Dear Editor,
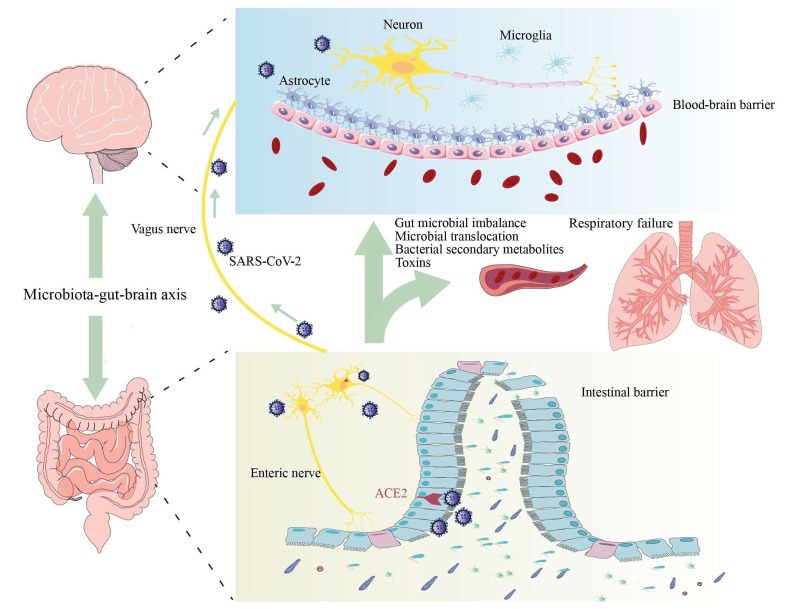
Many thanks for great interest in our published review (Wu et al., 2020). As we know, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) mainly infects the respiratory system, causing acute respiratory infectious diseases. It is worth noting that, like other coronaviruses, SARA-CoV-2 can do cause neurological damage. Various neurological manifestations of infected patients, including headache, anosmia, and anorexia, have been reported (Mao et al., 2020). Genome sequencing also confirmed the presence of SARS-CoV-2 in the cerebrospinal fluid. However, the mechanisms of neurological damage caused by SARS-CoV-2 remain unclear. Based on the existing findings, we proposed the following hypotheses: (1) directly entering the central nervous system (CNS) through olfactory nerve, (2) attacking the vagus nerve-nucleus of the solitary tract (NTS) via infecting the lung, and (3) affecting the brain via gut-brain axis (see Fig. 1 ).
Fig. 1.
The role and mechanism of gut-brain axis in neurological damage caused by SARS-CoV-2. ACE2: angiotensin-converting enzyme 2; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2.
Infectious humans often appear anosmia, which may be related to the virus attacking the CNS directly through the olfactory nerve. This viewpoint, however, may not be entirely correct. Firstly, nasal congestion, mucosal swelling, and local inflammation caused by viral upper respiratory infection can also cause dysosmia. Secondly, SARS-CoV-2 invades the target cells by binding its spike protein to the angiotensin-converting enzyme 2 (ACE2), but ACE2 level in the brain is lower expressed (Hikmet et al., 2020), and that the local immunity for the olfactory bulb is sufficient to resist the virus invasion. Collectively, these findings failed to support that SARS-CoV-2 could directly invade the CNS resulting in neurological damage.
The vagus nerve is widely distributed among the lung, sensing mechanical and chemical stimuli to the center. After the virus is inhaled into the lung, it directly invades the nerve endings and migrates into the NTS via the ascending fibers of vagus nerve. In addition, SARS-CoV-2 rapidly proliferation in the lung affects the gas exchange and then leads to hypoxia, finally causing brain dysfunction (Radnis et al., 2020, Wu et al., 2020).
The expression of ACE2 in intestinal epithelial cells and the enteric nervous system (ENS) is ample (Deffner et al., 2020). The intestine may be the target for SARS-CoV-2 infection (Esposito et al., 2020). Some COVID-19 patients were clinically characterized by gastrointestinal symptoms as the first manifestation, and that the viral RNA has been isolated from the feces of confirmed patients. The cluster analysis results also showed that the clinical manifestations of the digestive system are pathologically close to those of the CNS (Mirfazeli et al., 2020). We strongly agreed with the hypothesis that the SARS-CoV-2 is likely to affect the CNS through the intestine (Esposito et al., 2020).
Intestinal microbes have important biological functions, maintaining the intestinal epithelial barrier, immune homeostasis, and preventing pathogen invasion. In addition, intestinal microbes can affect the integrity and permeability of the blood–brain barrier (BBB) by increasing the expression of tight junction proteins. ACE2 is believed to be closely related to intestinal microbial homeostasis. Intestine infection and down-regulation of ACE2 expression caused by SARS-CoV-2 infection can lead to abnormal composition of gut microbiota, including decreased levels of Lactobacillus and Bifidobacterium (He et al., 2020). Microbial imbalance and intestinal inflammation are bound to affect intestinal barrier function and BBB integrity and permeability, thereby causing the translocation of intestinal bacteria, toxins and other intestinal microbial metabolites to enter the brain via blood circulation, finally resulting in brain dysfunction (Cryan et al., 2019).
The vagus nerve forms synaptic connections with neurons in the ENS, and transmits the intestinal information to the NTS, where the information is integrated and conveyed to the brain. Abnormalities in ENS not only cause gastrointestinal dysfunction, but also affect brain function through the gut-brain axis. A previous studies has shown that neurotropic viruses persistently infect the ENS and cause intestinal dysfunction (Brun et al., 2010). After entering the intestinal tract, SARS-CoV-2 invades the ENS by combining with ACE2, and then travels to the brain through the vagus nerve to affect the CNS function. More importantly, ENS injury leads to intestinal dysmotility, abnormal intestinal blood flow and epithelial barrier dysfunction, which further promotes the toxins from intestinal microbes and bacterial metabolites into the blood, aggravating brain damage.
The intestine involves the regulation of CNS function in various ways, such as nervous system, hormone system, and immune system. Increasing evidence shows that the SARS-CoV-2 can cause intestinal dysfunction, microbial imbalance, and immune disorder. Through the microbe-gut-brain axis, the intestine, especially the intestinal bacteria, is most likely to be the major approach for SARS-CoV-2 affecting the brain function. It is therefore likely that brain dysfunction would be secondary complication after SARS-CoV-2 infection. Future studies on the role of gut-brain axis in SARS-CoV-2 neuroinvasion are exceedingly required.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (grant numbers: 81703482 and 81974171 to C. Y.).
References
- Brun P., Giron M.C., Zoppellaro C., Bin A., Porzionato A., De Caro R., Barbara G., Stanghellini V., Corinaldesi R., Zaninotto G., Palù G., Gaion R.M., Tonini M., De Giorgio R., Castagliuolo I. Herpes simplex virus type 1 infection of the rat enteric nervous system evokes small-bowel neuromuscular abnormalities. Gastroenterology. 2010;138:1790–1801. doi: 10.1053/j.gastro.2010.01.036. [DOI] [PubMed] [Google Scholar]
- Cryan J.F., O'Riordan K.J., Cowan C.S.M., Sandhu K.V., Bastiaanssen T.F.S., Boehme M., Codagnone M.G., Cussotto S., Fulling C., Golubeva A.V., Guzzetta K.E., Jaggar M., Long-Smith C.M., Lyte J.M., Martin J.A., Molinero-Perez A., Moloney G., Morelli E., Morillas E., O'Connor R., Cruz-Pereira J.S., Peterson V.L., Rea K., Ritz N.L., Sherwin E., Spichak S., Teichman E.M., van de Wouw M., Ventura-Silva A.P., Wallace-Fitzsimons S.E., Hyland N., Clarke G., Dinan T.G. The microbiota-gut-brain axis. Physiol. Rev. 2019;99:1877–2013. doi: 10.1152/physrev.00018.2018. [DOI] [PubMed] [Google Scholar]
- Deffner F., Scharr M., Klingenstein S., Klingenstein M., Milazzo A., Scherer S., Wagner A., Hirt B., Mack A.F., Neckel P.H. Histological evidence for the enteric nervous system and the choroid plexus as alternative routes of neuroinvasion by SARS-CoV2. Front. Neuroanat. 2020;14 doi: 10.3389/fnana.2020.596439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esposito G., Pesce M., Seguella L., Sanseverino W., Lu J., Sarnelli G. Can the enteric nervous system be an alternative entrance door in SARS-CoV2 neuroinvasion? Brain Behav. Immun. 2020;87:93–94. doi: 10.1016/j.bbi.2020.04.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He L.H., Ren L.F., Li J.F., Wu Y.N., Li X., Zhang L. Intestinal Flora as a Potential Strategy to Fight SARS-CoV-2 Infection. Front. Microbiol. 2020;11:1388. doi: 10.3389/fmicb.2020.01388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hikmet F., Méar L., Edvinsson Å., Micke P., Uhlén M., Lindskog C. The protein expression profile of ACE2 in human tissues. Mol. Syst. Biol. 2020;16 doi: 10.15252/msb.20209610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirfazeli F.S., Sarabi-Jamab A., Jahanbakhshi A., Kordi A., Javadnia P., Shariat S.V., Aloosh O., Almasi-Dooghaee M., Faiz S.H.R. Neuropsychiatric manifestations of COVID-19 can be clustered in three distinct symptom categories. Sci. Rep. 2020;10:20957. doi: 10.1038/s41598-020-78050-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radnis C., Qiu S., Jhaveri M., Da Silva I., Szewka A., Koffman L. Radiographic and clinical neurologic manifestations of COVID-19 related hypoxemia. J. Neurol. Sci. 2020;418 doi: 10.1016/j.jns.2020.117119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Xu X., Chen Z., Duan J., Hashimoto K., Yang L., Liu C., Yang C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020;87:18–22. doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]