Abstract
Studies have pointed out that air pollution may be a contributing factor to the coronavirus disease 2019 (COVID-19) pandemic. However, the specific links between air pollution and severe acute respiratory syndrome-coronavirus-2 infection remain unclear. Here we provide evidence from in vitro, animal and human studies from the existing literature. Epidemiological investigations have related various air pollutants to COVID-19 morbidity and mortality at the population level, however, those studies suffer from several limitations. Air pollution may be linked to an increase in COVID-19 severity and lethality through its impact on chronic diseases, such as cardiopulmonary diseases and diabetes. Experimental studies have shown that exposure to air pollution leads to a decreased immune response, thus facilitating viral penetration and replication. Viruses may persist in air through complex interactions with particles and gases depending on: 1) chemical composition; 2) electric charges of particles; and 3) meteorological conditions such as relative humidity, ultraviolet (UV) radiation and temperature. In addition, by reducing UV radiation, air pollutants may promote viral persistence in air and reduce vitamin D synthesis. Further epidemiological studies are needed to better estimate the impact of air pollution on COVID-19. In vitro and in vivo studies are also strongly needed, in particular to more precisely explore the particle–virus interaction in air.
Short abstract
Our review highlights that both short- and long-term exposures to air pollution may be important aggravating factors for SARS-CoV-2 transmission and COVID-19 severity and lethality through multiple mechanisms https://bit.ly/395aS9w
Introduction
In late 2019, the city of Wuhan, China became the epicenter of a pneumonia epidemic due to a new coronavirus, severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2), which causes a disease given the name coronavirus disease 2019 (COVID-19) in February 2020 by the World Health Organization [1]. COVID-19 infections are not specific, ranging from completely asymptomatic to severe forms which might require mechanical ventilation and/or lead to death. Although older age and comorbidities have consistently been reported as risk factors for unfavourable COVID-19 prognosis, younger patients without known risk factors are also being admitted to intensive care, albeit to a lesser extent and with a different range of symptoms. Reported comorbidities include hypertension, obesity, diabetes, cardiovascular disease, COPD, chronic kidney disease and malignancy. Symptomatology, when present, is quite variable reflecting systemic involvement, including symptoms of lower respiratory tract infection (e.g. cough, fever and dyspnoea), neurological impairment, cutaneous manifestations, or gastro-intestinal issues especially in the elderly [2]. Multiple organ failure and cardiopulmonary complications, such as myopericarditis, pulmonary embolism and acute respiratory distress syndrome, represent some of the major complications of severe COVID-19. Some of the severe forms and deaths attributed to SARS-CoV-2 infection are suspected to arise from a disproportionate inflammatory response that leads to a major release of pro-inflammatory cytokines, also referred to as a “cytokine storm” [3].
Emerging epidemiological and experimental data are now suggesting the involvement of air pollution in COVID-19-related outcomes. As suggested by the existing literature on multiple respiratory viruses, air pollution may play a role in COVID-19-related morbidity and mortality according to specific mechanisms.
Air pollution is a complex mixture of gaseous and particulate constituents that vary both spatially and temporally. COVID-19-related epidemiological studies have so far investigated impacts based on particulate matter (PM) of two sizes, e.g. inhalable particles (particles with a 50% cut-off aerodynamic diameter of 10 µm (PM10)) and fine particles (particles with a 50% cut-off aerodynamic diameter of 2.5 µm (PM2.5)). It is also worthwhile mentioning here that both PM10 and PM2.5 are a proxy for ultrafine particles (diameter <0.1 µm). The size is important in determining the PM transportability and final destination within the respiratory tract or bloodstream. Depending on its source, PM can have different compositions. Carbonaceous PM from coal, fuel or wood combustion is the most harmful and is responsible for many chronic diseases, including cardiopulmonary and metabolic diseases, neurodegenerative conditions, cancer and low birthweight [4, 5]. The gaseous pollutants investigated so far at the population level in relation to COVID-19 and other respiratory viruses include nitrogen oxides (NOx) and ozone (O3). NO2 is a major air pollutant in urban environments, primarily arising from traffic, in particular from diesel cars, and has been associated with asthma, COPD, bronchiolitis and cardiovascular diseases [6]. Tropospheric O3 levels are dependent on other emitted pollutants, including NOx and VOCs, as well as on climate parameters, and have been associated with excess cardiorespiratory mortality and morbidity [7].
In this paper, we provide a comprehensive overview and synthesis of key data, bringing in evidence that sheds light on the question of whether or not air pollution and COVID-19 are related.
Methods
We conducted a review of the published literature on the relationship between ambient air pollution and viral respiratory infections with particular emphasis on SARS-CoV-2 using evidence from in vitro, animal and human studies. Literature was gathered using available online academic databases such as Pubmed, Science Direct and Google Scholar, with no restrictions on publication dates. We focused our review on outdoor air pollution (including the air pollutants PM, NO2 and O3), using MeSH and keywords such as “particulate matter and SARS-CoV-2/COVID-19”, “air pollution and SARS-CoV-2/COVID-19”, “air pollution and viral respiratory disease”, “air pollution and SARS-CoV-1 ”, “particulate matter and viral respiratory diseases”, “ozone and respiratory viruses”, “nitrogen dioxide and respiratory viruses”, “relative humidity, temperature and respiratory viral diseases”.
In reviewing the epidemiological studies, we considered the STROBE (Strengthening the Reporting of OBservational studies in Epidemiology) checklist of items that should be detailed in reports of observational studies, namely: study design, setting, study participants, variables (i.e. exposures, health outcomes and confounding variables), data sources, methods of assessment, statistical methods, main results, sensitivity analyses, limitations, and generalisability [8].
We also included published reviews exploring air pollution and respiratory virus and editorial letters referring to air pollution and COVID-19. However, because the COVID-19 pandemic disease is a current major health issue of urgent concern, we have also included preprint articles. Although environmental tobacco smoke exposure and indoor air pollution are important issues, in this review, we focused on the relationship between outdoor air pollution and SARS-CoV-2/COVID-19 in terms of morbidity-mortality, physicochemical interactions and atmospheric propagation.
First, we present the epidemiological literature on the relationship of outdoor air pollution exposure to respiratory viral infections with particular attention on: COVID-19 morbidity and mortality; the major diseases and organs targeted by both air pollutants and COVID-19; the potential biological mechanisms involved; and the interactions between viruses and air pollutants inside the human body. Secondly, we will review the mode of transport/transmission of SARS-CoV-2, the possible interactions between air pollutants and viruses in ambient air, and discuss whether air pollution plays a role in viral transmission.
Results
Does air pollution exposure increase COVID-19 morbidity and mortality? Evidence from population studies
Air pollution and viral respiratory infections
A century after the Spanish Influenza Pandemic, a study reported that cities that used more coal had tens of thousands of excess flu deaths compared to cities that used less coal, taking into account confounders such as socio-economic and baseline health status [9]. Long-term exposure to air pollution has been associated with increased influenza mortality in the USA [10]. In 2003, a Chinese study on SARS-CoV-1 showed that people living in the most polluted areas had a two-fold increased risk of dying from SARS compared to people living in less polluted areas [11]. In 2015, analysis conducted in 195 countries concluded that air pollution is a significant contributor to the burden of lower respiratory tract infections [12]. As demonstrated for influenza, exposure to air pollution increases the severity of viral respiratory infections [13]. In addition, several studies have demonstrated that increases in air pollutant concentrations were associated with an increased occurrence of respiratory viral diseases among children and adults, in particular when the viral infection was concomitant to a short-term increase in exposure to air pollution. Increases in PM2.5 concentrations have been associated with increased rates of viral infection, namely influenza, respiratory syncytial virus (RSV) and measles. In these studies, concentrations of PM2.5 were correlated with the number of new cases of respiratory viral infections with a lag time of a few days [14, 15].
Air pollution and COVID-19
Various studies have also reported a link between air pollution exposure and COVID-19 morbidity and mortality in humans (table 1). However, from an epidemiological point of view, several concerns arose from non-randomised studies that have investigated this relationship. First, the values for the number of confirmed COVID-19 cases, including the number of deaths, have been determined without standardised methodologies and tools, and without reliable testing in many cases during the first months of the COVID-19 pandemic, which may have led to an underestimation in the total number of COVID-19 outcomes. Secondly, the studies suffer from air pollution exposure misclassification. Available assessments come from either satellites or local monitoring stations without any detail on individual exposures. Aside from the study by Travaglio et al. [32] with individual level data from the UK Biobank, all epidemiological studies are ecological with aggregated assessments at the population levels (countries, counties and cities); thus, more longitudinal studies with individual level data are needed [36]. Ambient outdoor pollution concentrations can be a general indicator of air quality, but they are not a reliable substitute for the exposure experienced by a given individual. In this context, using exceedance levels established by public authorities is also limiting, although it can be considered as a proxy of chronic exposure to elevated levels of air pollution. Another weakness encountered arises from the fact that both air pollution and infections are autocorrelated both spatially and temporally, which has not been taken into account so far by the existing investigations. Studies lacking a control for spatio-temporal autocorrelations have been shown to bias the estimation of the results. In this context it has to be underlined that COVID-19 cases mainly arise from clusters, i.e. at work, that are not taken into account in epidemiological studies [36]. Furthermore, most analyses have not taken into account important confounders, i.e. factors related to both air pollution and COVID-19, such as population density [36].Viral spread may vary considerably across different areas depending on population density, the time of the virus introduction, and the time of infection control measures, such as physical distancing, mask requirements, test policies or stay-at-home directives. All these factors influence the dynamics of the disease that can be estimated through the daily number of new cases or through the calculation of the basic reproductive number (R0) and effective reproductive number, both depending on transmissibility, contact rates and duration of contagiousness, as well as on virus mutation time. As air pollution may also influence both transmissibility and duration of infectiousness, the analysis of the impact of air pollution on the dynamic of COVID-19 has to take into account confounders such as population density, the time of the virus introduction and the time of introduction of infection control measures [37].
TABLE 1.
Epidemiological evidence on air pollution exposure and COVID-19 events
| First author [ref.] | Study location | Period | Air pollutants exposure | COVID-19 outcome | Findings: quantified results | Comments |
| Short-term exposure | ||||||
| Setti [16] | Italy | 7 Feb to 15 March 2020 | Daily PM10, concentrations higher than the daily limit value (50 µg·m−3) according to the national monitoring system | Daily number of confirmed cases | Positive correlation between the number of cases in each province and the average number of exceedances of PM10 daily limit value (R2=0.98) | Only exceedance data and only one air pollutant (PM10) |
| Bontempi [17] | Italy (Piedmont, Lombardy, 12 cities) | 10 Feb to 27 March 2020 | Daily PM10 concentrations according to the national air quality monitoring system | Daily number of confirmed cases | No evidence of correlations between the presence of high quantities of PM10 and cases on the basis of visual graphs | Lack of statistical test of the correlation |
| Zoran [18] | Italy (Milan) | 1 Jan to 30 April 2020 | Daily average concentrations of O3, NO2 according to the national air quality monitoring system | Daily total number of confirmed cases, new positive cases and total deaths | Positive correlation of O3 (Pearson coefficient=0.64, 0.50, 0.69) but negative correlation of NO2 (Pearson coefficient=−0.55, −0.35, −0.58) with all outcomes | Taking into account humidity, temperature and lockdown (before and after) |
| Zoran [19] | Italy (Milan) | 1 Jan to 30 April 2020 | Daily average concentrations of PM2.5, PM10, daily maximum PM10 according to the national air quality monitoring system and AQI | Daily total number of confirmed cases, new positive cases and total deaths | Positive correlations between daily new cases and daily maximum PM10 (R2=0.51), daily average PM2.5 (R2=0.25) and daily AQI (R2=0.43) | Statistical significance not reported (no p-value) |
| Frontera [20] | Italy | 1 Feb to 31 March 2020 | PM2.5 mean concentration in February (data from Italian Civil Protection Agency) | Total number of cases and deaths | Positive correlation between PM2.5 concentration in February and total number of cases (Pearson coefficient=0.64, p<0.0074) and death numbers (Pearson coefficient=0.53, p<0.032) on 31 March 2020 | No quantification of the correlation and no adjustment for confounders such as population density |
| Conticini [21] | Italy | AQI based on concentration values for up to five key pollutants, including: PM10, PM2.5, O3, SO2 and NO2 | Death rate | Mortality rate in Lombardy and Emilia Romagna (highly polluted by NO2) higher than in the rest of Italy (12% versus 4.5%) | AQI as a proxy of exposure. No statistical tests |
|
| Fronza [22] | Europe (47 regional European capitals and 107 major Italian cities) | 10 Feb to 10 April 2020 | Hourly concentrations of PM2.5, PM10, O3 and NH3# | Daily confirmed cases per province and region | Positive correlation between number of cases per million and PM2.5, PM10 and NH3 (0.58≤ r ≤0.68) but negative correlation with O3, (r=−0.44). | Introduction of a binary classifier based on an artificial neural network to explain spatial differences |
| Li [23] | China (Wuhan and Xiao Gan) | 26 Jan to 29 Feb 2020 | AQI and four ambient air pollutants (PM2.5, PM10, NO2 and CO) according to the national air quality monitoring system | Daily number of new cases (incidence) | Incidence correlated with: AQI in both Wuhan (R2=0.13) and Xiao Gan (R2=0.223); PM2.5 and NO2 in both cities (R=0.329 for NO2 in Wuhan; R2=0.117 for PM2.5 in Xiao Gan); PM10 (R2=0.105) | Low values of R2 |
| Jiang [24] | China (Wuhan, Xiao Gan and Huang Gang) | 25 Jan to 29 Feb 2020 | Daily data of eight ambient air pollutants (PM2.5, PM10, SO2, CO, NO2, and 8-h O3) according to the national air quality monitoring system | Daily number of new cases (incidence) | Positive association (RR between PM2.5 1.036 (95% CI 1.032–1.039), 1.059 (1.046–1.072) and 1.144 (1.12–1.169)) and daily incidence in Wuhan, Xiao Gan and Huang Gang | Quantification of the risk |
| Yao [25] | China (63 cities) | 27 Jan to 26 Feb 2020 | Hourly NO2 data according to the national air quality monitoring system | Number of confirmed cases and basic reproduction number (R0) | Positive association of R0 with NO2 in all cities (Chi-squared=10.18, p=0.037) and with 12-day time lag in 11 cities (r>0.51, p<0.005) | Adjustment for temperature and humidity Other confounders that influence R0 (timing of viral introduction, timing of COVID-19 control policies, etc.) not taken into account Only Chi-squared for global test of association |
| Wang [26] | China (72 cities) | 20 Jan to 2 March 2020 | Daily concentrations of PM2.5 and PM10 according to the national air quality monitoring system | Daily confirmed cases | Short-term (lag 7 and 14 days) increase of 10 μg·m−3 in PM2.5 and PM10 associated with daily cases (RR 1.64 (95% CI 1.47–1.82) and 1.47 (1.34–1.61)) | Quantification of the risk controlled for ambient temperature, absolute humidity and migration scale index |
| Zhu [27] | China (120 cities) | 23 Jan to 29 Feb 2020 | Daily concentrations of PM2.5, PM10, SO2, CO, NO2 and O3 according to the national air quality monitoring system | Daily number of confirmed cases | Short-term increase 10-μg·m−3 (lag 0–14) in PM2.5, PM10, NO2 and O3 associated with a 2.24% (95% CI 1.02–3.46), 1.76% (0.89–2.63), 6.94% (2.38–11.51) and 4.76% (1.99–7.52) increase in the daily counts of confirmed cases | Models (GAMs) adjusted for temperature, humidity, wind speed, air pressure and time trend estimating the associations between the moving average (lag 0–7) concentrations of air pollutants Time series analysis allowing to take daily data and lags into account |
| Adhikari [28] | USA (Queens, NY) | 1 March to 20 April 2020 | Daily maximum 8-h O3, daily average PM2.5 according to the national air quality monitoring system | Number of confirmed cases and deaths | Positive association between O3 and cases (10.51% increase (95% CI 7.47–13.63) but negative relationship between PM2.5 and new cases (a one-unit increase in the moving average of PM2.5 associated with a 33.11% (95% CI 31.04–35.22) decrease in the daily new COVID- 19 cases) | Adjusted for meteorological factors, day trends and lagged outcome to account for the potential autocorrelation of the time series of new cases (deaths) |
| Long-term exposure | ||||||
| Fattorini [29] | Italy (regions) | 2010–2019 | Daily data on distribution of NO2, O3, PM2.5 and PM10 and days exceeding regulatory limits during the last 4 years, and during the last decade (2010–2019) with limits exceeded for at least 35 days according to the national air quality monitoring system | Daily number of confirmed cases | Positive correlations in up to 71 provinces between PM2.5, PM10, O3, and NO2 and cases (0.23 ≤R2 ≤0.34) | No adjustments for meteorological factors and population density |
| Wu [30] | USA (all inland counties) | Up to 4 April 2020 | County-level long-term average of PM2.5 between 2000 and 2016 from prediction models using national air quality monitoring system | COVID-19 death rate | A 1 μg·m−3 increase in PM2.5 associated with an 8% increase in the COVID-19 death rate (95% CI 2–15%) | Main analysis adjusted by 20 potential confounding factors including population density, household income, ethnic group and education, median house value, age, sex, BMI, smoking, temperature, relative humidity, number of individuals tested for COVID-19 Possible bias: county-level adjustment factors excluded institutionalised residents |
| Liang [31] | USA (3122 US counties) | 22 Jan 2020 to 29 April 2020 | Long-term (2010–2016) county-level exposures to NO2, PM2.5 and O3 according to the national air quality monitoring system | COVID-19 case-fatality rate and mortality rate | IQR (∼4.6 ppb) increase in NO2 associated with increase of 7.1% (95% CI 1.2–13.4%) and 11.2% (95% CI 3.4–19.5%) in COVID-19 case-fatality and mortality rates No significant association for PM2.5 and O3 |
Both single and multipollutant models and controlled for spatial trends and a comprehensive set of potential confounders including state-level test positive rate, county-level healthcare capacity, phase-of-epidemic, population mobility, sociodemographic, socioeconomic status, behaviour risk factors and meteorological factors |
| Travaglio [32] | UK Biobank data sources | 2018 to 2019 | Annual average of daily measurements for NO2, NO and O3 according to the national air quality monitoring system and higher resolution air pollution estimate (<2 km away from self-reported address) | Number of confirmed cases allowing to compute the infectivity rate and deaths | Association between SO2, PM2.5, PM10 and infectivity rate (OR 1.316 (95% CI 1.141–1.521), 1.120 (1.036–1.211) and 1.074 (1.017–1.136)) Smaller association for NO2 association between SO2, NO2, O3 and COVID-19 mortality (OR 1.172 (95% CI 1.005–1.369), 1.200 (1.026–1.414) and 8.503 (2.029–35.626)) |
Adjusted for population density and individual-level data from UK Biobank |
| Ogen [33] | Europe (66 administrative regions in four countries: Italy, France, Germany, Spain) | Jan to Feb 2020 | Tropospheric concentrations of NO2 (Sentinel-5P data) taking into account vertical airflow | Number of deaths collected from each country | Data from the Sentinel-5P showed two main NO2 hotspots over Europe: Northern Italy and Madrid metropolitan area, regions in which COVID-19 mortality has been particularly high | Long-term exposure defined as a 2-month period (Jan to Feb 2020) |
| Cole [34] | The Netherlands (355 municipalities) | Up to 5 June 2020 | Annual concentrations of PM2.5, NO2 or SO2, averaged over the period 2015–2019 | Number of cases, hospital admissions and deaths | A 1 μg·m−3 increase in PM2.5 concentrations associated with 9.4 more COVID-19 cases, 3.0 more hospital admissions and 2.3 more deaths | The relationship was observed in rural settings and persisted after controlling for a wide range of explanatory variables and a number of sensitivity and robustness exercises including instrumenting pollution to mitigate potential endogeneity and modelling spatial spill-overs using econometric techniques |
| Pozzer [35] | Worldwide | Up to June 2020 | Chronic exposure to PM2.5 in the years prior to the COVID-19 outbreak estimated on the basis of satellite observations over the year 2019 | Mortality rate ratios attributed to air pollution in the COVID-19 pandemic [34] and the SARS-CoV-1 epidemic [11] | PM2.5 contributes 15% (95% CI 7–33%) to COVID-19 mortality worldwide, 27% (95% CI 13–46%) in East Asia, 19% (95% CI 8–41%) in Europe and 17% (95% CI 6–39%) in North America | Relative risk (or hazard ratio) of excess COVID-19 mortality for USA and SARS-CoV-1 in China (assuming that SARS and COVID-19 mortality are similarly affected by long-term exposure to air pollution) from long-term exposure to air pollution using the exposure–response function of the WHO to estimate the attributable fraction |
COVID-19: coronavirus disease 2019; PMx: particles with a 50% cut-off aerodynamic diameter of x µm; AQI: Air Quality Index; SARS-CoV-1: severe acute respiratory syndrome-coronavirus-1; GAM: generalised additive model; R2: coefficient of determination defined as the proportion of the variance in the dependent variable (COVID-19 outcomes) that is predictable from the independent variable(s); IQR: interquartile range; BMI: body mass index; WHO: World Health Organization. #: according to the ENSEMBLE multi-model that combines the values of other seven models: CHIMERE METNorway, EMEP RIUUK, EURADIM KNMI/TNO, LOTOS-EUROS SMHI, MATCH FMI, SILAM Météo-France and MOCAGE UKMET (www.regional.atmosphere.copernicus.eu).
Among the studies addressing short-term effects of air pollution, Setti et al. [16] were the first to observe an association between the number of Italian provinces with daily averaged PM10 concentrations exceeding the European limit values of 50 µg·m−3 and the subsequent number of COVID-19. Significant associations were found between mean concentrations of PM2.5 during the month of February 2020 and the total number of Italian COVID-19 cases on 31 March 2020 [20]. However, a similar study focusing on the Lombardy and Piedmont regions in Northern Italy from 10 February to 27 March 2020 reported a null association between the number of COVID-19 cases and PM10 concentrations measured several days before the COVID emergency explosion [17]. This study is less robust than the others as it is based only on a visual approach without any statistical tests. A study exploring COVID-19 cases in Italy, France, Germany and Spain reported a positive association between PM2.5 and PM10 concentrations between February and April 2020 and the number of SARS-CoV-2 infected cases, while a negative association was found for O3 [33].
In China, city-specific effects of PM10 and PM2.5 on daily confirmed COVID-19 cases were examined in more than 70 cities. Short-term lagged (7 and 14 days) increases in PM2.5 were associated with daily COVID-19 confirmed cases, and the magnitude of effect was greater for PM2.5 compared to PM10 [26]. Another study in China looked at short-term levels of six different pollutants (PM2.5, PM10, SO2, CO, NO2 and O3) in 120 cities to determine their association with daily COVID-19 confirmed cases. The study reported an approximate 2% increase in daily COVID-19 cases for every 2-week lagged 10 μg·m−3 increase in PM2.5 and PM10 [27]. A pre-print study also reported a positive association between NO2 levels measured with a 12-day time-lag and the spread of COVID-19 (estimated with the basic reproduction number R0) in 63 Chinese cities, after adjustment of temperature and humidity [25].
Regarding the role of long-term exposure to air pollution, an Italian study reported that mean levels of NO2, O3, PM2.5 and PM10 during the past 4 years, as well as the number of days exceeding the limit values established by the European Commission during the past 3 years, were both correlated with the number of COVID-19 cases in Italy [29]. A UK study using individual data from the UK Biobank of 1450 participants, including 669 COVID-19 confirmed cases, that adjusted the models for confounding variables, reported significant positive associations of PM2.5 and PM10 with SARS-CoV-2 infectivity [32].
In addition, to explore the link between air pollution and the spread of COVID-19, several studies have analysed the effect of air pollution on the prognosis of COVID-19. Although these studies answer a different question and may also have their own weaknesses, exploring the prognosis of COVID-19 is less influenced by the dynamics of infection and thus leads to less internal validity threats such as unmeasured confounding. In terms of COVID-19 mortality, NO2 was found to be related to COVID-19 in a study covering Italy, Spain, Germany and France that revealed 83% of COVID-19 fatalities occurred in the regions with the highest NO2 levels [33]. In an Italian study, COVID-19 mortality on 31 March 2020 was twice as high in the regions with the highest levels of PM2.5 compared to that in the regions with the lowest levels of PM2.5 [20].
Interpretations of these short- and long-term effects should be made in the context of their respective epidemiologic designs. Since all of the previously mentioned studies were ecological and cross-sectional, individual-level inferential statements cannot be derived. The currently available ecological studies have an inherent internal validity threat, unmeasured confounding bias. Potential confounders such as population density, socioeconomic composition, access to healthcare and social distancing measures could also explain the findings. However, a US study examined the link between long-term ambient fine particulate concentrations and COVID-19 mortality taking these into account. This study, adjusted for multiple county-level confounders, covered more than 3000 US counties and examined whether the COVID-19-related mortality per capita varied by estimated county level PM2.5 concentrations across the 2000–2016 period. After adjusting for population density, average body mass index, levels of poverty, smoking rates, average temperature, humidity, and the number of tests performed in each state, the study concluded that a 1 µg·m3 increase in long-term PM2.5 concentration was associated with an 8% increase in COVID-19 mortality, which is a factor of 10 higher than all-cause mortality rates in the same counties reported in a previous analysis [30]. By taking into account the time of virus introduction in each county, this study also took into account the dynamic of the disease. Although this study adjusted for potential confounders, it is based on the assumption that previous PM2.5 concentrations were still representative of those during 2016–2020. Another limitation comes from the fact that many of the county-level adjustment factors resulted from the Behavioral Risk Factor Survey that excluded institutionalised residents, whereas they represent a large part of COVID-19 deaths [36].
Recently, another large study conducted at the county level in the USA showed a significant relationship between long-term exposure to NO2 and COVID-19 mortality [31]. This study used both single and multi-pollutant models and controlled for spatial trends and a comprehensive set of potential confounders including state-level test positive rates, county-level healthcare capacity, phase-of-epidemic, population mobility, socio-demographics, socioeconomic status, behaviour risk factors, and meteorological factors. Furthermore, the previously mentioned UK study, adjusted for population density, found that the levels of SO2 and NO2 recorded between 2018 and 2019, were positively linked to COVID-19 cases and COVID-19 mortality, while O3 was negatively correlated [32]. Although population density is important because it can promote both viral spread and air pollution, it fails to explain why some highly populated regions have larger numbers of COVID-19 cases and fatalities, pointing to socioeconomic and racial inequalities, including disparities related to environmental exposure. Recent data from the USA indicate that the number of COVID-19 cases and fatalities experienced by Black citizens were higher (up to 75%) than in the rest of the population, although they constitute only a small proportion (32% at maximum) of the overall population [38]. Outside conventional epidemiology, comprehensive econometric methods were applied to study the effect of PM2.5 and COVID-19 cases, hospitalisations and deaths in all 355 municipalities in the Netherlands [34]. The authors attempted a number of sensitivity analyses to account for unmeasured confounding (or omitted variable bias as commonly known in econometrics), measurement error in the exposure and the outcome and spatial spill-over. They found that a 1 µg·m−3 increase in long-term PM2.5 is associated with nine additional cases, three additional hospital admissions, and two additional deaths. Interestingly, this study found that the relationships between air pollution and COVID- 19 are also observed in rural zones, which suggests that air pollution plays a direct role in impacting COVID-19, independent of an urban setting with all its characteristics, such as density or crowding. In those rural zones, PM2.5 mainly arises from agricultural sources and especially from intensive livestock farming that leads to NH3 release. By interacting with other gaseous pollutants such as NOx, atmospheric NH3 leads to PM2.5 formation. In Europe, agriculture is one of the main sources of PM2.5 [39].
Additional limitations of these observational studies include the fact that most environmental health studies estimate conditional associations between non-randomised environmental exposures and health outcomes by directly regressing observed data without conceptual and design stages [40], an approach that is well documented to not guarantee valid causal inferences in general, especially in air pollution studies in which environmental exposures can be correlated with each other and effects are small [41, 42]. Disentangling independent air pollutant effects has been a challenge for observational studies, but can be achieved if the latter is embedded into a hypothetical multi-pollutant randomised experiment [42]. Moreover, to assess the robustness of epidemiological conclusions, air pollution studies should systematically consider sensitivity analyses that either: 1) vary the magnitude of the relationships between the unmeasured background covariates and both the exposure assignment mechanism and the outcome; or 2) study deviations from the assumed exposure assignment mechanism. Further investigations taking into account these limitations are needed to better understand the relationship of air pollution exposure to COVID-19.
Potential mechanisms explaining the relationship between air pollution and COVID-19 morbidity and mortality
How does air pollution affect COVID-19 comorbidities?
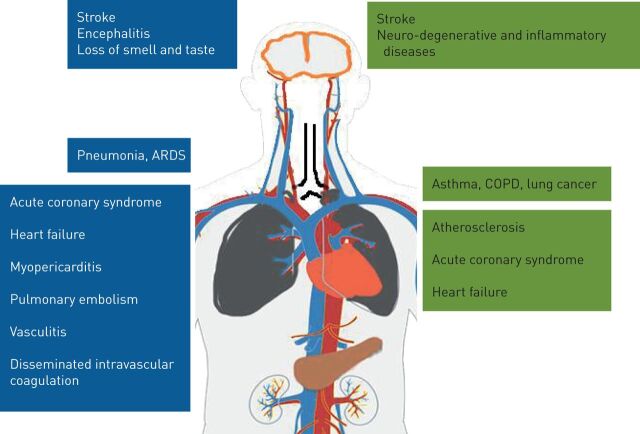
Air pollution and COVID-19 impact the same organs (figure 1). The course of COVID-19 indicates the very important role of the existence of comorbidities, which significantly increase a patient's risk profile. These include many chronic diseases such as cardiopulmonary and metabolic diseases, which air pollution adversely contributes to through both long- and short-term/acute air pollution exposure. This may explain why exposure to air pollution could indirectly predispose people to severe and more lethal forms of COVID-19, as suggested by epidemiological studies. In addition, air pollution may facilitate viral entry into the body via several processes. Herein, we review the main physiological systems affected by both air pollutants and SARS-CoV-2 and discuss the underlying mechanisms.
FIGURE 1.
Target organs and the main diseases that coronavirus disease 2019 (blue) and air pollution (green) share. ARDS: acute respiratory distress syndrome.
Impact on the respiratory system
Respiratory effects of COVID-19 range from mild illness of the upper airways to severe pneumonia and acute respiratory distress syndrome (ARDS). Air pollution affects the respiratory system through lung inflammation and oxidative stress that lead to short- and long-term effects on obstructive airway diseases such as asthma and COPD, and restrictive lung parenchyma diseases such as fibrosis [43, 44]. By weakening the respiratory system, air pollution could increase the severity of SARS-CoV-2 pneumonia, as suggested by the fact that children exposed to high NO2 concentrations are more likely to have severe forms of virus-induced asthma [45]. Additionally, it is well-documented that air pollution is correlated with chronic rhinitis and rhinosinusitis, which may increase airway mucosal permeability and allow for an easier penetration of SARS-CoV-2 [46, 47]. Recently, it has been suggested that nasal epithelial cells may be the primary access for SARS-CoV-2 [48]. SARS-CoV-2 uses angiotensin converting enzyme-2 (ACE2) receptors in the nasal and upper airways for cellular entry, which in turn, requires the cleavage of the spike protein by proteases and then, the binding of the spike protein to a specific cellular receptor such as ACE2 [48]. A study reported that inhibitors of proteases could be efficient to block viral entry [49]. As discussed later in this review (see the section In vitro studies), air pollution may actually activate proteases and thus facilitate viral entry in the cells. ACE2 receptors are found in several organs targeted by COVID-19, including the cardiovascular system [50].
Impact on the cardiovascular and metabolic system
Long-term exposure to air pollution increases all-cause mortality, particularly cardiovascular mortality. An annual increase of 10 µg·m−3 in PM2.5 has been associated with an 11% increase in cardiovascular mortality [51]. Similarly, a short-term increase in PM2.5 was associated with an increase in acute cardiovascular events [52]. In particular, stroke and ischaemic heart diseases drive the majority of air pollution-related mortality [53, 54]. The underlying mechanisms for the adverse cardiovascular effects of air pollution include oxidative stress and systemic inflammatory reactions that can be measured in the lungs and in the blood, such as increases in interleukin (IL)-1, IL-6, IL-8, IL-18, tumour necrosis factor-α, vascular endothelial growth factor, or intercellular adhesion molecule-1 [4, 55]. Systemic oxidative stress through reactive oxygen and nitrogen species can lead to endothelial dysfunction dysfunction, characterised by vasoconstriction/vasodilation imbalance, platelet activation, and leukocyte vascular adhesion that finally lead to atherothrombosis, myocardial infarction and stroke [4].
Several studies have reported that serious forms of COVID-19 are also associated with vascular endothelial inflammation and dysfunction [56]. Cardiovascular impairment of COVID-19 includes myopericarditis, acute coronary syndromes and heart failure, as well as vascular thrombotic diseases such as pulmonary embolism, vasculitis and disseminated intravascular coagulation that can lead to multiple organ failure. Severe forms of COVID-19 are also associated with systemic inflammation, and increased pro-thrombotic factors and markers of inflammation such as antiphospholipid antibodies, fibrinogen and D-dimer. Systemic acute inflammation in severe forms of COVID-19 is associated with increased levels of IL-6 and macrophage activation syndrome (also referred to as cytokine storms) that can lead to multiple organ failure [3]. Moreover, systemic inflammation in COVID-19 may play a role in destabilising coronary plaques resulting in acute coronary syndrome [57].
Air pollution is also involved in developing cardiometabolic risk factors like hypertension and insulin resistance [58, 59]. Chinese studies have found that hypertension or coronary heart disease were prevalent in about 38% of COVID-19 patients, whereas diabetes was prevalent in approximately 19% of patients [60]. Additionally, these patients showed higher risk of mortality from COVID-19 [61].
Impact on the neurological system
Neurological impairments in COVID-19, other than cerebrovascular diseases, include loss of smell and taste and encephalitis, and it has been described that SARS-CoV-2 could also be a neurotropic invasive virus that is site specific for the olfactory system [62]. In addition, evidence shows that ultrafine particulate matter can enter the olfactory epithelium then translocate to the olfactory bulb and potentially migrate to the olfactory cortex, thus implicating air pollution in the development of neurodegenerative disorders and neuroinflammatory diseases [63, 64]. However, although ultrafine particles and SARS-CoV-2 share the same mechanisms of migration through the olfactory nerves, to date, there is actually no evidence that exposure to ultrafine particles increases the risk of neurological impairment by SARS-CoV-2.
How might air pollution interact with SARS-CoV-2 in the human body? Evidence from experimental studies
Aside from the impact of air pollution on COVID-19 comorbidities at the population level, air pollutants may also decrease the immune response and facilitate respiratory virus replication as suggested by experimental studies that have explored the effect of concomitant exposure to air pollutants and respiratory viruses.
Human studies
A study involving 222 children reported that those living close to an electronic waste area with high PM2.5 concentrations had significantly decreased levels of salivary agglutinin (one of the primary antimicrobial proteins and peptides) compared to children living in a less polluted area [65]. In a randomised double-blind study in which volunteers were exposed to either ambient air or air with high levels of NO2, in an environmental chamber, analysis of bronchoalveolar lavage has demonstrated that macrophages were less efficient in inactivating the influenza virus when individuals were previously exposed to NO2 compared to ambient air [66]. In another randomised double-blind study, nonsmoking volunteers were exposed to wood smoke particles or filtered air before nasal inoculation with a vaccine dose of live attenuated influenza virus. When exposed to smoke particles, nasal lavage revealed reduced levels of interferon-γ-induced protein-10, which may lead to a decrease in immune response to viral infection, as interferon-γ-induced protein-10 is involved in the recruitment of cytotoxic lymphocytes [67]. In another double-blind randomised study including both healthy volunteers and volunteers with allergic rhinitis, nasal inoculation of live attenuated influenza virus was performed after 2 h of exposition to diesel exhaust or clean air and concluded that diesel exposure led to eosinophil activation and increased viral replication in the allergic group, highlighting the fact that diesel exhaust promotes infllamation and reduces virus clearance in allergic patients [68].
In vitro studies
As suggested by human studies, in vitro studies have also reported that air pollution may reduce antimicrobial activity through the downregulation of antimicrobial proteins and peptides (e.g. salivary agglutinin and surfactant protein D) in respiratory mucosal fluid [69]. In the epithelial lining fluid of the human respiratory tract, air pollutants, in particular O3 and PM, induce oxidative stress and the formation of reactive oxygen species that lead to antioxidant and surfactant depletion [70]. A similar experimental study demonstrated that when RSV is associated with ultrafine particles from combustion sources (e.g. those found in diesel exhaust) and then incubated with an epithelial cell culture, the inflammatory response was elevated compared to that from RSV alone [13, 71]. Another in vitro study examining human lung epithelial cells infected by the H1N1 virus also showed that in vitro exposure to secondary organic aerosols, which are part of combustion-derived particles (e.g. traffic or heating), led to cell apoptosis and increased viral replication [72].
Studies using environmental chambers have investigated the interactions between fine particles and different types of bacteriophage viruses, which were mixed with each other and brought into contact with host bacteria. When the air and virus mixture was previously filtered through a high-efficiency particulate absorbing filter, which removes a large fraction of fine particles, the activity of the viruses on the host bacteria was significantly modified: for some viruses, the presence of fine particles had a potentiating effect on viral infection, whereas for other viruses, fine particles significantly reduced viral replication [73]. This study illustrates the fact that the chemical and electrical characteristics of both particles and viruses play an important role in virus–particulate matter interactions. Regarding NO2 and O3 and concomitant exposure to rhinovirus, a major increase in the production of IL-8 in human nasal and bronchial epithelial cells was seen, as well as a decrease in macrophage activity [13]. In human nasal epithelial cells exposed to influenza A, pre-exposure to O3 increases viral replication due to an increase in cellular protease activity resulting from oxidative stress [74]. Proteases play an essential role in driving the cleavage of the viral membrane protein hemagglutinin and of the spike protein, necessary for viral entry into the host cells [75, 76]. This proteolysis plays an important role in the spread of several respiratory viruses, including both SARS-CoV-1 and SARS-CoV-2 [48, 49].
Animal studies
When mice are exposed to RSV after being exposed to ultrafine particles from combustion sources, studies found a significant decrease in immune response, specifically, a decrease in tumour necrosis factor-α, lymphocytes and interferons in bronchoalveolar lavage fluid, which was followed over the next 7 days, by an exacerbation of inflammation and infection [13]. In another study, authors observed a significant reduction in the phagocytosis capacity of macrophages when they were previously exposed to ultrafine particles from combustion sources [13]. Similar results were observed when mice were previously exposed to high concentrations of NO2 for 2 days and then exposed to murine cytomegalovirus [13]. The amount of virus needed in order to become infected was 100 times lower than that for mice exposed to ambient air. O3 exposure in mice led to different results depending on time and duration of exposure. In particular, when exposure to O3 occurred at the same time as viral infection, O3 may have had an antiviral activity [13, 77, 78]. This paradoxical protective effect of O3 may be explained by its oxidising behaviour, degrading viral macromolecules such as lipids, proteins and nucleic acids. As an example, O3 has an antimicrobial effect in high concentrations and is used to disinfect waterborne pathogens, e.g. ozonated water used in dental medicine and O3 treatment for sewage water [79, 80]. However, for O3 to decrease viral suspension in the air, the O3 must be present in high concentrations, which are not seen by current ambient levels. Thus, we do not expect the overall effect of O3 to decrease the viral load within humans or animals, but rather to impair immune response.
In rats, exposure to PM, especially ultrafine particles, induces the expression of the renin-angiotensin-aldosterone system (RAAS) and increases the expression of AT1R, a receptor of angiotensin 2, which is involved in lung and heart injuries [81, 82]. RAAS is involved in several diseases: while angiotensin 2 (through AT1 receptors) leads to vasoconstriction and inflammation, ACE2 receptor converts angiotensin 2 into protective angiotensin 1–7 with anti-inflammatory and vasodilating properties [83]. Severe forms of COVID-19, such as ARDS, have been associated with ACE2/AT1R imbalances [83, 84]. Although the exact role of RAAS on the severity of COVID-19 is still unclear, we know that SARS-CoV-2 uses the ACE2 receptor to enter the cells, which may lead to an increase in bioavailability and toxicity of angiotensin 2 through AT1R. In addition to the possible increase of AT1R expression due to PM, previous studies on rats have also reported that NO2 exposure induces the expression of ACE (angiotensin-converting enzyme that converts angiotensin 1 into angiotensin 2) and increases the liaison of angiotensin 2 to AT1R [85]; however, further studies are needed to explore the exact role of RAAS on the severity of COVID-19 and the possible role of air pollution.
How does air pollution interact with SARS-CoV-2 in the air?
In addition to their interactions with the body, air pollutants and respiratory viruses show complex interactions in ambient air which can influence the persistence of the virus in the air. Of extreme importance in the investigation of the relationship between air pollution and SARS-CoV-2 are: 1) the mode of transport or transmission of SARS-CoV-2; and 2) the possible physicochemical interactions between respiratory viruses and pollutants present in ambient air.
Droplets and aerosols
The physical size of SARS-CoV-2 is approximately 70–130 nm and its transmission mainly arises through the emission of macro droplets of 50–100 µm, e.g. through coughing and sneezing. Macro droplets consist of the virus in the centre, surrounded by water molecules and also mucus or cellular debris. These macro droplets are heavy and fall rapidly to the ground, which is why a safety distance of 2 m has been established by the health authorities in order to limit viral transmission. Macro droplets can also be deposited on solid surfaces where viral RNA is still quantifiable several hours later [86]. As has been demonstrated for influenza and measles, and suspected for SARS-CoV-1, recent studies are also considering SARS-CoV-2 transmission by aerosols formed by micro- and nano-droplets, also called droplet nuclei and defined as a suspended system of solid or liquid particles <5 µm in the air [87–90]. Depending on the particle mass, aerosols are able to remain suspended in the air for several hours and studies have found that SARS-CoV-2 RNA is still detected in the air 3 h after being emitted [86]. The role of airborne transmission of SARS-CoV-2 in COVID-19 is getting more recognition now. Some doubt remains about the pathogenicity of SARS-CoV-2 in suspensions because aerosols are smaller than macro droplets, and may contain fewer viable viruses but, in a recent study, increases in viral SARS-CoV-2 RNA during cell culture of the virus from recovered aerosol emitted by patients through breathing or speaking demonstrated the presence of infectious, replicating virions in <1 µm aerosol samples at a significant level (p<0.05) [91]. Western blot and transmission electron microscopy) analysis of these samples also showed evidence of viral proteins and intact virions. The infectious nature of aerosols collected in this study suggests that airborne transmission of COVID-19 is possible, and that aerosol prevention measures are necessary to effectively stem the spread of SARS-CoV-2. Other studies are needed to support it.
Airborne SARS-CoV-2 and PM and related mechanisms
Regardless of infectivity, SARS-CoV-2 emitted in aerosol form, for example from breathing, should behave like ultrafine particles arising from other sources, such as road traffic and heating. Viruses may mix with other suspended ultrafine particles, and thus may agglomerate to form particles of various sizes through aggregation condensation of ultrafine particles and nucleation of gaseous precursors [92, 93]. It is also well known that particles may serve as vectors for various substances such as pollen and microorganisms, including bacteria, fungi and viruses [94, 95]. It has previously been demonstrated that viruses are carried by fine particles. For example, a study has shown that the avian influenza virus was transmitted by dust particles between separated chicken groups [96]. SARS-CoV-2 RNA has recently been detected on PM in the Bergamo Province, Northern Italy, collected over a continuous 3 week period by two independent air samplers [97]. However, the presence of viral RNA does not necessarily mean viral infectivity, which depends on viral structural integrity, such as surface glycoproteins and sufficient viral concentration [98].
How do weather-related conditions impact the air pollution–virus interaction?
Meteorological conditions may also play an important role in air pollution–virus interactions. Although, a recent study did not find any evidence of an association between solar UV radiation and COVID-19, several studies have shown that UV radiation has antibacterial and antiviral impacts, both through a direct effect and through the formation of O3 and reactive oxygen species [99, 100]. Air pollution, especially PM, can reduce UV penetration, as demonstrated by several studies which reported that air pollutants such as PM significantly reduce the amount of UVB received by the body and thus, lead to reduced vitamin D synthesis [101–105]. In high concentrations, O3 can also filter UVB and can lead to decreased vitamin D levels. Vitamin D has an antioxidant activity, which has protective effects against respiratory viral infections and against oxidative stress due to air pollution [106, 107]. In addition, studies also reported that vitamin D modulates the inflammatory response to viral infections, regulates the RAAS and that reduced levels of vitamin D may play a role in the severity of COVID-19 [108, 109].
Regarding the influence of relative humidity, several studies have reported negative associations with SARS-CoV-2 transmission [110, 111]. As viral droplets contain water, humidity may play a role in evaporation and rehydration of viral aerosols, and thus be involved in viral stability, but its actual role in viral transmission and viral activity remains unknown [112]. Although there is as yet no consensus on the subject, it seems likely that a decrease in humidity leads to dehydration of macro droplets into smaller droplets that are able to remain in suspension in the ambient air, while an increase in humidity may lead to increased water content of viral droplets, and thus increased weight, enabling them to fall to the ground faster.
Whereas colder conditions lead to an increase in other respiratory viral transmissions such as influenza, we have found contradictory results on the link between COVID-19 and temperature [113]. Although several studies found that temperature was negatively associated with COVID-19 mortality and transmission, another study did not find any evidence of an association [23, 109, 111]. As recently reported, dry (relative humidity <20%) and cold air affects both immunity and viral spread owing to the fact that dry air may facilitate airborne viral transport, and that cold conditions, in addition to low humidity, may impair the functioning of airways ciliated cells [113]. Relative humidity and temperature also have an influence on ambient air pollution concentrations. In a cold season, the dense cold air causes a temperature inversion, which reduces air mixing and thus, exacerbates existing particulate pollution. The persistence of air pollutants can also be influenced by the lack of wind, preventing dispersion. Finally, atmospheric conditions that promote PM formation and stagnation may also promote SARS-CoV-2 persistence in the air [22]. In contrast, sunny days with hot temperatures and solar UV radiation increase the oxidative potential of the atmosphere, leading to higher O3 concentrations and may also lead to a reduced viability of SARS-CoV-2 in the air.
Discussion
Interactions between air pollutants and SARS-CoV-2 are summarised in figure 2. Existing literature suggests that PM and gaseous pollutants may act on COVID-19 in several ways. Air pollution may increase COVID-19 morbidity and mortality through its action on associated comorbidities. Experimental studies conducted for other respiratory viruses support the hypothesis that air pollution exposure may facilitate the occurrence of COVID-19 infection through a decrease in immune response. In vitro, animal and human studies have reported that exposure to air pollutants leads to increased mucosal permeability and oxidative stress, decreased antioxidants and surfactant antimicrobial proteins, as well as impaired macrophage phagocytosis. In addition, SARS-CoV-2 entry in host cells through ACE 2 requires the cleavage of the viral spike protein by proteases, and such protease activity may be increased by air pollution, as is documented for several other respiratory viruses.
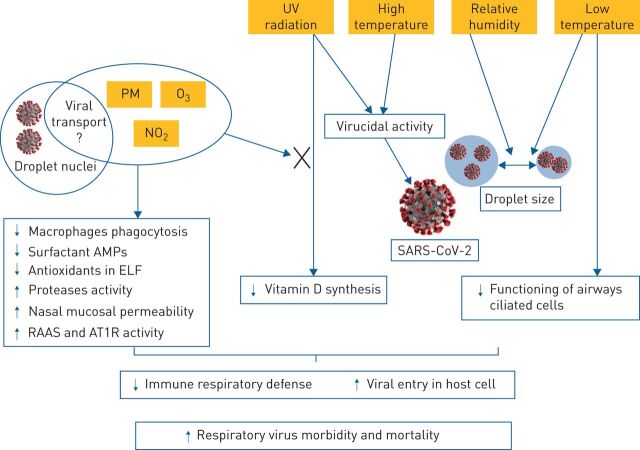
FIGURE 2.
Air pollutants/virus interaction according to atmospheric conditions. Relative humidity plays a role in the desiccation or hydration of viral droplet and, thus, influences the size of the droplet and the persistence of respiratory viruses in the air. Solar ultraviolet (UV) radiations have in vitro antiviral activity and lead to an increase in vitamin D synthesis. Atmospheric air pollutants may lead to decreased UV penetration leading to reduced vitamin D synthesis. Temperature influences the size of the viral droplet. In addition, low temperatures decrease the functioning of airways ciliated cells, while high temperatures may have antiviral activity. Droplet nuclei refers to viral droplets ≤5μm, it is also called viral airborne or viral aerosol. In addition to the common effect of air pollutants, which lead to a decrease in immune respiratory defence, particulate matter (PM) may be involved in respiratory virus transport. AMP: antimicrobial proteins and peptides; ELF: epithelial lining fluid; RASS: renin-angiotensin-aldosterone system; AT1R: angiotensin 2 receptor type 1.
In ambient air, pollutants and respiratory viruses show complex interactions. According to their composition, PM and viruses can interact and modify viral activity as shown by in vitro studies. In addition, PM is known to carry microorganisms such as viruses, and SARS-CoV-2 RNA has been identified on PM. It is still unclear, however, how long this virus remains infectious in ambient air and whether the low viral load contained within the aerosol is sufficient to induce infection. Atmospheric interactions between gases and viruses are also complex and depend on meteorological contributing factors such as UV radiation and relative humidity. By reducing UV radiations (which have antiviral activity) air pollutants may promote viral persistence in ambient air and also reduce vitamin D synthesis, and thus may play a role in the immune response against viral infections.
Although there is a need for more specific studies exploring interactions between air pollutants and SARS-CoV-2 in the ambient air and their impact on human health, this review highlights that both short- and long-term exposures to air pollution may be important aggravating factors for SARS-CoV-2 transmission and COVID-19 severity and lethality through multiple mechanisms. Future studies should also examine the role of indoor air pollution, in particular biomass and tobacco smoke, in COVID-19. The fact that both biology and atmospheric chemistry are separately implicated suggests that a more holistic approach to disease management and mitigation is necessary both in addressing the current COVID-19 pandemic and future viral epidemics. We cannot ignore that our surrounding environment may be exacerbating not only chronic disease, but infectious disease as well. In the light of these relationships between air pollution and COVID-19, and by virtue of the precautionary principle, we recommend the reduction of air pollution from all sources, especially that from road traffic and heat generation, through the reinforcement of public health policy.
Footnotes
Provenance: Submitted article, peer reviewed
Author contributions: T. Bourdrel conceived the idea for the review. T. Bourdrel designed and undertook the literature review and extracted the data with help from I. Annesi-Maesano and M-A. Bind. T. Bourdrel wrote the first draft of the manuscript with input from I. Annesi-Maesano and M-A. Bind. I. Annesi-Maesano synthesised this review. T. Bourdrel created the drawings and pictures. B. Alahmad and C.N. Maesano reviewed, edited and provided insights on the overall manuscript.
Conflict of interest: T. Bourdrel has nothing to disclose.
Conflict of interest: I. Annesi-Maesano has nothing to disclose.
Conflict of interest: B. Alahmad has nothing to disclose.
Conflict of interest: C.N. Maesano has nothing to disclose.
Conflict of interest: M-A. Bind has nothing to disclose.
Support statement: Research reported in this publication was supported by the John Harvard Distinguished Science Fellow Program within the FAS Division of Science of Harvard University, and by the Office of the Director, National Institutes of Health under Award Number DP5OD021412. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Zhu N, Zhang D, Wang W, et al. . A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382: 727–733. doi: 10.1056/NEJMoa2001017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Guan WJ, Liang WH, Zhao Y, et al. . Comorbidity and its impact on 1590 patients with Covid-19 in China: a nationwide analysis. Eur Respir J 2020; 55: 2000547. doi: 10.1183/13993003.00547-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McGonagle D, Sharif K, O'Regan A, et al. . The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun Rev 2020; 19: 102537. doi: 10.1016/j.autrev.2020.102537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bourdrel T, Bind M-A, Béjot Y, et al. . Cardiovascular effects of air pollution. Arch Cardiovasc Dis 2017; 110: 634–642. doi: 10.1016/j.acvd.2017.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schraufnagel DE, Balmes JR, Cowl CT, et al. . Air pollution and noncommunicable diseases: a review by the Forum of International Respiratory Societies’ Environmental Committee, Part 1: The Damaging Effects of Air Pollution. Chest 2019; 155: 409–416. doi: 10.1016/j.chest.2018.10.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Atkinson RW, Butland BK, Anderson HR, et al. . Long-term concentrations of nitrogen dioxide and mortality: a meta-analysis of cohort studies. Epidemiology 2018; 29: 460–472. doi: 10.1097/EDE.0000000000000847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Atkinson RW, Butland BK, Dimitroulopoulou C, et al. . Long-term exposure to ambient ozone and mortality: a quantitative systematic review and meta-analysis of evidence from cohort studies. BMJ Open 2016; 6: e009493. doi: 10.1136/bmjopen-2015-009493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.STROBE Statement . Strobe checklists. Version 4. www.strobe-statement.org/index.php?id=available-checklists Date last updated: Oct/Nov 2007.
- 9.Clay K. Pollution, Infectious Disease, and Mortality: Evidence from the 1918 Spanish Influenza Pandemic. J Economic Hist 2018; 78: 1179–1209. 10.1017/S002205071800058X. [DOI] [Google Scholar]
- 10.Pope CA, Burnett RT, Thurston GD, et al. . Cardiovascular mortality and long-term exposure to particulate air pollution: epidemiological evidence of general pathophysiological pathways of disease. Circulation 2004; 109: 71–77. doi: 10.1161/01.CIR.0000108927.80044.7F [DOI] [PubMed] [Google Scholar]
- 11.Cui Y, Zhang Z-F, Froines J, et al. . Air pollution and case fatality of SARS in the People's Republic of China: an ecologic study. Environ Heath 2003; 2: 15. doi: 10.1186/1476-069X-2-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Troeger C, Forouzanfar M, Rao PC, et al. . Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect Dis 2017; 17: 1133–1161. doi: 10.1016/S1473-3099(17)30396-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ciencewicki J, Jaspers I. Air pollution and respiratory viral infection. Inhal Toxicol 2007; 19: 1135–1146. doi: 10.1080/08958370701665434 [DOI] [PubMed] [Google Scholar]
- 14.Su W, Wu X, Geng X, et al. . The short-term effects of air pollutants on influenza-like illness in Jinan, China. BMC Public Health 2019; 19: 1319. doi: 10.1186/s12889-019-7607-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen G, Zhang W, Li S, et al. . Is short-term exposure to ambient fine particles associated with measles incidence in China? A multi-city study. Environ Res 2017; 156: 306–311. doi: 10.1016/j.envres.2017.03.046 [DOI] [PubMed] [Google Scholar]
- 16.Setti L, Passarini F, De Gennaro G, et al. . The potential role of particulate matter in the spreading of COVID-19 in Northern Italy: first evidence-based research hypotheses. medRxiv 2020; preprint [ 10.1101/2020.04.11.20061713]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bontempi E. First data analysis about possible COVID-19 virus airborne diffusion due to air particulate matter (PM): the case of Lombardy (Italy). Environ Res 2020; 186: 109639. doi: 10.1016/j.envres.2020.109639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zoran MA, Savastru RS, Savastru DM, et al. . Assessing the relationship between ground levels of ozone (O3) and nitrogen dioxide (NO2) with coronavirus (COVID-19) in Milan, Italy. Sci Total Environ 2020; 740: 140005. doi: 10.1016/j.scitotenv.2020.140005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zoran MA, Savastru RS, Savastru DM, et al. . Assessing the relationship between surface levels of PM2.5 and PM10 particulate matter impact on COVID-19 in Milan, Italy. Sci Total Environ 2020: 139825. doi: 10.1016/j.scitotenv.2020.139825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Frontera A, Cianfanelli L, Vlachos K, et al. . Severe air pollution links to higher mortality in COVID-19 patients: the “double-hit” hypothesis. J Infect 2020; 81: 255–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Conticini E, Frediani B, Caro D. Can atmospheric pollution be considered a co-factor in extremely high level of SARS-CoV-2 lethality in Northern Italy? Environ Pollut 2020; 261: 114465. doi: 10.1016/j.envpol.2020.114465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fronza R, Lusic M, Schmidt M, et al. . Spatial–temporal variations in atmospheric factors contribute to SARS-CoV-2 outbreak. Viruses 2020; 12: 588. doi: 10.3390/v12060588 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li H, Xu X-L, Dai D-W, et al. . Air pollution and temperature are associated with increased COVID-19 incidence: a time series study. Int J Infect Dis 2020; 97: 278–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang Y, Wu XJ, Guan YJ. Effect of ambient air pollutants and meteorological variables on COVID-19 incidence. Infect Control Hosp Epidemiol 2020; 41: 1011–1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yao Y, University F, China S, et al. . Ambient nitrogen dioxide pollution and spread ability of COVID-19 in Chinese cities. Ecotoxicol Environ Saf 2021; 208: 111421. doi: 10.1016/j.ecoenv.2020.111421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang B, Liu J, Fu S, et al. . An effect assessment of airborne particulate matter pollution on COVID-19: a multi-city Study in China. medRxiv 2020; preprint [ 10.1101/2020.04.09.20060137]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhu Y, Xie J, Huang F, et al. . Association between short-term exposure to air pollution and COVID-19 infection: evidence from China. Sci Total Environ 2020; 727: 138704. doi: 10.1016/j.scitotenv.2020.138704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adhikari A, Yin J. Short-term effects of ambient ozone, PM2.5, and meteorological factors on COVID-19 confirmed cases and deaths in Queens, New York. Int J Environ Res Public Health 2020; 17: 4047. doi: 10.3390/ijerph17114047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fattorini D, Regoli F. Role of the chronic air pollution levels in the Covid-19 outbreak risk in Italy. Environ Pollut 2020; 264: 114732. doi: 10.1016/j.envpol.2020.114732 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu X, Nethery RC, Sabath B, et al. . Exposure to air pollution and COVID-19 mortality in the United States: A nationwide cross-sectional study. Sci Advances 2020; 45: eabd4049. doi: 10.1126/sciadv.abd4049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liang D, Shi L, Zhao J, et al. . Urban Air Pollution May Enhance COVID-19 Case-Fatality and Mortality Rates in the United States. Innovation (N Y) 2020:10004. medRxiv 2020; preprint [ 10.1101/2020.05.04.20090746]. [DOI] [PMC free article] [PubMed]
- 32.Travaglio M, Popovic R, Yu Y, et al. . Links between air pollution and COVID-19 in England. Environ Pollut 2021; 268: 115859. 10.1016/j.envpol.2020.115859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ogen Y. Assessing nitrogen dioxide (NO2) levels as a contributing factor to coronavirus (COVID-19) fatality. Sci Total Environ 2020; 726: 138605. doi: 10.1016/j.scitotenv.2020.138605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cole MA, Ozgen C, Strobl E. Air Pollution Exposure and Covid-19, IZA Discussion Papers No. 13367. Bonn, Insitute of Labor Economics, 2020. [Google Scholar]
- 35.Pozzer A, Dominici F, Haines A, et al. . Regional and global contributions of air pollution to risk of death from COVID-19. Cardiovascular Res 2020; 116: 2247–2253. doi: 10.1093/cvr/cvaa288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Villeneuve PJ, Goldberg MS. Methodological considerations for epidemiological studies of air pollution and the SARS and COVID-19 coronavirus outbreaks. Environ Health Perspect 2020; 128: 95001. doi: 10.1289/EHP7411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Heederik DJJ, Smit LAM, Vermeulen RCH. Go slow to go fast: a plea for sustained scientific rigor in air pollution research during the COVID-19 pandemic. Eur Respir J 2020; 56: 2001361. doi: 10.1183/13993003.01361-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Brandt EB, Beck AF, Mersha TB. Air pollution, racial disparities, and COVID-19 mortality. J Allergy Clin Immunol 2020; 146: 61–63. doi: 10.1016/j.jaci.2020.04.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lelieveld J, Evans J, Fnais M, et al. . The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015; 525: 367–371. doi: 10.1038/nature15371 [DOI] [PubMed] [Google Scholar]
- 40.Bind MAC, Rubin DB. Bridging observational studies and randomized experiments by embedding the former in the latter. Stat Methods Med Res 2019; 28: 1958–1978. doi: 10.1177/0962280217740609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rubin DB. The design versus the analysis of observational studies for causal effects: Parallels with the design of randomized trials. Stat Med 2007; 26: 20–36. doi: 10.1002/sim.2739 [DOI] [PubMed] [Google Scholar]
- 42.Bind M-A. Causal modeling in environmental health. Annu Rev Public Health 2019; 40: 23–43. doi: 10.1146/annurev-publhealth-040218-044048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Guan WJ, Zheng XY, Chung KF, et al. . Impact of air pollution on the burden of chronic respiratory diseases in China: time for urgent action. Lancet 2016; 388: 1939–1951. doi: 10.1016/S0140-6736(16)31597-5 [DOI] [PubMed] [Google Scholar]
- 44.Conti S, Harari S, Caminati A, et al. . The association between air pollution and the incidence of idiopathic pulmonary fibrosis in Northern Italy. Eur Respir J 2018; 51: 1700397. doi: 10.1183/13993003.00397-2017 [DOI] [PubMed] [Google Scholar]
- 45.Chauhan AJ, Inskip HM, Linaker CH, et al. . Personal exposure to nitrogen dioxide (NO2) and the severity of virus-induced asthma in children. Lancet 2003; 361: 1939–1944. doi: 10.1016/S0140-6736(03)13582-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Annesi-Maesano I, Hulin M, Lavaud F, et al. . Poor air quality in classrooms related to asthma and rhinitis in primary schoolchildren of the French 6 Cities Study. Thorax 2012; 67: 682–688. doi: 10.1136/thoraxjnl-2011-200391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.London NR, Lina I, Ramanathan M. Aeroallergens, air pollutants, and chronic rhinitis and rhinosinusitis. World J Otorhinolaryngol Head Neck Surg 2018; 4: 209–215. doi: 10.1016/j.wjorl.2018.08.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sungnak W, Huang N, Bécavin C, et al. . SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Med 2020; 26: 681–687. doi: 10.1038/s41591-020-0868-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hoffmann M, Kleine-Weber H, Schroeder S, et al. . SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181: 271–280. doi: 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zheng YY, Ma YT, Zhang JY, et al. . COVID-19 and the cardiovascular system. Nat Rev Cardiol 2020; 17: 259–260. doi: 10.1038/s41569-020-0360-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hoek G, Krishnan RM, Beelen R, et al. . Long-term air pollution exposure and cardio- respiratory mortality: a review. Environ Health 2013; 12: 43. doi: 10.1186/1476-069X-12-43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nawrot TS, Perez L, Künzli N, et al. . Public health importance of triggers of myocardial infarction: a comparative risk assessment. Lancet 2011; 377: 732–740. doi: 10.1016/S0140-6736(10)62296-9 [DOI] [PubMed] [Google Scholar]
- 53.Feigin VL, Roth GA, Naghavi M, et al. . Global burden of stroke and risk factors in 188 countries, during 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet Neurol 2016; 15: 913–924. doi: 10.1016/S1474-4422(16)30073-4 [DOI] [PubMed] [Google Scholar]
- 54.Lelieveld J, Klingmüller K, Pozzer A, et al. . Cardiovascular disease burden from ambient air pollution in Europe reassessed using novel hazard ratio functions. Eur Heart J 2019; 40: 1590–1596. doi: 10.1093/eurheartj/ehz135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bind M-A, Baccarelli A, Zanobetti A, et al. . Air pollution and markers of coagulation, inflammation, and endothelial function: associations and epigene-environment interactions in an elderly cohort. Epidemiology 2012; 23: 332–340. doi: 10.1097/EDE.0b013e31824523f0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Varga Z, Flammer AJ, Steiger P, et al. . Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020; 395: 1417–1418. doi: 10.1016/S0140-6736(20)30937-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ferrari R, Di Pasquale G, Rapezzi C. Commentary: what is the relationship between Covid-19 and cardiovascular disease? Int J Cardiol 2020; 310: 167–168. doi: 10.1016/j.ijcard.2020.03.074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yang B, Guo Y, Markevych I, et al. . Association of long-term exposure to ambient air pollutants with risk factors for cardiovascular disease in China. JAMA Netw Open 2019; 2: e190318. doi: 10.1001/jamanetworkopen.2019.0318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Münzel T, Sørensen M, Gori T, et al. . Environmental stressors and cardio-metabolic disease: part II–mechanistic insights. Eur Heart J 2017; 38: 550–556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhou F, Yu T, Du R, et al. . Articles Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: 1054–1062. doi: 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020; 323: 1239–1242. [DOI] [PubMed] [Google Scholar]
- 62.Xydakis MS, Dehgani-Mobaraki P, Holbrook EH, et al. . Smell and taste dysfunction in patients with COVID-19. Lancet Infect Dis 2020; 20: 1015–1016. doi: 10.1016/S1473-3099(20)30293-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ajmani GS, Suh HH, Pinto JM. Effects of ambient air pollution exposure on olfaction: a review. Environ Health Perspect 2016; 124: 1683–1693. doi: 10.1289/EHP136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Peeples L. News feature: How air pollution threatens brain health. Proc Natl Acad Sci 2020: 202008940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhang S, Huo X, Zhang Y, et al. . Ambient fine particulate matter inhibits innate airway antimicrobial activity in preschool children in e-waste areas. Environ Int 2019; 123: 535–542. doi: 10.1016/j.envint.2018.12.061 [DOI] [PubMed] [Google Scholar]
- 66.Frampton MW, Smeglin AM, Roberts NJ, et al. . Nitrogen dioxide exposure in vivo and human alveolar macrophage inactivation of influenza virus in vitro. Environ Res 1989; 48: 179–192. doi: 10.1016/S0013-9351(89)80033-7 [DOI] [PubMed] [Google Scholar]
- 67.Rebuli ME, Speen AM, Martin EM, et al. . Wood smoke exposure alters human inflammatory responses to viral infection in a sex-specific manner: a randomized, placebo-controlled study. Am J Respir Crit Care Med 2019; 199: 996–1007. doi: 10.1164/rccm.201807-1287OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Noah TL, Zhou H, Zhang H, et al. . Diesel exhaust exposure and nasal response to attenuated influenza in normal and allergic volunteers. Am J Respir Crit Care Med 2012; 185: 179–185. doi: 10.1164/rccm.201103-0465OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang S, Huo X, Zhang Y, et al. . The association of PM2.5 with airway innate antimicrobial activities of salivary agglutinin and surfactant protein D. Chemosphere 2019; 226: 915–923. doi: 10.1016/j.chemosphere.2019.04.032 [DOI] [PubMed] [Google Scholar]
- 70.Lakey PSJ, Berkemeier T, Tong H, et al. . Chemical exposure-response relationship between air pollutants and reactive oxygen species in the human respiratory tract. Sci Rep 2016; 6: 32916. doi: 10.1038/srep32916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Cruz-Sanchez TM, Haddrell AE, Hackett TL, et al. . Formation of a stable mimic of ambient particulate matter containing viable infectious respiratory syncytial virus and its dry-deposition directly onto cell cultures. Anal Chem 2013; 85: 898–906. doi: 10.1021/ac302174y [DOI] [PubMed] [Google Scholar]
- 72.Wang S, Zhang X, Chu H, et al. . Impact of Secondary Organic Aerosol Exposure on the Pathogenesis of Human Influenza Virus (H1N1). American Geophysical Union, Fall Meeting, 2018, GH13B-0943. [Google Scholar]
- 73.Groulx N, Urch B, Duchaine C, et al. . The Pollution Particulate Concentrator (PoPCon): a platform to investigate the effects of particulate air pollutants on viral infectivity. Sci Total Environ 2018; 628–629: 1101–1107. doi: 10.1016/j.scitotenv.2018.02.118 [DOI] [PubMed] [Google Scholar]
- 74.Kesic MJ, Meyer M, Bauer R, et al. . Exposure to ozone modulates human airway protease/antiprotease balance contributing to increased influenza A infection. PLoS One 2012; 7: e35108. doi: 10.1371/journal.pone.0035108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Simmons G, Zmora P, Gierer S, et al. . Proteolytic activation of the SARS-coronavirus spike protein: cutting enzymes at the cutting edge of antiviral research. Antiviral Res 2013; 100: 605–614. doi: 10.1016/j.antiviral.2013.09.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Limburg H, Harbig A, Bestle D, et al. . TMPRSS2 is the major activating protease of influenza A virus in primary human airway cells and influenza B virus in human type II pneumocytes. J Virol 2019; 93: e00649-19. doi: 10.1128/JVI.00649-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Jakab GJ, Spannhake EW, Canning BJ, et al. . The effects of ozone on immune function. Environ Health Perspect 1995; 103: Suppl. 2, 77–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Dubuis ME, Dumont-Leblond N, Laliberté C, et al. . Ozone efficacy for the control of airborne viruses: bateriophage and norovirus models. PLoS One 2020; 15: e0231164. doi: 10.1371/journal.pone.0231164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Elvis AM, Ekta JS. Ozone therapy: a clinical review. J Nat Sci Biol Med 2011; 2: 66–70. doi: 10.4103/0976-9668.82319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gray NF. Ozone Disinfection. Percival S, Yates MV, Williams DD, et al., eds. In: Microbiology of Waterborne Diseases. Microbiological Aspects and Risk. 2nd Edn. 2013. Cambridge, MA, Academic press; pp. 599–615. [Google Scholar]
- 81.Aztatzi-Aguilar OG, Uribe-Ramírez M, Arias-Montaño JA, et al. . Acute and subchronic exposure to air particulate matter induces expression of angiotensin and bradykinin-related genes in the lungs and heart: angiotensin-II type-I receptor as a molecular target of particulate matter exposure. Part Fibre Toxicol 2015; 12: 17. doi: 10.1186/s12989-015-0094-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Morales-Rubio RA, Alvarado-Cruz I, Manzano-León N, et al. . In utero exposure to ultrafine particles promotes placental stress-induced programming of renin-angiotensin system-related elements in the offspring results in altered blood pressure in adult mice. Part Fibre Toxicol 2019; 16: 7. doi: 10.1186/s12989-019-0289-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cheng H, Wang Y, Wang G-Q. Organ-protective effect of angiotensin-converting enzyme 2 and its effect on the prognosis of COVID-19. J Med Virol 2020; 92: 726–730. doi: 10.1002/jmv.25785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rivellese F, Prediletto E. ACE2 at the centre of COVID-19 from paucisymptomatic infections to severe pneumonia. Autoimmun Rev 2020; 19: 102536. doi: 10.1016/j.autrev.2020.102536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Alifano M, Alifano P, Forgez P, et al. . Renin-angiotensin system at the heart of COVID-19 pandemic. Biochimie 2020; 174: 30–33. doi: 10.1016/j.biochi.2020.04.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.van Doremalen N, Bushmaker T, Morris DH, et al. . Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 2020; 382: 1564–1567. doi: 10.1056/NEJMc2004973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Liu Y, Ning Z, Chen Y, et al. . Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 2020; 582: 557–560. [DOI] [PubMed] [Google Scholar]
- 88.National Academies of Sciences, Engineering and Medicine . Rapid Expert Consultation on the Possibility of Bioaerosol Spread of SARS-CoV-2 for the COVID-19 Pandemic (April 1, 2020). Washington, DC, National Academies Press, 2020. 10.17226/25769]. [DOI] [Google Scholar]
- 89.Setti L, Passarini F, De Gennaro G, et al. . Airborne transmission route of COVID-19: why 2 meters/6 feet of inter-personal distance could not be enough. Int J Environ Res Public Health 2020; 17: 2932. doi: 10.3390/ijerph17082932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Morawska L, Cao J. Airborne transmission of SARS-CoV-2: the world should face the reality. Environ Int 2020; 139: 105730. doi: 10.1016/j.envint.2020.105730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Santarpia JL, Herrera VL, Rivera DN, et al. . The infectious nature of patient-generated SARS-CoV-2 aerosol. MedRxiv 2020; preprint [ 10.1101/2020.07.13.20041632]. [DOI] [Google Scholar]
- 92.Dominici F, Wang Y, Correia AW, et al. . Chemical composition of fine particulate matter and life expectancy: in 95 US counties between 2002 and 2007. Epidemiology 2015; 26: 556–564. doi: 10.1097/EDE.0000000000000297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kelly FJ, Fussell JC. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos Environ 2012; 60: 504–526. doi: 10.1016/j.atmosenv.2012.06.039 [DOI] [Google Scholar]
- 94.Cao C, Jiang W, Wang B, et al. . Inhalable microorganisms in Beijing's PM2.5 and PM10 pollutants during a severe smog event. Environ Sci Technol 2014; 48: 1499–1507. doi: 10.1021/es4048472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kalisa E, Archer S, Nagato E, et al. . Chemical and biological components of urban aerosols in Africa: current status and knowledge gaps. Int J Environ Res Public Health 2019; 16: 941. doi: 10.3390/ijerph16060941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Spekreijse D, Bouma A, Koch G, et al. . Quantification of dust-borne transmission of highly pathogenic avian influenza virus between chickens. Influenza Other Respi Viruses 2013; 7: 132–113. doi: 10.1111/j.1750-2659.2012.00362.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Setti L, Passarini F, De Gennaro G, et al. . SARS-Cov-2 RNA found on particulate matter of Bergamo in Northern Italy: first preliminary evidence. Environ Res 2020; 188: 109754. doi: 10.1016/j.envres.2020.109754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cheval S, Mihai Adamescu C, Georgiadis T, et al. . Observed and potential impacts of the COVID-19 pandemic on the environment. Int J Environ Res Public Health 2020; 17: 4140. doi: 10.3390/ijerph17114140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Cantwell RE, Hofmann R, Templeton MR. Interactions between humic matter and bacteria when disinfecting water with UV light. J Appl Microbiol 2008; 105: 25–35. doi: 10.1111/j.1365-2672.2007.03714.x [DOI] [PubMed] [Google Scholar]
- 100.Yao Y, Pan J, Liu Z, et al. . No association of COVID-19 transmission with temperature or UV radiation in Chinese cities. Eur Respir J 2020; 55: 2000517. doi: 10.1183/13993003.00517-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Welliver RC. Temperature, humidity, and ultraviolet B radiation predict community respiratory syncytial virus activity. Pediatr Infect Dis J 2007; 26: Suppl. 11, S29–S35. doi: 10.1097/INF.0b013e318157da59 [DOI] [PubMed] [Google Scholar]
- 102.Welch D, Buonanno M, Grilj V, et al. . Far-UVC light: a new tool to control the spread of airborne-mediated microbial diseases. Sci Rep 2018; 8: 2752. doi: 10.1038/s41598-018-21058-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Mousavi SE, Amini H, Heydarpour P, et al. . Air pollution, environmental chemicals, and smoking may trigger vitamin D deficiency: evidence and potential mechanisms. Environ Int 2019; 122: 67–90. doi: 10.1016/j.envint.2018.11.052 [DOI] [PubMed] [Google Scholar]
- 104.Hosseinpanah F, Pour SH, Heibatollahi M, et al. . The effects of air pollution on vitamin D status in healthy women: a cross sectional study. BMC Public Health 2010; 10: 519. doi: 10.1186/1471-2458-10-519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Baïz N, Dargent-Molina P, Wark JD, et al. . Gestational exposure to urban air pollution related to a decrease in cord blood vitamin D levels. J Clin Endocrinol Metab 2012; 97: 4087–4095. doi: 10.1210/jc.2012-1943 [DOI] [PubMed] [Google Scholar]
- 106.Péter S, Holguin F, Wood L, et al. . Nutritional solutions to reduce risks of negative health impacts of air pollution. Nutrients 2015; 7: 10398–10416. doi: 10.3390/nu7125539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Grant WB, Lahore H, McDonnell SL, et al. . Evidence that vitamin D supplementation could reduce risk of influenza and covid-19 infections and deaths. Nutrients 2020; 12: 988. doi: 10.3390/nu12040988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Laird E, Rhodes J, Kenny RA. Vitamin D and inflammation: potential implications for severity of Covid-19. Ir Med J 2020; 113: 81. [PubMed] [Google Scholar]
- 109.Mccartney DM, Byrne DG. Optimisation of Vitamin D status for enhanced immuno-protection against Covid-19. Ir Med J 2020; 113: 58. [PubMed] [Google Scholar]
- 110.Ma Y, Zhao Y, Liu J, et al. . Effects of temperature variation and humidity on the death of COVID-19 in Wuhan, China. Sci Total Environ 2020; 724: 138226. doi: 10.1016/j.scitotenv.2020.138226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wang J, Tang K, Feng K, et al. . High Temperature and High Humidity Reduce the Transmission of COVID-19. arXiv 2020; preprint [https://arxiv.org/abs/2003.05003]. arXiv:2003.05003v4. [Google Scholar]
- 112.Haddrell AE, Thomas RJ. Aerobiology: experimental considerations, observations, and future tools. Appl Environ Microbiol 2017; 83: e00809-17. doi: 10.1128/AEM.00809-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Moriyama M, Hugentobler WJ, Iwasaki A. Seasonality of respiratory viral infections. Annu Rev Virol 2020; 7: 83–101. doi: 10.1146/annurev-virology-012420-022445 [DOI] [PubMed] [Google Scholar]