Abstract
Primary intraventricular haemorrhage (PIVH) is an uncommon type of intracerebral haemorrhage, accounting for only 0.31% of all strokes and 3.1% of all intracerebral haemorrhages. Due to the low incidence of PIVH, little is known about its clinical characteristics, risk factors, aetiologies, prognosis and treatment. Acute hydrocephalus is common and is associated with a poor prognosis. External ventricular drainage (EVD) could promptly reduce intracranial pressure by diverting cerebrospinal fluid and intraventricular blood; however, the incidence of complications such as central nervous system infection, catheter occlusion and rebleeding are relatively common. Despite being an invasive procedure, frontal minicraniotomy is an available therapeutic option to avoid complications of EVD. The authors report a case of a PIVH managed with frontal minicraniotomy and perform a literature review about epidemiological data, clinical features and treatment of PIVH.
Keywords: coma and raised intracranial pressure, hydrocephalus
Background
Primary intraventricular haemorrhage (PIVH) is an uncommon type of intracerebral haemorrhage, accounting for only 0.31% of all strokes and 3.1% of all intracerebral haemorrhages.1 PIVH was first described by Sanders in 1881 as a haemorrhagic stroke confined to the ventricular system without laceration of the ventricular walls.2 It is commonly diagnosed in preterm newborns as a complication of birth but is relatively rare in adults.3 Prior to the introduction of CT and MRI, knowledge concerning PIVH was derived from postmortem examination. The development of new brain imaging techniques allows early diagnose and treatment. The low incidence of PIVH is responsible for the lack of information about risk factors, aetiologies, prognosis and treatment of this condition. In fact, the majority of series published in the literature have a small sample size or include traumatic and secondary IVH.
The appropriate treatment for PIVH remains controversial. According to the guidelines of the American Heart Association/American Stroke Association, an external ventricular drain (EVD) is recommended as treatment for acute hydrocephalus in patients with a decreased level of consciousness (Class IIa, Level of Evidence B);4 however, it is associated with relative high rates of rebleeding, central nervous system infection and catheter obstruction.5 6 Direct removal of the intraventricular clot by minicraniotomy is a therapeutic option to manage obstructive acute hydrocephalus related to PIVH; however, little is known about the outcomes of this specific surgical technique in PIVH. In this article, the authors aim to describe the practical use of frontal minicraniotomy in a patient with PIVH and acute hydrocephalus and review the available literature regarding epidemiological data, clinical features and treatment options for PIVH.
Case presentation
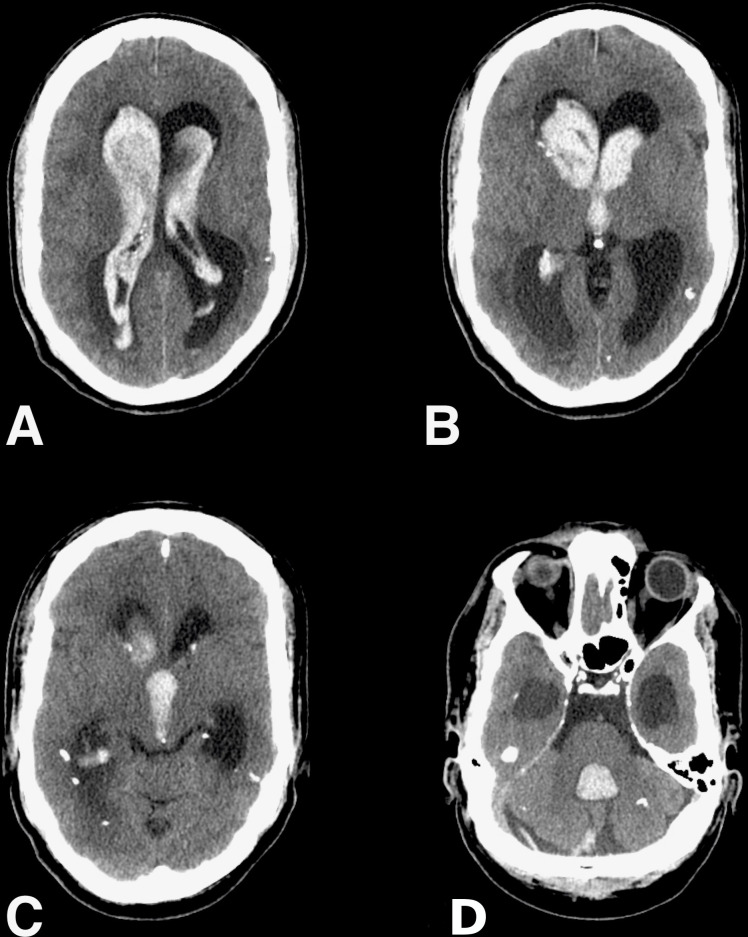
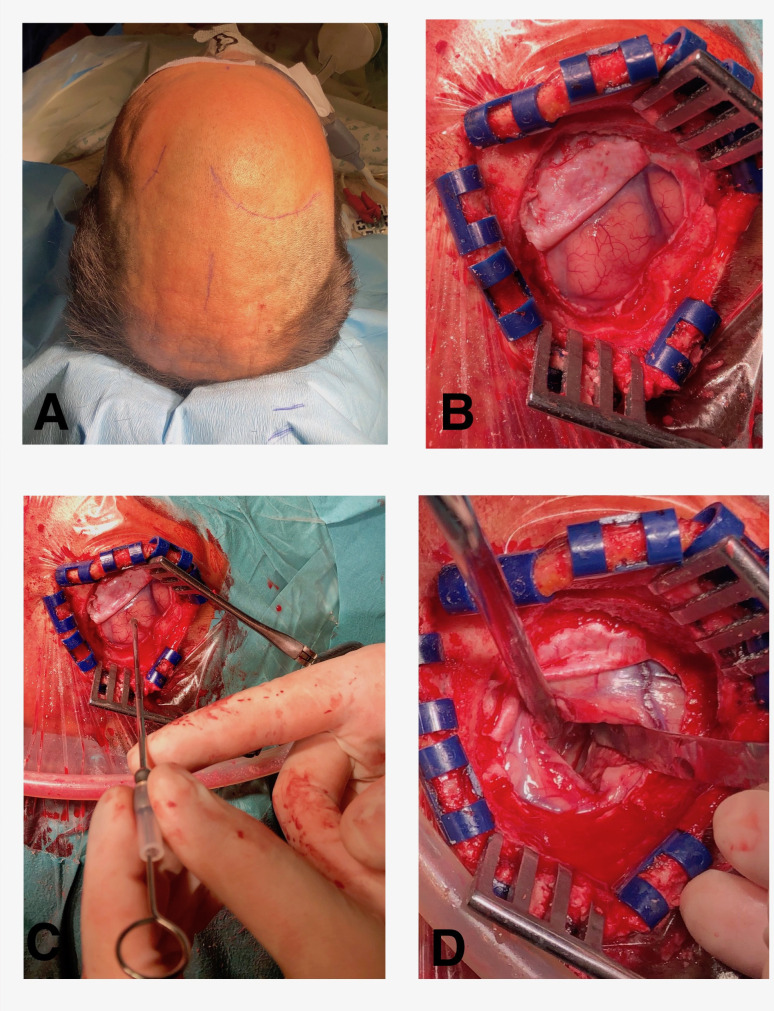
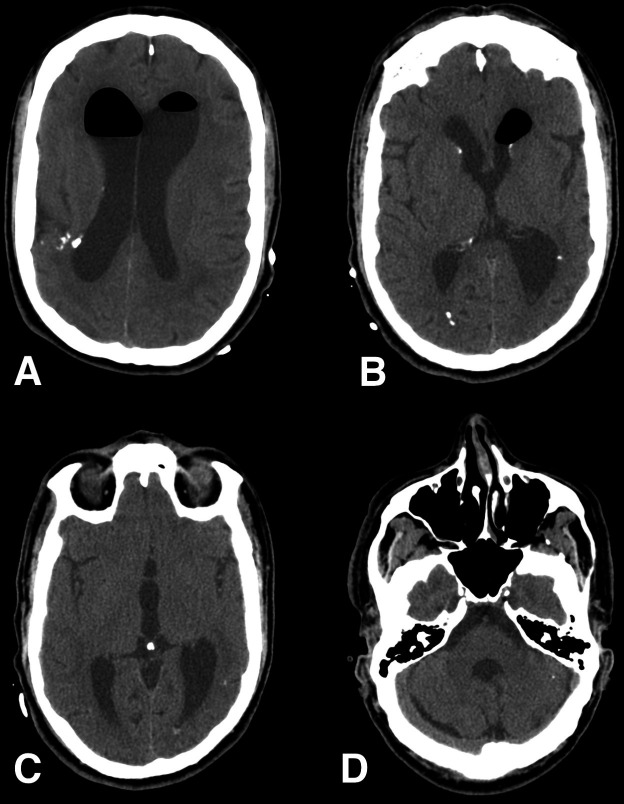
A 40-year-old man presented unconscious in the emergency department after a thunderclap headache witnessed by his relatives. He had a previous history of cerebral palsy related to paediatric bacterial meningitis that did not prevent him from having a part-time job and leading a relatively autonomous life. At initial physical examination, the breathing pattern was abnormal (Cheyne–Stokes respiration), the blood pressure was found to be elevated at 202/100 mm Hg and the cardiac rate was 38 bpm. The Glasgow Coma Scale was 6t (E1M4Vt), with bilateral withdrawal to pain and supranuclear upward gaze palsy. Pupilar, corneal and gag reflexes were preserved. A CT scan showed an IVH confined to all the ventricular system and obstructive acute hydrocephalus. (figure 1) The Graeb Score (GS) was 11 out of 12. He was immediately transferred to the operating theatre to perform clot evacuation. On the operating table, the patient was placed in the dorsal decubitus position with the head in the midline supported by a horseshoe headrest. As both lateral ventricles an Monro foramen were filled with blood, we planned the insertion of an EVD in the left lateral ventricle and a right frontal craniotomy 2 cm anterior to the coronal suture to perform direct clot drainage. The right later ventricle was accessed through a transcortical approach supported by a Cushing needle. The blood inside the right lateral ventricle was drained to allow cerebrospinal fluid circulation through the foramen of Monro to the third ventricle. (figure 2) Both EVDs were removed 24 hours after the operation. During hospitalisation, the patient recovered gradually to his initial condition. Intracranial MR angiography and digital subtraction angiography did not reveal any vascular abnormalities or space-occupying lesions. He was discharged 10 days after surgery with a Modified Rankin Scale (mRS) score of 1. A controlled CT scan was performed and showed considerable drainage of the intraventricular clot without signs of acute hydrocephalus (figure 3). No complications were recorded during hospitalisation. Twenty-24 hour ambulatory blood pressure monitoring of the patient in primary care revealed the need for antihypertensive medication. Coagulopathy and bleeding disorders were also excluded. The final diagnosis was idiopathic PIVH, arterial hypertension being the most likely risk factor.
Figure 1.
Pr-operative axial view CT scan showing intraventricular haemorrhage in the lateral ventricles (A), Monroe foramens (B) third ventricle (C) and fourth ventricle (D) with acute hydrocephalus.
Figure 2.
(A) Preoperative position and planned incisions. (B) Mini-fontal craniotomy and durotomy. (C) Transcortical approach of the right lateral ventricle with Cushing needle. (D) Intraventricular clot evacuation.
Figure 3.
Seven day postoperative CT scan showing complete resolution of the intraventricular haemorrhage without signs of hydrocephalus. (A) Lateral ventricles with pneumocephalus related to surgical approach. (B) Monroe Foramen free of blood allowing cerebrospinal fluid circulation to the third (C) and fourth (D) ventricles.
Outcome and follow-up
Recently, at the 24-month follow-up, the patient had an mRS score of 0 compared with the period before the vascular event. He recovered his normal function and gait pattern with the implementation of a daily rehabilitation programme.
Discussion
Regarding IVH, approximately 70% are secondary. Secondary IVH is often found as a complication of intraparenchymal haematomas (40%) or in association with subarachnoid haemorrhage (10%) and has worst functional and vital prognosis.7 PIVH is a rare type of haemorrhagic stroke confined to the ventricular system, arising from an intraventricular source, usually the choroid plexus. The anatomical structures of the ventricular walls should be preserved.2
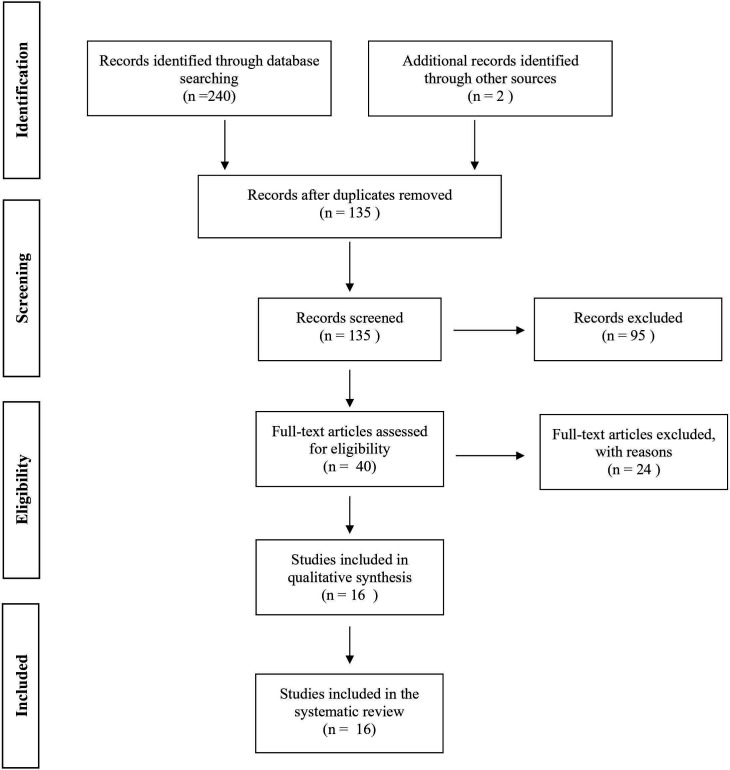
We conducted a systematic review of PIVH according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses. A systematic search via Science Direct, PubMed, Web of Science and Google Scholar was carried out for literature published between 1995 and 2019. The following terms in various combinations were used: ‘primary’, ‘intraventricular haemorrhage’ ‘hydrocephalus’ and ‘management’. All identified articles were then systematically assessed against the inclusion and exclusion criteria by two independent reviewers. Inclusion criteria were articles published in English presenting demographic and clinical features of patients with PIVH. Exclusion criteria included secondary IVH, PIVH diagnosed during autopsies, case reports with single patients and PIVH of preterm and newborns. Eligibility criteria were met by 16 studies8–24 (figure 4), totalling 564 patients with PIVH; however, complete data were not available for all subjects. Baseline demographics are presented in tables 1 and 2. These numbers also serve as the denominators for calculated percentages.
Figure 4.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses diagram for systematic review.
Table 1.
Patient demographics, presenting symptoms, imagological characteristics and outcome of patients with primary intraventricular haemorrhage
| Variable | Value |
| Total number of patients | 564 |
| Age, years (n=550) | |
| Mean±SD | 55.76±10.03 |
| Range | 9–90 |
| Sex | |
| Male | 303 (53.72%) |
| Female | 261 (46.28%) |
| Presenting symptoms (n=452) | |
| Headache | 303 (67.03%) |
| Altered mental status | 154 (34.07%) |
| Nausea/vomiting | 146 (32.30%) |
| Seizure | 26 (5.75%) |
| Risk factors/aetiologic agents (n=448) | |
| Hypertension | 240 (53.57%) |
| Arteriovenous malformation | 74 (16.00%) |
| Coagulation disorders | 58 (13.00%) |
| Moyamoya disease | 56 (12.50%) |
| Intraventricular aneurysm | 18 (4,01%) |
| Dural arteriovenous fistula | 9 (2,00%) |
| Intraventricular tumour | 4 (0,89%) |
| Mean Graeb Score ±SD (n=243) | 5.11±1.21 |
| Location of haemorrhage (n=160) | |
| Lateral ventricles | 138 (86.25%) |
| Third ventricle | 91 (56.88%) |
| Fourth ventricle | 71 (44.38%) |
| Outcome (n=180) | |
| Death | 91 (50.55%) |
| Asymptomatic | 67 (37.22%) |
| Memory/cognitive problems | 16 (8.89%) |
| Haemiparesis | 6 (3.33%) |
Regarding the degree of missingness present in the data, parentheses in the variable column indicate the total number of patients with data reported for that variable.
Table 2.
Outcome and treatment of patients with hydrocephalus
| Variable | Value |
| Number of patients with HC | 239 (42.38%) |
| Age, years (n=32) | |
| Mean±SD | 64.11±5.42 |
| Mean Graeb Score ±SD (n=81) | 7.12±4.14 |
| Mortality of patients with HC (n=49) | 36 (73.47%) |
| Hydrocephalus treatment (n=239) | |
| EVD | 168 (70.29%) |
| Conservative treatment | 53 (22.18%) |
| Craniotomy | 18 (7.53%) |
| Ventriculoperitoneal shunt (n=97) | 41 (42.23%) |
| EVD morbidity and mortality | |
| Rebleeding (n=10) | 5 (50.00%) |
| Central Nervous System infection (n=30) | 7 (23.33%) |
| Mortality rate (%) (n=33) | 18 (54.54%) |
Regarding the degree of missingness present in the data, parentheses in the variable column indicate the total number of patients with data reported for that variable.
EVD, external ventricular derivation; HC, hydrocephalus.
Patients ages ranged from 9 to 90 years with an average of 55.76±10.03 years. At least six patients were paediatric. Concerning gender, there were 303 men and 261 women. Initial symptoms were described for 452 subjects. The most common presenting symptoms of PIVH were headache (67.03%), altered mental status (34.07%) nausea and/or vomiting (32.30%). Seizures were described in only 5.75% of patients. Details about risk factors and etiological agents were available for 448 patients. Arterial hypertension was the main aetiological factor (53.57%) followed by intraventricular arteriovenous malformation (16%), moyamoya disease (12.5%) and intraventricular aneurysms (7.5% patients). Digital subtraction angiography provides morphological information on the intracranial circulation, being the gold-standard technique for the detection of cerebral aneurysms, moyamoya disease and vascular malformations. The distribution of IVH was reported in six studies (37.5%). IVH was localised inside the lateral ventricles or the third ventricle in 138 and 91 patients, respectively. Blood inside the fourth ventricle was observed in 71 patients. The mean GS score was 5.10±1.21. Of the 180 patients for whom outcome was described, the mortality rate was 50.55% and 37.22% was discharge without new neurological deficit.
Hydrocephalus was present in 239 (42.38%) patients. To relieve intracranial pressure (ICP) an EVD was placed in 168 patients and direct aspiration of the haematoma was performed in 18 patients. Of 42.23% of subjects needed a long-term ventriculoperitoneal shunt. The remaining subjects underwent conservative treatment. Data on EVD morbidity and mortality were available in six articles.10 12 14 15 18 19 The mortality rate of patients with EVD was 54.54%. The most common complication was rebleeding that occur in 5 (50%) of 10 patients for whom the complications were documented as either present or absent. Central nervous system infection was present in 7 (23.33%) of 30 subjects (table 2). Specific data about the outcome for the 18 patients treated with frontal craniotomy were not present in any article.
According to Sang-Hoon, older age, a low initial Glasgow Coma Scale, a high GS score, hydrocephalus and performance of EVD are all significantly associated with poor outcomes in patients with PIVH.20
Tamaki carried out the only retrospective study comparing the efficacy of frontal minicraniotomy in the treatment of IVH compared with EVD alone. Patients treated by frontal minicraniotomy were less likely to require long-term ventricular drainage and they had less subjects with meningitis (minicraniotomy: three cases vs EVD: eight cases, p=0.007), a high haematoma evacuation rate (minicraniotomy: 68.6±15% vs EVD: 12.7%±9.8%, p=0.0003) and a lower mortality rate compared with those undergoing EVD alone (minicraniotomy: 11% vs EVD: 27%).24
In a study developed by Halely, patients receiving EVD had an increased length of stay (21±17 days vs 9.5±10.4, p<0.005) and were 3.4 times more likely to experience a poor outcome compared with patients who did not require EVD (OR=3.4; p<0.001). Overall, 85% of the patients with IVH receiving EVD had a poor outcome.25
In our case, we decided to use direct ventricular clot aspiration. Different from secondary IVH, in PIVH, the brain parenchyma and the basal ganglia are preserved. Thus, if acute hydrocephalus was promptly resolved, the odds of low mRS are great. Moreover, EVD does not remediate the underlying pathophysiological agent, which is the clot itself. The presence of blood inside the ventricles and the increase in ICP promote dysfunction of the ependymal cells and of the blood brain barrier, which in turn modify the cerebrospinal fluid protein content and the osmotic pressure in the brain. Inflammation of the arachnoid granulations by inflammatory cytokines such as transforming growth factors 1 and 2 decreases cerebrospinal fluid absorption due to obliterative arachnoiditis and fibrotic blockage.26 So, in short term, EVD could decrease ICP, however, in long term, the patient’s likelihood of developing communicating hydrocephalus is relatively high. Moreover, EVD is associated with high rates of catheter occlusion, low rates of clot drainage and ventriculitis. The risk for ventriculitis is 10%–20% and is directly related to the duration of EVD placement. EVD replacement is common due to catheter occlusion and is associated with intraventricular rebleeding. The data obtained from this systematic review show that EVD is associated with relatively high mortality and morbidity, however, we do not had access to the patients’ initial neurological status and comorbidities. Moreover, acute hydrocephalus is generally associated with worst functional outcomes. The benefit of using a frontal craniotomy to treat hydrocephalus in PIVH is not demonstrated. Therefore, more case–control studies should be planned in order to understand its effects on the patient outcome and what subjects will benefit from this surgical procedure.
Learning points.
Primary intraventricular haemorrhage is a rare type of intracerebral haemorrhage confined to the ventricular system, with preservation of the brain parenchyma and the basal ganglia. Acute hydrocephalus should be solved as early as possible in order to avoid high Modified Rankin Scale scores.
Digital subtraction angiography is the gold-standard imaging modality for delineating intraventricular vascular malformations.
External ventricular drainage (EVD) could promptly reduce intracranial pressure; however, it is associated with a high rate of complications.
Central nervous system infection, catheter occlusion and rebleeding are common issues relate do EVD.
In patients with PIVH, the authors suggest the use of frontal minicraniotomy to preform direct aspiration of the intraventricular clot, coagulation of the source of bleeding and prompt resolution of acute hydrocephalus.
Footnotes
Contributors: All author contibute to the work. JPP, DRC, DM: conceptualisation, methodology, statistics, writing. JPP, RP: writing original draft, review and editing.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Darby DG, Donnan GA, Saling MA, et al. Primary intraventricular hemorrhage: clinical and neuropsychological findings in a prospective stroke series. Neurology 1988;38:68–75. 10.1212/WNL.38.1.68 [DOI] [PubMed] [Google Scholar]
- 2.Sanders E A study of primary, immediate, or direct hemorrhage into the ventricles of the brain. Am J Med Sci 1881;82:85–128. 10.1097/00000441-188107000-00008 [DOI] [Google Scholar]
- 3.Barnaure I, Liberato AC, Gonzalez RG, et al. Isolated intraventricular haemorrhage in adults. Br J Radiol 2017;90:1069. 10.1259/bjr.20160779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Morgenstern LB, Hemphill JC, Anderson C, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American heart Association/American stroke association. Stroke 2010;41:2108–29. 10.1161/STR.0b013e3181ec611b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dasic D, Hanna SJ, Bojanic S, et al. External ventricular drain infection: the effect of a strict protocol on infection rates and a review of the literature. Br J Neurosurg 2006;20:296–300. 10.1080/02688690600999901 [DOI] [PubMed] [Google Scholar]
- 6.Maniker AH, Vaynman AY, Karimi RJ, et al. Hemorrhagic complications of external ventricular drainage. Neurosurgery 2006;59:ONS419-24; discussion ONS424-5. 10.1227/01.NEU.0000222817.99752.E6 [DOI] [PubMed] [Google Scholar]
- 7.Hanley DF, Fisher M, Lees K. Intraventricular hemorrhage: severity factor and treatment target in spontaneous intracerebral hemorrhage. Stroke 2009;40:1533–8. 10.1161/STROKEAHA.108.535419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Angelopoulos M, Gupta SR, Azat Kia B. Primary intraventricular hemorrhage in adults: clinical features, risk factors, and outcome. Surg Neurol 1995;44:433–7. 10.1016/0090-3019(95)00261-8 [DOI] [PubMed] [Google Scholar]
- 9.Martí-Fàbregas J, Piles S, Guardia E, et al. Spontaneous primary intraventricular hemorrhage: clinical data, etiology and outcome. J Neurol 1999;246:287–91. 10.1007/s004150050348 [DOI] [PubMed] [Google Scholar]
- 10.Passero S, Ulivelli M, Reale F. Primary intraventricular haemorrhage in adults. Acta Neurol Scand 2002;105:115–9. 10.1034/j.1600-0404.2002.1o118.x [DOI] [PubMed] [Google Scholar]
- 11.Hameed B, Khealani BA, Mozzafar T, et al. Prognostic indicators in patients with primary intraventricular haemorrhage. J Pak Med Assoc 2005;55:315–7. [PubMed] [Google Scholar]
- 12.Kiymaz N, Demir O, Cirak B. Is external ventricular drainage useful in primary intraventricular hemorrhages? Adv Ther 2005;22:447–52. 10.1007/BF02849864 [DOI] [PubMed] [Google Scholar]
- 13.Flint AC, Roebken A, Singh V. Primary intraventricular hemorrhage: yield of diagnostic angiography and clinical outcome. Neurocrit Care 2008;8:330–6. 10.1007/s12028-008-9070-2 [DOI] [PubMed] [Google Scholar]
- 14.Giray S, Sen O, Sarica FB, et al. Spontaneous primary intraventricular hemorrhage in adults: clinical data, etiology and outcome. Turk Neurosurg 2009;19:338–44. [PubMed] [Google Scholar]
- 15.Ahn SW, Hwang S-K. Non-traumatic primary intraventricular hemorrhage. Kor J Cerebrovascular Surgery 2010;12:159–64. [Google Scholar]
- 16.Arboix A, García-Eroles L, Vicens A, et al. Spontaneous primary intraventricular hemorrhage: clinical features and early outcome. ISRN Neurol 2012;2012:1–7. 10.5402/2012/498303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Srivastava T, Sannegowda RB, Satija V, et al. Primary intraventricular hemorrhage: clinical features, risk factors, etiology, and yield of diagnostic cerebral angiography. Neurol India 2014;62:144–8. 10.4103/0028-3886.132333 [DOI] [PubMed] [Google Scholar]
- 18.Guo R, Ma L, Shrestha BK, et al. A retrospective clinical study of 98 adult idiopathic primary intraventricular hemorrhage cases. Medicine 2016;95:e5089. 10.1097/MD.0000000000005089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang S, Jia B, Li H, et al. Primary intraventricular hemorrhage in adults: etiological causes and prognostic factors in Chinese population. J Neurol 2017;264:382–90. 10.1007/s00415-016-8367-x [DOI] [PubMed] [Google Scholar]
- 20.Lee S-H, Park K-J, Park D-H, et al. Factors associated with clinical outcomes in patients with primary intraventricular hemorrhage. Med Sci Monit 2017;23:1401–12. 10.12659/MSM.899309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Weinstein R, Ess K, Sirdar B, et al. Primary intraventricular hemorrhage: clinical characteristics and outcomes. J Stroke Cerebrovasc Dis 2017;26:995–9. 10.1016/j.jstrokecerebrovasdis.2016.11.114 [DOI] [PubMed] [Google Scholar]
- 22.Pan K, Pandit A, Bhattacharyya B. Primary intraventricular haemorrhage: clinical and aetiological profile with predictors of outcome-A hospital based study. J Clin Diagnostic Res 2018;12:1–4. 10.7860/JCDR/2018/32328.11593 [DOI] [Google Scholar]
- 23.Guo R, Chen R, Yu Z, et al. Clinical features and prognosis of primary intraventricular hemorrhage in elderly: single-center experience. World Neurosurg 2019;124:e445–52. 10.1016/j.wneu.2018.12.114 [DOI] [PubMed] [Google Scholar]
- 24.Tamaki T, Node Y. Frontal Mini-craniotomy for evacuation of spontaneous thalamic and intraventricular hemorrhage. Surg Cereb Stroke 2010;38:181–5. 10.2335/scs.38.181 [DOI] [Google Scholar]
- 25.Hallevi H, Albright KC, Aronowski J, et al. Intraventricular hemorrhage: anatomic relationships and clinical implications. Neurology 2008;70:848–52. 10.1212/01.wnl.0000304930.47751.75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bu Y, Chen M, Gao T, et al. Mechanisms of hydrocephalus after intraventricular haemorrhage in adults. Stroke Vasc Neurol 2016;1:e000003:23–7. 10.1136/svn-2015-000003 [DOI] [PMC free article] [PubMed] [Google Scholar]