Abstract
Background
The recent novel conception of neoadjuvant immunotherapy has generated interest among surgeons worldwide, especially the lack of experience involving surgical treatment for the neoadjuvant immunotherapy population.
Methods
Patients with non-small cell lung cancer (NSCLC), who underwent neoadjuvant immunotherapy or chemo-immunotherapy, were retrospectively collected between September 2018 and April 2020. Demographic data, pathological and clinical features, therapeutic regimens and outcome data of all individuals were collected by retrospective chart review. Operative details, information of neoadjuvant therapy, were also abstracted.
Results
In total, 31 patients were included in the final analysis. The patients’ median age was 61 years. In total, 29 of the patients were males, while 2 were females. Patients received a median of 3 doses before resection. The median duration from final treatment to surgery was 34 days. After neoadjuvant treatment, post-treatment computed tomography scan showed that 24 patients had partial response. In total, 12 of 31 patients had a major pathological response, 15 pathological downstaging. Three patients had no residual viable tumor. A positive surgical margin was identified in 7 cases. One or more postoperative complications occurred in 18 of all 31 patients. In total, 26 patients underwent next-generation sequencing before surgery in total. Among them, 2 patients were detected STK11 mutations, none of whom had a major pathological response by final pathological examination.
Conclusions
Pulmonary resection after neoadjuvant immunotherapy or chemo-immunotherapy for resectable NSCLC appears to be safe with low operative mortality and morbidity rate in the current population.
Keywords: Neoadjuvant treatment, Immunotherapy, Non-small cell lung cancer
Introduction
Globally, lung cancer is one of the most common malignancies, with its high incidence, mortality and low 5-year survival rate [1]. Non-small cell lung cancer (NSCLC) is responsible for more than 85% of all cases of lung cancer, and approximately one-third of NSCLC cases are diagnosed at a locally advanced stage [2]. For patients with early stage (stage I and II) and appropriately selected locally advanced disease (stage IIIA and IIIB), the standard approach is multidisciplinary treatment including complete surgical resection, chemotherapy, radiotherapy, targeted therapy and so on [3].
Traditionally, platinum-based chemotherapy is mainly used in advanced or metastatic NSCLC [4]. More recently, PD-(L)1 immune checkpoint inhibitors have changed the treatment paradigm of patients with advanced or metastatic NSCLC [5, 6]. The development of anticancer immunotherapy targeting PD-1/PD-L1 and CTLA-4 immune checkpoints has achieved encouraging therapeutic effects in many cancers, including melanoma, NSCLC, renal cell cancer and Hodgkin disease [7]. With evidence that targeting the host immune response to lung cancer, and development of immune checkpoint blockage agents, immunotherapy has become the first-line therapy for the majority of patients with metastatic NSCLC [8].
Neoadjuvant therapy in the treatment of NSCLC is well established as part of the guidelines [9]. Recent published pilot data on preoperative use of checkpoint inhibition in patients with NSCLC suggested the safety and feasibility of inductive immunotherapy with surgically resectable cases [10–17]. The novel conception of neoadjuvant immunotherapy has generated interest among surgeons worldwide [18]. However, the lack of experience involving surgical treatment for the neoadjuvant immunotherapy population, caused substantial concerns of surgical perspective regarding the inflammatory action of these agents, such as pneumonitis and endocrinopathies, and subsequent technical challenges [19]. To address this, the current study was conducted to report the analysis of surgical perspective outcome data after neoadjuvant immunotherapy followed by surgery for resectable NSCLC.
Patients and methods
The current retrospective study was conducted at Shanghai Chest Hospital, an ultra-high-volume tertiary thoracic surgery center in Shanghai, China. The institutional review board approved the retrospective analysis of anonymous patient data. The data were retrospectively collected on patients admitted to Shanghai Chest Hospital between September 2018 and April 2020. Patients with histologic diagnosis of NSCLC underwent neoadjuvant immunotherapy or chemo-immunotherapy and with available clinicopathological characteristics were included. Exclusion criteria included patients recruited in blind clinical trials, unavailable clinicopathological characteristics.
The treatment regimens, including initial and subsequent regimens, indication for surgery were always discussed and determined by a multidisciplinary team. Demographic data, pathological and clinical features, therapeutic regimens and outcome data of all individuals were collected by retrospective chart review.
For all patients, preoperative assessment approaches should contain contrast-enhanced computed tomography (CT) scan or positron emission tomography (PET)/CT scan, brain imaging with CT or magnetic resonance imaging, pretreatment tumor biopsy, invasive mediastinal nodal staging with endobronchial ultrasound or mediastinoscopy. Operative details, including operative approach, extent of resection, operative time, hospital length of stay, postoperative morbidity, rationale for conversion of minimally invasive surgical procedures to thoracotomy if necessary and other details related to the operative experience, were recorded in detail. Information of neoadjuvant therapy, such as agents, courses, duration of final neoadjuvant treatment to surgery, was also abstracted.
Patients were staged according to AJCC Lung Cancer Staging, 8th edition (2017) [20]. Operative time was obtained by review of the anesthesia and operating room nurse record, which was defined as the time from skin incision to closure. Surgical complications were evaluated and recorded according to the criteria defined by the Society of Thoracic Surgeons and the European Society of Thoracic Surgeons general thoracic surgery databases [21]. Briefly, complete pathological response was defined as 0% viable tumor cells in residual tumor, while major pathological response as 10% remaining [22].
Statistical analysis
Data were expressed as median and range unless otherwise indicated, and P < 0.05 was considered significant. Statistical calculations were conducted with SPSS software (IBM SPSS Statistics for Windows, version 22.0., IBM Corp., Armonk, NY, USA).
Result
Of the 14,901 patients underwent lung resection in our institute during the study period, 31 patients received neoadjuvant immunotherapy or chemo-immunotherapy for at least 2 courses were included in the study. Majority of these 14,901 patients were treated with surgery only, instead of adjuvant or neoadjuvant treatment, due to their early-stage diseases. The demographic and clinical characteristics of the overall cohort are listed in Table 1. The patients’ median age at the time of the surgery was 61 years (range, 38–77 years). In total, 29 of the patients were males (93.5%), while 2 were females (6.5%). Squamous cell carcinoma (n = 22, 71.0%) was the most common histologic subtype and adenocarcinoma (n = 9, 29.0%) second most common. In total, the majority of patients had stage IIIA disease (16, 51.6%), and 10 patients (32.3%) with stage IIIB disease, 4 (12.9%) with stage IIB disease, 1 (3.2%) with stage IIA disease at the stage of treatment naive. Moreover, pathological responses were observed regardless of tumor PD-L1 expression, and PD-L1 expression did not appear to be predictive of a treatment benefit (Table 1, Fig. 1).
Table 1.
Demographic and clinical characteristics of the overall cohort
| Characteristic | < = 10% viable tumor (n = 12) | > 10% viable tumor (n = 19) | p value |
|---|---|---|---|
| Median age (range), ys | 60 (48–77) | 61 (38–66) | 0.408 |
| Sex | 0.510 | ||
| Female | 0 | 2 | |
| Male | 12 | 17 | |
| Median BMI (range) | 23.81 (21.05–27.77) | 24.49 (17.72–28.39) | 0.805 |
| Clinical stage | 0.455 | ||
| IIA | 0 | 1 | |
| T2bN0 | 0 | 1 | |
| IIB | 3 | 1 | |
| T2aN1 | 1 | 1 | |
| T2bN1 | 1 | 0 | |
| T3N0 | 1 | 0 | |
| IIIA | 6 | 10 | |
| T2aN2 | 3 | 5 | |
| T2bN2 | 0 | 1 | |
| T3N1 | 2 | 1 | |
| T4N0 | 0 | 2 | |
| T4N1 | 1 | 1 | |
| IIIB | 3 | 7 | |
| T3N2 | 3 | 3 | |
| T4N2 | 0 | 4 | |
| Histologic subtype | 0.101 | ||
| Squamous cell carcinoma | 11 | 11 | |
| Adenocarcinoma | 1 | 8 | |
| Smoking history | 0.523 | ||
| Never | 8 | 16 | |
| Former | 3 | 1 | |
| Current | 1 | 2 | |
| PD-L1 expression | 0.352 | ||
| < 1% | 5 | 5 | |
| 1—50% | 1 | 4 | |
| > 50% | 2 | 5 | |
| NA | 4 | 5 |
BMI body mass index, NA Unknown
Fig. 1.
Detailed information involving radiographic response, pathological response, pretreatment PD-L1 expression and NGS result of all 31 patients. NGS next-generation sequencing; PR partial response; SD stable disease; PD progressive disease; pCR pathological complete remission; NA not applicable
None of the patients received preoperative radiotherapy. The most commonly used neoadjuvant regimens were chemo-immunotherapy (n = 27, 87.1%), followed by immunotherapy alone (n = 4, 12.9%). The most commonly prescribed checkpoint inhibitor was pembrolizumab (16, 51.6%), the others as nivolumab (15, 48.4%). The frequencies of the most commonly used drugs and drug classes are shown in Table 2. Patients received a median of 3 doses before resection (range, 2–4 doses). The median duration from final treatment to surgery was 34 days (range, 4–93 days). After neoadjuvant treatment, post-treatment CT scan showed that 24 patients (77.4%) had partial response, five patients (16.1%) stable disease and two (6.5%) radiographic progression of disease (Table 2 and Fig. 1).
Table 2.
Neoadjuvant characteristics of the overall cohort
| Characteristic | ≤ 0% viable tumor (n = 12) | > 10% viable tumor (n = 19) | p value |
|---|---|---|---|
| Neoadjuvant regimens | 0.999 | ||
| Chemo-immunotherapy | 11 | 16 | |
| Immunotherapy alone | 1 | 3 | |
| Prescribed checkpoint inhibitor | 0.552 | ||
| Pembrolizumab | 5 | 10 | |
| Nivolumab | 7 | 9 | |
| Median doses (range) | 3 (2–3) | 2 (2–4) | 0.782 |
| Median duration from final treatment to surgery (range), days | 32 (4–93) | 36 (23–86) | 0.314 |
| Radiographic response assessment | 0.352 | ||
| PR | 10 | 14 | |
| SD | 2 | 3 | |
| PD | 0 | 2 | |
PR partial response; SD stable disease; PD progressive disease
Details of surgical intervention and tumor location for the patients are listed in Table 3. The most common procedure was lobectomy (18, 58.1%), followed by sleeve lobectomy (7, 22.6%), bilobectomy (4, 12.9%) and pneumonectomy (2, 6.5%). Minimally invasive approaches, including video-assisted thoracic surgery (VATS) in 8 patients, and robot-assisted thoracic surgery (RATS) in 1 patient, were attempted in 9 resections (7 lobectomies, 1 sleeve lobectomy, 1 bilobectomy). In total, only 1 patient (3.2%) with clinical stage IIB tumor, who was initially attempted to underwent VATS procedure, was converted to open thoracotomy due to dense adhesions of hilar lymph node causing difficulty dissecting the blood vessel. Therefore, the conversion rate from minimally invasive procedures to thoracotomy in the present population was 3.2%. Median operative time was 158 min (range, 77–279 min). Median estimated blood loss was 200 mL (range, 50–1600 mL). Two patients (%) required blood transfusion. Median hospital length of stay after surgery was 7 days (range, 2–29 days).
Table 3.
Surgical and postoperative characteristics of the overall cohort
| Characteristic | ≤ 10% viable tumor (n = 12) | > 10% viable tumor (n= 19) | p value |
|---|---|---|---|
| Tumor location | 0.250 | ||
| LUL | 2 | 3 | |
| LLL | 1 | 2 | |
| RUL | 5 | 12 | |
| RML | 0 | 1 | |
| RLL | 4 | 1 | |
| Extent of resection | 0.181 | ||
| Lobectomy | 4 | 14 | |
| Bilobectomy | 3 | 1 | |
| Sleeve lobectomy | 5 | 2 | |
| Pneumonectomy | 0 | 2 | |
| Approach | 0.930 | ||
| Open thoracotomy | 9 | 13 | |
| VATS | 2 | 6 | |
| RATS | 1 | 0 | |
| Median operative time (range), min | 159 (77–223) | 157 (82–279) | 0.994 |
| Median estimated blood loss (range), ml | 200 (50–200) | 200 (50–1600) | 0.265 |
| Median hospital length of stay after surgery (range), days | 6 (2–25) | 8 (4–29) | 0.906 |
| Pathological downstaging | 0.106 | ||
| No | 4 | 12 | |
| Yes | 8 | 7 | |
| Surgical margin | 0.029 | ||
| R0 | 12 | 12 | |
| R1 | 0 | 4 | |
| R2 | 0 | 3 | |
| Thirty-day mortality | 0 | 0 | NA |
| Ninety-day mortality | 0 | 0 | NA |
| Postoperative complications | 0.749 | ||
| Prolonged air leak | 6 | 8 | |
| Arrhythmia | 1 | 2 | |
| Pneumonia | 0 | 1 | |
| Chylothorax | 1 | 0 | |
| Wound infection | 0 | 1 | |
| Adjuvant treatment | 0.068 | ||
| Chemo-immunotherapy | 4 | 7 | |
| Chemotherapy | 3 | 2 | |
| Chemoradiotherapy | 0 | 4 | |
| Radiotherapy | 0 | 1 | |
| Immunotherapy alone | 1 | 0 | |
| Targeted therapy | 0 | 1 | |
| Active surveillance | 4 | 4 |
LUL left upper lobe; LLL left lower lobe; RUL right upper lobe; RML right middle lobe; RLL right lower lobe; VATS video-assisted thoracic surgery; RATS robot-assisted thoracic surgery; NA not applicable
Final pathological examination demonstrated 12 of 31 patients (38.7%) had a major pathological response (MPR) (defined as ≤ 10% residual viable tumor), 15 (48.4%) pathological downstaging. Three patients (9.7%) had no residual viable tumor, which means pathological complete remission, while viable tumor cells in 28 patients (90.3%) (Fig. 2). A positive surgical margin (R1 or R2) was identified in 7 (22.6%) cases, including 4 cases of R1 resection and 3 cases of R2 resection (Table 3). All 4 patients were classified as R1 resections because of positive highest mediastinal lymph nodes. Even though intraoperative pathology was conducted as clinical routine activity, positive surgical margin status was inevitable in these 3 patients identified as R2 resection, due to their limited lung function intolerant of bilobectomy or pneumonectomy. One or more postoperative complications occurred in 18 of all 31 patients (overall morbidity, 58.1%) (Table 3). The most common complication was prolonged air leak, occurring in 14 patients (45.2%), followed by arrhythmia (n = 3, 9.7%), pneumonia (n = 1, 3.2%), chylothorax (n = 1, 3.2%) and wound infection (n = 1, 3.2%). No early deaths (within 90 days) were reported among the whole cohort. There was no significant difference in complication rates between the two groups, patients had ≤10% viable tumor remaining on final pathological assessment, and those who had > 10% viable tumor (p = 0.981).
Fig. 2.
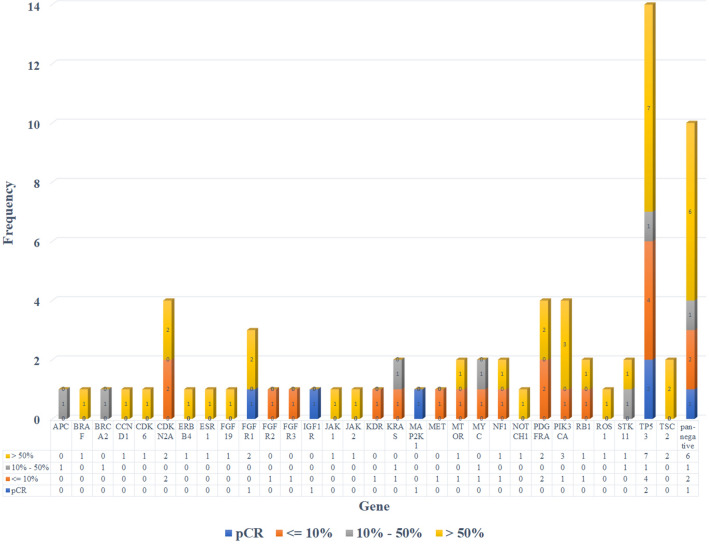
Detailed information about pathological response of 26 patients with NGS result. NGS next-generation sequencing
Following completion of surgical therapy, 11 patients (35.5%) received adjuvant chemo-immunotherapy, 5 patients (16.1%) chemotherapy, 4 patients (12.9%) chemoradiotherapy, 1 patient (3.2%) postoperative radiotherapy, 1 patient (3.2%) immunotherapy alone, 1 patient (3.2%) targeted therapy, while the remaining 8 patients (25.8%) underwent active surveillance. Remarkably, for these 7 patients with positive surgical margin (R1 or R2), postoperative radiotherapy or chemoradiotherapy was conducted following NCCN guidelines. Totally, 19 of 31 patients (61.3%) in the current cohort discontinued checkpoint inhibitor treatment postoperatively. Notably, one female 57-year-old patient diagnosed as stage IIIA adenocarcinoma, whose initial gene assay showed wild-type EGFR and KRAS, negative for ALK and ROS1 gene rearrangement, was prescribed chemo-immunotherapy (carboplatin-pemetrexed and nivolumab). After 3 doses neoadjuvant treatment, right upper lobe lobectomy was conducted. It is interesting that postoperative gene assay indicated EGFR exon 19 deletion. Therefore, adjuvant targeted therapy was suggested to this individual.
Additionally, 26 patients underwent next-generation sequencing (NGS) before surgery in total (Figs. 1, 2). Among them, 2 patients (7.7%) were detected STK11 mutations. None of these 2 individuals had an MPR by final pathological examination, which also confirmed that STK11 mutations might be an unfavorable factor to neoadjuvant immunotherapy [14] (Figs. 1, 2).
Discussion
In the current study, it is safe and feasible of the application of neoadjuvant immunotherapy or chemo-immunotherapy before surgery for patients with resectable NSCLC.
As is well known, with the rapid development of modern tumor immunology, immunotherapy has become an increasingly important weapon and achieving great success in tumor therapy [23]. The role of immunotherapy has restructured the treatment approach to numerous malignancies. Immune checkpoint inhibitors have revolutionized the therapeutic landscape in oncology and have shown significant clinical benefit in several cancer types [24]. The clinical application of immune checkpoint inhibitors has dramatically changed the treatment landscape for patients with advanced cancers, including advanced NSCLC [25].
Prior to immunotherapy, treatment for advanced NSCLC had not changed significantly since the broad uptake of chemotherapy over best supportive care in the mid-1990s [26]. Neoadjuvant chemotherapy has already been widely accepted as a comprehensive therapeutic strategy, especially for local advanced lung cancer [27]. Immunotherapy with immune checkpoint inhibitors has brought real progress in the treatment of solid tumors including lung cancer, which have revolutionized the management of patients with NSCLC [28]. At present, numerous ongoing investigations of neoadjuvant immunotherapy or chemo-immunotherapy demonstrate the promising future of immunotherapy in the complex management paradigm of advanced NSCLC, which represents one of the next frontiers in cancer immunotherapy [10, 29, 30].
With concerns of potential pulmonary and extrapulmonary toxicity after immunotherapy administration, such as pneumonitis, adrenal insufficiency and hyperthyroidism [31], for patients who have received neoadjuvant immunotherapy or chemo-immunotherapy, the safety of following pulmonary resection is the primary concern for thoracic surgeons [19]. According to previous studies, secondary analyses of former small pilot prospective studies, the approach of lung resection after neoadjuvant therapy is safe and reliable [10, 11, 32]. Our initial series demonstrated low rates of mortality and major intraoperative and postoperative complications, similar to those published in previous neoadjuvant immunotherapy or chemotherapy settings [19, 27]. In our current cohort, the rates of chest tube air leak (45.2%), arrhythmia (n = 3, 9.7%), pneumonia (3.2%), reintubation (0%) and respiratory failure (0%) were acceptable and comparable to previous studies [19, 27]. Compared with patients without neoadjuvant treatment, the complication profile was similar in the current population. The complications occurred at similar levels in the present neoadjuvant group, comparable with previous reports of without neoadjuvant cohort as 42.5–68.3% [33]. In addition, a similar trend is observed for the distribution of postoperative complications between the two systems [33]. In summary, patients who underwent surgery after neoadjuvant immunotherapy or chemo-immunotherapy did not have out-of-range intraoperative and postoperative complications rate, which indicates that patients can be safely operated with satisfied surgical outcomes after prescribed neoadjuvant immunotherapy or chemotherapy agents.
Since it is hard to quantitatively evaluate the difficulty of the surgery, there are some significant manifestations, such as operating time, amount of blood loss, conversion rate and perioperative complications [21]. In the present study, all similar manifestations as previously reported in the literature [11, 19, 27, 32], including comparable operation time, minimal intraoperative blood loss, low level of conversion rate and low rates of mortality and complications, indicate that neoadjuvant immunotherapy or chemo-immunotherapy does not increase the complexity and difficulty of the operation.
As one of the most significant predictors of prolonged survival, radical (R0) resection rate is critical to underline the feasibility and utility of neoadjuvant therapeutical strategy [27, 34]. Expect for the 77.4% R0 resection rate in the present cohort, it is worth noting that 12 patients (38.7%) have no evidence of residual tumor cell at microscopic examination. The present pilot data confirmed that STK11 mutations might be an unfavorable factor to neoadjuvant immunotherapy [14]. Consequently, the identification and characterization of novel markers that can predict residual disease after neoadjuvant therapy is critical to investigate new management approaches to select appropriate patient and improve clinical outcome.
As a real-world study focusing on patients with clinical practice, instead of clinical trial, the current report suggests the safety of surgical resection after neoadjuvant immunotherapy or chemo-immunotherapy in resectable NSCLC patients. Considering that neoadjuvant immunotherapy continues to be studied both individually and in combination with chemotherapy, these results would have a pertinence for thoracic oncologists and surgeons.
What remains unclear is the optimum neoadjuvant doses and interval from final administration of neoadjuvant agents to operation. In the situation of lacking evidence-based algorithms, current study could provide implications for clinical practice. Based on our data, neoadjuvant immunotherapy or chemo-immunotherapy was prescribed for 2–4 courses, followed by radiographic assessment of residual cancer and treatment response. After the last prescription, patients were often observed for a drug-free interval period of approximately 30 days to ensure they are fully prepared for the subsequent surgery. In the current cohort, two patients experienced a relevant long drug-free interval of 86 and 93 days, because of the Chinese Lunar New Year holiday and COVID-19 pandemic since January 2020 [35]. However, after control of COVID-19 outbreak in China [36], both of them were admitted and underwent lobectomy and sleeve lobectomy.
The study involved several limitations. The present retrospective observational study has inherent limitations. Although there is an increasing number of patients who underwent surgery following immunotherapy, sufficiently large sample size report is lacking, and enough experiences still expected. Future research including prospective data and follow-up assessments is in progress.
Conclusively, pulmonary resection after neoadjuvant immunotherapy or chemo-immunotherapy for resectable NSCLC appears to be safe with low operative mortality and morbidity rate in the current population. Because of possible post-neoadjuvant treatment adhesions or fibrosis, concerns that surgical treatment for these patients might be challenging have raised. However, based on the current data, difficulty of operation appears to be similar to those treated with other neoadjuvant agents. Future studies with larger sample numbers will address new perspectives in the setting of neoadjuvant immunotherapy or chemo-immunotherapy.
Acknowledgements
We wish to thank Prof Pamela Derish, Scientific Publications Manager of UCSF Department of Surgery (University of California, San Francisco), for language editing. The Institutional Review Board of Shanghai Chest Hospital approved the retrospective analysis of anonymous patient data.
Author contributions
LJ, JH, SSJ and QQL conceived the presented idea and designed the work. LJ, WWR and YFS conducted the data collection. LJ, CWL and YT performed the data analysis and interpretation. LJ and JWN drafted the article. LJ, XKC, YHY, ZPD and QQL conducted the operative procedures. ZML prescribed the neoadjuvant immunotherapy or chemo-immunotherapy. QQL encouraged to investigate and supervised the findings of this work. All authors discussed the results and contributed to the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81702251, 81972176 and 81800073), the Science Foundation of Shanghai (No.18ZR1435100) and Shanghai Hospital Development Center (SHDC12016113).
Data availability material
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no competing interests.
Informed consent
Not required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Long Jiang, Jia Huang, Shanshan Jiang, Wenwen Rong and Yaofeng Shen have contributed equally to this work.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30. doi: 10.3322/caac.21590. [DOI] [PubMed] [Google Scholar]
- 2.Bagcchi S. Lung cancer survival only increases by a small amount despite recent treatment advances. Lancet Respir Med. 2017;5:169. doi: 10.1016/S2213-2600(17)30041-3. [DOI] [PubMed] [Google Scholar]
- 3.Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553:446–54. doi: 10.1038/nature25183. [DOI] [PubMed] [Google Scholar]
- 4.Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ, Jr, Wu YL, Paz-Ares L. Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389:299–311. doi: 10.1016/S0140-6736(16)30958-8. [DOI] [PubMed] [Google Scholar]
- 5.Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced nonsmall-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 Study. J Clin Oncol. 2019;37:2518–27. doi: 10.1200/JCO.19.00934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gandhi L, Rodriguez-Abreu D, Gadgeel S, et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med. 2018;378:2078–92. doi: 10.1056/NEJMoa1801005. [DOI] [PubMed] [Google Scholar]
- 7.DeBerardinis RJ. Tumor microenvironment, metabolism, and immunotherapy. N Engl J Med. 2020;382:869–71. doi: 10.1056/NEJMcibr1914890. [DOI] [PubMed] [Google Scholar]
- 8.Lin JJ, Shaw AT. Raising the bar on first-line immunotherapy in lung cancer. Lancet Oncol. 2017;18:2–3. doi: 10.1016/S1470-2045(16)30594-0. [DOI] [PubMed] [Google Scholar]
- 9.Ettinger DS, Wood DE, Aggarwal C, et al. NCCN guidelines Insights: non-small cell lung cancer, version 1.2020. J Natl Compr Canc Netw. 2019;17:1464–72. doi: 10.6004/jnccn.2019.0059. [DOI] [PubMed] [Google Scholar]
- 10.Forde PM, Chaft JE, Smith KN, et al. Neoadjuvant PD-1 blockade in resectable lung cancer. N Engl J Med. 2018;378:1976–86. doi: 10.1056/NEJMoa1716078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bott MJ, Yang SC, Park BJ, et al. Initial results of pulmonary resection after neoadjuvant nivolumab in patients with resectable non-small cell lung cancer. J Thorac Cardiovasc Surg. 2019;158:269–76. doi: 10.1016/j.jtcvs.2018.11.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Provencio M, Nadal E, Insa A, et al. OA01.05 phase II study of neo-adjuvant chemo/immunotherapy for resectable stages IIIA non-small cell lung cancer- Nadim study-SLCG. J Thoracic Oncol. 2018;13:320. doi: 10.1016/j.jtho.2018.08.236. [DOI] [Google Scholar]
- 13.Altorki N, Borczuk A, Saxena A, et al. P2.04–92 Neoadjuvant Durvalumab with or without sub-ablative stereotactic radiotherapy (SBRT) in patients with resectable NSCLC ( NCT02904954) J Thoracic Oncol. 2019;14:746. doi: 10.1016/j.jtho.2019.08.1597. [DOI] [Google Scholar]
- 14.Shu CA, Gainor JF, Awad MM, et al. Neoadjuvant atezolizumab and chemotherapy in patients with resectable non-small-cell lung cancer: an open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol. 2020;21:786–95. doi: 10.1016/S1470-2045(20)30140-6. [DOI] [PubMed] [Google Scholar]
- 15.Cascone T, William WN, Weissferdt A, et al. Neoadjuvant nivolumab (N) or nivolumab plus ipilimumab (NI) for resectable non-small cell lung cancer (NSCLC): clinical and correlative results from the NEOSTAR study. J Clin Oncol. 2019;37:8504. doi: 10.1200/JCO.2019.37.15_suppl.8504. [DOI] [Google Scholar]
- 16.Kwiatkowski DJ, Rusch VW, Chaft JE, et al. Neoadjuvant atezolizumab in resectable non-small cell lung cancer (NSCLC): Interim analysis and biomarker data from a multicenter study (LCMC3) J Clin Oncol. 2019;37:8503. doi: 10.1200/JCO.2019.37.15_suppl.8503. [DOI] [Google Scholar]
- 17.Provencio M, Nadal E, Insa A, et al. Neoadjuvant chemo-immunotherapy for the treatment of stage IIIA resectable non-small-cell lung cancer (NSCLC): A phase II multicenter exploratory study—final data of patients who underwent surgical assessment. J Clin Oncol. 2019;37:8509. doi: 10.1200/JCO.2019.37.15_suppl.8509. [DOI] [Google Scholar]
- 18.Broderick SR. Adjuvant and neoadjuvant immunotherapy in non-small cell lung cancer. Thorac Surg Clin. 2020;30:215–20. doi: 10.1016/j.thorsurg.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 19.Broderick SR, Bott MJ. Neoadjuvant immunotherapy in patients with resectable non-small cell lung cancer. J Thorac Cardiovasc Surg. 2019;158:1471–4. doi: 10.1016/j.jtcvs.2019.06.114. [DOI] [PubMed] [Google Scholar]
- 20.Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung cancer staging project: proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11:39–51. doi: 10.1016/j.jtho.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 21.Fernandez FG, Falcoz PE, Kozower BD, Salati M, Wright CD, Brunelli A. The society of thoracic surgeons and the European Society of Thoracic Surgeons general thoracic surgery databases: joint standardization of variable definitions and terminology. Ann Thorac Surg. 2015;99:368–76. doi: 10.1016/j.athoracsur.2014.05.104. [DOI] [PubMed] [Google Scholar]
- 22.Hellmann MD, Chaft JE, William WN, Jr, et al. Pathological response after neoadjuvant chemotherapy in resectable non-small-cell lung cancers: proposal for the use of major pathological response as a surrogate endpoint. Lancet Oncol. 2014;15:e42–50. doi: 10.1016/S1470-2045(13)70334-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Popovic A, Jaffee EM, Zaidi N. Emerging strategies for combination checkpoint modulators in cancer immunotherapy. J Clin Invest. 2018;128:3209–18. doi: 10.1172/JCI120775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Havel JJ, Chowell D, Chan TA. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer. 2019;19:133–50. doi: 10.1038/s41568-019-0116-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359:1350–5. doi: 10.1126/science.aar4060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Non-small Cell Lung Cancer Collaborative Group Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. Non-small Cell Lung Cancer Collaborative Group BMJ. 1995;311:899–909. [PMC free article] [PubMed] [Google Scholar]
- 27.Gilligan D, Nicolson M, Smith I, et al. Preoperative chemotherapy in patients with resectable non-small cell lung cancer: results of the MRC LU22/NVALT 2/EORTC 08012 multicentre randomised trial and update of systematic review. Lancet. 2007;369:1929–37. doi: 10.1016/S0140-6736(07)60714-4. [DOI] [PubMed] [Google Scholar]
- 28.Hellmann MD, Nathanson T, Rizvi H, et al. Genomic features of response to combination immunotherapy in patients with advanced non-small-cell lung cancer. Cancer Cell. 2018;33(843–52):e4. doi: 10.1016/j.ccell.2018.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Eichhorn F, Klotz LV, Bischoff H, Thomas M, Lasitschka F, Winter H, Hoffmann H, Eichhorn ME. Neoadjuvant anti-programmed death-1 immunotherapy by pembrolizumab in resectable nodal positive stage II/IIIa non-small-cell lung cancer (NSCLC): the NEOMUN trial. BMC Cancer. 2019;19:413. doi: 10.1186/s12885-019-5624-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gao S, Li N, Gao S, et al. Neoadjuvant PD-1 inhibitor (Sintilimab) in NSCLC. J Thorac Oncol. 2020;15:816–26. doi: 10.1016/j.jtho.2020.01.017. [DOI] [PubMed] [Google Scholar]
- 31.Kennedy LB, Salama AKS. A review of cancer immunotherapy toxicity. CA Cancer J Clin. 2020;70:86–104. doi: 10.3322/caac.21596. [DOI] [PubMed] [Google Scholar]
- 32.Yang CJ, McSherry F, Mayne NR, et al. Surgical outcomes after neoadjuvant chemotherapy and ipilimumab for non-small cell lung cancer. Ann Thorac Surg. 2018;105:924–9. doi: 10.1016/j.athoracsur.2017.09.030. [DOI] [PubMed] [Google Scholar]
- 33.Thorsteinsson H, Alexandersson A, Oskarsdottir GN, Skuladottir R, Isaksson HJ, Jonsson S, Gudbjartsson T. Resection rate and outcome of pulmonary resections for non-small-cell lung cancer: a nationwide study from Iceland. J Thorac Oncol. 2012;7:1164–9. doi: 10.1097/JTO.0b013e318252d022. [DOI] [PubMed] [Google Scholar]
- 34.Dy GK, Bogner PN, Tan W, et al. Phase II study of perioperative chemotherapy with cisplatin and pemetrexed in non-small-cell lung cancer. J Thorac Oncol. 2014;9:222–30. doi: 10.1097/JTO.0000000000000062. [DOI] [PubMed] [Google Scholar]
- 35.Pan A, Liu L, Wang C, et al. Association of public health interventions with the epidemiology of the COVID-19 outbreak in Wuhan. China: JAMA; 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen S, Yang J, Yang W, Wang C, Barnighausen T. COVID-19 control in China during mass population movements at New Year. Lancet. 2020;395:764–6. doi: 10.1016/S0140-6736(20)30421-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.