Abstract
As of October 2020, there are over 40 million confirmed cases, and more than 1 million confirmed deaths of Covid-19 worldwide. The main cause of death in hospitalized patients is a respiratory failure due to acute respiratory distress syndrome. It has been suggested that the very intense immune response induces diffuse alveolar damage that far exceeds the harm that would have been caused by virus replication per se, resulting in lethal tissue destruction. We present a detailed report of the histopathological findings on cryo transbronchial biopsy in the patient with persistent (3 months) interstitial pneumonitis and severe CD8 positive cell infiltration in the lungs due to SARS-CoV-2 infection. CD8 positive T-lymphocytes have a great potential to damage tissue either through direct cytotoxicity or through cytokines release.
Keywords: SARS-CoV-2, Pneumonitis, CD8 positive T-lymphocytes
1. Introduction
As of October 2020, there are over 40 million confirmed cases, and more than 1 million confirmed deaths of Covid-19 worldwide [1]. The main reportable cause of death in hospitalized patients is a respiratory failure on the background of acute respiratory distress syndrome (ARDS) [2,3]. However, the long-term effects of SARS-CoV-2 infection in the lungs are not fully investigated so far, especially in antemortem tissue.
To our knowledge, there are few cases of antemortem lung biopsies on Covid-19 patients [4]. We report a case of a patient with persistent (3 months) interstitial inflammatory infiltrates and pneumonitis due to SARS-CoV-2 infection and detailed histopathological findings on cryo transbronchial biopsy.
2. Case summary
A 49-year old male with known chronic lymphocytic leukemia (CLL) (since 2010 with good disease response) presented to an emergency department of the Froedtert & the Medical College of Wisconsin regional hospital at the beginning of August 2020 with fever, chills, slight cough. He appeared overall well, and his vital signs did not indicate a severe systemic infection at that time. Physical exam and lab tests were unremarkable as well, but chest x-ray showed a subtle linear opacity involving the right midlung and left base. The patient tested positive for SARS-CoV-2. Chest CT showed extensive peripheral patchy and heterogeneous groundglass opacities consistent with SARS-CoV-2 pneumonia. However, the patient was hesitant and decided to be discharged despite possible benefits from a hospital admission.
Two weeks later the patient developed a fever of 40.3 °C, dry cough, pleuritic chest pain with deep breathing, and dyspnea. CT revealed progression of the bilateral patchy parenchymal groundglass opacities in the lungs with some confluent opacities within the lower lobes. He was immediately admitted to the inpatient unit. At this point the patient re-tested positive for SARS-CoV-2 by nasal swab, but extensive workup for additional infectious agents was negative. Despite appropriate management (aggressive steroid regiment, prophylactic antibiotics, IV antibiotics, ruxolitinib)., fever and bilateral pulmonary infiltrates persisted without improvement and there were additional areas in the lungs that have over the next 10 days after hospitalization. The signs of progression justified bronchoscopy and cryo transbronchial biopsy on the 11th day of admission. There were no endobronchial lesions or gross mucosal abnormalities visualized during the bronchoscopy procedure.
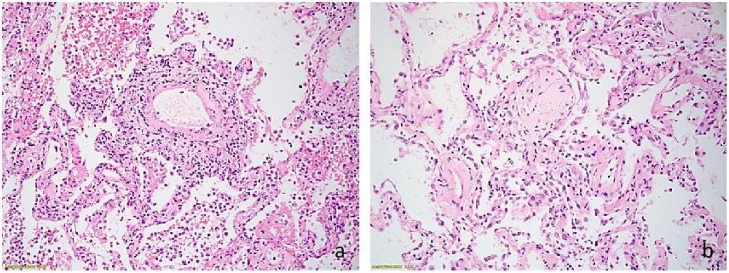
Tissue biopsies were formalin-fixed, paraffin-embedded, sectioned, stained with H&E, and subsequently examined in a light microscope. All lung samples showed interstitial inflammatory infiltrate and pneumonitis (Fig. 1 ). Inflammatory infiltrate was associated with patchy fibrin deposition, prominent fibroblastic foci, and occasional alveolar macrophages. Some small airways showed additional polypoid projections of granulation tissue. Reactive pneumocytes demonstrated nuclear enlargement and occasional syncytial changes.
Fig. 1.
a. Interstitial lymphocytic infiltration of the lung and alveolar fibrin (Hematoxylin-Eosin, 20x). b. Fibroblastic foci (Hematoxylin-Eosin, 20x).
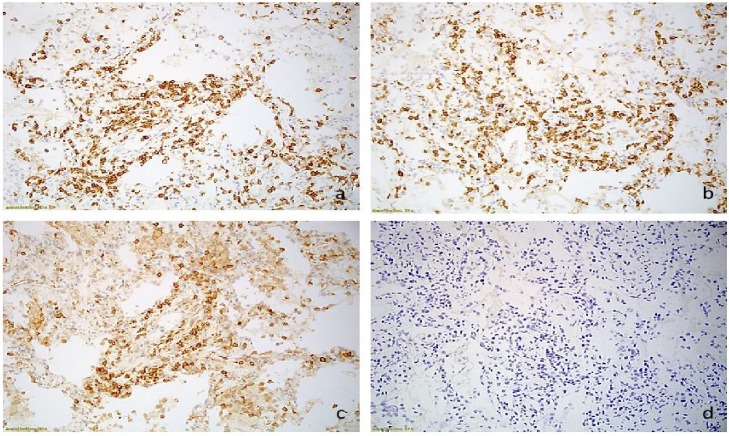
Immunnohistochemical studies were performed that revealed dominant prevalence of CD3 and CD8 positive T-lymphocytes with a very limited number of the CD4 positive T-cells (Fig. 2 ). There were virtually no CD20 positive B-lymphocytes. Immunohistochemical stains for CD10, CD20, CD23 and CD138 stains were negative. We concluded that the main histopathologic findings were markedly severe interstitial T-cell lymphocytic infiltration with fibrinous and organizing pneumonia and proliferative bronchiolitis consistent with SARS-CoV-2 infection. There was no evidence of vasculitis, CLL or other malignancy, granulomas, or aspiration. Stains for bacteria (Gram), acid-fast bacilli (AFB) and fungal organisms (GMS) were negative. Additional flow cytometry of the bronchoalveolar lavage fluid and peripheral blood did not reveal abnormalities.
Fig. 2.
Inflammatory infiltrate consist of CD3 positive T-lymphocytes (a), predominantly CD8 positive cytotoxic cells (b), and minor component of CD4 positive helper cells (c). CD20 positive B-cells were absent (d).
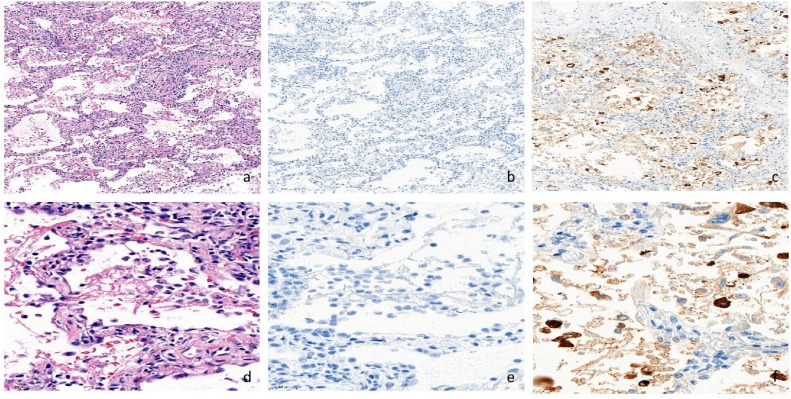
Sensitive and validated immunohistochemistry for SARS-CoV-2 nucleocapsid protein [5] was negative (Fig. 3 ), indicating that virus had been cleared from lungs at this stage.
Fig. 3.
No residual SARS-COV-2 is detected in post-COVID-19 lung sy from the patient. a, d. Lung tissue (Hematoxylin-Eosin). b, e. Immunohistochemistry of lung tissue is negative for SARS-CoV-2 nucleocapsid protein. c, f. Positive immunohistochemistry control for SARS-CoV-2 nucleocapsid protein.
The patient was started on high-dose steroid therapy and showed clinical improvement the next day with improved shortness of breath and no fever. Despite the steroid therapy, the patient had recurrent fever to 39.1 °C on the 6th day and experienced a new episode of shortness of breath with severe hypoxemia with an SpO2 of 77%. He was transferred to the ICU for increasing oxygen requirements. CT scan was repeated and a marked increase in bilateral patchy areas of consolidation with surrounding zones of groundglass opacity and intralobular septal thickening ("crazy paving") was noted. In spite of intensive supportive therapy, the patient had a refractory hypoxic respiratory failure and persistent positivity for Covid-19, and he died at the end of October. An autopsy was not performed.
3. Discussion
Literature review revealed reports of patients that died due to lung complications of Covid-19. The most common findings include diffuse alveolar damage, hyaline membrane formation, atypical enlarged pneumocytes with large nuclei or damaged pneumocytes with focal sloughing and formation of syncytial giant cells, type II pneumocyte hyperplasia, intra-alveolar hemorrhages, cluster or plug formation with accumulated fibrin. Pneumocytes may contain the virus within the cytoplasm and may show amphophilic granular cytoplasm and prominent nucleoli characteristic of viral cytopathic-like changes. There may be the presence of congested vessels, fibrinoid necrosis of the small vasculature, hyaline thrombi in microvessels. Edema in the respiratory mucosa along with infiltration by lymphocytes highlights the presence of severe inflammation [6,7].
In our case, the patient had persistent positivity for Covid-19 for 3 months and an antemortem cryo transbronchial biopsy that found areas of the interstitial inflammation with intensive lymphocytic infiltration by cytotoxic CD8 positive T-cells during the histopathological examination.
Mortality in patients with Covid-19 has been linked to ARDS and massive tissue damage due to the excessive production of proinflammatory cytokines induced by the virus. The three most important pro-inflammatory cytokines produced by tissue macrophages, endothelial, and epithelial cells are IL-1, IL-6, and TNF-α. As shown by previous data in the literature, increased levels of IL-1, IFNγ, TNF-α, IL-6, IL-12 strongly pointed toward activation of T-lymphocytes and influx of T-cells from the circulation into the site of viral infection [8].
A CD8 positive T-cell leads to virus clearance with minimal damage to the infected host. These T-lymphocytes become evident as early as 4–5 days after infection, with the peak numbers usually observed between 7–14 days. However, in some cases, the very intense immune response induces diffuse alveolar damage that far exceeds the harm that would have been caused by viral replication per se, and induces lethal tissue destruction or ireversible changes like fibrosis [9]. CD8 positive T-lymphocytes have great potential to damage tissues either through direct cytotoxicity or through cytokines release. There are two main pathways of cell apoptosis initiated by CD8 positive T-cell in response to a viral infection, the intrinsic, or mitochondrial, and the extrinsic pathways. The intrinsic pathway is mediated by BCL-2 proteins that regulate the permeability of the outer mitochondrial membrane and activate apoptotic protease-activation factor 1, cytochrome c, and caspase 9. The extrinsic pathway is induced through FAS (apoptosis antigen 1) receptor that belongs to the TNF receptor family and FAS ligand. The contributions of the intrinsic and extrinsic apoptosis pathways depend on the nature of the viral infection (acute or chronic) [10].
A recent report characterizes the immunological features of Covid-19 patients showing that the number of T-lymphocytes progressively decreases in peripheral blood in relation with increasing severity of disease. These data correlate with our antemortem histopathological examination of the lung that revealed the dominant infiltration of the tissue by CD8 positive T-cells. Observations are accumulating showing the prognostic value of cytokines and indicating that cytokine patterns are predictive of survival and mortality independently of comorbidities [11]. Early cytokine measurements and antemortem cryo transbronchial biopsy can be reliable predictors of outcome and might help to determine which patients are likely to develop respiratory failure. If confirmed, these factors could be used to select patients for clinical trials to aimed to disrupt the underlying pathomorphological events. According to these findings, anti-inflammatory therapy with IL-6 inhibitors, corticosteroids, checkpoint inhibitors, cytokine-adsorption devices, intravenous immunoglobulin could be very useful and reliable in decreasing the mortality in Covid-19 patients, especially if it started early enough after getting appropriate histopathological and laboratory evidence [12].
Although some patients with ARDS survived at the acute phase, there is a possibility of a long-term complication of Covid-19-related pulmonary fibrosis and death [13]. Lung biopsy may provide substantial insights relevant for patients with ongoing and eliminated SARS-CoV-2 infection by recognizing acute and chronic histopathological changes in the lungs. This can open a new possibility of initiating antifibrotic treatment to prevent further progression of pulmonary fibrosis [14].
We have described a novel lung injury pattern associated with SARS-CoV-2 infection. It manifests as a T-cell predominantly cytotoxic inflammatory infiltration. This observation could provide information that could be used to improve the clinical outcome of the COVID-19 patients.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sector.
Declaration of Competing Interest
Authors have declared that no competing interests exist. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1.World Health Organization - Coronavirus disease (COVID-19), situation reports. https://covid19.who.int/.
- 2.Hwang D.M., Chamberlain D.W., Poutanen S.M., Low D.E., Asa S.L., Butany J. Pulmonary pathology of severe acute respiratory syndrome in Toronto. Mod. Pathol. 2005;18(January 1):1–10. doi: 10.1038/modpathol.3800247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Calabrese F., Pezzuto F., Fortarezza F., Hofman P., Kern I., Panizo A., von der Thüsen J., Timofeev S., Gorkiewicz G., Lunardi F. Pulmonary pathology and COVID-19: lessons from autopsy. The experience of European pulmonary pathologists. Virchows Arch. 2020;477(September 3):359–372. doi: 10.1007/s00428-020-02886-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Doglioni C., Ravaglia C., Rossi G., Dubini A., Pedica F., Piciucchi S., Vizzuso A., Pecciarini L. Acute lung injury evolution in Covid-19. MedRxiv. 2021 doi: 10.1101/2020.08.09.20170910. [DOI] [Google Scholar]
- 5.Sun Y., Ge L., Rau M., Patton M., Gallan A., Felix J., Rui H. 2021. Sensitive and Specific Immunohistochemistry Protocols for Detection of SARS-CoV-2 Nucleocapsid and Spike Proteins in Formalin-Fixed, Paraffin-Embedded COVID-19 Patient Tissues. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Deshmukh V., Motwani R., Kumar A., Kumari C., Raza K. Histopathological observations in COVID-19: a systematic review. J. Clin. Pathol. 2020:1–8. doi: 10.1136/jclinpath-2020-206995. 0. [DOI] [PubMed] [Google Scholar]
- 7.Copin M.C., Parmentier E., Duburcq T., Poissy J., Mathieu D. Lille COVID-19 ICU and anatomopathology group. Time to consider histologic pattern of lung injury to treat critically ill patients with COVID-19 infection. Intensive Care Med. 2020;46(June 6):1124–1126. doi: 10.1007/s00134-020-06057-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ragab D., Salah Eldin H., Taeimah M., Khattab R., Salem R. The COVID-19 cytokine storm; what we know so far. Front. Immunol. 2020;11(June):1446. doi: 10.3389/fimmu.2020.01446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Coperchini F., Chiovato L., Croce L., Magri F., Rotondi M. The cytokine storm in COVID-19: an overview of the involvement of the chemokine/chemokine-receptor system. Cytokine Growth Factor Rev. 2020;53(June):25–32. doi: 10.1016/j.cytogfr.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kulinski J.M., Tarakanova V.L., Verbsky J. Regulation of antiviral CD8 T-cell responses. Crit. Rev. Immunol. 2013;33(6):477–488. doi: 10.1615/critrevimmunol.2013007909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Del Valle D.M., Kim-Schulze S., Huang H.H., Beckmann N.D., Nirenberg S., Wang B., Lavin Y., Swartz T.H., Madduri D., Stock A., Marron T.U., Xie H. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020;26(October 10):1636–1643. doi: 10.1038/s41591-020-1051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tang Y., Liu J., Zhang D., Xu Z., Ji J., Wen C. Cytokine storm in COVID-19: the current evidence and treatment strategies. Front. Immunol. 2020;11(July):1708. doi: 10.3389/fimmu.2020.01708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schwensen H.F., Borreschmidt L.K., Storgaard M., Redsted S., Christensen S., Madsen L.B. Fatal pulmonary fibrosis: a post-COVID-19 autopsy case. J. Clin. Pathol. 2020;(July):206879. doi: 10.1136/jclinpath-2020-206879. [DOI] [PubMed] [Google Scholar]
- 14.George P.M., Wells A.U., Jenkins R.G. Pulmonary fibrosis and COVID-19: the potential role for antifibrotic therapy. Lancet Respir. Med. 2020;8(August 8):807–815. doi: 10.1016/S2213-2600(20)30225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]