Abstract
Background:
Beginning in October 2016, the Centers for Medicare & Medicaid Services (CMS) issued expanded guidance requiring accrediting organizations and state survey agencies to report serious infection control breaches to relevant state health departments. This project sought to characterize and summarize The Joint Commission’s early experiences and findings in applying this guidance to facilities accredited under the ambulatory and office-based surgery programs in 2017.
Methods:
Surveyor notes were retrospectively reviewed to identify individual breaches, and then the Centers for Disease Control and Prevention’s Infection Prevention Checklist for Outpatient Settings was used to categorize and code documented breaches.
Results:
Of 845 ambulatory organizations, 39 (4.6%) had breaches observed during the survey process and reported to health departments. Within these organizations, surveyors documented 356 breaches, representing 52 different breach codes. Common breach domains were sterilization of reusable devices, device reprocessing observation, device reprocessing, disinfection of reusable devices, and infection control program and infrastructure. Eight of the 39 facilities (20.5%) were cited for not performing the minimum level of reprocessing based on the items’ intended use, reusing single-use devices, and/or not using aseptic technique to prepare injections.
Conclusion:
The CMS infection control breach reporting requirement has helped highlight some of the challenges faced by ambulatory facilities in providing a safe care environment for their patients. This analysis identified numerous opportunities for improved staff training and competencies as well as leadership oversight and investment in necessary resources. More systematic assessments of infection control practices, extending to both accredited and nonaccredited ambulatory facilities, are needed to inform oversight and prevention efforts.
As the volume and complexity of ambulatory procedures and settings continue to grow, so does the risk for harm stemming from serious breaches in infection prevention and control (IPC). Poor IPC practices can cause significant patient harm and outbreaks, which can lead to large patient notification events of potentially exposed patients and staff. For example, the Centers for Disease Control and Prevention’s (CDC’s) “One & Only Campaign” noted at least 50 outbreaks of viral hepatitis or bacterial infections associated with unsafe injection practices in outpatient settings from 2001 through 2011.1 Breaches related to medical equipment reprocessing have also led to large-scale patient notification events.2 Staff exposure from failure to use proper personal protective equipment (PPE) may also occur due to inadequate supplies or training. Previous studies have reported that IPC breaches occur in a variety of outpatient settings, including ambulatory surgery centers,3 and freestanding general and specialty clinics.4–7
Compared with Medicare-certified facilities (for example, hospitals, dialysis clinics, nursing homes), many types of freestanding ambulatory facilities, such as office-based surgery centers and multispecialty practices, are subject to less standardized regulatory oversight and performance monitoring requirements.8, 9 Accreditation is voluntary for ambulatory settings, except in a few states that require accreditation for some ambulatory or office-based surgery centers. Nevertheless, among facilities that seek Joint Commission accreditation, IPC–related problems remain the most frequently cited reason for noncompliance with standards.10 In part, this is because nonhospital-affiliated ambulatory facilities have substantially fewer IPC staff resources, and IPC staff often have multiple additional responsibilities.11 Staff often face space and equipment constraints and pressure for rapid turnover of patients, leading to substandard adherence to IPC practices such as appropriate reprocessing of medical equipment.12, 13 These clinics are often located in building spaces that were not originally designed to serve as health care facilities. As such, there may be functional limitations within the facility related to its architecture or ventilation, plumbing, and other systems. For example, room size and configuration may not allow for the process to flow from dirty to clean. This can compromise adherence to many aspects of IPC, ranging from hand hygiene to equipment reprocessing.
In October 2016 the Centers for Medicare & Medicaid Services (CMS) issued guidance expanding requirements for accrediting organizations and state survey agencies to report serious infection control breaches directly to the relevant state public health authorities.14 Engagement with health departments was intended to facilitate risk assessment and, if necessary, to provide assistance with implementing patient notification activities. Breaches that must be reported are those that could potentially expose patients to the blood or body fluids of another. This includes unsafe injection practices, such as the reuse of needles, syringes, lancet pens, or other injection devices for more than one individual. In addition, the guidance recommends reporting breaches when the accrediting organization or survey agency believes that additional public health assessment is needed, citing infection control lapses such as improper cleaning and disinfection of endoscopic equipment or sterilization of surgical instruments.14 In many cases, circumstances resulting in this type of public health reporting are multifactorial, reflecting a compilation of multiple breaches that together indicate systemic deficiencies rather than a single isolated error.
To date, no published studies have described findings from accrediting bodies or survey agencies regarding IPC breaches reported to health departments. The objective of this project was to characterize the major themes of IPC breaches that were identified by The Joint Commission among facilities seeking ambulatory accreditation and reported to state health departments in 2017. Information on the types of reported breaches can be used to help freestanding ambulatory facilities identify common infection control lapses and help oversight organizations concerned with IPC at state and national levels to target their resources for surveillance and prevention of the IPC lapses that place patients at risk.
METHODS
This project was a retrospective review of IPC breaches reported to state health departments. We included facilities that were evaluated for accreditation by The Joint Commission between February 1 and December 31, 2017, under the Ambulatory Health Care Accreditation Program (n = 739) or Office-Based Surgery Accreditation Program (n = 106). Facilities eligible for ambulatory accreditation included those providing primary and specialty medical and dental services (for example, Federally Qualified Health Centers, oncology centers), diagnostic and therapeutic services (for example, imaging, radiation therapy), surgical services (for example, ambulatory surgery centers), episodic care (for example, urgent care and freestanding emergency departments), and telehealth services. Ambulatory surgery centers had the option to pursue accreditation and Medicare certification simultaneously through The Joint Commission’s deemed survey process. Facilities eligible for office-based surgery accreditation were those that provided surgical and anesthesia services, not eligible for Medicare certification, and were licensed under business occupancy (defined as an occupancy that can have only three or fewer individuals at the same time who are either rendered incapable of self-preservation in an emergency or undergoing general anesthesia).15
Facilities were identified through review of electronic notification messages sent by The Joint Commission’s State Relations Department to the designated state health department contacts, typically the state health care-associated infection (HAI) coordinator. The reported breaches were compiled into a Microsoft Excel spreadsheet to which facility demographic information and surveyor comments were added. A coding structure for individual breaches was derived from the domains and sub-elements contained in the CDC’s Infection Prevention Checklist for Outpatient Settings.16 For example, if during review of the sterilization process a surveyor noted “brushes used were not cleaned/disinfected per manufacturer instructions,” this breach would be coded using the CDC outpatient checklist category 12D, described as “cleaning brushes are not disposable or, if reusable, not cleaned and high-level disinfected or sterilized (per manufacturer’s instructions) after use.” Additional project-specific codes within existing domains were created for breaches not addressed in the CDC’s outpatient checklist and for which surveyors used Joint Commission accreditation standards–related phrasing. This resulted in a total of 133 possible code options. Appendix 1 (available in online article) provides the complete coding structure.
NVivo 11 (QSR International [Americas] Inc., Burlington, Massachusetts) was used to assign codes that describe specific breaches. Members of the project team [B.B, S.C., K.D.] assigned a code for each surveyor comment that described a unique breach when there were multiple breaches within a facility. To promote accuracy and consistency, three project team members agreed on assigned codes during team meetings, with input from The Joint Commission’s infection preventionist [S.G.] as needed.
Statistical analysis was performed using SAS 9.4 (SAS Institute Inc., Cary, North Carolina). Simple descriptive statistics were calculated, and the Pearson correlation coefficient was used to examine the association between number of services provided by an organization (for example, primary care, radiation oncology, and podiatry) and number of breaches per organization. This project was not considered to be human subjects research because of the focus on organizational practices; no patient or staff identifiers were collected.
RESULTS
During February-December 2017, 845 organizations were evaluated for accreditation, of which 39 (4.6%) had breaches that met the CMS reporting requirements. Of the 39 organizations, 18 were ambulatory surgery centers (13 under the deemed survey process), 7 were office-based surgery centers, 11 were medical and/or dental clinics, and 3 were other types (Table 1).
Table 1.
Types of Facilities and Number of Breaches Among Joint Commission-Accredited Ambulatory Care Facilities That Were Subject to CMS-Mandated Breach Reporting, 2017
| Main Group Type | Type of Ambulatory Organization | Number of Facilities | Number of Breaches | Average Breaches per Facility |
|---|---|---|---|---|
| Ambulatory surgery center (ASC) | ASC deemed—multispecialty | 8 | 79 | 10 |
| ASC deemed—single specialty | 5 | 48 | 10 | |
| ASC non-deemed—multispecialty | 4 | 33 | 8 | |
| ASC non-deemed—single specialty | 1 | 13 | 13 | |
| Total | 18 | 173 | ||
| Office-based surgery center | Multispecialty | 3 | 30 | 10 |
| Single specialty | 4 | 37 | 9 | |
| Total | 7 | 67 | ||
| Medical/dental clinic | Federally Qualified Health Center (FQHC) | 5 | 41 | 8 |
| Multispecialty | 3 | 8 | 3 | |
| Single specialty | 3 | 28 | 9 | |
| Total | 11 | 77 | ||
| Other | Freestanding emergency room/urgent care | 1 | 23 | 23 |
| Medical imaging | 2 | 16 | 8 | |
| Total | 3 | 39 |
CMS, Centers for Medicare & Medicaid Services.
Twenty-eight of the 39 facilities with reported breaches (71.8%) provided multiple types of services (Table 2). The average number of different services provided was 4.3 (standard deviation [SD] = 3.3, median = 4, range: 1–14). Across the 39 facilities, 32 different types of services were offered, with the most common being orthopedic surgery (n = 13), gynecological procedures (n = 11), plastic surgery (n = 11), pain management (n = 11), podiatric surgery (n = 10), general surgery (n = 9), endoscopy (n = 8), urological procedures (n = 8), dentistry (n = 7), ear, nose, and throat (ENT) procedures (n = 6), family practice (n = 6), gastroenterology (n = 6), short stay observation/recovery services (n = 6), general practice (n = 5), internal medicine (n = 5), obstetrics (n = 5), pediatric medicine (n = 5), and pharmacy dispensary (n = 5).
Table 2.
Type of Ambulatory Facility and Examples of Services and Instruments Associated with Infection Control Breaches Reported to Health Departments During 2017
| Main Group Type | Type of Ambulatory Organization | Number of Coded Breaches | Services Provided at Facility | Devices or Instruments Associated with the Breach(es) When Specified in Notes | |
|---|---|---|---|---|---|
| 1 | Ambulatory surgery center | ASC | 13 | Urological procedures | cystoscopes |
| 2 | Ambulatory surgery center | ASC | 12 | Lithotripsy; Endoscopy; Surgery and procedures: ENT, GI, General, GYN, OPHTH, OMFS, ORTHO, PAIN, Plastic, POD, URO | eye instruments, endoscope |
| 3 | Ambulatory surgery center | ASC | 5 | Endoscopy; Surgery and procedures: GI, General, GYN, ORTHO, PAIN, Plastic, POD, URO | endoscopes, arthroscopes |
| 4 | Ambulatory surgery center | ASC deemed | 7 | Endoscopy | endoscopes |
| 5 | Ambulatory surgery center | ASC deemed | 3 | Surgery and procedures: GYN, OPHTH, ORTHO, Plastic, POD, Short stay observation | bronchoscope, laryngeal mask airway, vaginal ultrasound transducers |
| 6 | Ambulatory surgery center | ASC deemed | 21 | Urological procedures | cystoscopes |
| 7 | Ambulatory surgery center | ASC deemed | 10 | Endoscopy; Surgery and procedures: ENT, GI, General, GYN, OPHTH, ORTHO, PAIN, Plastic, URO, Short stay observation | dental instruments |
| 8 | Ambulatory surgery center | ASC deemed | 9 | Surgery and procedures: ORTHO, PAIN, POD | surgical implants |
| 9 | Ambulatory surgery center | ASC deemed | 6 | Pediatric dental | intraoral instruments |
| 10 | Ambulatory surgery center | ASC deemed | 8 | Endoscopy | endoscopes |
| 11 | Ambulatory surgery center | ASC deemed | 6 | PAIN | radio frequency ablation probes |
| 12 | Ambulatory surgery center | ASC deemed | 5 | Cardiac CATH; Endoscopy; Surgery and procedures: ENT, GI, General, GYN, ORTHO, PAIN, Plastic, Short stay observation, URO | vaginal ultrasound transducers |
| 13 | Ambulatory surgery center | ASC deemed | 2 | Surgery and procedures: ENT, General, ORTHO, PAIN, POD, Short stay observation | hardware implants |
| 14 | Ambulatory surgery center | ASC deemed | 24 | Endoscopy; Surgery and procedures: ENT, GI, General, GYN, OPHTH, Plastic, POD | eye instruments, laryngeal mask airways, endoscope |
| 15 | Ambulatory surgery center | ASC deemed | 15 | Surgery and procedures: Hand and spine, ORTHO, PAIN | UTD |
| 16 | Ambulatory surgery center | ASC deemed | 11 | Surgery and procedures: ENT, General, ORTHO, PAIN, POD | bronchoscope, other orthopedic instruments |
| 17 | Ambulatory surgery center | Oral maxillo-facial surgery |
8 | Surgery and procedures: Anesthesia, Dentistry, OMFS | dental handle |
| 18 | Ambulatory surgery center | Surgery center | 8 | Surgery and procedures: Short stay observation, URO | cystoscope |
| 19 | Multispecialty medical/dental | FQHC | 8 | Dentistry; Family and general practice; GYN; OB; Optometry; Pediatric; Pharmacy | vaginal ultrasound transducers, dental instruments |
| 20 | Multispecialty medical/dental | FQHC | 8 | Dentistry; Family and general practice; Pharmacy | UTD |
| 21 | Multispecialty medical/dental | FQHC | 10 | Dentistry; Family practice, Pediatric; Pharmacy | vaginal ultrasound transducers, dental instruments |
| 22 | Multispecialty medical/dental | FQHC | 13 | Family and general practice; GYN; Internal medicine; OB; Pediatric; Pharmacy | UTD |
| 23 | Multispecialty medical/dental | FQHC | 2 | Dentistry; Family practice; GYN; Internal medicine; OB; Pediatric; Pharmacy; POD | hemoglobin point of care test, vaginal ultrasound transducer |
| 24 | Multispecialty medical/dental | Medical center/clinic | 1 | Dentistry; Family and general practice; GYN; OB; Pediatric medicine; POD | vaginal ultrasound transducer |
| 25 | Multispecialty medical/dental | Medical center/clinic | 6 | General practice; GYN; Internal medicine; OB | glucometer, endoscope |
| 26 | Multispecialty medical/dental | Multispecialty group | 1 | Internal medicine; Neurology; ORTHO; Other—general surgery | ultrasound transducer used for needle biopsies of thyroid |
| 27 | Office-based surgery center | OBS—p lastic surgery | 15 | Plastic surgery | UTD |
| 28 | Office-based surgery center | OBS—oral & maxillofacial & plastic surgery | 13 | Surgery and procedures: OMFS, Plastic | cosmetic and oral and maxillofacial surgery (arts) and body cosmetic surgery, dental hand piece |
| 29 | Office-based surgery center | OBS—oral & maxillofacial surgery, endoscopy & pain management |
3 | Endoscopy; Surgery and procedures: OMFS, PAIN, Short stay observation | dental/oral surgery |
| 30 | Office-based surgery center | OBS—urology | 10 | Urological procedures | cystoscopes |
| 31 | Office-based surgery center | OBS—plastic surgery | 8 | Plastic surgery | surgical instruments |
| 32 | Office-based surgery center | OBS—plastic surgery | 4 | Plastic surgery | UTD |
| 33 | Office-based surgery center | Orthopedic | 14 | Surgery and procedures: General, ORTHO, PAIN, Plastic, POD, Spine | UTD |
| 34 | Other single specialty/ diagnostic/ therapeutic | Freestanding ER | 23 | Diagnostic imaging: CT, CTA, ultrasound, x-ray; Emergency care 24/7 | gynecological ring forceps, vaginal ultrasound transducer, glucometer |
| 35 | Other single specialty/ diagnostic/ therapeutic | Medical center/clinic | 9 | Internal medicine; diagnostic imaging: x-ray, sleep diagnostic | UTD |
| 36 | Other single specialty/ diagnostic/ therapeutic | Medical imaging | 8 | Diagnostic imaging: CT, MRI, mammography, ultrasound, x-ray | vaginal ultrasound transducer |
| 37 | Other single specialty/ diagnostic/ therapeutic | Medical imaging | 8 | Diagnostic imaging: CT, MRI, ultrasound, x-ray | vaginal ultrasound transducer |
| 38 | Other single specialty/ diagnostic/ therapeutic | Oncology related | 13 | Radiation oncology | brachytherapy vaginal CT/MR applicator |
| 39 | Other single specialty/ diagnostic/ therapeutic | Oncology related | 6 | Radiation oncology | broncho-laryngoscope |
ASC, ambulatory surgery center; CATH, catheterization; CT, computed tomography; CTA, computed tomography angiography; ENT, ears, nose, throat; ER, emergency room; FQHC, Federally Qualified Health Center; GI, gastroenterology; GYN, gynecology; MRI, magnetic resonance imaging; OB, obstetrics; OBS, office-based surgery; OMFS, oral maxillofacial surgery; OPHTH, ophthalmology; ORTHO, orthopedics; PAIN, pain management (invasive); POD, podiatry; URO, urology; UTD, unable to determine.
Table 2 provides examples of the medical devices and instruments mentioned in surveyor notes related to the infection control breaches when this information was available. These included endoscopes (for example, cystoscopes, bronchoscopes), vaginal ultrasound transducers, and dental, ophthalmic, and surgical instruments. There was no correlation between the total number of different types of services offered and the total number of coded breaches identified by facility.
COMMONLY IDENTIFIED BREACHES
Across the 39 facilities, 356 breaches were coded using 52 different breach codes. There were on average 9.1 breaches per facility (SD = 5.5, median = 8, range: 1–24). Approximately 20% (n = 8) of facilities were cited for the most serious breaches, including not performing the minimum level of reprocessing based on the items’ intended use, reusing single-use devices, and/or not using aseptic technique to prepare injections.
The majority of breaches were coded under the following domains (as defined in Figure 1): sterilization of reusable devices (111, 31.2%), device reprocessing observation (85, 23.9%), disinfection of reusable devices (62, 17.4%), device reprocessing (62, 17.4%), and infection control program and infrastructure (26, 7.3%). Examples of breaches within the most frequently identified CDC outpatient checklist domains are described below. See Table 3 for a full listing of breaches and frequencies identified under each domain.
Figure 1:
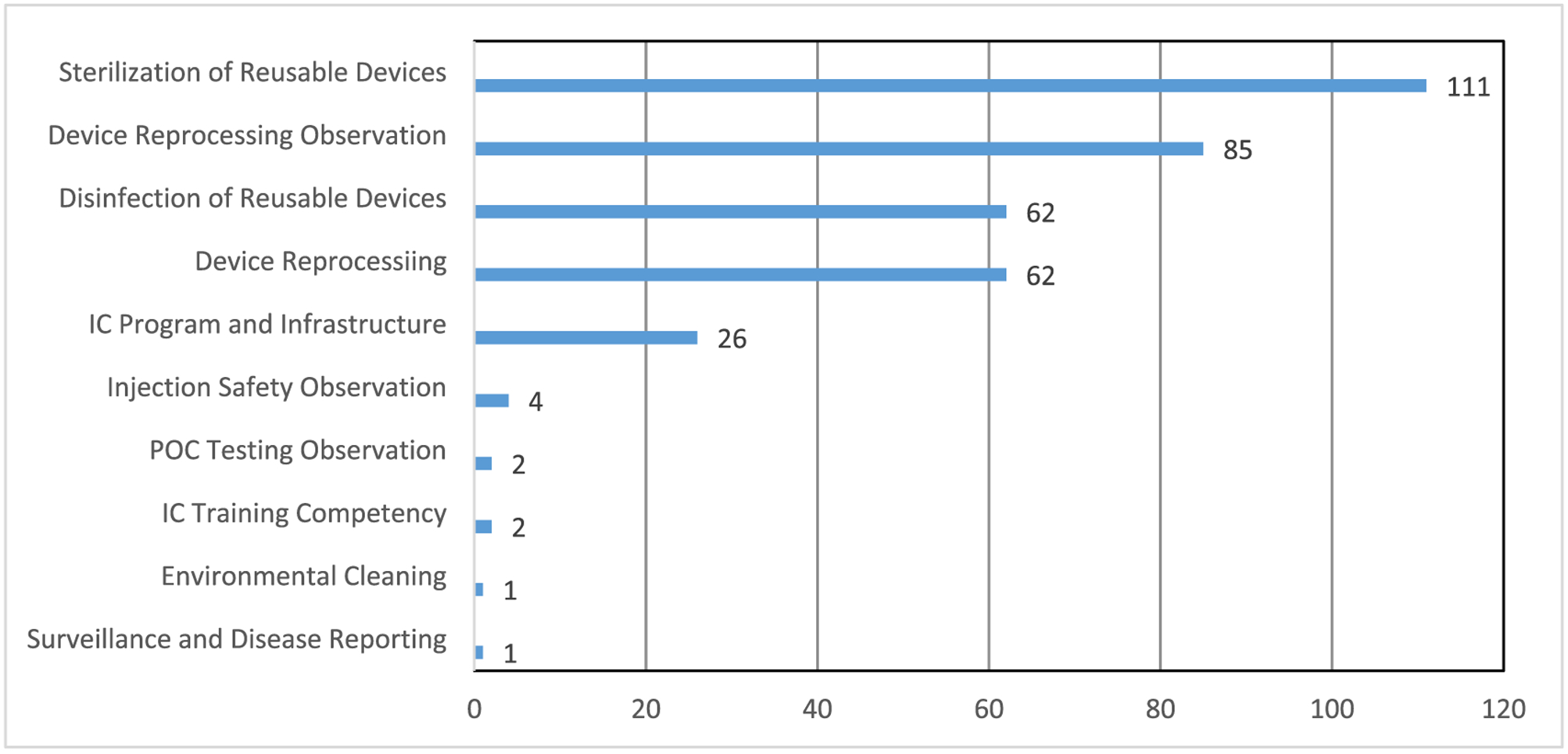
As shown in this figure, more than half of the 356 breaches were coded under the domains of sterilization of reusable devices (31.2%) and device reprocessing observation (23.9%).
* Source for domains is the Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care from the Centers for Disease Control and Prevention (CDC): https://www.cdc.gov/infectioncontrol/pdf/outpatient/guide.pdf (accessed May 27, 2020). There are several additional domains included in the CDC’s Infection Prevention Checklist for Outpatient Settings but not listed above. These can be found in Appendix 1, available in online article.
Table 3.
Domains, Description, and Frequency of Coded Infection Control Breaches Reported to Health Departments During 2017 Among Ambulatory Care Facilities Undergoing Joint Commission Accreditation Surveys
| Domains* | Code ID† | Breach Description | Frequency |
|---|---|---|---|
| Sterilization of reusable devices | 12P | Project-specific code (breaches not elsewhere specified). Assesses other elements related to sterilization. Specific improvement areas noted include transport in open tray; BI not placed correctly; contaminated instruments hand-carried into decontamination room; expired supplies; and sterilization instruments, including implants, not put into circulation prior to BI results. | 26 |
| 12J | Sterilization logs are not current and/or do not include results from each load. | 24 | |
| 12G | Failed to use a biological indicator, intended specifically for the type and cycle parameters of the sterilizer, at least weekly for each sterilizer and with every load containing implantable items. | 12 | |
| 12E | After cleaning, instruments are not appropriately wrapped/packaged for sterilization (for example, package system selected is compatible with the sterilization process being performed, items are not placed correctly into the basket, shelf or cart of the sterilizer so as not to impede the penetration of the sterilant, hinged instruments are not open, instruments are not disassembled if indicated by the manufacturer). | 12 | |
| 12B | Cleaning is not performed as soon as practical after use (for example, at the point of use) to prevent soiled materials from becoming dried onto devices. | 9 | |
| 12C | Enzymatic cleaner or detergent is not used for cleaning and discarded according to manufacturer’s instructions (typically after each use). | 9 | |
| 12D | Cleaning brushes are not disposable or, if reusable, not cleaned and high-level disinfected or not sterilized (per manufacturer’s instructions) after use. | 6 | |
| 12A | Devices are not thoroughly cleaned according to manufacturer instructions and visually inspected for residual soil prior to sterilization. | 5 | |
| 12F | A chemical indicator (process indicator) is not placed correctly in the instrument packs in every load. | 5 | |
| 12I | Sterile packs are not labeled with a load number that indicates the sterilizer used, the cycle or load number, the date of sterilization, and, if applicable, the expiration date. | 2 | |
| 12O | The facility does not have a process to perform initial cleaning of devices (to prevent soiled materials from becoming dried onto devices) prior to transport to the off-site reprocessing facility. | 1 | |
| Device reprocessing observation | 11bI | Project-specific code. Reusable medical devices are not cleaned, reprocessed (disinfection or sterilization) and not maintained according to evidence-based guidelines. | 25 |
| 11bB | Reusable medical devices are not cleaned, reprocessed (disinfection or sterilization) and maintained according to the manufacturer instructions. | 22 | |
| 11bA | Policies, procedures, and manufacturer reprocessing instructions for reusable medical devices used in the facility are not available in the reprocessing area(s). | 11 | |
| 11bD | Reprocessing area: | 8 | |
| i. Adequate space is not allotted for reprocessing activities. | |||
| ii. A workflow pattern is not followed such that devices clearly flow from | |||
| high-contamination areas to clean/sterile areas (that is, there is no clear separation between soiled and clean workspaces). | |||
| 11bJ | Project-specific code. Facility is not doing high-level disinfection (HLD) or sterilization when it should be done. | 8 | |
| 11bH | Project-specific code. Device reprocessing breaches not elsewhere specified include lack of consistent implementation of policy, following outdated guidelines, and no sterilizer on-site. | 7 | |
| 11bC | Single-use devices are not discarded after use and/or were used for more than one patient. | 2 | |
| 11bE | Adequate time for reprocessing is not allowed to ensure adherence to all steps recommended by the device manufacturer, including drying and proper storage. | 1 | |
| 11bF | Health care personnel (HCP) engaged in device reprocessing do not wear appropriate personal protective equipment (PPE) to prevent exposure to infectious agents or chemicals (PPE can include gloves, gowns, masks, and eye protection). | 1 | |
| Device reprocessing | 11aC | HCP responsible for reprocessing reusable medical devices do not receive hands-on training on proper selection and use of PPE and recommended steps for reprocessing assigned devices: | 26 |
| i. Upon hire, prior to being allowed to reprocess devices | |||
| ii. Annually | |||
| iii. When new devices are introduced or policies/procedures change. | |||
| 11aD | HCP are not required to demonstrate competency with reprocessing procedures (that is, correct technique is observed by trainer) following each training. | 18 | |
| 11aI | Routine maintenance for reprocessing equipment (for example, automated endoscope reprocessors, steam autoclave) is not performed by qualified personnel in accordance with manufacturer instructions; availability of maintenance records not confirmed. | 15 | |
| 11aE | Facility does not regularly audit (monitor and document) adherence to reprocessing procedures. | 2 | |
| 11aA | Facility has no policies and procedures to ensure that reusable medical devices are cleaned and reprocessed appropriately prior to use on another patient. | 1 | |
| Disinfection of reusable devices | 13O | Project-specific code. HLD breaches not elsewhere specified include expired test strips. scopes transported improperly, stained sponges, and lack of documentation that HLD quality monitoring parameters were performed. | 14 |
| 13F | For chemicals used in HLD, manufacturer instructions are not followed for: | 8 | |
| i. Preparation | |||
| ii. Testing for appropriate concentration, and | |||
| iii. Replacement (that is, upon expiration or loss of efficacy) | |||
| 13L | After HLD, devices not stored in a manner to protect from damage or contamination. Note: Endoscopes should be hung in a vertical position. | 7 | |
| 13B | Devices are not thoroughly cleaned according to manufacturer instructions and visually inspected for residual soil prior to HLD | 6 | |
| 13A | Flexible endoscopes are not inspected for damage and leak tested as part of each reprocessing cycle. (Any device that fails the leak test is removed from clinical use and repaired.) | 4 | |
| 13C | Cleaning is not performed as soon as practical after use (for example, at the point of use) to prevent soiled materials from becoming dried onto instruments. | 4 | |
| 13D | Enzymatic cleaner or detergent is not used and discarded according to manufacturer instructions (typically after each use). | 4 | |
| 13J | After high-level disinfection, devices are appropriately rinsed as specified by the manufacturer. | 4 | |
| 13E | Cleaning brushes are not disposable or, if reusable, are not cleaned and high-level disinfected or sterilized (per manufacturer instructions) after use. | 3 | |
| 13K | Devices are not dried thoroughly prior to reuse. Note: For lumened instruments (for example, endoscopes) this includes flushing all channels with alcohol and forcing air through channels. | 3 | |
| 13H | Devices are not disinfected for the appropriate length of time as specified by manufacturer instructions. | 2 | |
| 13M | Facility does not maintain a log for each endoscopy procedure that includes patient’s name and medical record number (if available), procedure, date, endoscopist, system used to reprocess the endoscope (if more than one system could be used in the reprocessing area), and serial number or other identifier of the endoscope used. | 2 | |
| 13I | Devices are not disinfected at the appropriate temperature as specified by manufacturer instructions. | 1 | |
| Infection control | 1E | Project-specific code. Infection control program and infrastructure breaches not | 13 |
| program and infrastructure | elsewhere specified include inadequate facility leadership oversight and accountability regarding evidence-based manufacturer instruction-supported sterilization processes, reprocessing of surgical instruments, and HLD. | ||
| 1F | Project-specific code. Infection control program and infrastructure breaches not elsewhere specified include failure to perform or inadequate risk assessment and lack of specific job qualifications. | 6 | |
| 1C | Facility does not have at least one individual trained in infection prevention employed by or regularly available (for example, by contract) to manage the facility’s infection control program. | 4 | |
| 1B | Infection prevention policies and procedures are not reassessed at least annually or according to state or federal requirements and updated if appropriate. | 2 | |
| 1A | Written infection prevention policies and procedures are not available, current, and based on evidence-based guidelines (for example, CDC/HICPAC), regulations, or standards. | 1 | |
| Injection safety observation | 7bA | Injections are not prepared using aseptic technique in a clean area free from 1 contamination or contact with blood, body fluids, or contaminated equipment. | 1 |
| 7bB | Needles and syringes are not used for only one patient (this includes 1 manufactured prefilled syringes and cartridge devices such as insulin pens). | 1 | |
| 7bH | Multidose vials to be used for more than one patient are not kept in a centralized 1 medication area and/or entered the immediate patient treatment area (for example, operating room, patient room/cubicle). | 1 | |
| 7bI | All sharps are not disposed of in a puncture-resistant sharps container. | ||
| Point-of-care testing observation | 9bA | New single-use, auto-disabling lancing device is not used for each patient. | 1 |
| 9bB | If used for more than one patient, the point-of-care testing meter is not cleaned and disinfected after every use according to manufacturer’s instructions. | 1 | |
| Environmental cleaning | 10aG | Operating rooms are not terminally cleaned after last procedure of the day. | 2 |
| Surveillance and disease reporting | 4D | Project-specific code. Surveillance and disease reporting breaches not elsewhere specified include lack of follow-up after surgical site infections. | 1 |
BI, biological indicator; CDC/HICPAC, Centers for Disease Control and Infection, Healthcare Infection Control Practices Advisory Committee.
Domains were derived from the Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care , from the Centers for Disease Control and Prevention.(CDC):https://www.cdc.gov/infectioncontrol/pdf/outpatient/guide.pdf (accessed May 27, 2020).
Numbers and letters correspond to items within domains on the CDC’s Infection Prevention Checklist for Outpatient Settings. For more information see Appendix 1, available in online article.
Sterilization of reusable devices: Includes inappropriate transport or hand-carrying of contaminated instruments, biological indicators not placed correctly, premature release of instruments, failure to document cycle parameters, failure to use appropriate biological indicators, and instruments packaged in a manner that would not allow equipment to be exposed to sterilant.
Device reprocessing observation: Includes lack of cleaning, reprocessing, and maintaining devices per evidence-based guidelines; not following manufacturer instructions for reprocessing; failure to make policies, procedures, and manufacturer reprocessing instructions available in the reprocessing area; having inadequate space or failure to ensure that devices flow from high-contamination areas to clean/sterile areas; and not doing any high-level disinfection (HLD) or sterilization when it should be done.
Disinfection of reusable devices: Includes use of expired test strips, improper transportation of sponges, stained sponges, lack of documentation of HLD quality monitoring parameters, inappropriate storage of disinfectants and cabinets not vented per manufacturer instructions, not following manufacturer instructions for the preparation of HLD chemicals, not cleaning devices prior to HLD, and devices not stored in a manner to protect from damage or contamination.
Device reprocessing: Includes lack of training of health care providers on selection and use of PPE and reprocessing, not requiring health care providers to demonstrate competency with reprocessing procedures, lack of qualified personnel to perform routine maintenance for reprocessing equipment in accordance with manufacturer instructions, and/or maintenance records unavailable.
Surveyor findings related to the CDC checklist’s “Infection Control Program and Infrastructure” domain are not typically considered breaches, but surveyors frequently reported such findings as lack of adequate leadership oversight and accountability, failure to perform a risk assessment or performing an inadequate risk assessment, and lack of specific job qualifications.
Finally, breaches reported from two other domains with low frequency but high risk concern were injection safety observation and point-of-care testing. Under the domain of injection safety observation, one breach was reported for each of the following:
Injections not prepared using aseptic technique in a clean area free from contamination
Needles or syringes used for more than one patient
Multidose vials not kept in a centralized medication area and/or entered the immediate patient treatment area
Not all sharps were disposed of in a puncture-resistant sharps container.
Similarly, under the point-of-care testing domain, one breach was reported for each of the following:
Single-use auto-disabling lancing devices used for more than one patient
Point-of-care testing meter was not cleaned and disinfected after every use according to manufacturer’s instructions.
DISCUSSION
This project characterized and summarized the IPC breaches that were identified by Joint Commission surveyors during the ambulatory health care and office-based surgery accreditation process and were reported to state health departments in 2017. That year was the first full calendar year of experience with the expanded CMS guidance on infection control breach reporting. Surveyors identified several common breaches that could potentially cause patient harm and that can help outpatient facilities target prevention efforts. The breaches identified in this project can be considered leading indicators of underlying weaknesses in IPC system and processes that may pose patient risk if unaddressed.
The most frequently reported breaches were in the domains of sterilization of reusable devices, HLD of reusable devices, and other aspects of device reprocessing. Specific areas of concern were not using biological indicators appropriately for sterilization and inadequate documentation of quality monitoring parameters on HLD logs. Lack of adherence to manufacturers’ instructions and evidence-based guidelines were also common themes across several domains. Approximately 20% of facilities were cited for serious breaches such as not performing the minimum level of reprocessing, reusing single-use devices, or not using aseptic techniques to prepare injections. Such practices can pose risk to patients and highlight the need for regular and systematic assessments of IPC practices in ambulatory settings.
IPC program infrastructure was another domain in which deficiencies were commonly identified, including lack of leadership, oversight, and accountability. Related concerns were inadequate competency-based training of staff and lack of IPC expertise of supervisors. For example, oversight of IPC may be done by a physician, nursing supervisor, or even infection preventionist who has never had formal training in the required HLD and/or sterilization processes. Findings also highlighted the need for development and periodic review of IPC policies and procedures and making these available to staff.
Our findings are generally consistent with other studies in freestanding ambulatory settings.3,4,6,17 We found relatively few instances of serious injection safety breaches, such as reuse of needles or single-dose vials, or mishandling of fingerstick devices and glucometers, all of which have been associated with bloodborne pathogen outbreaks. This finding might be due to a combination of factors. Although unannounced, on-site visits for the purposes of accreditation are routinely conducted outside the setting of an ongoing outbreak, therefore making it less likely that such assessments will identify the types of higher-risk infection control breaches more likely to be seen during an active, ongoing outbreak. Also, expectations for safe injection practices have been communicated with increasing detail in guidance from the CDC and other agencies and organizations,1,18–21 increasing awareness among health care providers and facilities. These are reflected in CMS and accrediting organization standards and have been the subject of extensive educational efforts over the past decade.
One implication of our findings for practice is that ambulatory settings should consider investing more resources in IPC staff training and competency. Health care facility leadership should strongly consider having at least one individual with training in IPC employed or regularly available (for example, by contract) to oversee the facility’s IPC program. If not employed by the facility, persons with IPC expertise can be accessed through consultation from professional societies or other groups, as well as local hospitals and health departments.22 The CDC and others offer free IPC guidance documents and checklists tailored for ambulatory facilities and several specialties such as oncology, dental, orthopedic and pain management, and podiatry.19 Relevant CDC-sponsored IPC trainings are available online at no cost.23,24 Notably, some of the most commonly identified breaches that we identified in this project have been addressed by recent guidance. For example, medical device reprocessing lapses have been associated with outbreaks25–27 and the CDC Healthcare Infection Control Practices Advisory Committee recently issued recommendations and tools to assist health care facilities in achieving a reliable, high-quality reprocessing program. Though these materials focus on endoscopes, the advice is broadly applicable to medical device reprocessing.28 There is also recent US Food and Drug Administration (FDA) guidance on reprocessing of ultrasound probes.29,30 Additional effort is needed to raise awareness of these resources and tools, along with incentives to ensure that they reach the appropriate staff and are used to drive improvements.
This project has several limitations. Breaches reported to health departments do not represent all IPC-related breaches and instances of noncompliance with standards identified by Joint Commission surveyors in these settings, but only those consistent with the reporting requirements specified by CMS.14 A review of all Joint Commission ambulatory health care and office-based surgery survey findings (extending to facility surveys that did not prompt health department reporting) was beyond the scope of this project. Thus the findings reported here do not necessarily correspond to noncompliance with Joint Commission IPC standards related to leadership, hand hygiene, and other topics evaluated on-site. Further, the reporting requirements for device reprocessing breaches are somewhat subjective and open to interpretation. Areas of accreditation survey emphasis may also vary with time and by setting. During this time period, surveyors were instructed to visit all areas that perform HLD and sterilization because it was emerging as a known high-risk area. Reported breaches come from the first year of a new process. Information on specific services and instruments associated with breaches was sometimes missing from surveyor notes; therefore, those described should be considered examples rather than an absolute indication of frequency or risk.
In addition, the small number of facilities in our analysis precludes drawing conclusions about the entire population of ambulatory facilities. We cannot say which types of facilities had the most reported breaches because we do not have sufficient information on the underlying population of ambulatory facilities using the groupings of facility type reported here. It is worth noting that most ambulatory facilities, including physician offices and specialty clinics, are not subject to regular survey visits as part of an accreditation or Medicare certification process. Only ambulatory surgery centers are eligible for Medicare deemed status and thus may have a stronger incentive to become accredited. While some state laws and policies extend accreditation requirements to select outpatient facility types, most states do not. Other studies suggest that organizations likely to pursue accreditation in the absence of deemed status drivers (for example, nursing care centers) may provide higher-quality services or be more motivated to improve.31 Therefore, facilities accredited by The Joint Commission and eligible for inclusion in the project reported here are not representative of all ambulatory facilities. Before drawing conclusions about higher-risk facilities and services, it would be helpful to see a more extensive examination of outpatient IPC practices and conditions that encompasses both accredited and nonaccredited facilities across a variety of models and specialties. Similarly, future studies should compare hospital-affiliated ambulatory facilities with freestanding ambulatory facilities.
CONCLUSION
This study shows that there are opportunities to improve IPC practices in ambulatory facilities, particularly in relation to device reprocessing, including sterilization and HLD, staff training and competency, and leadership oversight. Identification and remediation of existing weaknesses in systems and processes can prevent outbreaks and improve safety for both patients and staff. By highlighting the need for public health assessment of serious IPC breaches, including considerations for patient notifications, the CMS reporting requirement has helped raise the profile of health department HAI programs and their role in helping guide corrective actions. More systematic assessments of IPC practices, extending to both accredited and nonaccredited ambulatory facilities, are needed to help identify issues and inform efforts to protect patients from infection risks in the outpatient environment.
Supplementary Material
Acknowledgments.
The authors wish to thank Scott Williams, PsyD; David Baker, MD. MPH. FACP; Ana McKee, MD; and Lisa Waldowski, DNP, APRN, CIC, for their guidance on and participation in this project, and Hasina Hafiz, MPH, for editorial assistance.
Footnotes
Conflicts of Interest. All authors report no conflicts of interest.
SUPPLEMENTARY MATERIALS
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jcjq.2020.05.005.
Contributor Information
Barbara I. Braun, Department of Research, The Joint Commission, Oakbrook Terrace, Illinois..
Salome O. Chitavi, Department of Research, The Joint Commission..
Kiran M. Perkins, US Public Health Service, and Team Lead, Outbreak and Response, Division of Healthcare Quality Promotion, Centers for Disease Control and Prevention (CDC), Atlanta..
Joseph F. Perz, Public Health Programs, Division of Healthcare Quality Promotion, CDC..
Ruth Link-Gelles, Public Health Service, and Epidemiologist, Division of Healthcare Quality Promotion, CDC..
Jennifer Hoppe, Division of Support Operations, The Joint Commission..
Kristine M. Donofrio, Division of Healthcare Quality Evaluation, The Joint Commission..
Yanhong Shen, Department of Quality Measurement, The Joint Commission..
Sylvia Garcia-Houchins, Office of Quality and Patient Safety, The Joint Commission..
REFERENCES
- 1.Centers for Disease Control and Prevention. One & Only Campaign. (Updated: Dec 3, 2019.) Accessed May 27, 2020 https://www.cdc.gov/injectionsafety/one-and-only.html.
- 2.Centers for Disease Control and Prevention, Health Alert Network. Immediate Need for Healthcare Facilities to Review Procedures for Cleaning, Disinfecting, and Sterilizing Reusable Medical Devices, Sep 11 2015. Accessed May 27, 2020 https://emergency.cdc.gov/han/han00382.asp .
- 3.Schaefer MK, et al. Infection control assessment of ambulatory surgical centers. JAMA. 2010. Jun 9;303:2273–2279 . [DOI] [PubMed] [Google Scholar]
- 4.Ross K, et al. Outbreak of septic arthritis associated with intra-articular injections at an outpatient practice—New Jersey, 2017. MMWR Morb Mortal Wkly Rep.. 2017. Jul 28;66:777–779 Erratum in: MMWR Morb Mortal Wkly Rep. 2018 Apr 13;67:432. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ross KM, et al. Bacterial septic arthritis infections associated with intra-articular injection practices for osteoarthritis knee pain—New Jersey, 2017. Infect Control Hosp Epidemiol.. 2019;40:1013–1018 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beaudoin AL, et al. Invasive group A Streptococcus infections associated with liposuction surgery at outpatient facilities not subject to state or federal regulation. JAMA Intern Med. 2014;174:1136–1142 . [DOI] [PubMed] [Google Scholar]
- 7.Perz JF, et al. US outbreak investigations highlight the need for safe injection practices and basic infection control. Clin Liver Dis. 2010;14:137–151 . [DOI] [PubMed] [Google Scholar]
- 8.Barie PS. Infection control practices in ambulatory surgical centers. JAMA. 2010. Jun 9;303:2295–2297 . [DOI] [PubMed] [Google Scholar]
- 9.Centers for Disease Control and Prevention. Outpatient Settings Policy Options for Improving Infection Prevention: Key Policy Elements for Best Practices, Oct 2015. Accessed May 27, 2020 https://www.cdc.gov/hai/pdfs/prevent/Outpatient-Settings-Policy-Options.pdf .
- 10.The Joint Commission Full-Year 2019 Top Noncompliance Data. Jt Comm Perspect. 2020;40(4):3–30 . [Google Scholar]
- 11.Pogorzelska-Maziarz M, Kalp EL. Infection prevention outside of the acute care setting: results from the MegaSurvey of infection preventionists. Am J Infect Control. 2017. Jun 1;45:597–602 . [DOI] [PubMed] [Google Scholar]
- 12.Anderson L, et al. Challenges to safe injection practices in ambulatory care. Infect Control Hosp Epidemiol. 2017;38:614–616 . [DOI] [PubMed] [Google Scholar]
- 13.Steinkuller F, et al. Outpatient infection prevention: a practical primer. Open Forum Infect Dis. 2018. May 2 5:ofy053. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Centers for Medicare & Medicaid Services Center for Clinical Standards and Quality/Survey & Certification Group. Infection Control Breaches Which Warrant Referral to Public Health Authorities.. May 30, 2014. (Updated: Oct 28, 2016.) Accessed May 27, 2020 https://www.cms.gov/Medicare/Provider-Enrollment-and-Certification/SurveyCertificationGenInfo/Downloads/Survey-and-Cert-Letter-14-36.pdf .
- 15.The Joint Commission. Introduction: How The Joint Commission Can Help You Move Toward High Reliability (IN-TRO) In Comprehensive Accreditation Manual for Office-Based Surgery Practices (E-dition). Oak Brook, IL: Joint Commission Resources, 2020. . [Google Scholar]
- 16.Centers for Disease Control and Prevention. Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care: Appendix A: Infection Prevention Checklist for Outpatient Settings, ver. 2.3, Sep 2016. Accessed May 27, 2020 https://www.cdc.gov/infectioncontrol/pdf/outpatient/guidechecklist.pdf .
- 17.Centers for Disease Control and Prevention Outbreaks and Patient Notifications in Outpatient Settings. Selected Examples. Jul 10, 2015:2010–2014. Accessed May 27, 2020 https://www.cdc.gov/hai/settings/outpatient/outbreaks-patient-notifications.html .
- 18.Centers for Disease Control and Prevention. Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care, ver. 2.3, Sep 2016. Accessed May 27, 2020 https://www.cdc.gov/infectioncontrol/pdf/outpatient/guide.pdf .
- 19.Centers for Disease Control and Prevention. Tools for Healthcare Settings, Jul 10, 2019. (Updated:.) Accessed May 27, 2020 https://www.cdc.gov/infectioncontrol/tools/index.html .
- 20.The Joint Commission Preventing infection from the misuse of vials. Sentinel Event Alert. Jun 16, 2014(52). Accessed May 27, 2020 https://www.jointcommission.org/assets/1/6/SEA_52.pdf . [PubMed]
- 21.Dolan SA, et al. APIC position paper: Safe injection, infusion, and medication vial practices in health care. Am J Infect Control. 2016. Jul 1;44:750–757 . [DOI] [PubMed] [Google Scholar]
- 22.Taylor D, et al. Evolution of ELC healthcare-associated infections/antimicrobial resistance (HAI/AR) programs, 2009–2018 Paper presented at the Council of State and Territorial Epidemiologists Annual Conference, Jun 11, 2018. Abstract accessed May 27, 2020 https://cste.confex.com/cste/2018/meetingapp.cgi/Paper/9828 .
- 23.Centers for Disease Control and Prevention. CDC/STRIVE Infection Control Training. (Updated: Nov 21, 2019.) Accessed May 27, 2020 https://www.cdc.gov/infectioncontrol/training/strive.html.
- 24.CDC Train. Nursing Home Infection Preventionist Training Course. Feb 15, 2019–Feb 25, 2032, 2020. Accessed May 27 https://www.train.org/cdctrain/training_plan/3814.
- 25.Wendelboe AM, et al. Outbreak of cystoscopy related infections with Pseudomonas aeruginosa: New Mexico, 2007. J Urol.. 2008;180:588–592 . [DOI] [PubMed] [Google Scholar]
- 26.Aumeran C, et al. Multidrug-resistant Klebsiella pneumoniae outbreak after endoscopic retrograde cholangiopan-creatography. Endoscopy. 2010;42:895–899 . [DOI] [PubMed] [Google Scholar]
- 27.Gastmeier P, Vonberg RP. Klebsiella spp. in endoscopy-associated infections: we may only be seeing the tip of the iceberg. Infection.. 2014;42:15–21 . [DOI] [PubMed] [Google Scholar]
- 28.Centers for Disease Control and Prevention, Healthcare Infection Control Practices Advisory Committee. Essential Elements of a Reprocessing Program for Flexible Endoscopes—Recommendations of the Healthcare Infection Control Practices Advisory Committee. (Updated: Jun 28, 2017.) Accessed May 27, 2020 https://www.cdc.gov/hicpac/pdf/flexible-endoscope-reprocessing.pdf.
- 29.US Food and Drug Administration. Press Release: FDA Recommends Health Care Facilities and Manufacturers Begin Transitioning to Duodenoscopes with Disposable Components to Reduce Risk of Patient Infection. Aug 29, 2019. Accessed May 27, 2020 https://www.fda.gov/news-events/press-announcements/fda-recommends-health-care-facilities-and-manufacturers-begin-transitioning-duodenoscopes-disposable.
- 30.US Food and Drug Administration, Center for Devices and Radiological Health. Marketing Clearance of Diagnostic Ultrasound Systems and Transducers: Guidance for Industry and Food and Drug Administration Staff. Jun 27, 2019. Accessed May 27, 2020 https://www.fda.gov/media/71100/download .
- 31.Williams SC, et al. Comparing public quality ratings for accredited and nonaccredited nursing homes. J Am Med Dir Assoc. 2017;18:24–29 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


