Abstract
Aromatic aldehydes elicit their antisickling effects primarily by increasing the affinity of hemoglobin (Hb) for oxygen (O2). However, challenges related to weak potency and poor pharmacokinetic properties have hampered their development to treat sickle cell disease (SCD). Herein, we report our efforts to enhance the pharmacological profile of our previously reported compounds. These compounds showed enhanced effects on Hb modification, Hb-O2 affinity, and sickling inhibition, with sustained pharmacological effects in vitro. Importantly, some compounds exhibited unusually high antisickling activity despite moderate effects on the Hb-O2 affinity, which we attribute to an O2-independent antisickling activity, in addition to the O2-dependent activity. Structural studies are consistent with our hypothesis, which revealed the compounds interacting strongly with the polymer-stabilizing αF-helix could potentially weaken the polymer. In vivo studies with wild-type mice demonstrated significant pharmacologic effects. Our structure-based efforts have identified promising leads to be developed as novel therapeutic agents for SCD.
Graphical Abstract

INTRODUCTION
Sickle cell disease (SCD) is an inherited genetic disorder that is caused by a mutation at the sixth position of the β-chain of normal hemoglobin (HbA), which results in substitution of a polar glutamic acid (Glu) residue for a hydrophobic valine (Val) to form sickle Hb (HbS).1 Under hypoxic conditions, the concentration of the low affinity deoxygenated HbS (deoxyHbS) increases, which polymerizes into long, rigid, and insoluble fibers with concomitant sickling of red blood cells (RBCs). The polymerization process is initiated by a primary interaction between β2Val6 from one deoxyHbS molecule and a hydrophobic acceptor pocket in the region of β1Ala70, β1Phe85, and β1Leu88 of another deoxyHbS molecule.2-4 The polymerization process is further worsened because of high levels of 2,3-diphosphoglycerate present in sickle RBCs, which is an adaptive process to increase tissue oxygenation in SCD patients.5-8 The hypoxia-induced RBC sickling leads to multiple secondary pathophysiological events, for example, adhesion of RBCs to tissue endothelium, oxidative stress/damage, hemolysis of RBCs, hemolytic anemia, inflammation, vaso-occlusion, impaired microvascular blood flow, decreased vascular nitric oxide (NO) bioavailability, painful crises, multiorgan damage, morbidity, and mortality.9-11
Several secondary contacts between the deoxyHbS molecules also play important roles in stabilizing the HbS polymers, as demonstrated by a number of naturally occurring mutations that disrupt these polymer-stabilizing contacts and in turn reduce HbS polymerization and RBC sickling. For example, αAsn78 → Lys (Hb Stanleyville) leads to increase in the solubility of deoxyHbS heterotetramers, mitigating the severity of SCD.12-14
Approximately, 100,000 Americans have SCD, and the Centers for Disease Control and Prevention (CDC) has declared this disease as a major public health concern.15 Four drugs have currently been approved for treating SCD. These include hydroxyurea (HU),16,17 which has been used for about two decades. There is reported lack of response to HU in up to 30% of patients due to a complex variety of reasons.18 l-Glutamine (Endari), an antioxidant, was approved in 2017 by the US FDA and expected to reduce painful crises and adverse events;19 however, the European regulatory body recommended against approval of the medication because of limited evidence of efficacy in phase III trials.20 GBT-440 (Aka Voxelotor or Oxbryta),21 (Figure 1), and Crizanlizumab22 were approved in 2019. The latter is a monoclonal antibody targeting P-selectin, to reduce the frequency of painful vaso-occlusive crises. As intracellular polymerization of deoxyHbS remains the principal cause of the pathophysiology-associated SCD, GBT-440, which directly prevents hypoxia-induced RBC sickling by increasing Hb affinity for oxygen, offers immense therapeutic potential.23 Although, in the clinic, GBT-440 showed proof-of-principle disease-modifying potential by increasing Hb levels and reducing hemolysis in patients, there was no evidence of efficacy against vaso-occlusion. Thus, there is a need for novel aromatic aldehydes for treating SCD with better clinical outcomes.
Figure 1.

Structures of 5-HMF, vanillin, INN-298, INN-312, SAJ-310, TD-7, and GBT-440.
Prior to GBT-440, several aromatic aldehydes were studied as allosteric effectors of hemoglobin (AEHs) for their antisickling activities.24,25 Specifically, these compounds form Schiff-base adducts with the Val1 amines of both α-subunits of Hb increase Hb-O2 affinity and ameliorate the primary pathophysiology of SCD, that is, RBC sickling. Several challenges relating to the application of aromatic aldehydes for treating SCD became apparent during the early years of their development. These include nonspecific binding to plasma proteins and susceptibility of the active aldehyde moiety to rapid oxidative metabolism by several enzymes, for example, aldehyde oxidase (AO) and aldehyde dehydrogenases (ALDHs),26-32 that compromise the effectiveness of these compounds dramatically. Another major challenge is the difficulty in designing pharmaceutically applicable agents that are capable of sustained modification of the large amounts of intracellular Hb (~5 mmol) in the body. These problems led to termination of clinical trials of vanillin and 5-HMF (Aes103)29 for SCD. GBT-440 appeared to have overcome some of these problems. While the long-term benefits of GBT-440 are still unclear, the need remains critical for additional investigations into strategies to further enhance this class of compounds. In our continuous efforts to develop aromatic aldehydes for SCD, we have adopted a structure-based approach to modify the antisickling agent, vanillin, resulting in several generations of potent, allosteric, and covalent-binding antisickling derivatives.33-39 From vanillin to the INN33 or SAJ39 series and then to TD35 series of compounds (Figure 1), significantly improved antisickling activities have been observed. Among these, the INN and SAJ compounds are pyridyl derivatives of vanillin, while the TD compounds represent further modifications of the INN compounds (with a hydroxymethyl group on the pyridine ring) rationalized to stereospecifically inhibit deoxyHbS polymer formation by perturbing the αF-helix and contribute to the antisickling activity of the compounds through the O2-independent antisickling mechanism. At every stage, co-crystallization of these compounds in complex with Hb revealed insights into their binding and guided our future structure-based modifications. These compounds, however, still underwent significant metabolism leading to suboptimal pharmacokinetics properties, for example, short duration of action and low bioavailability, mainly because of the highly susceptible aldehyde pharmacophore. Nonetheless, the development of vanillin,40 5HMF,38,41 and GBT-44042 demonstrated that these allosteric modulators of Hb carry no significant toxicity even at high doses and thus established aromatic aldehydes as a scaffold to design potential therapeutics for SCD.
In this current work, we aimed to design a series of aromatic aldehydes that potently improve the antisickling activity of aromatic aldehydes while addressing some of the challenges associated with the rapid metabolism of the aldehyde pharmacophore.
RESULTS AND DISCUSSION
Molecular Design.
Two molecules of vanillin bind to the α-cleft of Hb in a symmetry-related fashion, forming Schiff-base interactions with the N-terminal amines of the two α-subunits. Our previous studies identified several vanillin derivatives, for example, SAJ-310,39 INN-312,34 and INN-29834 (Figure 1) with improved in vitro antisickling potency compared to vanillin.35 Some of these compounds, for example, INN-312 with the pyridinylmethoxy substitution ortho to the aldehyde group, showed a second novel O2-independent antisickling activity, although weak, which was attributed to weak hydrophobic interactions with a surface-located αF-helix. Based on structural studies, we subsequently modified the SAJ and INN series of compounds to generate another series of compounds termed TD, by incorporating the hydroxymethyl group on the pyridine ring. The rationale was to increase protein interactions with the α-cleft residues to potently increase Hb oxygen affinity (O2-dependent antisickling activity), as well as strongly interact with the surface-located αF-helix to directly weaken polymer interactions (O2-independent antisickling activity). The most promising compound, TD-7, however undergoes significant metabolism leading to suboptimal pharmacokinetics properties, for example, short duration of pharmacologic action and low bioavailability. In addition, the co-crystal structure of TD-7 with Hb revealed that the hydroxymethyl group did not make interactions with the αF-helix as anticipated.35 Therefore, we decided to pursue further structure-based modifications to design better pharmacological agents.
In the current study, we structurally modified TD-7 by first replacing the hydroxymethyl group on the pyridine ring with a methyl ester group, as well as varying its position on the pyridine ring while maintaining the meta-methoxy on the benzaldehyde ring (Table 1, compounds 1–7). Introduction of the methyl ester group was expected to lead to closer interactions with the αF-helix and increase polymer destabilization and/or increase interactions with Hb to improve both the O2-independent and O2-dependent antisickling activities. Next, to reduce oxidative metabolism of the aldehyde moiety, we introduced an ortho-hydroxy group on the benzaldehyde ring (Table 1, compounds 8–14). As previously observed, the ortho-hydroxy is expected to form an intramolecular hydrogen-bond interaction with the aldehyde moiety to reduce enzymatic metabolism.43,44
Table 1.
Structures of Target Compounds
 | |||||
|---|---|---|---|---|---|
| Compd. | R1 | R2 | X | Y | R3 |
| 1 | H | −OCH3 | N | CH | |
| 2 | H | −OCH3 | N | CH | |
| 3 | H | −OCH3 | N | CH | |
| 4 | H | −OCH3 | N | CH | |
| 5 | H | −OCH3 | CH | N | |
| 6 | H | −OCH3 | CH | N | |
| 7 | H | −OCH3 | CH | N | |
| 8 | −OH | H | N | CH | |
| 9 | −OH | H | N | CH | |
| 10 | −OH | H | N | CH | |
| 11 | −OH | H | N | CH | |
| 12 | −OH | H | CH | N | |
| 13 | −OH | H | CH | N | |
| 14 | −OH | H | CH | N | |
In summary, two series of compounds, one with the 5-methoxy group (1–7) and second with the 2-hydroxy group (8–14) on the aromatic aldehyde ring, were synthesized, as shown in Table 1. Each series contained 2- and 3-pyridine-substituted benzaldehydes with the methyl esters at different positions on the pyridine ring. Because of unavailability of reagents as well as limitations in the synthetic feasibility, the 4-substituted pyridine derivatives were not included.
Chemistry.
All target compounds in Table 1 were synthesized in two steps. The first step was a radical-catalyzed Wohl–Ziegler bromination reaction of a methyl-substituted pyridylmethyl ester to give bromomethyl-substituted pyridylmethyl ester. The starting materials used for these reactions were commercially available methyl-substituted methyl esters of nicotinic acid and picolinic acid. N-Bromosuccinimide (NBS) was used as the brominating agent, while 2,2′-azobis(2-methylpropionitrile) (AIBN) was applied as the radical initiator. The bromomethyl pyridine intermediates obtained were then used to form an ether linkage between the phenolic oxygen of either 2-hydroxy-5-methoxybenzaldehyde or 2,6-dihydroxybenzaldehyde via Williamson’s synthesis using the previously reported procedure (Scheme 1).35,39 The synthesis of TD-7, as a positive control, has been reported.33,39 The compounds were obtained in reasonable yields (75–90%), fully characterized, and then investigated for binding interactions with Hb, as well as in vitro and in vivo pharmacokinetics and/or pharmacodynamics (PDs) studies using normal and sickled blood or wild-type mice.
Scheme 1.

Synthetic Route for Target Compounds
Compounds Potently Increase Hb Affinity for Oxygen.
Aromatic aldehydes are known to bind to Hb allosterically and modify it, either to increase its O2 affinity [stabilizing the relaxed (R) state] or increase O2 delivery [stabilizing the tense (T) state].25,45-50 Increasing the Hb-O2 affinity is the primary mechanism by which these compounds elicit antisickling effects as the R-state Hb is resistant to polymerization. Thus, oxygen equilibrium curve (OEC) studies were first conducted using normal human whole blood to determine the effects of the compounds on Hb’s affinity for oxygen.
Dose-dependent studies were carried out using 0.5, 1, and 2 mM concentrations of the compounds and normal whole blood (hematocrit adjusted to 30%) at 37 °C as previously reported.33 The concentration of Hb in blood (~5.0 mmol) warrants the use of such high concentrations of the tested compounds. Whole blood comprises of plasma proteins, enzymes, and RBC cell membrane, which may play an interfering role on the activity of the compounds as compared to purified Hb. Thus, whole blood was used in this study to obtain results more representative of the native system.
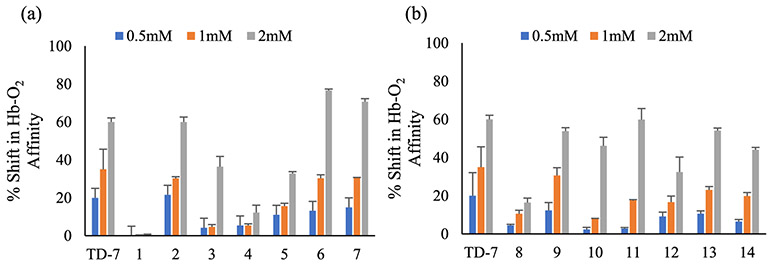
As shown in Figure 2, the compounds increased Hb affinity for O2 in normal whole blood to varying degrees. The low or insignificant activity shown by compounds 1 and 8 was likely because of their low solubility in most solvents, including dimethyl sulfoxide (DMSO). Thus, compounds 1 and 8 were not studied further. The low activity of compounds 3, 5, 10, and 12 compared to TD-7 could be due to the low intrinsic affinity for Hb or due to plasma protein binding and/or difficulty in RBC membrane permeation. It could also be due to significant metabolism by enzymes in the blood and/or RBCs that are known to play deteriorating roles in the bioavailability and potency of antisickling aromatic aldehydes.26,29,30,41 In comparison, compounds 2, 6, 7, 9, 11, and 13 showed significant increase in the Hb-O2 affinity. Vanillin, at 2 mM concentration, only increased Hb O2 affinity by 21%,33 whereas TD-7 showed almost similar potency as most of the best compounds. These observations confirmed that the structural modifications have conserved the potent Hb-O2 affinity in vitro activity previously observed in TD-7, the parent lead.
Figure 2.
Concentration-dependent shift in the Hb-O2 affinity of (a) m-methoxy-substituted and, (b) o-hydroxy-substituted compounds in normal whole blood after 1.5 h incubation.
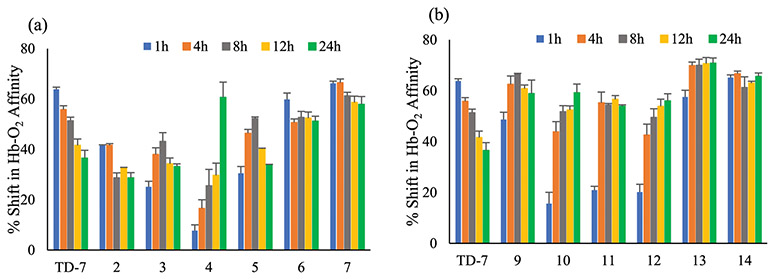
As pointed out above, aromatic aldehydes are highly susceptible to oxidative metabolism of the active aldehyde function,26-30 resulting in most of these compounds having poor oral bioavailability, which has stymied their development. Therefore, one of the major objectives of this work was to identify compounds that may be less susceptible to the oxidative metabolism of the aldehyde pharmacophore. For this reason, time-dependent Hb-O2 affinity studies were conducted with 2 mM concentrations of the compounds with human blood (hematocrit adjusted to 30%) at 37 °C for 24 h using TD-7 as control. Although, oxidative metabolism of aldehydes occurs by several enzymes in various tissues including AO,31,32 it is primarily by ALDH in RBCs;26-28 therefore, whole blood serves as an appropriate matrix for the measure of oxidative metabolism in vitro. Among the meta-methoxy derivatives, compound 2 showed optimal activity between 1 and 4 h which then decreased to about 70% of its activity by 8 h and sustained through the 24 h. Compounds 3 and 5 showed maximum potency at 8 h followed by a gradual decrease over the course of 24 h to about 70 and 60% of its original activity, respectively. Interestingly, three of the meta-methoxy derivatives, including compounds 4, 6, and 7 appear to be stable against oxidative metabolism in whole blood. Compound 4 shows a very slow onset and reached its highest potency at 24 h. Such a slow onset may not be optimal because other metabolizing enzymes, such as in the liver, could deactivate this compound before reaching therapeutic potency. Compounds 6 and 7, on the other hand, have quick onset and showed a sustained effect during the 24 h experiment (Figure 3a). In comparison, TD-7, similar to some of the meta-methoxy derivatives, also after reaching a maximal effect at 1 h gradually decreased in potency, and at 24 h, showed a loss of almost 45% of its activity. Clearly, the meta-methoxy-substituted derivatives show varying oxidative metabolism in whole blood and/or RBC, probably because of differences in their affinity to the metabolizing enzymes, ALDH. What is not clear is whether, when administered in vivo, compounds 4, 6, and 7 would still maintain their resistance to oxidative metabolism of the aldehyde.
Figure 3.
Time-dependent shift in the Hb-O2 affinity of (a) m-methoxy-substituted and, (b) o-hydroxy-substituted compounds in normal whole blood at a concentration of 2 mM.
Unlike the meta-methoxy-substituted compounds (Figure 3a), all the ortho-hydroxy-substituted compounds (Figure 3b) showed sustained action over the duration of the experiment, suggesting all compounds are resistant to oxidative metabolism in whole blood. Interestingly, compounds 10, 11, and 12 showed relatively slow onset of action compared to the other compounds, which we speculate to be due to slow RBC membrane permeation. As previously reported, formation of intramolecular hydrogen bonding between the aldehyde carbonyl group and the ortho-hydroxy group acts as a protection to prevent undue metabolism of the aldehyde.43,51 It is clear from the forgoing that not only did we maintain the Hb-O2 potencies of these compounds but also achieved one of our primary objectives of increasing the metabolic stability of the aldehyde moiety.
The compounds have a major effect of Hb cooperativity as the shape of the OEC changed from sigmoidal to hyperbolic with addition of the compound (Supporting Information S1), as a result of the stabilization of the R-state Hb and restraining the cross talk between the Hb subunits that is important for the R → T transition.24,25
Compounds Potently Increase In Vitro Antisickling, Hb-Modification, and Hb-O2 Affinity Activities with Sickle RBCs.
Because the pathophysiology of SCD, including RBC sickling, is driven by hypoxia-induced polymerization of deoxyHbS, we further characterized these high-O2 affinity compounds for their O2-dependent antisickling properties. We incubated the compounds at 0.5, 1.0, and 2.0 mM concentrations with blood from individuals with homozygous SS (hematocrit 20%), under hypoxic conditions (2.5% O2/97.5% N2) at 37 °C for 2 h. The concentration-dependent antisickling effect was observed for most of the tested compounds (Figure 4). Although compounds 4, 12, and 14 showed dose-dependent sickling inhibition, their antisickling effects were significantly weaker when compared to the other compounds. At a lower concentration of 0.5 mM, compounds 2, 5, 9, and 13 showed more than 35% sickling inhibition as compared to only 26% by TD-7. At 1.0 mM concentration, compounds 5, 6, 9, and 13 showed more than 70% inhibition as compared to 47% by TD-7, with compounds 5 and 13 almost exhibiting 100% sickling inhibition (Figure 4a,b). At 2.0 mM concentration, several of the tested compounds showed more than 90% sickling inhibition.
Figure 4.
Inhibition of sickling by (a) m-methoxy-substituted and, (b) o-hydroxy-substituted compounds in SS RBCs.
This observation clearly indicates that the structural modifications have led to improvement in increasing the antisickling potencies of the compounds when compared to TD-7. In comparison, GBT-440 at 0.5 and 1.0 mM decreased the O2-dependent sickling by 56% and 98%, respectively, suggesting that at lower concentration, GBT440 shows better potency. This enhanced potent activity of GBT-440 has been suggested to be because of reduced binding stoichiometry, that is, it binds Hb at a 1:1 ratio52 while our compounds bind Hb at a 2:1 ratio.
The remarkable enhanced antisickling activity of the compounds compared to TD-7 led us to investigate whether such an improvement is solely the consequence of their ability to bind Hb and modify it to the higher O2 affinity form. Aliquot samples from the above antisickling study were hemolyzed and utilized for dose-dependent Hb-O2 affinity and Hb modification studies. As observed with normal whole blood (Figure 2), the compounds increased Hb affinity for oxygen and Hb modification in dose-dependent fashions (Figure 5). The modification effect by the compounds were in most instances similar or better than observed with TD-7. For example, compounds 7, 9, 10, 11, 13, and 14 showed almost complete modification of HbS at 2.0 mM concentration compared to the ~80% by TD-7. At 1.0 mM concentration, compounds 5, 6, 7, 9, and 14 modified HbS similar to TD-7 (~60%), while compounds 10, 11, and 13 showed more than 80% HbS modification at the same concentration (Figure 5). In few instances, such as with compounds 2, 3, 4, and 12, TD-7 showed better a Hb modification effect. Interestingly, while compounds 4 and 12 expectedly showed weaker antisickling effects than TD-7, compounds 2 and 3 appear to show comparable or stronger antisickling effects when compared to TD-7. In comparison, GBT-440 as expected from its potent antisickling activity showed very high Hb modification to the high affinity adduct form of 55% (0.5 mM) and 100% (1.0 mM).
Figure 5.
Concentration-dependent Hb modification (adduct formation) by (a) m-methoxy-substituted and, (b) o-hydroxy-substituted compounds in SS RBCs.
Of most interest is that despite the fact that most of these compounds, with the exception of 4, 12, and 14, show better antisickling effects than TD-7, they conversely increase Hb affinity for oxygen to a lesser degree when compared to TD-7 (Figures 4 and 6). Similar to TD-7, GBT-440’s high antisickling activity (56% and 98% at 0.5 and 1.0 mM) also corresponded to very high P50 shifts of 41 and 80%, respectively. The unusual weak correlation between the antisickling and P50 shift of our newer derivative compounds as discussed below is likely because of a second mechanism of O2-independent antisickling activity.
Figure 6.
Concentration-dependent shift in Hb-O2 affinity by (a) m-methoxy-substituted and, (b) o-hydroxy-substituted compounds in SS RBCs.
The pharmacologic properties of aromatic aldehydes, including their ability to modify Hb, increase Hb affinity for oxygen, and inhibit hypoxia-induced RBC sickling are known to be highly correlated. For example 5-HMF, when incubated with sickle blood results in perfect direct correlations (r2 = 1) between any two of the biological parameters, that is, Hb adduct, P50 shift, or sickling inhibition.41 With the newer derivative compounds, even though we observed a relatively good correlation between the Hb adduct and P50 shift (r2 = 0.8–0.9) (Figure 7), it is weaker than reported for 5-HMF41 or GBT-440. Even more so, the correlation between antisickling activity and Hb modification are weaker and especially between antisickling activity and P50 shift (Figure 7). The weak correlation between the PD parameters of the compounds presented here clearly suggests additional unique mechanism of antisickling action that is particularly independent of Hb oxygen affinity. Compounds 2, 3, 5, 6, 9, 10, and 13 demonstrated the most potent antisickling effects despite only marginally increasing Hb affinity for oxygen (Figure 7, Table 2). For example, while compound 9 inhibits sickling and increases Hb-O2 affinity by 43 and 12% at 0.5 mM (71 and 19% at 1.0 mM), respectively, GBT-440 on the other hand inhibits sickling and increases Hb oxygen affinity by 56 and 41%, at 0.5 mM (100 and 80% at 1 mM), respectively. This substantial antisickling activity, but at a relatively much lower Hb-O2 affinity increase by some of our compounds, we propose, is because of the polymer destabilization effect that potentially led to an O2-independent antisickling activity contribution, which is currently under further investigation.
Figure 7.
Linear correlation between increase in the Hb-O2 affinity, antisickling, and Hb-modification properties for 0.5 mM (blue), 1 mM (green), and 2 mM (gray) concentrations of compounds.
Table 2.
Sickling Inhibition, Hb-O2 Affinity, and Hb-Modification Studies Using SS RBCs with the Seven Compounds
| % sickling inhibition |
% Hb-O2 affinity |
% Hb-modification |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| compd | 0.5 mM | 1 mM | 2 mM | 0.5 mM | 1 mM | 2 mM | 0.5 mM | 1 mM | 2 mM |
| 2 | 41.8 ± 0.7 | 71.7 ± 0.9 | 99 ± 2.6 | 5.9 ± 0.7 | 12.8 ± 1.9 | 21.3 ± 2.1 | 32.1 ± 0.5 | 49.9 ± 1.6 | 67.5 ± 2.2 |
| 3 | 17.5 ± 1.1 | 48.2 ± 1.2 | 88 ± 5.5 | 4.8 ± 2.1 | 20.1 ± 1.3 | 37.1 ± 2.5 | 20.9 ± 1.5 | 41.7 ± 1.8 | 63.8 ± 3.2 |
| 5 | 37.6 ± 0.9 | 97.7 ± 1.5 | 99.1 ± 1.1 | 4.4 ± 0.9 | 13.9 ± 1.5 | 25.4 ± 1.1 | 30.2 ± 0.5 | 62.3 ± 1.9 | 79.2 ± 0.6 |
| 6 | 29.1 ± 1.2 | 81.6 ± 1.8 | 96.5 ± 0.7 | 9.9 ± 0.7 | 19.5 ± 0.8 | 32.3 ± 0.7 | 36.1 ± 1.1 | 64 ± 1.3 | 81.3 ± 1.4 |
| 9 | 43.3 ± 4.1 | 70.6 ± 4.1 | 98.8 ± 1.8 | 12.2 ± 2.1 | 18.8 ± 2.1 | 39.6 ± 1.3 | 46 ± 3.2 | 64.4 ± 2.7 | 90.8 ± 1.2 |
| 10 | 7.6 ± 1.1 | 54.2 ± 0.1 | 93.2 ± 4.4 | 5.4 ± 1.4 | 29.2 ± 1.1 | 48.6 ± 0.4 | 29.2 ± 1.6 | 81.1 ± 0.6 | 100 ± 0.7 |
| 13 | 39.5 ± 12 | 90.7 ± 11 | 98.9 ± 2.1 | 16.3 ± 12 | 44.2 ± 1.6 | 60.1 ± 2.1 | 47.8 ± 4.6 | 92.5 ± 5 | 100 ± 1.1 |
Stability Studies of Compounds Using Whole Blood Show Limited Hydrolysis of the Parent Methyl Ester.
The parent compounds reported here contain the methylester moiety, which could potentially hydrolyze into their corresponding acids and affect the PK or PD properties. We therefore studied representative compounds, 9 and 10, for their stability in buffer and whole blood using reversed-phase high-performance liquid chromatography (HPLC). The compounds at 0.5 and 1 mM concentrations were incubated in normal whole blood (hematocrit adjusted to 30%) at 37 °C, and aliquots obtained at 0, 4, 8, and 24 h were subjected to deproteinization and extraction of the compounds in organic solvent (acetonitrile). The purified extracts were subjected to HPLC analyses to detect and quantify the amount of the parent compound present, as well as any metabolized derivatives. The acid analogue of compound 10 was used as a control, which as expected eluted before the parent compound (Figure 8). Both parent compounds showed formation of the carboxylic acid analogue, but at very low levels (Figure 8). Compound 10 demonstrated a time-dependent hydrolysis of the methyl ester to the acid analogue, with 89 ± 4.4% (0.5 mM) and 83 ± 1.7% (1 mM) of parent compound 10 retained at 24 h (Figure 8a,c). Interestingly, compound 9 did not show any observable metabolism until 24 h, with 81 ± 1.4% (0.5 mM) and 78 ± 2.4% (1 mM) of the parent compound 9 being retained (Figure 8a,b). Thus, during the course of 24 h, only approximately 20% of compounds 9 and 10 metabolized into the carboxylic acid form, indicating that the antisickling activity of both compounds (and in general all compounds) which were conducted within 3 h of experimentation is likely exclusively due to the parent methyl esters.
Figure 8.
Time-dependent recovery of parent compounds 9 and 10 at 0.5 and 1 mM; (a) representative chromatograms of 1 mM compound 9 and (b) compound 10 at 0, 4, 8, and 24 h.
X-ray Crystallography Shows Compounds Bind as Predicted and Provide Atomic Basis for Their Potent Antisickling Activities.
To verify our molecular design, which was intended toward these compounds interacting with the αF-helix of Hb, as well as explain the unusual functional/biological effects of the compounds, we selected two compounds, one each from the meta-methoxy-substituted and ortho-hydroxy-substituted benzaldehyde series, namely, compounds 6 and 9 respectively, for co-crystallization studies with CO-liganded Hb (COHb) in the R2-state conformation. COHb was used for the crystallographic study because the structures of O2-Hb and COHb complexes are very similar, while the latter is comparatively easy to purify, more stable, and easier to work with. Also, normal Hb (HbA) instead of HbS was used for the crystallographic study because the quaternary structures as well as the size and geometry of the α-cleft binding site are similar in the two Hb structures.4,52 Freshly made solution of the compounds was added to deoxygenated Hb (30 mg/mL protein) at an Hb tetramer-compound ratio of 1:10. The complex mixture was saturated with CO and allowed to incubate for 2 h to form the COHb-compound complex. Co-crystallization experiments were carried out under low salt precipitant conditions (10–20% PEG6000, 100 mM HEPES buffer, pH 7.4), where cherry red needle crystals, isomorphous to previous aromatic aldehyde Hb-R2 state complex crystals, appeared in 1–3 days and were used to collect X-ray diffraction data.35,39 The crystal structures of the COHb complexes with compounds 6 and 9 were determined by the molecular replacement method with the Phenix program53 using the native R2-state crystal structure (PDB ID 1BBB) as a search model. Structure refinement was carried out using both Phenix and CNS,54 and model correction was carried out using COOT.55 The crystal structures of COHb-6 (PDB ID 6XDT) and COHb-9 (PDB ID 6XE7) complexes were refined to 1.9 and 2.0 Å, respectively. The overall tetrameric structures of both complexes were indistinguishable from 1BBB (RMSD 0.3 Å), and their detailed crystallographic data are summarized in Table 3.
Table 3.
Crystallographic Data and Refinement Statistics for COHb-6 and COHb-9 Complexes
| compound | 6 (PDB ID6XDT) | 9 (PDB ID6XE7) |
|---|---|---|
| Data Collection Statistics | ||
| space group | P212121 | P212121 |
| unit-cell a, b, c (c5) | 63.68, 82.98, 105.03 | 62.64, 82.76, 104.77 |
| resolution (c5) | 29.32–1.90 (1.97–1.90) | 29.29–2.00 (2.07–2.00) |
| unique reflections | 40,600 (3764) | 37,109 (3683) |
| redundancy | 4.27 | 4.04 |
| completeness (%) | 92.5 (86.9) | 98.9 (99.7) |
| average I/σ(I) | 13.4 (3.3) | 8.5 (2.3) |
| Rmerge (%)a | 5.5 (37.3) | 7.9 (42.8) |
| Refinement Statistics | ||
| resolution (c5) | 30.00–1.90 | 30.00–2.00 |
| no. of reflections | 40,573 | 37,580 |
| Rwork (%) | 18.81 | 19.94 |
| Rfree (%)b | 23.91 | 25.14 |
| rmsd bonds (c5) | 0.006 | 0.006 |
| rmsd angles (deg) | 0.97 | 0.83 |
| Dihedral Angles | ||
| most favored (%) | 98.00 | 98.00 |
| allowed (%) | 1.78 | 1.96 |
| Average B (c52)/Atoms | ||
| all atoms | 32.00 | 34.90 |
| protein | 31.50 | 34.40 |
| ligand | 29.50 | 35.00 |
| water | 36.90 | 39.00 |
Rmerge = ΣhklΣi∣Ii(hkl) − ⟨I(hkl)⟩∣/ΣhklΣiIi(hkl).
Rfree was calculated from 5% randomly selected reflection for cross-validation. All other measured reflections were used during refinement.
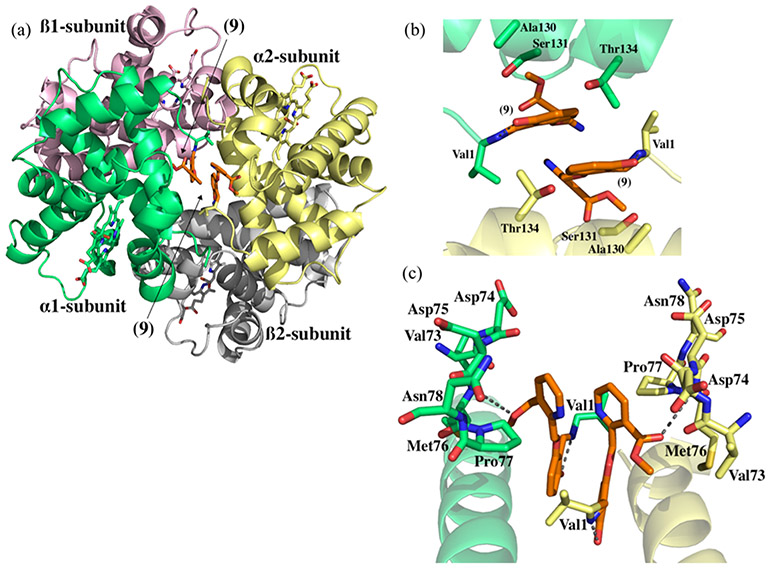
As expected, compounds 6 and 9 bind in a symmetry-related fashion, forming Schiff base interactions between the aldehyde group of the compounds with the αVal1 nitrogens (~1.5 c5) of both α1- and α2-subunits at the α-cleft of Hb. Also, as anticipated, the ortho-substituted pyridinylmethoxy-methyl ester groups directed upward toward the surface of the α-cleft and close to the αF-helix (Figures 9 and 10). Because both molecules in their respective structures bind in a symmetrical fashion, detailed interaction analyses with Hb will be focused on α2Val1 binding, which appears to be more ordered than the α1Val1 binding, especially with compound 9.
Figure 9.
Crystal structure of Hb in the R2 conformation in complex with two molecules of compound 9 bound at the α-cleft (a) overall crystal structure of Hb in the R2 conformation in the complex with two molecules of compound 9 in the central water cavity. (b) Close-up view of compound 9 bound at the α-cleft of Hb. (c) Close-up view of compound 9 showing interactions with the residues of the αF-helix. (Hb is shown as cartoon with α1 subunit in green, α2 in yellow, β1 in pink, and β2 in gray; hemes are shown as sticks; compound 9 is shown in orange sticks).
Figure 10.
Crystal structure of Hb in the R2 conformation in complex with two molecules of compound 6 bound at the α-cleft (a) overall crystal structure of Hb in the R2 conformation in complex with two molecules of compound 6 in the central water cavity. (b) Closeup view of compound 6 bound at the α-cleft of Hb. (c) Closeup view of compound 6 showing interactions with the residues of the αF-helix. (Hb is shown as cartoon with α1 subunit in green, α2 in yellow, β1 in pink, and β2 in gray; hemes are shown as sticks; compound 6 is shown in cyan sticks; water molecules are shown as red spheres).
The benzaldehyde ring of both compounds 6 and 9 superimposed closely, making both intra- and inter-subunit hydrophobic interactions with α2Ala130, α2Ser131, α2Thr134, and α1Thr134 (Figures 9-11). The pyridine rings of the two bound molecules formed extensive face-to-face π–π stacking interactions (3.4–3.8 c5) (Figures 9 and 10). These interactions serve to stabilize the R-state and increase the oxygen affinity of Hb, as reported previously.25
Figure 11.
Structural comparison of compounds 6 and 9 bound at the α-cleft of Hb. (a) superposition of compounds 6 and 9 molecules showing interactions with residues from αF-helix. (b) 90° rotated view of (a). (Hb is shown as cartoon with α1 subunit in green and α2 in yellow; compounds 6 and 9 are shown in cyan and orange sticks, respectively).
Interestingly, the ortho-hydroxy group of compound 9 makes both direct and water-mediated hydrogen bonding interactions with the αVal1 nitrogen. This interaction may help increase the binding affinity of compound 9 with the protein (Figure 3). The pyridinylmethoxy-methyl ester group in compound 9, which is directed toward the surface of the α-cleft, is involved in a hydrogen bonding interaction with the backbone nitrogen of α2Met76 of the αF-helix (3.5 c5) (Figure 9), which is missing in TD-7 binding.35 In addition, the pyridinylmethoxy-methyl ester group is also involved in hydrophobic interactions with α2Val73, α2Asp74, and α2Pro77 (2.7–4.0 Å) residues, which in TD-7 is relatively weaker (3.5–4.3 Å).35 These αF-helix interactions, we believe lead to perturbation of the helix to destabilize the polymer, explaining the unusually high antisickling effect (through O2-independent mechanism) at the relatively low P50 shift of this compound. As noted above, GBT-440 also binds to the α-cleft of the R2-state Hb to make Schiff-base interaction with the αVal1 amine. Interestingly, unlike our compounds and previously studied aromatic aldehydes that bind two molecules, one each to α1-subunit and α2-subunit, GBT-440 only binds one molecule at the α-cleft because of the isopropyl–pyrazole substituent that sterically prevents two molecules from binding.52 Nonetheless, unlike compound 9, GBT-440 does not make any close contact with the αF-helix, and thus, we do not expect this molecule to have any significant O2-independent antisickling activity, consistent with our comparative study that show strong correlation between its P50 shift and antisickling activity, and also as reported in the literature.52
Similar to compound 9, compound 6 also binds with the pyridinylmethoxy-methyl ester group orientated toward the αF-helix. Nonetheless, the ester group forms stronger intra-subunit hydrophobic interactions with α2Pro77 (3.1 c5) compared to compound 9 (3.5 c5) (Figures 10 and 11). In addition, the methoxy groups of the ester form several water-mediated hydrogen bonding interactions with the backbone atoms of α2Val73, α2Asp75, and α2Met76 of the αF-helix (Figure 10). Similar to compound 9, compound 6 interactions with the αF-helix likely lead to perturbation of the αF-helix, consistent with this compound unusually potent antisickling effect when compared to the relatively small effect on the Hb oxygen affinity.
Compounds Showed Improved In Vivo PD Effects.
Through systematic SAR studies, we successfully identified several compounds that showed significantly enhanced pharmacologic activity in vitro compared to the previous precursor TD-7. Several of these compounds showed sustained action over 24 h. Among the most potent compounds identified, compounds 9 and 10 were selected for further in vivo PD studies using wild-type mice (C57BL/6). The mice received a single IP dose of 150 mg/kg (a total of 6 mice). Blood samples were obtained prior to treatment (0), and l, 3, and 6 h post treatment, via submandibular bleeding and subjected to hemolysis using standard methods. Clarified lysates, free of cell debris and RBC ghosts, were analyzed by cation-exchange HPLC to determine the levels of drug-modified Hb (adducts) and to conduct OEC studies to determine the degree of P50 shift or Hb-O2 affinity.
All compounds showed significant in vivo modification of intracellular Hb with increasing levels from l h to the 6 h experimental period (Figure 12). Mice treated with compound 9 demonstrated the highest levels of modified Hb (16.9 ± 1.0% at 6 h), followed by compound 10 (9.6 ± 1.9%). Only 1.5 ± 0.3% modified Hb was observed in mice treated with the control TD-7. Corresponding changes were observed in oxygen affinity at the measured time points of 3 and 6 h (Figure 13). Mice treated with compound 9 showed the highest levels of shifts (14.5% ± 2.2 at 6 h). Compound 10 also increased Hb-O2 affinity, although relatively lower by 5.5% ± 1.9. In contrast, only approximately 1% ± 0.2 shift in the Hb-O2 affinity was observed in the mice treated with TD-7. These preliminary in vivo studies demonstrate improvement in the pharmacologic profile of our aromatic aldehyde class of allosteric effectors of Hb.
Figure 12.
Time-dependent modification of Hb in wild-type mice (n = 6) after 150 mg/kg IP administration of test compounds.
Figure 13.
Time-dependent Hb-O2 affinity shifts in the wild-type mice (n = 6) after 150 mg/kg IP administration of test compounds.
CONCLUSIONS
Intracellular polymerization of deoxygenated sickle hemoglobin remains the principal cause of the pathophysiology associated with SCD. Naturally occurring and synthetic AEHs have been investigated as potential therapeutic agents for the treatment of SCD.33,35,36,39-41,52 Even though this class of compounds showed significant potency in vitro, several of them lack good PK properties and therefore are not feasible to treat a chronic disease that requires significant levels of Hb modification for efficacy. These drawbacks have severely hampered the clinical development and utility of aromatic aldehydes to treat SCD. For example, our group studied 5-HMF; however, because of metabolic instability of the aldehyde that translated to poor oral bioavailability, the phase II clinical study was terminated.24,56 In previous reports, our group has demonstrated a systematic and rational structure-based approach to develop several aromatic aldehydes that exhibited excellent antisickling activity. In the current report, we modified the TD compounds to overcome the disadvantages associated with potency and oxidative metabolism by derivatizing the methyl alcohol on the pyridines to a methyl ester as well as protecting the aromatic aldehyde with an ortho-hydroxy group. These compounds exhibited several novel properties that are missing from the previous generation compounds. They showed sustained pharmacologic activity in vitro, as well as both sustained and significantly improved activity in vivo. Remarkably, some of the compounds potentially exhibited additional mechanism of action independent of the primary O2-dependent antisickling effect. Structural studies of compounds 6 and 9 complexes provided plausible explanations for these enhanced antisickling activities that involves perturbing the αF-helix leading to O2-independent antisickling activity. These novel results are encouraging and provide guidance for the further development of potent antisickling lead compounds. Compounds 9, 10, and 13 are currently undergoing formulation for detailed in vivo PD studies.
EXPERIMENTAL SECTION
Chemistry.
All reagents used in the synthesis and functional assays were purchased from Sigma-Aldrich (St. Louis, MO) or Thermo Fisher Scientific (Waltham, MA) and utilized without additional purification. 1H NMR and 13C NMR spectra were obtained on a Bruker 400 MHz spectrometer, and tetramethylsilane was used as an internal standard. Peak positions are given in parts per million (δ). Column chromatography was performed on silica gel (grade 60 mesh; Bodman Industries, Aston, PA). Routine thin-layer chromatography was performed on silica gel GHIF plates (250 μm, 2.5 × 10 cm; Analtech Inc., Newark, DE). MS spectra were obtained from a PerkinElmer Flexar UHPLC with an AxION 2 time-of-flight mass spectrometer, and the molecular weight of the compounds was within 0.005% of calculated values. Infrared spectra were obtained on Thermo Nicolet iS10 FT-IR. Purity of the compounds was determined by HPLC using Varian Microsorb 100-5 C18 column (250 × 4.6 mm), using Prostar 325 UV–vis (210 nm) as the detector. The HPLC parameters used were injection volume =15 μL, sample concentration = 3 mM, mobile phase = 60MeCN–40H2O, flow rate = 1 mL/min.
The compounds were synthesized according to the general procedures for similar compounds.33,35,39
General Procedure for the Synthesis of Compounds i–vii.
A mixture of the methyl-substituted pyridylmethyl ester (1 equiv) and α,α′-azoisobutyronitrile (AIBN) (10%) was dissolved in carbon-tetrachloride (CCl4). The solution was heated at 48 °C, and NBS (1.1 equiv) solution in CCl4 was added dropwise and stirred for 5 h. The reaction was cooled to room temperature, and the solvent evaporated. The mixture was then extracted using dichloromethane and water followed by washing the organic layer with brine. The organic layer was dried over sodium sulfate, filtered, evaporated, and the crude product was purified using column chromatography.
Methyl 5-(bromomethyl)nicotinate (vi) and methyl 2-(bromomethyl)benzoate (vii) were purchased from Sigma-Aldrich (St. Louis, MO).
Methyl 6-(Bromomethyl)nicotinate (i).57
The title compound was prepared following the general procedure as a white solid in 66% yield. 1H NMR (400 MHz, DMSO-d6): δ 9.16 (d, J = 1.56 Hz, 1H), 8.30 (dd, J = 8.12, 2.2 Hz, 1H), 7.53 (dd, J = 8.12, 0.48 Hz, 1H), 4.58 (s, 2H), 3.96 (s, 3H). MS (ESI) m/z: 229.98 [M + H]+.
Methyl 2-(Bromomethyl)nicotinate (ii).58
The title compound was prepared following the general procedure as an orange solid in 65% yield. 1H NMR (400 MHz, DMSO-d6): δ 8.71 (dd, J = 4.8, 1.76 Hz, 1H), 8.28 (dd, J = 7.92, 1.76 Hz, 1H), 7.33 (m, 1H), 5.04 (s, 2H), (s, 3H). MS (ESI) m/z: 229.98 [M + H]+.
Methyl 6-(Bromomethyl)picolinate (iii).59
The title compound was prepared following the general procedure as a white solid in 66% yield. 1H NMR (400 MHz, DMSO-d6): δ 8.05 (d, J = 7.68 Hz, 1H), (t, J = 7.72 Hz, 1H), 7.77 (d, J = 7.68 Hz, 1H), 4.67 (s, 2H), 3.98 (s, 3H). MS (ESI) m/z: 229.98 [M + H]+.
Methyl 2-(Bromomethyl)isonicotinate (iv).60
The title compound was prepared following the general procedure as a dark blue solid in 66% yield. 1H NMR (400 MHz, DMSO-d6): δ 8.77 (dd, J =5, 0.64 Hz, 1H), 8.02 (m, 1H), 7.77 (dd, J =5, 1.56 Hz, 1H), 4.81 (s, 2H), 3.91 (s, 3H). MS (ESI) m/z: 229.98 [M + H]+.
Methyl 5-(Bromomethyl)picolinate (v).61
The title compound was prepared following the general procedure as a white solid in 66% yield. 1H NMR (400 MHz, DMSO-d6): δ 8.78 (t, J = 1.48 Hz, 1H), 8.06 (d, J =1.36 Hz, 2H), 4.82 (s, 2H), 3.89 (s, 3H). MS (ESi) m/z: 229.98 [M + H]+.
General Procedure for Synthesis of Compounds 1–14.
A mixture of 2-hydroxy-5-methoxybenzaldehyde or 2,6-dihydroxybenzaldehyde (1 equiv) and corresponding bromo-pyridinylmethoxy-methyl ester (1 equiv) was dissolved in anhydrous N,N-dimethylformamide (DMF). Anhydrous potassium carbonate (K2CO3) (1.2 equiv) was added to this mixture, and the reaction was stirred at room temperature for 8–10 h. The solvent was then evaporated, and the reaction mixture extracted with ethyl acetate and water. The organic layer was dried over sodium sulfate, filtered, solvent evaporated, and the crude product was purified using column chromatography.
Methyl 6-((2-Formyl-4-methoxyphenoxy)methyl)nicotinate (1).
The title compound was prepared following the general procedure as a white solid in 82% yield. IR (Diamond, cm−1): 2922, 2867, 1721, 1669, 1620, 1585, 1499, 1446, 1391, 1370, 1286, 1224, 1174, 1141, 1121; 1H NMR (400 MHz, DMSO-d6): δ 10.49 (s, 1H), 9.08 (m, 1H), 8.36 (dd, J = 8.16, 2.16 Hz, 1H), 7.80 (d, J = 8.12 Hz, 1H), 7.25 (m, 3H), 5.42 (s, 2H), 3.91 (s, 3H), 3.77 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.94, 165.00, 161.04, 154.62, 153.62, 149.59, 137.82, 125.03, 124.77, 122.85, 121.31, 115.97, 110.78, 71.02, 55.59, 52.38. Mass spectrum m/z: found 324.07 [M + Na]+; calcd, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 96% pure.
Methyl 2-((2-Formyl-4-methoxyphenoxy)methyl)nicotinate (2).
The title compound was prepared following the general procedure as a pale yellow solid in 82% yield. IR (Diamond, cm−1): 2920, 1719, 1684, 1667, 1622, 1583, 1535, 1492, 1445, 1404, 1372, 1276, 1215, 1168, 1142; 1H NMR (400 MHz, DMSO-d6): δ 10.25 (s, 1H), 8.74 (dd, J = 4.76, 1.48 Hz, 1H), 8.23 (dd, J = 7.8, 1.44 Hz, 1H), 7.55 (m, 1H), 7.2 (m, 3H), 5.56 (s, 2H), 3.78 (s, 3H), 3.75 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.73, 166.14, 155.39, 155.29, 153.46, 151.55, 138.47, 126.17, 124.86, 123.66, 122.98, 116.13, 110.06, 70.98, 55.61, 52.54. Mass spectrum m/z: found 302.11 [M + H]+, 324.09 [M + Na]+; calcd, 302.10 [M + H]+, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 98% pure.
Methyl 6-((2-Formyl-4-methoxyphenoxy)methyl)picolinate (3).
The title compound was prepared following the general procedure as a white solid in 82% yield. IR (Diamond, cm−1): 2953, 2851, 1720, 1682, 1618, 1585, 1494, 1396, 1369, 1296, 1222, 1169, 1141; 1H NMR (400 MHz, DMSO-d6): δ 10.47 (s, 1H), 8.03 (m, 2H), 7.87 (d, J = 7.32 Hz, 1H), 7.26 (m, 3H), 5.38 (s, 2H), 3.90 (s, 3H), 3.76 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 189.03, 164.99, 156.95, 154.68, 153.59, 146.99, 138.57, 125.05, 125.01, 123.95, 123.84, 115.99, 110.76, 71.08, 55.59, 52.39. Mass spectrum m/z: found 302.10 [M + H]+, 324.08 [M + Na]+; calcd, 302.10 [M + H]+, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 98% pure.
Methyl 2-((2-Formyl-4-methoxyphenoxy)methyl)isonicotinate (4).
The title compound was prepared following the general procedure as a white solid in 82% yield. IR (Diamond, cm−1): 2923, 1729, 1686, 1608, 1566, 1450, 1382, 1292, 1276, 1216, 1190, 1171; 1H NMR (400 MHz, DMSO-d6): δ 10.43 (s, 1H), 8.81 (dd, J = 5, 0.56 Hz, 1H), 8.02 (s, J = 1.44 Hz, 1H), 7.80 (dd, J = 5.04, 1.56 Hz, 1H), 7.24 (m, 3H), 5.41 (s, 2H), 3.91 (s, 3H), 3.76 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.81, 165.02, 157.80, 154.78, 153.63, 150.44, 137.79, 125.09, 122.92, 121.82, 120.33, 116.25, 110.73, 71.27, 55.58, 52.81. Mass spectrum m/z: found 302.10 [M + H]+, 324.08 [M + Na]+; calcd, 302.10 [M + H]+, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 98% pure.
Methyl 3-((2-Formyl-4-methoxyphenoxy)methyl)picolinate (5).
The title compound was prepared following the general procedure as a pale yellow solid in 80% yield. IR (Diamond, cm−1): 2897, 1712, 1671, 1607, 1567, 1490, 1445, 1398, 1365, 1275, 1232, 1165, 1139, 1106; 1H NMR (400 MHz, DMSO-d6): δ 10.35 (s, 1H), 8.65 (dd, J = 4.6, 1.28 Hz, 1H), 8.2 (dd, J = 7.84, 0.76 Hz, 1H), 7.66 (dd, J = 7.88, 4.68 Hz, 1H), 7.24 (m, 3H), 5.49 (s, 2H), 3.83 (s, 3H), 3.77 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.81, 166.12, 154.66, 153.55, 148.49, 146.69, 136.89, 133.04, 126.43, 124.89, 122.96, 115.79, 110.68, 67.44, 55.56, 52.34. Mass spectrum m/z: found 324.08 [M + Na]+; calcd, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 98% pure.
Methyl 5-((2-Formyl-4-methoxyphenoxy)methylpicolinate) (6).
The title compound was prepared following the general procedure as a pale yellow solid in 80% yield. IR (Diamond, cm−1): 2962, 2864, 2767, 1719, 1684, 1673, 1588, 1489, 1458, 1395, 1355, 1274, 1218, 1198, 1150, 1127; 1H NMR (400 MHz, DMSO-d6): δ 10.40 (s, 1H), 8.84 (d, J = 1.36 Hz, 1H), 8.10 (m, 2H), 7.28 (m, 2H), 7.2 (d, J = 2.92 Hz, 1H), 5.39 (s, 2H), 3.89 (s, 3H), 3.76 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.77, 154.89, 154.57, 148.62, 135.93, 135.67, 135.93, 135.67, 125.92, 125.17, 123.28, 114.92, 111.30, 68.52, 55.90, 52.97. Mass spectrum m/z: found 302.10 [M + H]+, 324.09 [M + Na]+; calcd, 302.10 [M + H]+, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 98% pure.
Methyl 5-((2-Formyl-4-methoxyphenoxy)methyl)nicotinate (7).
The title compound was prepared following the general procedure as a white solid in 82% yield. IR (Diamond, cm−1): 2969, 1718, 1670, 1602, 1572, 1498, 1435, 1405, 1382, 1279, 1217, 1185, 1160, 1119; 1H NMR (400 MHz, DMSO-d6): δ 10.37 (s, 1h), 9.06 (d, J = 2 Hz, 1H), 8.97 (d, J = 2.04 Hz, 1H), 8.42 (t, J = 2.04 Hz, 1H), 7.29 (m, 2H), 7.2 (d, J = 3.08 Hz, 1H), 5.38 (s, 2H), 3.9 (s, 3H), 3.77 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 188.87, 165.04, 154.64, 153.64, 152.90, 149.49, 135.99, 132.63, 125.44, 125.13, 122.88, 116.21, 110.71, 67.83, 55.58, 52.49. Mass spectrum m/z: found 324.08 [M + Na]+; calcd, 324.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 6-((2-Formyl-3-hydroxyphenoxy)methyl)nicotinate (8).
The title compound was prepared following the general procedure as a white solid in 58% yield. IR (Diamond, cm−1): 3299, 2959, 1726, 1694, 1620, 1458, 1437, 1382, 1343, 1285, 1233, 1172, 1122; 1H NMR (400 MHz, DMSO-d6): δ 11.73 (s, 1H), 10.45 (s, 1H), 9.09 (s, 1H), 8.34 (d, J = 8.12, 1H), 7.81 (d, J = 8.28 Hz, 1H), 7.51 (t, J = 8.4 Hz, 1H), 6.67 (d, J = 8.52 Hz, 1H), 6.56 (d, J = 8.36 Hz) 5.41 (s, 2H), 3.90 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 193.85, 165.39, 163.86, 160.84, 160.41, 150.66, 138.40, 138.12, 125.47, 120.52, 111.05, 110.86, 102.24, 70.91, 52.47. Mass spectrum m/z: found 288.08 [M + H]+, 310.08 [M + Na]+; calcd, 288.08 [M + H]+, 309.06 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 2-((2-Formyl-3-hydroxyphenoxy)methyl)nicotinate (9).
The title compound was prepared following the general procedure as a pale yellow solid in 58% yield. IR (Diamond, cm−1): 2956, 1713, 1618, 1637, 1571, 1459, 1435, 1396, 1370, 1287, 1238, 1170, 1141; 1H NMR (400 MHz, DMSO-d6): δ 11.67 (s, 1H), 10.19 (s, 1H), 8.75 (dd, J = 4.8, 1.68 Hz, 1H), 8.25 (dd, J = 7.84, 1.68 Hz, 1H), 7.55 (m, 2H), 6.66 (d, J = 8.2 Hz, 1H), 6.53 (d, J = 8.4 Hz, 1H), 5.58 (s, 2H), 3.79 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 193.68, 166.15, 162.26, 161.44, 155.02, 151.73, 138.75, 138.31, 126.02, 123.60, 110.66, 109.49, 103.51, 70.59, 52.50. Mass spectrum m/z: found 288.09 [M + H]+, 310.07 [M + Na]+; calcd, 288.08 [M + H]+, 309.06 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 6-((2-Formyl-3-hydroxyphenoxy)methyl)picolinate (10).
The title compound was prepared following the general procedure as a pale yellow solid in 60% yield. IR (Diamond, cm−1): 3065, 3010, 2956, 2890, 1738, 1697, 1640, 1614, 1583, 1462, 1454, 1400, 1358, 1297, 1242, 1190, 1171, 1150; 1H NMR (400 MHz, DMSO-d6): δ 11.74 (s, 1H), 10.44 (s, 1H), 8.04 (m, 2H), 7.91 (dd, J = 7.48, 1.12 Hz, 1H), 7.52 (t, J = 8.4 Hz, 1H), 6.7 (d, J = 8 Hz, 1H), 6.56 (d, J = 8.4 Hz, 1H), 5.39 (s, 2H), 3.90 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 193.79, 164.99, 162.43, 160.69, 160.62, 149.59, 138.65, 137.84, 124.81, 121.26, 110.79, 109.81, 103.42, 70.49, 52.41. Mass spectrum m/z: found 288.09 [M + H]+, 310.07 [M + Na]+; calcd, [M + H]+, 309.06 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 2-((2-Formyl-3-hydroxyphenoxy)methyl)isonicotinate (11).
The title compound was prepared following the general procedure as a pale yellow solid in 58% yield. IR (Diamond, cm−1): 2961, 1733, 1716, 1639, 1620, 1577, 1460, 1441, 1373, 1342, 1293, 1235, 1215, 1174; 1H NMR (400 MHz, DMSO-d6): δ 11.68 (s, 1H), 10.39 (s, 1H), 8.81 (dd, J =5, 0.6 Hz, 1H), 8.03 (d, J = 0.56 Hz, 1H), 7.81 (dd, J = 5, 1.56 Hz 1H), 7.51 (t, J = 8.4 Hz, 1H), 5.41 (s, 2H), 3.91 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 193.51, 165.02, 162.37, 160.80, 157.43, 150.43, 138.66, 137.84, 121.89, 120.38, 110.83, 109.74, 103.56, 70.69, 52.83. Mass spectrum m/z: found [M + H]+, 310.07 [M + Na]+; calcd, 288.08 [M + H]+, 309.06 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 3-((2-Formyl-3-hydroxyphenoxy)methyl)picolinate (12).
The title compound was prepared following the general procedure as a pale yellow solid in 54% yield. IR (Diamond, cm−1): 3092, 2923, 2851, 1774, 1712, 1632, 1599, 1566, 1515, 1478, 1366, 1276, 1231, 1180, 1136, 1072; 1H NMR (400 MHz, DMSO-d6): δ 11.72 (s, 1H), 10.32 (s, 1H), 8.65 (dd, J = 4.64, 1.52 Hz, 1H), 8.22 (m, 1H), 7.66 (dd, J = 7.88, 4.64 Hz, 1H), 7.54 (t, J = 8.4 Hz, 1H), 6.66 (d, J = 8 Hz, 1H), 6.57 (d, J = 8.4 Hz, 1H), 5.51 (s, 2H), 3.84 (s, 3H), 13C NMR (100 MHz, DMSO-d6): δ 193.59, 166.09, 162.46, 160.68, 148.53, 146.54, 138.72, 136.83, 132.78, 126.44, 110.74, 109.79, 103.26, 67.04, 52.35. Mass spectrum m/z: found 310.08 [M + Na]+; calcd, 309.06 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 5-((2-Formyl-3-hydroxyphenoxy)methyl)picolinate (13).
The title compound was prepared following the general procedure as a white solid in 54% yield. IR (Diamond, cm−1): 2954, 1731, 1679, 1644, 1618, 1574, 1458, 1440, 1392, 1357, 1289, 1249, 1177, 1144, 1122; 1H NMR (400 MHz, DMSO-d6): δ 11.74 (s, 1H), 10.37 (s, 1H), 8.86 (s, 1H), 8.12 (m, 2H), 7.54 (t, J = 8.4 Hz, 1H), 6.72 (d, J = Hz, 1H), 6.56 (d, J = 8.4 Hz, 1H), 5.40 (s, 2H), 3.89 (s, 3H); 13C NMR (100 MHz, DMSO-d6): δ 193.83, 162.42, 160.69, 148.72, 147.01, 138.64, 136.32, 135.91, 135.78, 124.64, 109.79, 103.37, 67.31, 52.39. Mass spectrum m/z: found 310.08 [M + Na]+; calcd, 310.08 [M + Na]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Methyl 5-((2-Formyl-3-hydroxyphenoxy)methyl)nicotinate (14).
The title compound was prepared following the general procedure as a white solid in 54% yield. IR (Diamond, cm−1): 2907, 1719, 1643, 1617, 1584, 1460, 1429, 1397, 1370, 1292, 1246, 1179, 1152, 1121;1H NMR (400 MHz, DMSO-d6): δ 11.72 (s, 1H), 10.33 (s, 1H), 9.05 (d, J = 1.8 Hz, 1H), 8.99 (d, J = 1.76 Hz, 1H), 8.43 (t, J = 2 Hz, 1H), 7.55 (t, J = 8.2 Hz, 1H), 6.73 (d, J = 8.2 Hz, 1H), 6.56 (d, J = 8.44 Hz, 1H), 5.39 (s, 2H), 3.91 (s, 3H); 13C NMR (100 MHz, DMSO-β): δ 193.74, 176.82, 165.04, 162.40, 160.72, 152.87, 149.53, 138.63, 135.99, 132.32, 110.80, 109.76, 103.42, 67.28, 52.49. Mass spectrum m/z: found 288.08 [M + H]+; calcd, 288.08 [M + H]+. The purity of the compound was checked by HPLC and was found to be 99% pure.
Blood.
Normal whole blood was collected from adult donors at the Virginia Commonwealth University after informed consent, in accordance with regulations of the IRB for Protection of Human Subjects. Hb was purified from discarded normal blood samples using standard procedures.62 Leftover blood samples from individuals with homozygous SS were obtained and utilized, based on an approved IRB protocol at the Children’s Hospital of Philadelphia, with informed consent.
Oxygen Equillibrium Curve Studies.
The OEC study to determine the effect of the compounds on Hb affinity for oxygen was conducted following the standard procedure.34,41 Whole blood (30% hematocrit) was incubated in the absence or presence of compounds (0.5, 1.0, and 2.0 mM) solubilized in DMSO. Following, the blood-compound samples were incubated in an IL 237 tonometer (Instrumentation Laboratories, Inc., Lexington, MA) for about 7 min at 37 °C against a gas mixture containing O2 concentrations of 0.804, 2.935, and 5.528% and allowed to equilibrate at O2 tensions of 6, 20, and 40 mmHg. After equilibration, the sample was removed via syringe and aspirated into a ABL 700 series table top automated blood gas analyzer (Radiometer America, Inc., Westlake, OH) to determine total hemoglobin (tHb), hematocrit (Hct), pH, pCO2, partial pressure of oxygen (pO2), and the Hb-O2 saturation (sO2) values. The measured values of pO2 and sO2 at each oxygen saturation level were then subjected to nonlinear regression analysis using the software Scientist (Micromath, Salt Lake City UT) with the following equation
This equation was used to calculate P50 and Hill coefficient (N) values. ΔP50 (%) was determined as
For concentration-dependent OEC studies, 0.5, 1.0, and 2 mM concentrations of each compound to be tested were used, whereas for time-dependent OEC studies, 2 mM of each compound was used. Aliquots of each sample was tested at 1, 2, 4, 8, 12, and 24 h to obtain the P50 and ΔP50 values.
Hemoglobin Modification, Oxygen Equilibrium, and Antisickling Studies Using Human Sickle Blood.
Blood suspensions from subjects with homozygous SCD (hematocrit 20%) were incubated under air in the absence or presence of compounds (0.5, 1, and 2 mM) at 37 °C for 1 h to ensure that binding has attained equilibrium. Following, the suspensions were incubated under hypoxic conditions (certified gas mix of 2.5% O2/97.5% N2) at 37 °C for 2 h. Aliquot samples were fixed with 2% glutaraldehyde solution without exposure to air and then subjected to microscopic morphological analysis of bright field images (at 40× magnification) of single layer cells on an Olympus BX40 microscope fitted with an Infinity 2 camera (Olympus) and the coupled Image Capture software. The residual samples were washed in phosphate-buffer saline and hemolyzed in hypotonic lysis buffer for subsequent analyses.38,41
For oxygen equilibrium studies, approximately, 100 μL aliquot samples from the above-clarified lysate were added to 4 mL of 0.1 M potassium phosphate buffer, pH 7.0, in cuvettes and subjected to hemoximetry analysis using the Hemox Analyzer (TCS Scientific Corp., New Hope, PA) to assess P50 shifts. Degree of Hb-O2 affinity shift (ΔP50) was expressed as percentage fractions of control DMSO-treated samples.
Finally, for the Hb adduct formation studies, the above-clarified lysates were subjected to cation-exchange HPLC (Hitachi D-7000 Series, Hitachi Instruments, Inc., San Jose, CA), using a weak cation-exchange column (Poly CAT A: 30 mm × 4.6 mm, Poly LC, Inc., Columbia, MD). A commercial standard consisting of approximately equal amounts of composite HbF, HbA, HbS, and HbC (Helena Laboratories, Beaumont, TX) was utilized as reference isotypes. The areas of new peaks, representing HbS adducts, were obtained, calculated as percentage fractions of total Hb area and reported as levels of modified Hb.
Stability Studies of Compounds Using Whole Blood.
Normal whole blood (hematocrit 30%) was incubated in the absence and presence of 0.5 and 1 mM of compounds 9 and 10. Aliquots were removed at 0, 4, 8, and 24 h of incubation, and deproteinization was carried out using 0.1 M formic acid in acetonitrile (HPLC grade) at 60–65 °C for 30 min. Following this, the samples were centrifuged at 12,000 rpm for 20 min to obtain a clear supernatant which was sequentially filtered through 0.43 and 0.22 μM filters to further clean the samples for HPLC analysis. Compounds 9, 10, and the corresponding carboxylic acid derivative of compound 10 were used as the positive controls, while blood in the absence of any compound was used as the negative control. HPLC analysis was carried out at 261 nm wavelength using the Phenomenex Luna C18 5μ 100A 150 × 4.6 mm column with a gradient consisting 0.1% formic acid in water (Buffer A) and 0.1% formic acid in acetonitrile (Buffer B) and a flow rate of 1.5 mL/min. Elution times for compounds and their metabolites were approximately 9.6 and 11.8 min, respectively.
X-ray Crystallography.
Co-crystals of liganded Hb with compounds 6 and 9 were obtained following similar reported procedures.35,39 In short, freshly made solution of compound in DMSO was added to deoxygenated (deoxy) Hb (30 mg/mL protein) at an Hb tetramer-compound ratio of 1:10. Following, the complex mixture was saturated with carbon monoxide and allowed to incubate for 2 h to form the COHb-compound complex. Sodium cyanoborohydride (NaBH3CN) was then added to this mixture to reduce the Schiff-base adduct formed between the protein and compound to the corresponding irreversible alkylamine covalent bond. The resulting solution was crystallized using 10–20% PEG6000, 100 mM HEPES buffer, and pH 7.4 using the batch method as previously published. Single-cherry red needle crystals were formed in 1–3 days and were used to collect X-ray diffraction data at 100 K using Rigaku MicroMax 007HF X-ray Generator, Eiger R 4M Detector, and Oxford Cobra Cryo-system (The Woodlands, TX). The crystals were first cryoprotected with 80 μL mother liquor mixed with 62 μL of 50% PEG6000. The data set was processed with the CrysAlis PRO 2015 program (Rigaku Oxford Diffraction Ltd. Yarnton, Oxfordshire, England) and the CCP4 suite of programs.63 The crystal structures of the COHb-compound complexes were determined by a molecular replacement method with the Phenix program,53,64 using the native R2-state crystal structure (PDB ID 1BBB) as a search model. The structures were refined using both Phenix and CNS, while model building and correction were carried out using COOT.54,55
In Vivo Pharmacologic Effect of Compounds in Wild-Type Mice.
We tested in vivo pharmacologic effects of compounds 9 and 10 (and TD-7 as control) in C57BL/6 mice. All animal experiments were performed according to the institutional ethical guidelines on animal care and approved by the Institute Animal Care and Use Committee at the Children’s Hospital of Philadelphia (approval number: IAC 19-000113). Equal numbers of male (n = 3) and female (n = 3) mice were used for the study. The animals were treated with a single intraperitoneal (IP) (150 mg/kg) dose of the compounds. Blood samples were obtained prior to treatment (0) and 1, 3, and 6 h posttreatment, via submandibular bleeding, and subjected to hemolysis using standard methods. Clarified lysates, free of cell debris, and RBC ghosts were analyzed by cation-exchange HPLC to determine the levels of drug-modified hemoglobin (adducts) and utilized to conduct oxygen equilibrium studies to determine degrees of shift in P50 values using methodologies described earlier for in vitro studies on human samples.
Statistical Analyses.
All functional and biological assays evaluating antisickling properties, Hb modification, and oxygen affinity changes were conducted in three biological replicates. Results are reported as mean values with standard deviations, from triplicate analyses.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by NIH/NIMHD grant MD009124 (M.K.S.). Structural biology resources were provided by NIH Shared Instrumentation Grant S10OD021756 (M.K.S.) and Virginia General Assembly Higher Education Equipment Trust Fund (HEETF) to Virginia Commonwealth University (M.K.S.).
ABBREVIATIONS
- Hb
hemoglobin
- SCD
sickle cell disease
- HbS
sickle hemoglobin
- RBC
red blood cell
- HU
hydroxyurea
- AEH
allosteric effector of hemoglobin
- ALDH
aldehyde dehydrogenase
- OEC
oxygen equilibrium curve
- SS
homozygous sickle cell trait
- HbA
normal hemoglobin
- pO2
partial pressure of oxygen
- pCO2
partial pressure of carbon dioxide
- sO2
oxygen-saturation
- Hct
hematocrit
- tHb
total hemoglobin
- P50
partial pressure of oxygen at which 50% of hemoglobin is saturated with oxygen
Footnotes
Supporting Information
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.jmedchem.0c01287.
Molecular formula strings (CSV)
OEC of final compounds; final 2Fo – Fc electron density map of COHb-6 and COHb-9 complexes; representative plot for cation-exchange HPLC patterns of modified Hb; spectral data of final compounds (1H and 13C graphs); and purity data of final compounds (HPLC graphs) (PDF)
Accession Codes
The atomic coordinate and structure factor files have been submitted to the Protein Data Bank under accession codes 6XDT and 6XE7 for the complex structures of compounds 6 and 9, respectively. Authors will release the atomic coordinates and experimental data upon article publication.
The authors declare the following competing financial interest(s): Virginia Commonwealth University has filed a patent related to compounds 1-14.
Contributor Information
Piyusha P. Pagare, Department of Medicinal Chemistry, Virginia Commonwealth University, Richmond, Virginia, 23298, United States.
Mohini S. Ghatge, Department of Medicinal Chemistry and The Institute for Structural Biology, Drug Discovery, and Development, School of Pharmacy, Virginia Commonwealth University, Richmond, Virginia 23298, United States
Qiukan Chen, Division of Hematology, The Children’s Hospital of Philadelphia, Philadelphia, Pennsylvania 19104, United States.
Faik N. Musayev, Department of Medicinal Chemistry and The Institute for Structural Biology, Drug Discovery, and Development, School of Pharmacy, Virginia Commonwealth University, Richmond, Virginia 23298, United States
Jurgen Venitz, Department of Pharmaceutics, Virginia Commonwealth University, Richmond, Virginia 23298, United States.
Osheiza Abdulmalik, Division of Hematology, The Children’s Hospital of Philadelphia, Philadelphia, Pennsylvania 19104, United States.
Yan Zhang, Department of Medicinal Chemistry and The Institute for Structural Biology, Drug Discovery, and Development, School of Pharmacy, Virginia Commonwealth University, Richmond, Virginia 23298, United States.
Martin K. Safo, Department of Medicinal Chemistry and The Institute for Structural Biology, Drug Discovery, and Development, School of Pharmacy, Virginia Commonwealth University, Richmond, Virginia 23298, United States.
REFERENCES
- (1).Pauling L; Itano HA; Singer SJ; Wells IC Sickle Cell Anemia, a Molecular Disease. Science 1949, 110, 543–548. [DOI] [PubMed] [Google Scholar]
- (2).Cretegny I; Edelstein SJ Double Strand Packing in Hemoglobin S Fibers. J. Mol. Biol 1993, 230, 733–738. [DOI] [PubMed] [Google Scholar]
- (3).Ferrone FA Polymerization and Sickle Cell Disease: A Molecular View. Microcirculation 2004, 11, 115–128. [DOI] [PubMed] [Google Scholar]
- (4).Ghatge MS; Ahmed MH; Omar ASM; Pagare PP; Rosef S; Kellogg GE; Abdulmalik O; Safo MK Crystal Structure of Carbonmonoxy Sickle Hemoglobin in R-State Conformation. J. Struct. Biol 2016, 194, 446–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Rogers SC; Ross JGC; d’Avignon A; Gibbons LB; Gazit V; Hassan MN; McLaughlin D; Griffin S; Neumayr T; Debaun M; DeBaun MR; Doctor A Sickle Hemoglobin Disturbs Normal Coupling among Erythrocyte O2 Content, Glycolysis, and Antioxidant Capacity. Blood 2013, 121, 1651–1662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Poillon WN; Kim BC; Welty EV; Walder JA The Effect of 2,3-Diphosphoglycerate on the Solubility of Deoxyhemoglobin S. Arch. Biochem. Biophys 1986, 249, 301–305. [DOI] [PubMed] [Google Scholar]
- (7).Poillon W; Kim B 2,3-Diphosphoglycerate and Intracellular PH as Interdependent Determinants of the Physiologic Solubility of Deoxyhemoglobin S. Blood 1990, 76, 1028–1036. [PubMed] [Google Scholar]
- (8).Jensen FB The Dual Roles of Red Blood Cells in Tissue Oxygen Delivery: Oxygen Carriers and Regulators of Local Blood Flow. J. Exp. Biol 2009, 212, 3387–3393. [DOI] [PubMed] [Google Scholar]
- (9).Habara A; Steinberg MH Minireview: Genetic Basis of Heterogeneity and Severity in Sickle Cell Disease. Exp. Biol. Med 2016, 241, 689–696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (10).Thein MS; Igbineweka NE; Thein SL Sickle Cell Disease in the Older Adult. Pathology 2017, 49, 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Chaturvedi S; DeBaun MR Evolution of Sickle Cell Disease from a Life-Threatening Disease of Children to a Chronic Disease of Adults: The Last 40 Years. Am. J. Hematol 2016, 91, 5–14. [DOI] [PubMed] [Google Scholar]
- (12).Nagel RL; Johnson J; Bookchin RM; Garel MC; Rosa J; Schiliro G; Wajcman H; Labie D; Moo-Penn W; Castro O β-Chain contact sites in the haemoglobin S polymer. Nature 1980, 283, 832–834. [DOI] [PubMed] [Google Scholar]
- (13).Rhoda M-D; Martin J; Blouquit Y; Garel M-C; Edelstein SJ; Rosa J Sickle Cell Hemoglobin Fiber Formation Strongly Inhibited by the Stanleyville II Mutation (A78 Asn → Lys). Biochem. Biophys. Res. Commun 1983, 111, 8–13. [DOI] [PubMed] [Google Scholar]
- (14).Benesch RE; Kwong S; Edalji R; Benesch R Alpha Chain Mutations with Opposite Effects on the Gelation of Hemoglobin S. J. Biol. Chem 1979, 254, 8169–8172. [PubMed] [Google Scholar]
- (15).CDC. Data & Statistics on Sickle Cell Disease, CDC; https://www.cdc.gov/ncbddd/sicklecell/data.html (accessed May 2, 2020). [Google Scholar]
- (16).Goldberg MA; Brugnara C; Dover GJ; Schapira L; Lacroix L; Bunn HF Hydroxyurea and Erythropoietin Therapy in Sickle Cell Anemia. Semin. Oncol 1992, 19, 74–81. [PubMed] [Google Scholar]
- (17).Platt OS Hydroxyurea for the Treatment of Sickle Cell Anemia. N. Engl. J. Med 2008, 358, 1362–1369. [DOI] [PubMed] [Google Scholar]
- (18).Sinha CB; Bakshi N; Ross D; Krishnamurti L From Trust to Skepticism: An In-Depth Analysis across Age Groups of Adults with Sickle Cell Disease on Their Perspectives Regarding Hydroxyurea. PLoS One 2018, 13, No. e0199375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Niihara Y; Zerez CR; Akiyama DS; Tanaka KR Oral L-Glutamine Therapy for Sickle Cell Anemia: I. Subjective Clinical Improvement and Favorable Change in Red Cell NAD Redox Potential. Am. J. Hematol 1998, 58, 117–121. [DOI] [PubMed] [Google Scholar]
- (20).Kaufman MB Pharmaceutical Approval Update. P T 2017, 42, 620–621. [PMC free article] [PubMed] [Google Scholar]
- (21).Vichinsky E; Hoppe CC; Ataga KI; Ware RE; Nduba V; El-Beshlawy A; Hassab H; Achebe MM; Alkindi S; Brown RC; Diuguid DL; Telfer P; Tsitsikas DA; Elghandour A; Gordeuk VR; Kanter J; Abboud MR; Lehrer-Graiwer J; Tonda M; Intondi A; Tong B; Howard J A Phase 3 Randomized Trial of Voxelotor in Sickle Cell Disease. N. Engl. J. Med 2019, 381, 509–519. [DOI] [PubMed] [Google Scholar]
- (22).Ataga KI; Kutlar A; Kanter J; Liles D; Cancado R; Friedrisch J; Guthrie TH; Knight-Madden J; Alvarez OA; Gordeuk VR; Gualandro S; Colella MP; Smith WR; Rollins SA; Stocker JW; Rother RP Crizanlizumab for the Prevention of Pain Crises in Sickle Cell Disease. N. Engl. J. Med 2017, 376, 429–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (23).Ferrone FA GBT440 Increases Haemoglobin Oxygen Affinity, Reduces Sickling and Prolongs RBC Half-Life in a Murine Model of Sickle Cell Disease. Br. J. Haematol 2016, 174, 499–500. [DOI] [PubMed] [Google Scholar]
- (24).Safo MK; Kato GJ Therapeutic Strategies to Alter the Oxygen Affinity of Sickle Hemoglobin. Hematol. Oncol. Clin. North Am 2014, 28, 217–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (25).Safo MK; Bruno S Allosteric Effectors of Hemoglobin: Past, Present and Future In Chemistry and Biochemistry of Oxygen Therapeutics; Mozzarelli A, Bettati S, Eds.; John Wiley & Sons, Ltd.: Chichester, West Sussex, England, 2011; pp 285–300. [Google Scholar]
- (26).Godfrey VB; Chen LJ; Griffin RJ; Lebetkin EH; Burka LT Distribution and Metabolism of (5-Hydroxymethyl)Furfural in Male F344 Rats and B6C3F1 Mice after Oral Administration. J. Toxicol. Environ. Health, Part A 1999, 57, 199–210. [DOI] [PubMed] [Google Scholar]
- (27).Vasiliou V; Pappa A; Petersen DR Role of Aldehyde Dehydrogenases in Endogenous and Xenobiotic Metabolism. Chem.-Biol. Interact 2000, 129, 1–19. [DOI] [PubMed] [Google Scholar]
- (28).Yoshida A; Rzhetsky A; Hsu LC; Chang C Human Aldehyde Dehydrogenase Gene Family. Eur. J. Biochem 1998, 251, 549–557. [DOI] [PubMed] [Google Scholar]
- (29).Stern W; Mathews D; McKew J; Shen X; Kato GJ A Phase 1, First-in-Man, Dose-Response Study of Aes-103 (5-HMF), an Anti-Sickling, Allosteric Modifier of Hemoglobin Oxygen Affinity in Healthy Norman Volunteers. Blood 2012, 120, 3210. [Google Scholar]
- (30).Parikh A; Venitz J Novel In vitro Target-site Drug Disposition (TSDD)/Pharmacodynamic (PD) Model for 5-Hydroxymethyl Furfural (5-HMF) in Human Whole Blood.: PII-068. Clin. Pharmacol. Ther 2014, 95. [Google Scholar]
- (31).Pryde DC; Tran T-D; Jones P; Duckworth J; Howard M; Gardner I; Hyland R; Webster R; Wenham T; Bagal S; Omoto K; Schneider RP; Lin J Medicinal Chemistry Approaches to Avoid Aldehyde Oxidase Metabolism. Bioorg. Med. Chem. Lett 2012, 22, 2856–2860. [DOI] [PubMed] [Google Scholar]
- (32).Pryde DC; Dalvie D; Hu Q; Jones P; Obach RS; Tran T-D Aldehyde Oxidase: An Enzyme of Emerging Importance in Drug Discovery. J. Med. Chem 2010, 53, 8441–8460. [DOI] [PubMed] [Google Scholar]
- (33).Nnamani IN; Joshi GS; Danso-Danquah R; Abdulmalik O; Asakura T; Abraham DJ; Safo MK Pyridyl Derivatives of Benzaldehyde as Potential Antisickling Agents. Chem. Biodiversity 2008, 5, 1762–1769. [DOI] [PubMed] [Google Scholar]
- (34).Abdulmalik O; Ghatge MS; Musayev FN; Parikh A; Chen Q; Yang J; Nnamani I; Danso-Danquah R; Eseonu DN; Asakura T; Abraham DJ; Venitz J; Safo MK Crystallographic Analysis of Human Hemoglobin Elucidates the Structural Basis of the Potent and Dual Antisickling Activity of Pyridyl Derivatives of Vanillin. Acta Crystallogr., Sect. D: Biol. Crystallogr 2011, 67, 920–928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (35).Deshpande TM; Pagare PP; Ghatge MS; Chen Q; Musayev FN; Venitz J; Zhang Y; Abdulmalik O; Safo MK Rational Modification of Vanillin Derivatives to Stereospecifically Destabilize Sickle Hemoglobin Polymer Formation. Acta Crystallogr., Sect. D: Struct. Biol 2018, 74, 956–964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Xu GG; Pagare PP; Ghatge MS; Safo RP; Gazi A; Chen Q; David T; Alabbas AB; Musayev FN; Venitz J; Zhang Y; Safo MK; Abdulmalik O Design, Synthesis, and Biological Evaluation of Ester and Ether Derivatives of Antisickling Agent 5-HMF for the Treatment of Sickle Cell Disease. Mol. Pharm 2017, 14, 3499–3511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Abdulmalik O; Deshpande T; Ghatge M; Zhang Y; Venitz J; Parikh A; Safo MK Novel Structurally-Modified Allosteric Effectors of Hemoglobin Exhibit Superior Antisickling Properties. Blood 2014, 124, 218. [Google Scholar]
- (38).Safo MK; Abdulmalik O; Danso-Danquah R; Burnett JC; Nokuri S; Joshi GS; Musayev FN; Asakura T; Abraham DJ Structural Basis for the Potent Antisickling Effect of a Novel Class of Five-Membered Heterocyclic Aldehydic Compounds. J. Med. Chem 2004, 47, 4665–4676. [DOI] [PubMed] [Google Scholar]
- (39).Pagare PP; Ghatge MS; Musayev FN; Deshpande TM; Chen Q; Braxton C; Kim S; Venitz J; Zhang Y; Abdulmalik O; Safo MK Rational Design of Pyridyl Derivatives of Vanillin for the Treatment of Sickle Cell Disease. Bioorg. Med. Chem 2018, 26, 2530–2538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (40).Abraham D; Mehanna A; Wireko F; Whitney J; Thomas R; Orringer E Vanillin, a Potential Agent for the Treatment of Sickle Cell Anemia. Blood 1991, 77, 1334–1341. [PubMed] [Google Scholar]
- (41).Abdulmalik O; Safo MK; Chen Q; Yang J; Brugnara C; Ohene-Frempong K; Abraham DJ; Asakura T 5-Hydroxymethyl-2-Furfural Modifies Intracellular Sickle Haemoglobin and Inhibits Sickling of Red Blood Cells. Br. J. Haematol 2005, 128, 552–561. [DOI] [PubMed] [Google Scholar]
- (42).ASCO Post Staff. FDA Approves Voxelotor for Sickle Cell Disease; FDA, 2019. [Google Scholar]
- (43).Metcalf B; Chuang C; Dufu K; Patel MP; Silva-Garcia A; Johnson C; Lu Q; Partridge JR; Patskovska L; Patskovsky Y; Almo SC; Jacobson MP; Hua L; Xu Q; Gwaltney SL; Yee C; Harris J; Morgan BP; James J; Xu D; Hutchaleelaha A; Paulvannan K; Oksenberg D; Li Z Discovery of GBT440, an Orally Bioavailable R-State Stabilizer of Sickle Cell Hemoglobin. ACS Med. Chem. Lett 2017, 8, 321–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (44).Wireko FC; Abraham DJ X-Ray Diffraction Study of the Binding of the Antisickling Agent 12C79 to Human Hemoglobin. Proc. Natl. Acad. Sci U.S.A 1991, 88, 2209–2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (45).Safo MK; Ahmed MH; Ghatge MS; Boyiri T Hemoglobin-Ligand Binding: Understanding Hb Function and Allostery on Atomic Level. Biochim. Biophys. Acta 2011, 1814, 797–809. [DOI] [PubMed] [Google Scholar]
- (46).Jenkins JD; Musayev FN; Danso-Danquah R; Abraham DJ; Safo MK Structure of Relaxed-State Human Hemoglobin: Insight into Ligand Uptake, Transport and Release. Acta Crystallogr., Sect. D: Biol. Crystallogr 2009, 65, 41–48. [DOI] [PubMed] [Google Scholar]
- (47).Silva MM; Rogers PH; Arnone A A Third Quaternary Structure of Human Hemoglobin A at 1.7 A Resolution. J. Biol. Chem 1992, 267, 17248–17256. [PubMed] [Google Scholar]
- (48).Perutz MF Nature of Haem-Haem Interaction. Nature 1972, 237, 495–499. [DOI] [PubMed] [Google Scholar]
- (49).Perutz MF Hemoglobin Structure and Respiratory Transport. Sci. Am 1978, 239, 92–125. [DOI] [PubMed] [Google Scholar]
- (50).Perutz MF; Wilkinson AJ; Paoli M; Dodson GG The Stereochemical Mechanism of the Cooperative Effects in Hemoglobin Revisited. Annu. Rev. Biophys. Biomol Struct 1998, 27, 1–34. [DOI] [PubMed] [Google Scholar]
- (51).Beddell CR; Goodford PJ; Kneen G; White RD; Wilkinson S; Wootton R Substituted Benzaldehydes Designed to Increase the Oxygen Affinity of Human Haemoglobin and Inhibit the Sickling of Sickle Erythrocytes. Br. J. Pharmacol 1984, 82, 397–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (52).Oksenberg D; Dufu K; Patel MP; Chuang C; Li Z; Xu Q; Silva-Garcia A; Zhou C; Hutchaleelaha A; Patskovska L; Patskovsky Y; Almo SC; Sinha U; Metcalf BW; Archer DR GBT440 Increases Haemoglobin Oxygen Affinity, Reduces Sickling and Prolongs RBC Half-Life in a Murine Model of Sickle Cell Disease. Br. J. Haematol 2016, 175, 141–153. [DOI] [PubMed] [Google Scholar]
- (53).Echols N; Grosse-Kunstleve RW; Afonine PV; Bunkóczi G; Chen VB; Headd JJ; McCoy AJ; Moriarty NW; Read RJ; Richardson DC; Richardson JS; Terwilliger TC; Adams PD Graphical Tools for Macromolecular Crystallography in PHENIX. J. Appl. Crystallogr 2012, 45, 581–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (54).Brünger AT; Adams PD; Clore GM; DeLano WL; Gros P; Grosse-Kunstleve RW; Jiang JS; Kuszewski J; Nilges M; Pannu NS; Read RJ; Rice LM; Simonson T; Warren GL Crystallography & NMR System: A New Software Suite for Macromolecular Structure Determination. Acta Crystallogr., Sect. D: Biol. Crystallogr 1998, 54, 905–921. [DOI] [PubMed] [Google Scholar]
- (55).Emsley P; Lohkamp B; Scott WG; Cowtan K Features and Development of Coot. Acta Crystallogr., Sect. D: Biol. Crystallogr 2010, 66, 486–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (56).Kato GJ New Insights into Sickle Cell Disease: Mechanisms and Investigational Therapies. Curr. Opin. Hematol 2016, 23, 224–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (57).Drewry JA; Fletcher S; Hassan H; Gunning PT Novel Asymmetrically Functionalized Bis-Dipicolylamine Metal Complexes: Peripheral Decoration of a Potent Anion Recognition Scaffold. Org. Biomol. Chem 2009, 7, 5074–5077. [DOI] [PubMed] [Google Scholar]
- (58).De LS; Liu W; Lo HY; Nemoto PA Benzimidazole Inhibitors of Leukotriene Production. WO2012061169A1, October 5, 2012. [Google Scholar]
- (59).Zhang D; Bousquet B; Mulatier J-C; Pitrat D; Jean M; Vanthuyne N; Guy L; Dutasta J-P; Martinez A Synthesis, Resolution, and Absolute Configuration of Chiral Tris(2-Pyridylmethyl)Amine-Based Hemicryptophane Molecular Cages. J. Org. Chem 2017, 82, 6082–6088. [DOI] [PubMed] [Google Scholar]
- (60).Cheng L; Schell P Isoxazol-3(2h)-One Analogs as Plasminogen Inhibitors and Their Use in the Treatment of Fibrinolysis Related Diseases. WO2012047156A1, December 4, 2012. [Google Scholar]
- (61).Gaillard P; Seenisamy J; Liu-Bujalski L; Caldwell RD; Potnick J; Qiu H; Neagu C; Jones R; Won AC; Goutopoulos A; Sherer B; Johnson TL; Gardberg A Heteroaryl Compounds as Btk Inhibitors and Uses Thereof. U.S. Patent 2,016,096,834 A1, October 6, 2015.
- (62).Safo MK; Abraham DJ X-ray Crystallography of Hemoglobins Hemoglobin Disorders; Methods in Molecular Biology; Humana Press, Totowa, NJ, USA, 2003; pp 1–19. [DOI] [PubMed] [Google Scholar]
- (63).Winn MD; Ballard CC; Cowtan KD; Dodson EJ; Emsley P; Evans PR; Keegan RM; Krissinel EB; Leslie AGW; McCoy A; McNicholas SJ; Murshudov GN; Pannu NS; Potterton EA; Powell HR; Read RJ; Vagin A; Wilson KS Overview of the CCP4 suite and current developments. Acta Crystallogr., Sect. D: Biol. Crystallogr 2011, 67, 235–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (64).Adams PD; Afonine PV; Bunkóczi G; Chen VB; Echols N; Headd JJ; Hung L-W; Jain S; Kapral GJ; Grosse Kunstleve RW; McCoy AJ; Moriarty NW; Oeffner RD; Read RJ; Richardson DC; Richardson JS; Terwilliger TC; Zwart PH The Phenix Software for Automated Determination of Macromolecular Structures. Methods 2011, 55, 94–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.