Abstract
Senescence marker protein-30 (SMP30), a novel ageing marker, suppresses oxidative stress in the liver. However, studies on phytochemical-mediated regulation of SMP30 expression are lacking. Here, we showed that epigallocatechin gallate (EGCg), a polyphenol abundant in green tea, positively regulates SMP30 expression in the rat hepatoma-derived Fao cells. EGCg maintained SMP30 expression even in the presence of cycloheximide, a protein synthesis inhibitor. Furthermore, treatment of cells with tert-butyl hydroperoxide (tert-BHP), an oxidative promoter, decreased SMP30 expression and ERK1/2 phosphorylation, while EGCg treatment inhibited these effects. Male mice (7-week-old) were divided into 4 groups—Control (saline), tert-BHP (1.5 mmol/kg tert-BHP), EGCg + tert-BHP (30 mg/kg/day of EGCg and 1.5 mmol/kg tert-BHP), and EGCg (30 mg/kg/day). After oral EGCg administration for 6 consecutive days, EGCg + tert-BHP group mice were administered tert-BHP. The tert-BHP-administered mice showed decreased SMP30 expression in the liver and increased aspartate aminotransferase and alanine transaminase (hepatic injury marker enzymes) activities; however, EGCg treatment attenuated these changes. Thus, EGCg-induced SMP30 upregulation may alleviate tert-BHP-induced liver injury. The findings of this study offer new perspectives of the anti-ageing properties of EGCg.
Keywords: senescence marker protein-30, epigallocatechin gallate, tert-BHP-induced liver injury
Introduction
Ageing is a physiological phenomenon in all living species characterized by a progressive loss of physiological integrity, leading to impaired body functions.(1,2) Ageing is accompanied by an increased frequency of age-related disorders including cardiovascular disease, neurodegeneration, cancer, and osteoporosis.(3–7) The fundamental mechanism of ageing is related to the process of cellular senescence. Senescent cells are considered to be in a state of irreversible growth arrest, although they remain metabolically active.(8) Senescence is caused by increased oxidative stress, including hydrogen peroxide, genetic toxicity, and oncogene activation. Therefore, we must first understand how to suppress oxidative stress to understand the mechanisms driving ageing.
Senescence marker protein-30 (SMP30), also known as regucalcin, is a 34 kDa protein. The gene encoding SMP30 is located on the X chromosome and was originally discovered in the rat liver.(9) The SMP30 is highly conserved in various species, including human, mouse, and rat, and is highly expressed in the liver and kidney.(10) SMP30 exhibits gluconolactonase activity, involving it in l-ascorbic acid biosynthesis in the liver, while it also suppresses oxidative stress.(11–13) In fact, acute oxidative stress such as carbon tetrachloride treatment suppresses SMP30 expression in the liver.(14) In addition, SMP30-knockout mice show increased reactive oxygen species (ROS) accumulation and a decreased lifespan, and exhibit distinct ageing-associated characteristics.(15) In contrast, dietary restriction reduces oxidative stress, prolongs lifespan, and increases SMP30 expression.(16) Handa et al.(17) reported that SMP30 over-expression decreases ROS formation and antioxidant enzyme activities, including superoxide dismutase (SOD) and glutathione (GSH), in hepatic carcinoma HepG2 cells. Thus, SMP30 may play a role in anti-ageing by suppressing oxidative stress. However, to the best of our knowledge, there has been no research on the regulation of SMP30 expression using phytochemicals.
Tea and tea polyphenols have been reported to possess potent radical scavenging and anti-inflammatory effects.(18,19) Specially, the most abundant polyphenol in green tea, (–)-epigallocatechin-3-gallate (EGCg), has diverse pharmacological activities, and is preventative against several chronic diseases including cancer, inflammation, diabetes, fatty liver disease, and neurodegenerative diseases.(20–24) It also protects cells from ROS-induced injury.(25) EGCg could reduce oxidative stress and cell damage by increasing SMP30 expression. However, there is little knowledge about the effect of EGCg on SMP30 expression.
The aim of this study was to determine whether EGCg positively regulates SMP30 expression and prevents liver injury due to tert-butyl hydroperoxide (tert-BHP), an oxidative stress inducer in Fao cells (in vitro) and in mice (in vivo).
Materials and Methods
Reagents
EGCg (>98.0%) was purchased from Nacalai Tesque (Kyoto, Japan) for use in both in vitro and in vivo examinations. Cycloheximide (CHX) was purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). tert-BHP and N-acetyl cysteine (NAC) were obtained from Sigma-Aldrich (St. Louis, MO).
Cell culture
Fao rat liver cell line was cultured in DMEM/F-12 Ham’s (Gibco BRL/Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (Biowest, Nuaillé, France), 100 U/ml penicillin, and 100 µg/ml streptomycin (Gibco BRL/Invitrogen) at 37°C in 5% CO2.
Cell viability assay
Fao cells (1.0 × 105 cells/well) were cultured in 96-well plates. Cells were treated with various concentrations of EGCg for 24 h. Cell viability was assessed using the Cell Counting Kit-8 (CCK-8; DOJINDO, Kumamoto, Japan). The effect of EGCg on cell viability was expressed as percent cell viability, with that of cells treated with a methanol vehicle set at 100%.
Cytotoxicity evaluation (lactate dehydrogenase assay)
Fao cells (1.0 × 105 cells/well) were cultured in 96-well plates. Cells were treated with various EGCg concentrations for 24 h. After 24 h, 50 µl of culture medium from each well was collected and transferred to another 96-well culture plate. A total of 50 µl of lactate dehydrogenase (LDH) reagent (DOJINDO, Kumamoto, Japan) was added to each well and the solution was incubated at room temperature for 30 min. After the reaction was complete, absorbance at 490 nm was measured using a microplate reader. The effect of EGCg on decreasing LDH release was expressed as a percent with tert-BHP-treated cells set at 100%.
Animal studies
This study was approved by the Tokyo University of Agriculture Animal Use Committee, and mice were maintained in accordance with the guidelines of the university for the care and use of laboratory animals. A total of 28 specific pathogen-free male ICR mice (7 weeks old, 32–34 g) were obtained from a commercial animal breeder (Japan SLC, Inc., Hamamatsu, Japan) and acclimated for one week in our facility prior to the experiment. All animals were housed in a controlled environment at 22 ± 2°C and 60 ± 5% relative humidity under a 12/12 h light/dark cycle. All mice were fed the same amount of AIN-93G diet with ad-libitum access to tap water. Mice were randomly divided into four groups (n = 7 for each group)—Control (saline injection), tert-BHP (1.5 mmol/kg tert-BHP injection), EGCg + tert-BHP (30 mg/kg/day of EGCg and 1.5 mmol/kg tert-BHP injection), and EGCg (30 mg/kg/day of EGCg). EGCg and tert-BHP were dissolved in saline. Saline alone or saline with EGCg was administered by oral gavage to mice for six consecutive days before tert-BHP injection. At 24 h after the final oral administration of EGCg, severe acute liver injury was induced by intraperitoneally injecting 1.5 mmol/kg tert-BHP dissolved in saline. Saline alone was also intraperitoneally administered to the control and EGCg groups. Mice were sacrificed 24 h after intraperitoneal administration and whole blood was collected via an abdominal vein under three types of mixed anesthesia. The liver was removed and immediately weighed before fixing or storing at −80°C until downstream western blot analysis.
Western blot analysis
Liver homogenate was prepared by homogenizing 0.5 g of liver in 1.5 ml of RIPA buffer [10 mM Tris-HCl, pH 7.6, 150 mM sodium chloride, 0.1% (w/v) sodium deoxycholate, 0.1% (w/v) sodium dodecyl sulfate, 1.0% (w/v) NP-40 substitute]. Fao cells were washed twice with PBS and lysed with RIPA buffer. Samples were separated using SDS-PAGE before transfer to a PVDF membrane. The membranes were blocked with 5% bovine serum albumin in Tris-buffered saline with 0.05% Tween-20 (TBST) before incubating with anti-SMP30 (Santa Cruz Biotechnology, Dallas, TX), anti-ERK1/2, anti-phospho-ERK1/2 (Thr202/Tyr204) (Cell Signaling Technology, Danvers, MA), and β-actin (Santa Cruz Biotechnology) antibodies in TBST. Next, membranes were incubated with HRP-conjugated secondary antibodies (Santa Cruz Biotechnology) in TBST before visualization using the EzWestLumi Plus (ATTO, Tokyo, Japan). Band intensities were quantified using ImageJ (National Institutes of Health, Bethesda, MD).
Aspartate aminotransferase and alanine transaminase measurement
Blood was collected from the inferior vena cava and serum was obtained by centrifuging at 3,000 × g for 10 min at 4°C. Aspartate aminotransferase (AST) and alanine transaminase (ALT) (FUJIFILM Wako Pure Chemical Corporation, Osaka, Japan) were used as biochemical markers of hepatotoxicity and were determined using a commercially available spectrophotometric assay (λmax = 555 nm) according to the manufacturer’s instructions.
Statistical analysis
Results are presented as mean ± SE. All data presented represent the mean of three independent experiments. Multiple comparisons were performed using Tukey’s test or Tukey-Kramer’s test after one-way analysis of variance (ANOVA). P<0.05 was considered statistically significant.
Results
EGCg regulated SMP30 expression in a dose-dependent manner
Fao cells were treated with EGCg at various concentrations, and cell viability was evaluated using the CCK-8 assay. As shown in Fig. 1A, EGCg did not induce toxicity in Fao cells. The results of this assay indicated that the maximum concentration of EGCg that can be used in our subsequent studies was 100 µM. Next, we examined SMP30 expression after EGCg treatment. Interestingly, EGCg treatment caused an increase in SMP30 expression in a dose-dependent manner until 50 µM, but the upregulation stopped at 100 µM (Fig. 1B). Subsequently, we used 30 µM of EGCg as the maximum concentration for further experiments.
Fig. 1.
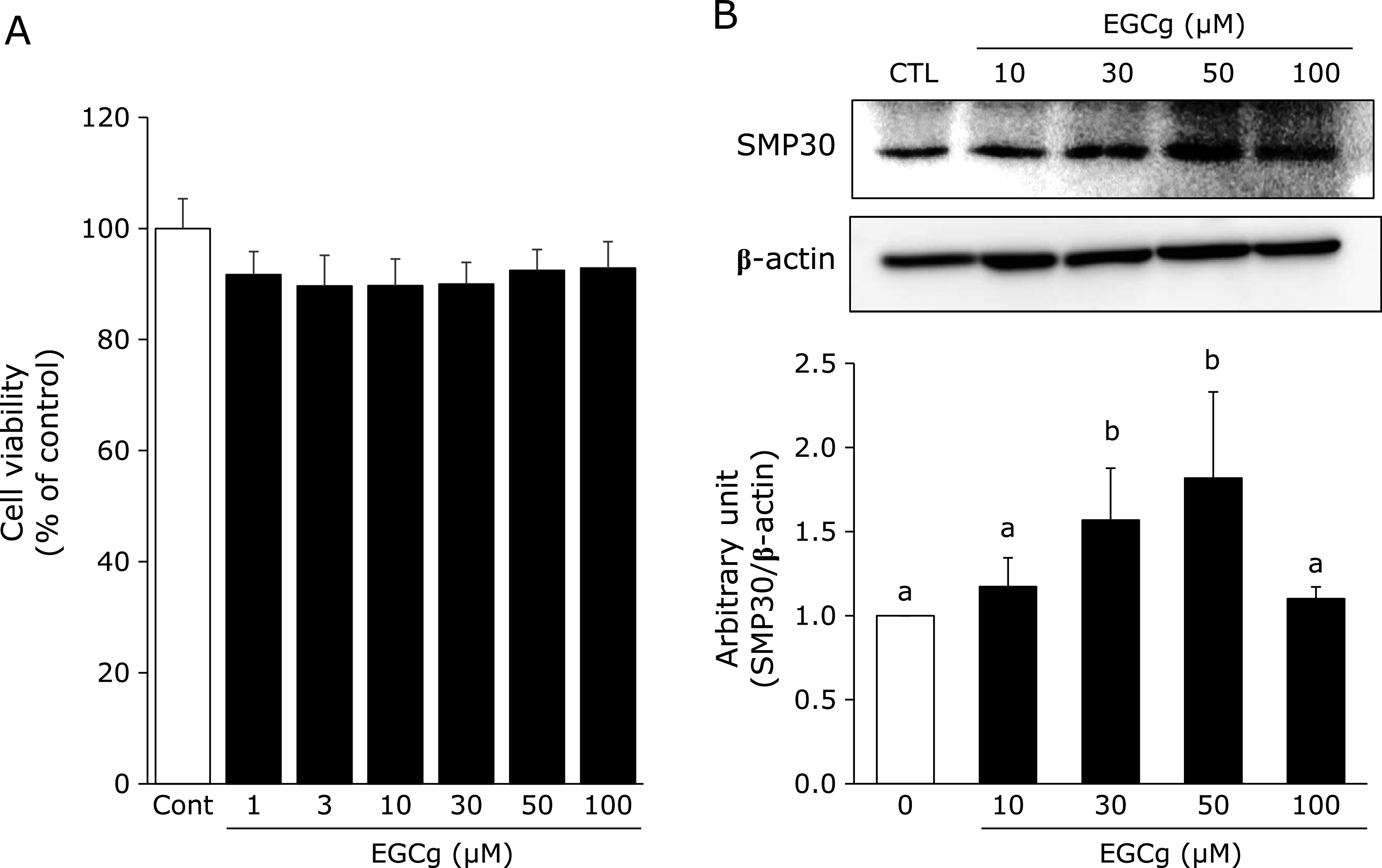
Effects of EGCg on cell toxicity and SMP30 expression. (A) Cell viability of EGCg: Fao cells (1.0 × 105 cells/well) were cultured in a 96-well plate before treating with various EGCg concentrations for 24 h. Cell viability was assessed using the CCK-8 assay. Cell viability was expressed as a percentage of the values obtained for EGCg-untreated cells. (B) Protein expression of SMP30: Fao cells were treated with EGCg (0–100 µM) for 24 h, and the cell lysate was immunoblotted with each indicated antibody. Densitometry quantification of SMP30 was performed using ImageJ. SMP30 levels were normalized to β-actin. Data are shown as the means ± SE from at least three independent experiments. Unmatching letters denote significantly different, p<0.05.
EGCg promotes SMP30 stabilization
To determine whether EGCg treatment results in SMP30 stability, Fao cells were pre-treated with CHX, an inhibitor of protein synthesis. Cells were treated with EGCg or a vehicle and protein lysates were collected after 24 h. EGCg was able to promote SMP30 stabilization even in CHX-co-treated cells (Fig. 2A and B).
Fig. 2.
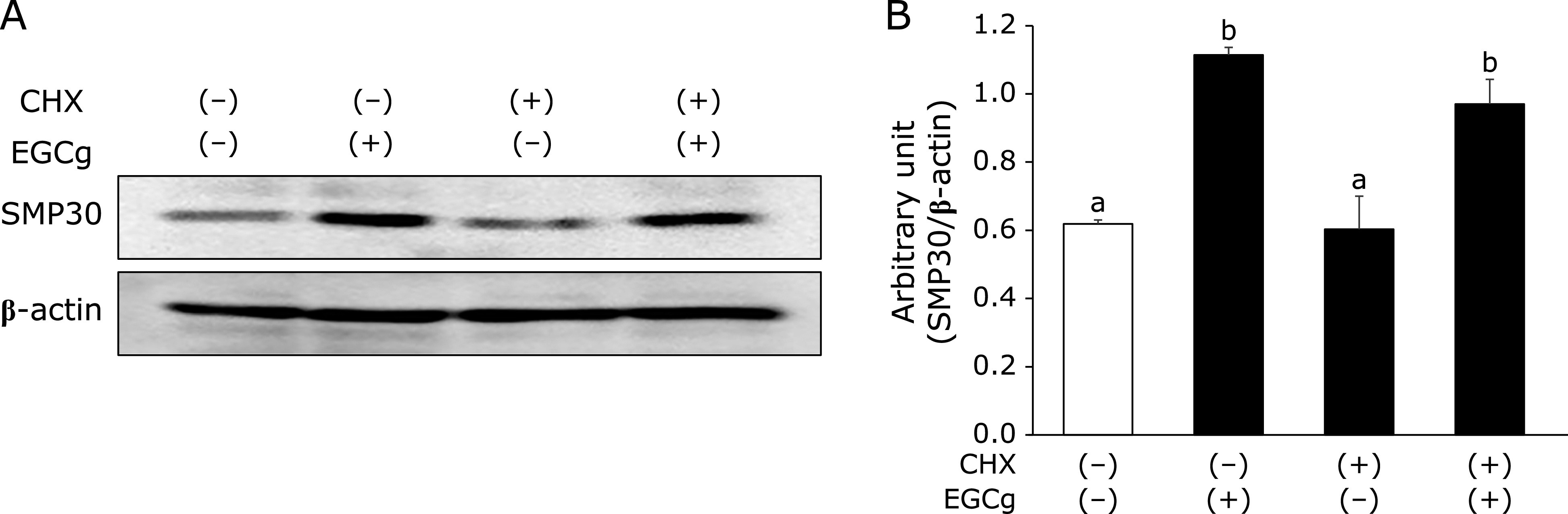
Effects of EGCg on SMP30 up-regulation and stabilization in Fao cells. (A) Fao cells were pre-treated with or without CHX (10 mg/ml) for 0.5 h before stimulating with or without 30 µM EGCg for 24 h. (B) Densitometry quantification was performed using ImageJ. SMP30 levels were normalized to β-actin. Data are shown as the means ± SE from at least three independent experiments. Unmatching letters denote significantly different, p<0.05.
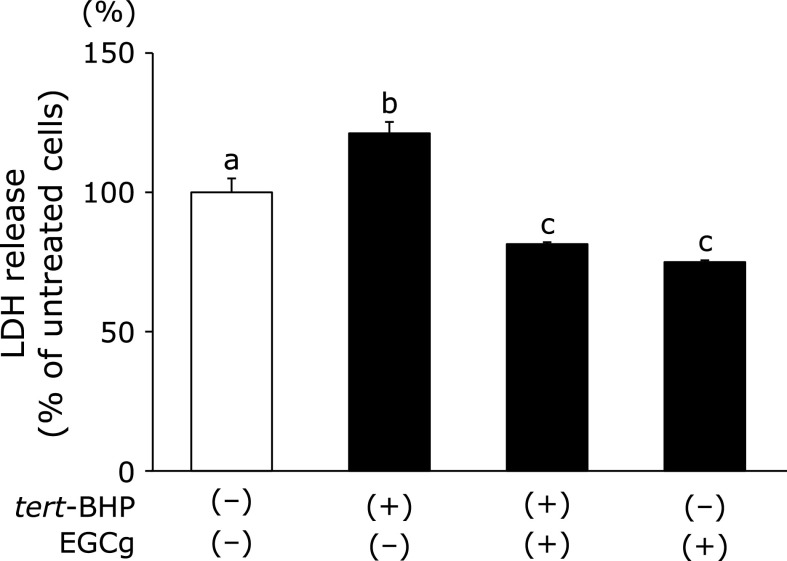
EGCg attenuates LDH release in tert-BHP-treated cells
Next, we examined whether EGCg attenuates liver injury in tert-BHP-treated cells. LDH release, which is a marker of intracellular damage, was measured in Fao cells. Fao cells were pre-treated with EGCg to assess the protective effect of EGCg against tert-BHP induced cell damage. Although LDH release was observed after stimulation with tert-BHP treatment, LDH release was lower in the EGCg-treated cells than in the cells treated with tert-BHP alone (Fig. 3).
Fig. 3.
Effects of EGCg on LDH release with tert-BHP-induced oxidative stress in Fao cells. Fao cells were pre-treated with 30 µM EGCg for 8 h. After incubating with EGCg, cells were treated with 100 µM tert-BHP with or without 30 µM EGCg for 24 h. Quantitative LDH level analyses were performed in the cell culture supernatant. Data are shown as the means ± SE from at least three independent experiments. Unmatching letters denote significantly different, p<0.05.
EGCg attenuates the downregulation of SMP30 and ERK1/2 phosphorylation in tert-BHP-treated cells
Previously, Jung et al.(16) reported that downregulation of SMP30 is likely involved in the ERK signaling pathway. In addition, tert-BHP induces ERK1/2 phosphorylation.(26) Thus, we examined whether EGCg treatment increases SMP30 expression and/or suppresses ERK1/2 phosphorylation in Fao cells. Interestingly, tert-BHP downregulated SMP30 expression and induced ERK1/2 phosphorylation, while EGCg pre-treatment attenuated these effects (Fig. 4A and B). Furthermore, decreased SMP30 expression is accompanied by increased ROS generation.(27) Therefore, we assessed whether NAC, a nitroxide antioxidant reagent, could attenuate the downregulation of SMP30 expression by tert-BHP treatment in Fao cells, since nitroxides are known to have high anti-oxidative activities.(28) The results indicated that SMP30 expression was downregulated in tert-BHP-treated Fao cells with 30 µM EGCg, while treatment with 0.5 mM NAC increased SMP30 expression (Fig. 4C and D).
Fig. 4.
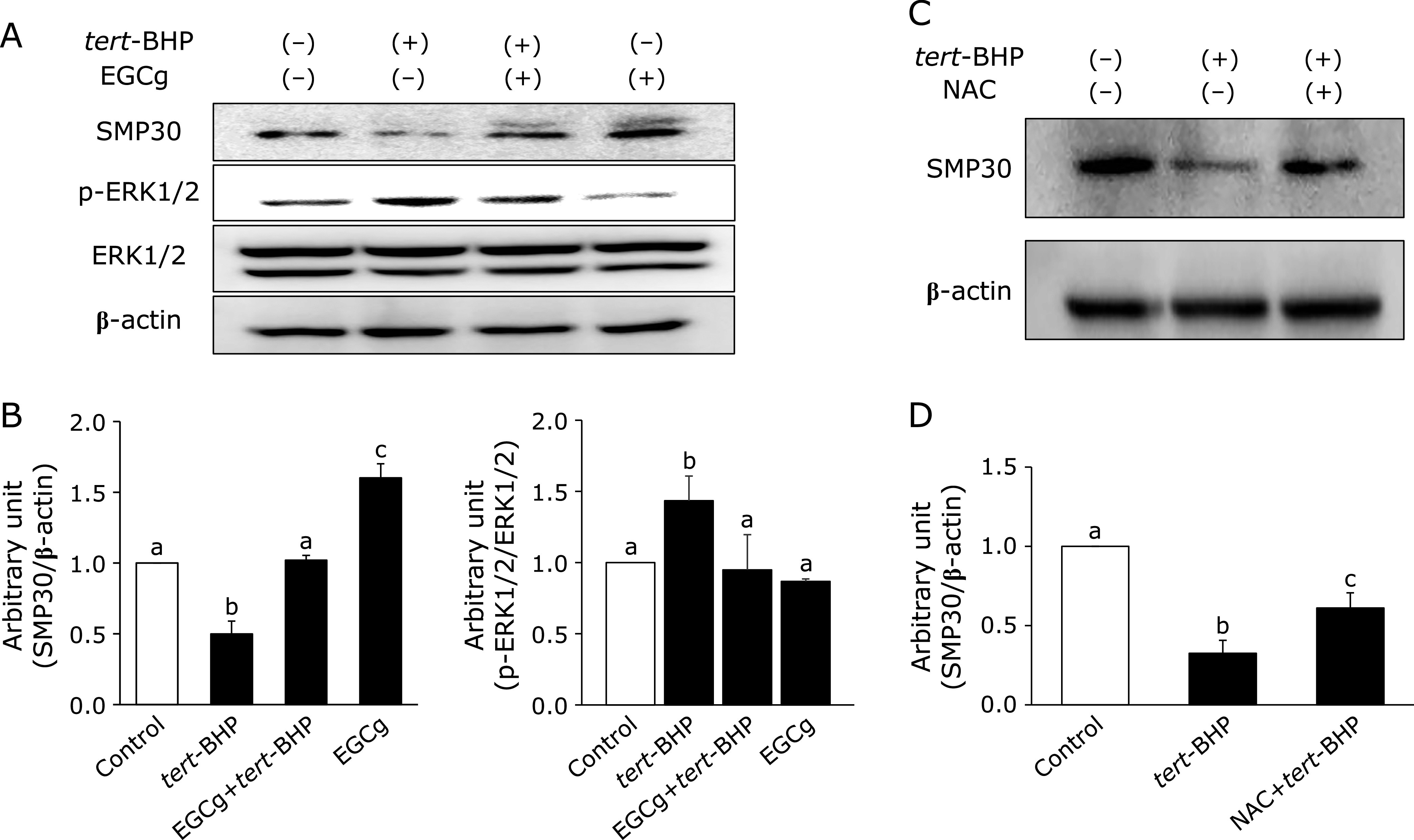
Effects of EGCg on SMP30 expression and ERK1/2 phosphorylation with tert-BHP-treated Fao cells. (A) Cells were pre-treated with 30 µM EGCg for 8 h. After incubating with EGCg, cells were treated with 100 µM tert-BHP with or without 30 µM EGCg for 24 h. Cells were homogenized using RIPA buffer, and the supernatants were separated to measure SMP30, phosphorylated ERK1/2 (activation form), and ERK1/2 expression. (B) Densitometry quantification was performed using ImageJ. SMP30 levels were normalized to β-actin. p-ERK1/2 levels (Thr202/Tyr204) were normalized to ERK1/2. (C) Cells were treated with 100 µM tert-BHP with/without 0.5 mM NAC for 24 h. The supernatants were separated for SMP30 expression measurement. (D) Densitometry quantification was performed using ImageJ. SMP30 levels were normalized to β-actin. Data are shown as the means ± SE from at least three independent experiments. Unmatching letters denote significantly different, p<0.05.
EGCg attenuates the downregulation of SMP30 and liver injury in tert-BHP-administered mice
tert-BHP has been used to induce oxidative stress in previous in vivo and in vitro studies.(29,30) As shown in Fig. 5A, we first examined the effect of EGCg oral administration (30 mg/kg/day) for 1 week on SMP30 expression and serum AST and ALT levels, which reflect hepatic functions, in tert-BHP-administered (1.5 mmol/kg) mice. There were no significant differences in food intake, final body weight, or organ weight between the control and the tert-BHP-administered mice with or without EGCg (Table 1). Hepatic SMP30 expression in the tert-BHP-administered group was decreased compared to that in the control group, while that in the EGCg oral administration group was unchanged (Fig. 5B). Subsequently, AST and ALT levels were higher in the tert-BHP group than in the control group, while EGCg oral administration significantly attenuated this increase (Fig. 5C and D). Importantly, in the EGCg group, liver injury did not occur and liver SMP30 expression was not increased.
Fig. 5.
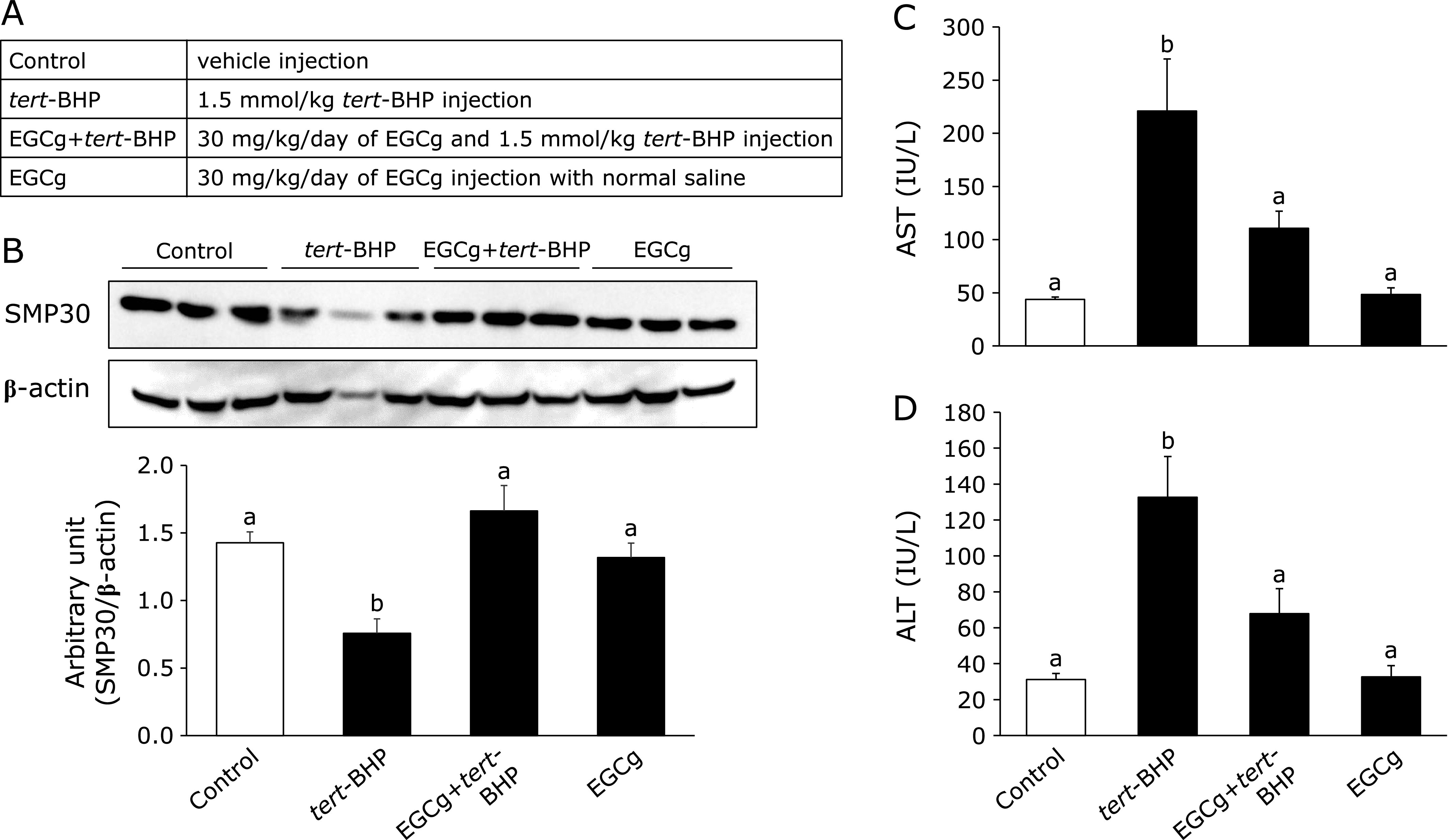
Effects of EGCg on SMP30 expression and hepatic toxicity in tert-BHP-injected mice. (A) All groups were fed an AIN93G diet as a basal diet and given tap water for 7 days. EGCg (30 mg/kg body weight) was orally administered to the EGCg and EGCg + tert-BHP groups for 6 days. After 6 days, the tert-BHP and EGCg + tert-BHP groups were intraperitoneally injected with tert-BHP for 24 h. (B) Livers were homogenized using RIPA buffer, and the supernatants were separated for SMP30 expression measurement. Densitometry quantification was performed using ImageJ. SMP30 levels were normalized to β-actin. Data are shown as the means ± SE. (C) Blood was collected and serum separated to determine serum AST and (D) ALT levels. Unmatching letters denote significantly different, p<0.05.
Table 1.
Food intake, body weight, and organ weights
| Control | tert-BHP | EGCg + tert-BHP | EGCg | |
|---|---|---|---|---|
| Total food intake (g) | 47.19 ± 0.90 | 45.72 ± 1.78 | 46.73 ± 1.82 | 46.69 ± 1.14 |
| Final body weight (g) | 36.93 ± 2.30 | 36.55 ± 1.62 | 36.92 ± 2.73 | 36.81 ± 1.39 |
| Liver weight (g/100 g body weight) | 1.891 ± 0.05 | 1.933 ± 0.06 | 1.947 ± 0.08 | 1.871 ± 0.08 |
| Kidney weight (g/100 g body weight) | 0.595 ± 0.03 | 0.600 ± 0.04 | 0.624 ± 0.02 | 0.623 ± 0.02 |
Data are shown as the means ± SE (n = 7 each).
Discussion
In the present study, we assessed whether EGCg positively regulates SMP30 expression and prevents liver injury in vitro and in vivo. Here, we indicated that Fao liver cell lines treated with EGCg have increased SMP30 expression. It has previously been reported that SMP30 expression is downregulated by increased ROS.(14,27) In contrast, EGCg possesses free radical-scavenging activity and inhibits protein aggregation via proteasomal degradation.(31,32) Here, we determined how EGCg upregulates SMP30 expression. In fact, we found that EGCg can upregulate SMP30 expression even when cells are treated with CHX, an inhibitor of protein synthesis. Therefore, these data propose that the ubiquitin-proteasome pathway is involved in regulating the turnover of SMP30 and that EGCg increases SMP30 stability via post-translational mechanisms, resulting in decreased proteasome-dependent protein degradation. To support this hypothesis, it was reported that ester bond-containing green tea polyphenols, including EGCg, potently inhibit cellular proteasome activity.(33) Thus, we suggest a novel mechanism in which EGCg acts to regulate SMP30 stabilization at the intracellular level.
As previously described, tert-BHP, an oxidant reagent, can induce acute liver injury.(29,30) To confirm the positive effect of EGCg under oxidative stress conditions, SMP30 expression was measured in tert-BHP-treated Fao cells. We observed that EGCg treatment attenuated the downregulation of SMP30 expression in tert-BHP-treated cells.
Jung et al.(16) reported that the downregulation of SMP30 expression is likely involved in the ERK signaling pathway. Thus, we examined whether EGCg attenuated ERK1/2 activation and inhibited the downregulation of SMP30 expression due to tert-BHT treatment. In fact, our data suggested that EGCg upregulates SMP30 expression by inactivating ERK1/2 signaling. Interestingly, over-expression of SMP30 in HepG2 cells contributes to a marked decrease in ROS formation accompanied by decreased lipid peroxidation, SOD activity and GSH, whereas SMP30 itself does not exert radical scavenging ability.(17) In contrast, SMP30 protects intestinal epithelial cells from inflammation-induced cell death by enhancing the activity of Nrf2, a key antioxidant molecule.(34) In addition, EGCg activates Nrf2 signaling and promotes antioxidant enzymes such as SOD and catalase to work against oxidative stress-induced hepatotoxicity in vivo and in vitro.(35) Therefore, we propose that EGCg may be involved in upregulating and stabilizing SMP30 and could act as an antioxidant signal activator in tert-BHP-treated liver cells.
We confirmed that AST and ALT, liver injury markers, are drastically increased in tert-BHP-treated mice compared to that in untreated mice; however, EGCg inhibited the increase in these markers. Of note, we also observed that EGCg administration significantly inhibited the downregulation of SMP30 expression in the liver of tert-BHP-administered mice, suggesting that it plays a role in attenuating tert-BHP-induced liver injury. In contrast, several studies have reported that high-dose EGCg causes hepatotoxicity.(36–39) Here, treating mice with EGCg alone did not modulate AST and ALT levels, and EGCg-treated mice did not increase SMP30 expression in the liver compared to that in the control mice. Thus, oral EGCg administration for 6 days may be insufficient. Another study showed that treatment with EGCg (25 mg/kg/day) for long duration extends lifespan by improving age-associated inflammation and oxidative stress in healthy rats.(40) In any case, we found that EGCg positively regulates SMP30 stabilization at the intracellular level to protect against tert-BHP-induced liver injury. Based on in vitro data, EGCg could regulate SMP30 turnover and stability via post-translational mechanisms.
In conclusion, the present study demonstrated that EGCg upregulates and stabilizes SMP30 and reduces tert-BHP-induced liver injury in vitro. Importantly, our in vivo data also shows that EGCg treatment improves the downregulation of SMP30 and liver injury markers in mice with tert-BHP-induced liver injury. These findings could offer new perspectives for novel therapeutic approaches against age-related diseases by EGCg-induced upregulation and stabilization of SMP30.
Author Contributions
Study concept and design: HI, KA, and MU; acquisition of data: HI, KA; analysis and interpretation of data: HI, KA, and MU; drafting of the manuscript: HI, KA, and MU; critical revision of the manuscript for important intellectual content: MT and NT; statistical analysis: HI, KA; administrative, technical, or material support: RK-T and NT; study supervision: MU.
Acknowledgments
This study was supported in part by JSPS KAKENHI (Grant No. 15H01767). The authors thank T. Takagi and S. Fujii (Tokyo University of Agriculture) for technical assistance.
Abbreviations
- ALT
alanine transaminase
- AST
aspartate aminotransferase
- CHX
cycloheximide
- EGCg
epigallocatechin gallate
- NAC
N-acetyl cysteine
- ROS
reactive oxygen species
- SMP30
senescence marker protein-30
- tert-BHP
tert-butyl hydroperoxide
Conflict of Interest
No potential conflicts of interest were disclosed.
References
- 1.Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of cellular senescence. Trends Cell Biol 2018; 28: 436–453. [DOI] [PubMed] [Google Scholar]
- 2.Baar MP, Brandt RMC, Putavet DA, et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 2017; 169: 132–147.e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.vB Hjelmborg J, Iachine I, Skytthe A, et al. Genetic influence on human lifespan and longevity. Hum Genet 2006; 119: 312–321. [DOI] [PubMed] [Google Scholar]
- 4.Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation 2003; 107: 139–146. [DOI] [PubMed] [Google Scholar]
- 5.Du LY, Chang LY, Ardiles AO, et al. Alzheimer’s disease-related protein expression in the retina of octodon degus. PLoS One 2015; 10: e0135499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16UNK4a. Cell 1997; 88: 593–602. [DOI] [PubMed] [Google Scholar]
- 7.Chen K, Yang YH, Jiang SD, Jiang LS. Decreased activity of osteocyte autophagy with aging may contribute to the bone loss in senile population. Histochem Cell Biol 2014; 142: 285–295. [DOI] [PubMed] [Google Scholar]
- 8.Kwon SM, Hong SM, Lee YK, Min S, Yoon G. Metabolic features and regulation in cell senescence. BMB Rep 2019; 52: 5–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ishigami A, Maruyama N. Significance of SMP30 in gerontology. Geriatr Gerontol Int 2007; 7: 316–325. [Google Scholar]
- 10.Fujita T, Shirasawa T, Uchida K, Maruyama N. Gene regulation of senescence marker 30 (SMP30): coordinated up-regulation with tissue maturation and gradual down-regulation with aging. Mech Ageing Dev 1996; 87: 219–229. [DOI] [PubMed] [Google Scholar]
- 11.Fujita T, Uchida K, Maruyama N. Purification of senescence marker protein-30 (SMP30) and its androgen-independent decrease with age in the rat liver. Biochim Biophys Acta 1992; 1116: 122–128. [DOI] [PubMed] [Google Scholar]
- 12.Kondo Y, Masutomi H, Noda Y, et al. Senescence marker protein-30/superoxide dismutase 1 double knockout mice exhibit increased oxidative stress and hepatic steatosis. FEBS Open Bio 2014; 4: 522–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sato Y, Amano A, Kishimoto Y, et al. Ascorbic acid prevents protein oxidation in livers of senescence marker protein-30/gluconolactonase knockout mice. Geriatr Gerontol Int 2014; 14: 989–995. [DOI] [PubMed] [Google Scholar]
- 14.Feng D, Kondo Y, Ishigami A, Kuramoto M, Machida T. Senescence marker protein-30 as a novel antiaging molecule. Ann N Y Acad Sci 2004; 1019: 360–364. [DOI] [PubMed] [Google Scholar]
- 15.Kondo Y, Inai Y, Sato Y, et al. Senescence marker protein 30 functions as gluconolactoase in L-ascorbic acid biosynthesis, and its knockout mice are prone to scurvy. Proc Natl Acad Sci U S A 2006; 103: 5723–5728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jung KJ, Maruyama N, Ishigami A, Yu BP, Chung HY. The redox-sensitive DNA binding sites responsible for age-related downregulation of SMP30 by ERK pathway and reversal by calorie restriction. Antioxid Redox Signal 2006; 8: 671–680. [DOI] [PubMed] [Google Scholar]
- 17.Handa S, Maruyama N, Ishigami A. Over-expression of senescence marker protein-30 decreases reactive oxygen species in human hepatic carcinoma Hep G2 cells. Biol Pharm Bul 2009; 32: 1645–1648. [DOI] [PubMed] [Google Scholar]
- 18.Nanjo F, Goto K, Seto R, Suzuki M, Sakai M, Hara Y. Scavenging effects of tea catechins and their derivatives on 1,1-diphenyl-2-picrylhydrazyl radical. Free Radic Biol Med 1996; 8: 895–902. [DOI] [PubMed] [Google Scholar]
- 19.Pérez-Cano FJ, Castell M. Flavonoids, inflammation and immune system. Nutrients 2016; 8: 659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chung LY, Cheung TC, Kong SK, et al. Induction of apoptosis by green tea catechins in human prostate cancer DU145 cells. Life Sci 2001; 68: 1207–1214. [DOI] [PubMed] [Google Scholar]
- 21.Hosakawa Y, Hosakawa I, Ozaki K, Nakanishi T, Nakae H, Matsuo T. Tea polyphenols inhibit IL-6 production in tumor necrosis factor superfamily 14-stimulated human gingival fibroblasts. Mol Nutr Food Res 2010; 54 Suppl 2: S151–S158. [DOI] [PubMed] [Google Scholar]
- 22.Hwang JT, Park IJ, Shin JI, et al. Genistein, EGCG, and capsaicin inhibit adipocyte differentiation process via activating AMP-activated protein kinase. Biochem Biophys Res Commun 2005; 338: 694–699. [DOI] [PubMed] [Google Scholar]
- 23.Ushiroda C, Naito Y, Takagi T, et al. Green tea polyphenol (epigallocatechin-3-gallate) improves gut dysbiosis and serum bile acids dysregulation in high-fat diet-fed mice. J Clin Biochem Nutr 2019; 65: 34–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rezai-Zadeh K, Aredash GW, Hou H, et al. Green tea epigallocatechin-3-gallate (EGCG) reduces beta-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res 2008; 1214: 177–187. [DOI] [PubMed] [Google Scholar]
- 25.Nakagawa T, Yokozawa T, Sano M, Takeuchi S, Kim M, Minamoto S. Acitivity of (–)-epigallocatechin 3-O-gallate against oxidative stress in rats with adenine-induced renal failure. J Agric Food Chem 2004; 52: 2103–2107. [DOI] [PubMed] [Google Scholar]
- 26.Li YB, Gao JL, Zhang QW, Hoi PM, Wang YT. Bisdemethoxycurcumin protects endothelial cells against t-BHP-induced cell damage by regulating the phosphorylation level of ERK1/2 and Akt. Int J Mol Med 2011; 27: 205–211. [DOI] [PubMed] [Google Scholar]
- 27.Jung K, Ishigami A, Maruyama N, et al. Modulation of gene expression of SMP-30 by LPS and calorie restriction during aging process. Exp Gerontol 2004; 39: 1169–1177. [DOI] [PubMed] [Google Scholar]
- 28.Shinto S, Matsuoka Y, Yamato M, Yamada KI. Antioxidant nitroxides protect hepatic cells from oxidative stress-induced cell death. J Clin Biochem Nutr 2018; 62: 132–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Garcia-Cohen EC, Marin J, Diez-Picazo LD, Baena AB, Salaices M, Rodriguez-Martinez MA. Oxidative stress induced by tert-butyl hydroperoxide causes vasoconstriction in the aorta from hypertensive and aged rats: role of cyclooxygenase-2 isoform. J Pharmacol Exp Ther 2000; 293: 75–81. [PubMed] [Google Scholar]
- 30.Li YB, Zhong ZF, Chen MW, et al. Bisdemethoxycurcumin increase sirt1 to antagonize t-BHP-induced premature senescence in WI38 fibroblast cells. Evid Based Complement Alternat Med 2013; 2013: 851714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Abd-ElSalam HA, Al-Ghobashy MA, Al-Shorbagy M, Nassar N, Zaazaa HE, Ibrahim MA. Correlation of in vitro and in vivo assay results for assessment of free radical scavenging activity of green tea nutraceuticals. J Food Sci 2016; 81: C1707–C1715. [DOI] [PubMed] [Google Scholar]
- 32.Quan Y, Li L, Dong L, et al. Epigallocatechin-3-gallate (EGCG) inhibits aggregation of pulmonary fibrosis associated mutant surfactant protein A2 via a proteasomal degradation pathway. Int J Biochem Cell Biol 2019; 116: 105612. [DOI] [PubMed] [Google Scholar]
- 33.Kuhn DJ, Burns AC, Kazi A, Dou QP. Direct inhibition of the ubiquitin-proteasome pathway by ester bond-containing green tea polyphenols is associated with increased expression of sterol regulatory element-binding protein 2 and LDL receptor. Biochim Biophys Acta 2004; 1682: 1–10. [DOI] [PubMed] [Google Scholar]
- 34.Choo J, Heo G, Kim SJ, et al. Senescence marker protein 30 protects intestinal epithelial cells against inflammation-induced cell death by enhancing Nrf2 activity. Biochim Biophys Acta Mol Basis Dis 2018; 1864: 3668–3678. [DOI] [PubMed] [Google Scholar]
- 35.Han XD, Zhang YY, Wang KL, Huang YP, Yang ZB, Liu Z. The involvement of Nrf2 in the protective effects of (–)-epigallocatechin-3-gallate (EGCG) on NaAsO2-induced hepatotoxicity. Oncotarget 2017; 8: 65302–65312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Suganuma M, Okabe S, Oniyama M, Tada Y, Ito H, Fujiki H. Wide distribution of [3H] (–)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998; 19: 1771–1776. [DOI] [PubMed] [Google Scholar]
- 37.Mazzanti G, Menniti-Ippolito F, Moro PA, et al. Hepatotoxicity from green tea: a review of the literature and two unpublished cases. Eur J Clin Pharmacol 2009; 65: 331–341. [DOI] [PubMed] [Google Scholar]
- 38.Li GX, Chen YK, Hou Z, et al. Pro-oxidative activities and dose-response relationship of (–)-epigallocatechin-3-gallate in the inhibition of lung cancer growth: a comparative study in vivo and in vitro. Carcinogenesis 2010; 31: 902–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Inoue H, Maeda-Yamamoto M, Nesumi A, Tanaka T, Murakami A. Low and medium but not high dose of green tea polyphenols ameliorated dextran sodium sulfate-induced hepatotoxicity and nephrotoxicity. Biosci Biotechnol Biochem 2013; 77: 1223–1228. [DOI] [PubMed] [Google Scholar]
- 40.Niu Y, Na L, Feng R, et al. The phytochemical, EGCG, extends lifespan by reducing liver and kidney function damage and improving age-associated inflammation and oxidative stress in healthy rats. Aging Cell 2013; 12: 1041–1049. [DOI] [PubMed] [Google Scholar]