Abstract
In recent years a revival of interest has emerged in the health benefits of intermittent fasting and long-term fasting, as well as of other related nutritional strategies. In addition to meal size and composition a new focus on time and frequency of meals has gained attention. The present review will investigate the effects of the main forms of fasting, activating the metabolic switch from glucose to fat and ketones (G-to-K), starting 12–16 h after cessation or strong reduction of food intake. During fasting the deactivation of mTOR regulated nutrient signalling pathways and activation of the AMP protein kinase trigger cell repair and inhibit anabolic processes. Clinical and animal studies have clearly indicated that modulating diet and meal frequency, as well as application of fasting patterns, e.g. intermittent fasting, periodic fasting, or long-term fasting are part of a new lifestyle approach leading to increased life and health span, enhanced intrinsic defences against oxidative and metabolic stresses, improved cognition, as well as a decrease in cardiovascular risk in both obese and non-obese subjects. Finally, in order to better understand the mechanisms beyond fasting-related changes, human studies as well as non-human models closer to human physiology may offer useful clues.
KEY-MESSAGES
Biochemical changes during fasting are characterised by a glucose to ketone switch, leading to a rise of ketones, advantageously used for brain energy, with consequent improved cognition.
Ketones reduce appetite and help maintain effective fasting.
Application of fasting patterns increases healthy life span and defences against oxidative and metabolic stresses.
Today’s strategies for the use of therapeutic fasting are based on different protocols, generally relying on intermittent fasting, of different duration and calorie intake.
Long-term fasting, with durations between 5 and 21 days can be successfully repeated in the course of a year.
Keywords: Glucose-to-ketones metabolic switch, intermittent fasting, long-term fasting, periodic fasting, prolonged fasting, zero calorie diet
1. Introduction
In the evolution alternation of food availability and food scarcity has been the consequence of seasonal cycles with variations in sunlight exposure. Before humans could efficiently conserve food, physiological and behavioural adaptations to survive periods of food limitation, have led to the development of the metabolic fasting programmes. Storing food as fat in the adipose tissue instead of carbohydrates or proteins, was a highly efficient solution for humans and animals to cope with the absence of food supply and it permitted to fuel prolonged periods of fasting [1,2]. In free living animals fasting is coupled with migration, hibernation or huddling to save energy. Bears [3] and ground squirrels [4] reduce energy expenditure by hibernating and undergo prolonged fasting periods of months with no or little food [5], whereas Emperor penguins fast during moulting and breeding. During fasting, they can stand colder conditions better than any other bird, while maintaining their usual body temperature [6] thanks to the huddling behaviour [7,8]. Birds can undertake strenuous efforts during long distance migration, while flying hundreds of kilometres without food or drink [9].
Humans also had to adapt to natural cycles of food scarcity for millions of years and only with the onset of agriculture in the Neolithic era, more continuous food supply became available. This culminated in today’s technologies allowing humans in privileged countries to have access to any type of food at any time during the whole year. Extreme situations such as famine [10], chronic malnutrition, hunger strikes or long course of anorectic denial of food [11,12] have also provided information about the human capacity to cope with long periods of food shortage. In addition to being a necessity for human survival, the ability to fast has been known in most religions as traditionally ritualised periods lasting from hours to weeks [13]. Religions used fasting empirically for its effects on mind and body, as well as an important factor in community cohesion [14]. In medicine fasting for 2–21 days or more was known for its numerous therapeutic effects that led to multidisciplinary fasting programmes [15], well-known in the public less in the scientific community. Total fasting safely practiced for 31 days has been documented scientifically at the beginning of the twentieth century [16]. Later on, in the 1960s, as obesity started to be a medical issue on a large scale, water fasting to treat morbid obesity and co-morbidities emerged under the names of zero calorie diet or total fasting [17]. The effects of this type of fasting, lasting weeks or months, particularly on obesity, have been extensively documented [18].
Nowadays a revival of the interest for fasting emerges from a different perspective. Instead of focussing only on weight loss in morbid obesity, the new focus is on the effects of the main fasting regimens, that activate the metabolic switch from liver-derived glucose to adipose cell-derived ketones (G-to-K) and its reversal K-to-G [19], on longevity, health span expansion, multi-stress resistance and antioxidant defence stimulation, improved performance and cellular regeneration in animal models and humans.
The present review investigates, in animal models and humans, how application of several types of fasting strategies, e.g. intermittent fasting, periodic fasting, or long-term fasting could be part of a new medical approach leading to improved healthy life span and cognition, as well as to a decrement in cardiovascular risk in both obese and non-obese subjects.
2. Fasting classification
Fasting is defined as the voluntary abstinence or strong limitation of caloric ingestion for a limited period of time, triggering the G-to-K switch and major changes in the activity of signalling pathways. Further effects also take place on refeeding when the K-to-G switch occurs [19]. The big challenge when it comes to classify fasting regimens is that duration has a very different meaning if we consider animals, such as non-obese mice, or humans. One day fasting brings the mouse almost to starvation (ketones decrease and protein catabolism increases), whereas a day in humans is considered as intermittent fasting [20].
Table 1 summarises the main forms of fasting and other restrictive diets. Calorie restriction (CR) refers to a daily reduction by 15–40% of calorie intake without malnutrition. Intermittent fasting (IF) refers to fasting lengths between 16 and 48 h, alternated with usual food intake. The most frequently studied IF procedure is alternate day fasting (ADF) [21], whereby normal food intake occurs on one day and restricted food the next day. Time restricted eating/feeding (TRE/TRF) is characterised by intake of food occurring within a time window of 8–12 h per day or less [22]. Periodic fasting (PF) describes cycles of fasting or calorie restricted diets, i.e. 5:2 diet (5:2), referring to two days per week, consecutive or not, in which food intake is drastically reduced to approximately 600 kcal [23]. In some classifications 5:2 is considered as IF. Periods of fasting lasting from many days to weeks have sometimes been referred to as periodic and can be repeated every year [24]. Other Authors referred to them as long-term or prolonged fasting [25]. Considering that these adjectives do not take into consideration differences in the baseline nutritional status and between species, in the present review article we refer to long-term fasting (LF) as a food abstinence from 2 to 21 days or more during which no or minimal amounts of calories, up to 200–250 kcal/days are given within appropriate schedules. LF has been well documented in obese and non-obese subjects [24] as well as in animals [7,26] and during the 1960s in morbidly obese subjects [27]. Other regimens, derived from fasting strategies, are the very-low-calorie-diets (VLCD) [28], a hypocaloric formula diet providing 80–100 g of proteins and an average of 1000 kcal/days, designed to treat obesity and to avoid a negative nitrogen balance. Furthermore, the fasting-mimicking diet (FMD) [29,30] is a hypocaloric (800–1100 kcal), low protein, ketogenic diet leading to weight loss and to some of the effects of fasting. It is worth mentioning that nutrient restricted normocaloric diets like the ketogenic diet (carbohydrate restriction) and the protein or amino acid (methionine) restricted diet [31] are being also intensively evaluated.
Table 1.
Main forms of fasting and related diets.
| Fasting and related diets | Calorie intake | Length |
|---|---|---|
| Calorie Restriction (CR) | ∼70 % of normocaloric intake without malnutrition (with possible variations) | Optional |
| Intermittent Fasting (IF) | ||
| Alternate Day Fasting (ADF) | 0 kcal/alternating with ad libitum (with possible variations) | Optional; alternation of fasting and ad libitum food intake every other day |
| Time Restricted Feeding/Eating (TRF/TRE) 16:8 | Ad libitum with possible variations | Optional; daily food intake is reduced to a time window of 8 h (with possible variations) |
| Periodic Fasting (PF) | ||
| 5:2 diet (5:2) | 600 kcal/2 days per week/ad libitum | Two consecutive or independent days of calorie restriction every week |
| Long-term (LF) or prolonged Fasting | ||
| Buchinger Wilhelmi fasting | 200–250 kcal/day | >2 days to weeks according to baseline nutritional and health status |
| Water-only fasting | 0 kcal/day | |
| Zero calorie diet | 0 kcal/day | Weeks to months. Weight-loss program practiced between the 1960s and the 1970s |
| Very Low Calorie Diet* (VLCD) | 800–1000 kcal | High protein formula weight-loss program with reduced calorie intake for 6 -12 weeks |
| Fasting Mimicking Diet* (FMD) | 800–1100 kcal | 5 days of calorie reduced low protein diet (1 to 6 cycles per year) |
Diets derived from fasting strategies.
3. Metabolic and cellular responses to fasting
3.1. The metabolic switch
The onset of fasting is characterised by the metabolic switch, that defines the fuel switch occurring when an organism commutes from the eating mode, including generally around 50% carbohydrates, to the fasting mode [19]. From 12 to 16 h after interrupting food absorption, glucose levels drop, followed by a decrease of insulin levels and at the same time of circulating amino acid levels. The onset of the metabolic switch depends on the liver glycogen content at the beginning of the fast as well as on the composition of the preceding meal, energy expenditure, and physical activity [19]. Lipids in adipocytes (triacylglycerol and diacylglycerol) are metabolised to free fatty acids (FFAs) whose levels increase in fasting blood, to be partly oxidised in most tissues, such as muscles, kidneys, heart and partly transformed in the liver to ketones (β-hydroxybutyrate, acetoacetate and acetone) [32]. Ketones are not only fuels, since they have also signalling effects and regulate expression and activity of transcription factors like the peroxisome proliferator-activated receptor γ coactivator 1α (PGC-1α), sirtuins (SIRTs) [33], poly-adenosine diphosphate [ADP]-ribose polymerase 1 (PARP1), and ADP ribosyl cyclase [34], as well as fibroblast growth factor 21 [35,36] and nicotinamide adenine dinucleotide (NAD+). A rise of brain-derived neurotrophic factor (BDNF) in the central nervous system (CNS) can enhance brain health [37].
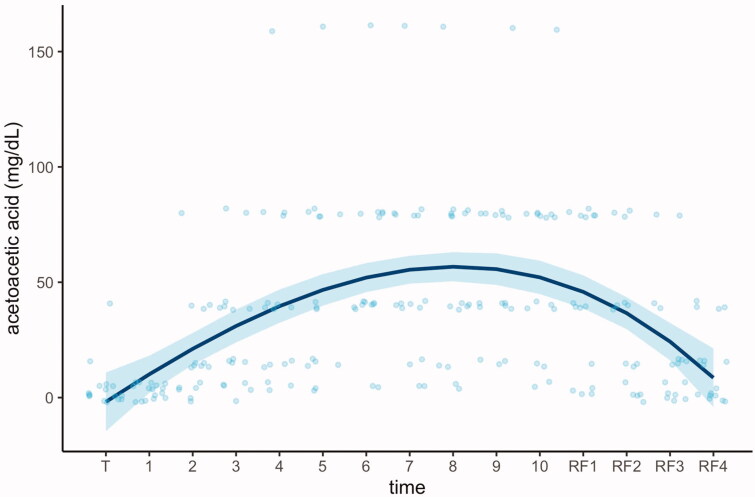
Ketones are effectively oxidised by the fasting brain, more reluctant than other tissues to renounce glucose and metabolise FFAs [38]. The use of FFAs and ketones as energy sources reduces the respiratory-exchange ratio (RER), compared with the fed state, indicating a greater metabolic efficiency of energy production during weight loss leading to ketosis [25]. The decreased ATP/AMP ratio will activate AMP-activated protein kinase (AMPK) as well as create a mild oxidative eustress [39] leading to the activation of antioxidant and cytoprotective enzymes (i.e. superoxide dismutase, catalase, peroxidase, sulfiredoxin 1, thioredoxin reductase 1, haem oxygenase-1, NAD(P)H quinone oxidoreductase 1, glutamate-cysteine ligase, glutathione S-transferases, and uridine 5′- diphospho-glucuronosyltransferases). During prolonged periods of fasting, ketosis has been shown to reach a plateau after 4 days, an effect that can last for several hours or days, and to decrease when food is reintroduced (Figure 1) [40].
Figure 1.
Metabolic switch from carbohydrates to fatty acids and ketones induced by 10 days of fasting (daily energy intake of about 250 kcal and multidisciplinary programme). A regression spline was fitted on individual acetoacetic values to show the variations in ketosis during the course of the study. T: transition to the fasting mode; RF: progressive reintroduction of food [40].
The G-to-K switch is the key mechanism allowing to spare proteins by reducing protein utilisation, as reflected by the changes in nitrogen balance [41]. It is regulated by inhibition of the mTOR (mechanistic/mammalian target of Rapamycin) signalling pathway, thus decreasing protein synthesis and enhancing autophagy, that leads to the recycling of endogenous proteins [25]. Relying on ketones for energy during weight loss has the advantage of promoting retention of lean mass; the same may not occur after plain CR [42]. This conclusion was not supported in the most extensive ADF studies to date [21]. At present, the substantial benefit of fasting regimens on lean mass retention needs to be reconsidered. While the adaptation to long-term fasting can promote lean mass retention, this has not been definitely shown to occur preferentially after IF rather than with CR [43]. It should be noted that ketosis can be also obtained with low energy and/or high fat diets [44]. Normocaloric strict high fat ketogenic diets are successfully prescribed to epileptic children who do not respond to drug treatments [45].
Switching from a carbohydrate to a lipid rich diet has been successfully applied for weight reduction, although, based on current evidence, low-CHO and very-low-CHO diets do not appear to be clearly superior to other dietary approaches [44]. Ketones serve as an energy source, being metabolised to acetyl coenzyme A (AcCoA) entering the tricarboxylic acid cycle generating adenosine triphosphate (ATP). They sustain the function of muscle and brain cells during fasting as well as extended periods of physical exertion [46]. Furthermore, ketosis leads to a fully compensated acidosis, believed to be responsible for the characteristic absence of hunger during fasting [47–49]. Absence of hunger enhances compliance [24]. Blood glucose stabilises at the lower normal level and will remain stable during the whole fasting period, as long as fat reserves can fuel metabolism and the protein pool remains at the physiological limits [46].
Concomitantly, in a well-orchestrated manner, an increase in the hormonal secretion of glucagon (stimulation of glycogenolysis and gluconeogenesis) [50], growth hormone (implicated in lipolysis) [51], cortisol and adrenaline regulate the course of fasting [52]. Furthermore, along with a decrement in the adipokine leptin and a rise in adiponectin [53], fasting significantly reduces insulin like-growth factor (IGF)-1. Only restrictions of 50% or more of normal daily energy requirements can reduce IGF-1 levels [54]. Low levels of IGF-1 reduce the intracellular mitogenic signalling pathways, and lead mammalian cells to enter either a non-dividing or a low-dividing state and invest energy resources into cellular protection against various insults (multi-stress resistance) [55].
Since during fasting very few or no exogenous proteins or sugars enter the system, nutrient dependent signalling pathways are shut down [mTOR and Ras-adenylate cyclase (AC)-protein kinase A (PKA)], along with the de-repression of transcription factors e.g. SIRT, and forkhead box O1 (FOXO1), leading to the many peculiar features of the fasting mode [37]. The promotion of the multi-stress resistance is characterised by improved antioxidant defences, increased DNA repair and diminished inflammation [56]. Moreover, most of cardiovascular risk factors (waist circumference, lipids, blood pressure and insulin resistance) are improved by CR, IF or LF [57,58].
The fasting mode will be reversed as soon as a mixed diet is reintroduced, triggering the K-to-G switch: glucose and insulin levels increase, ketones drop, mTOR is reactivated leading to increased protein synthesis and mitochondrial biogenesis, as well as decreased autophagy. Cell regeneration in multiple systems occurs, with increased mesenchymal and progenitor cells [29]. In this refeeding phase, specific effects take place concomitant to cell growth and plasticity, leading to functional tissue remodelling [25] and offering a unique opportunity, in adult life, to boost cellular and tissue regeneration [30]. In some cases, an inappropriate pattern of food reintroduction can lead to relapse of symptoms, as seen in the case of LF prescription to treat polyarthritis [59].
3.2. Signalling pathways modulated by fasting
In the fasting mode, cellular and metabolic processes are controlled by a complex network of transcriptional regulators. Major regulators are SIRTs, nuclear factor erythroid 2–related factor 2 (NRF2), FOXO1, nuclear factor ‘kappa-light-chain-enhancer’ of activated β-cells (NFkB), hypoxia inducible factor 1 α (HIF-1α), heat shock factor (HSF-1). The decrease of the protein responsive signalling pathway mTOR and of its downstream effector, the ribosomal protein S6 kinase β-1, leads to global protein synthesis inhibition and recycling of macromolecules by autophagy stimulation [60]. In the brain, in addition to raised neuronal stress resistance through bolstered mitochondrial function there is an improvement in antioxidant defences, DNA repair, and stimulation of BDNF production [61,62]. BDNF regulates hippocampal neurogenesis, dendrite morphology and synapse plasticity, and increases production of new neurons from neural stem cells [63]. The decrease in glucose levels in parallel to the rise in ketones during fasting is associated with a decrement in the glucose responsive Ras-AC-PKA pathway, implicated in life span extension [64].
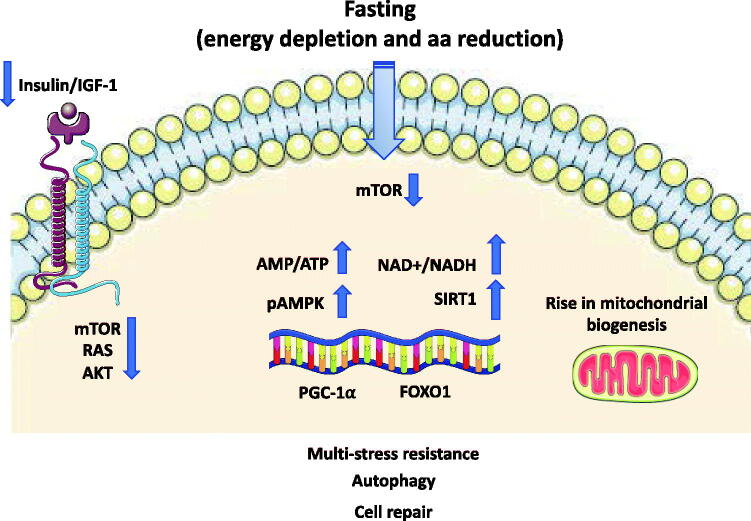
Other crucial consequences of fasting are the decrease in insulin/IGF-1 signalling, leading to a reduction in anabolic processes and decrement in the ATP/AMP ratio with consequent activation of AMPK. This last step triggers repair and inhibition of the anabolic processes [65] (Figure 2). Another mechanism associated to the fasting-induced activation of AMPK is autophagy of muscle cells, a process preserving blood glucose levels during LF. As a proof-of-concept, the selective skeletal muscle depletion of AMPK resulted in hypoglycaemia and hyperketosis, an effect not due to an impairment in fatty acid oxidation, but instead to a reduction in autophagy of muscle cells, leading to reduced circulating levels of alanine, an essential amino acid required for gluconeogenesis [66].
Figure 2.
Representationof signalling pathways modulated fasting. The reduced levels of circulating amino acids and of IGF-1 consequent to fasting repress the activity of mTOR and its downstream effector leading to an inhibition of global protein synthesis and promote recycling of macromolecules by autophagy stimulation. There is a rise in the AMP-to-ATP ratio leading to the activation of AMPK. SIRT1-driven deacetylation of PGC-1α and FOXO1 transcription factors provides a mechanism by which mitochondrial and lipid oxidation genes can be dynamically controlled in response to energy demand. AMPK: AMP-activated protein kinase; FOXO1: forkhead box O1; IGF: insulin-like growth factor; NAD+: nicotinamide adenine dinucleotide; PGC-1α: peroxisome proliferator–activated receptor γ coactivator 1α; mTOR: mammalian target of Rapamycin; SIRT: sirtuin.
Among the many activated transcriptional regulators quoted above, hepatic SIRT1 protein levels are raised during fasting and reduced with refeeding [67]. SIRT1-driven deacetylation of PGC-1α [68] and the increased FOXO1 transcription factor provides a mechanism by which mitochondrial and lipid oxidation genes can be dynamically controlled in response to energy demand [69]. A specific liver knock down of SIRT1 results in a decreased fasting-dependent down-regulation of the lipogenic gene expression (SREBP-1c), suggesting this mechanism for the regulation by SIRT1 of multiple SREBP-1 target genes [70]. SIRT1 suppresses glucose production by inhibiting the CREB regulated transcription coactivator 2 (CRTC2)-mediated gluconeogenesis, activated during the G-to-K switch [71]. Similar to SIRT1, the mitochondrial deacetylase SIRT3 regulates metabolic homeostasis during fasting and CR [72]. SIRT3 is critical for fatty acid oxidation and ketogenesis during fasting, by regulating the deacetylation state and the activity of mitochondrial enzymes involved in the metabolic switch. Having thus a major role in liver ketogenesis, mice lacking SIRT3 display lower plasma β-hydroxybutyrate levels during fasting [72].
4. Evidence from experimental animals
The life and health span extension produced by CR has been an area of interest since the late 1970s [73]. Compared to mice assigned to an ad libitum regimen, 30% CR and TRF (single-meal feeding) enhanced longevity, regardless of diet composition and showed improvements in morbidity and mortality [74]. Prevention of age-related declines, as evaluated by tests of motor coordination (rotarod) and learning (complex maze) took place in mice exposed to a 50% CR regimen. Increased locomotor activity in a runwheel cage, regardless of age was observed, without effects on exploratory activity in a novel arena [75]. This type of functional improvement has been related to anatomical changes, such as a rise in synapse numbers and stimulation of mitochondrial biogenesis, correlated with increased BDNF levels [76]. 3-hydroxybutyrate may also mediate adaptive responses of neurons to fasting, exercise and ketogenic diets [77]. In mice, hippocampus dependent spatial learning and memory deficit improve upon a daily TRF with ameliorated behavioural changes, in particular anxiety-like behaviours. These improvements, found in old mice, may rely on the enhanced cerebral blood flow and blood-brain barrier function, similar to that seen in young mice at 5–6 months of age following CR [78].
Relative to TRE, the study of mechanisms leading to changes in the molecular circadian clock genes, activated or deactivated according to light and diet, has opened new horizons [65]. These circadian rhythms, displaying oscillations over a period of 24 h, are evolutionarily conserved and driven by the need to synchronise biological activity with the ever-changing, but predictable, environment of the rotating Earth. In mammals, feeding behaviour is cyclic with periods of fasting separating feeding bouts, leading the organism to switch from nutrient storage during feeding periods to the use of stored nutrients during fasting [22]. Studies on the effects of a disruption of metabolic circadian cycles, showed that under TRF, in which mice are fed an equivalent amount of food as the ad libitum cohort but for a defined period of 8 h, mice were protected against obesity, hyperinsulinemia, hepatic steatosis, and inflammation [60]. Compared to rats fed ad libitum, those exposed to ADF present lower resting heart rate and blood pressure (BP), this last remaining lower after exposure to immobilisation and swim stressors [79], and equal to or greater than that previously obtained with exercise training regimens in rats [80].
Other Authors confirmed that TRF regimens, compared to prolonged ad libitum feeding are associated to an improved hemodynamic profile, i.e. reduced heart rate and systolic and diastolic BP. TRF decreases the sympathetic autonomic nervous system activity and raises the parasympathetic or vagal tone [81]. Another TRF regimen of 4 h-feeding/day in mice restored the expression of clock genes, leading to reduced body weight, cholesterolaemia, tumour necrosis factor-α and improved insulin sensitivity [82]. These findings suggest that TRF can normalise the expression of genes involved in fatty acid metabolism, β-oxidation, PPARγ and antioxidant defences in the liver. The correlation between clock genes and TRF has recently been documented by a study in mice lacking a circadian clock. When these are provided with access to high energy food ad libitum they gain weight quickly, but when subjected to a TRF regimen (10 h), they are protected from the weight gain and metabolic diseases driven by a high-energy diet [83].
Prolonged ADF can upregulate the hemo-oxygenase1 and glucose-regulated protein 7B, both reduced by aging, in the cerebral cortex and striatum. These cytoprotective proteins against metabolic and oxidative stresses [84] rise after ADF and appear to sustain expression/responsivity of genes involved in adaptive neuroplasticity and cognition [85]. Similarly, in the case of animal models of neurologic disorders, including epilepsy, Alzheimer, Parkinson disease and stroke, application of an IF regimen (feeding for 8 h/day) appears to protect against the neurological damage in ischaemic stroke, with circulating leptin as a possible mediator [86]. Mice maintained on a chronic IF for 11 months exhibited a superior cognitive ability in the Barnes maze test of spatial memory [53]. This type of positive response also occurred after a late-onset short-term IF dietary restriction for three months in old rats (animals fasted overnight) [87].
A better cognitive performance was also reported when mice were fed cycles of FMD lasting 4 days, followed by a standard ad libitum diet. Compared to control mice, FMD resulted in a rise in life span, i.e. up to 28.3 vs 25.5 months. Bi-monthly application of this dietary approach reduced visceral fat, cancer incidence and skin lesions, rejuvenated the immune system, and retarded bone mineral density loss; when applied later in life, there was improved cognitive performance [29].
5. Therapeutic fasting: clinical impact in contemporary medicine
Modulating diet and meal frequency as well as several patterns of fasting can represent a new paradigm in today’s medical approaches. As recently highlighted by de Cabo and Mattson, Homo sapiens has adapted to fasting in ways that enable the organism to tolerate or overcome challenges and then restore homeostasis [25]. An increasing number of physiological effects linked to IF can be applied also to LF, at least in the initial phase, leading to (i) weight loss and metabolic reset, (ii) increased insulin sensitivity, (iii) reduction in inflammation and oxidative damage to proteins, lipids and DNA, as well as (iv) enhanced immune system function [88]. All these mechanisms are definitely of value in the management of obese as well as non-obese subjects.
5.1. LF – a general overview
Whereas the metabolic switch and the changes in signalling pathways above described also apply at the beginning of LF, the question is whether these effects persist, decrease or increase in the course of LF. Evidently, it will depend on the duration of fasting, the individual profile including age, nutritional and health status, as well as personal inclination for the procedure [89]. One of the first detailed scientific observations of a non-obese voluntary subject on total fasting ended safely after 31 days [16]. Other case reports of persons fasting during several weeks were published and brought stupefaction in the medical community and the public. Many Authors have stressed the well-being, the absence of hunger and lifted mood reported by fasting obese and non-obese subjects [90]. The human capacity to live without energy intake for periods of almost 40 days has been often reported in non-obese subjects during hunger strikes [91].
A medically supervised practice of LF has a long tradition in Europe and in particular in Germany. The safety of this programme has been recently documented in large cohort studies [15,24]. This type of LF, generally lasting from 4 to 21 days, has been studied in various clinical conditions: chronic inflammatory disorders [92,93] and rheumatoid arthritis [59,94], hypertension [95], irritable bowel syndrome [96], insulin resistance, type 2 diabetes and metabolic syndrome [97,98], fibromyalgia [99], breast and ovarian cancer [100], osteoarthritis [101], obesity [102], and fatty liver [103]. Furthermore, MRI evaluation documented significant changes in body composition after 14 days of fasting [104].
LF was first documented in morbidly obese subjects in numerous publications from 1959 [90] to 1975 [41], reporting fasting periods from several days up to 249 or, in an extreme case report, to 382 days [105,106], called “zero calorie diets” or “total fasting,” sometimes with administration of multi-vitamins and potassium supplements. The main concern at that time was to elucidate how humans could fast that long, having a limited possibility to break down their protein pool and at the same time the need to provide their CNS with energy: the daily calorie needs of the brain in the eating phases are 400–570 kcal delivered by 100–145 g glucose [46]. If these needs have to be met by gluconeogenesis, since fatty acids cannot be transformed into glucose, this would mean catabolizing 200 g of proteins daily. Since the human protein body pool is roughly 6000–8000 g [107] and cannot drop more than 1/3 [108], this would reduce the ability to fast for long periods. In fasting subjects, after depletion of the glycogen stores, a rapid decrease in carbohydrate oxidation and protein catabolism takes place. Measurement of urinary nitrogen excretion allowed to determine protein utilisation. Whereas at the beginning of fasting 35 g of nitrogen were excreted per day, these values tended to fall to 2–4 g/day (12–25 g protein/d) after 4 weeks [109]. The brain was increasingly fed with ketone bodies, especially β-hydroxybutyrate, keeping it alert in order to stay able to face challenges [46,110].
The protein sparing mechanism is induced by the metabolic switch of G-to-K, as described by Longo & Mattson decades later [111]. Cahill demonstrated the fuel change occurring in the brain after 38–41 days of fasting [46]. The brain system metabolises only 30% of glucose supplied by the glycerol molecule, liberated after cleavage of three fatty acids from triglycerides by lipoprotein lipase. Altogether, during total fasting, up to 41 days, 86 g/24h glucose is produced: half of this comes from blood cells, producing lactate and pyruvate through glycolysis, then resynthesized to glucose in the liver. The other half comes from glycerol and gluconeogenesis, taking place first in the liver and then in the kidneys from amino acids, respectively alanine and glutamine [112]. This shift produces ammonium to titrate ketone bodies [113].
Three phases have been described in the course of LF in animals, such as the Emperor penguin, that undergo yearly, during 35 years (average life span) fasting periods lasting several weeks until exhaustion of energy reserves [114]. An initial phase 1, when the metabolic switch occurs, is followed by a phase of protein sparing called phase 2, lasting several days to weeks. Phase 3 finally starts when fat and protein reserves have reached a threshold, forcing animals to refeed [115]. Phase 3 is characterised by a high catabolic activity, as well as adrenergic stimulation and underlines the urgent need to replenish energy stores. Nevertheless, the process is fully reversible. In humans these three phases seem to exist in a similar way. Under physiological condition of therapeutic fasting, phase 3 is not reached. Phase 3 has been described in anorexia [11].
The zero-calorie diet, where only non-caloric beverages are permitted, was relatively well tolerated for several weeks or even months, even though obese persons were often without exercise, psychological support or nutritional education to prevent relapses. Pioneers of this type of fasting to treat obesity made even the recommendation – surprising for our contemporaries – that obese subjects should not fast longer than 100 days without nitrogen balance monitoring [116]. The publication of a single case of death caused by acute intractable heart arrest happened on the seventh refeeding day in a 20-year-old woman [117]. She had been on a 30 week zero calorie diet, reducing her weight from 118 to 60 kg. By simple calculation this would mean a protein utilisation of 4000 g, i.e. more than 50% of her protein pool, this being the most plausible death aetiology. At autopsy a decrease of myofibril diameter in the heart with gross fragmentation was diagnosed. Isolated cases of death by very prolonged zero calorie diet periods of several weeks or months in morbidly obese had not always such a clear aetiology; other possible causes of death include factors such as potassium or vitamin deficiencies, imperfect patient selection or compliance, associated medications or pre-existing disease [118–120].
For several years, zero calorie diets were followed by a medical team in hospital wards. Despite the polymorbidity of the patients, the procedure seemed to be safe for periods of 60–100 days. Because of the long duration, the costs for hospitalisation were considered as too elevated and, as a consequence, people started total fasting on their own. Furthermore, obese subjects could also buy protein supplemented diet formulas in supermarkets, without any supervision. One of these products consisting of a liquid based hydrolysate of collagen proteins, the so called “liquid protein diet” (LPD) was linked to deaths from cardiac arrest in 32 out of 44 casualties [118]. Again, the histological diagnosis was myofibrillar gross fragmentation but, unlike the first documented case, the subjects were still obese. The poor protein quality of LPD might have played an accelerating role in protein pool depletion or else in depletion of specific amino acids.
After these events, the depletion of the protein pool was incriminated and this led to a strict regulation of what has been called VLCD (very low calorie diet), a hypocaloric formula providing 80–100 g good quality protein/day. The main objective was to avoid a negative nitrogen balance measured by 24 h urinary excretion [109]. VLCD were prescribed to last not more than 6 weeks. By reflection, this reduction of the fasting period alone could have sufficed to reduce risk drastically. Protein formulas provided a market allowing to design ambulatory multidisciplinary programmes. The need to provide proteins was not questioned any more until today.
5.1.1. Impact of LF on health and well-being
The practice of LF has reached a wide network of utilisation in Europe. Among 1422 subjects who followed fasting periods (daily calorie intake of 200–250 kcal accompanied by a multidisciplinary lifestyle programme) lasting between 4 and 21 days, there were significant reductions in body weight, between 3.2 ± 0.0 kg after 5 and 8.6 ± 0.3 kg after 20 days of fasting, as well as in abdominal circumference, i.e. between 4.6 ± 0.1 cm and 8.8 ± 0.8 cm, respectively. BP decreased for the whole group from 131.6 ± 0.7 to 120.7 ± 0.4 for systolic BP and from 83.7 ± 0.4 to 77.9 ± 0.3 for diastolic BP [24]. A reduction of total cholesterol (−0.4 ± 0.0 mmol/L), triglycerides (−0.4 ± 0.0 mmol/L), glucose (−0.7 ± 0.1 mmol/L) and HbA1c (−1.2 ± 0.1 mmol/mol) was reported. The absence of hunger feeling, documented in 93.2% of the subjects and an increase of emotional and physical well-being was documented. None of the subjects dropped out of the fasting procedure. Adverse effects, e.g. cardiac arrhythmia, were reported in less than 1% and mild symptoms like headache and fatigue occurred rarely and mainly in the first days. In another study on 174 hypertensive patients, who underwent a water-only fasting programme (approximately 10 days), 90% of the subjects achieved a reduction of BP to below 140/90 mm Hg [121].
An improved fatty liver index (FLI), a surrogate marker of non-alcoholic fatty liver disease (NAFLD), has been recently described after LF in subjects with and without type 2 diabetes [103]. In a series of 697 subjects, out of whom 264 had a baseline FLI > 60 (threshold for fatty liver), a LF of 8.5 ± 4.0 days was carried out, providing 250 kcal/day and large quantities of water. FLI decreased in the whole cohort significantly (−14.02 ± 11.67), the largest benefit being noted in diabetics (−19.15 ± 11.0). BMI decreased by 1.51 ± 0.82 kg/m2, 50% of the subjects losing more than 5% body weight. Improvement of FLI was significantly correlated with the number of fasting days and with the magnitude of BMI reduction [103].
5.1.2. LF and microbiota
The contribution of gut microbiota to human diseases is being intensively studied, such as in the case of inflammatory bowel disease, gastric ulcers, NAFLD, obesity, metabolic syndrome as well as associated neurologic disorders [122]. This has led to a detailed evaluation of dietary changes since gut microbiota relies, almost entirely, on host diet composition and food processing capacity, to obtain the metabolic substrates needed to cover its energy requirements (150–450 kcal/d, 628–1883 kJ/d). Thus, it seems inevitable that periods of fasting may have an impact on gut microbiota [123]. Ten days of fasting in 15 healthy men led to a decrease in abundance of Lachnospiraceae and Ruminococcaceae, as shown by faecal 16S rRNA gene amplicon sequencing, with a concomitant rise in Bacteroidetes and Proteobacteria (Escherichia coli and Bilophila wadsworthia). These changes were associated with an increase in faecal branched-chain amino acids (BCAA) by 18%, possibly coming from host-derived compounds such as desquamated cells attacked by the microbiota. The effects were reversed three months after fasting [40]. In obese women on a VLCD (800 kcal/day) for 4 weeks major consistent changes in dominant faecal bacterial communities were reported [124].
A potential benefit of microbiomal changes after fasting is the amelioration of the altered gut microbiome in relapsing-remitting multiple sclerosis [125]. Since CR has a clear anti-inflammatory potential and chronic CR has been shown to attenuate autoimmune encephalomyelitis [126], therapeutic fasting may be of value in the management of CNS autoimmunity. At present, experimental data have shown raised gut bacterial richness after IF, with enhanced anti-oxidative microbial metabolic pathways [127]. In the same report the Authors describe similar microbiomal changes following IF in a series of MS patients. There are at present only plans to test PF. A controlled clinical trial on LF is ongoing in metabolic syndrome (NCT02099968), as well as on IF in relapsing-remitting multiple sclerosis (NTC03539094).
5.2. Impact of CR and IF on cardiometabolic risk factors in non-obese and obese subjects
The CALERIE (Comprehensive Assessment of the Long-Term Effects of Reducing Intake of Energy) study [128,129], enrolled participants with BMI between 22 and 28 kg/m2, proposing a 25% CR throughout a 2-year period with a control group staying on their current diet. CR significantly improved general health and mood [130]. Weight, body fat, fat mass, and fat free mass as well as cardiometabolic risk factors, i.e. lipids, BP, C-reactive protein, and insulin sensitivity index were significantly reduced compared to controls [131–133]. Concerning obese subjects, the TEMPO (Type of Energy Manipulation for Promoting Optimum Metabolic Health and Body Composition in Obesity) trial compared the long-term impact of a 25–35% CR during 12 months vs a 65–75% CR during 4 months followed by 8 months of a more moderate energy restriction. Both interventions had a prescribed protein intake of 1.0 g/kg of baseline body weight per day. Among the 101 recruited postmenopausal women, those on the 65–75% CR lost more abdominal subcutaneous and visceral adipose tissue, but they also lost more whole body and thigh muscle lean mass, proportional to the total weight loss. Moreover, participants allocated to the severe group lost more total hip mineral density. Overall, although these changes were not related to differences in muscle strength [134], prudence should be exerted as in any manipulation of food intake in men or postmenopausal women with sarcopenia or osteoporosis.
Relative to the ADF approach, this was proven to be acceptable and effective (weight loss being 2.5% and fat mass 4%) in non-obese subjects who underwent fasting every other day for 22 days [135]. Most recently, following a similar approach, Stekovic et al. [136] investigated the direct effects of ADF during 4 weeks in a series of normal weight individuals, leading to an average 37% reduction of the daily calorie intake. Improved sense of well-being and improvement of cardiovascular risk markers, in particular triglycerides and LDL-cholesterol, with reduced trunk fat, fat-to-lean ratio and of the inflammatory marker soluble inter-cellular adhesion molecule-1 (sICAM) were detected. An increase of β-hydroxybutyrate occurred even in non-fasting days. Interestingly, the pro-aging amino acid methionine was periodically depleted.
Another study compared the effects of CR vs ADF enrolling 100 participants. These were randomised to adhere for 1 year (6-month intervention followed by 6-month weight maintenance) to one of the following regimens: (i) ADF (25% of energy needs on fasting days and 125% on alternating feast days), and (ii) 75% CR. Compared to the control group, no differences between interventions were found either in weight loss, −6.0% vs − 5.3%, respectively or in systolic and diastolic BPs, heart rate, triglycerides, fasting glucose, C-reactive protein. Overall, the Authors concluded that ADF is not superior to a CR approach and leads to a higher rate of dropouts, i.e. 38% (13/34) vs 29% (10/35), respectively [21]. It should be recalled that, as elsewhere reviewed [19], IF generally results in significant reductions in body weight and fat mass, the magnitude of weight losses being quite large (≥ 5.0 kg), except in trials less than 8 weeks in duration.
In obese individuals (BMI ≥ 30 kg/m2), Catenacci and colleagues compared ADF with CR during an 8 week-intervention [137]. Neither weight loss nor body composition, lipids and insulin sensitivity differed between groups. After 24 weeks of unsupervised follow-up aimed at assessing weight regain, there were no significant differences but changes in percent fat mass and lean mass were more favourable in the ADF group. Patients exercising more during the follow-up had a better weight maintenance [137], an often documented observation [138].
As above described, IF regimens have been shown to reduce global fat mass and visceral fat, both linked to a reduced risk of diabetes development [139] and thus of cardiovascular outcomes [140]. In overweight or obese patients with type 2 diabetes, 12 weeks of the 5:2 diet (400–600 Kcal/two days/week) did not differ from continuous energy restriction (1200–1400 kcal/day) in reducing glycated haemoglobin (−0.7%). The 5:2 group showed an increased tendency to hypoglycaemia despite medication reduction [141].
Application of ADF in obese subjects for 10 weeks reduced total cholesterol, LDL-cholesterol and triglycerides with a switch towards a rise of LDL size by 5% and a reduction of small LDL by 9% [142]. A similar lipid improvement was found in obese patients following a modified protocol of ADF with an overall 25% reduction of energy needs. Furthermore, a decrement in systolic BP as well as in C-reactive protein were also found [58,143]. In overweight women, daily energy restriction (600–650 kcal/days for 2 days/week) vs intermittent energy and carbohydrate restriction was superior in improving body fat reduction and insulin sensitivity [144].
Finally, in a very recent trial, enrolling 19 metabolic syndrome patients, application of TRE, i.e. a reduction of daily eating window from ≥ 14 h to a self-selected 10 h window over 12 weeks, led to healthier body composition, lowered BP, and decreased levels of cardiovascular risk associated lipids [145]. Moreover, TRE led to a modest, but not significant increase in sleep duration with no impact on sleep efficiency and a trend towards a reduction in physical activity.
Conclusions
While clinical studies on fasting have generally shown positive effects on health and possibly on life span, documenting benefits and challenges of long-term fasting still needs further studies. The question whether repeated cycles of fasting, as it happens spontaneously in animals, can enhance benefits, is still awaiting a definite answer. After having been rated, in the 1960s, as a successful strategy to treat obesity and comorbidities, additional benefits of fasting other than weight loss have been uncovered. Among others are improvements in glucose regulation, BP and heart rate, as well as abdominal fat loss. The key point in the clinical approach of fasting will be dependent upon acceptance and compliance, as well as on safety. These issues are linked to the emotional and physical well-being, the absence of hunger and the presence of professional guidance. TRE and IF can well fit in everyday life and may be possibly adopted as a lifelong eating behaviour. Long-term fasting, in fact, requires specialised settings, possibly away from the usual environment.
An emerging aspect, in the planning of the fasting strategy, is the after-fast period, when food is reintroduced. When the fasting process is reversed, symptom relapses can occur in the absence of specific nutritional guidelines. This may partially reduce the numerous health benefits of fasting. The individual limits of this procedure need to be carefully analysed and, similar to any manipulation of food intake, prudence is necessary, particularly in older subjects and in the case of low BMI, sarcopenia or eating disorders.
Disclosure statement
M.R. received honoraria from Buchinger Wilhelmi Clinic (BWC) for a scientific presentation (January 2020). F.W.T. and F.G. are employees of BWC. C.R.S. has no financial interests.
References
- 1.Liao C-Y, Rikke BA, Johnson TE, et al. Fat maintenance is a predictor of the murine lifespan response to dietary restriction. Aging Cell. 2011;10(4):629–639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mitchell SJ, Madrigal-Matute J, Scheibye-Knudsen M, et al. Effects of sex, strain, and energy intake on hallmarks of aging in mice. Cell Metab. 2016;23(6):1093–1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stirling I, Derocher AE. Effects of climate warming on polar bears: a review of the evidence. Glob Chang Biol. 2012;18(9):2694–2706. [DOI] [PubMed] [Google Scholar]
- 4.Hindle AG, Grabek KR, Epperson LE, et al. Metabolic changes associated with the long winter fast dominate the liver proteome in 13-lined ground squirrels. Physiol Genomics. 2014;46(10):348–361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Storey KB. Out cold: biochemical regulation of mammalian hibernation - a mini-review. Gerontology. 2010;56(2):220–230. [DOI] [PubMed] [Google Scholar]
- 6.Le Maho Y, Delclitte P, Chatonnet J. Thermoregulation in fasting emperor penguins under natural conditions. Am J Physiol. 1976;231(3):913–922. [DOI] [PubMed] [Google Scholar]
- 7.Groscolas R, Robin JP. Long-term fasting and re-feeding in penguins. Comp Biochem Physiol Part A Mol Integr Physiol. 2001;128(3):645–655. [DOI] [PubMed] [Google Scholar]
- 8.Lewden A, Enstipp MR, Bonnet B, et al. Thermal strategies of king penguins during prolonged fasting in water. J Exp Biol. 2017;220(Pt 24):4600–4611. [DOI] [PubMed] [Google Scholar]
- 9.Vaillancourt E, Weber JM. Fuel metabolism in Canada geese: effects of glucagon on glucose kinetics. Am J Physiol Regul Integr Comp Physiol. 2015;309(5):R535–R543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lettieri Barbato D, Aquilano K. Feast and famine: adipose tissue adaptations for healthy aging. Ageing Res Rev. 2016;28:85–93. [DOI] [PubMed] [Google Scholar]
- 11.Rigaud D, Hassid J, Meulemans A, et al. A paradoxical increase in resting energy expenditure in malnourished patients near death: the king penguin syndrome. Am J Clin Nutr. 2000;72(2):355–360. [DOI] [PubMed] [Google Scholar]
- 12.Westmoreland P, Krantz MJ, Mehler PS. Medical complications of anorexia nervosa and bulimia. Am J Med. 2016;129(1):30–37. [DOI] [PubMed] [Google Scholar]
- 13.Trepanowski JF, Bloomer RJ. The impact of religious fasting on human health. Nutr J. 2010;9:57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Persynaki A, Karras S, Pichard C. Unraveling the metabolic health benefits of fasting related to religious beliefs: a narrative review. Nutrition. 2017;35:14–20. [DOI] [PubMed] [Google Scholar]
- 15.Wilhelmi de Toledo F, Buchinger A, Burggrabe H, et al. Fasting therapy - an expert panel update of the 2002 consensus guidelines. Forsch Komplementmed. 2013;20(6):434–443. [DOI] [PubMed] [Google Scholar]
- 16.Benedict FG. Chemical and physiological studies of a man fasting thirtyone days. Proc Natl Acad Sci USA. 1915;1(4):228–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stewart WK, Fleming LW, Robertson PC. Massive obesity treated by intermittent fasting. A metabolic and clinical study. Am J Med. 1966;40(6):967–986. [DOI] [PubMed] [Google Scholar]
- 18.Lignot J-H, Le Maho Y. A history of modern research into fasting, starvation, and inanition. In: McCue M, editor. Comparative physiology of fasting, starvation, and food limitation. Berlin: Springer-Verlag; 2012. [Google Scholar]
- 19.Anton SD, Moehl K, Donahoo WT, et al. Flipping the metabolic switch: understanding and applying the health benefits of fasting. Obesity (Silver Spring). 2018;26(2):254–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cuendet GS, Loten EG, Cameron DP, et al. Hormone-substrate responses to total fasting in lean and obese mice. Am J Physiol. 1975;228(1):276–283. [DOI] [PubMed] [Google Scholar]
- 21.Trepanowski JF, Kroeger CM, Barnosky A, et al. Effect of alternate-day fasting on weight loss, weight maintenance, and cardioprotection among metabolically healthy obese adults: a randomized clinical trial. JAMA Intern Med. 2017;177(7):930–938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chaix A, Manoogian ENC, Melkani GC, et al. Time-restricted eating to prevent and manage chronic metabolic diseases. Annu Rev Nutr. 2019;39:291–315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Patterson RE, Laughlin GA, LaCroix AZ, et al. Intermittent fasting and human metabolic health. J Acad Nutr Diet. 2015;115(8):1203–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wilhelmi de Toledo F, Grundler F, Bergouignan A, et al. Safety, health improvement and well-being during a 4 to 21-day fasting period in an observational study including 1422 subjects. PLoS One. 2019;14(1):e0209353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.de Cabo R, Mattson MP. Effects of intermittent fasting on health, aging, and disease. N Engl J Med. 2019;381(26):2541–2551. [DOI] [PubMed] [Google Scholar]
- 26.Chazarin B, Storey KB, Ziemianin A, et al. Metabolic reprogramming involving glycolysis in the hibernating brown bear skeletal muscle. Front Zool. 2019;16:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Drenick EJ, Swendseid ME, Blahd WH, et al. prolonged starvation as treatment for severe obesity. JAMA. 1964;187:100–105. [DOI] [PubMed] [Google Scholar]
- 28.Jo E, Worts PR, Elam ML, et al. Resistance training during a 12-week protein supplemented VLCD treatment enhances weight-loss outcomes in obese patients. Clin Nutr. 2019;38(1):372–382. [DOI] [PubMed] [Google Scholar]
- 29.Brandhorst S, Choi IY, Wei M, et al. A periodic diet that mimics fasting promotes multi-system regeneration, enhanced cognitive performance, and healthspan. Cell Metab. 2015;22(1):86–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cheng C-W, Villani V, Buono R, et al. Fasting-mimicking diet promotes Ngn3-driven β-cell regeneration to reverse diabetes. Cell. 2017;168(5):775–788 e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Longchamp A, Mirabella T, Arduini A, et al. Amino acid restriction triggers angiogenesis via GCN2/ATF4 regulation of VEGF and H2S production. Cell. 2018;173(1):117–129 e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jaspers RT, Zillikens MC, Friesema ECH, et al. Exercise, fasting, and mimetics: toward beneficial combinations? FASEB J. 2017;31(1):14–28. [DOI] [PubMed] [Google Scholar]
- 33.Imai SI, Guarente L. It takes two to tango: NAD(+) and sirtuins in aging/longevity control. NPJ Aging Mech Dis. 2016;2:16017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lee HC. Physiological functions of cyclic ADP-ribose and NAADP as calcium messengers. Annu Rev Pharmacol Toxicol. 2001;41:317–345. [DOI] [PubMed] [Google Scholar]
- 35.Galman C, Lundåsen T ,Kharitonenkov A, et al. The circulating metabolic regulator FGF21 is induced by prolonged fasting and PPARalpha activation in man. Cell Metab. 2008;8(2):169–174. [DOI] [PubMed] [Google Scholar]
- 36.Osataphan S, Macchi C, Singhal G, et al. SGLT2 inhibition reprograms systemic metabolism via FGF21-dependent and -independent mechanisms. JCI Insight. 2019;4(5):e123130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mattson MP, Moehl K, Ghena N, et al. Intermittent metabolic switching, neuroplasticity and brain health. Nat Rev Neurosci. 2018;19(2):63–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Barry D, Ellul S, Watters L, et al. The ketogenic diet in disease and development. Int J Dev Neurosci. 2018;68:53–58. [DOI] [PubMed] [Google Scholar]
- 39.Sies H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol. 2017;11:613–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mesnage R, Grundler F, Schwiertz A, et al. Changes in human gut microbiota composition are linked to the energy metabolic switch during 10 d of Buchinger fasting. J Nutr Sci. 2019;8:e36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wechsler JG, Wenzel H, Swobodnik W, et al. Nitrogen balance studies during modified fasting. Postgrad Med J. 1984;60 (Suppl 3):66–73. [PubMed] [Google Scholar]
- 42.Varady KA. Intermittent versus daily calorie restriction: which diet regimen is more effective for weight loss? Obes Rev. 2011;12(7):e593–e601. [DOI] [PubMed] [Google Scholar]
- 43.Tinsley GM, La Bounty PM. Effects of intermittent fasting on body composition and clinical health markers in humans. Nutr Rev. 2015;73(10):661–674. [DOI] [PubMed] [Google Scholar]
- 44.Kirkpatrick CF, Bolick JP, Kris-Etherton PM, et al. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: a scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J Clin Lipidol. 2019;13(5):689–711 e1. [DOI] [PubMed] [Google Scholar]
- 45.D’Andrea Meira I, Romão TT, Pires do Prado HJ, et al. Ketogenic diet and epilepsy: what we know so far. Front Neurosci. 2019;13:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cahill GF. Jr. Fuel metabolism in starvation. Annu Rev Nutr. 2006;26:1–22. [DOI] [PubMed] [Google Scholar]
- 47.Leiter LA, Marliss EB. Survival during fasting may depend on fat as well as protein stores. JAMA. 1982;248(18):2306–2307. [PubMed] [Google Scholar]
- 48.Marliss EB, Murray FT, Nakhooda AF. The metabolic response to hypocaloric protein diets in obese man. J Clin Invest. 1978;62(2):468–479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Johnstone AM, Horgan GW, Murison SD, et al. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am J Clin Nutr. 2008;87(1):44–55. [DOI] [PubMed] [Google Scholar]
- 50.Marliss EB, Aoki TT, Unger RH, et al. Glucagon levels and metabolic effects in fasting man. J Clin Invest. 1970;49(12):2256–2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sakharova AA, Horowitz JF, Surya S, et al. Role of growth hormone in regulating lipolysis, proteolysis, and hepatic glucose production during fasting. J Clin Endocrinol Metab. 2008;93(7):2755–2759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Anson RM, Guo Z, de Cabo R, et al. Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake. Proc Natl Acad Sci USA. 2003;100(10):6216–6220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li L, Wang Z, Zuo Z. Chronic intermittent fasting improves cognitive functions and brain structures in mice. PLoS One. 2013;8(6):e66069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rahmani J, Kord Varkaneh H, Clark C, et al. The influence of fasting and energy restricting diets on IGF-1 levels in humans: a systematic review and meta-analysis. Ageing Res Rev. 2019;53:100910. [DOI] [PubMed] [Google Scholar]
- 55.Buono R, Longo VD. Starvation, stress resistance, and cancer. Trends Endocrinol Metab. 2018;29(4):271–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Redman LM, Huffman KM, Landerman LR, et al. Effect of caloric restriction with and without exercise on metabolic intermediates in nonobese men and women. J Clin Endocrinol Metab. 2011;96(2):E312–E321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Walford RL, Mock D, Verdery R, et al. Calorie restriction in biosphere 2: alterations in physiologic, hematologic, hormonal, and biochemical parameters in humans restricted for a 2-year period. J Gerontol A Biol Sci Med Sci. 2002;57(6):B211–B224. [DOI] [PubMed] [Google Scholar]
- 58.Varady KA, Bhutani S, Klempel MC, et al. Alternate day fasting for weight loss in normal weight and overweight subjects: a randomized controlled trial. Nutr J. 2013;12(1):146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Muller H, de Toledo FW, Resch KL. Fasting followed by vegetarian diet in patients with rheumatoid arthritis: a systematic review. Scand J Rheumatol. 2001;30(1):1–10. [DOI] [PubMed] [Google Scholar]
- 60.Hatori M, Vollmers C, Zarrinpar A, et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012;15(6):848–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Menzies FM, Fleming A, Caricasole A, et al. Autophagy and neurodegeneration: pathogenic mechanisms and therapeutic opportunities. Neuron. 2017;93(5):1015–1034. [DOI] [PubMed] [Google Scholar]
- 62.Mattson MP, Arumugam TV. Hallmarks of brain aging: adaptive and pathological modification by metabolic states. Cell Metab. 2018;27(6):1176–1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tornese P, Sala N, Bonini D, et al. Chronic mild stress induces anhedonic behavior and changes in glutamate release, BDNF trafficking and dendrite morphology only in stress vulnerable rats. The rapid restorative action of ketamine. Neurobiol Stress. 2019;10:100160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fontana L, Partridge L, Longo VD. Extending healthy life span-from yeast to humans. Science. 2010;328(5976):321–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Di Francesco A, Di Germanio C, Bernier M, et al. A time to fast. Science. 2018;362(6416):770–775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bujak AL, Crane JD, Lally JS, et al. AMPK activation of muscle autophagy prevents fasting-induced hypoglycemia and myopathy during aging. Cell Metab. 2015;21(6):883–890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Rodgers JT, Puigserver P. Fasting-dependent glucose and lipid metabolic response through hepatic sirtuin 1. Proc Natl Acad Sci USA. 2007;104(31):12861–12866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rodgers JT, Lerin C, Haas W, et al. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature. 2005;434(7029):113–118. [DOI] [PubMed] [Google Scholar]
- 69.Canto C, et al. Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab. 2010;11(3):213–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Walker AK, Yang F, Jiang K, et al. Conserved role of SIRT1 orthologs in fasting-dependent inhibition of the lipid/cholesterol regulator SREBP. Genes Dev. 2010;24(13):1403–1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Liu Y, Dentin R, Chen D, et al. A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange. Nature. 2008;456(7219):269–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Shimazu T, Hirschey MD, Hua L, et al. SIRT3 deacetylates mitochondrial 3-hydroxy-3-methylglutaryl CoA synthase 2 and regulates ketone body production. Cell Metab. 2010;12(6):654–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fernandes G, Yunis EJ, Good RA. Suppression of adenocarcinoma by the immunological consequences of calorie restriction. Nature. 1976;263(5577):504–507. [DOI] [PubMed] [Google Scholar]
- 74.Mitchell SJ, Bernier M, Mattison JA, et al. Daily fasting improves health and survival in male mice independent of diet composition and calories. Cell Metab. 2019;29(1):221–228 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ingram DK, Weindruch R, Spangler EL, et al. Dietary restriction benefits learning and motor performance of aged mice. J Gerontol. 1987;42(1):78–81. [DOI] [PubMed] [Google Scholar]
- 76.Stranahan AM, Lee K, Martin B, et al. Voluntary exercise and caloric restriction enhance hippocampal dendritic spine density and BDNF levels in diabetic mice. Hippocampus. 2009;19(10):951–961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Marosi K, Kim SW, Moehl K, et al. 3-Hydroxybutyrate regulates energy metabolism and induces BDNF expression in cerebral cortical neurons. J Neurochem. 2016;139(5):769–781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Parikh I, Guo J, Chuang K-H, et al. Caloric restriction preserves memory and reduces anxiety of aging mice with early enhancement of neurovascular functions. Aging (Albany NY). 2016;8(11):2814–2826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wan R, Camandola S, Mattson MP. Intermittent food deprivation improves cardiovascular and neuroendocrine responses to stress in rats. J Nutr. 2003;133(6):1921–1929. [DOI] [PubMed] [Google Scholar]
- 80.Kinnick TR, Youngblood EB, O'Keefe MP, et al. Modulation of insulin resistance and hypertension by voluntary exercise training in the TG(mREN2)27 rat. J Appl Physiol (1985). 2002;93(2):805–812. discussion 797. [DOI] [PubMed] [Google Scholar]
- 81.Mager DE, Wan R, Brown M, et al. Caloric restriction and intermittent fasting alter spectral measures of heart rate and blood pressure variability in rats. FASEB J. 2006;20(6):631–637. [DOI] [PubMed] [Google Scholar]
- 82.Sherman H, Genzer Y, Cohen R, et al. Timed high-fat diet resets circadian metabolism and prevents obesity. FASEB J. 2012;26(8):3493–3502. [DOI] [PubMed] [Google Scholar]
- 83.Chaix A, Lin T, Le HD, et al. Time-restricted feeding prevents obesity and metabolic syndrome in mice lacking a circadian clock. Cell Metab. 2019;29(2):303–319 e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Arumugam TV, Phillips TM, Cheng A, et al. Age and energy intake interact to modify cell stress pathways and stroke outcome. Ann Neurol. 2010;67(1):41–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Fusco S, Ripoli C, Podda MV, et al. A role for neuronal cAMP responsive-element binding (CREB)-1 in brain responses to calorie restriction. Proc Natl Acad Sci USA. 2012;109(2):621–626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Manzanero S, Erion JR, Santro T, et al. Intermittent fasting attenuates increases in neurogenesis after ischemia and reperfusion and improves recovery. J Cereb Blood Flow Metab. 2014;34(5):897–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Singh R, Lakhanpal D, Kumar S, et al. Late-onset intermittent fasting dietary restriction as a potential intervention to retard age-associated brain function impairments in male rats. Age (Dordr). 2012;34(4):917–933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mattson MP, Wan R. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. J Nutr Biochem. 2005;16(3):129–137. [DOI] [PubMed] [Google Scholar]
- 89.Kjeldsen-Kragh J, Haugen M, Borchgrevink CF, et al. Controlled trial of fasting and one-year vegetarian diet in rheumatoid arthritis. Lancet. 1991;338(8772):899–902. [DOI] [PubMed] [Google Scholar]
- 90.Bloom WL. Fasting as an introduction to the treatment of obesity. Metab Clin Exp. 1959;8(3):214–220. [PubMed] [Google Scholar]
- 91.Frommel D, Gautier M, Questiaux E, et al. Voluntary total fasting: a challenge for the medical community. Lancet. 1984;1(8392):1451–1452. [DOI] [PubMed] [Google Scholar]
- 92.Lithell H, Bruce A, Gustafsson IB, et al. A fasting and vegetarian diet treatment trial on chronic inflammatory disorders. Acta Derm Venereol. 1983;63(5):397–403. [PubMed] [Google Scholar]
- 93.Schmidt S, Stange R, Lischka E, et al. [Uncontrolled clinical study of the efficacy of ambulant fasting in patients with osteoarthritis]. Forsch Komplementmed. 2010;17(2):87–94. [DOI] [PubMed] [Google Scholar]
- 94.Fraser DA, Thoen J, Rustan AC, et al. Changes in plasma free fatty acid concentrations in rheumatoid arthritis patients during fasting and their effects upon T-lymphocyte proliferation. Rheumatology (Oxford), 1999;38(10):948–952. [DOI] [PubMed] [Google Scholar]
- 95.Goldhamer AC, Lisle DJ, Sultana P, et al. Medically supervised water-only fasting in the treatment of borderline hypertension. J Altern Complement Med. 2002;8(5):643–650. [DOI] [PubMed] [Google Scholar]
- 96.Kanazawa M, Fukudo S. Effects of fasting therapy on irritable bowel syndrome. Int J Behav Med. 2006;13(3):214–220. [DOI] [PubMed] [Google Scholar]
- 97.Stange R, Pflugbeil C, Michalsen A, et al. Therapeutic fasting in patients with metabolic syndrome and impaired insulin resistance. Forsch Komplementmed. 2013;20(6):421–426. [DOI] [PubMed] [Google Scholar]
- 98.Li C, Ostermann T, Hardt M, et al. Metabolic and psychological response to 7-day fasting in obese patients with and without metabolic syndrome. Forsch Komplementmed. 2013;20(6):413–420. [DOI] [PubMed] [Google Scholar]
- 99.Michalsen A, Li C. Fasting therapy for treating and preventing disease - current state of evidence. Forsch Komplementmed. 2013;20(6):444–453. [DOI] [PubMed] [Google Scholar]
- 100.Bauersfeld SP, Kessler CS, Wischnewsky M, et al. The effects of short-term fasting on quality of life and tolerance to chemotherapy in patients with breast and ovarian cancer: a randomized cross-over pilot study. BMC Cancer. 2018;18(1):476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Drinda S, Franke S, Schmidt S, et al. AGE-RAGE interaction does not explain the clinical improvements after therapeutic fasting in osteoarthritis. Complement Med Res. 2018;25(3):167–172. [DOI] [PubMed] [Google Scholar]
- 102.Wilhelmi de Toledo F. Eat, fast, and live longer! Forsch Komplementmed. 2013;20(6):462–464. [DOI] [PubMed] [Google Scholar]
- 103.Drinda S, Grundler F, Neumann T, et al. Effects of periodic fasting on fatty liver index-a prospective observational study. Nutrients. 2019;11(11):2601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Viallon M, et al. Chemical-shift-encoded magnetic resonance imaging and spectroscopy to reveal immediate and long-term multi-organs composition changes of a 14-days periodic fasting intervention: a technological and case report. Front Nutr. 2019;6:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thomson TJ, Runcie J, Miller V. Treatment of obesity by total fasting for up to 249 days. Lancet. 1966;2(7471):992–996. [DOI] [PubMed] [Google Scholar]
- 106.Stewart WK, Fleming LW. Features of a successful therapeutic fast of 382 days’ duration. Postgrad Med J. 1973;49(569):203–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Cahill GJ, Jr., Owen OE, Morgan AP. The consumption of fuels during prolonged starvation. Adv Enzyme Regul. 1968;6:143–150. [DOI] [PubMed] [Google Scholar]
- 108.Cahill GF, Jr., Marliss EB, Aoki TT. Fat and nitrogen metabolism in fasting man. Horm Metab Res. 1970;2(Suppl 2):181–185. [PubMed] [Google Scholar]
- 109.Fisler JS, Drenick EJ, Blumfield DE, et al. Nitrogen economy during very low calorie reducing diets: quality and quantity of dietary protein. Am J Clin Nutr. 1982;35(3):471–486. [DOI] [PubMed] [Google Scholar]
- 110.Cahill GF, Jr., Owen OE. Starvation and survival. Trans Am Clin Climatol Assoc. 1968;79:13–20. [PMC free article] [PubMed] [Google Scholar]
- 111.Longo VD, Mattson MP. Fasting: molecular mechanisms and clinical applications. Cell Metab. 2014;19(2):181–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Owen OE, Felig P, Morgan AP, et al. Liver and kidney metabolism during prolonged starvation. J Clin Invest. 1969;48(3):574–583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Goodman MN, Larsen PR, Kaplan MM, et al. Starvation in the rat. II. Effect of age and obesity on protein sparing and fuel metabolism. Am J Physiol. 1980;239(4):E277–E286. [DOI] [PubMed] [Google Scholar]
- 114.Bertile F, Fouillen L, Wasselin T, et al. The safety limits of an extended fast: lessons from a non-model organism. Sci Rep. 2016;6:39008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Koubi HE, Robin JP, Dewasmes G, et al. Fasting-induced rise in locomotor activity in rats coincides with increased protein utilization. Physiol Behav. 1991;50(2):337–343. [DOI] [PubMed] [Google Scholar]
- 116.Ditschuneit H, Ditschuneit HH, Wechsler J. Management of obesity–zero diet or calorie-reduced diet? Zero diet. Internist (Berl). 1979;20(3):151–158. [PubMed] [Google Scholar]
- 117.Garnett ES, Barnard DL, Ford J, et al. Gross fragmentation of cardiac myofibrils after therapeutic starvation for obesity. Lancet. 1969;1(7601):914–916. [DOI] [PubMed] [Google Scholar]
- 118.Frank A, Graham C, Frank S. Fatalities on the liquid-protein diet: an analysis of possible causes. Int J Obes. 1981;5(3):243–248. [PubMed] [Google Scholar]
- 119.Lantigua RA, Amatruda JM, Biddle TL, et al. Cardiac arrhythmias associated with a liquid protein diet for the treatment of obesity. N Engl J Med. 1980;303(13):735–738. [DOI] [PubMed] [Google Scholar]
- 120.Van Itallie TB, Yang MU. Cardiac dysfunction in obese dieters: a potentially lethal complication of rapid, massive weight loss. Am J Clin Nutr. 1984;39(5):695–702. [DOI] [PubMed] [Google Scholar]
- 121.Goldhamer A, Lisle D, Parpia B, et al. Medically supervised water-only fasting in the treatment of hypertension. J Manipulative Physiol Ther. 2001;24(5):335–339. [DOI] [PubMed] [Google Scholar]
- 122.Busnelli M, Manzini S, Chiesa G. The gut microbiota affects host pathophysiology as an endocrine organ: a focus on cardiovascular disease. Nutrients. 2019;12(1):79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Riedl RA, Atkinson SN, Burnett CML, et al. The gut microbiome, energy homeostasis, and implications for hypertension. Curr Hypertens Rep. 2017;19(4):27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ott B, Skurk T, Hastreiter L, et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci Rep. 2017;7(1):11955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jangi S, Gandhi R, Cox LM, et al. Alterations of the human gut microbiome in multiple sclerosis. Nat Commun. 2016;7:12015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Piccio L, Stark JL, Cross AH. Chronic calorie restriction attenuates experimental autoimmune encephalomyelitis. J Leukoc Biol. 2008;84(4):940–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Cignarella F, Cantoni C, Ghezzi L, et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. 2018;27(6):1222–1235 e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Rochon J, Bales CW, Ravussin E, et al. Design and conduct of the CALERIE study: comprehensive assessment of the long-term effects of reducing intake of energy. J Gerontol A Biol Sci Med Sci. 2011;66A(1):97–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Stewart TM, Bhapkar M, Das S, et al. Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy Phase 2 (CALERIE Phase 2) screening and recruitment: methods and results. Contemp Clin Trials. 2013;34(1):10–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Martin CK, Bhapkar M, Pittas AG, et al. Effect of calorie restriction on mood, quality of life, sleep, and sexual function in healthy nonobese adults: the CALERIE 2 randomized clinical trial. JAMA Intern Med. 2016;176(6):743–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Das SK, Roberts SB, Bhapkar MV, et al. Body-composition changes in the Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE)-2 study: a 2-y randomized controlled trial of calorie restriction in nonobese humans. Am J Clin Nutr. 2017;105(4):913–927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sparks LM, et al. Effects of 12 months of caloric restriction on muscle mitochondrial function in healthy individuals. J Clin Endocrinol Metab. 2017;102(1):111–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Kraus WE, Bhapkar M, Huffman KM, et al. 2 years of calorie restriction and cardiometabolic risk (CALERIE): exploratory outcomes of a multicentre, phase 2, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(9):673–683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Seimon RV, Wild-Taylor AL, Keating SE, et al. Effect of weight loss via severe vs moderate energy restriction on lean mass and body composition among postmenopausal women with obesity: the TEMPO diet randomized clinical trial. JAMA Netw Open. 2019;2(10):e1913733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Heilbronn LK, Smith SR, Martin CK, et al. Alternate-day fasting in nonobese subjects: effects on body weight, body composition, and energy metabolism. Am J Clin Nutr. 2005;81(1):69–73. [DOI] [PubMed] [Google Scholar]
- 136.Stekovic S, Hofer SJ, Tripolt N, et al. Alternate day fasting improves physiological and molecular markers of aging in healthy, non-obese humans. Cell Metab. 2019;30(3):462–476 e5. [DOI] [PubMed] [Google Scholar]
- 137.Catenacci VA, Pan Z, Ostendorf D, et al. A randomized pilot study comparing zero-calorie alternate-day fasting to daily caloric restriction in adults with obesity. Obesity (Silver Spring). 2016;24(9):1874–1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Ostendorf DM, Lyden K, Pan Z, et al. Objectively measured physical activity and sedentary behavior in successful weight loss maintainers. Obesity (Silver Spring). 2018;26(1):53–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Neeland IJ, Turer AT, Ayers CR, et al. Dysfunctional adiposity and the risk of prediabetes and type 2 diabetes in obese adults. JAMA. 2012;308(11):1150–1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Cosentino F, Grant PJ, Aboyans V, et al. 2019 ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020;41:255–323. [DOI] [PubMed] [Google Scholar]
- 141.Corley BT, Carroll RW, Hall RM, et al. Intermittent fasting in type 2 diabetes mellitus and the risk of hypoglycaemia: a randomized controlled trial. Diabet Med. 2018;35(5):588–594. [DOI] [PubMed] [Google Scholar]
- 142.Klempel MC, Kroeger CM, Varady KA. Alternate day fasting increases LDL particle size independently of dietary fat content in obese humans. Eur J Clin Nutr. 2013;67(7):783–785. [DOI] [PubMed] [Google Scholar]
- 143.Varady KA, Bhutani S, Church EC, et al. Short-term modified alternate-day fasting: a novel dietary strategy for weight loss and cardioprotection in obese adults. Am J Clin Nutr. 2009;90(5):1138–1143. [DOI] [PubMed] [Google Scholar]
- 144.Harvie M, Wright C, Pegington M, et al. The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. Br J Nutr. 2013;110(8):1534–1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wilkinson MJ, Manoogian ENC, Zadourian A, et al. Ten-hour time-restricted eating reduces weight, blood pressure, and atherogenic lipids in patients with metabolic syndrome. Cell Metab. 2020;31(1):92–104 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]