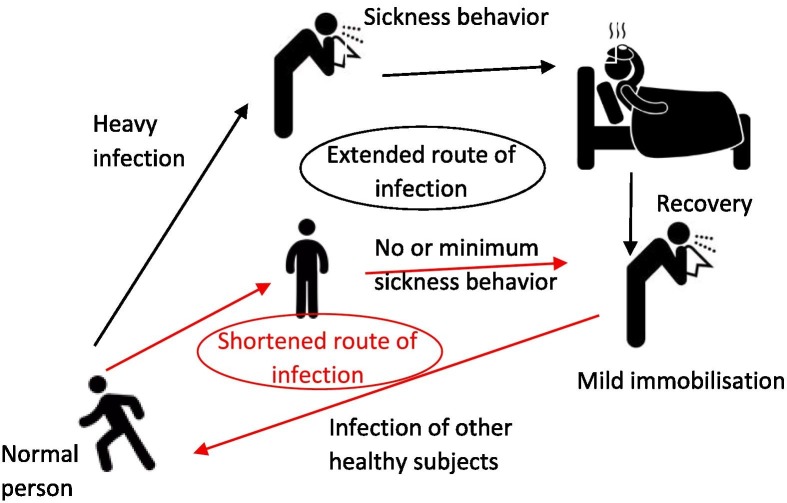
Sickness behavior, including social disinterest, isolation, reduced physical activity, fatigue, increased sleep duration, lethargy and reduced appetite, is an adaptive evolutionary conserved response developed by sick individuals to cope with infectious diseases; but also to slow the spread of the infection in the community by decreasing social interactions (Fig. 1 ). Sickness symptoms may also act as an alarm signal, eliciting to healthy individuals protective demeanor that permit them to avoid infection. However, sickness behavior of the novel coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), varies markedly between individuals. The huge variability of cytokine-induced sickness symptoms in COVID-19 is due to the hyper-inflammatory state that has been reported only in patients with moderate to critical illness but not in asymptomatic, pre-symptomatic and in individuals with mild forms of COVID-19 (Gandhi et al., 2020).
Fig. 1.
Accelerated COVID-19 infection – bypassing the slow and long disease circle of heavily infected individuals by mild or absent sickness behavior. Cytokine-induced sickness behavior, a form of behavioral defense, is a universal adaptive response to infectious pathogens that conserves energy to enhance the efficiency of the immune system. Sickness response includes fever, lethargy/fatigue, depression (or similar signs), irritability, discomfort, pain, nausea, cognitive disturbances including effects on memory and reaction time, and loss of interest in food and drink intake, social interactions and sex. These are triggered in sick individuals, primarily due to a sense of self-preservation and to better cope with pathogens, but evolutionary also to slow the spread of the infection in the community. This is typically achieved by reducing social interactions of sick individuals and further activating in healthy individuals their “behavioral immune system”, resulting in protective behaviors that may favor the avoidance or even expelling of the sick individual (e.g. by maintaining distance or avoiding encounters). Humans are able to infer infection risk from perceptual cues and recognize even subtle signs of sickness in others, including facial, olfactory and vocal changes.
Unlike its close relative SARS-CoV (the causative agent of SARS), whose shedding occurs during phases of strong sickness behavior, and usually by patients with more severe symptoms (i.e., after 10 days of illness), the SARS-CoV-2 shedding is rather highest early, at the time of symptom onset to day 5 of illness (Cevik et al., 2020). SARS-CoV-2 is less pathogenic but far more transmissible than SARS-CoV, and also than MERS-CoV, another pathogenic coronavirus, peaking at 7–10 days of illness (Cevik et al., 2020). In this context, evidence has shown that COVID-19 cases experiencing only mild sickness behavior constitute an important link in the transmission chain (Bouayed and Bohn, 2020), probably as mild-COVID-19 forms are insufficient to reduce social interaction and physical activity. Thus, mild sickness behavior might confer to the new coronavirus a high potential of person-to-person transmission, and would play to the advantage of the evolutionary success of the virus.
Although COVID-19 appears as a flu-like illness, with cough, sweat, rhinitis and muscle aches as the most common experienced symptoms, substantial differences exist regarding the timing of the onset of sickness response. While the median incubation period was estimated at 5.1 days, and 97.5% of patients develop symptoms within 11.5 days after infection (Lauer et al., 2020), a meta-analysis has shown that sickness symptoms start during the first day after inoculation of healthy volunteers with human influenza virus and peaked on the third day (Carrat et al., 2008). The average viral shedding increased during the first day and consistently peaked on the following (Carrat et al., 2008). Intriguingly, it has been highlighted in a recent perspective article that influenza vaccine increases significantly human social behavior pre-symptomatically (i.e., in the 48 h immediately post-exposure), with significant large-group interactions, when compared to 48 hours pre-exposure (Bouayed and Bohn, 2020). In accordance, increased social interactions during the asymptomatic phase, especially in super-spreaders, was hypothesized to constitute a paramount route of SARS-CoV-2 transmission (Bouayed and Bohn, 2020). During the period of SARS-CoV-2 incubation, i.e., before displaying sickness symptoms, which can last up to 2 weeks, the fact that pre-symptomatic patients are potentially infectious constitutes an additional factor accelerating the transmission of COVID-19. It has been estimated that pre-symptomatic SARS-CoV-2 infections alone are almost sufficient to sustain epidemic growth (Ferretti et al., 2020). Therefore, infectiousness may peak prior to sickness behavior onset and COVID-19 patients may be infectious 1 to 3 days prior to showing symptoms (Gandhi et al., 2020). This constitutes an additional and possibly key factor providing the new coronavirus an extraordinary advantage for its silent spread.
While sickness response in COVID-19 individuals has been classified to range from mild to critical, it is estimated that up to 40% of people infected with SARS-CoV-2 did not experience any signs of disease (Beck and Aksentijevich, 2020). Interestingly, the viral loads of asymptomatic persons were similar to those in symptomatic individuals but with faster viral clearance and potential transmissibility at the onset of infection (Cevik et al., 2020). Thus, even when a large population is somewhat protected from COVID-19 outbreak and heavy symptoms, this enhances the risk of human-to-human transmission in the community, especially to vulnerable individuals. Indeed, it was estimated that both asymptomatic and pre-symptomatic SARS-CoV-2 infections combined account for up to 56% of all transmissions (Candido et al., 2020). Therefore, the absence of sickness behavior in many SARS-CoV-2 carriers constitutes an important factor determining COVID-19 transmission rate. Consequently, without sickness behavior or even with mild-symptoms, infected individuals may not reduce their social interaction or seek medical care.
Children are less vulnerable to the novel coronavirus as they develop less symptomatic COVID-19 (typically mild symptoms) and therefore undergo less frequently hospitalization compared to other age groups. Nevertheless, it has been shown that nasopharyngeal SARS-CoV-2 viral loads in infected children were similar to those in other age groups, suggesting that children could be as infectious as adults (Jones et al., 2020). However, shedding duration is positively correlated with age, and no viable SARS-CoV-2 was detected beyond 9 days of sickness symptoms (Cevik et al., 2020). Thus, it is yet unclear whether viral load in children also implies high infectiousness, as the virus particles may not necessarily be viable. Theoretically, children could act as an important link in the transmission chain of SARS-CoV-2. This assumption is reinforced by the fact that sickness behavior is usually not experienced in infected children. In India, it was suggested from contact-tracing data that children below 14 years of age are active participants in the spread of SARS-CoV-2 to contacts of similar-ages (Laxminarayan et al., 2020). Contrary to what was assumed, it has been inferred from a cluster in a nursery in Poland that children could be effective drivers of SARS-CoV-2 infection between adults (Okarska-Napierała et al., 2021). Indeed, 5 asymptomatic children 1–2 years of age might have infected 9 adult relatives (Okarska-Napierała et al., 2021). However, given the controversial findings on children, further studies are needed to clarify whether children are less important drivers of transmission than adults or whether they are rather silent spreaders of SARS-CoV-2 into the community.
In summary, sickness behavior is anticipated to slow transmission by reducing social interactions and the limited activity of the sick individuals, and further by alerting healthy persons to trigger protective avoidance behavior. During the ongoing pandemic, asymptomatic and pre-symptomatic SARS-CoV-2 infections, and mild forms of COVID-19 constitute the key component of the transmission chain, especially when coinciding with early high viable viral load. In this respect, it was estimated that 10% of the cases have been super-spreaders, resulting in 80% of viral spread (Bouayed and Bohn, 2020). Without severe or moderate sickness behavior and when infectiousness precedes considerably cytokine-induced symptoms, infected individuals have the potential to be super-spreaders. Thus, person-to-person transmission of the new coronavirus can continue, contributing to making COVID-19 a rapidly spreading infectious worldwide pandemic.
References
- Beck D.B., Aksentijevich I. Susceptibility to severe COVID-19. Science. 2020;370(6515):404–405. doi: 10.1126/science.abe7591. [DOI] [PubMed] [Google Scholar]
- Bouayed, J., Bohn, T., 2020. Behavioral manipulation-key to the successful global spread of the new coronavirus SARS-CoV-2? J. Med. Virol. doi:10.1002/jmv.26446. [DOI] [PMC free article] [PubMed]
- Candido D.S., Claro I.M., de Jesus J.G., Souza W.M., Moreira F.R.R., Dellicour S., du Mellan T.A., Plessis L., Pereira R.H.M., et al. Evolution and epidemic spread of SARS-CoV-2 in Brazil. Science. 2020;369(6508):1255–1260. doi: 10.1126/science.abd2161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrat F., Vergu E., Ferguson N.M., Lemaitre M., Cauchemez S., Leach S., Valleron A.J. Time lines of infection and disease in human influenza: a review of volunteer challenge studies. Am. J. Epidemiol. 2008;167(7):775–785. doi: 10.1093/aje/kwm375. [DOI] [PubMed] [Google Scholar]
- Cevik M., Tate M., Lloyd O., Maraolo A.E., Schafers J., Ho A. SARS-CoV-2, SARS-CoV, and MERS-CoV viral load dynamics, duration of viral shedding, and infectiousness: a systematic review and meta-analysis. Lancet Microbe. 2020 doi: 10.1016/S2666-5247(1020)30172-30175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferretti L., Wymant C., Kendall M., Zhao L., Nurtay A., Abeler-Dörner L., Parker M., Bonsall D., Fraser C. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science. 2020;368(6491):eabb6936. doi: 10.1126/science:abb6936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandhi R.T., Lynch J.B., del Rio C. Mild or moderate Covid-19. N. Engl. J. Med. 2020;383(18):1757–1766. doi: 10.1056/NEJMcp2009249. [DOI] [PubMed] [Google Scholar]
- Jones, T.C., Muhlemann, B., Veith, T., Biele, G., Zuchowski, M., Hoffmann, J., Stein, A., Edelmann, A., Corman, V.M., Drosten, C., 2020. An analysis of SARS-CoV-2 viral load by patient age. medRxiv https://doi.org/10.1101/2020.1106.1108.20125484.
- Lauer S.A., Grantz K.H., Bi Q., Jones F.K., Zheng Q., Meredith H.R., Azman A.S., Reich N.G., Lessler J. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann. Intern. Med. 2020;172(9):577–582. doi: 10.7326/M20-0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laxminarayan R., Wahl B., Dudala S.R., Gopal K., Mohan B.C., Neelima S., Jawahar Reddy K.S., Radhakrishnan J., Lewnard J.A. Epidemiology and transmission dynamics of COVID-19 in two Indian states. Science. 2020;370(6517):691–697. doi: 10.1126/science.abd7672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okarska-Napierała M., Mańdziuk J., Kuchar E. SARS-CoV-2 Cluster in nursery, Poland. Emerg. Infect. Dis. 2021;27(1):317–319. doi: 10.3201/eid2701.203849. [DOI] [PMC free article] [PubMed] [Google Scholar]