Abstract
Objective This study aimed to describe two cases of acute respiratory distress syndrome (ARDS) secondary to novel coronavirus disease 2019 (COVID-19) in pregnant women requiring extracorporeal membrane oxygenation (ECMO), and resulting in premature delivery.
Study Design The clinical course of two women hospitalized with ARDS due to COVID-19 care in our intensive care (ICU) is summarized; both participants provided consent to be included in this case series.
Results Both women recovered with no clinical sequelae. Neonatal outcomes were within the realm of expected for prematurity with the exception of coagulopathy. There was no vertical transmission to the neonates.
Conclusion This case series highlights that ECMO is a feasible treatment in the pregnant woman with severe COVID-19 and that delivery can be performed safely on ECMO with no additional risk to the fetus. While ECMO carries its natural risks, it should be considered a viable option during pregnancy and the postpartum period.
Key Points
COVID-19 may present with a more severe course in pregnancy.
ECMO may be used in pregnant woman with severe COVID-19.
Delivery can be performed on ECMO without added fetal risk.
Keywords: severe acute respiratory syndrome-coronavirus-2, COVID-19, extracorporeal membrane oxygenation, respiratory distress syndrome, pregnancy
Severe acute respiratory syndrome–coronavirus-2 (SARS-CoV-2) has a variable clinical course, with the majority of patients treated as outpatients and a select group requiring intensive care unit (ICU) admission and ventilatory support. 1 2 Similar to other viral respiratory illnesses, novel coronavirus disease 2019 (COVID-19) infection during pregnancy has been associated with increased ICU admission and need for ventilatory support. 3 4 A pregnancy surveillance study reported a 10% need for respiratory support. 5
Experience with the use of extracorporeal membrane oxygenation (ECMO) in COVID-19 has described mortality rates ranging from 15 to 50%. 6 7 Global outcomes of ECMO in the treatment of COVID-19 are currently being tracked, with over 2,000 cases included to date. 8 Data on ECMO in pregnancy is even more limited. A 2020 systematic review included 221 studies (358 patients) requiring ECMO in pregnancy for all indications, with the most common indication being acute respiratory distress syndrome (ARDS). The study noted an overall 30-day maternal survival rate of 75%, with a 3 to 18% rate of major maternal complications, and 49% rate of preterm delivery. 9 We report the maternal and fetal outcomes of two critically ill pregnant women who required ECMO support for COVID-19.
Case A
Mother
A 27-year-old gravida-3 para-0 African-American woman with obesity presented at 29 6/7 weeks of gestation endorsing productive cough and dyspnea for 1 week. She had a positive nasopharyngeal (NP) reverse-transcript polymerase chain reaction (RT-PCR) swab for severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) and chest radiograph (CXR) notable for extensive bilateral infiltrates ( Fig. 1A ). Fetal status was reassuring. She was admitted, placed on high-flow nasal cannula, and broad-spectrum antibiotics. Her respiratory status worsened, requiring transfer to the ICU on hospital day (HD)-3 and intubation on HD5. In the ICU, she was proned and treated with tocilizumab, remdesivir, and steroids (dexamethasone and betamethasone) and placed on a heparin. On HD7, she was transferred to our quaternary care ICU for continued clinical worsening.
Fig. 1.
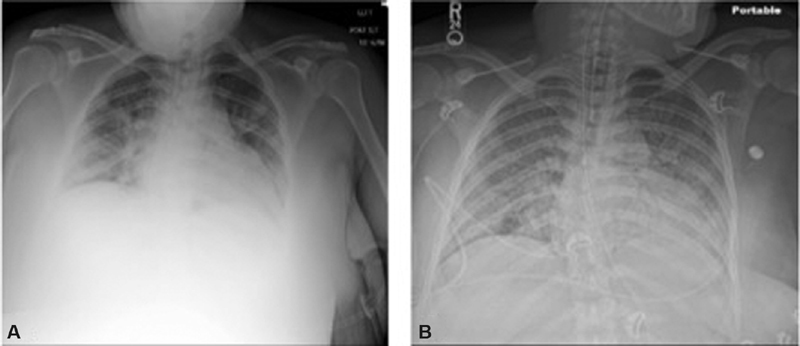
( A ) Initial maternal chest radiograph for case A with extensive bilateral infiltrates. ( B ) Maternal chest radiograph for case A in the ICU with diffuse airspace opacification, likely multifocal pneumonia and ARDS. ARDS, acute respiratory distress syndrome; ICU, intensive care unit.
On presentation to our ICU, she was sedated and paralyzed. Her vital signs were notable for temperature, 37.3°C; heart rate, 110 beats/min; respiratory rate, 28 breaths/min; and SpO 2 , 89% on 100% oxygen. Arterial blood gas (ABG) demonstrated a pH of 7.38, pCO 2 of 43 mm Hg, pO 2 of 71 mm Hg, and HCO 3 of 24.8 mEq/L. She had severe-range blood pressures and an elevated urine protein-to-creatinine ratio (P:Cr) of 0.7. CXR showed diffuse airspace opacification consistent with multifocal pneumonia and ARDS ( Fig. 1B ). Maternal-fetal medicine (MFM) was consulted, and ultrasound demonstrated a live fetus size consistent with estimated dates. Given worsening hypoxia on maximum ventilatory support, the patient was placed on venovenous (VV) ECMO via bilateral femoral veins. She was continued on remdesivir to complete a 5-day course, dexamethasone, and broad-spectrum antibiotics and received convalescent plasma.
On ECMO, her hemoglobin dropped from 12.4 to 9.4 mg/dL, platelets from 279 to 96 mg/dL, fibrinogen from 347 to 150 mg/dL, and creatinine increased from 0.3 to 1.36 mg/dL on HD15. Given thrombocytopenia, heparin-associated platelet antibodies were collected ruling out heparin-induced thrombocytopenia. Given the laboratory findings and sustained severe-range blood pressures, she was diagnosed with HELLP (hemolysis, elevated liver enzymes, and low platelets) syndrome. After discussion with MFM and critical care teams, plan was made to proceed with delivery to possibly improve maternal respiratory status. Heparin was held 6 hours prior to delivery with normalization of the PTT.
On HD15, 31 6/7 weeks of gestation, patient underwent cesarean delivery under general anesthesia with an estimated blood loss (EBL) of 950 cc. Placental pathology demonstrated focal areas of mild villous edema, with no evidence of infection.
Postpartum, her blood pressures improved and she developed disseminated intravascular coagulation (DIC), that required 11 units of packed red blood cells (PRBC), 2 units of platelets, and one unit of cryoprecipitate. Coagulation parameters normalized by HD21/postoperative day 6 (POD6).
On HD18/POD3, the patient was weaned-off of ECMO and decannulated. A tracheostomy was placed on HD19/POD4 and she was weaned to high-flow oxygen. Her hospital course was complicated by Pseudomonas species ventilator-associated pneumonia, for which she received antibiotics. By HD36/POD21, she was transitioned to room air and her tracheostomy was removed. The patient remained on isolation during her entire hospital stay and repeat SARS-CoV-2 RT-PCR testing on HD36/POD21 was negative. The patient was discharged home in stable condition without any oxygen requirements on HD37/POD22 ( Fig. 2A ).
Fig. 2.
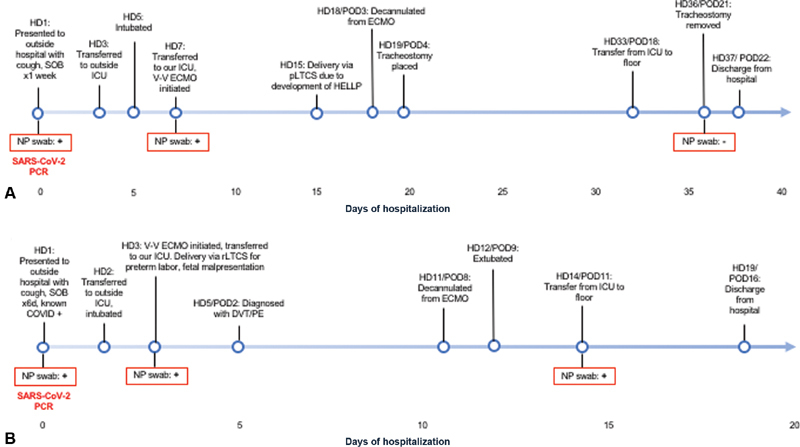
Maternal timeline of events for case A ( A ) and case B ( B ).
Neonate
The baby weighed 1,465 g (21% Fenton) and was initially managed on (continuous positive airway pressure) CPAP, but required escalation to positive-pressure ventilation for poor respiratory effort. Due to continued poor oxygenation and intermittent respiratory effort, the infant was intubated 4 minutes after birth. The 1- and 5-minute Apgar's scores were 4 and 7. After securing the airway and stabilization, the infant was enclosed in the isolette and transferred to the neonatal ICU (NICU). The infant required up to 60% oxygen and ground glass opacities were appreciated on CXR. Surfactant was administered via the endotracheal tube and antibiotics were administered for possible sepsis given clinical risk factors.
The baby had significant abdominal and extremity ecchymoses. Coagulation studies showed international normalized ratio (INR) of 3.8, partial thromboplastin time (PTT) of 52.3 seconds, and fibrinogen of 113 mg/dL. Fresh frozen plasma (FFP) and cryoprecipitate were given. A head ultrasound (HUS) at 12 hours of life showed diffuse periventricular echogenicity, but repeat HUS obtained 7 days after birth was normal. Repeat coagulation studies 24 hours after birth were normal.
SARS-CoV-2 NP RT-PCR were sent at birth, 24 hours, 48 hours, and 7 days after birth which were all negative. SARS-CoV-2 serology studies showed the baby was immunoglobulin (Ig)-G positive. The infant was extubated within 24 hours after birth, and was transitioned to room air within a week. The patient remained in the NICU for routine care for prematurity and had no other significant medical issues outside of those related to prematurity.
Case B
Mother
A 43-year-old gravida-2 para-1 Filipino women at 26 3/7 weeks of gestation with a medical history significant for hypertension, gestational diabetes mellitus, and obesity presented to an outside hospital reporting 6 days of headache, cough, and fever. She had a positive NP swab for SARS-CoV-2 3 days prior to presentation.
On presentation, she was tachypneic and diaphoretic. Respiratory rate was 53 breaths/min, and oxygen saturation was initially 79% and improved to 97% on 15 L/min supplemental oxygen. ABG on 15 L/min oxygen showed pH of 7.4, CO 2 of 36.1 mm Hg, O 2 of 28.7 mm Hg, and HCO 3 of 21.7 mEq/dL. CXR demonstrated patchy bibasilar infiltrates. She was managed with supplemental oxygen and received remdesivir, dexamethasone, and broad-spectrum antibiotics. On HD2, she developed worsening hypoxia and was transferred to the ICU, started on high-flow nasal cannula, and ultimately intubated. Given worsening hypoxia on maximum ventilator settings, the decision was made to proceed with VV ECMO initiation. The patient was cannulated via bilateral femoral veins on HD3 and was transferred to our ICU for ECMO management.
On presentation to our ICU, she was intubated, sedated, and on 100% oxygen on ECMO. Initial blood gas showed CO 2 of 37 mm Hg, O 2 of 97 mm Hg, and oxygen saturation of 97%. CXR was notable for diffuse bilateral lung opacification consistent with COVID-19 pneumonia and acute lung injury ( Fig. 3 ).
Fig. 3.
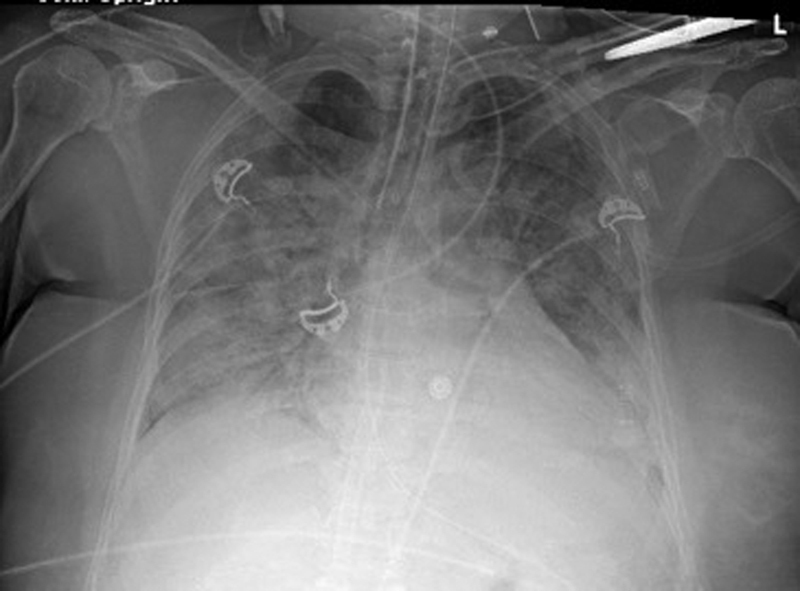
Maternal chest radiograph for case B with diffuse bilateral lung opacification.
Fetal heart rate tracing demonstrated minimal variability, no accelerations, and recurrent late decelerations with regular contractions. The cervix was closed on initial examination. She was started on vasopressors to maintain a mean arterial pressure (MAP) above 65 mm Hg for fetal resuscitation. Over the next few hours, the fetal heart rate tracing remained nonreassuring with recurrent late decelerations. She continued to contract and repeat cervical exam was notable for preterm labor with 4 cm of cervical dilation. Ultrasound demonstrated a fetus in backup transverse presentation. Given preterm labor with malpresentation and nonreassuring fetal heartrate tracing, cesarean delivery was recommended. Heparin was held 2 hours prior to delivery for PTT normalization.
On HD3, at 27 5/7 weeks of gestation, the patient underwent repeat cesarean delivery in the main operating room. A live male infant was delivered under general anesthesia with an EBL of 900 cc. Placental pathology was unremarkable without evidence of acute chorioamionitis, vilitis, or decidual maternal/fetal vasculopathy.
On HD4/POD1, her platelets dropped from 258 to 130K/uL. Urine P:Cr was elevated at 0.4. Despite elevated P:Cr, platelets normalized, blood pressures and serum preeclampsia laboratories were unremarkable. Heparin was restarted 24 hours after delivery. On HD5/POD2, the patient was noted to have asymmetric lower extremity swelling and Doppler demonstrated a deep venous thrombosis from the left popliteal to the common femoral vein. Computed tomography angiography was notable for a right lower lobe lobar and segmental pulmonary embolism, and she was continued on therapeutic anticoagulation with heparin. She was transitioned to enoxaparin and then apixiban, as she did not plan to breastfeed.
On HD11/POD8, she was weaned-off of ECMO and was decannulated. Pseudomonas aeruginosa ventilator-associated pneumonia was treated with antibiotics. She was extubated on HD12/POD9 and weaned-off oxygen completely by HD19/POD16. She remained on isolation through her hospitalization. SARS-CoV-2 RT-PCR testing on HD1, HD3, and HD14 were positive. She was discharged home on HD19/POD16 ( Fig. 2B ).
Neonate
The neonate was delivered weighing 1,000 g (67% Fenton). At delivery, the heart rate was undetectable and positive pressure ventilation was initiated and intubated by 2 minutes after birth. Apgar's scores were 1, 5, 6, and 7 at 1, 5, 10, and 15 minutes. The infant was enclosed in the isolette. Surfactant was administered to the neonate, given the clinical FiO 2 requirement and CXR. Antibiotics were initiated, given the risk factors contributed by the severity of the mother's clinical illness.
Coagulation studies at birth demonstrated INR of 5.0, PTT of 83.9 seconds, and fibrinogen of 123 mg/dL. FFP and cryoprecipitate were administered. HUS obtained at 24 hours and 7 days after birth were both interpreted as normal for gestational age, without evidence of intraventricular hemorrhage.
SARS-CoV-2 NP swabs were sent at birth, 24 and 48 hours, and 7, and 14 after birth and were negative. SARS-CoV-2 serology studies were IgG negative. The patient was removed from isolation 14 days after birth as per infectious disease team consultation. The infant was extubated within 5 days after birth. The patient remains hospitalized for issues related to prematurity but remains clinically well without signs or symptoms of COVID-19 infection.
Discussion
We report two cases of severe ARDS secondary to SARS-CoV-2. During their hospital course, these patients both demonstrated worsening respiratory failure necessitating ECMO. While data are limited, it appears that COVID-19 infection in pregnancy is more likely to result in a severe course compared with nonpregnant patients. 3 4 Both of these patients had preexisting conditions that may have increased their risk for more severe COVID-19-related illness.
Case A was complicated by development of suspected HELLP syndrome. An observational study by Mendoza et al demonstrated that SARS-CoV-2 can produce systemic effects including hypertension, acute kidney injury, thrombocytopenia, and liver injury due to vasoconstriction, in contrast to the endothelial damage secondary to oxidative stress noted in preeclampsia/HELLP syndrome. 10 The development of this “preeclampsia-like” syndrome may be difficult to distinguish due to overlapping clinical features and may potentially lead to overdiagnosis of preeclampsia/HELLP syndrome among pregnant women with COVID-19 infection. Case B also developed low platelets after delivery and similarly had proteinuria; however, thrombocytopenia was felt to be reflective of her acute illness, rather than preeclampsia.
The indication for the deliveries were different in our patients. In case A, delivery timing was based on the development of suspected HELLP syndrome with the lack of improvement of respiratory status. For case B, delivery occurred due to preterm labor, a known risk both with COVID-19 infection and in patients on ECMO. 5 9 Current recommendations suggest that timing and mode of delivery in COVID-19-positive pregnant women be based on routine obstetrical indications, and delivery should not be used as a treatment to improve maternal respiratory symptoms, except in cases of maternal cardiac arrest; however, in the setting of ECMO, while delivery is not recommended to improve maternal respiratory status, the risks and benefits of preterm delivery versus remaining on ECMO for a prolonged period of time must be considered. 11 Pharmacologic treatment options such as remdesivir, interleukin (IL)-6 modulators, and convalescent plasma has not been extensively studied in pregnant women but use is appropriate in critically ill women as compassionate use. 11 12
This case series is consistent with other reports of the low vertical transmission rate of SARS-CoV-2. 13 14 15 16 17 18 19 Clinical presentation of SARS-CoV-2 in infants can range from asymptomatic to neonatal respiratory distress requiring mechanical ventilation. 15 16 17 18 19 20 Here, both infants had negative NP swabs, but one infant had IgG antibodies to COVID-19. It is possible that the presence of IgG antibodies in one infant resulted from transplacental passage given the mother's prolonged clinical illness versus the shorter course and lack of adequate maternal production or placental passage of IgG in our second dyad. 19 Neonatal IgG could be from the maternal convalescent plasma as part of her treatment plan. Placental transfer of convalescent plasma and theoretic immunity of the neonate has not been studied.
In terms of neonatal outcomes after maternal ECMO (for any indication), fetal survival is reported to be around 70%. 9 21 22 23 Reviews on modern use of ECMO in pregnancy have not comprehensively reported on bleeding risk to the neonate. 9 21 22 23 Theoretically, due to the necessity for anticoagulation of the mother while on the ECMO circuit, the neonates had prolonged exposure of in utero anticoagulant, albeit reversed for delivery and heparin does not cross the placenta. Evaluation for abnormal coagulation studies may be advisable in neonates born to mothers with COVID-19, especially given the risk of intraventricular hemorrhage in the extremely or very low birthweight neonates.
Conclusion
We describe two cases of ARDS secondary to COVID-19 in pregnancy leading to ECMO and preterm delivery. Both mothers had a significant improvement in their clinical status after ECMO. Neonatal outcomes were within the realm of expected for prematurity with the exception of coagulopathy. We highlight that ECMO is a feasible treatment in the pregnant woman with severe COVID-19 disease and that delivery can be performed safely on ECMO with no apparent additional risk to the fetus/neonate. While ECMO carries inherent risks, it should be considered a viable option during pregnancy and puerperium.
Acknowledgments
We thank all study participants for agreeing to participate.
Footnotes
Conflict of Interest None declared.
References
- 1.Centers for Disease Control and Prevention Coronavirus disease 2019 (COVID-19): COVIDViewAccessed September 25, 3030 from:https://www.cdc.gov/coronavirus/2019-ncov/covid-data/covidview/index.html
- 2.Lewnard J A, Liu V X, Jackson M L. Incidence, clinical outcomes, and transmission dynamics of severe coronavirus disease 2019 in California and Washington: prospective cohort study. BMJ. 2020;369:m1923. doi: 10.1136/bmj.m1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Favre G, Pomar L, Musso D, Baud D.2019-nCoV epidemic: what about pregnancies? Lancet 2020395(10224):e40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention Data on COVID-19 during Pregnancy 2020. Accessed Aug 19, 2020 at:https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/special-populations/pregnancy-data-on-covid-19.html
- 5.UK Obstetric Surveillance System SARS-CoV-2 Infection in Pregnancy Collaborative Group . Knight M, Bunch K, Vousden N. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107. doi: 10.1136/bmj.m2107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zeng Y, Cai Z, Xianyu Y, Yang B X, Song T, Yan Q. Prognosis when using extracorporeal membrane oxygenation (ECMO) for critically ill COVID-19 patients in China: a retrospective case series. Crit Care. 2020;24(01):148. doi: 10.1186/s13054-020-2840-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jacobs J P, Stammers A H, St Louis J. Extracorporeal membrane oxygenation in the treatment of severe pulmonary and cardiac compromise in coronavirus disease 2019: experience with 32 patients. ASAIO J. 2020;66(07):722–730. doi: 10.1097/MAT.0000000000001185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.ECMO in COVID-19Accessed Aug 19, 2020 at:https://www.elso.org/COVID19.aspx
- 9.Naoum E E, Chalupka A, Haft J. Extracorporeal life support in pregnancy: a systematic review. J Am Heart Assoc. 2020;9(13):e016072. doi: 10.1161/JAHA.119.016072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mendoza M, Garcia-Ruiz I, Maiz N. Pre-eclampsia-like syndrome induced by severe COVID-19: a prospective observational study. BJOG. 2020;127(11):1374–1380. doi: 10.1111/1471-0528.16339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Halscott T, Vaught J, Miller E.Society for Maternal Fetal Medicine. Management considerations for pregnant patients with COVID-19Accessed Aug 19, 2020 at:https://s3.amazonaws.com/cdn.smfm.org/media/2415/SMFM_COVID_Management_of_COVID_pos_preg_patients_7-2-20.PDF_.pdf
- 12.Committee on Ethics . ACOG committee opinion no. 646: ethical considerations for including women as research participants. Obstet Gynecol. 2015;126(03):e100–e107. doi: 10.1097/AOG.0000000000001150. [DOI] [PubMed] [Google Scholar]
- 13.Karimi-Zarchi M, Neamatzadeh H, Dastgheib S A. Vertical transmission of coronavirus disease 19 (COVID-19) from infected pregnant mothers to neonates: a review. Fetal Pediatr Pathol. 2020;39(03):246–250. doi: 10.1080/15513815.2020.1747120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen H, Guo J, Wang C.Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records Lancet 2020395(10226):809–815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Parazzini F, Bortolus R, Mauri P A, Favilli A, Gerli S, Ferrazzi E. Delivery in pregnant women infected with SARS-CoV-2: A fast review. Int J Gynaecol Obstet. 2020;150(01):41–46. doi: 10.1002/ijgo.13166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zeng L, Xia S, Yuan W. Neonatal early-onset infection with SARS-CoV-2 in 33 neonates born to mothers with COVID-19 in Wuhan, China. JAMA Pediatr. 2020;174(07):722–725. doi: 10.1001/jamapediatrics.2020.0878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mullins E, Evans D, Viner R M, O'Brien P, Morris E. Coronavirus in pregnancy and delivery: rapid review. Ultrasound Obstet Gynecol. 2020;55(05):586–592. doi: 10.1002/uog.22014. [DOI] [PubMed] [Google Scholar]
- 18.Savasi V M, Parisi F, Patanè L. Clinical findings and disease severity in hospitalized pregnant women with coronavirus disease 2019 (COVID-19) Obstet Gynecol. 2020;136(02):252–258. doi: 10.1097/AOG.0000000000003979. [DOI] [PubMed] [Google Scholar]
- 19.Kotlyar A, Grechukhina O, Chen A. Vertical transmission of COVID-19: a systematic review and meta-analysis. Am J Obstet Gynecol. 2020 doi: 10.1016/j.ajog.2020.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alzamora M C, Paredes T, Caceres D, Webb C M, Valdez L M, La Rosa M. Severe COVID-19 during pregnancy and possible vertical transmission. Am J Perinatol. 2020;37(08):861–865. doi: 10.1055/s-0040-1710050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nair P, Davies A R, Beca J. Extracorporeal membrane oxygenation for severe ARDS in pregnant and postpartum women during the 2009 H1N1 pandemic. Intensive Care Med. 2011;37(04):648–654. doi: 10.1007/s00134-011-2138-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sharma N S, Wille K M, Bellot S C, Diaz-Guzman E. Modern use of extracorporeal life support in pregnancy and postpartum. ASAIO J. 2015;61(01):110–114. doi: 10.1097/MAT.0000000000000154. [DOI] [PubMed] [Google Scholar]
- 23.Zhang J JY, Ong J A, Syn N L. Extracorporeal membrane oxygenation in pregnant and postpartum women: a systematic review and meta-regression analysis. J Intensive Care Med. 2019 doi: 10.1177/0885066619892826. [DOI] [PubMed] [Google Scholar]


