Abstract
BACKGROUND
There is scarce literature on point-of-care ultrasound (POCUS) assessment characteristics in coronavirus disease 2019 (COVID-19) pneumonia with hypoxic respiratory failure.
METHODS
This study was an observational, prospective, single-center study, including adults suspected to have COVID-19 who were transferred to the intensive care unit (ICU). An intensivist in critical care ultrasound performed lung ultrasound (LUS) and echocardiology within 12 hours of patients' admission to the ICU. We calculated the trans mitral E/A ratio, E/e′, left ventricular ejection fraction (EF), inferior vena cava (IVC) diameter, right ventricle (RV) size and systolic function.
RESULTS
In the group of patients with confirmed COVID-19 pneumonia, echocardiographic findings revealed normal E/e′, deceleration time (DT), and transmittal E/A ratio compared to those in the non-COVID-19 patients (p = 0.001, 0.0001, and 0.0001, respectively). IVC diameter was < 2 cm with > 50% collapsibility in 62 (81%) patients with COVID-19 pneumonia; a diameter of > 2 cm and < 50% collapsibility was detected among those with non-COVID-19 pneumonia (p-value of 0.001). In patients with COVID-19 pneumonia, there were 3 cases of myocarditis (3.9%) with poor EF, severe RV systolic dysfunction was seen in 9 cases (11.6%), and 3 cases exhibited RV thrombus. Lung US revealed 4 signs suggestive of COVID-19 pneumonia in 77 patients (98.6%) (sensitivity 96.9%; confidence interval, 85%–99.5%) when compared with reverse transcriptase-polymerase chain reaction results.
CONCLUSIONS
POCUS plays an important role in the bedside diagnosis, hemodynamic assessment and management of patients with acute hypoxic respiratory and circulatory failure with COVID-19 pneumonia.
Keywords: Diagnostic ultrasound, SARS-CoV, Pneumonia, Echocardiography
INTRODUCTION
Although the lung is the primary organ involved in coronavirus disease 2019 (COVID-19) infection, cardiac involvement is also frequently reported in patients with acute hypoxic respiratory failure.1) Thus, rapid bedside assessment of the heart and lungs using point-of-care ultrasound (POCUS) provides a clinician with a more accurate initial diagnosis for patients presenting with acute hypoxic respiratory failure in the context of the COVID-19 pneumonia pandemic.
The cardiac component of ultrasound examination is technically challenging because the cardiac structures need to be imaged from redundant scan planes, indirectly obtaining left ventricular filling pressure (LVFP) using Doppler. Echocardiography views are obtained along with non-cardiac views, such as the chest (lung, pleura) and inferior vena cava (IVC). In infection with severe acute respiratory syndrome coronavirus 2, POCUS may help confirm the diagnosis of COVID-19 pneumonia to triage dyspneic and hypoxic patients to help determine the need for subsequent management.
Many modified versions of POCUS employ echocardiography in COVID-19 patients,2),3) but we preferred to add LVFPs and IVC, as these parameters inform our approach for fluid responsiveness. A modified version of the American Society of Echocardiography (ASE) POCUS protocol may be of value in suspected or confirmed COVID-19 infection.3)
METHODS
Setting and study population
This was an observational, prospective, single-center study conducted in the intensive care unit (ICU) of Adan General Hospital from May 1st, 2020, to June 25, 2020. This study protocol was approved by the Ethical Committee of the Ministry of Health in Kuwait, and informed consent was obtained from every patient or from his or her next of kin.
Patients were included if they were age > 18 years of age, suspected to have COVID-19 and had been transferred to the ICU with fever or suspected respiratory infection and exhibited one of the following: respiratory rate > 30 breaths/min; severe respiratory distress; or SpO2 < 93% on room air.4) Patients were admitted to the ICU directly from the emergency department after reverse transcriptase-polymerase chain reaction (RT-PCR) testing was performed at the central virology laboratory in Kuwait. Clinical data were entered on a separate standardized data collection form at the time of patient enrollment by the treating critical care physician. Clinical data included the patient's age and sex, presenting symptoms, medical history, oxygen saturation from pulse oximetry and chest radiograph. An intensivist certified in critical care ultrasound who was blinded to the RT-PCR results, if available at the time of examination, performed the lung ultrasound (LUS) within 12 hours of the patient's admission to the ICU.
LUS
We performed lung ultrasonography for every patient admitted to the ICU with suspicion of COVID-19 using a 12-zone method.5) There were 6 zones for each hemithorax: 2 anterior, 2 axillary, and 2 posterior zones on each side. The anterior chest wall was defined as extending from the parasternal line to the anterior axillary line. This zone was divided into upper and lower regions at the third intercostal space. The lateral area from the anterior to the posterior axillary line was divided into upper and lower halves. The posterior zone was identified from the posterior axillary line to the paravertebral line. The ultrasound images were saved to a hard drive and were reviewed by a senior intensivist trained in critical care ultrasound. Ultrasound imaging was performed using a portable ultrasound machine (GE Vivid iq, Horten, Norway) equipped with a 3.5-MHz broadband curvilinear transducer. The probe was placed in an oblique position on the intercostal space, and the pleural line was centered in the middle of the image by adjusting the depth settings. The oblique position of the probe on the intercostal space allows visualization of a large portion of the pleural line without interruption by rib shadows.
Measurements
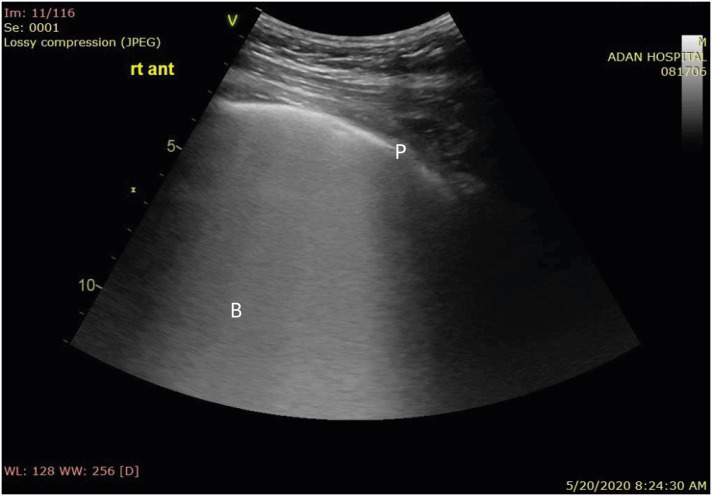
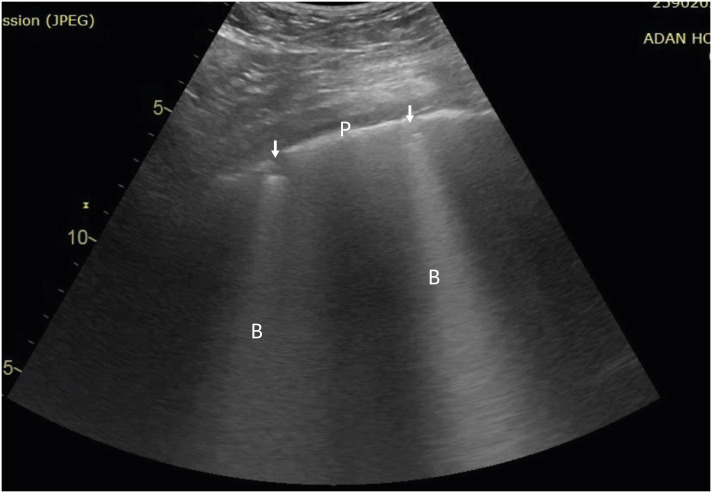
Pleural sliding and A-lines (repetitive lines parallel to the pleural line) on ultrasound are seen in normal healthy lungs.4) Interstitial syndrome is indicated by the presence of multiple B-lines (more than 3 lines in one region). The 4 signs of COVID-19 pneumonia on LUS evaluation are as follows (Figures 1 and 2)5):
Figure 1. Coalescent B-lines giving the appearance of a shining white lung with irregular pleura. The B-lines maintain their brightness until the end of the screen.
P: pleura, B: B-lines.
Figure 2. Separated B-lines with irregular pleura.
P: pleura, B: B-lines, arrow: subpleural consolidation.
1. Bilateral B-lines in both separate and coalescent forms, sometimes patchy, frequently giving the appearance of a shining white lung. The B-lines maintain their brightness until the end of the screen. They arise either directly from limited sliding pleura or from a small subpleural consolidation.
2. Bilateral diffuse irregularities of the pleural line.
3. Absence of significant pleural effusion.
4. Presence of multiple subpleural consolidations of various sizes.
Each lung zone was assigned a score to predict overall lung aeration.6)
• Score 0: predominant A-lines or < 3 separated B-lines.
• Score 1: at least 3 B-lines or coalescent B-lines occupying ≤ 50% of the screen without a clearly irregular pleural line.
• Score 1p: at least 3 B-lines or coalescent B-lines occupying ≤ 50% of the screen with a clearly irregular pleural line.
• Score 2: coalescent B-lines occupying > 50% of the screen without a clearly irregular pleural line.
• Score 2p: coalescent B-lines occupying > 50% of the screen with a clearly irregular pleural line.
• Score 3: large consolidations (at least > 1 cm).
The letters ‘p’ is irregular pleural line and are not counted in the score: this is a more qualitative information, which is useful because they are very frequent in COVID-19, which is compatible with the pathophysiology of the condition.6) The aeration score was calculated for each patient, and the mean was compared between the groups.
We counted the total number of SPCs in the 12 lung zones and calculated the mean difference between the groups.
Echocardiography
Elevated LVFP is indirectly evaluated by echocardiography, reflecting myocardial relaxation and stiffness diseases of the LV.7),8) Tissue Doppler imaging of early mitral annular velocity (e′) is a good indicator of LV myocardial relaxation. After measuring the transmitral peak early filling velocity, E is the ratio of E/e′ and is used as an indirect measure of LVFP. E/e′ lateral > 12, E/e′ mean > 13, or E/e′ septal > 15 indicates elevated LVFP, whereas E/e′ < 8 (any location) indicates normal LVFP.4)
In both systole and diastole, E and e′ velocities are measured from the apical 4-chamber view by placing a 5-mm sample volume over the lateral or medial region of the mitral annulus to cover the longitudinal excursion of the mitral annulus. The velocity scale is set to approximately 20 cm/sec above and below the zero-velocity baseline, and we reduced this to the minimum angulation between the plane of cardiac motion and the ultrasound beam. The recommendation for spectral recordings is a sweep speed of 50 to 100 mm/sec at end expiration.7)
The average from 3 consecutive cycles was measured for all reported echocardiographic measurements. LV volume and LV ejection fraction (EF) were assessed as recommended by the ASE.5) Mitral inflow was analyzed for peak E (early diastolic) and peak A (late diastolic) velocities, E/A ratio, and deceleration time (DT) of E velocity.
Right ventricle (RV) systolic function assessment depends primarily on the presence of 3 main measurements. 1) Tricuspid annular plane systolic excursion (TAPSE) represents longitudinal shortening of the RV. TAPSE is measured in the apical 4-chamber view by placing an M-mode cursor on the lateral tricuspid annulus and measuring the peak distance traveled by this reference point during systole with the normal reference limit being a TAPSE of ≥ 1.6 cm. 2) Tricuspid annular velocity (S′), reflects the longitudinal displacement of the tricuspid annulus during systole. S′ reflects the longitudinal velocity of the tricuspid annulus during systole and is measured in the apical 4-chamber view by placing a tissue Doppler cursor on the lateral tricuspid annulus and measuring the peak velocity of this point during systole. The normal reference limit is an S′ of ≥ 9.5 cm/s. 3) Fractional area change (FAC) is the percent change in RV area from diastole to systole in the apical 4-chamber view. It reflects the systolic function of the inflow and apical portions of the RV and is measured by manually tracing the contour of the RV at end-diastole and at end-systole. The FAC was calculated as follows: FAC = (end-diastolic RV area − end-systolic RV area)/end-diastolic RV. The normal reference limit for FAC is ≥ 35%.9)
Data analysis
Statistical analyses were performed using Statistical Package for the Social Sciences (IBM SPSS 19; IBM, Chicago, IL, USA). Patients with acute hypoxic respiratory failure admitted with suspicion of COVID-19 were divided into 2 groups: those with confirmed COVID-19 pneumonia and those with non-COVID-19 disease. Student's t-test was used to assess differences between the groups in the case of a normal distribution. Fisher's exact test was used for categorical data. Statistical significance was assumed at p < 0.05. Sensitivity, specificity, and positive and negative likelihood ratios of LUS and RT-PCR for the diagnosis of COVID-19 pneumonia were calculated.
RESULTS
Of 92 patients suspected to have COVID-19 pneumonia, 77 (84%) cases were confirmed. The median age of patients with COVID-19 pneumonia was 53 (36–82) years, and 64 (83%) were men. The median age of the patients without COVID-19 pneumonia was 68 (25–80), and 7 (47%) were men. The clinical characteristics of patients in relation to COVID-19 pneumonia confirmation are shown in Table 1.
Table 1. Clinical characteristics of the patients.
| Characteristics | Confirmed COVID-19 (77 cases; 84.4%) | Non-COVID-19 (15 cases; 15.5%) | Total (92 cases) | p-value | |
|---|---|---|---|---|---|
| Median age (IQR, years) | 53 (36–82) | 68 (25–80) | 0.001 | ||
| Male | 64 (83) | 7 (46) | 71 (77) | 0.005 | |
| Medical history | |||||
| IHD | 9 (11.6) | 10 (66.6) | 19 (20) | < 0.001 | |
| CABG | 2 (2.6) | 8 (53) | 10 (10.8) | 0.001 | |
| Hypertension | 20 (25.9) | 12 (80) | 32 (34.7) | 0.002 | |
| Diabetes mellitus | 25 (32) | 14 (93) | 39 (42) | < 0.001 | |
| COPD | 3 (3) | 3 (20) | 2 (2) | 0.020 | |
| Chronic renal impairment | 25 (32) | 10 (66.6) | 35 (38) | 0.015 | |
| Cancer | 0 | 1 (6.6) | 1 (1) | 0.091 | |
| Status on admission to ICU | |||||
| Acute MI | 2 (2.6) | 10 (66.6) | 12 (13) | 0.001 | |
| Acute PE | 9 (11.6) | 0 | 9 (9.7) | 0.186 | |
| Duration of symptoms (median in days) | 5 (2–10) | 2 (3–4) | |||
| Hypoxemia | All | All | 92 (100) | 0.020 | |
| PO2/FiO2 (mean) | 145 | 226 | 0.026 | ||
| HFNC | 35 (45.4) | 2 (13) | 50 (54) | 0.002 | |
| IV | 21 (27) | 4 (26.6) | 25 (27) | ||
| Facemask | 17 (22) | 2 (13) | 19 (20.6) | ||
| SOFA score (mean) | 7.7 | 6 | 0.390 | ||
| Echocardiogram findings (mean) | |||||
| E/A | 1.10 ± 9.56 | 1.80 ± 0.26 | 0.001 | ||
| DT (ms) | 252 ± 44 | 151 ± 39 | < 0.001 | ||
| E/e′ | 9.70 ± 4.00 | 19.00 ± 2.98 | < 0.001 | ||
| LVEF (%) | 66.0 ± 9.6 | 55.0 ± 14.8 | 0.010 | ||
| IVC < 2 cm | 62 (80) | 0 | < 0.001 | ||
| IVC > 2 cm | 15 (19.4) | 100 | < 0.001 | ||
| US chest finding | |||||
| Aeration score | 27.0 ± 9.0 | 21.0 ± 6.5 | 0.018 | ||
| SPC | 13.000 ± 10.700 | 1.000 ± 0.258 | < 0.001 | ||
Values are presented as number (%) or mean ± standard deviation.
COVID-19: coronavirus disease 2019; IQR: interquartile range; IHD: ischemic heart disease; CABG: coronary bypass surgery; COPD: chronic obstructive pulmonary disease; ICU: intensive care unit; MI: myocardial infarction ; PE: pulmonary embolism; PO2/FiO2: partial pressure of oxygen in arterial blood/fraction of inspired oxygen; HFNC: high-flow nasal cannula; IV: invasive ventilation; SOFA score: sequential organ failure assessment score; E/A: early diastolic transmitral flow/late diastolic transmitral flow by Doppler; DT: deceleration time; E/e': early diastolic transmitral flow by Doppler/early diastolic relaxation; LVEF: left ventricle ejection fraction; IVC: inferior vena cava; SPC: subpleural consolidation; US: ultrasound.
In the group of patients with confirmed COVID-19 pneumonia (Table 1), echocardiographic findings showed normal E/e′, DT, and E/A compared to non-COVID-19 patients (p = 0.001, 0.0001, 0.0001, respectively). The IVC diameter was < 2 cm with > 50% collapsibility in 62 (81%) patients with COVID-19 pneumonia, whereas patients with non-COVID-19 pneumonia had a diameter of > 2 cm and < 50% collapsibility, with a p-value of 0.001. There were 3 cases of myocarditis (3.9%) with poor EF, severe RV systolic dysfunction was seen in 9 cases (11.6%), and 3 cases exhibited RV thrombus. Acute myocardial infarction was observed in 2 cases of COVID-19 pneumonia (2.5%) and acute CVA in 6 (7.8%). Pulmonary hypertension with normal RV systolic function was identified in 11 patients (14%). We observed mild pericardial effusion with no constriction in 4 COVID-19 patients and none in non-COVID-19 cases.
The mean LVEF in non-COVID-19 pneumonia was 55%, but 3 cases had LVEF < 45%. The diagnoses among those with non-COVID pneumonia included one case of non-Hodgkin lymphoma postradiation pulmonary fibrosis that presented with hypertensive pulmonary edema. The other cases were 4 cases of chronic dialysis and pulmonary edema, a 25-year-old woman with miliary tuberculosis and severe myocarditis, 3 cases of NSTEMI and pulmonary edema, a 26-year-old man with uncontrolled hypertension, renal failure and possible vasculitis, one case of obstructive sleep apnea and right basal pneumonia and another 4 cases of hypertensive pulmonary edema with UTI.
Lung US revealed 4 signs suggestive of COVID-19 pneumonia in 77 patients (98.6%) (sensitivity 96.9%; confidence interval, 85%–99.5%) compared to RT-PCR results. The aeration score was significantly higher for COVID-19 pneumonia (p = 0.018), as was the total number of subpleural consolidations in the 12 zones of the chest (p < 0.0001) than for the non-COVID patients.
DISCUSSION
POCUS is of great importance as a bedside tool for immediately identifying types of acute respiratory failure and shock with guided hemodynamic management, and many protocols have been adopted and validated.10),11) Some protocols have adopted only LUS for fluid management and diagnosis of respiratory failure.12) In patients with COVID-19 pneumonia, LUS is utilized primarily for diagnostic purposes and is difficult to apply for fluid assessment due to lung pathology. The most common types of shock in COVID-19 pneumonia patients are septic, cardiogenic and massive pulmonary embolism with right ventricular dysfunction.2) Hypovolemia and hypovolemic shock should always be considered and can be assessed with echocardiography (decreased cardiac output, hyperdynamic left ventricle, IVC collapse and high respiratory variability, and LV filling pressures).
The frontline intensivist may grossly evaluate the level of LV filling pressure based on the qualitative interpretation of the mitral Doppler pattern (DT, E/A, E/e′), which helps differentiate between hydrostatic pulmonary edema and acute respiratory distress syndrome (ARDS).12) Accurate prediction of a predefined level of invasive pulmonary artery occlusion pressure by Doppler indices is of clinical value in both ventilated and nonventilated patients presenting with acute respiratory failure.13) Accordingly, combined Doppler indices appear to be of additional value for estimating LV filling pressure, principally in critically ill patients with underlying cardiac diseases known to alter diastolic properties and predict reload fluid responsiveness.14) Our COVID-19 positive patients were characterized by normal filling pressures, in contrast to patients with non-COVID-19 pneumonia. In patients with COVID-19, pneumonia presenting with hypotension and normal filling pressures can allow fluid administration and follow-up of the response.
The size variations of the IVC (collapsibility and distensibility) with respiration can serve as a predictor of a patient volume status. Greater than 50% collapse of the IVC correlates with intravascular volume depletion.15) Most of our patients had changes in IVC > 50% in diameter with respiration, except for patients with LV and RV dysfunction, which might indicate volume depletion at presentation. IVC variability combined with transmitral Doppler can assess the volume status of patients with circulatory instability and acute hypoxic respiratory failure.
The different forms of cardiac involvement observed in our prospective study are consistent with the cardiovascular disease observed in patients with other severe viral respiratory infections.16) Right ventricular dysfunction was observed in 11.6% of COVID-19 patients. Confirmed acute pulmonary embolism and right ventricular acute dilatation were observed in 9 patients, and thrombus was seen in the RV in 3 patients with acute deterioration and hypoxic respiratory and circulatory failure. These are likely to reflect severe respiratory disease in COVID-19 pneumonia and clinical and subclinical pulmonary thromboembolism due to coagulation dysfunction.17)
LV dysfunction was present in one-third of patients in the form of myocarditis and cardiogenic shock, pericarditis and acute myocardial infarctions. These results are in agreement with recently published data from the European Society of Cardiology, although the pathology of LV dysfunction in COVID-19 needs more clarification.18)
The sonographic appearance of the lungs in COVID-19 patients depends upon the time course of the illness. We chose to assess patients with early signs after admission because the method is important for early diagnosis, and the results are expected to be unique to COVID-19 pneumonia before the development of ARDS or secondary infection after admission to the ICU. Volpicelli et al.19) reported irregularity of the pleural line, bilateral patchy distribution of multiform clusters of B-lines, and multiple small peripheral consolidations.
Multiple consolidations of variable size were observed in the subpleural lesions. The subpleural consolidations were bilateral, and the echogenicity in the lesions was homogeneous and inhomogeneous. The subpleural consolidations are considered segmental infarcts.20) The subpleural consolidation was previously described in viral pneumonia and now in COVID-19 pneumonia, but not with other interstitial lung diseases.6) The grade 3 in aeration score, large subpleural consolidations > 1 cm, is considered, giving the higher aeration score in cases with COVID-19 pneumonia.6) On the other hand, we counted the small subpleural consolidations less than 1 cm, which does not contribute to the aeration score. The aeration score was higher in COVID-19 pneumonia, and SPC counting was almost zero in non-COVID-19 pneumonia patients, suggesting these factors as characteristic sonographic findings in COVID-19 pneumonia.
The limitation of our prospective study is the small number of patients and unequal patient numbers between the groups, as well as its categorization as a single-center study.
The majority of patients with COVID-19 pneumonia have normal to low LV filling pressures based on echocardiography. POCUS plays an important role in bedside diagnosis, hemodynamic assessment, and management of patients with acute hypoxic respiratory and circulatory failure in patients with COVID-19 pneumonia. Therefore, LUS may play an important role in the diagnosis of COVID-19 pneumonia.
Footnotes
Funding: The research was performed as part of the employment of the authors in Ministry of Health and Kuwait Oil Company.
Conflict of Interest: The authors have no financial conflicts of interest.
- Data curation: Bamasood OM.
- Methodology: Bitar ZI.
- Project administration: Bitar ZI.
- Resources: Bamasood OM, Maadarani OS.
- Supervision: Shamsah M, Bamasood OM, Alfoudri H.
- Validation: Maadarani OS.
- Visualization: Bitar ZI, Shamsah M.
- Writing - original draft: Bitar ZI.
- Writing - review & editing: Bitar ZI, Shamsah M, Alfoudri H.
References
- 1.Clerkin KJ, Fried JA, Raikhelkar J, Sayer G, Griffin JM, Masoumi A, et al. COVID-19 and Cardiovascular Disease. Circulation. 2020;141:1648–1655. doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]
- 2.Peng QY, Wang XT, Zhang LN Chinese Critical Care Ultrasound Study Group (CCUSG) Using echocardiography to guide the treatment of novel coronavirus pneumonia. Crit Care. 2020;24:143. doi: 10.1186/s13054-020-02856-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Johri AM, Galen B, Kirkpatrick JN, Lanspa M, Mulvagh S, Thamman R. ASE statement on point-of-care ultrasound during the 2019 novel coronavirus pandemic. J Am Soc Echocardiogr. 2020;33:670–673. doi: 10.1016/j.echo.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization. Volume 2. IMAI district clinician manual. Hospital care for adolescents and adults [Internet] Geneva: World Health Organization; 2011. [Accessed March 4, 2020]. Available at: https://apps.who.int/iris/bitstream/handle/10665/77751/9789241548290_Vol2_eng.pdf?sequence=3. [Google Scholar]
- 5.Peng QY, Wang XT, Zhang LN Chinese Critical Care Ultrasound Study Group (CCUSG) Findings of lung ultrasonography of novel corona virus pneumonia during the 2019-2020 epidemic. Intensive Care Med. 2020;46:849–850. doi: 10.1007/s00134-020-05996-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gargani L, Soliman-Aboumarie H, Volpicelli G, Corradi F, Pastore MC, Cameli M. Why, when, and how to use lung ultrasound during the COVID-19 pandemic: enthusiasm and caution. Eur Heart J Cardiovasc Imaging. 2020;21:941–948. doi: 10.1093/ehjci/jeaa163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nagueh SF, Appleton CP, Gillebert TC, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography. J Am Soc Echocardiogr. 2009;22:107–133. doi: 10.1016/j.echo.2008.11.023. [DOI] [PubMed] [Google Scholar]
- 8.Levitov AB, Mayo PH, Vastardis L. Echocardiographic Assessment of Left Ventricular Systolic and Diastolic Function, Critical Care Ultrasonography. 2nd ed. New York, NY: McGraw-Hill Global Education Holdings, LLC; 2015. [Google Scholar]
- 9.Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23:685–713. doi: 10.1016/j.echo.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 10.Perera P, Mailhot T, Riley D, Mandavia D. The RUSH exam: Rapid Ultrasound in SHock in the evaluation of the critically lll. Emerg Med Clin North Am. 2010;28:29–56. doi: 10.1016/j.emc.2009.09.010. [DOI] [PubMed] [Google Scholar]
- 11.Manson W, Hafez NM. The rapid assessment of dyspnea with ultrasound: RADiUS. Ultrasound Clin. 2011;6:261–276. [Google Scholar]
- 12.Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest. 2015;147:1659–1670. doi: 10.1378/chest.14-1313. [DOI] [PubMed] [Google Scholar]
- 13.Bouhemad B, Nicolas-Robin A, Benois A, Lemaire S, Goarin JP, Rouby JJ. Echocardiographic Doppler assessment of pulmonary capillary wedge pressure in surgical patients with postoperative circulatory shock and acute lung injury. Anesthesiology. 2003;98:1091–1100. doi: 10.1097/00000542-200305000-00011. [DOI] [PubMed] [Google Scholar]
- 14.Boussuges A, Blanc P, Molenat F, Burnet H, Habib G, Sainty JM. Evaluation of left ventricular filling pressure by transthoracic Doppler echocardiography in the intensive care unit. Crit Care Med. 2002;30:362–367. doi: 10.1097/00003246-200202000-00016. [DOI] [PubMed] [Google Scholar]
- 15.Lang RM, Bierig M, Devereux RB, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18:1440–1463. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 16.Madjid M, Miller CC, Zarubaev VV, et al. Influenza epidemics and acute respiratory disease activity are associated with a surge in autopsy-confirmed coronary heart disease death: results from 8 years of autopsies in 34,892 subjects. Eur Heart J. 2007;28:1205–1210. doi: 10.1093/eurheartj/ehm035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18:1421–1424. doi: 10.1111/jth.14830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dweck MR, Bularga A, Hahn RT, et al. Global evaluation of echocardiography in patients with COVID-19. Eur Heart J Cardiovasc Imaging. 2020;21:949–958. doi: 10.1093/ehjci/jeaa178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Volpicelli G, Gargani L. Sonographic signs and patterns of COVID-19 pneumonia. Ultrasound J. 2020;12:22. doi: 10.1186/s13089-020-00171-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zotzmann V, Lang CN, Bamberg F, Bode C, Staudacher DL. Are subpleural consolidations indicators for segmental pulmonary embolism in COVID-19? Intensive Care Med. 2020;46:1109–1110. doi: 10.1007/s00134-020-06044-z. [DOI] [PMC free article] [PubMed] [Google Scholar]