Correction to: J Neuroinflammation 18, 21 (2021)
https://doi.org/10.1186/s12974-020-02048-0
Following publication of the original article [1], the authors noticed an incorrect Fig. 1 image and incorrect panel “e” background color on the image of Fig. 3 in the published version of this article. Presented here are the corrected Figs. 1 and 3. The original article has been updated.
Fig. 1.
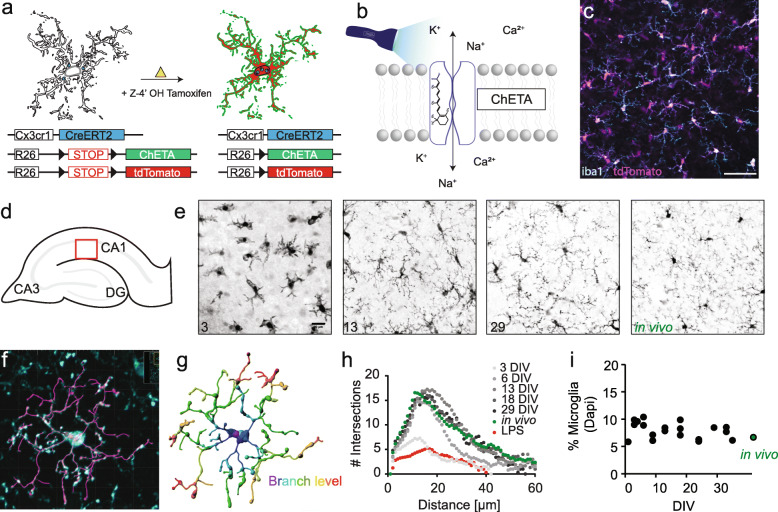
Microglia-specific ChETA expression in organotypic hippocampal slice cultures. a Schematic overview of microglia-specific expression. The microglia-driver line Cx3cr1-CreERT2 (blue) is crossed with a reporter mouse line (R26-LSL-tdTomato, red) and the ChETA mouse line (R26-LSL-ChETA, green). After injection of (Z)-4-hydroxytamoxifen, the tdTomato and ChETA are expressed in microglia. b Illustration of the Channelrhodopsin-variant ChETA activated by blue light. Scale bar 25 μm. c Immunostaining using antibodies against the reporter (tdTomato - red) and microglia (iba1 - cyan). d Graphic illustration of the hippocampal structure and the investigated area for microglia morphology in e (red square). e Z-projection of confocal images acquired for Sholl analysis of microglia at 3, 13, 29 DIV, and in vivo. Scale bar: 25 μm. f Confocal image of a microglia cell in organotypic slice culture which was fixed with PFA and stained against the microglia marker iba1. Overlay with IMARIS analysis (magenta). g Result of microglia branch detection with color coding by branching level. h Sholl analysis of microglia over time (number of intersections versus distance from cell body). i Quantification of % microglia cells between dentate gyrus and CA1 relative to total cell count (DAPI)
Fig. 3.
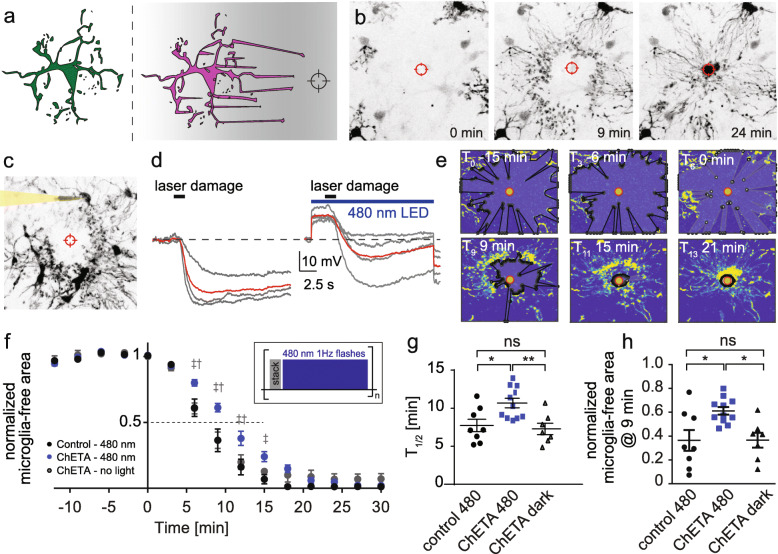
Optogenetic microglia depolarization decelerates chemotactic response kinetics. a Graphic illustration and representative images of microglia chemotaxis towards an induced laser-damage. b Two-photon maximum projections of the chemotactic response 0, 9, and 24 min after the laser-damage. c Two-photon z-projection of a patched microglia during chemotaxis. d Voltage-clamp recordings of patched microglia during chemotaxis. Gray: Individual microglial responses from four experiments, red: Average of all experiments. Left: no light stimulation during laser-damage, right: with light stimulation during laser damage. e Automated MATLAB analysis of chemotaxis quantified as the reduction in microglia-free area around the laser damage (black polygon) at different time points of the experiment. f Relative laser damage response measured as microglia-free area. Black: Control slices (no construct) with light stimulation (n = 8 areas, 5 slices). Gray: Experiments with ChETA expression in microglia, but without light stimulation (n = 7 areas, 4 slices). Blue: Slices with ChETA expression in microglia combined light stimulation (n = 11 areas, 7 slices). Insert: Graphic representation of light stimulation protocol between stack acquisitions. 2-way ANOVA (‡ control480 – ChETA480, p < 0.001, († ChETA no light – ChETA480, p < 0.01). g Time to 50% engulfment was prolonged by optogenetic depolarization. h 9 min after injury, the microglia-free area was larger when microglia were depolarized. g, h One-way ANOVA with Tukey’s post hoc comparison (*p < 0.05, **p < 0.01, ***p < 0.001)
Contributor Information
Laura Laprell, Email: laura.laprell@zmnh.uni-hamburg.de.
Thomas G. Oertner, Email: thomas.oertner@zmnh.uni-hamburg.de
Reference
- 1.Laprell L, Schulze C, Brehme ML, et al. The role of microglia membrane potential in chemotaxis. J Neuroinflammation. 2021;18:21. doi: 10.1186/s12974-020-02048-0. [DOI] [PMC free article] [PubMed] [Google Scholar]