Abstract
Background
Paroxysmal tonic upgaze (PTU) is an infantile-onset paroxysmal neurological disorder that is characterized by episodes of sustained conjugate upward eye deviation. The paroxysmal abnormal eye movements need to be differentiated from seizures. We report a case of PTU with occipital discharge on electroencephalography (EEG), which made the diagnosis more complicated.
Case presentation
A 6-month-old girl presented with paroxysmal upward deviation or left strabismus of the eyes, with a bowed head, lowered jaw, raised eyebrows, closed lips, and slight grin. Each episode lasted for a few seconds, and episodes occurred multiple times per day. EEG showed spike waves in the right occipital region, and the girl was initially misdiagnosed with epilepsy. After further analysis using video EEG, we corrected her diagnosis as PTU and stopped the administration of an antiepileptic drug.
Conclusion
PTU accompanied by discharge on EEG may lead to a misdiagnosis. Video EEG monitoring, and especially the analysis of EEG traces synchronized with attacks, can provide evidence to distinguish between seizures and non-epileptic events.
Keywords: Paroxysmal tonic upgaze, dystonia, electroencephalography, epilepsy, seizures, childhood, pediatric
Introduction
The symptoms of seizures are diverse, and are similar to those of some episodic dystonia and physiological behaviors. Although the electroencephalography (EEG) of most patients with non-epileptic events are normal, there are sometimes some discharges between attacks. In these cases, it is more likely that the non-epileptic events are misdiagnosed as seizures.
Paroxysmal tonic upgaze (PTU) is an infantile-onset paroxysmal neurological disorder that is characterized by episodes of variably sustained conjugate upward eye deviation, with neck flexion, down-beating saccades in attempted downgaze, and preserved horizontal eye movements.1 Herein, we report a case of PTU who was initially misdiagnosed with epilepsy because of paroxysmal ocular movements and occipital discharge on EEG.
Case report
The girl was born at 40 weeks’ gestation as the first child of healthy, nonconsanguineous Chinese parents. Her birth history was not noteworthy. There was no family history of hereditary disorders such as migraine or episodic ataxia. She had normal developmental milestones.
At 6 months of age, the patient began to exhibit intermittent upward deviation or left strabismus of the eyes, with a bowed head, lowered jaw, raised eyebrows, closed lips, and slight grin. Each episode lasted for a few seconds. These episodes occurred multiple times per day (up to dozens of times), followed by a spontaneous resumption of normal ocular movements. The episodes occurred in small clusters or in isolation, and were not accompanied by any clouding of consciousness. These attacks increased during drowsiness, and disappeared during sleep. There was no history of infection or drug ingestion. In the weeks subsequent to her initial presentation, the episodes increased in number.
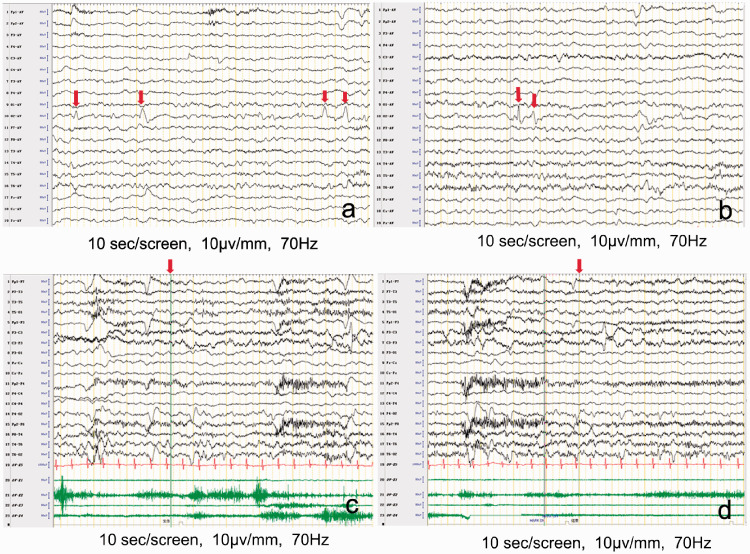
Neurological and ophthalmological examinations were normal. Routine blood test, biochemical examinations, and serum lactate results were normal. Brain magnetic resonance imaging revealed no abnormalities. However, video EEG revealed spike waves of medium amplitude scattered in the right occipital area. The ipsilateral posterior temporal region or the contralateral occipital region were involved. These abnormal discharges were sometimes associated with blinking (Figure 1a,b). In contrast, there was no epileptic electrical activity synchronized with tonic upward gaze episodes (Figure 1c,d).
Figure 1.
Electroencephalography (EEG) between episodes and during an episode. a, b: Scattered spike waves in the occipital region between episodes on EEG. c, d: Normal EEG synchronized with a paroxysmal tonic upgaze episode. Arrows show the start and end points of the episodes.
The patient was initially misdiagnosed with epilepsy because of abnormal discharges in the occipital region and her paroxysmal eye movements. However, after further analysis using video EEG, we corrected her diagnosis as PTU, based on both the normal EEG synchronized with episodes and the typical symptoms of PTU. Following this new diagnosis, we stopped the administration of an antiepileptic drug. The patient’s episodes gradually decreased during the following 2 months, until they had disappeared completely.
We performed whole-exome sequencing and a dynamic mutation test of 10 genes related to ataxia, including CACNA1A (calcium voltage-gated channel subunit alpha-1A gene). The results did not reveal any candidate disease-causing mutations in CACNA1A, GRID2 (glutamate receptor, ionotropic, delta 2 gene), or SEPSECS (O-phosphoseryl-tRNA(Sec) selenium transferase gene). Moreover, the trio-exome analysis did not reveal any protein-altering de novo mutations. However, a search for compound heterozygous variants in this case revealed some uncertain variants (Table 1).
Table 1.
Compound heterozygous variants.
| Gene | Nucleotide changes | Pathogenicity(ACMG) |
|---|---|---|
| GPR56 | c.685(exon 6)G>A | uncertain |
| c.132(exon 4)C>T | uncertain | |
| DOCK7 | c.3207 + 15(IVS26)G>A | uncertain |
| c.874(exon 8)G>A | uncertain | |
| CUBN | c.10577(exon 66)G>C | uncertain |
| c.41(exon 1)C>T | uncertain |
ACMG, American College of Medical Genetics; CUBN, cubilin gene; DOCK7, dedicator of cytokinesis protein 7 gene; GPR56, G protein-coupled receptor 56 gene; IVS26, intron 26.
Discussion
PTU is a childhood syndrome that was originally reported by Ouvrier and Billson in 1988.1–3 It is a kind of paroxysmal dystonia, and is characterized by sudden ocular movements with a sustained upward deviation of the eyes, without disturbance of consciousness. The symptoms of PTU are similar to epileptic attacks of focal seizures or absence in childhood. It is therefore difficult for clinicians to judge whether they are seizures or non-epileptic events. Video EEG monitoring can be used to provide evidence for the diagnosis of PTU.
The results of EEG are normal in most cases of PTU.1 However, in the present case, EEG suggested abnormal discharges in the occipital region. Notably, the discharges were not synchronized with the paroxysmal upgaze.
There are a number of reasons for this child being initially misdiagnosed with epilepsy. In terms of symptomatology, focal seizures in young children are often characterized by sluggishness and decreased movements accompanied by upward gaze;4 these are very similar to PTU symptoms. However, focal seizures are usually accompanied by a reduction in response, and patients may appear pale, cyanotic, and salivating; our patient has never presented these symptoms. In terms of EEG manifestations, occipital discharges occurred during the intervals between attacks in the present case. This is another factor that led to a misdiagnosis. In some normal children, abnormal electrical activity can occur in Rolandic and occipital areas on EEG; this activity is mostly related to genetic and developmental factors.5 Thus, this phenomenon is not direct evidence for the existence of epilepsy. When children present with paroxysmal symptoms, both EEG and the symptoms during episodes are very important for distinguishing between epileptic seizures and non-epileptic events.
To date, the cause of PTU remains unknown. The existence of some familial cases with autosomal recessive and autosomal dominant inheritance suggest a genetic basis. Mutations in CACNA1A, GRID2, and SEPSECS have been reported to be associated with PTU.6–8 A mutation in the CACNA1A gene, causing calcium channelopathy, has been reported in some patients with PTU.6 CACNA1A is also linked to other paroxysmal disorders, such as paroxysmal torticollis of infancy, episodic ataxia, and hemiplegic migraine.6 In addition, a mutation in the GRID2 gene, involving the disturbance of synapse organization, has been reported in two families with spinocerebellar ataxia type 18, progressive cerebellar atrophy, and PTU.7 Furthermore, SEPSECS mutations have been reported to be linked with cerebello–cerebral atrophy and the facultative occurrence of PTU.8
Three cases of PTU have also been reported to have chromosome 15 abnormalities. The first was a 3-month-old infant with partial tetrasomy of chromosome 15.9 The second was a 2-year-old girl with Angelman syndrome caused by a maternal deletion of 15q11-13.10 The third was a 13-year-old girl with Prader–Willi syndrome as a result of a paternal deletion on 15q11-13.11
The reported results of imaging studies for most children with PTU have been normal, with the exception of some cases with hypomyelination or delayed myelination, or with lesions within the oculomotor pathway,12,13 which is mostly located within the midbrain and brainstem.
Most patients with PTU have been reported to follow a benign course, with the resolution of all symptoms within 12 months after onset. However, children with additional symptoms, such as developmental delays or ataxia, appear to have poorer prognoses.3,14,15 In our case, the onset symptoms resolved spontaneously without any medical treatment, and the patient’s development level was normal.
In summary, PTU is a rare paroxysmal dystonia in childhood. Video EEG monitoring, and especially EEG analysis synchronized with PTU episodes, can provide evidence to distinguish between seizures and non-epileptic events.
Footnotes
Ethics statement: This study was approved by the Clinical Research Ethics Committee of the First Hospital of Jilin University, China (reference number 2020-394) and the patient’s parents.
Declaration of conflicting interest: The authors declare that there is no conflict of interest.
Funding: This study was supported by the Department of Science and Technology of Jilin Province of China (20180101159JC, 20200201452JC).
ORCID iD: Yan-Feng Zhang https://orcid.org/0000-0002-3301-6784
References
- 1.Ouvrier RA, Billson F. Benign paroxysmal tonic upgaze of childhood. J Child Neurol 1988; 3: 177–180. DOI: 10.1177/088307388800300305. [DOI] [PubMed] [Google Scholar]
- 2.Kartal A. Paroxysmal tonic upgaze in children: three case reports and a review of the literature. Pediatr Emerg Care 2019; 35: e67–e69. DOI: 10.1097/PEC.0000000000001327. [DOI] [PubMed] [Google Scholar]
- 3.Salmina C, Taddeo I, Falesi M, et al. Paroxysmal tonic upgaze in normal children: a case series and a review of the literature. Eur J Paediatr Neurol 2012; 16: 683–687. DOI: 10.1016/j.ejpn.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 4.Johns Hopkins Medicine. Seizures and epilepsy in children, https://www.hopkinsmedicine.org/health/conditions-and-diseases/epilepsy/seizures-and-epilepsy-in-children. (2020, accessed 25 December 2020).
- 5.Liu X. Clinical electroencephalography. Beijing: People's Medical Publishing House, 2017; 14: 331. [Google Scholar]
- 6.Blumkin L, Leshinsky-Silver E, Michelson M, et al. Paroxysmal tonic upward gaze as a presentation of de-novo mutations in CACNA1A. Eur J Paediatr Neurol 2015; 19: 292–297. DOI: 10.1016/j.ejpn.2014.12.018. [DOI] [PubMed] [Google Scholar]
- 7.Hills LB, Masri A, Konno K, et al. Deletions in GRID2 lead to a recessive syndrome of cerebellar ataxia and tonic upgaze in humans. Neurology 2013; 81: 1378–1386. DOI: 10.1212/WNL.0b013e3182a841a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Agamy O, Ben Zeev B, Lev D, et al. Mutations disrupting selenocysteine formation cause progressive cerebello-cerebral atrophy. Am J Hum Genet 2010; 87: 538–544. DOI: 10.1016/j.ajhg.2010.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Joseph K, Avallone J, Difazio M. Paroxysmal tonic upgaze and partial tetrasomy of chromosome 15: a novel genetic association. J Child Neurol 2005; 20: 165–168. DOI: 10.1177/08830738050200021601. [DOI] [PubMed] [Google Scholar]
- 10.Fukumura S, Watanabe T, Takayama R, et al. Paroxysmal tonic upward gaze complicating Angelman syndrome. Pediatr Neurol 2015; 52: 125–127. DOI: 10.1016/j.pediatrneurol.2014.08.022. [DOI] [PubMed] [Google Scholar]
- 11.Gross I, Gross-Tsur V. Paroxysmal tonic upward gaze at adolescence: a girl with Prader-Willi syndrome. Isr Med Assoc J 2016; 18: 703–704. [PubMed] [Google Scholar]
- 12.Blumkin L, Lev D, Watemberg N, et al. Hypomyelinating leukoencephalopathy with paroxysmal tonic upgaze and absence of psychomotor development. Mov Disord 2007; 22: 226–230. DOI: 10.1002/mds.21277. [DOI] [PubMed] [Google Scholar]
- 13.Senbil N, Yilmaz D, Yuksel D, et al. Paroxysmal tonic upgaze presenting as a clinical isolated syndrome. J Child Neurol 2009; 24: 600–602. DOI: 10.1177/0883073808327836. [DOI] [PubMed] [Google Scholar]
- 14.Deonna T, Roulet E, Meyer HU. Benign paroxysmal tonic upgaze of childhood–a new syndrome. Neuropediatrics 1990; 21: 213–214. DOI: 10.1055/s-2008-1071499. [DOI] [PubMed] [Google Scholar]
- 15.Sousa L, Gonorazky S. [Paroxysmal upgaze deviation syndrome]. Arch Argent Pediatr 2010; 108: e108–e110. DOI: 10.1590/S0325-00752010000500012. [DOI] [PubMed] [Google Scholar]