Abstract
Focal segmental glomerulosclerosis (FSGS) is a serious condition leading to kidney failure. We aimed to investigate the clinical characteristics of FSGS and its differences compared with minimal change disease (MCD) using cross-sectional data from the Japan Renal Biopsy Registry. In Analysis 1, primary FSGS (n = 996) were stratified by age into three groups: pediatric (< 18 years), adult (18–64 years), and elderly (≥ 65 years), and clinical characteristics were compared. Clinical diagnosis of nephrotic syndrome (NS) was given to 73.5% (97/132) of the pediatric, 41.2% (256/622) of the adult, and 65.7% (159/242) of the elderly group. In Analysis 2, primary FSGS (n = 306) and MCD (n = 1303) whose clinical diagnosis was nephrotic syndrome (NS) and laboratory data were consistent with NS, were enrolled. Logistic regression analysis was conducted to elucidate the variables which can distinguish FSGS from MCD. On multivariable analysis, higher systolic blood pressure, higher serum albumin, lower eGFR, and presence of hematuria associated with FSGS. In Japanese nationwide registry, primary FSGS patients aged 18–64 years showed lower rate of NS than those in other ages. Among primary nephrotic cases, FSGS showed distinct clinical features from MCD.
Subject terms: Medical research, Nephrology
Introduction
Focal segmental glomerulosclerosis (FSGS) presents with proteinuria, often accompanied by nephrotic syndrome (NS), and may lead to end-stage renal disease1. FSGS originally referred to steroid-resistant nephrotic syndrome in pediatric patients with segmental obliteration of glomerular capillaries, with or without hyalinosis on light microscopy2. The entity of FSGS has been expanded and FSGS is currently regarded as a group of kidney diseases sharing common glomerular lesions3. There are two pathophysiological types of FSGS: primary and secondary ones. In primary FSGS, it is postulated that circulating permeability factors cause podocyte injury, inducing NS3,4. Whereas, secondary cases have underlying etiologies such as hypertension, obesity, viruses, drugs, genetic mutations, or adaptive conditions4,5, and they do not always manifest NS. Therefore, the clinical presentations of FSGS are diverse. Cases with NS (usually the primary form) show a different clinical course compared with minimal change disease (MCD) i.e. poor therapeutic response, worse renal prognosis1,6, and rapid recurrence of proteinuria after kidney transplantation7. However, it is difficult to clearly distinguish nephrotic FSGS from MCD because of their similar clinical presentations and the sampling limitations of pathological specimens8,9. To date, few studies have described the clinical features of FSGS, as well as the distinction between nephrotic FSGS and MCD using a large sample size.
The Japan Renal Biopsy Registry (J-RBR) is a nationwide, web-based, registry of renal biopsies established by the Japanese Society of Nephrology in 200710. Cross-sectional studies using this database have described the clinical features of several kidney diseases in Japan11–19.
The present study aimed to clarify the clinical characteristics of FSGS patients in Japan by age stratification of them and comparison with MCD patients through an analysis of data from the J-RBR compiled between July 2007 and June 2016.
Results
Among 30,949 patients who registered to J-RBR between July 2007 and June 2016, 1,410 cases were extracted for the evaluation of FSGS. Additionally, 3656 patients were also extracted for the evaluation of MCD (Fig. 1).
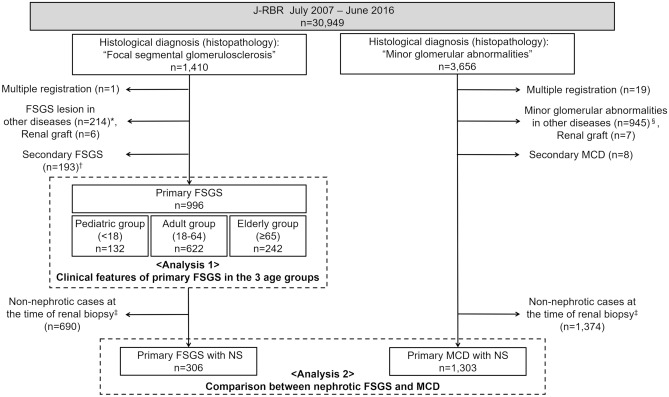
Figure 1.
Flow of patient selection in present study. *The details of distribution of histopathologically diagnosed “focal segmental glomerulosclerosis” including FSGS lesion in other diseases are shown in Supplementary Fig. S1 and Supplementary Table S1. †The details of secondary FSGS are shown in Supplementary Fig. S3. ‡Nephrotic cases at the time of renal biopsy was defined as the cases whose clinical diagnosis was nephrotic syndrome and who fitted the laboratory criteria of nephrotic syndrome. Laboratory criteria for nephrotic syndrome, for pediatric patients (age < 18): urinary protein ≥ 40 mg/h/m2 or ≥ 2.0 g/gCr and serum albumin ≤ 2.5 g/dL; for adult and elderly patients (age ≥ 18): urinary protein ≥ 3.5 g/day or ≥ 3.5 g/gCr and serum albumin ≤ 3.0 g/dL. §The details of distribution of histopathologically diagnosed “minor glomerular abnormalities” including minor glomerular abnormalities in other diseases are shown in Supplementary Fig. S2 and Supplementary Table S2. J-RBR Japan Renal Biopsy Registry, FSGS focal segmental glomerulosclerosis, MCD minimal change disease, NS nephrotic syndrome.
General demographics of FSGS in the J-RBR
After removing a patient with multiple registration, 1409 patients were registered as histologically registered as focal segmental glomerulosclerosis in J-RBR. Of these, 996 were primary FSGS and 193 were secondary FSGS (Supplementary Fig. S1). The annual incidence of FSGS accounted for 3.5–4.5% of all registered cases in the J-RBR database. The percentage of primary FSGS were constant at approximately 3% of all during 2007 to 2016 (Fig. 2). The details of secondary FSGS were summarized in Supplementary Fig. S3.
Figure 2.
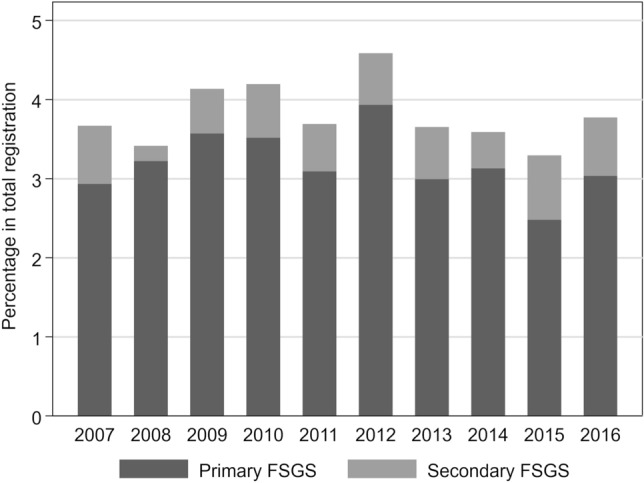
Annual incidence of FSGS in the J-RBR. J-RBR Japan Renal Biopsy Registry, FSGS focal segmental glomerulosclerosis.
Analysis 1: description of clinical features of primary FSGS in the three age groups
For Analysis 1, 996 primary FSGS cases were divided into three age groups: pediatric group (n = 132), adult group (n = 622), and elderly group (n = 242). The detailed age-stratified distribution of the prevalence and proportion of patients with FSGS and MCD are shown in Supplementary Fig. S4. The clinical features of the patients are summarized in Table 1. Patients who underwent biopsy more than twice were the most common in the pediatric group (26.5%). In clinical diagnosis, NS was the most common (51.4%), followed by chronic nephritic syndrome (44.4%) in total. A clinical diagnosis of NS was 73.5% in the pediatric group, 41.2% in the adult group, and 65.7% in the elderly group. In the adult group, prevalence of chronic nephritic syndrome (54.3%) was higher than that of NS. Body mass index was greater and kidney function was worse in the adult and elderly groups than in the pediatric group (P < 0.001). The elderly group showed the highest systolic blood pressure and highest prevalence of concomitant hypertension on antihypertensive drugs (68.8%) and diabetes mellitus (18.8%). Baseline estimated glomerular filtration rate (eGFR) was highest in the pediatric group, followed by the adult group and elderly group (eGFR: 106, 64, and 40 mL/min/1.73 m2, respectively). Fewer patients who were consistent with NS in laboratory data at biopsy were found in the pediatric group and adult group than in the elderly group (24.2%, 27.5%, and 51.2%, respectively).
Table 1.
Clinical features of primary FSGS in the Japan Renal Biopsy Registry.
| Obs | Overall (n = 996) | Age groups | P-value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pediatric group (n = 132) | Adult group (n = 622) | Elderly group (n = 242) | ||||||||
| Patient characteristics | ||||||||||
| Age | 996 | 47 | [89–64] | 10 | [4–15] | 43 | [32–55] | 72 | [68–77] | < 0.001 |
| Sex (male) | 996 | 592 | (59.4) | 80 | (60.6) | 360 | (57.9) | 152 | (62.8) | 0.40 |
| Number of biopsies | ||||||||||
| First | 996 | 516 | (51.8) | 52 | (39.4) | 322 | (51.8) | 142 | (58.7) | < 0.001 |
| Second | 66 | (6.6) | 24 | (18.2) | 33 | (5.3) | 9 | (3.7) | ||
| ≥ 3 times | 26 | (2.6) | 11 | (8.3) | 14 | (2.6) | 1 | (0.4) | ||
| Unknown | 388 | (39.0) | 45 | (34.1) | 253 | (40.7) | 90 | (37.2) | ||
| Clinical diagnosis | ||||||||||
| Nephrotic syndrome | 996 | 512 | (51.4) | 97 | (73.5) | 256 | (41.2) | 159 | (65.7) | < 0.001 |
| Chronic nephritic syndrome | 442 | (44.4) | 31 | (25.5) | 338 | (54.3) | 73 | (30.2) | ||
| Others | 42 | (4.2) | 4 | (3.0) | 28 | (4.5) | 10 | (4.1) | ||
| Body Mass Index | 982 | 22.9 | [20.3–26.2] | 18.5 | [16.3–21.5] | 23.5 | [20.8–27.1] | 23.6 | [21.4–25.9] | < 0.001 |
| Systolic blood pressure | 828 | 129 | [118–142] | 113 | [103–121] | 129 | [119–141] | 138 | [125–150] | < 0.001 |
| Diastolic blood pressure | 828 | 78 | [68–86] | 68 | [60–78] | 80 | [70–88] | 77 | [70–86] | < 0.001 |
| Antihypertensive drugs | 817 | 414 | (50.7) | 31 | (34.1) | 244 | (46.6) | 139 | (68.8) | < 0.001 |
| Diabetes mellitus | 717 | 99 | (13.8) | 4 | (4.8) | 61 | (13.5) | 34 | (18.8) | 0.013 |
| Laboratory data | ||||||||||
| Total protein, g/dL | 988 | 6.2 | [4.9–7.0] | 6.1 | [5.0–6.9] | 6.5 | [5.0–7.0] | 5.5 | [4.8–6.6] | < 0.001 |
| Albumin, g/dL | 986 | 3.5 | [2.2–4.1] | 3.6 | [2.6–4.2] | 3.7 | [2.3–4.1] | 2.6 | [2.0–3.75 | < 0.001 |
| Total cholesterol, mg/dL | 975 | 235 | [195–323] | 250 | [177–399] | 232 | [195–308] | 242 | [195–325] | 0.53 |
| HbA1c (NGSP), % | 608 | 5.7 | [5.4–6.0] | 5.5 | [5.3–5.8] | 5.6 | [5.4–6.0] | 5.8 | [5.4–6.0] | 0.043 |
| Creatinine, mg/dL | 994 | 0.96 | [0.70–1.31] | 0.44 | [0.31–0.66] | 0.95 | [0.73–1.26] | 1.24 | [0.93–1.69] | < 0.001 |
| eGFR, mL/min/1.73 m2 | 987 | 60 | [41–84] | 106 | [83–134] | 64 | [47–83] | 40 | [29–57] | < 0.001 |
| Urinary protein, g/gCr | 697 | 2.9 | [1.02–6.93] | 1.86 | [0.16–6.16] | 2.27 | [0.98–5.46] | 5.66 | [2.83–9.70] | < 0.001 |
| Urinary protein, g/day | 680 | 2.24 | [0.84–5.49] | 1.65 | [0.17–6.80] | 1.96 | [0.84–5.01] | 3.68 | [1.24–6.44] | < 0.001 |
| Consistent with NSa | 996 | 327 | (32.8) | 32 | (24.2) | 171 | (27.5) | 124 | (51.2) | < 0.001 |
| Urinary occult blood | ||||||||||
| (−) | 996 | 323 | (32.4) | 70 | (53.0) | 205 | (33.0) | 48 | (19.8) | < 0.001 |
| (±) | 143 | (14.4) | 14 | (10.6) | 94 | (15.1 | 35 | (14.5) | ||
| (1+) | 170 | (17.1) | 10 | (7.6) | 100 | (16.1 | 60 | (24.8) | ||
| (2+) | 208 | (20.9) | 20 | (15.2) | 129 | (20.7) | 59 | (24.4) | ||
| (3+) | 152 | (15.3) | 18 | (13.6) | 94 | (15.1) | 40 | (16.5) | ||
| Urinary occult blood presentb | 996 | 530 | (53.2) | 48 | (36.4) | 323 | (51.9) | 159 | (65.7) | < 0.001 |
| Urinary RBC/HFP | ||||||||||
| (−) | 996 | 154 | (15.5) | 32 | (24.2 | 94 | (15.1) | 28 | (11.6) | 0.025 |
| < 5 | 448 | (45.0) | 49 | (37.1) | 287 | (46.1) | 112 | (46.3) | ||
| 5–10 | 163 | (16.4) | 18 | (13.6) | 97 | (15.6) | 48 | (19.8) | ||
| 10–30 | 129 | (13.0) | 16 | (12.1) | 77 | (12.4) | 36 | (14.9) | ||
| Many | 102 | (10.2) | 17 | (12.9) | 67 | (10.8) | 18 | (7.4) | ||
| Urinary RBC presentc | 996 | 394 | (39.6) | 51 | (38.6) | 241 | (38.8) | 102 | (42.2) | 0.64 |
| Hematuria presentd | 996 | 363 | (36.5) | 41 | (31.1) | 226 | (36.3) | 96 | (39.7) | 0.26 |
Data are presented as median [interquartile range] for continuous variables and count (percentage) for categorical variables.
obs number of observations, eGFR estimated glomerular filtration rate, NGSP National Glycohemoglobin Standardization Program, NS nephrotic syndrome, RBC red blood cell, HPF high powered field.
aLaboratory criteria for nephrotic syndrome, for pediatric patients (age < 18): urinary protein ≥ 40 mg/h/m2 or ≥ 2.0 g/gCr and serum albumin ≤ 2.5 g/dL; for adult and elderly patients (age ≥ 18): urinary protein ≥ 3.5 g/day or ≥ 3.5 g/gCr and serum albumin ≤ 3.0 g/dL.
bUrinary occult blood present, (1+), (2+), (3+) on dipstick.
cUrinary RBC present, ≥ 5/HPF in urine sediment.
dHematuria present, (1+), (2+), (3+) on dipstick and ≥ 5/HPF in sediment.
Analysis 2: comparison between nephrotic FSGS and MCD
Of 1410 patients with “focal segmental glomerulosclerosis” and 3656 with “minor glomerular abnormalities” on histological diagnosis (histopathology), 306 with nephrotic FSGS and 1303 with MCD were included in Analysis 2 (Fig. 1). The characteristics of nephrotic FSGS and MCD at the biopsy are summarized in Table 2. FSGS cases were older (FSGS: 58 vs. MCD: 44 years), had a higher prevalence of hypertension on antihypertensive drugs (56.2 vs. 28.7%), and lower eGFR (53 vs. 72 mL/min/1.73 m2). FSGS patients also had higher serum albumin (1.9 vs. 1.7 g/dL), lower daily urinary protein levels (6.28 vs. 7.00 g/day) and higher prevalence of hematuria (52.9 vs. 29.8%) at biopsy.
Table 2.
Comparison between nephrotic FSGS and MCD cases.
| Nephrotic FSGS | MCD | P-value | |||||
|---|---|---|---|---|---|---|---|
| (n = 306) | (n = 1303) | ||||||
| Obs | Median, n | [IQR], (%) | Obs | Median, n | [IQR], (%) | ||
| Patient characteristics | |||||||
| Age | 306 | 58 | [34–71] | 1303 | 44 | [26–66] | < 0.001 |
| Sex (male) | 306 | 184 | (60.1) | 1303 | 749 | (57.5) | 0.40 |
| Body Mass Index | 305 | 22.9 | [20.5–26.1] | 1296 | 23.4 | [20.8–26.2] | 0.41 |
| Systolic blood pressure | 265 | 136 | [121–148] | 1125 | 123 | [111–135] | < 0.001 |
| Diastolic blood pressure | 265 | 80 | [70–90] | 1125 | 74 | [66–83] | < 0.001 |
| Antihypertensive drugs | 242 | 136 | (56.2) | 1087 | 312 | (28.7) | < 0.001 |
| Diabetes mellitus | 215 | 33 | (15.4) | 971 | 131 | (13.5) | 0.48 |
| Laboratory data | |||||||
| Total protein, g/dL | 304 | 4.7 | [4.1–5.3] | 1299 | 4.5 | [4.0–5.0] | < 0.001 |
| Albumin, g/dL | 306 | 1.9 | [1.5–2.4] | 1303 | 1.7 | [1.3–2.1] | < 0.001 |
| Total cholesterol, mg/dL | 302 | 348 | [275–430] | 1297 | 411 | [328–499] | < 0.001 |
| HbA1c (NGSP), % | 204 | 5.6 | [5.3–6.0] | 887 | 5.6 | [5.4–6.0] | 0.32 |
| Creatinine, mg/dL | 304 | 1.03 | [0.75–1.64] | 1302 | 0.85 | [0.64–1.11] | < 0.001 |
| eGFR, mL/min/1.73 m2 | 302 | 53 | [32–73] | 1289 | 72 | [51–90] | < 0.001 |
| Urinary protein, g/gCr | 243 | 8.17 | [5.50–11.00] | 1042 | 8.47 | [5.50–11.92] | 0.24 |
| Urinary protein, g/day | 232 | 6.28 | [4.54–8.87] | 983 | 7.00 | [4.75–10.20] | 0.015 |
| Urinary occult blood | |||||||
| (−) | 306 | 27 | (8.8) | 1303 | 258 | (19.8) | < 0.001 |
| (±) | 40 | (13.1) | 221 | (17.0) | |||
| (1+) | 82 | (26.8) | 272 | (20.9) | |||
| (2+) | 104 | (34.0) | 403 | (30.9) | |||
| (3+) | 53 | (17.3) | 149 | (11.4) | |||
| Urinary occult blood present* | 306 | 239 | (78.1) | 1303 | 824 | (63.2) | < 0.001 |
| Urinary RBC/HPF | |||||||
| (−) | 306 | 23 | (7.5) | 1303 | 201 | (15.4) | < 0.001 |
| < 5 | 114 | (37.3) | 688 | (52.8) | |||
| 5–10 | 81 | (26.5) | 214 | (16.4) | |||
| 10–30 | 56 | (18.3) | 142 | (10.9) | |||
| Many | 32 | (10.5) | 58 | (4.5) | |||
| Urinary RBC presenta | 306 | 169 | (55.2) | 1303 | 414 | (31.8) | < 0.001 |
| Hematuria presenta | 306 | 162 | (52.9) | 1303 | 388 | (29.8) | < 0.001 |
FSGS focal segmental glomerulosclerosis, MCD minimal change disease, eGFR estimated glomerular filtration rate, RBC red blood cell, HPF high powered field.
aDefinition: Urinary occult blood present, (1+), (2+), (3+) on dipstick; Urinary RBC present, ≥ 5/HPF in urine sediment; Hematuria present, (1+), (2+), (3+) on dipstick and ≥ 5/HPF in sediment.
As summarized in Table 3, univariate logistic regression analysis revealed that age, systolic blood pressure, serum albumin, serum total cholesterol, serum creatinine (log-transformation), eGFR and presence of urinary red blood cells and hematuria were significantly associated with a diagnosis of FSGS. The correlations between continuous variables are shown in Supplementary Table S3. Patient age, systolic blood pressure, serum albumin, eGFR, daily urinary protein level (log-transformation), and presence of hematuria were used in the multivariate model (Table 3). Because of the strong correlation between patient age and eGFR, we evaluated two models that took one of them: Model 1 and Model 2. In Model 1, FSGS was associated with higher systolic blood pressure (OR 1.25, 95% confidence interval [CI] 1.14–1.36, for every increase of 10 mmHg), higher serum albumin (OR 2.13, 95% CI 1.56–2.91), lower eGFR (OR 0.90, 95% CI 0.85–0.95, every increase of 10 mL/min/1.73m2), and presence of hematuria (OR 1.92, 95% CI 1.36–2.69). In Model 2, higher systolic blood pressure (OR 1.31, 95% CI 1.19–1.44, for every increase of 10 mmHg), higher serum albumin (OR 2.02, 95% CI 1.49–2.73), and presence of hematuria (OR 2.18, 95% CI 1.56–3.04).
Table 3.
Associating factors with FSGS vs. MCD.
| Variables | Univariate | Multivariable | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Model 1b | Model 2c | ||||||||
| OR | [95% CI] | P-value | OR | [95% CI] | P-value | OR | [95% CI] | P-value | |
| Age (every 10 years) | 1.13 | [1.07–1.20] | < 0.001 | 0.98 | [0.90–1.07] | 0.72 | |||
| Sex (male) | 1.12 | [0.87–1.44] | 0.40 | ||||||
| Body Mass Index | 1.00 | [0.98–1.03] | 0.82 | ||||||
| Systolic BP (every 10 mmHg) | 1.38 | [1.28–1.48] | < 0.001 | 1.25 | [1.14–1.36] | < 0.001 | 1.31 | [1.19–1.44] | < 0.001 |
| Anti-hypertensive drugs | 3.19 | [2.39–4.24] | < 0.001 | ||||||
| Diabetes mellitus | 1.16 | [0.77–1.76] | 0.48 | ||||||
| HbA1c (NGSP) | 0.86 | [0.68–1.09] | 0.21 | ||||||
| Total protein | 1.11 | [0.99–1.22] | 0.066 | ||||||
| Albumin | 2.24 | [1.79–2.82] | < 0.001 | 2.13 | [1.56–2.91] | < 0.001 | 2.02 | [1.49–2.73] | < 0.001 |
| Total cholesterol (every 10 mg/dL) | 0.96 | [0.95–0.97] | < 0.001 | ||||||
| Log_creatinine | 1.96 | [1.59–2.41] | < 0.001 | ||||||
| eGFR (every 10 mL/min/1.73 m2) | 0.86 | [0.83–0.90] | < 0.001 | 0.90 | [0.85–0.95] | < 0.001 | |||
| Log_urinary protein (g/gCr) | 0.89 | [0.72–1.11] | 0.31 | ||||||
| Log_urinary protein (g/day) | 0.81 | [0.65–1.01] | 0.061 | 0.78 | [0.59–1.04] | 0.093 | 0.81 | [0.61–1.08] | 0.146 |
| urinary occult blood presenta | 2.07 | [1.55–2.78] | < 0.001 | ||||||
| urinary RBC presenta | 2.65 | [2.06–3.41] | < 0.001 | ||||||
| Hematuria presenta | 2.65 | [2.06–3.42] | < 0.001 | 1.92 | [1.36–2.69] | < 0.001 | 2.18 | [1.56–3.04] | < 0.001 |
FSGS focal segmental glomerulosclerosis, OR odds ratio, CI confidence interval, BP blood pressure, eGFR estimated glomerular filtration rate, RBC red blood cell, HPF high powered field.
aDefinition: Urinary occult blood present, (1+), (2+), (3+) on dipstick; Urinary RBC present, ≥ 5/HPF in urine sediment; Hematuria present, (1+), (2+), (3+) on dipstick and ≥ 5/HPF in sediment.
bMultivariable model 1: Adjusted for systolic blood pressure, albumin, eGFR, daily urinary protein (log-transformed), hematuria.
cMultivariable model 2: Adjusted for age, systolic blood pressure, albumin, daily urinary protein (log-transformed), hematuria.
Discussion
The present study used the data from a Japanese nationwide kidney biopsy registry. This is a unique study focusing on the clinical manifestations of FSGS with a large sample size. Furthermore, this study is the first one to describe the clinical features of primary FSGS from two different viewpoints: age stratification and comparison between nephrotic FSGS and MCD.
In the J-RBR database, FSGS occupied 3.4–4.5% in total number of kidney biopsy. The Research Group on Progressive Renal Disease from the Ministry of Health, Labor and Welfare of Japan reported that the annual number of native kidney biopsy in Japan was estimated 18,000 to 21,00020. Therefore, approximately 800 patients per year were inferred to be diagnosed with FSGS by biopsy in our country. The prevalence of FSGS varied among races and countries21. An international survey revealed that FSGS accounted for 19.1% of primary glomerular disease in North America, 14.9% in Europe, 6.9% in Asia, and 15.8% in Latin America22. A part of this epidemiological difference could be explained by the presence of genetic variants in apolipoprotein L1 (APOL1) among people with sub-Saharan ancestry23. Additionally, several studies reported that the incidence of FSGS was increasing, especially in the US24–26. The present study shows that incidence of FSGS in Japan was lower than that in reports from other regions21 and it had not changed for the past decade.
To evaluate the population with FSGS, it is necessary to distinguish primary FSGS from secondary FSGS27. However, distinguishing secondary FSGS is challenging and the incidence of secondary FSGS remains unclear. D’Agati reported that 10–20% of FSGS patients are secondary ones3. In J-RBR, secondary cases constituted 16.2% of the total FSGS patients, and our results showed that the etiologies in these patients were different among the age groups; 10.8%, 17.4% and 16.0% among the pediatric, adult and elderly group, respectively. As the etiology, hypertension and obesity were the most common in total or among the adult and elderly group. Unilateral kidney or renal dysplasia, low birth weight and genetic disorders were leading causes of FSGS among the pediatric group (Supplementary Fig. S3).
Analysis 1 described the clinical characteristics of primary FSGS in three age groups. We showed that the proportion of NS was different in each age group. Previous studies reported that nephrotic-range proteinuria is more frequently seen in children than in adults in FSGS8. In our study, the pediatric group showed the highest prevalence of NS (73.5%). However, the urinary protein level was the lowest in this group. This discrepancy between clinical diagnosis and laboratory data could be explained by the effect of immunosuppressive treatment. Because of the quite high occupancy of MCD in NS of pediatric age, children with NS typically receive empiric treatment centering glucocorticoids and the indication of kidney biopsy are limited to the cases with refractory clinical course i.e., steroid-resistant or frequent relapse. Therefore, it is possible that most pediatric FSGS patients had already received immunosuppressive treatment by the time of biopsy. However, we cannot distinguish whether the data was obtained before or after initiation of immunosuppressive treatment, in the J-RBR data. The adult group showed the lowest prevalence of NS (41.2%) and the highest prevalence of chronic nephritic syndrome (54.3%). Whereas, the elderly group showed a higher prevalence of NS (65.7%) than the adult group. This suggested the differences in indications for renal biopsy among age groups. In Japan, a nationwide health examination program for all community residents, including urinalysis screening, has been conducted for over 40 years28. This regular screening enables early detection of urine abnormalities and early referral to a nephrologist. The suggested indication for renal biopsy in adult patients in Japan is ≥ 0.5 g/day of proteinuria or any amount of proteinuria with hematuria29. Many patients in the adult group may have undergone biopsy based on these suggested indications. Therefore, it is likely that the non-nephrotic cases with a substantial number of secondary FSGS were included to this age group. In contrast, it was also possible that the threshold of indications for renal biopsy in elderly patients might be higher i.e., clinicians attempted to perform biopsy for NS patients but not for the non-nephrotic patients in this age group. The clinical features of overall FSGS patients including secondary FSGS in the J-RBR and their age-group comparison were shown in Supplementary Table S4.
Analysis 2 compared the clinical features of nephrotic FSGS and MCD at biopsy. In this analysis, nephrotic FSGS was considered as primary FSGS because their laboratory data (nephrotic proteinuria with hypoalbuminemia) were consistent with NS5,30 and secondary cases with identifiable etiologies had been excluded. Even though classic articles suggested that hypertension, renal insufficiency and hematuria at the onset of disease are helpful to distinguish FSGS from MCD1, none have ever been statistically evaluated for their clinical significance in FSGS with a large sample size. We found that higher blood pressure, higher serum albumin, and incidence of hematuria at biopsy were associated with nephrotic FSGS in two multivariable models. FSGS was associated with older age and lower eGFR in univariate analysis. However, association between higher age and FSGS was not observed in multivariable Model 2, which suggested that there was confounding between age and eGFR. As described above, pediatric patients could represent biased population with the necessity of single or repeat biopsy for steroid-resistant or frequent relapse NS. We conducted sub-analysis only including the patients aged ≥ 18 years (nephrotic FSGS: n = 277, MCD: n = 1135) and obtained similar results as original analysis (Supplementary Tables S5, S6). These results suggest that lower eGFR but not higher age was an independent factor discriminating primary FSGS from MCD.
This study has several limitations. First, the J-RBR system did not collect detailed information regarding the etiology of FSGS. All the information was provided by the local coinvestigators. The J-RBR also lacked the information of the findings in electron microscopy and detailed genetic testing that could help discriminating primary and secondary FSGS. It is possible that primary FSGS may include some secondary cases and, therefore, the percentage of secondary FSGS may be underestimated in our study. Second, the J-RBR did not collect the information about total number of glomeruli. Therefore, it was possible that we included the patients without adequate number of glomeruli to distinguish FSGS from MCD in the present study. Third, this study was based on cross-sectional data. Although eGFR was lower in FSGS than in MCD, it was difficult to determine whether the impaired kidney function indicated chronic kidney disease or acute kidney injury. Fourth, as mentioned above, the J-RBR data do not include information on whether the data were obtained before or after the initiation of immunosuppressive treatment. Modification of the J-RBR system has been conducted to achieve a more accurate description of pathological diagnoses, including etiological information31. Another limitation of this study is the lack of information regarding pathological subgroups of FSGS: variants of the Columbia classification27. Previous studies reported that morphologic variants of FSGS demonstrate distinct features in their clinical presentation and prognosis32–37. A longitudinal investigation is currently underway based on J-RBR database including the additional information regarding Columbia classification.
In conclusion, nation-wide registry system revealed the characteristics and clinical features of FSGS in Japan. The results show that the incidence of FSGS in Japan is lower than that in other countries. When primary FSGS patients were divided into three age groups, the incidence of NS was lowest in the adult group (18–64 years), suggesting that the indications for renal biopsy might have been stricter among pediatric and elderly patients. When the analysis was focused on primary NS at the time of renal biopsy, FSGS showed distinct clinical features, such as younger age, higher blood pressure, higher serum albumin, lower eGFR, lower daily urinary protein level and presence of hematuria compared with MCD.
Methods
Overview of the J-RBR system
This cross-sectional study used the data from the J-RBR. The details of the J-RBR system were described in a previous publication10. As of June 2016, 143 nephrology centers participated in this registry, which included 30,949 patients. The J-RBR enters patient clinical information at biopsy. The J-RBR diagnosis consists of three components: (i) a clinical diagnosis, (ii) a histological diagnosis by pathogenesis, and (iii) a histological diagnosis by histopathology.
Patients
Among the patients who registered to J-RBR between July 2007 and June 2016, cases whose histological diagnosis (histopathology) was “focal segmental glomerulosclerosis” were extracted for the evaluation of FSGS. Additionally, patients whose histological diagnosis (histopathology) was “minor glomerular abnormalities” were also extracted for the evaluation of MCD (Fig. 1).
General demographics of FSGS in the J-RBR
Primary FSGS and secondary FSGS in the J-RBR database were included. From the patients with histological diagnosis (histopathology) of “focal segmental glomerulosclerosis” without multiple registration (n = 1409), the patients whose histological diagnosis (pathogenesis) was “primary glomerular disease” were defined as primary FSGS unless they had information of etiologies. The patients with identified information of their etiologies (i.e. obesity, hypertension, etc.) were defined as secondary FSGS. For example, when hypertension was considered to be a cause of FSGS, the case was classified into secondary FSGS. When hypertension was considered as just a concomitant condition, the case was classified into primary FSGS. FSGS lesions in other diseases (i.e. diabetic nephropathy, lupus nephritis, etc.) and renal graft were excluded. The details of selection for FSGS patients were described in Supplementary Fig. S1 and Supplementary Table S1.
The annual incidence of FSGS and proportion of primary and secondary FSGS and were described. Then, two main analyses were conducted as follows.
Analysis 1: description of clinical features of primary FSGS in the three age groups
The cases with primary FSGS were divided into three age groups: pediatric group (< 18 years), adult group (18–64 years), and elderly group (≥ 65 years) (Fig. 1). Clinical parameters were compared among the age groups.
Analysis 2: comparison between nephrotic FSGS and MCD
To compare the clinical characteristics of nephrotic FSGS with those of MCD, patients who fulfilled the following criteria were included in Analysis 2 (Fig. 1).
Nephrotic FSGS
(i) primary cases, (ii) clinical diagnosis of NS, and (iii) laboratory data consistent with NS, which was defined as urinary protein ≥ 40 mg/h/m2 or ≥ 2.0 g/gCr and serum albumin ≤ 2.5 g/dL for pediatric patients (age < 18) and urinary protein ≥ 3.5 g/day or ≥ 3.5 g/gCr and serum albumin ≤ 3.0 g/dL for adult and elderly patients (age ≥ 18). The body surface area (m2) of pediatric patients was calculated by the DuBois formula: height (cm)0.725 × body weight (kg)0.425 × 0.007184.
Nephrotic MCD
From the patients with histological diagnosis (histopathology) of “minor glomerular abnormalities”, following patients were included: (i) primary MCD, (ii) clinical diagnosis of NS, and (iii) laboratory data consistent with NS. Patients with multiple registrations, secondary MCD, other diseases with no obvious light microscopic glomerular findings (i.e., thin basement membrane disease, Class I lupus nephritis, etc.), and renal graft were excluded. The details of selection for MCD patients were described in Supplementary Fig. S2 and Supplementary Table S2.
Clinical parameters were compared between nephrotic FSGS and MCD patients.
Data collection
Clinical data at biopsy such as patient characteristics (age, sex, height, body weight, blood pressure), comorbidities (concomitant hypertension and diabetes), urinary findings (urinalysis, daily proteinuria), and blood test findings (serum creatinine, total protein, albumin, total cholesterol) were extracted from the J-RBR database.
eGFR was calculated using equations based on serum creatinine (sCr) level for Japanese children (age < 18)38: eGFR [mL/min/1.73 m2] = 110.2 × reference-sCr/sCr (mg/dL) + 2.93, reference-sCr = − 1.259 × Height (m)5 + 7.815 × Height (m)4 − 18.57 × Height (m)3 + 21.9 × Height (m)2 − 11.71 × Height (m) + 2.628 (if male), reference sCr = − 4.536 × Height (m)5 + 27.16 × Height (m)4 − 63.47 × Height (m)3 + 72.43 × Height (m)2 − 40.06 × Height (m) + 8.778 (if female) and adults (age ≥ 18)39: eGFR [mL/min/1.73 m2] = 194 × sCr (mg/dL)−1.094 × Age−0.287 (if male), eGFR [mL/min/1.73 m2] = 194 × sCr (mg/dL)−1.094 × Age−0.287 × 0.739 (if female). Additional diagnostic information, including details of etiology were also extracted.
Statistics
Analysis 1: description of clinical features of primary FSGS in the three age groups
Clinical characteristics were expressed as median/interquartile range and frequency number/percentage. We used the Kruskal–Wallis test to compare continuous variables and the chi-squared test to compare the proportions of categorical variables among the age groups.
Analysis 2: comparison between nephrotic FSGS and MCD
We used the Mann–Whitney U test to compare continuous variables and the chi-squared test to compare the proportions of categorical variables between nephrotic FSGS and MCD. Logistic regression analysis was used to evaluate the association between each variable and diagnosis of FSGS compared to MCD. Variables with P < 0.05 in univariate analysis and variables with clinical importance (age, eGFR, and urinary protein level) were included in the multivariable model, while we excluded variables with strong correlations (Pearson’s correlation coefficients > 0.40) or clinical relevance.
Statistical analyses were conducted using STATA IC version 14.0 (StataCorp LLC, College Station, TX, USA). The statistical significance level was set at P < 0.05.
Ethics
Written informed consent was obtained from all participants and/or their legal guardians. All procedures performed in the present study were in accordance with the standards of the ethics committee of the Japanese Society of Nephrology (approval number: 40, J-RBR201604) and the ethics committee of Nagoya University (approval number: 2016-0492-2), and with the Helsinki Declaration of 1975 and its later amendments. J-RBR is registered in the UMIN Clinical Trial Registry (UMIN000000618).
Supplementary Information
Acknowledgements
The authors are grateful to all the colleagues who collected the data for the J-RBR in participated facilities (Supplementary Note). This study was supported in part by a Grant-in-Aid for Intractable Renal Diseases Research, Research on Rare and Intractable Diseases, and Health and Labour Sciences Research Grants from the Ministry of Health, Labour, and Welfare of Japan (H29-nanchi-ippan-017); Japan Agency for Medical Research and Development (18ek0109354h0001).
Author contributions
All authors contributed to the conception and design of the work; H.Y., H.Sugiyama, H.Sato contributed to data extraction and supervision of the project; T.O. performed statistical analysis; All authors contributed to the interpretation of the results; T.O. and S.M. wrote the main manuscript and all authors reviewed and revised it; all authors approved the final version of the manuscript.
Data availability
The datasets generated and analyzed during the current study are not publicly available because the consent obtained from the participants does not cover unlimited public sharing of the data but are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-020-80931-9.
References
- 1.Korbet SM. Clinical picture and outcome of primary focal segmental glomerulosclerosis. Nephrol. Dial Transplant. 1999;14:68–73. doi: 10.1093/ndt/14.suppl_3.68. [DOI] [PubMed] [Google Scholar]
- 2.Churg J, Habib R, White RR. Pathology of the nephrotic syndrome in children. Lancet. 1970;295:1299–1302. doi: 10.1016/S0140-6736(70)91905-7. [DOI] [PubMed] [Google Scholar]
- 3.D’Agati VD, Kaskel FJ, Falk RJ. Focal segmental glomerulosclerosis. N. Engl. J. Med. 2011;365:2398–2411. doi: 10.1056/NEJMra1106556. [DOI] [PubMed] [Google Scholar]
- 4.De Vriese AS, Sethi S, Nath KA, Glassock RJ, Fervenza FC. Differentiating primary, genetic, and secondary FSGS in adults: A clinicopathologic approach. J. Am. Soc. Nephrol. 2018;29:759–774. doi: 10.1681/ASN.2017090958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.D’Agati VD. The many masks of focal segmental glomerulosclerosis. Kidney Int. 1994;46:1223–1241. doi: 10.1038/ki.1994.388. [DOI] [PubMed] [Google Scholar]
- 6.Gipson DS, et al. Complete remission in the nephrotic syndrome study network. Clin. J. Am. Soc. Nephrol. 2016;11:81–89. doi: 10.2215/CJN.02560315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ponticelli C. Recurrence of focal segmental glomerular sclerosis (FSGS) after renal transplantation. Nephrol. Dial Transplant. 2010;25:25–31. doi: 10.1093/ndt/gfp538. [DOI] [PubMed] [Google Scholar]
- 8.Bose B, Cattran D. Glomerular diseases: FSGS. Clin. J. Am. Soc. Nephrol. 2014;9:626–632. doi: 10.2215/CJN.05810513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Corwin HL, Schwartz MM, Lewis EJ. The importance of sample size in the interpretation of the renal biopsy. Am. J. Nephrol. 1988;8:85–89. doi: 10.1159/000167563. [DOI] [PubMed] [Google Scholar]
- 10.Sugiyama H, et al. Japan Renal Biopsy registry: The first nationwide, web-based, and prospective registry system of renal biopsies in Japan. Clin. Exp. Nephrol. 2011;15:493–503. doi: 10.1007/s10157-011-0430-4. [DOI] [PubMed] [Google Scholar]
- 11.Yokoyama H, Taguchi T, Sugiyama H, Sato H. Membranous nephropathy in Japan: Analysis of the Japan Renal Biopsy Registry (J-RBR) Clin. Exp. Nephrol. 2012;16:557–563. doi: 10.1007/s10157-012-0593-7. [DOI] [PubMed] [Google Scholar]
- 12.Komatsu H, et al. Clinical manifestations of Henoch-Schönlein purpura nephritis and IgA nephropathy: Comparative analysis of data from the Japan Renal Biopsy Registry (J-RBR) Clin. Exp. Nephrol. 2016;20:552–560. doi: 10.1007/s10157-015-1177-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yokoyama H, Narita I, Sugiyama H, Nagata M. Drug-induced kidney disease: A study of the Japan Renal Biopsy Registry from 2007 to 2015. Clin. Exp. Nephrol. 2015;20:720–730. doi: 10.1007/s10157-015-1201-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hiromura K, et al. Clinical and histological features of lupus nephritis in Japan: A cross-sectional analysis of the Japan Renal Biopsy Registry (J-RBR) Nephrology (Carlton) 2017;22:885–891. doi: 10.1111/nep.12863. [DOI] [PubMed] [Google Scholar]
- 15.Nishi S, et al. A clinical evaluation of renal amyloidosis in the Japan renal biopsy registry: A cross-sectional study. Clin. Exp. Nephrol. 2017;21:624–632. doi: 10.1007/s10157-016-1326-0. [DOI] [PubMed] [Google Scholar]
- 16.Ichikawa K, Konta T, Sato H, Ueda Y, Yokoyama H. The clinical and pathological characteristics of nephropathies in connective tissue diseases in the Japan Renal Biopsy Registry. Clin. Exp. Nephrol. 2017;21:1024–1029. doi: 10.1007/s10157-017-1398-5. [DOI] [PubMed] [Google Scholar]
- 17.Nakagawa N, et al. Clinical features and pathogenesis of membranoproliferative glomerulonephritis: A nationwide analysis of the Japan renal biopsy registry from 2007 to 2015. Clin. Exp. Nephrol. 2017;22:797–807. doi: 10.1007/s10157-017-1513-7. [DOI] [PubMed] [Google Scholar]
- 18.Okabayashi Y, et al. Distribution of nephrologists and regional variation in the clinical severity of IgA nephropathy at biopsy diagnosis in Japan: A cross-sectional study. BMJ Open. 2018;8:1–8. doi: 10.1136/bmjopen-2018-024317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Katsuno T, et al. A nationwide cross-sectional analysis of thrombotic microangiopathy in the Japan Renal biopsy registry (J-RBR) Clin. Exp. Nephrol. 2020;24:789–797. doi: 10.1007/s10157-020-01896-7. [DOI] [PubMed] [Google Scholar]
- 20.Sugiyama H, et al. Japan Renal Biopsy Registry and Japan Kidney Disease Registry: Committee report for 2009 and 2010. Clin. Exp. Nephrol. 2013;17:155–173. doi: 10.1007/s10157-012-0746-8. [DOI] [PubMed] [Google Scholar]
- 21.Woo KT, et al. Global evolutionary trend of the prevalence of primary glomerulonephritis. Nephron. Clin. Pract. 2010;116:c337–c346. doi: 10.1159/000319594. [DOI] [PubMed] [Google Scholar]
- 22.O’Shaughnessy MM, et al. Glomerular disease frequencies by race, sex and region: Results from the International Kidney Biopsy Survey. Nephrol. Dial Transplant. 2017;33:661–669. doi: 10.1093/ndt/gfx189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Genovese G, et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science. 2010;329:841–845. doi: 10.1126/science.1193032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Haas M, Meehan SM, Karrison TG, Spargo BH. Changing etiologies of unexplained adult nephrotic syndrome: A comparison of renal biopsy findings from 1976–1979 and 1995–1997. Am. J. Kidney Dis. 1997;30:621–631. doi: 10.1016/S0272-6386(97)90485-6. [DOI] [PubMed] [Google Scholar]
- 25.Kitiyakara C, Eggers P, Kopp JB. Twenty-one-year trend in ESRD due to focal segmental glomerulosclerosis in the United States. Am. J. Kidney Dis. 2004;44:815–825. doi: 10.1016/S0272-6386(04)01081-9. [DOI] [PubMed] [Google Scholar]
- 26.Sim JJ, et al. Distribution of biopsy-proven presumed primary glomerulonephropathies in 2000–2011 among a racially and ethnically diverse US population. Am. J. Kidney Dis. 2016;68:533–544. doi: 10.1053/j.ajkd.2016.03.416. [DOI] [PubMed] [Google Scholar]
- 27.D’Agati VD, Fogo AB, Bruijn JA, Jennette JC. Pathologic classification of focal segmental glomerulosclerosis: A working proposal. Am. J. Kidney Dis. 2004;43:368–382. doi: 10.1053/j.ajkd.2003.10.024. [DOI] [PubMed] [Google Scholar]
- 28.Imai E, et al. Kidney disease screening program in Japan: History, outcome, and perspectives. Clin. J. Am. Soc. Nephrol. 2007;2:1360–1366. doi: 10.2215/CJN.00980207. [DOI] [PubMed] [Google Scholar]
- 29.Nihon Jinzo Gakkai Special issue: Evidence-based practice guideline for the treatment of CKD. Nihon Jinzo Gakkai Shi. 2013;55:585–860. [PubMed] [Google Scholar]
- 30.Praga M, et al. Absence of hypoalbuminemia despite massive proteinuria in focal segmental glomerulosclerosis secondary to hyperfiltration. Am. J. Kidney Dis. 1999;33:52–58. doi: 10.1016/S0272-6386(99)70257-X. [DOI] [PubMed] [Google Scholar]
- 31.Ozeki T, et al. The revised version 2018 of the nationwide web-based registry system for kidney diseases in Japan: Japan Renal Biopsy Registry and Japan Kidney Disease Registry. Clin. Exp. Nephrol. 2020;24:1058–1068. doi: 10.1007/s10157-020-01932-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chun MJ. Focal segmental glomerulosclerosis in nephrotic adults: Presentation, prognosis, and response to therapy of the histologic variants. J. Am. Soc. Nephrol. 2004;15:2169–2177. doi: 10.1097/01.ASN.0000135051.62500.97. [DOI] [PubMed] [Google Scholar]
- 33.Thomas DB, et al. Clinical and pathologic characteristics of focal segmental glomerulosclerosis pathologic variants. Kidney Int. 2006;69:920–926. doi: 10.1038/sj.ki.5000160. [DOI] [PubMed] [Google Scholar]
- 34.Stokes MB, Valeri AM, Markowitz GS, D’Agati VD. Cellular focal segmental glomerulosclerosis: Clinical and pathologic features. Kidney Int. 2006;70:1783–1792. doi: 10.1038/sj.ki.5001903. [DOI] [PubMed] [Google Scholar]
- 35.Deegens JKJ, Steenbergen EJ, Borm GF, Wetzels JFM. Pathological variants of focal segmental glomerulosclerosis in an adult Dutch population—Epidemiology and outcome. Nephrol. Dial Transplant. 2008;23:186–192. doi: 10.1093/ndt/gfm523. [DOI] [PubMed] [Google Scholar]
- 36.D’Agati VD, et al. Association of histologic variants in FSGS clinical trial with presenting features and outcomes. Clin. J. Am. Soc. Nephrol. 2013;8:399–406. doi: 10.2215/CJN.06100612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tsuchimoto A, et al. Utility of Columbia classification in focal segmental glomerulosclerosis: renal prognosis and treatment response among the pathological variants. Nephrol. Dial Transplant. 2019 doi: 10.1093/ndt/gfy374. [DOI] [PubMed] [Google Scholar]
- 38.Uemura O, et al. Creatinine-based equation to estimate the glomerular filtration rate in Japanese children and adolescents with chronic kidney disease. Clin. Exp. Nephrol. 2014;18:626–633. doi: 10.1007/s10157-013-0856-y. [DOI] [PubMed] [Google Scholar]
- 39.Matsuo S, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am. J. Kidney Dis. 2009;53:982–992. doi: 10.1053/j.ajkd.2008.12.034. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available because the consent obtained from the participants does not cover unlimited public sharing of the data but are available from the corresponding author on reasonable request.