Abstract
Objectives
Infection with the SARS-COV2 virus (COVID-19) may be complicated by thrombotic diathesis. This complication often involves the pulmonary microcirculation. While macrovascular thrombotic complications of the lung may include pulmonary artery embolism, pulmonary artery thrombus in situ has also been hypothesized. Pulmonary vein thrombosis has not been described in this context.
Methods/Results
Herein, we provide a case of an otherwise healthy male who developed an ischemic stroke with left internal carotid thrombus. Further imaging revealed pulmonary emboli with propagation through the pulmonary veins into the left atrium. This left atrial thrombus provides a source of atypical “paradoxic arterial embolism”.
Conclusions
Thrombotic outcomes in the setting of severe COVID 19 pneumonia may include macrovascular venous thromboembolism, microvascular pulmonary vascular thrombosis and arterial thromboembolism. Pulmonary vein, herein described, provides further mechanistic pathway for potential arterial embolic phenomenon.
Keywords: Venous thromboembolism, Pulmonary vein thrombosis, Stroke, COVID-19
Highlights
SARS-COV2 (COVID-19) infection may be complicated by thrombotic diathesis.
While the primary site of thrombosis involves the pulmonary microcirculation, macrovascular thrombosis is well described.
Pulmonary artery thrombosis may be either embolic or thrombus in situ
Pulmonary venous thrombus propagation is herein described as a potential source of arterial embolism.
Timely diagnosis and aggressive management may prevent catastrophic embolic complications.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causing COVID-19 primarily causes pulmonary disease but is known to cause extrapulmonary manifestations including a thromboembolic propensity. The systemic inflammatory “cytokine storm” contributes to vascular endothelial injury and coagulation activation [1, 2] which predisposes patients to arterial and venous thrombosis [3]. A recent patient admitted with severe pulmonary involvement of COVID 19 infection complicated by pulmonary vein thrombosis and pulmonary embolism underscores the prothrombotic nature of this infection.
Clinical presentation
A 51-year-old gentleman with no significant past medical history developed cough and fatigue and tested positive for COVID-19 by PCR of nasal swab sample. After a 2 week self-isolation, he was found unresponsive at his home. On examination, he was noted to have right hemiplegia. Initial laboratory assessment revealed leukocytosis (white blood count 16.1X 10(9)/L), polycythemia (hemoglobin 18.0 g/dl), with elevated creatinine kinase (633U/L), and troponin (22 ng/L without significant delta). His D-Dimer was elevated (18,819 ng/ml FEU). Chest Xray showed bilateral pulmonary infiltrates. Non-contrast CT head demonstrated a large middle cerebral artery stroke with hemorrhagic transformation (Fig. 1). Carotid ultrasound demonstrated a non-occlusive thrombus in the left carotid bifurcation extending into the internal carotid artery. A transthoracic echocardiogram revealed a severely enlarged right ventricle, severe pulmonary hypertension and right ventricular strain. CT angiogram chest showed bilateral main pulmonary artery embolus (Fig. 2). A filling defect was noted in the inferior right pulmonary vein extending into the left atrium consistent with pulmonary vein thrombus in cross-sectional (Panel A) and coronal imaging (Fig. 3). Venous ultrasound of lower extremities showed right popliteal vein deep venous thrombosis. Complete thrombophilia testing including lupus anticoagulant, anticardiolipin and beta 2 glycoprotein 1 antibodies were negative. The patient tolerated moderate intensity intravenous unfractionated heparin without bleeding complications and was discharged 25 days later on warfarin therapy. Patient will complete at least 6 months of anticoagulation therapy and follow up with neurology and primary care.
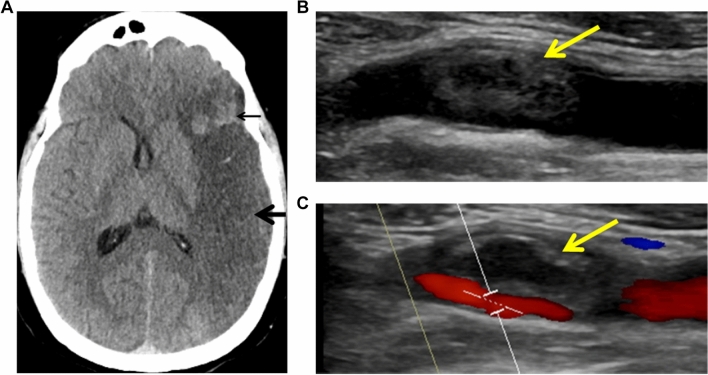
Fig. 1.
Large left middle cerebral artery stroke. A large region of hypoattenuation (Panel a) involving the left frontal, insular, parietal, and temporal lobes consistent with a left MCA territory infarct (thick arrow). An area of associated intraparenchymal hemorrhage in the anterior left frontal lobe is also noted (thin arrow). Near-occlusive thrombus (yellow arrows) in the left carotid bulb extending into the proximal internal carotid artery (Panel b) accentuated by color Doppler (Panel c). Minimal underlying atherosclerotic plaque was noted in the carotid arterial wall
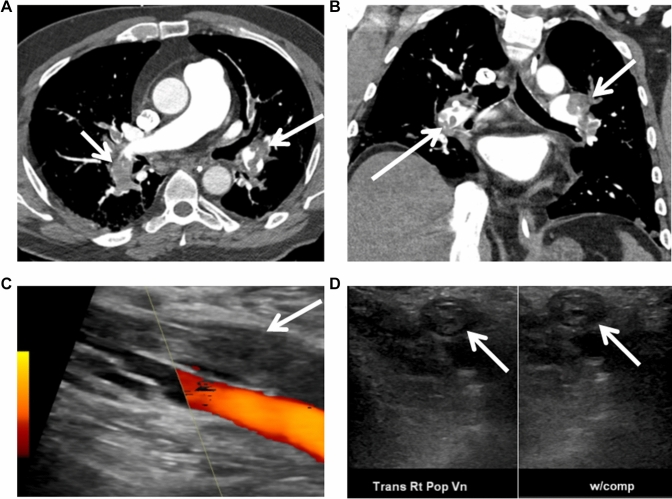
Fig. 2.
Venous thromboembolism. Large burden acute bilateral pulmonary thromboemboli (arrows) are noted in cross-sectional (Panel a) and coronal images (Panel b). Acute deep vein thrombosis is noted in the right popliteal vein (Panel c and d)
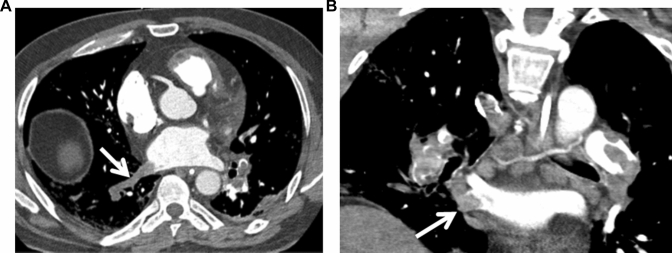
Fig. 3.
Pulmonary vein thrombus. Filling defect in the inferior right pulmonary vein (arrow) consistent with pulmonary vein thrombus in cross-sectional (Panel a) and coronal imaging (Panel b)
Discussion
The reported incidence of arterial and venous thromboembolism (VTE) associated with COVID-19 coagulopathic state varies considerably by author, management, cohort ascertainment (ICU, ward, ambulatory) and imaging strategy [4]. Proposed thromboembolic mechanisms include: (1) complement activation with vascular injury, (2) neutrophil activation with neutrophil extracellular traps (NETs) formation and uncontrolled positive signaling loop between macrophages and neutrophils resulting in a “cytokine storm” and (3) hypoxia mediated activation of hypoxia inducible factors (HIFs). Infection leads to activation of coagulation cascade utilizing humoral and cellular pathways causing microvascular thrombosis contributing to multi-organ failure [5]. COVID-19 patients are also at a risk of arterial thrombosis including ischemic stroke and myocardial infarction. Apart from hypercoagulable state, stress induced cardiomyopathy, septic embolization and atrial fibrillation contribute to cardiovascular morbidity and mortality [6–8].
Pulmonary vein thrombus (PVT) is an extremely rare thrombotic event usually diagnosed after lung surgery, pulmonary malignancy, or as a complication of pulmonary vein isolation for treatment of atrial fibrillation [9]. Patient had no history of cardiothoracic surgery or anatomic abnormality of his pulmonary venous circulation. In this initial case of pulmonary vein thrombosis complicating COVID-19 pneumonia, the underlying thrombotic mechanism may be multifactorial. Whereas the pulmonary vein thrombosis was co-localized to the same lung segments as the pulmonary embolism, it is likely that the pulmonary embolism occurred first and that the pulmonary venous thrombosis followed afterwards. It is noteworthy that pulmonary vein thrombosis rarely if ever complicates a pulmonary embolic event apart from this infection. Under normal conditions, pulmonary arterioles and capillaries are a rich source of tPA (tissue plasminogen activator) and fibrinolytic activity and serve as a biochemical barrier against thrombus propagation following pulmonary embolism. The thrombotic signal within the infected lung may have been augmented by local endothelial injury from complement mediated membrane attack complexes and associated coagulation activation. Endothelial infection by the virus may have triggered local recruitment of neutrophils entrapping platelets in the process of NETosis. The ensuing signaling between neutrophils and macrophages may have amplified the cytokine storm known to augment thrombosis. Hypoxia inducible factors (HIF) are also known to stimulate leukocytes and platelets, induce local tissue factor expression and impair fibrinolytic pathways further promoting thrombus propagation. In this case, it is unclear if the carotid artery thrombus resulted from embolized PVT or an in-situ thrombus.
Funding
None.
Compliance with ethical standards
Conflict of interest
The author declares that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Delabranche X, Helms J, Meziani F. Immunohaemostasis: a new view on haemostasis during sepsis. Ann Intensive Care. 2017;7:117. doi: 10.1186/s13613-017-0339-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iba T, Levy JH. Inflammation and thrombosis: roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J Thromb Haemost. 2018;16:231–241. doi: 10.1111/jth.13911. [DOI] [PubMed] [Google Scholar]
- 3.Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;75:2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rosovsky RP, Grodzin C, Channick R et al (2020) Diagnosis and treatment of pulmonary embolism during the COVID-19 Pandemic: a position paper from the National PERT Consortium. Chest [DOI] [PMC free article] [PubMed]
- 5.Jackson SP, Darbousset R, Schoenwaelder SM. Thromboinflammation: challenges of therapeutically targeting coagulation and other host defense mechanisms. Blood. 2019;133:906–918. doi: 10.1182/blood-2018-11-882993. [DOI] [PubMed] [Google Scholar]
- 6.Yaghi S, Ishida K, Torres J, et al. SARS-CoV-2 and stroke in a new york healthcare system. Stroke. 2020;51:2002–2011. doi: 10.1161/STROKEAHA.120.030335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mohamud AY, Griffith B, Rehman M et al (2020) Intraluminal carotid artery thrombus in COVID-19: another danger of cytokine storm? AJNR Am J Neuroradiol [DOI] [PMC free article] [PubMed]
- 8.Viguier A, Delamarre L, Duplantier J, Olivot JM, Bonneville F. Acute ischemic stroke complicating common carotid artery thrombosis during a severe COVID-19 infection. J Neuroradiol. 2020;47:393–394. doi: 10.1016/j.neurad.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wu JP, Wu Q, Yang Y, Du ZZ, Sun HF. Idiopathic pulmonary vein thrombosis extending to left atrium: a case report with a literature review. Chin Med J (Engl) 2012;125:1197–1200. [PubMed] [Google Scholar]