Abstract
With the alarming rise of infected cases and deaths, COVID-19 is a pandemic, affecting 220 countries worldwide. Until now, no specific treatment is available against SARS-CoV-2. The causal virus SARS-CoV-2 primarily infects lung cells, leading to respiratory illness ranging in severity from the common cold to deadly pneumonia. This, with comorbidities, worsens the clinical outcome, particularly for immunosuppressed individuals with COVID-19. Interestingly, the commensal gut microbiota has been shown to improve lung infections by modulating the immune system. Therefore, fine-tuning of the gut microbiome with probiotics could be an alternative strategy for boosting immunity and treating COVID-19. Here, we present a systematic biological network and meta-analysis to provide a rationale for the implementation of probiotics in preventing and/or treating COVID-19. We have identified 90 training genes from the literature analysis (according to PRISMA guidelines) and generated an association network concerning the candidate genes linked with COVID-19 and probiotic treatment. The functional modules and pathway enrichment analysis of the association network clearly show that the application of probiotics could have therapeutic effects on ACE2-mediated virus entry, activation of the systemic immune response, nlrp3-mediated immunomodulatory pathways, immune cell migration resulting in lung tissue damage and cardiovascular difficulties, and altered glucose/lipid metabolic pathways in the disease prognosis. We also demonstrate the potential mechanistic domains as molecular targets for probiotic applications to combat the viral infection. Our study, therefore, offers probiotics-mediated novel preventive and therapeutic strategies for COVID-19 warfare.
Keywords: COVID-19, SARS-CoV-2, Probiotics, Gut-lung axis, Biological network analysis, Meta-analysis
Introduction
The coronavirus disease-2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has been declared as a pandemic by the World Health Organization (WHO). To date, more than 67 million confirmed cases of COVID-19 have been reported in 220 countries, with more than 1.5 million confirmed deaths (as of 4:22 pm CET, 9 December 2020, https://covid19.who.int/). The death toll has climbed up since no specific treatment for COVID-19 is currently available. The global emergency of the pandemic therefore urgently demands the investigation of the novel therapeutic strategy effective for SARS-CoV-2.
SARS-CoV-2 is a positive-sense RNA virus and belongs to pathogenic beta-coronavirus. The other two beta-coronavirus that caused earlier major outbreaks of lethal pneumonia are SARS-CoV and MERS-CoV. SARS-CoV-2 shares almost 80% of the genome with SARS-CoV. However, SARS-CoV-2 is more distant from MERS-CoV, with only 50% identity [1]. Epidemiology indicates SARS-CoV-2 is more infectious but less fatal (2‒5%) than SARS-CoV (9.4%) and MERS-CoV (34.5%) [2]. Nevertheless, similar to them, SARS-CoV-2 primarily infects alveolar epithelial cells of the lung, leading to a respiratory syndrome with a variable degree of severity, ranging from upper respiratory tract infection (URTI) to severe interstitial pneumonia and acute respiratory distress syndrome (ARDS) [3]. Older people and immunocompromised individuals with existing medical conditions like diabetes, hypertension, and cardiovascular complications are critically affected by the disease [4].
Interestingly, respiratory infections, sepsis, and ARDS are associated with a change in gut microbiota composition, indicating their possible role in pulmonary health [5]. It is also shown that the macrophage response to the respiratory viruses during the viral infection depends on the composition of gut microbes [6]. Therefore, the gut and lung are interconnected organs and influence their homeostasis through immunological communication [7]. Strikingly, similar cross-talk among gut and lungs appears in COVID-19 instances as well [7]. A clear decrease in cell number of two common gut bacteria, Lactobacillus and Bifidobacterium spp., has also been found in COVID-19 patients [8]. Next, it is also reported that bronchoalveolar lavage fluid samples of COVID-19 and community-acquired-pneumonia patients are dominated by bacteria that are common in the oral cavity and upper respiratory tract (URT) [7]. Hence, the cross-talk between the gut and the lung through the gut-lung axis may impart a significant role in SARS-CoV-2 infection [5]. Therefore, manipulation of the intestinal microbiota by application of probiotics could be a potential therapeutic strategy or adjuvant therapeutic option for maintaining health and preventing and/or treating the disease. Consumption of probiotics has also been shown to improve the URTI in numerous human clinical trials [9] (Table 1).
Table 1.
The probiotic products with formulation supplied in human clinical trials in the prevention of upper respiratory tract infection (URTI)
| References | Probiotic strain | Formulation | Age group | Dosage | Mode of administration | Function |
|---|---|---|---|---|---|---|
| Laursen et al. [13] | Bifidobacterium animalis subsp. lactis (BB-12) | B. animalis subsp. lactis (BB-12) and L. rhamnosus (LGG) |
Infants 8‒14 months (healthy) |
1 g of maltodextrin powder with 1 × 109 cfu each of BB-12 and LGG for 6 months | Oral | No significant effect |
| Leyer et al. [14] | Bifidobacterium animalis subsp. lactis Bi-07 (ATCC PTA-4802) | B. animalis subsp. lactis Bi-07 (ATCC PTA-4802), L. acidophilus NCFM (ATCC 700396) | Children 3‒5 years (healthy) | 1 g sachet with 1 × 1010 cfu of each bacterium with 120 ml 1% fat milk twice daily for 6 months | Oral |
Reduced fever, rhinorrhoea, cough incidence and antibiotic requirement |
| Pregliasco et al. [15] | Bifidobacterium lactis (Probial BS 01-LMG P-21384) |
L. plantarum (Probial LP 02-LMG P-21020), L. rhamnosus (Probial LR 04-DSM 16605) and B. lactis (Probial BS 01-LMG P-21384) |
Person 15‒56 years (healthy) | 1 sachet (0.1 g) with 10 × 109 cfu of each bacterium daily for 90 days | Oral | Significantly decreased URTI |
| Hojsak et al. [17] | Bifidobacterium animalis subsp. lactis (BB-12®) | Individual | Children 1‒18 years (hospitalized) | 1 g maltodextrin powder with 1 × 109 cfu for the entire duration of the hospital stay | Oral | No significant effect |
| Hojsak et al. [16] | Bifidobacterium animalis subsp. lactis (BB-12) | Individual | Children 1.43‒7.48 years (healthy) | 1 g maltodextrin powder with 1 × 109 cfu for 90 days | Oral | No significant effect |
| Li et al. [18] | Bifidobacterium tetravaccine tablets |
Bifidobacterium infantis, Lactobacillus acidophilus, Enterococcus faecalis and Bacillus cereus |
Children > 11 years (recurrent respiratory tract infected) | Bifidobacterium tetra vaccine tablets (live) for 2 months | Oral | Reduced the frequency of URTI |
| Cazzola et al. [19] | Bifidobacterium bifidum R0071 | L. helveticus R0052, B. longum subsp. infantis R0033, B. bifidum R0071 | Children 3‒7 years (healthy) | 1.5 g sachet with 5 × 109 cfu live cells daily for 3 months | Oral | Prevented usual acute infectious illnesses, decreased the risk of occurrence of common infectious viral diseases including common cold, flu, respiratory problems |
| Garaiova et al. [20] | Lactobacillus acidophilus CUL60 (NCIMB 30157), Lactobacillus acidophilus CUL21 (NCIMB 30156) | L. acidophilus CUL21 (NCIMB 30156), L. acidophilus CUL60 (NCIMB 30157), B. bifidum CUL20 (NCIMB 30153), and B. animalis subsp. lactis CUL34 (NCIMB 30172) | Children 3–6 years (healthy) | 1 tablet with 1 × 1010 cfu of Lactobacillus spp. and 0.25 × 1010 cfu of Bifidobacterium spp. and 50 mg vitamin C daily for 6 months | Oral | Potential preventives for URTI |
| Gerasimov et al. [21] | Lactobacillus acidophilus DDS-1 (NCIMB 30333) | L. acidophilus DDS-1 and B. lactis UABLA-12 (NCIMB 30334) | Children 3‒12 years (healthy) | ∼1 g of powder with 5 × 109 cfu of Lactobacillus spp. and Bifidobacterium spp. in 1:4 ratio and 50 mg FOS for 7 months | Oral | Reduced acute respiratory infection (ARI) |
| Strasser et al. [22] | Lactobacillus acidophilus W22 | B. bifidum W23, B. lactis W51, E. faecium W54, L. acidophilus W22, L. brevis W63, and Lactococcus lactis W58 | Adults 20‒35 years (healthy) | 4 g sachet with 1 × 1010 cfu of each bacterium daily for 12 weeks | Oral | Reduced the incidence of URTI |
| Strasser et al. [22] | Lactococcus lactis W58 | B. bifidum W23, B. lactis W51, E. faecium W54, L. acidophilus W22, L. brevis W63, and L. lactis W58 | Adults 20‒35 years (healthy) | 4 g sachet with 1 × 1010 cfu of each bacterium daily for 12 weeks | Oral | Reduced the incidence of URTI |
| Jespersen et al. [23] | Lactobacillus paracasei subsp. paracasei, L. casei 431 | Individual | Person 18‒60 years (healthy) | 100 ml milk with 1 × 109 cfu live cells once daily for 6 weeks | Oral | Reduced the duration of the common cold and influenza-like illness (ILI) episodes in healthy adults |
| Pu et al. [24] | Lactobacillus paracasei (N1115) | Individual | Older person ≥ 45 (healthy) | 3.6 × 107 cfu/mL live cells for 12 weeks | Oral | Reduced the risk of acute upper tract infections in the elderly. Enhanced T-cell-mediated natural immune defense |
| Corsello et al. [25] | Lactobacillus paracasei CBA L74 | Individual | Children 12‒48 months (healthy) | 150 ml of milk or water with 5.9 × 109 cfu/g live cells for 3 months | Oral | Reduced the risk of acute upper tract infections in the elderly. Enhanced T-cell-mediated natural immune defense |
| Nocerino et al. [26] | Lactobacillus paracasei CBA L74 | Individual | Children 12‒48 months (healthy) | 150 ml of milk or water with 5.9 × 109 cfu/g live cells for 3 months | Oral | Reduced the risk of acute upper tract infections in the elderly. Enhanced T-cell-mediated natural immune defense |
| Berggren et al. [27] | Lactobacillus paracasei 8700:2 (DSM 13434) | L. paracasei 8700:2 (DSM 13434) and L. plantarum HEAL 9 (DSM 15312) | Person 18‒65 years (healthy) | 1 g maltodextrin and lyophilized bacteria with 1 × 109 cfu/day live cells for 12 weeks | Oral | Reduced frequency and duration of common cold, URTI |
| Szymanski et al. [28] | Lactobacillus plantarum PL02 | B. longum PL03 (33%), L. rhamnosus KL53A (33%), and L. plantarum PL02 (34%) | Children 5 months to 16 years (with respiratory tract infection) | 1 tablet with 1 × 108 cfu cells twice daily for 4 weeks | Oral | No significant function was observed |
| Hirose et al. [29] | Lactobacillus plantarum L-137 | heat-killed L. plantarum L-137 (HK L-137) | Older person 40‒64 years (healthy) | 1 tablet with 50 mg of bacteria daily for 12 weeks | Oral | The decreased URTI incidence in healthy subjects through augmentation of immune functions |
| Tubelius et al. [30] | Lactobacillus reuteri protectis (ATCC 55730) | Individual | Person 18‒65 years (healthy) | 100 ml liquid with 1 × 108 cfu live cells for 80 days | Oral | Shortened duration of respiratory diseases |
| Pregliasco et al. [15] | Lactobacillus rhamnosus (Probial LR 04-DSM 16605) | L. plantarum (Probial LP 02-LMG P-21020), L. rhamnosus (Probial LR 04-DSM 16605), and B. lactis (Probial BS 01-LMG P-21384) | Person 15‒ 62 years (healthy) | 1 capsule (5 g) with 0.1 g = 10 × 109 cfu L. plantarum; 0.1 g = 10 × 109 cfu L. rhamnosus; 0.1 g = 10 × 109 cfu B. lactis; 3 g FOS and 1.7 g glucose/maltodextrin daily for 3 months | Oral | Improved health by reducing the incidence and severity of respiratory diseases |
| Kukkonen et al. [31] | Lactobacillus rhamnosus GG | L. rhamnosus GG, L. rhamnosus LC705, B. breve Bb99 and P. freudenreichii ssp. shermanii JS | new born infants (healthy) | 1 capsule with 8–9 × 109 cfu of each bacterium for 6 weeks | Oral | Increased resistance to respiratory infections |
| Hojsak et al. [32] | Lactobacillus rhamnosus GG | Individual | Children 13‒86 months (healthy) | 100 ml of fermented milk with 1 × 109 cfu cells daily for 3 months | Oral | Reduced the risk of URTI |
| Hojsak et al. [33] | Lactobacillus rhamnosus GG | Individual | Children > 12 months (non-healthy) | 100 ml of fermented milk with 1 × 109 cfu cells daily for 3 months | Oral | Reduced the risk of URTI |
| Kumpu et al. [34] | Lactobacillus rhamnosus GG | Individual | Children 2‒6 years (healthy) | Milk with 6.7 × 105 to 1.9 × 106 cfu/ml cells for 28 weeks (amount of milk consumed by each child was recorded) | Oral | Reduced the risk of URTI |
| Smith et al. [35] | Lactobacillus rhamnosus LGG | L. rhamnosus LGG® and B. animalis ssp. lactis BB-12® | Adults 18‒24 years (Susceptible to upper respiratory infections) |
5 g powder (stick) with 1 × 109 cfu cells each of LGG and BB-12 daily for 12 weeks |
Oral |
Mitigated decrements in health-related quality of life (HRQL) during upper respiratory infections (URI) |
| Wang et al. [36] | Lactobacillus rhamnosus GG | Individual | Older person ≥ 65 years (hospital residents) | 2 capsules with 1 × 1010 cfu cells daily for 6 months | Oral | Reduced the risk of URTI |
| Shida et al. [37] |
Lactobacillus casei strain Shirota (LcS, YIT 9029) |
Individual | Person 30‒49 years (healthy) | 1 drink with 1 × 1011 cfu live cells daily for 12 weeks | Oral | Reduced risk of URTI and common infectious diseases (CID) |
| Fujita et al. [38] | Lactobacillus casei strain Shirota (LcS) | Individual | Person 18‒67 years (healthy) | 80 ml fermented milk with 4 × 1010 cfu cell of LcS per day | Oral | Reduced the duration of acute URTIs |
| Guillemard et al. [39] | Lactobacillus casei DN-114 001 | L. casei DN-114 001, S. thermophilus, and L. delbrueckii subsp. bulgaricus | Older person ≥ 70 years (healthy) | 2 bottles of 100 g/d with 1 × 1010 cfu/100 g live cells for 112 days | Oral | Reduced risk of URTI and common infectious diseases (CID) |
| Guillemard et al. [40] | Lactobacillus casei DN-114 001 | L. casei DN-114 001, S. thermophilus, and L. delbrueckii subsp. bulgaricus | Person 18‒65 years (healthy) | 2 bottles of 100 g/d with 1 × 1010 cfu/100 g live cells for 112 days | Oral | Reduced risk of URTI and common infectious diseases (CID) |
Since the specific strains of probiotics, applied with appropriate dosage and mode of administration, can efficiently treat respiratory complications, we hypothesize that the application of probiotics could be effective against SARS-CoV-2 infection. Probiotics build up a protective mucus barrier, impart healthy immune response to all age group individuals minimizing the probability of infectious disease occurrence [10]. Probiotics also maintain the balance of the gut microbial community and protect the respiratory system by preventing secondary bacterial infections [7]. Recently, Tiwari et al. (2020) have reviewed the beneficial strains of lactic acid bacteria (LAB) and their potential antiviral properties [11]. Metabolites like short-chain fatty acids (SCFAs), lactic acid, hydrogen peroxide, and antimicrobial peptides (AMPs) like bacteriocins produced by the LAB have been suggested to be beneficial in restricting the viral entry and/or reducing the viral load [11]. Additionally, such AMPs are also important in the colonization of probiotics in the GI tract, cell signaling, and host defense [11]. The activities of bacteriocins in reinforcing the immunity with their immunomodulatory effects further suggest the prophylactic use of probiotics against viral infections, including COVID-19 [11].
Network analysis offers an effective approach to identify molecular mechanisms and connections between genes and their pathways from dynamic networks [12]. Therefore, in the present network and meta-analysis study, a biological association network is generated concerning training genes of SARS-CoV-2 infection and probiotic treatment. The meta-analysis shows the rational justification for the implementation of the probiotics approach in respiratory infections. The analysis of functional modules, pathway enrichment, and topological network parameters reveal that probiotics could have tremendous therapeutic potential during the pathophysiological events of COVID-19. The comprehensive network analysis study indicates that the application of probiotics at the significant domains of the infection mechanism could be beneficial in the prevention and treatment of SARS-CoV-2 infection.
Materials and Methods
Despite the genomic similarity with the SARS-CoV, high-throughput data and an unambiguous model for SARS-CoV-2 immunopathology are unavailable. The literature search for the study was performed according to the PRISMA guideline (Fig. 1). The rationale for probiotics as a cure for the global pandemic was justified by the meta-analysis study (Fig. 2). Only a few clinical trials concerning the direct application of other probiotics like yeast (Saccharomyces boulardii), Bacilli spp. (Bacillus coagulans), and Enterococcus spp. (Enterococcus faecalis) on URTI patients are available. Therefore, the study was restricted to the most common probiotic strains Lactobacillus spp. and Bifidobacterium spp. to increase the robustness and accuracy of the network meta-analysis. Further, a network-analysis study was considered to evaluate the probable bioprotective mechanisms of probiotics against SARS-CoV-2 infection. The network module analysis revealed functionally related genes and provided key domains for the potential mechanism of probiotics against SARS-CoV-2 infection. The entire methodology is summarized in Fig. S1.
Fig. 1.
Systematic literature search selection process. The PRISMA diagram details the applied search and selection process during this study
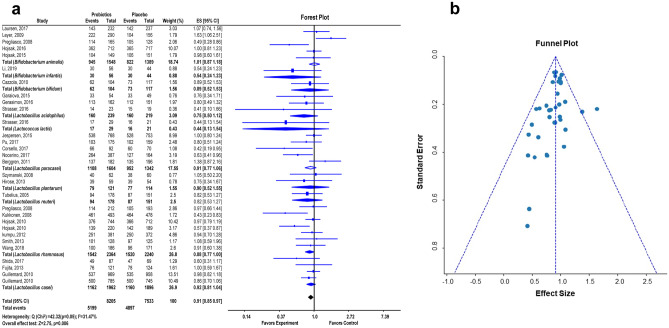
Fig. 2.
Meta-analysis study rationalizes the application of probiotics as a preventive and treatment strategy in COVID-19. Forest plot showing pooled mean difference and 95% confidence intervals for the effect of probiotics-based therapy versus placebo controls (after adjustment for heterogeneity) on the patients with upper respiratory tract infection (URTI) (a). Funnel plot of the standard error plotted against the effect size to examine publication bias shows the effect of probiotics on upper respiratory tract infection (URTI) in different clinical trials (b). The perpendicular line to the x-axis represents the pooled effect size. The studies outside the triangle represent positive or negative bias. The lack of significant asymmetry in the funnel plot suggests the absence of publication bias
Rational for Application of Probiotics as Potential Preventive and an Alternative Treatment Strategy for SARS-CoV-2 Infection: A Meta-analysis
The meta-analysis was conducted to analyze similar studies that are already available in the literature. In our study concerning probiotic treatment on URTI patients, Q-statistics is used to assess whether all effect sizes in the sample of single studies are homogeneous (belong to the same population). The I2 index indicates the degree of heterogeneity. Further, the heterogeneity on the pooled estimates of the individual outcomes of the meta-analysis was assessed by the forest plot. The effects of probiotics depend on specific strains, age-group, clinical dosages, and the mode of administration. Clinical studies with randomized, double-blind placebo-controlled human trials were screened. The overall effects of specific probiotic strains were measured as mean difference (MD) or standardized mean difference (SMD), effects size, 95% confidence interval (CI), and weight percentage for studies by using a random-effects model. The mean change, i.e., the standard deviation in the number of healthy individuals and patients, was used to calculate the MD between the probiotics (or synbiotics) and the placebo groups. The statistical analyses were performed using MetaPython. The p value of Q statistic < 0.05 was defined as an indicator of heterogeneity, and data were considered heterogeneous for I2 values higher than 40%. Begg’s funnel plot was performed to examine the publication bias. All the reported p values were two-sided, and p values ≤ 0.05 were regarded as statistically significant for all included studies.
Literature Search Strategy: Data-Mining
The literature study was conducted following the PRISMA guidelines (Fig. 1). The comparatively unexplored functional genes (training genes) relevant to the viral infection were detected from the structured databases by data-mining strategy. The primary search strategy included manual analysis of the published literature indexed in the electronic search database PubMed archive (http://www.ncbi.nlm.nih.gov/pubmed/) using reference management software EndNote X7 (Bld 7072) (endnote.com). The search terms or MeSH terms used in EndNote included “probiotics” AND “coronavirus.” In the scarcity of the available studies regarding SARS-CoV-2 immunopathology and antiviral probiotic application against them, the literature search was continued using the NCBI PubMed database. The search terms or keywords used were “probiotics” ∩ “COVID-19”, “probiotics” ∩ “severe acute respiratory syndrome coronavirus 2”, “probiotics” ∩ “SARS-CoV-2,” and “probiotics” ∩ “coronavirus.” The search was limited to the studies conducted on humans, and available literature from April 18 to June 8, 2020 was considered for the present study. Therefore, data-mining considered the immunopathology of SARS-CoV-2 infection and the application of probiotics to mitigate the disease severity. This approach allowed us to pull out the training genes associated with novel coronavirus infection and probiotic treatment. The top-ranked genes were thus selected and used for text-mining.
Association Network Construction and Selection of the Candidate Genes: Text-Mining
An efficient and integrated combination of data-mining and text-mining approaches was used to retrieve documents eliminating biases toward vigorously investigated disease phenotypes. The unstructured biomedical texts were regained with simultaneous construction of interaction networks by text-mining strategy. Thus, the candidate genes associated with SARS-CoV-2 infection and antiviral probiotic treatment were constructed in an association network by screening the training genes from literature based on the text-mining approach. An open-source bioinformatics tool, Cytoscape, and the metasearch plug-in Agilent Literature Search was used to visualize the molecular interaction networks, integrating the gene expression profiles of the respective training genes. Agilent Literature Search software fetched documents based on the entered query using multiple text-based search engines, parsed into sentences. The software generated the association network based on a lexicon set definition which defines the gene names of the parsed sentences as “concepts” and the interaction terms of interests as “verbs.” The network was generated by extracting an association for every parsed sentence containing at least two “concepts” and one “verb” or known interaction term. The gene names and association terms represented nodes and edges respectively of the text-mining–based network, generated through Cytoscape. In this work, Cytoscape 3.7.1 App and Agilent Literature Search 3.1.1 beta (LitSearch version 2.69) were used to analyze the existing data source from the published literature available in the PubMed database. The training genes were used as “search terms”; Max Engine Matches was set at 10; the “use context” and the “concept lexicon restrict search” options were selected as search controls; “Homo sapiens” was used as “extraction control”.
Functional Module Determination: MCODE Analysis
Any disease pathogenesis manifests the complex interaction of biological events modulated by the disease-related association network. Identification of tightly interconnected nodes or genes from a very densely connected network was useful for understanding significant biological events in terms of modules and their interconnections. The association network generated with SARS-CoV-2 pathogenesis candidate genes was subdivided into modules by MCODE plug-in of Cytoscape according to local neighborhood density. The extracted modules were graphically displayed as an isolated, more considerable dense region with functionally similar genes. The network view of the clusters enabled us to understand the local topology and functional features concerning the whole network consisting of several other candidate genes and edges. Network modules with MCODE score of more than three and a minimum of four nodes were considered as significant and carried forward for further analysis of functional annotation. The functional annotations and the pathways associated with candidate genes of the selected MCODE clusters were determined by GeneCards and NCBI.
Quantitative Data Synthesis: Network Analysis
The network topology parameter details of the association network were obtained by analyzing the association network with the NetworkAnalyzer tool in Cytoscape. The node degree and clustering coefficient of the individual node genes together represented the number of connections associated with a node and the degree of involvement of a node in the participating clusters. This tool showed the degree of participation of individual genes in the whole network and participating clusters. The functional annotations of the highest MCODE score with clustered candidate genes were determined by the integrative human gene database GeneCards (version 4.14). It provided gene-centric data of the annotated and predicted human genes with the functions and pathways associated with the candidate genes.
Assessment of Overrepresentation of Gene Ontology Categories by BiNGO
To identify enriched biological processes that are affected by COVID-19 immunopathology, a gene ontology (GO) functional enrichment analysis was performed using the Biological Networks Gene Ontology tool (BiNGO, version 3.0.3) in Cytoscape 2.8.0 (http://www.cytoscape.org/), with a threshold of p < 0.001. BiNGO is a tool to determine statistically overrepresented GO terms for a set of genes associated with any biological processes as recorded in the Gene Ontology database. The BiNGO analysis was done with the MCODE-derived gene clusters of the association network. The statistical test was set to “hypergeometric test,” multiple testing correction as “Benjamini & Hochberg False Discovery Rate (FDR) correction,” the significance level was set to 0.05, the categories to be visualized were “overrepresented categories after correction,” reference set was “use the whole annotation as reference set,” ontology file was “GO_Biological_Process,” and finally organism/annotation was selected as “Homo sapiens.”
Results
Overall Effects of Probiotics on URTI
URTI is one of the principal symptoms of SARS-CoV-2 infection. Therefore, in all the meta-analysis studies, the duration and severity of URTI were compared between individual probiotic or probiotic formulation (or synbiotics) treatment and placebo-controlled groups, including children and adult individuals [13–40]. The forest plot showed the effect of probiotic treatment on URTI (Fig. 2a). As indicated by an I2 value of 31.47% and Q (chi-square) statistics of 42.32 (p = 0.05), there was no statistical heterogeneity. The overall effect size of the study was 2.75, with p = 0.006. The effect of probiotic treatment on URTI was plotted according to specific strains of probiotics. The effects size (ES), confidence level (95% CI), and the weight percentage of clinical trials concerning the specific strains were also depicted. Thus, the forest plot of the meta-analysis study showed statistically significant effects of probiotic treatment on URTI.
Publication Bias
The funnel plot showed minimal evidence of publication bias among the selected studies associated with randomized, double-blind, placebo-controlled human clinical trials of probiotic application on URTI (Fig. 2b). The funnel plot with strain-specific probiotic treatment on URTI patients was symmetrical, which indicated that no significant asymmetry was detected in any of the analyses.
Identification of the Respective Training Genes Associated with COVID-19 Pathophysiology and Antiviral Approaches with Probiotic Treatment
The data-mining exploratory technique found out about 36 data, including clinical trials, in vitro experiments, bioinformatics analysis, reviews, mini-reviews, and editorial letters associated with COVID-19 pathophysiology and probiotic treatment from the literature [1, 41–47]. The use of specific MeSH terms in EndNote and keywords used in the PubMed database excluded non-specific search results. This confirmed the accuracy of the mining study performed with the reports exclusively on SARS-CoV-2 infection and the application of probiotics. Manual analysis of the literature shortlisted 90 training genes of interest associated with COVID-19 pathogenesis (Table 2). These genes were used for the generation of the association network and further analysis.
Table 2.
Training gene set entangled with SARS-CoV-2 infection and probiotic treatment
| References | Training genes |
|---|---|
| Wang et al. [41] | ifnγ, il4, cd11c, cd80, cd86 |
| Chai et al. [42] | il1β, il2, il6, il8, il10, ifnα, tnfα, tlr3, tgev |
| Romano et al. [43] | il1, il12, il15, il13, il17, nf-κb, stat3 |
| Catanzaro et al. [1] | ace2, tmprss2, il6, il1β, il8, g-csf, gmcsf, ip-10, mcp-1, ccl3, tnfα, hs-crp, ighv3–23, ighv3–7, ighv3–15, ighv3–30, igkv3–11, ighv3–23-ighj4, il1ra, il9, fgf, ifnγ, mip-1α, mip-1β, pdgf, vegf, g-csf,, mcp-1, mip-1α, tnfα, ifn-α2, ifnγ il1ra il2, 4, 7, 10, 12, 17, g-csf, m-csf, tlr3, tlr7, tlr8, rig-i, mda5, cxcl2,, jak, ccl3 myd88, inos, cd86, gp130, adam17, il-6rα, sil-6rα, egfr, aak1, gak, s1p, s1prs, ras/erk, pi3k/akt/enos, plc/ca2 + , trif, at1, ccl2, s1p1, ifnβ |
| Young et al. [44] | crp, hmgcoa, app, scfa, gpr43, hdac |
| Akour [45] | g-csf, ip10/cxcl10, mcp-1, thp-1, il22, il8, tnfα, mpo, inos, cox-2, nf-κb, il6 |
| Renzo et al. [46] | ace2, il10, il17, mtor |
| Shi et al. [47] | hla, has2 |
Generation of Association Network with the Candidate Genes of COVID-19 Pathological Mechanisms and Antiviral Treatments with Probiotics
All the 90 shortlisted training genes were used for the development of a text-mining–based association network. The primary association network obtained through Cytoscape with training genes showed 453 nodes representing the candidate genes and 1273 edges. Fig. S2 indicated the topology of the central association network, highlighting the listed training genes involved in the SARS-CoV-2 infection. Table 3 shows the network parameter statistics. According to the network topology, “the number of connected components” represented the network connectivity, which reflected a pairwise connection among all the nodes. Hence, a lower number of connected components of 27, obtained by the network statistics, suggested robust connectivity of the association network (Table 3). The clustering coefficient 0.633 designated the average clustering coefficient or degree involvement of respective nodes in the participating cluster of the network, with 5.620 average number of neighbors. The tendency to form high degree nodes or hub nodes was described by high “network heterogeneity” of 1.137 and the absence of any isolated nodes. The presence of hub nodes indicated a real functional network with multiple biologically relevant pathways associated with the novel coronavirus infection. The text-mining approach in conjunction with Cytoscape has been successfully adopted to illustrate actin dynamics during the post-ejaculatory life of spermatozoa [48], to demonstrate the synergistic mechanisms of therapeutic herbs for rheumatic arthritis [49], and to find out the effects of psychological stress on innate immunity and metabolism in human [50].
Table 3.
Network statistics of the association network of the probiotics-COVID-19 axis
| Topology feature details | |||||||
|---|---|---|---|---|---|---|---|
| Network | Network heterogeneity | Number of Nodes | Nodes Edges | No. of isolated nodes | Avg. num. of neighbors | Clustering coefficient | Number of connected components |
| Training gene set | 1.137 | 453 | 1273 | 0 | 5.620 | 0.633 | 27 |
The table contains details of the primary association network obtained by text-mining results using an Agilent Literature search (ALS) plugin. “Network clustering coefficient” is the average of the clustering coefficients for all nodes in the network. The “average number of neighbors” indicates the average connectivity of a node in the network
Cluster Analysis of the Association Network Identified the Groups of Promising Genes Related to COVID-19 Clinical Spectrum and Antiviral Probiotic Treatment
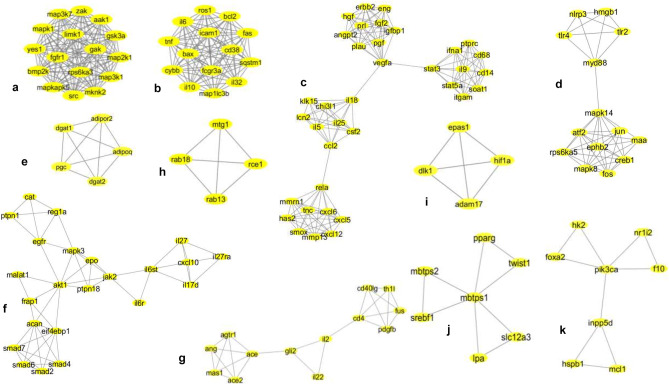
MCODE tool of Cytoscape derived 38 densely bridged modules from the first network (Fig. S3) by an algorithm called “top overlap.” It made groups of “genes of interest” according to their correlation coefficient and highest-scoring edge among all other candidate genes of the network. The algorithm also minimized the chances of false-positive results, increasing the robustness and accuracy of the analysis [51]. The individual clusters were further selected based on their scores for functional studies with the removal of lower confidence data points. The details of the modules are given in table S1. Among the 38 derived modules, 11 resultant clusters with MCODE score ≥ 3, nodes ≥ 4, edges ≥ 6 were chosen for functional annotation (Fig. 3). Module 1 with MCODE score 16, 16 nodes, and 120 edges was the highest-scoring module, whereas module 2 with MCODE score 14, 14 nodes, and 91 edges was the second-highest, and module 3 with score 8.457, 36 nodes, and 147 edges was the third-highest. The network matrices like the node degree, the clustering coefficient of the primary network, and MCODE-derived modules provided the topological parameters to understand the importance of nodes for pathway analysis. The highest-scoring module 1 (MCODE score 16) consisted of 16 nodes (src, limk1, rps6ka3, aak1, mapk1, mknk2, map3k1, gak, map2k1, fgfr1, mapkapk5, map3k7, bmp2k, zak, gsk3, yes1) (Fig. 3a). The highest number of nodes or edges containing module 3 (cxcl5, cxcl12, tnc, has2, cxcl6, mmrn1, mmp13, smox, rela, ccl2, chi3l1, csf2, il5, il25, klk15, lcn2, il18, vegfa, eng, plau, pgf, prl, angpt2, igfbp1, hgf, erbb2, fgf2, stat3, stat5, ifna1, soat1, cd68, il9, itgam, ptprc, cd14) was preferred as the most significant functionally relevant clusters for SARS-CoV-2 infection. It is worth mentioning here that cluster analysis (using MCODE algorithm) is widely adopted for biological interpretation and elucidation of complex molecular networks (see Discussion) [52].
Fig. 3.
Network topologies of the most significant eleven MCODE clusters derived from association network of SARS-CoV-2 pathogenesis and probiotic treatment selected for further GO enrichment and pathway analysis. MCODE derived cluster 1: score 16, nodes 16 (src, limk1, rps6ka3, aak1, mapk1, mknk2, map3k1, gak, map2k1, fgfr1, mapkapk5, map3k7, bmp2k, zak, gsk3, yes1), and edges 120 (a). MCODE-derived cluster 2: score 14, nodes 14 (tnf, il6, map1lc3b, ros1, cd38, bax, fas, sqstm1, il10, fcgr3a, cybb, il32, bcl2, icam1), and edges 91 (b). MCODE-derived cluster 3: score 8.457, nodes 36 (cxcl5, cxcl12, tnc, has2, cxcl6, mmrn1, mmp13, smox, rela, ccl2, chi3l1, csf2, il5, il25, klk15, lcn2, il18, vegfa, eng, plau, pgf, prl, angpt2, igfbp1, hgf, erbb2, fgf2, stat3, stat5, ifna1, soat1, cd68, il9, itgam, ptprc, cd14), and edges 148 (c). MCODE-derived cluster 5: score 6.769, nodes 14 (tlr2, tlr4, nlrp3, myd88, hmgb1, mapk14, creb1, rps6ka5, jun, ephb2, mapk8, atf2, fos, maa), and edges 44 (d). MCODE-derived cluster 6: score 5, nodes 5 (dgat2, adipor2, dgat1, pgc, adipoq), and edges 10 (e). MCODE-derived cluster 9: score 4.818, nodes 23 (il27ra, il27, cxcl10, il17d, il6st, il6r, jak2, ptpn18, mapk3, epo, akt1, egfr, reg1a, ptpn1, cat, frap1, malat1, acan, eif4ebp1, smad4, smad6, smad2, smad7), and edges 53 (f). MCODE-derived cluster 10: score 4.167 and 4, nodes 13 (ace2, mas1, ace, ang, agtr1, il22, il2, gli2, cd4, cd40lg, fus, th1l, pdgfb) (g). MCODE-derived cluster 11: score 4, nodes 5 (rab18, rab13, mtg1, rce1), edges 25 (h). MCODE-derived cluster 13: score of 4, nodes 4 (hif1a, dlk1, adam17, epas1), and edges 6 (i). MCODE-derived cluster 35: score of 3, nodes 7 (pparg, twist1, lpa, slc12a, mbtps1, srebf1, mbtps2), and edges 9 (j). MCODE-derived cluster 38: score of 2.857, nodes 8 (hk2, nr1i2, foxa2, pik3ca, f10, inpp5d, hspb1, mcl1), and edges 10 (k)
Determination of Functional Annotation Profile of the Sub-clusters to Elucidate the Protective Avenues of Probiotics Against SARS-CoV-2 Infection
A higher-scoring MCODE module with a high number of nodes and edges implied a possible multi-functional role. Such modules could be assigned in the essential regulatory functions of the disease pathogenesis and thereby could be the target for the probiotic treatment.
The genomic information of the nodes showed that MCODE cluster 1 candidate genes src, limk1, rps6ka3, aak1, mapk1, mknk2, map3k1, gak, map2k1, fgfr1, mapkapk5, map3k7, bmp2k, zak, gsk3, yes1 were involved in the receptor-mediated endocytosis and phagocytosis, stress-mediated cellular metabolic pathways (Fig. 3a). Therefore, probiotic supplementation could heal the disrupted intestinal barrier and prevent virus entry.
MCODE cluster 2 consisting of 14 nodes (tnf, il6, map1lc3b, ros1, cd38, bax, fas, sqstm1, il10, fcgr3a, cybb, il32, bcl2, icam1) (Fig. 3b) had functions in the activation of immune cells (B cells, T cells, natural killer cells) in response to stress stimuli, induction of the NF-κb-mediated inflammatory pathways, and subsequent apoptosis as well as the production of cytokines. Several probiotic strains are reported to balance the immune activations and inflammatory cytokines, which could have immense implications in the SARS-CoV-2-induced immunological complications.
Although MCODE cluster 3 was the third in rank based on score, it contained the highest number of nodes and edges that might indicate multiple cellular pathways (Fig. 3c). The GeneCards-derived functional profile of nodes expectedly showed the interconnections of four cellular events upon the viral infection: (a) activation of principal antiviral interferons (IFNs) and innate immune cells (stat3, stat5, ifna1, soat1, cd68, il9, itgam, ptprc, cd14); (b) activation of proinflammatory cytokines and their downstream signaling through JAK-STAT pathway (ccl2, chi3l1, csf2, il5, il25, klk15, lcn2, il18), (c) activation of proinflammatory cytokines and chemokines resulting in the immune cell (neutrophil, monocyte) migration and infiltration in lung tissues, and (d) synthesis of lung-damaging hyaluronan by has2 or hyaluronan synthase-2 (cxcl5, cxcl12, tnc, has2, cxcl6, mmrn1, mmp13, smox, rela), a proinflammatory cytokine-induced growth factor, responsible for vascular permeability and pulmonary dysfunction (vegfa, eng, plau, pgf, prl, angpt2, igfbp1, hgf, erbb2, fgf2). Although limited studies are available on the ability of probiotics to degrade glycosaminoglycan molecules, probiotics could have an impact on the degradation of hyaluronan substances synthesized by SARS-CoV-2-induced proinflammatory cytokines.
The candidate genes of cluster 5 (tlr2, tlr4, nlrp3, myd88, hmgb1, mapk14, creb1, rps6ka5, jun, ephb2, mapk8, atf2, fos, maa) represented the genes responsible for TLR-mediated innate immune responses and subsequent activation of inflammasome complex, with stress-induced MAPK signaling pathway (Fig. 3d). The virus entry might activate the host’s innate immune system through TLR-myd88-mediated pathways and eventually activate the cellular inflammasome complex. The functional annotations of cluster 6 candidate genes (dgat2, adipor2, dgat1, pgc, adipoq) indicated glucose and lipid metabolic pathways and innate immune responses (Fig. 3e). Host defense mechanisms by the proliferation of Th1 cell suppressing Th2 and Th17 cells, isotype switching and anti-inflammatory cytokine production, and induction of growth regulatory pathways were represented by MCODE cluster 9 candidate genes (il27ra, il27, cxcl10, il17d, il6st, il6r, jak2, ptpn18, mapk3, epo, akt1, egfr, reg1a, ptpn1, cat, frap1, malat1, acan, eif4ebp1, smad4, smad6, smad2, smad7) (Fig. 3f). The genomic functional annotations of cluster 10 (ace2, mas1, ace, ang, agtr1, il22, il2, gli2, cd4, cd40lg, fus, th1l, pdgfb) and cluster 11 (rab18, rab13, mtg1, rce1) candidate genes showed the reported ACE2-ADAM17-mediated entry of SARS-CoV-2 in the host cell with the resulting dysregulation of the renin-angiotensin system (RAS) involving ace2, ace, mas1, ang genes (Fig. 3g and h). Moreover, cluster 13 candidate genes (hif1a, dlk1, adam17, epas1) indicated the receptor-mediated endocytosis of the virus entry with the consequent activation of oxidative stress-responsive pathways mediated by hif1a (Fig. 3i). Probiotic supplementation could alter the adverse conditions of the infection by regulating host metabolic pathways through the secretion of SCFAs. Additionally, probiotics could balance the inflammatory cytokines and chemokine levels in the body by regulating oxidative stress, altering the SARS-CoV-2 infection condition. The effects of probiotics in controlling blood pressure may significantly alter ace2-mediated dysregulation of RAS. Finally, the candidate genes of MCODE cluster 35 (pparg, twist1, lpa, slc12a, mbtps1, srebf1, mbtps2) and cluster 38 (hk2, nr1i2, foxa2, pik3ca, f10, inpp5d, hspb1, mcl1) designated the involvement of glucose and lipid metabolic pathways, adipocyte differentiation, VEGF signaling pathway, and induction of atherosclerosis (Fig. 3j and k). Although the MCODE score of cluster 38 was <3, the module was selected for analysis due to the 8 nodes and 10 edges that might reflect significant function and pathways in the disease. Treatment with probiotic formulations might reverse any metabolic abnormality upon SARS-CoV-2 infections by releasing SCFAs. The molecular events and pathways indicated by MCODE clusters were supported by the published scientific shreds of evidence available for SARS-CoV-2 infection and pathogenesis, which elucidated the promising yet unexplored functional pathways of these genes.
GO Enrichment Analysis to Assess the Overrepresentation of Gene Ontology Categories by the BiNGO
The enrichment or overrepresented GO terms associated with the MCODE cluster candidate genes, designated by p values in BiNGO enrichment analysis, are detailed in table S2. The analysis showed that cluster 1, cluster 2, cluster 3, and cluster 5 involved the pathways related to viral infection, pathogen-associated molecular patterns (PAMPs)–mediated immune response to extracellular signals, stress, leukocyte migration, activation of cellular oxidative stress and inflammatory processes through phosphate metabolic processes, regulation of MAPKKK cascade, and jun kinase activity. Therefore, these physiological events associated with SARS-CoV-2 infections could be modulated by probiotic supplementation. Probiotics could heal the intestinal barriers and minimize viral entry, as well as balance the immune responses and inflammatory conditions. Cluster 6, cluster 9, cluster 10, cluster 11, and cluster 13 were involved in the regulation of innate immune cell differentiation, regulation of glucose and lipid transport, cholesterol efflux, glycerol, glycerolipid, acylglycerol, triglyceride, TNF, and cytokine signaling by JAK-STAT pathway in addition to regulation of renin-angiotensin–mediated blood pressure. Probiotic supplementation could play an immense role in immune cell development and differentiation, regulation of inflammatory processes, metabolic activity, and the regulation of blood pressure through RAS. Cluster 35 and cluster 38 genes were involved in lipid, cholesterol, steroid, glucose, and monosaccharide metabolism, xenobiotic drug transport, regulation of monocyte and neutrophil differentiation, and response to cytokine IL6. This enrichment analysis of the GO terms of candidate genes enlightened us on the major biological processes associated with SARS-CoV-2 infection pathogenesis. It provided the cellular domains for antiviral probiotic mechanisms to defeat the viral infection. Several research groups have used similar BiNGO enrichment analysis to understand breast cancer susceptibility or to detect key pathways responsible for gastric cancer or analyze the protein-protein network [12].
Discussion
The successful application of probiotics in respiratory infections of human clinical trials encourages us to study the probiotics application to alleviate SARS-CoV-2 infection. The meta-analysis is an epidemiological study design that quantitatively examines the available outcomes and estimates the effect of treatment much precisely than a single study contributing to the pooled analysis. In this study, by using a systematic network and meta-analysis, we provide a promising probiotic mechanism that can reinforce the immunity and mitigate the SARS-CoV-2 infection.
Probiotics Could Confine the Virus Entry and Reduce the Adverse Effects Caused by Antiviral Immune Activation and Dysregulation of RAS
ACE2 receptor is a master regulator of RAS and blood pressure. However, SARS-CoV-2 enters the host enterocytes and lung tissues through ADAM17-mediated proteolytic cleavage of the ACE2 receptor [53]. The disruption of the protective “gut-lung axis” increases the propensity of the infection in the underlying ACE2 containing epithelial cells. SARS-CoV-2 infection may downregulate cellular ACE2 expression with the resulting accumulation of angiotensin II (Ang II). Therefore, infected lung alveolar and small intestinal epithelial cells, heart, kidney, vascular endothelial, and smooth muscle cells (highly expressing ACE2) cause vasoconstriction, tissue inflammation, and oxidative stress, which worsen the health conditions of COVID-19 patients [53].
Our functional annotations and enrichment analysis of candidate genes of MCODE cluster 10 (ace2, mas1, ace, ang, agtr1, il22, il2, gli2, cd4, cd40lg, fus, th1l, pdgfb) and MCODE cluster 13 (hif1a, dlk1, adam17, epas1) (Fig. 3g and i; Table S3) indicate that probiotics could have potential roles in limiting viral entry through ADAM17-ACE2–mediated receptor endocytosis. Further enrichment analysis shows that the application of probiotics could mitigate the adverse effects of dysregulated RAS system, hif1a-mediated oxidative stress [54], activated immune cells (monocytes and NK cells), and elevated proinflammatory cytokines.
Probiotics are known to heal the damaged epithelial barrier and thereby protect the underlying ACE2 receptor-expressing cells [55]. Bacteriocin-producing probiotic strains can colonize in the GI tract better than bacteriocin non-producers. Therefore, bacteriocins also play a crucial role in microbial colonization [11]. Probiotics have been reported to reduce nitric oxide (NO) production [56], hypertension [57] and oxidative stress [58], and secrete ACE-inhibitory peptides [59], SCFAs (acetate, propionate, and butyrate) to induce anti-inflammation and control the blood pressure [60]. Recombinant probiotic L. paracasei in conjugation with ACE2 has been applied for the cardiovascular protective role of ACE2 in mice [61]. Additionally, cinnamycin-like lantibiotics (bacteriocins) mediate inflammatory responses by inactivating phospholipase A2. One such lantibiotic ancovenin is reported to inhibit ACE, thereby preventing the conversion of Ang I to Ang II and regulate the blood pressure [11].
Probiotics Could Play a Protective Role in Cytokine Storm and Lung Injury Caused by SARS-CoV-2 Infection
Frequent interaction with the external environment in terms of food and air alters the microbial composition of intestinal microbiota as well as lungs. Several GI disorders also have manifestations of respiratory complications. The high expression of ACE2 receptor in gut and lung epithelial cells with the prevalence of respiratory and GI tract infections in COVID-19 patients strongly suggests the bi-directional cross-talk of the “gut-lung axis” [7]. After entry, the viral RNAs activate the cellular innate immune system (TLRs) [62] and inflammatory pathways (NLRP3 and NF-κB) as a protective mechanism. Activated TLRs promote first-line antiviral response through MYD88 and IRF3/7-mediated type-I IFN production [63, 64]. The NLRP3 inflammasome complex assists in the secretion of proinflammatory IL1b and IL18, which activates T-cells or macrophages to secrete IL6 and TNFα. The released proinflammatory cytokines (IL1B, IL18, IL6, TNFα) further convert naive T-cells to Th1/CTLs/CD8+ or Th17 and triggers the secretion of proinflammatory IFNγ and IL17. Therefore, the gut microbiome controls innate and adaptive immune responses against the respiratory pathogens [7]. However, the NF-κB pathway can be turned on in either of two ways: activated NLRP3 or TLR4 and stress-induced MAPK signaling (ERK1/2, JNKs, and p38/MAPK14) pathway [65, 66]. The activated NF-κB pathway contributes to proinflammatory cytokine secretion and apoptosis in enterocytes and lung tissues. The resulting tissue injury activates local circulatory innate immune cells and establishes a proinflammatory feed-forward loop of cytokines called cytokine storm [65]. The surge of cytokines and chemokines induces VEGF, IL8, and additional IL6 and reduces E-cadherin expression on endothelial cells ensuing vascular permeability, while it also elicits leukocyte trafficking and migration of monocytes, neutrophils, NK cells, macrophages, and dendritic cells in the lung cells. The resulting immune cells and cytokines induce hyaluronan synthesis, which participates in the pathophysiology of ARDS, the hallmark of SARS-CoV-2 infection [67]. The “gut-lung axis” facilitates the migration of immune cells from the gut to the lung through circulation and enforces immunity in health and disease. Therefore, the immunological coordination between the gut and lung affects each other’s homeostasis and influences the acquired immunity in health and disease of the host.
Our functional annotations and enrichment analysis of MCODE cluster 5 candidate genes (tlr2, tlr4, nlrp3, myd88, hmgb1, mapk14, creb1, rps6ka5, jun, ephb2, mapk8, atf2, fos, maa) indicate that probiotics could be associated with TLR-mediated innate immune response during SARS-CoV-2 infection. TLR-3, 7/8, reported to recognize the SARS-CoV-2 RNA, was also present in the association network but not in the MCODE cluster (Fig. 3d; Table S4). The presence of nlrp3 in cluster 5 hints the activation of inflammatory and antiviral IFNs which is further supported by cluster 2 (tnf, il6, map1lc3b, ros1, cd38, bax, fas, sqstm1, il10, fcgr3a, cybb, il32, bcl2, icam1) and cluster 3 (cxcl5, cxcl12, tnc, has2, cxcl6, mmrn1, mmp13, smoxlus, rela, ccl2, chi3l1, csf2, il5, il25, klk15, lcn2, il18, vegfa, eng, plau, pgf, prl, angpt2, igfbp1, hgf, erbb2, fgf2, stat3, stat5, ifna1, soat1, cd68, il9, itgam, ptprc, cd14) candidate genes (Fig. 3b and c; Table S5). The highest node degree of IL6 and all the cytokine and chemokine genes confirm their significant participation indicating reported elevation in the immunopathology of COVID-19. Additionally, apoptotic genes like bax, bcl2, fas, and cybb may indicate TNFα, IL6-mediated programmed cell death of T-cells depicting the observed global lymphocytopenia in the COVID-19 patients [68]. However, receptors like cd68 (NK cells), cd14 (monocytes), and has2 gene may depict the rapid migration and infiltration of innate immune cells in the lung tissue with injury in response to chemoattractant chemokines. The candidate genes of cluster 9 (il27ra, il27, cxcl10, il17d, il6st, il6r, jak2, ptpn18, mapk3, epo, akt1, egfr, reg1a, ptpn1, cat, frap1, malat1, acan, eif4ebp1, smad4, smad6, smad2, smad7) and cluster 1 (src, limk1, rps6ka3, aak1, mapk1, mknk2, map3k1, gak, map2k1, fgfr1, mapkapk5, map3k7, bmp2k, zak, gsk3, yes1) show the involvement of MAPK signaling and proinflammatory cytokine-induced growth regulatory pathways (Figs. 3f and S1; Table S6). Therefore, our analysis indicates that probiotics could play a protective role in the above-mentioned signaling pathways.
Probiotics protect the intestinal barrier by inhibiting cytokine-induced intestinal epithelial cell apoptosis [69]. A probiotics mixture consisting of L. acidophilus, L. casei, L. reuteri, B. bifidium, and Streptococcus thermophilus is reported to induce both T-cell and B-cell hyporesponsiveness and downregulate T helper (Th) Th1, Th2, and Th17 cytokines without inducing apoptosis [70]. Further, probiotic-derived molecules (i.e., bacteriocins) are also known to trigger CD4(+) and CD8(+) T cell activation and modulate interleukin production [11]. Thus, probiotics could have a promising role against lymphocytopenia or reduction of T cell count, which is observed in COVID-19 patients. Moreover, probiotic formulations can balance pro- and anti-inflammatory cytokine secretion, which is the key factor for robust immune (adaptive and innate) system. Although few reports are available on the immunomodulatory effects of bacteriocins, the activity of nisin, nisaplin, and pediocin is well documented [11]. Several clinical trials on URTI individuals have reported the decreased proinflammatory and increased anti-inflammatory cytokine upon probiotic application. Limited search has been done, which reveals the ability of probiotics to degrade glycosaminoglycan molecules [71] that may result in reduced expression of has2 and hyaluronan breakdown. Probiotics are also proven to downregulate the NF-κB signaling pathway by regulating MAPK and ERK pathways reducing systemic inflammation [72].
Probiotics Could Improve Cardiovascular Complications and Lipidomic Abnormalities
COVID-19 comorbidities like obesity, metabolic syndrome (e.g., IBD), are associated with an inadequate gut-microbial composition linked to disruption of the bi-directional communication network in the gut-heart axis. Such dysbiosis reduces gut-barrier integrity, accelerates intestinal inflammation, and contributes as an extragenomic factor to cardiovascular complications [73]. Dysregulation of RAS due to SARS-CoV-2 entry promotes inflammation, vasoconstriction, hypertrophy, proliferation, and fibrosis, all factors that contribute to the development and progression of cardiopulmonary diseases [74]. Chronic elevation of IL6 and cytokine storm promotes macrophages to release MCP-1, which aids atherogenesis, expression of cell adhesion molecules, and proliferation and migration of vascular smooth muscle cells resulting in cardiovascular diseases (CVDs) (e.g., coronary atherosclerosis, inflammation in the vascular system, and thrombosis) [75]. Enhanced angiotensin II also causes EGFR transactivation-induced vascular remodeling [76]. The induction of CVDs further functionally damages intestinal epithelial cells (IECs), which, together with barrier dysfunction, allows translocation of gut bacteria–derived endotoxins (lipopolysaccharide or LPS) and metabolites into the circulation. The recognition of LPS by receptors like TLR4 on cardiomyocytes induces tissue inflammation and increases cytokine secretion (TNFα). The pathophysiology of coronary atherosclerosis is also associated with aberrant production of metabolites like betaine, choline, and TMAO by microbial digestion. These metabolites and LPS affect cholesterol and lipoprotein (LDL) metabolic pathways, which amplifies inflammation and worsen SARS-CoV-2 infection [73].
The candidate genes encoding growth factors of MCODE cluster 3 (vegfa, eng, plau, pgf, prl, angpt2, igfbp1, hgf, erbb2, fgf2, stat3, stat5) along with overrepresentation of GO terms indicates the role of probiotics in the mitigation of elevated angiotensin II-induced cardiovascular complications (Fig. 3c; Table S5). The functional annotations and enrichment analysis of the candidate genes of MCODE cluster 6 (dgat2, adipor2, dgat1, pgc, adipoq), cluster 35 (pparg, twist1, lpa, slc12a, mbtps1, srebf1, mbtps2), and cluster 38 (hk2, nr1i2, foxa2, pik3ca, f10, inpp5d, hspb1, mcl1) indicates that probiotics could have potential roles in glucose and cholesterol or lipoprotein metabolism (Fig. 3e, j, k; Table S7). The lipidomic and cholesterol metabolic abnormalities due to SARS-CoV-2 infection are poorly documented. The limited reports indicate enhanced glucose and lipid need for viral replication and metabolism since viruses hijack the host’s metabolic processes. The overrepresentation of GO terms associated with lipid and cholesterol metabolism reflects the lipid-demanding processes (viral replication, endocytosis, and exocytosis) involved in SARS-CoV-2 infection [77]. They also indicate cardiovascular complications due to gut-dysbiosis and COVID-19.
Probiotic supplementation has been proved to reduce total cholesterol, LDL, and triglycerides and increase HDL count. The antihypertension capability of the probiotics makes them an affordable and adjunctive treatment option in hypertension, diabetes, and cardiovascular diseases, and other dyslipidemia-associated health issues [78]. SCFAs released by gut bacteria and probiotics are reported to regulate blood pressure (BP). Receptors like GPR41 and Olfr78 are responsible for hypo- and hypertensive effects of SCFAs, respectively [73]. A number of LAB probiotic strains are reported to exhibit antiviral activity by producing bacteriocins, which mostly inhibit viral replication that indicates their potential role in reducing SARS-CoV-2 infection [11].
Conclusion
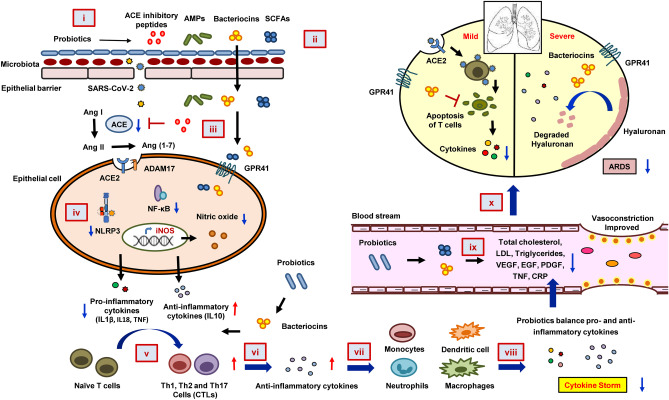
Our systematic network and meta-analysis study aid us to propose a mechanistic model of probiotic actions in the alleviation of COVID-19 (Fig. 4). Probiotic consumption could reduce the propensity of viral entry by healing the ACE2 containing epithelial barrier. The antimicrobial peptides or bacteriocins, SCFAs, and ACE inhibitory peptides released by beneficial bacteria could balance the dysregulated RAS. Hence, the blood pressure or cardiovascular complications can be mitigated. The reduced NO production could decrease cellular oxidative stress. This can lead to the downregulation of inflammatory (NLRP3 and NF-κB) pathways and eventually, the secretion of proinflammatory cytokines or chemokines. Probiotics and bacteriocins might balance pro- and anti-inflammatory cytokine levels and increase the T-cell count in the SARS-CoV-2-infected patients. Finally, probiotics might also degrade the hyaluronan and hence could improve ARDS. Therefore, probiotics could be considered a potential preventive and alternative treatment strategy for both mild and severe stages of COVID-19. However, further research is required for elucidation.
Fig. 4.
The mechanistic model of probiotics action against COVID-19. Probiotics induce a stronger epithelial barrier that prevents viral entry through the gut (i), probiotics modulate gut microbiota and induce the synthesis of SCFAs that regulate blood pressure and inflammation (ii). Probiotics also release ACE-inhibitory peptides that could reduce angiotensin II (Ang II) expression, thereby inhibiting viral entry into the cell (iii). Probiotics induce anti-inflammation by suppressing NF-κb signaling and reducing the levels of IL1β, IL18, NO, and TNF (iv). Bacteriocin and other anti-, as well as proinflammatory cytokines produced by the effects of probiotics modulate Th1, Th2, and Th17 cells (v). Which, in turn, help in the production of more anti-inflammatory cytokines (vi). The anti-inflammatory cytokines regulate monocytes, macrophages, dendritic cells, and neutrophils (vii) to downregulate SARS-CoV-2 infection–mediated cytokine storm (viii), resulting in decreased total cholesterol, LDL, triglycerides, VEGF, EGF, PDGF, TNF, and CRP level in the blood stream (ix). The reduced cytokine storm and inflammation exerted by probiotics cause the reduction in hyaluronan synthesis, which eventually could improve the ARDS condition in SARS-CoV-2 infection (x)
Supplementary Information
Below is the link to the electronic supplementary material.
Abbreviations
- COVID-19
Coronavirus disease of 2019
- SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2
- PRISMA
Preferred reporting items for systematic reviews and meta-analyses
- ACE2
Angiotensin-converting enzyme 2
- WHO
World Health Organization
- SARS-CoV
Severe acute respiratory syndrome coronavirus
- MERS-CoV
Middle East respiratory syndrome coronavirus
- URTI
Upper respiratory tract infection
- ARDS
Acute respiratory distress syndrome
- MCODE
Molecular complex detection
- BiNGO
Biological Networks Gene Ontology tool
- IL
Interleukin
- FOS
Fructooligosaccharide
Funding
This study was partially supported by the intramural funding provided by IIT Bhubaneswar. S.P. and S.S. receive Institutional fellowship from IIT Bhubaneswar. B.P. is supported by the endowment grant to IIT Bhubaneswar from Dr. Dash Foundation, USA.
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declarations
Competing Interests
The authors declare that there is no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12602-021-09748-w.
References
- 1.Catanzaro M, Fagiani F, Racchi M, Corsini E, Govoni S, et al. Immune response in COVID-19: addressing a pharmacological challenge by targeting pathways triggered by SARS-CoV-2. Signal Transduct Target Ther. 2020;5(1):1–10. doi: 10.1038/s41392-020-0191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu YC, Chen CS, Chan YJ. The outbreak of COVID-19: an overview. J Chin Med Assoc. 2020;83(3):217–220. doi: 10.1097/JCMA.0000000000000270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhu N, Zhang D, Wang W, Li X, Yang B, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kumar S, Zhi K, Mukherji A, Gerth K. Repurposing antiviral protease inhibitors using extracellular vesicles for potential therapy of COVID-19. Viruses. 2020;12(5):486–501. doi: 10.3390/v12050486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dhar D, Mohanty A. Gut microbiota and Covid-19-possible link and implications. Virus Res. 2020;37:198018–198024. doi: 10.1016/j.virusres.2020.198018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hanada S, Pirzadeh M, Carver KY, Deng JC. Respiratory viral infection-induced microbiome alterations and secondary bacterial pneumonia. Front Immunol. 2018;9:2640–2655. doi: 10.3389/fimmu.2018.02640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahlawat S, Sharma KK. Immunological co-ordination between gut and lungs in SARS-CoV-2 infection. Virus Res. 2020;37:198103–198114. doi: 10.1016/j.virusres.2020.198103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu K, Cai H, Shen Y, Ni Q, Chen Y, et al. Management of corona virus disease-19 (COVID-19): the Zhejiang experience. J Zhejiang Univ Sci B. 2020;49(1):147–157. doi: 10.3785/j.issn.1008-9292.2020.02.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kanauchi O, Andoh A, AbuBakar S, Yamamoto N. Probiotics and paraprobiotics in viral infection: clinical application and effects on the innate and acquired immune systems. Curr Pharm Des. 2018;24(6):710–717. doi: 10.2174/1381612824666180116163411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Baud D, Agri VD, Gibson GR, Reid G, Giannoni E. Using probiotics to flatten the curve of coronavirus disease COVID-2019 pandemic. Front Public Health. 2020;8:1–5. doi: 10.3389/fpubh.2020.00186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tiwari SK, Dicks LM, Popov IV, Karaseva A, Ermakov AM, et al. Probiotics at war against viruses: what is missing from the picture? Front Microbiol. 2020;11:1877–1898. doi: 10.3389/fmicb.2020.01877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guo L, Song C, Wang P, Dai L, Zhang J, et al. A systems biology approach to detect key pathways and interaction networks in gastric cancer on the basis of microarray analysis. Mol Med Rep. 2015;12(5):7139–7145. doi: 10.3892/mmr.2015.4242. [DOI] [PubMed] [Google Scholar]
- 13.Laursen RP, Larnkjær A, Ritz C, Hauger H, Michaelsen KF, et al. Probiotics and child care absence due to infections: a randomized controlled trial. Pediatrics. 2017;140(2):486–501. doi: 10.1542/peds.2017-0735. [DOI] [PubMed] [Google Scholar]
- 14.Leyer GJ, Li S, Mubasher ME, Reifer C, Ouwehand AC. Probiotic effects on cold and influenza-like symptom incidence and duration in children. Pediatrics. 2009;124(2):e172–e179. doi: 10.1542/peds.2008-2666. [DOI] [PubMed] [Google Scholar]
- 15.Pregliasco F, Anselmi G, Fonte L, Giussani F, Schieppati S, et al. A new chance of preventing winter diseases by the administration of synbiotic formulations. J Clin Gastroenterol. 2008;42:S224–S233. doi: 10.1097/MCG.0b013e31817e1c91. [DOI] [PubMed] [Google Scholar]
- 16.Hojsak I, Tokić Pivac V, Močić Pavić A, Pasini AM, Kolaček S. Bifidobacterium animalis subsp. lactis fails to prevent common infections in hospitalized children: a randomized, double-blind, placebo-controlled study. Am J Clin Nutr. 2015;101(3):680–684. doi: 10.3945/ajcn.114.102004. [DOI] [PubMed] [Google Scholar]
- 17.Hojsak I, Pavić AM, Kos T, Dumančić J, Kolaček S. Bifidobacterium animalis subsp. lactis in prevention of common infections in healthy children attending day care centers–randomized, double blind, placebo-controlled study. Clin Nutr. 2016;35(3):587–591. doi: 10.1016/j.clnu.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 18.Li KL, Wang BZ, Li ZP, Li YL, Liang JJ. Alterations of intestinal flora and the effects of probiotics in children with recurrent respiratory tract infection. World J Pediatr. 2019;15(3):255–261. doi: 10.1007/s12519-019-00248-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cazzola M, Pham-Thi N, Kerihuel JC, Durand H, Bohbot S. Efficacy of a synbiotic supplementation in the prevention of common winter diseases in children: a randomized, double-blind, placebo-controlled pilot study. Ther Adv Respir Dis. 2010;4(5):271–278. doi: 10.1177/1753465810379010. [DOI] [PubMed] [Google Scholar]
- 20.Garaiova I, Muchová J, Nagyová Z, Wang D, Li J, et al. Probiotics and vitamin C for the prevention of respiratory tract infections in children attending preschool: a randomised controlled pilot study. Eur J Clin Nutr. 2015;69(3):373–379. doi: 10.1038/ejcn.2014.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gerasimov S, Ivantsiv V, Bobryk L, Tsitsura O, Dedyshin L, et al. Role of short-term use of L. acidophilus DDS-1 and B. lactis UABLA-12 in acute respiratory infections in children: a randomized controlled trial. Eur J Clin Nutr. 2016;70(4):463–469. doi: 10.1038/ejcn.2015.171. [DOI] [PubMed] [Google Scholar]
- 22.Strasser B, Geiger D, Schauer M, Gostner JM, Gatterer H, et al. Probiotic supplements beneficially affect tryptophan–kynurenine metabolism and reduce the incidence of upper respiratory tract infections in trained athletes: a randomized, double-blinded, placebo-controlled trial. Nutrients. 2016;8(11):752–767. doi: 10.3390/nu8110752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jespersen L, Tarnow I, Eskesen D, Morberg CM, Michelsen B, et al. Effect of Lactobacillus paracasei subsp. paracasei, L. casei 431 on immune response to influenza vaccination and upper respiratory tract infections in healthy adult volunteers: a randomized, double-blind, placebo-controlled, parallel-group study. Am J Clin Nutr. 2015;101(6):1188–1196. doi: 10.3945/ajcn.114.103531. [DOI] [PubMed] [Google Scholar]
- 24.Pu F, Guo Y, Li M, Zhu H, Wang S, et al. Yogurt supplemented with probiotics can protect the healthy elderly from respiratory infections: a randomized controlled open-label trial. Clin Interv Aging. 2017;12:1223–1231. doi: 10.2147/CIA.S141518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Corsello G, Carta M, Marinello R, Picca M, De Marco G, et al. Preventive effect of cow’s milk fermented with Lactobacillus paracasei CBA L74 on common infectious diseases in children: a multicenter randomized controlled trial. Nutrients. 2017;9(7):669–684. doi: 10.3390/nu9070669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nocerino R, Paparo L, Terrin G, Pezzella V, Amoroso A, et al. Cow’s milk and rice fermented with Lactobacillus paracasei CBA L74 prevent infectious diseases in children: a randomized controlled trial. Clin Nutr. 2017;36(1):118–125. doi: 10.1016/j.clnu.2015.12.004. [DOI] [PubMed] [Google Scholar]
- 27.Berggren A, Ahrén IL, Larsson N, Önning G. Randomised, double-blind and placebo-controlled study using new probiotic lactobacilli for strengthening the body immune defence against viral infections. Eur J Nutr. 2011;50(3):203–210. doi: 10.1007/s00394-010-0127-6. [DOI] [PubMed] [Google Scholar]
- 28.Szymański H, Armańska M, Kowalska-Duplaga K, Szajewska H. Bifidobacterium longum PL03, Lactobacillus rhamnosus KL53A, and Lactobacillus plantarum PL02 in the prevention of antibiotic-associated diarrhea in children: a randomized controlled pilot trial. Digestion. 2008;78(1):13–17. doi: 10.1159/000151300. [DOI] [PubMed] [Google Scholar]
- 29.Hirose Y, Yamamoto Y, Yoshikai Y, Murosaki S. Oral intake of heat-killed Lactobacillus plantarum L-137 decreases the incidence of upper respiratory tract infection in healthy subjects with high levels of psychological stress. J Nutr Sci. 2013;2:1–8. doi: 10.1017/jns.2013.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tubelius P, Stan V, Zachrisson A. Increasing work-place healthiness with the probiotic Lactobacillus reuteri: a randomised, double-blind placebo-controlled study. Environ Health. 2005;4(1):1–5. doi: 10.1186/1476-069X-4-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kukkonen K, Savilahti E, Haahtela T, Juntunen-Backman K, Korpela R, et al. Long-term safety and impact on infection rates of postnatal probiotic and prebiotic (synbiotic) treatment: randomized, double-blind, placebo-controlled trial. Pediatrics. 2008;122(1):8–12. doi: 10.1542/peds.2007-1192. [DOI] [PubMed] [Google Scholar]
- 32.Hojsak I, Snovak N, Abdović S, Szajewska H, Mišak Z, et al. Lactobacillus GG in the prevention of gastrointestinal and respiratory tract infections in children who attend day care centers: a randomized, double-blind, placebo-controlled trial. Clin Nutr. 2010;29(3):312–316. doi: 10.1016/j.clnu.2009.09.008. [DOI] [PubMed] [Google Scholar]
- 33.Hojsak I, Abdović S, Szajewska H, Milošević M, Krznarić Ž, et al. Lactobacillus GG in the prevention of nosocomial gastrointestinal and respiratory tract infections. Pediatrics. 2010;125(5):e1171–e1177. doi: 10.1542/peds.2009-2568. [DOI] [PubMed] [Google Scholar]
- 34.Kumpu M, Kekkonen R, Kautiainen H, Järvenpää S, Kristo A, et al. Milk containing probiotic Lactobacillus rhamnosus GG and respiratory illness in children: a randomized, double-blind, placebo-controlled trial. Eur J Clin Nutr. 2012;66(9):1020–1023. doi: 10.1038/ejcn.2012.62. [DOI] [PubMed] [Google Scholar]
- 35.Smith TJ, Rigassio-Radler D, Denmark R, Haley T, Touger-Decker R. Effect of Lactobacillus rhamnosus LGG® and Bifidobacterium animalis ssp. lactis BB-12® on health-related quality of life in college students affected by upper respiratory infections. Br J Nutr. 2013;109(11):1999–2007. doi: 10.1017/S0007114512004138. [DOI] [PubMed] [Google Scholar]
- 36.Wang B, Hylwka T, Smieja M, Surrette M, Bowdish DM, et al. Probiotics to prevent respiratory infections in nursing homes: a pilot randomized controlled trial. J Am Geriatr Soc. 2018;66(7):1346–1352. doi: 10.1111/jgs.15396. [DOI] [PubMed] [Google Scholar]
- 37.Shida K, Sato T, Iizuka R, Hoshi R, Watanabe O, et al. Daily intake of fermented milk with Lactobacillus casei strain Shirota reduces the incidence and duration of upper respiratory tract infections in healthy middle-aged office workers. Eur J Nutr. 2017;56(1):45–53. doi: 10.1007/s00394-015-1056-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fujita R, Iimuro S, Shinozaki T, Sakamaki K, Uemura Y, et al. Decreased duration of acute upper respiratory tract infections with daily intake of fermented milk: a multicenter, double-blinded, randomized comparative study in users of day care facilities for the elderly population. Am J Infect Control. 2013;41(12):1231–1235. doi: 10.1016/j.ajic.2013.04.005. [DOI] [PubMed] [Google Scholar]
- 39.Guillemard E, Tondu F, Lacoin F, Schrezenmeir J. Consumption of a fermented dairy product containing the probiotic Lactobacillus casei DN-114 001 reduces the duration of respiratory infections in the elderly in a randomised controlled trial. Br J Nutr. 2010;103(1):58–68. doi: 10.1017/S0007114509991395. [DOI] [PubMed] [Google Scholar]
- 40.Guillemard E, Tanguy J, Flavigny AL, de la Motte S, Schrezenmeir J. Effects of consumption of a fermented dairy product containing the probiotic Lactobacillus casei DN-114 001 on common respiratory and gastrointestinal infections in shift workers in a randomized controlled trial. J Am Coll Nutr. 2010;29(5):455–468. doi: 10.1080/07315724.2010.10719882. [DOI] [PubMed] [Google Scholar]
- 41.Wang X, Wang L, Huang X, Ma S, Yu M, et al. Oral delivery of probiotics expressing dendritic cell-targeting peptide fused with porcine epidemic diarrhea virus COE antigen: a promising vaccine strategy against PEDV. Viruses. 2017;9(11):312–328. doi: 10.3390/v9110312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chai W, Burwinkel M, Wang Z, Palissa C, Esch B, et al. Antiviral effects of a probiotic Enterococcus faecium strain against transmissible gastroenteritis coronavirus. Arch Virol. 2013;158(4):799–807. doi: 10.1007/s00705-012-1543-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Romano L, Bilotta F, Dauri M, Macheda S, Pujia A et al (2020) Short report–medical nutrition therapy for critically ill patients with COVID-19. Eur Rev Med Pharmacol Sci 24(7):4035‒4039. 10.26355/eurrev_202004_20874 [DOI] [PubMed]
- 44.Young RP, Hopkins RJ, Marsland B. The gut–liver–lung axis. Modulation of the innate immune response and its possible role in chronic obstructive pulmonary disease. Am J Respir Cell Mol Biol. 2016;54(2):161–169. doi: 10.1165/rcmb.2015-0250PS. [DOI] [PubMed] [Google Scholar]
- 45.Akour A. Probiotics and COVID-19: is there any link? Lett Appl Microbiol. 2020;71:1–6. doi: 10.1111/lam.13334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Di Renzo L, Merra G, Esposito E, De Lorenzo A (2020) Are probiotics effective adjuvant therapeutic choice in patients with COVID-19?. Eur Rev Med Pharmacol Sci 24(8):4062‒4063. 10.26355/eurrev_202004_20977 [DOI] [PubMed]
- 47.Shi Y, Wang Y, Shao C, Huang J, Gan J, et al. COVID-19 infection: the perspectives on immune responses. Cell Death Differ. 2020;27:1451–1454. doi: 10.1038/s41418-020-0530-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bernabò N, Ordinelli A, Ramal Sanchez M, Mattioli M, Barboni B. Networks models of actin dynamics during spermatozoa postejaculatory life: a comparison among human-made and text mining-based models. Biomed Res Int. 2016;2016:1–9. doi: 10.1155/2016/9795409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu XX, Bi JP, Ping L, Li P, Li F. A network pharmacology approach to determine the synergetic mechanisms of herb couple for treating rheumatic arthritis. Drug Des Devel Ther. 2018;12:967–979. doi: 10.2147/DDDT.S161904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Priyadarshini S, Aich P. Effects of psychological stress on innate immunity and metabolism in humans: a systematic analysis. PLoS ONE. 2012;7(9):e43232. doi: 10.1371/journal.pone.0043232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bader GD, Hogue CW. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics. 2003;4(1):2–28. doi: 10.1186/1471-2105-4-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fang HT, Farran CAE, Xing QR, Zhang LF, Li H, et al. Global H3. 3 dynamic deposition defines its bimodal role in cell fate transition. Nat Commun. 2018;9(1):1–17. doi: 10.1038/s41467-018-03904-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bourgonje AR, Abdulle AE, Timens W, Hillebrands JL, Navis GJ, et al. Angiotensin-converting enzyme-2 (ACE2), SARS-CoV-2 and pathophysiology of coronavirus disease 2019 (COVID-19) J Pathol. 2020;251:228–248. doi: 10.1002/path.5471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wilkerson RG, Adler JD, Shah NG, Brown R. Silent hypoxia: a harbinger of clinical deterioration in patients with COVID-19. Am J Emerg Med. 2020;38:2243.e2245–2243.e2246. doi: 10.1016/j.ajem.2020.05.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lukic J, Chen V, Strahinic I, Begovic J, Lev-Tov H, et al. Probiotics or pro-healers: the role of beneficial bacteria in tissue repair. Wound Repair Regen. 2017;25(6):912–922. doi: 10.1111/wrr.12607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kang CH, Han SH, Kim JS, Kim Y, Jeong Y, et al. Inhibition of nitric oxide production, oxidative stress prevention, and probiotic activity of lactic acid bacteria isolated from the human vagina and fermented food. Microorganisms. 2019;7(4):109–119. doi: 10.3390/microorganisms7040109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Dong JY, Szeto IM, Makinen K, Gao Q, Wang J, et al. Effect of probiotic fermented milk on blood pressure: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1188–1194. doi: 10.1017/S0007114513001712. [DOI] [PubMed] [Google Scholar]
- 58.Mikelsaar M, Zilmer M. Lactobacillus fermentum ME-3–an antimicrobial and antioxidative probiotic. Microb Ecol Health Dis. 2009;21(1):1–27. doi: 10.1080/08910600902815561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ramchandran L, Shah NP. Proteolytic profiles and angiotensin-I converting enzyme and α-glucosidase inhibitory activities of selected lactic acid bacteria. J Food Sci. 2008;73(2):M75–M81. doi: 10.1111/j.1750-3841.2007.00643.x. [DOI] [PubMed] [Google Scholar]
- 60.Daliri EB-M, Lee BH, Oh DH. Current perspectives on antihypertensive probiotics. Probiotics Antimicrob Proteins. 2017;9(2):91–101. doi: 10.1007/s12602-016-9241-y. [DOI] [PubMed] [Google Scholar]
- 61.Verma A, Xu K, Du T, Zhu P, Liang Z, et al. Expression of human ACE2 in Lactobacillus and beneficial effects in diabetic retinopathy in mice. Mol Ther Methods Clin Dev. 2019;14:161–170. doi: 10.1016/j.omtm.2019.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.de Marcken M, Dhaliwal K, Danielsen AC, Gautron AS, Dominguez-Villar M. TLR7 and TLR8 activate distinct pathways in monocytes during RNA virus infection. Sci Signal. 2019;12(605):1–19. doi: 10.1126/scisignal.aaw1347. [DOI] [PubMed] [Google Scholar]
- 63.Zhao C, Zhao W. NLRP3 inflammasome—a key player in antiviral responses. Front Immunol. 2020;11:211–219. doi: 10.3389/fimmu.2020.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Olejnik J, Hume AJ, Mühlberger E. Toll-like receptor 4 in acute viral infection: too much of a good thing. PLoS Pathog. 2018;14(12):e1007390. doi: 10.1371/journal.ppat.1007390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jamilloux Y, Henry T, Belot A, Viel S, Fauter M, et al. Should we stimulate or suppress immune responses in COVID-19? cytokine and anti-cytokine interventions. Autoimmun Rev. 2020;19:102567–102581. doi: 10.1016/j.autrev.2020.102567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Liu Y, Shepherd EG, Nelin LD. MAPK phosphatases—regulating the immune response. Nat Rev Immunol. 2007;7(3):202–212. doi: 10.1038/nri2035. [DOI] [PubMed] [Google Scholar]
- 67.Bell TJ, Brand OJ, Morgan DJ, Salek-Ardakani S, Jagger C, et al. Defective lung function following influenza virus is due to prolonged, reversible hyaluronan synthesis. Matrix Biol. 2019;80:14–28. doi: 10.1016/j.matbio.2018.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kivela P. Paradigm shift for COVID-19 response: identifying high-risk individuals and treating inflammation. West J Emerg Med. 2020;21(3):473–476. doi: 10.5811/westjem.2020.3.47520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yan F, Polk DB. Probiotic bacterium prevents cytokine-induced apoptosis in intestinal epithelial cells. J Biol Chem. 2002;277(52):50959–50965. doi: 10.1074/jbc.M207050200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yan F, Polk D. Probiotics and immune health. Curr Opin Gastroenterol. 2011;27(6):496–501. doi: 10.1097/MOG.0b013e32834baa4d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kawai K, Kamochi R, Oiki S, Murata K, Hashimoto W. Probiotics in human gut microbiota can degrade host glycosaminoglycans. Sci Rep. 2018;8(1):1–13. doi: 10.1038/s41598-018-28886-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Dai C, Zhao DH, Jiang M. VSL# 3 probiotics regulate the intestinal epithelial barrier in vivo and in vitro via the p38 and ERK signaling pathways. Int J Mol Med. 2012;29(2):202–208. doi: 10.3892/ijmm.2011.839. [DOI] [PubMed] [Google Scholar]
- 73.Ahlawat S, Sharma KK. Gut–organ axis: a microbial outreach and networking. Lett Appl Microbiol. 2020;71:1–33. doi: 10.1111/lam.13333. [DOI] [PubMed] [Google Scholar]
- 74.Cole-Jeffrey CT, Liu M, Katovich MJ, Raizada MK, Shenoy V. ACE2 and microbiota: emerging targets for cardiopulmonary disease therapy. J Cardiovasc Pharmacol. 2015;66(6):540–550. doi: 10.1097/FJC.0000000000000307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wainstein MV, Mossmann M, Araujo GN, Gonçalves SC, Gravina GL, et al. Elevated serum interleukin-6 is predictive of coronary artery disease in intermediate risk overweight patients referred for coronary angiography. Diabetol Metab Syndr. 2017;9(1):1–7. doi: 10.1186/s13098-017-0266-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Eguchi S, Kawai T, Scalia R, Rizzo V. Understanding angiotensin II type 1 receptor signaling in vascular pathophysiology. Hypertension. 2018;71(5):804–810. doi: 10.1161/HYPERTENSIONAHA.118.10266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Abu-Farha M, Thanaraj TA, Qaddoumi MG, Hashem A, Abubaker J, et al. The role of lipid metabolism in COVID-19 virus infection and as a drug target. Int J Mol Sci. 2020;21(10):3544–3555. doi: 10.3390/ijms21103544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sivamaruthi BS, Kesika P, Chaiyasut C. A mini-review of human studies on cholesterol-lowering properties of probiotics. Sci Pharm. 2019;87(4):26–39. doi: 10.3390/scipharm87040026. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.