The case
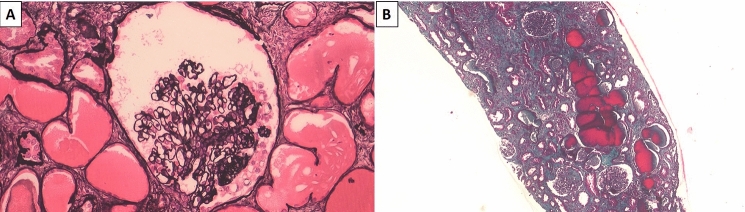
A 57-year-old patient of Congolese origin with a medical history of hypertension under triple therapy was referred to the emergency department due to significant deterioration in general condition, loss of 10 kg in one month, fever, anosmia and ageusia. He also reported a few episodes of non-bloody diarrhea and oliguria. Laboratory findings revealed severe acute kidney failure (serum creatinine at 30.3 mg/dL versus 0.8 mg/dL one month earlier, blood urea nitrogen at 536 mg/dL), hyperkaliemia at 7.4 mmol/L, and low bicarbonatemia at 13 mmol/L (Supplemental table). There was glomerular-range proteinuria with urinary protein-to-creatinine ratio (uPCR) at 1.2 g/g including 0.6 g/g of albuminuria and microscopic hematuria (41/mm3). Normocytic anemia was also noticed (10 g/dL), as was lymphopenia (300/mm3), marked inflammatory syndrome (C-reactive protein 2.5 mg/dL, fibrinogen 0.52 g/dL), and hypoalbuminemia (2.5 g/dL). Etiological assessment showed significant activation of the alternate complement pathway (low C3, normal C4), non-significant titer of anti-nuclear antibodies (1/80), and negative anti-neutrophil cytoplasmic, anti-glomerular basement membrane and anti-PLA2R antibodies. Biological findings also revealed polyclonal hypergammaglobulinemia, normal light chain ratio, and negative cryoglobulinemia. Testing for human immunodeficiency, hepatitis B/C viruses, and syphilis were negative, as were CMV, HHV6, HHV7 and Parvovirus B19 detection by PCR. Finally, nasal and tracheal PCR confirmed acute SARS-CoV-2 infection. The patient initially needed two dialysis sessions. He did not require any oxygen support nor specific treatment for COVID-19 infection. However, given the persisting kidney dysfunction one month later (serum creatinine 2.5 mg/dL and increased uPCR 4.9 g/g), a kidney biopsy was performed, revealing a collapsing variant of focal and segmental glomerulosclerosis (FSGS): histological examination was significant for diffuse hypertrophy and vacuolation of podocyte cytoplasm, diffuse interstitial lymphoplasmacytic infiltrate, and acute tubular necrosis associated with several microcystic tubular dilations (Fig. 1). There was no sign of cellular proliferation within the glomeruli. Immunofluorescence testing revealed mild peripheral and polytypic IgM and C3 deposits within the glomeruli. Electron microscopy did not reveal any foot process effacement, immune deposition, or endothelial tubulo-reticular inclusion. In this context, APOL1 genotyping was performed on a peripheral blood sample with respect to G0 wild type, G1 (rs73885319 and rs60910145) and G2 (rs71785313) SNPs by direct Sanger sequencing. This analysis revealed the rare APOL1 genotype G1GM/G1G+. Four months after admission, renal outcome was globally favorable with serum creatinine stabilization at 1.6 mg/dL and sequelae proteinuria at 1.6 g/g under ACE inhibitors. On a physical level, the patient fully recovered.
Fig. 1.
Kidney biopsy findings. a Jones Methenamine Silver, magnification × 200. Glomerulus presenting a typical lesion of collapsing variant of focal and segmental glomerulosclerosis. Collapsed capillary loops with overlying podocyte hypertrophy and hyperplasia; b Masson’s Trichrome, magnification × 50. Moderate lesions of tubular atrophy and interstitial fibrosis. Microcystic dilations of renal tubules that are reminiscent of HIVAN lesions. Some lymphocytes may also be noticed on the background
Lessons for the clinical nephrologist
We report a case of severe acute renal failure complicating SARS-CoV-2 infection and revealing a collapsing variant of FSGS. Genetic analysis showed the presence of a very rare APOL1 high risk genotype G1GM/G1G+, which was, for the first time to the best of our knowledge, associated with COVID-19-related nephropathy. Our case thus provides further arguments for the pathophysiological hypothesis of a double-hit mechanism and represents an opportunity to summarize the current evidence on this emerging cause of glomerulopathy.
What about ‘COVID-19 associated nephropathy’ (COVAN)?
Since the start of the COVID-19 pandemic, many teams have reported a renal tropism of the infection. A prospective Chinese cohort of 707 hospitalized patients revealed that proteinuria, hematuria, and acute renal failure were present in 44, 27 and 14% of cases, respectively [1]. Renal involvement was also recognized as a poor prognostic factor [1]. Most of the histological lesions corresponded to acute tubular necrosis [2]. However, a few authors have also reported glomerular involvement, in the form of collapsing glomerulopathy [3]. The similarities between these lesions and those observed in HIV-associated nephropathy (HIVAN) have led to the proposal of the name ‘COVAN’ for ‘COVID-19 associated nephropathy’ [4]. Most of the patients presented with severe acute kidney failure, leading in many cases to dialysis, with frequent failure to recover kidney function [5]. Our patient had the particularity of a favorable outcome, with the sole need for two initial dialysis sessions, followed by a gradual decrease in serum creatinine (up to 1.6 mg/dL). On a histological level, in addition to collapsing FSGS, he exhibited very severe tubular involvement, with major microcystic tubular dilations. These lesions, described in other collapsing glomerulopathies, have also been reported in previous cases of COVAN [6]. Electron microscopy did not reveal any tubulo-reticular inclusion within endothelial cells (also called ‘interferon footprint’) [7]. Taken together, the predominance of tubular involvement and the absence of interferon footprint lesions could explain the spontaneous, favorable renal outcome. Interestingly, the cases previously described in the literature often reported kidney impairment alone (only 12/34 patients requiring oxygen-support and 3/34 hospitalized in intensive care), despite the presence of major comorbidities (30/34 with diabetes, hypertension, CKD and/or obesity) that should have put them at higher risk of severe disease course (Table 1). Like the previous case reports, our patient also experienced isolated severe kidney impairment.
Table 1.
Main characteristics of the previously described cases of COVAN (literature review)
| Author | Age | Sex | Ethnicity | APOL1 | Kidney | Comorbidities | AKI | sCr (mg/dL) | uPCR (g/g) | sAlb (g/L) | O2-therapy | ICU | RRT | LFU sCr (mg/dL) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gaillard et al. [10] | 79 | M | African | NA | NK | HT, CKD | Yes | 2.5 | 11.4 | 29 | NA | No | Yes | RRT |
| Kissling et al. [3] | 63 | M | African | G1/G1 | NK | HT | Yes | 8.4 | 5 | NA | Yes | No | No | 5,5 |
| Larsen et al. [7] | 44 | F | African | G1/G1 | NK | HT, DM, CKD | Yes | 4 | 25 | 25 | Yes | No | Yes | RRT |
| Peleg et al. [11] | 46 | M | African | G1/G1 | NK | Ob | Yes | 12.5 | 5.8 | 21 | No | No | Yes | RRT |
| Nlandu et al. [12] | 48 | M | African | NA | NK | HT, DM | Yes | 15.9 | 18 | 23 | No | No | Yes | 5 |
| Noble et al. [13] | 54 | M | African | NA | NK | HT, Ob, CKD | Yes | 13.6 | 3.59 | NA | No | No | Yes | 8,1 |
| 45 | M | Donor: NA | NA | KT | HT, DM, Ob, CKD | Yes | 14.1 | NA | NA | Yes | No | Yes | RRT | |
| Malhotra et al. [14] | 64 | M | African | G0/G1 | NK | HT, DM, Ob, CKD, treated HIV | Yes | 2.3 | 2.7 | NA | Yes | Yes | Yes | RRT |
| Wu et al. [6] | 63 | M | African | G1/G1 | NK | HT: n = 4 DM: n = 3 CKD: n = 3 | Yes | 4.9 | 12.7 | 21 | 2 patients in ICU for ARDS | Yes | RRT | |
| 64 | F | African | G2/G2 | NK | Yes | 4.2 | 4,6 | 24 | No | 3,1 | ||||
| 65 | F | African | G1/G1 | NK | Yes | 2.9 | 13.6 | 26 | Yes | Died | ||||
| 44 | M | African | G1/G1 | NK | Yes | 11.4 | 25 | 25 | Yes | RRT | ||||
| 37 | M | African | G1/G2 | NK | Yes | 9 | NA | 30 | Yes | Died | ||||
| 56 | M | African | G1/G1 | NK | Yes | 6.7 | 3.6 | 29 | Yes | 2,8 | ||||
| Sharma et al. [15] | 77 | F | African | NA | NK | HT | Yes | 8.3 | 1.5 | NA | Yes | No | Yes | 3 |
| Kudose et al. [16] | 46 | M | African | G1/G1: n = 2 G1/G2: n = 1 NA: n = 3 | NK | Ob, OSA, CKD | Yes | 12.5 | 5.8 | 31 | No | No | Yes | RRT |
| 62 | M | African | NK | HT, CKD | Yes | 10.7 | 12.1 | 31 | No | No | No | 3,8 | ||
| 62 | M | African | NK | HT, DM, CKD | Yes | 11.6 | 19.2 | 24 | Yes | No | No | 2,3 | ||
| 57 | M | African | NK | HT, HCV, CKD | Yes | 4.9 | 6.2 | 25 | Yes | No | No | 4,9 | ||
| 61 | M | African | NK | HT, Ob | Yes | 14 | 9 | 25 | No | No | Yes | RRT | ||
| Gupta et al. [17] | 71 | M | Asian Indian | NA | NK | HT, DM, CKD | Yes | 4.5 | 18.4 | 20 | No | No | Yes | RRT |
| 54 | M | African | NA | NK | HT, DM, CKD | Yes | 4.7 | 16 | 16 | No | No | No | 4,5 | |
| Akilesh et al. [5] | 46 | M | African | G1/G1 | NK | HT | Yes | 8.7 | 13.7 | NA | No | NA | Yes | RRT |
| 60 | F | African | NA | NK | HT | Yes | 5.7 | 21 | NA | No | NA | NA | NA | |
| 58 | F | African | NA | NK | HT | Yes | 10.2 | 20 | NA | No | NA | Yes | RRT | |
| 44 | M | Hispanic | G2/G2 | NK | HT | Yes | 12 | 11.4 | NA | No | NA | Yes | RRT | |
| 58 | M | African | NA | NK | None | Yes | 11.3 | 4 | NA | Yes | NA | Yes | RRT | |
| 47 | M | African | NA | NK | None | Yes | 6.6 | 7.6 | NA | No | NA | Yes | RRT | |
| 63 | F | African | NA | NK | HT | Yes | 6 | 20 | NA | No | NA | Yes | RRT | |
| Deshmukh et al. [18] | 42 | M | Asian Indian | NA | NK | None | No | 1 | 8 | 19 | No | No | No | 1 |
| Lazareth et al. [19] | 29 | M | Donor: NA | Donor: G0/G2 | KT | HT, CKD | Yes | 6.1 | 8 | 28 | No | No | No | 3,7 |
| Kadosh et al. [20] | 56 | M | African | NA | NK | Heart Tx, CKD | Yes | 7.8 | 7.3 | NA | No | No | No | NA |
| Magoon et al. [21] | 28 | F | African | G1/G1 | NK | None | Yes | 6.6 | 2 | 16 | Yes | No | Yes | NA |
| 56 | M | African | G1/G2 | NK | HT, CKD | Yes | 7.7 | 21 | 8 | Yes | No | Yes | NA | |
| Summary | 54 | ≈ 3H/1F | Afr: 29/34 |
HMZ/HTZ comp: 16/18; HTZ: 2/18; WT: 0/18 |
32 NK/ 2 KT | HT, DM, Ob or CKD: 30/34 | 33/34 | 8.0 mg/dL | 11.3 g/g | 23.6 g/L | 12/34 | 3/34 | 24/34 |
RRT: 16/34, Death: 2/34, sCr: 3.5 mg/dL |
Afr African ethnicity, AKI acute kidney injury, ARDS acute respiratory distress syndrome, CKD chronic kidney disease, DM diabetes mellitus, F female, HCV hepatitis C virus, HIV human immunodeficiency virus, HMZ Homozygous, HTZ Heterozygous, HT hypertension, ICU intensive care unit, KT kidney transplant, LFU last follow-up, M male, NA non acquired data, NK native kidney, Ob obesity, OSA obstructive sleep apnea, RRT renal replacement therapy, sAlb serum albumin, sCr serum creatinine, uPCR urinary protein-to-creatinine ratio, WT Wild type
Why should we care about APOL1 genotype when facing COVAN?
To date, there are less than 40 COVAN cases described in the literature, mainly in men between 30 and 80 years old, and almost exclusively of African ethnicity (Table 1). Among all the case reports, more than half were tested for APOL1: almost all were of African origin (29/34) and all of them were positive for APOL1 risk variant [6] (16/18 homozygous or compound heterozygous, 2/18 heterozygous) (Table 1). In fact, G1 and G2 variants confer to the heterozygous carriers a selective advantage against African trypanosomiasis, at the cost of an increased risk of developing FSGS (17-fold higher risk) or HIVAN (29-fold higher risk) for the homozygous or compound heterozygous patients [8]. These variants were indeed highly prevalent (23 and 13% respectively) in an African American population with confirmed FSGS [8]. In a recessive experimental model, APOL1 risk variants act as a ‘first hit’, which in the context of type 1 interferon release (‘second hit’), leads to podocyte distress and collapsing glomerular lesions [9]. HIV, lupus erythematosus and interferon-based therapy were the main recognized ‘second hit’ triggers, before SARS-CoV-2 infection was recently added to the list.
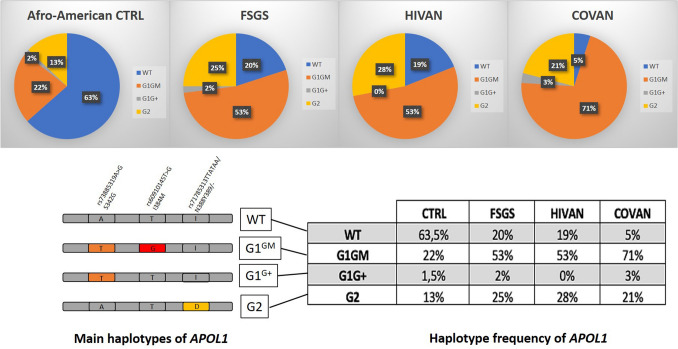
In our report, the patient was of African origin, but presented a particularly rare APOL1 genotype (G1GM/G1G+), which associated one common risk haplotype called G1GM (comprising two missense variants, rs73885319 -S342G- and rs60910145 -I384M-) and the G1G+ very rare haplotype (comprising the rs73885319 -S342G- variant only) [8]. Kopp et al. showed that among 271 African American patients with FSGS or HIVAN, the G1GM/G1G+ genotype was very rare (7/271 patients, 2.5%), but seemed to have the same impact on the development of glomerulopathy [8] (Fig. 2). To our knowledge, this is the first description of COVAN associated with this rare APOL1 risk variant. This G1GM/G1G+ genotype could potentially explain the unusual, favorable clinical course of our patient compared to the other patients described, most of whom remained dialysis-dependent (16/34; mean serum creatinine for those withdrawing dialysis: 3.5 mg/dL) (Table 1).
Fig. 2.
Frequency of APOL1 risk haplotypes in an African American population, according to their clinical condition: healthy donors, FSGS, HIVAN, and COVAN. Schematic representation of four main APOL1 haplotypes: Wild type (G0), G1GM (with 2 missense risk alleles), G1G+ (with 1 missense risk allele) and G2D6 (with 6-bp deletion risk allele). APOL1 haplotype frequency in African American control population, FSGS, HIVAN and COVAN. Data regarding FSGS, HIVAN and healthy donors are extracted from the Kopp et al. study [8]. COVAN data correspond to the literature review including our case (Table 1). COVAN COVID-19-associated nephropathy, FSGS Focal Segmental Glomerulosclerosis, HIVAN HIV-associated nephropathy, WT Wild type
Which lessons could be drawn from this case report?
Although most kidney lesions related to COVID-19 infection are due to acute tubular necrosis, kidney biopsy remains essential. As illustrated by our report, some cases of collapsing FSGS called ‘COVAN’ have been described, frequently with poor renal prognosis but also mild clinical form. The association of COVID-19 and collapsing glomerulopathy is even more frequent in patients of African origin, in whom the prevalence of risk variants of APOL1 is higher. Our report thus highlights the contribution of genetic analysis in the context of COVAN, (and the need of not simply reporting the most frequent risk variants). The genetic results may thus have a prognostic implication for the patients, (as well as a prevention interest for their relatives). Unlike most of the previously described cases of COVAN, our patient presented an overall favorable outcome, potentially explained by the presence of a very rare risk variant of APOL1.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgments
The authors would like to thank Dr F. Moulonguet, Dr C. Gauvain, Dr M. Maanaoui, Dr R. Lenain and Dr F. Provôt for proofreading and for their insightful advice on this work.
Author contributions
TL, NP, AH, FG Conceptualization. TL, NP, JBG Data acquisition. TL, AH Manuscript drafting. TL, NP, JBG, FG, AH Critical revision. All the authors Manuscript approval.
Compliance with ethical standards
Conflict of interest
The authors declare that the patient provided consent for the report of his medical data in this manuscript.
Ethical approval
This case was reported under approval of the hospital ethics committee.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
4/3/2021
A Correction to this paper has been published: 10.1007/s40620-021-01037-7
References
- 1.Cheng Y, Luo R, Wang K, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97:829–839. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Su H, Yang M, Wan C, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98(1):219–227. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kissling S, Rotman S, Gerber C, et al. Collapsing glomerulopathy in a COVID-19 patient. Kidney Int. 2020;98:228–239. doi: 10.1016/j.kint.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Velez JCQ, Caza T, Larsen CP. COVAN is the new HIVAN: the re-emergence of collapsing glomerulopathy with COVID-19. Nat Rev Nephrol. 2020;16:565–567. doi: 10.1038/s41581-020-0332-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Akilesh S, Nast CC, Yamashita M et al (2020) Multicenter clinicopathology correlation of kidney biopsies performed in COVID-19 patients presenting with acute kidney injury of proteinuria. Am J Kidney Dis. S0272-6386(20)31014–3 [DOI] [PMC free article] [PubMed]
- 6.Wu H, Larsen CP, Hernandez-Arroyo CF, et al. AKI and collapsing glomerulopathy associated with COVID-19 and APOL1 high risk genotype. J Am Soc Nephrol. 2020;31(8):1688–1695. doi: 10.1681/ASN.2020050558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Larsen CP, Bourne TD, Wilson JD, et al. Collapsing glomerulopathy in patient with COVID-19. Kidney Int Rep. 2020;5(6):935–939. doi: 10.1016/j.ekir.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kopp JB, Nelson GW, Sampath K, et al. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol. 2011;2011(22):2129–2137. doi: 10.1681/ASN.2011040388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Freedman BI, Limou S, Ma L, et al. APOL1-associated-nephropathy: a key contributor to racial disparities in CKD. Am J Kidney Dis. 2018;72(5):S8–S16. doi: 10.1053/j.ajkd.2018.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gaillard F, Ismael S, Sannier A, et al. Tubuloreticular inclusions in COVID-19-related collapsing glomerulopathy. Kidney Int. 2020;98(1):241. doi: 10.1016/j.kint.2020.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Peleg Y, Kudose S, d’Agati V, et al. Acute kidney injury due to collapsing glomerulopathy following COVID-19 infection. Kidney Int Rep. 2020;5(6):940–945. doi: 10.1016/j.ekir.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nlandu YM, Makulo JRR, Pakasa NM, et al. First case of COVID-19-associated collapsing glomerulopathy in sub-Saharan Africa. Case Rep Nephrol. 2020;2020:1–5. doi: 10.1155/2020/8820713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Noble R, Tan MY, McCulloch T, et al. Collapsing glomerulopathy affecting native and transplant kidney in individuals with COVID-19. Nephron. 2020;144(11):589–594. doi: 10.1159/000509938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Malhotra V, Magoon S, Troyer DA, et al. Collapsing focal segmental glomerulosclerosis and acute oxalate nephropathy in a patient with COVID-19: a double whammy. J Investig Med High Impact Case Rep. 2020;8:2324709620963635. doi: 10.1177/2324709620963635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sharma P, Uppal NN, Wanchoo R, et al. COVID-19-associated kidney injury: a case series of kidney biopsy findings. J Am Soc Nephrol. 2020;31(9):1948–1958. doi: 10.1681/ASN.2020050699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kudose S, Batal I, Santoriello D, et al. Kidney biopsy findings in patients with COVID-19. J Am Soc Nephrol. 2020;31(9):1959–1968. doi: 10.1681/ASN.2020060802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gupta RK, Bhargava R, Shaukat AA, et al. Spectrum of podocytopathies in new-onset nephrotic syndrome following COVID-19 disease: a report of 2 cases. BMC Nephrol. 2020;21(1):326. doi: 10.1186/s12882-020-01970-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Deshmukh S, Zhou XJ, Hiser W. Collapsing glomerulopathy in a patient of Indian descent in the setting of COVID-19. Ren Fail. 2020;42(1):877–880. doi: 10.1080/0886022X.2020.1811122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lazareth H, Péré H, Binois Y, et al. COVID-19-related collapsing glomerulopathy in a kidney transplant recipient. Am J Kidney Dis. 2020;76(4):590–594. doi: 10.1053/j.ajkd.2020.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kadosh BS, Pavone J, Wu M, et al. Collapsing glomerulopathy associated with COVID-19 infection in a heart transplant recipient. J Heart Lung Transplant. 2020;39(8):855–857. doi: 10.1016/j.healun.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Magoon S, Bichu P, Malhotra V, et al. COVID-19-related glomerulopathy: a report of 2 cases of collapsing focal segmental glomerulosclerosis. Kidney Med. 2020;2(4):488–492. doi: 10.1016/j.xkme.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.