Abstract
Importance
Initial guidelines recommended prompt endotracheal intubation rather than non-invasive ventilation (NIV) for COVID-19 patients requiring ventilator support. There is insufficient data comparing the impact of intubation versus NIV on patient-centered outcomes of these patients.
Objective
To compare all-cause 30-day mortality for hospitalized COVID-19 patients with respiratory failure who underwent intubation first, intubation after NIV, or NIV only.
Design
Retrospective study of patients admitted in March and April of 2020.
Setting
A teaching hospital in Brooklyn, New York City.
Participants
Adult COVID-19 confirmed patients who required ventilator support (non-invasive ventilation and/or endotracheal intubation) at discretion of treating physician, were included.
Exposures
Patients were categorized into three exposure groups: intubation-first, intubation after NIV, or NIV-only.
Primary outcome
30-day all-cause mortality, a predetermined outcome measured by multivariable logistic regression. Data are presented with medians and interquartile ranges, or percentages with 95% confidence intervals, for continuous and categorical variables, respectively. Covariates for the model were age, sex, qSOFA score ≥ 2, presenting oxygen saturation, vasopressor use, and greater than three comorbidities. A secondary multivariable model compared mortality of all patients that received NIV (intubation after NIV and NIV-only) with the intubation-first group.
Results
A total of 222 were enrolled. Overall mortality was 77.5% (95%CI, 72–83%). Mortality for intubation-first group was 82% (95%CI, 73–89%; 75/91), for Intubation after NIV was 84% (95%CI, 70–92%; 37/44), and for NIV-only was 69% (95%CI, 59–78%; 60/87). In multivariable analysis, NIV-only was associated with decreased all-cause mortality (odds ratio [OR]: 0.30, 95%CI, 0.13–0.69). No difference in mortality was observed between intubation-first and intubation after NIV. Secondary analysis found all patients who received NIV to have lower mortality than patients who were intubated only (OR: 0.44, 95%CI, 0.21–0.95).
Conclusions & Relevance
Utilization of NIV as the initial intervention in COVID-19 patients requiring ventilatory support is associated with significant survival benefit. For patients intubated after NIV, the mortality rate is not worse than those who undergo intubation as their initial intervention.
Keywords: Non-invasive positive pressure ventilation, Endotracheal intubation, Hypoxemia, COVID-19
1. Introduction
The high volume of critically ill patients suffering from the novel coronavirus disease 2019 (COVID-19) has presented new clinical challenges. The reported mortality rates for COVID-19 Intensive Care Unit (ICU) patients has ranged from 26% to 69% [[1], [2], [3], [4], [5]]. Respiratory failure is a frequent cause of mortality in COVID-19 victims [6]. A study on COVID-19 patients receiving non-invasive ventilation (NIV1 ) and intubation found 28-day mortality rates of 79% and 86%, respectively [6]. Many of the initial case series and descriptions of COVID-19 patients have characterized the lung damage from COVID-19 in terms of Acute Respiratory Distress Syndrome (ARDS) [7,8]. ARDS can develop from a variety of etiologies causing diffuse inflammation, increased pulmonary vascular permeability, and resulting loss of gas exchange and hypoxemia [8]. ARDS has defined criteria measurable with radiographic and laboratory findings. COVID-19 patients who decompensate generally meet the criteria for ARDS [9]. Given COVID-19 is a novel pathogen with no data to base treatment recommendation on, the basis for initial guidance in managing COVID-19 patients with respiratory failure was mainly from ARDS studies.
1.1. Ventilatory support for ARDS, pneumonia, & COVID-19 patients
Much research over the past few years has focused on the question of appropriate ventilatory interventions for ARDS and pneumonia patients. Late intubation has been associated with increased mortality [10]. NIV techniques such as continuous positive airway pressure (CPAP) and biphasic positive airway pressure (BiPAP) have been criticized in recent years as not applicable to use in ARDS [11]. Attempts to use NIV in ARDS patients, therefore delaying intubation, have shown negative outcomes [12]. Similar to ARDS, the use of NIV in pandemic viral pneumonia is not recommended [13]. Studies show a higher mortality for NIV than invasive mechanical ventilation in patients with severe pneumonia, and over 90% of Middle East Respiratory Syndrome (MERS) patients who initially received NIV eventually required intubation [14,15].
Early guidance for respiratory support in COVID-19 recommended caution in the use of NIV [16] and noted that early intubation was preferable [17]. Later reports however, showed lower mortality for COVID-19 patients using NIV and high-flow nasal oxygen than with intubation [18]. Other authors have used a stepwise approach, advocating non-invasive ventilation or high flow nasal cannula oxygenation prior to intubation, in an effort to stave off need for it [19]. From an Infection Control standpoint, studies show NIV disperses air from the lungs a meter from the device and droplets can remain suspended in room air for the duration of use [[20], [21], [22], [23]]. This risk of aerosolization was an important consideration in the initial recommendations to avoid NIV [16,17] in order to protect the healthcare staff.
1.2. Our experience
Beginning in March of 2020, as the COVID-19 pandemic was declared, a wave of patients in respiratory distress arrived in the emergency department at our academic medical center in Brooklyn, New York. Within days, hospital guidelines were established calling for early intubation over NIV. Our institution was following national [17] and international [16] COVID-19 guidelines based on studies of acute respiratory distress syndrome (ARDS) and around infection control concerns [[20], [21], [22], [23]]. Notable among these COVID-19 victims were those with “silent hypoxia” who were also called “happy hypoxic” due to minimal respiratory distress despite extremely low oxygen saturations measured by pulse oximetry [24]. Witnessing the deleterious effect of prolonged hypoxia on patients, aggressive management of hypoxia became a priority early on. Noting high mortality rates among intubated patients, some physicians elected to first initiate NIV, against hospital guidelines recommending prompt intubation. Initiation of NIV before endotracheal intubation became popular with anecdotal success.
Therefore, we designed this study to examine the outcome of patients undergoing different initial methods of ventilator support in COVID-19 patients in respiratory distress.
2. Methods
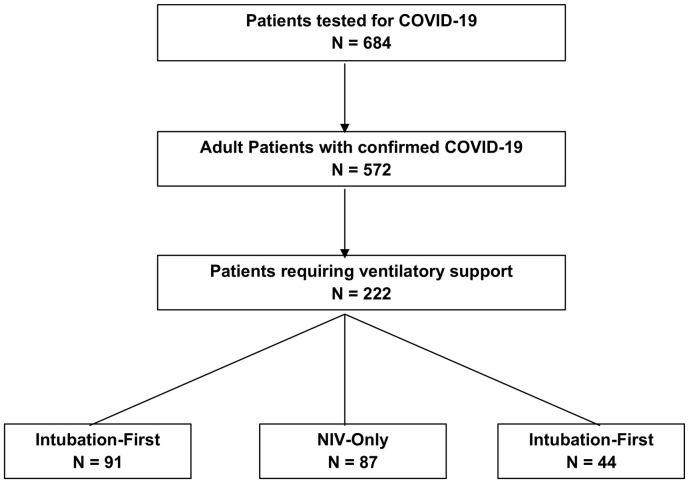
Study Design: The study design is a retrospective cohort study of a sample of patients, age 18 or older, with COVID-19 confirmed by real-time polymerase chain reaction between March 12th and April 13th, 2020. All these identified patients were followed up through May 12th, 2020, if they were still hospitalized. Patients were included if they required ventilatory support (intubation or NIV in the hospital) due to respiratory failure. Respiratory failure and initiation of ventilatory support was determined by the treating physician. See Fig. 1 for an exclusion flowchart. The State University of New York - Downstate Health Sciences University (SUNY – Downstate) Institutional Review Board approved the study and waived the written informed consent requirement.
Fig. 1.
Exclusion flowchart.
Study setting: The study was conducted at SUNY - Downstate, an academic medical center in Central Brooklyn, which was designated by the state authorities as a COVID-only center. The area of Central Brooklyn is inhabited by a mostly under- or un-insured and underserved population of African- and Caribbean-Americans.
Group Comparisons: Three categories defined the exposures in the primary analysis: those who were initially intubated (Intubation-First), those who were initially on NIV and did not require intubation (NIV-Only), and those who were initially on NIV and required subsequent intubation (NIV-Intubation).
Outcome: The study outcome was all-cause in-hospital or 30-day mortality (whichever came first). The outcome was determined by reviewing the institutional electronic medical records (EMR).
Study Protocol: Data was collected and reviewed from EMR according to a set study protocol by a trained team of physicians and medical students. Patient confidentiality was protected by systematically de-identifying patients and storing patient data in a HIPAA-compliant, institution-managed network drive only accessible to those with an institution-associated account and permission given by the principal investigator.
NIV and intubation were defined as either an order or physician documentation of the use of CPAP/BiPAP or endotracheal intubation. In cases of these interventions, when orders and physician documentation disagreed, written documentation was used for data entry. If an order regarding ventilatory intervention was found in the chart but no documentation supported the order having been carried out, it was assumed such interventions were not performed. For the purposes of categorizing exposure groups, we assumed that if NIV and intubation were ordered on the same date, that NIV took place first. This assumption is based on the typical sequence of escalating interventions in the clinical setting, and an informal review of the data showing that nearly all of these patients remained intubated beyond the date of intubation, precluding use of NIV. Patients intubated in the field were categorized in the Intubation-First group.
All demographic variables were collected from EMR demographic data. Older age, Black race, and male sex have all been correlated with increased mortality among COVID-19 patients [2,4,25]. The data to calculate qSOFA and SpO2 were collected from EMR records of arrival vital signs and mental status. Higher mortality is seen in patients with a qSOFA score of two or greater; this is validated for hospital and ED patients with infectious symptoms [26,27]. Lower SpO2 is correlated with increased mortality in COVID-19 patients [28]. Separately, the first recorded ratio of arterial oxygen to the fraction of inspired oxygen (P/F ratio) was collected from the EMR. Pressor use, associated with shock, at any time during the patients' hospital stay was assessed from their medication orders.
Comorbidities were collected from patients' ICD-10 codes as recorded in the EMR. Medical comorbidities included hypertension, diabetes mellitus, congestive heart failure, coronary artery disease, chronic pulmonary disease, chronic kidney disease, Immunocompromised patients (due to HIV or other reasons), cancer, and obesity (body mass index [BMI] ≥30). Each comorbidity was assigned a score of 1, and if 3 or more the comorbidities were present, a patient was categorized as ≥3 comorbidities for analysis. These comorbidities have been found at higher rates in COVID-19 patients than in the general population [29]. Comorbidities including obesity, diabetes, coronary artery disease, and hypertension are also associated with increased mortality [30,31], and likelihood of having been intubated [2,31], among COVID-19 patients. Mental status on presentation and body mass index (BMI), both components of other variables, were also collected for bivariate analysis.
Statistical analysis: Data are presented with medians and interquartile ranges, or percentages with 95% confidence intervals, for continuous and categorical variables respectively. Case-wise deletion was used to handle missing data. Bivariate analysis was carried out using a chi-square test to assess the association of categorical variables with mortality. Continuous variables were analyzed using a Kruskal-Wallis H test. Multivariable logistic regression was used to establish the odds of mortality. The variables included in the regression model were chosen based on previous literature or if they reached a p value of <0.1 in the bivariate analysis [32]. The association of each variable with the study outcome is reported with odds ratio (OR) and 95% confidence interval (CI). Patients who were intubated first were the reference group for both models.
We established significance of the model at the 0.05 α level. Statistical analysis was carried out using SAS 9.4.
3. Results
During the study period, a total of 574 patients tested positive for COVID-19. Of these patients, 222 adult patients met the inclusion criteria (respiratory failure requiring ventilatory support in the -hospital). All but one of the included patients had a disposition (discharged alive or died) by the end of the follow-up period. The overall mortality was 77.5% (95%CI, 72–83%). Mortality for Intubation-First group was 82% (95%CI, 73–89%; 75/91), for NIV-Intubation was 84% (95%CI, 70–92%; 37/44), and for NIV-Only was 69% (95%CI, 59–78%; 60/87). The baseline characteristics of the study cohort are listed in Table 1 . The most common comorbidities were hypertension, diabetes, and asthma.
Table 1.
Baseline characteristics of the study Cohort
| COVID-19 Patient Characteristics and Conditions | ||
|---|---|---|
| Characteristics | N = 222 (%, or IQR* for continuous variables) | 95% Confidence Interval |
| Exposure | ||
| Intubation First | 91 (41%) | (0.34, 0.48) |
| NIV to intubation | 44 (20%) | (0.15, 0.26) |
| NIV Only | 87 (39%) | (0.33, 0.46) |
| Age | 69.5 (62–78) | (67.2, 70.5) |
| Sex | ||
| Male | 129 (58%) | (0.51, 0.65) |
| Female | 93 (42%) | (0.35, 0.49) |
| Race/Ethnicity | ||
| Black/African American | 194 (87%) | 0.82, 0.91) |
| White | 12 (5%) | 0.02, 0.09) |
| Hispanic | 1 (<1%) | 0.0001, 0.02) |
| Asian | 1 (<1%) | 0.0001, 0.02) |
| Undisclosed/Unknown | 14 (6%) | 0.03, 0.10) |
| BMI | ||
| BMI continuous | 29.39 (25.5–33) | (29.4, 31.4) |
| BMI ≥ 30 | 83 (44%) | (0.37, 0.51) |
| Symptoms | ||
| SpO2 ≤ 90 | 98 (46%) | (0.41, 0.55) |
| ED SpO2 | 91 (86–97) | (88.4, 90.9) |
| Fever ≥100.4 | 78 (36%) | (0.30, 0.42) |
| qSOFA ≥2 | 58 (26%) | (0.21, 0.33) |
| Altered Mental Status | 77 (35%) | (0.28, 0.41) |
| Pressor Use | 74 (33%) | (0.27, 0.40) |
| Comorbidities | ||
| Diabetes | 136 (61%) | (0.55, 0.68) |
| Hypertension | 169 (76%) | (0.71, 0.82) |
| Coronary Artery Disease | 37 (17%) | (0.12, 0.22) |
| Asthma | 22 (44%) | (0.30, 0.58) |
| COPD | 23 (43) | (0.29,0.56) |
| Any Pulmonary Disease | 52 (27%) | (0.21, 0.34) |
| ≥ 3 Comorbidities | 125 (56%) | (0.50, 0.63) |
*Interquartile range values (IQR) are medians (IQR)
† Missing values for BMI variables, Fever, qSOFA ≥2, ≥3 comorbidities, Coronary Artery Disease, Asthma, COPD_Emphysema & SpO2
*Comorbidities included hypertension, diabetes, congestive heart failure, coronary artery disease, chronic pulmonary disease, kidney disease, HIV, immunocompromised from another etiology, cancer, or obesity (BMI ≥ 30).
- Abbreviations: NIV: Non-invasive ventilation; IQR: Inter-quartile range; SPO2: Pulse Oxymetry; qSOFA: quick Sepsis Related Organ Failure Assessment; BMI: Body Mass Index.
Table 2 presents the characteristics of the cohort categorized by the exposure group. Age, mental status, vasopressor use, diabetes, obesity, and presenting SpO2 varied by exposure group. The P/F ratio was recorded for 140 patients; the median value was 138 (IQR, 74–241).
Table 2.
Patient characteristics stratified by ventilatory support method
| Variables | Exposure Groups Stratified |
|||
|---|---|---|---|---|
| Intubation-first |
NIV-Only |
NIV-intubation |
P-value | |
|
N = 91 (41%) |
N = 87 (39%) |
N = 44 (20%) |
||
| N = 222 (% with 95% CI or Median with IQR) | ||||
| Age (Median) | 67 (60–76) | 67 (65–82) | 69 (58–75) | 0.004 |
| Male gender | 55 (60) | 51 (59) | 23 (52) | 0.66 |
| (0.50, 0.71) | (0.48, 0.69) | (0.37, 0.68) | ||
| Black/African American race | 80 (88) | 72 (83) | 42 (95) | 0.12 |
| (0.79, 0.94) | (0.73, 0.90) | (0.85, 0.99) | ||
| SpO2 < 90.5 | 37 (41) | 37 (45) | 24 (62) | 0.15 |
| (0.33, 0.55) | (0.34, 0.57) | (0.45, 0.77) | ||
| Temperature ≥ 100.4 | 35 (38) | 25 (29) | 18 (41) | 0.23 |
| (0.30, 0.51) | (0.20, 0.40) | (0.26, 0.57) | ||
| Triage SpO2 (Median) | 93 (87–97) | 91.5 (86–96) | 88 (81–94) | 0.008 |
| qSOFA ≥ 2 | 29 (32) | 21 (24) | 8 (18) | 0.21 |
| (0.22, 0.42) | (0.16, 0.35) | (0.08, 0.33) | ||
| Altered Mental Status | 40 (44) | 27 (31) | 10 (23) | 0.03 |
| (0.34, 0.54) | (0.22, 0.41) | (0.13, 0.37) | ||
| Pressor Use | 50 (55) | 2 (2) | 22 (50) | <0.0001 |
| (0.45, 0.65) | (0.014, 0.80) | (0.36, 0.64) | ||
| BMI (Median) | 30 (26–35) | 28 (25–30) | 31 (27–34) | 0.09 |
| Diabetes | 49 (54) | 52 (60) | 35 (80) | 0.02 |
| (0.43, 0.64) | (0.49, 0.70) | (0.68, 0.91) | ||
| Hypertension | 65 (71) | 71 (83) | 33 (77) | 0.28 |
| (0.62, 0.81) | (0.73, 0.90) | (0.64, 0.89) | ||
| Coronary Artery Disease | 10 (12) | 20 (23) | 7 (16) | 0.14 |
| (0.04, 0.19) | (0.14, 0.32) | (0.05, 0.27) | ||
| Asthma | 8 (36) | 11 (58) | 3 (33) | 0.30 |
| (0.36, 0.80) | (0.30, 0.64) | (0.16, 0.56) | ||
| COPD/Emphysema | 8 (35) | 10 (53) | 5 (42) | 0.51 |
| (0.15, 0.54) | (0.30, 0.75) | (0.14, 0.70) | ||
| Any Pulmonary Disease | 19 (25) | 24 (31) | 9 (25) | 0.68 |
| (0.15, 0.35) | (0.05, 0.21) | (0.11, 0.39) | ||
| ≥ 3 Comorbidities | 48 (53) | 49 (56) | 28 (63) | 0.49 |
| (0.42, 0.63) | (0.45, 0.67) | (0.48, 0.78) | ||
| BMI ≥ 30 | 38 (50) | 25 (32) | 20 (56) | 0.03 |
| (0.38, 0.62) | (0.22, 0.44) | (0.38, 0.72) | ||
| Mortality | 75 (82) | 60 (69) | 37 (84) | 0.0501 |
| (0.73, 0.90) | (0.58, 0.78) | (0.70, 0.93) | ||
Multivariable Analysis: In the multivariable adjusted logistic regression model, we found a 70% reduction in odds of mortality among COVID-19 patients who received NIV only (OR 0.30, 95% CI 0.13–0.69) compared to those who received intubation without NIV. No association was found for those who were intubated after initial therapy with NIV, as compared to those intubated without prior NIV (Table 3 ).
Table 3.
Adjusted Odds Ratios for Mortality among select COVID-19 Patient Characteristics and Conditions
| Variables | Adjusted Odds Ratio, 95% CI | P-value* |
|---|---|---|
| Intubation-first | 1.00 | – |
| NIV-only | 0.30 | 0.004 |
| (0.13, 0.69) | ||
| NIV-intubation | 1.39 | 0.58 |
| (0.44, 4.39) | ||
| Age | 1.06 | <0.001 |
| (1.03, 1.09) | ||
| Male gender | 1.18 | 0.67 |
| (0.55, 2.53 | ||
| ≥ 3 Comorbidities | 1.28 | 0.51 |
| (0.61, 2.68) | ||
| SpO2 ≤ 90 | 1.34 | 0.43 |
| (0.64, 2.81) | ||
| qSOFA ≥ 2 | 1.67 | 0.28 |
| (0.66, 4.22) | ||
| Pressor Use | 1.92 | 0.21 |
| (0.70, 5.28) |
Of all 131 patients receiving NIV as initial ventilatory support, 44 (33.6%) required intubation after NIV treatment failed. Secondary analysis (data not shown) found no difference in crude mortality for patients who received NIV as an initial intervention, as compared to those who were intubated without NIV. In the secondary multivariable adjusted logistic regression model, we found 66% reduced odds of mortality among COVID-19 patients who received NIV initially (OR 0.44, 95% CI 0.21–0.95) compared to those who received intubation without NIV.
4. Discussion
This study compared - all-cause 30-day mortality for hospitalized COVID-19 patients with respiratory failure who underwent intubation without prior NIV, intubation after NIV, or NIV alone. Our analysis of a sample of 222 COVID-19 adult inpatients at a teaching hospital in Brooklyn, New York found that utilization of NIV as the first step of the ventilatory support, was associated with improved survival. Our study also revealed that the mortality rate in patients who were intubated first was similar to mortality rates in patients who were intubated after an initial attempt of NIV. In the setting of initial NIV failing the patient, the risk of death would be the same as for those who are intubated first. Other similar studies show initiating NIV prior to intubation is presenting with some success [23,24,26]. In contrast to data from ARDS studies, particularly those looking at ICU patients with severe ARDS [33], using NIV as the first step did not increase mortality of the patients reviewed in our study [12].
This study contributes to the body of evidence suggesting that COVID-19 patients may respond differently to NIV than patients suffering from ARDS or severe viral pneumonia [9,34]. In this light, the interchangeability of clinical guidance for these three groups needs to be re-evaluated. If NIV prior to intubation is not significantly associated with an increased risk of mortality, as our results indicate, then it may be clinically prudent to use intubation as a last resort, as has been suggested [35]. Furthermore, there were significantly lower odds of mortality for patients who only receive NIV. More research is needed to determine which subset of patients may indeed benefit from intubation, and when it should be performed.
Treatment measures at the individual level must also weigh the risks to the overall response in a disaster. Respiratory support that further spreads infection to staff and patients will increase the number of victims with COVID-19 while decreasing the number of healthcare staff available to manage the event. Clinicians can attempt to mitigate this with the use of helmet ventilators, negative pressure rooms, and appropriate PPE for hospital staff.
There are practical concerns of ventilator availability unrelated to pulmonary management that affect the level of respiratory support used in COVID-19 patients. In the setting of a national deficit of ventilators, NIV is an alternative treatment option to conserve ventilators [36]. Furthermore, the staff needed to manage a ventilated patient is a great strain on a system that is faced with staffing shortages at baseline. The overall response of the COVID-19 disaster depends on not wasting valuable resources such as ventilators and critical care staff on patients who clinically do not require them for survival.
4.1. Limitations
While Brooklyn has been one of the epicenters of the global COVID-19 pandemic, our cohort mostly consists of African-American and Caribbean-American population with high prevalence of comorbidities. Therefore, our findings may not be generalizable to some other parts of the United States or to global populations at large. Older average age together with high prevalence of comorbidities, particularly diabetes, obesity, and asthma, are likely contributors to the high mortality rates seen in our study population. These mortality rates do, however, appear to be in line with those seen in intubated patients at other New York City area hospitals [37], although higher than seen in other New York City studies [2]. The severity of disease in our sample maybe further described by the median P/F ratio which, if discussed in terms of ARDS, falls into the category of moderate ARDS [38]. The P/F ratio as a data point has limitations in our study however, as this retrospective study could only use one snippet of a dynamic marker.
The statistical significance of our results may be affected by a relatively small sample size. Despite the high volume of COVID-19 patients presenting at our hospital, the limited timeframe available to capture data and the necessary filtering of patients to obtain the appropriate sample curtailed our sample size. Our smaller sample precluded the use of matching and sub-group analysis. Larger future studies may find a significant difference in mortality with regard to the use of NIV prior to intubation. We were able to follow almost all patients through their hospitalization course (via the EMR) as only one patient remained hospitalized after thirty days. We were unable to account for patients who may have died after discharge within 30 days if their death was not recorded in the EMR.
There are a number of limitations which affect our ability to address in closer detail the respiratory interventions in question. CPAP and BiPAP orders were not differentiated in our EMR, and so NIV has here been defined as the broad category encompassing both of these modalities. We hope that future studies may be able to provide a closer analysis of differences in outcomes between these two modalities. Likewise, while the EMR can show that patients were intubated in the hospital, we were unable to assess whether any of these patients had previously been intubated or given CPAP (EMS in our study area typically do not have access to BiPAP) by EMS prior to arrival. There are, especially with a disease where best practices are still being established, many possible variations in clinical care by physician which may be related to both ventilatory management decisions and outcomes. Other issues not related to clinical course, including institutional protocols and crowding of the intensive care unit. It is difficult to assess how these limitations may impact the results of our study, but a future study examining patient outcomes as they vary by vent settings, lung compliance, physician, unit, or hospital, may help to clarify these variations.
The data for time of ventilatory interventions were collected from the orders placed in the EMR. It is likely, especially given the strains placed on hospital staff in the context of pandemic conditions, that the ordered times do not accurately reflect the time of the intervention. For this reason, we cannot directly speak to the use of “early” intubation, and instead can only discuss the sequence of ventilatory interventions.
We attempted to introduce appropriate covariates into our model to account for factors which may affect both mortality and the type of respiratory intervention a patient receives. The decision to choose invasive versus non-invasive ventilation is a complex one which heavily relies on clinical judgement based on consideration of many factors. Some of these confounders might not have been accounted for in our analysis and the potential for residual confounding exists. The chosen covariates themselves are subject to the limitations of retrospective data collection. A qSOFA score is calculated based on respiratory rate, systolic blood pressure, and mental status. Respiratory rate, in particular, may be incorrectly recorded in the EMR, especially in high-volume situations like those into which the patients in this study arrived at the hospital. Pulse oximetry readings vary depending on patient and equipment use characteristics, and whether a patient is receiving oxygen supplementation, factors for which we were unable to account for due to limitations of documentation. Some of the patients in our sample arrived at the hospital already intubated, and some arrived with CPAP already provided by emergency medical services. Future studies examining the ratio of arterial oxygen to the fraction of inspired oxygen may get a better approximation of patient severity on presentation.
The predominance of hypoxemia and respiratory failure in patients hospitalized with severe COVID-19 poses a challenge to physicians caring for these patients. Our study is highly relevant to the current climate as it examines the association of different ventilatory support modalities to a patient-centered outcome (mortality). Ultimately, randomized controlled trials comparing these ventilatory support mechanisms should shed light on the definitive management of respiratory failure in COVID-19 patients.
5. Conclusion
Utilizing NIV as an initial therapy in COVID-19 patients requiring ventilatory support is associated with significant survival benefit. For failed NIV treatment, patients' mortality rate is not worse than for those who undergo intubation first.
Declaration of Competing Interest
None.
Acknowledgements
We would like to acknowledge the contributions through data collection.
of:
-
•
Alvin Oomen, 4th year Medical Student, SUNY Downstate Health Sciences University, Brooklyn, NY, USA, alvin.oomen@downstate.edu
-
•
Jessica Delahanty, 4th year Medical Student, SUNY Downstate Health Sciences University, Brooklyn, NY, USA, jessica.delahanty@downstate.edu
As well as the following for their contribution to framing and discussion of on-the-ground.
treatments in the ED:
-
•
Surriya Ahmad, MD, SUNY Downstate Health Sciences University, Brooklyn, NY, USA, surriya.ahmad@downstate.edu
Footnotes
NIV refers to the use of ventilatory positive airway pressure through a mask as opposed to an endotracheal tube
References
- 1.Arentz M., Yim E., Klaff L., et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA. 2020 Apr 28;323(16):1612–1614. doi: 10.1001/jama.2020.4326. Published online March 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goyal P., Choi J.J., Pinheiro L.C., et al. Clinical characteristics of Covid-19 in New York City. N Engl J Med. 2020 Apr;17 doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang D., Hu B., Hu C., et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grasselli G., Zangrillo A., Zanella A., et al. Baseline characteristics and outcomes of 1591 Patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323(16):1574–1581. doi: 10.1001/jama.2020.5394. Published online April 06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bhatraju P.K., Ghassemieh B.J., Nichols M., et al. Covid-19 in critically ill Patients in the Seattle region - case series. N Engl J Med. 2020 Mar 30 doi: 10.1056/NEJMoa2004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yang Xiaobo, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. The Lancet Respiratory Medicine. 2020;8(5):P475–481. doi: 10.1016/S2213-2600(20)30079-5. May 01, 2020; Published:February 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang C., Wang Y., Li X., et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 Feb 15;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.The ARDS Definition Task Force* Acute respiratory distress syndrome: The Berlin definition. JAMA. 2012;307(23):2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 9.Marini J.J., Gattinoni L. Management of COVID-19 respiratory distress. JAMA. 2020;323(22):2329–2330. doi: 10.1001/jama.2020.6825. Published online April 24. [DOI] [PubMed] [Google Scholar]
- 10.Kangelaris K.N., Ware L.B., Wang C.Y., et al. Timing of intubation and clinical outcomes in adults with acute respiratory distress syndrome. Crit Care Med. 2016;44(1):120–129. doi: 10.1097/CCM.0000000000001359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nava S., Hill N. Non-invasive ventilation in acute respiratory failure. Lancet. 2009 Jul 18;374(9685):250–259. doi: 10.1016/S0140-6736(09)60496-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garpestad E., Schumaker G., Hill N.S. Noninvasive ventilation for acute respiratory distress syndrome: breaking down the final frontier? Crit Care Med. 2007 Jan;35(1):288–290. doi: 10.1097/01.CCM.0000251637.35325.F5. [DOI] [PubMed] [Google Scholar]
- 13.Rochwerg B., et al. Official ERS/ATS clinical practice guidelines: noninvasive ventilation for acute respiratory failure. European Respiratory Journal. 2017;50(2) doi: 10.1183/13993003.02426-2016. [DOI] [PubMed] [Google Scholar]
- 14.Valley T.S., Walkey A.J., Lindenauer P.K., Wiener R.S., Cooke C.R. Association between noninvasive ventilation and mortality among older Patients with pneumonia. Crit Care Med. 2017;45(3):e246–e254. doi: 10.1097/CCM.0000000000002076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alraddadi B.M., Qushmaq I., Al-Hameed F.M., et al. Noninvasive ventilation in critically ill patients with the Middle East respiratory syndrome. Influenza Other Respi Viruses. 2019;13(4):382–390. doi: 10.1111/irv.12635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clinical management of severe acute respiratory infection when COVID-19 is suspected. World Health Organization. Available at https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected. Accessed May 8th, 2020.
- 17.COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available at https://www.covid19treatmentguidelines.nih.gov/. Accessed May 8th, 2020. [PubMed]
- 18.Wang K., Zhao W., Li J., et al. The experience of high-flow nasal cannula in hospitalized patients with 2019 novel coronavirus-infected pneumonia in two hospitals of Chongqing, China. Ann Intensive Care. 2020;10:37. doi: 10.1186/s13613-020-00653-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sun Q., Qiu H., Huang M., Yang Y. Lower mortality of COVID-19 by early recognition and intervention: experience from Jiangsu Province. Ann Intensive Care. 2020;10(1):33. doi: 10.1186/s13613-020-00650-2. Published 2020 Mar 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hui D.S., Chow B.K., Lo T., et al. Exhaled air dispersion during noninvasive ventilation via helmets and a total facemask. Chest. 2015;147(5):1336–1343. doi: 10.1378/chest.14-1934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Simonds A.K., Hanak A., Chatwin M., et al. Evaluation of droplet dispersion during non-invasive ventilation, oxygen therapy, nebuliser treatment and chest physiotherapy in clinical practice: implications for management of pandemic influenza and other airborne infections. Health Technol Assess. 2010 Oct;14(46):131–172. doi: 10.3310/hta14460-02. [DOI] [PubMed] [Google Scholar]
- 22.World Health Organization 2020. Infection prevention and control during health care when novel coronavirus (nCoV) infection is suspected: interim guidance. World Health Organization 2020. Date accessed: April 14, 2020.
- 23.Tran K., Cimon K., Severn M., Pessoa-Silva C.L., Conly J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: a systematic review. PLoS One. 2012;7(4) doi: 10.1371/journal.pone.0035797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Levitan R. The Infection That’s Silently Killing Coronavirus Patients. The New York Times. 2020 Apr 20 [Google Scholar]
- 25.Wadhera R.K., Wadhera P., Gaba P., et al. Variation in COVID-19 hospitalizations and deaths across New York City boroughs. JAMA. 2020;323(21):2192–2195. doi: 10.1001/jama.2020.7197. Published online April 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Seymour C.W., Liu V.X., Iwashyna T.J., et al. Assessment of clinical criteria for Sepsis: for the third international consensus definitions for Sepsis and septic shock (Sepsis-3) JAMA. 2016;315(8):762–774. doi: 10.1001/jama.2016.0288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Freund Y., Lemachatti N., Krastinova E., et al. Prognostic accuracy of Sepsis-3 criteria for in-hospital mortality among Patients with suspected infection presenting to the emergency department. JAMA. 2017;317(3):301–308. doi: 10.1001/jama.2016.20329. [DOI] [PubMed] [Google Scholar]
- 28.Xie J., Covassin N., Fan Z., et al. Association Between Hypoxemia and Mortality in Patients With COVID-19. Mayo Clin Proc. 2020;95(6):1138–1147. doi: 10.1016/j.mayocp.2020.04.006. [published online ahead of print, 2020] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Killerby M.E., Link-Gelles R., Haight S.C., et al. Characteristics associated with hospitalization among Patients with COVID-19 — metropolitan Atlanta, Georgia, March–April 2020. MMWR Morb Mortal Wkly Rep. 2020;69:790–794. doi: 10.15585/mmwr.mm6925e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Guan W.J., Liang W.H., Zhao Y., et al. Comorbidity and its impact on 1590 patients with Covid-19 in China: A Nationwide Analysis published online ahead of print. Eur Respir J. 2020;2020:2000547. doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nakeshbandi M., Maini R., Daniel P., et al. The impact of obesity on COVID-19 complications: a retrospective cohort study. Int J Obes (Lond) 2020;44:1832–1837. doi: 10.1038/s41366-020-0648-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vittinghoff E., Glidden D.V., Shiboski S.C., et al. Springer; New York: 2015. Regression methods in biostatistics: Linear, logistic, survival, and repeated measures models. [Google Scholar]
- 33.Bellani G., Laffey J.G., Pham T., LUNG SAFE Investigators; ESICM Trials Group, et al. Noninvasive ventilation of Patients with acute respiratory distress syndrome. Insights from the LUNG SAFE study. Am J Respir Crit Care Med. 2017 Jan 1;195(1):67–77. doi: 10.1164/rccm.201606-1306OC. [DOI] [PubMed] [Google Scholar]
- 34.Bartosch J. UChicago medicine doctors see ‘truly remarkable’ success using ventilator alternatives to treat COVID-19. U Chicago Medicine Forefront. 2020 Apr 22 https://www.uchicagomedicine.org/forefront/coronavirus-disease-covid-19/uchicago-medicine-doctors-see-truly-remarkable-success-using-ventilator-alternatives-to-treat-covid19 (Online publication, accessed April 30, 2020) [Google Scholar]
- 35.Tobin M.J. Basing respiratory management of Coronavirus on physiological principles. Am J Respir Crit Care Med. 2020 Jun 1;201(11):1319–1320. doi: 10.1164/rccm.202004-1076ED. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ranney M.L., Griffeth V., Jha A.K. Critical supply shortages - The need for ventilators and personal protective equipment during the Covid-19 pandemic. N Engl J Med. 2020 Apr 30;382(18) doi: 10.1056/NEJMp2006141. [DOI] [PubMed] [Google Scholar]
- 37.Richardson S., Hirsch J.S., Narasimhan M., et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020;323(20):2052–2059. doi: 10.1001/jama.2020.6775. Published online April 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Horovitz J.H., Carrico C.J., Shires G.T. Pulmonary response to major injury. Arch Surg. 1974 Mar;108(3):349–355. doi: 10.1001/archsurg.1974.01350270079014. [DOI] [PubMed] [Google Scholar]