Abstract
Chimeric antigen receptor-modified T cells (CAR-T) have emerged as a promising immunotherapeutic approach in relapsed/refractory haematolgical malignancies. Broader application is limited by unique toxicities, notably, neurotoxicity (NTX). Language dysfunction is among the most frequent symptoms of NTX, the underlying mechanisms of which remain to be elucidated. Electroencephalogram (EEG) is an important tool to monitor for NTX and may provide insights into language dysfunction.
Aim
We aimed to characterise language dysfunction and define electroencephalographic signatures after CAR-T cell therapy.
Methods
We reviewed the clinical presentation and EEG findings of 20 adult patients presenting with language dysfunction after CAR-T cell infusion. The cohort included a subset of patients treated with investigational CD19-directed CAR-T cells for non-Hodgkin’s lymphoma (n=17), acute lymphoblastic leukaemia (n=1), follicular lymphoma (n=1) and chronic lymphocytic leukaemia (n=1).
Results
Language dysfunction presented within 14 days of CAR-T cell infusion in 16 (84%) patients. Ten (50%) patients had mild word-finding difficulties and 10 (50%) had marked dysphasia with profound word-finding difficulties; the latter were all associated with generalised rhythmic delta activity or generalised periodic discharges on EEG.
Conclusions
Language dysfunction after CAR-T cell therapy is associated with generalised EEG abnormalities.
Keywords: EEG, aphasia, oncology
Introduction
Chimeric antigen receptor-modified T cells (CAR-T) have emerged as a promising immunotherapeutic approach in relapsed and refractory haematological malignancies.1 Broader application is limited by unique toxicities, most markedly, neurotoxicity (NTX).2 Language dysfunction is among the most frequent symptoms of NTX,3 the underlying mechanisms of which remain to be elucidated. Electroencephalography (EEG) is a useful tool to monitor NTX and can provide pathophysiological insights into language dysfunction after CAR-T cell therapy.4
Methods
Participants
We reviewed the clinical presentation and EEGs of 20 adult patients, a subset from a larger cohort, who were all treated with investigational CD19-directed CAR-T cells. The 20 patients included in this subanalysis included all of those who presented with language dysfunction after CAR-T cell infusion from a total of 25 who presented with NTX to our institution between 2016 and 2018 and 17 of whom have previously been reported.3 The timing of onset of language dysfunction was also reviewed. Patients were treated at the Massachusetts General Hospital, USA, and Kings College Hospital, UK, for non-Hodgkin’s lymphoma (n=17), acute lymphoblastic leukaemia (n=1), follicular lymphoma (n=1) and chronic lymphocytic leukaemia (n=1). Patients with central nervous system disease were excluded. Written informed consent was obtained from all patients.
Data collection
The EEG recordings of the 20 patients who developed language dysfunction after CAR-T cell infusion between 2016 and 2018 were analysed retrospectively following institutional database searches. EEG recordings were available from most patients at the time of clinical identification of language dysfunction, including long-term monitoring (LTM; 17 patients, 83%) or a 30 min portable recording (3 patients, 17%) depending on EEG availability at the treating centre. LTM was initiated within 4 hours of language dysfunction identification with the aim to continue until the patient returned to their neurological baseline. EEG electrode placement was according to the 10–20 international system using 21 electrodes with a 240 Hz sampling rate. A notch filter (60 Hz) and bandpass filter (0.5–70 Hz) were applied. Non-physiological artefacts were identified and excluded. The review Software used was the Natus Neuroworks software. Inter-reporter variability was reduced by retrospective re-review of all EEGs by a clinical neurophysiologist (ES) in both referential and bipolar montages and report nomenclature was consistent with the American Clinical Neurophysiology Society (ACNS) guidance.5 Participants who did not have an EEG within 4 hours of clinical identification of language dysfunction were excluded.
Specifics of language dysfunction were determined by detailed retrospective analysis of the patients’ medical records and neurological examination. The type of language dysfunction at presentation (prior to the development of severe NTX), was classified as (1) mild expressive dysphasia (no impairment of comprehension, word-finding difficulties related to one or two words in a sentence or mildly decreased fluency), or (2) marked expressive dysphasia/aphasia (no impairment of language comprehension, disruption of verbal fluency with inability to express more than two words in a sentence or complete inability to verbalise). Aphasia and dysphasia were distinguished from encephalopathy without specific language deficit by detailed neurological examination. The time point of onset and progression of the NTX symptoms was recorded and graded as mild NTX grade 1–2 or severe NTX grade 3–4 as it has been previously reported.3 Available imaging studies of the neuraxis were also reviewed.
Results
EEG monitoring at the time of onset of language dysfunction (prior to the development of severe NTX) was available in 18/20 patients (90%). Two patients (10%) did not have an EEG carried out at this time and were therefore excluded from the EEG analysis.
Language dysfunction presented within 4 days (n=5; 26%), 7 days (n=10; 53%), 14 days (n=16; 84%) and 21 days (n=17; 89%) of CAR-T cell infusion (figure 1). We herein indicated the fraction of patients who had language dysfunction at this specific time point. Two patients (11%) had a recurrence of language dysfunction outside of this 3-week period, at day 90 and day 210 post infusion. Ten (50%) of the patients had mild word-finding difficulties and 10 (50%) had marked dysphasia with word-finding difficulties with only a few words of expressive speech or complete aphasia at initial presentation. Nineteen (95%) of the patients with language dysfunction went on to develop encephalopathic states (severe NTX grade 3–4). The mean duration of language dysfunction was 8 days. The patients were all metabolically and haemodynamically stable and non-infectious at onset of language dysfunction. Five of the patients had myoclonus, six had clinically identified convulsive seizures and one had non-convulsive status epilepticus during the 3-week postinfusion period.
Figure 1.
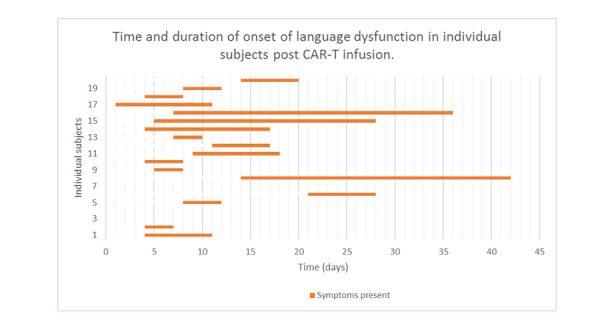
Time and duration of onset of language dysfunction in individual subjects post chimeric antigen receptor-modified T cells (CAR-T) infusion.
At the time of identification of language dysfunction within the first 30 s epoch, the EEG patterns revealed GRDA in 14 (78%) patients, generalised periodic discharges (GPDs) and/or bilateral period discharges in 7 patients (39%), or an alternating pattern between GRDA and GPDs in 6 (33%) patients. Nine (64%) patients at the time of onset of language dysfunction had EEGs which displayed either multifocal or focal sharp waves. The focal sharp waves were bifrontal and independent with shifting hemispheric predominance in four (22%) patients. In three (17%) patients, focal sharp waves were seen over either the right or left frontotemporal region. Most patients (78%) had an EEG signature which was profoundly slowed with GRDA at symptom identification (as recognised by the LTM only); 43% of these had convulsive seizures. The 10 patients who had marked dysphasia all had GRDA at the time of presentation which persisted through the duration of their presentation identified during LTM. None of the patients had a normal EEG at the time of clinical identification of language dysfunction. None of the patients who had a 30 min portable recording had GRDA or GPDs and only focal EEG changes were identified. Fourteen (70%) patients had a brain MRI at the time of identification of language dysfunction, two (10%) had a CT brain scan and four (20%) did not have brain imaging. Of the 14 (70%) patients who had a brain MRI, 2 were abnormal. The first with a pre-existing infarct in the left precentral gyrus which was considered temporally and pathophysiologically unrelated to the presentation of language dysfunction. The second was a patient who had presented with two episodes of expressive dysphasia (day 9 and 6 months post infusion). Both episodes were clinically similar with initial word finding difficulty, progressing to an encephalopathic state. On both occasions, EEG showed prolonged periods of 1–2 Hz GPDs consistent with non-convulsive status epilepticus. A brain MRI done at the 6-month interval showed areas of elevated T2/fluid attenuated inversion recovery (FLAIR) signal without restricted diffusion in the juxtacortical white matter of the right superior parietal lobule which may have influenced his language networks.
Discussion
Our main finding is that language dysfunction after CAR-T cell therapy is associated with generalised EEG abnormalities such as GRDA and GPDs as seen on LTM studies. Patients most commonly developed language dysfunction between 4 and 14 days post CAR-T cell infusion, consistent with the literature.6 The language dysfunction seen included acute onset expressive dysphasia or aphasia. Fifty per cent of patients presented with acute onset aphasia and developed mutism, which has previously been reported.7
All patients had EEG abnormalities at the time of clinical identification of expressive dysphasia or aphasia. Four (22%) patients had isolated focal EEG abnormalities, with three (17%) having epileptiform discharges over the frontotemporal regions. None of the patients with focal epileptiform discharges were reported to have clinical seizures. Most patients (78%) had an EEG signature which was profoundly slowed with GRDA at symptom onset. These generalised changes were identified during LTM (mean duration 106 hours±14) but not in patients undergoing 30 min EEG recording. Six of the patients with this EEG signature were identified as having convulsive seizures. Runs of GPDs or GRDAs may be on the ictal–interictal continuum concerning for electrographic seizures, consistent with the literature.6 All patients identified to have GRDA or GPDs went on to develop encephalopathy (severe NTX grade 3–4). Studies have demonstrated a strong relationship between seizure activity and GPDs, thus those seen in our study may well be ictal; however, the relationship between GRDA and seizures is less clear.8 9 The ACNS guidelines state that any pattern of evolving activity (>4 Hz) can be regarded as ictal5; however, the significance of slower rhythmic activity such as GRDA remains uncertain.9 Patients with GPDs were treated with antiepileptic medications including levetiracetam and many demonstrated neurological improvement thereafter, suggesting that these were epileptiform discharges. GPDs or GRDA in the setting of encephalopathy may be mediated by both cortical or subcortical dysfunction, including T-cell mediated toxicity or cytokine-mediated disruption of the thalamocortical network.4 10 11
Little is known regarding EEG findings in CAR-T patients. However, a recent case series demonstrated the presence of GPDs in four patients on LTM in this context, including one patient with acute onset aphasia as the presenting symptom.4
Two of the patients in our study had a second and delayed presentation of expressive dysphasia: at 90 and 210 days post infusion, both of which were related to a disease relapse. Both recurrent episodes were clinically similar in their presentation with initial word-finding difficulty, progressing to encephalopathy. In the case presenting at 90 days, the repeat LTM showed prolonged periods of 1–2 Hz GPDs consistent with non-convulsive status epilepticus as in the initial study. This patient was loaded with levetiracetam and intravenous benzodiazepine and made an electroclinical recovery within 48 hours. Brain MRI revealed a subtle right superior parietal lobule lesion possibly contributing to the language dysfunction and EEG findings. In the second case of delayed language alteration at 210 days, LTM identified GRDA, similar to the initial EEG.
In selected cases, NTX has been described months after CAR T-cell infusion.3 At 1 year post infusion, 70% of our cohort made good clinical progress. Unfortunately, 30% of our cohort died within 30 days of CAR-T cell therapy, a third of whom had severe NTX grade 3–4 with encephalopathy and GPDs on the EEG at the time of death. As such, GPDs appeared to be associated with worse outcomes.
Most patients had normal cranial brain MR imaging despite marked neurological decline, suggesting a functional rather than structural underlying mechanism.12
Limitations of our study include the retrospective nature of our study, and the initiation of EEG recordings after onset of language dysfunction, although all patients were monitored within 4 hours of clinical identification of language dysfunction. Inter-rater variability also may remain an issue in relation to clinical identification of language dysfunction in a uniform and timely manner. It may be that those described as having acute onset marked dysphasia or aphasia, in fact had milder symptoms several hours earlier. Additionally, it is possible that cortical/subcortical dysfunction had already generalised by the time EEG recording was initiated without identifying a focal onset of EEG abnormality. Moreover, some patients may have had (asymptomatic) EEG abnormalities at baseline prior to CAR-T cell treatment, which cannot be ruled out given the design of our study. LTM had a mean duration of 106 hours which was limited due to resources. Ideally, monitoring would be for the entire duration of clinical symptoms.
It remains unclear why areas of expressive language function are exquisitely vulnerable in the setting of CAR-T mediated NTX. However, 78% (n=14) of our cohort demonstrated more generalised EEG changes, supporting the notion that cortical dysfunction may be a more global than focal phenomenon.10–12
LTM for patients undergoing CAR-T therapy from time of infusion (ideally prior to onset of NTX until resolution of NTX, resource permitting) should be considered to better characterise the temporal and spatial patterns of cortical dysfunction in this vulnerable patient population and guide appropriate management such as initiation of steroids or antiepileptic drugs. A repeat EEG study could be considered if there is a clinical deterioration or symptom recurrence. Ninety-five per cent of patients with language dysfunction at presentation went on to develop grade 3–4 NTX and this presentation may therefore predict more profound NTX. Given that language dysfunction and EEG findings may represent an early finding in CAR-T cell therapy-related NTX, EEG monitoring is emerging as a useful tool in monitoring patients undergoing CAR-T cell therapy.
Footnotes
Contributors: ES and JD are responsible for the manuscript content and can act as guarantors. They planned the work together and were responsible for data interpretation and result analysis, as well as reporting. PK was responsible for data interpretation and result analysis and editing and revision of the manuscript. The other coauthors including RB, RDMD, RCDE, LD, DA, VP, AP, AH, MF, RS, SI and AC contributed to the conduct, reporting and manuscript editing and meet ICMJE criteria for authorship.
Funding: The study was supported by institutional funds from the Massachusetts General Hospital and the HCA International Foundation. JD received support from the American Cancer Society, the American Academy of Neurology and the Amy Gallagher Foundation.
Competing interests: JD is a consultant for Blue Earth Diagnostics and Unum Therapeutics. JD is an author for UpToDate and received royalties from Wolters Kluwer.
Patient consent for publication: Not required.
Ethics approval: IRB approval was obtained for this study and retrospective analysis of electronic records and EEG recordings.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request. The data that support the findings of this study are available from the corresponding author Dr Elisaveta Sokolov upon reasonable request. email: elisaveta.sokolov@nhs.net.
References
- 1.Brudno JN, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for lymphoma. Nat Rev Clin Oncol 2018;15:31–46. 10.1038/nrclinonc.2017.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene Ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–44. 10.1056/NEJMoa1707447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Karschnia P, Jordan JT, Forst DA, et al. Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood 2019;133:2212–21. 10.1182/blood-2018-12-893396 [DOI] [PubMed] [Google Scholar]
- 4.Herlopian A, Dietrich J, Abramson JS, et al. Eeg findings in car T-cell therapy-related encephalopathy. Neurology 2018;91:227–9. 10.1212/WNL.0000000000005910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hirsch LJ, LaRoche SM, Gaspard N, et al. American clinical neurophysiology Society's standardized critical care EEG terminology: 2012 version. J Clin Neurophysiol 2013;30:1–27. 10.1097/WNP.0b013e3182784729 [DOI] [PubMed] [Google Scholar]
- 6.Maus MV, Levine BL. Chimeric antigen receptor T-cell therapy for the community oncologist. Oncologist 2016;21:608–17. 10.1634/theoncologist.2015-0421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rubin DB, Danish HH, Ali AB, et al. Neurological toxicities associated with chimeric antigen receptor T-cell therapy. Brain 2019;142:1334–48. 10.1093/brain/awz053 [DOI] [PubMed] [Google Scholar]
- 8.Rodriguez Ruiz A, Vlachy J, Lee JW, et al. Critical care EEG monitoring research Consortium. Association of periodic and rhythmic electroencephalographic patterns with seizures in critically ill patients. JAMA Neurol 2017;74:181–8. [DOI] [PubMed] [Google Scholar]
- 9.Johnson EL, Kaplan PW. Population of the ictal-interictal zone: the significance of periodic and rhythmic activity. Clin Neurophysiol Pract 2017;2:107–18. 10.1016/j.cnp.2017.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kroschinsky F, Stölzel F, von Bonin S. New drugs, new toxicities: severe side effects of modern targeted and immunotherapy of cancer and their management. intensive care in hematological and oncological patients (iCHOP) Collaborative group. Crit Care 2017;21:89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Foreman B, Claassen J, Abou Khaled K, et al. Generalized periodic discharges in the critically ill: a case-control study of 200 patients. Neurology 2012;79:1951–60. 10.1212/WNL.0b013e3182735cd7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Valand HA, Huda F, Tu RK. Chimeric antigen receptor T-cell therapy: what the Neuroradiologist needs to know. AJNR Am J Neuroradiol 2019;40:766–8. 10.3174/ajnr.A6042 [DOI] [PMC free article] [PubMed] [Google Scholar]


