathological conditions, and found promising results as tocilizumab reported quick control of symptoms, damage decrease in imaging findings and improved respiratory function in most of the patients of the study9. These data suggest that cytokine inhibitors, an immunomodulatory therapy, is a feasible therapeutic approach in COVID-19 patients with immune compromise, as it regulates the positive feedback loop between cytokines and immune cells and thus reduces in a significant way the proinflammatory state and immune related injury of the disease. Nevertheless, treatment implies targeting only those cytokines implicated in the CS and not interfering with the viral clearance dependent on IL-15, IL-12, IL-21, IFN-y and IFN type I10.
Therefore, modulating Th17 immune responses, particularly IL-17 associated responses, could provide potential treatment in patients with COVID-19. There are several ways to downregulate IL-17 effects, including blocking IL-17 itself, the IL-17 receptor or indirectly targeting the IL-17 pathway. For instance, secukinumab is a monoclonal antibody against IL-17A and brodalumab targets the IL-17R, but there are other inhibitors in the “upstream” signaling pathway such as JAK2 inhibitors (fedratinib) and antagonists of interleukins that induce Th17 differentiation, including IL-1 (anakinra), IL-6 (tocilizumab) and IL-23 (ustekinumab). The mechanism by which these agents could block SARS-CoV-2 immune related effects need to be well dilucidated given the fact that these inhibitors may provoke selective immunosuppression that may affect antiviral protective responses, as with ustekinumab that can also block IL-12 responses, which affects Th1 cell differentiation and interferon production. Otherwise, targeting JAK2/STAT3 pathway could be beneficial in several aspects since JAK2 does not interfere with type I interferons response that support antiviral immunity7, and then JAK2 inhibitors can be propitious to mitigate the proinflammatory function of existing Th17 cells, prevent the SARS-CoV-2 entrance to the cells and the immune activation in the initial stages of COVID-19. Furthermore, the benefits of targeted Th17 cell and IL-17 therapy strategies have to be weighed against the potential risks derived from the immunosuppression, adverse effects, success rate and cost-effectiveness of the therapy, for which much more research regarding the pathological role of IL-17 in COVID-19 is needed and randomized controlled trials with a large number of patients and minimal risk of bias should be performed.
Declaration of Competing Interest
None.
Acknowledgments
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1.Qing Ye, Bili Wang, Jianhua Mao. The pathogenesis and treatment of the ‘Cytokine Storm’’ in COVID-19. J Infect. 2020;80(6) doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yingxia Liu, Cong Zhang, Fengming Huang, Yang Yang, Fuxiang Wang, Yuan Jing. 2019-novel coronavirus (2019-nCoV) infections trigger an exaggerated cytokine response aggravating lung injury. ChinaXiv. 2020 doi: 10.12074/202002.00018. [DOI] [Google Scholar]
- 3.Chaolin Huang, Yeming Wang, Xingwang Li, Lili Ren, Jianping Zhao, Yi Hu. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahesh Manjunath Gouda, Prabhakar Bhandary Yashodhar. IL-17A Mediated Downstream Changes During Acute Lung Injury. Biochem Mol Biol J. 2018;04(02) doi: 10.21767/2471-8084.100062. [DOI] [Google Scholar]
- 5.Taku Kuwabara, Fumio Ishikawa, Kondo Motonari, Terutaka Kakiuchi. The Role of IL-17 and Related Cytokines in Inflammatory Autoimmune Diseases. Mediators Inflamm. 2017 doi: 10.1155/2017/3908061. 3908061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Suniti Bhaumik, Rajatava Basu. Cellular and molecular dynamics of Th17 differentiation and its developmental plasticity in the intestinal immune response. Front Immunol. 2017;8(254) doi: 10.3389/fimmu.2017.00254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dandan Wu, O Yang Xuexian. TH17 responses in cytokine storm of COVID-19: An emerging target of JAK2 inhibitor Fedratinib. J Microbiol Immunol Infect. 2020 doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tao Ma Wen, Ting Yao Xiao, Qun Peng, Kun Chen De. The protective and pathogenic roles of IL-17 in viral infections: Friend or foe. Open Biol. 2019 doi: 10.1098/rsob.190109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xiaoling Xu, Mingfeng Han, Tiantian Li, Wei Sun, Dongsheng Wang, Binqing Fu. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci. 2020;117(20):10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Georg Schett, Michael Sticherling, Neurath Markus F. COVID-19: risk for cytokine targeting in chronic inflammatory diseases. Nat Rev Immunol. 2020 doi: 10.1038/s41577-020-0312-7. [DOI] [PMC free article] [PubMed] [Google Scholar]