Abstract
As the airways of SARS-CoV-2 infected patients contain a high viral load, bronchoscopy is associated with increased risk of patient to health care worker transmission due to aerosolised viral particles and contamination of surfaces during bronchoscopy. Bronchoscopy is not appropriate for diagnosing SARS-CoV-2 infection and, as an aerosol generating procedure involving a significant risk of transmission, has a very limited role in the management of SARS-CoV-2 infected patients including children. During the SARS-CoV-2 pandemic rigid bronchoscopy should be avoided due to the increased risk of droplet spread. Flexible bronchoscopy should be performed first in SARS-CoV-2 positive individuals or in unknown cases, to determine if rigid bronchoscopy is indicated. When available single-use flexible bronchoscopes may be considered for use; devices are available with a range of diameters, and improved image quality and degrees of angulation. When rigid bronchoscopy is necessary, jet ventilation must be avoided and conventional ventilation be used to reduce the risk of aerosolisation. Adequate personal protection equipment is key, as is training of health care workers in correct donning and doffing. Modified full face masks are a practical and safe alternative to filtering facepieces for use in bronchoscopy. When anaesthetic and infection prevention control protocols are strictly adhered to, bronchoscopy can be performed in SARS-CoV-2 positive children.
Keywords: Bronchoscopy, COVID-19, SARS-CoV-2, Single-use flexible bronchoscopes, Personal protection equipment
Introduction
The emergence and rapid global spread of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-COV-2) resulting in Coronavirus Disease 2019 (COVID-19) initially reported in Wuhan, China has been well documented. Since the start of the outbreak in December 2019 there is an ever-increasing number of infections worldwide.
SARS-CoV-2 spreads primarily through respiratory droplets and microdroplets generated by the infected person, although aerosol transmission may also occur [1], [2], [3]. Aerosol generating procedures (AGPs) pose the greatest risk of transmission [4]. The use of bronchoscopy is associated with increased risk of patient-to-health care worker (HCW) transmission, due to aerosolised viral particles, which may be inhaled but also results in environmental contamination of surfaces during bronchoscopy [5], [6], [7], [8]. The airways of SARS-CoV-2 infected patients contain a very high viral load [9]. Airway procedures result in the aerosolisation of SARS-CoV-2 placing HCWs at high risk of getting infected [10], [11], [12].
The first SARS-CoV-2 transmission to an HCW was described in January 2020 and the first reported HCW death related to SARS-CoV-2 was an otolaryngologist from Wuhan, China [13].
Limited information is available about the risk for front-line workers during previous viral pandemics. Only one case-control study about the risk for front-line HCWs caring for SARS-CoV-1 patients has been performed and in this study, conducted in China, tracheostomies were performed by HCWs. This study demonstrated with a univariate analysis, that in HCWs working with SARS-CoV-1 patients, 6/85 cases (who had IgG against SARS-CoV-1) versus 11/646 controls (who did not have IgG against SARS-CoV-1) had performed tracheostomies during the epidemic (odds ratio 4.15, 95% CI 2.75, 7.54). In this study the risk during bronchoscopy did not reach significance (pooled OR 1.3, 95% CI 0.5, 14.2) [14].
Thompson et al measured the amount of viral RNA in the air in the vicinity of H1N1 positive patients during bronchoscopy and compared it to controls. They could not demonstrate increased viral RNA during bronchoscopy [15].
It seems that the evidence for bronchoscopy to be listed as an AGP by the World Health Organization is based on a study comparing the rate of tuberculin skin test conversion among pulmonology and infectious diseases fellows graduating in 1983 during a resurgence of tuberculosis in the United States [16], [17].
Suctioning during bronchoscopy creates air currents, which generate droplets as they move across the surface of the liquid lining of the airways. The size of the droplets is inversely proportional to the velocity of the air [16].
The American Association of Bronchology and Interventional Pulmonology (AABIP) released a statement that bronchoscopy has a limited diagnostic role in SARS-CoV-2 infected patients, due to substantial risk of contamination. The collection of upper respiratory samples via nasopharyngeal and oropharyngeal swabs is non-invasive and less contaminating. When patients are intubated, respiratory specimen collection can include tracheal aspirates and non-bronchoscopic bronchoalveolar lavage. The statement highlights that bronchoscopy is not an appropriate tool to diagnose SARS-CoV-2 infection and the benefits are far outweighed by the risks [18].
Bronchoscopy use in SARS-CoV-2 infected adults has reportedly mainly been applied for the removal of airway plugs and the adaption of antimicrobial agents [19], [20], [21].
The role of children in the transmission of SARS-CoV-2 in the community is thought to be limited, yet the risk of transmission to HCWs performing AGPs on SARS-CoV-2 infected children is likely to be significant [22], [23].
Bronchoscopy has a very limited role in the management of SARS-CoV-2 positive children. It has been reported that most children will have mild respiratory disease and do not need ventilation or require bronchoscopy. Xia et al. reported that COVID-19 in children is characterised by mild pneumonia, with only 10% of patients developing tachypnoea. Imaging findings of pneumonic changes were confirmed by chest CT scan as in most children the chest x-ray appearance was normal [24].
Children requiring emergency bronchoscopy procedures may however be SARS-CoV-2 positive, but asymptomatic. In an emergency situation the SARS-CoV-2 test may not be available. These emergency indications for bronchoscopy include foreign body inhalation, tuberculosis with airway obstruction and life threatening congenital or acquired airway obstruction. Only one report of four children infected with SARS-CoV-2 who required bronchoscopy is available in the literature [25]. Both paediatric and some of the adult studies have reported that bronchoscopy can be performed safely with no transmission to HCWs. In contrast, Tomego et al. have reported on a bronchoscopy study in adults on mechanical ventilation where one of the pulmonologists became infected with SARS-CoV-2 [19].
Rigid bronchoscopy should be avoided, due to the increased risk of droplet spread during the SARS-CoV-2 pandemic [26]. Flexible bronchoscopy should be performed first in SARS-CoV-2 positive individuals or in unknown cases, to determine if rigid bronchoscopy is indicated. Foreign bodies often can also be removed with flexible bronchoscopy, but rigid bronchoscopy should be always available. When rigid bronchoscopy is used jet ventilation must be avoided and conventional ventilation must rather be used to reduce the risk of aerosolisation.
Equipment
Single-use flexible bronchoscopes (SUFBs)
Several companies produce single-use bronchoscopes with some of them currently fourth-generation devices that have improved image quality and degrees of angulation, and with a range of diameters available. They are produced with a small, portable, reusable screen that is easy to clean and from which videos or images can easily be saved or downloaded [27]. The efficacy of SUFBs has been studied in an anaesthetic setting [28], [29] and in bronchoalveolar lavage (BAL) [30] in healthy volunteers but has not been studied in complex respiratory procedures.
The risk of transmission of infectious particles to HCWs is reduced with SUFBs, as reprocessing is not necessary and there is less exposure to aerosols and fomites.
Thus, the AABIP has recommended avoiding the use of reusable flexible bronchoscopes in the setting of the SARS-CoV-2 pandemic and using SUFBs instead [18].
Adult studies have reported on SUFBs in SARS-CoV-2 positive patients [21], [27]. Some of these studies reused the SUFBs for the same patient due to cost factors. Currently there are no paediatric studies on the use of SUFBs in SARS-CoV-2 positive children.
The disinfection of reuseable bronchoscopes is done in the usual fashion because the virus is sensitive to most disinfectants used.
Personal protection equipment (PPE)
The SARS-CoV-2 era is a challenging time for respiratory teams to protect their patients and themselves. For SARS-CoV-2 infected or suspected patients undergoing AGPs, the European Resuscitation Council guidelines advocate the use of a long-sleeved waterproof gown; one pair of non-sterile gloves; a filtering facepiece (FFP3 or N99 respirator; FFP2 or N95 if FFP3 not available); and eye and face protection (full-face shield/visor or polycarbonate safety glasses). The gloves should cover the cuff of the gown. Where filtering facepieces are not available, the resuscitation council guidelines recommend use of powered air-purifying respirators (PAPR) with hoods [31]. However, owing to global PPE shortages and supply chain challenges, many hospitals have had to procure alternatives to traditional PPE. The problem with using an N95 mask and face shield is that the vision is not optimal. This limitation is significant during complicated interventions, such as removing foreign bodies and doing endobronchial ultrasound.
PAPRs are expensive and cost about 1400 USD each. They contain a battery-powered high-efficiency particulate air filter that delivers clean air into a hood and blows off exhaled air. The advantages of PAPRs include that the risk of leakage is very small, thus there is no need for a fit test and it can be used by those who fail a fit test, the respirator can be used with beards and no additional eye protection is required (Fig. 1 ) [32].
Fig. 1.

PAPR devices being worn by the bronchoscopy team during the SARS-CoV-2 pandemic.
The SEAC® Libera (SEACSUB, Italy) full face snorkel mask (with adaptor) was fitted with an Intersurgical Clear-Guard filter (Intersurgical, South Africa) and has been adapted by replacing the snorkel with a 3D printed adaptor and the addition of a bacterial/viral filter (Fig. 2 ). This SEAC® PPE mask conforms to the provisions of the EU regulation 425/2016 regarding PPE. These masks are ideal for PPE due to their ability to withstand the intrusion of water. The silicone face skirt is hypoallergenic, and the mask has a low internal volume. The safety of the SEAC® mask is ensured by separate air inlet and outlet channels, thereby decreasing breathing effort, simulating natural breathing and preventing CO2 retention. ETCO2 and ETO2 have shown to be stable in two case studies with only single participants, as well as in similar masks that have been used in endoscopic procedures [33], [34], [35].
Fig. 2.

An adapted Scuba diving mask and full PPE with filter being used during bronchoscopy.
HCWs should receive training in donning, doffing and decontamination of the masks and should ensure a proper fit before using the mask: the crown strap should be on the occiput of the head, with the straps below the pinnae of the ears. To ensure a proper seal, hair, head scarves and scrub caps should remain on the outside of the silicone skirt of the mask. HCWs wearing spectacles need to switch to contact lenses. Verbal communication is mildly impaired, requiring slower, slightly louder and properly articulated speech [36]. The mask seal should be tested using the modified quantitative negative-pressure test prior to entry into the clinical environment [36]. A hand (or two) is placed on the HME filter(s) and a deep breath is taken. If the mask seals adequately, a vacuum is created inside the mask and a sensation of stifling is experienced. Visual check for condensation of the mask’s eye plate should be performed, as this is a warning sign of a leak or CO2 build-up [36].
General measures
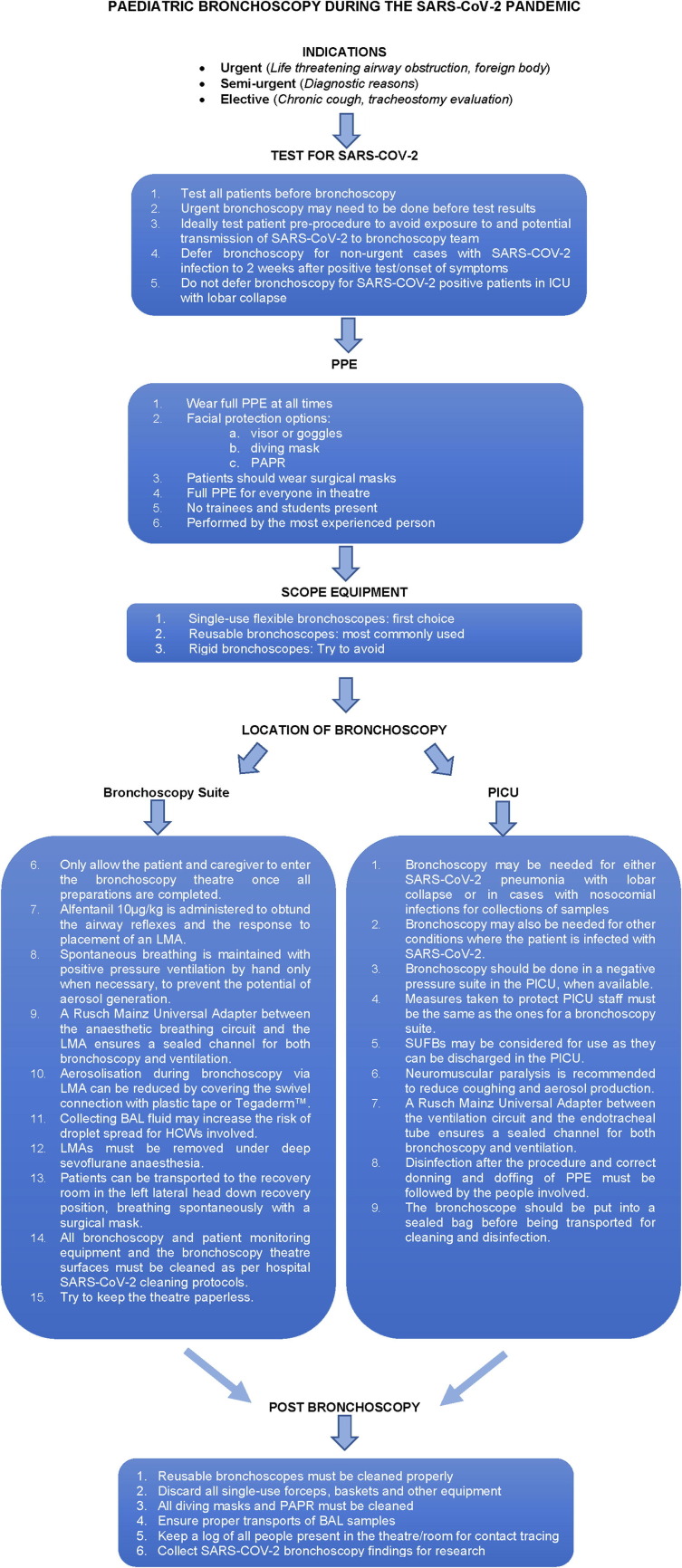
SARS-CoV-2 testing of all children needing bronchoscopy should be done pre-operatively when possible. If results are not available patients must be managed as if potentially SARS-CoV-2 positive, due to the risk of asymptomatic infection. The viral load may also vary during the course of the disease, which may affect the sensitivity of the test. There may be variability in the quality of the sampling, due to difficulty in swabbing the nasopharynx. Patients may even become infected during their hospital admission for the indicated procedure. The patient and the patient’s caregiver must wear a surgical mask when they enter the bronchoscopy theatre. Bronchoscopy should be done in an adequately ventilated room with air flow of at least 160 L/s per patient or in a negative pressure room with at least 12 air changes per hour. This may not be available in many parts of the world. The number of people in the theatre must be limited to essential people, especially during induction of anaesthetics. There should be no trainees and students present and the bronchoscopy should be performed by the most experienced person to reduce the duration of the procedure. Correct donning and doffing of PPE should be done by everybody who is present in the theatre (Fig. 3 ).
Fig. 3.
Flow diagram for paediatric bronchoscopy during the SARS-CoV-2 pandemic.
Anaesthetic techniques for a bronchoscopy suite
Patients and caregivers should only be allowed to enter the bronchoscopy theatre once all preparations are complete, thereby reducing the exposure time for staff. Induction of anaesthesia, on the lap of a caregiver, is completed with sevoflurane gas induction and an FiO2 of 0.5. Standard ASA monitoring is applied, including capnography. Intravenous access is established and alfentanil 10 μg/kg administered to obtund the airway reflexes and the response to placement of a laryngeal mask airway (LMA). The LMA is held in position by the anaesthetist ensuring a tight seal. Spontaneous breathing is maintained with positive pressure ventilation by hand only when necessary, to prevent the potential of aerosol generation. Placement of a Rusch Mainz Universal Adapter between the anaesthetic breathing circuit and the LMA ensures a sealed channel for both bronchoscopy and ventilation while minimising the risk of aerosolisation.
Lignocaine 1% is injected onto the vocal cords via the bronchoscope working channel, to a maximum dose of 7 mg/kg.
Aerosolisation during bronchoscopy via LMA can be reduced by covering the swivel connection with plastic tape or Tegaderm™. Collecting bronchoalveolar lavage fluid may increase the risk of droplet spread for HCWs involved. This should be done with care and only if it will benefit the patient. Adult guidelines suggest reducing the amount of fluid needed for BAL to reduce the risk of aerosolisation [37].
LMAs must be removed under deep sevoflurane anaesthesia. Once stable the spontaneously breathing patients can be transported to the recovery room in the left lateral head down recovery position, wearing a surgical mask. All bronchoscopy and patient monitoring equipment and the bronchoscopy theatre surfaces must be cleaned as per hospital SARS-CoV-2 cleaning protocols. Where possible an attempt must be made to keep the theatre paperless.
Bronchoscopy in a paediatric intensive care unit (PICU)
Bronchoscopy in a PICU may be needed for either SARS-CoV-2 pneumonia with lobar collapse or in cases who develop nosocomial co-infections and require samples to be collected. Bronchoscopy in SARS-CoV-2 positive children might also be indicated for conditions other than the SARS-CoV-2 infection. These may include cardiac comorbidities, tuberculosis, suspected foreign body inhalation and congenital lung anomalies. Bronchoscopy should be done in a negative pressure suite in the PICU, when available. Measures taken to protect PICU staff must be the same as in the bronchoscopy suite. When available SUFBs may be considered for use as the instruments can be discharged in the PICU, compared to a re-useable bronchoscope that needs be to send for cleaning. Neuromuscular paralysis is recommended to reduce coughing and aerosol production, unless contraindicated. The placement of a Rusch Mainz Universal Adapter between the ventilation circuit and the endotracheal tube ensures a sealed channel for both bronchoscopy and ventilation while minimising the risk of aerosolisation. The room used for the bronchoscopy needs to be disinfected after the procedure and correct donning and doffing of PPE must be followed by the medical staff involved. Reusable bronchoscopes should be placed into a sealed bag before being transported for cleaning and disinfection.
Ear, nose and throat (ENT) procedures
Diagnostic endoscopy of the upper airways is listed as an AGP. Due to the high viral loads in the upper aerodigestive tract of SARS-CoV-2 infected patients aerosolised particles would likely contain viral material. The highest viral loads occur in the nasal cavities [38].
Furthermore, coughing may produce even more aerosols. Due to the close proximity of the endoscopist performing outpatient ENT procedures to an infected patient’s mouth and nose they are at increased risk of infection [39], [40].
Most endoscopes used for the upper aerodigestive tract do not require suctioning, which might reduce the risk; however full PPE is indicated, as for bronchoscopy.
Conclusion
Bronchoscopy is a high-risk procedure, with a great potential for direct SARS-CoV-2 exposure and hospital-acquired infection among HCWs during the SARS-CoV-2 pandemic.
Many patients infected with SARS-CoV-2 will be completely asymptomatic. We have long passed the stage in the pandemic where travel history was a reliable indicator of the risk associated with the patient. False negative SARS-CoV-2 test results further complicate the pre-operative risk analysis of each patient. It is thus not unreasonable for HCWs doing bronchoscopy procedures during the SARS-CoV-2 pandemic to implement airborne precautions regardless of the child’s SARS-CoV-2 status. Bronchoscopy can be performed in SARS-CoV-2 positive children if infection prevention protocols are strictly adhered to and HCWs wear appropriate PPE at all times. Modified full face masks are a practical and safe alternative to filtering facepieces for use in bronchoscopy. It is advisable to adhere to this high standard of PPE until the pandemic has become a part of modern day history.
Educational aims
The reader will come to appreciate that:
-
•
Bronchoscopy is a high risk procedure in the COVID-19 pandemic, associated with increased risk of patient to health care worker transmission due to aerosolised viral particles and contamination of surfaces during bronchoscopy.
-
•
Bronchoscopy has a very limited role in the management of SARS-CoV-2 infected children.
-
•
While the role of children in SARS-CoV-2 transmission in the community appears to be limited, the risk of transmission to health care workers performing aerosol generating procedures on SARS-CoV-2 infected children is likely to be significant.
-
•
Rigid bronchoscopy should be avoided, due to the increased risk of droplet spread; when necessary, jet ventilation must be avoided.
-
•
When available single-use flexible bronchoscopes may be considered for use.
Directions for future research
-
•
Assessing the efficacy of single-use flexible bronchoscopes in complex procedures.
-
•
Developing effective prevention methods for future viral pandemics.
Funding
None.
Declarations of interest
None.
References
- 1.Bourouiba L. Turbulent gas clouds and respiratory pathogen emissions: potential implications for reducing transmission of COVID-19. JAMA. 2020;323(18):1837–1838. doi: 10.1001/jama.2020.4756. [DOI] [PubMed] [Google Scholar]
- 2.Asadi S., Bouvier N., Wexler A.S., Ristenpart W.D. The coronavirus pandemic and aerosols: does COVID-19 transmit via expiratory particles? Aerosol Sci Technol. 2020;54(6):635–638. doi: 10.1080/02786826.2020.1749229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stadnytskyi V., Bax C.E., Bax A., Anfinrud P. The airborne lifetime of small speech droplets and their potential importance in SARS-CoV-2 transmission. Proc Natl Acad Sci USA. 2020;117(22):11875–11877. doi: 10.1073/pnas.2006874117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Somsen G.A., van Rijn C., Kooij S., Bem R.A., Bonn D. Small droplet aerosols in poorly ventilated spaces and SARS-CoV-2 transmission. Lancet Respir Med. 2020;8(7):658–659. doi: 10.1016/S2213-2600(20)30245-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.World Health Organization (WHO). Transmission of SARS-CoV-2: implications for infection prevention precautions. 9 July 2020. file:///C:/Users/AEC1/AppData/Local/Temp/WHO-2019-nCoV-Sci_Brief-Transmission_modes-2020.3-eng.pdf.
- 6.Harding H., Broom A., Broom J. Aerosol generating procedures and infective risk to healthcare workers: SARS-CoV-2 - the limits of the evidence. J Hosp Infect. 2020;105(4):717–725. doi: 10.1016/j.jhin.2020.05.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schünemann H.J., Khabsa J., Solo K., Khamis A.M., Brignardello-Petersen R., El-Harakeh A., Darzi A., Hajizadeh A., Bognanni A., Bak A., Izcovich A., Cuello-Garcia C.A., Chen C., Borowiack E., Chamseddine F., Schünemann F., Morgano G.P., Muti-Schünemann G.E.U., Chen G., Zhao H., Neumann I., Brozek J., Schmidt J., Hneiny L., Harrison L., Reinap M., Junek M., Santesso N., El-Khoury R., Thomas R., Nieuwlaat R., Stalteri R., Yaacoub S., Lotfi T., Baldeh T., Piggott T., Zhang Y., Saad Z., Rochwerg B., Perri D., Fan E., Stehling F., Akl I.B., Loeb M., Garner P., Aston S., Alhazzani W., Szczeklik W., Chu D.K., Akl E.A. Ventilation techniques and risk for transmission of coronavirus disease, including COVID-19: a living systematic review of multiple streams of evidence. Ann Intern Med. 2020;173(3):204–216. doi: 10.7326/M20-2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mick P., Murphy R. Aerosol-generating otolaryngology procedures and the need for enhanced PPE during the COVID-19 pandemic: a literature review. J Otolaryngol Head Neck Surg. 2020;49(1) doi: 10.1186/s40463-020-00424-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zou L., Ruan F., Huang M., Liang L., Huang H., Hong Z., Yu J., Kang M., Song Y., Xia J., Guo Q., Song T., He J., Yen H.L., Peiris M., Wu J. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med. 2020;382(12):1177–1179. doi: 10.1056/NEJMc2001737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization (WHO). Modes of transmission of virus causing COVID‐19: implications for IPC precaution recommendations [scientific brief]. https://www.who.int/publications-detail/modes-oftransmission-of-virus-causing-covid-19-implications-foripc-precaution-recommendations; [accessed 21 April 2020].
- 11.Tran K., Cimon K., Severn M., Pessoa-Silva C.L., Conly J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: a systematic review. PLoS One. 2012;7(4) doi: 10.1371/journal.pone.0035797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Frauenfelder C., Butler C., Hartley B., Cochrane L., Jephson C., Nash R., Hewitt R., Albert D., Wyatt M., Hall A. Practical insights for paediatric otolaryngology surgical cases and performing microlaryngobronchoscopy during the COVID-19 pandemic. Int J Pediatr Otorhinolaryngol. 2020;134:110030. doi: 10.1016/j.ijporl.2020.110030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chan J.Y.K., Wong E.W.Y., Lam W. Practical aspects of otolaryngologic clinical services during the 2019 novel coronavirus epidemic: an experience in Hong Kong. JAMA Otolaryngol Head Neck Surg. 2020;146(6):519–520. doi: 10.1001/jamaoto.2020.0488. [DOI] [PubMed] [Google Scholar]
- 14.Mehta AC, Prakash UB, Garland R, Haponik E, Moses L, Schaffner W, Silvestri G. American College of Chest Physicians and American Association for Bronchology [corrected] consensus statement: prevention of flexible bronchoscopy-associated infection. Chest 2005;128(3):1742-55. https://doi: 10.1378/chest.128.3.1742. Erratum in: Chest. 2005 Nov;128(5):3779. PMID: 16162783; PMCID: PMC7094662. [DOI] [PMC free article] [PubMed]
- 15.Thompson KA, Pappachan JV, Bennett AM, Mittal H, Macken S, Dove BK, Nguyen-Van-Tam JS, Copley VR, O'Brien S, Hoffman P, Parks S, Bentley A, Isalska B, Thomson G. EASE Study Consortium. Influenza aerosols in UK hospitals during the H1N1 (2009) pandemic--the risk of aerosol generation during medical procedures. PLoS One 2013;8(2):e56278. https://doi:10.1371/journal.pone.0056278. [DOI] [PMC free article] [PubMed]
- 16.World Health Organization. Infection prevention and control of epidemic- and pandemic-prone acute respiratory diseases in health care; WHO Interim Guidelines, https://www.who.int/csr/resources/publications/WHO_CDS_EPR_2007_6c.pdf?ua=1; Geneva; 2007. [PubMed]
- 17.Malasky C., Jordan T., Potulski F., Reichman L.B. Occupational tuberculous infections among pulmonary physicians in training. Am Rev Respir Dis. 1990;142(3):505–507. doi: 10.1164/ajrccm/142.3.505. [DOI] [PubMed] [Google Scholar]
- 18.Wahidi MM, Lamb C, Murgu S, Musani A, Shojaee S, Sachdeva A, Maldonado F, Mahmood K, Kinsey M, Sethi S, Mahajan A, Majid A, Keyes C, Alraiyes AH, Sung A, Hsia D, Eapen G. American Association for Bronchology and Interventional Pulmonology (AABIP) statement on the use of bronchoscopy and respiratory specimen collection in patients with suspected or confirmed COVID-19 infection. J Bronchol Interv Pulmonol. 2020 27(4):e52-e54. https://doi: 10.1097/LBR.0000000000000681. [DOI] [PMC free article] [PubMed]
- 19.Torrego A., Pajares V., Fernández-Arias C., Vera P., Mancebo J. Bronchoscopy in patients with COVID-19 with invasive mechanical ventilation: a single-center experience. Am J Respir Crit Care Med. 2020;202(2):284–287. doi: 10.1164/rccm.202004-0945LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Su Z.Q., Ye T.S., Chen D.F., Deng X.L., Chen H.J., Li S.Y. The role of bronchoscopy in COVID-19. Respiration. 2020;99(8):697–698. doi: 10.1159/000507402. [DOI] [PubMed] [Google Scholar]
- 21.Bruyneel M., Gabrovska M., Rummens P., Roman A., Claus M., Stevens E., et al. Bronchoscopy in COVID-19 intensive care unit patients. Respirology. 2020;25(12):1313–1315. doi: 10.1111/resp.13932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li X., Xu W., Dozier M., He Y., Kirolos A., Theodoratou E. UNCOVER. The role of children in transmission of SARS-CoV-2: a rapid review. J Glob Health. 2020;10(1):011101. doi: 10.7189/jogh.10.011101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Diercks G.R., Park B.J., Myers L.B., Kwolek C.J. Asymptomatic COVID-19 infection in a child with nasal foreign body. Int J Pediatr Otorhinolaryngol. 2020;135:110092. doi: 10.1016/j.ijporl.2020.110092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xia W., Shao J., Guo Y.u., Peng X., Li Z., Hu D. Clinical and CT features in pediatric patients with COVID‐19 infection: different points from adults. Pediatr Pulmonol. 2020;55(5):1169–1174. doi: 10.1002/ppul.24718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goussard P., Van Wyk L., Burke J., Malherbe A., Retief F., Andronikou S., Mfingwana L., Ruttens D., Van der Zalm M., Dramowski A., Da Costa A., Rabie H. Bronchoscopy in children with COVID‐19: a case series. Pediatr Pulmonol. 2020;55(10):2816–2822. doi: 10.1002/ppul.25015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Francom C.R., Javia L.R., Wolter N.E., Lee G.S., Wine T., Morrissey T., Papsin B.C., Peyton J.M., Matava C.T., Volk M.S., Prager J.D., Propst E.J. Pediatric laryngoscopy and bronchoscopy during the COVID-19 pandemic: A four-center collaborative protocol to improve safety with perioperative management strategies and creation of a surgical tent with disposable drapes. Int J Pediatr Otorhinolaryngol. 2020;134:110059. doi: 10.1016/j.ijporl.2020.110059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barron S.P., Kennedy M.P. Single-use (disposable) flexible bronchoscopes: the future of bronchoscopy? Adv Ther. 2020;37(11):4538–4548. doi: 10.1007/s12325-020-01495-8. Epub 2020 Sep 17. PMID: 32944885; PMCID: PMC7497855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chan J.K., Ng I., Ang J.P., Koh S.M., Lee K., Messavia P., et al. Randomised controlled trial comparing the Ambu aScopeTM2 with a conventional fibreoptic bronchoscope in orotrachealintubation of anaesthetised adult patients. Anaesth Intensive Care. 2015;43(4):479–484. doi: 10.1177/0310057X1504300410. [DOI] [PubMed] [Google Scholar]
- 29.Kristensen M.S., Fredensborg B.B. The disposable Ambu aScope vs. a conventional flexible videoscope for awake intubation - a randomised study: disposable vs. standard flexible scopes. Acta Anaesthesiol Scand. 2013;57(7):888–895. doi: 10.1111/aas.12094. [DOI] [PubMed] [Google Scholar]
- 30.Zaidi S.R., Collins A.M., Mitsi E., Reiné J., Davies K., Wright A.D., Owugha J., Fitzgerald R., Ganguli A., Gordon S.B., Ferreira D.M., Rylance J. Single use and conventional bronchoscopes for Broncho alveolar lavage (BAL) in research: a comparative study ( NCT 02515591) BMC Pulm Med. 2017;17(1) doi: 10.1186/s12890-017-0421-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.European Resuscitation Council COVID‐19 Guidelines. https://erc.edu/sites/5714e77d5e615861f00f7d18/content_entry5ea884fa4c84867335e4d1ff/5ea885f34c84867335e4d20e/files/ERC_covid19_pages.pdf?1588257310; [accessed 24 April 2020]. [DOI] [PMC free article] [PubMed]
- 32.Board on Health Sciences Policy; Institute of Medicine. The Use and Effectiveness of Powered Air Purifying Respirators in Health Care: Workshop Summary. Washington (DC): National Academies Press (US); 2015 May 7. PMID: 25996018. [PubMed]
- 33.Greig P.R., Carvalho E.B., Ramessur S. Safety testing improvised COVID-19 personal protective equipment based on a modified full-face snorkel mask. Anaesthesia. 2020;75:962–977. doi: 10.1111/anae.15085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kechli M.K., Lerman J., Ross M.M. Modifying a full-face snorkel mask to meet N95 respirator standards for use with coronavirus disease 2019 patients. A Practice. 2020;14(7):e01237. doi: 10.1213/XAA.0000000000001237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kusano C., Goddard J., Gotoda T. Experience for use of modified full‐face snorkel mask as personal protective equipment during endoscopic procedures in the era of coronavirus disease pandemic. Dig Endosc. 2020;32(6):1000. doi: 10.1111/den.13784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Germonpre P., Van Rompaey D., Balestra C. Evaluation of protection level, respiratory safety, and practical aspects of commercially available snorkel masks as personal protection devices against aerosolized contaminants and SARS-CoV2. Int J Environ Res Public Health. 2020;17(12):4347. doi: 10.3390/ijerph1712434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Flick H, Arns BM, Bolitschek J, Bucher B, Cima K, Gingrich E, Handzhiev S, Hochmair M, Horak F, Idzko M, Jaksch P, Kovacs G, Kropfmüller R, Lamprecht B, Löffler-Ragg J, Meilinger M, Olschewski H, Pfleger A, Puchner B, Puelacher C, Prior C, Rodriguez P, Salzer H, Schenk P, Schindler O, Stelzmüller I, Strenger V, Täubl H, Urban M, Wagner M, Wimberger F, Zacharasiewicz A, Zwick RH, Eber E.
- 38.Management of patients with SARS-CoV-2 infections and of patients with chronic lung diseases during the COVID-19 pandemic (as of 9 May 2020) : Statement of the Austrian Society of Pneumology (ASP). [DOI] [PMC free article] [PubMed]
- 39.Wien Klin Wochenschr. 2020 Jul;132(13-14):365-386. doi: 10.1007/s00508-020-01691-0. [DOI] [PMC free article] [PubMed]
- 40.World Health Organization. Infection prevention and control during health care when COVID-19 is suspected. Interim guidance, file:///C:/Users/AEC1/OneDrive%20-%20Stellenbosch%20University/Documents/Downloads/WHO-2019-nCoV-IPC-2020.3-eng.pdf; 19 March 2020. Geneva; 2020 [cited 2020 Apr 10]. [accessed 6 May 2020].