Abstract
Background.
There is widespread interest in discriminating indolent from aggressive ductal carcinoma in situ (DCIS). We sought to evaluate collagen organization in the DCIS tumor microenvironment in relation to pathologic characteristics and patient outcomes.
Methods.
We retrieved fixed tissue specimens for 90 DCIS cases within the population-based Vermont DCIS Cohort. We imaged collagen fibers within 75 μm of the tumor/stromal boundary on H&E-stained slides using multiphoton microscopy with second harmonic generation. Automated software quantified collagen fiber length, width, straightness, density, alignment, and angle to the tumor/stroma boundary. Factor analysis identified linear combinations of collagen fiber features representing composite attributes of collagen organization.
Results.
Multiple collagen features were associated with DCIS grade, necrosis pattern, or periductal fibrosis (P<0.05). After adjusting for treatments and nuclear grade, risk of recurrence (defined as any second breast cancer diagnosis) was lower among cases with greater collagen fiber width (hazard ratio [HR]=0.57 per one standard deviation increase; 95% CI:0.39-0.84) and fiber density (HR=0.60; 95% CI:0.42-0.85), while risk was elevated among DCIS cases with higher fiber straightness (HR=1.47; 95% CI:1.05-2.06) and distance to the nearest two fibers (HR=1.47; 95% CI:1.06-2.02). Fiber length, alignment, and fiber angle were not associated with recurrence (P>0.05). Five composite factors were identified, accounting for 72.4% of the total variability among fibers; three were inversely associated with recurrence (HRs ranging from 0.60-0.67; P ≤ 0.01).
Conclusions.
Multiple aspects of collagen organization around DCIS lesions are associated with recurrence risk.
Impact.
Collagen organization should be considered in the development of prognostic DCIS biomarker signatures.
Keywords: Breast cancer, Ductal carcinoma in situ, DCIS, collagen, second harmonic generation
INTRODUCTION
Ductal carcinoma in situ (DCIS) is considered the earliest form of breast cancer, in which malignant cells are confined within the basement membrane of the breast duct system (1). DCIS is typically detected by mammography and accounts for >25% of screen-detected breast cancers (2). DCIS is considered a non-obligate precursor to invasive breast cancer (3). Due to uncertainty in the natural history of DCIS (4-10), there is widespread concern regarding overtreatment (11). It is not currently possible to determine which DCIS lesions are likely to progress to a potentially lethal invasive stage; thus, most women diagnosed with DCIS undergo surgical, radiation, and endocrine hormone therapies similar to women diagnosed with invasive breast cancer (12).
Identification of DCIS prognostic markers could permit personalized treatment strategies that spare women unnecessary treatments (3). Most prior research on DCIS biomarkers has focused on the malignant epithelial cancer cells: their genetic characteristics, protein- and RNA-expression, morphology, and growth patterns (13-15). Chromosomal abnormalities in DCIS are remarkably similar to invasive carcinoma (16). This suggests other factors are involved in DCIS progression.
Several lines of evidence suggest that the surrounding stromal tissue is an active and necessary accomplice in promoting invasion (17-21). Collagen is a major component of the stromal tissue that surrounds breast ducts. Laboratory studies have shown that fibrillar collagen plays a key role in promoting tumor initiation and metastasis (22). Prior studies have suggested that collagen fiber orientation and alignment are prognostic markers in invasive breast cancer patients (23, 24). We previously found that collagen fiber alignment in DCIS specimens was associated with poor prognosis pathology characteristics, but not with recurrence (1). To our knowledge, other aspects of collagen organization have not been evaluated in DCIS specimens. We sought to comprehensively characterize collagen structural organization around DCIS lesions and evaluate its association with DCIS pathology and outcomes within a well-characterized population-based cohort.
METHODS
Study population
The Vermont DCIS cohort (25) includes 1,252 women diagnosed with DCIS in Vermont during 1994-2012, identified via the Vermont Breast Cancer Surveillance System (VBCSS). The VBCSS is one of six active breast imaging registries within the Breast Cancer Surveillance Consortium (26, 27) and collects breast imaging data from all radiology facilities in Vermont, linked to pathology records and the Vermont Cancer Registry (28, 29). This study was approved by the University of Vermont Institutional Review Board with a waiver of consent.
The Vermont DCIS cohort includes women diagnosed with DCIS as a first primary breast cancer (25). Women with a concurrent invasive breast cancer diagnosis are excluded. For this study, a roster of 148 DCIS cases was initially identified from the Vermont DCIS Cohort. This included 74 patients who had experienced a second event (defined as any second breast cancer diagnosis (in situ or invasive, ipsilateral or contralateral) occurring at least 6 months after the initial DCIS diagnosis and 74 who had not experienced a second event. Ultimately, adequate tissue specimens for collagen fiber analysis were available for 90 cases, including 51 who experienced a second event and 39 who had not (Table 1). Reasons for unavailability of tissue specimens included: pathology facility no longer retained the tissue specimen (e.g., for cases >15 years old at certain facilities); no DCIS remained in the tissue specimen; or the case was released for other research and paraffin blocks could not be located.
Table 1.
Characteristics of study participants from the Vermont DCIS Cohort.
| Characteristics | N | % |
|---|---|---|
| Age at diagnosis | ||
| <50 | 36 | 40.0 |
| 50-69 | 40 | 44.4 |
| 70+ | 14 | 15.6 |
| Mode of detection | ||
| Screening | 62 | 68.9 |
| Symptoms | 13 | 14.4 |
| Missing | 15 | 16.7 |
| Treatment | ||
| Mastectomy | 5 | 5.6 |
| BCS only | 38 | 42.2 |
| BCS & radiation therapy | 26 | 28.9 |
| BCS & endocrine therapy | 9 | 10.0 |
| BCS & radiation & endocrine therapy | 12 | 13.3 |
| Recurrence | ||
| No | 39 | 43.3 |
| Yes – Ipsilateral | 38 | 42.2 |
| Yes – Contralateral | 12 | 13.3 |
| Yes – Bilateral | 1 | 1.1 |
| Type of Recurrence | ||
| None | 39 | 43.3 |
| DCIS | 25 | 27.8 |
| Invasive breast cancer | 26 | 28.9 |
BCS, breast conserving surgery
Clinical data
All participants completed a questionnaire at breast imaging visits, providing information on demographics and treatment information. Radiological exam results reported by the radiology facility included indication (i.e., screening vs. diagnostic) and assessments, which were used to determine DCIS method of detection (30). Pathology reports for all breast specimens evaluated at pathology facilities in Vermont were linked to study participants, with standardized data abstracted on specimen type and histology. Pathology slides and blocks for the index DCIS were reviewed centrally by a single pathologist (DLW) to confirm the diagnosis and record standardized data on nuclear grade, necrosis, inflammation, and periductal fibrosis. Among the 73 cases with grade available from clinical pathology reports, there was 81% agreement with grade from the central review. Participants were also linked to the Vermont Cancer Registry, providing consolidated information on stage at diagnosis and treatment data.
Treatment was categorized based on the clinical questionnaire, pathology record, and cancer registry data, as previously described (25). Data from subsequent pathology reports and the Vermont Cancer Registry were used to identify second breast cancer diagnoses occurring ≥6 months after the index DCIS diagnosis. Second events included DCIS or invasive breast cancer diagnosed in the ipsilateral or contralateral breast.
Collagen assessment
The study pathologist (DLW) identified a representative tumor block to re-cut an H&E stained slide for collagen analysis. The slide was digitized and annotated by the pathologist to identify areas of DCIS. Specific imaging locations within these relatively large areas were chosen by the microscopist (MWC) based on the criteria that they include adequate stromal collagen that could be clearly ascribed to a specific DCIS tumor/stromal boundary, without interference from blood vessels, fatty areas, or non-annotated areas of epithelium. The microscopist was blinded to the cases’ recurrence status. One to five 770 μm x 770 μm locations were imaged per case, with 91% of cases having ≥3 locations imaged. Second-harmonic generation (SHG) on a multiphoton microscope (see detailed imaging protocol in the Supplement) was used to image collagen fibers. The frequency doubling of the incident laser light caused by the unique tertiary structure of collagen allows for this, producing an image of only collagen fibers (Figure 1). In order to alleviate out of focal plane issues due to the unevenness of the tissue slice, 3 z-planes were captured per SHG image and then maximum-intensity projected to capture the entire axial field of view. The open-source image analysis software packages CT-FIRE and CurveAlign were used to quantify collagen fiber features, as previously described (24, 31, 32). CT-Fire determined the total length (i.e., following the contour of the fiber), end-to-end length (i.e., straight distance between one end of the fiber to the other end), and width of each collagen fiber in the SHG image. The ratio of end-to-end length to total length was computed as a measure of straightness, and the difference between total and end-to-end length was computed as another measure of the fiber’s curviness. CurveAlign measured fiber alignment, fiber density, distance to the nearest two fibers, and the angle of fibers with respect to the tumor/stromal boundary, as previously described (33). Briefly, fiber alignment (i.e., anisotropy) is calculated as coefficients ranging from 0 to 1, with 1 indicating perfectly aligned fibers and 0 representing randomly oriented fibers. Fiber density was defined on three different scales: counts of fibers within a 24 μm, 48 μm, or 96 μm square box centered on the fiber centroid. All collagen measures used in analyses were restricted to fibers within 100 pixels (75.19 μm) from the tumor/stromal boundary. A brief summary of the definitions of each collagen feature used in the study is provided in Supplementary Table S1. A detailed description of the computational methods used to make these measurements, along with a validation of their accuracy using a synthetic test set of fibers has been previously published (34).
Figure 1.
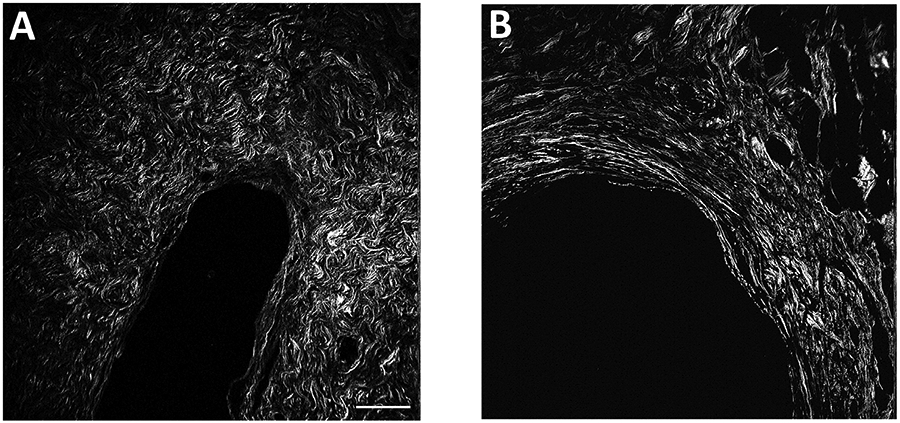
Sample multiphoton microscopy images of collagen fibers, illustrating cases with different collagen features. Panel A shows an area of DCIS surrounded by short, curvy, dense fibers. Panel B shows an area of DCIS surrounded with long, straight, high alignment fibers. Scale bar, 100 μm.
Statistical analysis
Summary statistics were used to describe the distribution of collagen features of individual fibers across imaged locations and their mean values for each woman were computed. Factor analysis was performed on a set of 11 collagen features of each fiber to identify linear combinations that account for the covariance among features and potentially represent composite attributes of collagen organization. The principal component factoring method was used, and five factors with eigenvalues greater than 1.0 were retained. Varimax rotation was used to obtain orthogonal factors. Values of the five factors were computed for each fiber, and the mean value determined across all fibers from each woman. Associations of collagen features and factor scores with DCIS pathology characteristics were examined by ANOVA. Cox regression was used to assess associations of collagen features and factor scores with time to occurrence of a second breast cancer event, adjusted for nuclear grade and treatment. In sensitivity analyses we adjusted for additional variables, including age, menopausal status, body mass index, breast density, method of detection, and surgical margin status. Disease-free survival was defined as the time from diagnosis to a second breast cancer diagnosis, with censoring at the date of the last VBCSS breast imaging or pathology record. All statistical analyses were performed in SAS Version 9.4 (SAS Institute Inc., Cary, NC), all tests were two-sided, and P values ≤0.05 were considered statistically significant.
RESULTS
Forty percent of the participants were under age 50 at diagnosis, and 42% were treated only with breast-conserving surgery (Table 1). The majority of second events (74.5%) occurred in the ipsilateral breast, and approximately half of second breast cancer diagnoses were invasive. The mean time to second event was 5.4 years (range, 0.5-15.3 years); mean follow-up time among cases without a second event was 12.7 years (range, 2.4-20.9 years).
Collagen fiber features
37,523 fibers were identified among the 90 imaged DCIS lesions. All collagen features were highly variable except fiber straightness, which is a ratio of two highly correlated measures and had a coefficient of variation (CV) across all fibers and women of 5.5%. CVs for all other collagen features ranged from 19% to 92% (Supplemental Table S2). For all features, differences between fibers within an imaged location accounted for most of the variability (88.1% to 98.6% of the total variance). Differences between locations within a woman’s DCIS sample contributed 0.6% to 9.5% to the total variance, while differences between women contributed 0.2% to 5.3%. Despite the between-fiber variation in individual features, differing patterns of collagen organization were evident among women (Figure 1).
Four fiber features were inversely related to DCIS nuclear grade and necrosis pattern: total minus end-to-end length, width, box density 48×48μm, and box density 96×96 μm (Table 2). Distance to the nearest two fibers was positively associated with nuclear grade and necrosis pattern. Straightness, box density 24×24 μm, alignment of the nearest 4 fibers, and angle to the tumor/stromal boundary were associated with necrosis pattern but not nuclear grade. The presence of inflammation was associated with a smaller difference between fiber total length and end-to-end length, greater straightness, more narrow fiber width, and higher alignment to the nearest four fibers. Only total minus end-to-end fiber length was significantly related to periductal fibrosis, with lower values observed for DCIS lesions having concentric cellular fibrosis.
Table 2.
Association between collagen features and DCIS pathology characteristics, Vermont DCIS Cohort.
| N | Total length (μm) |
Total minus end-to-end length (μm) |
aStraightness | Width (μm) | Distance to nearest 2 fibers (μm) |
bBox density 24×24 μm |
bBox density 48×48 μm |
bBox density 96×96 μm |
Alignment of nearest 4 |
cBox alignment 24×24 μm |
Distance to tumor/stromal boundary (μm) |
Angle to tumor/stromal boundary (°) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nuclear Grade | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | Mean (SE) | |
| Low | 7 | 41.63 (0.60) | 3.55 (0.11) | 0.916 (0.002) | 3.33 (0.09) | 21.06 (0.68) | 1.41 (0.04) | 3.30 (0.19) | 10.62 (0.79) | 0.705 (0.025) | 0.918 (0.012) | 38.82 (0.83) | 25.67 (1.82) |
| Intermediate | 45 | 42.54 (0.45) | 3.53 (0.05) | 0.918 (0.001) | 3.20 (0.02) | 21.67 (0.30) | 1.39 (0.01) | 3.19 (0.06) | 10.21 (0.23) | 0.738 (0.009) | 0.928 (0.004) | 37.93 (0.30) | 23.25 (0.61) |
| High | 36 | 41.98 (0.53) | 3.37 (0.05) | 0.920 (0.001) | 3.08 (0.02) | 23.17 (0.38) | 1.36 (0.01) | 2.96 (0.06) | 9.19 (0.25) | 0.756 (0.010) | 0.935 (0.005) | 38.13 (0.30) | 22.67 (0.69) |
| p = 0.62 | p = 0.02 | p =0.18 | p < .001 | P = 0.007 | p = 0.17 | p =0 .01 | p = 0.01 | p =0.10 | p = 0.31 | p = 0.59 | p = 0.23 | ||
| Necrosis | |||||||||||||
| Absent | 25 | 42.64 (0.60) | 3.57 (0.05) | 0.918 (0.001) | 3.28 (0.04) | 21.21 (0.38) | 1.42 (0.02) | 3.30 (0.08) | 10.60 (0.34) | 0.724 (0.014) | 0.920 (0.008) | 38.67 (0.45) | 24.57 (0.95) |
| Focal | 21 | 40.95 (0.60) | 3.52 (0.07) | 0.915 (0.002) | 3.14 (0.04) | 21.98 (0.45) | 1.38 (0.02) | 3.13 (0.08) | 10.00 (0.34) | 0.718 (0.013) | 0.928 (0.006) | 37.85 (0.38) | 24.41 (0.88) |
| Central necrosis | 41 | 42.61 (0.45) | 3.38 (0.04) | 0.921 (0.001) | 3.10 (0.02) | 23.03 (0.38) | 1.36 (0.01) | 2.97 (0.05) | 9.25 (0.23) | 0.767 (0.007) | 0.937 (0.004) | 37.84 (0.30) | 21.79 (0.55) |
| p = 0.10 | p = 0.01 | p = 0.003 | p < .001 | p = .009 | p =0 .03 | p = 0.003 | p = 0.004 | p = 0.002 | p = 0.07 | p = 0.27 | p = 0.01 | ||
| Inflammation | |||||||||||||
| Present | 32 | 42.57 (0.53) | 3.37 (0.05) | 0.921 (0.001) | 3.11 (0.03) | 22.72 (0.38) | 1.37 (0.01) | 3.04 (0.07) | 9.54 (0.27) | 0.760 (0.011) | 0.936 (0.005) | 38.17 (0.30) | 22.67 (0.80) |
| Absent | 56 | 42.05 (0.38) | 3.53 (0.04) | 0.917 (0.001) | 3.19 (0.02) | 21.96 (0.30) | 1.39 (0.01) | 3.14 (0.05) | 9.99 (0.22) | 0.733 (0.008) | 0.927 (0.004) | 38.04 (0.23) | 23.51 (0.54) |
| p = 0.45 | p = 0 .01 | p = 0.01 | p = 0.05 | P = 0.15 | p = 0.29 | p = 0.24 | p = 0.21 | p = 0.04 | p = 0.19 | p = 0.78 | p = 0.37 | ||
| Periductal fibrosis | |||||||||||||
| No | 39 | 42.55 (0.53) | 3.50 (0.05) | 0.919 (0.001) | 3.19 (0.03) | 22.37 (0.38) | 1.37 (0.02) | 3.07 (0.07) | 9.74 (0.30) | 0.743 (0.011) | 0.933 (0.006) | 37.78 (0.30) | 23.08 (0.70) |
| Yes-concentric/not cellular | 6 | 42.41 (0.75) | 3.65 (0.13) | 0.914 (0.002) | 3.15 (0.05) | 21.71 (0.98) | 1.42 (0.02) | 3.16 (0.15) | 10.12 (0.63) | 0.742 (0.011) | 0.920 (0.005) | 38.41 (0.60) | 22.95 (1.57) |
| Yes-concentric/cellular | 31 | 41.84 (0.45) | 3.36 (0.05) | 0.920 (0.001) | 3.11 (0.02) | 22.57 (0.38) | 1.37 (0.01) | 3.06 (0.05) | 9.59 (0.24) | 0.753 (0.010) | 0.932 (0.005) | 38.38 (0.30) | 22.90 (0.80) |
| dYes-other | 10 | 42.52 (0.68) | 3.62 (0.06) | 0.917 (0.002) | 3.19 (0.06) | 21.37 (0.60) | 1.40 (0.03) | 3.28 (0.10) | 10.37 (0.39) | 0.723 (0.015) | 0.922 (0.008) | 38.08 (0.68) | 24.00 (1.04) |
| p = 0.81 | p = 0.01 | p = 0.16 | p = 0.24 | P = 0.53 | p = 0.61 | p = 0.39 | p = 0.56 | p = 0.62 | p = 0.60 | p = 0.69 | p = 0.92 |
SE, standard error. P-values were computed from ANOVA tests of whether the mean collagen feature differed among groups based on the DCIS pathology characteristic. P-values less than 0.05 were considered statistically significant and are indicated in bold font.
Straightness is defined as the ratio of the end-to-end length to the total length of the fiber.
Box density refers to the number of collagen fibers contained within box of the specified size overlaid on the fiber centroid.
Alignment of fibers contained within a 24×24 μm box overlaid on the fiber centroid.
Includes periductal fibrosis that is neither cellular nor concentric, as well as periductal fibrosis with unknown concentricity/cellularity.
Composite measures
Factor analysis identified five linear combinations of 11 collagen features (factors) that accounted for 72.4% of the total variability of the features among fibers. The standardized scoring coefficients for computing factor scores are shown in Table 3 and provide insight into the collagen attribute being measured by indicating the relative strength of the correlation (loading) between each fiber feature and factor score. These relationships are also summarized in Supplemental Table S1. Factor 1 loaded positively on box density 24×24 μm and negatively on box alignment 24×24 μm, such that cases with high values of Factor 1 had localized areas of collagen with a high density of non-aligned fibers. Factor 2 loaded most highly on box density 96×96 μm and distance from the tumor/stromal boundary, indicating areas of dense collagen closer to the outer boundary of the area measured (75.19 μm) from the tumor/stromal boundary. Total minus end-to-end fiber length and straightness loaded positively and negatively on Factor 3, respectively, indicative of curved or serpentine fibers. Factor 4 loaded positively on alignment of the nearest four fibers and negatively on angle to the tumor/stromal boundary, indicating the presence of aligned fibers that tended to be parallel to the tumor boundary. Factor 5 loaded most heavily on fiber length and width.
Table 3.
Standardized scoring coefficients for computing composite measures identified from factor analysis, Vermont DCIS Cohort.
| Factor 1 | Factor 2 | Factor 3 | Factor 4 | Factor 5 | |
|---|---|---|---|---|---|
| Total length | 0.048 | −0.143 | 0.195 | 0.117 | 0.482 |
| Total minus end-to-end length | 0.006 | −0.026 | 0.489 | 0.005 | 0.092 |
| aStraightness | 0.038 | −0.087 | −0.487 | 0.077 | 0.291 |
| Width (μm) | −0.071 | 0.083 | −0.141 | −0.137 | 0.675 |
| Distance to nearest 2 fibers | −0.244 | −0.197 | 0.007 | −0.190 | 0.025 |
| bBox density 24×24 μm | 0.478 | −0.139 | 0.011 | 0.051 | −0.034 |
| bBox density 48×48 μm | 0.158 | 0.270 | −0.010 | 0.130 | −0.035 |
| bBox density 96×96 μm | −0.055 | 0.424 | 0.001 | 0.043 | 0.058 |
| Alignment of the nearest 4 fibers | 0.004 | 0.103 | −0.012 | 0.591 | −0.220 |
| cBox alignment 24×24 μm | −0.424 | 0.226 | 0.007 | 0.236 | −0.035 |
| Distance to tumor/stromal boundary (μm) | −0.286 | 0.438 | 0.031 | −0.162 | −0.040 |
| Angle relative to tumor/stromal boundary (°) | −0.046 | 0.052 | 0.023 | −0.479 | −0.119 |
Bold font is used to highlight collagen features with the highest coefficients (absolute value >0.4).
Straightness is defined as the ratio of the end-to-end length to the total length of the fiber.
Box density refers to the number of collagen fibers contained within a box of the specified size overlaid on the fiber centroid.
Alignment of fibers contained within a 24×24 μm box overlaid on the fiber centroid.
Mean values of factors 2 and 5, averaged across all fibers for each woman, had inverse relationships with nuclear grade (Table 4). All five factors were associated with necrosis patterns. Only Factor 3 was significantly associated with inflammation and periductal fibrosis.
Table 4.
Association of collagen composite factors with DCIS pathology characteristics, Vermont DCIS Cohort.
| Mean Factor Value (SE) | ||||||
|---|---|---|---|---|---|---|
| N | Factor 1 (high density, low alignment) |
Factor 2 (high density, long distance to tumor/stromal boundary) |
Factor 3 (Serpentine, not straight) |
Factor 4 (high alignment, parallel orientation to tumor boundary) |
Factor 5 (Long, wide fibers) |
|
| Nuclear Grade | ||||||
| Low | 7 | 0.27 (0.35) | 0.53 (0.45) | −0.04 (0.45) | −0.62 (0.42) | 0.82 (0.48) |
| Intermediate | 45 | 0.15 (0.14) | 0.20 (0.14) | 0.13 (0.15) | 0.02 (0.15) | 0.23 (0.15) |
| High | 36 | −0.28 (0.16) | −0.38 (0.15) | −0.18 (0.16) | 0.15 (0.16) | −0.46 (0.13) |
| p = 0.10 | p = 0.01 | p = 0.36 | p = 0.18 | p < .001 | ||
| Necrosis | ||||||
| Absent | 25 | 0.35 (0.22) | 0.47 (0.20) | 0.08 (0.19) | −0.25 (0.23) | 0.64 (0.20) |
| Focal | 21 | 0.04 (0.19) | 0.07 (0.20) | 0.41 (0.22) | −0.32 (0.22) | −0.24 (0.21) |
| Central necrosis | 41 | −0.27 (0.14) | −0.35 (0.14) | −0.27 (0.14) | 0.36 (0.12) | −0.32 (0.13) |
| p = 0.04 | p = 0.004 | p = 0.03 | p = 0.01 | p < .001 | ||
| Inflammation | ||||||
| Present | 32 | −0.19 (0.16) | −0.17 (0.17) | −0.30 (0.16) | 0.25 (0.18) | −0.24 (0.16) |
| Absent | 56 | 0.08 (0.14) | 0.08 (0.14) | 0.16 (0.13) | −0.11 (0.13) | 0.12 (0.14) |
| p = 0.21 | p = 0.26 | p = 0.03 | p = 0.10 | p = 0.10 | ||
| Periductal fibrosis | ||||||
| No | 39 | −0.11 (0.18) | −0.07 (0.18) | 0.00 (0.16) | 0.01 (0.18) | 0.17 (0.19) |
| Yes, concentric, not cellular | 6 | 0.32 (0.30) | 0.13 (0.39) | 0.80 (0.34) | 0.05 (0.23) | −0.13 (0.28) |
| Yes, concentric and cellular | 31 | −0.11 (0.13) | −0.12 (0.15) | −0.29 (0.16) | 0.14 (0.18) | −0.33 (0.14) |
| aYes, other | 10 | 0.33 (0.28) | 0.30 (0.25) | 0.40 (0.21) | −0.16 (0.25) | 0.21 (0.33) |
| p = 0.46 | p = 0.66 | p = 0.03 | p = 0.87 | p = 0.19 | ||
SE, standard error. P-values were computed from ANOVA tests of whether the mean collagen composite factor differed among groups based on the DCIS pathology characteristic. P-values less than 0.05 were considered statistically significant and are indicated in bold font.
Includes periductal fibrosis that is neither cellular nor concentric, as well as periductal fibrosis with unknown concentricity/cellularity.
Associations with risk of recurrence
Risk of a second breast cancer event was related to several fiber features as well as three of the factors (Table 5). After adjusting for treatments and nuclear grade, risk of any second event was lower among cases with greater collagen fiber width (HR=0.57 per one standard deviation increase; 95% CI:0.39-0.84) and fiber density within a 96 μm x 96 μm box (HR=0.60; 95% CI:0.42-0.85), while risk was elevated among DCIS cases with higher fiber straightness (HR=1.47; 95% CI:1.05-2.06) and distance to the nearest two fibers (HR=1.47; 95% CI:1.06-2.02). Total minus end-to-end length was also associated with reduced risk (HR=0.55; 95% CI:0.39-0.76). Fiber length, alignment, and angle relative to the tumor-stroma boundary were not associated with recurrence (P>0.05). Factors 2, 3, and 5 were associated with reduced risk of any second breast cancer event (HRs ranging from 0.60-0.67). Hazard ratios for ipsilateral second events were similar to those for any second event. Adjustment for additional covariates had little influence on the results (Supplemental Table S3). Collagen factors were also associated with risk of contralateral second events (Supplemental Table S4).
Table 5.
Associations between collagen organization and risk of recurrence, Vermont DCIS Cohort. All hazard ratios correspond to an increase of one standard deviation in the collagen measure.
| Any second event | Ipsilateral second event | |||||||
|---|---|---|---|---|---|---|---|---|
| Adjusted for treatment | Adjusted for treatment and grade | Adjusted for treatment | Adjusted for treatment and grade | |||||
| HR (95% CI) | p-value | HR (95% CI) | p-value | HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Directly measured collagen features | ||||||||
| Total length | 0.76 (0.54 - 1.06) | 0.10 | 0.77 (0.55 - 1.08) | 0.13 | 0.74 (0.50 - 1.10) | 0.14 | 0.74 (0.50 - 1.10) | 0.13 |
| Total minus end-to-end length | 0.51 (0.36 - 0.71) | <.001 | 0.55 (0.39 - 0.76) | <.001 | 0.53 (0.37 - 0.76) | <.001 | 0.57 (0.39 - 0.82) | 0.003 |
| aStraightness | 1.52 (1.09 - 2.10) | 0.01 | 1.47 (1.05 - 2.06) | 0.03 | 1.54 (1.05 - 2.24) | 0.03 | 1.49 (1.01 - 2.21) | 0.05 |
| Width | 0.53 (0.37 - 0.76) | <.001 | 0.57 (0.39 - 0.84) | 0.01 | 0.50 (0.32 - 0.78) | 0.002 | 0.54 (0.34 - 0.86) | 0.01 |
| Distance to nearest 2 fibers | 1.57 (1.15 – 2.14) | .005 | 1.47 (1.06 - 2.02) | 0.02 | 1.82 (1.22 – 2.73) | 0.004 | 1.70 (1.14 – 2.54) | 0.009 |
| bBox density 24×24 μm | 0.68 (0.48 - 0.95) | 0.03 | 0.73 (0.52 - 1.04) | 0.08 | 0.69 (0.46 - 1.02) | 0.06 | 0.74 (0.50 - 1.09) | 0.13 |
| bBox density 48×48 μm | 0.55 (0.39 - 0.78) | <.001 | 0.58 (0.41 - 0.83) | 0.003 | 0.55 (0.36 - 0.82) | 0.004 | 0.58 (0.38 - 0.88) | 0.01 |
| bBox density 96×96 μm | 0.57 (0.41 - 0.79) | <.001 | 0.60 (0.42 - 0.85) | 0.004 | 0.55 (0.37 - 0.82) | 0.003 | 0.57 (0.38 - 0.88) | 0.01 |
| Alignment of the nearest 4 fibers | 1.07 (0.78 - 1.46) | 0.68 | 0.94 (0.67 - 1.33) | 0.73 | 1.14 (0.79 - 1.64) | 0.48 | 1.03 (0.69 - 1.54) | 0.88 |
| cBox alignment 24×24 μm | 1.19 (0.86 - 1.65) | 0.30 | 1.07 (0.77 - 1.51) | 0.68 | 1.27 (0.88 - 1.84) | 0.21 | 1.17 (0.80 - 1.72) | 0.42 |
| Distance to tumor/stromal boundary | 0.84 (0.65 - 1.08) | 0.17 | 0.87 (0.67 - 1.13) | 0.29 | 0.87 (0.65 - 1.16) | 0.33 | 0.93 (0.69 - 1.27) | 0.66 |
| Angle relative to tumor/stromal boundary | 0.93 (0.67 - 1.29) | 0.66 | 1.07 (0.75 - 1.52) | 0.73 | 0.88 (0.59 - 1.30) | 0.52 | 1.01 (0.65 - 1.55) | 0.98 |
| Composite measuresd | ||||||||
| Factor 1 (high density, low alignment) | 0.72 (0.52 - 1.00) | 0.05 | 0.78 (0.56 - 1.09) | 0.14 | 0.68 (0.46 - 1.00) | 0.05 | 0.73 (0.50 - 1.06) | 0.10 |
| Factor 2 (high density, long distance to tumor/stromal boundary) | 0.57 (0.42 - 0.78) | <0.001 | 0.60 (0.43 - 0.83) | 0.002 | 0.56 (0.39 - 0.82) | 0.003 | 0.59 (0.40 - 0.88) | 0.01 |
| Factor 3 (Serpentine, not straight) | 0.68 (0.51 - 0.90) | 0.01 | 0.67 (0.50 - 0.91) | 0.01 | 0.68 (0.50 - 0.94) | 0.02 | 0.68 (0.48 - 0.95) | 0.02 |
| Factor 4 (high alignment, parallel orientation to tumor boundary) | 0.99 (0.72 - 1.36) | 0.95 | 0.86 (0.61 - 1.21) | 0.38 | 1.05 (0.73 - 1.51) | 0.80 | 0.92 (0.62 - 1.37) | 0.68 |
| Factor 5 (Long, wide fibers) | 0.57 (0.41 - 0.81) | 0.002 | 0.62 (0.43 - 0.90) | 0.01 | 0.54 (0.36 - 0.82) | 0.004 | 0.58 (0.37 - 0.89) | 0.01 |
P-values less than 0.05 were considered statistically significant and are indicated in bold font.
Straightness is defined as the ratio of the end-to-end length to the total length of the fiber.
Box density refers to the number of collagen fibers contained within box of the specified size overlaid on the fiber centroid.
Alignment of fibers contained within a 24×24 μm box overlaid on the fiber centroid.
Composite measures are linear combinations of the directly measured collagen features identified by factor analysis; see text for details.
DISCUSSION
Our results demonstrate that numerous aspects of collagen organization around DCIS lesions are associated with DCIS pathologic characteristics and patient outcomes. Collagen fiber straightness, width, and density appear to be particularly relevant to prognosis. In addition, factor analysis identified five linear combinations of collagen fiber characteristics, representing more complex aspects of collagen organization, which were also associated with DCIS pathologic characteristics and patient outcomes. These results indicate that collagen organization should be further evaluated to provide novel insights regarding the biology of DCIS epithelial/stromal interactions and as a potential prognostic marker for breast cancer recurrence.
Previous studies of collagen organization in breast cancer have focused on collagen fiber alignment and orientation with respect to the tumor-stromal boundary. Early studies used ex-vivo imaging of intact, unfixed mouse mammary tumors to reveal cell invasion occurring along aligned collagen fibers radiating perpendicular to the tumor-stromal boundary (36). Tumor explants cultured in a randomly organized collagen matrix led to active realignment of the collagen and migration of individual tumor cells outward along radially aligned fibers (36). Likewise, when cells were seeded into microchannels filled with random or aligned polymerized collagen, cells migrated with greater directional persistence in the aligned collagen matrices (37). In a sample of 196 human invasive breast cancer biopsy specimens, a phenotype of perpendicularly aligned collagen fibers was identified that was associated with reduced disease-free survival (23). A subsequent study found that this phenotype did not have a statistically significant association with disease-free survival in a cohort of 371 invasive breast cancer cases, though it was associated with overall survival (24). The same study also found that collagen density in the tumor microenvironment was inversely related to risk of disease progression, consistent with our findings.
We are aware of two prior studies that have examined collagen in relation to DCIS specifically. Among 227 DCIS cases from the Wisconsin In Situ Cohort, greater collagen fiber angles were associated with negative hormone receptor status and presence of comedo necrosis but not with nuclear grade or disease-free survival (1). Among 776 DCIS cases in a UK cohort (38), investigators developed an image-based collagen prognostic index based on the orientation of collagen fibers relative to the DCIS boundary, fiber density, and alignment. A poor collagen prognostic index score – indicating more perpendicular fibers relative to the fiber boundary, greater fiber density, and higher alignment – was associated with higher nuclear grade, comedo type necrosis, hormonal receptor negativity, HER2 positivity, high proliferation index, and higher risk of recurrence. That study did not report results for fiber angle, density, and alignment individually; thus, direct comparison to our results is not possible.
As described above, prior studies have focused largely on fiber angle with respect to the tumor boundary (1, 23, 24, 38), which was not associated with outcomes in our study (nor in two of the three prior studies to directly evaluate fiber angle). We investigated a wide range of collagen fiber features. Our findings suggest that collagen fiber density is particularly relevant to DCIS pathology and outcomes. This is consistent with the Esbona et al. study of invasive breast cancer described above (24), though somewhat surprising based on prior ex-vivo mechanistic studies suggesting that collagen density was associated with increased tumor initiation and metastasis (22). This may be a reflection of differences in how collagen density was assessed, but also may indicate that the impact of collagen density may depend on other aspects of collagen organization (e.g., alignment of fibers). In addition to fiber density, fiber width, alignment, straightness, and total minus end-to-end length (another measure of straightness/curvature) were also associated with DCIS histopathology and outcomes in our study. The plasticity of the collagen matrix and its association with traditional DCIS prognostic factors (grade, necrosis) and the presence or absence inflammation appears to follow a trend where higher nuclear grade, central necrosis, and absent inflammation are associated with less curved, less dense, narrower fibers that are further apart from one another.
Our results are also consistent with laboratory studies investigating the role of collagen in mediating the association of parity with breast cancer risk (39, 40). Maller et al. showed that the abundance of collagen in the extracellular matrix from parous rats was associated with decreased tumor growth and reduced tumor cell invasion compared to ECM from nulliparous rats. Using methods including second harmonic generation imaging, the authors showed that abundant collagen in the mammary glands of parous rats is less linearized, and that high density collagen induces tumor suppressive attributes. These results have a striking consistency with our findings, indicating that linearized/straight collagen has tumor promoting properties whereas dense collagen is protective.
We used factor analysis to evaluate combinations of multiple collagen features. Factor identification did not use any information on recurrence, yet a number of these composite factors were indeed associated with recurrence. These factors could cluster DCIS cases into distinct phenotypes based on multiple aspects of their collagen organization. We did not identify any specific histopathologic patterns of periductal fibrosis associated with specific factors; however, some associations were plausibly explanatory. For example, one case with concentric, loosely packed wavy fibers reminiscent of unraveling or fraying rope had low scores for Factors 1, 2, and 5, a high score for Factor 4 (localized alignment) and an intermediate score for Factor 3 (curved, serpentine fibers). Further exploration of these factors may help to understand the patterns of collagen organization that are most relevant to DCIS epithelial/stromal biology and prediction of outcomes.
The biological mechanisms driving changes in collagen organization around breast cancer lesions remain unclear. Notably, our findings were robust to statistical adjustment for a number of patient characteristics, including age, menopausal status, and breast density, thereby suggesting that collagen organization is independently associated with DCIS outcomes. Future studies are required to directly evaluate a wide range of patient factors and molecular characteristics in relation to collagen organization around DCIS. Numerous pathways have been identified by which cancer cells can influence collagen synthesis and organization or by which collagen can influence cancer cell behavior (41, 42). Immunohistochemical staining of DCIS specimens has revealed variability in collagen deposition and the distribution of stromal cells in the tumor microenvironment (43). The role of carcinoma-associated fibroblasts (CAFs) is of particular interest as fibroblasts are important for producing collagen during mammary gland development and CAFs have been linked to breast cancer progression through various mechanisms (43). Other studies have identified stromal factors such as cyclooxygenase 2 (COX-2), CD68, and numerous extracellular matrix proteins that are correlated with collagen organization in breast cancer specimens (24, 44). Further research is needed to understand collagen-related signaling pathways involving both epithelial and stromal cells.
The multiphoton imaging approach used in our study allows for non-invasive and specific imaging of collagen fibers, and combined with quantitative image analysis tools, this provides a powerful method for assessing collagen organization (45). All fibrillar subtypes of collagen generate second harmonic signals, which comprise the majority of collagen isoforms with the notable exception of collagen type IV, a major constituent of the basement membrane. However, since the basement membrane represents a very small portion of the total collagen matrix imaged, and there are other collagen subtypes present in the basement membrane that are imaged, this exclusion should have minimal influence on our results.
We chose the risk of any subsequent event as the primary outcome in our study because women diagnosed with DCIS are concerned about their risk of any type of future breast cancer diagnosis (ipsilateral/contralateral, in situ/invasive). However, analyses of specific types of second events can inform our understanding of the biology and progression of DCIS related to collagen organization and could have treatment implications. We found similar risk associations for ipsilateral events as we did for the risk of any second event. Future studies with larger sample sizes are needed to specifically examine risk of ipsilateral invasive events and contralateral events separately.
Limitations of our study include the relatively small sample size, though it was adequate for the detection of statistically significant differences in outcomes. Data on hormone receptor and HER2 status was not available for the majority of cases, because these tests were not routinely performed clinically during the time period in which most of the index DCIS cases were diagnosed. The Vermont DCIS cohort is population-based but has very limited racial diversity. Validation of our findings in external study cohorts is needed to further support the development of a collagen signature that predicts DCIS outcomes. The ability of collagen signatures to add useful information to recurrence prediction models that include other known prognostic factors should be evaluated formally. Future studies of treated and untreated women are needed to better understand the nature of the association between collagen organization and DCIS outcomes. Trials of active surveillance for DCIS should offer unique opportunities for testing prognostic markers (46-48). It is also possible that collagen organization could influence recurrence risk as a moderator of treatment effectiveness (e.g., radiation or endocrine therapy).
Overall, our results demonstrate that multiple aspects of collagen organization around DCIS lesions, including collagen fiber straightness, width, and density, are associated with risk of recurrence. Collagen organization should be considered in the development of biomarker signatures to distinguish indolent from aggressive DCIS.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by the National Cancer Institute (grant number U01 CA196383 to B.L. Sprague and D.L. Weaver, U54 CA163303 to B.L. Sprague and D.L. Weaver, P01 CA154292 to B.L. Sprague, R01 CA199996 to M.W. Conklin, and P30 CA014520 to A. Trentham-Dietz); the Patient-Centered Outcomes Research Institute (grant number PCS-1504-30370 to B.L. Sprague); and the University of Vermont Cancer Center with funds generously awarded by the Lake Champlain Cancer Research Organization (pilot grant #032800 to B.L. Sprague). The collection of Vermont Cancer Registry data used in this study was supported by the Centers for Disease Control and Prevention (Cooperative Agreement No. NU58DP006322 to the Vermont State Agency for Human Services). None of the funding bodies had a role in the design of the study nor the collection, analysis, and interpretation of the data, nor the writing of the manuscript. The statements presented in this work are solely the responsibility of the authors and do not necessarily represent the official views of the National Cancer Institute, the National Institutes of Health, the Centers for Disease Control and Prevention, or PCORI, its Board of Governors or Methodology Committee. The authors wish to thank Drs. Andrew Goodwin, Brenda Waters, and Jill Warrington who participated in the centralized review of DCIS specimens; Alison Johnson and Jennifer Kachajian at the Vermont Cancer Registry; and Denis Nunez, Rachael Chicoine, John Mace, Mark Bowman, Mike Butler, Meghan Farrington, Cindy Groseclose, Kathleen Howe, Dawn Pelkey, Dusty Quick, and Tiffany Sharp who conduct and support data collection within the Vermont Breast Cancer Surveillance System. We also thank Drs. Yuming Liu, Bin Li and Jeremy Bredfeldt with technical support for SHG image analysis and image collection. The authors particularly would like to acknowledge the immeasurable contribution of the late Dr. Patti Keely to this project and for her impact on the field of the science of the tumor microenvironment and the role of collagen in tumor progression which motivated this study.
ABBREVIATIONS:
- BCS
breast conserving surgery
- DCIS
ductal carcinoma in situ
- HR
hazard ratio
- CI
confidence interval
- SE
standard error
- SHG
second harmonic generation
Footnotes
Author disclosures. Dr. Ted James reports being a scientific advisor for Perimeter Medical Imaging, Inc. The other authors have no disclosures to report.
REFERENCES
- 1.Conklin MW, Gangnon RE, Sprague BL, Van Gemert L, Hampton JM, Eliceiri KW, et al. Collagen Alignment as a Predictor of Recurrence after Ductal Carcinoma In Situ. Cancer Epidemiol Biomarkers Prev 2018;27(2):138–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Miglioretti DL, Zhu W, Kerlikowske K, Sprague BL, Onega T, Buist DS, et al. Breast Tumor Prognostic Characteristics and Biennial vs Annual Mammography, Age, and Menopausal Status. JAMA Oncol 2015;1(8):1069–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sprague BL, McLaughlin V, Hampton JM, Newcomb PA, Trentham-Dietz A. Disease-free survival by treatment after a DCIS diagnosis in a population-based cohort study. Breast cancer research and treatment 2013;141(1):145–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duffy SW, Agbaje O, Tabar L, Vitak B, Bjurstam N, Bjorneld L, et al. Overdiagnosis and overtreatment of breast cancer: estimates of overdiagnosis from two trials of mammographic screening for breast cancer. Breast Cancer Res 2005;7(6):258–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ernster VL, Barclay J. Increases in ductal carcinoma in situ (DCIS) of the breast in relation to mammography: a dilemma. J Natl Cancer Inst Monogr 1997, (22):151–6. [DOI] [PubMed] [Google Scholar]
- 6.Esserman L, Shieh Y, Thompson I. Rethinking screening for breast cancer and prostate cancer. JAMA 2009;302(15):1685–92. [DOI] [PubMed] [Google Scholar]
- 7.Betsill WL Jr., Rosen PP, Lieberman PH, Robbins GF. Intraductal Carcinoma: Long-Term Follow-up After Treatment by Biopsy Alone. JAMA 1978;239(18):1863–1867. [DOI] [PubMed] [Google Scholar]
- 8.Rosen PP, Braun DW Jr., Kinne DE. The clinical significance of pre-invasive breast carcinoma. Cancer 1980;46(4 Suppl):919–25. [DOI] [PubMed] [Google Scholar]
- 9.Page DL, Dupont WD, Rogers LW, Landenberger M. Intraductal carcinoma of the breast: follow-up after biopsy only. Cancer 1982;49(4):751–8. [DOI] [PubMed] [Google Scholar]
- 10.Eusebi V, Feudale E, Foschini MP, Micheli A, Conti A, Riva C, et al. Long-term follow-up of in situ carcinoma of the breast. Semin Diagn Pathol 1994;11(3):223–35. [PubMed] [Google Scholar]
- 11.Groen EJ, Elshof LE, Visser LL, Rutgers EJT, Winter-Warnars HAO, Lips EH, et al. Finding the balance between over- and under-treatment of ductal carcinoma in situ (DCIS). Breast 2017;31:274–283. [DOI] [PubMed] [Google Scholar]
- 12.Hwang ES, Esserman LJ. Management of ductal carcinoma in situ. Surg Clin North Am 1999;79(5):1007–30 viii. [DOI] [PubMed] [Google Scholar]
- 13.van de Vijver MJ, He YD, van’t Veer LJ, Dai H, Hart AA, Voskuil DW, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 2002;347(25):1999–2009. [DOI] [PubMed] [Google Scholar]
- 14.van ‘t Veer LJ, Dai H, van de Vijver MJ, He YD, Hart AA, Mao M, et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 2002;415(6871):530–6. [DOI] [PubMed] [Google Scholar]
- 15.Solin LJ, Gray R, Baehner FL, Butler SM, Hughes LL, Yoshizawa C, et al. A multigene expression assay to predict local recurrence risk for ductal carcinoma in situ of the breast. J Natl Cancer Inst 2013;105(10):701–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hwang ES, DeVries S, Chew KL, Moore DH 2nd, Kerlikowske K, Thor A, et al. Patterns of chromosomal alterations in breast ductal carcinoma in situ. Clin Cancer Res 2004;10(15):5160–7. [DOI] [PubMed] [Google Scholar]
- 17.Ronnov-Jessen L, Bissell MJ. Breast cancer by proxy: can the microenvironment be both the cause and consequence? Trends Mol Med 2009;15(1):5–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mueller MM, Fusenig NE. Friends or foes - bipolar effects of the tumour stroma in cancer. Nat Rev Cancer 2004;4(11):839–49. [DOI] [PubMed] [Google Scholar]
- 19.Bhowmick NA, Neilson EG, Moses HL. Stromal fibroblasts in cancer initiation and progression. Nature 2004;432(7015):332–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wiseman BS, Werb Z. Stromal effects on mammary gland development and breast cancer. Science 2002;296(5570):1046–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sgroi DC. Preinvasive breast cancer. Annu Rev Pathol 2010;5:193–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Provenzano PP, Inman DR, Eliceiri KW, Knittel JG, Yan L, Rueden CT, et al. Collagen density promotes mammary tumor initiation and progression. BMC Med 2008;6:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Conklin MW, Eickhoff JC, Riching KM, Pehlke CA, Eliceiri KW, Provenzano PP, et al. Aligned collagen is a prognostic signature for survival in human breast carcinoma. Am J Pathol 2011;178(3):1221–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Esbona K, Yi Y, Saha S, Yu M, Van Doorn RR, Conklin MW, et al. The Presence of Cyclooxygenase 2, Tumor-Associated Macrophages, and Collagen Alignment as Prognostic Markers for Invasive Breast Carcinoma Patients. Am J Pathol 2018;188(3):559–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sprague BL, Vacek PM, Herschorn SD, James TA, Geller BM, Trentham-Dietz A, et al. Time-varying risks of second events following a DCIS diagnosis in the population-based Vermont DCIS cohort. Breast Cancer Res Treat 2019;174(1):227–235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lehman CD, Arao RF, Sprague BL, Lee JM, Buist DS, Kerlikowske K, et al. National Performance Benchmarks for Modern Screening Digital Mammography: Update from the Breast Cancer Surveillance Consortium. Radiology 2017;283(1):49–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ballard-Barbash R, Taplin SH, Yankaskas BC, Ernster VL, Rosenberg RD, Carney PA, et al. Breast Cancer Surveillance Consortium: a national mammography screening and outcomes database. AJR. American journal of roentgenology 1997;169(4):1001–8. [DOI] [PubMed] [Google Scholar]
- 28.Sprague BL, Bolton KC, Mace JL, Herschorn SD, James TA, Vacek PM, et al. Registry-based study of trends in breast cancer screening mammography before and after the 2009 U.S. Preventive Services Task Force recommendations. Radiology 2014;270(2):354–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Geller B, Worden J, Ashley J, Oppenheimer R, Weaver D. Multipurpose statewide breast cancer surveillance system: the Vermont experience. Journal of Registry Management 1996;23:168–174. [Google Scholar]
- 30.American College of Radiology. ACR BI-RADS® - Mammography 5th Edition. In. ACR BI-RADS Atlas: Breast Imaging Reporting and Data System. Reston, VA: American College of Radiology; 2013. [Google Scholar]
- 31.Liu Y, Keikhosravi A, Mehta GS, Drifka CR, Eliceiri KW. Methods for Quantifying Fibrillar Collagen Alignment. Methods Mol Biol 2017;1627:429–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bredfeldt JS, Liu Y, Pehlke CA, Conklin MW, Szulczewski JM, Inman DR, et al. Computational segmentation of collagen fibers from second-harmonic generation images of breast cancer. J Biomed Opt 2014;19(1):16007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bredfeldt JS, Liu Y, Conklin MW, Keely PJ, Mackie TR, Eliceiri KW. Automated quantification of aligned collagen for human breast carcinoma prognosis. J Pathol Inform 2014;5(1):28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu Y, Keikhosravi A, Pehlke CA, Bredfeldt JS, Dutson M, Liu H, et al. Fibrillar Collagen Quantification With Curvelet Transform Based Computational Methods. Front Bioeng Biotechnol 2020;8:198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Keikhosravi A, Li B, Liu Y, Eliceiri KW. Intensity-based registration of bright-field and second-harmonic generation images of histopathology tissue sections. Biomed Opt Express 2020;11(1):160–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Provenzano PP, Eliceiri KW, Campbell JM, Inman DR, White JG, Keely PJ. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med 2006;4(1):38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Riching KM, Cox BL, Salick MR, Pehlke C, Riching AS, Ponik SM, et al. 3D collagen alignment limits protrusions to enhance breast cancer cell persistence. Biophys J 2014;107(11):2546–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Toss MS, Miligy IM, Gorringe KL, AlKawaz A, Mittal K, Aneja R, et al. Geometric characteristics of collagen have independent prognostic significance in breast ductal carcinoma in situ: an image analysis study. Mod Pathol 2019;32(10):1473–1485. [DOI] [PubMed] [Google Scholar]
- 39.Lyons TR, O’Brien J, Borges VF, Conklin MW, Keely PJ, Eliceiri KW, et al. Postpartum mammary gland involution drives progression of ductal carcinoma in situ through collagen and COX-2. Nat Med 2011;17(9):1109–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maller O, Hansen KC, Lyons TR, Acerbi I, Weaver VM, Prekeris R, et al. Collagen architecture in pregnancy-induced protection from breast cancer. J Cell Sci 2013;126(Pt 18):4108–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xu S, Xu H, Wang W, Li S, Li H, Li T, et al. The role of collagen in cancer: from bench to bedside. J Transl Med 2019;17(1):309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Conklin MW, Keely PJ. Why the stroma matters in breast cancer: insights into breast cancer patient outcomes through the examination of stromal biomarkers. Cell Adh Migr 2012;6(3):249–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nelson AC, Machado HL, Schwertfeger KL. Breaking through to the Other Side: Microenvironment Contributions to DCIS Initiation and Progression. J Mammary Gland Biol Neoplasia 2018;23(4):207–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yang N, Mosher R, Seo S, Beebe D, Friedl A. Syndecan-1 in breast cancer stroma fibroblasts regulates extracellular matrix fiber organization and carcinoma cell motility. Am J Pathol 2011;178(1):325–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen X, Nadiarynkh O, Plotnikov S, Campagnola PJ. Second harmonic generation microscopy for quantitative analysis of collagen fibrillar structure. Nat Protoc 2012;7(4):654–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Francis A, Fallowfield L, Rea D. The LORIS Trial: Addressing overtreatment of ductal carcinoma in situ. Clin Oncol (R Coll Radiol) 2015;27(1):6–8. [DOI] [PubMed] [Google Scholar]
- 47.Elshof LE, Tryfonidis K, Slaets L, van Leeuwen-Stok AE, Skinner VP, Dif N, et al. Feasibility of a prospective, randomised, open-label, international multicentre, phase III, non-inferiority trial to assess the safety of active surveillance for low risk ductal carcinoma in situ - The LORD study. Eur J Cancer 2015;51(12):1497–510. [DOI] [PubMed] [Google Scholar]
- 48.Hwang ES, Hyslop T, Lynch T, Frank E, Pinto D, Basila D, et al. The COMET (Comparison of Operative versus Monitoring and Endocrine Therapy) trial: a phase III randomised controlled clinical trial for low-risk ductal carcinoma in situ (DCIS). BMJ Open 2019;9(3):e026797. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


