Abstract
Sixteen volunteers each drank 700 ml sugar-containing soft drink during two successive periods and the blood sugar was measured at 10 min intervals together with electrical impedance spectroscopy and near infrared spectroscopy (NIR). A maximum correlation of 0.46 was found for the electrical measurements but no clear separation between low and high blood glucose levels were found in the NIR measurements. The latter was attributed to the experimental design where the NIR probe was removed from the skin between each measurement.
Keywords: Bioimpedance, near-infrared spectroscopy, NIR, glucose, diabetes
Introduction
The number of diabetes patients is increasing in numbers worldwide [1,2]. Even with adequate treatment and follow-up, morbidity and mortality is higher than in the general population [3,4,5,6]. To control blood glucose levels, patients may use the standard finger-prick method or one of the continuous blood glucose meters that have been developed over the later years [7]. Non-invasive methods (not involving piercing the skin) for measuring blood glucose have been investigated for decades [8], but a working device with adequate accuracy does not exist today. A method for easy and pain-free blood glucose measurement would be revolutionary in the treatment of diabetes.
Materials and methods
Sixteen test subjects participated in this study, eight female and eight male, mean age 24 (20–37) years. None of them suffered from diabetes or had health problems that were relevant for this study. They fasted overnight before partaking in the experiments.
The measurements were conducted in a laboratory with normal room temperature of around 22 °C. After a period of acclimatization (including information, informed consent, electrode attachment, etc.), their blood glucose was measured using a FreeStyle Freedome Lite device from Abbott Diabetes Care Ltd (Witney, UK).
Electrical impedance was then measured on the ventral side of the left underarm, using a frequency extended ISX-3 impedance analyzer from Sciospec Scientific Instruments GmbH (Bennewitz, Germany) and two KittyCat hydrogel electrodes (Kendall, KittyCat®, 1050NPSM) in a two-electrode system. One electrode was placed 2 cm below the elbow pit (antecubital fossa) and the other 10 cm further down the underarm towards the wrist. The measurements were done in the frequency range 100 Hz to 40 MHz using 400 mV p-p.
Near-infrared reflectometry was used on the same area of the right underarm using an Ocean Optics NIRQuest® 512 in the range 900 – 1700 nm (Ocean Insight, NY, USA).
This procedure was repeated every ten minutes. After the third measurement series, the test subjects were given 7 dl of a sugar-containing soft drink to drink (approximately 106 gram sugar per liter). The measurements then continued while the blood sugar level increased, peaked and then returned approximately to the initial level.
During this period, the test subjects were free to walk around or use a stationary bike for very light exercise between the measurements. They were then given another 7 dl of soft drink and the measurements continued until the measured blood sugar level had peaked and again showed a negative trend.
The statistical analysis presented in this article was performed using Matlab® 2018b.
Informed consent
Informed consent has been obtained from all individuals included in this study.
Ethical approval
The research related to human use has been complied with all relevant national regulations, institutional policies and in accordance with the tenets of the Helsinki Declaration, and has been approved by the authors’ institutional review board or equivalent committee.
Results
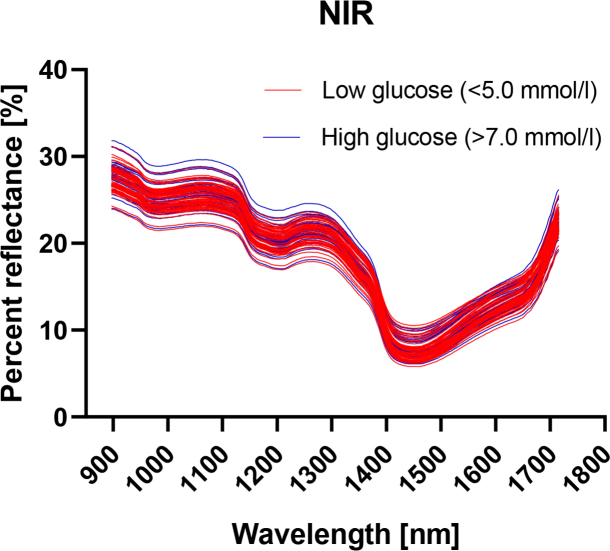
Figure 1 shows all the unprocessed NIR spectra for glucose levels below 5.0 mM (blue lines) or above 7.0 mM (red lines) plotted together. From visual inspection, there is no clear separation between the spectra for the two groups.
Fig.1.
Unprocessed NIR spectra for glucose levels below 5.0 mM (blue lines) or above 7.0 mM (red lines).
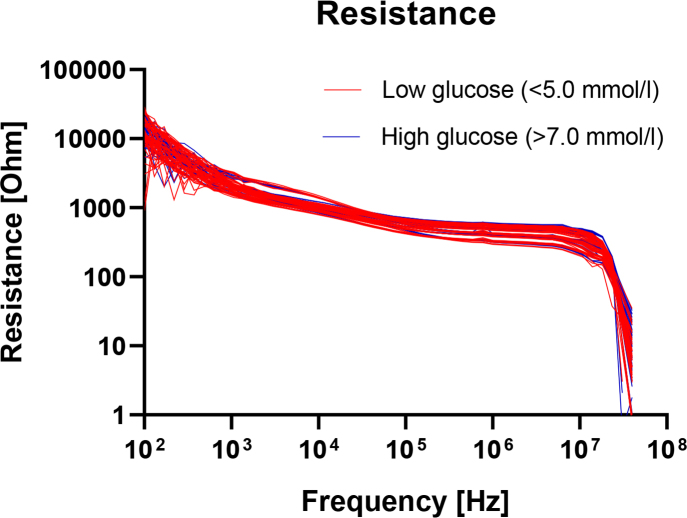
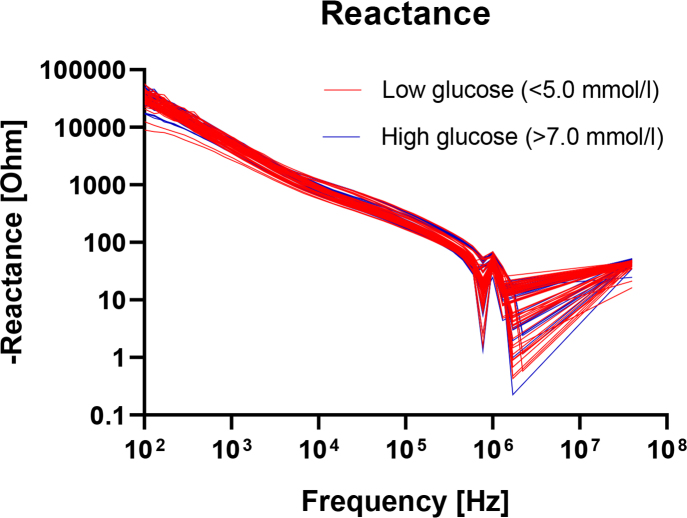
In figure 2, the real part of the measured electrical impedance, the resistance R, is plotted as a function of frequency. Here also, the curves are color coded with levels below 5.0 mM in blue and above 7.0 mM in red. The reactance X is correspondingly plotted in figure 3. There are no obvious differences between the groups in these plots either, when assessed purely by visual inspection.
Fig.2.
Measured resistance as a function of frequency for glucose levels below 5.0 mM (blue lines) or above 7.0 mM (red lines).
Fig.3.
Measured reactance as a function of frequency for glucose levels below 5.0 mM (blue lines) or above 7.0 mM (red lines).
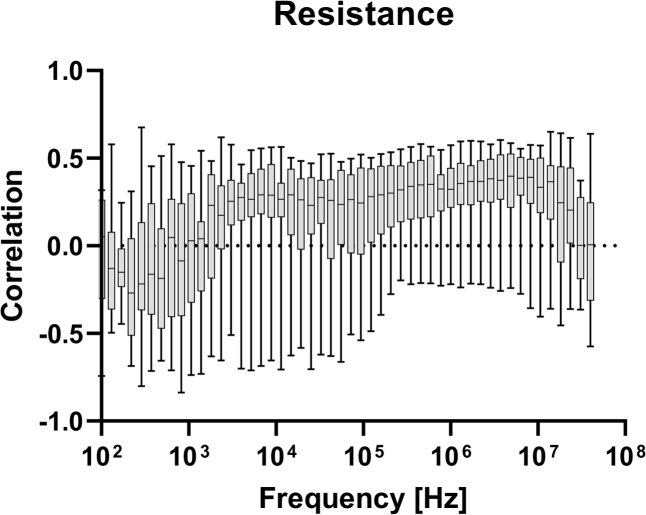
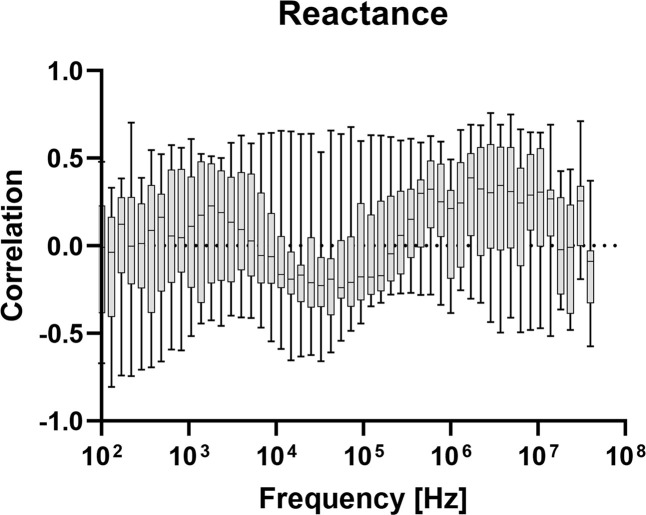
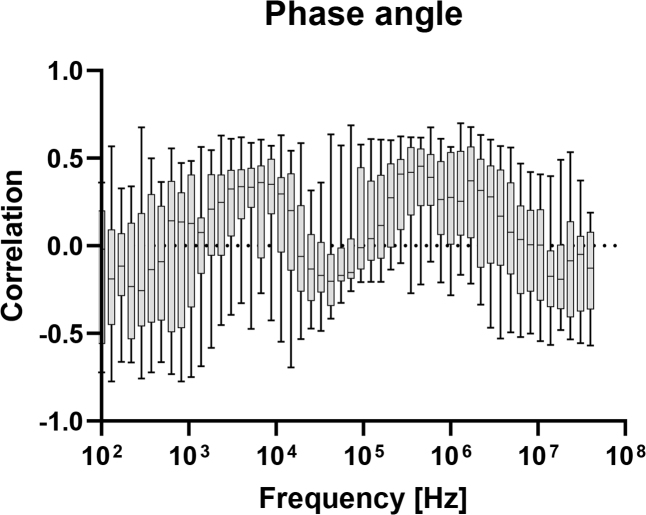
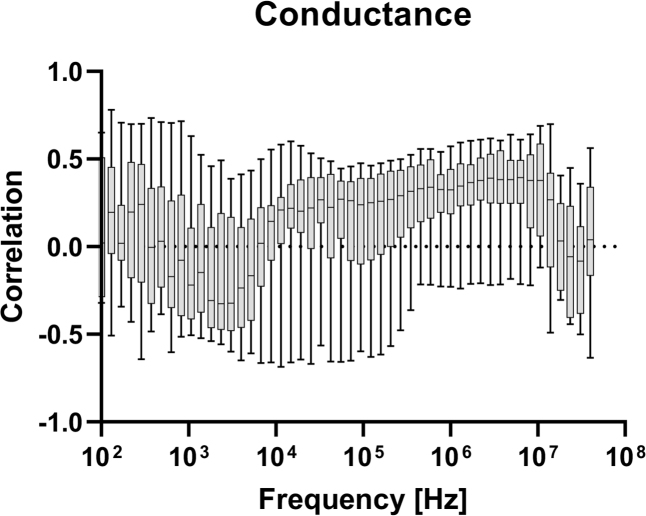
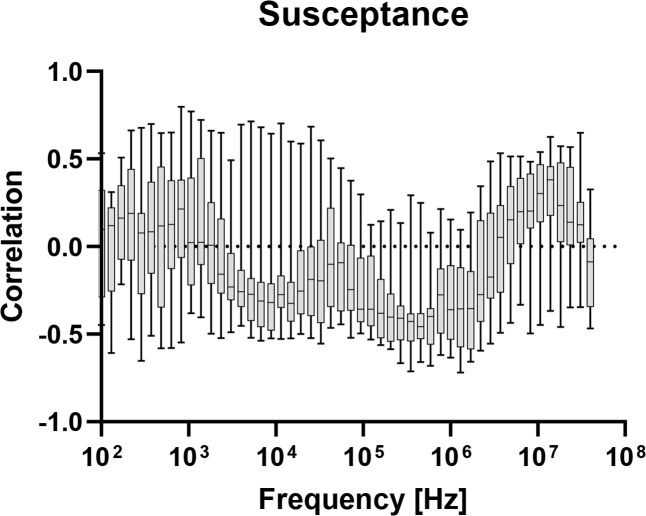
In figures 4–8, correlation plots have been made for the impedance and admittance components, namely resistance, reactance, phase angle, conductance and susceptance, respectively. They are all based on Pearson product-moment correlation coefficient calculations. Based on this analysis, susceptance at 455.5 kHz shows the highest correlation of about −0.46. This correlation is not high enough for prediction of blood glucose and hence multivariate models are tested.
Fig.4.
Correlation between measured resistance at different frequencies, and blood glucose level.
Fig.5.
Correlation between measured reactance at different frequencies, and blood glucose level.
Fig.6.
Correlation between measured phase angle at different frequencies, and blood glucose level.
Fig.7.
Correlation between measured conductance at different frequencies, and blood glucose level.
Fig.8.
Correlation between measured susceptance at different frequencies, and blood glucose level.
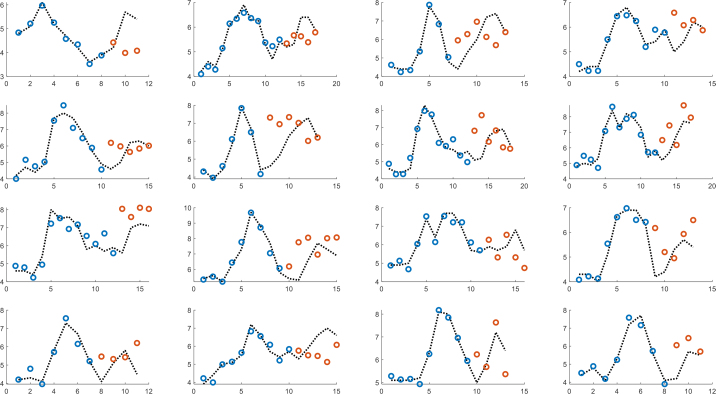
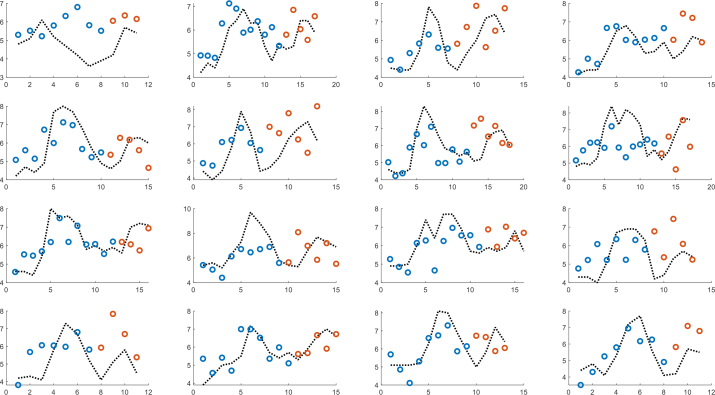
Figure 9 shows the results of multivariate partial least squares (PLS) regression, where the models were individually trained until the start of intake of sugar-containing soft drink for the second time and thereafter used for prediction. The model is based on resistance, reactance and NIR reflectance and three components are used. Correspondingly, figure 10 shows a globally trained PLS model with five components, where all individuals are part of the model.
Fig.9.
PLS model with three components based on NIR, resistance and reactance. Individually trained until the start of intake of sugar-containing soft drink for the second time (blue dots) and thereafter used for prediction (orange dots).
Fig.10.
PLS model with five components based on NIR, resistance and reactance. Globally trained until the start of intake of sugar-containing soft drink for the second time (blue dots) and thereafter used for prediction orange dots).
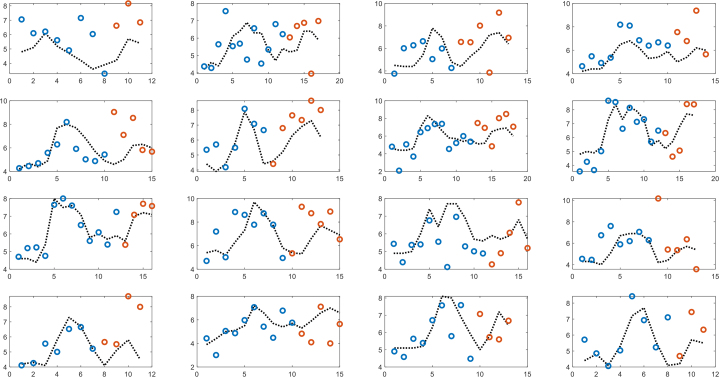
Similarly, figure 11 shows an artificial neural network (ANN) model with the same inputs (R, X, and NIR), which is globally trained until the start of intake of sugar-containing soft drink for the second time and thereafter used for prediction. The ANN has one hidden layer and five nodes, and the Tansig activation function and Levenberg-Marquardt training algorithm were used.
Fig.11.
ANN model with one hidden layer and five nodes based on NIR, resistance and reactance. Globally trained until the start of intake of sugar-containing soft drink for the second time (blue dots) and thereafter used for prediction orange dots).
Discussion
The NIR measurements did not reveal any clear separation in the reflection spectra for blood glucose levels below 5.0 mM and above 7.0 mM. Earlier studies have for example found a mean absolute relative difference (MARD) of 32.7% (see e.g. [9] for discussion of MARD) when using NIR and temperature to predict blood glucose [10]. Visual inspection of the data presented in figure 1 clearly demonstrates the lack of any meaningful correlation. There could be several reasons for this: The band used by Tronstad et al. [10] was 880 – 2200 nm. According to [11] and [12] the best optical wavelength bands for detection of the glucose molecule with NIR, are 1500 – 1850 nm and 2050 – 2392 nm. Hence Tronstad et al. [10] covered a part of the upper band that our study did not cover.
Another difference in the experimental designs was that we removed the NIR probe between each measurement. Although measures were taken to ensure placement of the probe on the same skin site each time, we cannot ignore the possibility that this lead to a higher variance between the measurements that is not related to the glucose level.
The highest correlation for the electrical admittance measurements was found for the suceptance at 455.5 kHz. At that frequency, the measurements will be dominated by deeper, viable tissue and the susceptance will mainly represent the dielectric charging of the cell membranes in the β dispersion range [13]. Hayashi et al. [14] explain how altered glucose level will change both the conductivity and permittivity of the cell membrane with positive or negative correlation dependent on the glucose concentration.
The PLS and ANN models show that the measured bioimpedance and NIR data are of limited value for prediction of blood glucose level. Individually trained models obviously have the potential to be more closely adapted to each subject and with this design where the glucose level increases in two distinct periods, this should indeed be possible. However, the number of available measurements per subject is very limited in this study. In that sense, the globally trained models have the advantage of a significantly higher number of measurements, but the combined data sets will contain individual variations that may act as noise in the model. In general, a global model would be preferable since it reduces the need for individual calibration.
Conclusion
The possible prediction of blood glucose was tested using NIR and bioimpedance spectroscopy. Although a moderate correlation was found for some of the electrical parameters, it was not sufficient to produce a satisfactory prediction model. The quality of the NIR measurements was probably reduced due to the removal and reattachment of the NIR probe between each measurement.
Footnotes
Conflict of interest
Authors state no conflict of interest.
References
- 1.Collaboration NCDRF. Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet. 2016;387:1513–1530. doi: 10.1016/S0140-6736(16)00618-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Egro FM. Why is type 1 diabetes increasing? J Mol Endocrinol. 2013;51:R1–13. doi: 10.1530/JME-13-0067. [DOI] [PubMed] [Google Scholar]
- 3.Jorgensen ME, Almdal TP, Carstensen B. Time trends in mortality rates in type 1 diabetes from 2002 to 2011. Diabetologia. 2013;56:2401–2404. doi: 10.1007/s00125-013-3025-7. https://doi.org/10.1007/s00125-013-3025-7 . [DOI] [PubMed] [Google Scholar]
- 4.Laing SP, Swerdlow AJ, Slater SD, Burden AC, Morris A, Waugh NR, Gatling W, Bingley PJ, Patterson CC. Mortality from heart disease in a cohort of 23,000 patients with insulin-treated diabetes. Diabetologia. 2003;46:760–765. doi: 10.1007/s00125-003-1116-6. https://doi.org/10.1007/s00125-003-1116-6 . [DOI] [PubMed] [Google Scholar]
- 5.Secrest AM, Becker DJ, Kelsey SF, LaPorte RE, Orchard TJ. All-cause mortality trends in a large population-based cohort with long-standing childhood-onset type 1 diabetes: the Allegheny County type 1 diabetes registry. Diabetes Care. 2010;33:2573–2579. doi: 10.2337/dc10-1170. https://doi.org/10.2337/dc10-1170 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Livingstone SJ, Looker HC, Hothersall EJ, Wild SH, Lindsay RS, Chalmers J, Cleland S, Leese GP, McKnight J, Morris AD, Pearson DW, Peden NR, Petrie JR, Philip S, Sattar N, Sullivan F, Colhoun HM. Risk of cardiovascular disease and total mortality in adults with type 1 diabetes: Scottish registry linkage study. PLoS Medicine. 2012;9:e1001321. doi: 10.1371/journal.pmed.1001321. https://doi.org/10.1371/journal.pmed.1001321 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rodbard D. Continuous Glucose Monitoring: A Review of Recent Studies Demonstrating Improved Glycemic Outcomes. Diabetes Technology & Therapeutics. 2017;19:S25–S37. doi: 10.1089/dia.2017.0035. https://doi.org/10.1089/dia.2017.0035 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lin T. Non-Invasive Glucose Monitoring: A Review of Challenges and Recent Advances. Curr Trends Biomedical Eng & Biosci. 2017;6(5) 555696. https://doi.org/10.19080/CTBEB.2017.06.555696 . [Google Scholar]
- 9.Danne Thomas. et al. International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care. 2017;40(12):1631–1640. doi: 10.2337/dc17-1600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tronstad C., Elvebakk O., Staal O.M., Kalvøy H., Høgetveit J.O., Jenssen T.G., Birkeland K.I., Martinsen Ø.G.. Non-invasive prediction of blood glucose trends during hypoglycemia. Analytica Chimica Acta. 2019;1052:37–48. doi: 10.1016/j.aca.2018.12.009. https://doi.org/10.1016/j.aca.2018.12.009 . [DOI] [PubMed] [Google Scholar]
- 11.Yadav J., Rani A., Singh V., Murari B.M.. Prospects and limitations of noninvasive blood glucose monitoring using near-infrared spectroscopy. Biomed. Signal Process. Control. 2015;18:2014–2227. https://doi.org/10.1016/j.bspc.2015.01.005 . [Google Scholar]
- 12.Rong L., Wenliang C., Kexin X.. Next step of non-invasive glucose monitor byNIR technique from the well controlled measuring condition and results. Opt. Quant. Electron. 2005;37:1305–1317. https://doi.org/10.1109/apbp.2004.1412258 . [Google Scholar]
- 13.Grimnes S, Martinsen ØG. Bioimpedance and bioelectricity basics. 3rd edition. Academic Press; 2015. https://doi.org/10.1016/b978-0-12-411470-8.00011-8 . [Google Scholar]
- 14.Hayashi Y., Livshits L., Caduff A., Feldman Y.. Dielectric spectroscopy study of specific glucose influence on human erythrocyte membranes. J. Phys. Appl. Phys. 2002;36:369–374. https://doi.org/10.1088/0022-3727/36/4/307 . [Google Scholar]