Abstract
SARS-CoV-2 infection or COVID-19 has become a worldwide pandemic; however, effective treatment for COVID-19 remains to be established. Along with acute respiratory distress syndrome (ARDS), new and old cardiovascular injuries are important causes of significant morbidity and mortality in COVID-19. Exploring new approaches managing cardiovascular complications is essential in controlling the disease progression and preventing long-term complications. Oxytocin (OXT), an immune-regulating neuropeptide, has recently emerged as a strong candidate for treatment and prevention of COVID-19 pandemic. OXT carries special functions in immunologic defense, homeostasis and surveillance. It suppresses neutrophil infiltration and inflammatory cytokine release, activates T-lymphocytes, and antagonizes negative effects of angiotensin II and other key pathological events of COVID-19. Additionally, OXT can promote γ-interferon expression to inhibit cathepsin L and increases superoxide dismutase expression to reduce heparin and heparan sulphate fragmentation. Through these mechanisms, OXT can block viral invasion, suppress cytokine storm, reverse lymphocytopenia, and prevent progression to ARDS and multiple organ failures. Importantly, besides prevention of metabolic disorders associated with atherosclerosis and diabetes mellitus, OXT can protect the heart and vasculature through suppressing hypertension and brain-heart syndrome, and promoting regeneration of injured cardiomyocytes. Unlike other therapeutic agents, exogenous OXT can be used safely without the side-effects seen in remdesivir and corticosteroid. Importantly, OXT can be mobilized endogenously to prevent pathogenesis of COVID-19. This article summarizes our current understandings of cardiovascular pathogenesis caused by COVID-19, explores the protective potentials of OXT against COVID-19-associated cardiovascular diseases, and discusses challenges in applying OXT in treatment and prevention of COVID-19.
Chemical compounds
Angiotensin-converting enzyme 2 (ACE2); atrial natriuretic peptide (ANP); cathepsin L; heparan sulphate proteoglycans (HSPGs); interferon; interleukin; oxytocin; superoxide dismutase; transmembrane serine protease isoform 2 (TMPRSS2).
Keywords: Heart, Hypothalamus, Immunology, SARS-CoV-2, Vasculature
Graphical abstract
1. Introduction
The outbreak of a novel coronavirus (SARS-CoV-2) infection (i.e. COVID-19) has become a worldwide pandemic since December 2019. The number of confirmed cases of COVID-19 in the world is >95.4 million including ~2 million deaths as of January 17, 2021 (https://www.worldometers.info/coronavirus/). The disease carries a high mortality among those who are older, immune-suppressed, or with underlying co-morbidities, predisposing them to acute respiratory distress syndrome (ARDS), cardiovascular (CV) complications, and multi-organ failure [1,2]. Given the medical emergency of a rapidly spreading contagion, its unpredictable nature with continuous viral mutations, lack of effective treatments, and current lack of data for long-term effects of vaccines [[3], [4], [5]], novel strategies to prevent its infection and improve survival rate among infected patients are urgently needed.
Among many potential treatments, oxytocin (OXT), a classical hypothalamic neuropeptide with pleiotropic actions [6], has emerged as a strong candidate against COVID-19. Moreover, evidences have emerged showing the CV protective effects of OXT [7] by targeting many key pathogenetic events in COVID-19. This is critical as CV complications contribute to significant morbidity and mortality among COVID-19 patients [[8], [9], [10]]. This article summarizes our current understandings of potential roles of OXT in prevention and treatment of CV complications in COVID-19.
2. Pathogenesis of COVID-19
2.1. Symptoms and clinical manifestations
Despite many asymptomatic cases, those with symptoms often present with fever, cough, fatigue, anosmia or hyposmia, nausea, and anorexia. Factors that are associated with mortality include male sex, advanced age, cytokine storm, ARDS, acute cardiac injury, acute kidney injury and the presence of comorbidities including hypertension, diabetes mellitus, CV diseases, and cerebrovascular diseases [11,12]. Patients often exhibit ground-glass opacity on chest computed tomography [13], troponinemia [14], increased cytokines, and lymphocytopenia [15] as well as hypometabolism in many brain regions including the hypothalamus [16]. All of these findings indicate the involvement of multiple organ systems.
2.2. Immunological disorders
The pathogenesis of COVID-19 involves dysfunction in immunological homeostasis, fibrinolysis, vessel contractility, inflammation, oxidative stress, and vascular permeability, etc. [1]. As reported, COVID-19 patients carry high levels of pro-inflammatory cytokines including interleukin (IL)-6 and tumor necrosis factor-α, atrophy of spleen and lymph nodes, higher neutrophil count and lower number of other types of leukocytes, particularly T-lymphocytes [17]. This compromised immune system causes destruction of pneumocytes following the invasion of SARS-CoV-2, which in turn increases neutrophil infiltration and even leads to the fatal “cytokine storm.” That is, an uncontrolled and excessive release of pro-inflammatory cytokines that causes autoimmune self-destructive response. The cytokine storm contributes to severe ARDS, multiple organ failure and eventually fatality [18]. For instance, IL-1 induces a significant number of metabolic and hematological changes such as hypotension, endothelial dysfunction, macrophage intervention and increased protein leakage from blood vessels, venous thromboembolism and shock [19]. The same importance applies to lymphocytopenia. For example, thymosin α1 supplement significantly reduces mortality of severe COVID-19 patients with the counts of CD8+ and CD4+ T cells in circulation lower than 400/μL and 650/μL, respectively. This is because thymosin α1 reverses T cell exhaustion and improves immune reconstitution by promoting thymus output during SARS-CoV-2 infection [20]. Thus, suppressing sustained release of cytokines and inflammatory response, and promoting T-lymphocyte functions are the essential therapeutic strategy to treat COVID-19 patients.
2.3. SARS-CoV-2 entry of cells
SARS-CoV-2 entry of host cells could involve host cell membrane-bound angiotensin-converting enzyme 2 (ACE2), transmembrane serine protease isoform 2 (TMPRSS2), lysosomal endopeptidase cathepsin L, and membrane bound heparan sulphate proteoglycans (HSPGs) and others [21]. As the receptor for SARS-CoV-2, ACE2 is present on the surface of lung alveolar epithelial cells, enterocytes of the small intestine, renal tubules, heart, arterial and venous endothelial cells, arterial smooth muscle cells, cerebral neurons and monocytes/macrophages [22,23]. Spike proteins give SARS-CoV-2 the ability for cellular infection [24], as human immunodeficiency virus (HIV) does [25]. SARS-CoV-2 enters human ACE2-expressing cells mainly via endocytosis, in which cathepsin L and other components are critical [26]. TMPRSS2 primes the viral spike protein in order to facilitate ACE2-mediated viral entry and extra-pulmonary spread [27]. Besides TMPRSS2, HSPGs are cell surface receptors that assist ACE2 in endocytosis-mediated coronavirus entry [28]. Thus, multiple cellular components are involved in the invasion of SARS-CoV-2 and can be potential targets in the treatment.
2.4. ACE2 loss and AT II effects
SARS-CoV-2 invasion destroys infected cells and reduces ACE2 activity. Resultantly, the capacity of ACE2 to convert angiotensin II (AT II) into inactive AT 1–7 decreases, which causes elevation of serum AT II levels, neutrophil accumulation, vascular hyper-permeability, and pulmonary edema seen in ARDS [29]. Infusion of ACE2 in ARDS patients can decrease AT II and IL-6 levels, and increase surfactant protein, thereby exerting protective functions [30]. Blocking viral endocytosis and excessive AT II actions are alternative measures for controlling the pathogenesis.
3. COVID-19 and CV complications
3.1. CV complications
CV disease represents a common comorbidity predisposing COVID-19 patients to high mortality [[8], [9], [10]]. The complications include acute cardiac injury, myocarditis, fibrosis, fulminant myocarditis rapidly evolving into depressed systolic left ventricular function, arrhythmias, hypercoagulable state, venous thromboembolism, and cardiomyopathies mimicking ST-elevation myocardial infarction [14,31]. Acute cardiac injury is commonly observed in severe cases and is strongly associated with mortality [11]. It is important to note that COVID-19 patients often carry pre-existing CV comorbidities. Among 45 studies including 14,358 adult participants, prevalent comorbidities are hypertension (19.1%), CV disease (17.9%), endocrine disorder (9.3%) and diabetes (9.2%) [13]. Patients with pre-existing conditions may face a greater risk of developing into the severe condition. Thus, old and new CV complications are worthy of special attention in COVID-19 patients.
3.2. Dissemination via endothelium of blood vessels
In the dissemination of SARS-CoV-2 from the lung to multiple organs, endothelial dysfunction of the CV system is a common cause. After infecting respiratory epithelial cells, SARS-CoV-2 gets into the circulation through endothelial cells of blood vessels in the lung and then enters tissues through the endothelial cells in capillaries again. COVID-19 patients exhibit abnormalities in the vascular endothelium, which alter the blood flow, and evoke platelet function abnormalities and hyper-viscosity [32]. The endothelial dysfunction and damage cause release of procoagulant components, which together with hypoxia, immune reactions and hypercoagulability contributes to thrombogenesis [33]. Thus, endothelium is a critical barrier in COVID-19 transmission and dissemination.
3.3. Mechanisms underlying CV injuries
CV injuries from viral infection are also mediated by ACE2 and cathepsin L [34]. In adult human heart, pericytes with high-expression of ACE2 serve as target cardiac cells for SARS-CoV-2. Infection in pericytes results in dysfunction of capillary endothelial cells and microvascular dysfunction. Patients with heart failure show increased ACE2 expression at both mRNA and protein levels [35]. Among patients with diabetes and CV disease, the expression levels of TMPRSS2 are elevated significantly in comparison to healthy individuals. Additionally, expression of viral entry-related genes is increased in the settings of hypertension across target organ systems [21]. Thus, if infected by the virus, these patients may have higher risk of CV complications and progression to critically-ill condition.
The etiology of CV injuries seems to be multifactorial including direct viral myocardial damage, hypoxia, hypoperfusion, enhanced inflammatory state, ACE2 down-regulation, drug toxicity, endogenous catecholamine adrenergic status and others [36]. In SARS-CoV-2 infected human, pluripotent stem cell-derived cardiomyocytes, double-stranded viral RNA and viral spike protein expressions are detected in intracellular space. In addition, viral spike protein and particles are present in living human heart slices after infection with SARS-CoV-2, which induces cytotoxic and proapoptotic reaction and abolishes cardiomyocyte activities. The subsequent virus-mediated myocardial injury is closely related to inflammatory hyperactivity. For example, cardiac injury is significantly associated with inflammation biomarkers such as IL-6, C-reactive protein, hyperferritinemia, and leukocytosis along with elevated troponin I levels [31]. Direct cardiac injury following viral entry through ACE2 receptors is closely associated with cytokine storm [37]. One of the currently used treatments, remdesivir, has been shown to inhibit viral infection of cardiomyocytes [34,38]. These findings highlight the key role of viral injury in initiating the CV pathogenesis in COVID-19 patients.
Further study also reveals that lung pathology can cause CV malfunctions. Clinical and experimental observations have shown that loss of ACE2 function aggravates pulmonary hypertension while restoring ACE2 function exerts protection on cardiopulmonary circulation. In addition, SARS-CoV-2 tropism and its interaction with the renin-angiotensin-aldosterone system (RAAS), through ACE2 receptor, may enhance inflammation response and cardiac aggression [31]. Lastly, SARS-CoV-2 infection can lead to right heart dysfunction by inducing pulmonary hypertension, pulmonary embolism, and ARDS.
4. Effects of OXT on CV activity
OXT, a nonapeptide synthesized in hypothalamic magnocellular neuroendocrine cells in the supraoptic and paraventricular nuclei (SON and PVN), is a well-known humoral factor regulating parturition and lactation [6,39]. In the brain, OXT terminals are found on large intracerebral arteries [40]. OXT-containing neural fibers are present in pia-arachnoids, blood vessels at the base of the brain and over the dorsal surface [41]. Thus, OXT can regulate cerebral blood flow directly. Moreover, OXT and OXT receptor (OTR) are identified in the pulmonary artery, vena cava and aorta of rats, dogs and sheep [42] as well as cardiac cells in rats [43] and endothelial cells of human umbilical vein endothelial cells [44]. They allow OXT to regulate CV activity at peripheral sites as well.
4.1. General effects of OXT on CV activity
OXT can modulate CV activity including the development of cardiomyocytes, ionotropic and chronotropic effects, dual role in cardiac output, endocrine activity (e.g. secretion of atrial natriuretic peptide, ANP) and direct cardioprotection [7].
In general, OXT exerts negative chronotropic and ionotropic effects on cardiac activity. OXT can reduce heart rate and cardiac contractility by activating intrinsic cardiac cholinergic neurons of the vagus and promoting nitric oxide (NO) production. This is because atropine and NO synthase inhibitor, L-NAME, can significantly inhibit this OXT effect [45]. Consistently, in OXT knockout mice, there were mild hypotension, higher sympathetic tone and higher heart rate [46], indicating activation of sympathetic outflow. At higher concentrations, OXT can directly reduce left ventricle pressure and heart rate in isolated rat hearts and decrease mean artery pressure in rats, which could involve the action of ANP [47]. Thus, OXT participates in the maintenance of tonic blood pressure and suppression of sympathetic reserve.
OXT can protect the CV system from social stress damages or the brain-heart syndrome via neural and endocrine approaches. Social stress is important etiology of CV malfunctions because it can disrupt normal behavior, neuroendocrine, and autonomic responses. By facilitating positive social interactions, suppressing sympathetic outflow and reducing fear and anxiety, OXT can counteract the deleterious effects of social stress and its associated unhealthy life-style, hypertension, and coronary artery diseases [48,49].
Another approach for OXT protection is its influencing the secretion of many cytokines [50,51]. For example, OXT infusion dose-dependently increases plasma ANP concentration as much as 4-fold after 20 min. ANP produced in the right atrium can prevent reperfusion arrhythmia and maintain ATP production in ischemic tissue while facilitating Na+ excretion to reduce after-load and blood pressure [52]. Moreover, OXT can suppress the activity of hypothalamic-pituitary-adrenocortical axis [53], and reduce the release of stress hormone. Through these approaches, OXT can reduce heart rate and blood pressure while increasing the reserve of cardiac output, thereby exerting CV protective effects.
4.2. Suppressing pathogenesis underlying CV diseases by OXT
OXT is a well-documented hormone of CV protection [54,55]. For example, in rats with myocardial infarction, OXT infusion results in suppression of inflammation by reducing neutrophils and macrophages, depressing the expression of tumor necrosis factor-α and IL-6, promoting transforming growth factor-β and reducing apoptosis and fibrotic deposits [56]. In rabbits of myocardial infarction and reperfusion, OXT treatment significantly improves left ventricular function, reduces infarct size, and increases the number of CD31-positive microvessels. These effects are associated with increased expression of OTR, prosurvival signals, and antifibrotic and angiogenic signals like phosphorylated-Akt protein kinase, phosphorylated-endothelial NO synthase, and vascular endothelial growth factor [57]. In addition, OXT can reduce oxygen consumption by its negative chronotropic and ionotropic effects and protect the heart from ischemia/reperfusion injuries. These facts support that OXT can be a potential treatment in patients with CV complications.
The protective effects of OXT can also be achieved through central approach. In a rat model of trans-ascending aortic constriction-induced left ventricular hypertrophy and heart failure, chronic activation of hypothalamic OXT neurons in the PVN to elevate parasympathetic tone can improve cardiac function, reduce IL-1β expression and lower myocardial collagen density (an indicator of cardiac fibrosis) as well as heart rate sensitivity to β-adrenergic stimulation [58]. Intracerebroventricular infusion of OXT or centrally released OXT can induce a preconditioning effect in ischemic-reperfused rat heart via brain OTR [59].
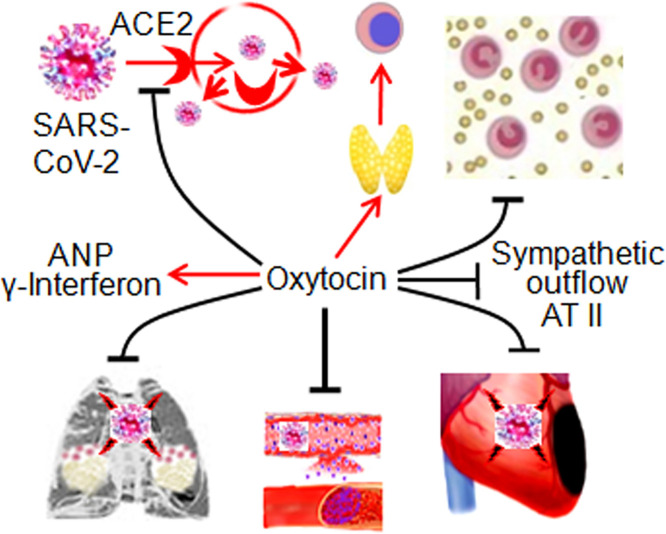
In OXT protection of the CV system, endothelial cells are an important target. OXT can elicit mesenchymal cells to express endothelial cell markers [60] and promote proliferation and migration of human dermal microvascular endothelial cells [61]. OXT is necessary for the formation of neurovascular interface in the pituitary by affecting endothelial morphogenesis [62]. In the posterior pituitary, OXT-containing axonal terminals express vascular endothelial growth factor A that can cause active proliferation of endothelial cells [63]. By protecting endothelial cells in blood vessels and stabilizing anticoagulant heparan [64], OXT could reduce the formation of thromboembolism and atherosclerosis while blocking pathogen invasion. Fig. 1 summarizes the CV protective effects and potential involvement of OXT in COVID-19-associated CV diseases.
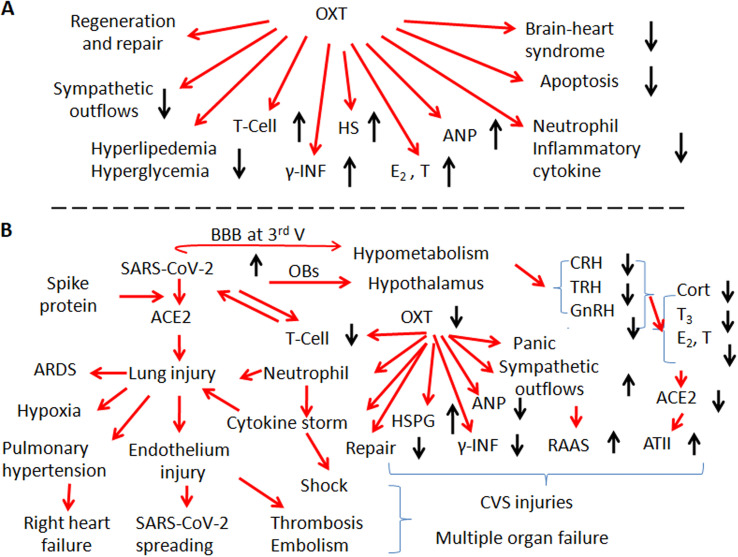
Fig. 1.
General protective functions of oxytocin (OXT) on the activity of cardiovascular system (CVS) and OXT-associated cardiovascular injuries evoked by SARS-CoV-2. A. General protective functions of OXT on CVS activity. B. Mechanisms underlying cardiovascular injuries in association with potentially abnormal activity of OXT/OXT receptor signaling events in COVID-19 patients. Abbreviations: 3rd V, the third ventricle; ACE2, angiotensin-converting enzyme 2; ANP, atrial natriuretic peptide; AT II, angiotensin II; ARDS, acute respiratory distress syndrome; BBB, blood-brain barrier; CRH, corticotropin-releasing hormone; E2, 17β-estradiol; GnRH, gonadotropin-releasing hormone; HS, heparan sulphate; HSPG, heparan sulphate proteoglycan; RAAS, renin-angiotensin-aldosterone system; γ-IFN, γ-interferon; OBs, olfactory bulbs; T, testosterone; T3, triiodothyronine; T-cells, T-lymphocytes; TRH, thyrotropin-releasing hormone.
5. OXT functions against COVID-19-associated CV injuries
The OXT-secreting system is considered as the higher neuroendocrine regulation center of the immune system [50,51] and key protective factor of CV system. Interruption of this system by COVID-19 partially accounts for the CV complications while reviving the OXT-secreting system or supplying OXT becomes a feasible strategy of inhibiting the pathogenesis of CV manifestations in COVID-19.
5.1. Evidence suggesting OXT involvement in COVID-19
The pathological changes in CV system of COVID-19 patients could result from dysfunction of the OXT-secreting system. In epidemiology, the decline in OXT and OTR with aging is likely responsible for the increased incidence of CV diseases since weakened OTR signaling can accelerate inflammatory and oxidative injuries, particularly among menopausal women [53,65]. In aged man, OXT secretion is also reduced, which is in parallel with increased incidence of hypertension, coronary artery disease, diabetes and cerebrovascular diseases. These diseases are also the common comorbidities that increase susceptibility to COVID-19 [11]. Consistently, plasma OXT level experiences a 3-fold decline in aged mice compared with young, and this decline is accompanied by similar decrease in OTR levels in muscle stem cells [66]. By contrast, exogenous estrogen application can increase OXT secretion, OTR mRNA expression in mouse brain [67], and intracardiac OTR signaling [68], which simulates the physiology of young women who are less susceptible than menopausal women to COVID-19. In addition, melatonin, a pineal hormone that has remarkable anti-inflammatory effects and can antagonize various viral infections including COVID-19 [69], can also regulate OXT release [70]. Thus, the susceptibility to COVID-19 is highly correlated with the activity of the OXT-secreting system.
In COVID-19 patients, there is also evidence indicating involvement of the hypothalamic-pituitary system. In two SARS-CoV-2 infected patients, the mammillary bodies and hypothalamus carried T2-hyperintensity, the pituitary gland was enlarged, and the upper pituitary stalk seemed globular in sagittal fluid-attenuated inversion recovery [71]. Since the mammillary bodies function as the upstream center of OXT neuronal activity, particularly its release in a pulsatile pattern [72], viral infection of the mammillary bodies unavoidably changes the downstream OXT neuronal activity and OXT secretion. In COVID-19 patients, many brain areas innervated by olfactory bulbs including the hypothalamus showed hypometabolism in 18F-FDG positron emission tomography scans [16], suggesting low activity of olfaction-associated hypothalamic neuroendocrine processes. It is also reported that activation of ACE2 in the PVN increases neuronal NO synthase and NO production, which can increase sympatho-inhibitory GABA activity and decrease sympatho-excitatory AT II signaling and glutamate activity [73]. ACE2 can reduce oxidative stress and cyclooxygenase-mediated neuroinflammation, increase antioxidant and NO signaling, and attenuate the development of neurogenic hypertension [74]. Studies on pathological changes in the hypothalamus of SARS-CoV-associated diseases also support the involvement of the hypothalamic neuroendocrine system in COVID-19. For example, in autopsies of 8 SARS patients, SARS-CoV was identified in the cytoplasm of numerous neurons in the hypothalamus and cortex [75]. In post-mortem tissues from HIV patients, OXT immunoreactivity in hypothalamic neurons decreased significantly [76]. Since these viruses use the same mechanisms of cellular infection and disruption as SARS-CoV-2 does, disruption of endogenous OXT production could be one etiology leading to the progression of COVID-19.
Involvement of the neuroendocrine hypothalamus is also supported by findings of endocrine disorders in COVID-19 patients as well. Testosterone and estrogen can increase the expression of ACE2, whereas serum total testosterone level is significantly decreased in COVID-19 patients [77]. This can result from the malfunction of sex glands as well as hypothalamic gonadotropin-releasing hormone. Levels of thyroid stimulating hormone and serum total triiodothyronine of COVID-19 patients are significantly lower than those of the healthy control group and non-COVID-19 pneumonia patients [78], which likely worsens the hypometabolism of the hypothalamus [16]. COVID-19 patients also have lower circulating levels of natriuretic peptides [79]. Since ANP is under facilitatory regulation of OXT, its reduction manifests that OXT or OTR signaling is reduced in COVID-19. In addition, systemic OXT levels are significantly suppressed following a peripheral infusion of AT II [80]. Since AT II is increased due to the reduction of ACE2, which can also reduce OXT secretion in COVID-19 patients. Decreases in these hypothalamus- and pituitary-associated hormones are in agreement with the hypometabolism found in the hypothalamus and pathological manifestations in COVID-19 patients [16,81], and they together indicate suppression of the hypothalamic neuroendocrine activity including the OXT-secreting system.
It is likely that SARS-CoV-2 enters the brain from the nose to the olfactory bulbs, and then infects neurons in the hypothalamus via the olfactory nerve [16]. The olfactory bulb involvement is supported by the high rates of anosmia or hyposmia in patients with COVID-19 [81] and by neural connections between the olfactory bulb and SON [82]. Alternatively, SARS-CoV-2 in the blood can enter the hypothalamus and pituitary through fenestrated capillaries of blood-brain barrier around the third ventricle and pituitary [83]. Direct evaluation of plasma OXT levels and examination of hypothalamic histology of COVID-19 patients are needed to further clarify this association.
5.2. Suppression of immunological disorders by OXT
Cytokine storm and lymphocytopenia are two major features of immunological disorders among COVID-19 patients. OXT has the potential to suppress cytokine storm. Diffuse pneumocyte and alveolar damage with pulmonary edema, proteinaceous exudate, inflammatory cell infiltrates, and release of pro-inflammatory cytokines can lead to cytokine storm [84]. This process can be suppressed by OXT administration. As shown in animal studies, exogenous OXT can decrease levels of IL-1β, IL-6, IL-18, and myeloperoxidase as well as incidence of acute lung injury in mice induced by lipopolysaccharide, an endotoxin [85]. OXT can downregulate neutrophil chemotactic molecules and myocardial neutrophil infiltration, and prevent myocardial injury by reducing inflammatory reaction, reactive oxygen species, and apoptosis caused by neutrophils [86]. In both smooth muscle and vascular endothelial cells, OXT can inhibit oxidative stress and pro-inflammatory cytokine release [87,88], thereby attenuating the cytokine storm and inflammation.
Additionally, OXT has the potential to block SARS-CoV-2 infection of cells and reduce SARS-CoV-2 loads. OXT can induce γ-interferon expression as shown in mouse spleen cell cultures [89]. Interferons can reduce viral infections by producing γ-interferon-inducible lysosomal thiol reductase that restricts the entry of selected enveloped RNA viruses via disrupting cathepsin L metabolism [90]. This potential of OXT is supported by the fact that at 24-h following weaning of suckling when OXT levels and pulsatility reduce, cathepsin L and cell apoptosis increase dramatically in mouse mammary glands [91]. Another potential approach of OXT blocking SARS-CoV-2 infection is its blocking HSPGs-mediated viral infection. OXT can induce the expression of extracellular superoxide dismutase [92], an antioxidant enzyme that has been shown to protect tissues from reactive oxygen species-induced heparin and heparan sulphate fragmentation, and thus the subsequent neutrophil chemotaxis [64]. Importantly, heparin and heparan sulphate can antagonize the binding of SARS-CoV-2 to HSPGs and block their cellular internalization [93]. Through these approaches, OXT has the potential to prevent SARS-CoV-2 entry and disruption of cells.
OXT can also reduce viral load by reversing lymphocytopenia. As reported, OXT acts on OTR in the thymus to promote the differentiation of T-lymphocytes [94]. OXT elicits a functional intracellular [Ca2+] response in T-lymphocytes to activate resident T-lymphocytes [95]. In women infected with HIV that has spike proteins highly homologous to COVID-19, high-level of OXT was associated with a positive correlation between stress and CD4 cell counts [96]. In a recent study, OXT was found to be more effective than hydroxychloroquine or lopinavir in modulating inflammation, and enhancing T-lymphocyte activation [97]. These findings suggest that OXT can prevent or alleviate lymphocytopenia in COVID-19 patients and thus reduce SARS-CoV-2 load, making OXT an ideal candidate for treatment of COVID-19.
5.3. Alleviating CV pathogenesis by increasing endothelial integrity
Besides immunological modulation, OXT may reduce CV injury from COVID-19 by increasing cellular integrity, particularly endothelium. First, OXT can prevent ACE2 loss by increasing T-lymphocytes that can in turn reduce SARS-CoV-2 viral load and cellular injuries [98]. Second, OXT maintains cell integrity by reducing neutrophil-mediated inflammatory injury and cardiac apoptosis [99]. Third, OXT may protect endothelial cells by increasing vascular endothelial growth factor [57] and promoting angiogenesis [44]. Fourth, OXT antagonizes the adverse effect of AT II on the CV system by increasing ANP production [7]. ANP can reduce renin and thus AT II activity in the RAAS, thus reducing damages in endothelial, pulmonary, and CV systems from COVID-19 [79]. Fifth, OXT can activate brainstem vagal neurons to increase parasympathetic output while decreasing RAAS activity [100] driven by sympathetic outflow [101] that are activated during COVID-19 [102]. Lastly, OXT can inhibit carbonic anhydrase activity that is associated with hypoxic pulmonary vasoconstriction, pulmonary hypertension right ventricle hypertrophy and fibrosis among COVID-19 patients [103]. While direct evidence on OXT modulation of ACE2 and AT II remains to be collected, OXT can be used to antagonize AT II effects in COVID-19 patients.
5.4. Suppression of COVID-19-associated CV injuries
CV system is a major target of COVID-19 that causes myocardial infarction, fulminant myocarditis, heart failure, arrhythmias, venous thromboembolism, and cardiomyopathies [14,31,104,105]. OXT exerts cardioprotective functions by reducing inflammation and cardiac fibrosis, improving left ventricular function, enhancing pro-survival kinases, elevating parasympathetic tone, reducing the infarct size, and protecting the heart from myocardial injury [7]. For example, OXT pre-treatment inhibits the degranulation of cardiac mast cells induced by ischemia and reperfusion injury, and down-regulates the expression of inflammatory factors [106]. OXT also functions as an anti-arrhythmic agent. For example, acute myocardial ischemia is accompanied by a rapid increase in electrical instability and often fatal ventricular arrhythmias. OXT can cause a significant and biphasic dose-dependent reduction in ectopic heart activity and arrhythmia score [107]. Intranasal application of OXT significantly reduces brain infarction and maintains the integrity of the blood-brain barrier in mice [108] and rats [109] undergoing transient middle cerebral artery occlusion. Additionally, OXT can promote angiogenesis, regeneration of cardiomyocytes, resistance to apoptosis and cardiac fibrosis [58]. For these reasons, OXT is able to increase the integrity of endothelial cells and thus, reduce the occurrence of thromboembolism. Furthermore, OXT significantly suppresses oxidized low-density lipoprotein-induced attachment of spontaneously immortalized THP-1 monocytes to human brain microvascular endothelial cells by reducing the expression of adhesion molecules, such as vascular cell adhesion protein 1 and E-selectin [110]. Thus, OXT carries the capacity to protect CV functions at all levels from COVID-19 injuries.
5.5. Weakening pre-existing comorbidities of COVID-19
Many COVID-19 patients have pre-existing comorbidities including hypertension, CV disease, endocrine disorder and diabetes. These comorbidities, particularly CV diseases, increase the morbidity and mortality of COVID-19 [3,11,104,111]. Inhibiting or improving these pre-existing conditions is important for reducing COVID-19 morbidity and mortality and thus, we further discuss the role of OXT in stabilization of these conditions.
5.5.1. Prevention of obesity, atherosclerosis and their complications
Metabolic and immunological disorders are essential etiologies underlying obesity, atherosclerosis and the associated diseases, such as hypertension, coronary artery disease and ischemic stroke [112]. OXT does not only carry anti-inflammatory effects as stated above but also reduces hyperlipidemia, an essential metabolic disorder for obesity and atherosclerosis. As reported, serum OXT levels were low in obese person [113]; OXT- or OTR-deficient mice developed late-onset obesity [114]. In response to subcutaneous injection of OXT, cholesterol levels decreased in rats [115]. Chronic OXT application also reduced weight gain in diet-induced obese mice and rats; chronic subcutaneous or intranasal OXT treatment was sufficient to elicit body weight loss in obese humans [116]. Thus, OXT may reduce individual susceptibility to COVID-19 by suppressing obesity, atherosclerosis and its complications.
5.5.2. Prevention of hypertension
OXT has the potential to inhibit the development of hypertension. In hypertensive rats, OXT and OXT mRNA expressions are reduced in the hypothalamus whereas, OXT injected subcutaneously or intracerebroventricularly for 5 days can significantly decrease blood pressure in rats [117]. Centrally released OXT can significantly reduce blood pressure and heart rate and antagonize acute stress-induced CV responses [118]. Moreover, chronic activation of OXT neurons restores the release of OXT from PVN neural fibers in the dorsal motor nucleus of the vagus, and prevents chronic intermittent hypoxia-evoked hypertension [119]. Based on these findings, we believe that OXT can exert anti-hypertensive effects through both central and peripheral approaches.
5.5.3. Anti-diabetic effects
Population with diabetes mellitus has high prevalence of CV diseases [120] and high susceptibility to COVID-19. In patients with type 2 diabetes mellitus, serum OXT levels are relatively low [113]. In a mouse model of type 2 diabetes mellitus, OXT, OTRs, ANP, and endothelial NO synthase gene expressions in the heart are low [121]. In fasting men, intranasal OXT application can attenuate the peak excursion of plasma glucose [122]. Mechanistically, OTR signaling attenuates the death of beta cells in pancreatic islets exposed to cytotoxic stresses [123] and increases beta-cell response to the glucose challenge [122]. Furthermore, OXT can promote glucose uptake in cultured cardiomyocytes from newborn and adult rats [124], in myocardial cells during hypoxia [125] and in mesenchymal stem cells [126]. These facts indicate that OXT plays a significant role in the regulation of glucose metabolism and can prevent development and progression of diabetes mellitus. Thus, OXT could be used to reduce COVID-19 morbidity through glycemic control and stabilization.
5.5.4. Anti-social stress
Anxiety and panic are common responses in COVID-19 patients especially with ARDS [127]. OXT can reduce the expression of panic-related behaviors (e.g. fear and escape) by acting on the medial amygdala and the dorsal periaqueductal gray as previously reviewed [51]. OXT is positively associated with diminished stress among securely attached participants and can attenuate perception of stress due to adverse life events in old age [128]. In stroke, social pairing in adult mice enhances hypothalamic OXT gene expression and leads to smaller infarct size by reducing neuroinflammation and oxidative stress [112]. OXT can reduce stress-elicited neuroendocrine, autonomic, and behavioral responses [53], which can be used to prevent the brain-heart syndrome [129]. In addition, OXT also has anti-depressive effects [130] and may be used to relieve social isolation-associated depression in COVID-19 patients [48]. Thus, OXT/OTR signaling contributes to cardio-protection through diverse approaches in COVID-19.
6. Further questions about CV protective potential of OXT in COVID-19
In prevention and treatment of COVID-19, particularly the CV complications, OXT or OTR agonist is an ideal candidate for clinical trials. The following questions should be considered before its trials in management of COVID-19.
6.1. Platelet aggregation and thrombosis
Some observations suggest that OXT can promote platelet aggregation. For example, inducing labor by using OXT tends to increase platelet aggregation and decrease disaggregation [131]; synthetic polyphosphate can inhibit OXT-induced platelet aggregation by reducing platelet Ca2+ levels and inhibiting the thromboxane A2 signaling pathway [132]. Notably, OXT has different effects on ADP-induced platelet aggregation at different reproductive states. That is, the aggregation is potentiated at low (<200 nM) and inhibited at high (>400 nM) ADP concentrations in nonpregnant women; in pregnant women, OXT does not modulate ADP-induced platelet aggregation [133]. Since most of these observations are associated with parturition and depending on the doses of OXT applied, the pro-coagulation effect of OXT may facilitate homeostasis during parturition. In contrast, OXT generally protects endothelial cells from immunological injuries [110], increases anticoagulant heparan sulphate stability, and reduces adhesion molecules [110], and thus could reduce the formation of thrombosis. These facts support the possibility that OXT may reduce the pro-thrombotic state in non-pregnant COVID-19 patients.
6.2. Biomarker or inducer of CV injuries
While OXT exhibits extensive protective potentials for CV complications in COVID-19, there are some observations showing controversial results about OXT protection of CV system. For example, left coronary artery ligation-induced myocardial infarction in rats increased OXT release, which was inhibited by melatonin [134], an agent having CV protective function. Acute myocardial infarction can increase brain release of OXT in the PVN which mediates sympatho-excitatory responses and the production of proinflammatory cytokines in rats [135]. Various stressors can provoke sudden CV effects and trigger the release of OXT in rats. However, administration of exogenous OXT on the ischemic-reperfused isolated heart of rats still reduced infarct size, levels of creatine kinase MB isoenzyme and lactate dehydrogenase in coronary effluent, and severity of ischemia and reperfusion injury [136]. As for the inhibition of OXT level by the CV protective melatonin [137], this effect is likely due to dose-dependent effects of melatonin on OXT secretion [70]. Taken together, these findings support that OXT is critical in immunologic surveillance [50] and the increased OXT levels function as a compensatory reaction to the acute hypoxia injury.
Another question is whether cardiac-specific over-expression of OTR is beneficial to myocardial functions. In mice with cardiac-specific over-expression of OTR, the left ventricular function was reduced, end-diastolic volume was larger and mortality was high along with cardiac fibrosis, atrial thrombus, and increased expression of pro-fibrogenic genes [138]. Interestingly, in rat dams separated from her pups, hypothalamic OXT secretion increased; however, plasma OXT levels were low. This is due to that over-activation of OTR signaling causes post-excitation inhibition and a subsequent reduction of the secretory activity of OXT neurons [130,139]. Thus, it is possible that over-expression or over-activation of OTR exerts an effect opposite to the physiological effect although normal OTR signaling is essential for CV protection.
The third question is about the effect of OXT on renin secretion. In denervated kidney, direct infusion of OXT can significantly increase renin secretion from the kidneys in association with the activation of β-adrenoceptors [140]. This effect is likely due to that the denervation of sympathetic nerve increases the expression of β-adrenoceptors or its sensitivity to adrenergic agonist, which amplifies a weaker OXT effects on renin secretion without the antagonism of secondary ANP release from the atrium and inhibition of sympathetic outflows from the PVN by OXT in vivo. As a whole, OXT increase during acute CV injuries can be viewed as a biomarker of cardiac stresses and a compensatory reaction of body defense system, but not an inducer of CV injuries.
6.3. The characteristics of OXT effects on the CV system
OXT in the blood could differentially modulate CV activities, depending on the amount and pattern of its secretion from the posterior pituitary. This is because the effects of OXT on its targets are time-, dose- and pattern-dependent [141]. That is, longer and higher dose of OXT stimulation can cause inhibition of target cells following initial excitation [142,143]; OXT released in bolus can cause stronger response than the same amount of OXT in lower dose for longer time [144]. In the CV system, the same characteristics are also present. For example, in isolated hearts, 10−7 M OXT caused 10% reduction of left ventricle pressure, but 10−10 to 10−8 M concentrations had no effects while diastolic pressure raised and heart rate fell only with 10−5 M OXT [47]. Relative to the physiological levels of plasma OXT in 10−12 to 10−11 M [145,146], the negative ionotropic effects of OXT are clearly based on pharmacological mechanism. For example, the pressor effect of high-dose OXT is actually mediated by vasopressin receptors [147]. Under different conditions, expressions of OXT and OTR are also different [43], which make OXT effects on CV system different following changes in CV activity, such as the effect of cardiac-specific over-expression of OTR in mice [138].
In addition, the action of OXT is tissue-specific. In isolated dog carotid arteries, OXT causes vasoconstriction of human basilar artery [41], likely by acting on smooth muscle cells [148]. It is also reported that OXT in 10−11–10−7 M levels causes relatively weaker contraction of the basilar artery compared to vasopressin and has no effects on mesenteric arteries in rats [149]. Intramuscular injection of OXT decreases uterine blood flow during uterine contractions in puerperal dairy cows [150], which occurs at postpartum day 2 but not day 5. Lastly, patterns of OXT actions are also important for its effect. For example, pulsatile but not tonic secretion of OXT plays the role of anti-precancerous lesions of the mammary glands in rat dams [151]; social desirability and milk transferred from the mother to the baby are largely dependent on OXT pulsatility [152]. Thus, in application of OXT, it is necessary to consider the location of target tissue- and organ-specific effect, the time- and dose-dependent effects as well as the patterns of application.
7. Strategies of OXT application in COVID-19
7.1. Clinical trials of OXT in COVID-19 patients
In management of COVID-19, many drugs have been considered, such as ACE2 blockers, anti-inflammatory drugs, antibodies against IL-1 and anti-IL-6, remdesivir, dexamethasone, hydroxychloroquine and vaccines [153]. In clinical studies, their efficacies are either under further evaluation or proved to be ineffective. For instance, COVID-19 patients' sera have only limited cross-neutralization [26], suggesting that recovery from one infection might not protect against the other. In patients with COVID-19, the hypothalamic-pituitary-adrenocortical axis is likely inhibited as stated above, which makes corticosteroid critical for prevention of cytokine storm [154]; however, corticosteroid delays virus clearing but does not convincingly improve survival or reduce hospitalization duration and the use of mechanical ventilation [155]. Hydroxychloroquine previously used to treat COVID-19 is known to prolong the QT interval and can have a proarrhythmic propensity [36]. Right now it is not given to COVID-19 patients as it is considered to be ineffective [156,157]. Lastly, COVID-19 vaccines have been developed extensively; however, their safety and efficacy profiles remain to be established. Notably, at 6 months after acute infection, COVID-19 survivors are troubled with fatigue, sleep difficulties, anxiety or depression and even severe impaired pulmonary diffusion capacities and abnormal finding on pulmonary imaging tests [158]. So far, there are currently no known specifically effective treatments for COVID-19. OXT, with broad-spectrum anti-COVID-19 potential, is relatively safe compared to antiviral drugs and corticosteroids, and inexpensive relative to antiviral therapies [3,111], and thus is worthy of clinical trial to validate its efficiency.
In treatment of CV diseases, many approaches of applying OXT have been proposed [7]. These considerations are also viable for treating CV complications in COVID-19. In brief, OXT or its agonists can be used exogenously by intravenous infusion, intramuscular injection or intranasal application. OXT can be used in a continuous low dose to minimize potential CV disturbances, or in bolus to maximize the efficiency without desensitizing OTR. OXT can also be used in nasal approach to activate the OXT-secreting system. Special caution should be taken for pregnant women around parturition, particularly for those who are allergic to OXT or have high basal AT II levels. As reported, acute systemic administration of low-dose OXT exerts a protective role; however, chronic subcutaneous administration of low-dose OXT (20 or 100 ng/kg/h, for 28 days) can enforce AT II-induced hypertension, cardiac hypertrophy, and renal damage in rats although OXT itself does not have these effects [159]. Future laboratory study and clinical trial of OXT usages are critical in translating its therapeutic potentials into reality, not only for COVID-19 but also for other unexpected viral infections.
7.2. Prevention of COVID-19 through mobilization of endogenous OXT
Besides the needs for treatment, prevention of COVID-19 is another irreplaceable potential of OXT. Human-to-human transmission via droplets and contaminated surfaces has been described, from both symptomatic and asymptomatic individuals. Correspondingly, wearing mask and social distance have been adopted as common prevention measures. While these measures are effective in reducing COVID-19 morbidity, they also raise concerns for massive social isolation and long-term psychological effects [160] that decrease immunological defense [50,51] and thus can increase COVID-19 susceptibility. It is known that social isolation can cause CV complications [161] while disrupting normal OXT secretion [[162], [163], [164]]. Importantly, application of OXT can alleviate social isolation-induced atherosclerosis and adipose tissue inflammation [165]. Currently, newly developed specific vaccines face the challenge of continued mutations in SARS-CoV-2 and immune evasion [166,167] while complete social isolation is difficult to achieve, which leave majority of the population remaining under the threats of COVID-19. Thus, strengthening the basic immunologic function is a measure that universally fits our human society, particularly for aged population who have reduced immunologic function and OXT production [168]. For this purpose, mobilization of endogenous OXT can be an optimal approach to improve our resistance to COVID-19 by blocking SARS-CoV-2 entry, reducing viral load, and reducing pre-existing comorbidities.
OXT is a “social hormone” and can be elicited by many types of healthy behavior via increasing vagal inputs and by conditioned reflex, such as massage [169], listening to music [170], brief mindfulness session [171], light therapy [134], and physical exercise [172,173]. Although the integrity of our human society can be disrupted by self-isolation and social distancing, mobilizing endogenous OXT functions can help us to rebuild it by giving individuals of high immunologic defense capacity and lower susceptibility to COVID-19 in the "re-opening."
8. Conclusion
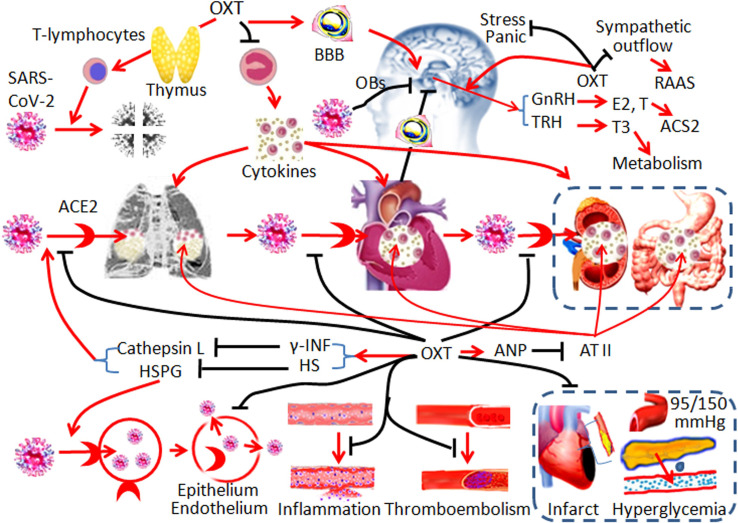
CV protective potentials of OXT allow reversal of lymphocytopenia, suppression of cytokine storm, prevention of ARDS, and reversal of multiple organ failures. While blocking SAR-CoV-2 infection, OXT also has the potential to treat CV complications in COVID-19 patients through multiple mechanisms (Fig. 2 ). For this reason, we should repurpose OXT for treatment and prevention of COVID-19.
Fig. 2.
Hypothetic mechanisms underlying the cardiovascular protection by OXT in COVID-19. The diagrams show the targets of OXT on different links of the pathogenesis. Black stop lines indicate inhibition or reduction; red arrows show activation or promotion. Other annotations refer to Fig. 1. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
CRediT authorship contribution statement
SCW conceived the study and wrote the first draft; YFW conceived the study and made the final revision.
Declaration of competing interest
The authors declare that they have no competing interests.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 31471113, YFW), the fund of “Double-First-Class” Construction of Harbin Medical University (key laboratory of preservation of human genetic resources and disease control in China). SCW is a Cardiology fellow of the University of California-Irvine.
References
- 1.Libby P., Luscher T. COVID-19 is, in the end, an endothelial disease. Eur. Heart J. 2020;41:3038–3044. doi: 10.1093/eurheartj/ehaa623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alharthy A., Faqihi F., Memish Z.A., Karakitsos D. Fragile endothelium and brain dysregulated neurochemical activity in COVID-19. ACS Chem. Neurosci. 2020;11:2159–2162. doi: 10.1021/acschemneuro.0c00437. [DOI] [PubMed] [Google Scholar]
- 3.Zhang J., Zhou L., Yang Y., Peng W., Wang W., Chen X. Therapeutic and triage strategies for 2019 novel coronavirus disease in fever clinics. Lancet Respir. Med. 2020;8:e11–e12. doi: 10.1016/S2213-2600(20)30071-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barlow A., Landolf K.M., Barlow B., Yeung S.Y.A., Heavner J.J., Claassen C.W., Heavner M.S. Review of emerging pharmacotherapy for the treatment of coronavirus disease 2019. Pharmacotherapy. 2020;40:416–437. doi: 10.1002/phar.2398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maciorowski D., Idrissi S.Z.E., Gupta Y., Medernach B.J., Burns M.B., Becker D.P., Durvasula R., Kempaiah P. A review of the preclinical and clinical efficacy of remdesivir, hydroxychloroquine, and lopinavir-ritonavir treatments against COVID-19. SLAS Discovery. 2020;25:1108–1122. doi: 10.1177/2472555220958385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yang H.P., Wang L., Han L., Wang S.C. Nonsocial functions of hypothalamic oxytocin. ISRN Neurosci. 2013;2013:179272. doi: 10.1155/2013/179272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang P., Wang S.C., Yang H., Lv C., Jia S., Liu X., Wang X., Meng D., Qin D., Zhu H., Wang Y.F. Therapeutic potential of oxytocin in atherosclerotic cardiovascular disease: mechanisms and signaling pathways. Front. Neurosci. 2019;13:454. doi: 10.3389/fnins.2019.00454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Duerr G.D., Heine A., Hamiko M., Zimmer S., Luetkens J.A., Nattermann J., Rieke G., Isaak A., Jehle J., Held S.A.E., Wasmuth J.C., Wittmann M., Strassburg C.P., Brossart P., Coburn M., Treede H., Nickenig G., Kurts C., Velten M. Parameters predicting COVID-19-induced myocardial injury and mortality. Life Sci. 2020;260:118400. doi: 10.1016/j.lfs.2020.118400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Luetkens J.A., Isaak A., Zimmer S., Nattermann J., Sprinkart A.M., Boesecke C., Rieke G.J., Zachoval C., Heine A., Velten M., Duerr G.D. Diffuse myocardial inflammation in COVID-19 associated myocarditis detected by multiparametric cardiac magnetic resonance imaging. Circulation Cardiovascular imaging. 2020;13 doi: 10.1161/CIRCIMAGING.120.010897. [DOI] [PubMed] [Google Scholar]
- 10.Lindner D., Fitzek A., Brauninger H., Aleshcheva G., Edler C., Meissner K., Scherschel K., Kirchhof P., Escher F., Schultheiss H.P., Blankenberg S., Puschel K., Westermann D. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020;5:1281–1285. doi: 10.1001/jamacardio.2020.3551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Madjid M., Safavi-Naeini P., Solomon S.D., Vardeny O. Potential effects of coronaviruses on the cardiovascular system: a review. JAMA Cardiol. 2020;5:831–840. doi: 10.1001/jamacardio.2020.1286. [DOI] [PubMed] [Google Scholar]
- 12.Gebhard C., Regitz-Zagrosek V., Neuhauser H.K., Morgan R., Klein S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex Differ. 2020;11:29. doi: 10.1186/s13293-020-00304-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bennett S., Tafuro J., Mayer J., Darlington D., Wai Wong C., Muntean E.A., Wong N., Mallen C., Shing Kwok C. Clinical features and outcomes of adults with COVID-19: a systematic review and pooled analysis of the literature. International journal of clinical practice. 2020 doi: 10.1111/ijcp.13725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bansal M. Cardiovascular disease and COVID-19. Diabetes & metabolic syndrome. 2020;14:247–250. doi: 10.1016/j.dsx.2020.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huang D., Lian X., Song F., Ma H., Lian Z., Liang Y., Qin T., Chen W., Wang S. Clinical features of severe patients infected with 2019 novel coronavirus: a systematic review and meta-analysis. Annals of translational medicine. 2020;8:576. doi: 10.21037/atm-20-2124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guedj E., Million M., Dudouet P., Tissot-Dupont H., Bregeon F., Cammilleri S., Raoult D. (18)F-FDG brain PET hypometabolism in post-SARS-CoV-2 infection: substrate for persistent/delayed disorders? European journal of nuclear medicine and molecular imaging. 2020;48:592–595. doi: 10.1007/s00259-020-04973-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020;71:762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Abdin S.M., Elgendy S.M., Alyammahi S.K., Alhamad D.W., Omar H.A. Tackling the cytokine storm in COVID-19, challenges and hopes. Life Sci. 2020;257:118054. doi: 10.1016/j.lfs.2020.118054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Conti P., Caraffa A., Gallenga C.E., Ross R., Kritas S.K., Frydas I., Younes A., Di Emidio P., Ronconi G., Toniato E. IL-1 induces throboxane-A2 (TxA2) in COVID-19 causing inflammation and micro-thrombi: inhibitory effect of the IL-1 receptor antagonist (IL-1Ra) J. Biol. Regul. Homeost. Agents. 2020;34:1623–1627. doi: 10.23812/20-34-4EDIT-65. [DOI] [PubMed] [Google Scholar]
- 20.Liu Y., Pang Y., Hu Z., Wu M., Wang C., Feng Z., Mao C., Tan Y., Liu Y., Chen L., Li M., Wang G., Yuan Z., Diao B., Wu Y., Chen Y. Thymosin alpha 1 (Talpha1) reduces the mortality of severe COVID-19 by restoration of lymphocytopenia and reversion of exhausted T cells. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 2020;71:2150–2157. doi: 10.1093/cid/ciaa630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Breidenbach J.D., Dube P., Ghosh S., Abdullah B.N., Modyanov N.N., Malhotra D., Dworkin L.D., Haller S.T., Kennedy D.J. Impact of comorbidities on SARS-CoV-2 viral entry-related genes. Journal of personalized medicine. 2020;10 doi: 10.3390/jpm10040146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. The Journal of pathology. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Annweiler C., Cao Z., Wu Y., Faucon E., Mouhat S., Kovacic H., Sabatier J.M. Counter-regulatory ‘renin-angiotensin’ system-based candidate drugs to treat COVID-19 diseases in SARS-CoV-2-infected patients. Infectious disorders drug targets. 2020;20:407–408. doi: 10.2174/1871526520666200518073329. [DOI] [PubMed] [Google Scholar]
- 24.Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280 e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.West A.P., Jr., Scharf L., Scheid J.F., Klein F., Bjorkman P.J., Nussenzweig M.C. Structural insights on the role of antibodies in HIV-1 vaccine and therapy. Cell. 2014;156:633–648. doi: 10.1016/j.cell.2014.01.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ou X., Liu Y., Lei X., Li P., Mi D., Ren L., Guo L., Guo R., Chen T., Hu J., Xiang Z., Mu Z., Chen X., Chen J., Hu K., Jin Q., Wang J., Qian Z. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020;11:1620. doi: 10.1038/s41467-020-15562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thunders M., Delahunt B. Gene of the month: TMPRSS2 (transmembrane serine protease 2) J. Clin. Pathol. 2020;73:773–776. doi: 10.1136/jclinpath-2020-206987. [DOI] [PubMed] [Google Scholar]
- 28.Zhang Q., Chen C.Z., Swaroop M., Xu M., Wang L., Lee J., Pradhan M., Shen M., Luo Z., Xu Y., Huang W., Zheng W., Ye Y. Targeting heparan sulfate proteoglycan-assisted endocytosis as a COVID-19 therapeutic option. bioRxiv: the preprint server for biology. 2020 doi: 10.1101/2020.07.14.202549. [DOI] [Google Scholar]
- 29.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J., Wang Z., Li J., Li J., Feng C., Zhang Z., Wang L., Peng L., Chen L., Qin Y., Zhao D., Tan S., Yin L., Xu J., Zhou C., Jiang C., Liu L. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci. China Life Sci. 2020;63:364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Khan A., Benthin C., Zeno B., Albertson T.E., Boyd J., Christie J.D., Hall R., Poirier G., Ronco J.J., Tidswell M., Hardes K., Powley W.M., Wright T.J., Siederer S.K., Fairman D.A., Lipson D.A., Bayliffe A.I., Lazaar A.L. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit. Care. 2017;21:234. doi: 10.1186/s13054-017-1823-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Azevedo R.B., Botelho B.G., Hollanda J.V.G., Ferreira L.V.L., Junqueira de Andrade L.Z., Oei S., Mello T.S., Muxfeldt E.S. Covid-19 and the cardiovascular system: a comprehensive review. J. Hum. Hypertens. 2020;35:4–11. doi: 10.1038/s41371-020-0387-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ahmed S., Zimba O., Gasparyan A.Y. Thrombosis in Coronavirus disease 2019 (COVID-19) through the prism of Virchow’s triad. Clin. Rheumatol. 2020;39:2529–2543. doi: 10.1007/s10067-020-05275-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Del Turco S., Vianello A., Ragusa R., Caselli C., Basta G. COVID-19 and cardiovascular consequences: is the endothelial dysfunction the hardest challenge? Thromb. Res. 2020;196:143–151. doi: 10.1016/j.thromres.2020.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bojkova D., Wagner J.U.G., Shumliakivska M., Aslan G.S., Saleem U., Hansen A., Luxan G., Gunther S., Pham M.D., Krishnan J., Harter P.N., Ermel U.H., Frangakis A.S., Milting H., Zeiher A.M., Klingel K., Cinatl J., Dendorfer A., Eschenhagen T., Tschope C., Ciesek S., Dimmeler S. SARS-CoV-2 infects and induces cytotoxic effects in human cardiomyocytes. Cardiovasc. Res. 2020;116:2207–2215. doi: 10.1093/cvr/cvaa267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen L., Li X., Chen M., Feng Y., Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020;116:1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kochi A.N., Tagliari A.P., Forleo G.B., Fassini G.M., Tondo C. Cardiac and arrhythmic complications in patients with COVID-19. J. Cardiovasc. Electrophysiol. 2020;31:1003–1008. doi: 10.1111/jce.14479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bader F., Manla Y., Atallah B., Starling R.C. Heart failure and COVID-19. Heart Fail. Rev. 2020;26:1–10. doi: 10.1007/s10741-020-10008-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C., Hohmann E., Chu H.Y., Luetkemeyer A., Kline S., Lopez de Castilla D., Finberg R.W., Dierberg K., Tapson V., Hsieh L., Patterson T.F., Paredes R., Sweeney D.A., Short W.R., Touloumi G., Lye D.C., Ohmagari N., Oh M.D., Ruiz-Palacios G.M., Benfield T., Fatkenheuer G., Kortepeter M.G., Atmar R.L., Creech C.B., Lundgren J., Babiker A.G., Pett S., Neaton J.D., Burgess T.H., Bonnett T., Green M., Makowski M., Osinusi A., Nayak S., Lane H.C., Members A.-S.G. Remdesivir for the treatment of Covid-19 - final report. N. Engl. J. Med. 2020;383:1813–1826. doi: 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Johnson Z.V., Young L.J. Oxytocin and vasopressin neural networks: implications for social behavioral diversity and translational neuroscience. Neurosci. Biobehav. Rev. 2017;76:87–98. doi: 10.1016/j.neubiorev.2017.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zimmerman E.A., Nilaver G., Hou-Yu A., Silverman A.J. Vasopressinergic and oxytocinergic pathways in the central nervous system. Fed. Proc. 1984;43:91–96. [PubMed] [Google Scholar]
- 41.Abrams G.M., Nilaver G., Recht L.R., Haldar J., Zimmerman E.A. Hypothalamic oxytocin: a cerebrovascular modulator in man? Neurology. 1985;35:1046–1049. doi: 10.1212/wnl.35.7.1046. [DOI] [PubMed] [Google Scholar]
- 42.Jankowski M., Wang D., Hajjar F., Mukaddam-Daher S., McCann S.M., Gutkowska J. Oxytocin and its receptors are synthesized in the rat vasculature. Proc. Natl. Acad. Sci. U. S. A. 2000;97:6207–6211. doi: 10.1073/pnas.110137497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wsol A., Kasarello K., Kuch M., Gala K., Cudnoch-Jedrzejewska A. Increased activity of the intracardiac oxytocinergic system in the development of postinfarction heart failure. Biomed. Res. Int. 2016;2016 doi: 10.1155/2016/3652068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhu J., Wang H., Zhang X., Xie Y. Regulation of angiogenic behaviors by oxytocin receptor through Gli1-indcued transcription of HIF-1alpha in human umbilical vein endothelial cells. Biomed. Pharmacother. 2017;90:928–934. doi: 10.1016/j.biopha.2017.04.021. [DOI] [PubMed] [Google Scholar]
- 45.Mukaddam-Daher S., Yin Y.L., Roy J., Gutkowska J., Cardinal R. Negative inotropic and chronotropic effects of oxytocin. Hypertension. 2001;38:292–296. doi: 10.1161/01.hyp.38.2.292. [DOI] [PubMed] [Google Scholar]
- 46.Michelini L.C., Marcelo M.C., Amico J., Morris M. Oxytocinergic regulation of cardiovascular function: studies in oxytocin-deficient mice. Am. J. Physiol. Heart Circ. Physiol. 2003;284:H2269–H2276. doi: 10.1152/ajpheart.00774.2002. [DOI] [PubMed] [Google Scholar]
- 47.Costa E.S.R.H., Pereira-Junior P.P., Oliveira P.F., Olivares E.L., Werneck-de-Castro J.P., Mello D.B., Nascimento J.H., Campos-de-Carvalho A.C. Cardiac effects of oxytocin: is there a role for this peptide in cardiovascular homeostasis? Regul. Pept. 2005;132:107–112. doi: 10.1016/j.regpep.2005.09.011. [DOI] [PubMed] [Google Scholar]
- 48.Grinevich V., Neumann I.D. Brain oxytocin: how puzzle stones from animal studies translate into psychiatry. Mol. Psychiatry. 2020;26:265–279. doi: 10.1038/s41380-020-0802-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Thomas PA, Kim S. Lost touch? Implications of physical touch for physical health. J. Gerontol. B Psychol. Sci. Soc. Sci. 2020:gbaa134. doi: 10.1093/geronb/gbaa134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang P., Yang H.P., Tian S., Wang L., Wang S.C., Zhang F., Wang Y.F. Oxytocin-secreting system: a major part of the neuroendocrine center regulating immunologic activity. J. Neuroimmunol. 2015;289:152–161. doi: 10.1016/j.jneuroim.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 51.Wang Y.-F. Center role of the oxytocin-secreting system in neuroendocrine-immune network revisited. Journal of Clinical & Experimental Neuroimmunology. 2016;1:102. [Google Scholar]
- 52.Ishii H., Amano T., Matsubara T., Murohara T. Pharmacological intervention for prevention of left ventricular remodeling and improving prognosis in myocardial infarction. Circulation. 2008;118:2710–2718. doi: 10.1161/CIRCULATIONAHA.107.748772. [DOI] [PubMed] [Google Scholar]
- 53.De Melo V.U., Saldanha R.R., Dos Santos C.R., De Campos Cruz J., Lira V.A., Santana-Filho V.J., Michelini L.C. Ovarian hormone deprivation reduces oxytocin expression in paraventricular nucleus preautonomic neurons and correlates with baroreflex impairment in rats. Front. Physiol. 2016;7:461. doi: 10.3389/fphys.2016.00461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Viero C., Shibuya I., Kitamura N., Verkhratsky A., Fujihara H., Katoh A., Ueta Y., Zingg H.H., Chvatal A., Sykova E., Dayanithi G. Review: oxytocin: crossing the bridge between basic science and pharmacotherapy. CNS Neurosci Ther. 2010;16:e138–e156. doi: 10.1111/j.1755-5949.2010.00185.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jankowski M., Broderick T.L., Gutkowska J. Oxytocin and cardioprotection in diabetes and obesity. BMC Endocr. Disord. 2016;16:34. doi: 10.1186/s12902-016-0110-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jankowski M., Bissonauth V., Gao L., Gangal M., Wang D., Danalache B., Wang Y., Stoyanova E., Cloutier G., Blaise G., Gutkowska J. Anti-inflammatory effect of oxytocin in rat myocardial infarction. Basic Res. Cardiol. 2010;105:205–218. doi: 10.1007/s00395-009-0076-5. [DOI] [PubMed] [Google Scholar]
- 57.Kobayashi H., Yasuda S., Bao N., Iwasa M., Kawamura I., Yamada Y., Yamaki T., Sumi S., Ushikoshi H., Nishigaki K., Takemura G., Fujiwara T., Fujiwara H., Minatoguchi S. Postinfarct treatment with oxytocin improves cardiac function and remodeling via activating cell-survival signals and angiogenesis. J. Cardiovasc. Pharmacol. 2009;54:510–519. doi: 10.1097/FJC.0b013e3181bfac02. [DOI] [PubMed] [Google Scholar]
- 58.Garrott K., Dyavanapalli J., Cauley E., Dwyer M.K., Kuzmiak-Glancy S., Wang X., Mendelowitz D., Kay M.W. Chronic activation of hypothalamic oxytocin neurons improves cardiac function during left ventricular hypertrophy-induced heart failure. Cardiovasc. Res. 2017;113:1318–1328. doi: 10.1093/cvr/cvx084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Moghimian M., Faghihi M., Karimian S.M., Imani A., Houshmand F., Azizi Y. Role of central oxytocin in stress-induced cardioprotection in ischemic-reperfused heart model. J. Cardiol. 2013;61:79–86. doi: 10.1016/j.jjcc.2012.08.021. [DOI] [PubMed] [Google Scholar]
- 60.Gutkowska J., Jankowski M. Oxytocin revisited: it is also a cardiovascular hormone. J Am Soc Hypertens. 2008;2:318–325. doi: 10.1016/j.jash.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 61.Cassoni P., Marrocco T., Bussolati B., Allia E., Munaron L., Sapino A., Bussolati G. Oxytocin induces proliferation and migration in immortalized human dermal microvascular endothelial cells and human breast tumor-derived endothelial cells. Molecular cancer research: MCR. 2006;4:351–359. doi: 10.1158/1541-7786.MCR-06-0024. [DOI] [PubMed] [Google Scholar]
- 62.Gutnick A., Blechman J., Kaslin J., Herwig L., Belting H.G., Affolter M., Bonkowsky J.L., Levkowitz G. The hypothalamic neuropeptide oxytocin is required for formation of the neurovascular interface of the pituitary. Dev. Cell. 2011;21:642–654. doi: 10.1016/j.devcel.2011.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Furube E., Mannari T., Morita S., Nishikawa K., Yoshida A., Itoh M., Miyata S. VEGF-dependent and PDGF-dependent dynamic neurovascular reconstruction in the neurohypophysis of adult mice. J. Endocrinol. 2014;222:161–179. doi: 10.1530/JOE-14-0075. [DOI] [PubMed] [Google Scholar]
- 64.Kliment C.R., Tobolewski J.M., Manni M.L., Tan R.J., Enghild J., Oury T.D. Extracellular superoxide dismutase protects against matrix degradation of heparan sulfate in the lung. Antioxid. Redox Signal. 2008;10:261–268. doi: 10.1089/ars.2007.1906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Light K.C., Grewen K.M., Amico J.A., Brownley K.A., West S.G., Hinderliter A.L., Girdler S.S. Oxytocinergic activity is linked to lower blood pressure and vascular resistance during stress in postmenopausal women on estrogen replacement. Horm. Behav. 2005;47:540–548. doi: 10.1016/j.yhbeh.2004.12.010. [DOI] [PubMed] [Google Scholar]
- 66.Elabd C., Cousin W., Upadhyayula P., Chen R.Y., Chooljian M.S., Li J., Kung S., Jiang K.P., Conboy I.M. Oxytocin is an age-specific circulating hormone that is necessary for muscle maintenance and regeneration. Nat. Commun. 2014;5:4082. doi: 10.1038/ncomms5082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Quinones-Jenab V., Jenab S., Ogawa S., Adan R.A., Burbach J.P., Pfaff D.W. Effects of estrogen on oxytocin receptor messenger ribonucleic acid expression in the uterus, pituitary, and forebrain of the female rat. Neuroendocrinology. 1997;65:9–17. doi: 10.1159/000127160. [DOI] [PubMed] [Google Scholar]
- 68.Jankowski M., Wang D., Danalache B., Gangal M., Gutkowska J. Cardiac oxytocin receptor blockade stimulates adverse cardiac remodeling in ovariectomized spontaneously hypertensive rats. Am. J. Physiol. Heart Circ. Physiol. 2010;299:H265–H274. doi: 10.1152/ajpheart.00487.2009. [DOI] [PubMed] [Google Scholar]
- 69.Bahrampour Juybari K., Pourhanifeh M.H., Hosseinzadeh A., Hemati K., Mehrzadi S. Melatonin potentials against viral infections including COVID-19: current evidence and new findings. Virus Res. 2020;287:198108. doi: 10.1016/j.virusres.2020.198108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Juszczak M., Wolak M., Bojanowska E., Piera L., Roszczyk M. The role of melatonin membrane receptors in melatonin-dependent oxytocin secretion from the rat hypothalamo-neurohypophysial system - an in vitro and in vivo approach. Endokrynol Pol. 2016;67:507–514. doi: 10.5603/EP.a2016.0035. [DOI] [PubMed] [Google Scholar]
- 71.Pascual-Goni E., Fortea J., Martinez-Domeno A., Rabella N., Tecame M., Gomez-Oliva C., Querol L., Gomez-Anson B. COVID-19-associated ophthalmoparesis and hypothalamic involvement. Neurology(R) Neuroimmunology & Neuroinflammation. 2020;7:e823. doi: 10.1212/NXI.0000000000000823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang Y.F., Negoro H., Higuchi T. Lesions of hypothalamic mammillary body desynchronise milk-ejection bursts of rat bilateral supraoptic oxytocin neurones. J. Neuroendocrinol. 2013;25:67–75. doi: 10.1111/j.1365-2826.2012.02368.x. [DOI] [PubMed] [Google Scholar]
- 73.Han Y., Sun H.J., Li P., Gao Q., Zhou Y.B., Zhang F., Gao X.Y., Zhu G.Q. Angiotensin-(1–7) in paraventricular nucleus modulates sympathetic activity and cardiac sympathetic afferent reflex in renovascular hypertensive rats. PLoS One. 2012;7 doi: 10.1371/journal.pone.0048966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sriramula S., Xia H., Xu P., Lazartigues E. Brain-targeted angiotensin-converting enzyme 2 overexpression attenuates neurogenic hypertension by inhibiting cyclooxygenase-mediated inflammation. Hypertension. 2015;65:577–586. doi: 10.1161/HYPERTENSIONAHA.114.04691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z., Zhuang H., Wu B., Zhong H., Shao H., Fang W., Gao D., Pei F., Li X., He Z., Xu D., Shi X., Anderson V.M., Leong A.S. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Langford D., Baron D., Joy J., Del Valle L., Shack J. Contributions of HIV infection in the hypothalamus and substance abuse/use to HPT dysregulation. Psychoneuroendocrinology. 2011;36:710–719. doi: 10.1016/j.psyneuen.2010.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cayan S, Uguz M, Saylam B, Akbay Effect of serum total testosterone and its relationship with other laboratory parameters on the prognosis of coronavirus disease 2019 (COVID-19) in SARS-CoV-2 infected male patients: a cohort study. Aging Male. 2020:1–11. doi: 10.1080/13685538.2020.1807930. [DOI] [PubMed] [Google Scholar]
- 78.Chen M, Zhou W, Xu W. Thyroid function analysis in 50 patients with COVID-19: a retrospective study. Thyroid. 2020;31:8–11. doi: 10.1089/thy.2020.0363. [DOI] [PubMed] [Google Scholar]
- 79.Currie M.G., Zimmer D.P., Halushka P.V. 2020. An impaired natriuretic peptide hormone system may play a role in COVID-19 severity in vulnerable populations. FASEB bioAdvances. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Srinivasa S., Aulinas A., O’Malley T., Maehler P., Adler G.K., Grinspoon S.K., Lawson E.A. Oxytocin response to controlled dietary sodium and angiotensin II among healthy individuals. Am. J. Physiol. Endocrinol. Metab. 2018;315:E671–E675. doi: 10.1152/ajpendo.00190.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chigr F., Merzouki M., Najimi M. Autonomic brain centers and pathophysiology of COVID-19. ACS Chem. Neurosci. 2020;11:1520–1522. doi: 10.1021/acschemneuro.0c00265. [DOI] [PubMed] [Google Scholar]
- 82.Liu X.Y., Cui D., Li D., Jiao R., Wang X., Jia S., Hou D., Li T., Liu H., Wang P., Wang Y.F. Oxytocin removes estrous female vs. male preference of virgin male rats: mediation of the supraoptic nucleus via olfactory bulbs. Front Cell Neurosci. 2017;11:327. doi: 10.3389/fncel.2017.00327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bougakov D., Podell K., Goldberg E. Multiple neuroinvasive pathways in COVID-19. Mol. Neurobiol. 2020 doi: 10.1007/s12035-020-02152-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.An X., Sun X., Hou Y., Yang X., Chen H., Zhang P., Wu J. Protective effect of oxytocin on LPS-induced acute lung injury in mice. Sci. Rep. 2019;9:2836. doi: 10.1038/s41598-019-39349-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Al-Amran F., Shahkolahi M. Oxytocin ameliorates the immediate myocardial injury in rat heart transplant through downregulation of neutrophil-dependent myocardial apoptosis. Transplant. Proc. 2013;45:2506–2512. doi: 10.1016/j.transproceed.2013.03.022. [DOI] [PubMed] [Google Scholar]
- 87.Szeto A., Nation D.A., Mendez A.J., Dominguez-Bendala J., Brooks L.G., Schneiderman N., McCabe P.M. Oxytocin attenuates NADPH-dependent superoxide activity and IL-6 secretion in macrophages and vascular cells. Am. J. Physiol. Endocrinol. Metab. 2008;295:E1495–E1501. doi: 10.1152/ajpendo.90718.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Szeto A., Rossetti M.A., Mendez A.J., Noller C.M., Herderick E.E., Gonzales J.A., Schneiderman N., McCabe P.M. Oxytocin administration attenuates atherosclerosis and inflammation in Watanabe Heritable Hyperlipidemic rabbits. Psychoneuroendocrinology. 2013;38:685–693. doi: 10.1016/j.psyneuen.2012.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Johnson H.M., Torres B.A. Regulation of lymphokine production by arginine vasopressin and oxytocin: modulation of lymphocyte function by neurohypophyseal hormones. J Immunol. 1985;135:773s–775s. [PubMed] [Google Scholar]
- 90.Chen D., Hou Z., Jiang D., Zheng M., Li G., Zhang Y., Li R., Lin H., Chang J., Zeng H., Guo J.T., Zhao X. GILT restricts the cellular entry mediated by the envelope glycoproteins of SARS-CoV, Ebola virus and Lassa fever virus. Emerging microbes & infections. 2019;8:1511–1523. doi: 10.1080/22221751.2019.1677446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jyotsna U.R., Medhamurthy R. Standardization and validation of an induced ovulation model system in buffalo cows: characterization of gene expression changes in the periovulatory follicle. Anim. Reprod. Sci. 2009;113:71–81. doi: 10.1016/j.anireprosci.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 92.Stralin P., Marklund S.L. Vasoactive factors and growth factors alter vascular smooth muscle cell EC-SOD expression. Am. J. Physiol. Heart Circ. Physiol. 2001;281:H1621–H1629. doi: 10.1152/ajpheart.2001.281.4.H1621. [DOI] [PubMed] [Google Scholar]
- 93.Tavassoly O., Safavi F., Tavassoly I. Heparin-binding peptides as novel therapies to stop SARS-CoV-2 cellular entry and infection. Mol. Pharmacol. 2020;98:612–619. doi: 10.1124/molpharm.120.000098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hansenne I., Rasier G., Pequeux C., Brilot F., Renard C., Breton C., Greimers R., Legros J.J., Geenen V., Martens H.J. Ontogenesis and functional aspects of oxytocin and vasopressin gene expression in the thymus network. J. Neuroimmunol. 2005;158:67–75. doi: 10.1016/j.jneuroim.2004.08.007. [DOI] [PubMed] [Google Scholar]
- 95.Ndiaye K., Poole D.H., Pate J.L. Expression and regulation of functional oxytocin receptors in bovine T lymphocytes. Biol. Reprod. 2008;78:786–793. doi: 10.1095/biolreprod.107.065938. [DOI] [PubMed] [Google Scholar]
- 96.Fekete E.M., Antoni M.H., Lopez C., Mendez A.J., Szeto A., Fletcher M.A., Klimas N., Kumar M., Schneiderman N. Stress buffering effects of oxytocin on HIV status in low-income ethnic minority women. Psychoneuroendocrinology. 2011;36:881–890. doi: 10.1016/j.psyneuen.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Imami A.S., O’Donovan S.M., Creeden J.F., Wu X., Eby H., McCullumsmith C.B., Uvnas-Moberg K., McCullumsmith R.E., Andari E. Oxytocin’s anti-inflammatory and proimmune functions in COVID-19: a transcriptomic signature-based approach. Physiol. Genomics. 2020;52:401–407. doi: 10.1152/physiolgenomics.00095.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Allegra A., Di Gioacchino M., Tonacci A., Musolino C., Gangemi S. Immunopathology of SARS-CoV-2 infection: immune cells and mediators, prognostic factors, and immune-therapeutic implications. International journal of molecular sciences. 2020;21:4782. doi: 10.3390/ijms21134782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Al-Amran F.F., Shahkolahi M. Oxytocin ameliorates the immediate myocardial injury in heart transplant through down regulation of the neutrophil dependent myocardial apoptosis. Heart Views. 2014;15:37–45. doi: 10.4103/1995-705X.137493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nielsen S.H., Magid E., Spannow J., Christensen S., Lam H.R., Petersen J.S. Renal function after myocardial infarction and cardiac arrest in rats: role of ANP-induced albuminuria? Acta Physiol. Scand. 1997;160:301–310. doi: 10.1046/j.1365-201X.1997.00162.x. [DOI] [PubMed] [Google Scholar]
- 101.Olszewski P.K., Klockars A., Schioth H.B., Levine A.S. Oxytocin as feeding inhibitor: maintaining homeostasis in consummatory behavior. Pharmacol. Biochem. Behav. 2010;97:47–54. doi: 10.1016/j.pbb.2010.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Porzionato A., Emmi A., Barbon S., Boscolo-Berto R., Stecco C., Stocco E., Macchi V., De Caro R. Sympathetic activation: a potential link between comorbidities and COVID-19. FEBS J. 2020;287:3681–3688. doi: 10.1111/febs.15481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Solaimanzadeh I. Acetazolamide, nifedipine and phosphodiesterase inhibitors: rationale for their utilization as adjunctive countermeasures in the treatment of coronavirus disease 2019 (COVID-19) Cureus. 2020;12 doi: 10.7759/cureus.7343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Gustafson D., Raju S., Wu R., Ching C., Veitch S., Rathnakumar K., Boudreau E., Howe K.L., Fish J.E. Overcoming barriers: the endothelium as a linchpin of coronavirus disease 2019 pathogenesis? Arterioscler. Thromb. Vasc. Biol. 2020;40:1818–1829. doi: 10.1161/ATVBAHA.120.314558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chatzis DG, Magounaki KT, Pantazopoulos IN, Johnson EO, Tsioufis KP. COVID-19 pandemic and cardiovascular disease: where do we stand? Minerva cardioangiologica. 2020;68:347–358. doi: 10.23736/S0026-4725.20.05298-6. [DOI] [PubMed] [Google Scholar]
- 106.Xiong W., Yao M., Zhou R., Qu Y., Yang Y., Wang Z., Song N., Chen H., Qian J. Oxytocin ameliorates ischemia/reperfusion-induced injury by inhibiting mast cell degranulation and inflammation in the rat heart. Biomed. Pharmacother. 2020;128:110358. doi: 10.1016/j.biopha.2020.110358. [DOI] [PubMed] [Google Scholar]
- 107.Houshmand F., Faghihi M., Imani A., Kheiri S. Effect of different doses of oxytocin on cardiac electrophysiology and arrhythmias induced by ischemia. J Adv Pharm Technol Res. 2017;8:131–137. doi: 10.4103/japtr.JAPTR_178_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Momenabadi S., Vafaei A.A., Bandegi A.R., Zahedi-Khorasani M., Mazaheri Z., Vakili A. Oxytocin reduces brain injury and maintains blood-brain barrier integrity after ischemic stroke in mice. NeuroMolecular Med. 2020;22:557–571. doi: 10.1007/s12017-020-08613-3. [DOI] [PubMed] [Google Scholar]
- 109.Etehadi Moghadam S., Azami Tameh A., Vahidinia Z., Atlasi M.A., Hassani Bafrani H., Naderian H. Neuroprotective effects of oxytocin hormone after an experimental stroke model and the possible role of calpain-1. J. Stroke Cerebrovasc. Dis. 2018;27:724–732. doi: 10.1016/j.jstrokecerebrovasdis.2017.10.020. [DOI] [PubMed] [Google Scholar]
- 110.Liu S., Pan S., Tan J., Zhao W., Liu F. Oxytocin inhibits ox-LDL-induced adhesion of monocytic THP-1 cells to human brain microvascular endothelial cells. Toxicol. Appl. Pharmacol. 2017;337:104–110. doi: 10.1016/j.taap.2017.10.022. [DOI] [PubMed] [Google Scholar]
- 111.Zhai P., Ding Y., Wu X., Long J., Zhong Y., Li Y. The epidemiology, diagnosis and treatment of COVID-19. International journal of antimicrobial agents. 2020;55 doi: 10.1016/j.ijantimicag.2020.105955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang S.C., Meng D., Yang H., Wang X., Jia S., Wang P., Wang Y.-F. Pathological basis of cardiac arrhythmias: vicious cycle of immune-metabolic dysregulation. Cardiovascular Disorders and Medicine. 2017;3:1–7. [Google Scholar]
- 113.Qian W., Zhu T., Tang B., Yu S., Hu H., Sun W., Pan R., Wang J., Wang D., Yang L., Mao C., Zhou L., Yuan G. Decreased circulating levels of oxytocin in obesity and newly diagnosed type 2 diabetic patients. J. Clin. Endocrinol. Metab. 2014;99:4683–4689. doi: 10.1210/jc.2014-2206. [DOI] [PubMed] [Google Scholar]
- 114.Takayanagi Y., Kasahara Y., Onaka T., Takahashi N., Kawada T., Nishimori K. Oxytocin receptor-deficient mice developed late-onset obesity. Neuroreport. 2008;19:951–955. doi: 10.1097/WNR.0b013e3283021ca9. [DOI] [PubMed] [Google Scholar]
- 115.Suva J., Caisova D., Stajner A. Modification of fat and carbohydrate metabolism by neurohypophyseal hormones. III. Effect of oxytocin on non-esterified fatty acid, glucose, triglyceride and cholesterol levels in rat serum. Endokrinologie. 1980;76:333–339. [PubMed] [Google Scholar]
- 116.Blevins J.E., Baskin D.G. Translational and therapeutic potential of oxytocin as an anti-obesity strategy: insights from rodents, nonhuman primates and humans. Physiol. Behav. 2015;152:438–449. doi: 10.1016/j.physbeh.2015.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Petersson M., Uvnas-Moberg K. Postnatal oxytocin treatment of spontaneously hypertensive male rats decreases blood pressure and body weight in adulthood. Neurosci. Lett. 2008;440:166–169. doi: 10.1016/j.neulet.2008.05.091. [DOI] [PubMed] [Google Scholar]
- 118.Wsol A, Cudnoch-Je drzejewska A, Szczepanska-Sadowska E, Kowalewski S, Dobruch J. Central oxytocin modulation of acute stress-induced cardiovascular responses after myocardial infarction in the rat. Stress. 2009;12:517–525. doi: 10.3109/10253890802687688. [DOI] [PubMed] [Google Scholar]
- 119.Jameson H., Bateman R., Byrne P., Dyavanapalli J., Wang X., Jain V., Mendelowitz D. Oxytocin neuron activation prevents hypertension that occurs with chronic intermittent hypoxia/hypercapnia in rats. Am. J. Physiol. Heart Circ. Physiol. 2016;310:H1549–H1557. doi: 10.1152/ajpheart.00808.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lehrke M., Marx N. Diabetes mellitus and heart failure. Am. J. Cardiol. 2017;120:S37–S47. doi: 10.1016/j.amjcard.2017.05.014. [DOI] [PubMed] [Google Scholar]
- 121.Gutkowska J., Broderick T.L., Bogdan D., Wang D., Lavoie J.M., Jankowski M. Downregulation of oxytocin and natriuretic peptides in diabetes: possible implications in cardiomyopathy. J. Physiol. 2009;587:4725–4736. doi: 10.1113/jphysiol.2009.176461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Klement J., Ott V., Rapp K., Brede S., Piccinini F., Cobelli C., Lehnert H., Hallschmid M. Oxytocin improves beta-cell responsivity and glucose tolerance in healthy men. Diabetes. 2017;66:264–271. doi: 10.2337/db16-0569. [DOI] [PubMed] [Google Scholar]
- 123.Watanabe S., Wei F.Y., Matsunaga T., Matsunaga N., Kaitsuka T., Tomizawa K. Oxytocin protects against stress-induced cell death in murine pancreatic beta-cells. Sci. Rep. 2016;6:25185. doi: 10.1038/srep25185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Florian M., Jankowski M., Gutkowska J. Oxytocin increases glucose uptake in neonatal rat cardiomyocytes. Endocrinology. 2010;151:482–491. doi: 10.1210/en.2009-0624. [DOI] [PubMed] [Google Scholar]
- 125.Shioi T., Kang P.M., Douglas P.S., Hampe J., Yballe C.M., Lawitts J., Cantley L.C., Izumo S. The conserved phosphoinositide 3-kinase pathway determines heart size in mice. EMBO J. 2000;19:2537–2548. doi: 10.1093/emboj/19.11.2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Noiseux N., Borie M., Desnoyers A., Menaouar A., Stevens L.M., Mansour S., Danalache B.A., Roy D.C., Jankowski M., Gutkowska J. Preconditioning of stem cells by oxytocin to improve their therapeutic potential. Endocrinology. 2012;153:5361–5372. doi: 10.1210/en.2012-1402. [DOI] [PubMed] [Google Scholar]
- 127.Javelot H., Weiner L. Panic and pandemic: narrative review of the literature on the links and risks of panic disorder as a consequence of the SARS-CoV-2 pandemic. L’Encephale. 2020:S0013–7006–5. doi: 10.1016/j.encep.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Emeny R.T., Huber D., Bidlingmaier M., Reincke M., Klug G., Ladwig K.H. Oxytocin-induced coping with stressful life events in old age depends on attachment: findings from the cross-sectional KORA Age study. Psychoneuroendocrinology. 2015;56:132–142. doi: 10.1016/j.psyneuen.2015.03.014. [DOI] [PubMed] [Google Scholar]
- 129.Wang X., Pei J., Hu X. The brain-heart connection in Takotsubo syndrome: the central nervous system, sympathetic nervous system, and catecholamine overload. Cardiol. Res. Pract. 2020;2020:4150291. doi: 10.1155/2020/4150291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Li D., Liu X., Liu H., Li T., Jia S., Wang X., Wang P., Qin D., Wang Y.F. Key roles of cyclooxygenase 2-protein kinase A-hyperpolarization-activated cyclic nucleotide-gated channel 3 pathway in the regulation of oxytocin neuronal activity in lactating rats with intermittent pup-deprivation. Neuroscience. 2021;452:13–25. doi: 10.1016/j.neuroscience.2020.10.016. [DOI] [PubMed] [Google Scholar]
- 131.Briel R.C., Kidess E., Kunz S., Dieter B. Studies on platelet function during different modes of administration of PgF2alpha in obstetrics and gynecology. Arch Gynakol. 1978;225:201–208. doi: 10.1007/BF00667202. [DOI] [PubMed] [Google Scholar]
- 132.Yang X., Wan M., Liang T., Peng M., Chen F. Synthetic polyphosphate inhibits endogenous coagulation and platelet aggregation in vitro. Biomed Rep. 2017;6:57–62. doi: 10.3892/br.2016.816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ailamazyan E.K., Petrishchev N.N., Vivulanets E.V., Maryutina L.V., Mindukshev I.V., Khachaturyan A.R. Effects of oxytocin and prostaglandin F(2alpha) (enzaprost) on platelet aggregation. Bull. Exp. Biol. Med. 2002;134:439–441. doi: 10.1023/a:1022677927836. [DOI] [PubMed] [Google Scholar]
- 134.Ciosek J., Drobnik J. Function of the hypothalamo-neurohypophysial system in rats with myocardial infarction is modified by melatonin. Pharmacol. Rep. 2012;64:1442–1454. doi: 10.1016/s1734-1140(12)70942-8. [DOI] [PubMed] [Google Scholar]
- 135.Du D., Jiang M., Liu M., Wang J., Xia C., Guan R., Shen L., Ji Y., Zhu D. Microglial P2X(7) receptor in the hypothalamic paraventricular nuclei contributes to sympathoexcitatory responses in acute myocardial infarction rat. Neurosci. Lett. 2015;587:22–28. doi: 10.1016/j.neulet.2014.12.026. [DOI] [PubMed] [Google Scholar]
- 136.Moghimian M., Faghihi M., Karimian S.M., Imani A. The effect of acute stress exposure on ischemia and reperfusion injury in rat heart: role of oxytocin. Stress. 2012;15:385–392. doi: 10.3109/10253890.2011.630436. [DOI] [PubMed] [Google Scholar]
- 137.El-Missiry M.A., El-Missiry Z.M.A., Othman A.I. Melatonin is a potential adjuvant to improve clinical outcomes in individuals with obesity and diabetes with coexistence of Covid-19. Eur. J. Pharmacol. 2020;882 doi: 10.1016/j.ejphar.2020.173329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Jung C., Wernly B., Bjursell M., Wiseman J., Admyre T., Wikstrom J., Palmer M., Seeliger F., Lichtenauer M., Franz M., Frick C., Andersson A.K., Elg M., Pernow J., Sjoquist P.O., Bohlooly Y.M., Wang Q.D. Cardiac-specific overexpression of oxytocin receptor leads to cardiomyopathy in mice. J. Card. Fail. 2018;24:470–478. doi: 10.1016/j.cardfail.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 139.Li D., Liu H., Liu X., Wang H., Li T., Wang X., Jia S., Wang P., Wang Y.F. Involvement of hyperpolarization-activated cyclic nucleotide-gated channel 3 in oxytocin neuronal activity in lactating rats with pup deprivation. ASN Neuro. 2020;12 doi: 10.1177/1759091420944658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Loichot C., Grima M., De Jong W., Helwig J.J., Imbs J.L., Barthelmebs M. Oxytocin-induced renin secretion by denervated kidney in anaesthetized rat. Eur. J. Pharmacol. 2002;454:241–247. doi: 10.1016/s0014-2999(02)02495-0. [DOI] [PubMed] [Google Scholar]
- 141.Hatton G.I., Wang Y.F. Neural mechanisms underlying the milk ejection burst and reflex. Prog. Brain Res. 2008;170:155–166. doi: 10.1016/S0079-6123(08)00414-7. [DOI] [PubMed] [Google Scholar]
- 142.Wang Y.F., Ponzio T.A., Hatton G.I. Autofeedback effects of progressively rising oxytocin concentrations on supraoptic oxytocin neuronal activity in slices from lactating rats. Am J Physiol Regul Integr Comp Physiol. 2006;290:R1191–R1198. doi: 10.1152/ajpregu.00725.2005. [DOI] [PubMed] [Google Scholar]
- 143.Wang Y.F., Hatton G.I. Dominant role of betagamma subunits of G-proteins in oxytocin-evoked burst firing. J. Neurosci. 2007;27:1902–1912. doi: 10.1523/JNEUROSCI.5346-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wakerley J.B., Clarke G., Summerlee A.J. In: The Physiology of Reproduction. Knobil E., Neill J.D., editors. Raven; New York: 1994. Milk ejection and its control; pp. 1131–1177. [Google Scholar]
- 145.Binay C., Paketci C., Guzel S., Samanci N. Serum irisin and Oxytocin as Predictors of Metabolic Parameters in Obese Children. Clin Res Pediatr Endocrinol. 2017;9:124–131. doi: 10.4274/jcrpe.3963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lee M.R., Scheidweiler K.B., Diao X.X., Akhlaghi F., Cummins A., Huestis M.A., Leggio L., Averbeck B.B. Oxytocin by intranasal and intravenous routes reaches the cerebrospinal fluid in rhesus macaques: determination using a novel oxytocin assay. Mol. Psychiatry. 2017;23:115–122. doi: 10.1038/mp.2017.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Petty M.A., Lang R.E., Unger T., Ganten D. The cardiovascular effects of oxytocin in conscious male rats. Eur. J. Pharmacol. 1985;112:203–210. doi: 10.1016/0014-2999(85)90497-2. [DOI] [PubMed] [Google Scholar]
- 148.D’Orleans-Juste P., Dion S., Mizrahi J., Regoli D. Effects of peptides and non-peptides on isolated arterial smooth muscles: role of endothelium. Eur. J. Pharmacol. 1985;114:9–21. doi: 10.1016/0014-2999(85)90515-1. [DOI] [PubMed] [Google Scholar]
- 149.Szabo C., Hardebo J.E. Regional differences in the contractile activity of neuropeptide Y, endothelin, oxytocin and vasopressin: comparison with non-peptidergic constrictors. An in vitro study in the basilar and mesenteric arteries of the rat. Acta Physiol. Hung. 1992;79:281–293. [PubMed] [Google Scholar]
- 150.Magata F., Hartmann D., Ishii M., Miura R., Takahashi H., Matsui M., Kida K., Miyamoto A., Bollwein H. Effects of exogenous oxytocin on uterine blood flow in puerperal dairy cows: the impact of days after parturition and retained fetal membranes. Vet. J. 2013;196:76–80. doi: 10.1016/j.tvjl.2012.08.010. [DOI] [PubMed] [Google Scholar]
- 151.Liu X., Jia S., Zhang Y., Wang Y.-F. Pulsatile but not tonic secretion of oxytocin plays the role of anti-precancerous lesions of the mammary glands in rat dams separated from the pups during lactation. M J Neuro. 2016;1:002. [Google Scholar]
- 152.Nissen E., Gustavsson P., Widstrom A.M., Uvnas-Moberg K. Oxytocin, prolactin, milk production and their relationship with personality traits in women after vaginal delivery or Cesarean section. J. Psychosom. Obstet. Gynaecol. 1998;19:49–58. doi: 10.3109/01674829809044221. [DOI] [PubMed] [Google Scholar]
- 153.Conti P., Younes A. Coronavirus COV-19/SARS-CoV-2 affects women less than men: clinical response to viral infection. J. Biol. Regul. Homeost. Agents. 2020;34:339–343. doi: 10.23812/Editorial-Conti-3. [DOI] [PubMed] [Google Scholar]
- 154.Ferrau F., Ceccato F., Cannavo S., Scaroni C. What we have to know about corticosteroids use during Sars-Cov-2 infection. J. Endocrinol. Investig. 2020:1–9. doi: 10.1007/s40618-020-01384-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Li H., Chen C., Hu F., Wang J., Zhao Q., Gale R.P., Liang Y. Impact of corticosteroid therapy on outcomes of persons with SARS-CoV-2, SARS-CoV, or MERS-CoV infection: a systematic review and meta-analysis. Leukemia. 2020;34:1503–1511. doi: 10.1038/s41375-020-0848-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Group RC, Horby P., Mafham M., Linsell L., Bell J.L., Staplin N., Emberson J.R., Wiselka M., Ustianowski A., Elmahi E., Prudon B., Whitehouse T., Felton T., Williams J., Faccenda J., Underwood J., Baillie J.K., Chappell L.C., Faust S.N., Jaki T., Jeffery K., Lim W.S., Montgomery A., Rowan K., Tarning J., Watson J.A., White N.J., Juszczak E., Haynes R., Landray M.J. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N. Engl. J. Med. 2020;383:2030–2040. doi: 10.1056/NEJMoa2022926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Self W.H., Semler M.W., Leither L.M., Casey J.D., Angus D.C., Brower R.G., Chang S.Y., Collins S.P., Eppensteiner J.C., Filbin M.R., Files D.C., Gibbs K.W., Ginde A.A., Gong M.N., Harrell F.E., Jr., Hayden D.L., Hough C.L., Johnson N.J., Khan A., Lindsell C.J., Matthay M.A., Moss M., Park P.K., Rice T.W., Robinson B.R.H., Schoenfeld D.A., Shapiro N.I., Steingrub J.S., Ulysse C.A., Weissman A., Yealy D.M., Thompson B.T., Brown S.M., National Heart L., Blood Institute PCTN, Steingrub J., Smithline H., Tiru B., Tidswell M., Kozikowski L., Thornton-Thompson S., De Souza L., Hou P., Baron R., Massaro A., Aisiku I., Fredenburgh L., Seethala R., Johnsky L., Riker R., Seder D., May T., Baumann M., Eldridge A., Lord C., Shapiro N., Talmor D., O’Mara T., Kirk C., Harrison K., Kurt L., Schermerhorn M., Banner-Goodspeed V., Boyle K., Dubosh N., Filbin M., Hibbert K., Parry B., Lavin-Parsons K., Pulido N., Lilley B., Lodenstein C., Margolin J., Brait K., Jones A., Galbraith J., Peacock R., Nandi U., Wachs T., Matthay M., Liu K., Kangelaris K., Wang R., Calfee C., Yee K., Hendey G., Chang S., Lim G., Qadir N., Tam A., Beutler R., Levitt J., Wilson J., Rogers A., Vojnik R., Roque J., Albertson T., Chenoweth J., Adams J., Pearson S., Juarez M., Almasri E., Fayed M., Hughes A., Hillard S., Huebinger R., Wang H., Vidales E., Patel B., Ginde A., Moss M., Baduashvili A., McKeehan J., Finck L., Higgins C., Howell M., Douglas I., Haukoos J., Hiller T., Lyle C., Cupelo A., Caruso E., Camacho C., Gravitz S., Finigan J., Griesmer C., Park P., Hyzy R., Nelson K., McDonough K., Olbrich N., Williams M., Kapoor R., Nash J., Willig M., Ford H., Gardner-Gray J., Ramesh M., Moses M., Ng Gong M., Aboodi M., Asghar A., Amosu O., Torres M., Kaur S., Chen J.T., Hope A., Lopez B., Rosales K., Young You J., Mosier J., Hypes C., Natt B., Borg B., Salvagio Campbell E., Hite R.D., Hudock K., Cresie A., Alhasan F., Gomez-Arroyo J., Duggal A., Mehkri O., Hastings A., Sahoo D., Abi Fadel F., Gole S., Shaner V., Wimer A., Meli Y., King A., Terndrup T., Exline M., Pannu S., Robart E., Karow S., Hough C., Robinson B., Johnson N., Henning D., Campo M., Gundel S., Seghal S., Katsandres S., Dean S., Khan A., Krol O., Jouzestani M., Huynh P., Weissman A., Yealy D., Scholl D., Adams P., McVerry B., Huang D., Angus D., Schooler J., Moore S., Files C., Miller C., Gibbs K., LaRose M., Flores L., Koehler L., Morse C., Sanders J., Langford C., Nanney K., MdalaGausi M., Yeboah P., Morris P., Sturgill J., Seif S., Cassity E., Dhar S., de Wit M., Mason J., Goodwin A., Hall G., Grady A., Chamberlain A., Brown S., Bledsoe J., Leither L., Peltan I., Starr N., Fergus M., Aston V., Montgomery Q., Smith R., Merrill M., Brown K., Armbruster B., Harris E., Middleton E., Paine R., Johnson S., Barrios M., Eppensteiner J., Limkakeng A., McGowan L., Porter T., Bouffler A., Leahy J.C., deBoisblanc B., Lammi M., Happel K., Lauto P., Self W., Casey J., Semler M., Collins S., Harrell F., Lindsell C., Rice T., Stubblefield W., Gray C., Johnson J., Roth M., Hays M., Torr D., Zakaria A., Schoenfeld D., Thompson T., Hayden D., Ringwood N., Oldmixon C., Ulysse C., Morse R., Muzikansky A., Fitzgerald L., Whitaker S., Lagakos A., Brower R., Reineck L., Aggarwal N., Bienstock K., Freemer M., Maclawiw M., Weinmann G., Morrison L., Gillespie M., Kryscio R., Brodie D., Zareba W., Rompalo A., Boeckh M., Parsons P., Christie J., Hall J., Horton N., Zoloth L., Dickert N., Diercks D. Effect of hydroxychloroquine on clinical status at 14 days in hospitalized patients with COVID-19: a randomized clinical trial. JAMA. 2020;324:2165–2176. doi: 10.1001/jama.2020.22240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Huang C., Huang L., Wang Y., Li X., Ren L., Gu X., Kang L., Guo L., Liu M., Zhou X., Luo J., Huang Z., Tu S., Zhao Y., Chen L., Xu D., Li Y., Li C., Peng L., Li Y., Xie W., Cui D., Shang L., Fan G., Xu J., Wang G., Wang Y., Zhong J., Wang C., Wang J., Zhang D., Cao B. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397:220–232. doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Phie J., Haleagrahara N., Newton P., Constantinoiu C., Sarnyai Z., Chilton L., Kinobe R. Prolonged subcutaneous administration of oxytocin accelerates angiotensin II-induced hypertension and renal damage in male rats. PLoS One. 2015;10 doi: 10.1371/journal.pone.0138048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Holmes E.A., O’Connor R.C., Perry V.H., Tracey I., Wessely S., Arseneault L., Ballard C., Christensen H., Cohen Silver R., Everall I., Ford T., John A., Kabir T., King K., Madan I., Michie S., Przybylski A.K., Shafran R., Sweeney A., Worthman C.M., Yardley L., Cowan K., Cope C., Hotopf M., Bullmore E. Multidisciplinary research priorities for the COVID-19 pandemic: a call for action for mental health science. Lancet Psychiatry. 2020;7:547–560. doi: 10.1016/S2215-0366(20)30168-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Knox S.S., Uvnas-Moberg K. Social isolation and cardiovascular disease: an atherosclerotic pathway? Psychoneuroendocrinology. 1998;23:877–890. doi: 10.1016/s0306-4530(98)00061-4. [DOI] [PubMed] [Google Scholar]
- 162.Harvey B.H., Regenass W., Dreyer W., Moller M. Social isolation rearing-induced anxiety and response to agomelatine in male and female rats: role of corticosterone, oxytocin, and vasopressin. J. Psychopharmacol. 2019;33:640–646. doi: 10.1177/0269881119826783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Tanaka K., Osako Y., Takahashi K., Hidaka C., Tomita K., Yuri K. Effects of post-weaning social isolation on social behaviors and oxytocinergic activity in male and female rats. Heliyon. 2019;5 doi: 10.1016/j.heliyon.2019.e01646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Taylor S.E., Gonzaga G.C., Klein L.C., Hu P., Greendale G.A., Seeman T.E. Relation of oxytocin to psychological stress responses and hypothalamic-pituitary-adrenocortical axis activity in older women. Psychosom. Med. 2006;68:238–245. doi: 10.1097/01.psy.0000203242.95990.74. [DOI] [PubMed] [Google Scholar]
- 165.Nation D.A., Szeto A., Mendez A.J., Brooks L.G., Zaias J., Herderick E.E., Gonzales J., Noller C.M., Schneiderman N., McCabe P.M. Oxytocin attenuates atherosclerosis and adipose tissue inflammation in socially isolated ApoE−/− mice. Psychosom. Med. 2010;72:376–382. doi: 10.1097/PSY.0b013e3181d74c48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Guruprasad L. Human SARS CoV-2 spike protein mutations. Proteins. 2021 doi: 10.1002/prot.26042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Gobeil S.M., Janowska K., McDowell S., Mansouri K., Parks R., Manne K., Stalls V., Kopp M.F., Henderson R., Edwards R.J., Haynes B.F., Acharya P. D614G mutation alters SARS-CoV-2 spike conformation and enhances protease cleavage at the S1/S2 junction. Cell reports. 2020;34 doi: 10.1016/j.celrep.2020.108630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Johnson J.L., Buisman-Pijlman F.T. Adversity impacting on oxytocin and behaviour: timing matters. Behav. Pharmacol. 2016;27:659–671. doi: 10.1097/FBP.0000000000000269. [DOI] [PubMed] [Google Scholar]
- 169.Yahya F.D., Ahmad M., Usman A.N., Sinrang A.W., Alasiry E., Bahar B. Potential combination of back massage therapy and acupressure as complementary therapy in postpartum women for the increase in the hormone oxytocin. Enferm Clin. 2020;30(Suppl. 2):570–572. [Google Scholar]
- 170.Nilsson U. Soothing music can increase oxytocin levels during bed rest after open-heart surgery: a randomised control trial. J. Clin. Nurs. 2009;18:2153–2161. doi: 10.1111/j.1365-2702.2008.02718.x. [DOI] [PubMed] [Google Scholar]
- 171.Bellosta-Batalla M., Del Carmen Blanco-Gandia M., Rodriguez-Arias M., Cebolla A., Perez-Blasco J., Moya-Albiol L. Brief mindfulness session improves mood and increases salivary oxytocin in psychology students. Stress and Health: journal of the International Society for the Investigation of Stress. 2020;36:469–477. doi: 10.1002/smi.2942. [DOI] [PubMed] [Google Scholar]
- 172.Michelini L.C. Differential effects of vasopressinergic and oxytocinergic pre-autonomic neurons on circulatory control: reflex mechanisms and changes during exercise. Clin. Exp. Pharmacol. Physiol. 2007;34:369–376. doi: 10.1111/j.1440-1681.2007.04589.x. [DOI] [PubMed] [Google Scholar]
- 173.Gutkowska J., Paquette A., Wang D., Lavoie J.M., Jankowski M. Effect of exercise training on cardiac oxytocin and natriuretic peptide systems in ovariectomized rats. Am J Physiol Regul Integr Comp Physiol. 2007;293:R267–R275. doi: 10.1152/ajpregu.00071.2007. [DOI] [PubMed] [Google Scholar]