Abstract
Background
Limited data is available addressing gastrointestinal (GI) ischemia in coronavirus disease 2019 (COVID-19). We reviewed the clinical and radiologic features of GI ischemia and its related complications in thirty-one COVID-19 patients reported in literature.
Methods
A systematic literature review was performed using a search strategy on all studies published from January 1, 2020, to June 13, 2020, and updated on September 6, 2020, on databases from PubMed, Scopus, Embase, Web of Science, and Google Scholar. Every study with at least one presentation of COVID-19-related GI ischemia complication and one GI imaging finding was included.
Results
In total, twenty-two studies and thirty-one patients with the mean age of 59 ± 12.7 (age range: 28–80) years old were included, of which 23 (74.2%) patients were male, 7 (22.5%) female, and one unknown gender. The significant GI imaging findings include mesenteric arterial or venous thromboembolism, followed by small bowel ischemia. Nine patients (29%) presented with arterial compromise due to superior mesenteric thromboembolism, resulting in bowel ischemia. Also, 6 patients (19.3%) demonstrated occlusive thrombosis of the portal system and superior mesenteric vein. More than two-thirds of patients (20, 64.5%) required laparotomy and bowel resection. Eventually, five (16.1%) patients were discharged, of whom four cases (12.9%) readmitted. Five (16.1%) patients remained ICU hospitalized at the report time and 12 (38.7%) patients died.
Conclusion
Macrovascular arterial/venous thrombosis is identified in almost half of COVID-19 patients with bowel ischemia. Overall mortality in COVID-19 patients with GI ischemia and radiologically evident mesenteric thrombotic occlusion was 38.7% and 40%, retrospectively.
Keywords: SARS-CoV-2; Coronavirus; Gastrointestinal; Ischemia; Abdominal pain; Tomography; X-ray, computed
1. Introduction
Since December 2019, the outbreak of the coronavirus disease 2019 (COVID-19) in Wuhan, China has become the leading cause of respiratory illnesses worldwide.1 , 2 From the latest world health organization (WHO) updated on November 1st, 2020, over 46 million cases of COVID-19 across 216 countries have been confirmed.3
COVID-19 manifestations are mostly respiratory with related complications ranging from mild non-pneumonic illnesses to respiratory failure.[4], [5], [6] However, several extrapulmonary presentations, including gastrointestinal (GI) symptoms, such as anorexia, diarrhea, vomiting, nausea, abdominal pain, and GI bleeding have been recently reported in COVID-19 patients.[7], 8., [9] Due to the prominence of the pulmonary presentations, extrapulmonary manifestations can be easily missed and overlooked, resulting in a delayed diagnosis of COVID-19 in patients with primary GI manifestations.9 Severe GI complications have been observed in critically ill patients with a higher frequency.7 , 10 Numerous GI complications, such as ileus, extensive hepatic necrosis, acute acalculous cholecystitis, and bowel ischemia have been reported in COVID-19 patients, particularly those admitted to intensive care unit (ICU).11
GI ischemia is an uncommon medical condition and results from any etiology that leads to bowel hypoperfusion.12 , 13 Recent studies suggest that the possible coagulopathy or vasculopathy caused by COVID-19 may increase the risk of thromboembolic events and associated ischemia.[14], [15], [16] Most of the published reports, however, include extremity venous thrombosis and pulmonary embolism.15 , [17], 18., [19] Thromboembolic events in the GI system, including mesenteric ischemia, are specifically important, because they constitute a potentially fatal clinical emergency with a high rate of mortality.[20], [21], [22]
Limited data is available addressing GI ischemia in the setting of COVID-19.23 , 24 Also, the exact underlying pathologic mechanism is not clear. Different pathophysiological mechanisms, such as inflammation, vasculopathy, immobilization, endothelial dysfunction, and hypercoagulable state have been considered to explain GI ischemia in COVID-19 patients.25 However, it is still unclear whether arterial thromboembolic events are direct complications of COVID-19 or simply a coincident. Considering the high mortality rate of GI ischemia, clinicians are encouraged to have a very high clinical suspicion index and low threshold for cross-sectional imaging and clinical screening of GI symptoms in the era of COVID-19 due to the possible risk of GI ischemia progression and its related complications that may require emergent surgical intervention. As comprehensive and inclusive data are not currently available to declare GI complications in COVID-19 patients, we systematically reviewed imaging findings and the outcome of confirmed COVID-19 patients with GI ischemic complications.
2. Materials and methods
2.1. Search strategies
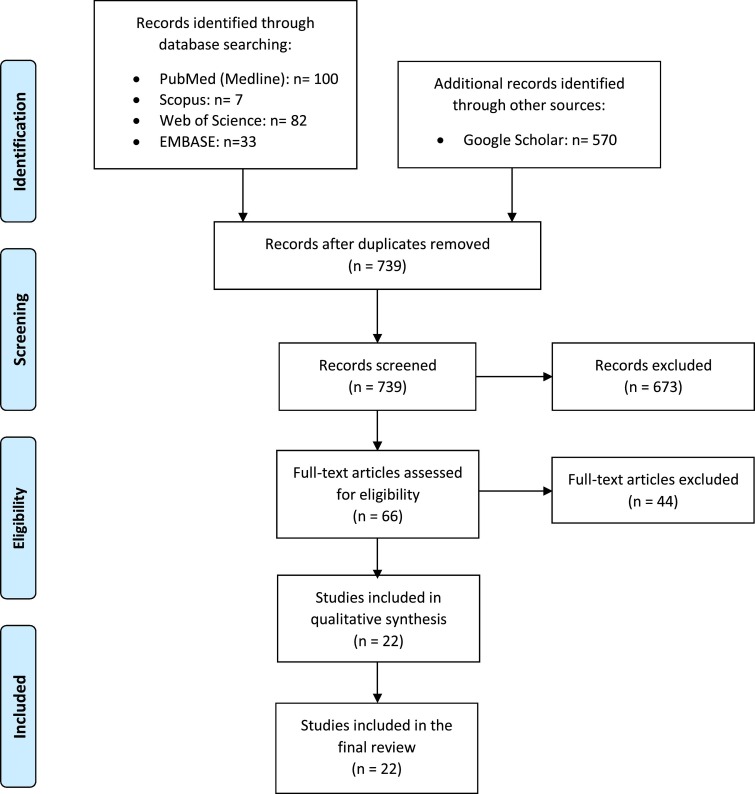
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guidelines.26 We firstly searched PubMed (MEDLINE), Scopus, Embase (Elsevier), Web of Science, and Google Scholar databases from January 1, 2020, to June 13, 2020, and updated on September 6, 2020, for studies on COVID-19 that reported the GI ischemia complications. Keywords for the literature search included “coronavirus disease,” “coronavirus infections,” “COVID-19”, “2019-nCoV”, “abdominal pain”, and “intestinal ischemia”. The full list of the keywords used is provided in Appendix A. Fig. 1 shows the flow diagram of the study selection process.
Fig. 1.
Flow diagram of study selection process.
2.2. Eligibility criteria
All studies published in the English language that included COVID-19 cases confirmed via reverse transcription-polymerase chain reaction (RT-PCR) or nasopharyngeal swabs, reported at least one presentation of GI ischemia complication, and with at least one GI imaging finding were eligible for inclusion. Correspondence or letters fulfilling the above criteria were also included in this study. Duplicates, conference abstracts, and studies without available full text were excluded.
2.3. Data extraction and quality assessment
Two authors independently extracted the data and then continued to cross-check the results. Disagreements were resolved by consensus. The following data from each study were extracted: name of the first author, study country, study design, the patient's age, gender, initial clinical presentations, imaging findings, operative procedure, course of the disease (such as ICU admission), and outcome. Furthermore, we reported a series of imaging findings from included studies23 , [27], [28], 29. (Fig. 2, Fig. 3, Fig. 4, Fig. 5, Fig. 6 ) (formal permissions obtained from the copyright holder). Quality of the included studies was assessed using the National Institutes of Health (NIH) quality assessment tool for case series/reports30 and the modified version of the Newcastle-Ottawa Quality Scale (NOS) for cohort studies.31 (Supplementary Tables 1&2).
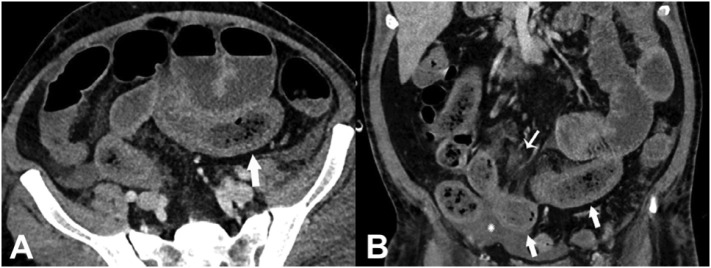
Fig. 2.
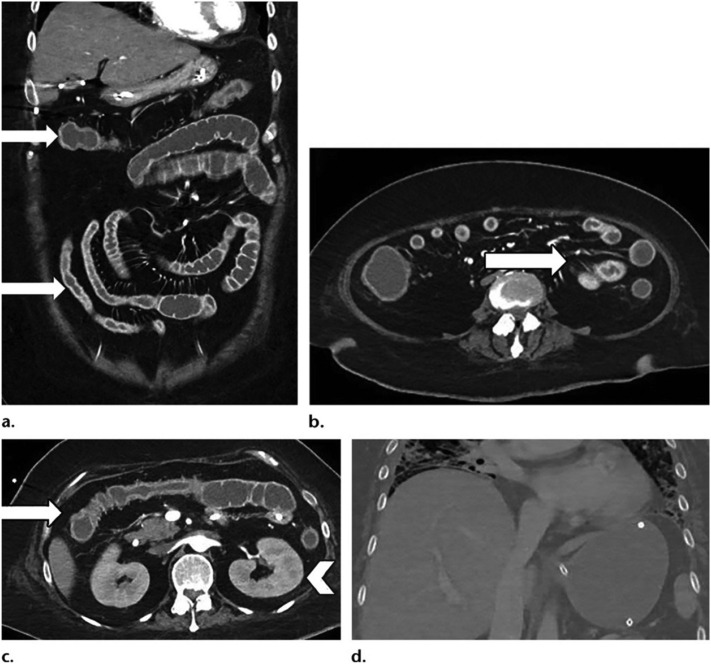
Axial (A) and coronal (B) CT of the abdomen and pelvis with IV contrast in a 57-year old man (patient no. 3) with a high clinical suspicion for bowel ischemia. There was generalized small bowel distension and segmental thickening (arrows), with adjacent mesenteric congestion (thin arrow in B), and a small volume of ascites (* in B). Findings are nonspecific but suggestive of early ischemia or infection. Images obtained from Bhayana et al.23 Radiology, published online May 11, 2020, copyright (2020), and permission to use granted by Ashley E. Daly, Senior Manager, Journal Rights & Communications Publications, Radiological Society of North America (RSNA).
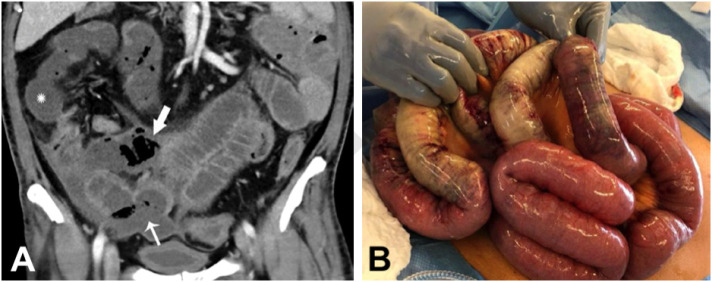
Fig. 3.
Coronal (A) CT of the abdomen and pelvis with IV contrast in a 47-year-old man (patient no. 4) with abdominal tenderness demonstrates typical findings of mesenteric ischemia and infarction, including pneumatosis intestinalis (arrow) and non-enhancing bowel (*). Frank discontinuity of a thickened loop of small bowel in the pelvis (thin arrow) is in keeping with perforation. These findings were confirmed at laparotomy (B), with the additional observation of atypical yellow discoloration of bowel. Images obtained from Bhayana et al. 23 Radiology, published online May 11, 2020, copyright (2020), and permission to use granted by Ashley E. Daly, Senior Manager, Journal Rights & Communications Publications, Radiological Society of North America (RSNA). (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
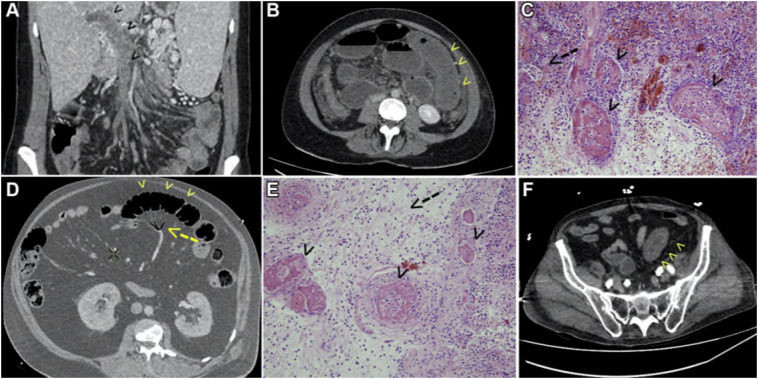
Fig. 4.
Abdominal CT scan and pathological findings in SARS-CoV-2 infected patients. (A) CT scan shows mesenteric and portal vein thrombosis (arrowheads) in a young female patient (patient no. 12) with essential thrombocythemia, as a first sign and prior to respiratory symptoms revealing the COVID-19. (B) CT scan shows bowel infarction in the first bowel loop (arrowheads) 5 days after admission of the patient no. 12, prompting emergency laparotomy and bowel resection. (C) Pathological findings of patient no. 12 (hematoxylin and eosin stain, 200 magnification): small bowel necrosis; arrowheads show microthrombi in the lamina propria and the submucosa; arrow shows glandular necrosis. (D) CT scan shows signs of bowel ischemia (arrowheads) and mesenteric venous gas (arrow) in the proximal jejunum in a 56-year-old male patient (patient no. 13) with acute respiratory distress syndrome during COVID-19. (E) Pathological findings of patient no. 13 (hematoxylin and eosin stain, 200 magnification): small bowel necrosis; arrowheads show micro-thrombi; and arrow shows edema and inflammatory infiltrates in the submucosa. (F) CT scan shows an inflammatory bowel loop with thickening and edema (arrowhead) in a 67-year-old male (patient no. 14) with acute respiratory distress syndrome during COVID-19. Images obtained from Ignat.27 Surgery published on July 1, 2020, Vol. 168 (1) P14–16 (copyright 2020), and permission to use granted by Elsevier and Copyright Clearance Center.
Fig. 5.
Cough and shortness of breath in a 46-year-old woman (patient no. 22) with diabetes mellitus who was initially diagnosed with COVID-19. The patient's condition clinically deteriorated, and the use of mechanical ventilation, extracorporeal membrane oxygenation (ECMO), and pressor support was required. The clinical course was complicated by acute toxic metabolic encephalopathy, pneumonia, acute respiratory distress syndrome, cardiogenic shock, acute kidney injury, and secondary adrenal insufficiency. Coronal (a) and axial (b, c) contrast-enhanced CT images show multifocal small and large bowel wall thickening (arrows) creating a ribbonlike appearance, likely related to small vessel ischemia. Note the altered enhancement of the kidneys (arrowhead in c) in the setting of acute kidney injury and multifocal peripheral airspace opacities at the lung bases on the coronal CT image (d). Images obtained from Olson et al.29 Radiology published online on July 10, 2020, Vol. 40, No. 5, copyright (2020), and permission to use granted by Ashley E. Daly, Senior Manager, Journal Rights & Communications Publications, Radiological Society of North America (RSNA).
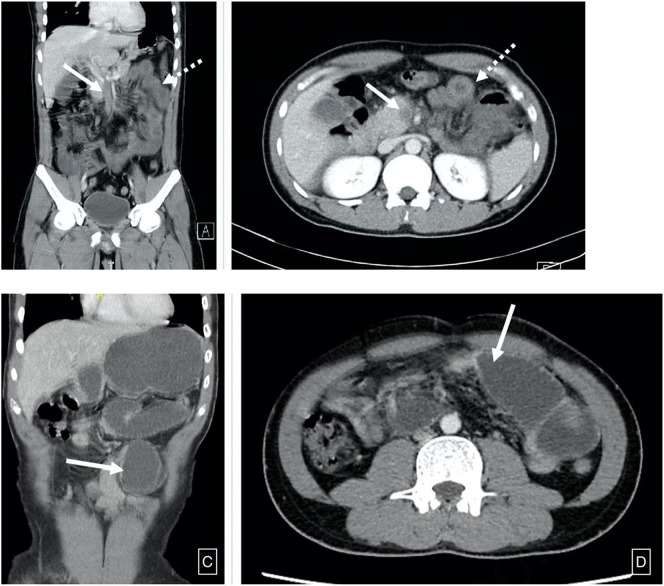
Fig. 6.
A computed tomography scan of the abdomen and pelvis revealed A, B: Superior mesenteric vein thrombosis (bold arrow) and diffuse mural thickening (dotted arrow), C, D: High grade small bowel obstruction with a transition point (bold arrow) seen in mid jejunum. Images obtained from Pang et al.28 Annals of Vascular Surgery available online on August 28, 2020, (copyright 2020), and permission to use granted by Elsevier and Copyright Clearance Center.
3. Results
A total of 792 articles were recognized in the initial search. After removing duplicates, 739 were screened by titles and abstracts. Next, unrelated articles were excluded and the remaining 57 were retrieved for full-text evaluation. In our search, we found twenty-two studies, including three cohort studies, three case series, twelve case reports, and four correspondence to the editors related to GI ischemia complications in patients with COVID-19. Thirty-one patients with the mean age of 59 ± 12.7 (age range: 28–80; age IQR: 51–70) years old were included, of which 23 patients were male (74.2%), 7 (22.5%) female, and one unknown gender. Table 1 summarizes the clinical characteristics, imaging findings, operative procedures, and outcomes of these patients.
Table 1.
Characteristics of twenty patients with COVID-19 infection according to gastrointestinal ischemia complications.
| Patient no./sex/age(y) | First author | Country | Initial symptoms | Imaging findings (Abdominal and pelvic CT scan) | Surgery procedure | Outcome |
|---|---|---|---|---|---|---|
| P1/NR/56 | Azouz et al.54 | France | Abdominal pain and vomiting | Intraluminal thrombus of the aortic arch, SMA thromboembolism, dilation and un-enhancement of small bowel loops (ischemia) | Endovascular thrombectomy, laparotomy (resection of 2 m of small bowel) | NR |
| P2/M/59 | Bianco et al.45 | Italy | Bilateral pneumonia, acute abdominal pain with nausea | Air-fluid levels in the small bowel loops associated with mesenteric edema, mild peritoneal free fluid, segmental small bowel ischemia | Laparotomy | Death |
| P3/M/57 | Bhayana et al.23 | USA | NR | Generalized small bowel distension and segmental thickening, adjacent mesenteric congestion, mild free fluid (ischemia) | Exploratory laparotomy | NR |
| P4/M/47 | Bhayana et al.23 | USA | Abdominal pain | Pneumatosis intestinalis and non-enhancing bowel, mesenteric ischemia and infarction, concomitant small bowel perforation | Exploratory laparotomy | NR |
| P5/M/52 | Bhayana et al.23 | USA | NR | Portal venous gas, suggesting small bowel infarction | Exploratory laparotomy | NR |
| P6/M/52 | Beccara et al.55 | Italy | Cough, fever, two episodes of diarrhea, vomiting, abdominal pain | Thrombosis of SMA branches, bowel distension | Intestinal resection with stapled side-to side anastomosis | Discharged, readmitted, and discharged |
| P7/M/47 | Gratland et al.24 | USA | Cough, SOB, fever, hypoxia, respiratory distress, increased abdominal pain | Small bowel ischemia with perforation | Exploratory laparotomy | Death |
| P8/M/60 | Vulliamy et al.56 | England | Loss of sensation and power in legs, cough, fever, tachypnoea and tachycardia | Acute thrombotic occlusion of the infrarenal aorta extending into the common iliac arteries | Thromboembolectomy | NR |
| P9/M/75 | Vulliamy et al.56 | England | Abdominal pain, vomiting, worsening of the cough, SOB | Intraluminal thrombus of descending thoracic aorta with embolic occlusion of SMA | Catheter-directed thrombolysis, laparotomy (resection of 150 cm of ischemic small bowel) | NR |
| P10/M/73 | Chun et al.57 | USA | Fever, nonproductive cough, SOB, bloody diarrhea, sudden left lower quadrant cramping pain | Mucosal hyperenhancement with mass-like thickening of the distal sigmoid colon, regional air within the mesenteric vessels (ischemic colitis) | Managed conservatively | Death |
| P11/M/70 | Farnia et al.58 | Italy | Diffuse abdominal pain, nausea, fever, cough, pharyngodynia, positive Blumberg sign | Occlusion of SMA, small bowel wall thinning and absence of contrast enhancement (ischemia) | NR | Death |
| P12/F/28 | Ignat et al.27 | France | Sudden increased abdominal pain and vomiting | Superior mesenteric and portal vein thrombosis followed by segmental small bowel ischemia | Bowel resection, temporary laparostomy (second-look procedure 48 h later); double jejunostomy and abdominal wall closure | Discharged |
| P13/M/56 | Ignat et al.27 | France | Acute respiratory distress | Mesenteric venous gas in proximal jejunum, small bowel ischemia | Bowel resection, laparostomy (second-look procedure and double ostomy 48 h later) | Hospitalized (ICU) |
| P14/M/67 | Ignat et al.27 | France | Acute respiratory distress | Single inflammatory bowel loop with wall thickening and edema, inflammatory segmental ileitis | NR | Hospitalized (ICU) |
| P15/M/62 | Norsa et al.59 | Italy | Abdominal pain and bilious vomiting | Thromboembolism of superior mesenteric vein, jejunal overdistension and un-enhancement, pneumatosis intestinalis (ischemia) | Small intestine resection | Death |
| P16/M/72 | Besutti et al.60 | Italy | Dry cough, SOB, severe abdominal pain | Thrombi in SMA and thoracic descending aorta, decreased or absent wall enhancement of small bowel loops (ischemia), associated splenic and renal infarcts | Resection of the ischemic bowel loop and splenectomy | Discharged and readmitted |
| P17/M/76 | Almeida et al.61 | Spain | Pneumonia, several episodes of hematochezia | Necrotizing pancreatitis, colonic wall thickening, absent of wall enhancement and mesenteric stranding (colonic ischemia) | NR | Death |
| P18/M/68 | Almeida et al.61 | Spain | Bilateral pneumonia, abdominal pain, paralytic ileus with signs of peritoneal irritation | Cecal perforation, pneumatosis of left colon | Laparotomy, peritoneal lavage an ileostomy | Death |
| P19/M/56 | Almeida et al.61 | Spain | Respiratory failure, abdominal distension with pain and guarding | Pneumoperitoneum, colonic pneumatosis | NR | Death |
| P20/M/46 | Dane et al.47 | USA | Fever, SOB, epigastric pain, weakness | Partial thrombosis of abdominal aorta and celiac trunk expending into common hepatic artery, SMA thrombosis, left sided renal and splenic infarcts | NR | NR |
| P21/M/51 | Olson et al.29 | USA | NR | Diffuse gastric wall thickening and pneumatosis with surrounding inflammation and portal vein thrombus and gas, esophagogastroscopic image shows gastric ischemia | NR | NR |
| P22/F/46 | Olson et al.29 | USA | NR | Multifocal small and large bowel wall thickening creating ribbonlike appearance, likely related to small vessel ischemia | NR | NR |
| P23/M/55 | Cheung et al.62 | USA | Fever, generalized abdominal pain, diarrhea, nausea | Low density clot causing high grade narrowing of the proximal SMA | Laparotomy, SMA thromboembolectomy (resection of 8 in. of necrotic small bowel followed by primary anastomosis) | Discharged and readmitted |
| P24/F/79 | De Barry et al.46 | France | Fever, epigastric abdominal pain, diarrhea, SOB | Spontaneous hyperdensity in the right portal vein (right portal vein thrombosis), proximal thrombosis of SMA, ischemia of caecum and small intestine | Thrombolysis and thrombectomy of the SMA | Death |
| P25/M/49 | English et al.63 | UK | Fever, SOB, cough | Hyperperfusion of the distal small bowel with intramural gas, ischemic distal small bowel | Laparotomy, laparoscopy | Hospitalized (ICU) |
| P26/M/56 | Seelinger et al.64 | France | NR | Small bowel ischemia was detected | Open small bowel resection, second look and double barrel ileostomy 48 h later | Hospitalized (ICU) |
| P27/F/70 | Seelinger et al.64 | France | NR | Sigmoid ischemia was detected, rectosigmoidoscopy found ulcerative and ischemic changes | Open rectosigmoid resection (Hartmann) | Hospitalized (ICU) |
| P28/F/61 | Karna et al.65 | India | NR | Thrombosis of distal SMA with dilated jejunoileal loops and normal enhancing bowel wall | Laparotomy | Death |
| P29/F/80 | Neto et al.66 | Brazil | Fever, cough, SOB, diffuse abdominal pain, stiffness | Extensive pneumoperitoneum | Laparotomy, rectosigmoidectomy with terminal colostomy | Death |
| P30/F/61 | Hoyo et al.67 | Spain | Severe acute abdominal pain, vomiting | Portal vein thrombosis, complete thrombosis of the splenoportal axis, mesenteric and splenic ischemia were observed in second CT scan | NR | Death |
| P31/M/30 | Pang et al.28 | Singapore | Colicky abdominal pain, vomiting | Superior mesenteric vein thrombosis with diffuse mural thickening and fat standing of multiple jejunal loops | Laparotomy | Discharged, readmitted, and discharged |
Noted. NR: Not reported, M: Male, F: Female, SOB: Shortness of breath, SMA: Superior mesenteric artery, ICU: Intensive care unit.
The most common initial presentations were fever (10, 32.2%), dry cough (9, 29%), and shortness of breath (8, 25.8%). Additionally, GI symptoms were compromised of abdominal pain (19, 61.3%) and nausea and vomiting (10, 32.2%). The significant GI imaging findings of these patients were comprised of mesenteric arterial or venous thromboembolism, followed by imaging signs of small bowel ischemia. Moreover, 11 patients (35.4%) required direct ICU admission. Nine patients (29%) demonstrated arterial compromise, resulting in bowel ischemia with evident superior mesenteric artery thromboembolism. Six patients (19.3%) demonstrated occluding thrombi in the portal system or superior mesenteric vein. In the remaining patients, no filling defect on imaging was identified to suggest occlusion of the large to medium size abdominal vasculature.
Eventually, five (16.1%) patients were discharged from the hospital, of whom 4 patients (12.9%) were readmitted. Five other patients (16.1%) remained hospitalized in ICU at the time of publication of the reports, and 12 (38.7%) patients died. The outcome is unknown/not reported in 9 patients (29%). Laparotomy and partial bowel resection were performed in twenty patients, of whom six patients died within few days after resection, eight patients transferred to ICU after the operation, and one patient (no. 12) was successfully discharged after resection of 80 cm long segment jejunal ischemia and 7 days of ICU stay. The outcome of the other five patients was not reported/published in the included articles. Patient no. 15 had a past medical history of diabetes type 2, obesity, arterial hypertension, and hepatic cirrhosis (non-alcoholic steatohepatitis) and presented with extensive thromboembolic involvement of superior mesenteric vein and associated pneumatosis intestinalis. He died 12 hours after small bowel resection due to refractory septic shock. Patient no. 18 had a past medical history of diabetes type 2 and hypertension; twelve days after admission to the ICU, the follow-up CT scan demonstrated a cecal perforation with left colonic pneumatosis. Laparotomy then confirmed fecaloid peritonitis, gangrenous cecal perforation, and diffuse small bowel and colon ischemia. The patient died a few days after the operation.
Patient no. 2 had an unremarkable past medical history, except for mild hypertension. Laparotomy and bowel resection (15-cm) with side-to-side manual anastomosis were performed to treat his ischemic bowel, but he died due to the multiorgan failure on the 4th postoperative day. Patient no. 6 improved clinically after 7th days of antiviral therapy after confirmed COVID-19 pneumonia and then discharged from the hospital. He was readmitted with severe clinical conditions like fever and several episodes of diarrhea without any signs of an acute abdomen. A contrast-enhanced abdominopelvic CT revealed arterial thrombosis involving SMA and bowel distension. Subsequently, intestinal resection with stapled side-to-side anastomosis was performed for the management of his intestinal ischemia. He was discharged uneventfully after surgery with regular GI function. Surgical exploration found the necrotic bowel extending from the ligament of Treitz to the transverse colon with perforation of the terminal Ileum in patient no. 7.
In total, 12 patients (38.7%) of the population included in our systematic review died from mesenteric ischemia. All of these cases demonstrated moderate to severe GI symptomatic presentation, of whom 11 patients (35.5%) died within a few days after bowel resection due to refractory septic shock or multiorgan failure. The only case who was treated non-surgically and died, was case no. 10, because the surgery consultants believed the surgery is of no added value. The contrast-enhanced abdominopelvic CT scan of this patient revealed ischemic colitis evident by thickening of the distal sigmoid colon along with pockets of gas within the mesenteric vessels. Reportedly, the patient was managed conservatively with bowel rest and antibiotic therapies; His hospital stay was complicated by cardiac arrest on day 5 of admission. He expired in 20 min despite advanced cardiovascular life support (ACLS).
Contrast-enhanced abdominopelvic CT scan of patient no. 13 revealed mesenteric venous gas and small bowel ischemia. The histopathologic evaluation confirmed the inflammatory necrosis of the mucosa, which was entirely replaced by phantom cells after laparotomy and bowel resection. This patient was suffering from multi-organ failure and was still admitted in ICU at the time of the submission of the manuscript. Patient no. 16 had a past medical history of diabetes type 2, hypertension, renal failure (stage III), coronary artery disease, and myocardial infarction. Subsequent to laparotomy, resection of the ischemic bowel loop, and splenectomy, he was discharged from the ICU, but readmitted due to recurrent mild abdominal pain.
In our systematically reviewed population, radiologically detectable mesenteric arterial thrombosis was detected in 9 patients (29%) and venous thrombosis in 6 patients (19.3%). In 16 patients (51.6%) no evident macroscopic thrombi were detected on contrast-enhanced CT abdomen and pelvis. The frequency of the radiologic findings of bowel ischemia in the reported cases (total of 41 imaging signs in 31 patients) are as follows: thrombosis in the arterial system (SMA and/or aorta) in 10 patients (24%), thrombosis in a portal system in 6 patients (14.6%), bowel wall unenhancement in 6 cases (14.6%), bowel wall thickening in 6 cases (14.6%), pneumatosis intestinalis in 5 cases (12%), mesenteric edema and stranding in 4 patients (9.7%), portal venous gas in 2 patients (4.8%), and pneumoperitoneum indicating perforation in 2 cases (4.8%).
4. Discussion
Life-threatening GI complications of COVID-19 infection mainly encompass ischemic infarction of the small and large bowel loops. In our study, radiologically detectable arterial/venous thrombosis was identified in almost half of patients with ischemic bowel. This study amplifies the importance of the macro- and microvascular involvement in the pathophysiology of the bowel ischemia in the background of SARS-CoV-2 infection.
According to genome sequence analysis, COVID-19 belongs to the Betacoronavirus genus, which also includes the severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS) coronaviruses.32 , 33 GI manifestations were reported in about one-third of hospitalized MERS patients according to the study in Saudi Arabia.34 The most frequent GI tract symptoms were diarrhea, followed by nausea, vomiting, and abdominal pain.35 More severe extrapulmonary manifestations and complications, such as GI complications, were reported in three ICU admitted MERS patients with respiratory failure. One patient developed ischemic bowel with pneumatosis intestinalis on CT, requiring hemicolectomy.36 Laparotomy findings for the other two patients were not significant. Also, elevated aminotransferase levels were present in some patients during hospitalization, which indicated liver damage.
Notably, enteric involvement was also demonstrated in patients with SARS infection, an epidemic in 2002 to 2003.37 , 38 GI symptoms in patients with SARS are overall the same as MERS36. Intestinal biopsy specimens from SARS patients showed significant active viral replication within the intestine cells.39 Mildly elevated aminotransferase levels due to liver dysfunction and acute abdomen requiring exploratory laparotomy were also reported in several cases of SARS.40
By detecting anal swabs and fecal samples positive for COVID-19, the fecal-oral route of the disease transmission has been suggested.41 , 42 Following the involvement of the GI tract mucosa by the virus, malabsorption, and diarrhea are the consequences of the infected enterocyte malfunction. Among the common presenting symptoms of the disease, GI-related complaints are seen in up to 38% of patients, among which diarrhea, nausea/vomiting and abdominal pain are the most common complaints.43
The imaging characteristics of the patients presenting with GI dominant manifestations include distended fluid-filled bowel loops with post-contrast enhancement and surrounding fat stranding/mesenteric inflammation.44 However, more severe cases of bowel wall necrosis leading to surgical resection have also been reported.45 Reports of the GI complications following COVID-19 are evolving.27 , 46 The most clinically significant imaging findings reported are pneumatosis intestinalis and portal venous gas, the two alarming signs of bowel ischemia. Although fluid-filled dilated loops have been frequently reported, this finding is associated with diarrhea, a common but nonspecific manifestation of the COVID-19 and other viral infections.23
In this study, nine patients demonstrated underlying radiologically detectable medium to extensive arterial thrombosis, and six patients were suffering from portal venous system thrombosis. An abdominal CT scan of patient no. 6 revealed thrombosis of far distal SMA branches. Moreover, in the category of radiologically visible arterial compromise, imaging finding of patient no. 11 was solely SMA occlusion by thromboembolism. Such arterial thromboembolic events, along with the mentioned six cases of the portal vein and superior mesenteric vein thrombosis, raise the suspicion of an underlying hypercoagulative state as a significant contributing factor in ischemic bowel disease in COVID-19 patients. Consistent with this review results, Dane et al. reported that solid organ infarction in COVID-19 patients follows vasculature patterns and may result from microthrombi.47 Among the cases of bowel ischemia in the setting of COVID-19 in our systematic review, the most common non-thrombotic radiologic findings were bowel wall unenhancement (which is a specific sign for ischemia) and bowel wall thickening (which is probably the more sensitive sign of ischemia).
Multiple factors are described in the pathogenesis of thrombotic events in patients with COVID-19. These factors include endothelial inflammation, thrombin formation, complement activation and initiation of the immune response, which result in thrombotic complications, such as deep venous thrombosis and arterial ischemia.48., [49], [50] Additionally, several contributing factors should be considered in the pathogenesis of bowel wall ischemia in COVID-19 patients, including ICU admissions, positive pressure ventilation, and viral enteritis. Microvascular thrombosis, non-occlusive mesenteric ischemia, and gross arterial and venous thrombosis have been suspected as culprits for fatal bowel wall necrosis. Furthermore, the COVID-19-associated coagulopathy in critically ill patients has been demonstrated and can result in microangiopathy, massive vessel thrombosis, and significant thromboembolic complications.23 , 45 , 51 , 52
Both macrovascular and microvascular thrombotic events are common in infected patients. It has been reported that the microvascular complications are detected in the early stages of the disease, and macrovascular events are frequently observed in severely ill patients. Now, it is well known that the hypercoagulable state induced by the COVID-19 infection results in micro and macrovascular complications.53 In recent studies, it has been suggested that all hospitalized COVID-19 patients should undergo routine coagulation profile monitoring and thromboembolism prophylaxis.51
Contrast-enhanced abdominopelvic CT scan has a pivotal role in the early diagnosis of ischemic bowel disease. It should be considered in any cases of COVID-19 with prominent GI signs and symptoms, especially those admitted in ICU. This modality allows to detect associated vascular findings and identifying those patients who may benefit from percutaneous endovascular thrombectomy as well.54 In our studied population, CT scan results revealed one patient (no. 14) with short segment bowel edema and diffuse wall thickening involving a single loop, concerning for inflammatory ileitis. Therefore, CT scan led the surgeon to a medical rather than surgical treatment approach. The patient underwent conservative management with a favorable outcome. Small or large bowel un-enhancement after contrast injection, along with pneumatosis intestinalis and portal venous gas, prompt lifesaving surgical intervention in the setting of acute bowel necrosis.24 By the progression of the COVID-19 pandemic, new aspects of the disease morbidity are being sought. Meanwhile, spotting different clinical and imaging characteristics of these complications help the physicians prognosticate and take action to prevent unfavorable outcomes.68 , 69 , 70
To our knowledge, this review study represents all cases of arterial/venous thrombosis in COVID-19 patients with bowel ischemia to date; it implies that further research is needed to identify patients most at risk of acute GI ischemia according to the published reports of increased thrombo-occlusive and thromboembolic events in COVID-19. Moreover, a larger sample of cases is needed for specifying the early imaging findings of GI ischemia in patients with COVID-19. Additionally, standardized management guidelines and patient selection strategies are needed considering prophylactic anticoagulant regimen in these patients.
5. Conclusion
Macrovascular arterial/venous thrombosis can be depicted in almost half of COVID-19 patients with bowel ischemia. Overall mortality in COVID-19 patients with GI ischemia and radiologically evident mesenteric ischemia was 38% and 40%, retrospectively. Although many attributing factors are considered in the pathogenesis of bowel ischemia, factors such as hypercoagulative state, non-occlusive mesenteric ischemia, and microvascular thrombosis constitute the underlying pathology in the majority of COVID-19 patients with GI ischemic presentation.
Abbreviations
Funding information
The authors state that this work has not received any funding.
Informed consent
Informed consent was not required because it was a literature study.
Ethical approval
Institutional Review Board approval was not required because it was a literature study.
CRediT authorship contribution statement.
Pedram Keshavarz: Investigation, Writing - original draft, Writing - review & editing, Data curation. Faranak Rafiee: Investigation, Writing - review & editing, Data curation. Hadiseh Kavandi: Writing - review & editing, Data curation. Sogand Goudarzi: Data curation, Writing - review & editing, Investigation. Firouzeh Heidari: Investigation, Data curation, Writing - review & editing. Ali Gholamrezanezhad: Data curation, Writing - original draft, Writing - review & editing, Conceptualization, Investigation.
Declaration of competing interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Acknowledgments
The authors would like to thank Dr. Rajesh Bhayana, Dr. Mihaela Ignat, Dr. Michael C. Olson, and Dr. Jolyn Hui Qing Pang for providing the images and figures for this study and also, thank to publisher holder of Radiology (RSNA), RadioGraphics, Surgery Journal, and Annals of Vascular Surgery for granting permission to use the images.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.clinimag.2020.11.054.
Appendix A. Supplementary data
Supplementary tables
References
- 1.Guan W.-j., Ni Z.-y., Hu Y., et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. Jama. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 3.World Health Organization Coronavirus disease (COVID-2019) situation reports. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/;
- 4.Wang D., Hu B., Hu C., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. Jama. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li Q., Guan X., Wu P., et al. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. N Engl J Med. 2020;382(13):1199–1207. doi: 10.1056/NEJMoa2001316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Salehi S., Abedi A., Balakrishnan S., Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19) imaging reporting and data system (COVID-RADS) and common lexicon: a proposal based on the imaging data of 37 studies. E Eur Radiol. 2020;1 doi: 10.1007/s00330-020-06863-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tian Y., Rong L., Nian W., He Y. Gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51(9):843–851. doi: 10.1111/apt.15731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterol 2020;158(6):1831–1833. e1833. doi:10.1053/j.gastro.2020.02.055. [DOI] [PMC free article] [PubMed]
- 9.Behzad S., Velez E., Najafi M.H., Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19) pneumonia incidentally detected on coronary CT angiogram: a do-not-miss diagnosis. Emerg Radiol. 2020;1 doi: 10.1007/s10140-020-01802-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pan L., Mu M., Yang P., et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: a descriptive, cross-sectional, multicenter study. Am J Gastroenterol. 2020;115(5):766–773. doi: 10.14309/ajg.0000000000000620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaafarani H.M., El Moheb M., Hwabejire J.O., et al. Gastrointestinal complications in critically ill patients with COVID-19. Ann Surg. 2020 doi: 10.1097/SLA.0000000000004004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Amini A., Nagalli S. StatPearls Publishing; 2020. Bowel ischemia. StatPearls [internet] [PubMed] [Google Scholar]
- 13.Corcos O., Nuzzo A. Gastro-intestinal vascular emergencies. Best prac Res Clin Gastroenterol. 2013;27(5):709–725. doi: 10.1016/j.bpg.2013.08.006. [DOI] [PubMed] [Google Scholar]
- 14.Connors J.M., Levy J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood, Am Soc Hematol. 2020;135(23):2033–2040. doi: 10.1182/blood.2020006000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Klok F., Kruip M., Van der Meer N., et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145–147. doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wichmann D., Sperhake J.-P., Lütgehetmann M., et al. Autopsy findings and venous thromboembolism in patients with COVID-19: a prospective cohort study. Ann Intern Med. 2020 doi: 10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Salehi S., Gholamrezanezhad A. Reply to "segmental pulmonary vascular changes in COVID-19 pneumonia". AJR Am. J. Roentgenol. 2020;W34 doi: 10.2214/ajr.20.23672. [DOI] [PubMed] [Google Scholar]
- 18.Salehi S, Abedi A, Gholamrezanezhad A. Reply to" Vascular Changes Detected With Thoracic CT in Coronavirus Disease (COVID-19) Might Be Significant Determinants for Accurate Diagnosis and Optimal Patient Management". AJR Am J Roentgenol 2020;215(1). doi:10.2214/AJR.20.23339. [DOI] [PubMed]
- 19.Eslambolchi A., Aghaghazvini L., Gholamrezanezhad A., Kavosi H., Radmard A.R. Coronavirus disease 2019 (COVID-19) in patients with systemic autoimmune diseases or vasculitis: radiologic presentation. J Thromb Thrombolysis. 2020:1–10. doi: 10.1007/s11239-020-02289-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grootjans J., Lenaerts K., Buurman W.A., Dejong C.H., Derikx J.P. Life and death at the mucosal-luminal interface: new perspectives on human intestinal ischemia-reperfusion. World J Gastroenterol. 2016;22(9):2760–2770. doi: 10.3748/wjg.v22.i9.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Roussel A., Castier Y., Nuzzo A., et al. Revascularization of acute mesenteric ischemia after creation of a dedicated multidisciplinary center. J Vasc Surg. 2020;62(5):1251–1256. doi: 10.1016/j.jvs.2015.06.204. [DOI] [PubMed] [Google Scholar]
- 22.Kooraki S., Hosseiny M., Myers L., Gholamrezanezhad A. Coronavirus (COVID-19) outbreak: what the department of radiology should know. J Am Coll Radiol. 2020 doi: 10.1016/j.jacr.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bhayana R., Som A., Li M.D., et al. Abdominal imaging findings in COVID-19: preliminary observations. Radiology. 2020;201908 doi: 10.1148/radiol.2020201908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gartland R.M., Velmahos G.C. Bowel necrosis in the setting of COVID-19. J Gastrointest Surg. 2020:1–2. doi: 10.1007/s11605-020-04632-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bikdeli B., Madhavan M.V., Jimenez D., et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up. JACC State-of-the-Art Review J Am Coll Cardiol. 2020;75(23):2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moher D., Liberati A., Tetzlaff J., Altman D.G., Group P. Reprint—preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Phys Ther. 2009;89:873–880. doi: 10.2522/ptj.2009.89.9.870. [DOI] [PubMed] [Google Scholar]
- 27.Ignat M., Philouze G., Aussenac-Belle L., et al. Small bowel ischemia and SARS-CoV-2 infection: an underdiagnosed distinct clinical entity. Surgery. 2020;168(1):14–16. doi: 10.1016/j.surg.2020.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.JHQ Pang, Tang J.H., Eugene-Fan B., Lee C.L., Low J.K. A peculiar case of small bowel stricture in a COVID-19 patient with congenital adhesion band and superior mesenteric vein thrombosis. Ann Vas Surg. 2020 doi: 10.1016/j.avsg.2020.08.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Olson MC, Lubner MG, Menias CO, Mellnick VM, Gettle LM, Kim DH, et al. RadioGraphics Update: Venous Thrombosis and Hypercoagulability in the Abdomen and Pelvis—Findings in COVID-19. RadioGraphics 2020;40(5):200119. doi:10.1148/rg.2020200119. [DOI] [PMC free article] [PubMed]
- 30.National Heart L aBIw Study quality assessment tools. http://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools.Accessed
- 31.Margulis A.V., Pladevall M., Riera-Guardia N., et al. Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the Newcastle–Ottawa scale and the RTI item bank. Clin Epidemiol. 2014;6:359–368. doi: 10.2147/CLEP.S66677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Munster V.J., Koopmans M., van Doremalen N., van Riel D., de Wit E. A novel coronavirus emerging in China—key questions for impact assessment. N Engl J Med. 2020;382(8):692–694. doi: 10.1056/NEJMp2000929. [DOI] [PubMed] [Google Scholar]
- 33.Hosseiny M., Kooraki S., Gholamrezanezhad A., Reddy S., Myers L. Radiology perspective of coronavirus disease 2019 (COVID-19): lessons from severe acute respiratory syndrome and Middle East respiratory syndrome. AJR Am J Roentgenol. 2020;214(5):1078–1082. doi: 10.2214/AJR.20.22969. [DOI] [PubMed] [Google Scholar]
- 34.Assiri A., Al-Tawfiq J.A., Al-Rabeeah A.A., et al. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect Dis. 2013;13(9):752–761. doi: 10.1016/S1473-3099(13)70204-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Drosten C., Seilmaier M., Corman V.M., et al. Clinical features and virological analysis of a case of Middle East respiratory syndrome coronavirus infection. Lancet Infect Dis. 2013;13(9):745–751. doi: 10.1016/S1473-3099(13)70154-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arabi Y.M., Arifi A.A., Balkhy H.H., et al. Clinical course and outcomes of critically ill patients with Middle East respiratory syndrome coronavirus infection. Ann Intern Med. 2014;160(6) doi: 10.7326/M13-2486. [DOI] [PubMed] [Google Scholar]
- 37.Booth C.M., Matukas L.M., Tomlinson G.A., et al. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. Jama. 2003;289(21):2801–2809. doi: 10.1001/jama.289.21.JOC30885. [DOI] [PubMed] [Google Scholar]
- 38.Tsang K.W., Ho P.L., Ooi G.C., et al. A cluster of cases of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348(20):1977–1985. doi: 10.1056/NEJMoa030666. [DOI] [PubMed] [Google Scholar]
- 39.Leung W.K., To K.-f., Chan P.K., et al. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterol. 2003;125(4):1011–1017. doi: 10.1016/j.gastro.2003.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang J.-Z. Severe acute respiratory syndrome and its lesions in digestive system. World J Gastroenterol: WJG. 2003;9(6):1135–1138. doi: 10.3748/wjg.v9.i6.1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang W., Du R.-H., Li B., et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9(1):386–389. doi: 10.1080/22221751.2020.1729071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lin L., Jiang X., Zhang Z., et al. Gastrointestinal symptoms of 95 cases with SARS-CoV-2 infection. Gut. 2020;69(6):997–1001. doi: 10.1136/gutjnl-2020-321013. [DOI] [PubMed] [Google Scholar]
- 43.Pamplona J., Solano R., Soler C., Sàbat M. Epidemiological approximation of the enteric manifestation and possible fecal-oral transmission in COVID-19: a preliminary systematic review. Infect Dis. 2020 doi: 10.21203/rs.3.rs-33873/v1. [DOI] [PubMed] [Google Scholar]
- 44.Behzad S., Aghaghazvini L., Radmard A.R., Gholamrezanezhad A. Extrapulmonary manifestations of COVID-19: radiologic and clinical overview. Clin Imaging. 2020;66:35–41. doi: 10.1016/j.clinimag.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bianco F., Ranieri A., Paterniti G., Pata F., Gallo G. Acute intestinal ischemia in a patient with COVID-19. Tech Coloproctol. 2020;1 doi: 10.1007/s10151-020-02255-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.de Barry O., Mekki A., Diffre C., Seror M., Hajjam M.E., Carlier R.-Y. Arterial and venous abdominal thrombosis in a 79-year-old woman with COVID-19 pneumonia. Radiol Case Rep. 2020;15(7):1054–1057. doi: 10.1016/j.radcr.2020.04.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dane B., Smereka P., Wain R., Kim D., Katz D.S. Hypercoagulability in COVID-19: identification of arterial and venous thromboembolism in the abdomen, pelvis, and lower extremities. AJR Am J of Roentgenol. 2020 doi: 10.2214/AJR.20.23617. [DOI] [PubMed] [Google Scholar]
- 48.Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res 2020;2020:1–13 doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed]
- 49.McFadyen J.D., Stevens H., Peter K. The emerging threat of (micro) thrombosis in COVID-19 and its therapeutic implications. Circ Res. 2020;127(4):571–587. doi: 10.1161/CIRCRESAHA.120.317447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Akbari H., Tabrizi R., Lankarani K.B., et al. The role of cytokine profile and lymphocyte subsets in the severity of coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. Life Sci. 2020 doi: 10.1016/j.lfs.2020.118167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Iba T., Levy J.H., Levi M., Connors J.M., Thachil J. Coagulopathy of coronavirus disease 2019. Crit Care Med. 2020 doi: 10.1097/CCM.0000000000004458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fogarty H., Townsend L., Ni Cheallaigh C., et al. COVID19 coagulopathy in Caucasian patients. Br J Haematol. 2020;189:1044–1049. doi: 10.1111/bjh.16749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Colling ME, Kanthi Y. COVID-19-associated coagulopathy: An exploration of mechanisms. Vascular Med 2020:1358863X20932640. doi:10.1177/1358863X20932640. [DOI] [PMC free article] [PubMed]
- 54.Azouz E., Yang S., Monnier-Cholley L., Arrivé L. Systemic arterial thrombosis and acute mesenteric ischemia in a patient with COVID-19. Intensive Care Med. 2020;1 doi: 10.1007/s00134-020-06079-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Beccara L., Pacioni C., Ponton S., Francavilla S., Cuzzoli A. Arterial mesenteric thrombosis as a complication of SARS-CoV-2 infection. Eur J of Case Rep Intern Med. 2020;7(5) doi: 10.12890/2020_001690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vulliamy P., Jacob S., Davenport R.A. Acute aorto-iliac and mesenteric arterial thromboses as presenting features of COVID-19. Br J Haematol. 2020;189:1050–1063. doi: 10.1111/bjh.16760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chan K.H., Lim S.L., Damati A., et al. Coronavirus disease 2019 (COVID-19) and ischemic colitis: an under-recognized complication. Am J Emerg Med. 2020 doi: 10.1016/j.ajem.2020.05.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Farina D., Rondi P., Botturi E., et al. Gastrointestinal: bowel ischemia in a suspected coronavirus disease (COVID-19) patient. J Gastroenterol Hepatol. 2020 doi: 10.1111/jgh.15094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Norsa L., Valle C., Morotti D., Andrea B.P., Indriolo A., Aurelio S. Intestinal ischemia in the COVID-19 era. Dig Liver Dis. 2020 doi: 10.1016/j.dld.2020.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Besutti G., Bonacini R., Iotti V., et al. Abdominal visceral infarction in 3 patients with COVID-19. Emerg Infect Dis. 2020;26(8) doi: 10.3201/eid2608.201161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Almeida A., Azcárate V.V., Justicia C.S., et al. Severe colon ischemia in patients with severe COVID-2019 infection: a report of three cases. Int J Colorectal Dis. 2020 Researchsquare. EPub ahead of print. [Google Scholar]
- 62.Cheung S, Quiwa JC, Pillai A, Onwu C, Tharayil ZJ, Gupta R. Superior mesenteric artery thrombosis and acute intestinal ischemia as a consequence of COVID-19 infection. Am J Case Rep 2020;21:e925753-1. doi:10.12659/AJCR.925753. [DOI] [PMC free article] [PubMed]
- 63.English W., Banerjee S. Coagulopathy and mesenteric ischaemia in severe SARS-CoV-2 infection. ANZ J Surg. 2020;90(9):1826. doi: 10.1111/ans.16151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Seeliger B., Philouze G., Benotmane I., Mutter D., Pessaux P. Is the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) present intraperitoneally in patients with coronavirus disease 2019 (COVID-19) infection undergoing emergency operations? Surgery. 2020 doi: 10.1016/j.surg.2020.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Karna S.T., Panda R., Maurya A.P., Kumari S. Superior mesenteric artery thrombosis in COVID-19 pneumonia: an underestimated diagnosis—first case report in Asia. Indian Journal of Surgery. 2020:1–3. doi: 10.21203/rs.3.rs-31156/v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.IJFC Neto, Viana K.F., da Silva M.B.S., et al. Perforated acute abdomen in a patient with COVID-19: an atypical manifestation of the disease. J Coloproctol. 2020;40(3):269–272. doi: 10.1016/j.jcol.2020.05.011. [DOI] [Google Scholar]
- 67.Del Hoyo J., López-Muñoz P., Fernández-de la Varga M., Garrido-Marín A., Valero-Pérez E., Prieto M., et al. Hepatobiliary and Pancreatic: A fatal case of extensive splanchnic vein thrombosis in a patient with Covid-19. J. Gastroenterol. Hepatol. 2020:1. doi: 10.1111/jgh.15174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Fields B.K.K., Demirjian N.L., Dadgar H., Gholamrezanezhad A. 2020. Imaging of COVID-19: CT, MRI, and PET. (Semin. Nucl. Med.). S0001-2998(20)30123-9. Epub ahead of print. PMID: 33288215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Katal S., Gholamrezanezhad A. Neuroimaging findings in COVID-19: A narrative review. Neurosci. Lett. 2020:135529. doi: 10.1016/j.neulet.2020.135529. Epub ahead of print. PMID: 33248161; PMCID: PMC7686796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lombardi A.F., Afsahi A.M., Gupta A., Gholamrezanezhad A. Severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), influenza, and COVID-19, beyond the lungs: a review article. Radiol. Med. 2020:1–9. doi: 10.1007/s11547-020-01311-x. Epub ahead of print. PMID: 33242204; PMCID: PMC7689190. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary tables