Abstract
Background
Tuberculosis (TB) is one of the major public health problems in Ethiopia. Determining discharge outcome of TB cases helps to understand the effectiveness of TB diagnosis effort to improve case management. The objective of this study was to assess discharge outcomes and factors associated with death among patients on TB treatment admitted in a referral hospital in southwest Ethiopia.
Methods
A retrospective study was conducted in Jimma Medical Center by reviewing medical records of TB patients (age ≥15 years) admitted to medical wards from January 2015 to December 2017. Demographic and related data was collected using data collection format. The clinical evaluation outcome of an individual patient at discharge was recorded. Descriptive statistics were used to summarize participant characteristics. Multivariate logistic regression analyses were used to evaluate the risk factors for TB mortality.
Results
Among 465 patients included in the review, 225 (48.4%) were pulmonary (PTB) and 240 (51.6%) extra-pulmonary TB (EPTB) cases. Overall, 190 (40.9%) had bacteriologically confirmed for TB. HIV status was documented for 340 (73.1%) of them; 93 (27.4%) were found to be positive. The prevalence of HIV infection among EPTB and PTB cases was 50/275 (18.2%) and 43/190 (22.8%), respectively. A quarter of the patient, 114 (24.5%), died in the hospital while the rest were discharged with clinical improvement. Compared to smear positive PTB, the risk of death was two times higher in patients with smear negative PTB cases (aOR 2.3, 95% CI: 1.3–4.2). TB patients with coronary obstructive pulmonary disease (COPD) (aOR 4.6, 95% CI: 1.3–16.7), diabetes mellitus (aOR 5.7, 95% CI: 1.5–23.7), heart disease (aOR 3.8, 95% CI: 1.4–10.4) had about four-fold increased risk of death. HIV-positive patients had a higher risk of mortality (aOR 2.9, 95% CI: 1.7–5.0) than HIV-negative patients. The risk of death was not affected by the type of TB diagnosed, as 27.6% of EPTB and 31.9% PTB were died (p = 0.457).
Conclusion
TB was associated with high inpatient mortality at Jimma Medical Center. Mortality was found to be higher among unconfirmed cases, those with COPD, diabetes mellitus, heart disease and HIV infection. Thus, any effort to curtail mortality in such high TB burden setting should focus on improving TB diagnosis and addressing major comorbid medical conditions.
Keywords: Tuberculosis, Mortality, Diagnosis, HIV, Ethiopia
1. Introduction
Tuberculosis (TB) is the disease caused by Mycobacterium tuberculosis (MTB) complex. With an estimated 10 million new TB cases and 1.3 million deaths in 2018, TB remains a public health problems worldwide. The 2018 global TB report, released by the World Health Organization (WHO) in 2019, reported that Ethiopia had the 10th highest incidence of TB in the world with a total of 114, 233 new cases and the highest incidence rate with 164 new cases per 100, 000 people [1].
The use of smear microscopy is not enough to detect active TB in patients suspected of pulmonary and extra-pulmonary disease. An estimated 50% of TB cases have been missed and left untreated due to the continued use of less sensitive smear microscopy, particularly in resource-constrained countries [2], [3], [4], [5]. The capacity to use culture for diagnosis of TB in all patients with a negative smear result is very scarce [3], [6]. However, to overcome the diagnosis efficacy limitations, an experienced physicians initiates a full course of anti-TB treatment in smear-negative or severely ill patients according to the national guideline recommendations [7].
Fortunately, outpatients started on empiric TB treatment (without bacteriological confirmation for TB) were associated with poor clinical outcomes. In a retrospective review conducted at Gondar and Dilla hospitals, Ethiopia, including 2196 and 816 smear-negative outpatients, respectively, revealed that 16.7% and 29.4% of patients showed unsuccessful outcomes [8], [9]. Another studies showed that coinfection with HIV, and infection with multidrug‑resistant MTB are the main risk factors for mortality among TB patients [10], [11], [12]. Consequently, patients on TB treatment could be associated with higher rates of complications and excessive mortality, particularly whereby more than 20% of notified TB patients were co-infected with HIV [13].
Despite the recommended effective anti-TB treatment, some patients with TB still die while on treatment for their disease. Thus, the magnitude of mortality and associated factors among hospitalized patients on TB treatment in southwest Ethiopia remains poorly documented. This study was to assess discharge outcome and its associated factors with death among patients on TB treatment admitted in a referral hospital in southwest Ethiopia during a three-year (2015–2017) period. The findings of the study could be valuable to monitor the performance of the national TB treatment program and to institute interventions depending on the identified factors associated with mortality.
2. Materials and methods
2.1. Study setting
The study was conducted from January 2015 to December 2017 at Jimma Medical Center (JMC), the only referral and teaching hospital in the southwest region of Ethiopia. JMC provides inpatient and outpatient services to over 15 million population [14]. The standard approach to diagnose TB in Ethiopia is symptom screening, followed by smear microscopy according to the national TB treatment guideline recommendation adopted from the 2007 WHO's diagnostic algorithm. Depending on the clinician's decision, a CXR paid by the individual seeking care, is used for clinical decision-making [7]. The capacity to use the molecular Xpert MTB/RIF assay for patients who tested negative for smear microscopy is limited during the study period. The presence of co-morbidities such as pneumonia, diabetes mellitus, cardiovascular disease and COPD were diagnosed by physicians who care a patients. For example, for most of the patients, COPD was diagnosed clinically if they had a chronic persistent cough with breathlessness, chest tightness, wheezing and sputum production with underlying major risk factors (smoking and age >35 years). For those who had spirometry test, it was diagnosed in patients who had all the following findings: (1) Forced expiratory volume in the first second (FEV1) <80% of the predicted value, (2) If the ratio of FEV1 and forced vital capacity (FVC) [FEV1/FVC] was <0.7 and (3) absence of reversibility of airway obstruction (documented FEV1 improvement of <15% or <200 ml 20 min after taking 2 puffs of inhaled salbutamol) by clinician providing patient care [15].
2.2. Study design
We conducted a retrospective cross-sectional review of hospitalized patients on TB treatment in JMC, Ethiopia. The primary outcomes were mortality related to TB at discharge. The risk factors examined were socio-demographic and clinical features extracted at the time of discharge or death.
2.3. Study population and data collection
The study population includes hospitalized patients with an age of ≥15 years, clinicians decided to admit in medical wards as bacteriologically or clinically confirmed cases of TB from 1st of January 2015 to 31th of December 2017, and who started on anti-TB treatment. In this study, bacteriologically and clinically confirmed TB cases were defined according to the Ethiopian national TB and Leprosy control program guideline [7]. Bacteriologically confirmed TB cases: a patient with at least one sputum specimen positive for acid-fast bacilli (AFB) using smear microscopy or a positive result for AFB on extra-pulmonary samples or body fluids using microscopy. Clinically confirmed TB cases: a patient with spot-morning-spot sputum specimens were negative for AFB on smear microscopy, no response to broad-spectrum antibiotic therapy, chest X-ray abnormalities consistent with TB and decision was made by an experienced clinician to treat with a full course of TB treatment.
All inpatients diagnosed by direct smear microscopy for clinical samples, chest X-ray, or based on clinical symptoms according to the national guideline recommendation and registered on the registration book by head nurse of medical wards within 24 h of starting TB treatment. A nurse followed the patients until they moved out of the hospital and advised to continue treatment near to their home at health care center. The clinicians decided clinical evaluation outcomes were registered on patient‘s medical cards and registration log book. A selected nurse as a data collector for this study reviewed the patients’ medical cards as well as registration log book, and documented all important information regarding outcome variable on data collection format.
2.4. Data analysis
Study population characteristics were summarized using descriptive statistics (frequency, mean, and standard deviation or median and inter-quartile ranges as appropriate) and tabulated overall and stratified by type of TB diagnosis. The proportion of inpatient discharge outcomes was compared between bacteriologically confirmed for TB and clinically diagnosed patients in whom TB treatment was initiated empirically. Logistic regression analysis was performed to determine the strength of association (odds ratio and its 95% confidence interval) between patient characteristics and mortality related to TB. Characteristics that showed an association in bivariate analysis (p < 0.20) were included in the multivariate logistic regression model. All data analysis was performed using R Statistical software version 3.6.1.
2.5. Ethical clearance
The patients did not consent for their individual-level data to be made public as this was a routine care service and not a formal research study where study patients would give such consent. However, the study was ethically approved by the Institutional Review Board (IRB) of the Institute of Health, Jimma University with Ref. no. JHRPGD 388/2018 and permission was obtained from JMC to access patients’ medical information.
3. Results
3.1. Baseline characteristics of the study population
A total of 465 patients admitted to the hospital as TB cases during the three years were enrolled in this study, with a median age of 35 years (interquartile: 25–50). Among all participants, more than half (54%) were male, and close to 2/3rd (63.2%) were from rural residents. Among the 340 (73.1%) patients with documented HIV status, 27.4% (93/340) were HIV positive; of which 71 (76.3%) were on anti-retroviral treatment (ART) for a mean of 5 years (±1.51) while 22 (23.7%) receiving cotrimoxazole prophylaxis. Overall, 240 (51.6%) were extra-pulmonary TB cases and 225 (48.4%) pulmonary TB cases, and 7.1% of TB patients were comorbid with pneumonia, 3.7% heart failure, 2.0% diabetes mellitus and 2.4% COPD (Table 1).
Table 1.
Characteristics of study participants.
| Characteristics | Number | Percent |
|---|---|---|
| Age in years (median, IQR) | 35 (25–50) | |
| Sex | ||
| Female | 214 | 46.0 |
| Male | 251 | 54.0 |
| Residence | ||
| Urban | 171 | 36.8 |
| Rural | 294 | 63.2 |
| HIV status | ||
| Positive | 93 | 20.0 |
| Negative | 247 | 53.1 |
| Unknown | 125 | 26.9 |
| On ART (n = 93) | 71 | 76.3 |
| On cotrimoxazole prophylaxis (n = 93) | 22 | 23.7 |
| Pneumonia | 33 | 7.1 |
| Heart failure a | 17 | 3.7 |
| Diabetes b | 9 | 2.0 |
| Viral hepatitis c | 5 | 1.1 |
| COPD | 11 | 2.4 |
| Types of TB diagnosis | ||
| Pulmonary TB cases | 225 | 48.4 |
| Extra pulmonary TB cases | 240 | 51.6 |
HIV = human immunodeficiency virus, COPD = coronary pulmonary obstructive disease. a including ischemic heart disease and cardiac failure, b including type 1 and 2 DM, c including hepatitis B and C.
3.2. Bacteriological confirmation of TB and its prediction by the clinical algorithm
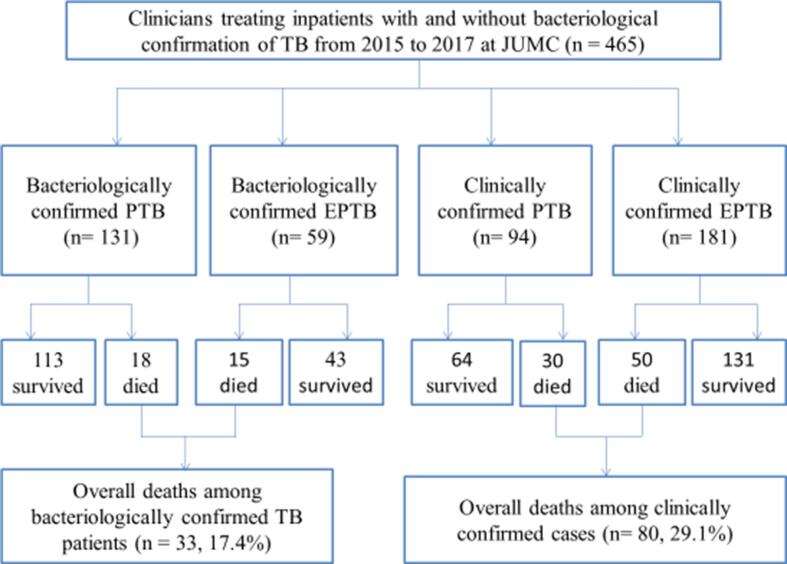
In line with a national guideline, a total of 190/465 (40.9%) hospitalized patients had bacteriologically confirmed for TB on smear microscopy (Fig. 1). Among all microbiologically confirmed cases, 68.9% (131/190) were PTB and 31% (59/190) were EPTB (Table 2) while 275/465 (59.1%) were diagnosed based on clinical suspicion only, with 34.2% (94/275) were PTB and 181 of the 277 (65.8%) were EPTB cases. The proportion of HIV infection among clinically confirmed TB cases who received empirical TB treatment was almost two times higher (24.0% (95% CI 19.1%, 29.5%) than those patients who were bacteriologically confirmed for TB (14.2% (95% CI 9.6%, 20.0%) (p = 0.009) (Table 3). The trend of bacteriological confirmed TB among hospitalized patients who treated for TB increased from 24 (12.6%) in 2015 to 94 (49.5%) in 2017.
Fig. 1.
Flowchart showing patient’s enrollment, TB diagnosis implications and hospital discharge outcome of patients admitted at JMC between 2015 and 2017. JUMC = Jimma University Medical Center, PTB = Pulmonary tuberculosis, EPTB = extrapulmonary tuberculosis.
Table 2.
Characteristics compared between bacteriologically confirmed PTB and EPTB inpatients started on TB treatment at JMC from 2015 to 2017.
| Characteristics | All (n = 190) | Bacteriologically confirmed, (n %) |
|
|---|---|---|---|
| PTB patients (n = 131) | EPTB patients (n = 59) | ||
| Age (years) | |||
| 15–40 | 106 (55.8) | 73 (55.7) | 33 (55.9) |
| 41–64 | 59 (31.1) | 42 (32.1) | 17 (28.8) |
| ≥65 | 25 (13.1) | 16 (12.2) | 9 (15.3) |
| Sex | |||
| Female | 94 (49.5) | 67 (51.1) | 27 (45.8) |
| Male | 96 (50.5) | 64 (48.9) | 32 (54.2) |
| Residence | |||
| Urban | 58 (30.5) | 37 (28.2) | 21 (35.6) |
| Rural | 132 (69.5) | 94 (71.8) | 38 (64.4) |
| HIV status | |||
| HIV-Negative | 119 (62.6) | 84 (64.1) | 35 (59.3) |
| HIV-Positive | 27 (14.2) | 15 (16.1) | 12 (20.3) |
| Unknown results | 44 (23.2) | 32 (24.4) | 12 (20.3) |
| Pneumonia | |||
| No | 176 (92.6) | 122 (93.1) | 54 (91.5) |
| Yes | 14 (7.4) | 9 (6.9) | 5 (8.5) |
| Heart failure a | |||
| No | 185 (97.4) | 127 (96.6) | 58 (98.3) |
| Yes | 5 (2.6) | 4 (3.1) | 1 (1.7) |
| Diabetes b | |||
| No | 185 (97.4) | 129 (98.5) | 56 (94.9) |
| Yes | 5 (2.6) | 2 (1.5) | 3 (5.1) |
| Viral hepatitis c | |||
| No | 189 (99.4) | 131 (100.0) | 58 (98.3) |
| Yes | 1 (0.5) | 0 (0.0) | 1 (1.7) |
| COPD | |||
| No | 184 (96.8) | 127 (96.9) | 57 (96.6) |
| Yes | 6 (3.2) | 4 (3.1) | 2 (3.4) |
| Year of admission | |||
| 2015 | 24 (12.6) | 15 (11.5) | 9 (15.3) |
| 2016 | 72 (37.9) | 43 (32.8) | 29 (49.2) |
| 2017 | 94 (49.5) | 73 (55.7) | 21 (35.6) |
PTB = pulmonary tuberculosis, EPTB = Extra pulmonary tuberculosis, COPD = coronary pulmonary obstructive disease. a including ischemic heart disease and cardiac failure, b including type 1 and 2 DM, c including hepatitis B and C.
Table 3.
Characteristics compared between clinically diagnosed PTB and EPTB inpatients started on TB treatment at JMC from 2015 to 2017.
| Characteristics | All (n = 275) | Clinically diagnosed, (n %) |
|
|---|---|---|---|
| PTB patients (n = 94) | EPTB patients (n = 181) | ||
| Age (years) | |||
| 15–40 | 174 (63.3) | 64 (68.1) | 110 (60.8) |
| 41–64 | 79 (28.7) | 25 (26.6) | 54 (29.8) |
| ≥65 | 22 (8.0) | 5 (5.3) | 17 (9.4) |
| Sex | |||
| Female | 121 (44.0) | 44 (46.8) | 77 (42.5) |
| Male | 154 (50.4) | 50 (53.2) | 104 (57.5) |
| Residence | |||
| Urban | 113 (41.1) | 41 (43.6) | 72 (39.8) |
| Rural | 162 (58.9) | 53 (56.4) | 109 (60.2) |
| HIV status | |||
| Negative | 129 (46.9) | 36 (38.3) | 93 (51.4) |
| Positive | 66 (24.0) | 28 (29.8) | 38 (21.0) |
| Unknown | 80 (29.1)) | 30 (31.9) | 50 (27.6) |
| Pneumonia | |||
| No | 256 (93.1)) | 88 (93.6) | 168 (92.8) |
| Yes | 19 (6.9) | 6 (6.4) | 13 (7.2) |
| Heart failurea | |||
| No | 264 (96.0) | 90 (95.7) | 174 (96.1) |
| Yes | 11 (4.0) | 4 (4.3) | 7 (3.9) |
| Diabetesb | |||
| No | 271 (98.5) | 93 (98.9) | 178 (98.3) |
| Yes | 4 (2.5) | 1 (1.1) | 3 (1.7) |
| Viral hepatitisc | |||
| No | 271 (98.5) | 93 (98.9) | 178 (98.3) |
| Yes | 4 (2.5) | 1 (1.1) | 3 (1.7) |
| COPD | |||
| No | 271 (98.5) | 92 (97.9) | 179 (98.9) |
| Yes | 4 (2.5) | 2 (2.1) | 2 (1.1) |
| Year of TB diagnosis | |||
| 2015 | 88 (32.0) | 28 (29.8) | 60 (33.1) |
| 2016 | 100 (36.4) | 35 (37.2) | 65 (35.9) |
| 2017 | 87 (31.6) | 31 (33.0) | 56 (30.9) |
PTB = pulmonary tuberculosis, EPTB = Extra pulmonary tuberculosis, COPD = coronary pulmonary obstructive disease. a including ischemic heart disease and cardiac failure, b including type 1 and 2 DM, c including hepatitis B and C.
3.3. Patient‘s discharge outcomes
About 3/4th of the patients, (75.5%, 95% CI: 71.3–79.3) were discharged alive; the rest (24.5%, 95% CI: 20.7–28.7) passed away in the hospital. Sixty-one percent of bacteriologically confirmed EPTB patients were discharged with improvement (Table 4). The risk of death was two times higher among bacteriologically confirmed EPTB cases than bacteriologically confirmed PTB (25.4% vs 13.7%, p = 0.049). Among clinically diagnosed PTB cases, 31.9% (95% CI: 22.7–42.3) were dying in-hospital, and 53.2% (95% CI: 42.6–63.6) were discharged with improvements. While the risk of death was not affected by the type of TB diagnosed, as 27.6% of EPTB and 31.9% PTB cases starting TB treatment based on clinical algorithm died (p = 0.457) (Table 5).
Table 4.
Discharge outcomes compared between bacteriologically confirmed PTB and EPTB inpatients started on TB treatment at JMC, Ethiopia.
| Discharge outcomes | All (n = 190) | Microbiologically confirmed |
|
|---|---|---|---|
| PTB patients (n = 131) | EPTB patients (n = 59) | ||
| Improved | 136 (71.6) | 100 (76.3) | 36 (61.0) |
| Died | 33 (17.4) | 18 (13.7) | 15 (25.4) |
| Left against medical advice | 9 (4.7) | 8 (6.1) | 1 (1.7) |
| Referred to higher facility | 3 (1.6) | 1 (0.8) | 2 (2.1) |
| Deteriorated | 9 (4.7) | 4 (3.1) | 5 (8.5) |
PTB = pulmonary tuberculosis, EPTB = Extra pulmonary tuberculosis.
Table 5.
Discharge outcomes compared between clinically diagnosed PTB and EPTB cases started on TB treatment at JMC, Ethiopia.
| Discharge outcomes | All (n = 275) | Clinically confirmed |
|
|---|---|---|---|
| PTB patients (n = 94) | EPTB patients (n = 181) | ||
| Improved | 157 (57.1) | 50 (53.2) | 107 (59.1) |
| Died | 80 (29.1) | 30 (31.9) | 50 (27.6) |
| Left against medical advice | 17 (6.2) | 8 (8.5) | 9 (5.0) |
| Referred to higher facility | 11 (4.0) | 2 (2.1) | 9 (5.0) |
| Deteriorated | 10 (3.6) | 4 (4.3) | 6 (3.3) |
PTB = pulmonary tuberculosis, EPTB = Extrapulmonary tuberculosis.
3.4. Associated factors with TB mortality
Hospitalized patients who had HIV co-infection (aOR 2.9, 95% CI: 1.7–5.0), cardiovascular disease (aOR 3.8, 95% CI: 1.4–10.4), diabetes mellitus (aOR 5.7, 95% CI: 1.5–23.7), COPD (aOR 4.6, 95% CI: 1.3–16.7) and bacteriologically unconfirmed PTB cases (aOR 2.3, 95% CI: 1.3–4.2) were independently associated with in-hospital mortality (Table 6).
Table 6.
Association between patient characteristics and mortality outcome among inpatients started on TB treatment in JMC, Ethiopia, from 2015 to 2017.
| Characteristics | Discharge outcomes |
OR (95% CI) | aOR (95%CI) | |
|---|---|---|---|---|
| Died (n = 114) | Survived (n = 351) | |||
| Age (years) | ||||
| 15–40 | 69 (60.5) | 211 (60.1) | Ref. | |
| 41–64 | 33 (28.9) | 104 (29.6) | 0.9 (0.6–1.6) | |
| ≥65 | 12 (10.5) | 36 (10.3) | 1.1 (0.5–2.1) | |
| Sex | ||||
| Female | 52 (45.6) | 162 (46.2) | Ref. | |
| Male | 62 (54.4) | 189 (53.8) | 1.1 (0.7–1.6) | |
| Residence | ||||
| Urban | 48 (42.1) | 123 (35.0) | Ref. | |
| Rural | 66 (57.9) | 228 (65.0) | 0.7 (0.5–1.2) | |
| HIV status | ||||
| Negative | 49 (43.0) | 198 (56.4) | Ref. | Ref. |
| Positive | 37 (32.4) | 56 (16.0) | 2.7 (1.6–4.5) | 2.9 (1.7–5.0) |
| Unknown | 28 (24.6) | 97 (27.6) | 1.2 (0.6–1.9) | 1.1 (0.6–1.8) |
| Pneumonia | ||||
| No | 106 (93.0) | 326 (92.9) | Ref. | |
| Yes | 8 (7.0) | 25 (7.1) | 0.9 (0.4–2.2) | |
| Heart failure | ||||
| No | 106 (93.0) | 342 (97.4) | Ref. | Ref. |
| Yes | 8 (7.0) | 9 (2.6) | 2.8 (1.1–7.7) | 3.8 (1.4–10.4) |
| Diabetes mellitus | ||||
| No | 109 (95.6) | 347 (98.9) | Ref. | Ref. |
| Yes | 5 (4.4) | 4 (1.1) | 3.9 (1.1–16.3) | 5.7 (1.5–23.7) |
| Viral hepatitis | ||||
| No | 112 (98.2) | 348 (99.1) | Ref. | |
| Yes | 2 (1.8) | 3 (0.9) | 2.1 (0.3–12.6) | |
| COPD | ||||
| No | 108 (94.7) | 346 (98.6) | Ref. | Ref. |
| Yes | 6 (5.3) | 5 (1.4) | 3.8 (1.2–13.5) | 4.6 (1.3–16.7) |
| Positive smear for PTB | ||||
| Yes | 18 (15.8) | 113 (32.2) | Ref. | Ref. |
| No | 96 (84.2) | 238 (67.8) | 2.5 (1.5–4.5) | 2.3 (1.3–4.2) |
| Positive smear for EPTB | ||||
| Yes | 15 (13.2) | 44 (12.5) | Ref. | |
| No | 99 (86.8) | 307 (87.3) | 0.9 (0.5–1.8) | |
| Clinically diagnosed PTB | ||||
| Yes | 30 (26.3) | 64 (18.2) | Ref. | |
| No | 84 (73.7) | 287 (81.8) | 0.6 (0.4–1.1) | |
| Clinically diagnosed EPTB | ||||
| Yes | 50 (43.9) | 131 (37.3) | Ref. | |
| No | 64 (56.1) | 220 (62.7) | 0.7 (0.5–1.2) | |
EPTB = Extrapulmonary TB, PTB = Pulmonary TB, COPD = Coronary obstructive pulmonary disease, OR = Odds ratio, aOR = Adjusted odds ratio, Ref. = reference category, 95% CI = 95% confidence interval.
4. Discussion
In the current study, the overall mortality among hospitalized patients on TB treatment was 24.5%. The previous studies done in Ethiopia reported that 7.7% to 13.9% of TB mortality among outpatients treated at TB clinics [16], [17], [18]. Our current study focused on hospitalized patients who expected to have a severe medical condition. Although, people in whom TB diagnosed based on signs and symptoms were more likely to die at the early stages of TB treatment initiated. The previous study reported a similar finding from Kenya [19] in which TB mortality was higher among patients diagnosed for TB based on clinical signs and received TB treatment empirically. This similarity might be due to the majority of unconfirmed cases were extra-pulmonary TB; on its own may be a severe form of TB with a higher risk of complications. On the other hand, those patients who were empirically treated for TB may have other diagnoses of chronic disease that may increase mortality due to either severe underlying diagnosis or delay in the treatment of the disease.
For decades, clinicians have used a clinical algorithm to overcome the limited sensitivity of smear microscopy and to avoid the negative consequences of delays in treatment initiation, especially in high TB/HIV prevalent areas. In the current study, the rate of TB mortality was two times higher among patients diagnosed for TB based on a clinical algorithm when compared with bacteriologically confirmed cases (29.1% versus 17.9%, respectively).
Hospitalized TB cases co-infected with COPD, heart failure, and diabetes mellitus were associated with higher mortality rates as they are the most significant factors that affects the survival chance of hospitalized patients diagnosed with TB. In this study, inpatients comorbid with COPD were 4.6 times more likely to die compared to those who had no COPD. The death of hospitalized patients was almost four times more likely among patient comorbid with cardiovascular disease compared to those who hadn't chronic cardiovascular disease. Diabetes mellitus was another independent risk factor for mortality among TB patients, with those who comorbid with diabetes mellitus were 5.7 times more likely to die. However, previously reported independent factors associated with deaths among outpatients were delay in diagnosis [20], type of TB diagnosis [16], older age and people with lower socioeconomic status [21] and advanced human HIV infection [22], [23] based on the data collected from TB treatment center. Identifying additional host risk factors of TB death in this study are important to improve TB outcome in this high-risk group.
TB patients co-infected with HIV were almost three times higher risk of dying compared to HIV-negative TB patients. Similarly, prior studies revealed higher death rates in HIV positive TB patients [16], [17], this is due to TB/HIV co-infected patients had depressed immunity that worsening the disease condition and delayed diagnosis. They may also have multiple opportunistic infections. Thus all of these factors contributed to high mortality [20], [21].
Even among HIV negative TB patients, the proportion of deaths in our study is higher than another study reported that 0.6% [24]. We believe that this difference is likely related to socio-economic status and difference in access to health facilities as there is a marked difference in urban settings like Addis Ababa and other rural and remote areas in the country. Our study revealed that important information regarding patient mortality and associated factors in hospitalized patients on TB treatment at a tertiary and teaching hospital in southwest Ethiopia that could have valuable information to monitor the performance of national TB control program and to initiate institute interventions.
Our study had some limitations. First, because we limited enrollment to hospitalized patient’s data, our results cannot be generalized to an outpatient setting. Second, we were not able to investigate the level of treatment adherence of each patient and the severity of comorbidities. Third, despite the presence of negative microscopy, it is still possible that some patients had not pulmonary or extra-pulmonary TB, which may have resulted in an overestimation of the proportion of overtreatment.
In conclusion, TB was associated with very high inpatient mortality at JMC. Mortality was found to be higher among bacteriologically unconfirmed cases, those with COPD and HIV infection. All hospitalized patients with presumptive TB should gain access to the more sensitive molecular tests to reduce TB related mortality and barriers to TB care. Thus, any effort to curtail mortality in such high TB burden setting should focus on improving diagnosis and addressing major comorbid medical conditions.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgement
The authors would like to acknowledge the assistance provided by members of staff of medical wards of JMC.
Author’s contribution statement
W.K. made substantial contribution from conception, design, data acquisition, analysis and manuscript writing. G.B. collect the data and gave approval of the final draft manuscript. E.K.G. and G.A. critical reviewed the manuscript and approve for publication. All authors read and approved the final manuscript.
References
- 1.World health organization . WHO; Geneva: 2019. Global tuberculosis report 2019. [Google Scholar]
- 2.Chadha V.K., Anjinappa S.M., Rade K., Baskaran D., Narang P., Kolappan C. Sensitivity and specificity of screening tools and smear microscopy in active tuberculosis case finding. Indian J Tubercul. 2019;66(1):99–104. doi: 10.1016/j.ijtb.2018.05.015. [DOI] [PubMed] [Google Scholar]
- 3.Gizaw N., Abera A., Sisay S., Desta K., Kreibich S., Gerwing-Adima L. The yield of Auramine O staining using led microscopy with bleach treated sputum samples for detection of pulmonary tuberculosis at St. Peter tuberculosis specialized hospital Addis Ababa, Ethiopia. J Clin Tuber Other Mycobact Dis. 2020;18 doi: 10.1016/j.jctube.2019.100140. 100140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kebede A.H., Alebachew Z., Tesfaye F., Lemma E., Abebe A., Agonafir M. The first population-based national tuberculosis prevalence survey in Ethiopia, 2010–2011. Nt J Tuberc Lung Dis. 2014;18(6):635–639. doi: 10.5588/ijtld.13.0417. [DOI] [PubMed] [Google Scholar]
- 5.MacLean E., Saravu K., Pai M. Diagnosing active tuberculosis in people living with HIV: an ongoing challenge. Curr Opin HIV AIDS. 2019;14(1):46–54. doi: 10.1097/COH.0000000000000512. [DOI] [PubMed] [Google Scholar]
- 6.World health organization . WHO; World bank: 2009. Approaches to improve sputum smear microscopy for tuberculosis diagnosis. [Google Scholar]
- 7.Federal Ministry of Health Ethiopia . MOH; Addis Ababa: 2014. Tuberculosis, leprosy and TB/HIV prevention and control programme. [Google Scholar]
- 8.Gebrezgabiher G., Romha G., Ejeta E., Asebe G., Zemene E., Ameni G. Treatment outcome of tuberculosis patients under directly observed treatment short course and factors affecting outcome in southern ethiopia: a five-year retrospective study. PLoS ONE. 2016;11(2) doi: 10.1371/journal.pone.0150560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tessema B., Muche A., Bekele A., Reissig D., Emmrich F., Sack U. Treatment outcome of tuberculosis patients at Gondar University Teaching Hospital, Northwest Ethiopia. A five-year retrospective study. BMC Public Health. 2009;9(1):371. doi: 10.1186/1471-2458-9-371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gaifer Z.A. Risk factors for tuberculosis mortality in a tertiary care center in Oman, 2006–2016. Int J Mycobacteriol. 2017;6(4):356–359. doi: 10.4103/ijmy.ijmy_123_17. PubMed PMID: 29171449. Epub 2017/11/25. eng. [DOI] [PubMed] [Google Scholar]
- 11.Hannah HA, Miramontes R, Gandhi NR. Sociodemographic and Clinical Risk Factors Associated With Tuberculosis Mortality in the United States, 2009-2013. Public health reports (Washington, DC: 1974). 2017 May/Jun;132(3):366-75. PubMed PMID: 28394707. Pubmed Central PMCID: PMC5415250. Epub 2017/04/11. eng. [DOI] [PMC free article] [PubMed]
- 12.Martin SJ, Sabina EP. Malnutrition and Associated Disorders in Tuberculosis and Its Therapy. Journal of dietary supplements. 2019;16(5):602-10. PubMed PMID: 29958051. Epub 2018/06/30. eng. [DOI] [PubMed]
- 13.Abebe G., Bonsa Z., Kebede W. Treatment outcomes and associated factors in tuberculosis patients at Jimma University Medical Center: a 5-year retrospective study. Int J Mycobacteriol. 2019;8(35–41) doi: 10.4103/ijmy.ijmy_177_18. [DOI] [PubMed] [Google Scholar]
- 14.Jimma University. Historical background Jimma Jimma University; Jimma Jimma University; 2015 [cited 2018 Dec 4]. Available from: http://www.ju.edu.et.
- 15.Global Initiative for Chronic Obstructive Lung Disease (GOLD). Pocket guied to COPD diagnosis, managment and prevention USA: Global initiative for chronic obstractive Lung disease, Inc; 2019.
- 16.Asgedom S.W., Tesfaye D., Nirayo Y.L., Atey T.M. Time to death and risk factors among tuberculosis patients in Northern Ethiopia. BMC Res Notes. 2018;11:696. doi: 10.1186/s13104-018-3806-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Beyene Y., Geresu B., Mulu A. Mortality among tuberculosis patients under DOTS programme: a historical cohort study. BMC Public Health. 2016;16:883. doi: 10.1186/s12889-016-3557-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Birlie A, Tesfaw G, Dejene T, Woldemichael K. Time to Death and Associated Factors among Tuberculosis Patients in Dangila Woreda, Northwest Ethiopia. PloS one. 2015;10(12):e0144244. PubMed PMID: 26669737. Pubmed Central PMCID: PMC4687639. Epub 2015/12/17. eng. [DOI] [PMC free article] [PubMed]
- 19.Huerga H, Ferlazzo G, Wanjala S, Bastard M, Bevilacqua P, Ardizzoni E, et al. Mortality in the first six months among HIV-positive and HIV-negative patients empirically treated for tuberculosis. BMC infectious diseases. 2019 19(1):132. PubMed PMID: 30744603. Pubmed Central PMCID: Pmc6369550. Epub eng. [DOI] [PMC free article] [PubMed]
- 20.Schneider D., McNabb S.J.N., Safaryan M., Davidyants V., Niazyan L., Orbelyan S. Reasons for delay in seeking care for tuberculosis, Republic of Armenia, 2006–2007. Interdiscip Persp Inf Dis. 2010;2010 doi: 10.1155/2010/412624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yen Y.F., Yen M.Y., Shih H.F., Hu B.S., Ho B.L., Li L.H. Prognostic factors associated with mortality before and during anti-tuberculosis treatment. Int J Tuberc Lung Dis. 2013;17(10):1310–1316. doi: 10.5588/ijtld.12.0888. [DOI] [PubMed] [Google Scholar]
- 22.Kamenju P., Aboud S. Tuberculosis-HIV co-infection among patients admitted at Muhimbili National Hospital in Dar es salaam, Tanzania. Tanzan J Health Res. 2011;13(1):21–26. doi: 10.4314/thrb.v13i1.60682. [DOI] [PubMed] [Google Scholar]
- 23.Shaweno D., Worku A. Tuberculosis treatment survival of HIV positive TB patients on directly observed treatment short-course in Southern Ethiopia: a retrospective cohort study. BMC Res Notes. 2012;5:682. doi: 10.1186/1756-0500-5-682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Getahun B., Ameni G., Biadgilign S., Medhin G. Mortality and associated risk factors in a cohort of tuberculosis patients treated under DOTS programme in Addis Ababa, Ethiopia. BMC Infect Dis. 2017;11:127. doi: 10.1186/1471-2334-11-127. [DOI] [PMC free article] [PubMed] [Google Scholar]