Abstract
Down syndrome is the most common genomic disorder of intellectual disability and is caused by trisomy of chromosome 21. Several genes in this chromosome repress mitochondrial biogenesis. The goal of this study was to evaluate whether early overexpression of these genes may cause a prenatal impairment of oxidative phosphorylation negatively affecting neurogenesis. Reduction in the mitochondrial energy production and a lower mitochondrial function have been reported in diverse tissues or cell types, and also at any age, including early fetuses, suggesting that a defect in oxidative phosphorylation is an early and general event in Down syndrome individuals. Moreover, many of the medical conditions associated with Down syndrome are also frequently found in patients with oxidative phosphorylation disease. Several drugs that enhance mitochondrial biogenesis are nowadays available and some of them have been already tested in mouse models of Down syndrome restoring neurogenesis and cognitive defects. Because neurogenesis relies on a correct mitochondrial function and critical periods of brain development occur mainly in the prenatal and early neonatal stages, therapeutic approaches intended to improve oxidative phosphorylation should be provided in these periods.
Keywords: Down syndrome, Brain development, Neurogenesis, Oxidative phosphorylation, Mitochondrial biogenesis
Graphical abstract
Highlights
-
•
Several chromosome 21-encoded proteins repress mitochondrial biogenesis.
-
•
These proteins are overexpressed in fetal brains of Down syndrome (DS) individuals.
-
•
Oxidative phosphorylation function is essential for neurogenesis.
-
•
Upregulation of these proteins adversely impact on neurogenesis.
-
•
Prenatal therapy with drugs inhibiting these proteins would increase DS neurogenesis.
1. Down syndrome and neurogenesis
Down syndrome (DS) is a multigene, multisystem disorder [1]. A partial or complete third copy of chromosome 21 (Hsa21), trisomy 21 (T21), is the major cause of DS. This condition is associated with intellectual disability, but other medical conditions, such as visual problems and hearing deficits, seizures, dementia, cardiac and muscle complications and gastrointestinal anomalies are also common in DS individuals [2,3].
Abnormalities in virtually every organ system are routinely treated in DS [4]. Intellectual disability is a key aspect of DS [4]. DS individuals show smaller brain volumes and reduced cortical surface area [1]. The brain of DS individuals exhibits a decline in proliferation and neuronal differentiation capacities of neural progenitor cells (NPCs) and an increased cell death. This deficient neurogenesis can already be observed in the fetal period [[5], [6], [7]]. To improve cognitive capabilities, active care programs using enriched environments are one of the most successful therapeutic interventions. However, results are limited and temporary [8].
2. Neurogenesis and oxidative phosphorylation
The oxidative phosphorylation (OXPHOS) system is the main energy provider to power activity of mature neurons [9]. For this reason, the central nervous system is one of the most affected tissues in OXPHOS disorders [10].
The modulation of maternal oxygenation in mice affects the fetal brain oxygen levels. Compared with normoxic brains, fetal brain volume of hypoxia-treated embryos was reduced whereas hyperoxia provoked a significant increase in brain volume [11]. Oxygen is mainly consumed by the OXPHOS system [12]. These observations suggest that OXPHOS function is also fundamental for brain development and the neurogenic process during prenatal development [13,14]. This biochemical pathway is required for neuronal differentiation [15,16]. In fact, pathological mutations in mouse or human OXPHOS-related genes impaired neuronal differentiation [[17], [18], [19]]. Confirming this effect, we have recently shown that neuronal differentiation of human cells overexpressing a mitochondrial ribosomal protein S12 (MRPS12) mutant gene was significantly reduced [20]. Moreover, the ribosomal antibiotic linezolid, that inhibits mitochondrial translation and decreases OXPHOS function, and cyanide, an OXPHOS poison, decreased neuronal differentiation of human cells [20,21].
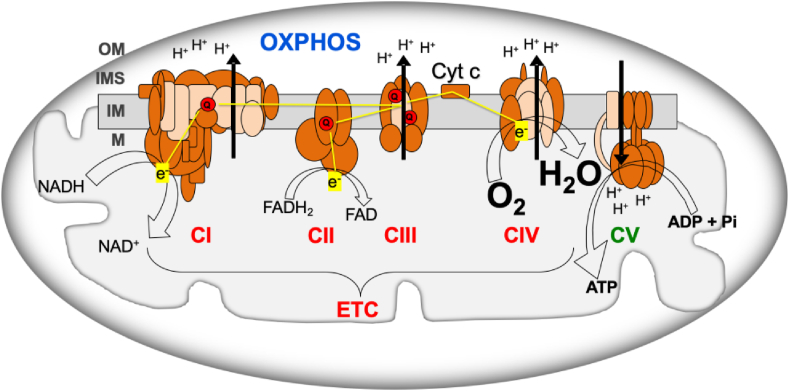
OXPHOS system consists of mitochondrial inner membrane electron transport chain (ETC) and ATP synthase (OXPHOS complex V, CV) (Fig. 1). ETC includes respiratory complexes I to IV (CI - CIV), cytochrome c and coenzyme Q (Q). Most of the mitochondrial proteins, including all the proteins required for mitochondrial DNA (mtDNA) replication, transcription and translation; assembly proteins of OXPHOS complexes; and most of the structural subunits of the OXPHOS complexes are coded in the nuclear chromosomes (nDNA). However, 13 OXPHOS subunits are mtDNA-encoded, along with the transfer and ribosomal RNAs (tRNAs and rRNAs) needed for their production. Electrons from the nutrients reduce flavin and nicotinamide coenzymes (FAD/FADH2 and NAD+/NADH) and, through CI and CII, reduce Q (ubiquinone/ubiquinol). Electrons from ubiquinol go through CIII, cytochrome c and CIV to reduce oxygen to water. The electron flow through the ETC determines the oxygen consumption and the mitochondrial inner membrane potential generated by the proton pumping from the mitochondrial matrix into the intermembrane space.
Fig. 1.
Oxidative phosphorylation system (OXPHOS). ADP, adenosine diphosphate; ATP, adenosine triphosphate; Cyt c, cytochrome c; CI-IV, respiratory complexes I-IV; CV, ATP synthase; ETC, electron transport chain; e−, electrons; FADH2/FAD, reduced and oxidized flavin adenine dinucleotide; H2O, water; H+, protons; IM, mitochondrial inner membrane; IMS, intermembrane space; M, mitochondrial matrix; NADH/NAD+, reduced and oxidized nicotinamide adenine dinucleotide; OM, mitochondrial outer membrane; O2, oxygen; Pi, phosphate; Q, coenzyme Q. Brown and pink colors are used for nDNA- and mtDNA-encoded subunits, respectively. Yellow lines, electron flow. Black arrows, proton pumping. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
3. Oxidative phosphorylation dysfunction is an early feature of down syndrome
3.1. Mouse models for down syndrome
There are several mouse models of DS [[22], [23], [24]], and some of them have been used to study neurogenesis and several mitochondrial parameters under this condition. It was published that the astrocytes of Ts1Cje mice, carriers of a trisomic segment that extends from Sod1 to Znf295 genes, showed a decrease in mitochondrial inner membrane potential and lower levels of ATP. The cerebral cortex of three-month-old Ts1Cje mice also had lower levels of ATP [25]. In Ts3Yah mice, which carry a tandem duplication of the Hspa13-App interval, there was a decrease in mRNAs of genes related to the OXPHOS system, in those related to mitochondrial biogenesis and in the amount of mtDNA in skeletal muscle [26]. The oxygen consumption with CI substrates was decreased in cerebral mitochondria of Ts65Dn mice, that harbor a trisomic segment between the App and Znf295 genes [27]. The levels of the p.MT-CO2 subunit from CIV were reduced in the soleus muscle of six-month-old Ts65Dn mice [28]. The oxygen consumption, CI and CIV activities, amount of ATP and mtDNA levels were shown decreased in NPCs of Ts65Dn mice. The impairment of the OXPHOS system was accompanied by a decrease in neuronal proliferation and differentiation capacity that has been suggested to be responsible for the reduced neuronal density found in the Ts65Dn mouse [[29], [30], [31]].
3.2. Down syndrome individuals
Several studies using differentiated cells from T21 human induced pluripotent stem cells (hiPSCs) showed that these models reflect the molecular and cellular phenotypes observed in primary cells and in individuals with DS [32]. T21 hiPSCs-derived neurons showed decreased mitochondrial inner membrane potential and increased production of amyloid peptide [33]. A reduction in mitochondrial inner membrane potential was also informed in lymphocytes and lymphoblastic cell lines from DS individuals [34,35], and also in neurons, astrocytes and fibroblasts of DS fetuses from 14 to 22 weeks of gestation [[36], [37], [38], [39], [40]]. A decrease in oxygen consumption was reported in fibroblasts and lymphocytes of DS individuals and fibroblasts of DS fetuses from 14 to 22 weeks of gestation [37,39,[41], [42], [43], [44], [45]]. The activity of ETC complexes was found decreased in fibroblasts, lymphocytes and platelets of DS individuals and fibroblasts of DS fetuses from 14 to 22 weeks of gestation [37,41,[43], [44], [45], [46], [47]]. The quantity of protein subunits for OXPHOS complexes was also reduced in brain, cerebellum and fibroblasts of DS adults or DS fetuses from 14 to 19 weeks of gestation [41,[48], [49], [50], [51]]. A lower Q quantity was reported in lymphocytes, platelets and plasma of DS individuals [52,53]. mRNA levels of a number of genes coding subunits of the OXPHOS complexes were decreased in T21 hiPSCs [54]. Supporting these data, a reduction in mRNA levels of several subunits of OXPHOS complexes was reported in DS cerebellum and fibroblasts [55,56]. Very interestingly, the down-regulation of genes for all OXPHOS complexes was also found in heart, fibroblasts and amniocytes of DS fetuses from 8 to 22 weeks of gestation [43,51,57,58]. This decline in mRNA levels was accompanied by a lower transcription of mtDNA-encoded genes and by a lower amount of mtDNA in brain from individuals with DS and Alzheimer disease (DSAD) and in DS fetuses of 22 weeks of gestation [59,60]. The levels of gene transcripts encoded in mtDNA were also found reduced in differentiated neurons from NPCs of DS individuals. This fact was accompanied by a reduced proliferative rate and a deviation of neuronal differentiation towards astrocytes [61].
All these modifications give place to a reduction in the mitochondrial energy production and a lower mitochondrial function [35,38,39,41,44,45,62,63]. As it can be noted from all these publications, OXPHOS defects were reported in distinct species, diverse tissues or cell types, and also at any age, including early fetuses. Therefore, it can be deduced that this defect is not a side effect but an early and general event in DS individuals. Moreover, many of the medical conditions associated with DS are also frequently found in patients with OXPHOS disease [64]. Thus, ‘red-flag’ features, which warrant the initiation of a baseline diagnostic evaluation for mitochondrial disease [65,66], such as epilepsia, intestinal dysmotility and hypotonia are also observed in DS individuals [2,3]. A multitude of other nonspecific symptoms that frequently occur in patients with mitochondrial disease [65,66], such as short stature, hearing loss, hypothyroidism and autism spectrum disorder (ASD) are also found in DS individuals [2,3]. Interestingly, similar to DS, ASD is a neurodevelopmental condition and the affectation of neurogenesis begins in the prenatal stage [67]. Moreover, an OXPHOS dysfunction is also found in ASD [68,69]. For all the aforementioned reasons, DS can be considered as an OXPHOS disorder.
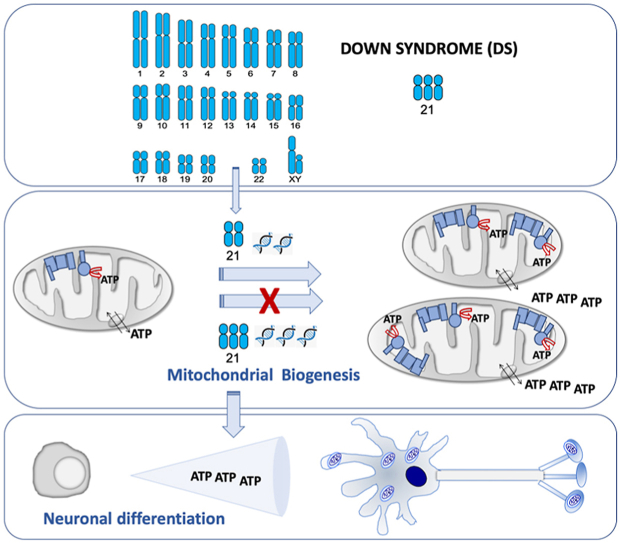
The overexpression of genes from Hsa21 is responsible for the DS pathogenesis. Some of these genes play a role in the expression of other genes and many of them are involved in the OXPHOS function. One of these genes is the amyloid precursor protein (APP) gene that is located in the chromosomal band 21q21.3 (Fig. 2A). This gene is overexpressed in different cell types from DS individuals [70,71]. While iPSCs had transcriptome profiles comparable to that of normal fetal brain development [61], T21 iPSCs had a marked upregulation of APP expression [61,72,73]. Moreover, its expression was also upregulated in NPCs, astrocytes and neurons derived from T21 hiPSCs [71,74]. Increased APP, and its carboxy-terminal fragment 99 (CTF99), levels were found in primary DS fetal brain cultures [36]. Increased APP mRNA and protein levels have been also detected in brains of fetuses with DS [[75], [76], [77], [78], [79]]. Moreover, amyloid beta (Aβ) was detected in fetuses with DS, but not in age-matched controls [80]. Increased CTF levels were also found in the brains of fetuses with DS [81]. The APP protein can associate with components of the mitochondrial protein translocation machinery and prevent the import of mitochondrial proteins, including nDNA-encoded OXPHOS subunits. Additionally, the APP-derived peptides, such as Aβ, amyloid precursor protein intracellular domain (AICD), and CTF99 can be present in mitochondria, interact with OXPHOS components and mtDNA and have detrimental consequences on bioenergetics function [82,83].
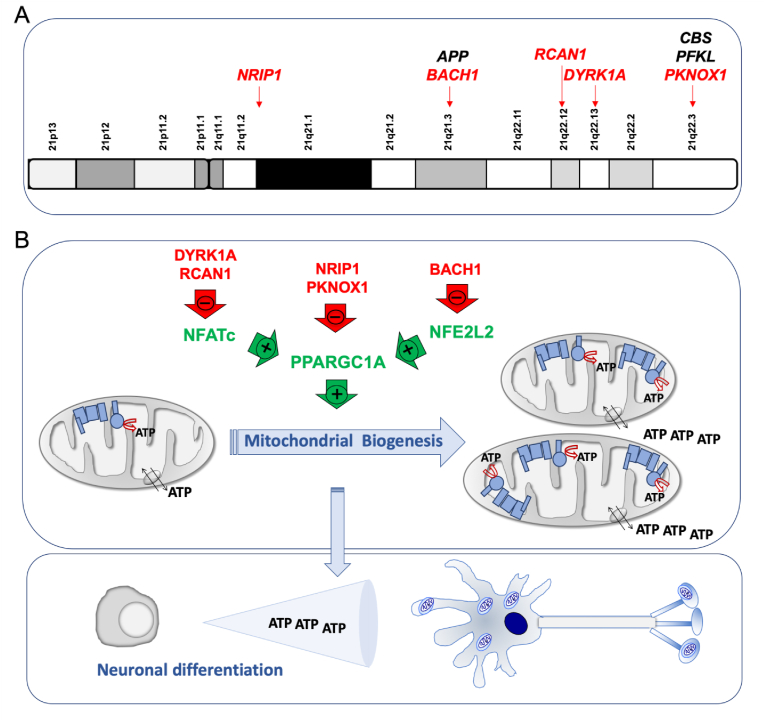
Fig. 2.
Chromosome 21 genes that repress mitochondrial biogenesis. A) Chromosome 21 location of genes that affect the oxidative phosphorylation function. Some of them, indirectly, through a negative effect on mitochondrial biogenesis (red color). B) Chromosome 21-endoced proteins that block mitochondrial biogenesis. Mitochondrial biogenesis leads to an overall increase in ATP synthesis. Neurogenesis requires high amount of energy. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
The cystathionine β synthase (CBS) gene is located in the 21q22.3 chromosomal band (Fig. 2A). This enzyme is responsible for the production of the gaseous transmitter hydrogen sulfide (H2S). An increased expression of CBS in many cells and tissues, including the brain, and H2S overproduction are well documented in DS individuals [71,[84], [85], [86], [87], [88], [89], [90], [91], [92]]. Human fetal brains from DS individuals showed increased CBS mRNA levels [78]. It was shown that a chronic exposure of pregnant wildtype animals to inhaled H2S produced significant neurodevelopmental alterations. This compound is an inhibitor of the respiratory CIV [92].
The liver type phosphofructokinase (PFK) gene, located in the chromosomal band 21q22.3 (Fig. 2A), is overexpressed in different cell types and the brain of DS individuals [70,74,88,91]. Its enzyme activity was shown increased in erythrocytes and fibroblasts from DS individuals [[93], [94], [95]]. PFK overexpression negatively regulated neurogenesis from NSCs and inhibited their neuronal differentiation [96]. PFK could play a key role in regulating the metabolic balance between glycolysis and OXPHOS [97].
The dual specificity tyrosine phosphorylation regulated kinase 1 A (DYRK1A) gene coded in the chromosomal band 21q22.13 (Fig. 2A) is overexpressed in different cell types [[70], [71], [72],74,79,98,99], adult brains [88,91,[100], [101], [102], [103]] and fetal brains of DS individuals [78,104,105]. An increase of DYRK1A protein and activity was found in NPCs derived of T21 hiPSCs obtained from fetal fibroblasts [6]. DYRK1A overexpression reduced the nuclear factor of activated T-cells (NFATc) activity (Fig. 2B) [43,106]. NFATc exerts a positive effect on the promoter of the peroxisome proliferator activated receptor gamma coactivator 1 alpha (PGC-1α or PPARGC1A) gene [107], which is a key modulator of mitochondrial biogenesis and OXPHOS function [108]. Therefore, high DYRK1A levels might reduce mitochondrial biogenesis.
The regulator of calcineurin 1 (RCAN1) gene, located in the chromosomal band 21q22.12 (Fig. 2A), is overexpressed in different cell types and the brain of DS individuals [70,72,88,99]. It is also overexpressed in different DS fetal cells and tissues, such as cultured amniocytes and chorionic villus cells [109,110], fibroblasts [111], liver and kidney [112], and brain [113]. Moreover, neural cells derived from T21 hiPSCs obtained from amniotic fluid stem cells overexpressed RCAN1 [114]. In Drosophila melanogaster, the overexpression of the RCAN1 homolog gene, nebula, reduced CIV activity and mitochondrial DNA content [59]. In embryonic rat striatal neurons ST14A, Rcan1 overexpression leaded to a reduction in mitochondrial mass and oxygen consumption rate [115]. The β-cells of mice overexpressing Rcan1 displayed reduced OXPHOS function [116]. In human umbilical vein endothelial cells (HUVEC) and cardiomyocytes, RCAN1 overexpression decreased mitochondrial mass [117,118]. In human neuroblastoma SH-SY5Y cells overexpressing RCAN1, oxygen consumption rate was decreased [119]. In addition, RCAN1 overexpression reduced NFATc activity (Fig. 2B and Table 1) [106].
Table 1.
Effect on OXPHOS function of the over- or under-expression of particular Hsa21 genes related to mitochondrial biogenesis.
The nuclear receptor interacting protein 1 (NRIP1) gene, which is located in 21q11.2-q21.1 chromosomal band (Fig. 2A), codes for a transcriptional corepressor protein. This gene is overexpressed in different cell types from DS individuals [70,71]. NRIP1 overexpression has been documented in fetal brains and fibroblasts of DS individuals [79,120,121]. It has been shown that Nrip1 levels affect OXPHOS function and mitochondrial biogenesis in different cells and tissues of mice and rats [[122], [123], [124], [125], [126], [127], [128], [129], [130], [131], [132], [133], [134], [135], [136], [137]]. A bias towards OXPHOS in human embryonic stem cells (hESCs) is consistent with increased PPARGC1A and decreased NRIP1 expression [138]. The NRIP1 overexpression in DS fetal fibroblasts represses PPARGC1A and decreases mRNA levels of many OXPHOS-related genes (Fig. 2B and Table 1) [120].
The PBX/knotted 1 homeobox 1 (PKNOX1) gene, located in the chromosomal band 21q22.3 (Fig. 2A), is overexpressed in different cell types from DS individuals [70], and fetal brain and fibroblasts of DS individuals [139,140]. This gene codes for a homeodomain transcription factor. Its muscle-specific ablation resulted in increased expression of ETC subunits, higher enzyme activity, and elevated PPARGC1A expression (Fig. 2B and Table 1) [141].
The BTB domain and CNC homolog 1 (BACH1) gene is located in 21q21.3 chromosomal band (Fig. 2A). This gene is overexpressed in different cell types from DS individuals [70,71,79,142]. It is also overexpressed in DS adult and fetal brain [88,91,143]. BACH1, a haem-binding transcription factor, binds to promoter regions of ETC genes and negatively regulates their transcription, thus reducing mitochondrial respiration [144]. On the contrary, the loss of BACH1 promotes mitochondrial respiration [144]. BACH1 is also a repressor of nuclear-erythroid-derived 2-like 2 (NRF2 or NFE2L2)-induced genes. NFE2L2 displaces BACH1 and upregulates a battery of genes. The PPARGC1A promoter contains NFE2L2 consensus sequences [145]. Therefore, BACH1 negatively regulates mitochondrial biogenesis (Fig. 2B and Table 1).
These observations suggest that several Hsa21 genes negatively regulate the PPARGC1A expression and/or activity [146], as shown in second trimester amniocytes derived from humans with DS [79]. This PPARGC1A downregulation would reduce mitochondrial biogenesis and lead to an OXPHOS defect.
4. Oxidative phosphorylation-targeted therapies in down syndrome
Critical periods of brain development, including neurogenesis and synapse formation, occur mainly in the prenatal and early neonatal stages. As already indicated, permanent neuromorphological alterations originate during fetal life in DS. Thus, this may be the period to positively impact neurogenesis and significantly improve the postnatal cognitive outcome in DS [4,29,32,147,148].
4.1. Mitochondrial biogenesis inducers
It has been shown that the activation of PPARGC1A and mitochondrial biogenesis protects against prenatal hypoxic-ischemic brain injury [149]. Similar therapies targeting mitochondria have shown to be beneficial in the treatment of DS phenotypes [[150], [151], [152]]. In the same way, the inhibition of overexpressed proteins in DS that reduce OXPHOS function, such as CBS, or mitochondrial biogenesis, such as DYRK1A, might be tested as a potential early therapy [153,154].
It was previously mentioned that PFK knockdown enhanced neuronal differentiation of NSCs and increased neurogenesis in the dentate gyrus of the mouse hippocampus [96]. It has been recently reported that tryptolinamide, a PFK inhibitor, activated mitochondrial respiration and rescued the defect in neuronal differentiation of iPSCs carrying mutant mtDNAs [97].
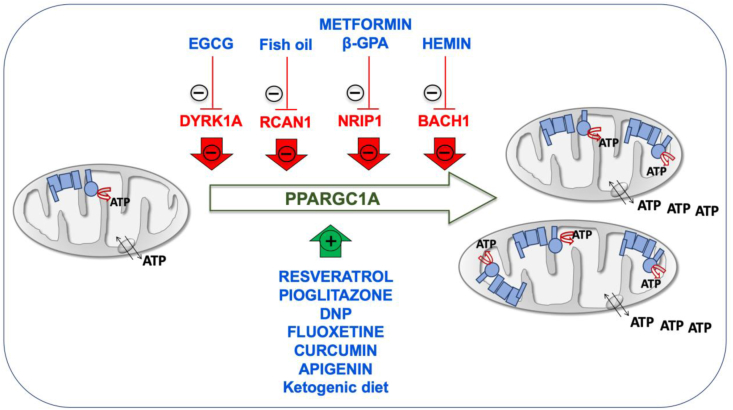
The promotion of mitochondrial biogenesis is an interesting therapeutic approach. Many drugs, such as epigallocatechin-3-gallate (EGCG), metformin, resveratrol, pioglitazone, β-guanadinopropionic acid (β-GPA) and 2,4-dinitrophenol (DNP) increase the PPARGC1A levels [30,39,146,155,156]. EGCG is a DYRK1A inhibitor and induces mitochondrial biogenesis (Fig. 3) [[157], [158], [159]]. This compound stimulated OXPHOS function in fibroblasts and lymphoblasts of DS individuals [44]. EGCG reversed the mitochondrial energetic deficit and rescued the altered neurogenesis of NPCs from a mouse model of DS or from T21 hiPSCs [6,30]. EGCG improved neurogenesis after ischemic stroke in adult mice [160]. Moreover, neonatal or prenatal treatment with EGCG restored neurogenesis and cognitive defects in mouse models of DS [161,162]. A mix of EGCG and omega-3 fatty acids restored ETC activity in lymphocytes from a DS child [163].
Fig. 3.
Inducers of mitochondrial biogenesis. Chromosome 21-encoded proteins that repress mitochondrial biogenesis are shown in red color. PPARGC1A, one of the main factors involved in mitochondrial biogenesis is represented in green color. Drugs enhancing mitochondrial biogenesis are indicated in blue color. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
Omega-3 fatty acids eicosapentaenoic (EPA) and docosahexaenoic (DHA) increased the Ppargc1a expression and mitochondrial biogenesis in the mouse C2C12 myoblast cell line [164]. Omega-3 containing fish oil supplementation starting at conception led to a reduction in hippocampal Rcan1 mRNA and protein levels in 1.5-month-old mice [165]. The prenatal administration of the omega-3 alpha-linolenic acid, that also increase PPARGC1A expression and mitochondrial biogenesis in fat of mice [166,167] and in human SH-SY5Y cells [168], reduced neuromorphological and cognitive alterations in Ts65Dn mice [169]. Alpha-linolenic acid is required in fetal stages of brain development in mice [170].
In mouse C2C12 cells, metformin downregulated NRIP1 and augmented the Ppargc1a levels (Fig. 3) [171]. Metformin also induced mitochondrial biogenesis in human aortic endothelial cells [172], stimulated OXPHOS function in fibroblasts of DS individuals [39], and improved neurogenesis of human bone marrow-mesenchymal stem cells [173]. β-GPA feeding led to marked reductions in both the protein content and mRNA expression of Nrip1 in fast-twitch rat triceps muscles and increased mitochondrial proteins, such as COXI, COXIV, and CS (Fig. 3) [127]. β-GPA treatment increased the mtDNA copy number in the cortex and ventral midbrain of mice, as well as the expression of several key metabolic enzyme indicators of mitochondria proliferation and the activation of Nfe2l2 signaling cascade [174].
Also related to an Hsa21 gene involved in mitochondrial biogenesis, hemin, the active ingredient of the FDA-approved drug Panhematin, binds to BACH1 and favors its release from DNA for nuclear export and subsequent degradation. It has been shown that hemin increased mitochondrial gene expression and induced oxygen consumption rate in triple negative breast cancer cells (Fig. 3) [144]. Kelch-like ECH associated protein 1 (KEAP1) sequesters NFE2L2 in the cytoplasm. In the presence of dimethylfumarate (DMF), KEAP1 is modified allowing NFE2L2 translocation to the nucleus and the dissociation of BACH1. It has been shown in mouse embryonic fibroblasts that DMF induced mitochondrial biogenesis and increased the mtDNA copy number, the transcription levels of mtDNA-encoded genes and the amount of OXPHOS subunits [175]. Interestingly, several pharmacologic therapies, such as fluoxetine, curcumin and apigenin have been already prenatally tested in mouse models of DS and shown to have beneficial effects on neurogenesis and behavioral performance [[176], [177], [178]]. These three drugs have been shown to improve OXPHOS function and mitochondrial biogenesis [[179], [180], [181], [182], [183], [184], [185]]. Although none of them has been related to the chromosome 21 genes that repress mitochondrial biogenesis, all of them increase the expression of NFE2L2 [[186], [187], [188], [189], [190], [191]].
Resveratrol and pioglitazone are inducers of mitochondrial biogenesis (Fig. 3) [158,192,193]. Pioglitazone stimulated OXPHOS function in fibroblasts of DS individuals [194], and improved neurogenesis of human neuroblastoma SH-SY5Y cells [195]. Resveratrol reversed the mitochondrial energetic deficit and rescued the altered neurogenesis of NPCs from a mouse model of DS [30].
Finally, the DNP activates mitochondrial biogenesis in mice and rats [196,197], and in human rhabdomyosarcoma cells (Fig. 3) [156]. DNP is a mild OXPHOS uncoupler that increased the synthesis of pyrimidine nucleotide-derived compounds to produce phospholipids, neuritogenesis and neuronal differentiation [[198], [199], [200]]. Interestingly, it was shown that the total phospholipid content was significantly reduced in central cortex and cerebellum of DS individuals [201]. It has been also reported that cerebral cortex cells (CTb) derived from mouse fetuses with trisomy 16, an animal model of DS, overexpressed App, because this protein is coded in the mouse chromosome 16 (Mmu16). DNP decreased intracellular accumulation of App in these cells [202].
In mouse hippocampus, the ketogenic diet upregulated the Ppargc1a mRNA and protein levels and other markers of mitochondrial biogenesis [203]. In cells from other species, including human fibroblasts, β-hydroxybutyrate increased the PPARGC1A levels and oxygen consumption [203]. In pigs, maternal ketosis increased fetal brain weight [204]. The ketogenic diet during the prenatal and early postnatal periods had beneficial effects on the brain development of pyruvate dehydrogenase complex-deficient mammalian progeny [205]. The ketogenic diet has been proposed as a potential therapeutic option for DS individuals [206].
4.2. OXPHOS products
Pyrimidine nucleotides are needed for RNA synthesis and, after reduction to deoxyribonucleotides, for DNA synthesis. In addition, these nucleotides are fundamental for the activation of the sugars that will give rise to the glycoproteins and glycolipids of the plasma membrane and also for the activation of compounds that will produce phospholipids and related molecules for the production of membrane lipids [21,207]. Uridine nucleotides are also involved in the N-acetylglucosamination of many proteins. If this process in disrupted, intellectual disability may ensue [208]. Pyrimidine nucleotides are essential in proliferating cells. On the other hand, neurons are non-proliferating differentiated cells. Its differentiation process includes the formation of axons and dendrites and the maintenance of its huge surface that requires a continuous membrane synthesis, even in already differentiated neurons [209]. Dihydroorotate dehydrogenase (DHODH) links the de novo pyrimidine biosynthesis pathway and OXPHOS function. Ubiquinone is the electrons’ acceptor in the oxidation of dihydroorotate to orotate by the DHODH [21,207]. A Q deficiency, as previously reported in DS, impairs the de novo synthesis of pyrimidine nucleotides [210]. DSAD individuals show a reduction in plasma uridine concentrations [211]. Hence, a deficit of the OXPHOS system hampering Q reoxidation might cause a reduction in the de novo pyrimidine biosynthesis [212], in the proliferation of neuronal precursor cells and in their differentiation into neurons. We observed that uridine was able to prevent the negative effects of OXPHOS dysfunction on neuronal differentiation. Uridine prevented the decline of neuronal differentiation in cells overexpressing a mutant MRPS12 gene, in linezolid-treated cells, in linezolid-treated cells overexpressing a mutant MRPS12 gene, or in cyanide-treated cells [20,21].
The choline prenatal administration has shown to have beneficial effects on neurogenesis and behavioral performance when tested in a mouse model of DS [213]. Along with cytidine triphosphate (CTP), derived from uridine triphosphate (UTP), choline is required for phospholipid synthesis and brain development [21,214]. Therefore, choline or uridine administration could increase this process [215].
5. Conclusions
Although DS is a multigenic disorder and many cell pathways are affected, the early prenatal OXPHOS dysfunction is a key pathologic mechanism in the intellectual disability. Moreover, OXPHOS is a ubiquitous cell function and its deficiency might affect every organ of the body and hence, be related to many of the medical conditions associated to DS. Therefore, any strategy developed to improve OXPHOS function could become a successful therapeutic approach in DS.
The invitation to pregnant women to participate in trials for fetal DS therapy should be based on adequate preclinical evidence. There should be convincing experimental evidence based on cell and animal studies that ensures the safety and efficacy of the therapy [4]. These models, with their related problems, are considered suitable approaches for the study of human pathophysiology and response to drugs in DS [23,24,32]. To test this hypothesis, the overexpression of each one of these genes that repress mitochondrial biogenesis in cells capable of differentiate into neurons would help to study their effect on OXPHOS function and on neurogenesis. On the other hand, iPSCs from DS individuals would allow to analyze the combined effect of all these genes on OXPHOS function and neuronal differentiation. The effect of the proposed drugs on these cellular parameters would also be studied in both cell models. If promising results are obtained, mouse models with DS would allow to study the prenatal effect of the proposed drugs. In fact, some results obtained with prenatal therapy in animal models encourage to dedicate more time, effort and resources in this field [216]. Finally, some of the proposed drugs have been demonstrated to be safe, have been approved and are normally administered in pregnant women for the treatment of other pathologies.
Funding sources
This work was supported by grants from Instituto de Salud Carlos III [FIS-PI17/00021, FIS-PI17/00166]; Fundación Mutua Madrileña [MMA17/01]; Precipita-FECYT crowdfunding program [PR194]; Gobierno de Aragón [LMP135_18, Grupos Consolidados B33_17R] and FEDER 2014–2020 “Construyendo Europa desde Aragón”. CIBERER is an initiative of the ISCIII. The sponsors had no role in the study design, collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
We would like to thank Santiago Morales for his assistance with figures.
Contributor Information
M. Pilar Bayona-Bafaluy, Email: pbayona@unizar.es.
Nuria Garrido-Pérez, Email: ngarrido@unizar.es.
Patricia Meade, Email: pmeade@unizar.es.
Eldris Iglesias, Email: eiglesia@unizar.es.
Irene Jiménez-Salvador, Email: jimsal@unizar.es.
Julio Montoya, Email: jmontoya@unizar.es.
Carmen Martínez-Cué, Email: martinec@unican.es.
Eduardo Ruiz-Pesini, Email: eduruiz@unizar.es.
References
- 1.Baburamani A.A., Patkee P.A., Arichi T., Rutherford M.A. New approaches to studying early brain development in Down syndrome. Dev. Med. Child Neurol. 2019;61:867–879. doi: 10.1111/dmcn.14260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Antonarakis S.E., Skotko B.G., Rafii M.S., Strydom A., Pape S.E., Bianchi D.W., Sherman S.L., Reeves R.H. Down syndrome. Nat. Rev. Dis. Prim. 2020;6:9. doi: 10.1038/s41572-019-0143-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bull M.J. Down syndrome. N. Engl. J. Med. 2020;382:2344–2352. doi: 10.1056/NEJMra1706537. [DOI] [PubMed] [Google Scholar]
- 4.de Wert G., Dondorp W., Bianchi D.W. Fetal therapy for Down syndrome: an ethical exploration. Prenat. Diagn. 2017;37:222–228. doi: 10.1002/pd.4995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guidi S., Bonasoni P., Ceccarelli C., Santini D., Gualtieri F., Ciani E., Bartesaghi R. Neurogenesis impairment and increased cell death reduce total neuron number in the hippocampal region of fetuses with Down syndrome. Brain Pathol. 2008;18:180–197. doi: 10.1111/j.1750-3639.2007.00113.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hibaoui Y., Grad I., Letourneau A., Sailani M.R., Dahoun S., Santoni F.A., Gimelli S., Guipponi M., Pelte M.F., Béna F., Antonarakis S.E., Feki A. Modelling and rescuing neurodevelopmental defect of Down syndrome using induced pluripotent stem cells from monozygotic twins discordant for trisomy 21. EMBO Mol. Med. 2014;6:259–277. doi: 10.1002/emmm.201302848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stagni F., Giacomini A., Emili M., Guidi S., Bartesaghi R. Neurogenesis impairment: an early developmental defect in Down syndrome. Free Radic. Biol. Med. 2018;114:15–32. doi: 10.1016/j.freeradbiomed.2017.07.026. [DOI] [PubMed] [Google Scholar]
- 8.Martínez Cué C., Dierssen M. Prog. Brain Res. Elsevier B.V.; 2020. Plasticity as a therapeutic target for improving cognition and behavior in Down syndrome; pp. 269–302. [DOI] [PubMed] [Google Scholar]
- 9.Hall C.N., Klein-Flugge M.C., Howarth C., Attwell D. Oxidative phosphorylation, not glycolysis, powers presynaptic and postsynaptic mechanisms underlying brain information processing. J. Neurosci. 2012;32:8940–8951. doi: 10.1523/JNEUROSCI.0026-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Keshavan N., Rahman S. Natural history of mitochondrial disorders: a systematic review. Essays Biochem. 2018;62:423–442. doi: 10.1042/EBC20170108. [DOI] [PubMed] [Google Scholar]
- 11.Wagenfuhr L., Meyer A.K., Braunschweig L., Marrone L., Storch A. Brain oxygen tension controls the expansion of outer subventricular zone-like basal progenitors in the developing mouse brain. Development. 2015;142:2904–2915. doi: 10.1242/dev.121939. [DOI] [PubMed] [Google Scholar]
- 12.Keeley T.P., Mann G.E. Defining physiological normoxia for improved translation of cell physiology to animal models and humans. Physiol. Rev. 2019;99:161–234. doi: 10.1152/physrev.00041.2017. [DOI] [PubMed] [Google Scholar]
- 13.Khacho M., Clark A., Svoboda D.S., MacLaurin J.G., Lagace D.C., Park D.S., Slack R.S. Mitochondrial dysfunction underlies cognitive defects as a result of neural stem cell depletion and impaired neurogenesis. Hum. Mol. Genet. 2017;26:3327–3341. doi: 10.1093/hmg/ddx217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khacho M., Harris R., Slack R.S. Mitochondria as central regulators of neural stem cell fate and cognitive function. Nat. Rev. Neurosci. 2019;20:34–48. doi: 10.1038/s41583-018-0091-3. [DOI] [PubMed] [Google Scholar]
- 15.O'Brien L.C., Keeney P.M., Bennett J.P. Differentiation of human neural stem cells into motor neurons stimulates mitochondrial biogenesis and decreases glycolytic flux. Stem Cell. Dev. 2015;24 doi: 10.1089/scd.2015.0076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lorenz C., Prigione A. Mitochondrial metabolism in early neural fate and its relevance for neuronal disease modeling. Curr. Opin. Cell Biol. 2017;49:71–76. doi: 10.1016/j.ceb.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 17.Kirby D.M., Rennie K.J., Smulders-Srinivasan T.K., Acin-Perez R., Whittington M., Enriquez J.-A., Trevelyan A.J., Turnbull D.M., Lightowlers R.N. Trans mitochondrial embryonic stem cells containing pathogenic mtDNA mutations are compromised in neuronal differentiation. Cell Prolif. 2009;42:413–424. doi: 10.1111/j.1365-2184.2009.00612.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hatakeyama H., Goto Y. Concise review: heteroplasmic mitochondrial DNA mutations and mitochondrial diseases: toward iPSC-based disease modeling, drug discovery, and regenerative therapeutics. Stem Cell. 2016;34:801–808. doi: 10.1002/stem.2292. [DOI] [PubMed] [Google Scholar]
- 19.Yokota M., Hatakeyama H., Ono Y., Kanazawa M., Goto Y. Mitochondrial respiratory dysfunction disturbs neuronal and cardiac lineage commitment of human iPSCs. Cell Death Dis. 2018;8:e2551. doi: 10.1038/cddis.2016.484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iglesias E., Bayona-Bafaluy M.P., Pesini A., Garrido-Pérez N., Meade P., Gaudó P., Jiménez-Salvador I., Montoya J., Ruiz-Pesini E. Uridine prevents negative effects of OXPHOS xenobiotics on dopaminergic neuronal differentiation. Cells. 2019;8 doi: 10.3390/cells8111407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pesini A., Iglesias E., Bayona-Bafaluy M.P., Garrido-Pérez N., Meade P., Gaudó P., Jiménez-Salvador I., Andrés-Benito P., Montoya J., Ferrer I., Pesini P., Ruiz-Pesini E. Brain pyrimidine nucleotide synthesis and Alzheimer disease. Aging (Albany. NY) 2019;11:8433–8462. doi: 10.18632/aging.102328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rueda N., Flórez J., Martínez-Cué C. Mouse models of down syndrome as a tool to unravel the causes of mental disabilities. Neural Plast. 2012:1–26. doi: 10.1155/2012/584071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Herault Y., Delabar J.M., Fisher E.M.C., Tybulewicz V.L.J., Yu E., Brault V. Rodent models in Down syndrome research: impact and future opportunities. Dis. Model. Mech. 2017;10:1165–1186. doi: 10.1242/dmm.029728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhao X., Bhattacharyya A. Human models are needed for studying human neurodevelopmental disorders. Am. J. Hum. Genet. 2018;103:829–857. doi: 10.1016/j.ajhg.2018.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shukkur E.A., Shimohata A., Akagi T., Yu W., Yamaguchi M., Murayama M., Chui D., Takeuchi T., Amano K., Subramhanya K.H., Hashikawa T., Sago H., Epstein C.J., Takashima A., Yamakawa K. Mitochondrial dysfunction and tau hyperphosphorylation in Ts1Cje, a mouse model for Down syndrome. Hum. Mol. Genet. 2006;15:2752–2762. doi: 10.1093/hmg/ddl211. [DOI] [PubMed] [Google Scholar]
- 26.Brault V., Duchon A., Romestaing C., Sahun I., Pothion S., Karout M., Borel C., Dembele D., Bizot J.-C., Messaddeq N., Sharp A.J., Roussel D., Antonarakis S.E., Dierssen M., Hérault Y. Opposite phenotypes of muscle strength and locomotor function in mouse models of partial trisomy and monosomy 21 for the proximal Hspa13-App region. PLoS Genet. 2015;11 doi: 10.1371/journal.pgen.1005062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bambrick L.L., Fiskum G. Mitochondrial dysfunction in mouse trisomy 16 brain. Brain Res. 2008;1188:9–16. doi: 10.1016/j.brainres.2007.10.045. [DOI] [PubMed] [Google Scholar]
- 28.Cowley P.M., Keslacy S., Middleton F.A., DeRuisseau L.R., Fernhall B., Kanaley J.A., DeRuisseau K.C. Functional and biochemical characterization of soleus muscle in Down syndrome mice: insight into the muscle dysfunction seen in the human condition. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012;303:R1251–R1260. doi: 10.1152/ajpregu.00312.2012. [DOI] [PubMed] [Google Scholar]
- 29.Bartesaghi R., Guidi S., Ciani E. Is it possible to improve neurodevelopmental abnormalities in Down syndrome? Rev. Neurosci. 2011;22:419–455. doi: 10.1515/RNS.2011.037. [DOI] [PubMed] [Google Scholar]
- 30.Valenti D., de Bari L., de Rasmo D., Signorile A., Henrion-Caude A., Contestabile A., Vacca R.A. The polyphenols resveratrol and epigallocatechin-3-gallate restore the severe impairment of mitochondria in hippocampal progenitor cells from a Down syndrome mouse model. Biochim. Biophys. Acta. 2016;1862:1093–1104. doi: 10.1016/j.bbadis.2016.03.003. [DOI] [PubMed] [Google Scholar]
- 31.Valenti D., Rossi L., Marzulli D., Bellomo F., De Rasmo D., Signorile A., Vacca R.A. Inhibition of Drp1-mediated mitochondrial fission improves mitochondrial dynamics and bioenergetics stimulating neurogenesis in hippocampal progenitor cells from a Down syndrome mouse model. Biochim. Biophys. Acta (BBA) - Mol. Basis Dis. 2017;1863:3117–3127. doi: 10.1016/j.bbadis.2017.09.014. [DOI] [PubMed] [Google Scholar]
- 32.Lee S.E., Duran-Martinez M., Khantsis S., Bianchi D.W., Guedj F. Challenges and opportunities for translation of therapies to improve cognition in down syndrome. Trends Mol. Med. 2020;26:150–169. doi: 10.1016/j.molmed.2019.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Murray A., Letourneau A., Canzonetta C., Stathaki E., Gimelli S., Sloan-Bena F., Abrehart R., Goh P., Lim S., Baldo C., Dagna-Bricarelli F., Hannan S., Mortensen M., Ballard D., Syndercombe Court D., Fusaki N., Hasegawa M., Smart T.G., Bishop C., Antonarakis S.E., Groet J., Nizetic D. Brief report: isogenic induced pluripotent stem cell lines from an adult with mosaic down syndrome model accelerated neuronal ageing and neurodegeneration. Stem Cell. 2015;33 doi: 10.1002/stem.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Abu Faddan N., Sayed D., Ghaleb F. T lymphocytes apoptosis and mitochondrial membrane potential in Down's syndrome. Fetal Pediatr. Pathol. 2011;30:45–52. doi: 10.3109/15513815.2010.505626. [DOI] [PubMed] [Google Scholar]
- 35.Coskun P., Helguera P., Nemati Z., Bohannan R.C., Thomas J., Samuel S.E., Argueta J., Doran E., Wallace D.C., Lott I.T., Busciglio J. Metabolic and growth rate alterations in lymphoblastic cell lines discriminate between down syndrome and alzheimer's disease. J. Alzheimers. Dis. 2017;55:737–748. doi: 10.3233/JAD-160278. [DOI] [PubMed] [Google Scholar]
- 36.Busciglio J., Pelsman A., Wong C., Pigino G., Yuan M., Mori H., Yankner B.A. Altered metabolism of the amyloid beta precursor protein is associated with mitochondrial dysfunction in Down's syndrome. Neuron. 2002;33:677–688. doi: 10.1016/s0896-6273(02)00604-9. [DOI] [PubMed] [Google Scholar]
- 37.Valenti D., Manente G.A., Moro L., Marra E., Vacca R.A. Deficit of complex I activity in human skin fibroblasts with chromosome 21 trisomy and overproduction of reactive oxygen species by mitochondria: involvement of the cAMP/PKA signalling pathway. Biochem. J. 2011;435:679–688. doi: 10.1042/BJ20101908. [DOI] [PubMed] [Google Scholar]
- 38.Helguera P., Seiglie J., Rodriguez J., Hanna M., Helguera G., Busciglio J. Adaptive downregulation of mitochondrial function in down syndrome. Cell Metabol. 2013;17:132–140. doi: 10.1016/j.cmet.2012.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Izzo A., Nitti M., Mollo N., Paladino S., Procaccini C., Faicchia D., Calì G., Genesio R., Bonfiglio F., Cicatiello R., Polishchuk E., Polishchuk R., Pinton P., Matarese G., Conti A., Nitsch L. Metformin restores the mitochondrial network and reverses mitochondrial dysfunction in Down syndrome cells. Hum. Mol. Genet. 2017;26:1056–1069. doi: 10.1093/hmg/ddx016. [DOI] [PubMed] [Google Scholar]
- 40.Takahashi D., Moriyama J., Nakamura T., Miki E., Takahashi E., Sato A., Akaike T., Itto-Nakama K., Arimoto H. AUTACs: cargo-specific degraders using selective autophagy. Mol. Cell. 2019;76:797–810. doi: 10.1016/j.molcel.2019.09.009. e10. [DOI] [PubMed] [Google Scholar]
- 41.Valenti D., Tullo A., Caratozzolo M.F., Merafina R.S., Scartezzini P., Marra E., Vacca R.A. Impairment of F1F0-ATPase, adenine nucleotide translocator and adenylate kinase causes mitochondrial energy deficit in human skin fibroblasts with chromosome 21 trisomy. Biochem. J. 2010;431:299–310. doi: 10.1042/BJ20100581. [DOI] [PubMed] [Google Scholar]
- 42.Aburawi E.H., Souid A.-K. Lymphocyte respiration in children with Trisomy 21. BMC Pediatr. 2012;12:193. doi: 10.1186/1471-2431-12-193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Piccoli C., Izzo A., Scrima R., Bonfiglio F., Manco R., Negri R., Quarato G., Cela O., Ripoli M., Prisco M., Gentile F., Calì G., Pinton P., Conti A., Nitsch L., Capitanio N. Chronic pro-oxidative state and mitochondrial dysfunctions are more pronounced in fibroblasts from Down syndrome foeti with congenital heart defects. Hum. Mol. Genet. 2013;22:1218–1232. doi: 10.1093/hmg/dds529. [DOI] [PubMed] [Google Scholar]
- 44.Valenti D., De Rasmo D., Signorile A., Rossi L., de Bari L., Scala I., Granese B., Papa S., Vacca R.A. Epigallocatechin-3-gallate prevents oxidative phosphorylation deficit and promotes mitochondrial biogenesis in human cells from subjects with Down's syndrome. Biochim. Biophys. Acta. 2013;1832:542–552. doi: 10.1016/j.bbadis.2012.12.011. [DOI] [PubMed] [Google Scholar]
- 45.Panagaki T., Randi E.B., Augsburger F., Szabo C. Overproduction of H2S, generated by CBS, inhibits mitochondrial Complex IV and suppresses oxidative phosphorylation in Down syndrome. Proc. Natl. Acad. Sci. U.S.A. 2019;116 doi: 10.1073/pnas.1911895116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Solmi R., Rossetti A., Talassi O., Tomasini G.L., Fato R., Estornell E., Lucarini G., Lenaz G., Simonelli L., Brunelli M.A., Biagini G. Gingival fibroblasts “in vitro” and Down's Syndrome. Cytotechnology. 1993;11:S59–S61. doi: 10.1007/BF00746056. [DOI] [PubMed] [Google Scholar]
- 47.Prince J., Jia S., Båve U., Annerén G., Oreland L. Mitochondrial enzyme deficiencies in Down's syndrome. J. Neural Transm. Park. Dis. Dement. Sect. 1994;8:171–181. doi: 10.1007/BF02260938. [DOI] [PubMed] [Google Scholar]
- 48.Kim S.H., Vlkolinsky R., Cairns N., Lubec G. Decreased levels of complex III core protein 1 and complex V beta chain in brains from patients with Alzheimer's disease and Down syndrome. Cell. Mol. Life Sci. 2000;57:1810–1816. doi: 10.1007/pl00000661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kim S.H., Fountoulakis M., Dierssen M., Lubec G. Decreased protein levels of complex I 30-kDa subunit in fetal Down syndrome brains. J. Neural. Transm. 2001;Suppl doi: 10.1007/978-3-7091-6262-0_9. [DOI] [PubMed] [Google Scholar]
- 50.Kim S.H., Vlkolinsky R., Cairns N., Fountoulakis M., Lubec G. The reduction of NADH ubiquinone oxidoreductase 24- and 75-kDa subunits in brains of patients with Down syndrome and Alzheimer's disease. Life Sci. 2001;68:2741–2750. doi: 10.1016/s0024-3205(01)01074-8. [DOI] [PubMed] [Google Scholar]
- 51.Liu Y., Borel C., Li L., Müller T., Williams E.G., Germain P.-L., Buljan M., Sajic T., Boersema P.J., Shao W., Faini M., Testa G., Beyer A., Antonarakis S.E., Aebersold R. Systematic proteome and proteostasis profiling in human Trisomy 21 fibroblast cells. Nat. Commun. 2017;8 doi: 10.1038/s41467-017-01422-6. 1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tiano L., Padella L., Carnevali P., Gabrielli O., Bruge F., Principi F., Littarru G.P. Coenzyme Q10 and oxidative imbalance in Down syndrome: biochemical and clinical aspects. Biofactors. 2008;32:161–167. doi: 10.1002/biof.5520320119. [DOI] [PubMed] [Google Scholar]
- 53.M.E. Zaki, H.T. El-Bassyouni, A.M.S. Tosson, E. Youness, J. Hussein, Coenzyme Q10 and pro-inflammatory markers in children with Down syndrome: clinical and biochemical aspects., J. Pediatr. 93 (n.d.) 100–104. 10.1016/j.jped.2016.04.012. [DOI] [PubMed]
- 54.Qiu J.-J., Liu Y.-N., Ren Z.-R., Yan J.-B. Dysfunctions of mitochondria in close association with strong perturbation of long noncoding RNAs expression in down syndrome. Int. J. Biochem. Cell Biol. 2017;92:115–120. doi: 10.1016/j.biocel.2017.09.017. [DOI] [PubMed] [Google Scholar]
- 55.Krapfenbauer K., Yoo B.C., Cairns N., Lubec G. Differential display reveals deteriorated mRNA levels of NADH3 (complex I) in cerebellum of patients with Down syndrome. J. Neural. Transm. Suppl. 1999;57:211–220. doi: 10.1007/978-3-7091-6380-1_13. [DOI] [PubMed] [Google Scholar]
- 56.Salemi M., Giambirtone M., Barone C., Salluzzo M.G., Russo R., Lo Giudice M., Cutuli S., Ridolfo F., Romano C. Mitochondrial mRNA expression in fibroblasts of Down syndrome subjects. Hum. Cell. 2018;31:179–181. doi: 10.1007/s13577-018-0205-2. [DOI] [PubMed] [Google Scholar]
- 57.Lee S.H., Lee S., Jun H.S., Jeong H.J., Cha W.T., Cho Y.S., Kim J.H., Ku S.Y., Cha K.Y. Expression of the mitochondrial ATPase6 gene and Tfam in Down syndrome. Mol. Cell. 2003;15:181–185. http://www.ncbi.nlm.nih.gov/pubmed/12803480 [PubMed] [Google Scholar]
- 58.Conti A., Fabbrini F., D'Agostino P., Negri R., Greco D., Genesio R., D'Armiento M., Olla C., Paladini D., Zannini M., Nitsch L. Altered expression of mitochondrial and extracellular matrix genes in the heart of human fetuses with chromosome 21 trisomy. BMC Genom. 2007;8:268. doi: 10.1186/1471-2164-8-268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chang K.T., Min K.-T. Drosophila melanogaster homolog of Down syndrome critical region 1 is critical for mitochondrial function. Nat. Neurosci. 2005;8:1577–1585. doi: 10.1038/nn1564. [DOI] [PubMed] [Google Scholar]
- 60.Coskun P.E., Wyrembak J., Derbereva O., Melkonian G., Doran E., Lott I.T., Head E., Cotman C.W., Wallace D.C. Systemic mitochondrial dysfunction and the etiology of Alzheimer's disease and down syndrome dementia. J. Alzheimers. Dis. 2010;20(Suppl 2):S293–S310. doi: 10.3233/JAD-2010-100351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sobol M., Klar J., Laan L., Shahsavani M., Schuster J., Annerén G., Konzer A., Mi J., Bergquist J., Nordlund J., Hoeber J., Huss M., Falk A., Dahl N. Transcriptome and proteome profiling of neural induced pluripotent stem cells from individuals with down syndrome disclose dynamic dysregulations of key pathways and cellular functions. Mol. Neurobiol. 2019;56:7113–7127. doi: 10.1007/s12035-019-1585-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Phillips A.C., Sleigh A., McAllister C.J., Brage S., Carpenter T.A., Kemp G.J., Holland A.J. Defective mitochondrial function in vivo in skeletal muscle in adults with Down's syndrome: a 31P-MRS study. PloS One. 2013;8 doi: 10.1371/journal.pone.0084031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pecze L., Randi E.B., Szabo C. Meta-analysis of metabolites involved in bioenergetic pathways reveals a pseudohypoxic state in Down syndrome. Mol. Med. 2020;26:102. doi: 10.1186/s10020-020-00225-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.McCormick E.M., Lott M.T., Dulik M.C., Shen L., Attimonelli M., Vitale O., Karaa A., Bai R., Pineda-Alvarez D.E., Singh L.N., Stanley C.M., Wong S., Bhardwaj A., Merkurjev D., Mao R., Sondheimer N., Zhang S., Procaccio V., Wallace D.C., Gai X., Falk M.J. Specifications of the ACMG/AMP standards and guidelines for mitochondrial DNA variant interpretation. Hum. Mutat. 2020 doi: 10.1002/humu.24107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Haas R.H., Parikh S., Falk M.J., Saneto R.P., Wolf N.I., Darin N., Cohen B.H. Mitochondrial disease: a practical approach for primary care physicians. Pediatrics. 2007;120:1326–1333. doi: 10.1542/peds.2007-0391. [DOI] [PubMed] [Google Scholar]
- 66.Zolkipli-Cunningham Z., Xiao R., Stoddart A., McCormick E.M., Holberts A., Burrill N., McCormack S., Williams L., Wang X., Thompson J.L.P., Falk M.J. Mitochondrial disease patient motivations and barriers to participate in clinical trials. PloS One. 2018;13 doi: 10.1371/journal.pone.0197513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Courchesne E., Gazestani V.H., Lewis N.E. Prenatal origins of ASD: the when, what, and how of ASD development. Trends Neurosci. 2020;43:326–342. doi: 10.1016/j.tins.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Giulivi C., Zhang Y.-F., Omanska-Klusek A., Ross-Inta C., Wong S., Hertz-Picciotto I., Tassone F., Pessah I.N. Mitochondrial dysfunction in autism. J. Am. Med. Assoc. 2010;304:2389. doi: 10.1001/jama.2010.1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Rossignol D.A., Frye R.E. Mitochondrial dysfunction in autism spectrum disorders: a systematic review and meta-analysis. Mol. Psychiatr. 2012;17:290–314. doi: 10.1038/mp.2010.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sullivan K.D., Lewis H.C., Hill A.A., Pandey A., Jackson L.P., Cabral J.M., Smith K.P., Liggett L.A., Gomez E.B., Galbraith M.D., DeGregori J., Espinosa J.M. Trisomy 21 consistently activates the interferon response. Elife. 2016;5 doi: 10.7554/eLife.16220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gonzales P.K., Roberts C.M., Fonte V., Jacobsen C., Stein G.H., Link C.D. Transcriptome analysis of genetically matched human induced pluripotent stem cells disomic or trisomic for chromosome 21. PloS One. 2018;13 doi: 10.1371/journal.pone.0194581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Briggs J.A., Sun J., Shepherd J., Ovchinnikov D.A., Chung T.-L., Nayler S.P., Kao L.-P., Morrow C.A., Thakar N.Y., Soo S.-Y., Peura T., Grimmond S., Wolvetang E.J. Integration-free induced pluripotent stem cells model genetic and neural developmental features of down syndrome etiology. Stem Cell. 2013;31:467–478. doi: 10.1002/stem.1297. [DOI] [PubMed] [Google Scholar]
- 73.Lu H.-E., Yang Y.-C., Chen S.-M., Su H.-L., Huang P.-C., Tsai M.-S., Wang T.-H., Tseng C.-P., Hwang S.-M. Modeling neurogenesis impairment in Down syndrome with induced pluripotent stem cells from Trisomy 21 amniotic fluid cells. Exp. Cell Res. 2013;319:498–505. doi: 10.1016/j.yexcr.2012.09.017. [DOI] [PubMed] [Google Scholar]
- 74.Ponroy Bally B., Farmer W.T., V Jones E., Jessa S., Kacerovsky J.B., Mayran A., Peng H., Lefebvre J.L., Drouin J., Hayer A., Ernst C., Murai K.K. Human iPSC-derived Down syndrome astrocytes display genome-wide perturbations in gene expression, an altered adhesion profile, and increased cellular dynamics. Hum. Mol. Genet. 2020;29:785–802. doi: 10.1093/hmg/ddaa003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tanzi R.E., Gusella J.F., Watkins P.C., Bruns G.A., St George-Hyslop P., Van Keuren M.L., Patterson D., Pagan S., Kurnit D.M., Neve R.L. Amyloid beta protein gene: cDNA, mRNA distribution, and genetic linkage near the Alzheimer locus. Science. 1987;235:880–884. doi: 10.1126/science.2949367. [DOI] [PubMed] [Google Scholar]
- 76.Tanzi R.E., Haines J.L., Watkins P.C., Stewart G.D., Wallace M.R., Hallewell R., Wong C., Wexler N.S., Conneally P.M., Gusella J.F. Genetic linkage map of human chromosome 21. Genomics. 1988;3:129–136. doi: 10.1016/0888-7543(88)90143-7. [DOI] [PubMed] [Google Scholar]
- 77.Sosa L.J., Postma N.L., Estrada-Bernal A., Hanna M., Guo R., Busciglio J., Pfenninger K.H. Dosage of amyloid precursor protein affects axonal contact guidance in Down syndrome. Faseb. J. 2014;28:195–205. doi: 10.1096/fj.13-232686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.El Hajj N., Dittrich M., Böck J., Kraus T.F.J., Nanda I., Müller T., Seidmann L., Tralau T., Galetzka D., Schneider E., Haaf T. Epigenetic dysregulation in the developing Down syndrome cortex. Epigenetics. 2016;11:563–578. doi: 10.1080/15592294.2016.1192736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Guedj F., LA Pennings J., Massingham L.J., Wick H.C., Siegel A.E., Tantravahi U., Bianchi D.W. An integrated human/murine transcriptome and pathway approach to identify prenatal treatments for down syndrome. Sci. Rep. 2016;6 doi: 10.1038/srep32353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Teller J.K., Russo C., DeBusk L.M., Angelini G., Zaccheo D., Dagna-Bricarelli F., Scartezzini P., Bertolini S., Mann D.M., Tabaton M., Gambetti P. Presence of soluble amyloid beta-peptide precedes amyloid plaque formation in Down's syndrome. Nat. Med. 1996;2:93–95. doi: 10.1038/nm0196-93. [DOI] [PubMed] [Google Scholar]
- 81.Russo C., Salis S., Dolcini V., Venezia V., Song X.H., Teller J.K., Schettini G. Amino-terminal modification and tyrosine phosphorylation of [corrected] carboxy-terminal fragments of the amyloid precursor protein in Alzheimer's disease and Down's syndrome brain. Neurobiol. Dis. 2001;8:173–180. doi: 10.1006/nbdi.2000.0357. [DOI] [PubMed] [Google Scholar]
- 82.Wilkins H.M., Swerdlow R.H. Amyloid precursor protein processing and bioenergetics. Brain Res. Bull. 2017;133:71–79. doi: 10.1016/j.brainresbull.2016.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lopez Sanchez M.I.G., van Wijngaarden P., Trounce I.A. Amyloid precursor protein-mediated mitochondrial regulation and Alzheimer's disease. Br. J. Pharmacol. 2019;176:3464–3474. doi: 10.1111/bph.14554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chadefaux B., Rethoré M.O., Raoul O., Ceballos I., Poissonnier M., Gilgenkranz S., Allard D. Cystathionine beta synthase: gene dosage effect in trisomy 21. Biochem. Biophys. Res. Commun. 1985;128:40–44. doi: 10.1016/0006-291x(85)91641-9. [DOI] [PubMed] [Google Scholar]
- 85.Taub J.W., Huang X., Matherly L.H., Stout M.L., Buck S.A., V Massey G., Becton D.L., Chang M.N., Weinstein H.J., Ravindranath Y. Expression of chromosome 21-localized genes in acute myeloid leukemia: differences between Down syndrome and non-Down syndrome blast cells and relationship to in vitro sensitivity to cytosine arabinoside and daunorubicin. Blood. 1999;94:1393–1400. http://www.ncbi.nlm.nih.gov/pubmed/10438727 [PubMed] [Google Scholar]
- 86.Ge Y., Jensen T.L., Matherly L.H., Taub J.W. Transcriptional regulation of the cystathionine-beta -synthase gene in Down syndrome and non-Down syndrome megakaryocytic leukemia cell lines. Blood. 2003;101:1551–1557. doi: 10.1182/blood-2002-07-2337. [DOI] [PubMed] [Google Scholar]
- 87.Ichinohe A., Kanaumi T., Takashima S., Enokido Y., Nagai Y., Kimura H. Cystathionine beta-synthase is enriched in the brains of Down's patients. Biochem. Biophys. Res. Commun. 2005;338:1547–1550. doi: 10.1016/j.bbrc.2005.10.118. [DOI] [PubMed] [Google Scholar]
- 88.Lockstone H.E., Harris L.W., Swatton J.E., Wayland M.T., Holland A.J., Bahn S. Gene expression profiling in the adult Down syndrome brain. Genomics. 2007;90:647–660. doi: 10.1016/j.ygeno.2007.08.005. [DOI] [PubMed] [Google Scholar]
- 89.E. Abdel-Salam, M. Aboul-Enein, A. Kotb, M. Elseify, A. Abul-Hassan, S. Ishaac, Hematologic changes in Down's syndrome during infantile and early childhood period., Gaz. Egypt Paediatr. Assoc. 24 (n.d.) 209–16. http://www.ncbi.nlm.nih.gov/pubmed/145981. [PubMed]
- 90.Pinilla J.M., Ayala-Ramírez P., García-Robles R., Olaya-C M., Bermúdez M. Expression of cystathionine beta-synthase and histopathological observations in placentas of patients with Down syndrome. J. Neonatal Perinat. Med. 2015;8:77–84. doi: 10.3233/NPM-15814092. [DOI] [PubMed] [Google Scholar]
- 91.Olmos-Serrano J.L., Kang H.J., Tyler W.A., Silbereis J.C., Cheng F., Zhu Y., Pletikos M., Jankovic-Rapan L., Cramer N.P., Galdzicki Z., Goodliffe J., Peters A., Sethares C., Delalle I., Golden J.A., Haydar T.F., Sestan N. Down syndrome developmental brain transcriptome reveals defective oligodendrocyte differentiation and myelination. Neuron. 2016;89:1208–1222. doi: 10.1016/j.neuron.2016.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Szabo C. The re-emerging pathophysiological role of the cystathionine-β-synthase - hydrogen sulfide system in Down syndrome. FEBS J. 2020;287:3150–3160. doi: 10.1111/febs.15214. [DOI] [PubMed] [Google Scholar]
- 93.Benson P.F., Linacre B., Taylor A.I. Erythrocyte ATP: D-fructose-6-phosphate 1-phosphotransferase (phosphofructokinase) activity in children with normałG trisomic mosaic Down's syndrome and in normal and Down's syndrome controls. Nature. 1968;220:1235–1236. doi: 10.1038/2201235a0. [DOI] [PubMed] [Google Scholar]
- 94.Pantelakis S.N., Karaklis A.G., Alexiou D., Vardas E., Valaes T. Red cell enzymes in trisomy 21. Am. J. Hum. Genet. 1970;22:184–193. http://www.ncbi.nlm.nih.gov/pubmed/4244917 [PMC free article] [PubMed] [Google Scholar]
- 95.Annerén K.G., Korenberg J.R., Epstein C.J. Phosphofructokinase activity in fibroblasts aneuploid for chromosome 21. Hum. Genet. 1987;76:63–65. doi: 10.1007/BF00283052. [DOI] [PubMed] [Google Scholar]
- 96.Zhang F., Qian X., Qin C., Lin Y., Wu H., Chang L., Luo C., Zhu D. Phosphofructokinase-1 negatively regulates neurogenesis from neural stem cells. Neurosci. Bull. 2016;32:205–216. doi: 10.1007/s12264-016-0032-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kobayashi H., Hatakeyama H., Nishimura H., Yokota M., Suzuki S., Tomabechi Y., Shirouzu M., Osada H., Mimaki M., Goto Y.-I., Yoshida M. Chemical reversal of abnormalities in cells carrying mitochondrial DNA mutations. Nat. Chem. Biol. 2020 doi: 10.1038/s41589-020-00676-4. [DOI] [PubMed] [Google Scholar]
- 98.Tlili A., Hoischen A., Ripoll C., Benabou E., Badel A., Ronan A., Touraine R., Grattau Y., Stora S., van Bon B., de Vries B., Menten B., Bockaert N., Gecz J., Antonarakis S.E., Campion D., Potier M.-C., Bléhaut H., Delabar J.-M., Janel N. BDNF and DYRK1A are variable and inversely correlated in lymphoblastoid cell lines from Down syndrome patients. Mol. Neurobiol. 2012;46:297–303. doi: 10.1007/s12035-012-8284-7. [DOI] [PubMed] [Google Scholar]
- 99.Granese B., Scala I., Spatuzza C., Valentino A., Coletta M., Vacca R.A., De Luca P., Andria G. Validation of microarray data in human lymphoblasts shows a role of the ubiquitin-proteasome system and NF-kB in the pathogenesis of Down syndrome. BMC Med. Genom. 2013;6:24. doi: 10.1186/1755-8794-6-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Dowjat W.K., Adayev T., Kuchna I., Nowicki K., Palminiello S., Hwang Y.W., Wegiel J. Trisomy-driven overexpression of DYRK1A kinase in the brain of subjects with Down syndrome. Neurosci. Lett. 2007;413:77–81. doi: 10.1016/j.neulet.2006.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu F., Liang Z., Wegiel J., Hwang Y.-W., Iqbal K., Grundke-Iqbal I., Ramakrishna N., Gong C.-X. Overexpression of Dyrk1A contributes to neurofibrillary degeneration in Down syndrome. Faseb. J. 2008;22:3224–3233. doi: 10.1096/fj.07-104539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Shi J., Zhang T., Zhou C., Chohan M.O., Gu X., Wegiel J., Zhou J., Hwang Y.-W., Iqbal K., Grundke-Iqbal I., Gong C.-X., Liu F. Increased dosage of Dyrk1A alters alternative splicing factor (ASF)-regulated alternative splicing of tau in Down syndrome. J. Biol. Chem. 2008;283:28660–28669. doi: 10.1074/jbc.M802645200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wegiel J., Kaczmarski W., Barua M., Kuchna I., Nowicki K., Wang K.-C., Wegiel J., Yang S.M., Frackowiak J., Mazur-Kolecka B., Silverman W.P., Reisberg B., Monteiro I., de Leon M., Wisniewski T., Dalton A., Lai F., Hwang Y.-W., Adayev T., Liu F., Iqbal K., Iqbal I.-G., Gong C.-X. Link between DYRK1A overexpression and several-fold enhancement of neurofibrillary degeneration with 3-repeat tau protein in Down syndrome. J. Neuropathol. Exp. Neurol. 2011;70:36–50. doi: 10.1097/NEN.0b013e318202bfa1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Guimera J., Casas C., Estivill X., Pritchard M. Human minibrain homologue (MNBH/DYRK1): characterization, alternative splicing, differential tissue expression, and overexpression in Down syndrome. Genomics. 1999;57:407–418. doi: 10.1006/geno.1999.5775. [DOI] [PubMed] [Google Scholar]
- 105.Kim M.-Y., Jeong B.C., Lee J.H., Kee H.J., Kook H., Kim N.S., Kim Y.H., Kim J.-K., Ahn K.Y., Kim K.K. A repressor complex, AP4 transcription factor and geminin, negatively regulates expression of target genes in nonneuronal cells. Proc. Natl. Acad. Sci. U.S.A. 2006;103:13074–13079. doi: 10.1073/pnas.0601915103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Arron J.R., Winslow M.M., Polleri A., Chang C.-P., Wu H., Gao X., Neilson J.R., Chen L., Heit J.J., Kim S.K., Yamasaki N., Miyakawa T., Francke U., Graef I.A., Crabtree G.R. NFAT dysregulation by increased dosage of DSCR1 and DYRK1A on chromosome 21. Nature. 2006;441:595–600. doi: 10.1038/nature04678. [DOI] [PubMed] [Google Scholar]
- 107.Handschin C., Rhee J., Lin J., Tarr P.T., Spiegelman B.M. An autoregulatory loop controls peroxisome proliferator-activated receptor gamma coactivator 1alpha expression in muscle. Proc. Natl. Acad. Sci. U.S.A. 2003;100:7111–7116. doi: 10.1073/pnas.1232352100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ventura-Clapier R., Garnier A., Veksler V. Transcriptional control of mitochondrial biogenesis: the central role of PGC-1alpha. Cardiovasc. Res. 2008;79:208–217. doi: 10.1093/cvr/cvn098. [DOI] [PubMed] [Google Scholar]
- 109.Altug-Teber O., Bonin M., Walter M., Mau-Holzmann U.A., Dufke A., Stappert H., Tekesin I., Heilbronner H., Nieselt K., Riess O. Specific transcriptional changes in human fetuses with autosomal trisomies. Cytogenet. Genome Res. 2007;119:171–184. doi: 10.1159/000112058. [DOI] [PubMed] [Google Scholar]
- 110.Volk M., Maver A., Lovrečić L., Juvan P., Peterlin B. Expression signature as a biomarker for prenatal diagnosis of trisomy 21. PloS One. 2013;8 doi: 10.1371/journal.pone.0074184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gimeno A., García-Giménez J.L., Audí L., Toran N., Andaluz P., Dasí F., Viña J., V Pallardó F. Decreased cell proliferation and higher oxidative stress in fibroblasts from Down Syndrome fetuses. Preliminary study. Biochim. Biophys. Acta. 2014;1842:116–125. doi: 10.1016/j.bbadis.2013.10.014. [DOI] [PubMed] [Google Scholar]
- 112.Baek K.-H., Zaslavsky A., Lynch R.C., Britt C., Okada Y., Siarey R.J., Lensch M.W., Park I.-H., Yoon S.S., Minami T., Korenberg J.R., Folkman J., Daley G.Q., Aird W.C., Galdzicki Z., Ryeom S. Down's syndrome suppression of tumour growth and the role of the calcineurin inhibitor DSCR1. Nature. 2009;459:1126–1130. doi: 10.1038/nature08062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Fuentes J.J., Genescà L., Kingsbury T.J., Cunningham K.W., Pérez-Riba M., Estivill X., de la Luna S. DSCR1, overexpressed in Down syndrome, is an inhibitor of calcineurin-mediated signaling pathways. Hum. Mol. Genet. 2000;9:1681–1690. doi: 10.1093/hmg/9.11.1681. [DOI] [PubMed] [Google Scholar]
- 114.Dashinimaev E.B., Artyuhov A.S., Bolshakov A.P., Vorotelyak E.A., V Vasiliev A. Neurons derived from induced pluripotent stem cells of patients with down syndrome reproduce early stages of alzheimer's disease type pathology in vitro. J. Alzheimers. Dis. 2017;56:835–847. doi: 10.3233/JAD-160945. [DOI] [PubMed] [Google Scholar]
- 115.Ermak G., Sojitra S., Yin F., Cadenas E., Cuervo A.M., Davies K.J.A. Chronic expression of RCAN1-1L protein induces mitochondrial autophagy and metabolic shift from oxidative phosphorylation to glycolysis in neuronal cells. J. Biol. Chem. 2012;287:14088–14098. doi: 10.1074/jbc.M111.305342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Peiris H., Duffield M.D., Fadista J., Jessup C.F., Kashmir V., Genders A.J., McGee S.L., Martin A.M., Saiedi M., Morton N., Carter R., Cousin M.A., Kokotos A.C., Oskolkov N., Volkov P., Hough T.A., Fisher E.M.C., Tybulewicz V.L.J., Busciglio J., Coskun P.E., Becker A., V Belichenko P., Mobley W.C., Ryan M.T., Chan J.Y., Laybutt D.R., Coates P.T., Yang S., Ling C., Groop L., Pritchard M.A., Keating D.J. A syntenic cross species aneuploidy genetic screen links RCAN1 expression to β-cell mitochondrial dysfunction in type 2 diabetes. PLoS Genet. 2016;12 doi: 10.1371/journal.pgen.1006033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sun L., Hao Y., An R., Li H., Xi C., Shen G. Overexpression of Rcan1-1L inhibits hypoxia-induced cell apoptosis through induction of mitophagy. Mol. Cell. 2014;37:785–794. doi: 10.14348/molcells.2014.0103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Yan L., Yang H., Li Y., Duan H., Wu J., Qian P., Li B., Wang S. Regulator of calcineurin 1-1L protects cardiomyocytes against hypoxia-induced apoptosis via mitophagy. J. Cardiovasc. Pharmacol. 2014;64:310–317. doi: 10.1097/FJC.0000000000000121. [DOI] [PubMed] [Google Scholar]
- 119.Lim S., Hwang S., Yu J.H., Lim J.W., Kim H. Lycopene inhibits regulator of calcineurin 1-mediated apoptosis by reducing oxidative stress and down-regulating Nucling in neuronal cells. Mol. Nutr. Food Res. 2017;61 doi: 10.1002/mnfr.201600530. [DOI] [PubMed] [Google Scholar]
- 120.Izzo A., Manco R., Bonfiglio F., Calì G., De Cristofaro T., Patergnani S., Cicatiello R., Scrima R., Zannini M., Pinton P., Conti A., Nitsch L. NRIP1/RIP140 siRNA-mediated attenuation counteracts mitochondrial dysfunction in Down syndrome. Hum. Mol. Genet. 2014;23:4406–4419. doi: 10.1093/hmg/ddu157. [DOI] [PubMed] [Google Scholar]
- 121.Wang X., Ren S., Yu W., Mu Q., Ye S., Cui C., Guo J. Cognitive impairments in adult mice with RIP140 overexpression in neural stem cells. Behav. Brain Res. 2020;393:112777. doi: 10.1016/j.bbr.2020.112777. [DOI] [PubMed] [Google Scholar]
- 122.Leonardsson G., Steel J.H., Christian M., Pocock V., Milligan S., Bell J., So P.-W., Medina-Gomez G., Vidal-Puig A., White R., Parker M.G. Nuclear receptor corepressor RIP140 regulates fat accumulation. Proc. Natl. Acad. Sci. U.S.A. 2004;101:8437–8442. doi: 10.1073/pnas.0401013101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Powelka A.M., Seth A., V Virbasius J., Kiskinis E., Nicoloro S.M., Guilherme A., Tang X., Straubhaar J., Cherniack A.D., Parker M.G., Czech M.P. Suppression of oxidative metabolism and mitochondrial biogenesis by the transcriptional corepressor RIP140 in mouse adipocytes. J. Clin. Invest. 2006;116:125–136. doi: 10.1172/JCI26040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Haden D.W., Suliman H.B., Carraway M.S., Welty-Wolf K.E., Ali A.S., Shitara H., Yonekawa H., Piantadosi C.A. Mitochondrial biogenesis restores oxidative metabolism during Staphylococcus aureus sepsis. Am. J. Respir. Crit. Care Med. 2007;176:768–777. doi: 10.1164/rccm.200701-161OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Puri V., Chakladar A., V Virbasius J., Konda S., Powelka A.M., Chouinard M., Hagan G.N., Perugini R., Czech M.P. RNAi-based gene silencing in primary mouse and human adipose tissues. J. Lipid Res. 2007;48:465–471. doi: 10.1194/jlr.D600033-JLR200. [DOI] [PubMed] [Google Scholar]
- 126.Seth A., Steel J.H., Nichol D., Pocock V., Kumaran M.K., Fritah A., Mobberley M., Ryder T.A., Rowlerson A., Scott J., Poutanen M., White R., Parker M. The transcriptional corepressor RIP140 regulates oxidative metabolism in skeletal muscle. Cell Metabol. 2007;6:236–245. doi: 10.1016/j.cmet.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Williams D.B., Sutherland L.N., Bomhof M.R., Basaraba S.A.U., Thrush A.B., Dyck D.J., Field C.J., Wright D.C. Muscle-specific differences in the response of mitochondrial proteins to beta-GPA feeding: an evaluation of potential mechanisms. Am. J. Physiol. Endocrinol. Metab. 2009;296:E1400–E1408. doi: 10.1152/ajpendo.90913.2008. [DOI] [PubMed] [Google Scholar]
- 128.Fritah A., Steel J.H., Nichol D., Parker N., Williams S., Price A., Strauss L., Ryder T.A., Mobberley M.A., Poutanen M., Parker M., White R. Elevated expression of the metabolic regulator receptor-interacting protein 140 results in cardiac hypertrophy and impaired cardiac function. Cardiovasc. Res. 2010;86:443–451. doi: 10.1093/cvr/cvp418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chen Y., Wang Y., Chen J., Chen X., Cao W., Chen S., Xu S., Huang H., Liu P. Roles of transcriptional corepressor RIP140 and coactivator PGC-1α in energy state of chronically infarcted rat hearts and mitochondrial function of cardiomyocytes. Mol. Cell. Endocrinol. 2012;362:11–18. doi: 10.1016/j.mce.2012.03.023. [DOI] [PubMed] [Google Scholar]
- 130.Hoshino D., Yoshida Y., Holloway G.P., Lally J., Hatta H., Bonen A. Clenbuterol, a β2-adrenergic agonist, reciprocally alters PGC-1 alpha and RIP140 and reduces fatty acid and pyruvate oxidation in rat skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012;302:R373–R384. doi: 10.1152/ajpregu.00183.2011. [DOI] [PubMed] [Google Scholar]
- 131.Furrer R., DE Haan A., Bravenboer N., Kos D., Lips P., Jaspers R.T. Effects of concurrent training on oxidative capacity in rat gastrocnemius muscle. Med. Sci. Sports Exerc. 2013;45:1674–1683. doi: 10.1249/MSS.0b013e31828fc65f. [DOI] [PubMed] [Google Scholar]
- 132.Zhang L., Chen Y., Yue Z., He Y., Zou J., Chen S., Liu M., Chen X., Liu Z., Liu X., Feng X., Li M., Liu P. The p65 subunit of NF-κB involves in RIP140-mediated inflammatory and metabolic dysregulation in cardiomyocytes. Arch. Biochem. Biophys. 2014;554:22–27. doi: 10.1016/j.abb.2014.05.005. [DOI] [PubMed] [Google Scholar]
- 133.Herbst E.A.F., Bonen A., Holloway G.P. Changes in nuclear receptor corepressor RIP140 do not influence mitochondrial content in the cortex. Appl. Physiol. Nutr. Metabol. 2015;40:1086–1088. doi: 10.1139/apnm-2015-0192. [DOI] [PubMed] [Google Scholar]
- 134.Miotto P.M., Frendo-Cumbo S., Sacco S.M., Wright D.C., Ward W.E., Holloway G.P. Combined high-fat-resveratrol diet and RIP140 knockout mice reveal a novel relationship between elevated bone mitochondrial content and compromised bone microarchitecture, bone mineral mass, and bone strength in the tibia. Mol. Nutr. Food Res. 2016;60 doi: 10.1002/mnfr.201500870. [DOI] [PubMed] [Google Scholar]
- 135.Chen Y., Chen S., Yue Z., Zhang Y., Zhou C., Cao W., Chen X., Zhang L., Liu P. Receptor-interacting protein 140 overexpression impairs cardiac mitochondrial function and accelerates the transition to heart failure in chronically infarcted rats. Transl. Res. 2017;180:91–102. doi: 10.1016/j.trsl.2016.08.005. e1. [DOI] [PubMed] [Google Scholar]
- 136.You J., Yue Z., Chen S., Chen Y., Lu X., Zhang X., Shen P., Li J., Han Q., Li Z., Liu P. Receptor-interacting Protein 140 represses Sirtuin 3 to facilitate hypertrophy, mitochondrial dysfunction and energy metabolic dysfunction in cardiomyocytes. Acta Physiol. 2017;220:58–71. doi: 10.1111/apha.12800. [DOI] [PubMed] [Google Scholar]
- 137.Park S.-S., Lee Y.-J., Kang H., Yang G., Hong E.J., Lim J.Y., Oh S., Kim E. Lactobacillus amylovorus KU4 ameliorates diet-induced obesity in mice by promoting adipose browning through PPARγ signaling. Sci. Rep. 2019;9 doi: 10.1038/s41598-019-56817-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Birket M.J., Orr A.L., Gerencser A.A., Madden D.T., Vitelli C., Swistowski A., Brand M.D., Zeng X. A reduction in ATP demand and mitochondrial activity with neural differentiation of human embryonic stem cells. J. Cell Sci. 2011;124:348–358. doi: 10.1242/jcs.072272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Sánchez-Font M.F., Bosch-Comas A., Gonzàlez-Duarte R., Marfany G. Overexpression of FABP7 in Down syndrome fetal brains is associated with PKNOX1 gene-dosage imbalance. Nucleic Acids Res. 2003;31:2769–2777. doi: 10.1093/nar/gkg396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Micali N., Longobardi E., Iotti G., Ferrai C., Castagnaro L., Ricciardi M., Blasi F., Crippa M.P. Down syndrome fibroblasts and mouse Prep1-overexpressing cells display increased sensitivity to genotoxic stress. Nucleic Acids Res. 2010;38:3595–3604. doi: 10.1093/nar/gkq019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Kanzleiter T., Rath M., Penkov D., Puchkov D., Schulz N., Blasi F., Schürmann A. Pknox1/Prep1 regulates mitochondrial oxidative phosphorylation components in skeletal muscle. Mol. Cell Biol. 2014;34:290–298. doi: 10.1128/MCB.01232-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.You S.-H., Lee Y.-S., Chang Y.-J., Lin C.-Y., Wang T.-H., Chang Y.-L. Gene expression profiling of amniotic fluid mesenchymal stem cells of monozygotic twins discordant for trisomy 21. Gene. 2020;738 doi: 10.1016/j.gene.2020.144461. [DOI] [PubMed] [Google Scholar]
- 143.Ferrando-Miguel R., Cheon M.S., Yang J.W., Lubec G. Overexpression of transcription factor BACH1 in fetal Down syndrome brain. J. Neural. Transm. 2003;Suppl:193–205. doi: 10.1007/978-3-7091-6721-2_17. [DOI] [PubMed] [Google Scholar]
- 144.Lee J., Yesilkanal A.E., Wynne J.P., Frankenberger C., Liu J., Yan J., Elbaz M., Rabe D.C., Rustandy F.D., Tiwari P., Grossman E.A., Hart P.C., Kang C., Sanderson S.M., Andrade J., Nomura D.K., Bonini M.G., Locasale J.W., Rosner M.R. Effective breast cancer combination therapy targeting BACH1 and mitochondrial metabolism. Nature. 2019;568:254–258. doi: 10.1038/s41586-019-1005-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.St-Pierre J., Drori S., Uldry M., Silvaggi J.M., Rhee J., Jäger S., Handschin C., Zheng K., Lin J., Yang W., Simon D.K., Bachoo R., Spiegelman B.M. Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell. 2006;127:397–408. doi: 10.1016/j.cell.2006.09.024. [DOI] [PubMed] [Google Scholar]
- 146.Mollo N., Cicatiello R., Aurilia M., Scognamiglio R., Genesio R., Charalambous M., Paladino S., Conti A., Nitsch L., Izzo A. Targeting mitochondrial network architecture in down syndrome and aging. Int. J. Mol. Sci. 2020;21 doi: 10.3390/ijms21093134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Guedj F., Bianchi D.W., Delabar J.-M. Prenatal treatment of Down syndrome: a reality? Curr. Opin. Obstet. Gynecol. 2014;26:92–103. doi: 10.1097/GCO.0000000000000056. [DOI] [PubMed] [Google Scholar]
- 148.Stagni F., Giacomini A., Guidi S., Ciani E., Bartesaghi R. Timing of therapies for Down syndrome: the sooner, the better. Front. Behav. Neurosci. 2015;9:265. doi: 10.3389/fnbeh.2015.00265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Jia L., Wang J., Cao H., Zhang X., Rong W., Xu Z. Activation of PGC-1α and mitochondrial biogenesis protects against prenatal hypoxic-ischemic brain injury. Neuroscience. 2020;432:63–72. doi: 10.1016/j.neuroscience.2020.02.035. [DOI] [PubMed] [Google Scholar]
- 150.Vacca R.A., Valenti D., Caccamese S., Daglia M., Braidy N., Nabavi S.M. Plant polyphenols as natural drugs for the management of Down syndrome and related disorders. Neurosci. Biobehav. Rev. 2016;71:865–877. doi: 10.1016/j.neubiorev.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 151.Valenti D., Braidy N., De Rasmo D., Signorile A., Rossi L., Atanasov A.G., Volpicella M., Henrion-Caude A., Nabavi S.M., Vacca R.A. Mitochondria as pharmacological targets in Down syndrome. Free Radic. Biol. Med. 2018;114:69–83. doi: 10.1016/j.freeradbiomed.2017.08.014. [DOI] [PubMed] [Google Scholar]
- 152.Vacca R.A., Bawari S., Valenti D., Tewari D., Nabavi S.F., Shirooie S., Sah A.N., Volpicella M., Braidy N., Nabavi S.M. Down syndrome: neurobiological alterations and therapeutic targets. Neurosci. Biobehav. Rev. 2019;98:234–255. doi: 10.1016/j.neubiorev.2019.01.001. [DOI] [PubMed] [Google Scholar]
- 153.Zuhra K., Augsburger F., Majtan T., Szabo C. Cystathionine-β-Synthase: molecular regulation and pharmacological inhibition. Biomolecules. 2020;10 doi: 10.3390/biom10050697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Feki A., Hibaoui Y. DYRK1A protein, A promising therapeutic target to improve cognitive deficits in down syndrome. Brain Sci. 2018;8 doi: 10.3390/brainsci8100187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Chaturvedi R.K., Calingasan N.Y., Yang L., Hennessey T., Johri A., Beal M.F. Impairment of PGC-1alpha expression, neuropathology and hepatic steatosis in a transgenic mouse model of Huntington's disease following chronic energy deprivation. Hum. Mol. Genet. 2010;19:3190–3205. doi: 10.1093/hmg/ddq229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Vaughan R.A., Garcia-Smith R., Bisoffi M., Trujillo K.A., Conn C.A. Effects of caffeine on metabolism and mitochondria biogenesis in rhabdomyosarcoma cells compared with 2,4-dinitrophenol. Nutr. Metab. Insights. 2012;5:59–70. doi: 10.4137/NMI.S10233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Shi W., Li L., Ding Y., Yang K., Chen Z., Fan X., Jiang S., Guan Y., Liu Z., Xu D., Wu L. The critical role of epigallocatechin gallate in regulating mitochondrial metabolism. Future Med. Chem. 2018;10:795–809. doi: 10.4155/fmc-2017-0204. [DOI] [PubMed] [Google Scholar]
- 158.Ruiz-Pesini E., Emperador S., López-Gallardo E., Hernández-Ainsa C., Montoya J. Increasing mtDNA levels as therapy for mitochondrial optic neuropathies. Drug Discov. Today. 2018;23:493–498. doi: 10.1016/j.drudis.2018.01.031. [DOI] [PubMed] [Google Scholar]
- 159.Arbones M.L., Thomazeau A., Nakano-Kobayashi A., Hagiwara M., Delabar J.M. DYRK1A and cognition: a lifelong relationship. Pharmacol. Ther. 2019;194:199–221. doi: 10.1016/j.pharmthera.2018.09.010. [DOI] [PubMed] [Google Scholar]
- 160.Zhang J.-C., Xu H., Yuan Y., Chen J.-Y., Zhang Y.-J., Lin Y., Yuan S.-Y. Delayed treatment with green tea polyphenol EGCG promotes neurogenesis after ischemic stroke in adult mice. Mol. Neurobiol. 2017;54:3652–3664. doi: 10.1007/s12035-016-9924-0. [DOI] [PubMed] [Google Scholar]
- 161.Stagni F., Giacomini A., Emili M., Trazzi S., Guidi S., Sassi M., Ciani E., Rimondini R., Bartesaghi R. Short- and long-term effects of neonatal pharmacotherapy with epigallocatechin-3-gallate on hippocampal development in the Ts65Dn mouse model of Down syndrome. Neuroscience. 2016;333:277–301. doi: 10.1016/j.neuroscience.2016.07.031. [DOI] [PubMed] [Google Scholar]
- 162.Souchet B., Duchon A., Gu Y., Dairou J., Chevalier C., Daubigney F., Nalesso V., Créau N., Yu Y., Janel N., Herault Y., Delabar J.M. Prenatal treatment with EGCG enriched green tea extract rescues GAD67 related developmental and cognitive defects in Down syndrome mouse models. Sci. Rep. 2019;9:3914. doi: 10.1038/s41598-019-40328-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Vacca R.A., Valenti D. Green tea EGCG plus fish oil omega-3 dietary supplements rescue mitochondrial dysfunctions and are safe in a Down's syndrome child. Clin. Nutr. 2015;34:783–784. doi: 10.1016/j.clnu.2015.04.012. [DOI] [PubMed] [Google Scholar]
- 164.Lee M.-S., Shin Y., Moon S., Kim S., Kim Y. Effects of eicosapentaenoic acid and docosahexaenoic acid on mitochondrial DNA replication and PGC-1α gene expression in C2C12 muscle cells. Prev. Nutr. Food Sci. 2016;21:317–322. doi: 10.3746/pnf.2016.21.4.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Zmijewski P.A., Gao L.Y., Saxena A.R., Chavannes N.K., Hushmendy S.F., Bhoiwala D.L., Crawford D.R. Fish oil improves gene targets of Down syndrome in C57BL and BALB/c mice. Nutr. Res. 2015;35:440–448. doi: 10.1016/j.nutres.2015.02.007. [DOI] [PubMed] [Google Scholar]
- 166.Zhou X., Wu W., Chen J., Wang X., Wang Y. AMP-activated protein kinase is required for the anti-adipogenic effects of alpha-linolenic acid. Nutr. Metab. 2015;12:10. doi: 10.1186/s12986-015-0006-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.You M., Fan R., Kim J., Shin S.-H., Chung S. Alpha-linolenic acid-enriched butter promotes fatty acid remodeling and thermogenic activation in the Brown adipose tissue. Nutrients. 2020;12 doi: 10.3390/nu12010136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Litwiniuk A., Domańska A., Chmielowska M., Martyńska L., Bik W., Kalisz M. The effects of alpha-linolenic acid on the secretory activity of astrocytes and β amyloid-associated neurodegeneration in differentiated SH-SY5Y cells: alpha-linolenic acid protects the SH-SY5Y cells against β amyloid toxicity. Oxid. Med. Cell. Longev. 2020;2020 doi: 10.1155/2020/8908901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.García-Cerro S., Rueda N., Vidal V., Puente A., Campa V., Lantigua S., Narcís O., Velasco A., Bartesaghi R., Martínez-Cué C. Prenatal administration of oleic acid or linolenic acid reduces neuromorphological and cognitive alterations in Ts65dn down syndrome mice. J. Nutr. 2020;150:1631–1643. doi: 10.1093/jn/nxaa074. [DOI] [PubMed] [Google Scholar]
- 170.Niculescu M.D., Lupu D.S., Craciunescu C.N. Maternal α-linolenic acid availability during gestation and lactation alters the postnatal hippocampal development in the mouse offspring. Int. J. Dev. Neurosci. 2011;29:795–802. doi: 10.1016/j.ijdevneu.2011.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Ljubicic V., Jasmin B.J. Metformin increases peroxisome proliferator-activated receptor γ Co-activator-1α and utrophin a expression in dystrophic skeletal muscle. Muscle Nerve. 2015;52:139–142. doi: 10.1002/mus.24692. [DOI] [PubMed] [Google Scholar]
- 172.Karnewar S., Neeli P.K., Panuganti D., Kotagiri S., Mallappa S., Jain N., Jerald M.K., Kotamraju S. Metformin regulates mitochondrial biogenesis and senescence through AMPK mediated H3K79 methylation: relevance in age-associated vascular dysfunction. Biochim. Biophys. Acta (BBA) - Mol. Basis Dis. 2018;1864:1115–1128. doi: 10.1016/j.bbadis.2018.01.018. [DOI] [PubMed] [Google Scholar]
- 173.Ahn M.-J., Cho G.-W. Metformin promotes neuronal differentiation and neurite outgrowth through AMPK activation in human bone marrow-mesenchymal stem cells. Biotechnol. Appl. Biochem. 2017;64:836–842. doi: 10.1002/bab.1584. [DOI] [PubMed] [Google Scholar]
- 174.Gureev A.P., Shaforostova E.A., Starkov A.A., Popov V.N. β-Guanidinopropionic acid stimulates brain mitochondria biogenesis and alters cognitive behavior in nondiseased mid-age mice. J. Exp. Neurosci. 2018;12 doi: 10.1177/1179069518766524. 1179069518766524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Ahuja M., Ammal Kaidery N., Yang L., Calingasan N., Smirnova N., Gaisin A., Gaisina I.N., Gazaryan I., Hushpulian D.M., Kaddour-Djebbar I., Bollag W.B., Morgan J.C., Ratan R.R., Starkov A.A., Beal M.F., Thomas B. Distinct Nrf2 signaling mechanisms of fumaric acid esters and their role in neuroprotection against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced experimental Parkinson’s-like disease. J. Neurosci. 2016;36:6332–6351. doi: 10.1523/JNEUROSCI.0426-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Guidi S., Stagni F., Bianchi P., Ciani E., Giacomini A., De Franceschi M., Moldrich R., Kurniawan N., Mardon K., Giuliani A., Calzà L., Bartesaghi R. Prenatal pharmacotherapy rescues brain development in a Down's syndrome mouse model. Brain. 2014;137:380–401. doi: 10.1093/brain/awt340. [DOI] [PubMed] [Google Scholar]
- 177.Guedj F., Siegel A.E., Pennings J.L.A., Alsebaa F., Massingham L.J., Tantravahi U., Bianchi D.W. Apigenin as a candidate prenatal treatment for trisomy 21: effects in human amniocytes and the Ts1Cje mouse model. Am. J. Hum. Genet. 2020;107:911–931. doi: 10.1016/j.ajhg.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Rueda N., Vidal V., García-Cerro S., Puente A., Campa V., Lantigua S., Narcís O., Bartesaghi R., Martínez-Cué C. Prenatal, but not postnatal, curcumin administration rescues neuromorphological and cognitive alterations in Ts65Dn down syndrome mice. J. Nutr. 2020;150:2478–2489. doi: 10.1093/jn/nxaa207. [DOI] [PubMed] [Google Scholar]
- 179.Liu L., Zhang W., Wang L., Li Y., Tan B., Lu X., Deng Y., Zhang Y., Guo X., Mu J., Yu G. Curcumin prevents cerebral ischemia reperfusion injury via increase of mitochondrial biogenesis. Neurochem. Res. 2014;39:1322–1331. doi: 10.1007/s11064-014-1315-1. [DOI] [PubMed] [Google Scholar]
- 180.Trujillo J., Granados-Castro L.F., Zazueta C., Andérica-Romero A.C., Chirino Y.I., Pedraza-Chaverrí J. Mitochondria as a target in the therapeutic properties of curcumin. Arch. Pharm. (Weinheim) 2014;347:873–884. doi: 10.1002/ardp.201400266. [DOI] [PubMed] [Google Scholar]
- 181.de Oliveira M.R., Jardim F.R., Setzer W.N., Nabavi S.M., Nabavi S.F. Curcumin, mitochondrial biogenesis, and mitophagy: exploring recent data and indicating future needs. Biotechnol. Adv. 2016;34:813–826. doi: 10.1016/j.biotechadv.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 182.Choi W.H., Son H.J., Jang Y.J., Ahn J., Jung C.H., Ha T.Y. Apigenin ameliorates the obesity‐induced skeletal muscle atrophy by attenuating mitochondrial dysfunction in the muscle of obese mice. Mol. Nutr. Food Res. 2017;61 doi: 10.1002/mnfr.201700218. [DOI] [PubMed] [Google Scholar]
- 183.Barros M. da L.D., Manhães-de-Castro R., Alves D.T., Quevedo O.G., Toscano A.E., Bonnin A., Galindo L. Long term effects of neonatal exposure to fluoxetine on energy balance: a systematic review of experimental studies. Eur. J. Pharmacol. 2018;833:298–306. doi: 10.1016/j.ejphar.2018.06.013. [DOI] [PubMed] [Google Scholar]
- 184.Simões‐Alves A.C., Silva‐Filho R.C., Braz G.R.F., Silva S.C.A., da Silva A.I., Lagranha C.J., Fernandes M.P. Neonatal treatment with fluoxetine improves mitochondrial respiration and reduces oxidative stress in liver of adult rats. J. Cell. Biochem. 2018;119:6555–6565. doi: 10.1002/jcb.26758. [DOI] [PubMed] [Google Scholar]
- 185.Braz G.R.F., Silva S.C. de A., Pedroza A.A. da S., de Lemos M.D., de Lima F.A., da Silva A.I., Lagranha C.J. Fluoxetine administration in juvenile overfed rats improves hypothalamic mitochondrial respiration and REDOX status and induces mitochondrial biogenesis transcriptional expression. Eur. J. Pharmacol. 2020;881 doi: 10.1016/j.ejphar.2020.173200. [DOI] [PubMed] [Google Scholar]
- 186.Scapagnini G., Sonya V., Nader A.G., Calogero C., Zella D., Fabio G. Modulation of nrf2/ARE pathway by food polyphenols: a nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol. Neurobiol. 2011;44:192–201. doi: 10.1007/s12035-011-8181-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Mendez-David I., Tritschler L., El Ali Z., Damiens M.-H., Pallardy M., David D.J., Kerdine-Römer S., Gardier A.M. Nrf2-signaling and BDNF: a new target for the antidepressant-like activity of chronic fluoxetine treatment in a mouse model of anxiety/depression. Neurosci. Lett. 2015;597:121–126. doi: 10.1016/j.neulet.2015.04.036. [DOI] [PubMed] [Google Scholar]
- 188.Hong C., Cao J., Wu C.-F., Kadioglu O., Schüffler A., Kauhl U., Klauck S.M., Opatz T., Thines E., Paul N.W., Efferth T. The Chinese herbal formula Free and Easy Wanderer ameliorates oxidative stress through KEAP1-NRF2/HO-1 pathway. Sci. Rep. 2017;7 doi: 10.1038/s41598-017-10443-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Perez-Leal O., Barrero C.A., Merali S. Pharmacological stimulation of nuclear factor (erythroid-derived 2)-like 2 translation activates antioxidant responses. J. Biol. Chem. 2017;292:14108–14121. doi: 10.1074/jbc.M116.770925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Robledinos-Antón N., Fernández-Ginés R., Manda G., Cuadrado A. Activators and inhibitors of NRF2: a review of their potential for clinical development. Oxid. Med. Cell. Longev. 2019:1–20. doi: 10.1155/2019/9372182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Yammine A., Zarrouk A., Nury T., Vejux A., Latruffe N., Vervandier-Fasseur D., Samadi M., Mackrill J.J., Greige-Gerges H., Auezova L., Lizard G. Prevention by dietary polyphenols (resveratrol, quercetin, apigenin) against 7-ketocholesterol-induced oxiapoptophagy in neuronal N2a cells: potential interest for the treatment of neurodegenerative and age-related diseases. Cells. 2020;9:2346. doi: 10.3390/cells9112346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Ghosh S., Patel N., Rahn D., McAllister J., Sadeghi S., Horwitz G., Berry D., Wang K.X., Swerdlow R.H. The thiazolidinedione pioglitazone alters mitochondrial function in human neuron-like cells. Mol. Pharmacol. 2007;71:1695–1702. doi: 10.1124/mol.106.033845. [DOI] [PubMed] [Google Scholar]
- 193.Komen J.C., Thorburn D.R. Turn up the power - pharmacological activation of mitochondrial biogenesis in mouse models. Br. J. Pharmacol. 2014;171:1818–1836. doi: 10.1111/bph.12413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Mollo N., Nitti M., Zerillo L., Faicchia D., Micillo T., Accarino R., Secondo A., Petrozziello T., Calì G., Cicatiello R., Bonfiglio F., Sarnataro V., Genesio R., Izzo A., Pinton P., Matarese G., Paladino S., Conti A., Nitsch L. Pioglitazone improves mitochondrial organization and bioenergetics in down syndrome cells. Front. Genet. 2019;10:606. doi: 10.3389/fgene.2019.00606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Miglio G., Rattazzi L., Rosa A.C., Fantozzi R. PPARgamma stimulation promotes neurite outgrowth in SH-SY5Y human neuroblastoma cells. Neurosci. Lett. 2009;454:134–138. doi: 10.1016/j.neulet.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 196.Cerqueira F.M., Laurindo F.R.M., Kowaltowski A.J. Mild mitochondrial uncoupling and calorie restriction increase fasting eNOS, akt and mitochondrial biogenesis. PloS One. 2011;6 doi: 10.1371/journal.pone.0018433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Schlagowski A.I., Singh F., Charles A.L., Gali Ramamoorthy T., Favret F., Piquard F., Geny B., Zoll J. Mitochondrial uncoupling reduces exercise capacity despite several skeletal muscle metabolic adaptations. J. Appl. Physiol. 2014;116:364–375. doi: 10.1152/japplphysiol.01177.2013. [DOI] [PubMed] [Google Scholar]
- 198.Farber S.A., Slack B.E., Blusztajn J.K. Acceleration of phosphatidylcholine synthesis and breakdown by inhibitors of mitochondrial function in neuronal cells: a model of the membrane defect of Alzheimer's disease. Faseb. J. 2000;14:2198–2206. doi: 10.1096/fj.99-0853. [DOI] [PubMed] [Google Scholar]
- 199.Wasilewska-Sampaio A.P., Silveira M.S., Holub O., Goecking R., Gomes F.C.A., Neto V.M., Linden R., Ferreira S.T., De Felice F.G. Neuritogenesis and neuronal differentiation promoted by 2,4-dinitrophenol, a novel anti-amyloidogenic compound. Faseb. J. 2005;19:1627–1636. doi: 10.1096/fj.05-3812com. [DOI] [PubMed] [Google Scholar]
- 200.Freitas-Correa L., V Lourenco M., Acquarone M., da Costa R.F.M., Galina A., Rehen S.K., Ferreira S.T. 2,4-Dinitrophenol induces neural differentiation of murine embryonic stem cells. Stem Cell Res. 2013;11:1407–1416. doi: 10.1016/j.scr.2013.09.016. [DOI] [PubMed] [Google Scholar]
- 201.Murphy E.J., Schapiro M.B., Rapoport S.I., Shetty H.U. Phospholipid composition and levels are altered in Down syndrome brain. Brain Res. 2000;867:9–18. doi: 10.1016/s0006-8993(00)02205-8. [DOI] [PubMed] [Google Scholar]
- 202.Paula Lima A.C., Arriagada C., Toro R., Cárdenas A.M., Caviedes R., Ferreira S.T., Caviedes P. Small-molecule aggregation inhibitors reduce excess amyloid in a trisomy 16 mouse cortical cell line. Biol. Res. 2008;41:129–136. doi: 10.4067/s0716-97602008000200001. [DOI] [PubMed] [Google Scholar]
- 203.Hasan-Olive M.M., Lauritzen K.H., Ali M., Rasmussen L.J., Storm-Mathisen J., Bergersen L.H. A ketogenic diet improves mitochondrial biogenesis and bioenergetics via the PGC1α-SIRT3-UCP2 Axis. Neurochem. Res. 2019;44:22–37. doi: 10.1007/s11064-018-2588-6. [DOI] [PubMed] [Google Scholar]
- 204.Rosebrough R.W., Steele N.C., Frobish L.T. Effect of ketogenic diets in gestation on some characteristics of carbohydrate metabolism in fetal pig brain and liver. Growth. 1981;45:42–57. http://www.ncbi.nlm.nih.gov/pubmed/6785157 [PubMed] [Google Scholar]
- 205.Pliss L., Jatania U., Patel M.S. Beneficial effect of feeding a ketogenic diet to mothers on brain development in their progeny with a murine model of pyruvate dehydrogenase complex deficiency. Mol. Genet. Metab. Reports. 2016;7:78–86. doi: 10.1016/j.ymgmr.2016.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Kaneko K. The ketogenic diet as a potential therapy in down syndrome. J. Pediatr. Pediatr. Med. 2018;2:11–17. doi: 10.29245/2578-2940/2018/2.1121. [DOI] [Google Scholar]
- 207.Pesini A., Iglesias E., Garrido N., Bayona-Bafaluy M.P., Montoya J., Ruiz-Pesini E. OXPHOS, pyrimidine nucleotides, and Alzheimer's disease: a pharmacogenomics approach. J. Alzheimers. Dis. 2014;42:87–96. doi: 10.3233/JAD-140384. [DOI] [PubMed] [Google Scholar]
- 208.Lagerlöf O. O-GlcNAc cycling in the developing, adult and geriatric brain. J. Bioenerg. Biomembr. 2018;50:241–261. doi: 10.1007/s10863-018-9760-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Pfenninger K.H. Plasma membrane expansion: a neuron's Herculean task. Nat. Rev. Neurosci. 2009;10:251–261. doi: 10.1038/nrn2593. [DOI] [PubMed] [Google Scholar]
- 210.López-Martín J.M., Salviati L., Trevisson E., Montini G., DiMauro S., Quinzii C., Hirano M., Rodriguez-Hernandez A., Cordero M.D., Sánchez-Alcázar J.A., Santos-Ocaña C., Navas P. Missense mutation of the COQ2 gene causes defects of bioenergetics and de novo pyrimidine synthesis. Hum. Mol. Genet. 2007;16:1091–1097. doi: 10.1093/hmg/ddm058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Gross T.J., Doran E., Cheema A.K., Head E., Lott I.T., Mapstone M. Plasma metabolites related to cellular energy metabolism are altered in adults with Down syndrome and Alzheimer's disease. Dev. Neurobiol. 2019;79:622–638. doi: 10.1002/dneu.22716. [DOI] [PubMed] [Google Scholar]
- 212.Gattermann N., Dadak M., Hofhaus G., Wulfert M., Berneburg M., Loeffler M.L., Simmonds H.A. Severe impairment of nucleotide synthesis through inhibition of mitochondrial respiration. Nucleos Nucleot. Nucleic Acids. 2004;23:1275–1279. doi: 10.1081/NCN-200027545. [DOI] [PubMed] [Google Scholar]
- 213.Velazquez R., Ash J.A., Powers B.E., Kelley C.M., Strawderman M., Luscher Z.I., Ginsberg S.D., Mufson E.J., Strupp B.J. Maternal choline supplementation improves spatial learning and adult hippocampal neurogenesis in the Ts65Dn mouse model of Down syndrome. Neurobiol. Dis. 2013;58:92–101. doi: 10.1016/j.nbd.2013.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Wang Y., Surzenko N., Friday W.B., Zeisel S.H. Maternal dietary intake of choline in mice regulates development of the cerebral cortex in the offspring. Faseb. J. 2016;30:1566–1578. doi: 10.1096/fj.15-282426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Wurtman R.J. Synapse formation in the brain can be enhanced by co-administering three specific nutrients. Eur. J. Pharmacol. 2017;817:20–21. doi: 10.1016/j.ejphar.2017.09.038. [DOI] [PubMed] [Google Scholar]
- 216.Tayebati S.K., Cecchi A., Martinelli I., Carboni E., Amenta F. Pharmacotherapy of down's syndrome: when and which? CNS Neurol. Disord. - Drug Targets. 2020;18:750–757. doi: 10.2174/1871527318666191114092924. [DOI] [PubMed] [Google Scholar]