Abstract
Abstract
Skin is the largest organ in the body, and directly contact with the external environment. Articles on the role of micro-current and skin have emerged in recent years. The function of micro-current is various, including introducing various drugs into the skin locally or throughout the body, stimulating skin wounds healing through various currents, suppressing pain caused by various diseases, and promoting blood circulation for postoperative muscle rehabilitation, etc. This article reviews these efforts. Compared with various physical and chemical medical therapies, micro-current stimulation provides a relatively safe, non-invasive therapy with few side effects, giving modern medicine a more suitable treatment option. At the same time, the cost of the electrical stimulation generating device is relatively low, which makes it have wider space to and more clinical application value. The current micro-current stimulation technology has become more and more mature, but there are still many problems in its research. The design of the experiment and the selection of the current parameters not standardized and rigorous. Now, clear regulations are needed to regulate this field. Micro-current skin therapy has become a robust, reliable, and well-structured system
Graphic Abstract
Keywords: Transdermal, Drug delivery, Transdermal drug delivery system, Wound healing, Electrical stimulation
Introduction
As the largest organ, the skin protects the human body and can receive external stimuli to provide feelings. In recent decades, there have been more and more research on skin irritation related to micro-current, which can be divided into the following four aspects, which is iontophoresis and electroporation for transdermal drug delivery, the promotion of skin wounds and chronic wound healing, transcutaneous electrical nerve stimulation (TENS) therapy for analgesia, and electrical muscle stimulation (EMS) therapy for postoperative rehabilitation and expanded applications.
In iontophoresis, direct current therapy is based on a reliable theory of electrical repulsion, and pulsed direct current has different waveform and duty cycles [1–3]. The delivery experiments carried out by researchers on various drugs and cosmetics have been very effective [4–6]. Electroporation therapy uses extremely high pulse voltage to introduce drugs into the skin. It has been widely used but takes certain risks. Its advantage is that it can transmit polymers [7–9]. Another application of electric current is to promote wound healing. When an external electric field acts on the human body's intrinsic electric field, it will affect the movement of cells involved in wound healing, thereby promoting the healing process, and the effect is very significant [10]. The research on chronic wounds is still lacking. As a safe non-invasive treatment, TENS therapy is divided into low-frequency TENS and high-frequency TENS clinically. It has a very obvious effect in inhibiting severe pain caused by many diseases. The principle of analgesia is to promote the secretion of analgesic substances and stimulate the sensory nerve in the body [11]. At present, TENS research still needs to standardize the experimental process and improve the system of pain evaluation. EMS was originally used for postoperative rehabilitation, and it has sufficient animal and clinical trial data to support it. Although there are still side effects during long-term treatment, it is far smaller than the previously mentioned current therapy [12]. In recent years, it has a wealth of applications in the game field, such as cooperating with virtual reality (VR) to create better game experience, improving sensory feedback mechanisms, and as an auxiliary device for sports [13].
This article will meticulously analyze the development and current status of various currents, as well as the future development direction. Compared to various physical and chemical medical therapies, micro-current stimulation provides a relatively safe, non-invasive therapy with few side effects. In the future, the skin's current stimulation will be more attractive, as a mature household device used by people to help people improve and enrich their lives.
Skin structure and stimulate function
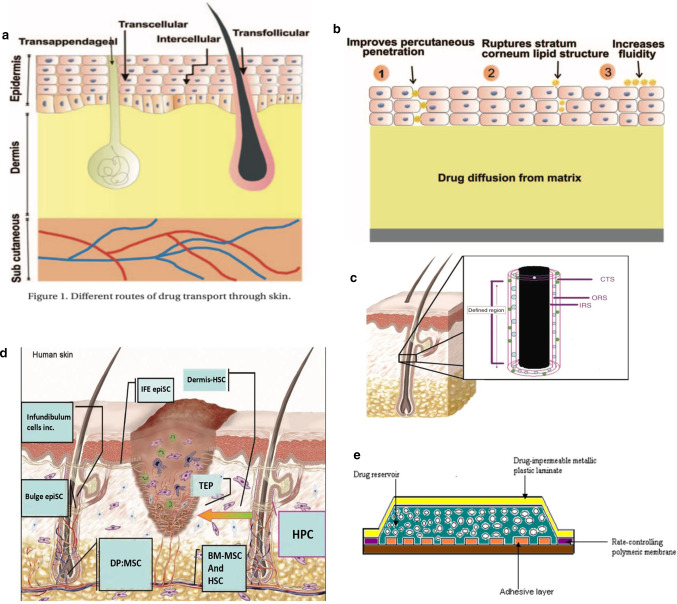
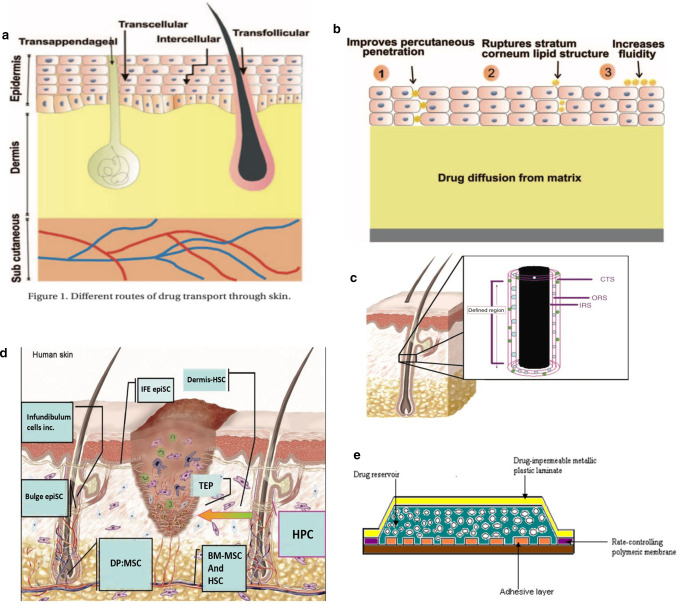
Skin is the largest organ in the body. It has a series of important functions such as protecting, regulating body temperature, excreting waste, and feeling stimuli. Anatomically, the skin is composed of three layers, namely the uppermost epidermal layer, the middle dermal layer, and the subcutaneous tissue, each of which functions differently [14]. The schematic diagram is shown in Fig. 1a [15].
Fig. 1.
a Structure of skin [15]. b Structure of the hair follicle: 1 – infundibulum; 2 – isthmus [16]. c Cross section of the hair follicle: Lanugo hairs are non- medullated and nonpigmented, present during intrauterine life [16]. d Protein levels of cytokines in BM-MSC-conditioned medium. (A) Antibody-based protein array analysis human dermal fibroblast (FB)- or BM-MSC-conditioned medium under hypoxic conditions. Similar results were obtained from three independent experiments and results from one of them are shown [18]. e Effect of BM-MSC-conditioned medium on wound closure. Representative images of wounds before treatment or 7 days after treatment with vehicle control medium (vehicle-M) (left), concentrated fibroblast (FB-M)- or BM-MSC-conditioned medium (MSC-M) (right) [18]
In recent decades, the formulations of transdermal drug patches have hardly increased, because the basic function of the skin is to protect and control the passage of substances, which hinders the delivery of drugs to the skin. Only a few drugs are suitable for molecular weight and transdermal absorption requirements, such as nitroglycerin for angina pectoris, clonidine for hypertension, and estradiol for estrogen deficiency. The transdermal drug delivery system (TDDs) may be the best new drug delivery system.
Skin structure
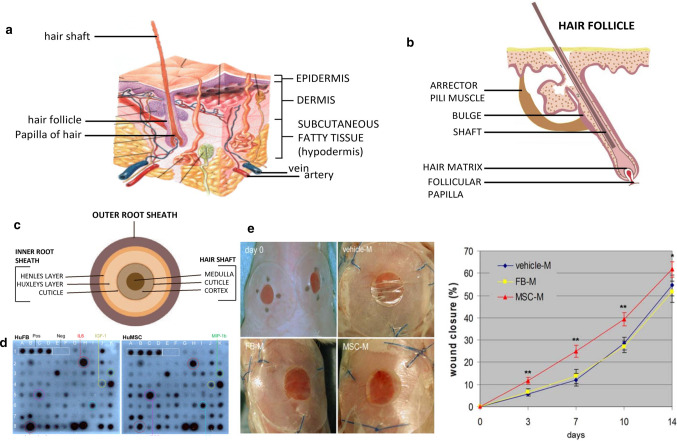
The epidermis is the superficial structure of the skin. The thickness of the epidermis is inconsistent throughout the body (0.06–0.8 mm), and plays an important barrier role. The outermost layer of the epidermal layer is the stratum corneum, and below it is the transparent layer, the granular layer, the spinous layer, and the base layer. The cell division at the base layer is active, and the continuously generated cells move upward from the granular layer to the stratum corneum, supplementing the aging and detached stratum corneum cells, undergoing keratinization and turning into dead cells without nucleus Lipid-containing adhesives stay together. On the other hand, as epidermal appendages, hair follicles are distributed on the entire body surface except the palms and sole. There are two types of hair, which is vellus hair and terminal hair. The terminal hair is thick, such as the hair of the scalp, eyebrows, eyelashes, and armpits, as shown in Fig. 1b, c, which is the structure of hair [16]. Each hair follicle consists of a hair shaft and a bulb. The ducts of the sebaceous and apocrine glands enter the hair follicle stem. Hair bulbs are composed of cells responsible for cell division, similar to the basal cells of the epidermis; then the cells differentiate to form an axis. The shaft is made of hard keratin. The mitotic rate of the hair matrix is greater than the mitotic rate of any other organ in the body. Hair growth is affected by any stress and disease that may alter mitotic activity. The effect of transdermal administration on vellus hair is negligible, but for terminal hair, the effect of mitosis must be considered. Although the blood vessels in the epidermal layer are very close to the skin surface, experiments show that the blood supply will not affect the epidermal penetration. The subcutaneous tissue is the hypodermis, which mainly supports the dermis and epidermis. The fat stored in it can regulate temperature and sense pressure, while protecting blood vessels and nerves.
Cells involved in wound healing
The skin contains numerous cells and tissues. Compared with the physical process of ion introduction, skin wound healing is spontaneous. Wound healing involves many types of cells, and it is a multi-party coordination and coordination process. When some of these factors are problematic, it can cause abnormal scars and chronic wounds. Adult stem cells (ASC) play a huge role in wound healing. Corneal epithelium healing and nerve regeneration involved in the repair process [17]. It is also a source of supplementary cells in wound healing. At the same time, tissue-specific ASCs also regulate other cells, such as inflammatory cells and tissue grandmother cells, to cooperate. Bone marrow derived mesenchymal stem cells (BM-MSC) have been shown to promote wound healing. Chen et al. [18] examined the paracrine factors released by BM-MSC and their effects on cells involved in wound healing, the antibody-based protein array analysis of fibroblasts or MSC-conditioned medium was performed under hypoxic conditions and normal oxygen conditions. As shown in Fig. 1d, fifteen cytokines with differential expression levels were found in 88 cytokines. Compared with normoxic conditions, hypoxic treatment significantly increases the release of several cytokines released by BM-MSC. The chemical attraction of MSC-conditioned medium and the promotion of mitotic wound healing requires the recruitment of cells from the surrounding tissues and blood into the wound. And then multiply. Wounds treated with vehicle medium or fibroblast conditioned medium were used as controls. Careful measurement of wounds on days 3, 7, 10, and 14 showed that MSC conditioned medium significantly accelerated wound closure compared to vehicle control medium or fibroblast conditioned medium, as shown in the Fig. 1e left, measurement of wound sizes is shown in the right of Fig. 1e [18, 19]. BM-MSCs releases high levels of cytokines and chemokines, which can enhance normal wound healing.
As key cells for wound healing, stem cells have different participants at different stages. Early injury is caused by inflammatory cell precursors from bone marrow and hair follicles, dermis and other locations, such as haematopoietic stem cells (HSC) from bone marrow (BM). Experiments in rats have shown that mast cell precursors in early wounds appear to be derived from BM and have been determined to respond to stem cell factor (SCF) [20]. These skin mast cell precursors and other haematopoietic progenitor cells (HPC) populations deposited in the dermis, then will participate in skin wound healing by supplying tissue macrophages and mature mast cells and dendritic cells Fig. 2d [21]. After this healing process is the re-epithelialization process, which also has multiple participants, of which keratinocytes come from two populations of skin epithelial stem cells (epiSC), which epiSC located in the base layer of interfollicular epidermis (IFE), hair follicle (HF) bulge epiSC in outer root sheath (ORS) Fig. 2c, the healing of the skin is not simply cell growth, but a variety of cells in the vicinity of the skin performing their duties, interconnected and coordinated work, and the regulation of this work is greatly affected by the electric field, which lays the foundation for the current treatment of skin wound foundation [21].
Fig. 2.
a Different routes of drug transport through skin [22]. b Percutaneous penetration of lipid vesicular carrier [22]. c Bulge region of a human hair follicle [21]. d Stem/progenitor cell populations that contribute to cutaneous wound healing [21]. e Cross-sectional view of polymer membrane permeation-controlled TDD systems [15]
Electrical stimulation on skin
The use of electric current to stimulate the skin is a large field of treatment in the medical field, and the results are quite fruitful, but there is not a clear enough distinction between the types and benefits of different micro-currents. Compared the benefits of current stimulation on skin, it can be divided into the following four aspects.
The research on transdermal current stimulation is the most extensive and thorough. The main therapies are iontophoresis and electroporation, and iontophoresis is common; Promotion of skin wound healing, which benefit has received increasing attention in recent years, and has gradually expanded to other fields, achieving attractive results; Transcutaneous electrical nerve stimulation for analgesia, with TENS percutaneous neurotherapy which is more often used as an adjuvant therapy and is combined with other therapies; The postoperative muscle recovery, using EMS electrical stimulation therapy, which is widely used in the postoperative recovery.
TDDs of micro-current stimulation
TDDs can be administered systemically or locally, which means the drug has penetrated through three layers enters the systemic circulation, or is applied topically only through the stratum corneum. Transdermal administration by passes liver metabolism, avoids the "peak and valley" effect of injection therapy.
There are four ways of transdermal administration, namely the transappendageal, transfolicular, transcellular, and intercellular routes. The different routes are shown in Fig. 2a [22]. Transcellular rout: Direct access to the dermis through keratinocytes and lipid bilayers, which includes active transport of ions, polar compounds, and endocytosis of macromolecules, which is the transport of molecules across epithelial cell membranes. Intercellular route: It is also called the paracellular route, which is a common way of drug molecular penetration. Cells are tightly connected by junctional adhesion molecules (JAMs). Closing proteins control the functioning of the barrier and select substances to transport between the cells. At this time, the drug stays in the lipid and is located around the keratin, so it is generally more suitable for the penetration of fat-soluble drugs. Transappendageal and transfolicular route: it is also called an appendage route, which includes sweat glands, hair follicles, and related sebaceous gland transportation. Although it avoids the stratum corneum, it only accounts for about 0.1% of the total skin area. The effect is secondary and is usually not considered in drug transport.
The path taken by drug penetration is mainly determined by the partition coefficient. The lipophilic molecules pass through the stratum corneum through a paracellular route. The hydrophilic molecules usually through transcellular route, but in fact the paracellular route is very tortuous, which greatly hinders the penetration of most drugs. The lipid vesicular system is another type of drug delivery method that can improve the bioavailability of encapsulated drug and extend the treatment time in a controlled manner. The transdermal penetration of lipid vesicle carriers is composed of physiological lipids. Vesicle carriers also destroy the lipid structure of stratum corneum, resulting in increased fluidity, thereby effectively transporting the carrier and drugs across the skin, increasing the transdermal permeation, as shown in Fig. 2b [22]. The penetration ability of drug nanocarriers (NCs) in biohydrogels will affect the efficiency of drug delivery [23]. It is worth mentioning that of the two main modes of transport, the transcellular route is relatively safe and easy to control. In addition to the nature of the molecule, the external environment can also affect transdermal administration. The stratum corneum of the skin is a great obstacle to transdermal administration, and it is composed of dead cells making it extremely resistant. Japanese scholars have studied the electrical properties of the stratum corneum skin, using cellophane to peel the stratum corneum layer by layer. With the peeling of the skin, the resistance decreases rapidly. When the number of strips reached 15 times, the change rate becomes constant, and the resistivity is approximately equal to the resistance of the subcutaneous tissue. Rate, that is, the part below the stratum corneum does not become a resistive layer [1].
At present, the medical community has used various methods, such as chemical accelerators, physical aids and delivery vehicles, to enhance the transdermal delivery of biologically active substances. The transdermal drug delivery (TDD) systems for polymer membrane penetration control, as shown in Fig. 2e, is a physical assistance method [15]. Allow the drug to penetrate through the rate control membrane. Electroosmosis therapy, as one of the physical auxiliary therapies, can reduce changes in drug plasma levels, and the effect is satisfied.
Iontophoresis
Iontophoresis is classified according to the flow of current. There are continuous direct current (CDC) and pulse depolarization iontophoresis (PDP). CDC is the earliest and common, but it will cause skin polarization and adverse irritation. PDP is a pulsed current that can alleviate the adverse effects and called depolarizing currents. Some literatures mention the use of alternating current (AC) for testing, which actually belong to pulsed direct current with direction change. In order to prevent confusion with the standard AC below, it is called a bidirectional pulse current.
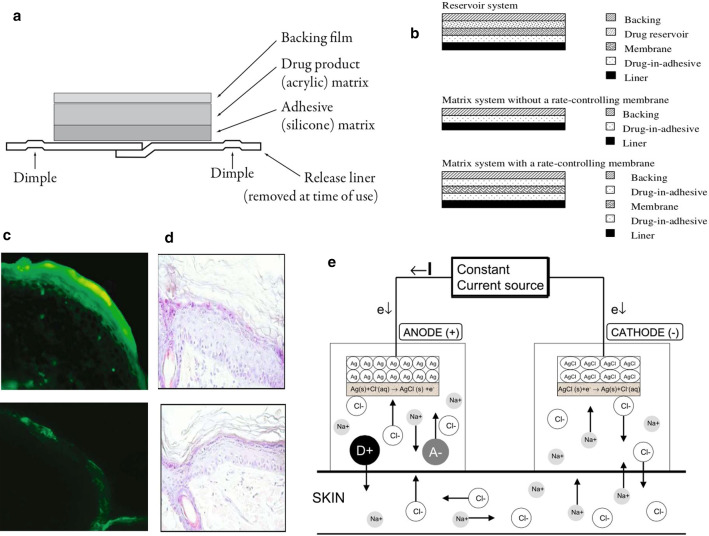
The very important point of skin medication is that transdermal drug delivery is needed to achieve a therapeutic dose, which makes a long time, also the single application time, and it will be affected by sweating and other reasons. Transdermal patches can well solve the problem of long-term application. The development of "matrix" patches combines drugs, polymer films and adhesives to form a single "matrix" layer. This design allows them to be small and thin, and avoid the use of irritating substances. For example, Fig. 3a is a cross-section of a typical matrix patch, including a base film, a vial, a matrix, an adhesive, and a release paper [24]. In general, patches can be classified as reservoir systems, matrix systems without rate-control membranes or matrix systems with rate-control membranes, as shown in Fig. 3b [25]. The storage system consists of three main parts: drug storage, rate control membrane and adhesive. The drug reservoir contains drugs and excipients. The drug penetrates the skin through the membrane and the adhesive. In the matrix system, the drug is located in adhesive. The adhesive serves as the basis for the formulation and the adhesive. In addition, the size of the patch limits the amount of drug that can be contained, and it can only deliver low molecular weight (≤ 500 daltons) and high lipophilic drugs [26]. Because of the obvious advantages and disadvantages of transdermal drug delivery, researchers have invented iontophoresis to aid the drug delivery and make up for its shortcomings.
Fig. 3.
a Cross-section of a typical matrix patch, in this case the rivastigmine transdermal patch (Exelon® patch, Novartis Pharma AG, Basel, Switzerland) [24]. b Types of transdermal drug delivery systems [25]. c In the experimental group, heparin after pulse current was observed in both the stratum corneum and the epidermis, and fluorescence was also observed in some areas of the dermis. Total magnification × 100 (right). In the control group, no fluorescence was observed in the epidermal layer and the dermal layer. Total magnification × 100 (left) [28]. d Histology of rat skin treated or untreated with pulsed current iontophoresis. Rat skin treated by pulsed current iontophoresis, total magnification × 200 (left), control (right) [28]. e Iontophoresis using a Ag/AgCl electrode system [27]
Iontophoresis is a non-invasive therapy. It places a small charge in an electroosmosis room containing similar charges, inserts a wet pad between the electrode plate and the skin to prevent the skin from being burned, and the anode repels positively charged chemicals. While the cathode repels negatively charged chemicals, the influence between the electrodes along the skin surface is negligible, and it is essentially active transport. In addition to its electrical repulsion, it can also bring about an electroosmotic flowing effect. Physiologically, the skin is negatively charged with PH, and the cations are selective. The specific example is the Ag/AgCl electrode system, as shown in Fig. 3e, in the anode compartment, the pharmaceutical preparation (D+A−) containing the ionizable drug D+ is placed in the electrode compartment with the same charge. The electrode chamber is placed at the far end of the skin, and an electric potential is applied to cause a current to flow through the circuit to introduce the same charged ions into the skin [27]. Pacini et al. [28] used pulsed current electroosmosis to introduce fluorescently labeled heparin into the skin of living rats, as shown in Fig. 3c. In the pulsed current electroosmosis experimental group (left), fluorescence was observed in the stratum corneum and the epidermis. For the marked heparin, fluorescence can also be observed in some areas of the dermis, and the contour of the fluorescence is distributed along the intercellular path, and the contour of the cells can be clearly seen, but no fluorescence was observed in the epidermis and dermis in the control group (right). Pacini et al. [28] also studied the effect of iontophoresis on the skin. After staining the skin after the experiment, they found that there was no significant difference between the treatment group (left) and the control group (right), as shown in Fig. 3d. Observed and well-preserved, the epidermal thickness of the experimental group was uniform and uninterrupted, and various connective cells were intact around. Iontophoresis can use a lower current to improve the transdermal delivery of drugs in a short period of time.
Under the influence of the current, molecules are transported through the electrolyte solution to penetrate the biofilm. This means that the CDC method or PDP method can use 0.5 mA/cm2 or less current to the skin, which can significantly speed up the drug delivery to the therapeutic dose, and at the same time, the human body can also accept such current levels [29]. Marro et al. [4] conducted a test of suitable skin for experiment for iontophoresis, using mannitol as the introduced drug, and found that pig skin is the most suitable for electroosmosis, and the permeability depends on voltage and current density. Bhatia and Singh used pigskin experiments and introduced luteinizing hormone-releasing hormone (LHRH) with iontophoresis. The results showed a significant increase in flux. The experimenter also used this method to study anti-anxiety, anti-diabetes, and cardiovascular diseases such as systemic systemic drug delivery [5]. Mutalik et al. [30] then continued to develop membrane-regulating systems for glipizide and matrix patch systems for the glipizide delivery, which was more effective than oral formulations, and showed that transdermal delivery of drugs using a combination of a drug and a polymer patch had good results and successfully prevented lower in the first few hours glucose and is also effective for chronic applications. Compared with oral administration, the stimulating effect of the transdermal route on the skin is negligible. The scanning electron microscope of the preparation patch showed that after in vitro penetration studies, pores were formed on the surface, as shown in Fig. 4a, the smooth surface of the film (A and B) penetrated and the surface became rough (A1 And B1).
Fig. 4.
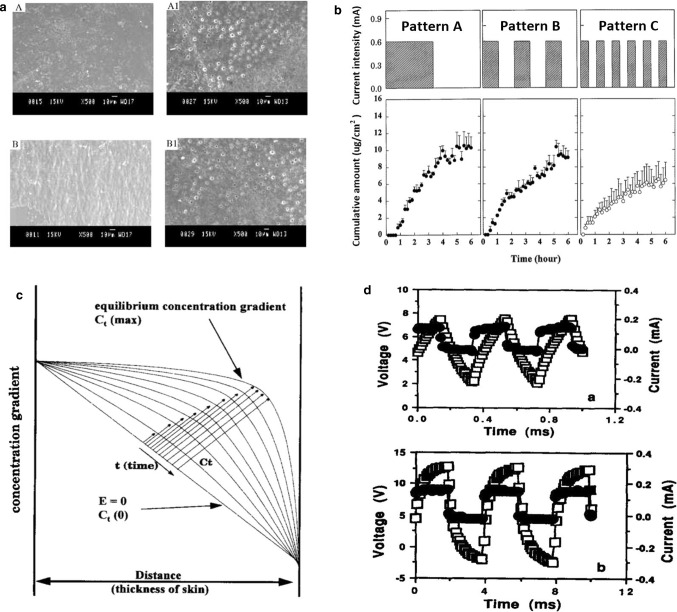
a SEM photographs of transdermal patches A: EC/PVP (3:2) before permeation studies; A1: EC/PVP (3:2) after permeation studies; B: ERL/ERS (4:1) before permeation studies; B1: ERL/ERS (4:1) after permeation studies [30]. b Comparison of transdermal iontophoretic skin permeation profiles of LHRH by three different application pattems with a current intensity of 0.6 mA [3]. c Schematic illustration of different concentration gradients across skin during various durations of current application. The transdermal iontophoretic skin permeation of LHRH is proportional to the concentration gradient, whose equilibrium concentration gradient is time-dependent [3]. d Current (filled circle) and voltage (open square) profiles 1 h after the initiation of lysine iontophoresis with 50% square-wave, unipolar, pulsed DC at frequencies of (up) 2500 Hz, (down) 250 Hz [31]
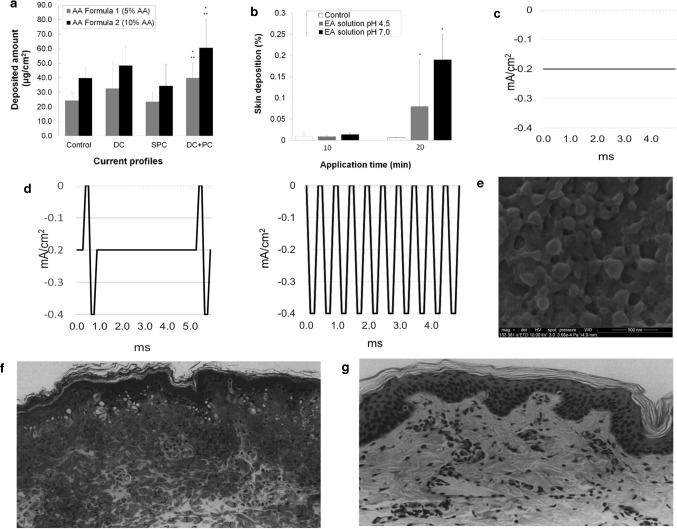
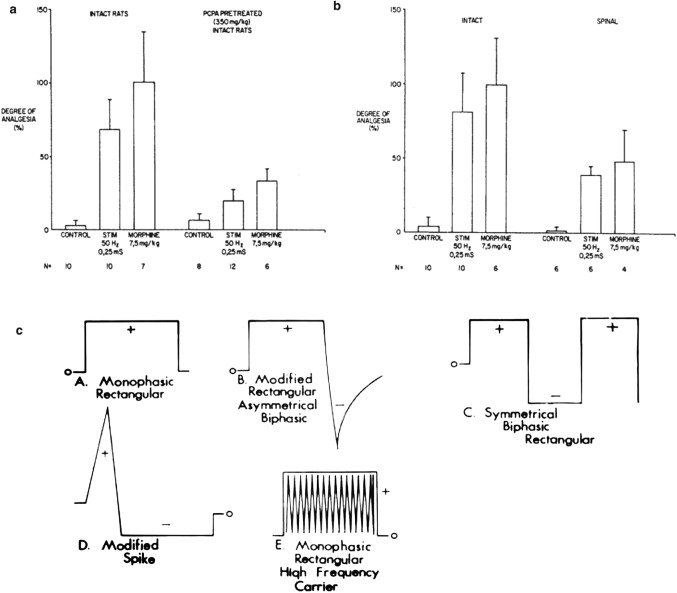
Regard to the effect of the parameters of iontophoresis on the efficacy, the current research is mature. According to Faraday's law, no matter whether it is a pulse current or a stationary current, the ion throughput is equal within the time when the average current is equal. What needs to pay attention, is the current should not be too large, so you need to increase the current intensity while making the skin tolerate. When the voltage is the same, decreasing the skin impedance increases the current. Plutohik pointed out that when the frequency is increased from 1 to 100 Hz, skin impedance will decrease from 130 kΩ to 30 kΩ [2]. For pulse current therapy, square wave pulses and exponential pulses are common. It is generally believed that square wave pulses can significantly increase drug permeability by increasing energy before reaching the energy saturation value. For exponential pulses, when the electroporation energy is unchanged, the transdermal rate of the drug will decrease over time. For AC pulsed electroosmosis therapy, Yamamoto found that when the frequency of the pulsed current is increased, the impedance on both sides of the skin decreases and the effective intensity on both sides increases when the current intensity is constant [1]. Chen and Chien [3] performed a pulsed direct current electric iontophoresis experiment of LHRH on the skin of hairless rats, using different waveform and duty cycles. The results show that the pulse waveform has little effect on penetration enhancement. However, the triangular wave has the smallest increase, and the higher the duty cycle, the more significant the enhancement of the penetration, the researchers used a polyacrylamide hydrogel storage device to characterize the effects of several electrical parameters on the transdermal iontophoresis of LHRH hairless rat skin. When current is applied continuously instead of in between, LHRH has a higher iontophoretic permeability. As shown in Fig. 4b, when direct current (DC) of 0.6 mA is continuously applied for 3 h, the total accumulation of LHRH in current application mode A is the highest, and the LHRH iontophoresis in mode C is the least. The schematic diagram of different concentration gradients on the skin during different durations of application is shown in Fig. 4c. LHRH transdermal iontophoresis skin penetration is proportional to the concentration gradient. The equilibrium concentration gradient is related to time. The difference between the various waveforms is not obvious. The on–off rate of pulsed direct current has a significant effect on the transdermal iontophoresis of LHRH, and proving that the higher the on–off rate (duty cycle), the greater the skin permeability of LHRH. For percutaneous iontophoresis, the duration of current application is more important than the amplitude and intensity of the current. It is worth noting that Hirvonen et al. [31] had the amino acid iontophoresis experiments show that constant direct current electroosmosis therapy has the highest efficiency under the same charge, and pulse electroosmosis cannot provide drugs that exceed the CDC current, and the frequency of the pulse does not significantly affect the delivery, as shown in Fig. 4d. It is a waveform of a unipolar square wave with a 50% duty cycle at different input frequencies. When the frequency increases, the voltage lags behind the current, witch due to the skin changes caused by electric current. The polarization of skin caused by osmotic therapy inevitably reduces its efficiency over time. Cazares-Delgadillo et al. [6] used different types of currents (DC, square unipolar pulse current (SPC), DC + pulse current (PC)) to perform skin experiments on cosmetics (ascorbic acid (AA) and ellagic acid (EA)). The current waveform is shown in Fig. 5c, d, when different percentages of AA solution were used, the import results are shown in the Fig. 5a, the group with a high AA content had more deposits, and the DC + PC group had the best effect. The DC group was second, and the SPC group had no advantage over the control group. In addition, the pH of the solution is also a factor that affects the amount of deposition in a certain period of time. As shown in the Fig. 5b, when the EA solution pH is 7, the EA molecules are mainly negatively charged, which increases compared to the amount of pH 4 [6].
Fig. 5.
a Deposited amount of AA obtained with AA Formula 1 (5%) and AA Formula 2 (10%) at each current profile (0.2 mA/cm2) for 5 min iontophoresis (n = 6) [6]. b Comparison of the deposition of 0.2% EA solution at pH 4.0 and pH 7.0 at 0.3 mA/cm2, applied for 10 min and 20 min iontophoresis. (l) Scanning electron microscope image of flufenamic acid-loaded FA-PLGA nanoparticles [6]. c Negative polarity current profiles, galvanic DC [6]. d Negative polarity current profiles, from left to right are galvanic direct current and pulse current (DC + PC), and SPC [6]. e Scanning electron microscope image of flufenamic acid-loaded FA-PLGA nanoparticles [44]. f Effect of lidocaine on porcine epidermis. Light micrograph of a 1 μm thick plastic section showing alteration of pig epidermis produced by iontophoresis of 4% aqueous lidocaine hydrochloride (3 mA for 45 min) in vivo. Note the dense cytoplasmic staining, the darkly stained flattened nuclei in the upper stratum spinosum and the stratum granulosum and the vacuolation of cells of the basal layer [32]. g Light micrograph showing control porcine skin [32]
The principle and efficacy of iontophoresis have been widely recognized, so safety and drug delivery toxicity are worthy of attention. Studies have shown that the rate of reduction in skin resistance depends on the applied voltage, and the time for reversible recovery depends on the size and duration of the electric field. The skin irritation in iontophoresis is partly due to the polarization caused by continuous DC induction. By applying current in a high-frequency pulse waveform, especially by briefly reversing the current during each cycle, this polarization can be avoided. Studies have shown that when lidocaine is released from sodium chloride aqueous solution for 10, 40 or 100 min within a certain current range, various macroscopic and microscopic changes occur in the pig epidermis, as shown in the Fig. 5f, g, vacuolation of basal layer cells occurs. Clinically, it is easy to observe various degrees of erythema and "petechia". The erythema below the anode usually disappears after 24 h, while it is very rare under the cathode. The cathode is associated with a higher degree of edema and the formation of some blisters [32]. Okabe et al. [33] carried out a study in volunteers and used beta-blockers for transdermal experiments, and no skin damage has been observed. The effects of transdermal administration are divided into two types: local delivery and systemic delivery. Other researchers have tried using electroosmosis to treat arrhythmias. They delivered an effective dose of verapamil, and concluded that electroosmosis would be safer and more reliable if delivered with high systemic toxicity [34]. A recent study shows that electrical stimulation can control the rate of peptide release, especially fibrin peptides used for tumor image diagnosis and inhibition of tumor cell migration, displaying a wide space for electrical stimulation therapy [35]. In fact, the PDP method is the mature therapy in iontophoresis, and local delivery greatly reduces adverse reactions such as skin polarization and redness, and systemic delivery is better than oral and injection. Iontophoresis is very compatible with other therapies and works well. Wangetal researched the efficacy of iontophoresis combined with the microneedle therapy, ultrasonic dialysis therapy, and chemotherapeutic therapy, and believed that combined therapy can increase the poor water solubility and the penetration efficiency of macromolecular compounds [36]. It is concluded that the combination of very low density current with penetration enhancers can significantly improve efficiency, and with this current density, the skin resistance can be restored as before. Nair et al. [37] studied the penetration of tartrate to enhance the penetration of chemical penetration enhancers combined with iontophoresis. Donohoe et al. [38] conducted vagus nerve stimulation (VNS) experiments with electrical impulses, which can alleviate seizures such as epilepsy, which is an advancement of existing therapeutic nerve stimulation. Oh and George [39] used conductive polymers for drug delivery to increase the nerve recovery after stroke. Wang et al. [40] designed a nanogenerator system to assist in the proliferation of osteoblasts, and proved the plasticity of P (VDF-TrFE) piezoelectric nanofiber NG, which can be used in a self-powered electrical stimulation system, and proposed its use in tissue and important potential applications in regeneration. Sebastian et al. [41] studied iontophoresis and lactic acid combined therapy, and pointed out that the ionic properties of the penetration enhancer molecules will affect the combined therapy. Bottoni [42] pointed out that although the use of silver ions and micro-current can reduce the number of bacteria in vitro, it is not clear that whether they can effectively reduce postoperative infections. Further research should be done when making micro-current silver electrodes. Mousavi et al. [43] developed a silk fibroin film-based drug delivery device and made it conductive, which can be electrochemically loaded, which is a new application of electrical stimulation. In addition, Gomez et al. [44] encapsulated flufenamic acid with negatively charged nanoparticles and introduced it into the body using iontophoresis. Figure 5e shows the negatively charged nanoparticles with FFA, although the results show that the penetration promoting effect is not ideal, but this shows the research potential of iontophoresis. Kolli et al. [45] combined microneedle technology with iontophoresis, and used microneedle as a pretreatment and then used iontophoresis. The results can improve the drug flux. Zhao et al. [46] pointed out in a review paper on micro-current that current electrical stimulation has a wide range of direct effects on biomolecules and cells, but the research on the basic mechanism of electrical stimulation lags behind experiments and applications. What is most needed is a stimulation program optimization and development of matching equipment. These studies show that electroosmosis still have much room for the expansion.
Electroporation
Electroporation therapy refers to the use of high-voltage short electrical pulses, which act on the lipid bilayer membrane to produce transient structural disturbances, which can enhance the penetration from small molecules to large molecules. It can also enhance the penetration of charged molecules and neutral molecules into the skin. There are two pathways for drug transport caused by electroporation, namely the curved pathway (for example, hydrophilic substances are transmitted through lipid domains) and the linear pathway (for example, hydrophilic substances are directly transmitted through lipid regions and electroporation holes on keratinocytes).
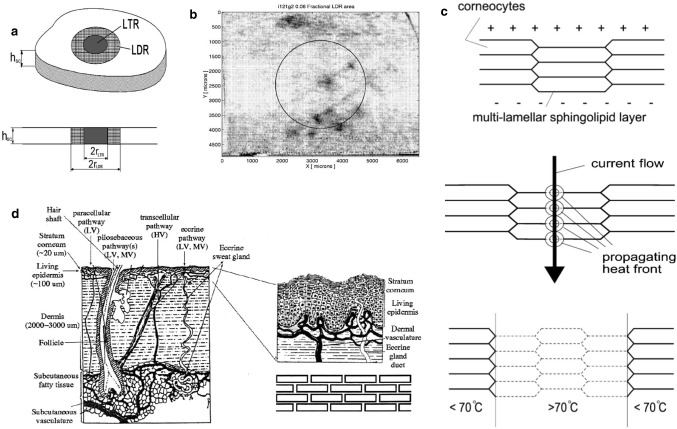
Under high-voltage pulses, the structure of the skin is divided into highly permeable local transport regions (LTRs) and non-permeable local peripheral regions (LDRs). Electroporation is considered a non-thermal phenomenon. However, the effects of related processes, such as Joule heating, should be considered. Because electroporation of the skin, especially the stratum corneum (SC), occurs at a highly localized location, the researchers predicted a local temperature increase and confirmed this through experiments. If voltage across SC reaches about 60 V, electrical breakdown related to electroporation may occur. As long as the pulse lasts, molecular transport can be generated by local electrophoresis or other driving forces. Small ion species dominate, and the transport of larger molecules is limited to one point within the LDR. This area is called LTR, as shown in Fig. 6a. The range of LDR is 1% to 30%, and the range of LTR is 0.1% to 10%. The heat distribution visualization is shown in Fig. 6b, which is the image acquired by the camera at the highest temperature. The circle indicates the area seen by the infrared detector. Keratinocytes are like bricks fixed together. The direction is shown by the arrow in Fig. 6c. The current will generate heat. The resulting electroporation reduces the electric field strength at the hole, and the possibility of electroporation increases with temperature. If the vicinity of LTR is heated, electroporation will occur. In the middle of the LTR, high temperature will enrich the water path in the lipid structure. After stopping the pulse, the channel in the vertical direction is still open [47]. Electroporation also has two types of exponentially decaying waves and square waves, and most cases are exponentially decaying waves. Electroporation is a new drug delivery method for macromolecular substances. Human skin is complex, with many functions and features, and candidate sites with multiple pathways are shown in Fig. 6d. A "brick wall model" is introduced to describe the passive diffusion of aliphatic molecules on SC. The lipid-based structure tends to exclude charged substances, the cell membrane of SC, the cell layer connected by tight junctions, and the multi-layer lipid bilayer membrane of SC all has relatively large electrical resistance, and electroporation occurs at a higher voltage, formation of water channels and electrical drive transmission of small ions and water-soluble molecules [48] voltage is usually several hundred volts, and the time constant is about 5 ms. Researchers have pointed out that in addition to short-term high-voltage pulses that cause electroporation, electroporation can also occur in conventional iontophoresis (when low voltage is applied for a long time), and analyzed the relationship between the location of electroporation and the electric field. As shown in Fig. 7d, when the induced electric field strength E = ET, the minimum transmembrane electric field strength required to cause dielectric breakdown. When E > ET, other electroporation is induced at a farther distance. If the motion is directed at the transmembrane electric field, the net negatively charged entities inside the spherical membrane will diffuse through the pores. The net positively charged entities inside the spherical membrane can diffuse out because the direction of motion is the same as the electric field near the hole [49].
Fig. 6.
a Schematic of the stratum corneum with LDR, a localized region (radius = 30–500 μm) where electric energy is dissipated, and which is characterized by a drop in resistance [47]. b A complete set of results from one experiment. The pulse parameters were Uskin = 83 V and τpulse = 95 ms [47]. c Model for the electrothermally induced changes at stratum corneum. (A) Stratum corneum before high-voltage pulse, (B) during electric current flow with propagating heat front starting at the center of maximal current density, and (C) partial recovery of the stratum corneum after pulse at sites where the phase transition of the lipid structure was not reached [47]. d Key features of skin, skin barriers and hypothetical aqueous pathways [48]
Fig. 7.
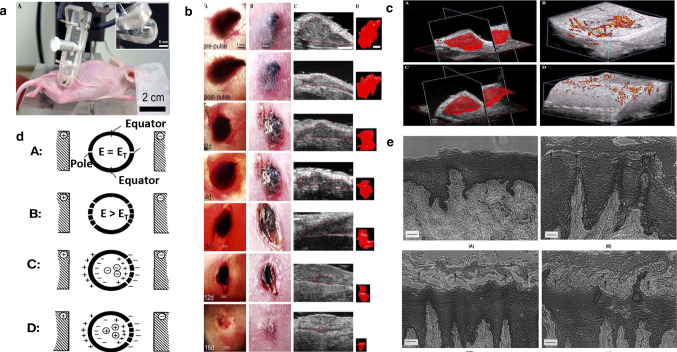
a Photograph of SKH-1 hairless mouse being treated with parallel plate electrode under isoflurane inhalation anesthesia. (Inset) Close-up of one of the plates of parallel plate electrode showing it recessed by 0.5 mm to allow a space for a conductive agar gel to be placed on it [7]. b Typical response of a melanoma to three applications of 100 pulses (300 ns, 40 kV/cm, 0.5 Hz) 30 min apart on day 0 followed by a single application on day 4 using a 5 mm diameter parallel plate electrode on mouse #102. Collection of seven matched sets of images of the same tumor all taken on the day indicated in the lower left corner of the transillumination image [7]. c Blood flow in melanoma before and after nsPEF application. (A) 3-D reconstruction of volume of melanoma; (B) power Doppler reconstruction of blood flow before field application. (C) 3-D reconstruction of volume of the same melanoma shown in (A) generated about 15 min after 100 pulses (300 ns, 40 kV/cm, 0.5 Hz). (D) Power Doppler reconstruction of blood flow in the same tumor shown in (B) generated about 15 min after 100 pulses (300 ns, 40 kV/cm, 0.5 Hz) [7]. d Location of electropores with respect to electric field. (A) One electropore at pole of each hemisphere when induced electric field strength, E, is equal to the minimum transmembrane electric field strength needed to cause dielectric breakdown, ET. (B) Additional electropores induced at greater distance from poles when E > ET. (C) Net negative charged entity inside spherical membrane is inhibited from diffusing through electropores if movement is against transmembrane electric field. (D) Net positive charged entity inside spherical membrane can diffuse out since movement is in same direction as electric field in vicinity of pore [49]. e Photographs of formalin-fixed, paraffin-embedded cross-sections of porcine buccal mucosae. Hematoxylin and eosin staining: (A) Control, untreated. (B) Control, passive permeation after an 8 h treatment period. Slightly enlargement of 2–3 cell layers (arrows) [50]
Electroporation can be used in combination with many other technologies and has a good effect. For example, electroporation can be combined with ultrasound or iontophoresis. The introduction of luteinizing hormone release hormone has a five-fold increase in permeability compared with the use alone, and reduces skin erythema, the same composite technology also has a good promotion effect on macromolecular drugs such as insulin and calmodulin [9].
High-frequency direct current may cause skin damage, causing erythema, herpes, and even burns. Jacobsen's in vitro three-chamber osmosis cell promoted atenolol hydrochloride ion penetration experiments showed that the outer layer of epithelial cells was disturbed after a treatment period of 1 mA at 8 h, as shown in Fig. 7) [50]. Compared with skin redness and swelling caused by CDC or PDP therapy, the effect of electroporation is relatively slight, which can cause focal intraepidermal edema and vacuoles, which increased with increasing voltage. However, for electroporation, skin redness, burns, fever, nerve irritation, and irreversible electroporation are clearly pointed out, and dose prediction between different drugs, the model is not universal and the prediction is more complicated [26]. Jacobsen [50] found that electroporation can treat skin tumors and cause the tumor cell nucleus to contract quickly and the tumor blood flow to stop. The hairless mice clamped by the parallel plate electrodes are shown in Fig. 7a, and the electrode diameter is 3–5 mm. These electrodes are coated with conductive agar to separate the skin from the electrodes. The distance between the two plates is 0.5–1 mm, the number of pulses is 100, the duration is 300 ns, the amplitude is 4–8 kV, and the frequency is 0.5 Hz. As shown in Fig. 7b, there was a difference between the treatment group and the control group two days after the experiment. The black SC appears on the stratum corneum of the pulse area and regenerates in the stratum corneum. It consists of coagulated red blood cells. After treatment, the tumor usually shrinks by 90% within two weeks. Figure 7c is a 3-D reconstruction of the volume of melanoma. Histology confirmed that red blood cells scatter in and around the melanoma tumor. This means that local blood vessels leak and red blood cells escape into the surrounding tissue. Tumor blood flow usually cannot be restored [7].
Iontophoresis and electroporation are widely used as two important therapies for transdermal introduction. Iontophoresis is mainly applied to drugs, and the movement of charged particles caused by electrostatic repulsion will incidentally change the structure of the skin, while electroporation changes the permeability of tissues, thereby increasing drug transport, regardless of the voltage applied electrode. Studies have shown that, from the applied voltage, electroporation contains ultrashort pulses with a duration of several milliseconds, with an intensity of several hundred volts, and a transdermal iontophoretic voltage of 3–12 V, which lasts from several minutes to several hours [51]. In effect, the mechanism of electroporation and iontophoresis is still controversial, such as Weaver believes that the difference lies in whether new channels are created, but from the foregoing, it can be seen that with low voltage for a long time similar pores also appear in the electroosmotic therapy, and Hui believes that both use the electric field to increase the permeability of skin accessory channels, there is no essential difference [52]. It is not accurate at all. Electroporation is obviously different from iontophoresis from the application mechanism to the effect, but there is an overlap in the phenomenon. Overall, these two therapies are now relatively mature.
Micro-current stimulation for wound healing
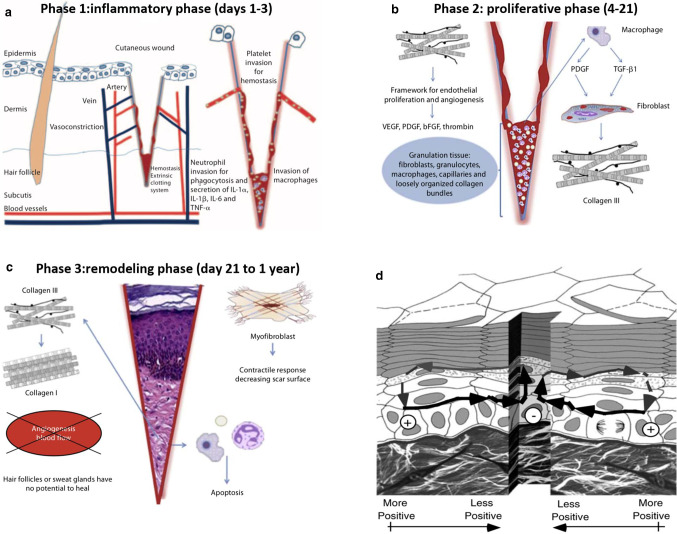
Human skin is the largest organ exposed, and injuries are very common. In addition, there are many diseases that can cause difficult-to-heal wounds on the skin, such as venous ulcers, which keeps the body in a morbid state and will cost huge medical expenses. The principle of wound healing is more complicated. The healing process consists of four stages (in order): the formation of temporary wound matrix (including hemostasis and coagulation), the accumulation of neutrophils and monocytes, the formation of granulation tissue, re-epithelial tissue and blood vessels Network recovery (proliferation and repair), reshaping phase. The first stage of wound healing is hemostasis and the formation of a temporary wound matrix. This process occurs immediately after injury. As shown in Fig. 8a, the wound healing inflammation stage is activated during the hemostasis and coagulation stages and can be roughly divided into neutrophil recruitment stage of monocytes and the late stage of monocyte appearance and transformation. The next step is the proliferative phase. The final step of the proliferation phase is the development of acute granulation tissue. As shown in Fig. 8b, the remodeling phase has begun. As a transitional tissue, it replaces the fibrin-based fibronectin-based temporary wound matrix and may cause scarring as it matures. Finally, during the remodeling phase, the formation of granulation tissue is stopped by apoptosis. During wound maturation, collagen III produced in the hyperplastic stage is now replaced by stronger collagen I. As shown in Fig. 8c, this type of collagen is oriented into small parallel bundles, and myofibroblasts pass through multiple attachments cause wound contraction and help reduce the surface of the scar that is forming [53]. There is a clear understanding of the mechanism of wound healing, but related therapies are not systematic. Modern medicine has greatly developed treatment of wounds. Therapy involves many aspects.
Fig. 8.
a Inflammatory phase after a cutaneous cut; hemostasis and invasion of inflammatory cells [53]. b Proliferative phase; organization of the thrombus, secretion of growth factors, synthesis of collagen III and the beginning of angiogenesis [53]. c Remodeling phase; regenerative processes fade and are followed by reorganization of the connective tissue and contractile response [53]. d Generation of skin wound electric fields. When wounded, the potential drives current flow through the newly formed low resistance pathway generating an electric field whose negative vector points toward the wound center at the lower portion of the epidermis and away from the wound on the upper portion below the stratum corneum [57]
Wound healing
The electric field plays a very important role in skin wound healing, and it can guide cells to complete the healing work naturally and orderly [10]. The microenvironment in the human body has a moderating effect on the biological behavior of cells [54]. At the same time, to study the specific impact of the microenvironment on cells, it is necessary to observe the microenvironment, such as the local oxygen microenvironment, which requires the assistance of materials such as hydrogel [55]. In both physiological and pathological conditions, cells can be observed to migrate in the interstices of tissues, including immune cells and cancer cells. Electrical stimulation can also regulate cell movement by promoting the formation of intercellular pores [56]. At present, research on skin wound healing is very rich. The types of currents including DC, high voltage pulsed current (HVPC), AC, and so on. Earlier studies on the effects of currents were messy and there were contradictions between conclusions.
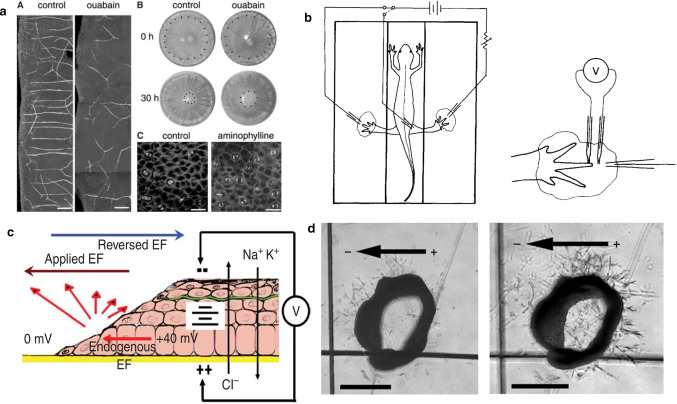
An endogenous DC electric field is naturally generated in the body. The research pointed out, that below the epidermis, the polarity of the electric field has a negative pole at the center of the wound, and above the epidermis, the waveform current flows in the opposite direction, as shown in Fig. 8d, these wound areas have some useful signal transmission characteristics, they immediately appear after injury, driving current out of any area of low resistance. The transverse electric field in Fig. 8d will continue to exist until the resistance increases as the wound heals. Therefore, long-lasting signals stimulate wound healing. Another very important experimental result is that the endogenous electric field near the wound has a very strong directional effect on the direction of sensory nerve germination and growth. Between 16 and 20 h after injury, a large number of nerve buds protrude directly from the wound incision edge in the entire rat cornea, as shown in Fig. 9a. In general, the electric field of a skin corneal wound is about 40–50 mV/mm, and the electric field of a skin wound is about 100-150 mV/mm. The experiment of wound current supply is shown in Fig. 9b (left). The newt is placed in the electronic room, and only the feet are extended into the pond. Minimize the shunt of the supplied current. The benzocaine-soaked pad and animal anesthetic were omitted. Figure 9b (right) is a view of a current supply electrode for potential measurement [57]. The polarity of the electrodes can have different effects on the wound. Song et al. [10] pointed out that the application of electric field (EF) (brown arrow) in the direction of wound healing will enhance the induction of endogenous EF (red arrow) in the wound and increase the wound healing rate. Conversely (blue) inhibits endogenous wound healing behavior, as shown in Fig. 9c. At the same time, their directed angiogenesis experiments showed that in the 3D armature chamber, the aorta of the vascular-like structure of the rat grows toward the anode, as shown in Fig. 9d. In the process of wound healing, low direct current will not damage healthy tissue. Among them, the proliferation and migration of fibroblasts are extremely important. In the early stage of wound healing, fibroblasts migrate to the wound area to proliferate and interact with surroundings (keratinocytes, adipocytes), etc. Fibroblasts differentiate and produce extracellular matrix (ECM), glycoproteins, adhesion molecules and various cytokines.
Fig. 9.
a Observations made near a wound in rat cornea. (A) Neuron outgrowths are strongly aligned by the endogenous electric field and exhibit a more random orientation when the field is reduced by ouabain addition. (B) The rate of corneal wound healing is reduced in the presence of ouabain. (C) The frequency and orientation of division planes is influenced by the field strength. Aminophylline increases the transcorneal potential and stimulates an increase in the rate of cell division and orientation of the axis of division perpendicular to the field [57]. b Diagram of the setup for current supply to the wounds. The newt was placed in a three-compartment chamber with only the feet immersed in pools of pond water. This minimized shunting of the supplied current (left). The benzocaine-soaked pads for keeping the head and trunk moist and the animal anesthetized are omitted for the sake of clarity (right). A view of one current-supplying electrode and the two microelectrodes used for potential measurements [57]. c The diagram illustrates the interaction between electric fields. The electric field can be directly attached to the endogenous electric field of the wound, where applying EF (brown arrow) in the default direction of wound healing will enhance wound-induced endogenous EF (red arrow), thus increasing the wound healing rate. In contrast, reversing the direction of EF (blue arrow) against the default wound healing direction will suppress the endogenous EF in the wound (red arrow), thus reducing or even stop the normal wound healing behaviors [10]. d Directional angiogenesis is a vascular-like structure of a rat placed in a 3D armature chamber with the aortic ring growing toward the anode. On the left is the aortic ring after embedding; on the right is day 3, after 200 mV/mm EF treatment. Rod is 500 μm [10]
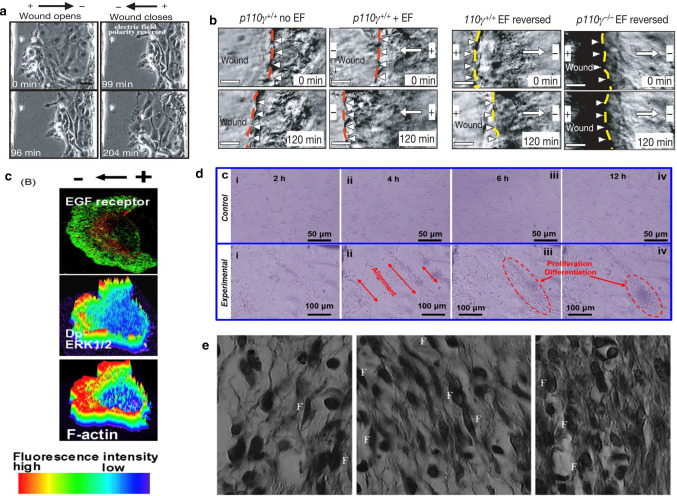
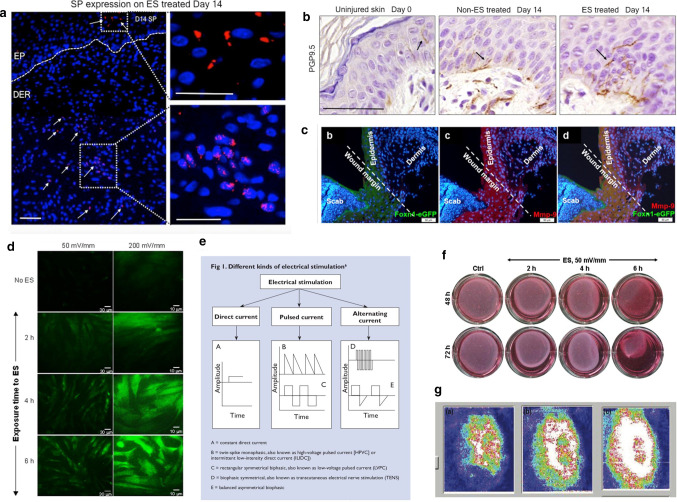
The rat experiments of Long et al. [58] show that electric fields can promote fibroblasts, to differentiate muscle cells and provide contractile force to close the wound. When fibroblasts are cultured separately, with 6 h electric field stimulation, fibroblasts can be observed to differentiate along the electric field lines. As shown in the Fig. 10d. Zhao et al. [59] also tested the effect of electrical signals on cell movement. The researchers observed cell migration in a single layer of epithelium. When using a polarity opposite to the default treatment direction, epithelial cells followed the electrical signal movement, the wound opened, and the polarity was reversed. After sexual wounds are closed, as shown in Fig. 10a, similarly, corneal cells move faster when the treatment electric field is applied, and wound healing speeds up, but when the opposite polarity electrode is applied, it will hinder healing, as shown in Fig. 10b, c. Another study by Zhao [60] explained the specific principle of how electric fields affect cells. The researcher fluorescent cells such as F-actin (which drives cell movement) and placed them in the electric field. The redistribution of fluorescent markers in keratinocytes and corneal epithelial cells was observed within minutes. As shown in Fig. 10e, the polarized agonist polymerized to cause the cells to move directionally, that is, the cells were depolarized on the cathode-facing side and hyperpolarized on one side of the anode. At the same time, study have pointed out that multi-cell behavior is more complicated than single cell behavior. The behavior of collective cells involves cell polarization, and this process is also regulated by electrical stimulation [61]. Embryonic stem cells differentiate in many directions, and electrical stimulation can induce cell differentiation [62]. The mobility of different types of cells varies greatly. In vivo and in vitro experiments have also proven that it induces epithelialization and promotes wound contractions. Positive polarity will enhance the migration of epithelial cells and cause the wound to close faster. Using the cathode as the active electrode can more effectively attract fibroblasts to the wound area. Figure 10f from left to the right is the control group, the cathode ES group and the anode ES group, 7 days after the experiment, the number of fibroblasts (F) in the cathode ES group was significantly higher than that in the control group, and more collagen could be produced. In addition, electrical stimulation can promote fibroblast differentiation [63]. Skin fibroblasts exposed to 50 mV/mm contracted the collagen gel matrix. After exposure to ES, fibroblasts added to type I collagen gel and cultured. The diameter of each collagen gel was measured at 48 h and 72 h. As shown in Fig. 11f, the experimental group contracted significantly. Another experiment revealed the effect of ES on the expression of α-SMA in fibroblasts. Skin fibroblasts were seeded on conductive membranes and exposed to different times of 50 or 200 mV/mm. Then, the cells were separated from the conductive membrane, washed, and seeded on a cover glass. Myoblasts labeled with cytoplasmic immunity were proposed. As shown in Fig. 11d, the number of labels increased significantly [64]. In addition, electrical stimulation can cause an increase in capillaries. ES has been shown to increase local blood circulation, increase tissue perfusion, and promote capillary growth. Recent studies have shown that electrical stimulation can lead to enhanced expression of neuroendocrine markers in the wound and can enlarge innervation. Studies by Sebastian et al. [65] showed that neuropeptide synthesis is up-regulated during ES treatment, as the treatment time increases, PGP9.5+ nerve fibers gradually increase in healing wounds Fig. 11b, especially on the 14th day after treatment, on the other hand, SP (substance P, neurotransmitter) expression increased under ES treatment Fig. 11a. Besides, for the first time, Lallyett et al. [66] conduct an analysis of the efficacy of electrical stimulation at the genetic level. The results of the study showed that a specific ES would cause the differential expression of 105 genes, and most of them were down-regulated.
Fig. 10.
a An EF directs migration of corneal epithelial cells in a monolayer model of wound healing (150 mV/mm) [59]. b Stratified corneal epithelium migrate in situ to heal a wound (towards the left) [59]. c Electric fields applied with polarity opposite to the default healing direction direct the wound edge to migrate away from the wound [59]. d Electrical stimulation experiments of fibroblasts cultured in the absence of (control) and (experimental) electric fields. It can be observed that fibroblasts differentiate along the electric field line [59]. e Polarized EGF receptors, dp-ERK (activated mitogen-activated protein kinase) and actin polymerization. Dp-ERK1/2 and F-actin are surface plots that represent fluorescence intensity [60]. f Light micrograph (hematoxylin and eosin, 100) of full-thickness wound in guinea pig 7 day after incision, control, cathodal ES, and anodal ES, respectively, from left to right. Cathodal and anodal DC ES (sensory intensity) was applied for 1 h per day, every other day, for 7 days. The number of fibroblasts (dark fusiform cells, F) was significantly higher in cathodal ES group compared with control group [63]
Fig. 11.
a Representative immunohistochemistry (IHC) of SP expression. Dotted lines indicate epidermal-dermal junction. Scale bar is 100 μm [65]. b IHC of PGP9.5 expression [65]. c Confocal microscopy imaging (A and A’) of colocalization of E-cadherin and vimentin-positive cells at day 6 after injury; insets provide its higher magnification [72]. d Effect of ES on fibroblast a-SMA expression. Dermal fibroblasts were seeded on conductive PPy/HE/PLLA membranes followed by exposure to 50 or 200 mV/mm for various periods. The cells were then detached from the conductive mesgmbranes, washed, seeded on coverslips, and cultured up to 70% confluence. The cells were then stained using relevant monoclonal antibodies. Cytoplasmically immunolabelled myofibroblasts are presented [64]. e A constant direct current. B double peak unidirectional current, that is, high-voltage pulse current [HPVC] or intermittent low-intensity direct current. C proves double-symmetric current, also called low-voltage pulse current. D biophasic symmetrical, which is a TENS. E balanced asymmetrical biophasic [69]. f Dermal fibroblasts exposed to 200 mV/mm highly contracted the collagen gel matrix. Following exposure to ES, the fibroblasts were added to a collagen type I gel and cultured. The diameter of each collagen gel was measured at 48 h and 72 h. Representative photos show the action of the ES-exposed and non- exposed cells on collagen gel contraction [64]. g Skin blood flow in a typical subject measured by the laser doppler flow imager before treatment (a), after 20 min wound heating (b) and just at the end of electrical stimulation (c). The whiter the color, the greater the blood flow. Blue represents the lowest blood flow [71]
Chronic wound
Chronic wounds are defined as wounds that have not been repaired after 3 months of appearance. Any disease may cause chronic wounds, and the cause is usually referred to as a reference therapy. Common causes of chronic wounds, such as neurological diseases, hemolytic anemia, rheumatoid arthritis, insufficient arterial blood supply, etc., are mostly related to blood problems [67]. Nair compiles clinical experimental studies on the treatment of chronic wounds by micro-current. The mechanism of action is the triggering or promoting effect of the healing process, and it has a more significant effect as an auxiliary therapy and has great potential [68]. Research on the healing of chronic wounds involves a wide variety of currents, including low-intensity direct current, low-voltage pulse current, alternating current, and transcutaneous TENS, as shown in Fig. 11e, and low-intensity direct current is the first therapeutic current used was tested in both animals and humans [69].
Gault et al. [70] used a constant low-intensity direct current (200–800 μA) to treat patients with ischemic skin ulcers. The researcher applied the negative electrode to the wound three days ago to play an antibacterial and clear necrotic role. After the wound are cleaned out, the positive electrode for the electrode was inverted the average healing time, after 2-h treatments three times a day for 4.7 weeks. The important cause of chronic wound formation is insufficient tissue oxygen supply and insufficient nutrition caused by poor blood flow. High frequency pulse current can greatly alleviate this situation. Petrofsky et al. [71] treated patients with foot ulcers, using ES (two-way sinusoidal current 250 μs, 30 Hz, 20 mA) and local dry heat therapy combined experiment, compared with a control using dry heat therapy after one month of treatment, the reduction of the average wound area in the experimental group was twice that of the experimental group, Fig. 11g shows the skin blood flow of a typical subject measured with a laser Doppler blood flow imager before treatment (a), after 20 min of wound stimulation (b), and at the end of electrical stimulation (c). The whiter the color means greater the blood flow. Blue represents the lowest blood flow. The results showed that the blood flow to the wound and the skin around the wound increased with the increase of ES. Cramp et al. [72] designed high-voltage pulsed electrical stimulation experiments showed that the use of electroporation to remove parts of the skin can be regenerated without scarring, and this method has few side effects, and at the same time, p63 (regulating the proliferation and differentiation factors of mature keratinocytes) had a significant expression after 1 day of electroporation Fig. 11c, the size of the cells marked by staining changed (pointed by the black arrow), and showed on the basement membrane migration phenomenon, the expression of p63 increased to a basic level after one week.
Research on biocompatible conductive materials is also a hot spot in recent years. The review by Huang et al. [73] pointed out that the cell microenvironment has become a key factor in determining the cell growth. The corresponding development of bionic materials is an important source of triggering and regulating cell behavior. Zhao et al. [74] have manufactured bio-materials with high electrical conductivity, wrapped in nano-layers, and can be widely used in the fields of cardiac tissue engineering and flexible electronic devices. Liu et al. [75] designed the electrostimulation wound dressing cloth by making non-proof cotton coated with silver oxide film and the zinc coating, which can significantly accelerate wound healing process, which has strong antibacterial ability and good cell compatibility. Studies have also show that intrinsically conducting polymers (CPs) show great advantages in promoting wound healing, can solve the problem of electrical stimulation applied to the skin, and can be combined with polymers. CPs has great potential in flexible wearable electrical stimulation therapy [76]. Kai et al. [77] use flexible enzyme electrodes and stretchable hydrogels to make bioelectric plasters with good compatibility. Experiments in mice prove that bioelectric plasters can accelerate wound healing. Qing et al. [78] developed a new type of heterostructure composite scaffold, composed of electrospun nanofibers with high conductivity and good biocompatibility on graphene-coated paper, which can induce and enhance the directional growth of neurons, providing graphene. This direction has great potential in the field of stem cell therapy. Lu et al. [79] studied polyHEMA/PPY hydrogel, which can be applied to the skin, and is stronger than ordinary ES electrodes in terms of antibacterial and enhanced wound healing, and can cover and stimulate the entire wound area. This conductive hydrogel has great advantages in the treatment of chronic wounds. The application of biomimetic materials currently covers tissue, cells, genes and other levels, with great potential, and compared to other tissues, skin is more suitable for conducting electrical stimulation experiments with conductive polymers.
At present, experiments on electrical stimulation to promote skin wound healing have achieved good results, but only in the field of low-voltage direct current has a sound and clear theoretical basis and research progress. Other types of current are mostly used to treat chronic diseases caused by various reasons. However, due to the complexity of the etiology and the variety of currents, the voltage and current parameters in this part of the study are not clear enough, and the experimental results are not convincing enough. Current research on current stimulation of chronic wounds requires authoritative experimental data with clear parameters.
Today, 20 years later, this problem has not been solved. Although the experimenters have gradually realized the possible side effects of the current, the analysis of the experimental results is far from rigorous. This makes the field of current stimulation to promote wound healing. Research is not clear enough.
Electrical nerve stimulation for analgesic
Pain is a symptom caused by human diseases, which in itself make the patient very distressed, and many acute and chronic diseases can cause pain, such as pancreatic inflammation, neuropathy, multiple sclerosis with back pain and rheumatoid arthritis, wrist pain and so on. TENS is a treatment method for electrically stimulating patients with pain symptoms to regulate the sensory nerves or to stimulate the valve mechanism or endogenous opioid peptide system. It can deal with many acute or chronic diseases clinically. Compared with painkillers and other invasive therapies, this therapy has almost no side effects. Rushton pointed out that TENS, as a safe non-invasive therapy, have a very obvious role in the suppression of severe pain caused by many conditions [11]. Siemens et al. [80] evaluated the effectiveness and safety of TENS therapy in the treatment of cancer pain, and conducted a single-blind experiment on patients. The results showed the safety of TENS, but the effectiveness is difficult to evaluate. Spinal cord stimulation (SCS) and peripheral nerve stimulation (PNS) also proved to be effective in the treatment of chronic pain [81]. In essence, TENS are a method of relieving pain by applying electrical stimuli of different strengths, different pulse times, and different frequencies to the skin. TENS therapy is based on the gating theory of pain described by Melzack and Wall in 1965 [82]. As shown in Fig. 12a, the T cell is an ascending neuron that may come from the vertebral thalamus, spinal membrane, or spinal midbrain. Substantia gelatinosa (SG) is a neuron in gelatin cells in the dorsal horn of the spinal cord. Open triangles indicate excitatory synapses, while solid triangles indicate inhibitory synapses. Large fibers represent the input of large diameter primary afferent fibers from the peripheral nervous system, and small fibers represent the input of small diameter nociceptive primary afferent fibers from the peripheral nervous system. Small-diameter fibers excite cells in the spinal cord, which sent information to higher centers to sense pain. Large-diameter fiber input reduces noxious sensory harmful inputs by activating inhibitory neurons in the spinal gelatin-like mass. In actual medical treatment, TENS have many modes, corresponding to different intensities and frequencies, and it covers a very wide range.
Fig. 12.
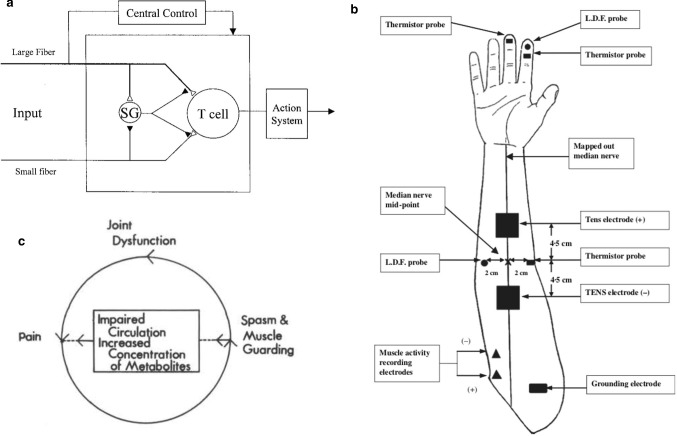
a Diagram showing the gate control theory of pain as originally described by Melzack and Wall, 1965 [82]. b Details of the experimental proce-dure, illustrating the placement of the TENS electrodes, skin thermistor probes, and the recording of muscle activity [83]. c Schematic illustration of a typical pain syndrome and its elements [87]
Regarding how TENS reduce pain, for Chen et al. [83] used a two-channel portable TENS device to conduct an asymmetric biphase wave through two self-adhesive electrodes for human trials, as shown in Fig. 12b. In all subjects, the intensity of TENS was normalized below the exercise threshold. Select the muscle innervated by the median nerve as the target muscle to monitor the motor response. Both the recording electrode and the ground dorsal horn. TENS and morphine produced similar analgesic effects in the complete vertebrate test. As shown in Fig. 13a, electrical stimulation at 50 Hz was similar to systemic morphine. Spinal transection reduced the amount of electrical stimulation or morphine inhibition by about 50%, in addition, TENS stimulation of large-diameter afferent nerves can suppress the nociceptive fiber evoked response in the para-chlorophenylalanine (PCPA) to consume the neurotransmitter serotonin (5-HT) significantly reduced the amount of analgesia produced by electrical stimulation or morphine, as shown in Fig. 13b [82]. Capogrosso et al. [84] developed a soft nerve interface that transmits electrical and chemical stimulation to the spinal cord to restore the movement of the paralyzed or paralyzed limb. TENS can also inhibit inflammatory pain by promoting or inhibiting the expression of related factors [85]. Clinically, TENS are divided into low-frequency TENS stimulation (generally < 10 Hz, 1 × 10–4-4 × 10–4 pulse width, high intensity) and high-frequency TENS stimulation (generally > 50 Hz, 50–100 s pulse width, medium intensity). The transmission frequency (1–4 Hz but the internal frequency of the waveform is high, pulse width 100–250 μs, high intensity) and hyperstimulation (1–4 Hz, 10–500 ms pulse width, high intensity) [86]. Its analgesic principle is to promote the release of pain-relieving drugs in the body and its direct effect on neural sensation. In current research, high-frequency electrical stimulation (such as 100 Hz) is a minority, and commonly it is low frequency alternating current (about 2 Hz) and direct current stimulation. Lampe pointed out that determining the efficacy of TENS includes the design of the exciter itself, the amplitude of the device output, the pulse width, the waveform, and the position of the electrode. Figure 13c describes the common waveforms found in most TENS devices today and provides different ranges of current amplitude, pulse width duration, and pulse rate. When evaluating the efficiency of each waveform clinically, various factors should be considered, and the most effective waveform has not yet been determined. When one waveform does not provide the best level of relief for the patient, another waveform can be used. As shown in Fig. 12c illustrates the typical pain syndrome and its elements. The syndrome includes pain, muscle spasms and joint dysfunction. One of the factors plus the other two will exacerbate the condition. Pain is usually accompanied by muscle cramps, which can lead to progressive joint dysfunction, which in turn leads to more pain [87]. Research by Melzack and Wall [88] show that, in general, pain is transmitted to the spinal cord through delta (small) fibers, while TENS stimulate the skin's beta (large) fibers, and use C-fiber to transmit signals faster to suppress pain signals from small fibers. Researchers have pointed out that low-frequency TENS can produce longer-lasting effects, and longer-term effects can be obtained with very low-frequency electrical stimulation. Compared to high frequency TENS, low frequency TENS can also significantly increase local skin blood flow. Cramp et al. [72] designed a double-blind experiment to determine the effect of TENS intensity on local and distal skin blood flow. The results show that the use of low-frequency (4 Hz, 200 μs) electrical stimulation for 15 min can significantly increase local skin blood flow. The effect on skin blood flow depends on whether it leads to muscle activity or whether it is above the exercise threshold. Siqueira et al. [89] show that low-frequency TENS had shown good efficacy in clinical trials for the treatment of throat pain, and the intensity of throat pain in the TENS treatment group was significantly reduced. At the same time, researchers have pointed out that surface electrodes are more effective than subcutaneous electrodes, and low frequency constant current stimulation is more effective in analgesia than high frequency. EMS therapy is associated with fewer side effects than other therapies and has mild symptoms, such as rashes associated with high-frequency TENS therapy, and electrode site burns encountered during low-frequency TENS therapy sense. TENS can also be used as an adjuvant therapy after surgery. Hush et al. [90] show that after thoracotomy in the intensive care unit (ICU), TENS adjuvant therapy can relieve pain in the first 12 h. As one of the difficult to treat diseases, eye pain usually cannot be relieved because of the difficulty of treating the ocular surface. In a recent TNES assisted long-term treatment of eye pain, the TENS experimental group reduced pain by 27.4% compared to the control group, which can assist in the treatment of eye pain [91]. Patellofemoral pain syndrome (PFPS) can cause muscle weakness and knee pain, which is a common disease among military personnel. The active exercises tested by Talbot et al. [92] are combined with TENS and neuromuscular electrical stimulation (NMES), compared with active exercise alone, it significantly increases the muscle strength.
Fig. 13.
a Bar graphs represent the analgesia produced by TENS and morphine in the tail flick test for animals that are intact and those that were spinalized. Fifty-hertz electrical stimulation produced an increase in the tail flick latency similar to that of systemic morphine. Spinal transection reduced the amount of inhibition by electrical stimulation or morphine by approximately 50% [82]. b Bar graphs represent the analgesia produced by 50 Hz electrical stimulation or morphine in the tail flick test in intact animals. Animals pretreated with PCPA to deplete the neurotransmitter serotonin (5-HT) showed a significant reduction in the amount of analgesia produced by either electrical stimulation or morphine. Control animals did not receive electrical stimulation or morphine but were still spinalized or pretreated with PCPA [82]. c Depiction of the wave-forms commonly found in most present-day TENS devices [87]
At present, there are still many experimental methods on TENS, among which blind experiments accepted by the medical community are difficult to carry out in TENS therapy. The safety and effectiveness of chronic pain had enough evidence to support, but the standardization and uniformity of the report needed to be improved [93]. High-frequency TENS will make a sound in the stimulation area. The subject feels low intensity and the experimenter can judge by sound. Low-frequency TENS and strong sudden TENS (similar to acupuncture) tend to have higher intensity, which will cause the subject to contract muscles. These problems make it difficult to carry out double-blind experiments with TENS.
The TENS therapeutic device are widely used in the medical field, but the analgesic effect of TENS is still not enough. On the one hand, this is because TENS are not easy to carry out blind experiments and the subject's pain evaluation system is not perfect; on the other hand, the analgesia of TENS in the research, for the understanding of the principle is not enough, and it is analyzed from the perspective of experiments. At the same time, regarding the analysis of the efficacy of TENS, the researchers pointed out that the amount of TENS used will cause sensory differences, and patients who use TENS for a long time and patients who receive short-term treatment have an impact on the experimental conclusions [94]. The status of TENS therapy in the modern medicine is slightly better than skin wound healing therapy. This is because the use of parameters such as current types (low frequency, high frequency) and pulses width in TENS therapy is very clear, but the research methods for these two are not strict enough. Strictly standardizing the experimental process and clear parameter selection is the focus of the next.
Postoperative electrical stimulation of the muscle
Muscle contraction is the response of resting the muscle tissue to neurophysiological stimuli. The activation of muscle fibers to generate tension is triggered by the organism's own electrical stimulation (action potential), which is transmitted in the nervous system. Skeletal muscle is innervated by peripheral motor nerves, and these motor nerve tissues are functional units called motor units. The motor unit is innervated by a single motor neuron and all muscle fibers of its axon branch. As shown in Fig. 14a, by applying current skin to skeletal muscle, it can cause normal involuntary skeletal muscle contraction. Each NMES circuit requires two electrodes, which provides an interface between the electrical stimulator and the patient through a coupling medium that enhances skin conductivity. The current from the stimulator enters at one electrode and flows back to the stimulator at the other electrode. The working schematic is shown in Fig. 14b. The flow of electrons from the stimulator is converted into a flow of ions in the motor nerve. If the stimulation is sufficient to depolarize the nerve to its threshold, an action potential is generated. Figure 14c shows some common waveform produced by commercial electrical stimulators, including various single-phase (onlone polarity), two-phase (alternating polarity), pulsed (isolated pulses between which no current flows) and pulse waveform (current burst) [95]. Action potentials are borne by peripheral motor nerves (motor neurons), and axon branches control all skeletal muscle fibers. When these motor units are triggered by the external current, this is called EMS. EMS is a technique widely used in sports training and clinical medicine. In principle, it generates muscle contraction by activating motor axons around the electrical stimulation location. In addition, EMS can also send sensory and pain signals to the motor cortex, sensory cortex and associated cortex through the activation of surrounding sensory axons, and also increase the excitability of the cortex. The symptom of muscle loss has now progressed to clinical symptoms, defined as low muscle mass, and is regarded as impaired physical function. The reason for muscle mass decline is not only its own morphological changes, but also neurological factors. In reduced-mass muscles, the diameter of type II fibers (the main source of muscle strength) decreases, while changes in the expression level of the associated myosin heavy chain are at a microscopic level of degradation. There are many animal experiments and clinical experiments to explore the role of EMS.
Fig. 14.
a Diagram of a motor unit, which is composed of the motor nerve, its branches, and all of the muscle fibers it innervates [95]. b Illustration of a NMES circuit [95]. c Examples of electrical waveforms commonly used in NMES. A, bursted biphasic (“Russian”) current; B, pulsed symmetric biphasic current; C, pulsed asymmetric biphasic current [95]. d Capillaries visualized byalkaline phosphatase in rabbit extensor digitorum [12]. e Transverse sections of rabbit EDL muscles stained for succinic dehydrogenase: (a) control and (b) stimulated for 14 days at 5 Hz continuously; (c) control and (d) stimulated for 21 days at 10 Hz intermittently [12]
EMS electrical stimulation is a treatment that acts on the skin surface clinically, and animal experiments provide more possibilities. It allows researchers to apply electrical stimulation for a long time and high intensity, and can be sectioned at any time. Micro-current stimulation at a certain frequency can promote the growth of capillaries, which is mentioned in various current therapies. The effects of electrical stimulation on capillaries and skeletal muscle have been studied in various animals, especially rabbits. Alkaline phosphatase was used to stain the long finger muscles of rabbits, as shown in Fig. 14d, the capillaries observed in the control (left) and 10 Hz stimulation (28 consecutive days). With the extension of the stimulation time, 10 Hz stimulation of fast muscles will gradually increase the capillary density, which is already obvious 4 days after the start of stimulation, and finally after 28 days of stimulation, the distribution of capillaries becomes slow Muscle capillaries are distributed. Figure 14e shows a cross-section of rabbit extensor digitorum longus (EDL) muscle stained with succinate dehydrogenase, group a is the control group, b is the continuous stimulation at 5 Hz for 14 days, group c is the control group, and d is stimulated intermittently at 10 Hz for 21 days. The results show that continuous stimulation at 5 Hz for 14 days can increase enzyme activity, which is equivalent to SDH activity produced by continuous 10 Hz stimulation (intermittent). There was almost no change in the muscles that was stimulated intermittently for 21 days at a frequency of 10 Hz (1440 stimuli per hour) [12]. Electrical stimulation results in increased blood flow in skeletal muscle and also increases capillary density [96]. The relevant explanation for this is that the increase in blood flow induced by stimulation can induce capillary growth.
Other animal experiments have shown that long-term electrical stimulation causes changes in muscle contraction characteristics. Salmons and Vrbova's experiments show that continuous electrical stimulation through implantation can slow down the contraction and relaxation of the tibialis anterior muscle and toe long extensor, and intermittent stimulation can significantly increase muscle tension [97]. In addition to changing the contractile properties of muscles, Pette et al. [98] found that after two weeks of continuous implantation stimulation, compared with the control group, the activity of muscle oxidative metabolism-related enzymes in the rabbit stimulation group increased, with the increase of myoglobin content. In addition, researchers through histochemical staining of succinate dehydrogenase in the cross section of rabbit tibialis anterior muscle found that after 28 days of stimulation, the succinate dehydrogenase activity of the experimental group was higher, which represents the aerobic of the overall tissue increased metabolic levels, and no white fibers were found in the experimental group, which indicates that long-term electrical stimulation will lead to the conversion of white muscle fibers to red muscle fibers. In addition, In the treatment of neurological diseases, electrical stimulation has proved to help the formation and growth of nerve synapses in petri dishes, which will provide new ideas for the treatment of neurodevelopmental diseases [99]. Animal experiments provided more rigorous and standardized experimental results, and provided a reference for further clinical experiments.
EMS postoperative rehabilitation
Postoperative rehabilitation is a very important topic in the medical field. Patients need bed rest after general surgery. This is a long process, especially for serious diseases such as sepsis and multiple organ failure. During this period, the patient's muscles will continue to decrease. For example, after the knee surgery and fixation, muscle weight loss is inevitable. At present, weight-bearing training is widely used in post-fracture rehabilitation training, but the specific load is still not scientifically quantified [100]. EMS became a very effective method of rehabilitation.
Willand et al. [101] used two groups of rats to recover immediately after the nerve was cut off, and then performed 600 muscle stimulation contractions (1 h/day) five days a week. The results show that EMS therapy can enhance the innervation ability of nerves, and it can be used at the earliest after injury. It was detected after two weeks that the intramuscular level of cellular neurotrophic factor mRNA in muscles of the EMS group was significantly increased compared to the control group, and two weeks after nerve repair, after the longitudinal sections of rat tibial nerves were observed under a microscope, nerve fibers containing green fluorescent protein (GFP) were seen, and compared with the control group (A, C), tibia received EMS therapy produced more fiber, as B and D in Fig. 15a.
Fig. 15.
a Longitudinal nerve sections. Representative sections were
taken from transected and surgically repaired tibial nerves from Thy1-GFP transgenic rats in which the gastrocnemius muscle was sham-stimulated (A) or subjected to daily EMS (B). The hashed line is the suture line. Higher magnification views of the boxed areas within the distal nerve stump are shown in (C) and (D) [101]. b Isoforce GT-330 (OG Giken), which was used to measure maximum muscle strength of both legs during CKC exercise (left). Cross-sectional areas of extensor and flexor muscles of injured and intact leg were measured in horizontal CT image (right) [102]. c A user is absorbed in his reading, not noticing the lamppost. Actuated navigation automatically steers him around the obstacle [108]. d Placement of the EMS electrodes and wearable control unit [109]. e Anatomic figure of the left forearm. Two electrode sleeves are placed on the skin. The electrodes cover several muscles. There are three active EMS channels: 3 + 4 → 12, 7 → 15, and 8 → 16. The blue electrodes are deactivated [110]
In essence, EMS can promote changes in the composition of muscle fibers, promote glucose absorption, increase oxidase activity, and also stimulate the growth of type II fibers, which is dominated by the muscle strength, because of the axon size of type II fibers, for it is larger than I-type fiber and has lower resistance, but it is also more prone to fatigue [95]. Research group, for patients with plaster cast on the legs after the ligament surgery, used a 30 Hz quadriceps electrical stimulation for the experimental group, 40 min each time, 3 times a week, and combined with the patient's own active contraction of the muscles. Results In the control group, the isometric stretching the muscle strength of knees was significantly reduced, and the relative fiber area and related enzyme activity of type I fibers were reduced, while the experimental group remained normal. Takahashi et al. [102] studied the rehabilitation of lower limb muscle strength after EMS rehabilitation therapy and traditional rehabilitation therapy after anterior cruciate ligament (ACL) surgery, using low-frequency electrical stimulation (0.5–3 Hz, 3 times a week for 3 months), and during this period Isoforce GT-330 (OG Giken) was used to measure the strength of the legs to assess muscle mass Fig. 15b (left), CT images of the muscles in the experimental group showed a significant improvement in quality, and the cross-sectional area of the extensor muscle increased more by 3 months, as shown in Fig. 15b (right) [22, 102].
Many controlled clinical trials have shown that in some specific diseases, EMS can significantly improve the muscle strength and increase exercise capacity. The researchers confirmed the role of EMS in promoting microcirculation in critical patients. After 45 min of EMS treatment of the lower limbs, the researchers clearly observed improvements in the intermuscular microcirculation using infrared spectroscopy [103]. The ventilator can assist in the treatment of chronic obstructive pulmonary disease (COPD), but it also takes a certain risk. The auxiliary treatment of COPD by muscle electrical stimulation therapy will greatly reduce the risk [104]. In addition, EMS therapy can prevent some diseases. Venous thrombosis is a disease of venous reflux disorders in the lower limbs. It refers to the abnormal clotting of blood in deep veins. Its pathogenesis depends on factors related to the composition of the vessel wall. At present, the medical community believes that the emergence of congestion is a major factor in the development of deep vein thrombosis. From the earliest walking-therapy to the leg compression therapy to EMS therapy, this field is constantly developing. In some cardiovascular diseases blood pressure is very important. Electrical stimulation can activate the baroreflex system and allow the brain to lower blood pressure through various mechanisms. This therapy has very few side effects [105]. In clinical practice, patients with atherosclerosis are prone to develop other severe illnesses, and EMS therapy can regulate blood vessel flow [106]. Sometimes EMS can also affect the whole body. Postprandial hyperglycemia is a major cause of death in patients with type 2 diabetes, so postprandial glucose control is considered to be an effective method, to reduce cardiovascular disease and myocardial infarction, especially it is post-prandial exercise. The contraction of a certain number of muscles can enhance the absorption of glucose by skeletal muscles. Hamada et al. [107] studied the effects of EMS electrical stimulation on postprandial hyperglycemia in patients with type 2 diabetes, and performed 30-min EMS for 30 min after a meal. Compared with rest after a meal, the post-prandial blood glucose level in the experimental group was significantly reduced (p < 0.05) And the concentration of C-phil, also decreased significantly (p < 0.01). And another set of experiments showed that the use of a single EMS (one-way square wave pulse, 20 min, 4 Hz frequency, duration 0.2 ms) can improve systemic glucose uptake, and can last more than 90 min after the EMS is stopped.
Expanded application of EMS
The use of electrical muscle stimulation has been greatly expanded in recent years. It has been used more and more widely from severe patients to recover from the light surgery to healthy people. There are studies using 4 Hz EMS to increase the quadriceps strength of sedentary adults, and some, such as Pfeiffer et al. [108] use electric stimulation of muscles (120 Hz, 100 μs, pulse voltage, 13–32 V, 39–66 mA) to volunteer whose walking control can bypass obstacles and navigate, as shown in Fig. 15c, volunteers avoid obstacles automatically.
In addition to navigation, the auxiliary applications of EMS equipment are more attractive. Wiehr et al. [109] designed a wearable running device, as shown in Fig. 15d it includes a force sensing resistor in the insole and an EMS electrode in the lower leg, in this way, the impact load can be reduced by hitting the middle foot or the front foot. Although this study is not practical enough, it provides an application idea for EMS. On the other hand, Duente et al. [110] in Germany conducted a more detailed study. They used 20 controllable channels to control muscles, as shown in Fig. 15e, each muscle has an independent electrode corresponding, and the electrical stimulation can be accurately control the finger, so that the experimenter can extend each finger individually, and can also extend multiple fingers at the same time as shown in Fig. 16a. Besides, Hao et al. [111] show that electrical stimulation can also induce tactile sensation, thereby providing sensory feedback treatment for amputees. Most of the above studies are unilateral control, and the current interactive EMS research has gradually attracted attention. Lopes et al. [13] designed a feedback system composed of the impact of the solenoid on the skin and the EMS stimulation, as shown in Fig. 16g. The surface of the additional solenoid on the back of the hand has a tip, and the user can be attacked and cooperate with VR to create a better gaming experience, in addition, when the user is attacked by a virtual character, apart from feeling the impact, the EMS device will stimulate the biceps to contract and make it feel resistance, as shown in Fig. 16f (right a), when the user attacks the opponent, the user will stop his hand in the air, causing a hit feeling as shown in Fig. 16f (right b) [13]. In addition to complex and real large-scale systems, there are also researchers dedicated to the research of portable game feedback devices. Lopes et al. [112] have developed portable equipment for mobile devices, as shown in Fig. 16e, which stimulated by electrodes connected to the arm, the muscles provide force feedback, simulating resistance to the aircraft in the game. At present, the research of EMS as an interactive auxiliary device has entered the right track, and it will be more widely used in the future.
Fig. 16.
a Two sleeves worn at the forearm. The upper sleeve has 22 electrodes, the lower sleeve has 18 electrodes [110]. b Example of overriding the conventional behavior: (a) As participants expected this door to open to the conventional side, they turned the handle right, (b) Affordance + + induced a left turning motion instead [113]. c Changing behavior according to the object’s state: (a) this cold cup affords grasping, (b) when hot it repels the user’s hand, but (c) its handle continues to afford grasping [113]. d An audience member guiding the artist’s brushstrokes by pressing a button on a connected tablet to activate one of the sets of EMS pads worn by the artist [114]. e Muscle-Propelled Force Feedback allows for miniaturized force feedback devices by electrically stimulating the user’s muscles, instead of using mechanical actuators [112]. f Impacto allows users to feel the impact of blocking the avatar’s hit by thrusting the user’s arm backwards by operating the user’s biceps (a). The same impacto unit allows simulating the sensation of attacking. In both cases the impacto unit activates the user’s biceps. This time, this causes the user’s hand to stop in mid-air, as if it had hit the opponent (b) [13]. g Additional solenoid unit mounted to the back of the hand featuring a surface tip allows the user to attack using jabs and uppercuts [13]
Other interactive EMS studies show even greater potential. Pedro Lopes et al. [113] assisted the control by detecting the actions of the EMS device user, which is called adaptation++, for example, as shown in Fig. 16c, when the experimenter held the cup of the cold cup, it is not controlling, and later will extend the palm to prevent the experimenter from holding the cup body when it is hot, only provides grip when the hand is close to the cup handle. In addition, adaptation++ can also correct the experimenter's movements, such as when the experimenter pulls the door handle from left to right (in the opposite direction), adaptation++ will give a left-turning stimulus as shown in Fig. 16b (right a), and when someone is in the door, the system will repel the hand near the door handle, and actively curl the finger into a fist, suggesting that the user knocking the door by hand Fig. 16b (right b). This is a human–computer interaction. Researchers from Lapland Rovaniemi Finland University [114] have conducted a more interesting human–machine–human experiment, for the EMS module is placed on the artist's arm, and the audience can control the electrodes through the tablet to influence the artist's pen to complete the painting, as shown in Fig. 16d.
The EMS muscle electrical stimulation technology has developed more and more mature. Although the comparison and evaluation between studies will be difficult to carry out due to differences in experimental equipment, electrode specifications, etc., its theory and efficacy are very clear and widely used in rehabilitation. At present, most of the research on EMS focuses on low-frequency and intermediate-frequency currents. This is because high-frequency currents are likely to cause muscle damage, and the effects of low-frequency currents are longer-lasting and have greater effects on muscle strength.
Perspective and summary
Micro-current stimulation, as a relatively safe and non-invasive noninvasive therapy, has been applied to many fields of medicine since its discovery, and has been greatly developed, allowing researchers to see new possibilities. Transdermal drug delivery, wound healing, analgesia and postoperative muscle rehabilitation has gradually expanded to non-medical fields in recent years. In current drug delivery therapy, the principle and efficacy of iontophoresis had been widely recognized, and the effect is very good. At present, the focus of research is turning to its safety and drug toxicity. The increase is also the introduction of micro-current, the electroporation therapy can achieve a larger dose of drug, but at the same time, it will cause more damage, which is not conducive to popularization. Another large area of micro-current application is to promote wound. Electric field has an extremely obvious effect on regulating and promoting healing, and can also increase the expression of neuroendocrine secretions. In recent years, the focus of attention has been on the application of biomimetic materials, especially conductive polymers. Hydrogels are widely used in this field due to their good biocompatibility. In the field of wound healing stimulated by micro-currents, another area that needs to be developed is the study of the mechanism of chronic wounds. At present, there are messy researches in this area and no experimental specification. As a safe non-invasive analgesic therapy, TENS can produce effects similar to morphine and can alleviate many refractory diseases. TENS therapeutic apparatus has been widely used in the medical field, but its analgesic effect and corresponding, it is difficult to communicate and describe. On the one hand, the evaluation system of pain is not yet complete, and the other is that the principle of analgesia is not clear. EMS is the safest electric current therapy compared to the previous ones. From its earliest use in postoperative muscle rehabilitation to the prevention of venous thrombosis and other diseases, its application field has been greatly expanded and can be used for sensory feedback in healthy subjects, as well as the control and feedback in the game, will be further miniaturized and flexible in the future and applied to the field of wear.
Footnotes
Executive Editor: Xi-Qiao Feng
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jianru Wang, Email: wjr104zah@sina.com.
Liang Guo, Email: guoliang@imech.ac.cn.
References
- 1.Yamamoto T, Yamamoto Y. Electrical-properties of epidermal stratum-corneum. Medi. Biol. Eng. 1976;14:151–158. doi: 10.1007/BF02478741. [DOI] [PubMed] [Google Scholar]
- 2.Plutchik R, Hirsch HR. Skin impedance and phase angle as a function of frequency and current. Science. 1963;141:927–928. doi: 10.1126/science.141.3584.927. [DOI] [PubMed] [Google Scholar]
- 3.Chen LLH, Chien YW. Transdermal iontophoretic permeation of luteinizing hormone releasing hormone: Characterization of electric parameters. J. Control. Release. 1996;40:187–198. doi: 10.1016/0168-3659(95)00181-6. [DOI] [Google Scholar]
- 4.Marro D, Guy RH, Delgado-Charro MB. Characterization of the iontophoretic permselectivity properties of human and pig skin. J. Control. Release. 2001;70:213–217. doi: 10.1016/S0168-3659(00)00350-3. [DOI] [PubMed] [Google Scholar]
- 5.Bhatia KS, Singh J. Mechanism of transport enhancement of lhrh through porcine epidermis by terpenes and iontophoresis: permeability and lipid extraction studies. Pharm. Res. 1998;15:1857–1862. doi: 10.1023/A:1011906022903. [DOI] [PubMed] [Google Scholar]
- 6.Cazares-Delgadillo J, Planard-Luong L, Gregoire S, et al. Investigation of different iontophoretic currents profiles for short-term applications in cosmetics. Pharmaceutics. 2018;10:266. doi: 10.3390/pharmaceutics10040266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nuccitelli R, Pliquett U, Chen XH, et al. Nanosecond pulsed electric fields cause melanomas to self-destruct. Biochem. Biophys. Res. Commun. 2006;343:351–360. doi: 10.1016/j.bbrc.2006.02.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Prausnitz MR, Bose VG, Langer R, et al. Electroporation of mammalian skin - a mechanism to enhance transdermal drug-delivery. Proc. Natl. Acad. Sci. USA. 1993;90:10504–10508. doi: 10.1073/pnas.90.22.10504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chang SL, Hofmann GA, Zhang L, et al. The effect of electroporation on iontophoretic transdermal delivery of calcium regulating hormones. J. Control. Release. 2000;66:127–133. doi: 10.1016/S0168-3659(99)00262-X. [DOI] [PubMed] [Google Scholar]
- 10.Song B, Gu Y, Pu J, et al. Application of direct current electric fields to cells and tissues in vitro and modulation of wound electric field in vivo. Nat. Protoc. 2007;2:1479–1489. doi: 10.1038/nprot.2007.205. [DOI] [PubMed] [Google Scholar]
- 11.Rushton DN. Electrical stimulation in the treatment of pain. Disabil. Rehabil. 2002;24:407–415. doi: 10.1080/09638280110108832. [DOI] [PubMed] [Google Scholar]
- 12.Brown MD, Cotter MA, Hudlicka O, et al. Effects of different patterns of muscle-activity on capillary density, mechanical-properties and structure of slow and fast rabbit muscles. Pflug. Archiv-Eur J Physiol. 1976;361:241–250. doi: 10.1007/BF00587288. [DOI] [PubMed] [Google Scholar]
- 13.Lopes, P., Ion, A., Baudisch, P.: Impacto: Simulating physical impact by combining tactile stimulation with electrical muscle stimulation. In: 28th Annual ACM Symposium on User Interface Software and Technology. pp.11–19 (2015)
- 14.Kanitakis J. Anatomy, histology and immunohistochemistry of normal human skin. Eur. J. Dermato. 2002;12:390–400. [PubMed] [Google Scholar]
- 15.Sharma N, Agarwal G, Rana A, et al. A review: Transdermal drug delivery system: a tool for novel drug delivery system. Int. J. Drug Dev. Res. 2011;3:70–84. [Google Scholar]
- 16.Zaidi Z, Lanigan SW. Dermatology in Cslinical Practice. London: Springer; 2010. pp. 1–15. [Google Scholar]
- 17.Funderburgh ML, Du YQ, Mann MM et al. (2005) Pax expression identifies progenitor cells for corneal keratocytes. Faseb J. 19, 1371–1373 [DOI] [PMC free article] [PubMed]
- 18.Chen LW, Tredget EE, Wu PYG, et al. Paracrine factors of mesenchymal stem cells recruit macrophages and endothelial lineage cells and enhance wound healing. PLoS ONE. 2008;3:12. doi: 10.1371/journal.pone.0001886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Badillo AT, Zhang L, Liechty KW. Stromal progenitor cells promote leukocyte migration through production of stromal-derived growth factor 1 alpha: a potential mechanism for stromal progenitor cell-mediated enhancement of cellular recruitment to wounds. J. Pediatr. Surg. 2008;43:1128–1133. doi: 10.1016/j.jpedsurg.2008.02.043. [DOI] [PubMed] [Google Scholar]
- 20.Yuan Q, Gurish MF, Friend DS, et al. Cutting edge: Generation of a novel stem cell factor-dependent mast cell progenitor. J. Immunol. 1998;161:5143–5146. doi: 10.4049/jimmunol.161.10.5143. [DOI] [PubMed] [Google Scholar]
- 21.Lau K, Paus R, Tiede S, et al. Exploring the role of stem cells in cutaneous wound healing. Exp. Dermatol. 2009;18:921–933. doi: 10.1111/j.1600-0625.2009.00942.x. [DOI] [PubMed] [Google Scholar]
- 22.Singh D, Pradhan M, Nag M, et al. Vesicular system: Versatile carrier for transdermal delivery of bioactives. Artif.Cells Nanomed. Biotechnol. 2015;43:282–290. doi: 10.3109/21691401.2014.883401. [DOI] [PubMed] [Google Scholar]
- 23.Tian F, Wang H, Li H, et al. Molecular simulation of diffusion of rigidity-tuned nanoparticles in biological hydrogels. Acta. Mech. Sin. 2019;35:376–383. doi: 10.1007/s10409-019-00858-x. [DOI] [Google Scholar]
- 24.Ale I, Lachapelle J-M, Maibach HI. Skin tolerability associated with transdermal drug delivery systems: an overview. Adv. Therapy. 2009;26:920–935. doi: 10.1007/s12325-009-0075-9. [DOI] [PubMed] [Google Scholar]
- 25.Wokovich AM, Prodduturi S, Doub WH, et al. Transdermal drug delivery system (tdds) adhesion as a critical safety, efficacy and quality attribute. Eur. J. Pharm. Biopharm. 2006;64:1–8. doi: 10.1016/j.ejpb.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 26.Vanbever R, Lecouturier N, Preat V. Transdermal delivery of metoprolol by electroporation. Pharm. Res. 1994;11:1657–1662. doi: 10.1023/A:1018930425591. [DOI] [PubMed] [Google Scholar]
- 27.Kalia YN, Naik A, Garrison J, et al. Iontophoretic drug delivery. Adv. Drug Deliv. Rev. 2004;56:619–658. doi: 10.1016/j.addr.2003.10.026. [DOI] [PubMed] [Google Scholar]
- 28.Pacini S, Punzi T, Gulisano M, et al. Transdermal delivery of heparin using pulsed current iontophoresis. Pharm. Res. 2006;23:114–120. doi: 10.1007/s11095-005-8923-z. [DOI] [PubMed] [Google Scholar]
- 29.Bellantone NH, Rim S, Francoeur ML, et al. Enhanced percutaneous-absorption via iontophoresis. 1. Evaluation of an invitro system and transport of model compounds. Int. J. Pharm. 1986;30:63–72. doi: 10.1016/0378-5173(86)90136-5. [DOI] [Google Scholar]
- 30.Mutalik S, Udupa N, Kumar S, et al. Glipizide matrix transdermal systems for diabetes mellitus: Preparation, in vitro and preclinical studies. Life Sci. 2006;79:1568–1577. doi: 10.1016/j.lfs.2006.05.002. [DOI] [PubMed] [Google Scholar]
- 31.Hirvonen J, Hueber F, Guy RH. Current profile regulates iontophoretic delivery of amino acids across the skin. J. Control. Release. 1995;37:239–249. doi: 10.1016/0168-3659(95)00081-X. [DOI] [Google Scholar]
- 32.Ledger PW. Skin biological issues in electrically enhanced transdermal delivery. Adv. Drug Deliv. Rev. 1992;9:289–307. doi: 10.1016/0169-409X(92)90027-N. [DOI] [Google Scholar]
- 33.Okabe K, Yamaguchi H, Kawai Y. New iontophoretic transdermal administration of beta-blocker metoprolol. J. Control. Release. 1986;4:79–85. doi: 10.1016/0168-3659(86)90043-X. [DOI] [Google Scholar]
- 34.Wearley L, Liu JC, Chien YW. Iontophoresis-facilitated transdermal delivery of verapamil. 2. Factors affecting the reversibility of skin permeability. J. Control. Release. 1989;9:231–242. doi: 10.1016/0168-3659(89)90091-6. [DOI] [Google Scholar]
- 35.Puiggali-Jou A, del Valle LJ, Aleman C. Encapsulation and storage of therapeutic fibrin-homing peptides using conducting polymer nanoparticles for programmed release by electrical stimulation. ACS Biomater. Sci. Eng. 2020;6:2135–2145. doi: 10.1021/acsbiomaterials.9b01794. [DOI] [PubMed] [Google Scholar]
- 36.Wang YP, Thakur R, Fan QX, et al. Transdermal iontophoresis: Combination strategies to improve transdermal iontophoretic drug delivery. Eur. J. Pharm. Biopharm. 2005;60:179–191. doi: 10.1016/j.ejpb.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 37.Nair A, Vyas H, Shah J, et al. Effect of permeation enhancers on the iontophoretic transport of metoprolol tartrate and the drug retention in skin. Drug Deliv. 2011;18:19–25. doi: 10.3109/10717544.2010.509361. [DOI] [PubMed] [Google Scholar]
- 38.Donohoe M, Jennings B, Balasubramaniam S. Deep brain drug-delivery control using vagus nerve communications. Comput. Netw. 2020;171:107137. doi: 10.1016/j.comnet.2020.107137. [DOI] [Google Scholar]
- 39.Oh B, George P. Conductive polymers to modulate the post-stroke neural environment. Brain Res. Bull. 2019;148:10–17. doi: 10.1016/j.brainresbull.2019.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang A, Hu M, Zhou L, et al. Self-powered well-aligned p(vdf-trfe) piezoelectric nanofiber nanogenerator for modulating an exact electrical stimulation and enhancing the proliferation of preosteoblasts. Nanomaterials. 2019;9:349. doi: 10.3390/nano9030349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sebastiani P, Nicoli S, Santi P. Effect of lactic acid and iontophoresis on drug permeation across rabbit ear skin. Int. J. Pharm. 2005;292:119–126. doi: 10.1016/j.ijpharm.2004.11.038. [DOI] [PubMed] [Google Scholar]
- 42.Bottoni CR. Editorial commentary: every cloud has a silver lining. But silver may not be the currency of choice: the bioelectric silver-zinc dressing requires additional investigation. Arthrosc. J. Arthrosc. Relat. Surg. 2018;34:2892–2893. doi: 10.1016/j.arthro.2018.07.039. [DOI] [PubMed] [Google Scholar]
- 43.Mousavi ST, Harper GR, Municoy S, et al. Electroactive silk fibroin films for electrochemically enhanced delivery of drugs. Macromol. Mater. Eng. 2020;305:2000130. doi: 10.1002/mame.202000130. [DOI] [Google Scholar]
- 44.Malinovskaja-Gomez K, Labouta HI, Schneider M, et al. Transdermal iontophoresis of flufenamic acid loaded plga nanoparticles. Eur. J. Pharm. Sci. 2016;89:154–162. doi: 10.1016/j.ejps.2016.04.034. [DOI] [PubMed] [Google Scholar]
- 45.Kolli CS, Xiao J, Parsons DL, et al. Microneedle assisted iontophoretic transdermal delivery of prochlorperazine edisylate. Drug Dev. Ind. Pharm. 2012;38:571–576. doi: 10.3109/03639045.2011.617753. [DOI] [PubMed] [Google Scholar]
- 46.Zhao S, Mehta AS, Zhao M. Biomedical applications of electrical stimulation. Cell. Mol. Life Sci. 2020;77:2681–2699. doi: 10.1007/s00018-019-03446-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pliquett UF, Martin GT, Weaver JC. Kinetics of the temperature rise within human stratum corneum during electroporation and pulsed high-voltage iontophoresis. Bioelectrochemistry. 2002;57:65–72. doi: 10.1016/S1567-5394(01)00177-3. [DOI] [PubMed] [Google Scholar]
- 48.Weaver JC, Vaughan TE, Chizmadzhev Y. Theory of electrical creation of aqueous pathways across skin transport barriers. Adv. Drug Deliv. Rev. 1999;35:21–39. doi: 10.1016/S0169-409X(98)00061-1. [DOI] [PubMed] [Google Scholar]
- 49.Sowers AE, Lieber MR. Electropore diameters, lifetimes, numbers, and locations in individual erythrocyte-ghosts. FEBS Lett. 1986;205:179–184. doi: 10.1016/0014-5793(86)80893-6. [DOI] [PubMed] [Google Scholar]
- 50.Jacobsen J. Buccal iontophoretic delivery of atenolol center dot hcl employing a new in vitro three-chamber permeation cell. J. Control. Release. 2001;70:83–95. doi: 10.1016/S0168-3659(00)00328-X. [DOI] [PubMed] [Google Scholar]
- 51.Wallace BM, Lasker JS. Drug delivery - stand and deliver - getting peptide drugs into the body. Science. 1993;260:912–913. doi: 10.1126/science.8493526. [DOI] [PubMed] [Google Scholar]
- 52.Hui SW. Low voltage electroporation of the skin, or is it iontophoresis? Biophys. J. 1998;74:679–680. doi: 10.1016/S0006-3495(98)73993-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Reinke JM, Sorg H. Wound repair and regeneration. Eur. Surg. Res. 2012;49:35–43. doi: 10.1159/000339613. [DOI] [PubMed] [Google Scholar]
- 54.Sun Y, Chen G, Lv Y. Effects of hypoxia on the biological behavior of mscs seeded in demineralized bone scaffolds with different stiffness. Acta. Mech. Sin. 2019;35:309–320. doi: 10.1007/s10409-019-00845-2. [DOI] [Google Scholar]
- 55.Wang M, Liu S, Li F. Imaging oxygen microenvironment in hydrogel microwell array. Acta. Mech. Sin. 2019;35:321–328. doi: 10.1007/s10409-018-0832-6. [DOI] [Google Scholar]
- 56.Xia Y, Pfeifer CR, Discher DE. Nuclear mechanics duringand after constricted migration. Acta. Mech. Sin. 2019;35:299–308. doi: 10.1007/s10409-018-00836-9. [DOI] [Google Scholar]
- 57.Chiang M, Cragoe EJ, Vanable JW. Intrinsic electric-fields promote epithelialization of wounds in the newt, notophthalmus-viridescens. Dev. Biol.. 1991;146:377–385. doi: 10.1016/0012-1606(91)90239-Y. [DOI] [PubMed] [Google Scholar]
- 58.Long Y, Wei H, Li J, et al. Effective wound healing enabled by discrete alternative electric fields from wearable nanogenerators. ACS Nano. 2018;12:12533–12540. doi: 10.1021/acsnano.8b07038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhao M, Song B, Pu J, et al. Electrical signals control wound healing through phosphatidylinositol-3-oh kinase-gamma and pten. Nature. 2006;442:457–460. doi: 10.1038/nature04925. [DOI] [PubMed] [Google Scholar]
- 60.Zhao M. Electrical fields in wound healing-an overriding signal that directs cell migration. Semin. Cell Dev. Biol. 2009;20:674–682. doi: 10.1016/j.semcdb.2008.12.009. [DOI] [PubMed] [Google Scholar]
- 61.He S, Li X, Ji B. Mechanical force drives the polarization and orientation of cells. Acta. Mech. Sin. 2019;35:275–288. doi: 10.1007/s10409-019-00864-z. [DOI] [Google Scholar]
- 62.Chen J, Wang N. Tissue cell differentiation and multicellular evolution via cytoskeletal stiffening in mechanically stressed microenvironments. Acta. Mech. Sin. 2019;35:270–274. doi: 10.1007/s10409-018-0814-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Torkaman G. Electrical stimulation of wound healing: a review of animal experimental evidence. Adv. Wound Care. 2014;3:202. doi: 10.1089/wound.2012.0409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rouabhia M, Park H, Meng S, et al. Electrical stimulation promotes wound healing by enhancing dermal fibroblast activity and promoting myofibroblast transdifferentiation. PLoS ONE. 2013;8:e71660. doi: 10.1371/journal.pone.0071660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sebastian A, Volk SW, Halai P, et al. Enhanced neurogenic biomarker expression and reinnervation in human acute skin wounds treated by electrical stimulation. J. Invest. Dermato. 2017;137:737–747. doi: 10.1016/j.jid.2016.09.038. [DOI] [PubMed] [Google Scholar]
- 66.Lallyett C, Yeung CYC, Nielson RH, et al. Changes in s100 proteins identified in healthy skin following electrical stimulation: relevance for wound healing. Adv. Skin Wound Care. 2018;31:322–327. doi: 10.1097/01.ASW.0000533722.06780.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Werdin F, Tenenhaus M, Rennekampff H-O. Chronic wound care. Lancet. 2008;372:1860–1862. doi: 10.1016/S0140-6736(08)61793-6. [DOI] [PubMed] [Google Scholar]
- 68.Nair HKR. Microcurrent as an adjunct therapy to accelerate chronic wound healing and reduce patient pain. J Wound Care. 2018;27:296–306. doi: 10.12968/jowc.2018.27.5.296. [DOI] [PubMed] [Google Scholar]
- 69.Ramadan A, Elsaidy M, Zyada R. Effect of low-intensity direct current on the healing of chronic wounds: a literature review. J. Wound Care. 2008;17:292–296. doi: 10.12968/jowc.2008.17.7.30520. [DOI] [PubMed] [Google Scholar]
- 70.Gault WR, Gatens PF., Jr Use of low intensity direct current in management of ischemic skin ulcers. Phys. Therapy. 1976;56:265–269. doi: 10.1093/ptj/56.3.265. [DOI] [PubMed] [Google Scholar]
- 71.Petrofsky JS, Lawson D, Berk L, et al. Enhanced healing of diabetic foot ulcers using local heat and electrical stimulation for 30 min three times per week. J. Diabet. 2010;2:41–46. doi: 10.1111/j.1753-0407.2009.00058.x. [DOI] [PubMed] [Google Scholar]
- 72.Cramp FL, McCullough GR, Lowe AS, et al. Transcutaneous electric nerve stimulation: The effect of intensity on local and distal cutaneous blood flow and skin temperature in healthy subjects. Arch. Phys. Med. Rehabil. 2002;83:5–9. doi: 10.1053/apmr.2002.27478. [DOI] [PubMed] [Google Scholar]
- 73.Huang G, Li F, Zhao X, et al. Functional and biomimetic materials for engineering of the three-dimensional cell microenvironment. Chem. Rev. 2017;117:12764–12850. doi: 10.1021/acs.chemrev.7b00094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhao G, Qing H, Huang G, et al. Reduced graphene oxide functionalized nanofibrous silk fibroin matrices for engineering excitable tissues. Npg Asia Materials. 2018;10:982–994. doi: 10.1038/s41427-018-0092-8. [DOI] [Google Scholar]
- 75.Liu S, Li J, Zhang S, et al. Template-assisted magnetron sputtering of cotton nonwovens for wound healing application. ACS Appl. Bio Mater. 2019;3:848–858. doi: 10.1021/acsabm.9b00942. [DOI] [PubMed] [Google Scholar]
- 76.Talikowska M, Fu X, Lisak G. Application of conducting polymers to wound care and skin tissue engineering: a review. Biosens. Bioelectron. 2019;135:50–63. doi: 10.1016/j.bios.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 77.Kai H, Yamauchi T, Ogawa Y, et al. Accelerated wound healing on skin by electrical stimulation with a bioelectric plaster. Adv. Healthc. Mater. 2017;6:1700465. doi: 10.1002/adhm.201700465. [DOI] [PubMed] [Google Scholar]
- 78.Qing H, Jin G, Zhao G, et al. Heterostructured silk-nanofiber-reduced graphene oxide composite scaffold for sh-sy5y cell alignment and differentiation. ACS Appl. Mater. Interfaces. 2018;10:39228–39237. doi: 10.1021/acsami.8b12562. [DOI] [PubMed] [Google Scholar]
- 79.Lu Y, Wang Y, Zhang J, et al. In-situ doping of a conductive hydrogel with low protein absorption and bacterial adhesion for electrical stimulation of chronic wounds. Acta Biomater. 2019;89:217–226. doi: 10.1016/j.actbio.2019.03.018. [DOI] [PubMed] [Google Scholar]
- 80.Siemens W, Boehlke C, Bennett MI, et al. Transcutaneous electrical nerve stimulation for advanced cancer pain inpatients in specialist palliative care-a blinded, randomized, sham-controlled pilot cross-over trial. Support. Care Cancer. 2020;28:5323–5333. doi: 10.1007/s00520-020-05370-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hofmeister M, Memedovich A, Brown S, et al. Effectiveness of neurostimulation technologies for the management of chronic pain: a systematic review. Neuromodulation. 2020;23:150–157. doi: 10.1111/ner.13020. [DOI] [PubMed] [Google Scholar]
- 82.Sluka KA, Walsh D. Transcutaneous electrical nerve stimulation: Basic science mechanisms and clinical effectiveness. J. Pain. 2003;4:109–121. doi: 10.1054/jpai.2003.434. [DOI] [PubMed] [Google Scholar]
- 83.Chen C-C, Johnson MI, McDonough S, et al. The effect of transcutaneous electrical nerve stimulation on local and distal cutaneous blood flow following a prolonged heat stimulus in healthy subjects. Clin. Physiol. Funct. Imaging. 2007;27:154–161. doi: 10.1111/j.1475-097X.2007.00731.x. [DOI] [PubMed] [Google Scholar]
- 84.Capogrosso M, Gandar J, Greiner N, et al. Advantages of soft subdural implants for the delivery of electrochemical neuromodulation therapies to the spinal cord. J. Neural Eng. 2018;15:026024. doi: 10.1088/1741-2552/aaa87a. [DOI] [PubMed] [Google Scholar]
- 85.Sovak G, Budgell B. Tens-like stimulation downregulates inflammatory cytokines in a pc-12 cell line. J. Manipulat. Physiol. Ther. 2017;40:381–386. doi: 10.1016/j.jmpt.2017.03.008. [DOI] [PubMed] [Google Scholar]
- 86.Osiri, M., Welch, V., Brosseau, L., et al.: Transcutaneous electrical nerve stimulation for knee osteoarthritis. In: The Cochrane Database of Systematic Reviews, CD002823-CD002823 (2000) [DOI] [PubMed]
- 87.Lampe GN. Introduction to use of trans-cutaneous electrical nerve-stimulation devices. Phys. Ther. 1978;58:1450–1454. doi: 10.1093/ptj/58.12.1450. [DOI] [PubMed] [Google Scholar]
- 88.Melzack R, Wall PD. Pain mechanisms: a new theory: a gate control system modulates sensory input from the skin before it evokes pain perception and response. Pain Forum. 1996;5:3–11. doi: 10.1016/S1082-3174(96)80062-6. [DOI] [Google Scholar]
- 89.Siqueira DLT, Ribeiro VV, Moreira MPA, et al. Effects of transcutaneous electrical nervous stimulation (tens) associated with vocal therapy on musculoskeletal pain of women with behavioral dysphonia: A randomized, placebo-controlled double-blind clinical trial. J. Commun. Disord. 2019;82:105923. doi: 10.1016/j.jcomdis.2019.105923. [DOI] [PubMed] [Google Scholar]
- 90.Husch HH, Watte G, Zanon M, et al. Effects of transcutaneous electrical nerve stimulation on pain, pulmonary function, and respiratory muscle strength after posterolateral thoracotomy: a randomized controlled trial. Lung. 2020;198:345–353. doi: 10.1007/s00408-020-00335-4. [DOI] [PubMed] [Google Scholar]
- 91.Zayan K, Aggarwal S, Felix E, et al. Transcutaneous electrical nerve stimulation for the long-term treatment of ocular pain. Neuromodulation. 2020;23:871–877. doi: 10.1111/ner.13146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Talbot LA, Solomon Z, Webb L, et al. Electrical stimulation therapies for active duty military with patellofemoral pain syndrome: a randomized trial. Mil. Med. 2020;185:E963–E971. doi: 10.1093/milmed/usaa037. [DOI] [PubMed] [Google Scholar]
- 93.Gibson, W., Wand, B.M., Meads, C., et al.: Transcutaneous electrical nerve stimulation (tens) for chronic pain - an overview of cochrane reviews. In: Cochrane Database of Systematic Reviews, CD011890 (2019) [DOI] [PMC free article] [PubMed]
- 94.Gladwell PW, Cramp F, Palmer S. Matching the perceived benefits of transcutaneous electrical nerve stimulation (tens) for chronic musculoskeletal pain against patient reported outcome measures using the international classification of functioning, disability and health (icf) Physiotherapy. 2020;106:128–135. doi: 10.1016/j.physio.2019.01.017. [DOI] [PubMed] [Google Scholar]
- 95.Brian R. The physiology of neuromuscular electrical stimulation. Pediatr. Phys. Therapy. 1997;9:96–102. [Google Scholar]
- 96.Ziada A, Hudlicka O, Tyler KR, et al. The effect of long-term vasodilatation on capillary growth and performance in rabbit heart and skeletal-muscle. Cardiovasc. Res. 1984;18:724–732. doi: 10.1093/cvr/18.12.724. [DOI] [PubMed] [Google Scholar]
- 97.Salmons S, Vrbova G. Influence of activity on some contractile characteristics of mammalian fast and slow muscles. J. Physiol. Lond. 1969;201:535–549. doi: 10.1113/jphysiol.1969.sp008771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Pette D, Smith ME, Staudte HW, et al. Effects of long-term electrical-stimulation on some contractile and metabolic characteristics of fast rabbit muscles. Pflug. Archiv-Eur. J. Physiol. 1973;338:257–272. doi: 10.1007/BF00587391. [DOI] [PubMed] [Google Scholar]
- 99.Zhang Q, Beirne S, Shu K, et al. Electrical stimulation with a conductive polymer promotes neurite outgrowth and synaptogenesis in primary cortical neurons in 3d. Sci. Rep. 2018;8:9855. doi: 10.1038/s41598-018-27784-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bai G-Y, Xu X-H, Wang J-H, et al. Single-leg weight limit of fixation model of simple supracondylar fracture of femur. Acta. Mech. Sin. 2019;35:926–939. doi: 10.1007/s10409-019-00855-0. [DOI] [Google Scholar]
- 101.Willand MP, Rosa E, Michalski B, et al. Electrical muscle stimulation elevates intramuscular bdnf and gdnf mrna following peripheral nerve injury and repair in rats. Neuroscience. 2016;334:93–104. doi: 10.1016/j.neuroscience.2016.07.040. [DOI] [PubMed] [Google Scholar]
- 102.Takahashi K, Hayashi M, Fujii T, et al. Early rehabilitation with weight-bearing standing-shaking-board exercise in combination with electrical muscle stimulation after anterior cruciate ligament reconstruction. Acta Med. Okayama. 2012;66:231–237. doi: 10.18926/AMO/48562. [DOI] [PubMed] [Google Scholar]
- 103.Gerovasili V, Stefanidis K, Vitzilaios K, et al. Electrical muscle stimulation preserves the muscle mass of critically ill patients: a randomized study. Crit. Care. 2009;13:R161. doi: 10.1186/cc8123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Aghasafari P, Pidaparti R. Influence of tidal-volume setting, emphysema and ards on human alveolar sacs mechanics. Acta. Mech. Sin. 2018;34:983–993. doi: 10.1007/s10409-018-0773-0. [DOI] [Google Scholar]
- 105.Yang S, Zhang LT, Hua C, et al. A prediction of in vivo mechanical stresses in blood vessels using thermal expansion method and its application to hypertension and vascular stenosis. Acta. Mech. Sin. 2018;34:1156–1166. doi: 10.1007/s10409-018-0780-1. [DOI] [Google Scholar]
- 106.Yang S, Wang Q, Shi W, et al. Numerical study of biomechanical characteristics of plaque rupture at stenosed carotid bifurcation: a stenosis mechanical property-specific guide for blood pressure control in daily activities. Acta. Mech. Sin. 2019;35:1279–1289. doi: 10.1007/s10409-019-00883-w. [DOI] [Google Scholar]
- 107.Hamada T, Sasaki H, Hayashi T, et al. Enhancement of whole body glucose uptake during and after human skeletal muscle low-frequency electrical stimulation. J. Appl. Physiol. 2003;94:2107–2112. doi: 10.1152/japplphysiol.00486.2002. [DOI] [PubMed] [Google Scholar]
- 108.Pfeiffer, M., Duente, T., Schneegass, S., et al.: Cruise control for pedestrians: Controlling walking direction using electrical muscle stimulation. In: 33rd Annual CHI Conference on Human Factors in Computing Systems. pp 2505–2514 (2015)
- 109.Wiehr, F., Kosmalla, F., Daiber, F., et al. Artopos - augmented reality terrain map visualization for collaborative route planning. ACM International Joint Conference on Pervasive and Ubiquitous Computing and ACM International Symposium on Wearable Computers 1047–1050 (2017)
- 110.Duente, T., Pfeiffer, M., Rohs, M., et al.: Zap++: A 20-channel electrical muscle stimulation system for fine-grained wearable force feedback. in: 19th International Conference on Human-Computer Interaction with Mobile Devices and Services. 13 (2017)
- 111.Hao M, Chou CH, Zhang J, et al. Restoring finger-specific sensory feedback for transradial amputees via non-invasive evoked tactile sensation. IEEE Open Journal of Engineering in Medicine and Biology. 2020;1:98–107. doi: 10.1109/OJEMB.2020.2981566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lopes, P., Butzmann, L., Baudisch, P.: Muscle-propelled force feedback: Bringing force feedback to mobile devices using electrical stimulation. In: 4th Augmented Human International Conference. pp. 231–232 (2013)
- 113.Lopes, P., Jonell, P., Baudisch, P. et al. Affordance plus plus : Allowing objects to communicate dynamic use. In: 33rd Annual CHI Conference on Human Factors in Computing Systems. pp. 2515–2524 (2015)
- 114.Colley, A., Leinonen, A., Forsman, M.-T., et al.: Ems painter: Co-creating visual art using electrical muscle stimulation. In: 12th International Conference on Tangible, Embedded, and Embodied Interaction. pp. 266–270 (2018)