Abstract

Waterborne acrylic resin is a kind of environmental protection resin, which is widely used in coatings, bridges, ships, and locomotives. In order to be better used in various fields, modification of waterborne acrylic resin has attracted much attention. In this paper, we introduce the method to synthesize waterborne acrylic resins, the composition of the resin, and basic properties of each monomer. According to the requirements of different properties of the resin, the modification mechanism and methods of the resin are discussed, including thermal performance, corrosion resistance, mechanical property, and water resistance. The applications of waterborne acrylic resin in the construction, automobile, metal anticorrosion, and furniture industries are discussed with detailed examples. Finally, the prospect of waterborne acrylic resin is proposed.
1. Introduction
Resin has attracted much attention as an important component of coating.1 Traditional solvent-based resins mainly use organic volatile solvents to reduce the viscosity of resins.2 However, with the increasing awareness of environmental protection, countries around the world have formulated strict environmental protection laws and policies,3 which limit the emission of volatile organic compounds (VOCs),4 restricting the development of traditional solvent-based coatings. An effective improvement is to replace traditional solvent resins with waterborne resin, which mainly use water as the solvent or dispersant and can significantly reduce the VOC emissions. Meanwhile, in the process of production and transportation, waterborne resin has the characteristics of being nonflammable and pollution-free and has attracted more and more attention.
There are many kinds of waterborne resins, among which waterborne acrylic resin is widely used in many fields, such as locomotives, ships, containers, bridges, military industry, and so on. Acrylic resin is synthesized by acrylate or methacrylate as the main monomers and exhibits strong light, heat, and chemical stability.5 Therefore, the coating prepared by acrylic resin has excellent low cost, weather resistance, pollution resistance, as well as acid and alkali resistance.6 In order to disperse well in water, hydrophilic groups need to be introduced into waterborne acrylic resin in the synthesis process, which leads to the formation of water and air that can easily enter the resin molecules. Thus, waterborne acrylic resin has many disadvantages, such as low solid content, poor water resistance, poor corrosion resistance, compactness, and so on.7 These disadvantages limit its application. In order to solve the problems, people use materials with excellent performances to modify waterborne acrylic resins.
This review summarizes the research on the modification of waterborne acrylic resin and analyzes and explores the composition and performance improvement mechanism and introduces the modification methods and application fields of waterborne acrylic resin.
2. Synthesis and Modification Principle of Waterborne Acrylic Resin
2.1. Principle for the Synthesis of Waterborne Acrylic Resin
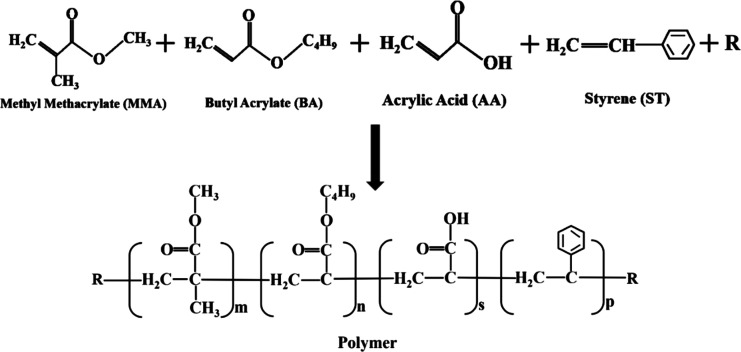
Waterborne acrylic resins are usually copolymerized by acrylic acid, methacrylic acid, and their esters or other derivatives, whose properties can be tailored by regulating the compositions (the basic composition and structure are shown in Figure 1), the ratio of various monomers, and the structure of raw materials. There are many ways to synthesize waterborne acrylic resins (including emulsion, solution polymerization, bulk polymerization, and suspension polymerization), of which emulsion polymerization is the most widely used. It is synthesized by monomers, emulsifiers, water, and initiators. The synthetic macromolecular chain is mainly composed of a carbon chain, which is divided into a soft segment and a hard segment. The hard segment is mainly composed of hard monomers (methacrylate, styrene, etc.), which can improve the hardness of the resin, and the soft segment mainly contains soft monomers such as acrylate, which can provide flexibility for the resin. The structure and composition of the resin framework affect the product performance. Other functional monomers are often used to copolymerize to provide different performance. Some unsaturated carboxylic acids such as acrylic acid, methacrylic acid, maleic anhydride, etc. are selected in the monomers of the carboxyl group, or the hydrophilic groups such as hydroxyl group, amide group, or ether bond are introduced to ensure the water solubility of the waterborne resin. Meanwhile, the acrylic was used to enhance the emulsion stability and the physical cross-linking via ionic clusters after the film formation. The addition of styrene and acrylonitrile can increase the impact resistance, wear resistance, and mechanical properties of the resin.8 The ring structure or benzene ring can improve the mechanical properties of the resin, such as glycidyl methacrylate. 2-Ethylhexyl acrylate and other long-chain alkanes can be selected to receive the flexible coating.
Figure 1.
Structure of waterborne acrylic resin and its monomers (R refers to other acrylate monomers).
2.2. Strategies for Modification of Waterborne Acrylic Resin
According to the improvement of various properties of waterborne acrylic resin explored by researchers in recent years, the modification strategies of four properties is summarized, which include thermal performance, corrosion resistance, mechanical property, and water resistance.
2.2.1. Thermal Performance
Waterborne acrylic resin belongs to thermosetting resin. Their thermal property is one of the key factors for their practical applications, including thermal stability, thermal oxidative stability, and flame retardancy.9 It mainly shows up in the heat resistance of the material and the deformation ability of the object under the influence of temperature. The smaller the deformation is, the higher the stability is. As is known, increasing the cross-linking density of thermosets is one of the most popular methods to improve the thermal stability of polymers. Researchers select the initiator and the active cross-linking monomer to accelerate the cross-linking reaction during the reaction process or add curing agent in the curing process to improve the cross-linking density. It has been found that the carbon layer formed during combustion can protect the material from further degradation at high temperatures, so the functional group with a high content or heteroatom is introduced into the resin during the synthesis process.10 They can form a compact and continuous carbon layer on the polymer surface to improve the thermal properties. At the same time, the polymer chain with a rigid structure also has certain thermal stability, which is one of the directions to solve the thermal performance.11 In addition, copolymerization with heat-resistant resin or composite with flame-retardant nanomaterials is also studied to improve the thermal performance of the resin. The addition methods of nanomaterials into polymers includes physical mixing (nanomaterials added as fillers), in situ polymerization, grafting by silane coupling agents, and so on. Physical mixing may cause poor dispersion of nanomaterials and affect the properties of polymers. The methods of in situ polymerization and grafting by coupling agents are often used to make nanomaterials grafted onto the chain segments of the resin through the action of chemical bonds to disperse evenly and enhance the properties of composite materials. For example, Romo-Uribe et al.12 synthesized waterborne acrylate bentonite (silicate layer) nanocomposites by in situ polymerization. As shown in Figure 2a, the bentonite expands in the water medium to provide the seed for further continuous growth. At the same time, polymerization would give rise to polymer chains intercalating the nanoplatelets and forming the intercalation morphology. The structure makes the nanoclay, which has good thermal properties, combine well with acrylic resin, increases the initial temperature of thermal degradation of the resin by 20 °C (Figure 2b), and enhances the thermal stability of the resin.
Figure 2.
Schematic diagram of nanoclay intercalation by polyacrylic macromolecules (a) and thermogravimetric analysis heating traces of acrylate–bentonite nanocomposites (b). Reproduced with permission from ref (12). Copyright 2016 Elsevier.
2.2.2. Corrosion Resistance
Metal corrosion will cause huge economic loss, and organic coating is one of the most effective ways to prevent corrosion.13 Due to its excellent color retention, environmental friendliness, acid alkali resistance, and chemical resistance, waterborne acrylic resins play an important role in the field of metal corrosion protection. Metal anticorrosion by resin coatings can be divided into two ways. One is external blocking, which can prevent the invasion of electrolyte by reinforcing the resin matrix or by adding nanomaterials or fluorine monomers to improve the hydrophobicity of the resin (Figure 3a). However, in the process of film formation, there are inevitably macro- or microdefects in the coating, and chemical or physical changes may occur to make the electrolyte penetrate the coating to metal surface, destroying the electrostatic interaction between the coating and the metal to weaken the adhesion of the coating and accelerate the corrosion of the metal, as shown in Figure 3b. Therefore, another way is internal interception to prevent further metal corrosion. The adhesion of the coating on the metal interface, especially the wet adhesion, can prevent further diffusion of corrosive ions, thus effectively protecting the metal (Figure 3c). The studies found that phosphate could be able to bind with metal substrate well and can also combine with carboxyl and hydroxyl groups in the coating to form a stable protective film between the coating and metal matrix to prevent the horizontal diffusion of corrosion medium and to slow down the corrosion and peeling of coating defects.14
Figure 3.
Schematic diagram of metal surface protection mechanism, external blocking (a), water entering (b), and internal interception (c).
2.2.3. Mechanical Property
The mechanical properties of the resin include rigidity, toughness and so on. The following methods can enhance the mechanical properties.
(1) Increasing the cross-linking density can enhance the mechanical properties of the resins. In the resin with a low cross-linking rate, the uncured functional group is helpful to absorb energy and can be used as a plasticizer to improve the fracture toughness and reduce the mechanical properties of the resin. On the contrary, the resin with a high cross-linking degree has strong mechanical properties. For example, Guo et al.15 used aminopropyltriethoxysilane (KH550) to cure the waterborne acrylic resin (the curing process of the resin is shown in Figure 4a) and found that glycidyl methacrylate (GMA) effectively increased the number of cross-link junctions between the resin and curing agent, enhanced the cross-linking density, and thus enhanced the tensile properties of the coatings (Figure 4b).
Figure 4.
Curing process of waterborne resin (a) and stress–strain curves of waterborne resin/GMA-KH550 (1:0.15) (b). Reproduced with permission from ref (15). Copyright 2018 Elsevier.
(2) Polymer–inorganic composite materials can improve the mechanical properties. In order to make the organic polymer and inorganic materials combine better, additives can be added, such as coalescing agent, silane coupling agent, plasticizer, etc. The additives mainly enhance the cross-linking effect, reduce the pore volume, refine the pore structure, reduce the porosity, and ensure the continuous and complete the membrane structure to improve the comprehensive mechanical properties of the composite. For example, the modified SiO2 nanoparticles can be used as the cross-linking sites of polymer chains, which reduces their mobility and leads to the formation of fixed and restricted migration areas around the nanoparticles, thus improving the tensile properties of the composite coating.16
(3) The ratio of hard and soft monomers affects the mechanical properties. For example, Drake et al.17 found that the tensile strength and elongation decrease with the increase of butyl acrylate. In addition, the hardness of the resin can be increased by properly increasing the content of hard monomer.
2.2.4. Water Resistance
Water resistance is also one of the important properties of waterborne resins. The water resistance of polymers mostly depends on their hydrophobic as well as the interactions between macromolecules. The hydrophobicity of the coating depends on the molecules with strong polarity and low surface energy in the resin and the roughness of the coating surface. For example, Yang et al.18 grafted silica onto fluorinated acrylic latex particles using silane coupling agent to connect nanomaterials and the resin matrix to improve the surface hydrophobicity (Figure 5a). Fluorine atoms have strong polarity and hydrophobicity, and silica can increase the roughness of the coating. At the same time, modified SiO2 could make the hydrophobic fluorinated chain exposed in the outer layer. Therefore, when the film is formed, more fluorinated chains gather on the surface of the film, making the contact angle of the coating increase from 83 to 110° (Figure 5b).
Figure 5.
Growth process of fluorinated polyacrylate/silica latex particle (a) and water contact angle of the films (b). Reproduced with permission from ref (18). Copyright 2015 Elsevier.
3. Application of Modified Waterborne Acrylic Resin
Waterborne acrylic resin has excellent comprehensive performance and environmental protection and is widely used in all aspects of people’s production and life, especially in the construction, automobile, metal anticorrosion, furniture, and plastic parts industries, etc. (Figure 6).
Figure 6.

Overview of the application of waterborne acrylic resin.
3.1. Application in the Construction Industry
Waterborne acrylic resin has the largest amount of categories in architectural coating because of its exquisite appearance, smoothness, full texture, good decoration, water washing, and dirt and scrub resistance. The biggest demand of outdoor architectural coating is to prevent aging. Aguirre et al.19 synthesized acrylic resin modified by nanomaterials; for example, CeO2 and ZnO can prevent photodegradation and aging. Meanwhile, the functional performance of waterborne acrylic resin in use is more and more prominent, such as acid rain protection on stone, waterproof and leakage protection in kitchens and bathrooms, scrub-resistant latex inner wall paint, weather resistant waterproofing, and antifouling for outer walls.20 Notably, the copolymer should have the ability to preserve either the vapor permeability or limited water absorbed by capillarity without occluding the marble pores, and it can also protect the surface of buildings from corrosion.
3.2. Application in the Automobile Industry
Since the specific environment of each part of the car is different, the car coating is also different. Modified waterborne acrylic resin can better meet the requirements of each part. There is often a temperature difference between the inside and outside of the car, which causes the water mist on the windshield and window glass to block vision. The surface tension of the acrylic coating is often reduced by the modification of organosilicon and organofluorine, so that the water vapor will not adhere and accumulate. Automobile shell coating must have good weather resistance and scratch resistance. High solid acrylic coating is suitable for the automobile shell, with high fullness, high gloss, good water resistance, and other excellent properties. Coatings for automobile interiors meet the requirements of heat resistance and water resistance. Automotive touch up paint is required to be fully and rapidly cured at ambient temperature to reduce capital investment and maintenance time of drying equipment.
3.3. Application in Metal Anticorrosion
A metal coating has a great effect on prolonging service life and decoration and can effectively avoid steel corrosion, copper oxidation, and other problems. Acrylic anticorrosive coating is widely used in steel structures, ships, metal plates, and other aspects. In the field of high anticorrosion requirements, the overall performance of waterborne acrylic resin is still not up to the level of solvent resin. However, in the field of industrial light anticorrosion, the single component waterborne acrylic resin can gradually reach the technical level of the same solvent-based resin on the performance, which has the characteristics of convenient construction and enters a rapid growth period. All of these emerge in an endless stream of research on emulsion polymerization. New emulsion polymerization technologies are emerging and maturing (such as group transfer polymerization, organic–inorganic composite emulsion polymerization, inverse emulsion polymerization, microemulsion polymerization, and so on), and the products that can be applied to metal fields emerge endlessly.
Organic fluorine modification, silicone modification, nanoparticle modification, alkyl resin modification, and other methods have been designed to enhance the corrosion resistance of waterborne acrylic resin and widened the application of waterborne acrylic resin in the field of metal corrosion protection.21 In addition, Li et al.22 prepared a new rust converter, which can be used in waterborne acrylic resin to prepare anticorrosive coatings. As one of the economic and effective methods, organosilicon modification has been widely used in the field of corrosion-resistant coatings. However, the cost of organic fluorine modification and phosphate functional monomer modification is high, and the process is complex, so the practical value of this research direction is not high. With the research and application of slippery liquid-infused porous surfaces, the anticorrosive coating modified by nanostructures is also gradually favored.23
3.4. Application in Wood Furniture
The application of waterborne acrylic resin in wood furniture has seen a rapid development. The sealing primer, transparent putty, primer, and finishing coat on wood furniture can all be used with acrylic resin. For example, Durmaz et al.24 found that waterborne acrylic coatings can improve the weatherability of wood–plastic composites (WPCs) and minimize the changes in color, mechanical strength, surface roughness, and surface degradation. As shown in Figure 7, acrylic coating protects the surface of the WPCs from weathering.
Figure 7.
Examination of WPCs exposed to accelerated weathering by light microscopy: (a) control samples, (b) formulation X (acrylic resin), and (c) formulation Y (acrylic emulsions). Reproduced with permission from ref (24). Copyright 2020 John Wiley and Sons.
Wood furniture cannot endure high temperature, and the coatings need to be cured by self-drying, low-temperature baking, or radiation curing. To solve this problem, one way is reducing the glass transition temperature of one component of the acrylic coating; for two-component waterborne acrylic resin, the appropriate curing agent can be selected to make the coating have the advantages of quick drying, good transparency, good light, and color retention. The other way is preparing self-cross-linking waterborne acrylic resin to achieve the aim of baking at low temperature or even curing by self-drying. The third way is using the waterborne polyurethane to modify waterborne acrylic resin. Waterborne polyurethane has excellent low-temperature film-forming and leveling properties. Using the complementary properties of waterborne polyurethane and waterborne acrylic resin, the two were organically combined to improve the comprehensive properties of the materials. In addition, the waterborne UV acrylic resin is a kind of resin containing a double bond or epoxy group and other active groups, which has a very good prospect. The initiator initiates the cross-linking with the condition of the light, giving the resin good performance, such as rapid curing, high content, no VOC, easy production, and high spraying efficiency. As the raw material of waterborne wood coatings, reducing the curing temperature and improving the hardness are still the key problems to be solved in the future.
3.5. Other Applications
There are also other applications such as leather processing, 3D printing, preparation of water-based ink, flame retardant materials, and so on. For example, Yong et al.25 reported a new type of film-forming acrylic resin. By controlling the morphology of the film at the submicron roughness level, it has low luster and high transparency. The polymer has good optical properties and thermal stability and is suitable for leather processing. At the same time, the rise of 3D materials makes the application of waterborne acrylic resin further expanded.
4. Conclusions and Prospects
With the increasingly strict environmental protection laws and regulations and the enhancement of consumers’ awareness of environmental protection, waterborne acrylic resin has become a product with rapid development and mature application in all waterborne coatings due to its excellent performance in recent years. In order to further improve the comprehensive properties of acrylic resin and broaden its application scope, various polymerization methods and advanced polymerization processes are adopted to modify acrylate to solve the actual problems, increase the development of waterborne acrylic resin products, and give it more excellent properties. In the future, waterborne acrylic resin still needs further development toward high performance, multifunction, and green environmental protection.
Biographies
Cuiyan Jiao received her Bachelor’s degree from Shandong University of Science and Technology in 2017 and is now studying for a Master’s degree at Shandong University of Science and Technology. Her research interest is in the development of waterborne acrylic resins, and she has published a highly cited SCI paper.
Li Sun received his Bachelor’s degree from Shandong University of Science and Technology in 2018 and is now studying for a Master’s degree at Shandong University of Science and Technology. His research interest is the photocatalytic degradation of dye liquor and the synthesis of waterborne resins. He has published one highly cited SCI paper.
Qian Shao, Professor of College of Chemical and Biological Engineering, Shandong University of Science and Technology. Her main research interest includes synthesis and applications of waterborne resins and their composites, synthesis and properties of photocatalysts, and their applications in degradation of organic pollutants.
Jiyong Song, associate professor of Shandong University of Science and Technology. His research interest is the synthesis and performance analysis of composite materials.
Qian Hu is now studying for a Ph.D. at Beijing Forestry University, University of Tennessee (Knoxville), and Zhengzhou University. His research interest is the energy recovery, wastewater treatment, and synthesis of waterborne resins and nanocomposites. He has published one highly cited SCI paper.
Nithesh Naik is faculty at Department of Mechanical and Manufacturing Engineering, Manipal Institute of Technology, Manipal Academy of Higher Education (Institute of Eminence) with 8+ years of academic experience. He has two years of industry experience in the field of planning and design of HVAC systems with ETA Engineering Private Limited, India. His research interest includes the development of polymers, green composites, finite element analysis, artificial intelligence, and design and product development techniques. He has applied for four patents at India’s patent office. Mr. Nithesh has published 50+ research publications in International Journals of repute and has keen interests in medical innovations. He received the award of South India’s most exciting young teacher. Nithesh has six patents applied at India’s patent office.
Zhanhu Guo earned his Ph.D. in chemical engineering from Louisiana State University in 2005. He received his three-year postdoctoral training in mechanical and aerospace engineering at the University of California Los Angeles. He is an Associate Professor in Chemical and Biomolecular Engineering and directs the Integrated Composites Laboratory at The University of Tennessee, Knoxville. His current research interests are in the areas of optoelectronic, electric, and magnetic materials for solar cells, catalysis, sensing, and electronics.
The authors declare no competing financial interest.
References
- Liu J.; Zhang J.; Tang J.; Pu L.; Xue Y.; Lu M.; Xu L.; Guo Z. Polydimethylsiloxane resin nanocomposite coating with alternating multilayer structure for corrosion protection performance. ES Mater. Manuf. 2020, 10, 29–38. 10.30919/esmm5f912. [DOI] [Google Scholar]
- Zhang D.; Williams B. L.; Santos V. H.; Lofink B. J.; Becher E. M.; Partyka A.; Peng X.; Sun L. Self-assembled intumescent flame retardant coatings: influence of pH on the flammability of cotton fabrics. Engi. Sci. 2020, 12, 106–112. 10.30919/es8d1134. [DOI] [Google Scholar]
- Li T.; Gao Y.; Zheng K.; Ma Y.; Ding D.; Zhang H. Achieving better greenhouse effect than glass: visibly transparent and low emissivity metal-polymer hybrid metamaterials. ES Energy Environ. 2019, 5, 102–107. 10.30919/esee8c325. [DOI] [Google Scholar]
- Chen M.; Zhang Y.; Zhang J.; Li K.; Lv T.; Shen K.; Zhu Z.; Liu Q. Facile lotus-leaf-templated synthesis and enhanced xylene gas sensing properties of Ag-LaFeO3 nanoparticles. J. Mater. Chem. C 2018, 6 (23), 6138–6145. 10.1039/C8TC01402G. [DOI] [Google Scholar]
- Karami S.; Motahari S.; Pishvaei M.; Eskandari N. Improvement of thermal properties of pigmented acrylic resin using silica aerogel. J. Appl. Polym. Sci. 2018, 135 (1), 45640. 10.1002/app.45640. [DOI] [Google Scholar]
- Du B.; Chen F.; Luo R.; Zhou S.; Wu Z. Synthesis and characterization of nano-TiO2/SiO2-acrylic composite resin. Adv. Mater. Sci. Eng. 2019, 2019, 6318623. 10.1155/2019/6318623. [DOI] [Google Scholar]
- Zhong S.; Li J.; Cai Y.; Yi L. Novel surfactant-free waterborne acrylic-silicone modified alkyd hybrid resin coatings containing nano-silica for the corrosion protection of carbon steel. Polym. Plast. Technol. Mater. 2019, 58 (8), 866–878. 10.1080/03602559.2018.1542711. [DOI] [Google Scholar]
- Xiong K.; Wang L.; Zhang A. The mechanical properties, compatibility, and thermal stabilities of POE-graft-methyl methacrylate and acrylonitrile(POE-g-MAN)/styrene-acrylonitrile copolymer (SAN resin) blends. J. Macromol. Sci., Part B: Phys. 2011, 50, 1350. 10.1080/00222348.2010.516178. [DOI] [Google Scholar]
- Yin Y.; Jiang B.; Zhu X.; Meng L.; Huang Y. Investigation of thermostability of modified graphene oxide/methylsilicone resin nanocomposites. Eng. Sci. 2018, 5, 73–78. 10.30919/es8d762. [DOI] [Google Scholar]
- Bai L.; Zheng S.; Bao R.; Liu Z.; Yang M.; Yang W. Effect of PLA crystallization on the thermal conductivity and breakdown strength of PLA/BN composites. ES Mater. Manuf. 2018, 3, 66–72. 10.30919/esmm5f195. [DOI] [Google Scholar]
- Yang K.; Chen X.; Zhang Z.; Yu X.; Naito K.; Zhang Q. Introducing rigid pyrimidine ring to improve the mechanical properties and thermal-oxidative stabilities of phthalonitrile resin. Polym. Adv. Technol. 2020, 31, 328–337. 10.1002/pat.4773. [DOI] [Google Scholar]
- Romo-Uribe A.; Santiago-Santiago K.; Zavala-Padilla G.; Reyes-Mayer A.; Calixto-Rodriguez M.; Arcos-Casarrubias J. A.; Baghdachi J. Waterborne layered silicate/acrylate nanocomposites by in-situ emulsion polymerization: Thermal and mechanical reinforcement. Prog. Org. Coat. 2016, 101, 59–70. 10.1016/j.porgcoat.2016.07.014. [DOI] [Google Scholar]
- Madhusudhana A. M.; Mohana K. N. S.; Hegde M. B.; Nayak S. R.; Rajitha K.; Swamy N. K. Functionalized graphene oxide-epoxy phenolic novolac nanocomposite: an efficient anticorrosion coating on mild steel in saline medium. Adv. Compos. Hybrid Mater. 2020, 3, 141–155. 10.1007/s42114-020-00142-8. [DOI] [Google Scholar]
- Zhong Z.; Yu Q.; Yao H.; Wu W.; Feng W.; Yu L.; Xu Z. Study of the styrene–acrylic emulsion modified by hydroxyl-phosphate ester and its stoving varnish. Prog. Org. Coat. 2013, 76 (5), 858–862. 10.1016/j.porgcoat.2013.02.008. [DOI] [Google Scholar]
- Guo X.; Ge S.; Wang J.; Zhang X.; Zhang T.; Lin J.; Zhao C. X.; Wang B.; Zhu G.; Guo Z. Waterborne acrylic resin modified with glycidyl methacrylate (GMA): Formula optimization and property analysis. Polymer 2018, 143, 155–163. 10.1016/j.polymer.2018.04.020. [DOI] [Google Scholar]
- Gharieh A.; Mirmohseni A.; Khorasani M. Preparation of UV-opaque, Vis-transparent acrylic–silica nanocomposite coating with promising physico-mechanical properties via miniemulsion polymerization. J. Coat. Technol. Res. 2019, 16 (3), 781–789. 10.1007/s11998-018-00155-5. [DOI] [Google Scholar]
- Drake I.; Cardoen G.; Hughes A.; Nakatani A. I.; Landes B.; Reffner J.; Even R. Polyurea-acrylic hybrid emulsions: Characterization and film properties. Polymer 2019, 181, 121761. 10.1016/j.polymer.2019.121761. [DOI] [Google Scholar]
- Yang W.; Zhu L.; Chen Y.; Ye H.; Ye Z. Surface topography and hydrophobicity of waterborne fluorinated acrylic/silica hybrid coatings. Colloids Surf., A 2015, 484, 62–69. 10.1016/j.colsurfa.2015.07.053. [DOI] [Google Scholar]
- Aguirre M.; Goikoetxea M.; Otero L. A.; Paulis M.; Leiza J. R. Accelerated ageing of hybrid acrylic waterborne coatings containing metal oxide nanoparticles: Effect on the microstructure. Surf. Coat. Technol. 2017, 321, 484–490. 10.1016/j.surfcoat.2017.05.013. [DOI] [Google Scholar]
- Khallaf M. K.; El-Midany A. A.; El-Mofty S. E. Influence of acrylic coatings on the interfacial, physical, and mechanical properties of stone-based monuments. Prog. Org. Coat. 2011, 72 (3), 592–598. 10.1016/j.porgcoat.2011.06.021. [DOI] [Google Scholar]
- Liu C.; Lin Y.; Dong Y.; Wu Y.; Bao Y.; Yan H.; Ma J. Fabrication and investigation on Ag nanowires/TiO2 nanosheets/graphene hybrid nanocomposite and its water treatment performance. Adv. Compos. Hybrid Mater. 2020, 3, 402–414. 10.1007/s42114-020-00164-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J.; Ge S.; Wang J.; Du H.; Song K.; Fei Z.; Shao Q.; Guo Z. Water-based rust converter and its polymer composites for surface anticorrosion. Colloids Surf., A 2018, 537, 334–342. 10.1016/j.colsurfa.2017.10.041. [DOI] [Google Scholar]
- Zhang M.; Dong M.; Chen S.; Guo Z. Slippery liquid-infused porous surface fabricated on aluminum maintains stable corrosion resistance at elevated temperatures. Eng. Sci. 2018, 3, 67–76. 10.30919/es8d732. [DOI] [Google Scholar]
- Durmaz S.; Özgenç Ö.; Avci E.; Hakki Boyaci İ. Weathering performance of waterborne acrylic coating systems on flat-pressed wood–plastic composites. J. Appl. Polym. Sci. 2020, 137 (14), 48518. 10.1002/app.48518. [DOI] [Google Scholar]
- Yong Q.; Liang C. Synthesis of an aqueous self-matting acrylic resin with low gloss and high transparency via controlling surface morphology. Polymers 2019, 11 (2), 322. 10.3390/polym11020322. [DOI] [PMC free article] [PubMed] [Google Scholar]