Abstract
Purpose of Review
The coronavirus disease-2019 (COVID-19) is a global pandemic which has not been seen in recent history, leaving behind deep socioeconomic damages and huge human losses with the disturbance in the healthcare sector. Despite the tremendous international effort and the launch of various clinical trials for the containment of this pandemic, no effective therapy has been proven yet.
Recent Findings
This review has highlighted the different traditional therapeutic techniques, along with the potential contribution of nanomedicine against the severe acute respiratory syndrome corovirus-2 (SARS-CoV-2). Repositioning of the drugs, such as remdesivir and chloroquine, is a rapid process for the reach of safe therapeutics, and the related clinical trials have determined effects against COVID-19. Various protein-based SARS-CoV-2 vaccine candidates have successfully entered clinical phases, determining positive results. The self-assembled and metallic nanovaccines mostly based on the antigenic properties of spike (S) protein are also approachable, feasible, and promising techniques for lowering the viral burden.
Summary
There are number of NP-based diagnostic systems have been reported for coronaviruses (CoVs) and specifically for SARS-CoV-2. However, extensive studies are still necessary and required for the nanoparticle (NP)-based therapy.
Keywords: COVID-19, SARS-CoV-2, Nanomedicine, Nanovaccines, Nano-based diagnosis
Introduction
The outbreak of coronavirus disease-2019 (COVID-19) began in Wuhan, China. The corona virus global crisis is posing unprecedented difficulties to humans [1]. This new social landscape requires collective consideration, where our actions are interrelated, interdependent, to trans-boundaries and cultural diversity [2, 3]. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) was recognized as the causative agent of this revolution and grown by smooth tissues such as the muzzle, mouth, and nose [4, 5]. The SARS-CoV-2 is a single-stranded ribonucleic acid (ssRNA) virus having a short genome of 26–32 kb with a diameter of 65–125 nm, and earlier known to be a 2019-novel coronavirus (2019-nCoV) [6, 7]. The infection expanded quickly everywhere in China within a month, and according to the World Health Organization (WHO), it is recently expanded to over 212 countries, regions, and provinces worldwide [8, 9]. Based on clinical evidence, there are two major requirements to control the COVID-19 pandemic: first to overcome infection rates and second to reduce mortality [10]. Recently, no approved drugs or vaccines are prepared for the treatment or prevention of COVID-19 [11]. Still, the ongoing vaccines are in the second and third phases of clinical trials. As the accessible therapies, including nonspecific antivirals, antibiotics, to treat secondary bacterial diseases, sepsis, and corticosteroids to decrease inflammation fail to treat the severe illness with COVID-19 [12, 13]. This appears as inflammatory lesions with ground-glass ambiguity on computed tomography (CT) scans. The current accessible drugs have been able to moderately handle indications. Therefore, the appropriate and effective medical treatment for the current COVID-19 infection is an immediate demand for mankind [10]. Early detection of the infection is essential for defence among all states including symptomatic and asymptomatic populace [3, 11, 14]. Numerous forthcoming drugs that are thought to be unswervingly appropriate, as well as new therapeutic drugs are being produced, including viral entry inhibitors, replication inhibitors, protease inhibitors, heterocyclic antivirals, nanodrug delivery systems, and biological therapeutic drugs [15].
In the name of health as our primary necessity, the common interest should be addressed in the future, keeping in mind the “One Health” concept, relying on the belief that the well-being of humans is stringent with animals’ connections and environment [16]. To tackle such a complex challenge, the collaboration between diverse researchers with complementary expertise is needed. The current challenge should be taken as an incredible opportunity to remind us of our globalized world, as shown in other scientific contexts, a multi-interdisciplinary methodology involving transversal disciplines of knowledge between countries [17]. Promoting exchanges and increasing diversity in teams will all be necessary to obtain new and important scientific solutions [18]. From this point of view, nanotechnology is inherently an area in which scientists with incredibly diverse backgrounds have converted into valuable cooperations for multifaceted difficulties. Now, nanotechnology is required to lay new foundations towards combating the recent global public health threat and rethinking a more sustainable future based on science [19]. Nanoparticles (NPs) are small, but have a large surface-to-volume ratio, giving them unprecedented, unique features. Because of these characteristics, nanoparticles have been used in the fields of biotechnology, medicine, drug delivery, sensors, and DNA labelling and are treated as a bridge between bulk materials [20]. The nano-strategies have been widely applied to enhance antiviral drug delivery and efficiency, individually in combination with nucleoside analogues which have potential applications against drug-resistant human immunodeficiency virus disease also [15, 21]. There are several available nano delivery systems can be used with newly developed drug formulations to efficiently deliver drugs with rapid therapeutic indices for COVID-19. However, this work is in growth phase, and no nanotechnology is recently available for the treatment of COVID-19 so far. Even nanotechnology plays a pivotal role in the analysis of COVID-19 [22]. In this review, authors explain the possible role of nanotechnology in the diagnosis, investigation, vaccine formulation, and reasonable treatment of COVID-19.
Nanoparticle-Based Diagnosis of Coronavirus
The diagnosis of coronaviruses (CoVs) including SARS-CoV-2 is primarily based on computing tomography scans and nucleic acid testing that utilize reverse transcription polymerase chain reaction (RT-PCR) technique [23, 24]. The rapid analysis and recognition of infected patients are the most ideal approaches to contain scourges and typical-to-traditional diagnostic methods because of inaccessibility of facilities, instruments, reagents, low yield, and technical speciality. Because of urgent need for fast, sensitive, and economic testing of COVID-19, nanotechnology can play a crucial role in the diagnosis of SARS-CoV-2 using nanoparticle-based (NP-based) electromagnetic properties (Table 1) [35]. The test procedures require robotized extraction of nucleic acids from samples to prevent cross-contamination or false-negative outcomes. Magnetic nanoparticles (MNPs) are also employed in separation of nucleic acids. For instance, Zhao et al. built up a one-step nucleic acid isolation procedure utilizing amino-adjusted MNPs along with polycarboxyl groups (PC-coated NH2-MNPs) to tie viral RNA [25]. Under influence of magnetic fields, nucleic acid combines easily and delivered from MNPs by the expansion of buffer. Utilizing COVID-19 pseudoviruses, polycarboxyl MNPs (10 ± 3nm, − 39 ± 1 mV) indicated superior retention and paramagnetic properties at a magnetic capture of 30 s. Superparamagnetic NPs were set up to give a magnetite core secured with silica of 80 nm combined with probe that is corresponding to the target cDNA (complementary DNA) of SARS-CoVs. The functionalized superparamagnetic NPs had the option to strengthen and separate target cDNA from samples utilizing magnetic field [26]. This separated DNA was intensified with polymerase chain reaction (PCR) so that it could be effectively detected through sandwich hybridization assessment through silica-coated fluorescence NPs (40 ± 5 nm) bringing about fluorescence intensity, which is legitimately identified with the target cDNA concentration.
Table 1.
Nanoparticle-based diagnosis for pathogenic coronaviruses
| Nanoparticles | Virus | Platform and ligand | Target | Description | Ref. |
|---|---|---|---|---|---|
| Magnetic nanoparticles (MNPs) | SARS-CoV-2 | Polycarboxyl-functionalized magnetic nanoparticles in polycarboxyl groups | Viral RNA | One-step, simple, sensitive, excellent paramagnetic characteristic, high pureness and fertility, no toxic reagents | [25] |
| SARS-CoV | Superparamagnetic nanoparticles in probe (complementary to cDNA) | Targeted cDNA | Quick process, high specificity, and sensitivity | [26] | |
| Gold nanoparticles (AuNPs) | MERS-CoV | AuNP-based colorimetric assay in thiolated ssDNA probe | Upstream of envelope protein gene and ORF-1A | Visible detection, cheap, quick (within 10 min), limit of detection is 1 pmol/μl | [27] |
| AuNP-modified carbon electrodes in thiolated ssDNA probe | Targeted DNA | Quick, sensitive, simplistic | [28] | ||
| Array of AuNP-modified carbon electrodes in MERS-CoV protein | Antibodies | Extremely selective, individual-step, genuine and sensitive | [29] | ||
| Streptavidin-AuNP conjugates in streptavidin | Fluoresce in isothiocyanate and biotin)-labelled RNA of MERS-CoV (nucleocapsid gene) | Upward flow detection, simple to operate, low-priced, quick | [30] | ||
| SARS-CoV-2 | SARS-CoV-2 antigen-AuNP conjugates (immunoassay strip) in SARS-CoV-2 antigens | IgG/IgM against SARS-CoV-2 | Chromatographic immunoassay is membrane-based, quick, affordable | [31] | |
| Antigens- SARS-CoV-2 conjugates (immunoassay strip) | IgG/IgM for SARS-CoV-2 | Oblique flow detection, sturdy readout signal | [32] | ||
| Silica-coated fluorescence nanoparticle in probe (complementary to cDNA) | Target cDNA | Quick process, high specificity and sensibility | [33] | ||
| AI-A, AV, CoV | Self-assembled star-shaped chiroplasmonic AuNP-quantum dot in virus-specific antibodies | Target virus | Chiro-immunosensor with exciton-plasmon interaction in chiral AuNP ultrasensitive | [34] |
SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SARS-CoV, severe acute respiratory syndrome coronavirus; MERS-CoV, Middle East respiratory syndrome coronavirus; AI-A, avian influenza-A; AV, adenovirus; CoV, coronavirus; cDNA, complementary DNA; AuNP, gold nanoparticle; ORF, open reading frame
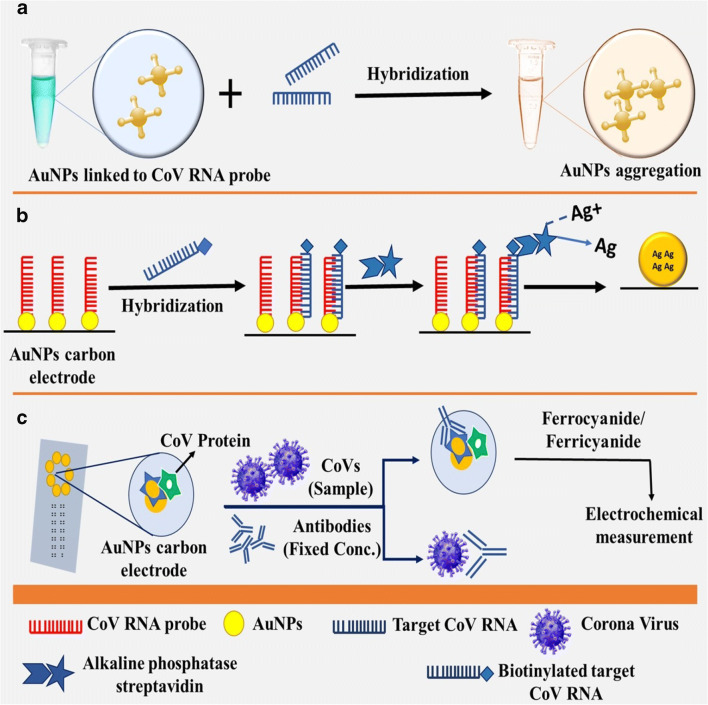
Gold nanoparticles (AuNPs) were investigated for developing nano assays for two reasons: (i) simplicity of electrostatic surface-design with different moieties, for example, antigens and antibodies; and (ii) SPR transition and change in colour [35, 36]. The AuNPs are most commonly utilized in colorimetric hybridization tests such as disulfide bond-based colorimetric assay designed by Kim et al. [27]. Utilizing thiolated single-stranded deoxyribonucleic acid (ssDNA) probes for targeting sites of Middle East respiratory syndrome coronavirus (MERS-CoV) genome for envelope protein (E) gene and open reading frame-1a (ORF-1a), framing a long self-assembled hybrid. This structure shields citrate ion-headed AuNPs from salt-actuated assembly. However, when target genes are absent, this protection is nonexistent, bringing about accumulation of AuNPs [27]. AuNP-based elementary hybridization assay was accounted for utilizing a DNA probe, which comprises thiolated-DNA comb immobilized on AuNP carbon terminal to hybridize biotinylated target DNA of SARS-CoV (Fig. 1) [28]. As a consequence of this, basic phosphatase streptavidin was formed through streptavidin-biotin association, catalyzing silver (Ag) ions and statement displacement of metallic Ag on anode surface. Metallic Ag can be estimated and it is in proportion to the concentration of alkaline phosphatase that target viral DNA [28].
Fig. 1.
Nanoparticle-based assays for coronaviruses. a Colorimetric hybridization assays, b Electrochemical hybridization assays, c elctrochemical immunosensor assays
Chiral gold nanohybrids (CAuNPs) with quantum dots (QDs) have been recently utilized to build star-shaped chiroplasmonic AuNPs that can detect distinctive viral infections including CoVs [34]. In this technique, each of CAuNPs and QDs were conjugated to two distinctive target virus-targeted antibodies. When target virus is present, a nano-sandwich configuration was produced, bringing about predominant plasmonic resonant coupling with the beta particle of QD [34]. In addition, AuNPs can be intended to identify CoV-specific antibodies utilizing electromagnetic immunosensor assay consisting of a chip and a C-terminal that contains a variety of AuNPs to identify CoV-explicit antibodies (Fig. 1) [29]. Over the surface of AuNP-anodes, MERS-CoV protein was immobilized in the presence of antibodies with specific concentration; emulation took place between the free viruses in the prototype with immobilized MERS-CoV protein. A ferrocyanide probe was utilized for electrochromic estimation. This chip was utilized for multiplexed recognition simultaneously through numerous terminals on a similar chip, with every electrode connected to various viral antigens [29]. New indicative tests for SARS-CoV-2, for example, NP-based flow detection strips, were too augmented to accelerate detection and also eliminate the prerequisite to send samples at laboratories. Vertical stream (VF) detection was done to visually recognize nucleoprotein (N) gene of MERS-CoV in association with reverse transcription loop-interceded isothermal amplification method (RT-LAMP-VF) [30]. RNA of MERS-CoV was enhanced by RT-LAMP; the amplicons were labelled with fluorescein isothiocyanate (FITC) and biotin to bind streptavidin-AuNP conjugates. This FITC-labelled complex was procured by anti-FITC antitoxin immobilized on strip, delivering a hued line in 10 min [30]. The COVID-19 outbreak laid a global pressure on healthcare system, and there is urgency for the detection of positive coronavirus cases and carry out viral surveillance. Diagnostic survey may assume a significant part in COVID-19 identification, permitting quick implementation of preventive measures that confine the transmission by identifying and isolating positive cases. Opacities on CT scans of COVID-19-infected lungs are different from that of patients with healthy lungs: the anatomy of the lung is denser and more profused [37–40]. Presently, COVID-19 is analyzed by means of reverse transcriptase (RT)-PCR and visualized through CT; however, every procedure has some limitations. Techniques involving molecular reactions can analyze coronaviruses more precisely than CT scans, and they have the ability to locate and classify various microorganisms [22, 37]. Nanotechnology brings new technics for delivering economical and versatile detection techniques with effective prescriptions. Nano sensors are presently a reality and can identify low bacterial and viral concentrations. Subsequently, healthcare professionals are helping to identify symptoms. As indicated by the joint WHO taskforce and China, since December 2019 to mid-February 2020, 104 SARS-CoV-2 viral strains were separated and sequenced with Illumina and Oxford nanopore sequencing method [38]. Illumina sequencing is a grouping that involves synthetic procedure and solid-phase bridge intensifications, though nanopore sequencing includes movement of DNA through a protein pore and assessing changes in voltage to identify the sequence. Research and innovation are the only tools to fight against COVID-19. The equipment’s used in nanotechnology can be reframed for the analysis of disease [22, 39]. Distinctive nanotechnologies have advantages over molecular methods for diagnosing COVID-19, and the current strategies to combat the recent pandemic are addressed in detail.
Reverse Transcriptase Loop-Mediated Isothermal Amplification
Coronavirus is one of the deadliest respiratory infections which is quickly propagating overseas and has caused global concern. Presently, it is analyzed by SARS-CoV-2 nucleic acid through reverse transcriptase loop-mediated isothermal amplification (RT-LAMP) combined with a nanoparticle-based biosensor (NBS) [41]. Other approaches are time-tedious and complex and require skilled specialists. Therefore, Yang et al. built up a RT-LAMP test to recognize triple genes for rapid diagnosis of SARS-CoV-2 [37]. In 2003, this technique was utilized to test for the SARS coronavirus and found to be simple and quick [42]. RT-LAMP, NBS-LAMP amplification method, and reverse transcription with nanoparticle-based biosensors enable diagnosis of COVID-19 in a single step. This technique requires 30 min for the amplification of nucleic acid at a consistent 60–65 °C utilizing various primers to distinguish the N, E, and ORF1ab gene sequence. Around ten sets of such primers have been configured per gene. The specificities of the COVID-19 RT-LAMP-NBS test was investigated by analysis of genomic sequence obtained from different microbes, including bacteria, fungi, and virus [22, 43].
Point-of-Care Testing
Purpose-of-care test procedure is followed when the patient samples are not sent to research facilities; this method does not require lab network to recognize patients who are infected. Lateral-flow antigen detection for SARS-CoV-2 is a consideration point being used for diagnosing COVID-19 [44]. Business lateral flow assays consist of a paper-like film strip covered with two lines: one consists of gold nanoparticle-antibody conjugate and another is used for antibody detection. Patient sample like blood, sputum, and urine are placed on the film; capillary movement attracts the protein through the line. At the point when the first line moves, antigens bind to the nanoparticle-antibody conjugate, after binding the complex streams across the film. The captured antibodies immobilize the complex after entering second side where the blue or red side is evident. Singular gold nanoparticles are red, while the coupled plasmon groups trigger the solvent enclosing the bundled gold nanoparticles to appear blue [23]. In research centres, numerous stages, for example, electrochemical sensors, paper-based frameworks, and surface-upgraded Raman spectrum-based systems are also established [22].
Optical Biosensor Nanotechnology
Optical biosensor nanotechnology is a new field of technology that enables the detection of the COVID-19 in only 30 min instantly from patients’ samples. This new innovation could undoubtedly decide if a patient is infected with the COVID-19 or flu infection [22]. This method will conceivably be utilized for procurement of patients and dealing with current pandemic situation. This novel biosensor device is also utilized for the analysis of different types of coronavirus diseases existing in animal body, for example, bats, cats, dogs, and sea animals, to identify and trace the possible evolutionary mechanism of these viruses for controlling such kinds of future outbreaks [22, 45].
Nanopore Target Sequencing
The nanopore target sequencing (NTS) technology detects SARS-CoV-2 with ten additional respiratory infections just in 6–10 h. This method is required contemporary for the diagnosis of COVID-19. Furthermore, the blueprint can also be extended to analyze different viruses and microorganisms. The NTS depends on intensification of eleven SARS-CoV-2 virulent virus and distinctive genomic sequence (e.g. ORF1ab) utilizing an essential band continued by sequencing of amplified section on a nanopore membrane [46]. This technique is based on a nanopore platform that sequences fragments of nucleic acid and simultaneously investigates the information near-real-time affirming SARS-CoV-2 diseases in few minutes by mapping the sequence read of SARS-CoV-2 genome for the analysis of identity and validity of outputs. The virulence locale (genome 21,563–29,674 bp; NC 045512.2) encoding spike (S, 1273 amino acid; AA), ORF3a (open reading frame 3a, 275 AA), envelope (E, 75 AA), membrane (M, 222 AA), ORF6 (66 AA), ORF7a (121 AA), ORF8 (121 AA), nucleocapsid (N, 419 AA), and ORF10 (38 AA) are helpful for the identification of SARS-CoV-2-virulent genomic sequence. The NTS is performed on one MinION sequencer chip (Oxford nanopore technique) to detect all test samples, the sequence info is determined through bioinformatics channelling at regular interval of timeframes. To expand plasmid concentration, all high-element reads were resolute by output reads on SARS-CoV-2 genome. As a norm for qPCR, NTS cannot decide that sample is positive for contagion via testing at one or two different domains; discoveries and information from all possible regions must be taken into consideration for further investigation [22, 47, 48].
Mechanism of Action of Antiviral Nanomaterials
Preclinical investigations have indicated virus nanoparticle reactions induce antiviral activity. Nanomaterials that work indirectly do not restrain virus alone; rather, they enhance the pharmacological action of antiviral medicines, and utilized for stability and better bioavailability [49–51]. Additionally, nanomaterials can likewise initiate an immune reaction for developing short-term immunity [52]. Interestingly, nanomaterials with direct activity act as dynamic compound, since they resist the viral infection alone, typically by changing the hereditary information of viral DNA. In 2005, Lara et al. demonstrated that receptors of HIV interact with AgNPs, restraining its infectivity [53]. They proposed that AgNPs bind to gp120 glycoproteins (GP) that prevent the virus to bind with cells, as noticed in the in vivo outcomes. Morris et al. has shown that AgNPs attach to surface GP of the virus preventing the association phenomenon by dropping its capability to attract cells [54]. Also, Cagno et al. proved that gold and iron oxide NP-coated ligands dislocate the configuration of numerous viruses [55], flouting the viral molecule that leads to denaturation of virus. Kim et al. exhibited that viral genes are suppressed by nanomaterials, the in vivo administration of anti-CCR5 siRNA/LFA-1 I-ts NPs silenced genes in leukocytes for approximately 10 days, inhibiting human immunodeficiency virus (HIV) disease [56]. Cojocaru et al. depicted the possible mode of action that permits nanomaterials to halt both DNA and RNA virus [57]. Among all the existing fundamental methods, inhibition of viral DNA/RNA is the major mechanism [58]. Nanomaterials show different characteristics of clinical intrigue, for example, upgraded chemical interactions, bio-compatibility, and controlled and targeted drug release, and were found safer when contrasted with the non-nanomaterials [59, 60].
Role of Nanotechnology in COVID-19 Treatment
COVID-19 outbreak requires impactful healthcare approaches for combating lethal lung infection. Until now, no particular medication is affirmed for the treatment of COVID-19 [61]. According to clinicians and therapeutic systems, a few immunomodulatory and antiviral drugs have been recommended and a number of drug verities are presently in phases of clinical trials. They include remdesivir, baricitinib, oseltamivir, EIDD-2801, favipiravir, darunavir/cobicistat, mRNA-1273, mycophenolate, tocilizumab, methylprednisolone, interferon, teicoplanin, ribavirin, hydroxychloroquine, and azithromycin [62]. On 28 March 2020, the Food and Drug Administration (FDA) permitted the use of hydroxychloroquine sulphate and chloroquine phosphate as an emergency prescription to fight against COVID-19 [63, 64]. As indicated by the most recent studies, regulations in Belgium, which additionally involved recommendations from Netherlands, Switzerland, France, and Italy, put forward the use of remdesivir, ritonavir/lopinavir, chloroquine or hydroxychloroquine, and tocilizumab for additional support; they also included use of O2 ventilator as a life support system and to save lungs from failure [65]. Favipiravir another antiviral medicine endorsed in China and Japan for curing flu is currently under audit for COVID-19 [66]. In addition to these prescriptions, Chinese drugs assume significant function to deal with coronavirus. The Chinese Academy of Sciences in collaboration with Shanghai Institute of Materia Medica recommended that SARS-CoV-2 can be treated utilizing an oral dose of Shuanghuanglian [67, 68]. The chemical composition of this fluid consists of baicalin, chlorogenic acid, and forsythin also demonstrated antiviral and bactericidal effects [69]. At present day 20 September 2020, none of the vaccine is approved for COVID-19; researches are ongoing in almost every country and are under pipeline, some are in phases I, II, and III. A vaccine developed by Oxford when tested on participant during clinical trial indicated serious neurological symptoms. Nanotechnology could have high chances for curing COVID-19 [69]. These can be utilized to decrease the severity and lethality of the disease (Table 2). Experimentations are under process and nano-based drugs will ideally be accessible soon.
Table 2.
List of various types of nanoparticles used for the treatment of various viral infections
| Target virus | Nanocomposites | Inhibition action | Inference or findings | Ref. |
|---|---|---|---|---|
| Poliovirus type-1 | Silver nanoparticles (AgNPs) | – | AgNP-based merchandises were absolute for biomedical utilization and to fight viral contagions | [70] |
| Human immunodeficiency virus-1 (HIV-1) | Silver nanoparticles | The basic relationship between thiol residues and Ag | Ag nanoparticles showed antiviral activity against HIV-1 bound to carbon coated, PVP coated, and bovine serum albumin. The interactions of Ag nanoparticles and viruses were size-dependent and involved thiol groups of glycoprotein 120. | [71] |
| Human immunodeficiency virus (HIV) | Amantadine (Ada), on the outermost layer of PVP-PEG-coated silver nanorods (Ada-PVP-PEG silver nanorods) | Amantadine surface-modified silver nanorods enhance HIV vaccine-triggered cytotoxic lymphocyte to generate approximately eight times more potent tumor necrosis factor-alpha in vivo. | Surface alterations of nanomaterials in essentially enhancing immunotherapy of the HIV vaccine towards HIV-infected cells | [72] |
| Polyvinylpyrrolidone (PVP)/stearic acid (SA)-polyethylene glycol (PEG) NP | Meritorious cellular internalization | Convenient loading, sustained deliverance, hemocompatibility and nontoxicity | [73] | |
| Lactoferrin NPs | – | Triple drug-loaded nanoparticles have numerous advantages towards solvable (free) drug mixtures that enhance bioavailability, enhance PK profile, and decrease drug-related toxicity. | [74] | |
| Amide functionalized alginate NPs | Represses viral transcriptase | Efficacious antiviral delivery | [75] | |
| Enterovirus 71 (EV71) | siRNA-modified Polyethylenimine (PEI) encapsulated AgNPs |
Prevent fragmentation of DNA, chromatin Condensation and activation of caspase-3 and obstacle approach of EV71 in the host cell |
Surface-decorated AgNPs with PEI and siRNA may be a potential silver species with antiviral characteristics for the treatment of viral infections. | [76] |
| Porcine epidemic diarrhea virus (PEDV) as a model of coronavirus | Glutathione-capped Ag2S nanoclusters (NCs) | Represses the synthesis of viral negative-strand RNA and viral budding | The results recommend the chance to grow efficient anti-SARS or anti-MARS reagents based on Ag2S NCs | [77] |
| Respiratory syncytial virus (RSV) | AgNP | NPs bind to viral glycoproteins and inhibit entry into the host cell. | AgNP-mediated decline in RSV replication, both in epithelial cell lines and in experimentally contaminated BALB/c mice. | [54] |
| Rabies virus | Nonlinear globular G2 dendrimer | – | Adjuvanticity effectiveness | [78] |
| Hepatitis C virus (HCV) | Ammonium-terminated amphiphilic Janus dendrimers | Repress HCV replication by combining with viral proteins | Nontoxic drug-loaded nano groups repress HCV replication at lowering camptothecin concentration. | [79] |
| Venezuelan equine encephalitis virus (VEEV) replicon RNAs Zika virus | Modified dendrimer NPs |
Activation of both CD8+ T-cell. Viral E protein-specific IgG responses |
This strategy can be utilized to evaluate fresh applicant antigens and recognize immune correlates without the usage of a live virus. | [80] |
| Pseudo typed viral particles (Ebola virus glycoprotein) | Glycodendrofullerenes Carbohydrate moieties (Mannose, Galactose) |
Dendritic cell-specific blocking Intracellular adhesion molecule 3 Non-integrated receptor grabbing |
Prohibition of viral attachment. Interrupted leading to the viral approach | [81] |
| H1N1, norovirus | Gold (Au)/iron oxide magnetic NP-decorated carbon nanotubes (CNTs) | DNA hybridization | Large sensitivity and selectivity apprehension of viral DNA | [82] |
| Herpes simplex virus | Monodispersed gold nanoparticles | Obviated viral accessory and entry into Vero cells | Gold nanoparticles obtained recommended as a safer alternative in virus chemotherapy | [83] |
| Porcine reproductive and respiratory syndrome virus (PRRSV) | Glycyrrhizic-acid-based carbon dots | Gly-CDs can repress PRRSV invasion and replication, stimulate antiviral innate immune responses, and prevent the accumulation of intracellular reactive oxygen species (ROS) due to PRRSV contamination. | Gly-CD has exceptional antiviral action with a multisite inhibitory mechanism, giving an encouraging applicant | [84] |
| Porcine epidemic diarrhea virus and pseudorabies virus | Gt, GtO, GO, rGO, GO-PDDA, GO-PVP | Electrostatic interaction and hydrophobic interaction with the sharp edged structure |
The antiviral action accompanied the sequence: GO ≈ rGO > GO-PVP > GtO. Gt and GO-PDDA did not exhibit antiviral action. GO represses viral infection by inactivating the virus before inserting the cell. The envelope and routine spikes were damaged upon virus incubation with GO for both viruses. |
[85] |
| Porcine epidemic diarrhea virus | Ag2 S NC |
Cells were contaminated with the virus and 12 hpi the plaque composition unit (PFU) was reduced from 3.8 × 105 to 2.5 × 102 PFU∙mL−1 upon treatment with Ag2 S NC. This result was associated with the expression of the PEDV N protein being downregulated in a concentration-dependent performance. Ag2 S NC inhibited proliferation of the virus through blockage of viral RNA synthesis and budding. |
[86] | |
| CD based on curcumin | Electrostatic interaction | CD based on curcumin resulted in better at reducing virus titers when correlated to the control CD. The nanosystem can repress viral approach, the synthesis of negative-stranded RNA, the budding of the virus, and the reactive oxygen species (ROS) produced by PEDV | [87] | |
| Feline coronavirus and infectious bursal disease virus | GO and GO-Ag | Electrostatic interactions and hydrophobic interactions within the lipid and the aromatic plane of GO. The basic relationship between thiol residues and Ag | GO and GO-Ag exhibited repression of FCoV with better results for the latter (only GO-Ag exhibited inhibition for IBDV). For clogged viruses, GO acts as a support and contributes towards the inhibition of the virus. No hydrophobic interactions exist for non-enveloped viruses and virus inhibition requires a divert bond. | [88] |
| Bacteriophages MS2 and ΦX174, murine norovirus, and adenovirus serotype 2 | Ag-MHC | The basic relationship between thiol residues and Ag, Ag+ ions can produce ROS and contribute towards virus inactivation | The Ag-MHC operation produced significant antiviral effects towards bacteriophages 4X174 and MNV, but not towards AdV2. Antiviral actions were diminished at pH 2 and 12. When estimating bacteriophage inactivation in tap and surface waters, Ag-MHC maintained its activity towards butX174, but it was diminished for MS2 correlated to regulators (distilled water). | [89] |
| Human coronavirus-229E | CQD modified with boronic acid moieties | Glycan borono-lectin interaction involving the S protein | CQDs were reactivated with boronic acid, phenylboronic acid, and 4-aminophenyl boronic acid, which determined antiviral action with better results for the latter. Viral repression action in the replication phase was determined. | [90] |
| Influenza A/PR/8/34 (H1N1) | SNP-DDAB | Electrostatic interaction | Nanosystems show antifungal, antibacterial, and antiviral action when applied to surfaces. The existence of any virus was not detected when contacted with nanosystems and influenza virus-coated glass coverslips. | [91] |
| Influenza strain X-31, A/Aichi/2/68 (H3N2) | NT-P | ROS upon irradiation | When NCI cells were treated with NT-P in the presence of a virus, the percentage of infection was reduced to 1% after 90 minutes of light irradiation. NT-P material can be used up to 5 times. The writers envisioned this substance as a disinfectant of surfaces and water. | [92] |
| Japanese encephalitis virus, dengue virus serotype 2, and influenza A | NCa, NCb, and NCc | Electrostatic interaction | NCcnanosystems essentially obstructed JEV infection at the onset of viral contamination. NCcnanosystem protected mice (80–100% protection) when vaccinated with JEV or DEN-2 prefabricated with NCc. In clinical studies, 6 hpi with JEV, a treatment with NCc, resulted in 60–80% protection of mice. | [93] |
| T7 bacteriophage | CNH-PL-T7 | Antibody-antigen interaction + photothermal effect | Both nanosystems (with and without antibodies) were able to eliminate T7 phage upon NIR irradiation. When using a thin suspension of nanohorns, only the targeted nanohorns were able to eliminate the T7 phage. | [94] |
| Hepatitis C virus | SNP-BA, MNP-BA, and ND-BA | Glycan borono-lectin interaction | None of the nanosystems exhibited important cell viability after 2 h. 58, 47, and 60% of viral repression were recorded for ND-BA, MNP-BA, and SNP-BA, sequentially. The nanosystems analyzed were able to diminish virus penetration. | [95] |
Gt, graphite; GtO, graphite oxide; GO, graphene oxide; rGO, reduced graphene oxide; GO-PDDA, graphene oxide/poly (diallyldimethylammonium chloride) composite; GO-PVP, graphene oxide-polyvinylpyrrolidone composite; GO-Ag, graphene oxide-silver nanocomposite; Ag-MHC, magnetic hybrid colloid decorated with Ag nanoparticles; CQD, carbon quantum dots; NC, nano-clusters; CD, carbon dots; SNP, silica nanoparticles; DDAB, didodecyldimethylammonium bromide; SNP-BA, silica nanoparticles-boronic acid; MNP-BA, iron oxide nanoparticle-boronic acid; ND-BA, nanodiamond-boronic acid; NCa, nanoclay with C18 tallow alkyl amine; NCb, nanoclay with triton X100; NCc, nanoclay with sodium dodecyl sulphate; CNH-PL-T7, carbon nanohorns functionalized with PEG distearoylphosphatidylethanolamine (PL) and T7 tag antibody; NT-P, multi-walled carbon nanotubes (MWNT) functionalized with protoporphyrin (PPIX)
Nano systems with that work against SARS-CoV-2 have been summarized in Table 1. There are two main approaches to deactivate enveloped viruses. The first includes interaction between viral surface and nanomaterial for its inactivation. Nano system consists of hydrophobic layer that interacts with the lipid membrane present on viral surface encouraging denaturing. Examples possibly will be 1 (rGO) and 5 (CD); electrostatic interactions, 2 (SNP-DDAB), 4 (NCc), and 6 (GO); 3 (CQD modified with boronic acid); borono-lectin-sugar interaction, 7 (GO-Ag); combined dative bond and 8 (Ag-MHC); and dative bond interaction. Second approach deals with the nano system that entirely executes the virus. Both approaches are effective to act against SARS-CoV-2 [96]. Another approach using nanosystems, as previously mentioned, has an additional feature in them that can be used to kill viruses. In this regard, electromagnetic radiation has been employed to activate this additional characteristic either by reactive species or by photothermal heating. Therefore, viruses can be killed by oxidation or denaturation processes [96].
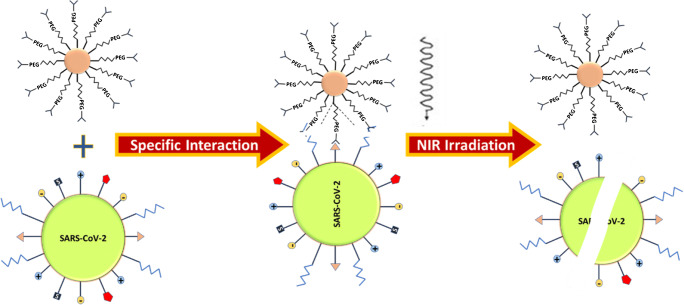
Miyako et al. [97] shaped carbon nanohorns (CNH) operationalized with a PEG derivative with PL (distearoylphosphatidylethanolamine) at one end and a T7 tag antibody (Ab) at the other end, NP sizes were of 50–150 nms. PEG has been incorporated to provide stability, PL to solubilize nanoparticles, and Ab to target T7 bacteriophage (T7 phage) as prototype virus. The overall impression (Fig. 2) signifies virus binding to the CNH, site-specific exclusion of the T7 phage exist via photothermal effect under NIR laser irradiation of 1064 nm. For control nanosystem, the Ab was not involved in NP system. Individually, both NS examined in presence and absence of Ab and found that they eliminated T7 phage upon NIR (near infrared) radiation (phage capsid proteins are denatured due to heat). When diluted suspensions of nanohorns were utilized, only marked NHs eliminated T7 phage [97].
Fig. 2.
Antibody-antigen interaction between a targeted nanosystem and a virus with NIR absorption for photothermal heating to inactivate the virus
Baneerjee et al. [98] synthesized multi-walled carbon nanotubes (MWNT) with protoporphyrin (PPIX) as nanomaterial (NT-P) to decrease potential of influenza-A virus for infecting mammalian cells. Influenza strain X-31, A/Aichi/2/68 (H3N2), viruses tested and pulmonary mucoepidermoid carcinoma cell line NCI-H292 (ATCC# CRL-1848) were used. The NPs designed initially by acid-functionalized MWNT aminated PPIX and reacted under the influence of thionyl chloride. When NCI cells treated with NT-P in virus prominence, as a result of 90-min light irradiation, the infection was dropped to 1% while 78% infection was accomplished in dark system. This defines release of ROS by PPIX due to light and not MWNT. As anticipated, photobleaching is a key component of NT-P that may perhaps be reused five times. Meanwhile, the system generates ROS (that influences protein oxidation, ruptures RNA genomes, and crosslinks protein-RNA [99]); it could likely work against all types of influenza virus strain.
Nano Vaccine Models of Relevance in the Fight Against COVID-19
The nanotechnology can play a prime role to fight against COVID-19 utilizing nano vaccines, which contain nanoparticles and act as carriers of Ag that could activate defensive immune reactions. Antigen processing cells have ability of capturing microparticulate Ag complex to boost adaptive autoimmune responses that subsist with pathogen after entering inside an organism. Nanomaterials have inbuilt immunomodulatory properties that can impact therapeutic action of vaccine [100]. Hence, these can be a good choice to evade viral diseases. In this context, VLPs could be a leading approach for the development of safer nano vaccines due to highly immunized macromolecular complex, imitating antigenic effects of microorganism. The recombinant vaccines based on VLPs (vaccines against hepatitis B virus and human papillomavirus) are very much commercialized [101]. In addition to VLPs, other NPs have also been utilized in the field of vaccinology; some examples are liposome, gold NPs, chitosan, and PLGA [102]. Many researchers are targeting vaccines for SARS-CoV-1 and MERS-CoV and described in the works; these reports can be preliminary facts to develop SARS-CoV-2; among all, VLPs were utmost discovered strategy. Real-time expression of S (spike), E (envelope), M (membrane), and N (nucleocapsid) proteins of SARS-CoV serve to produce VLPs engaging baculovirus/insect cells; they depicted similar homologous characters with that of SARS-CoV virion when observed under an electron microscope [103, 104]. One report suggested that VLPs are formed within insect expressing M and E protein barely [105]. A set of four subcutaneous doses of VLP (mixed with Freund’s adjuvant) immunized to mice model leads to release of high Ab titers counter to SARS-CoV. VLPs provoked cellular resistance by enhancing IFN-γ and IL-4 production [106]. In another in vitro research, DCs triggered production of IL-6 and TNF-α after VLPs concocted with E and M proteins from SARS-CoV and S protein from a bat-isolated CoV. Furthermore, DCs pre-exposed to VLPs bring about an upsurge in the quantity of IFN-γ + IL-4 + CD4 + T cells [107]. For mucosal immunization (being documented as the most relevant), VLPs from SARS-CoV were examined in a mouse prototype exposed to intraperitoneal or intranasal vaccination. Both lead to SARS-CoV-specific IgG upregulation with increased levels in case of intraperitoneal route (i.p. route). Contrasting parenteral administration, intranasal delivery prompts IgA response that can be traced in the GIT and lungs. IgA level was higher for the i.p. immunization in the intestinal tract rather than intranasal route (i.n. route). However, secretions from the venereal region for mice vaccinated intranasally exhibited low response [108]. A number of studies indicate that VLPs are synthesized in the baculovirus/insect cell system by co-expression of M1 influenza protein and the S protein from SARS-CoV. The resultant chimeric VLPs remained analogous to the wild-type SARS-CoV when linking size and structure. The major troublesome task is to develop immunogenicity and defensive effectiveness, the chimeric VLPs totally saved mice after i.m. and i.n. immunization of SARS protein S [104, 109]. Rhesus macaques when immunized with VLPs and Al adjuvant exhibited release of T-helper 1 (Th1) cell-mediated immunity [104, 110]. Silkworm larvae and Bm5 cells were confirmed to synthesize vaccine contenders with the help of MERS-CoV inherent protein S, E, and M. Despite that E and M proteins are released and secreted to the culture supernatant, the S protein is absent as VLPs. Nanosized vesicle (100–200 nm through immune-TEM) displayed that S protein was produced from Bm5 cells stating operational proteins of the S protein via surface active agent processing and physical ejection [111]. Thus, the nanoformulation of COVID-19 vaccines might be more effectively helpful in the immunization against the current pandemic among students of higher education and understudies stakeholders in school education system too [112].
Conclusions
The data presented in the current review depicted that nanotechnology has allowed development of various biosensors, nanovaccines, and antiviral composites that are highly effective for close-related viruses; thus, it is a valuable guide for the agent’s development against SARS-CoV-2. Therefore, there is much more that nanotechnology has to offer in the fight against COVID-19 pandemic and the following months will be critical for the exploitation of unique characteristics of nanosized sensors, antiviral composites, and vaccines in the fight against this unprecendented global health crisis at multiple levels, especially for viral spread prevention and establishment of infection, but also in an accurate and timely diagnostic. For the proper containment of COVID-19 or any other emerging coronaviral pandemic, the detailed understanding of the virus virulence and the transmission are required which will enable a better understanding of virus transfer between species along with the identification of the differently encoded nanostructural enzymes and proteins and the related mechanism of action. Accordingly, new therapeutic targets can be recognized and targeted by the use of surface-functionalized NPs. Also, the study of the lifecycle of the virus and the host’s response will help us to produce effective nanovaccine. Based on these studies, the future aspect of a broad-spectrum ‘universal’ NP-based vaccine or therapeutic is expected to be ready for the current and future CoV pandemics. It can be predicted interestingly that microfluidics will have the contribution in CoV detection, taking the benefits of miniaturization, rapid detection, and portability. As the infection and spread of the viruses is way faster than the effective vaccines, vaccines and drug study should be complementary to what has already been achieved in the previous CoV-related research. The infection-to-mortality ratio because of current global health emergency has crossed the limits, making it different from other viral infections. The tools of nanotechnology can be modified and used for detecting, treating, and preventing the spread of COVID-19. Nanotechnology offers a unique set of tools which can help in the contribution and in the understanding of viral diseases and towards the critical development of therapeutic and diagnostic platforms.
Acknowledgements
Authors would like to express their gratitude to the facilities provided by Maharishi Markandeshwar (Deemed to be University) Mullana-Ambala (Haryana), India, for carrying out this work.
Compliance with Ethical Standards
Conflict of Interest
None.
Human and Animal Rights and Informed Consent
The article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Clinical Pharmacology
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Adhikari SP, Meng S, Yu-Ju W, Yu-Ping M, Rui-Xue Y, Qing-Zhi W, Sun C, Sylvia S, Rozelle S, Raat H, Zhou H. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: A scoping review. Infect Dis Poverty. 2020;9:29. doi: 10.1186/s40249-020-00646-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Upadhyay SK, Singh R, Babita KG, Singh G. The outbreak and challenges of novel coronavirus (COVID-19): The global pandemic emergency of early 2K20 and Indian scenario. Int J Biol Pharm Allied Sci. 2020;9:1173–1199. doi: 10.31032/IJBPAS/2020/9.5.5126. [DOI] [Google Scholar]
- 3.Yadav R, Parihar RD, Dhiman U, Dhamija P, Upadhyay SK, Imran M, Behera SK, Prasad TSK. Docking of FDA approved drugs targeting NSP-16, N-protein and main protease of SARS-CoV-2 as dual inhibitors. Biointerface Res Appl Chem. 2020;11:9848–9861. doi: 10.33263/BRIAC113.98489861. [DOI] [Google Scholar]
- 4.Waris A, Khan AU, Ali M, Ali A, Baset A. COVID-19 outbreak: Current scenario of Pakistan. New Microbes New Infect. 2020;100681:100681. doi: 10.1016/j.nmni.2020.100681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sood S, Aggarwal V, Aggarwal D, Upadhyay SK, Sak K, Tuli HS, Kumar M, Kumar J, Talwar S. COVID-19 pandemic: From molecular biology, pathogenesis, detection and treatment to global societal impact. Curr Pharmacol Rep. 2020;6(5):212–227. doi: 10.1007/s40495-020-00229-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kaul D. An overview of coronaviruses including the SARS-2 coronavirus-Molecular biology, epidemiology and clinical implications. Curr Med Res Pract. 2020;10(2):54–64. doi: 10.1016/j.cmrp.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Upadhyay SK, Singh R, Singh M, Kumar V, Yadav M, Aggarwal D, Sehrawat N. COVID-19 in republic of India: A report on situation and precautionary strategies to global pandemic. Bull Environ Pharmacol Life Sci. 2020;9:39–48. [Google Scholar]
- 8.Curley M, Thomas N. Human security and public health in Southeast Asia: The SARS outbreak. Aust J Int Aff. 2004;58:17–32. doi: 10.1080/1035771032000184737. [DOI] [Google Scholar]
- 9.Kumar G, Singh G, Bhatnagar V, Gupta R, Upadhyay SK. Outcome of online teaching-learning over traditional education during covid-19 pandemic. Int J Adv Trends Comput Sci Eng. 2020;9(5):7704–7711. doi: 10.30534/ijatcse/2020/113952020. [DOI] [Google Scholar]
- 10.Kumar S, Rathi B. Coronavirus disease COVID-19: A new threat to public health. Curr Top Med Chem. 2020;20(8):599–600. doi: 10.2174/1568026620999200305144319. [DOI] [PubMed] [Google Scholar]
- 11.Dan S, Pant M, Upadhyay SK. The case fatality rate (CFR) in SARS-COV-2 patients with cardiovascular disease (CVD): A global health risk paradigm in COVID-19 pandemic. Curr Pharmacol Rep. 2020;6:315–324. doi: 10.1007/s40495-020-00239-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li G, De Clercq E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV) Nat Rev Drug Discov. 2020;19(3):149–150. doi: 10.1038/d41573-020-00016-0. [DOI] [PubMed] [Google Scholar]
- 13.Jha D, Sharma V, Sharma JK, Kumar S, Sharma V, Kamboj P, Upadhyay SK, Sharma AK. Plausible state-specific plans and recommendations to avert COVID-19 community transmission. Bull Pure Appl Sci Zool. 2020;39A(2):447–454. doi: 10.5958/2320-3188.2020.00051.0. [DOI] [Google Scholar]
- 14.Hashmi HAS, Asif HM. Early detection and assessment of COVID-19. Front Med. 2020;7:311. doi: 10.3389/fmed.2020.00311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Agarwal HK, Chhikara BS, Doncel GF, Parang K. Synthesis and anti-HIV activities of unsymmetrical long chain dicarboxylate esters of dinucleoside reverse transcriptase inhibitors. Bioorg Med Chem Lett. 2017;27:1934–1937. doi: 10.1016/j.bmcl.2017.03.031. [DOI] [PubMed] [Google Scholar]
- 16.Capua I, Cattoli G. One Health (r) evolution: Learning from the past to build a new future. Viruses. 2018;10:725. doi: 10.3390/v10120725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sumaryada T, Pramudita CA. Molecular docking evaluation of some Indonesian’s popular herbals for a possible COVID-19 treatment. Biointerface Res Appl Chem. 2020;11:9827–9835. doi: 10.33263/BRIAC113.98279835. [DOI] [Google Scholar]
- 18.Why interdisciplinary research matters. Nature. 2015;525:305. 10.1038/525305a. [DOI] [PubMed]
- 19.Weiss C, Carriere M, Fusco L, Capua I, Regla-Nava JA, Pasquali M, Scott JA, Vitale F, Unal MA, Mattevi C, Bedognetti D, Merkoçi A, Tasciotti E, Yilmazer A, Gogotsi Y, Stellacci F, Delogu LG. Toward nanotechnology-enabled approaches against the COVID-19 Pandemic. ACS Nano. 2020;14(6):6383–6406. doi: 10.1021/acsnano.0c03697. [DOI] [PubMed] [Google Scholar]
- 20.Gnanasangeetha D, Sarala TD. One pot synthesis of zinc oxide nanoparticles via chemical and green method. Res J Mater Sci. 2013;2320:6055. [Google Scholar]
- 21.Chhikara BS. Corona virus SARS-CoV-2 disease COVID-19: Infection, prevention and clinical advances of the prospective chemical drug therapeutics. Chem Biol Lett. 2020;7:63–72. [Google Scholar]
- 22.Waris A, Ali M, Khan AU, Ali A, Baset A. Role of nanotechnology in diagnosing and treating COVID-19 during the pandemic. Int J Clin Virol. 2020;4:065–070. doi: 10.29328/journal.ijcv.1001017. [DOI] [Google Scholar]
- 23.Udugama B, Kadhiresan P, Kozlowski HN, Malekjahani A, Osborne M, Li VYC, Chen H, Mubareka S, Gubbay JB, Chan WCW. Diagnosing COVID-19: The disease and tools for detection. ACS Nano. 2020;14(4):3822–3835. doi: 10.1021/acsnano.0c02624. [DOI] [PubMed] [Google Scholar]
- 24.Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet. 2020;395(10223):470–473. doi: 10.1016/S0140-6736(20)30185-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhao Z, Cui H, Song W, Ru X, Zhou W, Yu X. A simple magnetic nanoparticles-based viral RNA extraction method for efficient detection of SARS-CoV-2. Bio Rxiv. 2020. 10.1101/2020.02.22.961268.
- 26.Gong P, He X, Wang K, Tan W, Xie W, Wu P, Li H. Combination of functionalized nanoparticles and polymerase chain reaction-based method for SARS-CoV gene detection. J Nanosci Nanotechnol. 2008;8(1):293–300. doi: 10.1166/jnn.2008.18130. [DOI] [PubMed] [Google Scholar]
- 27.Kim H, Park M, Hwang J, Kim JH, Chung DR, Lee KS, Kang M. Development of label-free colorimetric assay for MERS-CoV using gold nanoparticles. ACS Sens. 2019;4(5):1306–1312. doi: 10.1021/acssensors.9b00175. [DOI] [PubMed] [Google Scholar]
- 28.Martinez-Paredes G, Gonzalez-Garcia MB, Costa-Garcia A. Genosensor for SARS virus detection based on gold nanostructured screen-printed carbon electrodes. Electroanalysis. 2009;21(3–5):379–385. doi: 10.1002/elan.200804399. [DOI] [Google Scholar]
- 29.Layqah LA, Eissa S. An electrochemical immunosensor for the corona virus associated with the Middle East respiratory syndrome using an array of gold nanoparticle-modified carbon electrodes. Mikrochim Acta. 2019;186(4):224. doi: 10.1007/s00604-019-3345-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang P, Wang H, Cao Z, Jin H, Chi H, Zhao J, Yu B, Yan F, Hu X, Wu F, Jiao C, Hou P, Xu S, Zhao Y, Feng N, Wang J, Sun W, Wang T, Gao Y, Yang S, Xia X. A rapid and specific assay for the detection of MERS-CoV. Front Microbiol. 2018;9(1101):1–9. doi: 10.3389/fmicb.2018.01101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Genie A. Rapid COVID-19 antibody detection tests: Principles and methods. http://www.assaygenie.com/rapid-covid19-antibody-detection-tests-principles-and-methods. Accessed Mar 2020.
- 32.Xiang J, Yan M, Li H, et al. Evaluation of enzyme-linked immunoassay and colloidal gold-immunochromatographic assay kit for detection of novel coronavirus (SARS-Cov-2) causing an outbreak of pneumonia (COVID-19). Med Rxiv. 2020. 10.1101/2020.02.27.20028787.
- 33.Radwan SH, Azzazy HM. Gold nanoparticles for molecular diagnostics. Expert Rev Mol Diagn. 2009;9(5):511–524. doi: 10.1586/erm.09.33. [DOI] [PubMed] [Google Scholar]
- 34.Ahmed SR, Nagy E, Neethirajan S. Self-assembled star-shaped chiroplasmonic gold nanoparticles for an ultrasensitive chiro-immunosensor for viruses. RSC Adv. 2017;7(65):40849–40857. doi: 10.1039/C7RA07175B. [DOI] [Google Scholar]
- 35.Abd Ellah NH, Gad SF, Muhammad K, Batiha GE, Hetta HF. Nanomedicine as a promising approach for diagnosis, treatment and prophylaxis against COVID-19. Nanomedicine. 2020;15(21):2085–2102. doi: 10.2217/nnm-2020-0247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Laromaine A, Koh L, Murugesan M, Ulijn RV, Stevens MM. Protease-triggered dispersion of nanoparticle assemblies. J Am Chem Soc. 2007;129(14):4156–4157. doi: 10.1021/ja0706504. [DOI] [PubMed] [Google Scholar]
- 37.Yang W, Dang X. Rapid detection of SARS-CoV-2 using reverse transcription RT-LAMP method. Med Rxiv. 2020. 10.1101/2020.03.02.20030130.
- 38.Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y, Ma X, Zhan F, Wang L, Hu T, Zhou H, Hu Z, Zhou W, Zhao L, Chen J, Meng Y, Wang J, Lin Y, Yuan J, Xie Z, Ma J, Liu WJ, Wang D, Xu W, Holmes EC, Gao GF, Wu G, Chen W, Shi W, Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chan WCW. Nano research for COVID-19. ACS Nano. 2020;14:3719–3720. doi: 10.1021/acsnano.0c02540. [DOI] [PubMed] [Google Scholar]
- 40.Chotiwan N, Brewster CD, Magalhaes T, Weger-Lucarelli J, Duggal NK, et al. Rapid and specific detection of Asian-and African-lineage Zika viruses. Sci Transl Med. 2017;9:eaag0538. doi: 10.1126/scitranslmed.aag0538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Law S, Leung AW, Xu C. Severe acute respiratory syndrome (SARS) and coronavirus disease-2019 (COVID-19): From causes to preventions in Hong Kong. Int J Infect Dis. 2020;94:156–163. doi: 10.1016/j.ijid.2020.03.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yang W, Dang X, Wang Q, Xu M, Zhao Q, et al. Rapid detection of SARS-CoV-2 using reverse transcription RT-LAMP method. Med Rxiv. 2020. 10.1101/2020.03.02.20030130.
- 43.Fleming AB, Raabe V. Current studies of convalescent plasma therapy for COVID-19 may underestimate risk of antibody-dependent enhancement. J Clin Virol. 2020;127:104388. doi: 10.1016/j.jcv.2020.104388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xiang J, Yan M, Li H, Liu T, Lin C, et al. Evaluation of enzyme-linked immunoassay and colloidal gold-immunochromatographic assay kit for detection of novel coronavirus (SARS-Cov-2) causing an outbreak of pneumonia (COVID-19). Med Rxiv. 2020. 10.1101/2020.02.27.20028787.
- 45.Zhu X, Wang X, Han L, Chen T, Wang L, et al. Reverse transcription loop-mediated isothermal amplification combined with nanoparticles based biosensor for diagnosis of COVID-19. Med Rxiv. 2020. 10.1101/2020.03.17.20037796. [DOI] [PMC free article] [PubMed]
- 46.Wang M, Fu A, Hu B, Tong Y, Liu R, et al. Nanopore target sequencing for accurate and comprehensive detection of SARS-CoV-2 and other respiratory viruses. Med Rxiv. 2020. 10.1101/2020.03.04.20029538.
- 47.Liu R, Fu A, Deng Z, Li Y, Liu T. Promising methods for detection of novel coronavirus SARS-CoV-2. View. 2020;1(1):e4. doi: 10.1002/viw2.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yu L, Tong Y, Shen G, Fu A, Lai Y, et al. Immunodepletion with hypoxemia: A potential high risk subtype of coronavirus disease 2019. Med Rxiv. 2020. 10.1101/2020.03.03.20030650.
- 49.Herzog C, Hartmann K, Künzi V, Kürsteiner O, Mischler R, Lazar H, Glück R. Eleven years of Inflexal V-a virosomal adjuvanted influenza vaccine. Vaccine. 2009;27(33):4381–4387. doi: 10.1016/j.vaccine.2009.05.029. [DOI] [PubMed] [Google Scholar]
- 50.Alconcel SNS, Baas AS, Maynard HD. FDA-approved poly(ethylene glycol)-protein conjugate drugs. Polym Chem. 2011;2:1442–1448. doi: 10.1016/j.vaccine.2009.05.029. [DOI] [Google Scholar]
- 51.Donalisio M, Leone F, Civra A, Spagnolo R, Ozer O, Lembo D, Cavalli R. Acyclovir-loaded chitosan nanospheres from nano-emulsion templating for the topical treatment of herpesviruses infections. Pharmaceutics. 2018;10:46. doi: 10.3390/pharmaceutics10020046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Seto WK, Yuen MF. New pharmacological approaches to a functional cure of hepatitis B. Clin Liver Dis (Hoboken) 2016;8:83–88. doi: 10.1002/cld.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lara HH, Ayala-Nuñez NV, Ixtepan-Turrent L, Rodriguez-Padilla C. Mode of antiviral action of silver nanoparticles against HIV-1. J Nanobiotechnol. 2010;8:1–10. doi: 10.1186/1477-3155-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Morris D, Ansar M, Speshock J, Ivanciuc T, Qu Y, Casola A, Garofalo R. Antiviral and immunomodulatory activity of silver nanoparticles in experimental RSV infection. Viruses. 2019;11(8):732. doi: 10.3390/v11080732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Cagno V, Andreozzi P, D’Alicarnasso M, Silva PJ, Mueller M, Galloux M, et al. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat Mater. 2018;17:195–203. doi: 10.1038/nmat5053. [DOI] [PubMed] [Google Scholar]
- 56.Kim SS, Peer D, Kumar P, Subramanya S, Wu H, Asthana D, Habiro K, Yang YG, Manjunath N, Shimaoka M, Shankar P. RNAi-mediated CCR5 silencing by LFA-1-targeted nanoparticles prevents HIV infection in BLT mice. Mol Ther. 2010;18:370–376. doi: 10.1038/mt.2009.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cojocaru FD, Botezat D, Gardikiotis I, Uritu CM, Dodi G, Trandafir L. Nanomaterials designed for antiviral drug delivery transport across biological barriers. Pharmaceutics. 2020;12:171. doi: 10.3390/pharmaceutics12020171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vazquez-Munoz R, Lopez-Ribot JL. Nanotechnology as an alternative to reduce the spread of COVID-19. Challenges. 2020;11(2):15. doi: 10.3390/challe11020015. [DOI] [Google Scholar]
- 59.Pulit-Prociak J, Banach M. Silver nanoparticles-A material of the future? Open Chem. 2016;14:76–91. doi: 10.1515/chem-2016-0005. [DOI] [Google Scholar]
- 60.Ali A, Ahmed S. A review on chitosan and its nanocomposites in drug delivery. Int J Biol Macromol. 2018;109:273–286. doi: 10.1016/j.ijbiomac.2017.12.078. [DOI] [PubMed] [Google Scholar]
- 61.Peeri NC, Shrestha N, Rahman MS, Zaki R, Tan Z, Bibi S, Baghbanzadeh M, Aghamohammadi N, Zhang W, Haque U. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: what lessons have we learned? Int J Epidemiol. 2020;49(3):717–726. doi: 10.1093/ije/dyaa033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ziaie S, Koucheck M, Miri M, Salarian S, Shojaei S, et al. Review of therapeutic agents for the treatment of COVID-19. J Cell Mol Anesth. 2020;5(1):32–36. doi: 10.22037/jcma.v5i1.29760. [DOI] [Google Scholar]
- 63.Song Z, Hu Y, Zheng S, Yang L, Zhao R. Hospital pharmacists’ pharmaceutical care for hospitalized patients with COVID-19: Recommendations and guidance from clinical experience. Res Soc Adm Pharm. 2020;17:2027–2031. doi: 10.1016/j.sapharm.2020.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tobaiqy M, Qashqary M, Al-Dahery S, et al. Therapeutic management of patients with COVID-19: A systematic review. Infect Prev Pract. 2020;2(3):100061. doi: 10.1016/j.infpip.2020.100061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Donders F, Lonnée-Hoffmann R, Tsiakalos A, Mendling W, Martinez de Oliveira J, et al. COVID I. ISIDOG recommendations concerning COVID-19 and pregnancy. Diagnostics. 2020;10(4):243. doi: 10.3390/diagnostics10040243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review. JAMA. 2020;323(18):1824–1836. doi: 10.1001/jama.2020.6019. [DOI] [PubMed] [Google Scholar]
- 67.Wang L, Wang Y, Ye D, Liu Q. Review of the 2019 novel coronavirus (SARS-CoV-2) based on current evidence. Int J Antimicrob Agents. 2020;55(6):105948. doi: 10.1016/j.ijantimicag.2020.105948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kaura KK, Allahbadiab G, Singh M. A comprehensive review on epidemiology, aetiopathogenesis, diagnosis and treatment of the novel coronavirus syndrome–COVID-19. Iberoamerican J Med. 2020:110–23. 10.5281/zenodo.3757110.
- 69.Dong L, Hu S, Gao J. Discovering drugs to treat coronavirus disease 2019 (COVID-19) Drug Discov Ther. 2020;14(1):58–60. doi: 10.5582/ddt.2020.01012. [DOI] [PubMed] [Google Scholar]
- 70.Huy TQ, Hien Thanh NT, Thuy NT, Chung PV, Hung PN, le AT, Hong Hanh NT. Cytotoxicity and antiviral activity of electrochemical-synthesized silver nanoparticles against poliovirus. J Virol Methods. 2017;241:52–57. doi: 10.1016/j.jviromet.2016.12.015. [DOI] [PubMed] [Google Scholar]
- 71.Dynamiker. Clinical value of IgG/IgM detection for COVID-19. http://en.dynamiker.com/index/index/news_info/aid/608.html. Accessed 16 Mar 2020.
- 72.Li W, Balachandran YL, Hao Y, Hao X, Li R, Nan Z, Zhang H, Shao Y, Liu Y. Amantadine surface-modified silver nanorods improves immunotherapy of HIV vaccine against HIV-infected cells. ACS Appl Mater Interfaces. 2018;10(34):28494–28501. doi: 10.1021/acsami.8b10948. [DOI] [PubMed] [Google Scholar]
- 73.Joshy KS, Snigdha S, Anne G, Nandakumar K, Laly AP, Sabu T, et al. Chem Phys Lipids. 2018;210:82–89. doi: 10.1016/j.chemphyslip.2017.11.003. [DOI] [PubMed] [Google Scholar]
- 74.Kumar P, Lakshmi YS, Kondapi AK. Triple drug combination of zidovudine, efavirenz and lamivudine loaded lactoferrin nanoparticles: An effective nano first-line regimen for HIV therapy. Pharm Res. 2017;34(2):257–268. doi: 10.1007/s11095-016-2048-4. [DOI] [PubMed] [Google Scholar]
- 75.Joshy KS, George A, Jose J, Kalarikkal N, Pothen LA, Thomas S. Novel dendritic structure of alginate hybrid nanoparticles for effective anti-viral drug delivery. Int J Biol Macromol. 2017;103:1265–1275. doi: 10.1016/j.ijbiomac.2017.05.094. [DOI] [PubMed] [Google Scholar]
- 76.Li Y, Lin Z, Xu T, Wang C, Zhao M, Xiao M, Wang H, Deng N, Zhu B. Delivery of VP1 siRNA to inhibit the EV71 virus using functionalized silver nanoparticles through ROS-mediated signaling pathways. RSC Adv. 2017;7:1453–1463. doi: 10.1039/c6ra26472g. [DOI] [Google Scholar]
- 77.Du T, Liang J, Dong N, Lu J, Fu Y, Fang L, Xiao S, Han H. Glutathione-capped Ag2S nanoclusters inhibit coronavirus proliferation through blockage of viral RNA synthesis and budding. ACS Appl Mater Interfaces. 2018;10:4369–4378. doi: 10.1021/acsami.7b13811. [DOI] [PubMed] [Google Scholar]
- 78.Asgary V, Shoari A, Afshar Moayad M, ShafieeArdestani M, Bigdeli R, Ghazizadeh L, Khosravy MS, Panahnejad E, Janani A, Bashar R, Abedi M, Ahangari CR. Evaluation of G2 citric acid-based dendrimer as an adjuvant in veterinary rabies vaccine. Viral Immunol. 2018;31:47–54. doi: 10.1089/vim.2017.0024. [DOI] [PubMed] [Google Scholar]
- 79.Lancelot A, Clavería-Gimeno R, Velázquez-Campoy A, Abian O, Serrano JL, Sierra T. Nanostructures based on ammonium-terminated amphiphilic Janus dendrimers as camptothecin carriers with antiviral activity. Eur Polym J. 2017;90:136–149. doi: 10.1016/j.eurpolymj.2017.03.012. [DOI] [Google Scholar]
- 80.Chahal JS, Fang T, Woodham AW, Khan OF. Ling J, Anderson DG, Ploegh HL. An RNA nanoparticle vaccine against Zika virus elicits antibody and CD8+ T cell responses in a mouse model. Sci Rep. 2017;7:1–9. doi: 10.1038/s41598-017-00193-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Muñoz A, Illescas BM, Luczkowiak J, Lasala F, Ribeiro-Viana R, Rojo J, Delgado R, Martín N. Antiviral activity of self-assembled glycodendro[60]fullerene monoadducts. J Mater Chem. 2017;5:6566–6571. doi: 10.1039/c7tb01379e. [DOI] [PubMed] [Google Scholar]
- 82.Lee J, Morita M, Takemura K, Park EY. A multi-functional gold/iron-oxide nanoparticle-CNT hybrid nanomaterial as virus DNA sensing platform. Biosens Bioelectron. 2018;102:425–431. doi: 10.1016/j.bios.2017.11.052. [DOI] [PubMed] [Google Scholar]
- 83.Halder A, Das S, Ojha D, Chattopadhyay D, Mukherjee A. Highly monodispersed gold nanoparticles synthesis and inhibition of herpes simplex virus infections. Mater Sci Eng. 2018;89:413–421. doi: 10.1016/j.msec.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 84.Tong T, Hu H, Zhou J, Deng S, Zhang X, Tang W, Fang L, Xiao S, Liang J. Glycyrrhizic-acid-based carbon dots with high antiviral activity by multisite inhibition mechanisms. Small. 2020;16:1–12. doi: 10.1002/smll.201906206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.FDA. Cellex qSARS-CoV-2 IgG/IgM rapid test. 2020. https://www.fda.gov/media/136625/download. Accessed Apr 2020.
- 86.EDI™. Novel coronavirus COVID-19 ELISA kits. Epitope Diagnistics Inc. 2020. http://www.epitopediagnostics.com/covid-19-elisa. Accessed Apr 2020.
- 87.SYBCL. SARS-CoV-2 solution. Shenzhen Yhlo Biotech Co. Ltd. 2020. https://www.amp-asbach.de/index_files/flyer_cov2.pdf. Accessed Apr 2020.
- 88.FDA. COVID-19 IgM/IgG system. DPP®. 2020. https://www.fda.gov/media/136963/download. Accessed Apr 2020.
- 89.USFDA. US food and drug administration VITROS immunodiagnostic product anti-SARS-CoV2 total reagent pack. Ortho-Clinical Diagnosis Inc. 2020. https://www.fda.gov/media/136970/download. Accessed Apr 2020.
- 90.Sure Biotech. Coronavirus (COVID-19) rapid test. 2020. https://www.surebiotech.com/rapid-test/coronavirus-covid-19-rapid-test/#intro. Accessed Apr 2020.
- 91.Ye S, Shao K, Li Z, Guo N, Zuo Y, Li Q, Lu Z, Chen L, He Q, Han H. Antiviral activity of graphene oxide: How sharp edged structure and charge matter. ACS Appl Mater Interfaces. 2015;7(38):21571–21579. doi: 10.1021/acsami.5b06876. [DOI] [PubMed] [Google Scholar]
- 92.LoczechinA SK. BarrasA, et al. Functional carbon quantum dots as medical countermeasures to human coronavirus. ACS Appl Mater. 2019;11(46):42964–42974. doi: 10.1021/acsami.9b15032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Elechiguerra JL, Burt JL, Morones JR, Camacho-Bragado A, Gao X, Lara HH, Yacaman M. Interaction of silver nanoparticles with HIV-1. J Nanobiotechnol. 2005;3(1):6. doi: 10.1186/1477-3155-3-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Galdiero S, Falanga A, Vitiello M, Cantisani M, Marra V, Galdiero M. Silver nanoparticles as potential antiviral agents. Molecules. 2011;16(10):8894–8918. doi: 10.3390/molecules16108894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chen YN, Hsueh YH, Hsieh CT, Tzou DY, Chang PL. Antiviral activity of graphene-silver nanocomposites against non-enveloped and enveloped viruses. Int J Environ Res Public Health. 2016;13(4):430. doi: 10.3390/ijerph13040430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Palestino G, García-Silva I, González-Ortega O, Rosales-Mendoza S. Can nanotechnology help in the fight against COVID-19? Expert Rev Anti-Infect Ther. 2020;18:1–16. doi: 10.1080/14787210.2020.1776115. [DOI] [PubMed] [Google Scholar]
- 97.Miyako E, Nagata H, Hirano K, Sakamoto K, Makita Y, Nakayama KI, Hirotsu T. Photoinduced antiviral carbon nanohorns. Nanotechnology. 2008;19(7):6. doi: 10.1088/0957-4484/19/7/075106. [DOI] [PubMed] [Google Scholar]
- 98.Banerjee I, Douaisi MP, Mondal D, Kane RS. Light-activated nanotube–porphyrin conjugates as effective antiviral agents. Nanotechnology. 2012;23(10):105101. doi: 10.1088/0957-4484/23/10/105101. [DOI] [PubMed] [Google Scholar]
- 99.Schneider JE, Tabatabaie T, Maidt L, Smith RH, Nguyen X, Pye Q, Floyd RA. Potential mechanisms of photodynamic inactivation of virus by methylene blue I. RNA–protein crosslinks and other oxidative lesions in Qβ Bacteriophage. Photochem Photobiol. 1998;67(3):350–357. doi: 10.1111/j.1751-1097.1998.tb05209.x. [DOI] [PubMed] [Google Scholar]
- 100.Bhardwaj P, Bhatia E, Sharma S, Ahamad N, Banerjee R. Advancements in prophylactic and therapeutic nanovaccines. Acta Biomater. 2020;108:1–21. doi: 10.1016/j.actbio.2020.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Gilca V, Sauvageau C, Boulianne N, de Serres G, Couillard M, Krajden M, Ouakki M, Murphy D, Trevisan A, Dionne M. Immunogenicity of quadrivalent HPV and combined hepatitis A and B vaccine when co-administered or administered one month apart to 9-10 year-old girls according to 0-6 month schedule. Hum Vaccine Immunother. 2014;10(8):2438–2445. doi: 10.4161/hv.29617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Singh R, Upadhyay SK, Singh M, Sharma I, Sharma P, Kamboj P, Saini A, Voraha R, Sharma AK, Upadhyay TK, Khan F. Chitin, chitinases and chitin derivatives in biopharmaceutical, agricultural and environmental perspective. Biointerface Res Appl Chem. 2021;11(3):9985–10005. doi: 10.33263/BRIAC113.998510005. [DOI] [Google Scholar]
- 103.Mortola E, Roy P. Efficient assembly and release of SARS coronavirus-like particles by a heterologous expression system. FEBS Lett. 2004;576(1-2):174–178. doi: 10.1016/j.febslet.2004.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wang C, Zheng X, Gai W, Zhao Y, Wang H, Wang H, Feng N, Chi H, Qiu B, Li N, Wang T, Gao Y, Yang S, Xia X. MERS-CoV virus-like particles produced in insect cells induce specific humoural and cellular imminity in rhesus macaques. Oncotarget. 2017;8(8):12686–12694. doi: 10.18632/oncotarget.8475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ho Y, Lin PH, Liu CY, Lee SP, Chao YC. Assembly of human severe acute respiratory syndrome coronavirus-like particles. Biochem Biophys Res Commun. 2004;318(4):833–838. doi: 10.1016/j.bbrc.2004.04.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lu X, Chen Y, Bai B, Hu H, Tao L, Yang J, Chen J, Chen Z, Hu Z, Wang H. Immune responses against severe acute respiratory syndrome coronavirus induced by virus-like particles in mice. Immunology. 2007;122(4):496–502. doi: 10.1111/j.1365-2567.2007.02676.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Bai B, Hu Q, Hu H, Zhou P, Shi Z, Meng J, Lu B, Huang Y, Mao P, Wang H. Virus-like particles of SARS-like coronavirus formed by membrane proteins from different origins demonstrate stimulating activity in human dendritic cells. PLoS One. 2008;3(7):e2685. doi: 10.1371/journal.pone.0002685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Lu B, Huang Y, Huang L, Li B, Zheng Z, Chen Z, Chen J, Hu Q, Wang H. Effect of mucosal and systemic immunization with virus-like particles of severe acute respiratory syndrome coronavirus in mice. Immunology. 2010;130(2):254–261. doi: 10.1111/j.1365-2567.2010.03231.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu YV, Massare MJ, Barnard DL, Kort T, Nathan M, Wang L, Smith G. Chimeric severe acute respiratory syndrome coronavirus (SARS-CoV) S glycoprotein and influenza matrix 1 efficiently form virus-like particles (VLPs) that protect mice against challenge with SARS-CoV. Vaccine. 2011;29(38):6606–6613. doi: 10.1016/j.vaccine.2011.06.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Schijns V, Fernandez-Tejada A, Barjaktarovic Z, Bouzalas I, et al. Modulation of immune responses using adjuvants to facilitate therapeutic vaccination. Immunol Rev. 2020;296:169–190. doi: 10.1111/imr.12889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kato T, Takami Y, Kumar Deo V, Park EY. Preparation of virus-like particle mimetic nanovesicles displaying the S protein of Middle East respiratory syndrome coronavirus using insect cells. J Biotechnol. 2019;306:177–184. doi: 10.1016/j.jbiotec.2019.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Garg S, Aggarwal D, Upadhyay SK, Kumar G, Singh G. Effect of COVID-19 on school education system: Challenges and opportunities to adopt online teaching and learning. Hum Soc Sci Rev. 2020;8(6):10–17. doi: 10.18510/hssr.2020.862. [DOI] [Google Scholar]