Abstract
Severe Acute Respiratory Syndrome Corona Virus 2 (SARS-CoV-2) outbreak is a major public health concern, which has accounted for >1.7 million deaths across the world. A surge in the case fatality ratio as compared with the infection ratio has been observed in most of the countries. The novel Coronavirus SARS-CoV-2 shares the most common sequence with SARS-CoV, but it has a higher rate of transmission. The SARS-CoV-2 pathogenesis is initiated by the binding of viral spike protein with the target receptor Angiotensin-Converting Enzyme 2 (ACE2) facilitating virus internalization within host cells. SARS-CoV-2 mainly causes alveolar damage ranging from mild to severe clinical respiratory manifestations. Most of the cases have revealed the association of Coronavirus disease with patients having earlier comorbidities like Hypertension, Diabetes mellitus, and Cerebrovascular diseases. Pharmacological investigation of the SARS-Cov-2 patients has revealed the frequent use of drugs belongs to Angiotensin-converting enzyme inhibitors (ACEi) and/or Angiotensin II type I receptor blockers (ARBs). Interestingly, a significant increase in ACE2 expression was noticed in patients routinely treated with the above group of drugs were also reported. To date, the association of ACEi and/or ARBs with the up-regulation of ACE2 expression has not been defined distinctively. The proposed review will focus on the pathways which are responsible for the upregulation of ACE2 and its impact on gravity of SARS-CoV-2 disease.
Keywords: COVID-19, ACE2, Comorbidities, ARB, ACEi, Pathways
1. Introduction
Severe Acute Respiratory Syndrome Corona Virus 2 (SARS-CoV-2) caused Corona Virus Disease 2019 (COVID-19) in more than ~77 million individuals with ~1.7 million fatalities globally. On March 11, 2020, the World Health Organization (WHO) declared COVID-19 a pandemic, with a significant surge in COVID-19 cases reported in most countries. The onset of COVID-19 infection actuates with the migrating of the virus into the respiratory tract and more prominently damaging the alveolar sacs of the respiratory system. SARS-CoV-2 is exceptionally infectious in nature and it spreads through respiratory droplets, thus social distancing has been recommended as a primary preventive measure. The time of incubation for the virus is 1–14 days within the hosts, yet the transmission can result in community spread due to high asymptomatic cases around the world. Statistical analysis has revealed that only 5–10% of the infected individuals show the severe manifestations of COVID-19 disease (Perella et al., 2019; Zhang et al., 2020a; Lai et al., 2020). The mortality of COVID-19 is ~0.2% among young healthy individuals, while the rate is high in old age people with preexisting hypertension and cardiovascular comorbidities. Moreover, SARS-CoV has a mortality rate of 10% as compared with MERS-CoV having a rate of 35%, but these earlier reported a low transmission rate of the virus Azhar et al., (2019); WHO (https://www.who.int/csr/sars/country/2003_08_15/en). But the Confirmed Fatality Ratio (CFR) of SARS-CoV-2 varies by country from less than 0.1%–28% (WHO/2019-nCoV/Sci_Brief/Mortality/2020.1). Major symptoms of SARS-CoV-2 infection comprise fever, headache, mild chest pain, loss of taste/smell, and breathing problems (Wang et al., 2020). Initial attachment of SARS-CoV-2 with the host cells is facilitated by the attachment of viral spike protein with the angiotensin-converting enzyme 2 (ACE2) receptors. The ACE2 expression pattern differs significantly among various tissues and organs. Thus the susceptibility of cells towards the infection eventually depends on the ease of viral internalization using ACE2 (Zhou et al., 2020). A case study in China on COVID-19 affected population reported a significant clinical history of Diabetes and Cerebrovascular comorbidities within 32 out of 52 patients under the Intensive Care Unit (ICU). Likewise, two independent studies on COVID-19 infected population reported the presence of single or multiple comorbidities like Diabetes Mellitus, Hypertension, and Cerebrovascular disease among 1099 patients (Yang et al., 2020; Zhang et al., 2020b). Further, clinical investigation on these groups of patients reported a prolonged use of angiotensin-converting enzyme inhibitors (ACEi) and/or angiotensin II type I receptor blockers (ARBs) for medicaments. Notably, the presence of ACE2 was considerably increased in patients with Type I or II Diabetes mellitus, due administration of ACEi and/or ARBs (Li et al., 2017). Similarly, medication for hypertension with ACEi and ARBs leads to abundant ACE2 expression (Wan et al., 2020). Earlier, a report has suggested the use of ARBs like Olmesartan may defend cardiovascular rearrangement by heart cardiac nitric oxide and accumulation of angiotensin-(1–7) mediated by high expression of ACE2 (Agata et al., 2006). Thus continuous use of ACEi and ARBs to treat the comorbidities leads to the overexpression of ACE2 was clearly reported in many instances, but not properly analyzed. Hence, we have attempted to analyze the plausible pathways relevant to the question in the form of review literature.
2. SARS-COV-2 proteome characteristics
The SARS-CoV-2 genome (~30 Kb) encodes for ORFs and divided into three parts based on the structural and functional roles in pathogenesis. Major portion of the genome codes for 16 non-structural proteins (nsp 1-16) which forms the replicase complex. One third of the SARS-CoV-2 genome codes for accessory (3a, 3 b, 6, 7a, 7 b, 8a, 8 b, 9 b, 9c) and four major structural (Spike, Envelope, Membrane, and Nucleocapsid) proteins (Lu et al., 2019; Zhang et al., 2020c). Among them, Spike protein (S) mainly facilitates the attachment with the host cells (Perlman and Netland, 2009). The size of Spike protein ranges between 180 and 200 kDa bearing an extracellular transmembrane and intracellular C-terminal domains (Bosch et al., 2003). Spike protein has 1273 residues with a major distribution of amino acids in the S1 and S2 subunit; The S1 subunit mainly comprises one N-terminal region (14–305) followed by the receptor-binding domain (319–541). S1/S2 subunit of the Spike proteins attaches to the ACE2 receptors and facilitate the formation of endosomes. Further, the S2 subunit covers the region of fusion peptide (788–806), transmembrane domain (1213–1237), and cytoplasmic domain (1237–1273) (Xia et al., 2020). Both subunits (S1 and S2) together shape the head and stalk structure of Spike protein (Tang et al., 2020).
2.1. ACE2 receptor and SARS-Cov-2 spike protein interactions
Spike protein of SARS-CoV-2 binds with human ACE2 receptors with higher affinity than the S proteins of SARS-CoV (2003). During infection, the S protein interacts with ACE2 through the receptor-binding domain of S1 subunit, facilitating the protein interaction with the host cells for viral attachment and pathogenesis. The high-affinity binding between Spike protein and ACE2 induces a three dimensional rearrangement in the Spike protein, followed by the splitting of the S1/S2 poly basic site by Cathepsin L protease (Wrapp et al., 2020; Wu et al., 2020; Liu et al., 2020). ACE2 mediated SARS-CoV-2 internalization within the host cell is the primary stage of virus infection, but it also requires the type II transmembrane serine protease (TMPRSS2) for the activity of the viral spike protein [21]. Clinical investigations confirmed the potential application of Camostat Mesylate (protease inhibitor) for TMPRSS2 in restricting the virus internalization and acting against SARS-CoV-2 infection (Hoffmann et al., 2020; Matsuyama et al., 2020). ACE2 acts against the actions of ACE by metabolizing its catalytic fibrogenic peptide AngII (angiotensin II) into Ang (1–7). Wang et al. studied the expression pattern for ACE2 through the analysis of genes in the specific tissues; standard range for the expression (−10 to 5). Mild to moderate expression (0–5) was observed in every tissue, but specifically higher expression (average to maximum limit; log2-transform) was in the blood vessel, lungs, ovary, adipose tissue, heart, small intestine, etc (Li et al., 2020). Under comorbid conditions like diabetes, cardiovascular diseases, and cancer, the ACE2 overexpression may balance the detrimental effects of the Angiotensin-II mediated AT1R signaling pathway. Further, studies revealed the abundance of ACE2 in type II alveolar cells and enterocytes, thus facilitating accelerated SARS-CoV-2 multiplication causing severe alveolar damage (Hamming et al., 2004; Turner et al., 2004). Thus, ACE2 acts as a key player in the SARS-CoV-2 pathogenesis on the basis of structural compatibility, stable interaction, and high-affinity binding.
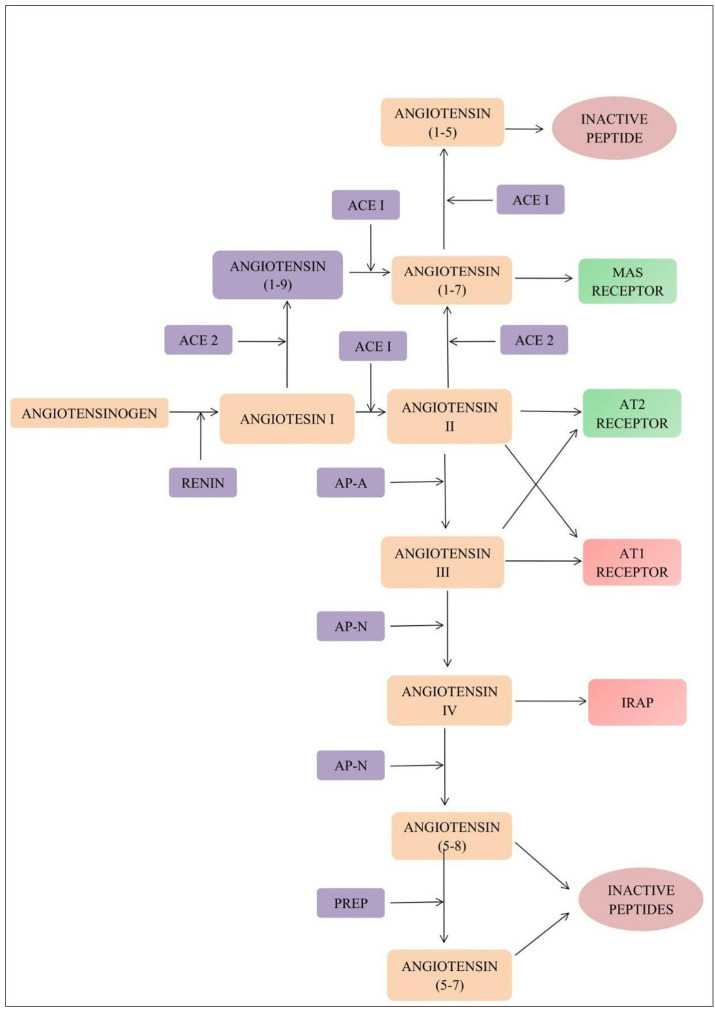
3. Role of ACE2 in the regulation of Renin-angiotensin system
Renin-Angiotensin System (RAS), a hormonal system that controls blood pressure, electrolyte, and volaemia is regulated by the classical angiotensin-converting-enzyme I (ACE1). Later, a homolog of ACE1 was recognized as angiotensin-converting enzyme 2 (ACE2) a carboxypeptidase that is vital for regulating cardiovascular homeostasis (Patel et al., 2012). It forms ang 1–9, ang 1–7 from angiotensin I and angiotensin II respectively (Donoghue et al., 2000). Further it controls the vasodilatation and reduces the hypertrophic effects (Tipnis et al., 2000). In counter-regulation, it cleaves the C-terminal residues from kinetensin, neurotensin, and des-Arg bradykinin peptides but remains ineffective against bradykinin (Vickers et al., 2000). ACE2 also cleaves peptides casomorphins, dynorphin A and apelins (Wang et al., 2016). The RAS system comprises two counter-regulatory mechanisms mediated by ACE, angiotensin type 1 (AT1R), ang II and ACE2, ang (1–7), angiotensin type 2 (AT2R) receptors followed by Mas receptor (MasR). ( Fig. 1 ). The classical enzyme ACE catalyze angiotensin I to angiotensin II which acts like some agonist in mediating vasoconstrictive and pro-inflammatory effects through AT1R. Angiotensin II acts on both AT1R and AT2R as part of the counter-regulatory mechanism, to maintain a proper balance between vasoconstriction and vasodilation in addition to other physiological roles. Angiotensin (Ang I) is degraded by the ACE to form Ang II a substrate for ACE2 (homolog of ACE). The major role of ACE2 is the depletion of Ang II which led the way for the emergence of angiotensin 1-7 to counter the effects of Ang II within the RAS system (Tikellis and Thomas, 2000; Mercure et al., 2008; Oudit and Penninger, 2011). Thus, ACE/Ang I and ACE2/ang 1–7 combos are part of the RAS system, and elevated ACE2 levels are shown to prevent heart failure, diabetes, and hypertension. The above findings confirm the pro-inflammatory activity of ACE2 along with Ang II-mediated AT1R signaling is proved as vital in the RAS system (Alenina et al., 2008; Bader, 2013). The entire RAS system is regulated by AT1R, AT2R, and MasR receptors. AT1R acts as a receptor for the regulation of anti-diuresis and vasoconstriction. But, AT2R and MasR both promote diuresis and vasodilatation. Moreover, MasR was shown to exhibit similar effects of AT2R and attenuates AT1R mediated signaling (Sampaio et al., 2007; Santos et al., 2007; Tallant et al., 2005; Zhang et al., 2014). In comparison, with the downstream signaling pathway of the ACE/Angiotensin II-mediated AT1R axis, the ACE2/Angiotensin 1–7/MasR and ACE2/Angiotensin 1–9/AT2R axis were reported as a physiological regulator against the former axis in activating and balancing the RAS system (Oudit et al. 2003, 2007; Lo et al., 2013; Danilczyk and Penninger, 2006). Based on the reports, ACE2 was determined not only to regulate the RAS system but also to limit the activity of Angiotensin II and ACE.
Fig. 1.
Diagrammatic portrayal of the Renin-angiotensin system. (AP-A: Glutamyl aminopeptidase, AP-N: Alanyl aminopeptidase, PREP: Prolyl endopeptidase, IRAP: Leucyl and cystinyl aminopeptidase).
4. ACEi and ARBs association with the ACE2 overexpression
ACE2 is the predominant regulator against increased vasoconstriction and pro-inflammatory responses induced by angiotensin II type 1 receptor axis. Based on ACE2 abundance in COVID-19 infected people, several studies have suggested the risk of developing COVID-19 with the administration of ACEi and ARBs as indirectly this therapeutics over produce the circulating ACE2 transcripts in the cells (Zheng et al., 2020; Fang et al., 2020; Watkins, 2020). Few studies have somehow suggested the risk of ACEi and ARBs with the development of ACE2 overexpression, but the evidence for approving alternative pharmacological agents over these inhibitors or blockers is still lacking and requires more clinical interventions. Salim S. Al-Rejaie et al. reported that the drug captopril (ACEi family drug) can regulate ACE2 overexpression while downregulating the angiotensin II-dependent AT1 receptor downstream signaling and RANKL expression in osteoporotic rats. The expression of ACE2 was considerably upregulated in comparison with classical ACE expression. Thus, captopril demonstrated a clinical role in the upregulation of the ACE2 dependent Mas receptor signaling cascade in restoring bone metabolism (Esler and Esler, 2020). The therapeutic application of captopril in treating hypertension and cardiovascular comorbidities exhibited positive clinical outcomes. Similarly, Enalapril a known ACEi was reported to increase ACE2 expression in the kidney tissues whereas no such significant fold change was observed in TMPRSS2 mRNA expression (Abuohashish et al., 2017; Saheb et al., 2020). Chappell and his group has reported increased expression of ACE2 mRNA, while optimum reduction in angiotensin II (plasma) expression upon treating the cardiac cells with lisinopril and losartan (Ferrario et al., 2005). A significant change was not observed in the cardiac ACE2 activity by the combination of both drugs. In the case of diabetes, renal ACE2 transcript levels were found to be bountiful after the administration of Losartan. Jessup JA et al. has characterized the ACE2 mRNA found within the heart and kidney of Losartan-treated transgenic rats (Jessup et al., 2006). ACEi and ARBs are individually used as medication for type II diabetes, but dual drug combined therapy can also auto-regulate the ACE2 expression. Further, the in vivo animal model study revealed a plausible association of ACE2 overexpression concerning anti-diabetic medication. Insulin administration in type I and II diabetes mellitus attenuates renal disintegrin and metalloproteinase domain 17 (ADAM-17) activities. ADAM-17 regulates ACE2 inactivation by cleaving off the ACE2 enzyme, but the reduced activity of ADAM-17 can also lead to an upregulated expression of ACE2. The overexpression of ACE2 in heart and lungs of the diabetic animal model was analyzed with Liraglutide administration (Wysocki et al., 2006; Xye-Ying and Jing Bo, 2016). Apart from ACE inhibitors and ARBs, thiazolidinediones also act as a vasodilator. Generally, ACE2 levels are elevated due to the intake of antidiabetic drugs. Similarly, higher expression of ACE2 was also found among the Insulin administered animal models. Miller HD et al. has described the relationship between ACE2 upregulation and administration of glucagon-like peptide 1 (GLP) agonist or dipeptidyl peptidase-4 (DPP) inhibitors to treat diabetes (Dambha-Miller et al., 2020). Further, type 2 antidiabetic drug thiazolidinedione was also reported to up-regulate ACE2 expression In vivo and In vitro models (Dambha-Miller et al., 2020). Thiazolidinediones improve vasodilation in diabetic patients by regulating the signaling cascade linking peroxisome proliferator-activated receptor-gamma () and Renin-Angiotensin System (Sarafidis et al., 2004; Pal and Bhadada, 2020). Roszer and Ricote has summarized and reported the association of and hepatic ACE2 expression with the drug pioglitazone, but a sufficient inference of ACE2 expression in COVID-19 comorbidities was not established (Roszer and Ricote, 2010). Administration of pioglitazone along with ARBs and/or ACEi can be performed in a SARS-CoV-2 based case-control study to determine the plausible pathways linked with ACE2 overexpression. In hypertension and diabetes, ventricular remodeling and dysregulated pathways are attenuated by Ang-(1–7) (Marques et al., 2011; Zhang et al., 2014). Yun Zhang et al. reported improved cardioprotection with perindopril treatment decreasing the formation of Ang II and also inhibiting the generation of angiotensin (1–5) from angiotensin (1–7) (Hao et al., 2015). Thus, perindopril along with the ACE2 activity was found to be associated with decreased formation of angiotensin II followed by ADAM17 down-regulation (Patel et al., 2014; Chappel, 2019). Moreover, treatment with perindopril remarkably improved angiotensin (1–7) levels in plasma. Solar MJ et al. have administered the Telmisartan (belongs to ARB) a drug used to prevent stroke, heart attack, and kidney problems in mice. This study shows the increased ACE2 transcript and protein levels in the kidney of Telmisartan treated mice (Soler et al., 2009). Irbesartan a drug used to treat hypertension was treated to C57BL/6 mice which also reported an increased aortic ACE2 mRNA as well as protein expression (Jin et al., 2012). Both studies establish the role of ACE2 in preventing hypertension. Also, Oudit G Y et al. reported the effect of irbesartan along with ang (1–7) on restoring cardioprotective effects in ACE2-null mice (Patel et al., 2012). Irbesartan blocks the angiotensin II type 1 receptor pathway thus reducing the vasoconstriction load and induces antihypertensive effects. Supplementation of ang (1–7) along with irbesartan reduced superoxide formation and attenuated NADPH oxidase activity. Further, it decreased p47phox and gp91phox activity and inhibited p47phox phosphorylation, thus attenuating NADPH oxidase activation and superoxide formation (Bendall et al., 2002; Zhong et al., 2004; Bodiga et al., 2011). Both, ACEi and ARBs are clearly established to increase the ACE2 levels. Likewise, several other medications such as Spironolactone and Eplerenone (both belong to Aldosterone antagonists) have been reported with ACE2 overexpression in experimental models (Keidar et al., 2005). Atorvastatin and Fluvastatin (belongs to Statin) were routinely used to reduce cholesterol can also upregulate renal and cardiac ACE2 expression (Tikoo et al., 2015; Shin et al., 2017). Notably, GLP agonists (liraglutide), DPP 4 inhibitors (Linagliptin), and NSAIDs (Ibuprofen) were reported to elevate ACE2 expression predominantly in the heart and lungs (Romani-Perez et al., 2015; Zhang et al., 2015; Qiao et al., 2015; Fandino et al., 2018). An alternative to ACEi and ARBs, all-trans retinoic acid (atRA) has notably ameliorated ACE2 articulation in the heart attributing towards the decrease of vasoconstriction in spontaneously hypertensive rats (SHR) suggesting that atRA might act as a therapeutic agent to prevent human essential hypertension (Beswick et al., 2001). Based on research and statistical investigations of COVID-19 infected people, several studies have mentioned important concerns over reconsidering the safety and leftover of ACEi and ARB's therapeutics in treating patients with hyper blood pressure and cardiovascular comorbidities. Though the association of ACEi and ARBs with the ACE2 upregulation in COVID-19 infected comorbidities is evident, researchers have suggested their concern on switching towards an alternative pharmacological agent for therapeutic intervention.
5. Regulatory pathways related to ACE2 expression
An official report proclaimed by the Italian National Institute of Health depicts that the most prevalent comorbidities among COVID patients were associated with arterial hypertension, diabetes mellitus, and ischemic cardiopathy (Onder et al., 2020). Further investigations confirm that 30% of patients exhibiting severe complications were taking ACEi drugs and 14% are taking ARB therapeutics. Recently, few clinical experts have revealed ACE2 overexpression among COVID-19 patient groups having hypertension, cardiovascular and diabetic comorbidities. ACE2 overexpression has also been noticed in patients taking ACEi/ARBs for their treatment. But the mild differences in expression are mainly depending upon the age and sex followed by monotherapy or combined therapy with ACEi and ARBs. ACE2 regulates the MasR signaling axis beside the ACE/Angiotensin II/AT1R regulation. The pathways associated with ACE2 regulation have shown that Ang II downregulates ACE2 (mRNA) transcripts in myocytes rather than fibroblasts. Tallant EA et al. reported the function of endothelin-1 in decreasing the ACE2 expression. The use of mitogen-activated protein kinase 1 (MAPK1) inhibitors with extracellular signal-regulated kinase 1/2 (ERK1/2) has also identified the role of endothelin-1 and Ang II in attenuating the ACE2 expression (Gallagher et al., 2008). In the case of mild to moderate inflammatory responses, non-steroidal anti-inflammatory drugs (NSAIDs) give relief for a period. However, long-term intake also increases the susceptibility to viral infections. Ibuprofen inhibits the prostaglandin synthesis from the arachidonic acid by non-selectively inhibiting the enzymatic activity of cyclooxygenase (COX) 1 and 2 (Bushra and Aslam, 2010). It mediates an increased sodium and water reabsorption due to the inhibition of prostaglandin synthesis, particularly Prostaglandin E2 (PGE2) and Prostaglandin I2 (PGI2) (Ricciotti and FitzGerald, 2011). Specifically, altered regulation in the ion and water leads to reduced renal perfusion followed by an upregulated ACE2 expression to facilitate a reverse action to reduce the perfusion (Turner, 2015; Mizuiri and Ohashi, 2015). Further, angiotensin II was reported to reduce the ACE2 activity while ADAM17 (referred to as TNFα-converting enzyme) upregulation in murine models. The signaling pathway was attenuated by blocking the Ang II/AT1R axis (Patel et al., 2014). Thus, receptor blockade exhibits a vital role in downregulating the kinase pathway of 1/2 and ADAM17 activity which prevents the shedding of ACE2. ADAM17 also regulates the shedding of extracellular domains and the activation of TNF-α, thereby promoting autocrine and paracrine activity. Therefore, TNF-α activation of tumor necrosis factor receptor (TNFRs) not only increases the ADAM17 activity but also increases the shedding of ACE2 in the RAS-based feedback loop (Gheblawi et al., 2020). Few reports have highlighted the role of the ACE2 signaling in regulating the activity of TNF-α and transforming growth factor-β (TGF-β) (cytokines) in the cases of cardiovascular condition and pulmonary hypertension (Grobe et al., 2007; Zeng et al., 2009; Purushothaman et al., 2013). Reduced expression of TGF-β, Smad, and membrane linked glycoprotein CD44 with telmisartan treatment demonstrated the pathways associated with negative control of ACE2 expression (Sriramula et al., 2011). Impact of ACE2 upregulation on the pro-inflammatory cytokines in the signaling cascade has not been analyzed in detail. However, the counter-regulatory role of sirtuin 1 in association with interleukin-1 has suggested an interlinked, yet investigatory pathway for ACE2 expression. In obese condition, the adipose tissue is subjected to hyperplasia and hypertrophy followed by endoplasmic stress. Irregular, vascularization results in hypoxia, necrosis, excess secretion of inflammatory adipokines, cytokines, and chemokines (Choe et al., 2016; Grant and Stephens, 2015). Based on adipocyte dysfunction, altered immunoregulation leads to the secretion of specific cytokines as well as TNF-α,interferon-gamma (IFN-γ) and interleukin-6 (IL-6) (Exley et al., 2014). In cases of obese adipocyte tissues, the excess secretion of proinflammatory cytokines are regulated by NLRP3 (NOD-, LRR-, and pyrin domain-containing protein 3) inflammasome. Several studies have reported the role of NLRP3 in immune regulation, lipid metabolism, and adipocyte function (Vandanmagsar et al., 2011). Further, ACE2 has been reported to be overexpressed in the pathophysiological condition of diabetes and obese adipocyte tissues. Excess accumulation of fatty acid not only influences the metabolic signaling but also facilitates the activation of crucial proteins like peroxisome proliferator-activated receptor gamma (PPARγ) (Kliewer et al., 1997). Thus, diabetes and obese patients are susceptible to the viral infection not only on the basis of ACEi and ARB medications but also irregular physiological conditions that facilitate irregular immunoregulation as well as deregulated metabolism. Further, the dysregulated pathophysiological condition results in the overexpression of ACE2 under cell energy stress. In a study, it was found that 5-amino-4-imidazole carboxamide riboside (AICAR) activated the AMPK pathway (Bai et al., 2016). AMP-activated protein kinase (AMPK) acts as an inhibitory switch to balance the energy-consuming pathways (Hardie et al., 2012). AMPK activates transcriptional mediators like sirtuin 1 and histone deacetylase that regulates metabolic pathways through multiple transcription factors (Tomas et al., 2002). Brookes Paul S et al. reported ACE2 overexpression after treating the ACE2 promoter with IL-1β in a time gap of 48hrs, thus the expression of ACE2 transcript (mRNA) was shown as highly time-dependent (Nadtochiy et al., 2011). Besides, in experimental hypertension conditions, rho-kinase inhibition by fasudil (inhibitor) has improved vasodilatation and ACE2 activity. Moreover, fasudil upregulated angiotensin (1–9) levels without any modifications in angiotensin (1–7) plasma levels (Clarke et al., 2014). Thus, the Rho-kinase pathway regulates the anti-hypertensive signaling cascade and improves cardioprotective effects (Ocaranza et al., 2011). Zhang and their group assessed the clinical significance of valsartan and enhancer-binding protein β (C/EBPβ) on the treatment of type I diabetes and ACE2 expression. The enhancer-binding protein β is a specific sequence CCAAT associated transcription factor which up-regulates of ACE2 expression by its overexpression. The transcription factor C/EBPβ also increased angiotensin (1–7) levels, but a less significant difference was determined (Tie et al., 2017).
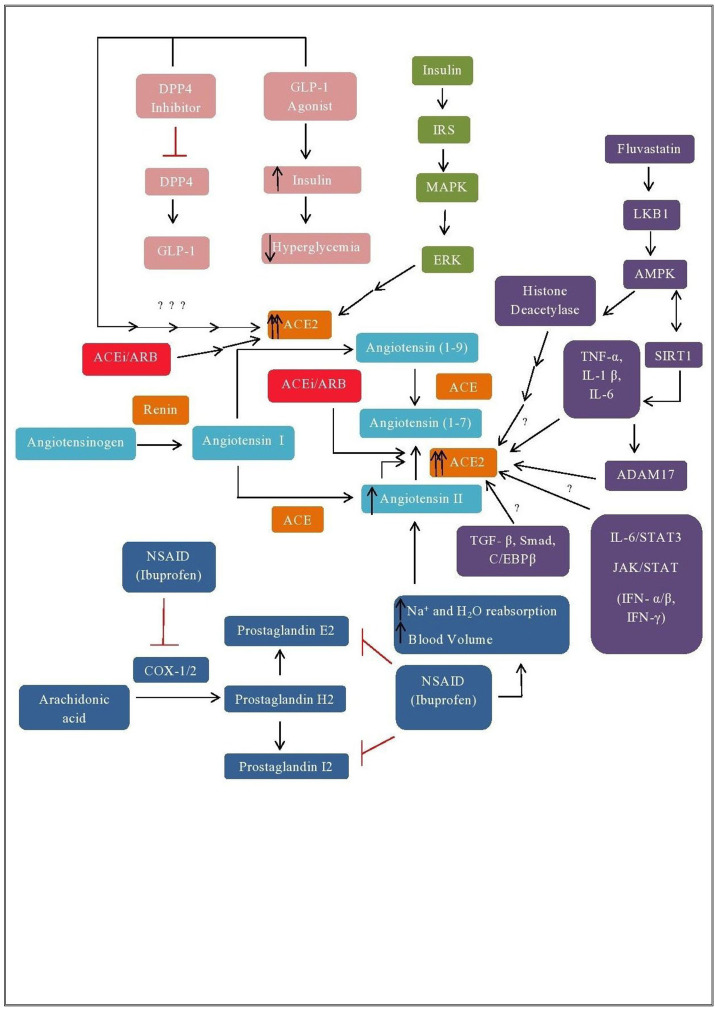
6. Data-mining on genes and pathways associated with ACE2 regulation
Text mining was performed with comorbidities i. e arterial hypertension, smoking, lung fibrosis, and bronchial asthma. This study has identified the association of histone modifiers with the ACE2 expression. The genes associated with the upregulation of ACE2 were assessed by pathway enrichment analysis. KDM5B was one of the genes linked with the ACE2 regulation. Histone modifications (acetylation/methylation) at H3K27/H3K4 were also associated with ACE2 regulation (Pinto et al., 2020). Moreover, research data highlighted the association of genes related to insulin secretion and interleukin-6 overexpression with SARS-CoV-2 comorbidities. Further, the insulin gene also regulates the Sirtuin 1 activity (Liang et al., 2009). Also, the upregulation of Sirtuin 1 was observed in the lungs of SARS-CoV-2 infected cases. It has also been demonstrated that Sirtuin 1 can epigenetically regulate ACE2 under an alarming situation of stress. Clinical studies have shown that histone deacetylase inhibitors may inhibit the expression of ACE2 (Dell’Oma et al., 2019). Based on data interpretation, genes that were positively correlated with ACE2 expression can facilitate epigenetic modification i. e histone (acetylation/deacetylation), gene activation, and chromatin dynamics. Mokuda S et al. revealed the association of IL-6 and Signal transducer and activator of transcription 3 (STAT3) signaling with ACE2 expression in rheumatoid synovium (Mokuda et al., 2020). Lee and Hennighausen has studied the link between interferon-α/β and γ along with STAT components in Janus kinase (JAK)/STAT signaling with reference to ACE2 activity in Type II Pneumocytes ( Fig. 2 ) ( Hennighausen and Lee, 2020). A recent report shows that Histone deacetylase inhibitor (HDACi) suppresses both ACE2 and ABO simultaneously (Takahashi et al., 2020). The interconnected pathways linked with ACE2 expression have been studied against pathological conditions, but a significant concern towards SARS-CoV-2 infection is still under investigation. Further, clinical approaches must be broadened for analyzing the parallel effect of inhibitors and inflammatory responses which in turn can auto-regulate significant cofactors concerning ACE2 overexpression. Several reports have speculated the increased expression of ACE2 due to ACEi/ARB therapeutic administration, but adequate evidence is still needed. Thus, the molecular paradigm of SARS-CoV-2 pathogenesis in association with ACE2 expression requires an integrative and elaborative analysis for complete understanding and effective treatment of the viral outbreak.
Fig. 2.
Schematic representation of RAAS associated AT1 and AT2/Mas receptor activity. ACEi inhibits the function of ACE I and ARBs inhibit the downstream signaling of AT1 receptor. AMPK/SIRT1 induces ACE2 regulation. Cytokines and Interferon's upregulating ACE2 expression through JAK/STAT signaling. IL-6 mediated STAT3 signaling upregulates ACE2. In comparison with the ACEi and ARBs, insulin, Non-steroidal anti-inflammatory drugs, dipeptidyl peptidase-4 inhibitor, glucagon-like peptide 1 receptor agonists in association with crucial molecular pathways regulate the ACE2 expression.
7. Conclusion
Attachment of SARS-CoV-2 with the ACE2 attenuates the receptor function, thus facilitating virus internalization. The counter-regulation of ng II regulated AT1 receptor signaling in pulmonary vasoconstriction and inflammatory damage is balanced by ACE2 activity to prevent cell damage. Research groups have analyzed alleviating ACE2 expression in SARS-CoV-2 infected patients who are dependent on ARBs and/or ACE inhibitors. In the case of diabetes mellitus, few in vivo studies have reported an association between ACEi/ARBs, and anti-diabetic medications on ACE2 upregulation, but significant clinical investigations are still required. ACEi and ARB linked ACE2 upregulation in hypertension and cardiovascular comorbidities also require significant evaluation. Imperative study on specific inflammatory molecules associated with positive regulation of ACE2 expression might provide the plausible pathways linked with SARS-CoV-2 pathophysiology. We have tried to address the significant molecular pathways associated with ACE2 expression which can be further clinically investigated to understand the elevated chance of developing SARS-CoV-2 in patients subjected to higher levels of ACE2. Currently, no guidelines have been provided by the health administrations and government agencies on shifting from ACEi and ARBs towards other alternative medications because of lack of evidence. Thus, patients with hypertension, diabetes mellitus, and cardiovascular problems should continue with ACEi and ARBs with caution. Further, investigations and distinctive molecular analyses are required to understand the molecular paradigm of ACE2 expression in association with SARS-CoV-2 pathogenesis.
Declaration of competing interest
None.
Acknowledgements
We thank Madurai Kamaraj University for providing the infrastructure through RUSA, PURSE programmes and UGC-INFLIBNET for literature subscriptions.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ejphar.2021.173899.
Funding sources and support
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author contributions
SRIDHAR Jayavel: Conceptualization, Supervision, Reviewing-editing.
RAHUL Parit: Investigation, Data curation, Original draft preparation.
All authors read and approved the final manuscript.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- Abuohashish H.M., Ahmed M.M., Sabry D., Khattab M.M., Al-Rejaie S.S. ACE-2/Ang1-7/Mas cascade mediates ACE inhibitor, captopril, protective effects in estrogen-deficient osteoporotic rats. Biomed. Pharmacother. 2017;92:58–68. doi: 10.1016/j.biopha.2017.05.062. PMID: 28531801. [DOI] [PubMed] [Google Scholar]
- Agata J., Ura N., Yoshida H., Shinshi Y., Sasaki H., Hyakkoku M., Taniguchi S., Shimamoto K. Olmesartan is an angiotensin II receptor blocker with an inhibitory effect on angiotensin-converting enzyme. Hypertens. Res. 2006;29(11):865–874. doi: 10.1291/hypres.29.865. [DOI] [PubMed] [Google Scholar]
- Alenina N., Xu P., Rentzsch B., Patkin E.L., Bader M. Genetically altered animal models for mas and angiotensin-(1–7) Exp. Physiol. 2008;93:528–537. doi: 10.1113/expphysiol.2007.040345. [DOI] [PubMed] [Google Scholar]
- Azhar E.I., Hui D.S.C., Memish Z.A., Drosten C., Zumla A. The Middle East respiratory syndrome (MERS) Infect. Dis. Clin. 2019;33(4):891–905. doi: 10.1016/j.idc.2019.08.001. PMCID: PMC7127753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bader M. ACE2, angiotensin-(1–7), and Mas: the other side of the coin. Pflügers Archiv. 2013;465(1):79–85. doi: 10.1007/s00424-012-1120-0. [DOI] [PubMed] [Google Scholar]
- Bai F., Pang X.F., Zhang L.H., Wang N.P., McKallip R.J., Garner R.E., Zhao Z.Q. Angiotensin II AT1 receptor alters ACE2 activity, eNOS expression and CD44-hyaluronan interaction in rats with hypertension and myocardial fibrosis. Life Sci. 2016;153:141–152. doi: 10.1016/j.lfs.2016.04.013. PMID: 27085217. [DOI] [PubMed] [Google Scholar]
- Bendall J.K., Cave A.C., Heymes C., Gall N., Shah A.M. Pivotal role of a gp91(phox)-containing NADPH oxidase in angiotensin II-induced cardiac hypertrophy in mice. Circulation. 2002;105(3):293–296. doi: 10.1161/hc0302.103712. [DOI] [PubMed] [Google Scholar]
- Beswick R.A., Dorrance A.M., Leite R., Webb R.C. NADH/NADPH oxidase and enhanced superoxide production in the mineralocorticoid hypertensive rat. Hypertension. 2001;38(5):1107–1111. doi: 10.1161/hy1101.093423. [DOI] [PubMed] [Google Scholar]
- Bodiga S., Zhong J.C., Wang W., Basu R., Lo J., Liu G.C., Guo D., Holland S.M., Scholey J.W., Penninger J.M., Kassiri Z., Oudit G.Y. Enhanced susceptibility to biomechanical stress in ACE2 null mice is prevented by loss of the p47(phox) NADPH oxidase subunit. Cardiovasc. Res. 2011;91(1):151–161. doi: 10.1093/cvr/cvr036. PMID: 21285291; PMCID: PMC3151662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch B.J., van der Zee R., de Haan C.A., Rottier P.J. The coronavirus spike protein is a class I virus fusion protein: structural and functional characterization of the fusion core complex. J. Virol. 2003;77(16):8801–8811. doi: 10.1128/JVI.77.16.8801-8811.2003. PMCID: PMC167208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bushra R., Aslam N. An overview of clinical pharmacology of Ibuprofen. Oman Med. J. 2010;25(3):155–1661. doi: 10.5001/omj.2010.49. PMCID: PMC3191627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chappell M.C. In: The Angiotensin-(1-7) Axis. Robson A.S.S., editor. Springer Nature Switzerland; 2019. formation and metabolism pathways; pp. 1–26. [DOI] [Google Scholar]
- Choe S.S., Huh J.Y., Hwang I.J., Kim J.I., Kim J.B. Adipose tissue remodeling: its role in energy metabolism and metabolic disorders. Front. Endocrinol. 2016;7:30. doi: 10.3389/fendo.2016.00030. PMCID: PMC4829583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke N.E., Belyaev N.D., Lambert D.W., Turner A.J. Epigenetic regulation of angiotensin-converting enzyme 2 (ACE2) by SIRT1 under conditions of cell energy stress. Clin. Sci. (Lond.) 2014;126(7):507–516. doi: 10.1042/CS20130291. [DOI] [PubMed] [Google Scholar]
- Dambha-Miller H., Albasri A., Hodgson S., Wilcox C.R., Khan S., Islam N., Little P., Griffin S.J. Currently prescribed drugs in the UK that could upregulate or downregulate ACE2 in COVID-19 disease: a systematic review. BMJ. Open. 2020;10(9) doi: 10.1136/bmjopen-2020-040644. PMCID: PMC7490921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danilczyk U., Penninger J.M. Angiotensin-converting enzyme II in the heart and the kidney. Circ. Res. 2006;98(4):463–471. doi: 10.1161/01.RES.0000205761.22353.5f. PMID: 16514079. [DOI] [PubMed] [Google Scholar]
- Dell'Omo G., Crescenti D., Vantaggiato C., Parravicini C., Borroni A.P., Rizzi N., Garofalo M., Pinto A., Recordati C., Scanziani E., Bassi F.D., Pruneri G., Conti P., Eberini I., Maggi A., Ciana P. Inhibition of SIRT1 deacetylase and p53 activation uncouples the anti-inflammatory and chemopreventive actions of NSAIDs. Br. J. Canc. 2019;120(5):537–546. doi: 10.1038/s41416-018-0372-7. PMCID: PMC6461760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donoghue M., Hsieh F., Baronas E., Godbout K., Gosselin M., Stagliano N., Donovan M., Woolf B., Robison K., Jeyaseelan R., Breitbart R.E., Acton S.A. Novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ. Res. 2000;87(5):e1–e9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- Esler M., Esler D. Can angiotensin receptor-blocking drugs perhaps be harmful in the COVID-19 pandemic? J. Hypertens. 2020;38(5):781–782. doi: 10.1097/HJH.0000000000002450. [DOI] [PubMed] [Google Scholar]
- Exley M.A., Hand L., O'Shea D., Lynch L. Interplay between the immune system and adipose tissue in obesity. J. Endocrinol. 2014;223(2):R41–R48. doi: 10.1530/JOE-13-0516. PMID: 25228503. [DOI] [PubMed] [Google Scholar]
- Fandiño J., Vaz A.A., Toba L., Romaní-Pérez M., González-Matías L., Mallo F., Diz-Chaves Y. Liraglutide enhances the activity of the ACE-2/ang(1-7)/mas receptor pathway in lungs of male pups from food-restricted mothers and prevents the reduction of SP-A. Internet J. Endocrinol. 2018:6920620. doi: 10.1155/2018/6920620. PMID: 30627159; PMCID: PMC6304858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020;8(4):e21. doi: 10.1016/S2213-2600(20)30116-8. PMCID: PMC7118626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrario C.M., Jessup J., Gallagher P.E., Averill D.B., Brosnihan K.B., Ann Tallant E., Smith R.D., Chappell M.C. Effects of renin-angiotensin system blockade on renal angiotensin-(1-7) forming enzymes and receptors. Kidney Int. 2005;68(5):2189–2196. doi: 10.1111/j.1523-1755.2005.00675.x. [DOI] [PubMed] [Google Scholar]
- Gallagher P.E., Ferrario C.M., Tallant E.A. Regulation of ACE2 in cardiac myocytes and fibroblasts. Am. J. Physiol. Heart Circ. Physiol. 2008;295(6):H2373–H2379. doi: 10.1152/ajpheart.00426.2008. PMCID:PMC2614534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gheblawi M., Wang K., Viveiros A., Nguyen Q., Zhong J.C., Turner A.J., Raizada M.K., Grant M.B., Oudit G.Y. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020;126(10):1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. PMCID: PMC7188049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant R.W., Stephens J.M. Fat in flames: influence of cytokines and pattern recognition receptors on adipocyte lipolysis. Am. J. Physiol. Endocrinol. Metab. 2015;309(3):E205–E213. doi: 10.1152/ajpendo.00053.2015. [DOI] [PubMed] [Google Scholar]
- Grobe J.L., Mecca A.P., Lingis M., Shenoy V., Bolton T.A., Machado J.M., Speth R.C., Raizada M.K., Katovich M.J. Prevention of angiotensin II-induced cardiac remodeling by angiotensin-(1-7) Am. J. Physiol. Heart Circ. Physiol. 2007;292(2):H736–H742. doi: 10.1152/ajpheart.00937.2006. [DOI] [PubMed] [Google Scholar]
- Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203(2):631–637. doi: 10.1002/path.1570. PMCID: PMC7167720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao P., Yang J., Liu Y., Zhang M., Zhang K., Gao F., Chen Y., Zhang C., Zhang Y. Combination of angiotensin-(1-7) with perindopril is better than single therapy in ameliorating diabetic cardiomyopathy. Sci. Rep. 2015;5:8794. doi: 10.1038/srep08794. PMID: 25740572; PMCID: PMC4350094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardie D.G., Ross F.A., Hawley S.A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 2012;13(4):251–262. doi: 10.1038/nrm3311. PMCID: PMC5726489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennighausen L., Lee H.K. Activation of the SARS-CoV-2 receptor Ace2 by cytokines through pan JAK-STAT enhancers during pregnancy. Cell Rep. 2020;32(13):108199. doi: 10.1016/j.celrep.2020.108199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280. doi: 10.1016/j.cell.2020.02.052. PMCID: PMC7102627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jessup J.A., Gallagher P.E., Averill D.B., Brosnihan K.B., Tallant E.A., Chappell M.C., Ferrario C.M. Effect of angiotensin II blockade on a new congenic model of hypertension derived from transgenic Ren-2 rats. Am. J. Physiol. Heart Circ. Physiol. 2006;291(5):H2166–H2172. doi: 10.1152/ajpheart.00061.2006. [DOI] [PubMed] [Google Scholar]
- Jin H.Y., Song B., Oudit G.Y., Davidge S.T., Yu H.M., Jiang Y.Y., Gao P.J., Zhu D.L., Ning G., Kassiri Z., Penninger J.M., Zhong J.C. ACE2 deficiency enhances angiotensin II-mediated aortic profilin-1 expression, inflammation and peroxynitrite production. PloS One. 2012;7(6) doi: 10.1371/journal.pone.0038502. PMCID: PMC3367919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keidar S., Gamliel-Lazarovich A., Kaplan M., Pavlotzky E., Hamoud S., Hayek T., Karry R., Abassi Z. Mineralocorticoid receptor blocker increases angiotensin-converting enzyme 2 activity in congestive heart failure patients. Circ. Res. 2005;97(9):946–953. doi: 10.1161/01.RES.0000187500.24964.7A. [DOI] [PubMed] [Google Scholar]
- Kliewer S.A., Sundseth S.S., Jones S.A., Brown P.J., Wisely G.B., Koble C.S., Devchand P., Wahli W., Willson T.M., Lenhard J.M., Lehmann J.M. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors α and γ. Proc. Natl. Acad. Sci. Unit. States Am. 1997;94(9):4318–4323. doi: 10.1073/pnas.94.9.4318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai C.C., Shih T.P., Ko W.C., Tang H.J., Hsueh P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int. J. Antimicrob. Agents. 2020;55(3):105924. doi: 10.1016/j.ijantimicag.2020.105924. PMCID: PMC7127800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M.Y., Li L., Zhang Y., Wang X.S. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Dis. Poverty. 2020;9(1):45. doi: 10.1186/s40249-020-00662-x. PMCID: PMC7186534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X.C., Zhang J., Zhuo J.L. The vasoprotective axes of the renin-angiotensin system: physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney diseases. Pharmacol. Res. 2017;125:21–38. doi: 10.1016/j.phrs.2017.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang F., Kume S., Koya D. SIRT1 and insulin resistance. Nat. Rev. Endocrinol. 2009;5(7):367–373. doi: 10.1038/nrendo.2009.101. [DOI] [PubMed] [Google Scholar]
- Liu T., Luo S., Libby P., Shi G.P. Cathepsin L-selective inhibitors: a potentially promising treatment for COVID-19 patients. Pharmacol. Ther. 2020;213:107587. doi: 10.1016/j.pharmthera.2020.107587. PMID: 32470470; PMCID: PMC7255230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo J., Patel V.B., Wang Z., Levasseur J., Kaufman S., Penninger J.M., Oudit G.Y. Angiotensin-converting enzyme 2 antagonizes angiotensin II-induced pressor response and NADPH oxidase activation in Wistar-Kyoto rats and spontaneously hypertensive rats. Exp. Physiol. 2013;98(1):109–122. doi: 10.1113/expphysiol.2012.067165. [DOI] [PubMed] [Google Scholar]
- Lu R., Zhao X., Li J., Niu P., Yang B., Wu H., Wang W., Song H., Huang B., Zhu N., Bi Y., Ma X., Zhan F., Wang L., Hu T., Zhou H., Hu Z., Zhou W., Zhao L., Chen J., Meng Y., Wang J., Lin Y., Yuan J., Xie Z., Ma J., Liu W.J., Wang D., Xu W., Holmes E.C., Gao G.F., Wu G., Chen W., Shi W., Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565–574. doi: 10.1016/S0140-6736(20)30251-8. PMID: 32007145; PMCID: PMC7159086, 10224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marques F.D., Ferreira A.J., Sinisterra R.D., Jacoby B.A., Sousa F.B., Caliari M.V., Silva G.A., Melo M.B., Nadu A.P., Souza L.E., Irigoyen M.C., Almeida A.P., Santos R.A. An oral formulation of angiotensin-(1-7) produces cardioprotective effects in infarcted and isoproterenol-treated rats. Hypertension. 2011;57(3):477–483. doi: 10.1161/HYPERTENSIONAHA.110.167346. [DOI] [PubMed] [Google Scholar]
- Matsuyama S., Nao N., Shirato K., Kawase M., Saito S., Takayama I., Nagata N., Sekizuka T., Katoh H., Kato F., Sakata M., Tahara M., Kutsuna S., Ohmagari N., Kuroda M., Suzuki T., Kageyama T., Takeda M. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. U. S. A. 2020;117(13):7001–7003. doi: 10.1073/pnas.2002589117. PMCID: PMC7132130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercure C., Yogi A., Callera G.E., Aranha A.B., Bader M., Ferreira A.J., Santos R.A., Walther T., Touyz R.M., Reudelhuber T.L. Angiotensin(1-7) blunts hypertensive cardiac remodeling by a direct effect on the heart. Circ. Res. 2008;103(11):1319–1326. doi: 10.1161/CIRCRESAHA.108.184911. [DOI] [PubMed] [Google Scholar]
- Mizuiri S., Ohashi Y. ACE and ACE2 in kidney disease. World J. Nephrol. 2015;4(1):74–82. doi: 10.5527/wjn.v4.i1.74. PMCID: PMC4317630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mokuda S., Tokunaga T., Masumoto J., Sugiyama E. Angiotensin-converting enzyme 2, a SARS-CoV-2 receptor, is upregulated by interleukin-6 via STAT3 signaling in rheumatoid synovium. J. Rheumatol. 2020;47(10):1593–1595. doi: 10.3899/jrheum.200547. PMID: 32611670. [DOI] [PubMed] [Google Scholar]
- Nadtochiy S.M., Yao H., McBurney M.W., Gu W., Guarente L., Rahman I., Brookes P.S. SIRT1-mediated acute cardioprotection. Am. J. Physiol. Heart Circ. Physiol. 2011;301(4):H1506–H1512. doi: 10.1152/ajpheart.00587.2011. PMID: 21856913; PMCID: PMC3197366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ocaranza M.P., Rivera P., Novoa U., Pinto M., González L., Chiong M., Lavandero S., Jalil J.E. Rho kinase inhibition activates the homologous angiotensin-converting enzyme-angiotensin-(1-9) axis in experimental hypertension. J. Hypertens. 2011;29(4):706–715. doi: 10.1097/HJH.0b013e3283440665. PMID: 21330937. [DOI] [PubMed] [Google Scholar]
- Onder G., Rezza G., Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. J. Am. Med. Assoc. 2020;323(18):1775–1776. doi: 10.1001/jama.2020.4683. [DOI] [PubMed] [Google Scholar]
- Oudit G.Y., Crackower M.A., Backx P.H., Penninger J.M. The role of ACE2 in cardiovascular physiology. Trends Cardiovasc. Med. 2003;13(3):93–101. doi: 10.1016/s1050-1738(02)00233-5. PMID: 12691672. [DOI] [PubMed] [Google Scholar]
- Oudit G.Y., Kassiri Z., Patel M.P. Chappell M, Butany J, Backx PH, Tsushima RG, Scholey JW, Khokha R, Penninger JM. Angiotensin II-mediated oxidative stress and inflammation mediate the age-dependent cardiomyopathy in ACE2 null mice. Cardiovasc. Res. 2007;75(1):29–39. doi: 10.1016/j.cardiores.2007.04.007. PMID: 17499227. [DOI] [PubMed] [Google Scholar]
- Oudit G.Y., Penninger J.M. Recombinant human angiotensin-converting enzyme 2 as a new renin-angiotensin system peptidase for heart failure therapy. Curr. Heart Fail. Rep. 2011;8(3):176–183. doi: 10.1007/s11897-011-0063-7. [DOI] [PubMed] [Google Scholar]
- Pal R., Bhadada S.K. Should anti-diabetic medications be reconsidered amid COVID-19 pandemic? Diabetes Res. Clin. Pract. 2020;163:108146. doi: 10.1016/j.diabres.2020.108146. PMID: 32283128; PMCID: PMC7151403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel V.B., Bodiga S., Basu R., Das S.K., Wang W., Wang Z., Lo J., Grant M.B., Zhong J., Kassiri Z., Oudit G.Y. Loss of angiotensin-converting enzyme-2 exacerbates diabetic cardiovascular complications and leads to systolic and vascular dysfunction: a critical role of the angiotensin II/AT1 receptor axis. Circ. Res. 2012;110(10):1322–1335. doi: 10.1161/CIRCRESAHA.112.268029. PMCID: PMC3746191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel V.B., Bodiga S., Fan D., Das S.K., Wang Z., Wang W., Basu R., Zhong J., Kassiri Z., Oudit G.Y. Cardioprotective effects mediated by angiotensin II type 1 receptor blockade and enhancing angiotensin 1-7 in experimental heart failure in angiotensin-converting enzyme 2-null mice. Hypertension. 2012;59(6):1195–1203. doi: 10.1161/HYPERTENSIONAHA.112.191650. [DOI] [PubMed] [Google Scholar]
- Patel V.B., Clarke N., Wang Z., Fan D., Parajuli N., Basu R., Putko B., Kassiri Z., Turner A.J., Oudit G.Y. Angiotensin II induced proteolytic cleavage of myocardial ACE2 is mediated by TACE/ADAM-17: a positive feedback mechanism in the RAS. J. Mol. Cell. Cardiol. 2014;66:167–176. doi: 10.1016/j.yjmcc.2013.11.017. PMID: 24332999. [DOI] [PubMed] [Google Scholar]
- Perlman S., Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat. Rev. Microbiol. 2009;7(6):439–450. doi: 10.1038/nrmicro2147. PMCID: PMC2830095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrella A., Carannante N., Berretta M., Rinaldi M., Maturo N., Rinaldi L. Novel Coronavirus 2019 (Sars-CoV2): a global emergency that needs new approaches? Eur. Rev. Med. Pharmacol. Sci. 2020;24(4):2162–2164. doi: 10.26355/eurrev_202002_20396. PMID: 32141586. [DOI] [PubMed] [Google Scholar]
- Pinto B.G.G., Oliveira A.E.R., Singh Y., Jimenez L., Gonçalves A.N.A., Ogava R.L.T., Creighton R., Peron J.P.S., Nakaya H.I. ACE2 expression is increased in the lungs of patients with comorbidities associated with severe COVID-19. J. Infect. Dis. 2020;222(4):556–563. doi: 10.1093/infdis/jiaa332. PMCID: PMC7276054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purushothaman K.R., Krishnan P., Purushothaman M., Wiley J., Alviar C.L., Ruiz F.J., Zubatov Y., Kini A.S., Sharma S.K., Fuster V., Moreno P.R. Expression of angiotensin-converting enzyme 2 and its end product angiotensin 1-7 is increased in diabetic atheroma: implications for inflammation and neovascularization. Cardiovasc. Pathol. 2013;22(1):42–48. doi: 10.1016/j.carpath.2012.05.004. [DOI] [PubMed] [Google Scholar]
- Qiao W., Wang C., Chen B., Zhang F., Liu Y., Lu Q., Guo H., Yan C., Sun H., Hu G., Yin X. Ibuprofen attenuates cardiac fibrosis in streptozotocin-induced diabetic rats. Cardiology. 2015;131(2):97–106. doi: 10.1159/000375362. [DOI] [PubMed] [Google Scholar]
- Ricciotti E., FitzGerald G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biol. 2011;31(5):986–1000. doi: 10.1161/ATVBAHA.110.207449. PMID: 21508345, PMCID: PMC3081099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romaní-Pérez M., Outeiriño-Iglesias V., Moya C.M., Santisteban P., González-Matías L.C., Vigo E., Mallo F. Activation of the GLP-1 receptor by liraglutide increases ACE2 expression, reversing right ventricle hypertrophy, and improving the production of SP-A and SP-B in the lungs of type 1 diabetes rats. Endocrinology. 2015;156(10):3559–3569. doi: 10.1210/en.2014-1685. PMID: 26196539. [DOI] [PubMed] [Google Scholar]
- Roszer T., Ricote M. PPAR. Res. ID; 2010. PPARs in the Renal Regulation of Systemic Blood Pressure; p. 698730. PMID: 20613959; PMCID: PMC2896854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saheb Sharif-Askari N., Saheb Sharif-Askari F., Alabed M., Tayoun A.A., Loney T., Uddin M., Senok A., Al Heialy S., Hamoudi R., Kashour T., Alsheikh-Ali A., Hamid Q., Halwani R. Effect of common medications on the expression of SARS-CoV-2 entry receptors in kidney tissue. Clin. Transl. Sci. 2020;13(6):1048–1054. doi: 10.1111/cts.12862. PMCID: PMC7461457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sampaio W.O., Souza dos Santos R.A., Faria-Silva R., da Mata Machado L.T., Schiffrin E.L., Touyz R.M. Angiotensin-(1-7) through receptor Mas mediates endothelial nitric oxide synthase activation via Akt-dependent pathways. Hypertension. 2007;49(1):185–192. doi: 10.1161/01.HYP.0000251865.35728.2f. [DOI] [PubMed] [Google Scholar]
- Santos R.A., Simoes e Silva A.C., Maric C., Silva D.M., Machado R.P., de Buhr I., Heringer-Walther S., Pinheiro S.V., Lopes M.T., Bader M., Mendes E.P., Lemos V.S., Campagnole-Santos M.J., Schultheiss H.P., Speth R., Walther T. Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc. Natl. Acad. Sci. U. S. A. 2003;100(14):8258–8263. doi: 10.1073/pnas.1432869100. PMCID: PMC166216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarafidis P.A., Lasaridis A.N., Nilsson P.M., Pagkalos E.M., Hitoglou-Makedou A.D., Pliakos C.I., Kazakos K.A., Yovos J.G., Zebekakis P.E., Tziolas I.M., Tourkantonis A.N. Ambulatory blood pressure reduction after rosiglitazone treatment in patients with type 2 diabetes and hypertension correlates with insulin sensitivity increase. J. Hypertens. 2004;22(9):1769–1777. doi: 10.1097/00004872-200409000-00022. [DOI] [PubMed] [Google Scholar]
- Shin Y.H., Min J.J., Lee J.H., Kim E.H., Kim G.E., Kim M.H., Lee J.J., Ahn H.J. The effect of fluvastatin on cardiac fibrosis and angiotensin-converting enzyme-2 expression in glucose-controlled diabetic rat hearts. Heart Ves. 2017;32(5):618–627. doi: 10.1007/s00380-016-0936-5. [DOI] [PubMed] [Google Scholar]
- Soler M.J., Ye M., Wysocki J., William J., Lloveras J., Batlle D. Localization of ACE2 in the renal vasculature: amplification by angiotensin II type 1 receptor blockade using telmisartan. Am. J. Physiol. Ren. Physiol. 2009;296(2):F398–F405. doi: 10.1152/ajprenal.90488.2008. [DOI] [PubMed] [Google Scholar]
- Sriramula S., Cardinale J.P., Lazartigues E., Francis J. ACE2 overexpression in the paraventricular nucleus attenuates angiotensin II-induced hypertension. Cardiovasc. Res. 2011;92(3):401–408. doi: 10.1093/cvr/cvr242. PMID: 21952934; PMCID: PMC3286198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi Y., Hayakawa A., Sano R., Fukuda H., Harada M., Kubo R., Okawa T., Kominato Y. Histone deacetylase inhibitors suppress ACE2 and ABO simultaneously suggesting a preventive potential against COVID-19. 2020. Scientific Reports, [preprint] [DOI] [PMC free article] [PubMed]
- Tallant E.A., Ferrario C.M., Gallagher P.E. Angiotensin-(1-7) inhibits growth of cardiac myocytes through activation of the mas receptor. Am. J. Physiol. Heart Circ. Physiol. 2005;289(4):H1560–H1566. doi: 10.1152/ajpheart.00941.2004. [DOI] [PubMed] [Google Scholar]
- Tang T., Bidon M., Jaimes J.A., Whittaker G.R., Daniel S. Coronavirus membrane fusion mechanism offers a potential target for antiviral development. Antivir. Res. 2020;178:104792. doi: 10.1016/j.antiviral.2020.104792. PMID: 32272173; PMCID: PMC7194977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tie Y., Zhai C., Zhang Y., Qin X., Yu F., Li H., Shan M., Zhang C. CCAAT/enhancer-binding protein β overexpression alleviates myocardial remodelling by regulating angiotensin-converting enzyme-2 expression in diabetes. J. Cell Mol. Med. 2017;22(3):1475–1488. doi: 10.1111/jcmm.13406. PMCID: PMC5824391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikellis C., Thomas M.C. Angiotensin-converting enzyme 2 (ACE2) is a key modulator of the renin angiotensin system in health and disease. Int. J. Pept. 2012:256294. doi: 10.1155/2012/256294. PMID: 22536270; PMCID: PMC3321295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikoo K., Patel G., Kumar S., Karpe P.A., Sanghavi M., Malek V., Srinivasan K. Tissue specific up regulation of ACE2 in rabbit model of atherosclerosis by atorvastatin: role of epigenetic histone modifications. Biochem. Pharmacol. 2015;93(3):343–351. doi: 10.1016/j.bcp.2014.11.013. [DOI] [PubMed] [Google Scholar]
- Tipnis S.R., Hooper N.M., Hyde R., Karran E., Christie G., Turner A.J. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J. Biol. Chem. 2000;275(43):33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- Tomás E., Lin Y.S., Dagher Z., Saha A., Luo Z., Ido Y., Ruderman N.B. Hyperglycemia and insulin resistance: possible mechanisms. Ann. N. Y. Acad. Sci. 2002;967:43–51. doi: 10.1111/j.1749-6632.2002.tb04262.x. 12079834. [DOI] [PubMed] [Google Scholar]
- Turner A.J. ACE2 cell biology, regulation, and physiological functions. The protective arm of the renin angiotensin system (RAS). 2015. 2015:185–189. doi: 10.1016/B978-0-12-801364-9.00025-0. PMCID: PMC7149539. [DOI] [Google Scholar]
- Turner A.J., Hiscox J.A., Hooper N.M. ACE2: from vasopeptidase to SARS virus receptor. Trends Pharmacol. Sci. 2004;25(6):291–294. doi: 10.1016/j.tips.2004.04.001. PMCID: PMC7119032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandanmagsar B., Youm Y.H., Ravussin A., Galgani J.E., Stadler K., Mynatt R.L., Ravussin E., Stephens J.M., Dixit V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011;17(2):179–188. doi: 10.1038/nm.2279. PMCID: PMC3076025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickers C., Hales P., Kaushik V., Dick L., Gavin J., Tang J., Godbout K., Parsons T., Baronas E., Hsieh F., Acton S., Patane M., Nichols A., Tummino P. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J. Biol. Chem. 2002;277(17):14838–14843. doi: 10.1074/jbc.M200581200. [DOI] [PubMed] [Google Scholar]
- Wan Y., Shang J., Graham R., Baric R.S., Li F. Receptor recognition by the novel coronavirus from Wuhan: an analysis based on decade-long structural studies of SARS coronavirus. J. Virol. 2020;94:e00127. doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., Wang B., Xiang H., Cheng Z., Xiong Y., Zhao Y., Li Y., Wang X., Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in wuhan, China. J. Am. Med. Assoc. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. PMCID: PMC7042881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W., McKinnie S.M., Farhan M., Paul M., McDonald T., McLean B., Llorens-Cortes C., Hazra S., Murray A.G., Vederas J.C., Oudit G.Y. Angiotensin-converting enzyme 2 metabolizes and partially inactivates pyr-apelin-13 and apelin-17: physiological effects in the cardiovascular system. Hypertension. 2016;68(2):365–377. doi: 10.1161/HYPERTENSIONAHA.115.06892. [DOI] [PubMed] [Google Scholar]
- Watkins J. Preventing a covid-19 pandemic. BMJ. 2020;368:m810. doi: 10.1136/bmj.m810. [DOI] [PubMed] [Google Scholar]
- Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367(6483):1260–1263. doi: 10.1126/science.abb2507. PMCID: PMC7164637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu F., Zhao S., Yu B., Chen Y.M., Wang W., Song Z.G., Hu Y., Tao Z.W., Tian J.H., Pei Y.Y., Yuan M.L., Zhang Y.L., Dai F.H., Liu Y., Wang Q.M., Zheng J.J., Xu L., Holmes E.C., Zhang Y.Z. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579(7798):265–269. doi: 10.1038/s41586-020-2008-3. PMCID: PMC7094943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wysocki J., Ye M., Soler M.J., Gurley S.B., Xiao H.D., Bernstein K.E., Coffman T.M., Chen S., Batlle D. ACE and ACE2 activity in diabetic mice. Diabetes. 2006;55(7):2132–2139. doi: 10.2337/db06-0033. [DOI] [PubMed] [Google Scholar]
- Xia S., Zhu Y., Liu M., Lan Q., Xu W., Wu Y., Ying T., Liu S., Shi Z., Jiang S., Lu L. Fusion mechanism of 2019-nCoV and fusion inhibitors targeting HR1 domain in spike protein. Cell. Mol. Immunol. 2020;17(7):765–767. doi: 10.1038/s41423-020-0374-2. PMCID: PMC7075278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue-Ying T., Jing-Bo H. ACEIs/ARBs for the prevention of type 2 diabetes in patients with cardiovasculardiseases: a systematic review and meta-analysis. Int. J. Clin. Exp. Med. 2016;9(5):7624–7637. [Google Scholar]
- Yang X., Yu Y., Xu J., Shu H., Xia J., Liu H., Wu Y., Zhang L., Yu Z., Fang M., Yu T., Wang Y., Pan S., Zou X., Yuan S., Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir. Med. 2020;8(5):475–481. doi: 10.1016/S2213-2600(20)30079-5. PMCID: PMC7102538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng W., Chen W., Leng X., He J.G., Ma H. Chronic angiotensin-(1-7) administration improves vascular remodeling after angioplasty through the regulation of the TGF-beta/Smad signaling pathway in rabbits. Biochem. Biophys. Res. Commun. 2009;389(1):138–144. doi: 10.1016/j.bbrc.2009.08.112. [DOI] [PubMed] [Google Scholar]
- Zhang J.J., Dong X., Cao Y.Y., Yuan Y.D., Yang Y.B., Yan Y.Q., Akdis C.A., Gao Y.D. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. 2020;75(7):1730–1741. doi: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- Zhang L.H., Pang X.F., Bai F., Wang N.P., Shah A.I., McKallip R.J., Li X.W., Wang X., Zhao Z.Q. Preservation of glucagon-like peptide-1 level attenuates angiotensin II-induced tissue fibrosis by altering AT1/AT 2 receptor expression and angiotensin-converting enzyme 2 activity in rat heart. Cardiovasc. Drugs Ther. 2015;29(3):243–255. doi: 10.1007/s10557-015-6592-7. [DOI] [PubMed] [Google Scholar]
- Zhang W., Du R.H., Li B., Zheng X.S., Yang X.L., Hu B., Wang Y.Y., Xiao G.F., Yan B., Shi Z.L., Zhou P. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg. Microb. Infect. 2020;9(1):386–389. doi: 10.1080/22221751.2020.1729071. PMID: 32065057; PMCID: PMC7048229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W., Xu Y.Z., Liu B., Wu R., Yang Y.Y., Xiao X.Q., Zhang X. Pioglitazone upregulates angiotensin converting enzyme 2 expression in insulin-sensitive tissues in rats with high-fat diet-induced nonalcoholic steatohepatitis. 2014. ScientificWorldJournal. ID: 603409. PMCID: PMC3914411. [DOI] [PMC free article] [PubMed]
- Zhang Y., Li B., Wang B., Zhang J., Wu J., Morgan T. Alteration of cardiac ACE2/Mas expression and cardiac remodelling in rats with aortic constriction. Chin. J. Physiol. 2014;57(6):335–342. doi: 10.4077/CJP.2014.BAD268. [DOI] [PubMed] [Google Scholar]
- Zhang Y.Z., Holmes E.C. A genomic perspective on the origin and emergence of SARS-CoV-2. Cell. 2020;181(2):223–227. doi: 10.1016/j.cell.2020.03.035. PMCID: PMC7194821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y.Y., Ma Y.T., Zhang J.Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020;17(5):259–260. doi: 10.1038/s41569-020-0360-5. PMCID: PMC7095524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong J.C., Huang D.Y., Yang Y.M., Li Y.F., Liu G.F., Song X.H., Du K. Upregulation of angiotensin-converting enzyme 2 by all-trans retinoic acid in spontaneously hypertensive rats. Hypertension. 2004;44(6):907–912. doi: 10.1161/01.HYP.0000146400.57221.74. PMID: 15477383. [DOI] [PubMed] [Google Scholar]
- Zhou P., Yang X.L., Wang X.G., et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.