Abstract
Faecal microbiota transplantation (FMT) is the transfer of screened and minimally processed faecal material from a ‘healthy’ donor to ‘diseased’ recipient. It has an established role, and is recommended as a therapeutic strategy, in the management of recurrent Clostridioides difficile infection (CDI). Recognition that gut dysbiosis is associated with, and may contribute to, numerous disease states has led to interest in exploiting FMT to ‘correct’ this microbial imbalance. Conditions for which it is proposed to be beneficial include inflammatory bowel disease, irritable bowel syndrome, liver disease and hepatic encephalopathy, neuropsychiatric conditions such as depression and anxiety, systemic inflammatory states like sepsis, and even coronavirus disease 2019. To understand what role, if any, FMT may play in the management of these conditions, it is important to consider the potential risks and benefits of the therapy. Regardless, there are several barriers to its more widespread adoption, which include incompletely understood mechanism of action (especially outside of CDI), inability to standardise treatment, disagreement on its active ingredients and how it should be regulated, and lack of long-term outcome and safety data. Whilst the transfer of faecal material from one individual to another to treat ailments or improve health has a history dating back thousands of years, there are fewer than 10 randomised controlled trials supporting its use. Moving forward, it will be imperative to gather as much data from FMT donors and recipients over as long a timeframe as possible, and for trials to be conducted with rigorous methodology, including appropriate control groups, in order to best understand the utility of FMT for indications beyond CDI. This review discusses the history of FMT, its appreciable mechanisms of action with reference to CDI, indications for FMT with an emerging evidence base above and beyond CDI, and future perspectives on the field.
Keywords: faecal microbiota transplantation (FMT), clostridioides difficile infection (CDI), dysbiosis, inflammatory bowel disease (IBD), irritable bowel syndrome (IBS), liver disease
A brief history of faecal microbiota transplantation
The first documented use of the ingestion of faecal material to treat illness comes from the Sanskrit text Charak Samhita. Panchgavya, a mixture of five cow products including dung, used in ayurvedic medicine for over 2000 years, claims to treat a variety of communicable and non-communicable diseases.1 It is unsurprising that consumption of products from a sacred animal was thought to improve health, nevertheless, a substantiated evidence base is lacking.
Chinese literature from the 4th century describes faecal material being used to treat food poisoning and diarrhoeal illnesses, and again in the 16th century, ‘golden syrup’, a mixture of faeces and water, was used with medicinal intent to cure abdominal illnesses.2 In Korea, ancient texts describe Ttsongul, a fermented rice wine made using faeces, believed to be effective in treating a wide range of problems from cuts through to epilepsy.3 Throughout other parts of Asia and the Middle East, Bedouins have treated dysentery with fresh camel dung for centuries.4 However, evidence as to the efficacy of any of these is at best anecdotal.
Faecal microbiota transplantation (FMT) first entered modern medicine in 1958 when Eiseman and colleagues successfully treated four patients with pseudomembranous colitis using faecal enema.5 Numerous case series and open-label trials followed with FMT used to treat inflammatory bowel disease (IBD) as well as and CDI (now known to be the predominant cause of pseudomembranous colitis), but it was not until 2013, that the first randomised control trial (RCT) evidence of FMT efficacy in the treatment of recurrent (r) CDI was published.6 The trial was stopped after an interim analysis demonstrated clear benefit of FMT compared with vancomycin. Subsequent RCTs and meta-analyses have confirmed that FMT (whether fresh, frozen or lyophilised, and given orally, via endoscopy or enema), is superior to antibiotics for treating relapsed or refractory CDI.7–13 Although most guidelines do not recommend FMT for treatment of any conditions other than rCDI,14 interest in using FMT for other indications is growing. Here, we review proposed mechanisms of action of FMT with a focus on CDI, the current evidence base for other indications, and consider future perspectives on the direction the field of FMT may take.
The constituents of FMT and proposed mechanisms of action
Microbiota is the descriptive term for all the organisms found in a particular niche, such as the human gut, including bacteria, viruses, archaea, and protozoa. The microbiome is the sum of the genetic material of these organisms. Modern sequencing techniques have enabled us to ‘see’ these organisms, as well as their relative abundance.15 The microbiota is a dynamic entity and varies not only from person to person (alpha diversity), but also over time in individuals (beta diversity). Metabolomic profiling, the ‘fingerprint’ left behind by the activities of the microbiota and its interaction with the host, adds further detail to our knowledge of this highly complex environment.16,17 Studies undertaken in health and in disease states have identified differences in all of these parameters.18 Deviations from the ‘norm’ have been termed dysbiosis (although this term has been criticised by some19) and can be seen locally, that is, within the gut, as well as systemically, for example, changes in lymphocyte subsets. The ability of FMT to ‘correct’ dysbiosis, or this ‘imbalance’, is of great interest.
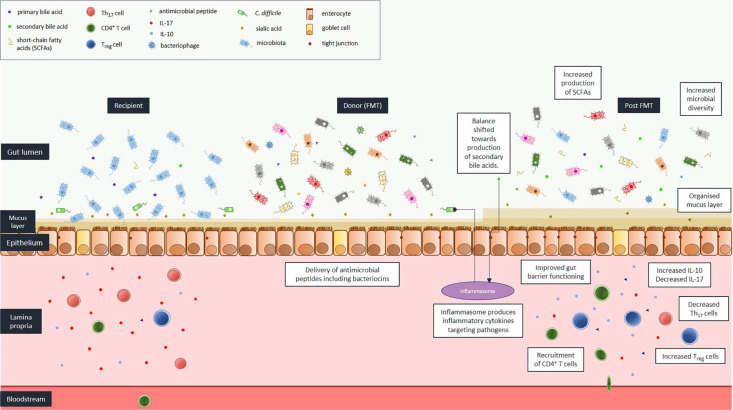
In CDI, there is, almost invariably, a recent alteration in gut microbiota following antimicrobial administration (usually several days to weeks before developing infection). For other disease states, for example, IBD, dysbiosis is often a more established process (months to years). This is an important difference, as FMT efficacy rates, and optimal dosing regimens may vary depending on the duration of dysbiosis. The proposed mechanisms of action of FMT, with specific reference to CDI, are depicted in Figure 1 and discussed below. The relative contribution of each is currently unknown. None, some, or all these mechanisms may be applicable to the activity of FMT for non-CDI conditions.
Figure 1.
Mechanisms of action of faecal microbiota transplantation (FMT) with reference to Clostridioides difficile infection (CDI).
C. difficile, Clostridioides difficile; IL, interleukin; SCFA, short-chain fatty acid; TH1, T-helper cell 1; Treg cell, regulatory T cell.
Colonisation resistance
Colonisation resistance is the protection afforded by healthy microbiota against invading pathogens. Antimicrobials are taken, for the most part, to kill, or at least prevent growth, of pathogenic microbes. Taken systemically, collateral disruption to the body’s microbiota occurs. Destabilising microbial communities disrupts microbiota-mucosal immunity interactions and promotes inflammation, allowing sites such as the gut to become colonised with pathogens including C. difficile. Ingestion of C. difficile spores leads to asymptomatic carriage, or in the worst-case scenario, to disease (CDI). Replenishing and diversifying the microbial community using FMT promotes colonisation resistance.20 A trend towards increased bacterial community diversity in recipients successfully treated with FMT suggests causality.21
Gut barrier
A healthy gut microbiota provides constant stimulation to the epithelial barrier leading to the production of an organised mucus layer, the maintenance of tight junctions between epithelial cells, and the production of antimicrobial peptides by the host. All these actions serve to compartmentalise the microbiota within the intestinal lumen. Disrupted microbiota-host crosstalk in dysbiosis means the integrity of the gut barrier is reduced, and in the most extreme cases, for example pseudomembranous colitis, there is total breakdown with translocation of residual microbiota. FMT can prevent, stabilise, and even reverse these effects.22
Bile acids
Bile acids are capable of directly influencing the microbiota and have been demonstrated to have antimicrobial activity,23,24 including affecting the growth of C. difficile. Primary bile acids (those initially released from the duodenum) stimulate germination of spores, whereas secondary bile acids (formed via 7α-dehydroxylation of primary bile acids by Lacnospiraceae and Ruminococcaceae families) inhibit this process.25 In faeces, secondary bile acids have been noted to be present in lower levels in individuals with CDI (especially those with recurrent disease) compared with those without, and FMT has been demonstrated to shift the balance from primary towards secondary bile acid synthesis in recipients.26 Bile acids interact with the host inflammasome, a multi-protein intracellular complex that detects pathogens, and induces the production of pro-inflammatory cytokines interleukin 1β (IL-1β) and IL-18. Inflammasome signalling is vital in recovery from CDI.27 Bile acids may also mitigate the effect of C. difficile exotoxins, thereby reducing epithelial damage.
Nutrients/carbohydrates (including short-chain fatty acids)/proteins (e.g. metal chelation)
The inner and outer mucous layers of the gut are made of complex carbohydrates, which are a ready source of energy for bacteria that possess the necessary digestive enzymes to break them down into compounds such as glucose and sialic acid. Loss of the bacteria with antimicrobial treatment, which efficiently scavenge these products, leaves them available for pathogens such as C. difficile, allowing germination and proliferation.28
Short-chain fatty acids (SCFAs) are a group of carbohydrates produced by anaerobic fermentation in the gut from dietary fibre and resistant starch, with acetate, propionate and butyrate being the most abundant.29 Their presence has been linked with improvement in gut barrier functioning and reduced inflammation by supporting peripheral regulatory T (Treg)-cell development.30 Alterations in levels of SCFAs, or bacteria which produce SCFAs, have been detected between individuals with CDI and healthy controls. Medium-chain fatty acids are predominantly derived from triglycerides and phospholipids in plant oils and milk products. Some of these have been shown to have antimicrobial activity, for example, lauric acid, inducing oxidative damage and cell lysis in C. difficile. Contribution to the efficacy of FMT still remains unclear.31
Certain proteins, for example, calprotectin, are capable of binding metal ions, for example, zinc, reducing their availability. Zinc (as well as calcium) are co-factors for bacterial enzymes, and transfer of faecal material which reduces free metal ion availability in the recipient may prevent the development of CDI by interfering with enzyme activity.32
Antimicrobial peptides
As well as competing with each other for nutrients, bacteria are capable of producing bacteriocins, antimicrobial proteins which target other, especially phylogenetically similar, bacterial species.33 Bacteriocins produced by Gram-positive bacteria are split into one of four classes; I [lantibiotics: small (<5 kDa) post-transcriptionally modified peptides with unusual amino acids], II [non-lantibiotics: small (<10 kDa) limited post-transcriptional modification and no unusual amino acids], III [large (>30 kDa), heat-labile lytic or non-lytic peptides], and IV (contain lipid or carbohydrate parts). Bacteriocins produced by Gram-negative bacteria include colicins (>10 kDa, produced by Escherichia coli), colicin-like (>10 kDa, produced by bacteria other than E. coli), microcins (molecular weight <10 kDa), and phage tail-like (very similar to phage tail structure). Further detail on their biology and spectrum of antimicrobial activity has recently been summarised.34
Several compounds that target C. difficile have been identified, including fidaxomicin, a drug given clinically to treat C. difficile. This is produced by the actinomycete, Dactylosporangium aurantiacum subspecies hamdenesis.35 Intestinal and immune cells have also been shown to produce compounds with antimicrobial activity such as defensins, cathelicidins and lysozyme.36
Bacteriophages
Bacteriophages are enveloped ribonucleic acid (RNA) or deoxyribonucleic acid (DNA) viruses that bind specific receptors (and thereby determine host specificity) on bacteria and archaea.37 They are capable of inducing cell lysis, and modifying virulence factors or gene expression, including in C. difficile.38 They have been utilised to treat antimicrobial-resistant organisms (AROs) where other lines of therapy have failed.39
Immunomodulation
The potential influence of FMT on the immune system was considered briefly above with respect to bile acids and SCFAs. Bacteria, too, have immunomodulatory actions, for example, Bacteroides fragilis, through its presentation of polysaccharide A (PSA), contributes to the maintenance of CD4+ T-lymphocyte development, balancing T-helper cell 1 (Th1) and Th2 responses, and lymphoid organogenesis. It can also inhibit the production of pro-inflammatory IL-17 (by inhibiting the Th17 response) and promote the production of anti-inflammatory IL-10 (by promoting the Treg cell response).40 Further description of the relationship between the immune system and microbiota is beyond the scope of this paper, but has recently been summarised elsewhere.41 Needless to say, the importance of a commensal microbiota in the development and maintenance of a functioning immune system is without question, as demonstrated by the defects in both gastrointestinal (GI) and systemic immunity in germ-free animal models.42,43
Regulation and safety of FMT
The regulatory landscape of FMT in the European Union and other jurisdictions has been reviewed elsewhere.44 There are widely varying approaches to its regulation and governance, with merits and disadvantages to each. As with any medical treatment, the benefits the patient stands to gain must be weighed against the risks of receiving FMT.
Of note, there has been a number of recent safety alerts relating to improperly screened donations which have resulted in transmission of infection.45 In light of the coronavirus disease 2019 (COVID-19) pandemic, donor material must now be screened for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), as the virus is detectable and culturable from the stool of infected individuals, so theoretically transmissible via FMT.46 FMT regulation must be carefully considered going forward, to ensure both the delivery of a safe product and the availability of the treatment to clinicians in a clinical and research capacity.
Indications with emerging evidence base
Severe (primary) CDI
Traditional management of severe CDI refractory to antibiotics has been colectomy. This is associated with high fatality rates, especially in the frail and elderly.47 The success of FMT for recurrent and relapsed CDI led to the trialling of FMT in severe and fulminant infection with excellent results. Overall clinical cure at 1 month was achieved in 91% patients in one trial, and 88% at 8 weeks in another, with a suggestion that multiple infusions may be superior to just one (cure rate 100% versus 75%). There were no serious adverse events related to FMT or its delivery.48,49 Expert opinion recommends consideration of FMT early in these cases,50 but at present there remains insufficient RCT evidence to support its use. However, it would be impractical and potentially unethical to recruit these critically unwell patients into trials to get a definitive answer, given the overwhelming evidence of benefit in rCDI.
Inflammatory bowel disease
There is emerging evidence that FMT can induce remission in ulcerative colitis (UC), one of the two main subtypes of IBD. Although encouraging, with success rates ranging from 25% to 45%,51,52 results fall well short of that seen in CDI. Individuals with IBD have reduced numbers of SCFA-producing obligate anaerobes and an increase in pro-inflammatory facultative anaerobes including E. coli.53 Interestingly, FMT from a donor with high colony counts of Ruminococcaceae, an obligate anaerobe and one of the main producers of butyrate in the gut, was more successful in inducing remission (39%) in UC compared with the other five donors (10%) in the study.54 These are observational data only and do not prove causality. It also does not explain why the majority (61%) failed to respond. The first RCT of FMT in Crohn’s disease (CD), the other main subtype of IBD, did not identify donor-specific response rates; however, sample size was small (21 participants). It did show a trend towards FMT promoting remission and a reduction in gut inflammation following steroid therapy, particularly if the recipient’s microbiome reflected that of their donor, compared with control, but results did not reach statistical significance.55 Larger studies are keenly anticipated. Defects in immune regulation including a reduction in Treg lymphocytes and enhanced Th17 and innate cell response associated with the production of pro-inflammatory cytokines have also been implicated in the pathogenesis of IBD. As described above, these can be moderated by certain bacteria or their components, which can be transmitted in FMT.
Primary sclerosing cholangitis
Primary sclerosing cholangitis (PSC) is a chronic, progressive cholestatic hepatobiliary disorder characterised by fibrotic destruction of bile ducts. It leads to biliary cirrhosis, portal hypertension and ultimately, liver failure. Concurrent IBD is common (80% patients), although the dysbiosis seen is distinct even from that seen in patients with IBD alone.56 It is proposed that bacterial translocation across an inflamed gut drives an inflammatory process in the biliary tree, leading to disease. This theory is supported in animal models.57 Attempts to modulate the gut microbiota of patients with PSC has been trialled with antimicrobials with mixed results, and it is not currently recommended as a therapeutic strategy.58,59 An open-label pilot study published last year demonstrated biochemical improvement (alkaline phosphatase levels fell by 50%) in recipients (3/10) of FMT in which there was donor engraftment out to 6 months post-transplant.60 The slowly progressive nature of disease means that long-term follow up will be necessary in future trials to establish any effect of FMT on patient outcomes.
Irritable bowel syndrome
Irritable bowel syndrome (IBS) is a heterogeneous chronic GI condition affecting around 1 in 10 people worldwide. Dysbiosis (for example high Firmicute: Bacteroidetes ratio) has been identified in many individuals, making correction with FMT an attractive proposition. Pooled results from RCTs do not show a conclusive benefit to date. However, if the delivery method of FMT is considered, there is a suggestion fresh or frozen donor stool may be beneficial, whereas capsulised FMT may cause harm.61–63 Holvoet et al. suggested microbiota modulation with FMT should be targeted at the subgroups with severe bloating and flatulence, in whom there is the most significant disturbance in gut microbial composition. They believe donor selection is important, short-lived effects can be augmented by repeat FMT delivery, but that certain individuals may be refractory to FMT. The study raises important questions: why should there be differences in response rates by sex (females were more likely to respond)? And, why individuals with a greater baseline diversity in microbes pre-FMT were more likely to have a positive response to FMT.64 This would seem counterintuitive if FMT worked by increasing microbial diversity; potentially other transplanted components were the reason for efficacy. Another recently published trial agrees that donor selection is important but suggests that efficacy can be achieved across IBS subtypes, that response is dose dependent, and is related to improving dysbiosis in the recipient.65 Patients in this trial had a lower IBS-SSS (symptom severity score) than the Dutch study and follow-up was limited to 3 months, so how durable the effect on symptoms is unclear. A further study suggests that despite longlasting microbiota changes, clinical improvement is small and transient with no significant effect on quality-of-life outcomes.66
Lastly, individuals with both IBS and chronic fatigue syndrome (CFS), also known as myalgic encephalomyelitis (ME), were assigned to receive either oral approaches (e.g. pre/probiotics, dietary advice, nutritional remedies) or anaerobically produced FMT delivered into the sigmoid colon via rectal catheter from 10 different donors in a non-randomised fashion. FMT recipients improved to a greater extent than those who received oral approaches alone according to the investigators’ assessment, suggesting a potential role for FMT in CFS.67 However, the retrospective nature of the study, and the lack of randomisation and an objective standard for assessing improvement, limit the generalisability of results.
Constipation
Slow-transit constipation (STC) is a common functional GI disorder and represents a subset of up to 30% individuals who suffer with constipation.68 Promising pilot study results with clinical improvement in 50–60% of participants up to 12 weeks69,70 were followed by the only RCT71 reported to date of FMT to treat STC, which again, showed favourable outcomes. Compared with control, the intervention group showed reduced transit time, and improved stool consistency. Overall, clinical improvement was seen in 53% of the intervention group (versus 20% in controls) and clinical cure in 37% (versus 13% in controls). However, the FMT regimen was intense (100 ml given daily for 6 days via naso-intestinal route) with a high rate of reported adverse events, and the study was not blinded, limiting attribution of the effect to FMT alone, as well as the broader application of results. FMT may also have a role in other types of constipation; an open-label study in patients with chronic intestinal pseudo-obstruction suggested that FMT could alleviate symptoms of pain and bloating.72
Eradication of antimicrobial-resistant organisms
It was noted that patients treated with FMT for rCDI who had concurrent gastrointestinal colonisation with various AROs cleared these organisms following the procedure.73,74 This sparked interest that FMT could be used to eradicate AROs from the gut. ARO colonisation rates in the community are low in Europe and America, but much higher in Africa and Asia75 and in healthcare settings.76 Travel (or hospital admission) to these areas is a risk factor for ARO colonisation, even in ‘healthy’ individuals. Once exposure is removed, a fully functioning gut microbiota appears to prevent persistent engraftment. However, antimicrobial use, or medical comorbidities, both of which contribute to continued dysbiosis, may allow resistant organisms to persist. Correcting imbalance with FMT seems a logical therapeutic strategy. Research to date, has been hampered by numerous factors. First, lack of RCTs: uncontrolled studies take no account for the spontaneous loss of ARO carriage and there is also implicit risk of publication bias against negative results; and second, the concurrent use of antimicrobials prior to FMT makes it is difficult to attribute causality to FMT alone.77–79
Immune-checkpoint-inhibitor-mediated colitis
Immune-checkpoint inhibitors are revolutionising the field of immuno-oncology, improving outcomes in a wide range of malignancies. A side effect of therapy is a colitis resembling IBD, both endoscopically and histologically, which usually responds to immunomodulatory treatment. Reports of refractory cases being successfully treated with FMT are emerging.80,81 One of these studies suggested possible mechanisms of action including FMT re-establishing the population of Treg cells in the gut mucosa, as well as gut microbiota changes away from pathogenic species such as Escherichia towards species such as Verrucomicrobiae with anti-inflammatory activity through the production of SCFAs.
Cirrhosis
The majority of research to date has focused on the prevention of hepatic encephalopathy, the most frequent manifestation of decompensated liver disease. RCTs have highlighted the safety and potential efficacy of FMT in improving both cognition and reducing hospital admissions in treatment versus control groups.82,83 Pre-treatment with antimicrobials does not appear necessary for FMT engraftment, and may actually cause harm (model for end-stage liver disease score transiently worsened post-antibiotics, but reverted to baseline post-FMT in one study).82 Studies reported to date have been limited by size, and included patients at the more severe end of the disease spectrum, so wider applicability remains unknown. FMT responders initially had an increase in secondary bile acids, possibly due to colonisation with Ruminococceae and Lachnospiraceae, and a rise in bacterial species such as Verrucomicrobiae, associated with the production of SCFAs. There was also evidence of reduced peripheral markers of inflammation (e.g. IL-6). However, by 12 months, many of these changes did not persist (although differences remained detectable between intervention and control groups); regardless, outcomes remained improved in terms of cognitive functioning and requirement for hospitalisation in patients who received FMT.84
Alcoholic hepatitis
As the name suggests, alcoholic hepatitis (AH) is liver inflammation secondary to excessive alcohol consumption. It is usually seen in chronic heavy drinkers and is frequently a precursor to the development of cirrhosis. In the extreme, acute cases can be life threatening, with complications including jaundice, ascites and hepatic encephalopathy. Current treatment options such as corticosteroids and liver transplantation are limited by inefficacy and patient ineligibility. Data are starting to emerge to suggest FMT may have a role to play in the management of AH, with a couple of small studies showing reduction in mortality (80% survival in intervention groups versus 30–40% in control groups) and disease-associated complications, including ascites and hepatic encephalopathy, up to 1 year of follow up.85,86
Hepatitis B infection
Chronic hepatitis B afflicts over 250 million individuals worldwide and is responsible for close to a million deaths per year.87,88 Results from two studies suggest FMT may aid hepatitis B virus e-antigen clearance (2/12 who received FMT versus 0/15 who did not in one study, and 3/5 FMT recipients versus 0/13 who did not in the other) in chronic carriers who fail to respond to anti-viral therapy. However, small sample sizes and the lack of participant randomisation make it difficult to draw any definitive conclusions as to whether FMT has a role in the setting of chronic hepatitis B infection at present.89,90 Results from an RCT completed in March 2018 [ClinicalTrials.gov identifier: NCT02689245] are awaited.
Obesity and the metabolic syndrome
Two recently published trials of capsulised FMT in obesity did not show benefit over control in a range of outcomes including body mass index and insulin sensitivity despite evidence of microbiota modification in FMT recipients questioning the role of FMT in this setting.91,92 This contrasts with historical studies which suggested FMT may be able to increase insulin sensitivity and improve glycaemic control, albeit only in the short term.93 There are numerous differences between studies including FMT delivery route and donor selection, which could go some way to explaining observed discrepancies, as well as the fact they were all small studies (the largest had 38 patients in total).
Non-alcoholic fatty liver disease is considered a hepatic manifestation of the metabolic syndrome and is associated with increased gut permeability. Allogeneic FMT from lean donors was superior to autologous FMT in improving gut barrier integrity in the short term (6 weeks), but not in increasing insulin sensitivity or reducing hepatic fat deposition.94 This was the first study outside of CDI to show a definitive reduction in intestinal permeability post-FMT and has potentially important ramifications with respect to other conditions.
Systemic inflammatory response syndrome (SIRS)
In critically unwell patients with acute respiratory distress syndrome, shock or intractable diarrhoea, for which no alternate aetiology can be found, it has been proposed that gut dysbiosis is, at least in part, contributing to, or even driving, the disease process. A reduction in bacteria that produce SCFAs and an increase in pathogenic bacteria such as Enterococcus and Escherichia has been found in patients admitted to intensive care.95 These changes may impair gut integrity and promote the release of toxins into the systemic circulation driving an inflammatory response. As previously discussed, FMT has been demonstrated to reverse changes such as these, and improvement in clinical condition post-FMT has been seen in a small number of cases.96 Further studies, including RCTs, are required to appreciate if this can truly be attributed to FMT.
Anxiety and depression
The gut–brain axis is a bidirectional signalling pathway which may be modulated by the gut microbiota. The current evidence base for the effect of FMT in psychiatric disorders (predominantly anxiety and depression) has been previously summarised.97 The overwhelming majority of in-human studies have enrolled patients with concurrent IBS98–102 or are limited to case reports,103–105 with only one study98 involving a control group and blinding, making it difficult to draw firm conclusions at present. Improvements were seen in depression, anxiety, quality-of-life and fatigue scores, although the effect may only be transient.
Autistic spectrum disorder
The high frequency of concurrent GI symptoms in individuals with autistic spectrum disorder (ASD), and their link to disease severity,106 has led to the suggestion that gut dysbiosis may be implicated in disease pathogenesis. Detectable differences in microbiota between children with ASD and controls have been identified,107,108 but interestingly not in studies with siblings as controls.109,110 FMT is proposed to work by increasing bacterial diversity, improving gut wall integrity and modulating blood metabolites.111 An open-label study which recruited 18 children with ASD and 20 age-matched ‘neurotypical’ controls showed improvement in both GI and behavioural symptoms post-FMT delivery. Limitations of the study include the use of antibiotics prior to FMT delivery (which has previously been shown to modulate symptoms112), the intensity of the treatment period (10 weeks), short-term follow up (8 weeks), and the lack of randomisation or blinding.
Parkinson’s disease
Parkinson’s disease (PD) is a progressive neurodegenerative disorder affecting neurons within the central, enteric, and peripheral autonomic nervous systems. Concurrent GI disturbance, usually constipation or slow transit, is common, and may precede motor symptoms.113 This has led to the proposition that the disease may even originate in the gut. A key component of disease aetiology is the aggregation of the protein, alpha-synuclein, a major component of Lewy bodies, in these neurons. It has been demonstrated (albeit in animal models) that alpha-synuclein is capable of being transported from the gut to the brain.114 This process is thought to be potentiated by gut inflammation.115 Numerous changes in the gut have been identified in patients with PD, including increased abundance of pro-inflammatory cytokines and bacteria,116 fewer anti-inflammatory-producing bacteria,115 and increased intestinal permeability,117 supporting this hypothesis. And, as discussed, FMT has the capacity to reverse these changes. Published in-human studies of FMT in PD to date are limited to a single case report, which showed a transient benefit in motor symptoms, but a more prolonged improvement in constipation.118 The results of presently recruiting clinical trials will be required to assess in FMT has a future role in PD.
Epilepsy
Differences in the microbiota of patients with treatment-refractory epilepsy, drug-sensitive epilepsy and healthy controls have been identified, and proven interventions which modulate the host microbiota (ketogenic diet) have been used to manage epilepsy.119,120 Studies show a relative increase in abundance of Firmicutes compared with Bacteroidetes in patients with treatment-refractory disease.120–122 Firmicutes may alter neurotransmitter levels, which could have an influence on seizure threshold. Higher levels of Bifidobacteria and Lactobacillus were associated with a reduced seizure frequency.122 In humans, studies are limited to a single case report of a patient with concurrent CD. Post-FMT there was a reduction in seizure frequency and an improvement in the activity index of the patient’s IBD.123
Multiple sclerosis
It has been suggested that patients with multiple sclerosis (MS) have a gut microbiota that is less able to induce Treg cells leading to a rise in Th1 and Th17.124 Elevated Th1 and Th17 cells are in turn hypothesised to induce central nervous system (CNS) inflammation and reduce blood–brain barrier permeability, potentiating further CNS inflammation.125 Modulation of the gut microbiota to induce more Treg cells could result in less activation of pathogenic T cells.126 Human studies so far have been restricted to case reports/series,127,128 and although they show promise, the results of ongoing clinical trials are awaited to understand if FMT will have a role in the management of MS.
Acute myeloid leukaemia
The combination of intensive chemotherapy and multiple courses of broad-spectrum antimicrobials for febrile neutropaenia result in significant gut dysbiosis in individuals with acute myeloid leukaemia. Autologous FMT harvested pre-treatment and given following induction treatment has been demonstrated to restore microbial diversity and potentially reduce systemic inflammation and the expression of antimicrobial-resistance genes. The study did not include a control group and there was a per-protocol analysis;129 nevertheless, results are encouraging, and RCTs are warranted to investigate further.
Future perspectives
Delivery route, dosage, and preparation techniques
The future role of FMT is likely to be shaped by how it can be delivered. Capsulised FMT has major advantages over fresh/frozen FMT delivered via colonoscopy, not least in terms of patient acceptability, but also practicality and scalability. This comes with the proviso that the active product is not detrimentally affected by the lyophilisation and/or encapsulation process and remains efficacious. There are further advantages if repeated dosing is required to achieve primary efficacy or durable effect, including delivery of therapy as an outpatient (the patient could store their treatment at home negating repeat trips to hospital). Results have emerged to suggest differences in efficacy for different preparations with respect to the indication for which FMT is being provided, for example, FMT appears to be effective via all routes/forms for CDI, but not for IBS.
Currently, there is a lack of clarity as to what dose of FMT is necessary to have therapeutic effect, and again, this may be indication dependent.130 A minimum of 30 g of faeces has been recommended for the treatment of rCDI.131 Further studies are necessary to identify minimum effective dose and redosing frequency for other indications.
Additionally, how FMT should be prepared remains subject to debate. Most colonic bacteria are obligate anaerobes, making the oxygen-rich environment of the outside world a suboptimal climate in which to preserve their viability. Therefore, anaerobic stool preparation has been trialled, with evidence of improved preservation of certain bacterial species using this technique.132 Although success rates in rCDI are unchanged by preparatory environment, it may be relevant for other interventions.133
Personalised therapy and standardisation
Recognition that success rates of FMT for indications other than CDI may be donor dependent has resulted in the idea that donor selection could influence FMT outcomes. Many of the stool donations in the described RCTs came from only one or two selected donors. This is beneficial in terms of creating a more universal product, but on the other hand, may mean that results are not widely applicable, and, if greater alpha diversity contributes to the efficacy of FMT, this approach may actually limit treatment effect. At present, there are many ideas about who is the ideal donor. They include a donor who was breastfed when an infant, is a non-smoker, is unrelated to recipient, has had minimal or no previous antibiotic exposure, has no recent travel history to an area with high rates of AROs, and so forth. Presently, there remains little hard evidence to confirm exactly what constitutes a ‘healthy microbiome’, and who is the model donor, and whether having multiple donors is, in fact, superior.
There is currently no licensed, industry-developed FMT-like product that can be administered to patients (outside of a clinical trial). Thus, we are reliant on stool banks set up by healthcare organisations, academia and in some cases, not-for-profit companies, together with the generosity of donors. This model is limited by strict screening criteria and a lack of stool banks to produce material for both routine clinical service and research studies.
Understandably creation of a ‘synthetic’ FMT is a desired goal in terms of reproducibility, safety, regulation and scalability, as well as commercial opportunity. Results of ‘manufactured’ FMT to date have shown promise,134,135 and further trials are ongoing [ClinicalTrials.gov identifiers: NCT03244644, NCT03788434 and NCT04208958 to list a few], but there remains a way to go before we are likely to see these entering widespread practice.
Other potential indications
Hypotheses, in vitro work, animal models, and in-human studies yet to be reported have suggested a potential role in a number of indications that we have not discussed in this article. These include, but are not limited to, Alzheimer’s disease, Guillain–Barré syndrome, stroke,136 allergy and atopy,137 psoriatic (and other inflammatory) arthropathy,138 colorectal cancer,139 and even COVID-19.140
Clinical trials
Supplementary Table 1 summarises trials in which FMT is being used as the investigational medicinal product (outside of CDI) that have completed, but for which no reported results could be identified. Supplementary Table 2 summarises the ongoing trials using FMT as of 17 August 2020 on ClinicalTrials.gov. The most common indication is IBD (particularly UC), with multiple RCTs with a target recruitment of >100 participants. There are also RCTs with >100 planned participants investigating FMT in eradication of AROs, IBS, cirrhosis, and gut dysbiosis post-Caesarean section and post-stem-cell transplant. Follow up in certain studies is for up to 10 years. These are all important steps in right direction to establishing a solid evidence base for the role of FMT beyond rCDI. Twenty-five trials yet to start recruiting include the additional indications of systemic sclerosis [ClinicalTrials.gov identifier: NCT04300426], alopecia [ClinicalTrials.gov identifier: NCT04238091] and hypertension [ClinicalTrials.gov identifier: NCT04406129].
Conclusion
Ingestion of faecal material has been used in as medicinal therapy for thousands of years, but it is not until the last decade that high-quality evidence to support the practice of FMT has emerged. FMT has multiple plausible mechanisms of action, including colonisation resistance, anti-inflammatory and immunomodulatory actions and direct antimicrobial properties from transmitted substances such as bacteriocins and organisms such as bacteriophages. However, the relative contribution of each proposed mechanism remains unclear, and probably varies according to the condition being treated.
There is a growing evidence base for the role of FMT for non-CDI indications; however, at present, the overwhelming majority of studies are limited to case reports/series or small pilot studies, and no firm conclusions as to the efficacy of FMT can be drawn. Carefully designed RCTs (many of which are in progress) will be necessary to truly begin to understand what future role, if any, it may play. Further work will also be required with respect to dosing, donor selection, and comparison of delivery routes and FMT preparations. Nevertheless, the early indications are there that FMT could be a promising therapy with established biological plausibility in IBD, hepatic encephalopathy, and a multitude of other conditions. It truly is an exciting and fascinating time to be working on the gut microbiota and faecal transplantation.
Supplemental Material
Supplemental material, sj-pdf-1-tai-10.1177_2049936120981526 for The role of faecal microbiota transplantation: looking beyond Clostridioides difficile infection by Simon D. Goldenberg and Blair Merrick in Therapeutic Advances in Infectious Disease
Footnotes
Author Contributions: SG and BM conceived the article, reviewed the literature, wrote, and critically reviewed the manuscript and all revisions. The corresponding author (SG) attests that no others meeting authorship criteria have been omitted.
Funding: The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement: Simon Goldenberg reports consulting fees from Astellas, Enterobiotix, MSD, Pfizer, and Shionogi.
ORCID iDs: Simon Goldenberg  https://orcid.org/0000-0002-6061-6064
https://orcid.org/0000-0002-6061-6064
Blair Merrick  https://orcid.org/0000-0003-0837-7382
https://orcid.org/0000-0003-0837-7382
Supplemental material: Supplemental material for this article is available online.
Contributor Information
Simon D. Goldenberg, Centre for Clinical Infection & Diagnostics Research, King’s College London and Guy’s & St. Thomas’ NHS Foundation Trust, 5th floor, North Wing, St Thomas’ hospital, Westminster Bridge Road, London, SE1 7EH, UK.
Blair Merrick, Centre for Clinical Infection & Diagnostics Research, King’s College London and Guy’s & St. Thomas’ NHS Foundation Trust, London, UK.
References
- 1. Dhama K, Rathore R, Chauhan RS, et al. Panchgavya (Cowpathy): an overview. Int J Cow Sci 2005; 1: 1–15. [Google Scholar]
- 2. Zhang F, Luo W, Shi Y, et al. Should we standardize the 1,700-year-old fecal microbiota transplantation? Am J Gastroenterol 2012; 107: 1755; author reply p.1755–1756. [DOI] [PubMed] [Google Scholar]
- 3. VICE. Korean Poo wine, https://www.vice.com/amp/en_us/article/7b7zmg/how-to-make-faeces-wine (2013, accessed 22 July 2020).
- 4. Lewin RA. More on Merde. Perspect Biol Med 2001; 44: 594–607. [DOI] [PubMed] [Google Scholar]
- 5. Eiseman B, Silen W, Bascom GS, et al. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery 1958; 44: 854–859. [PubMed] [Google Scholar]
- 6. Van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med 2013; 368: 407–415. [DOI] [PubMed] [Google Scholar]
- 7. Kelly CR, Khoruts A, Staley C, et al. Effect of fecal microbiota transplantation on recurrence in multiply recurrent Clostridium difficile infection: a randomized trial. Ann Intern Med 2016; 165: 609–616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Youngster I, Sauk J, Pindar C, et al. Fecal microbiota transplant for relapsing Clostridium difficile infection using a frozen inoculum from unrelated donors: a randomized, open-label, controlled pilot study. Clin Infect Dis 2014; 58: 1515–1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Quraishi MN, Widlak M, Bhala N, et al. Systematic review with meta-analysis: the efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection. Aliment Pharmacol Ther 2017; 46: 479–493. [DOI] [PubMed] [Google Scholar]
- 10. Moayyedi P, Yuan Y, Baharith H, et al. Faecal microbiota transplantation for Clostridium difficile-associated diarrhoea: a systematic review of randomised controlled trials. Med J Aust 2017; 207: 166–172. [DOI] [PubMed] [Google Scholar]
- 11. Kao D, Roach B, Silva M, et al. Effect of oral capsule- vs colonoscopy-delivered fecal microbiota transplantation on recurrent Clostridium difficile infection: a randomized clinical trial. JAMA 2017; 318: 1985–1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Cammarota G, Masucci L, Ianiro G, et al. Randomised clinical trial: faecal microbiota transplantation by colonoscopy vs. vancomycin for the treatment of recurrent Clostridium difficile infection. Aliment Pharmacol Ther 2015; 41: 835–843. [DOI] [PubMed] [Google Scholar]
- 13. Lee CH, Steiner T, Petrof EO, et al. Frozen vs fresh fecal microbiota transplantation and clinical resolution of diarrhea in patients with recurrent Clostridium difficile infection: a randomized clinical trial. JAMA 2016; 315: 142–149. [DOI] [PubMed] [Google Scholar]
- 14. Mullish BH, Quraishi MN, Segal JP, et al. The use of faecal microbiota transplant as treatment for recurrent or refractory Clostridium difficile infection and other potential indications: joint British Society of Gastroenterology (BSG) and Healthcare Infection Society (HIS) guidelines. J Hosp Infect 2018; 100(Suppl. 1): S1–S31. [DOI] [PubMed] [Google Scholar]
- 15. Malla MA, Dubey A, Kumar A, et al. Exploring the human microbiome: the potential future role of next-generation sequencing in disease diagnosis and treatment. Front Immunol 2018; 9: 2868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Vernocchi P, Del Chierico F, Putignani L. Gut microbiota profiling: metabolomics based approach to unravel compounds affecting human health. Front Microbiol 2016; 7: 1144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mallick H, Franzosa EA, McLver LJ, et al. Predictive metabolomic profiling of microbial communities using amplicon or metagenomic sequences. Nat Commun 2019; 10: 3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Shreiner AB, Kao JY, Young VB. The gut microbiome in health and in disease. Curr Opin Gastroenterol 2015; 31: 69–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Brüssow H. Problems with the concept of gut microbiota dysbiosis. Microb Biotechnol 2020; 13: 423–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Lawley TD, Walker AW. Intestinal colonization resistance. Immunology 2013; 138: 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Fuentes S, Van Nood E, Tims S, et al. Reset of a critically disturbed microbial ecosystem: faecal transplant in recurrent Clostridium difficile infection. ISME J 2014; 8: 1621–1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Khoruts A, Sadowsky MJ. Understanding the mechanisms of faecal microbiota transplantation. Nat Rev Gastroenterol Hepatol 2016; 13: 508–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Kakiyama G, Pandak WM, Gillevet PM, et al. Modulation of the fecal bile acid profile by gut microbiota in cirrhosis. J Hepatol 2013; 58: 949–955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Staley C, Kaiser T, Beura LK, et al. Stable engraftment of human microbiota into mice with a single oral gavage following antibiotic conditioning. Microbiome 2017; 5: 87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chiang JY. Bile acids: regulation of synthesis. J Lipid Res 2009; 50: 1955–1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Allegretti JR, Kearney S, Li N, et al. Recurrent Clostridium difficile infection associates with distinct bile acid and microbiome profiles. Aliment Pharmacol Ther 2016; 43: 1142–1153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Alimov I, Menon S, Cochran N, et al. Bile acid analogues are activators of pyrin inflammasome. J Biol Chem 2019; 294: 3359–3366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Ng KM, Ferreyra JA, Higginbottom SK, et al. Microbiota-liberated host sugars facilitate post-antibiotic expansion of enteric pathogens. Nature 2013; 502: 96–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Den Besten G, Van Eunen K, Groen AK, et al. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res 2013; 54: 2325–2340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Smith PM, Howitt MR, Panikov N, et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013; 341: 569–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Yang HT, Chen JW, Rathod J, et al. Lauric acid is an inhibitor of Clostridium difficile growth in vitro and reduces inflammation in a mouse infection model. Front Microbiol 2017; 8: 2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Zackular JP, Moore JL, Jordan AT, et al. Dietary zinc alters the microbiota and decreases resistance to Clostridium difficile infection. Nat Med 2016; 22: 1330–1334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Jack RW, Tagg JR, Ray B. Bacteriocins of gram-positive bacteria. Microbiol Rev 1995; 59: 171–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Simons A, Alhanout K, Duval RE. Bacteriocins, antimicrobial peptides from bacterial origin: overview of their biology and their impact against multidrug-resistant bacteria. Microorganisms 2020; 8: 639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kociolek LK, Gerding DN. Breakthroughs in the treatment and prevention of Clostridium difficile infection. Nat Rev Gastroenterol Hepatol 2016; 13: 150–160. [DOI] [PubMed] [Google Scholar]
- 36. McQuade R, Roxas B, Viswanathan VK, et al. Clostridium difficile clinical isolates exhibit variable susceptibility and proteome alterations upon exposure to mammalian cationic antimicrobial peptides. Anaerobe 2012; 18: 614–620. [DOI] [PubMed] [Google Scholar]
- 37. Dowah ASA, Clokie MRJ. Review of the nature, diversity and structure of bacteriophage receptor binding proteins that target gram-positive bacteria. Biophys Rev 2018; 10: 535–542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Fortier LC. Bacteriophages contribute to shaping Clostridioides (Clostridium) difficile species. Front Microbiol 2018; 9: 2033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Romero-Calle D, Guimarães Benevides R, Góes-Neto A, et al. Bacteriophages as alternatives to antibiotics in clinical care. Antibiotics (Basel) 2019; 8: 138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Frisbee AL, Petri WA., Jr. Considering the immune system during fecal microbiota transplantation for Clostridioides difficile infection. Trends Mol Med 2020; 26: 496–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res 2020; 30: 492–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Hall JA, Bouladoux N, Sun CM, et al. Commensal DNA limits regulatory T cell conversion and is a natural adjuvant of intestinal immune responses. Immunity 2008; 29: 637–649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Ivanov II, Atarashi K, Manel N, et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 2009; 139: 485–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Merrick B, Allen L, Zain NMM, et al. Regulation, risk and safety of faecal microbiota transplant. Infect Prev Pract 2020; 2: 100069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. DeFilipp Z, Bloom PP, Torres Soto M, et al. Drug-resistant E. coli bacteremia transmitted by fecal microbiota transplant. N Engl J Med 2019; 381: 2043–2050. [DOI] [PubMed] [Google Scholar]
- 46. Amirian ES. Potential fecal transmission of SARS-CoV-2: current evidence and implications for public health. Int J Infect Dis 2020; 95: 363–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Bhangu A, Nepogodiev D, Gupta A, et al. Systematic review and meta-analysis of outcomes following emergency surgery for Clostridium difficile colitis. Br J Surg 2012; 99: 1501–1513. [DOI] [PubMed] [Google Scholar]
- 48. Fischer M, Sipe B, Cheng YW, et al. Fecal microbiota transplant in severe and severe-complicated Clostridium difficile: a promising treatment approach. Gut Microbes 2017; 8: 289–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Ianiro G, Masucci L, Quaranta G, et al. Randomised clinical trial: faecal microbiota transplantation by colonoscopy plus vancomycin for the treatment of severe refractory Clostridium difficile infection-single versus multiple infusions. Aliment Pharmacol Ther 2018; 48: 152–159. [DOI] [PubMed] [Google Scholar]
- 50. Cheng YW, Fischer M. Fecal microbiota transplantation: redefining surgical management of refractory Clostridium difficile infection. Clin Colon Rectal Surg 2020; 33: 92–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Imdad A, Nicholson MR, Tanner-Smith EE, et al. Fecal transplantation for treatment of inflammatory bowel disease. Cochrane Database Syst Rev 2018; 11: CD012774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Costello SP, Hughes PA, Waters O, et al. Effect of fecal microbiota transplantation on 8-week remission in patients with ulcerative colitis: a randomized clinical trial. JAMA 2019; 321: 156–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Rajilic-Stojanovic M, Shanahan F, Guarner F, et al. Phylogenetic analysis of dysbiosis in ulcerative colitis during remission. Inflamm Bowel Dis 2013; 19: 481–488. [DOI] [PubMed] [Google Scholar]
- 54. Moayyedi P, Surette MG, Kim PT, et al. Fecal microbiota transplantation induces remission in patients with active ulcerative colitis in a randomized controlled trial. Gastroenterology 2015; 149: 102–109.e106. [DOI] [PubMed] [Google Scholar]
- 55. Sokol H, Landman C, Seksik P, et al. Fecal microbiota transplantation to maintain remission in Crohn’s disease: a pilot randomized controlled study. Microbiome 2020; 8: 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Sabino J, Vieira-Silva S, Machiels K, et al. Primary sclerosing cholangitis is characterised by intestinal dysbiosis independent from IBD. Gut 2016; 65: 1681–1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Tabibian JH, O’Hara SP, Lindor KD. Primary sclerosing cholangitis and the microbiota: current knowledge and perspectives on etiopathogenesis and emerging therapies. Scand J Gastroenterol 2014; 49: 901–908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Davies YK, Cox KM, Abdullah BA, et al. Long-term treatment of primary sclerosing cholangitis in children with oral vancomycin: an immunomodulating antibiotic. J Pediatr Gastroenterol Nutr 2008; 47: 61–67. [DOI] [PubMed] [Google Scholar]
- 59. Tabibian JH, Weeding E, Jorgensen RA, et al. Randomised clinical trial: vancomycin or metronidazole in patients with primary sclerosing cholangitis - a pilot study. Aliment Pharmacol Ther 2013; 37: 604–612. [DOI] [PubMed] [Google Scholar]
- 60. Allegretti JR, Kassam Z, Carrellas M, et al. Fecal microbiota transplantation in patients with primary sclerosing cholangitis: a pilot clinical trial. Am J Gastroenterol 2019; 114: 1071–1079. [DOI] [PubMed] [Google Scholar]
- 61. Xu D, Chen VL, Steiner CA, et al. Efficacy of fecal microbiota transplantation in irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol 2019; 114: 1043–1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Myneedu K, Deoker A, Schmulson MJ, et al. Fecal microbiota transplantation in irritable bowel syndrome: a systematic review and meta-analysis. United European Gastroenterol J 2019; 7: 1033–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ianiro G, Eusebi LH, Black CJ, et al. Systematic review with meta-analysis: efficacy of faecal microbiota transplantation for the treatment of irritable bowel syndrome. Aliment Pharmacol Ther 2019; 50: 240–248. [DOI] [PubMed] [Google Scholar]
- 64. Holvoet T, Joossens M, Vazquez-Castellanos JF, et al. Fecal microbiota transplantation reduces symptoms in some patients with irritable bowel syndrome with predominant abdominal bloating: short- and long-term results from a placebo-controlled randomized trial. Gastroenterology. Epub ahead of print 15 July 2020. DOI: 10.1053/j.gastro.2020.07.013. [DOI] [PubMed] [Google Scholar]
- 65. El-Salhy M, Hatlebakk JG, Gilja OH, et al. Efficacy of faecal microbiota transplantation for patients with irritable bowel syndrome in a randomised, double-blind, placebo-controlled study. Gut 2020; 69: 859–867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Lahtinen P, Jalanka J, Hartikainen A, et al. Randomised clinical trial: faecal microbiota transplantation versus autologous placebo administered via colonoscopy in irritable bowel syndrome. Aliment Pharmacol Ther 2020; 51: 1321–1331. [DOI] [PubMed] [Google Scholar]
- 67. Kenyon JN, Coe S, Izadi H. A retrospective outcome study of 42 patients with chronic fatigue syndrome, 30 of whom had irritable bowel syndrome. Half were treated with oral approached, and half were treated with faecal microbiome transplantation. Hum Microbiome J 2019; 13: 100061. [Google Scholar]
- 68. Frattini JC, Nogueras JJ. Slow transit constipation: a review of a colonic functional disorder. Clin Colon Rectal Surg 2008; 21: 146–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Tian H, Ding C, Gong J, et al. Treatment of slow transit constipation with fecal microbiota transplantation: a pilot study. J Clin Gastroenterol 2016; 50: 865–870. [DOI] [PubMed] [Google Scholar]
- 70. Zhang X, Tian H, Gu L, et al. Long-term follow-up of the effects of fecal microbiota transplantation in combination with soluble dietary fiber as a therapeutic regimen in slow transit constipation. Sci China Life Sci 2018; 61: 779–786. [DOI] [PubMed] [Google Scholar]
- 71. Tian H, Ge X, Nie Y, et al. Fecal microbiota transplantation in patients with slow-transit constipation: a randomized, clinical trial. PLoS One 2017; 12: e0171308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Gu L, Ding C, Tian H, et al. Serial frozen fecal microbiota transplantation in the treatment of chronic intestinal pseudo-obstruction: a preliminary study. J Neurogastroenterol Motil 2017; 23: 289–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Dubberke ER, Mullane KM, Gerding DN, et al. Clearance of vancomycin-resistant enterococcus concomitant with administration of a microbiota-based drug targeted at recurrent Clostridium difficile infection. Open Forum Infect Dis 2016; 3: ofw133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Tariq R, Pardi DS, Tosh PK, et al. Fecal microbiota transplantation for recurrent Clostridium difficile infection reduces recurrent urinary tract infection frequency. Clin Infect Dis 2017; 65: 1745–1747. [DOI] [PubMed] [Google Scholar]
- 75. Karanika S, Karantanos T, Arvanitis M, et al. Fecal colonization with extended-spectrum beta-lactamase-producing enterobacteriaceae and risk factors among healthy individuals: a systematic review and metaanalysis. Clin Infect Dis 2016; 63: 310–318. [DOI] [PubMed] [Google Scholar]
- 76. Feldman N, Adler A, Molshatzki N, et al. Gastrointestinal colonization by KPC-producing Klebsiella pneumoniae following hospital discharge: duration of carriage and risk factors for persistent carriage. Clin Microbiol Infect 2013; 19: E190–E196. [DOI] [PubMed] [Google Scholar]
- 77. Woodworth MH, Hayden MK, Young VB, et al. The role of fecal microbiota transplantation in reducing intestinal colonization with antibiotic-resistant organisms: the current landscape and future directions. Open Forum Infect Dis 2019; 6: ofz288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Huttner BD, De Lastours V, Wassenberg M, et al. A 5-day course of oral antibiotics followed by faecal transplantation to eradicate carriage of multidrug-resistant enterobacteriaceae: a randomized clinical trial. Clin Microbiol Infect 2019; 25: 830–838. [DOI] [PubMed] [Google Scholar]
- 79. Bar-Yoseph H, Carasso S, Shklar S, et al. Oral capsulized fecal microbiota transplantation for eradication of carbapenemase-producing enterobacteriaceae colonization with a metagenomic perspective. Clin Infect Dis. Epub ahead of print 8 June 2020. DOI: 10.1093/cid/ciaa737. [DOI] [PubMed] [Google Scholar]
- 80. Fasanello MK, Robillard KT, Boland PM, et al. Use of fecal microbial transplantation for immune checkpoint inhibitor colitis. ACG Case Rep J 2020; 7: e00360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Wang Y, Wiesnoski DH, Helmink BA, et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat Med 2018; 24: 1804–1808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Bajaj JS, Kassam Z, Fagan A, et al. Fecal microbiota transplant from a rational stool donor improves hepatic encephalopathy: a randomized clinical trial. Hepatology 2017; 66: 1727–1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Bajaj JS, Salzman NH, Acharya C, et al. Fecal microbial transplant capsules are safe in hepatic encephalopathy: a phase 1, randomized, placebo-controlled trial. Hepatology 2019; 70: 1690–1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Bajaj JS, Fagan A, Gavis EA, et al. Long-term outcomes of fecal microbiota transplantation in patients with cirrhosis. Gastroenterology 2019; 156: 1921–1923.e1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Philips CA, Pande A, Shasthry SM, et al. Healthy donor fecal microbiota transplantation in steroid-ineligible severe alcoholic hepatitis: a pilot study. Clin Gastroenterol Hepatol 2017; 15: 600–602. [DOI] [PubMed] [Google Scholar]
- 86. Indian Society of Gastroenterology. Indian J Gastroenterol 2016; 35(Suppl. 1): 1–111. [DOI] [PubMed] [Google Scholar]
- 87. Zampino R, Boemio A, Sagnelli C, et al. Hepatitis B virus burden in developing countries. World J Gastroenterol 2015; 21: 11941–11953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. World Health Organization. Global hepatitis report, 2017. Geneva: World Health Organization, 2017. [Google Scholar]
- 89. Chauhan A, Kumar R, Sharma S, et al. Fecal microbiota transplantation in Hepatitis B e antigen-positive chronic Hepatitis B patients: a pilot study. Dig Dis Sci. Epub ahead of print 11 April 2020. DOI: 10.1007/s10620-020-06246-x. [DOI] [PubMed] [Google Scholar]
- 90. Ren YD, Ye ZS, Yang LZ, et al. Fecal microbiota transplantation induces hepatitis B virus e-antigen (HBeAg) clearance in patients with positive HBeAg after long-term antiviral therapy. Hepatology 2017; 65: 1765–1768. [DOI] [PubMed] [Google Scholar]
- 91. Yu EW, Gao L, Stastka P, et al. Fecal microbiota transplantation for the improvement of metabolism in obesity: the FMT-TRIM double-blind placebo-controlled pilot trial. PLoS Med 2020; 17: e1003051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Allegretti JR, Kassam Z, Mullish BH, et al. Effects of fecal microbiota transplantation with oral capsules in obese patients. Clin Gastroenterol Hepatol 2020; 18: 855–863.e852. [DOI] [PubMed] [Google Scholar]
- 93. Zhang Z, Mocanu V, Cai C, et al. Impact of fecal microbiota transplantation on obesity and metabolic syndrome-a systematic review. Nutrients 2019; 11: 2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Craven L, Rahman A, Nair Parvathy S, et al. Allogenic fecal microbiota transplantation in patients with nonalcoholic fatty liver disease improves abnormal small intestinal permeability: a randomized control trial. Am J Gastroenterol 2020; 115: 1055–1065. [DOI] [PubMed] [Google Scholar]
- 95. Lankelma JM, Van Vught LA, Belzer C, et al. Critically ill patients demonstrate large interpersonal variation in intestinal microbiota dysregulation: a pilot study. Intensive Care Med 2017; 43: 59–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Limketkai BN, Hendler S, Ting PS, et al. Fecal microbiota transplantation for the critically ill patient. Nutr Clin Pract 2019; 34: 73–79. [DOI] [PubMed] [Google Scholar]
- 97. Chinna Meyyappan A, Forth E, Wallace CJK, et al. Effect of fecal microbiota transplant on symptoms of psychiatric disorders: a systematic review. BMC Psychiatry 2020; 20: 299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Johnsen PH, Hilpüsch F, Valle PC, et al. The effect of fecal microbiota transplantation on IBS related quality of life and fatigue in moderate to severe non-constipated irritable bowel: secondary endpoints of a double blind, randomized, placebo-controlled trial. EBioMedicine 2020; 51: 102562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Kurokawa S, Kishimoto T, Mizuno S, et al. The effect of fecal microbiota transplantation on psychiatric symptoms among patients with irritable bowel syndrome, functional diarrhea and functional constipation: an open-label observational study. J Affect Disord 2018; 235: 506–512. [DOI] [PubMed] [Google Scholar]
- 100. Mizuno S, Masaoka T, Naganuma M, et al. Bifidobacterium-rich fecal donor may be a positive predictor for successful fecal microbiota transplantation in patients with irritable bowel syndrome. Digestion 2017; 96: 29–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Mazzawi T, Lied GA, Sangnes DA, et al. The kinetics of gut microbial community composition in patients with irritable bowel syndrome following fecal microbiota transplantation. PLoS One 2018; 13: e0194904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Huang HL, Chen HT, Luo QL, et al. Relief of irritable bowel syndrome by fecal microbiota transplantation is associated with changes in diversity and composition of the gut microbiota. J Dig Dis 2019; 20: 401–408. [DOI] [PubMed] [Google Scholar]
- 103. Cai T, Shi X, Yuan LZ, et al. Fecal microbiota transplantation in an elderly patient with mental depression. Int Psychogeriatr 2019; 31: 1525–1526. [DOI] [PubMed] [Google Scholar]
- 104. De Clercq NC, Frissen MN, Davids M, et al. Weight gain after fecal microbiota transplantation in a patient with recurrent underweight following clinical recovery from anorexia nervosa. Psychother Psychosom 2019; 88: 58–60. [DOI] [PubMed] [Google Scholar]
- 105. Xie WR, Yang XY, Xia HH, et al. Hair regrowth following fecal microbiota transplantation in an elderly patient with alopecia areata: a case report and review of the literature. World J Clin Cases 2019; 7: 3074–3081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Adams JB, Johansen LJ, Powell LD, et al. Gastrointestinal flora and gastrointestinal status in children with autism–comparisons to typical children and correlation with autism severity. BMC Gastroenterol 2011; 11: 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Finegold SM, Dowd SE, Gontcharova V, et al. Pyrosequencing study of fecal microflora of autistic and control children. Anaerobe 2010; 16: 444–453. [DOI] [PubMed] [Google Scholar]
- 108. De Angelis M, Piccolo M, Vannini L, et al. Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified. PLoS One 2013; 8: e76993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Gondalia SV, Palombo EA, Knowles SR, et al. Molecular characterisation of gastrointestinal microbiota of children with autism (with and without gastrointestinal dysfunction) and their neurotypical siblings. Autism Res 2012; 5: 419–427. [DOI] [PubMed] [Google Scholar]
- 110. Son JS, Zheng LJ, Rowehl LM, et al. Comparison of fecal microbiota in children with autism spectrum disorders and neurotypical siblings in the simons simplex collection. PLoS One 2015; 10: e0137725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Hsiao EY, McBride SW, Hsien S, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013; 155: 1451–1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Sandler RH, Finegold SM, Bolte ER, et al. Short-term benefit from oral vancomycin treatment of regressive-onset autism. J Child Neurol 2000; 15: 429–435. [DOI] [PubMed] [Google Scholar]
- 113. Postuma RB, Aarsland D, Barone P, et al. Identifying prodromal Parkinson’s disease: pre-motor disorders in Parkinson’s disease. Mov Disord 2012; 27: 617–626. [DOI] [PubMed] [Google Scholar]
- 114. Braak H, Rub U, Gai WP, et al. Idiopathic Parkinson’s disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. J Neural Transm (Vienna) 2003; 110: 517–536. [DOI] [PubMed] [Google Scholar]
- 115. Keshavarzian A, Green SJ, Engen PA, et al. Colonic bacterial composition in Parkinson’s disease. Mov Disord 2015; 30: 1351–1360. [DOI] [PubMed] [Google Scholar]
- 116. Devos D, Lebouvier T, Lardeux B, et al. Colonic inflammation in Parkinson’s disease. Neurobiol Dis 2013; 50: 42–48. [DOI] [PubMed] [Google Scholar]
- 117. Forsyth CB, Shannon KM, Kordower JH, et al. Increased intestinal permeability correlates with sigmoid mucosa alpha-synuclein staining and endotoxin exposure markers in early Parkinson’s disease. PLoS One 2011; 6: e28032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Huang H, Xu H, Luo Q, et al. Fecal microbiota transplantation to treat Parkinson’s disease with constipation: a case report. Medicine (Baltimore) 2019; 98: e16163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Dahlin M, Prast-Nielsen S. The gut microbiome and epilepsy. EBioMedicine 2019; 44: 741–746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Lindefeldt M, Eng A, Darban H, et al. The ketogenic diet influences taxonomic and functional composition of the gut microbiota in children with severe epilepsy. NPJ Biofilms Microbiomes 2019; 5: 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Belvin MP, Anderson KV. A conserved signaling pathway: the Drosophila toll-dorsal pathway. Annu Rev Cell Dev Biol 1996; 12: 393–416. [DOI] [PubMed] [Google Scholar]
- 122. Peng A, Qiu X, Lai W, et al. Altered composition of the gut microbiome in patients with drug-resistant epilepsy. Epilepsy Res 2018; 147: 102–107. [DOI] [PubMed] [Google Scholar]
- 123. He Z, Cui BT, Zhang T, et al. Fecal microbiota transplantation cured epilepsy in a case with Crohn’s disease: the first report. World J Gastroenterol 2017; 23: 3565–3568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Jangi S, Gandhi R, Cox LM, et al. Alterations of the human gut microbiome in multiple sclerosis. Nat Commun 2016; 7: 12015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Dendrou CA, Fugger L, Friese MA. Immunopathology of multiple sclerosis. Nat Rev Immunol 2015; 15: 545–558. [DOI] [PubMed] [Google Scholar]
- 126. Berer K, Boziki M, Krishnamoorthy G. Selective accumulation of pro-inflammatory T cells in the intestine contributes to the resistance to autoimmune demyelinating disease. PLoS One 2014; 9: e87876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Makkawi S, Camara-Lemarroy C, Metz L. Fecal microbiota transplantation associated with 10 years of stability in a patient with SPMS. Neurol Neuroimmunol Neuroinflamm 2018; 5: e459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Borody T, Leis S, Campbell J, et al. Fecal microbiota transplantation (FMT) in multiple sclerosis (MS). Am J Gastroenterol 2011; 106: S352. [Google Scholar]
- 129. Mohamad M, Malard F, Vekhoff A, et al. The Odyssee study: prevention of dysbiosis complications with autologous fecal microbiota transfer (FMT) in acute myeloid leukemia (AML) patients undergoing intensive treatment: results of a prospective multicenter trial. Blood 2018; 132(Suppl. 1): 1444. [Google Scholar]
- 130. Ng SC, Kamm MA, Yeoh YK, et al. Scientific frontiers in faecal microbiota transplantation: joint document of Asia-Pacific Association of Gastroenterology (APAGE) and Asia-Pacific Society for Digestive Endoscopy (APSDE). Gut 2020; 69: 83–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Cammarota G, Ianiro G, Tilg H, et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut 2017; 66: 569–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Papanicolas LE, Choo JM, Wang Y, et al. Bacterial viability in faecal transplants: which bacteria survive? EBioMedicine 2019; 41: 509–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Rossen NG, Fuentes S, van der Spek MJ, et al. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology 2015; 149: 110–118e114. [DOI] [PubMed] [Google Scholar]
- 134. Petrof EO, Gloor GB, Vanner SJ, et al. Stool substitute transplant therapy for the eradication of Clostridium difficile infection: ‘RePOOPulating’ the gut. Microbiome 2013; 1: 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Ford C, Litcofsky K, McGovern B, et al. Engraftment of investigational microbiome drug, SER-262, in subjects receiving vancomycin is associated with reduced rates of recurrence after primary Clostridium difficile infection (CDI). Open Forum Infect Dis 2019; 6(Suppl. 2): S547–S548. [Google Scholar]
- 136. Vendrik KEW, Ooijevaar RE, De Jong PRC, et al. Fecal microbiota transplantation in neurological disorders. Front Cell Infect Microbiol 2020; 10: 98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Albuhairi S, Rachid R. Novel therapies for treatment of food allergy. Immunol Allergy Clin North Am 2020; 40: 175–186. [DOI] [PubMed] [Google Scholar]
- 138. Kragsnaes MS, Kjeldsen J, Horn HC, et al. Efficacy and safety of faecal microbiota transplantation in patients with psoriatic arthritis: protocol for a 6-month, double-blind, randomised, placebo-controlled trial. BMJ Open 2018; 8: e019231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Sui H, Zhang L, Gu K, et al. YYFZBJS ameliorates colorectal cancer progression in Apc(Min/+) mice by remodeling gut microbiota and inhibiting regulatory T-cell generation. Cell Commun Signal 2020; 18: 113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. McIlroy JR, Mullish BH, Goldenberg SD, et al. Intestinal microbiome transfer, a novel therapeutic strategy for COVID-19 induced hyperinflammation?: in reply to, ‘COVID-19: immunology and treatment options’, Felsenstein S, Herbert JA, McNamara PS, et al. 2020’. Clin Immunol 2020; 218: 108542. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-tai-10.1177_2049936120981526 for The role of faecal microbiota transplantation: looking beyond Clostridioides difficile infection by Simon D. Goldenberg and Blair Merrick in Therapeutic Advances in Infectious Disease