Introduction
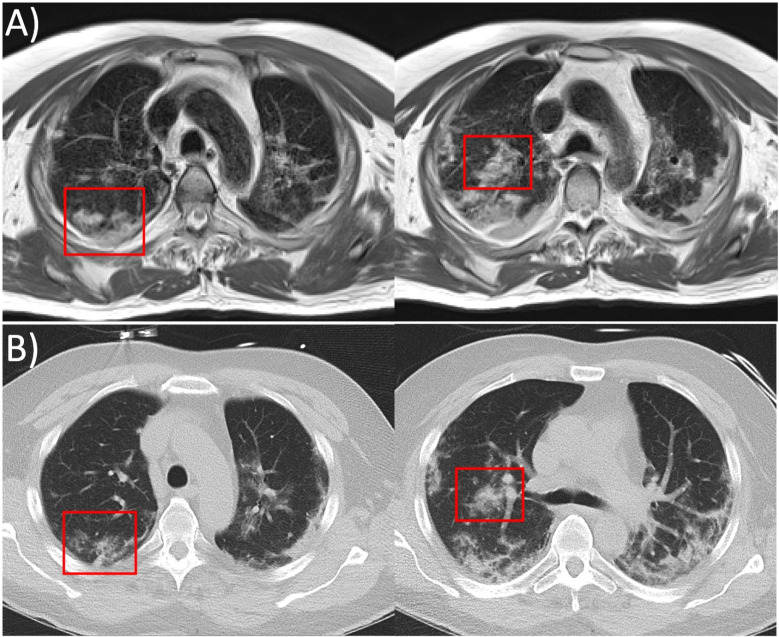
A 61-year-old man with COVID-19 infection underwent chest CT imaging and research MRI on a prototype 0.55T system on days 15 and 16, respectively, after symptom onset. Imaging was performed with local Institutional Review Board approval. We used a fast-BLADE turbo-spin echo MRI acquisition, gated to end-expiration. These MR images had intermediate image weighting between proton density and T2 (TE=48ms). CT images were reformatted to match the slice orientation and slice thickness of the MRI images for comparison. The information content provided by MRI was similar to CT (Figure, Movie [online]), including visualization of multifocal lesions equivalent to ground glass opacities predominately in upper lobes and peripheral distribution of consolidation predominantly in the lower lobes, characteristic of COVID-19 infection (1).
Figure:
Two axial slices from, A, 0.55T MRI (prototype MAGNETOM Aera, Siemens Healthineers, Erlangen, Germany) with spatial resolution 1.2mm x 1.2mm x 6mm (respiratory gated fast-BLADE turbo spin echo acquisition, TE/TR= 48ms/3471ms respiratory interval, acquisition time = 5min 15s) and, B, CT (SOMATOM Force, Siemens Healthcare, Erlangen, Germany) reformatted to 0.6mm x 0.6mm x 6mm spatial resolution. Multifocal lesions equivalent to ground glass opacities on CT and peripheral distribution of consolidative opacities characteristic of COVID-19 are observed using both imaging modalities. In some regions, the severity of the abnormality appears worse on MRI, as shown in the red boxes.
Movie.
Axial stack of slices from, A, 0.55T MRI and, B, CT.
One previous study has described ultrashort TE imaging at 3T in patients with COVID-19 infection (2), and another has demonstrated 0.55T MRI following recovery from COVID-19 infection (3). Lower field MRI systems equipped with high performance hardware and software offer promise to reduce distortion for lung MRI and to increase accessibility of MRI (4). This case demonstrates the capability of this MRI system configuration, compared with CT, for imaging COVID-19.
Acknowledgments
Acknowledgements
The authors would like to acknowledge the assistance of Siemens Healthcare in the modification of the MRI system for operation at 0.55T under an existing cooperative research agreement (CRADA) between NHLBI and Siemens Healthcare. We thank Christine Mancini, Kendall O’Brien, Peg Lowery, Jennifer Henry, and Doris Swaim for their assistance with this study.
Footnotes
Grant Support: This work was supported by the Division of Intramural Research, National Heart, Lung, and Blood Institute, National Institutes of Health (Z01-HL006213).
Disclosures: The authors are investigators on a US Government Cooperative Research and Development Agreement (CRADA) with Siemens Healthcare. Siemens participated in the modification of the MRI system from 1.5T to 0.55T.
The study was approved by the local Institutional Review Board, and all subjects provided written informed consent (Clinicaltrials.gov NCT03331380).
References
- 1.Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, Chung M, et al. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA - Secondary Publication. J Thorac Imaging. 2020;35(4):219-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yang S, Zhang Y, Shen J, Dai Y, Ling Y, Lu H, et al. Clinical Potential of UTE-MRI for Assessing COVID-19: Patient- and Lesion-Based Comparative Analysis. J Magn Reson Imaging. 2020;52(2):397-406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Heiss R, Grodzki DM, Horger W, Uder M, Nagel AM, Bickelhaupt S. High-performance low field MRI enables visualization of persistent pulmonary damage after COVID-19. Magn Reson Imaging. 2020;76:49-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Campbell-Washburn AE, Ramasawmy R, Restivo MC, Bhattacharya I, Basar B, Herzka DA, et al. Opportunities in Interventional and Diagnostic Imaging by Using High-performance Low-Field-Strength MRI. Radiology. 2019:190452. [DOI] [PMC free article] [PubMed] [Google Scholar]