Abstract
Background:
Broad spectrum sunscreens with a Sun Protection Factor (SPF) of 15 or greater are indicated to decrease the risk of skin cancer and early skin aging caused by the sun if used as directed with other sun protection measures. To determine whether sunscreen product performance is compromised under solar exposure and to test spectral uniformity of protection across the UVA spectrum, we tested broad spectrum sunscreens with a variety of active pharmaceutical ingredients (APIs) and in a variety of dosage forms.
Methods:
A cross-sectional market survey of 32 sunscreen drug products containing either organic or inorganic APIs with SPFs of 15, 30, 50 and 70 were tested. UV doses were delivered via natural sun in Silver Spring, Maryland between June and September of 2017.
Results:
Of the 32 sunscreen drug products, 6 products failed to meet their broad spectrum claim under solar exposure. Using FDA’s new proposal to strengthen sunscreen broad spectrum requirements, spectral uniformity based on the mean sunscreen absorbance of UVA1(340–400 nm)/UV (290–400 nm) indicated that ~40% of sunscreen drug products tested had suboptimal UVA protection.
Conclusion:
US consumers may unknowingly be receiving up to 36% more transmitted UVA when selecting between similarly labeled broad spectrum sunscreen drug products with equivalent SPF values. FDA’s new proposal may help decrease consumers’ overall lifetime UVA burden. Spectral absorbance data on sunscreen performance can be used to further improve the coupling of broad spectrum protection to a product’s SPF value so that consumers have improved proportional increases in UV protection.
Keywords: ultraviolet radiation, sunscreen, skin cancer, sun protection factor, broad spectrum
INTRODUCTION
U.S. consumers are faced with the challenge of preventing overexposure to UV radiation (UVR) while participating in most outdoor activities. To further complicate matters, everyone’s UV sensitivity profile is unique.1 Striking a balance between maintaining a healthy active lifestyle and mitigating risks of UVR overexposure is at the core of the Surgeon General’s Call to Action to Prevent Skin Cancer.2 It is no surprise, therefore, that topical protection using over-the-counter (OTC) sunscreen drug products is one of the most commonly used sun protection measures during intentional sun exposure. The use of OTC sunscreens that are effective at preventing sunburn (primarily from UVB (290 – 320 nm)), but less effective at protecting against the greater portion of sun exposure (UVA) may also be complicit in allowing chronic accumulation of UVA (320 – 400 nm) doses in consumers who believe themselves protected. UVR overexposure in consumers using sunscreens designed predominantly to protect against sunburn raises a concern of inadequate UVA protection with potential for long-term consequences.
The life-long use of sunscreens with greater frequency and in greater amounts across the gradient of skin types in the US population (including in young children and pregnant women) has led the FDA to seek additional safety data on 12 sunscreen active ingredients including whether they are dermally absorbed through human skin.3 Recent evidence from an exploratory study evaluating dermal absorption under maximal use conditions corroborates FDA’s request for additional safety data outlined in the recently issue proposed rule.3,4 Across this changing sunscreen landscape, some regions in the US (such as Hawaii and Key West, Florida) have passed legislation prohibiting distribution of sunscreens with certain active ingredients due to emerging ecological concerns.5,6 Ensuring the efficacy as well as the safety of sunscreens is a key factor in promoting public health and the basis for the proposal for improved SPF and broad spectrum protections in FDA’s proposed rule. Given the public health benefits of sunscreen use, FDA strongly recommends that consumers continue to use sunscreen in conjunction with other sun protective measures while data are developed.7
UVR is known to be a carcinogen.8,9 This is a particular concern for fair-skinned populations living in high ambient UVR areas where data shows increased non-melanoma skin cancer incidence rates.10,11 In contrast, the etiology of melanoma is suggested not to be determined by the cumulative lifetime exposure to UV, but rather the magnitude and interval of UV doses.12–15 Since the beginning of the decade, the scientific evidence linking UVA to skin cancer and related risks continues to grow16–23 and supports the hypothesis put forth earlier regarding excessive UVA exposure from sunlamps.24,25 In light of scientific evidence on UVA, the FDA has recently proposed (1) to require all sunscreen drug products SPF ≥ 15 become broad spectrum and (2) to strengthen the broad spectrum definition by requiring a UVAI/UV ratio of 0.7 or higher.3
Current dogma suggests that broad spectrum sunscreen drug products with increasing Sun Protection Factor (SPF) values also possess increasing magnitude of UVA protection under actual solar conditions. Broad spectrum sunscreen drug products commercially available in the U.S. are required to substantiate their broad spectrum label claim by passing the in vitro broad spectrum test procedure defined in the 21 CFR 201.327(j) using a standardized UV dose from a solar simulator. Regardless of the laboratory tested broad spectrum claim (which manufacturers must conduct and maintain testing records for), what is more critical is whether under actual solar conditions the proposed broad spectrum definition, in fact, discriminates products with more optimal UVA protection compared to the existing broad spectrum definition. Our objectives were to determine whether sunscreen product performance is compromised under actual solar exposure and to test spectral uniformity across the UVA spectrum. To further evaluate sunscreen performance, consumer storage of sunscreens under stress conditions in the hot environment of a motor vehicle was also evaluated. We hypothesized that spectral absorbance data on sunscreen performance can be used to further improve the coupling of a sunscreen’s SPF value to its broad spectrum protection so that consumers have improved proportional increases in UVB and UVA protection.
MATERIALS AND METHODS
OTC Sunscreen Drug Products:
Under outdoor solar exposure (Table 1), 32 commercially available broad spectrum sunscreens (from 23 sunscreen brands) with SPFs in the range of 15–70 were tested. All tested sunscreens contained at least one of the following UVA protective active ingredients (1) avobenzone, (2) titanium dioxide or (3) zinc oxide (Table 2). Due to the sample size and frequent reformulation of sunscreen products (requiring retesting for efficacy) that change ingredients/concentrations from year to year under the same brand label, the identities of the sunscreens were blinded. The in vitro broad spectrum test procedure defined in 21 CFR 201.327(j) (www.ecfr.gov) was followed, with the exception of substituting outdoor solar exposure for the solar simulator used during the sunscreen product pre-irradiation along with an additional intent-to-stress arm of the study (see Methods in Appendix A of Supplemental Materials). The need for IRB approval does not apply to this study.
Table 1.
Basic parameters of solar exposures used during sunscreen product pre-irradiation
| UVA (320–400 nm) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Solar Exposure Dates | UVA | UVB | UVA | UVB | Erythemogenic output1 | SEDs in 2hrs | UVA dose per 1 SED | UVA dose in 2 hrs |
| % | % | (W/m2) | (W/m2) | (W/m2, eff) | J/cm2 | |||
| 6/28/2017 | 95.3% | 4.7% | 37.04 | 1.83 | 0.12 | 8.79 | 3.04 | 26.67 |
| 6/30/2017 | 94.8% | 5.2% | 48.86 | 2.71 | 0.20 | 14.36 | 2.45 | 35.18 |
| 7/3/2017 | 95.7% | 4.3% | 46.29 | 2.10 | 0.15 | 10.77 | 3.09 | 33.33 |
| 7/20/2017 | 94.9% | 5.1% | 41.79 | 2.26 | 0.16 | 11.78 | 2.55 | 30.09 |
| 8/1/2017 | 94.9% | 5.1% | 49.23 | 2.63 | 0.19 | 13.67 | 2.59 | 35.45 |
| 9/25/2017 | 95.2% | 4.8% | 37.42 | 1.87 | 0.13 | 9.38 | 2.87 | 26.94 |
| 9/28/2017 | 95.3% | 4.7% | 39.00 | 1.94 | 0.13 | 9.64 | 2.91 | 28.08 |
To obtain erythemogenic outputs, the spectral irradiances (290–400 nm) were wavelength weighted using the International Commission on Illumination (CIE) Reference Action Spectrum for Erythema. The Standard Erythemal Dose (SED) equals 100 J/m2 erythema-effective.
Table 2.
OTC sunscreen drug product performance characteristics before and after solar exposure.
| Broad Spectrum SPF | Sunscreen ID1 | Active Pharmaceutical Ingredients2 (Concentration %) | Before
UV CW Mean |
After
UV CW Mean |
Before
UV UVA1/UV Mean |
After
UV UVA1/UV Mean3 |
Before
UV UVA/UVB Mean |
After
UV UVA/UVB Mean |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AVO | ECA | HMS | OMC | OS | OCT | OXY | TiO2 | ZnO | Total % | |||||||||
| 15 | L1 | 5 | 5 | 10 | 364.9 | 370.3 | 0.38 | 0.47 | *** | 0.29 | 0.34 | |||||||
| L2 | 2 | 2 | 10 | 14 | 376.0 | 374.2 | 0.84 | 0.74 | *** | 0.83 | 0.65 | |||||||
| SP1 | 1.5 | 5 | 3.5 | 10 | 376.2 | 375.0 | 0.81 | 0.78 | *** | 0.69 | 0.64 | |||||||
| L3 | 9.5 | 9.5 | 375.0 | 375.0 | 0.88 | 0.88 | ** | 0.83 | 0.82 | |||||||||
| L4 | 3 | 4 | 5 | 12 | 379.7 | 377.4 | 0.96 | 0.98 | *** | 0.96 | 0.97 | |||||||
| 30 | L5 | 7.5 | 4.5 | 3 | 15 | 349.8 | 355.8 | 0.25 | 0.30 | *** | 0.24 | 0.25 | ||||||
| L6 | 1 | 5 | 7.5 | 5 | 6 | 24.5 | 372.9 | 357.3 | 0.65 | 0.41 | *** | 0.52 | 0.35 | |||||
| L7 | 3 | 6.5 | 7.5 | 5 | 2 | 24 | 376.5 | 369.3 | 0.74 | 0.58 | *** | 0.59 | 0.44 | |||||
| ST1 | 2 | 15 | 7.5 | 5 | 29.5 | 370.7 | 370.1 | 0.60 | 0.57 | *** | 0.44 | 0.41 | ||||||
| ST2 | 3 | 10 | 10 | 23 | 374.2 | 375.0 | 0.76 | 0.76 | NS | 0.64 | 0.64 | |||||||
| L8 | 22 | 22 | 375.2 | 375.1 | 0.86 | 0.86 | NS | 0.80 | 0.80 | |||||||||
| SP2 | 12 | 12 | 376.3 | 376.7 | 0.73 | 0.74 | *** | 0.54 | 0.55 | |||||||||
| L9 | 3 | 10 | 5 | 10 | 28 | 379.9 | 377.0 | 0.85 | 0.78 | *** | 0.75 | 0.66 | ||||||
| P1 | 15 | 12 | 27 | 383.1 | 383.0 | 0.85 | 0.84 | ** | 0.73 | 0.73 | ||||||||
| 50 | L10 | 3 | 15 | 7.5 | 5 | 3 | 6 | 39.5 | 374.5 | 368.7 | 0.70 | 0.61 | *** | 0.57 | 0.53 | |||
| L11 | 7.5 | 10 | 17.5 | 369.3 | 369.4 | 0.62 | 0.62 | NS | 0.50 | 0.50 | ||||||||
| L12 | 7.5 | 5 | 14.5 | 27 | 370.1 | 369.8 | 0.56 | 0.57 | * | 0.41 | 0.42 | |||||||
| L13 | 2 | 17 | 19 | 373.0 | 372.3 | 0.74 | 0.74 | *** | 0.62 | 0.62 | ||||||||
| ST3 | 6 | 4.7 | 10.7 | 372.3 | 372.3 | 0.64 | 0.63 | NS | 0.51 | 0.50 | ||||||||
| F1 | 3 | 10 | 7.5 | 5 | 25.5 | 376.2 | 372.7 | 0.73 | 0.64 | *** | 0.57 | 0.47 | ||||||
| L14 | 7.3 | 4.3 | 11.6 | 371.9 | 373.3 | 0.63 | 0.64 | NS | 0.56 | 0.57 | ||||||||
| O1 | 3 | 10 | 7.5 | 5 | 25.5 | 377.1 | 376.1 | 0.80 | 0.77 | *** | 0.68 | 0.64 | ||||||
| L15 | 2.7 | 8 | 4.5 | 5 | 20.2 | 379.0 | 376.3 | 0.85 | 0.81 | *** | 0.73 | 0.69 | ||||||
| L16 | 3 | 10 | 5 | 10 | 28 | 376.9 | 376.3 | 0.77 | 0.77 | NS | 0.63 | 0.63 | ||||||
| SP3 | 3 | 10 | 5 | 4 | 5 | 27 | 379.4 | 377.5 | 0.83 | 0.80 | *** | 0.74 | 0.69 | |||||
| L17 | 19 | 19 | 378.0 | 379.0 | 0.91 | 0.91 | *** | 0.86 | 0.87 | |||||||||
| P2 | 14.2 | 4.9 | 19.1 | 380.1 | 380.2 | 0.73 | 0.73 | NS | 0.58 | 0.58 | ||||||||
| 70 | L18 | 2 | 15 | 5 | 2 | 6 | 30 | 377.9 | 371.9 | 0.79 | 0.67 | *** | 0.68 | 0.54 | ||||
| L19 | 3 | 15 | 5 | 10 | 6 | 39 | 376.1 | 372.6 | 0.76 | 0.68 | *** | 0.65 | 0.56 | |||||
| ST4 | 3 | 15 | 5 | 10 | 3 | 36 | 373.3 | 373.3 | 0.71 | 0.72 | * | 0.58 | 0.59 | |||||
| L20 | 3 | 10 | 5 | 4.5 | 4 | 26.5 | 377.1 | 374.4 | 0.79 | 0.75 | *** | 0.67 | 0.64 | |||||
| SP4 | 3 | 15 | 5 | 4 | 6 | 33 | 376.3 | 375.7 | 0.77 | 0.75 | *** | 0.66 | 0.64 | |||||
| Intent-to-stress | ||||||||||||||||||
| 15 | L1 | 5 | 5 | 10 | 361.6 | 368.5 | 0.33 | 0.42 | *** | 0.25 | 0.31 | |||||||
| L2 | 2 | 2 | 10 | 14 | 375.5 | 375.1 | 0.83 | 0.80 | *** | 0.81 | 0.74 | |||||||
| 50 | L13 | 2 | 17 | 19 | 373.3 | 373.0 | 0.74 | 0.74 | * | 0.62 | 0.62 | |||||||
| L14 | 7.3 | 4.3 | 11.6 | 375.8 | 376.1 | 0.72 | 0.71 | *** | 0.64 | 0.63 | ||||||||
| 70 | L18 | 2 | 15 | 5 | 2 | 6 | 30 | 377.0 | 371.6 | 0.77 | 0.67 | *** | 0.66 | 0.54 | ||||
| L19 | 3 | 15 | 5 | 10 | 6 | 39 | 375.9 | 372.6 | 0.75 | 0.68 | *** | 0.64 | 0.55 | |||||
| L20 | 3 | 10 | 5 | 4.5 | 4 | 26.5 | 376.4 | 374.3 | 0.78 | 0.74 | *** | 0.66 | 0.62 | |||||
The grey shaded rows indicate failure to meet broad spectrum claim (mean CW ≥ 370 nm) under solar exposure.
Dosage forms identified by first letter of ID: L = Lotion, SP = Spray, ST = Stick, F = Foam/Mousse, P = Powder, O = Oil
AVO = avobenzone, ECA = ecamsule, HMS = homosalate, OMC = octinoxate, OS = octisalate, OCT = octocrylene, OXY = oxybenzone, TiO2 = titanium dioxide, ZnO = zinc oxide
Statistical significant changes in spectral uniformity (UVAI/UV) before and after irradiation: *** = p < 0.0001, ** = p < 0.005, * = p < 0.05, NS = not significant
In vitro Broad Spectrum Test and Metrics of UVA protection:
Briefly, as defined in the in vitro broad spectrum test procedure in 21 CFR 201.327(j), sunscreen products were applied to optical-grade PMMA plates with a roughened three-dimensional surface topography (6 μm) at 0.75 mg/cm2. Pre-irradiation with a solar exposure with an equivalent erythemal effective dose of 800 J/m2 (i.e., 800 J/m2-eff) was used for each sunscreen. The broad spectrum test procedure in 21 CFR 201.327(j) requires only measurements of pre-irradiated sunscreen samples, but in this study the sunscreens were evaluated prior to solar exposure (unexposed controls) and after pre-irradiation with solar irradiation. Diffuse collection of sunscreen transmittance and absorbance were documented using a Lambda 1050 UV/Vis/NIR Spectrophotometer fitted with a 150 mm Spectralon integrating sphere (Perkin Elmer, Waltham, MA). Mean transmittance and absorbance were calculated for each wavelength between 290 nm to 400 nm for five different locations on three plates for a total of 15 measurements. Spectral transmittance (T(λ)) values were converted into absorbance (A(λ)) values using the relationship A(λ) = −log T(λ). The critical wavelength (CW) or λc is defined as the wavelength at which 90% of the integral of spectral absorbance curve is reached between 290 – 400 nm:
A mean CW ≥ 370 nm indicates that the tested sunscreen drug product meets its labeled broad spectrum protection claim. The absorbance profile, CW, photoinstability (ΔT (%)) and UVA protection metrics (UVA/UVB and UVAI/UV) were evaluated before and after solar irradiation. The calculation of the UVA/UVB and UVAI/UV ratio makes further use of the calculated mean A(λ) as defined in 21 CFR 201.327(j)(5). The following equations define the UVA/UVB ratio and UVAI/UV ratio:
The notation dλ is the wavelength interval during spectrophotometric measurements.
RESULTS
OTC sunscreen drug product pre-irradiation with solar exposure
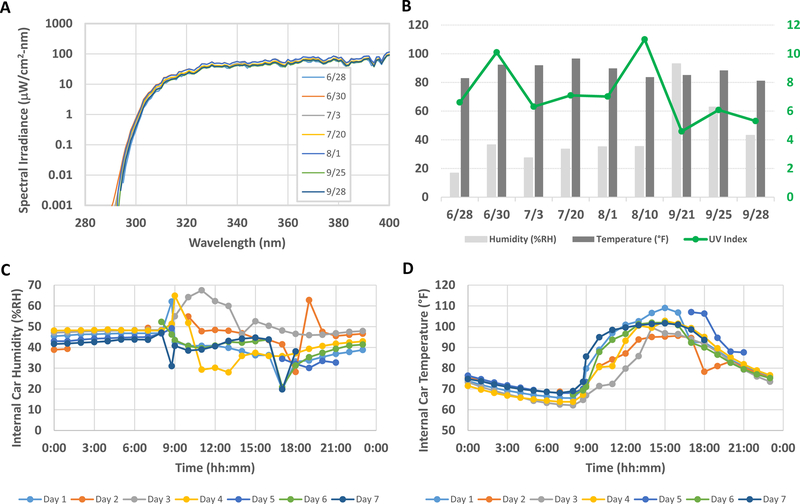
For this study, an equivalent fixed pre-irradiation dose of UVR delivered in the in vitro broad spectrum test (21 CFR 201.327(j)) of 800 J/m2-eff was accomplished using solar exposure on the FDA campus (Silver Spring, MD) during the summer months (Figure 1a and 1b, Table 1). The integrated spectral irradiances for both UVA and UVB for several of the solar exposures used to carry out the pre-irradiation of the OTC sunscreen drug products are shown in Table 1. The UVA and UVB outputs are consistent with historical solar irradiance values.24 Our solar pre-irradiation dose delivered in a time frame of 50–106 minutes depending on solar conditions meant our irradiation approach fell within the 2 hour reapplication period that is part of the directions for use on the Drug Facts Label of sunscreens.
Figure 1.
(a) Spectral irradiance from the sun versus wavelength for solar exposures between June 28 and Sept 28, 2017 in Silver Spring, Maryland (latitude 39.0 N). (b) Weather conditions including humidity (left y-axis), temperature (left y-axis), and UV Index (right y-axis) during solar exposure pre-irradiations of sunscreen samples. During the intent-to-stress arm of the study, fluctuation in humidity (c) and temperature (d) inside a motor vehicle exposed to the sun for one week was recorded by Hygrometer/Thermometer.
OTC sunscreen drug product characterization
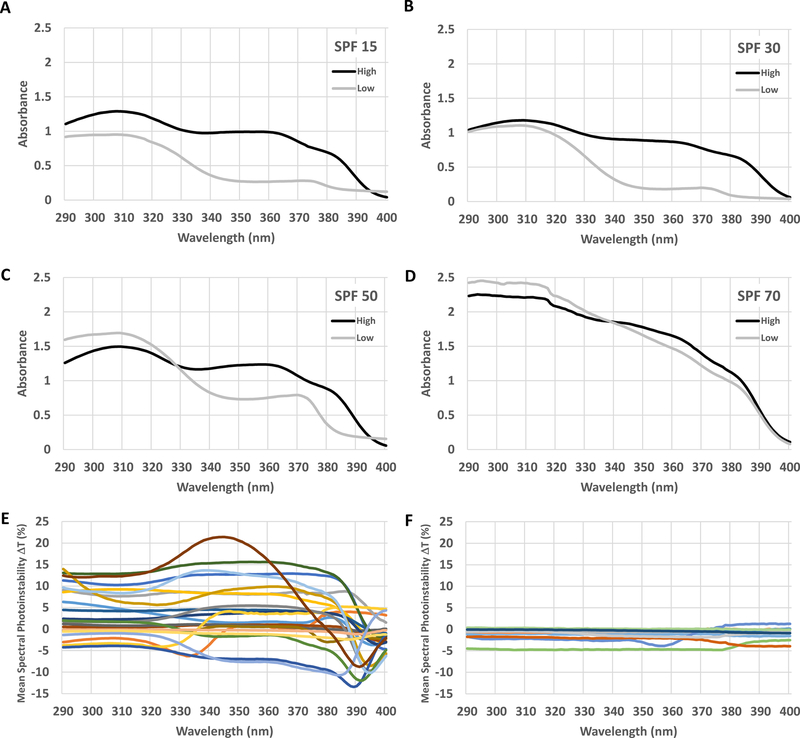
OTC sunscreen drug products are complex formulations composed of active pharmaceutical ingredients (APIs) and inactive ingredients in different dosage forms. Thirty-two U.S. commercially available OTC broad spectrum sunscreen drug products containing at least one UVA protective API of avobenzone, titanium dioxide and zinc oxide were evaluated for their performance under solar conditions. This small cross-sectional survey of the four major SPF value categories available in the commercial OTC market included 20 lotion sunscreens (SPF 15, 30, 50, 70), 4 spray sunscreens (SPF 15, 30, 50, 70), 4 sunscreen sticks (SPF 30, 50, 70), 2 sunscreen powders (SPF 30, 50), 1 sunscreen oil (SPF 50) and 1 foam/mousse sunscreen (SPF 50). Of the 32 sunscreen drug products tested, 6 showed failure to meet their labeled broad spectrum claim (mean CW ≥ 370 nm) under outdoor solar conditions (Table 2). The primary UVA protective API in these combination formulations showing failures involved 3 products containing avobenzone and another 3 products containing zinc oxide (Table 2). Ratio values that evaluate relative absorbance per unit wavelength were calculated from the area under the absorbance curve for different UV ranges (such as UVA or UVB) to further understand the extent of photodegradation and UVA protection of specific sunscreen products after solar irradiation. The UVA1(340–400 nm)/UV (290–400 nm) ratio of 0.70 equals the “high” category previously proposed by the FDA26 and is also FDA’s proposed3 delineating boundary between a product with high spectral uniformity and one with low spectral uniformity. Examples of sunscreen drug products with SPFs 15, 30, 50, and 70 tested after solar exposure highlight the UVA protection differences between products with high and low spectral uniformity (Figure 2 a, b, c, d). Additionally, the UVA/UVB ratio of at least 0.6 prior to UV exposure and a UVA/UVB ratio of at least 0.57 post-UV exposure is required to attain the United Kingdom (UK) Boots minimum 3-star rating (r2 = 0.97 with measured UVAI/UV). The UVA1/UV ratio of 0.70 also correlates with the UVA protection factor of 1/3 the SPF currently recommended in the European Union (EU) and the UK Boots 3 star rating.27,28 Spectral uniformity based on the mean sunscreen absorbance of UVA1/UV indicated that ~40% of sunscreen drug products tested (13 products) had suboptimal UVA protection (including all six products that failed to meet their broad spectrum claim) (Table 2). Seven products passed the current U.S. requirements for broad spectrum labeling, but did not demonstrate high spectral uniformity using a UVAI/UV of 0.7 (Table 2). Mean CWs generally either stayed the same or were reduced after UV irradiation; however, two sunscreen drug products showed increased CWs and, in those cases, also failed to demonstrate high spectral uniformity. Ultimately, one failed the broad spectrum test under outdoor solar conditions and the other failed the broad spectrum test after inclusion in the intent-to-stress arm of the study.
Figure 2.
Spectral profiles of OTC sunscreen drug products after solar exposure. Examples of absorbance profiles of broad spectrum SPF 15 (#SP1 and #L1) (a), 30 (#L9 and #L5) (b), 50 (#L15 and #L12) (c), 70 (#L20 and #L19) (d) sunscreen drug products exhibiting high spectral uniformity vs low spectral uniformity. Mean spectral photoinstability of organic (e) and inorganic (f) sunscreen APIs after an 800 J/m2-ery eff UV dose from the sun.
1-week temperature and humidity stress test inside a sun-exposed motor vehicle
In the case of sunscreen lotion dosage forms (which accounted for 20 sunscreen products), 7 sunscreen lotions that passed the broad spectrum test with a CW greater than 370 nm (but below 375 nm) were selected for the intent-to-stress test using the following actual use condition: fluctuating temperature and humidity inside a motor vehicle exposed to the sun for one week (Figure 1c and 1d). Only one product of the 7 sunscreen lotions failed to meet the CW of greater than 370 nm in the intent-to-stress arm of the study (Table 2). Three of the four products with low spectral uniformity maintained their low spectral uniformity profile.
OTC sunscreens in non-lotion dosage forms
In this study, several dosage forms were evaluated that are commercially available in the U.S. All of the sunscreen dosage forms tested were able to meet their labeled broad spectrum claim with CWs of 370 nm or higher after UV exposure (Table 2). Of the spray sunscreens tested, all four products exhibited CWs > 375 nm and high spectral uniformity. The two facial powder suncreens tested were the only products that tested with CWs > 380 nm and high spectral uniformity. Both of these powder sunscreens contained only combinations of titanium dioxide and zinc oxide APIs and also had the highest cost per ounce of any of the tested sunscreen products. This limited cross-section suggests that sunscreens in different dosage forms could be tested for broad spectrum claims according to the 21 CFR 201.327(j) method, but this should be interpreted with caution due to the limited number of products tested.
Change in spectral transmittance before and after pre-irradiation
Photostability that impacts sunscreen efficacy is in part addressed by the SPF test procedure where sunscreens are irradiated with UV to determine a protection factor. The broad spectrum test procedure also involves sunscreens being pre-irradiated with UV; therefore, it allows evaluation of the spectral changes of a sunscreen. Taken together, the in vivo SPF test procedure (21 CFR 201.327(i)) and the in vitro broad spectrum test procedure (21 CFR 201.327(j)) account for sunscreen drug product photostability under controlled laboratory conditions. Photodegradation is detected by lower SPF values or modified spectral absorption that may lead to a change in CW. The change in spectral transmittance before and after UV exposure to a defined erythemal dose has been described as spectral photoinstability with a mean ΔT (%) of ≥ 5% across wavelengths previously used as an arbitrary threshold value to distinguish between photoinstability and photostability of sunscreen drugs.29 Sunscreens with organic APIs exhibited more spectral photoinstability in both the UVA and UVB regions compared to sunscreen products containing only inorganic APIs (Figure 2e and 2f). However, in our dataset sunscreens which exhibited spectral photoinstability above 5% did not necessarily fail to meet the CW of 370 nm. In some cases, the opposite was true; sunscreens with CW above 370 nm and high spectral uniformity demonstrated photoinstability above 5%.
DISCUSSION
The challenge for most people has always been to incorporate several sun protection measures into an active lifestyle (e.g., limiting time in the sun, seeking shade, using sunscreen, wearing protective clothing, and sunglasses).30,31 Over the years, incremental steps have been taken to improve sunscreens through the addition of an in vitro broad spectrum test procedure and the addition of new directions including use with other sun protection measures. Despite educational efforts to mitigate preventable UV overexposure, the failure to impact sunburn or skin cancer incidence remains a serious clinical concern. While more needs to be done to encourage consumers to use appropriate sun protection measures, including sunscreen, improving the broad spectrum characteristics of marketed sunscreens may also help. With minimal effort and no modification to testing parameters, additional meaningful data can be extracted from the existing broad spectrum test as outlined in FDA’s Proposed Rule to better discriminate superior product performance. Further, our data demonstrate that it is possible for marketed U.S. sunscreens to meet the new FDA-proposed broad spectrum criteria.
The investigation into the performance of broad spectrum sunscreen drug products in actual solar conditions resulted in ~60% of tested products providing high UVA protection, but also a segment of products with suboptimal coverage across the UVA spectrum. The fact that 6 sunscreen drug products showed failure to reach a CW ≥ 370 nm after solar exposure pre-irradiation, does not necessarily imply that they would have failed to meet their labeled broad spectrum claim. The broad spectrum test procedure in 21 CFR 201.327(j) uses the controlled output of a solar simulator (rather than the solar exposure) to standardize the delivery of UV doses across laboratories during sunscreen efficacy testing. The detected sunscreen failures contained 2 to 6 APIs with no single excipient in common among them. Research evaluating APIs in combination determined that the performance of a single API was not predictive of multi-API combination performance in the formulated product.32 Given the limited products tested here, exposure under actual solar conditions and variety of API combinations in formulations, a common thread in sunscreen failures remains elusive and may not be attributable to a specific API, but rather to other aspects of formulation development.33
To examine overall UVA protection, we evaluated the amount of UVA being transmitted through broad spectrum sunscreens at an application amount of 0.75 mg/cm2 as defined by the broad spectrum test procedure in 21 CFR 201.327(j). An application amount of 0.75 mg/cm2 is just below the reported 0.79 mg/cm2 consumer application amount determined in a skin cancer prevention clinical trial and observed on a one-week sun holiday.34,35 Based on the tested sunscreens, the amount of UVA dose transmitted in two hours compared to using no sunscreen at all ranged from 30–66% of available UVA for SPF 15 and 30, and from 17–30% of available UVA for SPF 50 and 70 (Table 3). Sunscreens that exhibited a high spectral uniformity showed proportional attenuation of UVA with increasing SPF value after a pre-irradiation solar exposure with a range of 13–28% of available UVA over the course of two hours. This was not achieved with low spectral uniformity broad spectrum sunscreen drug products we tested.
Table 3:
UVA doses accumulated in 2 hours with 8 lotion broad spectrum SPF sunscreen drug products of low (UVAI/UV < 0.7) and high spectral uniformity (UVAI/UV > 0.7) at 2 different UV index conditions with sunscreen application amounts of 0.75 mg/cm2. The quantity of UVA in solar exposures for the lowest and highest spectral uniformity sunscreen drug products tested with SPFs 15 (#L1 vs #L4), 30 (#L5 vs #L9), 50 (#L12 vs #L15), and 70 (#L19 vs # L20) was calculated based on the transmission spectra with and without pre-irradiation of the sunscreen products multiplied by the solar spectrum.
| Amount of UVA in 2 hrs without pre-irradiation | Amount of UVA in 2 hrs with solar pre-irradiation | |||||||
|---|---|---|---|---|---|---|---|---|
| Spectral Uniformity | Spectral Uniformity | |||||||
| Low | High | Low | High | |||||
| % UVA received | % UVA received | % UVA received | % UVA received | |||||
| UV Index 6 | ||||||||
| 26.0 | 100% | 26.0 | 100% | 26.0 | 100% | 26.0 | 100% | |
| 15.1 | 58% | 8.6 | 33% | 14.3 | 55% | 7.3 | 28% | |
| 17.0 | 66% | 7.9 | 30% | 16.9 | 65% | 6.4 | 25% | |
| 7.8 | 30% | 7.6 | 29% | 8.0 | 31% | 5.0 | 19% | |
| 4.6 | 18% | 4.4 | 17% | 3.9 | 15% | 3.4 | 13% | |
| UV Index 10 | ||||||||
| 35.2 | 100% | 35.2 | 100% | 35.2 | 100% | 35.2 | 100% | |
| 20.3 | 58% | 11.6 | 33% | 19.3 | 55% | 9.8 | 28% | |
| 22.9 | 65% | 10.7 | 30% | 22.8 | 65% | 8.7 | 25% | |
| 10.5 | 30% | 10.2 | 29% | 10.7 | 31% | 6.7 | 19% | |
| 6.1 | 17% | 5.9 | 17% | 5.2 | 15% | 4.6 | 13% | |
Previously, it has been reported that the available annual solar UVA in the Washington DC metro area is about 19,250 J/cm2.24 Therefore, if one assumes an individual with frequent UV exposure were to receive 10% of that available UVA dose, a dose of 1,925 J/cm2 can be practically achieved during the summer in the Washington, DC area with no sunscreen (Table 3). In our example in Table 3, a broad spectrum SPF 30 with high spectral uniformity that further reduces the available UVA by 35–40% of the 1,925 J/cm2 for an individual with frequent UV exposure compared to a similarly labeled low spectral uniformity sunscreen drug product would lead to significant reduction in cumulative UVA burden over time. This reduction, over time, is especially important for consumers engaging in intentional sun exposure where increasing SPF values on the sunscreen bottle may increase their time in the sun as has been reported.36 Obviously, other factors will also affect the amount of UVA exposure for a given individual (time of year, geographic location, time of day, altitude, clouds, reflecting surfaces, individual’s UV sensitivity profile, and use of sun protection measures).
The problems of suboptimal UVA protection are not limited to the United States.37 In 2011, a small cross-sectional survey of European SPF 20 sunscreens indicated that only 7 of 15 sunscreens met voluntary EU recommendations38 for UVA protection (which correlates with tested products exhibiting a high spectral uniformity) before solar pre-irradiation and only 5 of 15 after solar pre-irradiation.39 The authors also highlighted the varying degrees of insufficient UVA protection for similarly labeled SPF 20 products based on absorbance profiles of the European sunscreens tested. Previously, another European study showed variable spectral profiles of sunscreen products after solar exposure in similarly labeled products ≤ SPF 15.40
Notwithstanding its limitation on the small number of products tested, testing across different days, and the use of solar exposure instead of UV from a solar simulator as defined in 21 CFR 201.327(j), the study described here provides valuable proof of principle on sunscreen performance in outdoor solar conditions and begins to scratch the surface of what happens to sunscreens after a 1-week stress test inside a sun-exposed motor vehicle. With the addition of the optional label claim of “broad spectrum” in the last decade, the expectation has always been that SPF and UVA protection are correlated, with higher SPF implying higher UVA protection to consumers. However, our data shows that this is not always the case. In fact, consumers may receive substantially different UVA protection levels from similarly labeled broad spectrum SPF sunscreens and may be unknowingly increasing their UVA burden depending on the sunscreen product they choose. Use of low spectral uniformity sunscreen under prolonged sun exposure may be one modifiable factor to decreasing risk of skin cancer and early skin aging from the long-term effects from UV. Therefore, any future clinical trials evaluating reduction in skin cancer risk from UV may need to consider spectral uniformity in sunscreen drug products.
Supplementary Material
SUMMARY STATEMENT.
How do commercial broad spectrum sunscreen drug products perform in actual outdoor solar conditions? Of the 32 commercial broad spectrum sunscreen drug products tested from the US market, 6 products failed broad spectrum testing and ~40% had suboptimal UVA protection when tested using solar exposure pre-irradiation (instead of UV exposure via a solar simulator). Topical photoprotection using sunscreen drug products is one of the most common methods used in skin cancer prevention; however, not all sunscreens labeled as broad spectrum provide increasing UVA protection in solar exposure conditions commensurate with the increasing labeled SPF value.
ACKNOWLEDGEMENTS:
This study was supported by the Regulatory Science & Review Enhancement Program of the Center for Drug Evaluation and Research, US Food and Drug Administration.
Role of the Funder/Sponsor: The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Footnotes
Conflict of Interest: The authors have no conflict of interest to declare
Publisher's Disclaimer: Disclaimer: The content of this publication does not necessarily reflect the views or policies of the US Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US government. This article reflects the views of the authors and should not be construed to represent FDA’s views or policies.
References
- 1.Tadokoro T, Kobayashi N, Zmudzka BZ, et al. UV-induced DNA damage and melanin content in human skin differing in racial/ethnic origin. FASEB J. 2003;17:1177–1179. [DOI] [PubMed] [Google Scholar]
- 2.U.S. Department of Health and Human Services. The Surgeon General’s Call to Action to Prevent Skin Cancer. Washington, DC: U.S. Dept of Health and Human Services, Office of the Surgeon General; 2014. [PubMed] [Google Scholar]
- 3.U.S. Food and Drug Administration. Sunscreen Drug Products for Over-The-Counter Human Use; Proposed Rule. In: Department of Health and Human Services, ed. Vol 84: Federal Register; 2019:6204–6275. [Google Scholar]
- 4.Matta MK, Zusterzeel R, Pilli NR, et al. Effect of Sunscreen Application Under Maximal Use Conditions on Plasma Concentration of Sunscreen Active Ingredients: A Randomized Clinical Trial. JAMA. May 6 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.29th Hawaii State Legislature. Environment; Water Pollution; Sunscreen; Oxybenzone; Octinoxate; Sale; Distribution; Prohibition. SB2571 SD2 HD2 CD1 2018; 1–6. Available at: https://www.capitol.hawaii.gov/Archives/measure_indiv_Archives.aspx?billtype=SB&billnumber=2571&year=2018. Accessed June 6, 2019. [Google Scholar]
- 6.City of Key West Florida. Requesting the Key West Sustainability Board to report to the City Commission on the efficacy of sunscreens that do not contain Oxybenzone and Octinoxate, and on other factors affecting the Coral Reef surrounding Key West. 19–3516 2019; https://keywest.legistar.com/LegislationDetail.aspx?ID=3847999&GUID=61C56230-C9F6-40AA-9AE4-9998D80064AF&Options=ID|Text|&Search=oxybenzone. Accessed June 6, 2019.
- 7.U.S. Food and Drug Administration. FDA advances new proposed regulation to make sure that sunscreens are safe and effective. 2019; https://www.fda.gov/news-events/press-announcements/fda-advances-new-proposed-regulation-make-sure-sunscreens-are-safe-and-effective. Accessed June 6, 2010.
- 8.National Toxicology Program. Report on Carcinogens Background Document for Ultraviolet (UV) Radiation, Broad Spectrum and UVA, UVB, and UVC. 2000:120. [PubMed]
- 9.International Agency for Research on Cancer. Solar and ultraviolet radiation. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol 100D: IARC Monogr Eval Carcinog Rish Chem Hum; 2012:68. [Google Scholar]
- 10.Qureshi AA, Laden F, Colditz GA, Hunter DJ. Geographic variation and risk of skin cancer in US women. Differences between melanoma, squamous cell carcinoma, and basal cell carcinoma. Arch Intern Med. 2008;168:501–507. [DOI] [PubMed] [Google Scholar]
- 11.Xiang F, Lucas R, Hales S, Neale R. Incidence of nonmelanoma skin cancer in relation to ambient UV radiation in white populations, 1978–2012: empirical relationships. JAMA Dermatol. 2014;150:1063–1071. [DOI] [PubMed] [Google Scholar]
- 12.Fears TR, Scotto J, Schneiderman MA. Mathematical models of age and ultraviolet effects on the incidence of skin cancer among whites in the United States. Am J Epidemiol. 1977;105:420–427. [DOI] [PubMed] [Google Scholar]
- 13.Dennis LK, Vanbeek MJ, Beane Freeman LE, Smith BJ, Dawson DV, Coughlin JA. Sunburns and risk of cutaneous melanoma: does age matter? A comprehensive meta-analysis. Ann Epidemiol. 2008;18:614–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chang YM, Barrett JH, Bishop DT, et al. Sun exposure and melanoma risk at different latitudes: a pooled analysis of 5700 cases and 7216 controls. Int J Epidemiol. 2009;38:814–830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure. Eur J Cancer. 2005;41:45–60. [DOI] [PubMed] [Google Scholar]
- 16.Brash DE. UV-induced Melanin Chemiexcitation: A New Mode of Melanoma Pathogenesis. Toxicol Pathol. 2016;44:552–554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hodis E, Watson IR, Kryukov GV, et al. A Landscape of Driver Mutations in Melanoma. Cell. 2012;150:251–263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Krauthammer M, Kong Y, Ha BH, et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nature Genetics. 2012;44:1006–1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Noonan FP, Zaidi MR, Wolnicka-Glubisz A, et al. Melanoma induction by ultraviolet A but not ultraviolet B radiation requires melanin pigment. Nat Commun. 2012;3:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Premi S, Wallisch S, Mano CM, et al. Photochemistry. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science. 2015;347:842–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tewari A, Grage MM, Harrison GI, Sarkany R, Young AR. UVA1 is skin deep: molecular and clinical implications. Photochem Photobiol Sci. 2013;12:95–103. [DOI] [PubMed] [Google Scholar]
- 22.Tewari A, Sarkany RP, Young AR. UVA1 induces cyclobutane pyrimidine dimers but not 6–4 photoproducts in human skin in vivo. J Invest Dermatol. 2012;132:394–400. [DOI] [PubMed] [Google Scholar]
- 23.Damian DL, Matthews YJ, Phan TA, Halliday GM. An action spectrum for ultraviolet radiation-induced immunosuppression in humans. Br J Dermatol. 2011;164:657–659. [DOI] [PubMed] [Google Scholar]
- 24.Miller SA, Hamilton SL, Wester UG, Cyr WH. An analysis of UVA emissions from sunlamps and the potential importance for melanoma. Photochem Photobiol. 1998;68:63–70. [PubMed] [Google Scholar]
- 25.Coelho SG, Hearing VJ. UVA tanning is involved in the increased incidence of skin cancers in fair-skinned young women. Pigment Cell Melanoma Res. 2010;23:57–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.U.S. Food and Drug Administration. Sunscreen Drug Products for Over-the-Counter Human Use; Proposed Amendment of Final Monograph. In: Department of Health and Human Services, ed. Vol 72: Federal Register; 2007:49070–49122. [Google Scholar]
- 27.Diffey B Spectral uniformity: a new index of broad spectrum (UVA) protection. Int J Cosmet Sci. 2009;31:63–68. [DOI] [PubMed] [Google Scholar]
- 28.Wang SQ, Stanfield JW, Osterwalder U. In vitro assessments of UVA protection by popular sunscreens available in the United States. J Am Acad Dermatol. 2008;59:934–942. [DOI] [PubMed] [Google Scholar]
- 29.Maier H, Schauberger G, Martincigh BS, Brunnhofer K, Honigsmann H. Ultraviolet protective performance of photoprotective lipsticks: change of spectral transmittance because of ultraviolet exposure. Photodermatol Photoimmunol Photomed. 2005;21:84–92. [DOI] [PubMed] [Google Scholar]
- 30.Guy GP Jr., Holman DM, Watson M. The Important Role of Schools in the Prevention of Skin Cancer. JAMA Dermatol. 2016;152:1083–1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Holman DM, Ding H, Guy GP Jr., Watson M, Hartman AM, Perna FM. Prevalence of Sun Protection Use and Sunburn and Association of Demographic and Behaviorial Characteristics With Sunburn Among US Adults. JAMA Dermatol. 2018;154:561–568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Maier H, Schauberger G, Brunnhofer K, Honigsmann H. Change of ultraviolet absorbance of sunscreens by exposure to solar-simulated radiation. J Invest Dermatol. 2001;117:256–262. [DOI] [PubMed] [Google Scholar]
- 33.Klein K In: Lowe NJ, Shaath NA, Pathak MA, ed. Sunscreens Development, Evaluation and Regulatory Aspects. Vol 15 2nd ed. New York: Marcel Dekker, Inc.; 1997:285–311. [Google Scholar]
- 34.Neale R, Williams G, Green A. Application patterns among participants randomized to daily sunscreen use in a skin cancer prevention trial. Arch Dermatol. October 2002;138:1319–1325. [DOI] [PubMed] [Google Scholar]
- 35.Petersen B, Datta P, Philipsen PA, Wulf HC. Sunscreen use and failures--on site observations on a sun-holiday. Photochem Photobiol Sci. 2013;12:190–196. [DOI] [PubMed] [Google Scholar]
- 36.Autier P, Boniol M, Dore JF. Sunscreen use and increased duration of intentional sun exposure: still a burning issue. Int J Cancer. 2007;121:1–5. [DOI] [PubMed] [Google Scholar]
- 37.Diffey B New Sunscreens and the Precautionary Principle. JAMA Dermatol. 2016;152:511–512. [DOI] [PubMed] [Google Scholar]
- 38.European Commission. COMMISSION RECOMMENDATION of 22 September 2006 on the efficacy of sunscreen products and the claims made relating thereto (2006/647/EC). Official Journal of the European Union. 2006;265:39–43. [Google Scholar]
- 39.Hojerova J, Medovcikova A, Mikula M. Photoprotective efficacy and photostability of fifteen sunscreen products having the same label SPF subjected to natural sunlight. Int J Pharm. 2011;408:27–38. [DOI] [PubMed] [Google Scholar]
- 40.Gonzalez H, Tarras-Wahlberg N, Stromdahl B, et al. Photostability of commercial sunscreens upon sun exposure and irradiation by ultraviolet lamps. BMC Dermatol. 2007;7:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.