Abstract
Background
Despite increased experience and technical developments in total knee arthroplasty (TKA), chronic postsurgical pain (CPSP) remains one of physicians’ biggest challenges. The aim of the present study was to evaluate the effectiveness of perineural injection therapy (PIT) in the management of CPSP after TKA.
Material/Methods
A total of 60 patients who had been surgically treated with TKA because of advanced knee osteoarthritis was included in the present study. The study included 2 groups. Group A consisted of patients who received 3 rounds of PIT combined with standard postoperative TKA protocol during the same period. Group B received standard postoperative TKA protocols (rehabilitation programs, oral and intravenous analgesics). Clinical effectiveness was evaluated via Western Ontario and McMaster Universities Arthritis Index (WOMAC) and Visual Analog Scale (VAS) at baseline and 1-, 3-, and 6-month follow-ups.
Results
All repeated measures showed significant improvements (P<0.001) in both groups for VAS and WOMAC scores. These scores were significantly better in group A in all follow-up periods compared with group B (P<0.001). Twenty-nine patients (93.5%) in group A reported excellent or good outcomes compared with 26 patients (89.6%) in group B.
Conclusions
PIT is a promising approach in CPSP with minimal cost, simple and secure injection procedures, minimal side effects, and higher clinical efficacy.
Keywords: Injections, Knee, Osteoarthritis, Pain
Background
Total knee arthroplasty (TKA) is the most successful and widely accepted method in the advanced stages of chronic knee osteoarthritis [1]. Chronic postsurgical pain (CPSP) is defined as moderate to severe pain lasting at least 3 months after surgery [2]. A visual analog scale (VAS) score greater than 40 mm 3 months after surgery indicates significant pain [3]. Despite increased experience and technical developments in this successful method, CPSP remains one of physicians’ biggest challenges [4–6]. At least 3 to 6 months of pain that considerably decreases quality of life after major surgical procedures is referred to as CPSP and it has a frequency varying from 10 to 34% after TKA [7–9]. The exact pathology of CPSP remains unclear. However, cessation of regular analgesia by patients because of intolerance or side effects of analgesics, implant failure, and damage to the sensorial nerves are some possible causes [10,11]. CPSP has a negative influence on life quality and the rehabilitation process and leads to decreased mobilization and joint stiffness. Thus, revision surgeries may become inevitable in some patients [10,12].
Most recent studies have focused on peri-, intra-, and postoperative acute pain control, and there is limited evidence regarding the optimal management of chronic pain after TKA [13–16]. In these limited studies, comprehensive and multidisciplinary approaches and patient-specific treatment modalities were recommended for CPSP [17]. Major treatment modalities for CPSP include medical treatment, physical therapy, pulsed radiofrequency, dry needling, and acupuncture. However, the success rate is low in some of these treatment modalities; scientific evidence is lacking for others [18,19].
Perineural injection therapy (PIT) is a novel restorative injection treatment, refined by Lyftogt [20] for the management of persistent and recurrent pain. In this treatment, 5% buffered dextrose is used near nerve injections to restore nerve function and movement. PIT targets peptidergic small fibers with transient receptor potential vanilloid 1 (TRPV1) expression in the skin and fascial tissues [21]. Voltage gating of TRPV1 stimulates release of proinflammatory neuropeptides calcitonin gene related peptide (CGRP) and substance P (SP) responsible for neurogenic inflammation, resulting in swelling and pain of the nerve trunk [22,23].
Proper management of CPSP improves clinical success and rehabilitation and decreases disability in this patient group [24]. In the present study we hypothesized that PIT could reduce the number of patients with CPSP, alleviate CPSP after TKA, and improve clinical success and the rehabilitation process. The aim of the present study was to evaluate effectiveness of PIT in the management of CPSP after TKA.
Material and Methods
A retrospective review of 60 patients with prospectively collected clinical and radiologic data was performed to evaluate the effectiveness of PIT in the treatment of CPSP after TKA. The local Ethics Committee approved all study protocols. An informed consent was signed by each patient enrolled in the study. Patients who received TKA because of knee osteoarthritis and whose ages varied from 40 to 90 years were included in the study. Patients with active infection, osteomyelitis, history of chronic infection around the knee joint, rheumatic diseases, immune diseases or other systemic inflammatory diseases, patients who had undergone previous operation on the knee, patients with bleeding tendency (hereditary or acquired), and pregnant patients were excluded from the study.
Setting
The present study included a total of 60 patients who had TKA due to knee osteoarthritis in January 2017–July 2018. Thirty-one patients received 3 rounds of PIT combined with standard postoperative TKA protocol consisting of rehabilitation programs, oral and intravenous analgesics (500 mg of acetaminophen every 8 h, 75 mg of diclofenac sodium every 8 h, and 100 mg of tramadol as needed) after TKA (group A), whereas another similar age- and gender-matched group of 29 patients received standard postoperative TKA protocol consisting of rehabilitation programs and oral and intravenous analgesics (500 mg of acetaminophen every 8 h, 75 mg of diclofenac sodium every 8 h and 100 mg of tramadol as needed) after TKA (group B). After 21 days of TKA, both groups of patients were recommended to use acetaminophen up to a maximum of 4 times a day and 500 mg when the pain became unbearable.
Intervention
Total Knee Arthroplasty
All surgeries were performed by the same surgeon (D.G.) using the medial parapatellar approach. Cement was used for fixing the implants. Fixed posterior cruciate ligament-substituting knee prosthesis (Smith-nephew, Geneses II Total Knee Replacement System, USA) was chosen for all patients. After the surgical procedure a compressive dressing was applied, and the knee was immobilized for 24 h. Then, continuous passive-motion and active and passive range-of-motion (ROM) exercises were started and the patients were mobilized the next day. The drain was taken off, and compressive dressing was terminated on the second day. All patients were recommended to use crutches or a walker for walking. The knee ROM exercises and weight bearing were increased gradually.
Perineural Injections and Physiotherapy Program
One researcher who had 10 years of clinical experience in orthopedic surgery and 8 years in prolotherapy (S.A.) performed all injection procedures. The injections were started 21 days after the surgery and repeated every 21 days for 3 seasons. The patients were placed in a supine position and the knee was flexed. Injections were performed under aseptic conditions using a 25-G hypodermic needle. The cutaneous nerves around the knee were palpated along its course and tender chronic constrictive injury (CCI) points (CCI points are tender points in the cutaneous nerves that occur when the nerves trapped in the penetration of the fascial layer at the fascial transition zone) were marked with a skin pen (Figure 1). Three milliliters of 5% dextrose (Koçak Farma, Turkey) solution was administered subcutaneously directly at the marked CCI points and tender points around the knee. All patients were given home exercises by an experienced physiotherapist including stretching, stabilization, and strength training exercises 3 times a week [25]. The patients were recommended to refrain from heavy daily activities and to rest the injected knee for 3 days. Anti-inflammatory drugs were prohibited except for acetaminophen, which could be used a maximum 4 times a day at 500 mg when the pain became unbearable.
Figure 1.
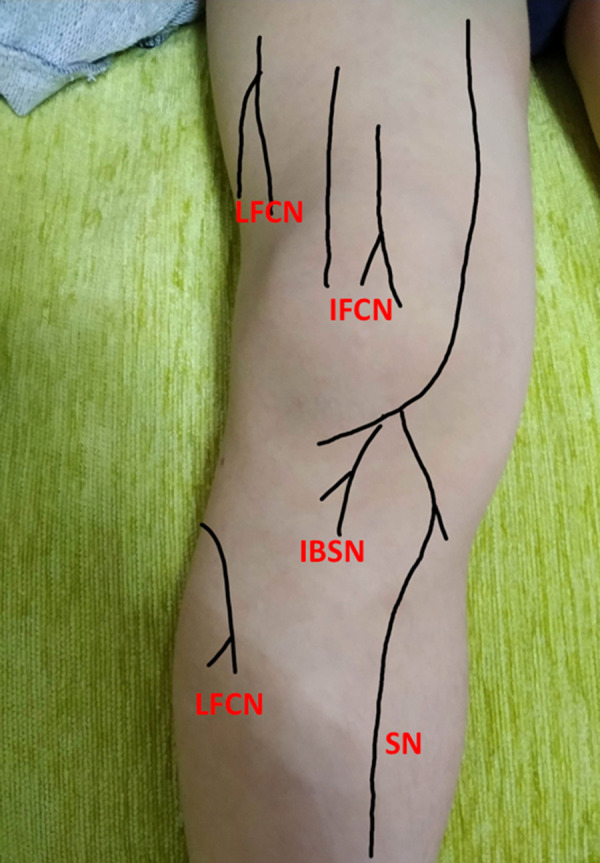
Tender chronic constrictive injury (CCI) points. LFCN – lateral femoral cutaneous nerve; IFCN – intermediate femoral cutaneous nerve; IBSN – infrapatellar branch of saphenous nerve; SN – saphenous nerve.
Outcomes
The outcomes were evaluated through face-to-face interviews by one of the authors (İ.G.) unaware of the treatment procedures. Pain was evaluated in all patients using VAS, in which intensity of the movement-evoked pain varied from 0 (painless) to 10 (extreme pain). Western Ontario and McMaster Universities Arthritis Index (WOMAC) was used to evaluate pain, physical function, and stiffness. This is a 24-item index commonly used for the evaluation of knee osteoarthritis. Follow-up examinations of the cases were performed independently by one of the coauthors at baseline 1 and 3 months after the treatment. The statistician and the evaluator were blind regarding the intervention.
The outcomes in the last follow-up were categorized as “excellent,” “good,” “fair,” or “poor” [26]. The patients were specifically evaluated for significant pain (>40 mm) for 3 months, which is considered an indicator of CPSP development [3].
Excellent: No ankle pain after daily activities or sport.
Good: Pain levels ≤50% of the original ankle pain.
Fair: Pain levels between 50% and 75% of the original ankle pain.
Poor: Pain levels ≥75% of the original ankle pain.
Statistical Analyses
Variables were expressed as mean±SD. Two-way repeated-measures analysis of variance was used for time comparison of group effects. The independent-samples t test was used to compare the data with continuous distribution. Analyses were conducted using a commercial software (IBM SPSS Statistics 19, SPSS Inc., an IBM Co., Somers, NY). P<0.05 was considered statistically significant.
Results
Demographic features of patients in groups A and B were similar (Table 1). All patients completed a minimum of 6-month follow-ups. Initial average VAS and WOMAC scores of the groups were similar (P=0.425 and 0.186, respectively) (Table 2).
Table 1.
General characteristics of variables.
| Variables | Group | P | |
|---|---|---|---|
| A | B | ||
| n | 31 | 29 | – |
| Sex (Male/Female ratio) | 6 (19.3)/25 (80.6) | 3 (10.3)/26 (89.6) | 0.285 |
| Side (right/left) | 17 (54.8)/14 (45.2) | 14 (48.3)/15 (51.7) | 0.309 |
| Age (years) | 67.17±6.8 | 66.17±6.1 | 0.279 |
Data are shown as mean±standard deviation or n (percentage). P – independent-samples t test or chi-square test were used.
Table 2.
Visual Analog Scale (VAS) and Western Ontario and McMaster Universities Arthritis Index (WOMAC) scores of 2 patient groups.
| Variables | Group | Difference (95% CI of difference) | P1 | |
|---|---|---|---|---|
| A | B | |||
| VAS_0 | 8.48±0.76a | 8.44±0.68a | 0.04 (−0.41–0.34) | 0.425 |
| VAS_1mo | 4.48±1.06b | 5.93±0.75b | 1.45 (0.97–1.93) | <0.001 |
| VAS_3mo | 2.32±0.97c | 3.86±1.21c | 1.54 (0.97–2.11) | <0.001 |
| VAS_6mo | 1.51±1.38d | 2.89±1.47d | 1.38 (0.64–2.12) | <0.001 |
| P2 | <0.001 | <0.001 | ||
| WOMAC_0 | 79.29±5.68a | 80.62±5.77a | 1.33 (−1.63–4.29) | 0.186 |
| WOMAC_1mo | 41.80±8.16b | 54.62±7.1b | 12.82 (8.85–16.78) | <0.001 |
| WOMAC _3mo | 24.32±6.52c | 36.24±10.25c | 11.92 (7.51–16.33) | <0.001 |
| WOMAC _6mo | 17.29±5.03d | 25.72±7.09d | 8.43 (5.26–11.59) | <0.001 |
| P2 | <0.001 | <0.001 | ||
Time×group interaction effect for VAS scores (F=23.530; P<0.001).
Time×group interaction effect for WOMAC scores (F=11.503; P<0.001).
| Measure of effect (change). | ||||
|---|---|---|---|---|
| Variables | Group | 95% CI of difference | P1 | |
| A | B | |||
| VAS 0–6 mo | −6.96±1.52 | −5.55±1.50 | 1.41 (0.63–2.20) | <0.001 |
| WOMAC 0–6 mo | −62.00±5.86 | −54.90±7.88 | 7.1 (3.53–10.68) | <0.001 |
| WOMAC subscores. | |||
|---|---|---|---|
| Variables | Group | P1 | |
| A | B | ||
| Pain_0 | 16.35±1.8a | 16.24±1.6a | 0.401 |
| Pain_1mo | 8.83±2.0b | 11.51±1.4b | <0.001 |
| Pain _3mo | 4.7±2.0c | 7.58±2.2c | <0.001 |
| Pain _6mo | 3.09±2.4d | 5.79±2.7d | <0.001 |
| P2 | <0.001 | <0.001 | |
| Stiffness_0 | 6.29±0.9a | 6.37±0.82a | 0.348 |
| Stiffness _1mo | 3.51±0.8b | 4.73±0.5b | <0.001 |
| Stiffness _3mo | 1.87±0.6c | 2.93±0.91c | <0.001 |
| Stiffness _6mo | 1.19±1.0d | 2.1±1.1d | <0.001 |
| P2 | <0.001 | <0.001 | |
| Physical function _0 | 56.67±3.6a | 58.0±4.39a | 0.103 |
| Physical function _1mo | 29.51±5.7b | 38.65±5.4b | <0.001 |
| Physical function _3mo | 17.70±4.1c | 25.72±7.2c | <0.001 |
| Physical function _6mo | 13.87±4.2d | 17.86±5.1d | <0.001 |
| P2 | <0.001 | <0.001 | |
P1 – between-subject comparison; P2 – within-subject comparison.
– in same column, different letters indicate statistically significant difference.
All repeated measures showed significant improvements (P<0.001) in both groups for WOMAC and VAS scores. These scores were significantly better in group A in all follow-up periods compared with group B (P<0.001) (Table 2).
Twenty-nine patients (93.5%) in group A reported good or excellent outcomes (good: n=25 and excellent: n=4) and 2 patients (6.4%) reported poor or fair outcomes (poor: n=1 and fair: n=1). Twenty-six patients (89.6%) in group B reported good or excellent outcomes (good: n=25 and excellent: n=1), whereas 3 patients (10.3%) reported poor or fair outcomes (poor: n=1 and fair: n=2). Two patients (6.4%) in group A and 6 patients (20.6%) in group B had significant pain (>40 mm).
In group B, superficial surgical site infection and wound drainage developed in one patient 3 weeks after TKA operation. This patient fully recovered after a 2-week hospital stay and intravenous ciprofloxacin (400 mg) and oral amoxicillin/clavulanic acid administrations. Insert dislocation occurred in another patient. The insert was replaced and lateral release was performed. In group A, superficial surgical site infection and drainage from the wound site were observed in 2 patients 10 days and 2 weeks after TKA operation. These patients fully recovered after 2 weeks of oral amoxicillin/clavulanic acid administration.
Discussion
CPSP remains one of the major problems after TKA. Unfortunately, there is limited explanation for CPSP and only a few methods are available for its treatment. We used PIT with 5% dextrose injections in the treatment of CPSP and found that this treatment was efficient and safe.
The exact cause of CPSP is not known [18]. Multiple variables are believed to come together in this condition, which develops as a mix of neuropathic pain (caused by a lesion or a disease of the somatosensory nervous system) and persistent muscular and ligamentous spasm and pain (caused by postoperative extreme inflammatory reaction) [23]. Because of tissue damage or a lesion of the somatosensory nervous system, neuropathic pain also coexists with CPSP [27,28]. Phillips et al stated that higher level of pain was the most common symptom in patients who were not satisfied with TKA, and almost half of them had possible neuropathic pain [29]. The National Institute for Health and Clinical Excellence has declared guidelines for the pharmacological treatment of neuropathic pain [30]. As the first-line treatment, use of pregabalin or amitriptyline is recommended. Some patients can benefit from these drugs. However, a considerable percentage of patients are refractory to these methods [19]. Moulin et al stated that only 23.7% of patients with severe neuropathic pain have experienced symptomatic relief with Canadian Pain Society drug protocols for neuropathic pain in their observational study [19]. In the management of this situation there is a need for comprehensive treatment modalities that alleviate neuropathic pain, restore healing, and rearrange postoperative injured muscular and ligamentous structures around the knee. In the present study we used PIT for the management of this problem. Pain and clinical scores were significantly improved compared with baseline (P<0.001). The clinical and pain scores of PIT group were also higher compared with the control group, which received no PIT after standard postsurgical protocol (P<0.001).
There are a limited number of studies dealing with injection therapy in CPSP treatment in the literature. These studies argue that the principles of multimodal therapies combining injection and physical therapy were superior to the unimodal ones [31,32]. In such a study, Núñez-Cortés et al [32] achieved clinically significant improvements in pain, range of motion, function, and myofascial trigger points in TKA patients who had dry needling treatment combined with therapeutic exercises. In parallel with the literature, we adopted the principle of multimodal therapy in the present study. VAS and WOMAC scores of the patients to which this principle was applied were significantly better than those of the group for which this principle was not applied (P<0.001). CPSP rate (significant pain [>40 mm]) of the patients who had PIT treatment dropped to one-third of the CPSP rate of the patients who did not (6.4 vs 20.6%).
Hilton’s law states that, as a result of embryological development, the joint, the muscles that move the joint, and the overlying skin share the same nerve supply [33]. According to this theory, injury of the superficial nerves, including surgical injury, can affect deeper structures. Somatosensory small fibers can be trapped, subjected to friction, and traumatized in the skin and fascial layers around muscles, ligaments, tendons, and joints [34]. The associated ischemia may cause an oxygen-glucose deprivation injury [35]. When C fibers run out of energy they depolarize and start to discharge. MacIver and Tanelian studied the peptidergic C fiber reaction to ischemia in 1992 and found a 652% increase of C fiber firing in response to hypoglycemia [36]. It is proposed that hypoglycemia-induced C fiber firing causes TRPV1 voltage gating. This in turn releases CGRP and SP, triggering neurogenic inflammation with pain and swelling of the nerve trunk. PIT injections have been successfully used for chronic and recurrent pain. The injections target the perineural area, reducing pain and swelling of nerve trunks and restoring nerve function, relieving pain, and improving movement. It has been proposed that the instant analgesic result of 5% dextrose is due to an effect on glucose-sensitive tandem pore potassium channels restoring repolarization [37]. In the present study, we palpated the cutaneous nerves along their course to mark tender nerve entrapments in CCI points. We injected 5% dextrose subcutaneously in all CCIs and tender points, providing significant reduction of pain scores and improved function compared with the control.
PIT has not been used before for CPSP. However, it was used for similar indications. First, Lyftogt used PIT with dextrose for the treatment of 300 patients with Achilles tendinopathy and achieved considerable pain control in these patients [20]. Abu-Zaid et al [38] used perineural injections for severe and moderate knee osteoarthritis in comparison with therapeutic continuous ultrasound and found that physical function, pain, ambulation, disability, and psychological status of the patients were better in perineural groups. Thor et al [39] treated 3 patients with complex regional pain syndrome (a patient with anterior talofibular ligament injury, a patient with shoulder injury, and a patient with a sustained traumatic amputation of fifth and fourth distal interphalangeal joints). All these patients benefited from the treatment and were able to actively participate in therapy and all of their symptoms were healed. In the present study, pain and functional scores of all patients in the PIT treatment group were better compared with those in the no-PIT treatment group. We believe that this therapeutic method holds promise to be more effective in patients with similar indications.
Because of CPSP, some patients discontinue rehabilitation and their mobilization becomes limited. Therefore, joint stiffness and movement limitation develop. In some patients, the lifetime of the prostheses shortens and patients may need revision surgeries [8]. Proper management of CPSP is believed to improve clinical success, rehabilitation, and decrease disability in this patient group [23]. In this context, we achieved significant success not only in pain scores but also in functional scores compared with the control group. With its clinic efficacy, simplicity, cost effectiveness, and safety, PIT is a promising method in the management of postsurgical pain syndromes.
Various concentrations of dextrose solutions have been used successfully in the treatment of musculoskeletal problems for a long time [40–43]. Concentrations up to 25% dextrose (hypertonic) have been used in most previous prolotherapy studies. In these studies, deep injections were applied to tendon-bone attachments, and healing was achieved by regenerative effects of hypertonic dextrose [44–47]. In these studies, some of the patients reported extreme pain, discomfort, and fatigue after the injections because of the inflammatory reaction arising from high concentrations of dextrose [40,48]. Nowadays, lower concentrations of dextrose have been increasingly used in clinical practice. PIT is the most prominent treatment method in this context. Buffered 5% dextrose (isotonic) is injected into the perineural area to relieve chronic and recurrent pain. PIT targets sensory peptidergic C fibers, decreasing TRPV1 activity and neurogenic pain. Repeated injections further improve neural functioning [37,49–51]. Lower concentrations of dextrose are also known to be effective in ligament-tissue healing. In an animal study, Jensen et al [52] showed that lower concentrations of dextrose provided proliferation without an inflammatory reaction. Güran et al [53] compared the effectiveness of lower and higher concentrations of dextrose solutions (low doses of 1%, 5%, and 10%, and high doses of 15%, 20%, and 25%) in vitro using human fibroblast cultures. They showed that in high-dextrose concentrations, up to 80% of fibroblasts died because of toxic conditions. In low-dextrose concentrations, on the other hand, gene expression of angiogenic and apoptotic factors in fibroblasts improved [53]. In the present study, none of the patients complained about excessive pain and fatigue. VAS and WOMAC scores were significantly better than those of the control group. We concluded that PIT provides excellent CPSP pain relief, rapid tissue healing, and functional improvement without excessive inflammation.
There is concern in surgical branches that injection practices might increase infection in patients undergoing surgery [54]. Similar to the previous studies, infection rates due to dextrose injection were minimal in the present study. In some previous studies, dextrose was applied especially in patients who had undergone surgery, and complications such as infection, cellulitis, or septic joint were not observed [55]. In the present study, infection occurred within the first 3 weeks in all groups before the injections. Two patients were completely cured before the injections in group B. We did not observe any indication of an injection-related infection in any of the patients. Therefore, and also in light of the literature, it could be safe to conclude that dextrose would not increase the risk of infection in patients undergoing surgery.
In the present study, fair or poor outcomes were obtained with only 2 patients (6.4%) in group B and 3 patients (poor: n=1 and fair: n=2) (10.3%) in group A. None of these patients experienced any serious complications such as bleeding, infection, cellulitis, or septic joint. In the literature, many factors associated with dissatisfaction such as comorbidity, psychological causes, and neuropathic pain were described in patients with knee prostheses [56,57]. Dissatisfaction rates between 10 to 20% were reported in previous studies [58–60]. What was observed for group A in the present study was similar to the ratios in previous reports. For group B in the present study, these ratios were lower. We assumed that PIT made positive contributions to success rates of TKA.
Limitations of the present study were its retrospective design, small sample size, relatively short follow-up periods, and lack of a placebo control group. Because of the retrospective design of the study, causal relationship between an intervention and observed outcomes could not be established. The effects of perineural injections were part of a combined treatment, so their isolated effects could not be determined. There was no placebo group, so the improvement in pain over time and function could only be explained by the natural history of pain after knee replacement. Patients were not blind to the treatment. Thus, expectation and nonspecific effects of PIT may have contributed to treatment effects. Therefore, more detailed studies with larger cohorts and longer follow-up periods could be useful.
Conclusions
Our study may be the first step in the exploration of an undiscovered field. Many therapeutic effects of dextrose injections have been described in the literature, indicating a variety of underlying mechanisms. Dextrose injections are promising in CPSP with their lower costs, simple and secure injection procedures, minimal side effects, and higher clinical efficacy.
Footnotes
Source of support: Departmental sources
References
- 1.Canovas F, Dagneaux L. Quality of life after total knee arthroplasty. Orthop Traumatol Surg Res. 2018;104:S41–46. doi: 10.1016/j.otsr.2017.04.017. [DOI] [PubMed] [Google Scholar]
- 2.Treede RD, Rief W, Barke A, et al. A classification of chronic pain for ICD-11. Pain. 2015;156:1003–7. doi: 10.1097/j.pain.0000000000000160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brander VA, Stulberg SD, Adams AD, et al. A prospective, observational study. Clin Orthop Relat Res. 2003;416:27–36. doi: 10.1097/01.blo.0000092983.12414.e9. [DOI] [PubMed] [Google Scholar]
- 4.Lenguerrand E, Wylde V, Gooberman-Hill R, et al. Trajectories of pain and function after primary hip and knee arthroplasty: The ADAPT cohort study. PLoS One. 2016;11:e0149306. doi: 10.1371/journal.pone.0149306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Halket A, Stratford PW, Kennedy DM, et al. Using hierarchical linear modeling to explore predictors of pain after total hip and knee arthroplasty as a consequence of osteoarthritis. J Arthroplasty. 2010;25:254–62. doi: 10.1016/j.arth.2009.01.007. [DOI] [PubMed] [Google Scholar]
- 6.Putman S, Argenson JN, Bonnevialle P, et al. Ten-year survival and complications of total knee arthroplasty for osteoarthritis secondary to trauma or surgery: A French multicentre study of 263 patients. Orthop Traumatol Surg Res. 2018;104(2):161–64. doi: 10.1016/j.otsr.2017.11.019. [DOI] [PubMed] [Google Scholar]
- 7.Werner MU, Kongsgaard UEI. Defining persistent post-surgical pain: Is an update required? Br J Anaesth. 2014;113:1–4. doi: 10.1093/bja/aeu012. [DOI] [PubMed] [Google Scholar]
- 8.Fletcher D, Stamer UM, Pogatzki-Zahn E, et al. An observational study. Eur J Anaesthesiol. 2015;32:725–34. doi: 10.1097/EJA.0000000000000319. [DOI] [PubMed] [Google Scholar]
- 9.Beswick AD, Wylde V, Gooberman-Hill R, et al. What proportion of patients report long-term pain after total hip or knee replacement for osteoarthritis? A systematic review of prospective studies in unselected patients. BMJ Open. 2012;2:e000435. doi: 10.1136/bmjopen-2011-000435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Toms AD, Mandalia V, Haigh R, et al. The management of patients with painful total knee replacement. J Bone Joint Surg Br. 2009;91(2):143–50. doi: 10.1302/0301-620X.91B2.20995. [DOI] [PubMed] [Google Scholar]
- 11.Wylde V, Blom AW. The failure of survivorship. J Bone Joint Surg. 2011;93:569–70. doi: 10.1302/0301-620X.93B5.26687. [DOI] [PubMed] [Google Scholar]
- 12.Remérand F, Godfroid HB, Brilhault J, et al. Chronic pain 1 year after foot surgery: Epidemiology and associated factors. Orthop Traumatol Surg Res. 2014;100(7):767–73. doi: 10.1016/j.otsr.2014.07.012. [DOI] [PubMed] [Google Scholar]
- 13.Ng FY, Chiu KY, Yan CH, et al. Continuous femoral nerve block versus patient-controlled analgesia following total knee arthroplasty. J Orthop Surg (Hong Kong) 2012;20(1):23–26. doi: 10.1177/230949901202000105. [DOI] [PubMed] [Google Scholar]
- 14.Marques EM, Jones HE, Elvers KT, et al. Local anaesthetic infiltration for perioperative pain control in total hip and knee replacement: Systematic review and meta-analyses of short- and long-term effectiveness. BMC Musculoskelet Disord. 2014;15:220. doi: 10.1186/1471-2474-15-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wylde V, Dennis J, Beswick AD, et al. Systematic review of management of chronic pain after surgery. Br J Surg. 2017;104:1293–306. doi: 10.1002/bjs.10601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thacoor A, Sandiford NA. Cryotherapy following total knee arthroplasty: What is the evidence? J Orthop Surg (Hong Kong) 2019;27(1):2309499019832752. doi: 10.1177/2309499019832752. [DOI] [PubMed] [Google Scholar]
- 17.Turk DC, Wilson HD, Cahana A. Treatment of chronic non-cancer pain. Lancet. 2011;377:2226–35. doi: 10.1016/S0140-6736(11)60402-9. [DOI] [PubMed] [Google Scholar]
- 18.Vas L, Khandagale N, Pai R. Successful management of chronic postsurgical pain following total knee replacement. Pain Med. 2014;15(10):1781–85. doi: 10.1111/pme.12508. [DOI] [PubMed] [Google Scholar]
- 19.Moulin DE, Clark AJ, Gordon A, et al. Canadian multi-centre cohort study to determine the long-term outcomes of the management of chronic neuropathic pain. Pain Res Manag. 2013;18(2):e39. [Google Scholar]
- 20.Lyftogt J. Subcutaneous prolotherapy for achilles tendinopathy: The best solution? Aust Musculoskeletal Med. 2007;12(2):107–9. [Google Scholar]
- 21.Kruger L, Silverman JD, Mantyh PW, et al. Cutaneous and deep terminations. J Comp Neurol. 1989;280(2):291–302. doi: 10.1002/cne.902800210. [DOI] [PubMed] [Google Scholar]
- 22.Brain SD, Moore DK. Pain and neurogenic inflammation. Birkhäuser; 1999. p. 115. [Google Scholar]
- 23.Malmberg AB, Bley KR. Turning up the heat on pain: TRPV1 receptors in pain and inflammation. Springer Science & Business Media. 2005:39. [Google Scholar]
- 24.Blom AW, Artz N, Beswick AD, et al. Improving patients’ experience and outcome of total joint replacement: The RESTORE programme. Southampton (UK): NIHR Journals Library; 2016. [PubMed] [Google Scholar]
- 25.Brotzman SB, Manske RC. Clinical orthopaedic rehabilitation: An evidence-based approach. 3rd ed. Philadelphia, PA: Elsevier/Mosby; 2011. [Google Scholar]
- 26.Kitaoka HB, Alexander IJ, Adelaar RS, et al. Clinical rating systems for the ankle-hindfoot, midfoot, hallux and lesser toes. Foot Ankle Int. 1994;15(7):349–53. doi: 10.1177/107110079401500701. [DOI] [PubMed] [Google Scholar]
- 27.Wylde V, Beswick A, Bruce J, et al. Chronic pain after total knee arthroplasty. EFORT Open Rev. 2018;3(8):461–70. doi: 10.1302/2058-5241.3.180004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McDermott AM, Toelle TR, Rowbotham DJ, et al. Results from a cross-sectional survey. Eur J Pain. 2006;10:127–35. doi: 10.1016/j.ejpain.2005.01.014. [DOI] [PubMed] [Google Scholar]
- 29.Phillips JR, Hopwood B, Arthur C, et al. The natural history of pain and neuropathic pain after knee replacement: A prospective cohort study of the point prevalence of pain and neuropathic pain to a minimum three-year follow-up. Bone Joint J. 2014;96-B(9):1227–33. doi: 10.1302/0301-620X.96B9.33756. [DOI] [PubMed] [Google Scholar]
- 30.National Institute for Health and Clinical Excellence. NICE guideline 96: Neuropathic pain: The pharmacological management of neuropathic pain in adults in non-specialist settings. Mar, 2010. http://guidance.nice.org.uk/CG96. [PubMed]
- 31.Feinberg BI, Feinberg RA. Persistent pain after total knee arthroplasty. Treatment with manual therapy and trigger point injections. J Musculoskelet Pain. 1998;6:85–95. [Google Scholar]
- 32.Núñez-Cortés R, Cruz-Montecinos C, Vásquez-Rosel Á, et al. Dry needling combined with physical therapy in patients with chronic postsurgical pain following total knee arthroplasty: A case series. J Orthop Sports Phys Ther. 2017;47:209–16. doi: 10.2519/jospt.2017.7089. [DOI] [PubMed] [Google Scholar]
- 33.Hilton J. On rest and pain. 2nd ed. New York: William Wood & Company; 1879. [Google Scholar]
- 34.Lundborg G, Dahlin LB. The pathophysiology of nerve compression. Hand Clin. 1992;8(2):215–27. [PubMed] [Google Scholar]
- 35.Lim TK, Shi XQ, Johnson JM, et al. Peripheral nerve injury induces persistent vascular dysfunction and endoneurial hypoxia, contributing to the genesis of neuropathic pain. J Neurosci. 2015;35(8):3346–59. doi: 10.1523/JNEUROSCI.4040-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.MacIver B, Tanelian D. Activation of C fibers by metabolic perturbations associated with tourniquet ischaemia. Anesthesiology. 1992;76:617–23. doi: 10.1097/00000542-199204000-00020. [DOI] [PubMed] [Google Scholar]
- 37.Burdakov D, Jensen LT, Alexopoulos H, et al. Tandem-pore K+ channels mediate inhibition of orexin neurons by glucose. Neuron. 2006;50(5):711–22. doi: 10.1016/j.neuron.2006.04.032. [DOI] [PubMed] [Google Scholar]
- 38.Abu-Zaid MH, Tabra SA, Elmorsy S. Effect of perineural injection therapy in moderate and severe knee osteoarthritis; A comparative study. Ann Rheum Dis. 2018;77:862–63. [Google Scholar]
- 39.Thor JA, Hanapi NHM, Halil H, et al. A sweet solution to pain. Pain Med. 2017;18:2041–45. doi: 10.1093/pm/pnx063. [DOI] [PubMed] [Google Scholar]
- 40.Akpancar S, Seven MM, Tuzun HY, et al. Current concepts of prolotherapy in orthopedic surgery. Arch Trauma Res. 2017;6(2):1–9. [Google Scholar]
- 41.Rahimzadeh P, Imani F, Faiz SHR, et al. The effects of injecting intra-articular platelet-rich plasma or prolotherapy on pain score and function in knee osteoarthritis. Clin Interv Aging. 2018;13:73–79. doi: 10.2147/CIA.S147757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hung CY, Chang KV, Öz¸akar L. Snapping hip due to gluteus medius tendinopathy: Ultrasound imaging in the diagnosis and guidance for prolotherapy. Pain Med. 2015;16(10):2040–41. doi: 10.1111/pme.12813. [DOI] [PubMed] [Google Scholar]
- 43.Kim WJ, Shin HY, Koo GH, et al. Ultrasound-guided prolotherapy with polydeoxyribonucleotide sodium in ischiofemoral impingement syndrome. Pain Pract. 2014;14(7):649–55. doi: 10.1111/papr.12215. [DOI] [PubMed] [Google Scholar]
- 44.Akpancar S, Gül D. Comparison of platelet rich plasma and prolotherapy in the management of osteochondral lesions of the talus: A retrospective cohort study. Med Sci Monit. 2019;25:5640–47. doi: 10.12659/MSM.914111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ersen O, Koca K, Akpancar S, et al. A randomized-controlled trial of prolotherapy injections in the treatment of plantar fasciitis. Turk J Phys Med Rehab. 2017;64(1):59–65. doi: 10.5606/tftrd.2018.944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Akpancar S, Örsçelik A, Seven MM, et al. The effectiveness of prolotherapy on failed rotator cuff repair surgery. Turk J Phys Med Rehab. 2019;65:1–8. doi: 10.5606/tftrd.2019.3222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gül D, Orsçelik A, Akpancar S. Treatment of osteoarthritis secondary to developmental dysplasia of the hip with prolotherapy injection versus a supervised progressive exercise control. Med Sci Monit. 2020;26:e919166. doi: 10.12659/MSM.919166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rabago D, Slattengren A, Zgierska A. Prolotherapy in primary care practice. Prim Care. 2010;37(1):65–80. doi: 10.1016/j.pop.2009.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Reeves KD, Lyftogt J. Prolotherapy: Regenerative injection therapy. In: Waldman SD, editor. Pain management. 2nd ed. Philadelphia: Saunders (Elsevier); 2011. pp. 1027–44. [Google Scholar]
- 50.Geppetti P, Holzer P. Neurogenic inflammation. Boca Raton, FL: CRC Press; 1996. [Google Scholar]
- 51.Lyftogt J. Pain conundrums: Which hypothesis? Central nervous system sensitization versus peripheral nervous system autonomy. Australas Musculoskel Med. 2008;13(2):72. [Google Scholar]
- 52.Jensen KT, Rabago DP, Best TM, et al. Early inflammatory response of knee ligaments to prolotherapy in a rat model. J Orthop Res. 2008;26(6):816–23. doi: 10.1002/jor.20600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Güran S, Coban ZD, Karasimav O, et al. Dextrose solution used for prolotherapy decreases cell viability and increases gene expressions of angiogenic and apoptotic factors. Gulhane Med J. 2018;60:42–46. [Google Scholar]
- 54.Papavasiliou AV, Isaac DL, Marimuthu R. Infection in knee replacements after previous injection of intra-articular steroid. J Bone Joint Surg Br. 2006;88(3):321–23. doi: 10.1302/0301-620X.88B3.17136. [DOI] [PubMed] [Google Scholar]
- 55.Solmaz İ, Akpancar S, Örsçelik A, et al. Dextrose injections for failed back surgery syndrome. A consecutive case series. Eur Spine J. 2019;28(7):1610–17. doi: 10.1007/s00586-019-06011-3. [DOI] [PubMed] [Google Scholar]
- 56.Lewis GN, Rice DA, McNair PJ, et al. A systematic review and meta-analysis. Br J Anaesth. 2015;114:551–61. doi: 10.1093/bja/aeu441. [DOI] [PubMed] [Google Scholar]
- 57.Singh JA, Lewallen DG. Medical and psychological comorbidity predicts poor pain outcomes after total knee arthroplasty. Rheumatology. 2013;52:916–23. doi: 10.1093/rheumatology/kes402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Scott CE, Oliver WM, MacDonald D, et al. Predicting dissatisfaction following total knee arthroplasty in patients under 55 years of age. Bone Joint J. 2016;98-B:1625–34. doi: 10.1302/0301-620X.98B12.BJJ-2016-0375.R1. [DOI] [PubMed] [Google Scholar]
- 59.Parvizi J, Nunley RM, Berend KR, et al. High level of residual symptoms in young patients after total knee arthroplasty. Clin Orthop Relat Res. 2014;472:133–37. doi: 10.1007/s11999-013-3229-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Aujla RS, Esler CN. Total knee arthroplasty for osteoarthritis in patients less than fifty-five years of age: A systematic review. J Arthroplasty. 2017;32:2598–603. doi: 10.1016/j.arth.2017.02.069. [DOI] [PubMed] [Google Scholar]


